Cerebral hemispheres
The cerebral hemispheres are the largest and most developed part of the human brain. They contain the primary motor and sensory cortices, the highest levels at which motor activities are controlled and to which general and special sensory systems project, and which provide the neural substrate for the conscious experience of sensory stimuli. Association areas are both modality-specific and multimodal, enabling complex analyses of the internal and external environment and of the relationship of the individual with the external world. The elements of the limbic system are particularly concerned with memory and the emotional aspects of behaviour, and provide an affective overtone to conscious experience as well as an interface with subcortical areas such as the hypothalamus, through which widespread physiological activities are integrated. Other cortical areas, primarily within the frontal region, are concerned with the highest aspects of cognitive function and contribute to personality, judgment, foresight and planning.
The external surface of each hemisphere is highly convoluted into a series of folds or gyri, separated by furrows or sulci (Fig. 25.1). The configuration of the main cerebral sulci and gyri provides the basis for dividing the hemispheres into frontal, parietal, occipital, temporal, insular and limbic lobes. The internal white matter contains association fibres limited to each hemisphere, commissural fibres linking corresponding areas of both hemispheres, and projection fibres connecting the cerebral cortex of each hemisphere with subcortical, brainstem and spinal cord nuclei. Some of these bundles (tracts, fasciculi) are relatively well defined macroscopically and microscopically, while others are less easy to identify. A detailed knowledge of the three-dimensional anatomical interrelationships of white matter tracts is a requisite for the planning, intraoperative monitoring and execution of neurosurgical resective procedures, e.g. for tumour surgery, and epilepsy and deep brain stimulation procedures. Current understanding of these relationships owes much to the seminal work of Josef Klingler and his meticulous dissection of white matter tracts using formalin-fixed, freeze-thawed brains (Agrawal et al 2011). Contemporary neurosurgical anatomical studies seek to define and delineate these fibre bundles, particularly in areas of complexity such as fibre crossing, by correlating anatomical findings obtained using Klingler's dissection techniques with the results obtained from diffusion-weighted magnetic resonance imaging, functional MRI (fMRI), intraoperative electrostimulation and behavioural analyses (Alarcon et al 2014, Jin et al 2014). Some details of the anatomy of the gyri, sulci, association fibres and the amygdaloid nuclear complex are described online only (Figs 25.2; 25.9–25.13; 25.14; 25.17; 25.18; 25.20; 25.22; 25.27; 25.36; 25.37; 25.46–25.49![]() ).
).
Cerebral Hemisphere Surfaces, Sulci and Gyri
Each hemisphere has superolateral, medial and inferior (basal) surfaces, separated by superomedial, inferolateral, medial orbital and medial occipital margins respectively.
The superolateral surface is convex and lies beneath the bones of the cranial vault; the frontal, parietal, temporal and occipital lobes correspond approximately in surface extent to the overlying bones from which they take their names. The frontal and parietal lobes are separated from the temporal lobe by the prominent lateral (Sylvian) fissure.
The inferior surface is divided by the anterior part of the lateral fissure into a small anterior orbital part and a larger posterior tentorial part. The orbital part is the concave orbital surface of the frontal lobe and rests on the floor of the anterior cranial fossa. The posterior part is formed by the basal aspects of the temporal and occipital lobes, and rests on the floor of the middle cranial fossa and the upper surface of the tentorium cerebelli, which separates it from the superior surface of the cerebellum. The medial surface is flat and vertical, separated from the opposite hemisphere by the longitudinal fissure and the falx cerebri. Anteriorly, the cerebral hemisphere terminates at the frontal and temporal poles, and posteriorly at the occipital pole.
The cerebral sulci delineate the brain gyri and are extensions of the subarachnoid space (Butler and Hodos 2005, Sarnat and Netsky 1981, Park et al 2007, Chi et al 1977, Nishikuni and Ribas 2013, Ono et al 1990, Catani and Thiebaut de Schotten 2012, Duvernoy 1991, Naidich et al 2013). When they are deep and anatomically constant, they are referred to as fissures. The main sulci have depths of 1–3 cm, and their walls harbour small gyri that connect with each other (transverse gyri). Sulci that separate the transverse gyri vary in length and depth, and may become visible as incisures at the surface of the brain. The indentations caused by cortical arteries can have an appearance similar to that of the incisures. The sulci of the superolateral and inferior surfaces of the hemisphere are usually orientated towards the nearest ventricular cavity.
On the brain surface, the sulci can be long or short, interrupted or continuous. Sulci that are usually continuous include the lateral fissure and the callosal, calcarine, parieto-occipital, collateral and, generally, the central sulcus.
Four main types of sulci have been described: large primary sulci (e.g. central, precentral, postcentral and continuous sulci); short primary sulci (e.g. rhinal, olfactory, lateral and occipital sulci); short sulci composed of several branches (e.g. orbital and subparietal sulci); and short, free supplementary sulci (e.g. medial frontal and lunate sulci) (Ono et al 1990). Sulci often have side branches that may be unconnected or connected (with end-to-side, end-to-end or side-to-side connections that can also join two neighbouring parallel sulci).
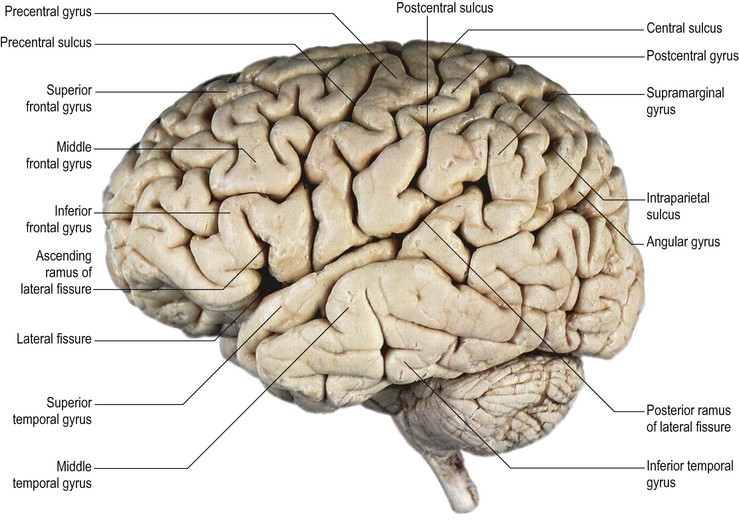
On the superolateral surface of the hemisphere, the frontal and temporal regions are each composed of three horizontal gyri (superior, middle and inferior frontal and temporal gyri). The central area is composed of two slightly oblique gyri (pre- and postcentral gyri). The parietal region is comprised of two semicircular lobules (superior and inferior parietal lobules, the inferior being formed by the supramarginal and angular gyri) (see Fig. 25.1). The occipital region is composed of two or three less well-defined gyri (superior, middle and inferior occipital gyri). The insula, which lies deep in the floor of the lateral fissure, consists of 4–5 diagonal gyri (short and long insular gyri) (Fig. 25.2![]() ).
).
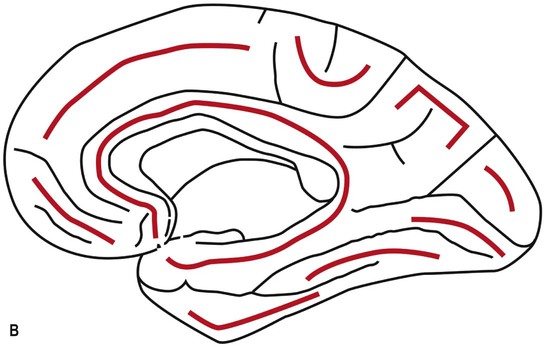
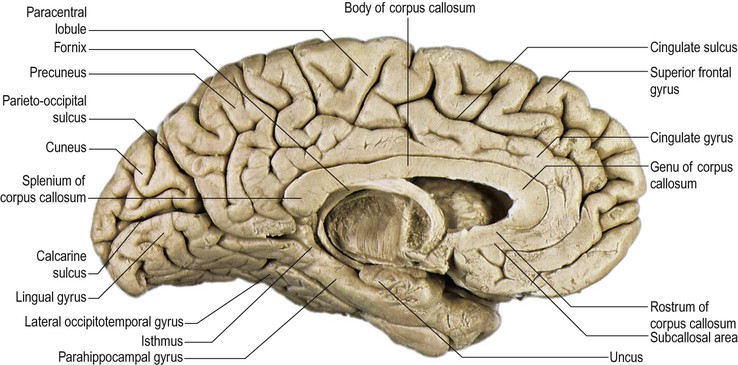
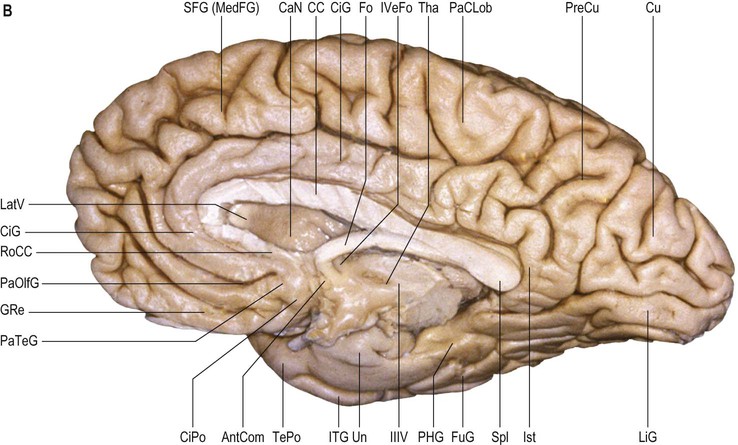
The orbital part of the inferior surface is covered by the orbital gyri and the basal aspect of the rectus gyri, and the tentorial part of the inferior surface is covered by the basal aspects of the inferior temporal, inferior occipital and lingual gyri, and the fusiform gyrus. The medial surface of the hemisphere is characterized by a very well defined C-shaped inner ring composed primarily of two continuous gyri (cingulate and parahippocampal gyri), surrounded by a much less well defined outer ring of gyri (medial aspects of rectus and superior frontal gyri, paracentral lobule, precuneus, cuneus, and medial aspect of lingual gyrus) (Fig. 25.3).
Microstructure of the Cortex
The microscopic structure of the cerebral cortex is an intricate complex of nerve cells and fibres, neuroglia and blood vessels. The neocortex essentially consists of three neuronal cell types. Pyramidal cells are the most abundant. Non-pyramidal cells, also called stellate or granule cells, are divided into spiny and non-spiny types. All types have been further subdivided on the basis of size and shape (Fig. 25.4; see Fig. 3.3).
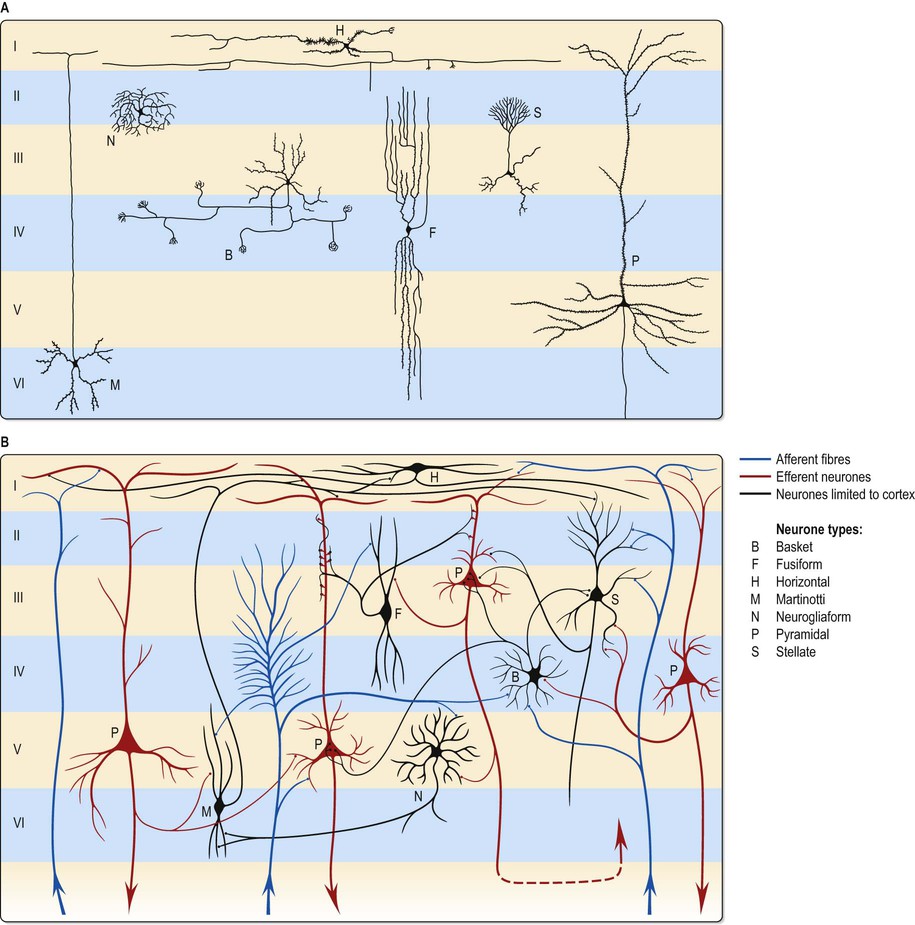
Pyramidal cells have a flask-shaped or triangular cell body ranging from 10 to 80 µm in diameter. The soma gives rise to a single thick apical dendrite and multiple basal dendrites. The apical dendrite ascends towards the cortical surface, tapering and branching, to end in a spray of terminal twigs in the most superficial lamina, the molecular layer. From the basal surface of the cell body, dendrites spread more horizontally, for distances up to 1 mm for the largest pyramidal cells. Like the apical dendrite, the basal dendrites branch profusely along their length. All pyramidal cell dendrites are studded with a myriad of dendritic spines. These become more numerous as distance from the parent cell soma increases. A single slender axon arises from the axon hillock, which is usually situated centrally on the basal surface of the pyramidal neurone. Ultimately, in the vast majority of cases, if not in all, the axon leaves the cortical grey matter to enter the white matter. Pyramidal cells are thus, perhaps universally, projection neurones. They use an excitatory amino acid, either glutamate or aspartate, as their neurotransmitter.
Spiny stellate cells are the second most numerous cell type in the neocortex and, for the most part, occupy lamina IV. They have relatively small multipolar cell bodies, commonly 6 to 10 µm in diameter. Several primary dendrites, profusely covered in spines, radiate for variable distances from the cell body. Their axons ramify within the grey matter predominantly in the vertical plane. Spiny stellate cells probably use glutamate as their neurotransmitter.
The smallest group comprises the heterogeneous non-spiny or sparsely spinous stellate cells. All are interneurones, and their axons are confined to grey matter. In morphological terms, this is not a single class of cell but a multitude of different forms, including basket, chandelier, double bouquet, neurogliaform, bipolar/fusiform and horizontal cells. Various types may have horizontally, vertically or radially ramifying axons.
Neurones with mainly horizontally dispersed axons include basket and horizontal cells. Basket cells have a short, vertical axon, which rapidly divides into horizontal collaterals, and these end in large terminal sprays synapsing with the somata and proximal dendrites of pyramidal cells. The cell bodies of horizontal cells lie mainly at the superficial border of lamina II, occasionally deep in lamina I (the molecular or plexiform layer). They are small and fusiform, and their dendrites spread short distances in two opposite directions in lamina I. Their axons often stem from a dendrite, then divide into two branches, which travel away from each other for great distances in the same layer.
Neurones with an axonal arborization predominantly perpendicular to the pial surface include chandelier, double bouquet and bipolar/fusiform cells. Chandelier cells have a variable morphology, although most are ovoid or fusiform and their dendrites arise from the upper and lower poles of the cell body. The axonal arborization, which emerges from the cell body or a proximal dendrite, is characteristic and identifies these neurones. A few cells in the more superficial laminae (II and IIIa) have descending axons, deeper cells (laminae IIIc and IV) have ascending axons, and intermediate neurones (IIIb) often have both. The axons ramify close to the parent cell body and terminate in numerous vertically orientated strings, which run alongside the axon hillocks of pyramidal cells, with which they synapse. Double bouquet (or bitufted) cells are found in laminae II and III and their axons traverse laminae II and V. Generally, these neurones have two or three main dendrites, which give rise to a superficial and deep dendritic tuft. A single axon arises usually from the oval or spindle-shaped cell soma and rapidly divides into an ascending and descending branch. These branches collateralize extensively, but the axonal arbor is confined to a perpendicularly extended, but horizontally confined, cylinder, 50–80 µm across. Bipolar cells are ovoid with a single ascending and a single descending dendrite, which arise from the upper and lower poles, respectively. These primary dendrites branch sparsely and their branches run vertically to produce a narrow dendritic tree, rarely more than 10 µm across, which may extend through most of the cortical thickness. Commonly, the axon originates from one of the primary dendrites, and rapidly branches to give a vertically elongated, horizontally confined axonal arbor, which closely parallels the dendritic tree in extent.
The principal recognizable neuronal type is the neurogliaform or spiderweb cell. These small spherical cells, 10–12 µm in diameter, are found mainly in laminae II–IV, depending on cortical area. Seven to ten thin dendrites typically radiate out from the cell soma, some branching once or twice to form a spherical dendritic field of approximately 100–150 µm diameter. The slender axon arises from the cell body or a proximal dendrite. Almost immediately, it branches profusely within the vicinity of the dendritic field (and usually somewhat beyond), to give a spherical axonal arbor up to 350 µm in diameter.
