Wound-healing physiology and factors that affect the repair process
Dorothy B. Doughty, Bonnie Sparks
Objectives
1. Compare and contrast wound-healing processes for each type of closure: primary intention, secondary intention, and tertiary intention.
2. Distinguish between partial-thickness wound repair and full-thickness wound repair, addressing the key components, phases, usual timeframes, and wound appearance.
3. Explain the difference between acute and chronic full-thickness wounds.
4. Describe four characteristics of a chronic wound.
5. Describe the role of the following cells in the wound repair process: platelets, polymorphonuclear leukocytes, macrophages, fibroblasts, endothelial cells, and keratinocytes.
6. Explain how bioactive agents (e.g., growth factors and cytokines) and extracellular matrix proteins (e.g., matrix metalloproteinases and tissue inhibitors of metalloproteinases) help to regulate the repair process.
7. Describe features and characteristics of scarless healing.
8. Distinguish between keloid and hypertrophic scarring and the treatment of each.
9. Identify conditions and comorbidities that affect the wound-healing process.
For centuries, wound healing was regarded as a mysterious process, with wound management based on practitioner preference as opposed to scientific principles. Research over the past 3 decades has contributed much information regarding the wound-healing process and the factors that facilitate it. We now know that repair is an extremely complex process involving hundreds, possibly thousands, of overlapping and “linked” processes (Goldman, 2004). It is critical for all wound care clinicians to base their interventions and recommendations on current data and to remain abreast of new findings and their implications for care. This chapter reviews the process of wound healing and discusses the implications for wound management.
Mechanism of wound healing
The ability to repair tissue damage is an important survival tool for any living organism. Regardless of the type or severity of injury, repair occurs by only two mechanisms—regeneration, or replacement of the damaged or lost tissue with more of the same, and scar formation, replacement of damaged or lost tissue with connective tissue that lacks some of the functions of the original tissue. Regeneration is the preferred mechanism of repair because normal function and appearance are maintained. Scar formation is a less satisfactory alternative and occurs when the tissues involved are incapable of regeneration. Many invertebrate and amphibian species have the ability to regenerate entire limbs. Humans have only limited capacity for regeneration, and most wounds heal by scar formation (Calvin, 1998; Mast and Schultz, 1996; Wilgus, 2007).
In humans, the mechanism of repair for any specific wound is determined by the tissue layers involved and their capacity for regeneration. Wounds that are confined to the epidermal and superficial dermal layers heal by regeneration because epithelial, endothelial, and connective tissue can be reproduced. In contrast, deep dermal structures (e.g., hair follicles, sebaceous glands, sweat glands), subcutaneous tissue, muscle, tendons, ligaments, and bone lack the capacity to regenerate; therefore loss of these structures is permanent, and wounds involving these structures must heal by scar formation (Martin, 1997; Mast and Schultz, 1996).
The standard repair mechanism for human soft tissue wounds is connective tissue (scar) formation; however, an exception to this rule is the early-gestation fetus. Intrauterine surgical procedures performed during the second trimester result in scarless healing, a phenomenon that has been consistently reproduced in laboratory studies. Interestingly, the fetus loses the ability to heal without scarring at approximately 22 to 24 weeks’ gestation. Repair during the third trimester and the postnatal period follows the “usual” rules for repair and results in scar formation. A number of differences in the molecular environment for repair in the early-gestation fetus are thought to contribute to scarless repair and are discussed throughout this chapter (Bullard et al, 2002; Dang et al, 2003; Wilgus, 2007; Yang et al, 2003).
The mechanism of healing for a specific wound depends on the tissue layers involved (partial-thickness vs. full-thickness), onset and duration (acute vs. chronic), and type of wound closure (primary, secondary, or tertiary intention).
Partial thickness/full thickness
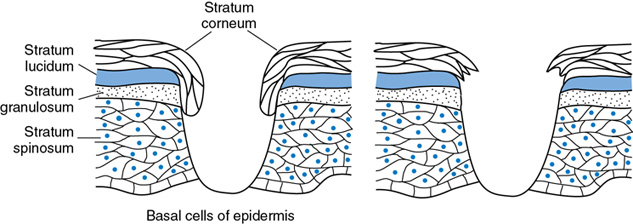
Partial-thickness wounds involve only partial loss of the skin layers; that is, they are confined to the epidermal and superficial dermal layers. Full-thickness wounds involve total loss of the skin layers (epidermis and dermis) and frequently involve loss of the deeper tissue layers as well (subcutaneous tissue, muscle, and bone) (see Figure 3-1). The timeframe for repair and the repair process itself differ significantly for partial-thickness and full-thickness wounds.