The majority of non-spiny or sparsely spinous non-pyramidal cells probably use γ-aminobutyric acid (GABA) as their principal neurotransmitter. This is almost certainly the case for basket, chandelier, double bouquet, neurogliaform and bipolar cells. Some are also characterized by the coexistence of one or more neuropeptides, including neuropeptide Y, vasoactive intestinal polypeptide (VIP), cholecystokinin (CCK), somatostatin and substance P. Acetylcholine is present in a subpopulation of bipolar cells, which may additionally be GABAergic and contain VIP.
Laminar Organization
The most obvious microscopic feature of a thin section of the neocortex stained to demonstrate cell bodies or fibres is its horizontal lamination. The extent to which this organization aids the understanding of cortical functional organization is debatable, but the use of cytoarchitectonic description to identify regions of cortex is common. Typical neocortex is described as having six layers, or laminae, lying parallel to the surface (Fig. 25.5). These are the molecular or plexiform layer; external granular lamina; external pyramidal lamina; internal granular lamina; internal pyramidal (ganglionic) lamina; and multiform (or fusiform/pleiomorphic) layer.
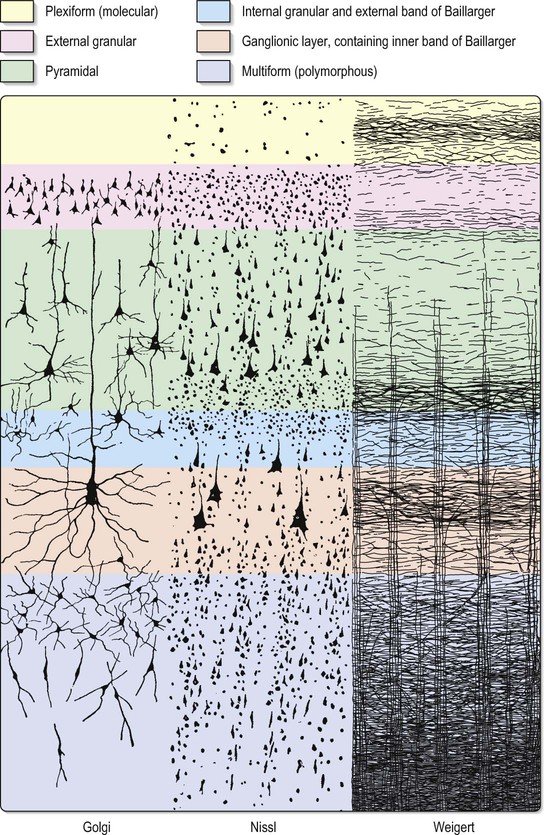
The molecular or plexiform layer is cell-sparse, containing only scattered horizontal cells and their processes enmeshed in a compacted mass of tangential, principally horizontal axons and dendrites. These are afferent fibres, which arise from outside the cortical area, together with intrinsic fibres from cortical interneurones, and the apical dendritic arbors of virtually all pyramidal neurones of the cerebral cortex. In histological sections stained to show myelin, layer I appears as a narrow horizontal band of fibres. The external granular lamina contains a varying density of small neuronal cell bodies, including both small pyramidal and non-pyramidal cells; the latter may predominate. Myelin fibre stains show mainly vertically arranged processes traversing the layer. The external pyramidal lamina contains pyramidal cells of varying sizes, together with scattered non-pyramidal neurones. The size of the pyramidal cells is smallest in the most superficial part of the layer and greatest in the deepest part. This lamina is frequently further subdivided into IIIa, IIIb and IIIc, with IIIa most superficial and IIIc deepest. As in layer II, myelin stains reveal a mostly vertical organization of fibres. The internal granular lamina is usually the narrowest of the cellular laminae. It contains densely packed, small, round cell bodies of non-pyramidal cells, notably spiny stellate cells and some small pyramidal cells. Within the lamina, in myelin stained sections, a prominent band of horizontal fibres (outer band of Baillarger) is seen. The internal pyramidal (ganglionic) lamina typically contains the largest pyramidal cells in any cortical area, though actual sizes vary considerably from area to area. Scattered non-pyramidal cells are also present. In myelin stains, the lamina is traversed by ascending and descending vertical fibres, and also contains a prominent central band of horizontal fibres (inner band of Baillarger). The multiform (or fusiform/pleiomorphic) layer consists of neurones with a variety of shapes, including recognizable pyramidal, spindle, ovoid and many other shapes of somata. Typically, most cells are small to medium in size. This lamina blends gradually with the underlying white matter, and a clear demarcation of its deeper boundary is not always possible.
Neocortical Structure
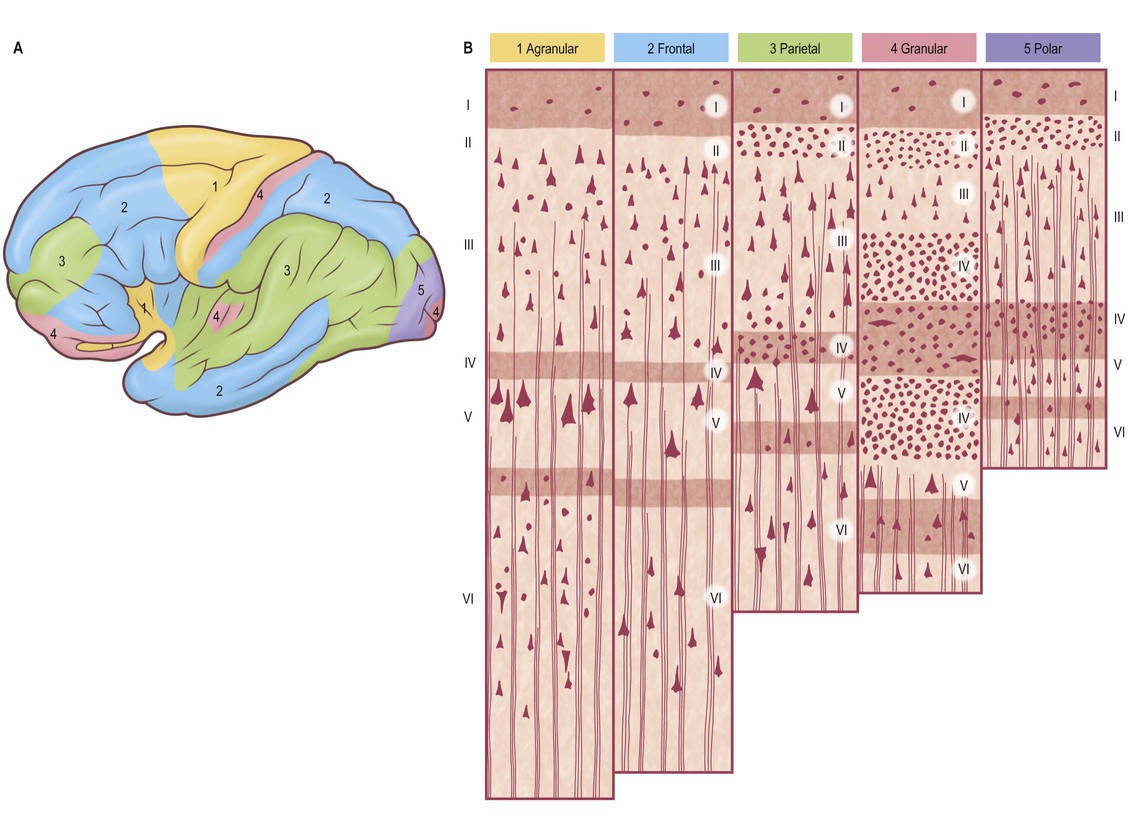
Five regional variations in neocortical structure are described (Fig. 25.6). While all are said to develop from the same six-layered pattern, two types – granular and agranular – are regarded as virtually lacking certain laminae, and are referred to as heterotypical. Homotypical variants, in which all six laminae are found, are called frontal, parietal and polar, names that link them with specific cortical regions in a somewhat misleading manner (e.g. the frontal type also occurs in parietal and temporal lobes).
The agranular type is considered to have diminished, or absent, granular laminae (II and IV) but always contains scattered stellate somata. Large pyramidal neurones are found in the greatest densities in agranular cortex, which is typified by the numerous efferent projections of pyramidal cell axons. Although it is often equated with motor cortical areas such as the precentral gyrus (area 4), agranular cortex also occurs elsewhere, e.g. areas 6, 8 and 44, and parts of the limbic system.
In the granular type of cortex the granular layers are maximally developed and contain densely packed stellate cells, among which small pyramidal neurones are dispersed. Laminae III and IV are poorly developed or unidentifiable. This type of cortex is particularly associated with afferent projections. However, it does receive efferent fibres, derived from the scattered pyramidal cells, although they are less numerous than elsewhere. Granular cortex occurs in the postcentral gyrus (somatosensory area), striate area (visual area) and superior temporal gyrus (acoustic area), and in small areas of the parahippocampal gyrus. Despite its very high density of stellate cells, especially in the striate area, it is almost the thinnest of the five main types. In the striate cortex, the external band of Baillarger (lamina IV) is well defined as the stria (white line) of Gennari.
The other three types of cortex are intermediate forms. In the frontal type, large numbers of small- and medium-sized pyramidal neurones appear in laminae III and V, and granular layers (II and IV) are less prominent. The relative prominence of these major forms of neurone varies reciprocally wherever this form of cortex exists.
The parietal type of cortex contains pyramidal cells, which are mostly smaller in size than in the frontal type. In marked contrast, the granular laminae are wider and contain more of the stellate cells: this kind of cortex occupies large areas in the parietal and temporal lobes. The polar type is classically identified with small areas near the frontal and occipital poles, and is the thinnest form of cortex. All six laminae are represented, but the pyramidal layer (III) is reduced in thickness and not so extensively invaded by stellate cells as it is in the granular type of cortex. In both polar and granular types, the multiform layer (VI) is more highly organized than in other types.
It is customary to refer to some discrete cortical territories not only by their anatomical location in relation to gyri and sulci, but also in relation to their cytoarchitectonic characteristics (Brodmann's areas) (Fig. 25.7). Some of the areas so defined, e.g. the primary sensory and motor cortices, have clear relevance in terms of anatomical connections and functional significance, others less so.
Cortical Lamination and Cortical Connections
The cortical laminae represent, to some extent, horizontal aggregations of neurones with common connections. This is most clearly seen in the lamination of cortical efferent (pyramidal) cells. The internal pyramidal lamina, layer V, gives rise to cortical projection fibres, most notably corticostriate, corticobulbar (including corticopontine) and corticospinal axons. In addition, a significant proportion of feedback corticocortical axons arise from cells in this layer, as do some corticothalamic fibres. Layer VI, the multiform lamina, is the major source of corticothalamic fibres. Supragranular pyramidal cells, predominantly layer III but also lamina II, give rise primarily to both association and commissural corticocortical pathways. Generally, short corticocortical fibres arise more superficially, and long corticocortical (both association and commissural) axons come from cells in the deeper parts of layer III. Major afferents to a cortical area tend to terminate in layers I, IV and VI. Quantitatively lesser projections end either in the intervening laminae II/III and V, or sparsely throughout the depth of the cortex. Numerically, the largest input to a cortical area tends to terminate mainly in layer IV. This pattern of termination is seen in the major thalamic input to visual and somatic sensory cortex. In general, non-thalamic subcortical afferents to the neocortex, which are shared by widespread areas, tend to terminate throughout all cortical layers, but the laminar pattern of their endings still varies considerably from area to area.
Columns and Modules
Experimental physiological and connectional studies have demonstrated an internal organization of the cortex, which is at right angles to the pial surface, with vertical columns or modules running through the depth of the cortex. The term ‘column’ refers to the observation that all cells encountered by a microelectrode penetrating and passing perpendicularly through the cortex respond to a single peripheral stimulus, a phenomenon first identified in the somatosensory cortex. In the visual cortex, narrow (50 µm) vertical strips of neurones respond to a bar stimulus of the same orientation (orientation columns), and wider strips (500 µm) respond preferentially to stimuli detected by one eye (ocular dominance columns). Adjacent orientation columns aggregate within an ocular dominance column to form a hypercolumn, responding to all orientations of stimulus for both eyes for one point in the visual field. Similar functional columnar organization has been described in widespread areas of neocortex, including motor cortex and association areas.
Cerebral Lobes
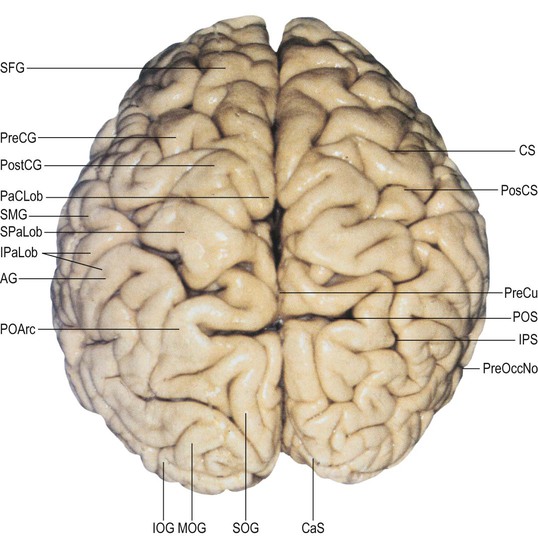
Each cerebral hemisphere is divided into six lobes: frontal, parietal, occipital, temporal, insular and limbic lobes. The surface features of the hemispheres exhibit considerable inter-individual variation in terms of the depth and size of their sulci and the resulting patterns of gyral separation (Ribas 2010). Connections between sulci are common; differing interpretations of these patterns of connectivity continue to contribute to inconsistencies in the literature, e.g. the use of different boundaries to demarcate the temporal, parietal and occipital lobes (Fig. 25.8).
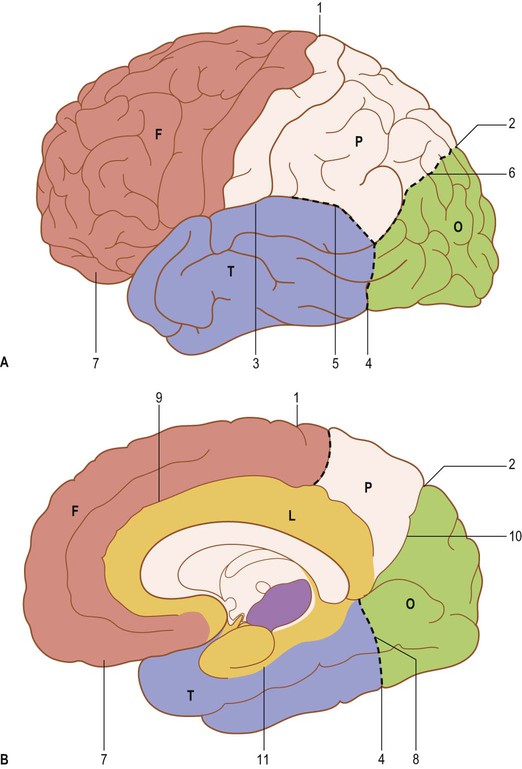
In what follows, each lobe will be described in terms of its external sulci and gyri, internal cortical structure and connectivity. Unless otherwise indicated in the caption, the dissections in this chapter display features in left cerebral hemispheres.
Frontal Lobe
The frontal lobe is the largest part of the cerebral hemisphere. It contains the primary motor area (MI) within the precentral gyrus, the supplementary motor area (SMA) anteriorly and medially, and the premotor areas anteriorly and laterally. While movement is thought to be initiated from within MI, the supplementary motor and premotor areas are believed to instruct the MI area. The most anterior and basal aspects of the frontal lobes are related to judgement and complex aspects of volitional behaviour.