Acute/chronic
Acute wounds typically are traumatic or surgical in origin. Acute wounds occur suddenly, and in most individuals, move rapidly and predictably through the repair process and result in durable closure. In contrast, chronic wounds fail to proceed normally through the repair process. Chronic wounds frequently are caused by vascular compromise, chronic inflammation, or repetitive insults to the tissue, and they either fail to close in a timely manner or fail to result in durable closure (Brissett and Hom, 2003; Pradhan et al, 2009).
Primary, secondary, tertiary
Classification of repair as primary-, secondary-, or tertiary-intention healing is based on the ideal of primary surgical closure for all wounds. Primary closure minimizes the volume of connective tissue deposition required for wound repair and restores the epithelial barrier to infection (Figure 4-1, A). Approximated surgical incisions are said to heal by primary intention; they usually heal quickly, with minimal scar formation, as long as infection and secondary breakdown are prevented. Wounds that are left open and allowed to heal by scar formation are classified as healing by secondary intention (Figure 4-1, B). These wounds heal more slowly because of the volume of connective tissue required to fill the defect. They are vulnerable to infection because they lack the epidermal barrier to microorganisms until late in the repair process. These wounds are characterized by prolongation of the inflammatory and proliferative phases of healing. Chronic wounds such as pressure ulcers and dehisced incisions typically heal by secondary intention. Wounds managed with delayed closure are classified as healing by tertiary intention, or delayed primary closure (Figure 4-1, C). This approach is sometimes required for abdominal incisions complicated by significant infection (Gabriel et al, 2010). Closure and/or approximation of the wound is delayed until the risk of infection is resolved and the wound is free of debris.

Wound-healing process
Wound healing is best understood as a cascade of events. Injury sets into motion a series of physiologic responses that are coordinated and sequenced and that, under normal circumstances, result in durable repair. Critical factors in the repair process include the cells that establish a clean wound bed and generate the tissue to fill the defect, the bioactive molecules that control cellular activity (growth factors and cytokines), and the wound-healing environment (extracellular matrix [ECM]). The wound-healing environment (matrix) provides a scaffold that promotes cell migration, and it contains a number of substances that regulate the activity of growth factors and cytokines and promote cell migration (e.g., matrix metalloproteinases) (Cheresh and Stupack, 2008; Gill and Parks, 2008; Macri and Clark, 2009).
Our current understanding of wound repair is based primarily on acute wound-healing models. Therefore the processes for partial-thickness wound healing and full-thickness wound healing by primary intention are presented first, followed by a discussion of the repair process for wounds that heal by secondary intention.
Partial-thickness wound repair
Partial-thickness wounds are shallow wounds involving epidermal loss and possibly partial loss of the dermal layer (see Plate 1). Partial-thickness wounds typically are less than 0.2 cm in depth. These wounds are moist and painful because of the loss of the epidermal covering and the resultant exposure of nerve endings. When the wound involves loss of the epidermis with exposure of the basement membrane, the wound base appears bright pink or red. In the presence of partial dermal loss, the wound base usually appears pale pink with distinct red “islets.” These “islets” represent the basement membrane of the epidermis, which projects deep into the dermis to line the epidermal appendages. These islands of epidermal basement membrane are important in partial-thickness wound healing because all epidermal cells are capable of regeneration, and each islet will serve as a source of new epithelium (Hunt and Van Winkle, 1997; Winter, 1979).
The major components of partial-thickness repair include an initial inflammatory response to injury, epithelial proliferation and migration (resurfacing), and reestablishment and differentiation of the epidermal layers to restore the barrier function of the skin (Monaco and Lawrence, 2003; Winter, 1979). If the wound involves dermal loss, connective tissue repair (granulation tissue formation) will proceed concurrently with epithelial repair (Table 4-1) (Jahoda and Reynolds, 2001).