Frontal lobe sulci and gyri
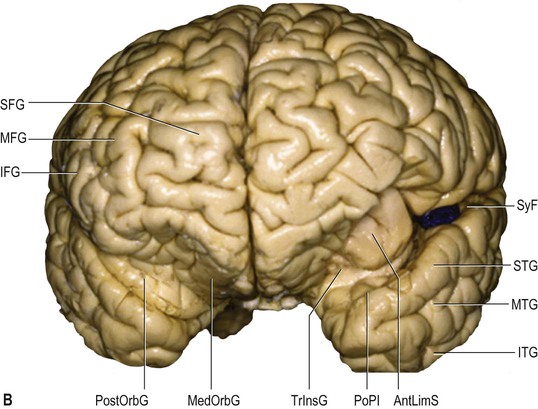
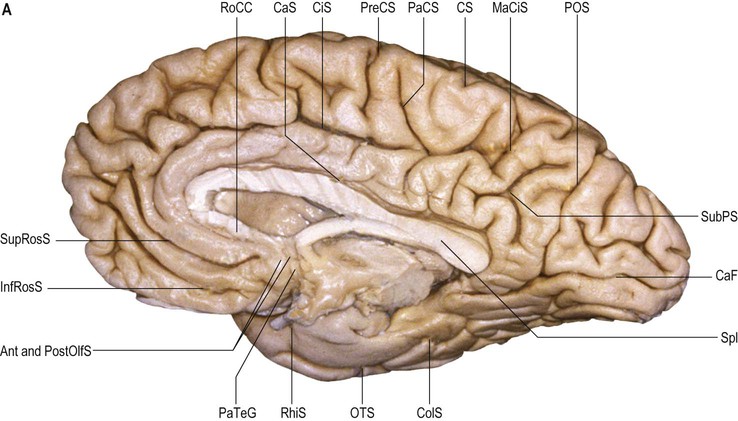
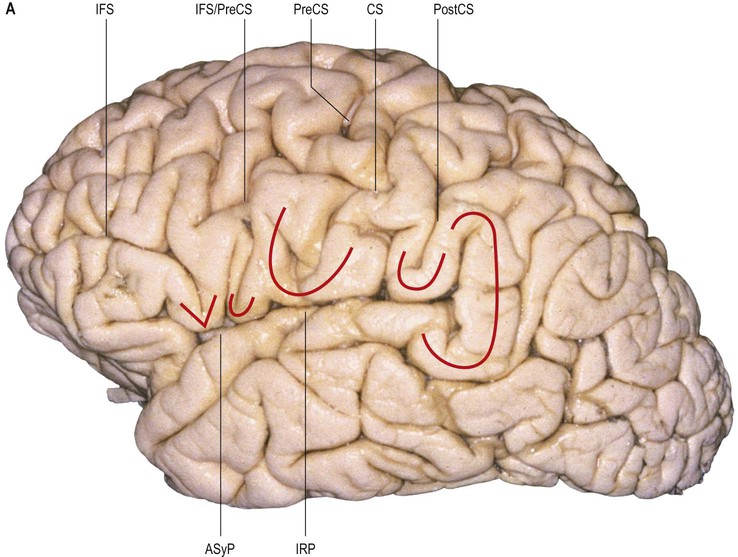
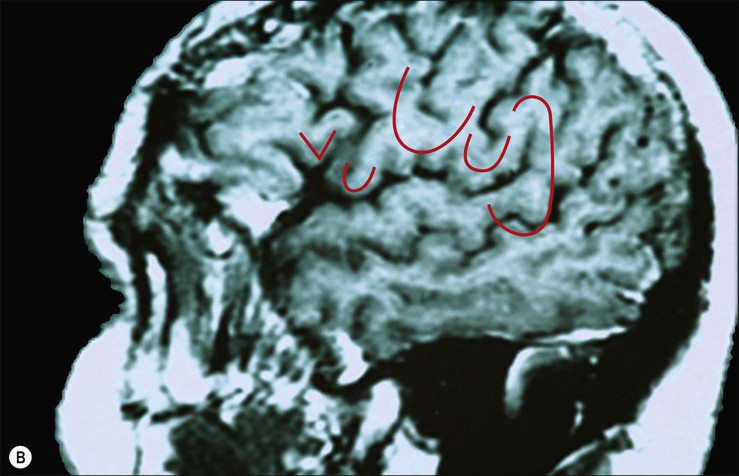
The frontal lobe is delimited posteriorly by the central sulcus, medially by the (great) longitudinal fissure and inferolaterally by the lateral fissure (Figs 25.9–25.10![]() ; see Figs 25.1, 25.8). The area of the frontal lobe anterior to the precentral gyrus is divided into longitudinal superior, middle and inferior frontal gyri; the frontal pole lies anterior to these gyri (see Figs 25.1, 25.8, 25.21B). Its superolateral (dorsal) surface is covered by the frontal bone. Its basal (ventral) surface lies over the orbital part of the frontal bone and the cribriform plate of the ethmoid bone, and displays the orbital and rectus gyri. The medial surface faces the falx cerebri.
; see Figs 25.1, 25.8). The area of the frontal lobe anterior to the precentral gyrus is divided into longitudinal superior, middle and inferior frontal gyri; the frontal pole lies anterior to these gyri (see Figs 25.1, 25.8, 25.21B). Its superolateral (dorsal) surface is covered by the frontal bone. Its basal (ventral) surface lies over the orbital part of the frontal bone and the cribriform plate of the ethmoid bone, and displays the orbital and rectus gyri. The medial surface faces the falx cerebri.
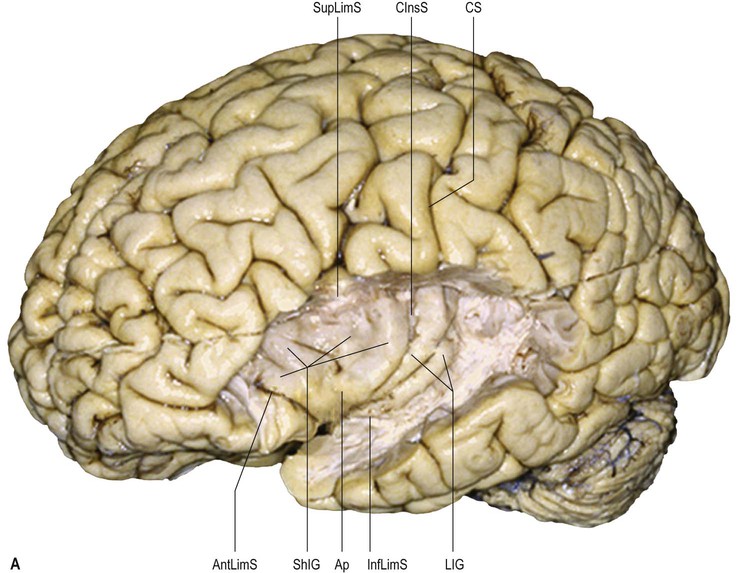
The central sulcus is the boundary between the frontal and parietal lobes. It demarcates the primary motor and somatosensory areas of the cortex, located in the precentral and postcentral gyri respectively. It starts in or near the superomedial border of the hemisphere, a little behind the midpoint between the frontal and occipital poles, and runs sinuously, resembling a lengthened letter S, downwards and forwards, to end usually a little above the posterior ramus of the lateral sulcus. The central sulcus is usually a continuous sulcus in both hemispheres.
The precentral gyrus lies obliquely over the superolateral surface of the cerebral hemisphere, its upper aspect extending on to the medial surface. It is continuous superiorly and inferiorly with the postcentral gyrus along connections that encircle both extremities of the central sulcus.
The inferior connection corresponds to the subcentral gyrus, delineated anteriorly and posteriorly by the anterior and posterior subcentral rami of the lateral fissure. It can either be situated completely over the lateral fissure or be in part internal to the fissure, in this situation giving the false impression that the central sulcus is a branch of the lateral fissure. The superior connection corresponds to the paracentral lobule (of Ecker) disposed along the medial surface of the hemisphere inside the interhemispheric fissure, delineated anteriorly by the paracentral sulcus and posteriorly by the ascending and distal part (marginal ramus) of the cingulate sulcus. Broca described a middle connection between the pre- and postcentral gyri (pli de passage moyen of Broca) that may be present as a gyral bridge, usually hidden within the central sulcus; on the cortical surface, this corresponds to the classic, posteriorly convex, middle genu of the central sulcus. When this middle connection is sufficiently developed so that it reaches the brain surface, it interrupts the central sulcus (Régis et al 2005).
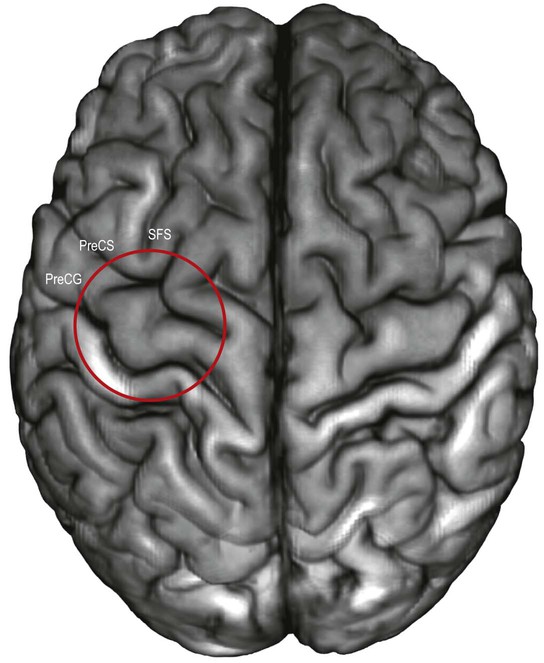
The localization of motor and sensory hand areas has been studied by correlating imaging cortical stimulation and postmortem cadaveric studies. The motor hand area has been localized by fMRI to a protrusion of the precentral gyrus that corresponded precisely to the middle genu of the central sulcus, at the distal end of the superior frontal sulcus (Yousry et al 1997, Ribas 2010) (Figs 25.11–25.12). Postmortem studies revealed that this protrusion was delimited by two anteriorly directed fissures that deepened towards the base of the protrusion. Hand sensory function has been localized to the postcentral component of the middle connection of the pre- and postcentral gyri (Boling and Olivier 2004, Boling et al 2008).
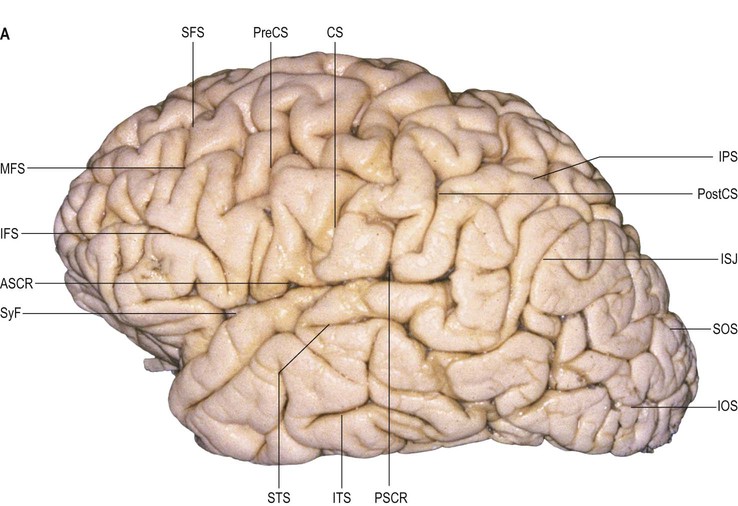
The precentral gyrus is delimited anteriorly by the precentral sulcus, itself divided into superior and inferior precentral sulci by the connection of the middle frontal gyrus with the precentral gyrus. Further connections of the superior, middle and inferior frontal gyri may divide the superior and the inferior precentral sulci into additional segments. The superior part of the precentral sulcus is very often interrupted superiorly by a connection between the superior frontal and precentral gyri, producing a more medial segment, the medial precentral sulcus, that corresponds to the sulcus precentralis medialis of Eberstaller. More dorsally, within the precentral region, the marginal precentral sulcus (sulcus precentralis marginalis of Cunningham) may merge with the superior precentral or central sulci. The inferior segment of the precentral sulcus always ends inside the opercular part of the inferior frontal gyrus, producing its characteristic U shape.
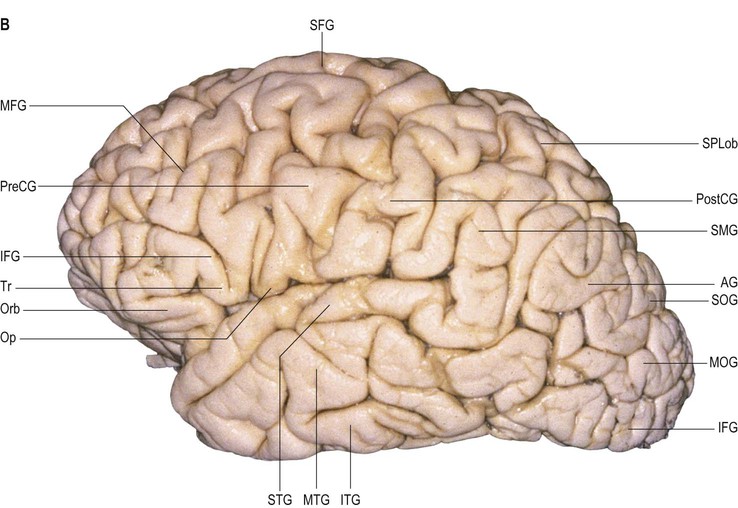
The pre- and the postcentral gyri are roughly parallel to the coronal suture; the precentral sulcus is located slightly posterior to it. The superior, middle and inferior frontal gyri are disposed longitudinally, anterior to the precentral gyrus, and are separated by superior and inferior frontal sulci (see Fig. 25.1); they are frequently referred to as F1, F2 and F3 respectively.
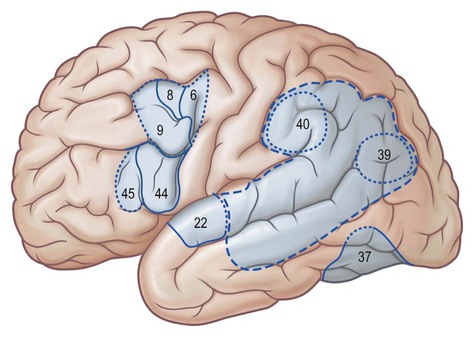
The superior frontal gyrus is continuous anteriorly and inferiorly with the rectus gyrus; it may also be connected to the orbital gyri and the middle frontal gyrus. Posteriorly, it is connected to the precentral gyrus by at least one fold, which most commonly lies medially along the interhemispheric fissure. Usually the superior longitudinal gyrus is subdivided into two longitudinal portions by a medial frontal sulcus; its medial portion is sometimes termed the medial frontal gyrus. The supplementary motor area is located along the most medial portion of the superior frontal gyrus, immediately facing the precentral gyrus; it varies between individuals and has poorly defined borders. The middle frontal gyrus is usually the largest of the frontal gyri, frequently connected superficially to the precentral gyrus by a prominent root that lies between the extremities of a marked interruption in the precentral sulcus. It harbours a complex of multiple shallow sulcal segments known collectively as the middle or intermediate frontal sulcus (Petrides 2012). Superiorly, the inferior frontal gyrus is crossed by various small branches of the interrupted inferior frontal sulcus; the triangular sulcus typically pierces the superior aspect of the triangular part. In the dominant hemisphere, the opercular and triangular parts of the inferior gyrus correspond to Broca's area, which is responsible for the production of spoken language (Fig. 25.13) (Quiñones-Hinojosa et al 2003). The most posterior aspect of the inferior frontal gyrus, identifiable by the connection of its opercular part with the precentral gyrus, corresponds to the ventral premotor cortical area; its bilateral stimulation causes speech arrest (Duffau 2011).
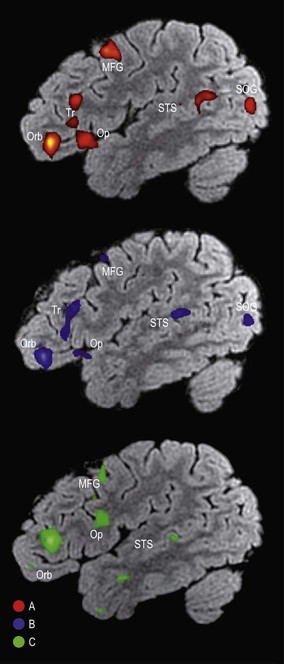
The superior frontal sulcus separates the superior and middle frontal gyri. It is very deep and is frequently continuous, ending posteriorly by encroaching on the precentral gyrus at the level of its omega region (corresponding to the motor cortical representation of the contralateral hand). The superior frontal sulcus therefore tends to point the way to the middle frontoparietal pli de passage, as well as to the middle genu of the precentral gyrus, where there is also a motor representation of the hand (Boling et al 1999).
The inferior frontal sulcus is always interrupted by the multiple connections running between the middle and inferior gyri and usually has three parts: orbital, triangular and opercular. The orbital part is the most prominent. The triangular part is usually more retracted, such that there is a small widening of the lateral fissure at its base corresponding to the anterior Sylvian point. It is characterized by horizontal and anterior ascending rami of the lateral fissure that consistently divide the lateral fissure into anterior and posterior branches. The opercular part is always U-shaped and harbours the inferior aspect of the precentral sulcus; it is continuous posteriorly with the basal aspect of the precentral gyrus over the anterior subcentral ramus of the lateral fissure (Fig. 25.14). The anterior basal portion of the opercular part is sometimes divided by another branch of the lateral fissure, the diagonal sulcus of Eberstaller.
Inferiorly, the orbital part continues with the lateral orbital gyrus, at times passing under a shallow sulcus known as the fronto-orbital sulcus. The basal apex of the triangular part is always superior to the lateral fissure; the base of the opercular part can be located either superiorly or within the fissure. Anteriorly, the inferior frontal gyrus terminates by merging with the anterior portion of the middle frontal gyrus. All of the frontal gyri are delineated anteriorly by the frontomarginal sulcus (frontomarginal sulcus of Wernicke), which lies superior and parallel to the supraciliary margin, separating the superolateral and orbital frontal surfaces. Posteriorly, the inferior frontal gyrus is connected to the precentral gyrus along the posterior aspect of its opercular part.
The olfactory sulcus lies longitudinally in a paramedian position on the frontobasal or orbital surface of each frontal lobe. It accommodates the olfactory tract and bulb. Posteriorly, the olfactory tract is divided into medial and lateral striae, which delineate the most anterior aspect of the anterior perforated substance (see Figs 25.20B, 25.32). The narrow gyrus rectus, medial to the olfactory sulcus, is considered to be the most anatomically constant of the cerebral gyri. The orbital gyri, lateral to the olfactory sulcus, account for the greatest proportion of the frontobasal surface. The anterior, posterior, medial and lateral orbital gyri are delineated by the lateral, medial and transverse orbital sulci and the cruciform sulcus of Rolando, which together form a characteristic H shape. The posterior orbital gyrus lies anterior to the anterior perforated substance and typically presents a configuration similar to a tricorn hat, a feature that may facilitate its identification in anatomical specimens where the H-shaped orbital sulcus is less obvious. The remaining orbital gyri are connected to the superior, middle and inferior frontal gyri along the frontal pole.
On its medial surface, the frontal lobe is limited inferiorly by the cingulate sulcus, which starts within the subcallosal region and extends over the cingulate gyrus. The cingulate sulcus may occasionally be double anteriorly and enclose a connection of the anterior aspect of the cingulate gyrus and the medial and anterior aspects of the superior frontal gyrus. It frequently has side branches that are directed inferiorly (see Fig. 25.10![]() ).