TABLE 4-1
| Level of Injury (Tissue Layers Involved) | Primary Phases of Repair | Outcomes |
| Epidermal loss | Inflammation (brief)
Epithelial resurfacing Restoration of normal epithelial thickness/repigmentation | No loss of function
No scar No change in skin appearance or color |
| Possible partial dermal loss | Dermal repair (if dermal loss involved) | Restoration of rete ridges/dermal papillae when dermis involved |
Epidermal repair.
Tissue trauma triggers the processes that result in epidermal repair: an acute inflammatory response followed by epidermal mitosis and migration (Harrison et al, 2006). The inflammatory response produces erythema, edema, and a serous exudate containing leukocytes. When this exudate is allowed to dry on the wound surface, a dry crust, commonly referred to as a “scab,” is formed. In partial-thickness wounds, the inflammatory response is limited, typically subsiding in less than 24 hours (Winter, 1979).
Epidermal resurfacing begins as the inflammatory phase subsides and is dependent on two processes: proliferation of epidermal cells throughout the wound bed and lateral migration of the epidermal cells at the leading edge. In order to migrate laterally, the keratinocytes at the wound edge must undergo several changes. First they must acquire a migratory phenotype. This involves breakdown of their attachments to adjacent cells, the basement membrane, and the underlying dermis. These attachments normally prevent lateral migration and provide epithelial stability. The keratinocytes then must undergo cytoskeletal alterations that support lateral movement. These alterations include flattening of the cells at the advancing edge of the epithelium and formation of protrusions known as lamellipodia, which attach to binding sites in the wound bed. The migratory keratinocytes then move across the wound bed by alternately attaching to the ECM and then detaching and reattaching at a more distal point (Harrison et al, 2006; Monaco and Lawrence, 2003).
The epithelial cells within the wound bed continue this pattern of lateral migration until they contact epithelial cells migrating from the opposite direction. Once the epithelial cells meet, lateral migration ceases; this phenomenon is known as contact inhibition (Hunt and Van Winkle, 1997; Monaco and Lawrence, 2003).
Epithelial resurfacing is supported by increased production of basal cells just behind the advancing edge and throughout the wound bed. The process typically peaks between 24 and 72 hours after injury. The rate of reepithelialization is affected by a number of factors of clinical significance. For example, resurfacing is promoted by maintenance of a moist wound surface. Winter (1979) found that partial-thickness wounds left open to air required 6 to 7 days to resurface, whereas moist wounds reepithelialized in 4 days. This is because cells can migrate much more rapidly in a moist environment (Figure 4-2). In contrast, when the surface of the wound is covered with a scab, migration is delayed while the epithelial cells secrete enzymes known as matrix metalloproteinases (MMPs). The MMPs lift the scab by cleaving the bonds that attach it to the wound bed, creating a moist pathway that supports keratinocyte migration (Harrison et al, 2006; Monaco and Lawrence, 2003; Winter, 1979). Interestingly, hypoxic conditions in the wound bed also serve to stimulate keratinocyte migration, probably through increased production of the MMPs that promote lateral migration. This positive response to wound bed hypoxia is lost in the elderly, which may be one factor contributing to slower rates of reepithelialization in the aged (O’Toole et al, 2008). In contrast, high bacteria levels may serve as an impediment to reepithelialization. Data indicate that keratinocyte migration is inhibited by bacterial byproducts such as lipopolysaccharide (Loryman and Mansbridge, 2008). In vivo evidence strongly suggests that elevated glucose levels are another major impediment to epidermal proliferation and migration. This may be one factor contributing to delayed healing of superficial wounds in diabetic patients with poorly controlled glucose levels (Lan et al, 2008).
The newly resurfaced epithelium appears pale, pink, and dry in people of all races (see Plate 1). Because it is only a few cell layers thick, the new epithelium is very fragile and requires protection against mechanical forces such as superficial shear and friction.
Once epithelial resurfacing is complete, the epithelial cells resume vertical migration and epidermal differentiation so that normal epidermal thickness and function are restored. The normal anchors to adjacent epidermal cells and to the basement membrane are reestablished (Harrison et al, 2006). The “new” epidermis gradually repigments, matching the individual’s normal skin tone (see Plate 1). This is clinically relevant, as the wound is not completely healed until repigmentation has occurred (Monaco and Lawrence, 2003).
Regulatory factors.