).
The paracentral lobule, bounded posteriorly by the marginal ramus and anteriorly by the paracentral sulcus (a branch of the cingulate sulcus), contains the distal part of the central sulcus and, inferior to it, the so-called paracentral fossa. Anterior to the paracentral lobule, the medial aspect of the superior frontal gyrus lies over the cingulate sulcus and the cingulate gyrus, merging inferiorly with the gyrus rectus. The latter is bounded superiorly by the superior rostral sulcus and accommodates the shallower inferior rostral sulcus along its surface. The cingulate gyrus systematically connects with the gyrus rectus around the posterior end of the superior rostral sulcus by a prominent U-shaped cortical fold known as the cingulate pole, which is located immediately anterior to the subcallosal gyri. Small supraorbital sulci lie within the medial surface of the frontal pole, superior to the superior rostral sulcus at the level of the genu of the corpus callosum.
Frontal lobe internal structure and connectivity
Primary motor cortex
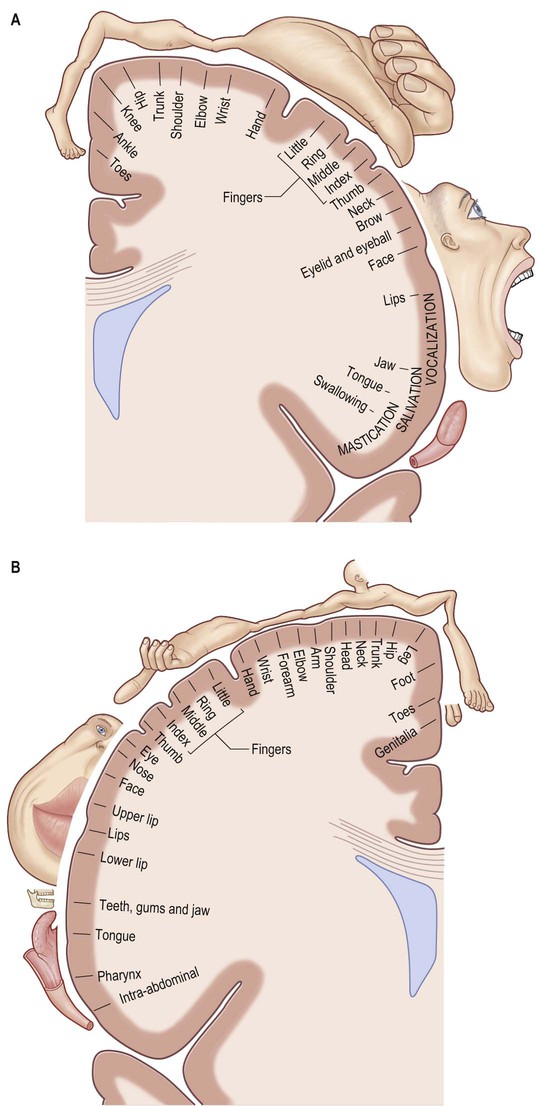
The primary motor cortex (MI) corresponds to the precentral gyrus (area 4), and is the area of cortex with the lowest threshold for eliciting contralateral muscle contraction by electrical stimulation. It contains a detailed topographically organized map (motor homunculus) of the opposite body half, with the head represented most laterally, and the leg and foot represented on the medial surface of the hemisphere in the paracentral lobule (Fig. 25.15). A striking feature is the disproportionate representation of body parts in relation to their physical size: large areas represent the muscles of the face and hand, which are capable of finely controlled or fractionated movements.
The cortex of area 4 is agranular, and layers II and IV are difficult to identify. The most characteristic feature is the presence in lamina V of some extremely large pyramidal cell bodies, Betz cells, which may approach 80 µm in diameter. These neurones project their axons into the corticospinal and corticonuclear tracts.
The major thalamic connections of area 4 are with the posterior part of the ventral lateral nucleus, which in turn receives afferents from the deep cerebellar nuclei. The posterior part of the ventral lateral nucleus also contains a topographic representation of the contralateral body half, which is preserved in its point-to-point projection to area 4, where it terminates largely in lamina IV. Other thalamic connections of area 4 are with the centromedian and parafascicular nuclei. These appear to provide the only route through which output from the basal ganglia, routed via the thalamus, reaches the primary motor cortex, since the projection of the internal segment of the globus pallidus to the ventral lateral nucleus of the thalamus is confined to the anterior division, and there is no overlap with cerebellothalamic territory. The anterior part of the ventral lateral nucleus projects to the premotor and supplementary motor areas of cortex with no projection to area 4.
The ipsilateral somatosensory cortex (SI) projects in a topographically organized way to area 4, and the connection is reciprocal. The projection to the motor cortex arises in areas 1 and 2, with little or no contribution from area 3b. Fibres from SI terminate in layers II and III of area 4, where they contact mainly pyramidal neurones. Evidence suggests that neurones activated monosynaptically by fibres from SI, as well as those activated polysynaptically, make contact with layer V pyramidal cells, including Betz cells, which give rise to corticospinal fibres. Movement-related neurones in the motor cortex that can be activated from SI tend to have a late onset of activity, mainly during the execution of movement. It has been suggested that this pathway plays a role primarily in making motor adjustments during a movement. Additional ipsilateral corticocortical fibres to area 4 from behind the central sulcus come from the second somatic sensory area (SII).
Neurones in area 4 are responsive to peripheral stimulation, and have receptive fields similar to those in the primary sensory cortex. Cells located posteriorly in the motor cortex have cutaneous receptive fields, whereas more anteriorly situated neurones respond to stimulation of deep tissues.
The motor cortex receives major frontal lobe association fibres from the premotor cortex and the supplementary motor area, and also fibres from the insula. It is probable that these pathways modulate motor cortical activity in relation to the preparation, guidance and temporal organization of movements. Area 4 sends fibres to, and receives fibres from, its contralateral counterpart, and also projects to the contralateral supplementary motor cortex.
Apart from its contribution to the corticospinal tract, the motor cortex has diverse subcortical projections. The connections to the striatum and pontine nuclei are heavy. It also projects to the subthalamic nucleus. The motor cortex sends projections to all nuclei in the brainstem, which are themselves the origin of descending pathways to the spinal cord: namely, the reticular formation, the red nucleus, the superior colliculus, the vestibular nuclei and the inferior olivary nucleus.
Corticospinal tract
The corticospinal or pyramidal tract provides direct control by the cerebral cortex over motor centres of the spinal cord (Ch. 20). A homologous pathway to the brainstem, the corticonuclear projection, fulfils a similar function in relation to motor nuclei of the brainstem (Ch. 21).
The percentage of corticospinal fibres that arise from the primary motor cortex may be in the region of 20–30%. They arise from pyramidal cells in layer V and give rise to the largest-diameter corticospinal axons. There is also a widespread origin from other parts of the frontal lobe, including the premotor cortex and the supplementary motor area. Many axons from the frontal cortex, notably the motor cortex, terminate in the ventral horn of the spinal cord. In cord segments mediating dexterous hand and finger movements, they terminate in the lateral part of the ventral horn, in close relationship to motor neuronal groups. A small percentage establish direct monosynaptic connections with α motor neurones. Between 40 and 60% of pyramidal tract axons arise from parietal areas, including area 3a, area 5 of the superior part of the parietal lobe, and SII in the parietal operculum. The majority of parietal fibres to the spinal cord terminate in the deeper layers of the dorsal horn.
Motor cortical neurones are active in relation to the force of contraction of agonist muscles; their relation to amplitude of movement is less clear. Their activity precedes the onset of electromyographic activity by 50–100 milliseconds, suggesting a role for cortical activation in generating rather than monitoring movement.
Premotor cortex
Brodmann's area 6 lies immediately in front of the primary motor cortex (Fig. 25.16). It extends on to the medial surface, where it becomes contiguous with area 24 in the cingulate gyrus, anterior and inferior to the paracentral lobule. A number of functional motor areas are contained within this cortical region. Lateral area 6, the area over most of the lateral surface of the hemisphere, corresponds to the premotor cortex.
The premotor cortex is divided into a dorsal and a ventral area (PMd and PMv respectively) on functional grounds, and on the basis of ipsilateral corticocortical association connections.
The major thalamic connections of the premotor cortex are with the anterior division of the ventrolateral nucleus and with the centromedian, parafascicular and centrolateral components of the intralaminar nuclei. Subcortical projections to the striatum and pontine nuclei are prominent, and this area also projects to the superior colliculus and the reticular formation. Both dorsal and ventral areas contribute to the corticospinal tract. Commissural connections are with the contralateral premotor, motor and superior parietal (area 5) cortex. Ipsilateral corticocortical connections with area 5 in the superior parietal cortex, and inferior parietal area 7b, are common to both dorsal and ventral subdivisions of the premotor cortex, and both send a major projection to the primary motor cortex. The dorsal premotor area also receives fibres from the posterior superior temporal cortex and projects to the supplementary motor cortex. The frontal eye field (area 8) projects to the dorsal subdivision. Perhaps the greatest functionally significant difference in connectivity between the two premotor area subdivisions is that the dorsal premotor area receives fibres from the dorsolateral prefrontal cortex, whereas the ventral subdivision receives fibres from the ventrolateral prefrontal cortex. All of these association connections are likely to be, or are known to be, reciprocal.
Neuronal activity in the premotor cortex in relation to both preparation for movement and movement itself has been extensively studied experimentally. Direction selectivity for movement is a common feature of many premotor neurones. In behavioural tasks, neurones in the dorsal premotor cortex show anticipatory activity and task-related discharge as well as direction selectivity, but little or no stimulus-related changes. The dorsal premotor cortex is probably important in establishing a motor set or intention, contributing to motor preparation in relation to internally guided movement. In contrast, ventral premotor cortex is more related to the execution of externally (especially visually) guided movements in relation to a specific external stimulus.
Frontal eye field
The frontal eye field corresponds to parts of Brodmann's areas 6, 8 and 9 (see Fig. 25.16). As its name implies, it is important in the control of eye movements. It receives its major thalamic projection from the parvocellular mediodorsal nucleus, with additional afferents from the medial pulvinar, the ventral anterior nucleus and the suprageniculate–limitans complex, and connects with the paracentral nucleus of the intralaminar group. The thalamocortical pathways to the frontal eye field form part of a pathway from the superior colliculus, the substantia nigra and the dentate nucleus of the cerebellum. The frontal eye field has extensive ipsilateral corticocortical connections, receiving fibres from several visual areas in the occipital, parietal and temporal lobes, including the medial temporal area (V5) and area 7a. There is also a projection from the superior temporal gyrus, which is auditory rather than visual in function. From within the frontal lobe, the frontal eye field receives fibres from the ventrolateral and dorsolateral prefrontal cortices. It projects to the dorsal and ventral premotor cortices and to the medial motor area, probably to the supplementary eye field adjacent to the supplementary motor area proper. It projects prominently to the superior colliculus, to the pontine gaze centre within the pontine reticular formation, and to other oculomotor related nuclei in the brainstem.
Supplementary motor cortex
The supplementary motor area (SMA; MII) lies medial to area 6, and extends from the most superolateral part to the medial surface of the hemisphere. Area 24 in the cingulate gyrus adjacent to area 6 contains several motor areas, which are termed cingulate motor areas. An additional functional subdivision, the pre-supplementary motor area, lies anterior to the supplementary motor area on the medial surface of the cortex. In the present discussion, these additional medial motor areas are included with the supplementary motor cortex.
The supplementary motor area receives its major thalamic input from the anterior part of the ventral lateral nucleus, which in turn is the major recipient of fibres from the internal segment of the globus pallidus. Additional thalamic afferents are from the ventral anterior nucleus, the intralaminar nuclei – notably the centrolateral and centromedial nuclei, and also from the mediodorsal nucleus. The connections with the thalamus are reciprocal. The supplementary motor cortex receives connections from widespread regions of the ipsilateral frontal lobe, including from the primary motor cortex, the dorsal premotor area, the dorsolateral and ventrolateral prefrontal, medial prefrontal and orbitofrontal cortex, and the frontal eye field. These connections are reciprocal but the major ipsilateral efferent pathway is to the motor cortex. Parietal lobe connections of the supplementary motor cortex are with the superior parietal area 5 and possibly inferior parietal area 7b. Contralateral connections are with the supplementary motor area, and motor and premotor cortices of the contralateral hemisphere. Subcortical connections, other than with the thalamus, pass to the striatum, subthalamic nucleus and pontine nuclei, the brainstem reticular formation and the inferior olivary nucleus. The supplementary motor area makes a substantial contribution to the corticospinal tract, contributing as much as 40% of the fibres from the frontal lobe.
The supplementary motor area contains a representation of the body in which the leg is posterior and the face anterior, with the upper limb between them. Its role in the control of movement is primarily in complex tasks that require temporal organization of sequential movements and in the retrieval of motor memory.
Stimulation of the supplementary motor area in conscious patients has been reported to elicit the sensation of an urge to move, or of anticipation that a movement is about to occur. A region anterior to the supplementary motor area for face representation (areas 44, 45) is important in vocalization and speech production (see Fig. 25.16).
Prefrontal cortex
The prefrontal cortex on the lateral surface of the hemisphere comprises predominantly Brodmann's areas 9, 46 and 45 (see Fig. 25.16). In non-human primates, two subdivisions of the lateral prefrontal cortex are recognized: a dorsal area equivalent to area 9, and perhaps including the superior part of area 46; and a ventral area, consisting of the inferior part of area 46 and area 45. Areas 44 and 45 are particularly notable in humans since, in the dominant hemisphere, they constitute the motor speech area (Broca's area) within the opercular and triangular parts of the inferior frontal gyrus, immediately anterior to the most inferior aspect of the precentral gyrus. Both the dorsolateral and ventrolateral prefrontal areas receive their major thalamic afferents from the mediodorsal nucleus, and there are additional contributions from the medial pulvinar, from the ventral anterior nucleus and from the paracentral nucleus of the anterior intralaminar group. The dorsolateral area receives long association fibres from the posterior and middle superior temporal gyrus (including auditory association areas), from parietal area 7a, and from much of the middle temporal cortex. From within the frontal lobe it also receives projections from the frontal pole (area 10), and from the medial prefrontal cortex (area 32) on the medial surface of the hemisphere. It projects to the supplementary motor area, the dorsal premotor cortex and the frontal eye field. All these thalamic and corticocortical connections are reciprocal. Commissural connections are with the homologous area and with the contralateral inferior parietal cortex. The ventrolateral prefrontal area receives long association fibres from both area 7a and area 7b of the parietal lobe, from auditory association areas of the temporal operculum, from the insula and from the anterior part of the lower bank of the superior temporal sulcus. From within the frontal lobe it receives fibres from the anterior orbitofrontal cortex and projects to the frontal eye field and the ventral premotor cortex. It connects with the contralateral homologous area via the corpus callosum. These connections are probably all reciprocal.
The cortex of the frontal pole (area 10) receives thalamic input from the mediodorsal nucleus, the medial pulvinar and the paracentral nucleus. It is reciprocally connected with the cortex of the temporal pole, the anterior orbitofrontal cortex and the dorsolateral prefrontal cortex. The orbitofrontal cortex connects with the mediodorsal, anteromedial, ventral anterior, medial pulvinar, paracentral and midline nuclei of the thalamus. Cortical association pathways come from the inferotemporal cortex, the anterior superior temporal gyrus and the temporal pole. Within the frontal lobe it has connections with the medial prefrontal cortex, the ventrolateral prefrontal cortex and medial motor areas. Commissural and other connections follow the general pattern for all neocortical areas.
The medial prefrontal cortex is connected with the mediodorsal, ventral anterior, anterior medial pulvinar, paracentral, midline and suprageniculate–limitans nuclei of the thalamus. It receives fibres from the anterior cortex of the superior temporal gyrus. Within the frontal lobe, it has connections with the orbitofrontal cortex, and the medial motor areas of the dorsolateral prefrontal cortex.
Parietal Lobe
The parietal lobe lies posterior to the central sulcus on the superolateral and medial surfaces of the cerebral hemisphere (see Figs 25.1, 25.8–25.10![]() ). This region is part of the complex language network (see below). Posterior to the supramarginal gyrus and again in the dominant hemisphere, the cortex of the angular gyrus is related to neuronal processing associated with reading and writing.
). This region is part of the complex language network (see below). Posterior to the supramarginal gyrus and again in the dominant hemisphere, the cortex of the angular gyrus is related to neuronal processing associated with reading and writing.
Parietal lobe sulci and gyri
The parietal gyri are morphologically poorly defined and tortuous; some are termed lobules. Posteriorly, the parietal lobe is delineated on the medial aspect by the parieto-occipital sulcus and on the lateral aspect by an imaginary line running from the point where the parieto-occipital sulcus emerges on to the superolateral border to the preoccipital notch (a small sulcus situated on the inferolateral border approximately 5 cm anterior to the occipital pole). The inferior boundary is the posterior ramus of the lateral fissure and its imaginary posterior prolongation.
The lateral aspect of the parietal lobe is divided into three areas by the postcentral and intraparietal sulci. The intraparietal sulcus lies predominantly longitudinally along the midportion of the parietal superolateral surface (see Fig. 25.1). It delineates the superior parietal lobule, continuous medially with the precuneus, and the inferior parietal lobule, made up of the supramarginal and angular gyri and a more posterior convolution continuous with the occipital lobe. The inferior aspect of the supramarginal gyrus within the inferior parietal lobule of the dominant hemisphere corresponds to Wernicke's area, which extends along the posterior aspect of the superior temporal gyrus.
The postcentral gyrus lies posterior to the precentral gyrus and is connected to it along the superior and inferior extremities of the central sulcus. It is usually narrower than the precentral gyrus. Both gyri are located obliquely on the superolateral surface of the hemisphere, just superior to the lateral fissure; their midportions correspond approximately to the anteroposterior centre of each cerebral hemisphere. The superior portions of the pre- and postcentral gyri, which constitute the paracentral lobule on the medial surface of the cerebral hemisphere, are topographically related to the ventricular atrium, situated posterior to the thalamus. The inferior portions of both gyri cover the posterior half of the insula and are topographically related to the body of the lateral ventricle, situated superior to the thalamus. The portion of the subcentral gyrus corresponding to the base of the postcentral gyrus consistently lies over the transverse gyri of Heschl, situated on the opercular surface of the temporal lobe (Wen et al 1999).
The postcentral sulcus delineates the posterior boundary of the postcentral gyrus. It is frequently interrupted by connections with the superior and inferior parietal lobules. The inferior part of the postcentral sulcus always ends at a basal connection between the postcentral and the supramarginal gyri (Ribas 2010). The intraparietal sulcus, which originates from around the midpoint of the postcentral sulcus, is prominent on the superolateral surface of the parietal lobe, running parallel with the superior margin of the hemisphere. Anteriorly, the intraparietal sulcus is usually continuous with the inferior portion of the postcentral sulcus and posteriorly it passes into the occipital lobe as the intra-occipital sulcus (superior occipital sulcus), which continues more posteriorly into the transverse occipital sulcus. The intraparietal sulcus divides the superolateral parietal surface into superior and inferior parietal lobules; along its length, it typically gives rise to superior and inferior vertical sulcal branches. The superior vertical sulcal branch (transverse parietal sulcus of Brissaud) divides the superior parietal lobule. The inferior vertical sulcal branch (intermediate sulcus of Jensen; sulcus intermedius primus of Jensen) separates the supramarginal gyrus anteriorly from the angular gyrus posteriorly.
The supramarginal gyrus is always a very well defined curved gyrus. It surrounds the distal portion of the lateral fissure (its posterior ascending branch) and becomes continuous with the posterior portion of the superior temporal gyrus. Above the distal end of the lateral fissure, the supramarginal gyrus is connected anteriorly to the postcentral gyrus through a fold that runs underneath the inferior aspect of the postcentral sulcus. Posteriorly, it occasionally rounds the inferior extremity of the intermediate sulcus and connects to the angular gyrus. The angular gyrus is a curved gyrus, often poorly defined morphologically. It always surrounds one of the distal segments of the superior temporal sulcus, usually the middle one (angular sulcus), and its most inferior portion is continuous with the middle temporal gyrus. The configuration of the angular gyrus is defined by the distal branching of the superior temporal sulcus, which typically ends as three continuous or interrupted caudal branches.
The most superior distal branch of the superior temporal sulcus has an ascending course; it may either penetrate the supramarginal gyrus or coincide with the intermediate sulcus of Jensen separating the supramarginal and angular gyri. The second branch is usually more horizontal and enters the angular gyrus as the angular sulcus. The most inferior caudal branch of the superior temporal sulcus is less evident and less constant. It courses underneath a posterior fold that frequently connects the angular gyrus and the most lateral aspect of the occipital lobe, and is usually continuous with the anterior occipital sulcus, lying predominantly vertically along the anterior edge of the middle occipital gyrus. The bulge of the supramarginal and angular gyri is responsible for the cranial parietal tuberosity (bossa).
The superior parietal lobule has a quadrangular shape. It is delineated anteriorly by the superior aspect of the postcentral sulcus and laterally by the intraparietal sulcus; medially, it is continuous with the precuneus gyrus along the superomedial border (Fig. 25.17). Anteriorly, it is typically connected to the postcentral gyrus via a fold that transects the most superior portion of the postcentral sulcus and, occasionally, via a fold that interrupts the postcentral sulcus more inferiorly. Posteriorly, the superior parietal lobule continues to the superior occipital gyrus via the prominent parieto-occipital arcus.
On the medial surface of each hemisphere, the precuneus lies posterior to the paracentral lobule as a medial extension of the superior parietal lobule. The precuneus and the medial aspect of the postcentral gyrus correspond to the medial portion of the parietal lobe (see Figs 25.3, 25.10B). The precuneus is quadrangular (quadrangular lobule of Foville), delineated anteriorly by the marginal branch of the cingulate sulcus, posteriorly by the parieto-occipital sulcus and inferiorly by multiple Y-shaped sulcal segments that constitute the subparietal sulcus. Inferior to the subparietal sulcus, the precuneus is connected to the isthmus of the cingulate gyrus, which is continuous with the parahippocampal gyrus.
The parieto-occipital sulcus separating the precuneus from the cuneus is deep and contains many small sulci and gyri. The precuneal limiting sulcus and the cuneal limiting sulcus delineate the inferior (posterior) limit of the precuneus and the superior (anterior) limit of the cuneus, respectively. They lie along the most superficial aspects of the superior and inferior margins of the parieto-occipital sulcus. Small cuneal gyri lie along its inner surfaces. The superior parietal lobule and the precuneus are also referred to as P1 and the supramarginal and angular gyri are referred to as P2 and Pc 3 or P3 respectively.
Parietal lobe internal structure and connectivity
Somatosensory cortex
The postcentral gyrus corresponds to the primary somatosensory cortex (SI; Brodmann's areas 3a, 3b, 1 and 2). Area 3a lies most anteriorly, apposing area 4, the primary motor cortex of the frontal lobe; area 3b is buried in the posterior wall of the central sulcus; area 1 lies along the posterior lip of the central sulcus; and area 2 occupies the crown of the postcentral gyrus.
The primary somatosensory cortex contains within it a topographical map of the contralateral half of the body. The face, tongue and lips are represented inferiorly, the trunk and upper limb are represented on the superolateral aspect, and the lower limb on the medial aspect of the hemisphere, giving rise to the familiar ‘homunculus’ map (see Fig. 25.15).
The somatosensory properties of SI depend on its thalamic input from the ventral posterior nucleus of the thalamus, which in turn receives the medial lemniscal, spinothalamic and trigeminothalamic pathways. The nucleus is divided into a ventral posterolateral part, which receives information from the trunk and limbs, and a ventral posteromedial part, in which the head is represented. Within the ventral posterior nucleus, neurones in the central core respond to cutaneous stimuli and those in the most dorsal anterior and posterior parts, which arch as a ‘shell’ over this central core, respond to deep stimuli. This is reflected in the differential projections to SI: the cutaneous central core projects to 3b, the deep tissue-responsive neurones send fibres to areas 3a and 2, and an intervening zone projects to area 1. Within the ventral posterior nucleus, anteroposterior rods of cells respond with similar modality and somatotopic properties. They appear to project to restricted focal patches in SI of approximately 0.5 mm width, which form narrow strips mediolaterally along SI. The laminar termination of thalamocortical axons from the ventral posterior nucleus is different in the separate cytoarchitectonic subdivisions of SI. In 3a and 3b, these axons terminate mainly in layer IV and the adjacent deep part of layer III, whereas in areas 1 and 2 they end in the deeper half of layer III, avoiding lamina IV. Additional thalamocortical fibres to SI arise from the intralaminar system, notably the centrolateral nucleus.
There is a complex internal connectivity within SI. An apparently stepwise hierarchical progression of information processing occurs from area 3b through area 1 to area 2. Outside the postcentral gyrus, SI has ipsilateral corticocortical association connections with a second somatosensory area (SII); area 5 in the superior part of the parietal lobe; area 4, the motor cortex, in the precentral gyrus; and the supplementary motor cortex in the medial part of area 6 of the frontal lobe.
SI has reciprocal commissural connections with its contralateral homologue, with the exception that the cortices containing the representation of the distal extremities are relatively devoid of such connections. Callosal fibres in SI arise mainly from the deep part of layer III and terminate in layers I–IV. Pyramidal cells contributing callosal projections receive monosynaptic thalamic and commissural connections.
SI has reciprocal subcortical connections with the thalamus and claustrum, and receives afferents from the nucleus basalis (basal nucleus of Meynert), the locus coeruleus and the midbrain raphe. It has other prominent subcortical projections. Corticostriatal fibres, arising in layer V, pass mainly to the putamen of the same side. Corticopontine and corticotectal fibres from SI arise in layer V. SI projects to the main pontine nuclei and to the nucleus reticularis tegmenti pontis (pontine tegmental reticular nucleus). In addition, axons arising in SI pass to the dorsal column nuclei and the spinal cord. Corticospinal pyramidal cells are found in layer V of SI. The topographical representation in the cortex is preserved in terms of the spinal segments to which different parts of the postcentral gyrus project. Thus, the arm representation projects to the cervical enlargement, the leg representation to the lumbosacral enlargement, and so on. Within the grey matter of the spinal cord, fibres from SI terminate in the dorsal horn, in Rexed's laminae 3–5; fibres from 3b and 1 end more dorsally, and those from area 2 more ventrally.
The second somatosensory area (SII) lies along the upper bank of the lateral fissure, posterior to the central sulcus. SII contains a somatotopic representation of the body, with the head and face most anteriorly, adjacent to SI, and the sacral regions most posteriorly. SII is reciprocally connected with the ventral posterior nucleus of the thalamus in a topographically organized fashion. Some thalamic neurones probably project to both SI and SII via axon collaterals. Other thalamic connections of SII are with the posterior group of nuclei and with the intralaminar central lateral nucleus. SII also projects to laminae IV–VII of the dorsal horn of the cervical and thoracic spinal cord, the dorsal column nuclei, the principal trigeminal nucleus, and the periaqueductal grey matter of the midbrain.
Within the cortex, SII is reciprocally connected with SI in a topographically organized manner and projects to the primary motor cortex. SII also projects in a topographically organized way to the lateral part of area 7 (area 7b) in the superior part of the parietal lobe, and makes connections with the posterior cingulate gyrus. Both right and left SII areas are interconnected across the corpus callosum, although distal limb representations are probably excluded. There are additional callosal projections to SI and area 7b.
Experimental studies show that neurones in SII respond particularly to transient cutaneous stimuli, e.g. brush strokes or tapping, which are characteristic of the responses of Pacinian corpuscles in the periphery. They show little response to maintained stimuli.
Superior and inferior parietal lobules
Posterior to the postcentral gyrus, the superior part of the parietal lobe is composed of areas 5, 7a and 7b (see Fig. 25.7). Area 5 receives a dense feed-forward projection from all cytoarchitectonic areas of SI in a topographically organized manner. The thalamic afferents to this area come from the lateral posterior nucleus and from the central lateral nucleus of the intralaminar group. Ipsilateral corticocortical fibres from area 5 go to area 7, the premotor and supplementary motor cortices, the posterior cingulate gyrus and the insular granular cortex. Commissural connections between area 5 on both sides tend to avoid the areas of representation of the distal limbs. The response properties of cells in area 5 are more complex than in SI, with larger receptive fields and evidence of submodality convergence. Area 5 contributes to the corticospinal tract.
In non-human primates, the inferior parietal lobule is area 7. In humans, this area is more superior; areas 39 and 40 intervene inferiorly. The counterparts for the latter areas in monkeys are unclear and little experimental evidence is available on their connections and functions. Their role in human cerebral processing is discussed below. In the monkey, area 7b receives somatosensory inputs from area 5 and SII. Connections pass to the posterior cingulate gyrus (area 23), insula and temporal cortex. Area 7b is reciprocally connected with area 46 in the prefrontal cortex and the lateral part of the premotor cortex. Commissural connections of area 7b are with the contralateral homologous area and with SII, the insular granular cortex and area 5. Thalamic connections are with the medial pulvinar nucleus and the intralaminar paracentral nucleus.
In monkeys, area 7a is not related to the cortical pathways for somatosensory processing but instead forms part of a dorsal cortical pathway for spatial vision. The major ipsilateral corticocortical connections to area 7a are derived from visual areas in the occipital and temporal lobes. In the ipsilateral hemisphere, area 7a has connections with the posterior cingulate cortex (area 24) and with areas 8 and 46 of the frontal lobe. Commissural connections are with its contralateral homologue. Area 7a is connected with the medial pulvinar and intralaminar paracentral nuclei of the thalamus. In experimental studies, neurones within area 7a are visually responsive; they relate largely to peripheral vision, respond to stimulus movement, and are modulated by eye movement.
Occipital Lobe
On the superolateral cerebral surface, the occipital lobe is situated posterior to an imaginary line connecting the point of emergence of the parieto-occipital fissure on the superomedial border of the cerebral hemisphere with the preoccipital notch of Meynert (approximately 5 cm anterior to the occipital pole) (see Fig. 25.8). On the medial surface, the occipital lobe is limited anteriorly by the parieto-occipital sulcus and by its prolongation towards the tentorium cerebelli. Along the inferior cerebral surface, the base of the occipital lobe is continuous with the base of the temporal lobe. The superolateral surface of the occipital lobe lies mostly beneath the squamous part of the occipital bone; its medial surface faces the most posterior aspect of the falx cerebri, and the inferior surface lies over the tentorium cerebelli. The sulci and gyri of the occipital lobe, especially of its superolateral surface, exhibit a greater degree of anatomical variation than is seen in the other lobes: this is reflected in an often confusing literature where the same sulcus may be identified using different names, and two obviously different sulci may be called by the same name. The particular complexity of sulcal, and therefore gyral, variability in this part of the occipital lobe has been attributed to variations in regional cortical folding that are reflected in the diversity of sulcal origin, degree of segmentation, length, depth and intrasulcal geometry. Regarding the major named sulci, there appears to be reasonable consensus that the parieto-occipital, calcarine, paracalcarine, lingual, transverse occipital, inferior lateral occipital and anterior occipital sulci, together with the occipital parts of the collateral and lateral occipitotemporal sulci, are constant features of the surface of the occipital lobe, whereas the superior occipital, superior lateral occipital, inferior occipital, lunate and occipitopolar sulci are less constant. (For further details, consult Iaria and Petrides (2007), Iaria et al (2008), Alves et al (2012), Malikovic et al (2012).)
Occipital lobe sulci and gyri
There are two or three gyri (superior, middle and inferior, or O1, O2 and O3, respectively) on the superolateral cerebral surface of the occipital lobe (see Figs 25.9B, 25.17![]() ). They converge posteriorly to form the occipital pole. Commonly, only the superior and inferior gyri are present; the area corresponding to the middle occipital gyrus lies between the inferior extension of the intra-occipital (or superior occipital or transverse occipital) sulcus and the lateral (or inferior occipital) sulcus.
). They converge posteriorly to form the occipital pole. Commonly, only the superior and inferior gyri are present; the area corresponding to the middle occipital gyrus lies between the inferior extension of the intra-occipital (or superior occipital or transverse occipital) sulcus and the lateral (or inferior occipital) sulcus.
The superior occipital gyrus is always well defined, and is continuous along the superomedial margin of the hemisphere with the cuneus. Superiorly, it is delimited by the depth of the parieto-occipital fissure on the superolateral hemispheric surface. It is continuous with the superior parietal lobule through the parieto-occipital arcus (corresponding to the first or superior parieto-occipital ‘pli de passage’ of Gratiolet). Laterally, the superior occipital gyrus may be delimited by either the intra-occipital, transverse occipital or superior occipital sulcus.
The inferior occipital gyrus lies horizontally along the inferolateral margin of the hemisphere, with its base lying over the tentorium cerebelli. Anteriorly, it is usually continuous with the inferior temporal gyrus; posteriorly, it extends medially around the occipital pole, becoming continuous with the lingual gyrus on the medial surface of the hemisphere. Superiorly, the inferior occipital gyrus is delimited by the lateral or inferior occipital sulcus.
The lateral occipital sulcus is a very evident horizontal sulcus. Anteriorly, it is frequently connected to the inferior temporal sulcus; inferiorly, it may be accompanied by a shorter accessory lateral occipital sulcus. Both of these sulci may be connected with a sulcal complex known as the anterior occipital sulcus, which, when present, lies along the anterior aspect of the middle occipital gyrus. The inferior occipital sulcus is sometimes described as a distinct and very small sulcus located near the inferior margin of the inferior occipital gyrus, but here the lateral and inferior occipital sulci are considered to be part of the same structure.
The intraparietal sulcus extends longitudinally and inferiorly into the occipital lobe, where it becomes the intra-occipital sulcus. The latter may occasionally descend to the occipital pole but it usually terminates on reaching the transverse occipital sulcus, dividing it into lateral and medial parts that penetrate the superior occipital gyrus (see Fig. 25.17). Since the lateral (inferior) occipital sulcus is always present and clearly divides the superolateral occipital surface into an inferior part, constituted by the inferior occipital gyrus, and a superior part, it has been suggested that the gyral pattern of the superior part depends mainly on the morphology of the lateral aspect of the transverse occipital sulcus. When this sulcal segment descends towards the occipital pole as an inferior extension of the intra-occipital sulcus, it divides the upper occipital convexity into superior and middle occipital gyri. The lunate sulcus, although conspicuous in monkeys and apes, is only sometimes identifiable in human brains, when it appears as a well-defined vertical and backward-curved sulcus anterior to the occipital pole.
Despite significant anatomical variation, the superolateral occipital convolutions are connected to the parietal and temporal convolutions by consistent cortical folds. According to the classic description by Gratiolet, four folds connect the parietal and temporal lobes with the occipital lobe: the superior parieto-occipital fold (parieto-occipital arcus) connects the superior parietal lobule with the superior occipital gyrus; the inferior parieto-occipital fold, a posterior extension of the angular gyrus, connects with the middle occipital gyrus and occasionally also with the superior occipital gyrus; the first temporo-occipital fold is the continuation of the middle temporal gyrus with the inferior occipital gyrus; and the second temporo-occipital fold is the continuation of the inferior temporal gyrus with the inferior occipital gyrus.
The medial surface of the occipital lobe shows less morphological variation than the superolateral surface. It is separated from the parietal lobe by the parieto-occipital sulcus and dominated by the calcarine sulcus. The dorsal part of this region, above the calcarine sulcus and posterior to the parieto-occipital fissure, is the cuneus. The ventral part of this region, lying below the calcarine sulcus and extending as far as the occipital extension of the collateral fissure, is the lingual gyrus (see Fig. 25.10B). The calcarine sulcus starts anteriorly underneath the splenium of the corpus callosum, delineating the inferior aspect of the isthmus of the cingulate gyrus, and runs posteriorly just above the inferomedial margin of the hemisphere. The parieto-occipital fissure emerges superiorly from the calcarine sulcus, separates the cuneus from the precuneus of the parietal lobe, and divides the calcarine sulcus into an anterior and a posterior part. The parieto-occipital and calcarine sulci appear continuous on the surface, but when their borders are retracted it becomes obvious that they are separated by one or more small gyri. The anterior part of the calcarine sulcus is classified as a complete sulcus because its depth creates an elevation (calcar avis) in the medial wall of the occipital horn of the lateral ventricle. The posterior part of the calcarine sulcus is considered an axial sulcus, given that its axis runs along the visual cortex. Only the posterior part includes the primary visual cortical areas, which are located on its superior (cuneal) and inferior (lingual) surfaces. This part of the calcarine sulcus frequently harbours the cuneolingual gyrus that links both gyri.
At the level of the occipital pole, the calcarine sulcus usually branches in a T or Y shape as the retrocalcarine sulcus. The gyrus descendens of Ecker lies posterior to and along the retrocalcarine sulcus and is occasionally bounded posteriorly by the occipitopolar sulcus. The retrocalcarine sulcus and its variations are sometimes referred to as external calcarine sulci.
Given the anatomical constancy of the calcarine and parieto-occipital fissures on the medial occipital surface, the cuneus (O6) is always a well-defined wedge-like convolution. The real anterior border of the cuneus is the cuneal limiting sulcus within the parieto-occipital fissure. Posteriorly, the cuneus rests over the posterior part of the calcarine sulcus and over the posterior aspect of the lingual gyrus. Superior to the posterior part of the calcarine sulcus, the cuneus harbours the paracalcarine or cuneal sulcus (the inferior sagittal sulcus of the cuneus of Retzius) and, further dorsally, the occipital paramedial sulcus (the paramesial sulcus of Elliot Smith or superior sagittal sulcus of Retzius).
The basal or inferior surface of the occipital lobe is continuous with the basal surface of the temporal lobe. It is formed, from medial to lateral, by the lingual, fusiform and inferior occipital gyri respectively (see Fig. 25.10B; Fig. 25.18). For details of the sulcal and gyral anatomy of the basal occipital-temporal lobe, see Chau et al (2014).
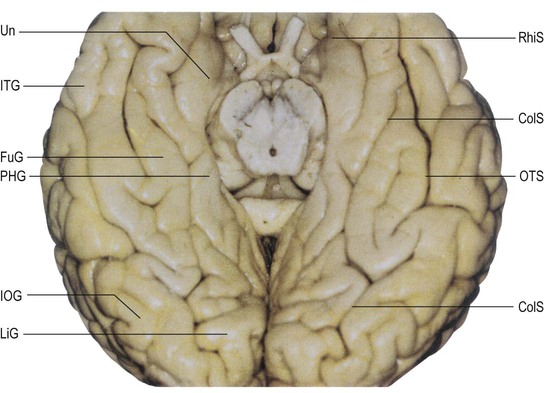
The lingual gyrus (medial temporo-occipital gyrus, O5) lies inferiorly along the entire length of the calcarine sulcus, forming the mediobasal portion of the occipital lobe. It is continuous anteriorly with the parahippocampal gyrus and its basal surface rests on the tentorium cerebelli. Posteriorly, it is frequently divided into a superior and inferior part by an intralingual sulcus, which may be a posteromedial ramus of the collateral sulcus.
The fusiform or lateral temporo-occipital gyrus lies along the temporo-occipital transition. Its posterior or occipital part is bounded medially by the collateral sulcus and laterally by the occipitotemporal sulcus; hence it lies between the lingual gyrus medially and the inferior occipital gyrus laterally. The occipital part of the fusiform gyrus (O4) lies over the tentorium cerebelli just posterior to the petrous part of the temporal bone. Topographically, it corresponds to the floor of the ventricular atrium; the temporal part of the gyrus lies underneath the temporal or inferior horn of the lateral ventricle.
The occipitotemporal sulcus rarely extends posteriorly as far as the occipital pole; both the collateral and occipitotemporal sulci frequently have secondary side branches and merge anteriorly. The inferior or basal aspect of the inferior occipital gyrus lies lateral to the fusiform gyrus and constitutes the most inferior portion of the lateral aspect of the occipital lobe. Along the inferolateral border of the hemisphere, the inferior temporal gyrus is continuous with the inferior occipital gyrus over the preoccipital notch, and the inferior occipital gyrus is continuous with the lingual gyrus along the occipital pole. Along the parietal and occipital aspects of the superomedial border of the hemisphere, the superior parietal lobule is continuous with the precuneus, and the superior occipital gyrus is continuous with the cuneus above the calcarine sulcus and with the lingual gyrus below the calcarine sulcus.
Occipital lobe internal structure and connectivity
The occipital lobe is composed almost entirely of Brodmann's areas 17, 18 and 19. Area 17, the striate cortex, is the primary visual cortex (VI). A host of other distinct visual areas reside in the occipital and temporal cortex. Functional subdivisions V2, V3 (dorsal and ventral) and V3A lie within Brodmann's area 18. Other functional areas at the junction of the occipital cortex with the parietal or temporal lobes lie wholly or partly in area 19.
The primary visual cortex is mostly located on the medial aspect of the occipital lobe and is coextensive with the subcortical nerve fibre stria of Gennari in layer IV; hence its alternative name, the striate cortex. It occupies the upper and lower lips and depths of the posterior part of the calcarine sulcus and extends into the cuneus and lingual gyrus.
The primary visual cortex receives afferent fibres from the lateral geniculate nucleus via the optic radiation (Fig. 25.19). The latter curves posteriorly and spreads through the white matter of the occipital lobe. Its fibres terminate in strict point-to-point fashion in the striate area. The cortex of each hemisphere receives impulses from two hemi-retinae, which represent the contralateral half of the binocular visual field. Superior and inferior retinal quadrants are connected with corresponding areas of the striate cortex. Thus, the superior retinal quadrants (representing the inferior half of the visual field) are connected with the visual cortex above the calcarine sulcus, and the inferior retinal quadrants (representing the upper half of the visual field) are connected with the visual cortex below the calcarine sulcus. The peripheral parts of the retinae activate the most anterior parts in the visual cortex. The macula impinges on a disproportionately large posterior part around the occipital pole.
The striate cortex is granular. Layer IV, bearing the stria of Gennari, is commonly divided into three sublayers. Passing from superficial to deep, these are IVA, IVB (which contains the stria) and IVC. The densely cellular IVC is further subdivided into a superficial IVCα and a deep IVCβ. Layer IVB contains only sparse, mainly non-pyramidal neurones. The input to area 17 from the lateral geniculate nucleus terminates predominantly in layers IVA and IVC. Other thalamic afferents, from the inferior pulvinar nucleus and the intralaminar group, pass to layers I and VI. Geniculocortical fibres terminate in alternating bands. Axons from geniculate laminae that receive information from the ipsilateral eye (laminae 2, 3 and 5) are segregated from those of laminae that receive input from the contralateral eye (laminae 1, 4 and 6). Neurones within layer IVC are monocular, i.e. they respond to stimulation of either the ipsilateral or contralateral eye, but not both. This horizontal segregation forms the anatomical basis of the ocular dominance column in that neurones encountered in a vertical strip from pia to white matter, although binocular outside layer IV, exhibit a preference for stimulation of one or other eye. The other major functional basis for visual cortical columnar organization is the orientation column. This describes the observation that an electrode passing through the depth of the cortex, at right angles to the plane from pia to white matter, encounters neurones that all respond preferentially to either a stationary or a moving straight line of a given orientation within the visual field. Cells with simple, complex and hypercomplex receptive fields occur in area 17. Simple cells respond optimally to lines in a narrowly defined position. Complex cells respond to a line anywhere within a receptive field, but with a specific orientation. Hypercomplex cells are similar to complex cells except that the length of the line or bar stimulus is also critical for an optimal response. There is a relationship between the complexity of response and the position of cells in relation to the cortical laminae. Simple cells are mainly in layer IV, and complex and hypercomplex cells predominate in either layers II and III or layers V and VI.
Ipsilateral corticocortical fibres pass from area 17 to a variety of functional areas in areas 18 and 19 and in the parietal and temporal cortices. Fibres from area 17 pass to area 18 (which contains visual areas V2, V3 and V3a); area 19 (which contains V4); the posterior intraparietal and the parieto-occipital areas; and to parts of the posterior temporal lobe, the middle temporal area and the medial superior temporal area. Subcortical efferents of the striate cortex pass to the superior colliculus, pretectum and parts of the brainstem reticular formation. Projections to the striatum (notably the tail of the caudate nucleus) and to the pontine nuclei are sparse. Geniculo- and claustrocortical projections are reciprocated by prominent descending projections, which arise in layer VI.
The second visual area (V2) occupies much of area 18 but is not coextensive with it. It contains a complete retinotopic representation of the visual hemifield, which is a mirror image of that in area 17, with the vertical meridian represented most posteriorly along the border between areas 17 and 18. The major ipsilateral corticocortical feed-forward projection to V2 comes from V1. Feed-forward projections from V2 pass to several other visual areas (and are reciprocated by feedback connections), including the third visual area (V3) and its various subdivisions (V3/V3d; VP/V3v; V3a); the fourth visual area (V4); areas in the temporal and parietal association cortices; and the frontal eye field. Thalamic afferents to V2 come from the lateral geniculate nucleus, the inferior and lateral pulvinar nuclei and parts of the intralaminar group of nuclei. Additional subcortical afferents are as for cortical areas in general. Subcortical efferents arise predominantly in layers V and VI. They pass to the thalamus, claustrum, superior colliculus, pretectum, brainstem reticular formation, striatum and pons. As for area 17, the callosal connections of V2 are restricted predominantly to the cortex, which contains the representation of the vertical meridian.
The third visual area (V3) is a narrow strip adjoining the anterior margin of V2, probably still within area 18 of Brodmann. V3 has been subdivided into dorsal (V3/V3d) and ventral (VP/V3v) regions on the basis of its afferents from area V1, myeloarchitecture, callosal and association connections, and receptive field properties. The dorsal subdivision receives from V1, whereas the ventral does not. Functionally, the dorsal part shows less wavelength selectivity, greater direction selectivity and smaller receptive fields than does the ventral subdivision. Both areas receive a feed-forward projection from V2 and are interconnected by association fibres. A further visual area, area V3a, lies anterior to the dorsal subdivision of V3. It receives afferent association connections from V1, V2, V3/V3d and VP/V3v, and has a complex and irregular topographic organization. All subdivisions project to diverse visual areas in the parietal, occipital and temporal cortices, including V4, and to the frontal eye fields.
The fourth visual area, V4, lies within area 19 anterior to the V3 complex. It receives a major ipsilateral feed-forward projection from V2. Colour selectivity as well as orientation selectivity may be transmitted to V4 and bilateral damage causes achromatopsia. V4 is more complex than a simple colour discrimination area because it is also involved in the discrimination of orientation, form and movement. It sends a feed-forward projection to the inferior temporal cortex and receives a feedback projection. It also connects with other visual areas that lie more dorsally in the temporal lobe, and in the parietal lobe. Thalamocortical connections are with the lateral and inferior pulvinar and the intralaminar nuclei. Other subcortical connections conform to the general pattern for all cortical areas. Callosal connections are with the contralateral V4 and other occipital visual areas.
Visual processing in inferior temporal and temporoparietal cortices involves two parallel pathways (dorsal and ventral visual streams) that emanate from the occipital lobe and are specialized for action and perception respectively (Goodale and Milner 1992, Goodale et al 2005). The dorsal pathway, concerned primarily with visuospatial discrimination, projects from V1 and V2 to the superior temporal and surrounding parietotemporal areas, and ultimately to area 7a of the parietal cortex; it mediates the sensory-motor transformations required to enable visually guided actions directed at an object. The fourth visual area, V4, is a key relay station for the ventral stream of projections, which is related to the perceptual identification of objects. Its connections pass sequentially along the inferior temporal gyrus in a feed-forward manner, from V4 to posterior, intermediate and then anterior, inferior temporal cortices. Ultimately, they feed into the temporal polar and medial temporal areas and so interface with the limbic system.
Temporal Lobe
The lateral surface of the temporal lobe is inferior to the lateral fissure, and lies beneath the squamous portion of the temporal bone. The basal surface lies over the floor of the middle cranial fossa posterior to the greater wing of the sphenoid bone. The opercular surface lies inside the lateral fissure (see Figs 25.1, 25.9![]() ). Along the superolateral surface of the hemisphere, the posterior limit of the temporal lobe is arbitrarily defined by imaginary lines that run from the superomedial part of the parieto-occipital sulcus to the preoccipital notch or incisure, and by a posterior prolongation of the lateral fissure. Functionally, the temporal lobe is bilaterally related with auditory functions and in the dominant hemisphere mostly with the comprehension of language. (For further reading on the microsurgical anatomy of the temporal lobe, see Kucukyuruk et al (2012).)
). Along the superolateral surface of the hemisphere, the posterior limit of the temporal lobe is arbitrarily defined by imaginary lines that run from the superomedial part of the parieto-occipital sulcus to the preoccipital notch or incisure, and by a posterior prolongation of the lateral fissure. Functionally, the temporal lobe is bilaterally related with auditory functions and in the dominant hemisphere mostly with the comprehension of language. (For further reading on the microsurgical anatomy of the temporal lobe, see Kucukyuruk et al (2012).)
Temporal lobe sulci and gyri
The lateral surface is composed of superior, middle and inferior temporal gyri (T1, T2 and T3 respectively), separated by the superior and inferior temporal sulci and all lying parallel to the lateral fissure (see Figs 25.1, 25.8). Anteriorly, the middle temporal gyrus is generally shorter; when this occurs, the superior and inferior gyri come together to form the temporal pole (see Fig. 25.9B; Fig. 25.20A![]() ).
).
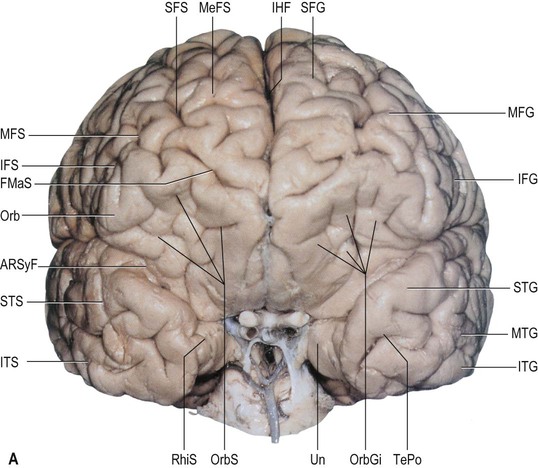
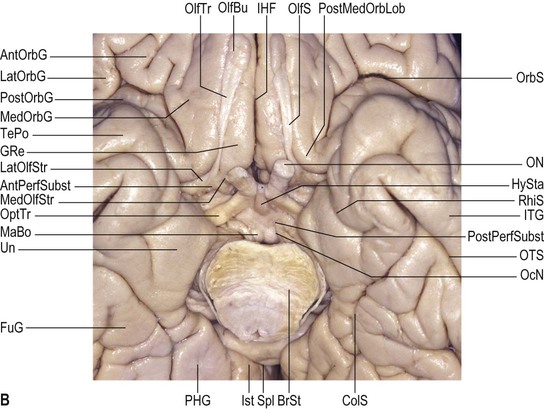
The superior temporal sulcus is well defined and deep. Typically, it is interrupted and composed of up to four segments but it may be continuous (Ochiai et al 2004). It terminates within the inferior parietal lobule, posterior to the end of the lateral fissure, by trifurcating into an ascending sulcal segment and an inferior branch that runs towards the occipital lobe. The superior temporal gyrus always continues posteriorly to the supramarginal gyrus encircling the terminal portion of the lateral fissure. The middle temporal gyrus is always connected to the angular gyrus beneath the distal and horizontal portion of the superior temporal sulcus. The inferior temporal gyrus is continuous posteriorly with the inferior occipital gyrus over the preoccipital notch. The inferior temporal sulcus is usually discontinuous. Both superior and inferior temporal sulci start at the most anterior aspect of the temporal pole and end posterior to the arbitrary border of the temporal lobe; both of them have their depths directed towards the inferior or temporal horn of the lateral ventricle. Topographically, the depth of the posterior part of the superior temporal sulcus is particularly related to the ventricular atrium.
The basal surface of the temporal lobe is composed laterally by the inferior surface of the inferior temporal gyrus and medially by the anterior or temporal portion of the fusiform or lateral temporo-occipital gyrus; the gyri are separated by the temporo-occipital sulcus. Medially, the fusiform gyrus is delimited by the collateral sulcus (inferior longitudinal sulcus of Huschke), which separates it from the parahippocampal gyrus of the limbic lobe (see Fig. 25.18). Short and secondary sulci (fusiform sulci) are found within its surface. Although not extensive, the fusiform gyrus has an anterior or temporal part, T4 (between the inferior and parahippocampal gyri), and a posterior or occipital part, O4 (between the inferior occipital and lingual gyri). The anterior part of the fusiform gyrus is typically curved or pointed, resembling an arrow; its anterior border usually lies close to the level of the cerebral peduncle. The temporal portion of the fusiform gyrus lies over the posterior aspect of the floor of the middle fossa and the upper surface of the petrous part of the temporal bone. The occipital part of the fusiform gyrus lies underneath the ventricular atrium.
Anterior to the fusiform gyrus, the collateral sulcus may be continuous with the rhinal sulcus. Alternatively, and more frequently, these sulci are separated by the so-called temporolimbic passage. The rhinal sulcus separates the entorhinal cortex of the uncus medially from the neocortex of the temporal pole laterally.
The superior or opercular surface of the temporal lobe is formed by the superior surface of the superior temporal gyrus, which lies within the lateral fissure and is composed of multiple transverse gyri. One of these is the voluminous transverse gyrus. It originates around the midpoint of the superior temporal gyrus and is orientated diagonally towards the posterior vertex of the floor of the lateral fissure, with its longest axis orientated towards the ventricular atrium. This gyrus, which may be single or double, is Heschl's gyrus; it is bounded anteriorly by the first transverse sulcus and posteriorly by the more defined sulcus of Heschl, and divides the temporal opercular surface into an anterior, polar plane and a posterior, temporal plane. The sulcus acusticus, an infrequent, small superior extension of the superior temporal sulcus anterior to Heschl's gyrus, indicates the anterior aspect of Heschl's gyrus on the superolateral surface of the temporal lobe. If the transverse temporal sulcus, which lies posterior to Heschl's sulcus within the temporal plane, reaches the lateral surface of the superior temporal gyrus, it indicates the posterior aspect of Heschl's gyrus. The transverse gyrus of Heschl and the most posterior aspect of the superior temporal gyrus correspond to the primary auditory cortex.
The surface of the polar plane is composed of multiple transverse gyri directed obliquely towards the inferior part of the insular circular sulcus (inferior limiting sulcus) (Fig. 25.21A). The temporal plane is flat, perpendicular to the brain surface, and triangular in shape. Its internal vertex corresponds to the posterior vertex of the base of the lateral (Sylvian) fissure, at the point where the superior part of the insular circular sulcus (superior limiting sulcus) meets the inferior part of the insular circular sulcus (inferior limiting sulcus), lying immediately over the atrium. The temporal plane is usually larger in the dominant hemisphere, supposedly reflecting its association with language functions (Geschwind and Levitsky 1968). Topographically, the oblique polar plane covers the insular surface, Heschl's gyrus underlies the opercular surface of the postcentral gyrus, and the flat surface of the temporal plane underlies the opercular surface of the supramarginal gyrus.
Temporal lobe internal structure and connectivity
The superior, middle and inferior temporal gyri correspond to Brodmann's areas 22, 21 and 20 respectively, and the temporal pole corresponds to area 38. The anterior transverse temporal gyrus and adjoining part of the superior temporal gyrus are auditory in function, and are considered to be Brodmann's area 42. The anterior gyrus is approximately area 41.
The temporal operculum houses the primary auditory cortex, AI. This is coextensive with the granular area 41 in the transverse temporal gyri. Surrounding areas constitute auditory association cortex. The primary auditory cortex is reciprocally connected with all subdivisions of the medial geniculate nucleus, and may receive additional thalamocortical projections from the medial pulvinar. The geniculocortical fibres terminate densely in layer IV. AI contains a tonotopic representation of the cochlea in which high frequencies are represented posteriorly and low frequencies anteriorly. Single-cell responses are to single tones of a narrow frequency band. Cells in single vertical electrode penetrations share an optimum frequency response.
The auditory cortex interconnects with prefrontal cortex, though the projections from AI are small. In general, posterior parts of the operculum project to areas 8 and 9. Central parts project to areas 8, 9 and 46. More anterior regions project to areas 9 and 46, to area 12 on the orbital surface of the hemisphere, and to the anterior cingulate gyrus on the medial surface. Contralateral corticocortical connections are with the same and adjacent regions in the other hemisphere. Onward connections of the auditory association pathway converge with those of the other sensory association pathways in cortical regions within the superior temporal sulcus.
The posterior cortical area of the dominant hemisphere, Wernicke's area, is intrinsically related to the auditory cortex. It is particularly responsible for the comprehension of language but its stimulation causes speech arrest. Wernicke's area is not well defined anatomically but occupies mainly the posterior aspect of the superior temporal gyrus and the basal aspect of the supramarginal gyrus. It may extend inferiorly along the middle temporal gyrus and anteriorly to within 3 cm of the temporal pole (Ojemann et al 1989).
Evidence suggests that area 21, the middle temporal cortex, is polysensory in humans, and that it connects with auditory, somatosensory and visual cortical association pathways. The auditory association areas of the superior temporal gyrus project in a complex ordered fashion to the middle temporal gyrus, as does the parietal cortex. The middle temporal gyrus connects with the frontal lobe: the most posterior parts project to posterior prefrontal cortex, areas 8 and 9, while intermediate regions connect more anteriorly with areas 19 and 46. Further forwards, the middle temporal region has connections with anterior prefrontal areas 10 and 46, and with anterior orbitofrontal areas 11 and 14. The most anterior middle temporal cortex is connected with the posterior orbitofrontal cortex, area 12, and with the medial surface of the frontal pole. Further forwards, this middle temporal region projects to the temporal pole and the entorhinal cortex. Thalamic connections are with the pulvinar nuclei and the intralaminar group. Other subcortical connections follow the general pattern for all cortical areas. Some projections (e.g. to the pons), particularly from anteriorly in the temporal lobe, are minimal. Physiological responses of cells in this middle temporal region show convergence of different sensory modalities, and many neurones respond to faces.
The inferior temporal cortex, area 20, is a higher visual association area. The posterior inferior temporal cortex receives major ipsilateral corticocortical fibres from occipitotemporal visual areas, notably V4. It contains a coarse retinotopic representation of the contralateral visual field, and sends a major feed-forward pathway to the anterior part of the inferior temporal cortex. The anterior inferior temporal cortex projects on to the temporal pole and to paralimbic areas on the medial surface of the temporal lobe. Additional ipsilateral association connections of the inferior temporal cortex are with the anterior middle temporal cortex, in the walls of the superior temporal gyrus, and with visual areas of the parietotemporal cortex. Frontal lobe connections are with area 46 in the dorsolateral prefrontal cortex (posterior inferior temporal) and with the orbitofrontal cortex (anterior inferior temporal). The posterior area also connects with the frontal eye field. Reciprocal thalamic connections are with the pulvinar nuclei; the posterior part is related mainly to the inferior and lateral nuclei, and the anterior part to the medial and adjacent lateral pulvinar. Intralaminar connections are with the paracentral and central medial nuclei. Other subcortical connections conform to the general pattern of all cortical regions. Callosal connections are between corresponding areas and the adjacent visual association areas of each hemisphere.
The cortex of the temporal pole receives feed-forward projections from widespread areas of temporal association cortex that are immediately posterior to it. The dorsal part receives predominantly auditory input from the anterior part of the superior temporal gyrus. The inferior part receives visual input from the anterior area of the inferior temporal cortex. Other ipsilateral connections are with the anterior insular, the posterior and medial orbitofrontal, and the medial prefrontal cortices. The temporal pole projects onwards into limbic and paralimbic areas. Thalamic connections are mainly with the medial pulvinar nucleus and with intralaminar and midline nuclei. Other subcortical connections are as for the cortex in general, although some projections, such as that to the pontine nuclei, are very small. Physiological responses of cells in this and more medial temporal cortex correspond particularly to behavioural performance and to the recognition of high-level aspects of social stimuli.
The cortex of the medial temporal lobe includes major subdivisions of the limbic system, such as the hippocampus and entorhinal cortex. Areas of neocortex adjacent to these limbic regions are grouped together as medial temporal association cortex.
Nuclei of the amygdala project to, and receive fibres from, neocortical areas, predominantly of the temporal lobe, and possibly inferior parietal cortex. The density of these pathways increases towards the temporal pole.
Insular Lobe
The insula forms the base of the Sylvian cistern and lies between the frontal and temporal lobes (see Fig. 25.21; Fig. 25.22![]() ).
).
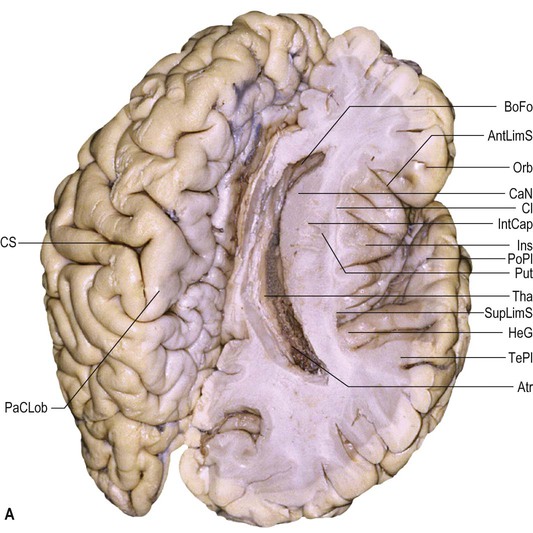
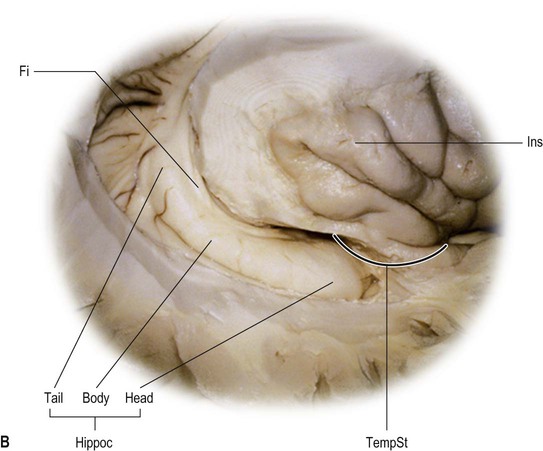
Insular lobe sulci and gyri
The insula has both lateral and anterior surfaces. The superior and inferior limiting sulci are morphologically true sulci that delineate the lateral insular surface from the frontoparietal operculum, the lateral insular surface and the temporal operculum (Türe et al 1999). The anterior limit of the insula is considerably deeper and morphologically characteristic of a true fissure or space and separates the anterior surface of the insula from the fronto-orbital operculum. The upper half of the fundus of the anterior limiting sulcus is separated from a true anterior recess of the lateral ventricle, anterior to the head of the caudate nucleus, by the fibres of the thin anterior limb of the internal capsule, whereas the fundus of the lower half continues to the ventral striatopallidal or anterior perforated substance region (Heimer 2003).
The anterior surface of the insula is covered by the fronto-orbital operculum (the posterior portion of the posterior orbital gyrus and the orbital part of the inferior frontal gyrus). Its lateral surface is covered superiorly by the frontoparietal operculum (triangular and opercular parts of the inferior frontal gyrus, subcentral gyrus, and anterior and basal part of the supramarginal gyrus) and inferiorly by the temporal operculum (polar plane of the superior temporal gyrus; see Fig. 25.14).
The lateral surface of the insula may be conceptualized as a pyramid with a triangular base; its anteroinferior vertex is the limen insulae and its summit is the insular apex. The limen insulae consists of a narrow strip of olfactory cortex that extends along the lateral aspect of the lateral olfactory stria, conjoining the insular cortex and anterior perforated substance. The surface of the insula is divided into anterior and posterior parts by a deep central sulcus that courses obliquely from the limen insulae towards the central sulcus of the cerebral hemisphere. The anterior part of the insular surface is composed of transverse, accessory and short insular gyri (anterior, middle and posterior short insular gyri), all arising from the region of the insular apex. The middle and posterior short insular gyri are separated by the precentral insular sulcus.
The transverse insular gyrus runs along the limen insulae, connecting the anterior insula with the posteromedial orbital lobule. The latter is the connection between the posterior portion of the medial orbital gyrus and the posterior orbital gyrus, and is located anterior to and along the lateral olfactory stria. The accessory gyrus extends from the anterior portion of the anterior short gyrus superiorly to the transverse insular gyrus, beneath the fronto-orbital operculum. Both gyri constitute the insular pole within the anterior aspect of the insula (Türe et al 1999, Türe et al 2000). The posterior part of the insula is located behind its central sulcus and is composed of anterior and posterior insular long gyri. Both of these gyri are separated by the postcentral insular sulcus; the anterior long gyrus is usually larger, and may be single and divided at its upper end.
The insular surface is delineated peripherally by the peri-insular sulcus (circular sulcus of Reil), which is interrupted by the transverse insular gyrus running across the limen insulae. Given the triangular shape of the insula, the peri-insular sulcus is usually divided into three parts, variously named either as the anterior, superior and inferior peri-insular sulci (Türe et al 1999), or the anterior, superior and inferior limiting sulci of the insula (Rhoton 2003).
Insular lobe internal structure and connectivity
Cytoarchitectonically, three zones are recognized within the insula. Anteriorly, and extending caudally into the central insula, the cortex is agranular. It is surrounded by a belt of dysgranular cortex, in which laminae II and III can be recognized, and this in turn is surrounded by an outer zone of homotypical granular cortex that extends to the caudal limit of the insula.
Thalamic afferents to the insula come from subdivisions of the ventral posterior nucleus and of the medial geniculate body, from the oral and medial parts of the pulvinar, the suprageniculate/nucleus limitans complex, the mediodorsal nucleus and the nuclei of the intralaminar and midline groups. It appears that the anterior (agranular) cortex is connected predominantly with the mediodorsal and ventroposterior nuclei, while the posterior (granular) cortex is connected predominantly with the pulvinar and the ventral posterior nuclei. The other nuclear groups appear to connect with all areas.
Ipsilateral cortical connections of the insula are diverse. Somatosensory connections are with SI, SII and surrounding areas, and areas 5 and 7b of the parietal lobe. The insular cortex also has connections with the orbitofrontal cortex. Several auditory regions in the temporal lobe interconnect with the posterior granular insula and the dysgranular cortex more anteriorly. Connections with visual areas are virtually absent. The anterior agranular cortex of the insula appears to have connections primarily with olfactory, limbic and paralimbic structures, including, most prominently, the amygdala.
The insula receives somatosensory, viscerosensory, homeostatic and nociceptive information from the entire body. (For a discussion of the role of the operculo-insular region in the processing of somatosensory inputs, see Mazzola et al (2012).)
Limbic Lobe
Limbus means edge or border; the descriptive term limbic was first used in the sixteenth century (Morgane and Mokler 2006), but is more usually associated with Broca, who described the cingulate and parahippocampal gyri as the greater limbic lobe, and considered the different sulci that limited these two gyri as parts of a single sulcus that he called the limbic sulcus (Broca 1877). On phylogenetic grounds, Broca considered that these structures might be associated with olfaction and for many years the term rhinencephalon (‘smell brain’) was used synonymously with limbic lobe; the term is no longer used.
The limbic lobe contains the cortical structures on the most medial edge of the hemisphere and includes the cingulate, parahippocampal and dentate gyri, the cingulate isthmus, the subcallosal area (paraolfactory and paraterminal gyri), the collateral and rhinal sulci, the uncus and the hippocampal formation. (The Terminologia Anatomica restricts the hippocampal representation in the limbic lobe to the fimbria of the hippocampus; other authors include the hippocampal formation (e.g. Chronister and Hardy 2006).)
Limbic lobe sulci and gyri
Within the inner aspect of the medial surface of each cerebral hemisphere, the cingulate gyrus wraps around the corpus callosum and continues posteriorly and inferiorly to the parahippocampal gyrus (see Fig. 25.10B![]() ). Lying above the callosal sulcus and below the cingulate sulcus, the cingulate gyrus starts within the subcallosal area below the rostrum of the corpus callosum and ascends around the genu of the corpus callosum, frequently connecting with the medial aspect of the superior frontal gyrus in its course. It is connected to the paracentral lobule as it lies over the body of the corpus callosum; more posteriorly, it is connected to the precuneus. Posterior to the splenium of the corpus callosum, it narrows to become the isthmus, and continues to the parahippocampal gyrus. Anteriorly and basally, the cingulate sulcus may be continuous with the anterior paraolfactory sulcus under the rostrum of the corpus callosum; when this occurs, the cingulate gyrus is continuous with the paraolfactory or subcallosal gyri. The cingulate sulcus may be double and parallel; the additional superior fold is the paracingulate sulcus.
). Lying above the callosal sulcus and below the cingulate sulcus, the cingulate gyrus starts within the subcallosal area below the rostrum of the corpus callosum and ascends around the genu of the corpus callosum, frequently connecting with the medial aspect of the superior frontal gyrus in its course. It is connected to the paracentral lobule as it lies over the body of the corpus callosum; more posteriorly, it is connected to the precuneus. Posterior to the splenium of the corpus callosum, it narrows to become the isthmus, and continues to the parahippocampal gyrus. Anteriorly and basally, the cingulate sulcus may be continuous with the anterior paraolfactory sulcus under the rostrum of the corpus callosum; when this occurs, the cingulate gyrus is continuous with the paraolfactory or subcallosal gyri. The cingulate sulcus may be double and parallel; the additional superior fold is the paracingulate sulcus.
Anteriorly, the parahippocampal gyrus folds back on itself medially to form the uncus, which is incorporated superiorly into the most lateral aspect of the frontobasal region via a well-defined neural peduncle anterior to the inferior horn of the lateral ventricle (see Figs 25.18![]() and 25.20B). Along its axial extension, the basal and medial surface of the parahippocampal gyrus curves laterally, forming a flat superior surface, the subiculum. The latter is slightly triangular with an anterior vertex; it corresponds to the floor of the lateral part of the transverse fissure. This portion of the transverse fissure harbours the so-called lateral wing of the ambient cistern. Laterally, the parahippocampal gyrus is contiguous with the fusiform gyrus underneath the depths of the collateral sulcus. Posteriorly, it is continuous with the lingual gyrus and the cingulate gyrus along its isthmus. Medially, it lies under the thalamus along the choroidal fissure. Superiorly, it is attached along the inferior aspect of the insular lobe via fibres that cover the inferior horn.
and 25.20B). Along its axial extension, the basal and medial surface of the parahippocampal gyrus curves laterally, forming a flat superior surface, the subiculum. The latter is slightly triangular with an anterior vertex; it corresponds to the floor of the lateral part of the transverse fissure. This portion of the transverse fissure harbours the so-called lateral wing of the ambient cistern. Laterally, the parahippocampal gyrus is contiguous with the fusiform gyrus underneath the depths of the collateral sulcus. Posteriorly, it is continuous with the lingual gyrus and the cingulate gyrus along its isthmus. Medially, it lies under the thalamus along the choroidal fissure. Superiorly, it is attached along the inferior aspect of the insular lobe via fibres that cover the inferior horn.
The indusium griseum is a thin layer of grey matter lying over the corpus callosum and covered by medial and lateral longitudinal striae running within the callosal sulcus beneath each cingulate gyrus. Anteriorly, the indusium griseum is connected to the paraterminal gyrus via the prehippocampal rudiment. Posteriorly, it circles the splenium of the corpus callosum and runs along the fasciolar gyrus on each side.
In the subcallosal area, the anterior and posterior paraolfactory gyri lie anterior to the paraterminal gyrus, separated by the anterior paraolfactory sulcus. Anteriorly, a consistent fold connects the most basal portion of the cingulate gyrus with the gyrus rectus, encircling the posterior end of the superior rostral sulcus, called the cingulate pole (Yasargil 1994). The paraterminal gyrus lies on the medial wall of the cerebral hemisphere posterior to the paraolfactory gyri, immediately facing the lamina terminalis, and delineated anteriorly by the short, vertical posterior olfactory sulcus. Inferiorly, the paraterminal gyrus extends along the diagonal band of Broca and the lateral olfactory stria.
The choroidal fissure extends between the entire fornix and thalamus, from the inferior choroidal point between the head and the body of the hippocampus to the interventricular foramen. Anterior to the inferior choroidal point (the point at which the anterior choroidal artery enters the temporal pole of the lateral ventricle (Tubbs et al 2010)), the anterior and mesial parts of the temporal lobe and the parahippocampal gyrus merge with the basal and lateral aspect of the frontal lobe through a neural peduncle that constitutes the so-called temporal stem (see below). Posterior to the inferior choroidal point, the choroidal fissure lies within the inferior horn between the fimbria of the fornix and the inferior aspect of the thalamus, along the parapeduncular space that harbours the ambient cistern. More posteriorly, the choroidal fissure within the atrium lies between the crura and the pulvinar, lateral to the quadrigeminal or pineal cistern. More superiorly and anteriorly, the choroidal fissure lies between the body of the fornix and the superior surface of the thalamus.
The collateral sulcus is a long, deep sulcus that extends along the basal temporal and occipital surface, with multiple side branches. Its temporal segment bulges into the ventricular floor as the collateral eminence, located lateral to the hippocampus. Its occipital segment corresponds to the collateral trigone that forms the triangular, flat surface of the ventricular atrium and posterior horn. The rhinal sulcus, which is not always readily identifiable, separates the uncus from the rest of the temporal pole. The subcallosal, cingulate, subparietal, anterior calcarine, collateral and rhinal sulci are frequently considered to form the limbic fissure.
Uncus
The uncus is triangular. It has a medial vertex; its anteromedial surface faces the carotid cistern and its posteromedial surface faces and encircles the mesencephalic peduncle (see Fig. 25.18![]() ). The semilunar gyrus, a lateral extension of the lateral olfactory stria, and the ambient gyrus form two small prominences on its medial and anterior surface. Both gyri cover the amygdala and are separated by the semi-annular sulcus, which harbours the anterior choroidal artery. Inferiorly, the free edge of the tentorium cerebelli is frequently associated with another depression. The most rostral and anterior aspects of the uncus correspond to the entorhinal cortex. The posterior half of the uncus contains the head of the hippocampus and is separated inferiorly from the parahippocampal gyrus by the uncal sulcus. The medial surface of the uncus faces the crural cistern. Its inferior surface is hidden inside the uncal sulcus, in which the posterior half of the uncus harbours the uncinate gyrus anteriorly and the uncal apex (intralimbic gyrus, hippocampus inversus) posteriorly, separated by the band of Giacomini. Collectively, these structures correspond to the extraventricular part of the head of the hippocampus. The band of Giacomini corresponds to the tail of the dentate gyrus, which vanishes on the medial aspect of the uncus. Along the cerebral base, the parahippocampal gyrus is delineated laterally by the collateral sulcus, separating it from the fusiform gyrus, and more anteriorly by the rhinal sulcus, which is occasionally continuous with the collateral sulcus.
). The semilunar gyrus, a lateral extension of the lateral olfactory stria, and the ambient gyrus form two small prominences on its medial and anterior surface. Both gyri cover the amygdala and are separated by the semi-annular sulcus, which harbours the anterior choroidal artery. Inferiorly, the free edge of the tentorium cerebelli is frequently associated with another depression. The most rostral and anterior aspects of the uncus correspond to the entorhinal cortex. The posterior half of the uncus contains the head of the hippocampus and is separated inferiorly from the parahippocampal gyrus by the uncal sulcus. The medial surface of the uncus faces the crural cistern. Its inferior surface is hidden inside the uncal sulcus, in which the posterior half of the uncus harbours the uncinate gyrus anteriorly and the uncal apex (intralimbic gyrus, hippocampus inversus) posteriorly, separated by the band of Giacomini. Collectively, these structures correspond to the extraventricular part of the head of the hippocampus. The band of Giacomini corresponds to the tail of the dentate gyrus, which vanishes on the medial aspect of the uncus. Along the cerebral base, the parahippocampal gyrus is delineated laterally by the collateral sulcus, separating it from the fusiform gyrus, and more anteriorly by the rhinal sulcus, which is occasionally continuous with the collateral sulcus.
Hippocampus
The hippocampus (hippocampus proper, cornu ammonis, Ammon's horn) is a convex elevation, approximately 5 cm long, within the parahippocampal gyrus inside the inferior (temporal) horn of the lateral ventricle (Duvernoy 1998) (Figs 25.23–25.24). Macroscopically, it can be divided into a head, a body and a tail (see Fig. 25.22B![]() ). Anteriorly, the head is expanded and bears two or three shallow grooves (pes hippocampi). The surface of the hippocampus, the alveus, is covered by the ependyma inside the ventricular cavity.
). Anteriorly, the head is expanded and bears two or three shallow grooves (pes hippocampi). The surface of the hippocampus, the alveus, is covered by the ependyma inside the ventricular cavity.
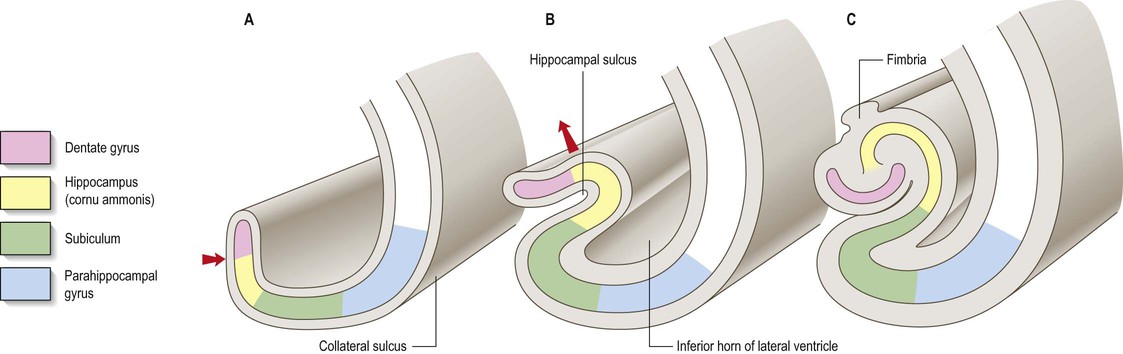
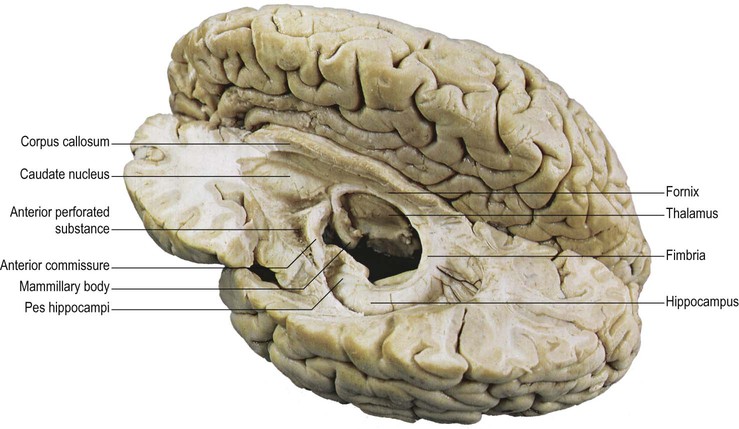
The dentate gyrus consists of small cortical prominences that form a chain along the medial aspect of the hippocampus (Fig. 25.25). Along its medial margin, it is separated from the subiculum of the parahippocampal gyrus by the hippocampal sulcus, usually a shallow sulcus that terminates anteriorly within the uncus (see Fig. 25.23). More superiorly and medially, the dentate gyrus is separated from the fimbria of the fornix by the fimbriodentate sulcus lying parallel to the hippocampal sulcus (Fig. 25.26).
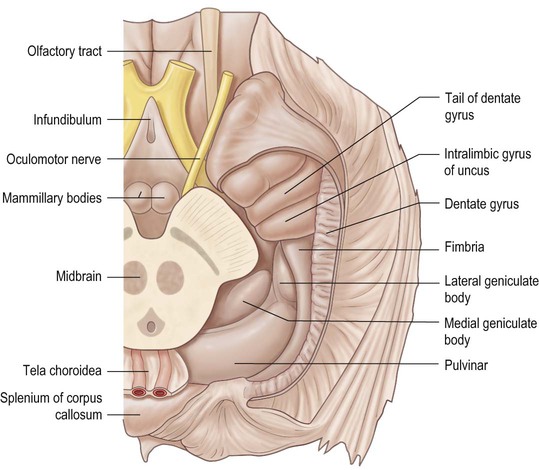
The main outflow bundle of the hippocampus, the fornix, wraps round the thalamus, from which it is separated by the choroidal fissure and the choroid plexus (see Fig. 25.3). It has several named parts: fimbria, crus, body and column (pillar). The fibres of the alveus converge to form the fimbria along the medial portion of the floor of the inferior horn of the lateral ventricle. At a point beneath the splenium of the corpus callosum, the white matter of the fimbria separates from the hippocampus to become the crus of the ipsilateral fornix. The two crura pass upwards and forwards beneath the corpus callosum, mirroring its arch but with a tighter curve. They are joined via fibres that cross in the commissure of the fornix (hippocampal commissure). Beyond this point, the crura merge to become the body of the fornix, which continues anteriorly within the roof of the third ventricle, below the lower border of the septum pellucidum and near the midline (Figs 25.27–25.28![]() ). At the level of the interventricular foramen (foramen of Munro) and the anterior commissure, the most anterior segment of the body diverges from its contralateral counterpart, and passes in an anterior, lateral and inferior direction as the column (pillar) of the fornix to penetrate the hypothalamic parenchyma towards the ipsilateral mammillary body. As they separate from the underlying thalamus, the columns of the fornix form the anterior margins of the interventricular foramen.
). At the level of the interventricular foramen (foramen of Munro) and the anterior commissure, the most anterior segment of the body diverges from its contralateral counterpart, and passes in an anterior, lateral and inferior direction as the column (pillar) of the fornix to penetrate the hypothalamic parenchyma towards the ipsilateral mammillary body. As they separate from the underlying thalamus, the columns of the fornix form the anterior margins of the interventricular foramen.
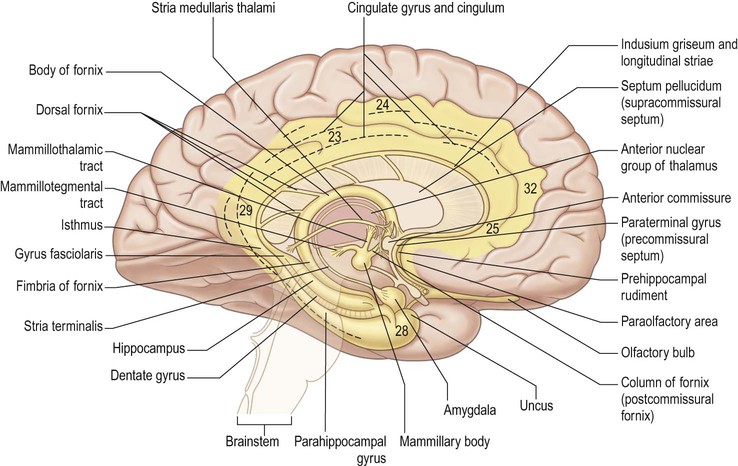
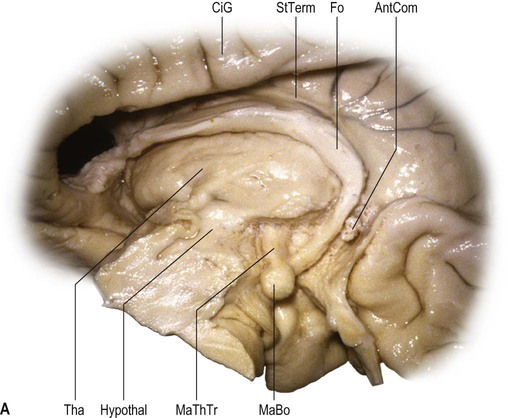
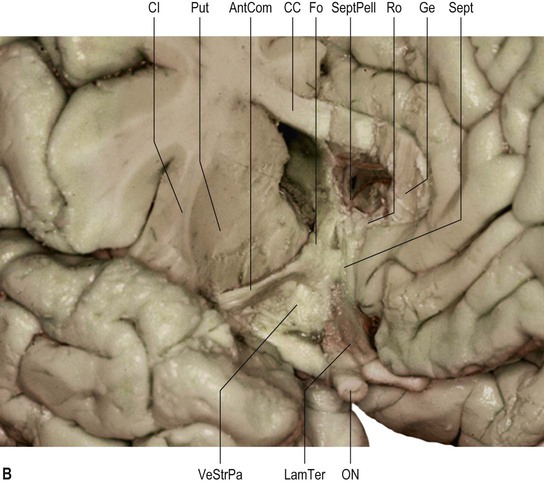
Limbic lobe internal structure and connectivity
Cingulate and parahippocampal cortex
The cingulate gyrus may be divided rostrocaudally into several cytoarchitectonically discrete areas: prelimbic (area 32) and infralimbic (area 25) cortices, anterior cingulate cortex (areas 23, 24, 32 and 33) and part of the posterior cingulate or retrosplenial cortex (area 29) (Commentary 3.1![]() ).
).
The cingulate gyrus contains specific motor areas and has extensive connections with neocortical areas of the frontal lobe and with somatosensory and visual association areas of the parietal, occipital and temporal lobes. The afferents to the cingulate gyrus are predominantly from neocortical areas on the lateral surface of the hemisphere. Within the cingulate cortex, most projections pass caudally, ultimately into the parahippocampal gyrus. Through this system, afferents from widespread areas of association cortex converge on the medial temporal lobe and hippocampal formation. There are parallel stepwise routes to these targets through cortical areas on the lateral surface. The anterior cingulate gyrus appears to be a component of a functional circuit that mediates pain perception and processing. The cingulate motor cortex subregion of the anterior cingulate gyrus is one of the first cortical brain regions activated in pain processing; its probable function is to mediate withdrawal actions and so minimize further injury. (For further reading, see Weston (2012).) The insula has strong functional connections with anterior and dorsal subregions of the anterior cingulate gyrus (areas 24 and 32),
The parahippocampal gyrus includes areas 27, 28 (entorhinal cortex), 35, 36, 48 and 49, and temporal cortical fields. It has complex interconnections with the cingulate cortex and with the hippocampal formation. In monkeys, the infralimbic cortex (area 25) has been shown to project to areas 24a and 24b. Area 25 also has reciprocal connections with the entorhinal cortex. Projections between the paralimbic area 32 and the limbic cortex (anterior, retrosplenial and entorhinal cortex) are somewhat less prominent. Areas 24 and 29 are connected with the paralimbic posterior cingulate area 23. In primates, the parahippocampal gyrus projects to virtually all association areas of the cortex; it also provides the major funnel through which polymodal sensory inputs converge on the hippocampus.