The processes of epithelial proliferation and migration are regulated by a complex interplay between various MMPs and selected growth factors. Specifically, lateral migration is dependent on normal levels and function of multiple MMPs, which serve to break the attachments that bind the edge keratinocytes to adjacent cells and the underlying wound bed. In addition to playing a key role in establishment of a migratory phenotype, MMPs facilitate continued resurfacing by assisting the migrating keratinocytes to detach from the wound bed. Repetitive detachment and distal reattachment are critical to the resurfacing process (Gill and Parks, 2008). Epithelial proliferation is dependent in part on keratinocyte attachment to the ECM and in part on exposure to growth factors. Keratinocyte attachment is required for the cell to exit from the G0 phase of mitosis, and growth factors are required to stimulate cellular reproduction. A number of growth factors promote keratinocyte proliferation includeing transforming growth factor-α (TGF-α), keratinocyte growth factor, platelet-derived growth factor (PDGF), and epidermal growth factor (EGF). See Chapter 20 for a detailed discussion of the molecular factors regulating wound repair.
Dermal repair.
In wounds involving both dermal and epidermal loss, dermal repair proceeds concurrently with reepithelialization. By the fifth day after injury, a layer of fluid separates the epidermis from the dermal tissue. New blood vessels begin to sprout, and fibroblasts become plentiful by about the seventh day. Collagen fibers are visible in the wound bed by the ninth day. Collagen synthesis continues to produce new connective tissue until about 10 to 15 days after injury. This new connective tissue grows upward into the fluid layer. At the same time, the flat epidermis falls down around the new vessels and collagen fibers, re-creating ridges at the dermal–epidermal junction. As the new connective tissue gradually contracts, the epidermis is drawn close to the dermis (Winter, 1979). Insulin and insulin-like growth factor may contribute to reformation of the dermal–epidermal junction.
Full-thickness wound repair: Wounds healing by primary intention
Full-thickness wounds, by definition, extend at least to the subcutaneous tissue layer and possibly as deep as the fascia–muscle layer or bone (see Plate 2). Full-thickness wounds may be either acute or chronic. This section addresses acute wound healing by primary intention, such as a surgical incision.
Many steps are involved in full-thickness repair, but they are commonly conceptualized as four major phases: hemostasis, inflammation, proliferation, and remodeling. Considerable overlap exists among the phases, and the cells involved in one phase produce the chemical stimuli and substances that serve to move the wound into the next phase. Thus normal repair is a complex and well-orchestrated series of events (Figure 4-3) and is affected by a number of systemic and local factors, including the wound environment.

Hemostasis.
Any acute injury extending beyond the epidermis causes bleeding, which activates a series of overlapping events designed to control blood loss, establish bacterial control, and seal the defect. Immediately upon injury, disruption of blood vessels exposes the subendothelial collagen to platelets, which trigger platelet activation and aggregation. Simultaneously, injured cells in the wound area release clotting factors that activate both the intrinsic and extrinsic coagulation pathways. As part of the coagulation pathway, circulating prothrombin is converted to thrombin, which is used to convert fibrinogen to fibrin. The end result is formation of a clot composed of fibrin, aggregated platelets, and blood cells. Hemostasis is further accomplished by a brief period of vasoconstriction mediated by thromboxane A2 and prostaglandin 2-α, substances released by the damaged cells and activated platelets. Clot formation seals the disrupted vessels so that blood loss is controlled. The clot also provides a temporary bacterial barrier, a reservoir of growth factors, and an interim matrix that serves as scaffolding for migrating cells (Brissett and Hom, 2003; Monaco and Lawrence, 2003; Phillips, 2000).
Clot formation, followed by fibrinolysis (clot breakdown), is a critical event in the sequence of wound healing (Figure 4-4). The activation and degranulation of platelets cause the α-granules and dense bodies of the platelets to rupture, releasing a potent “cocktail” of energy-producing compounds and cytokines/growth factors (including complement factor C5a). These substances attract the cells needed to begin the repair process. They also provide fuel for the energy-intensive process of wound healing. The platelet-derived substances thought to be most critical to repair include PDGF, TGF-β, and fibroblast growth factor-2. Thus hemostasis, which is the body’s normal response to tissue injury, actually initiates the entire wound-healing cascade. The importance of hemostasis to wound healing is underscored by the finding that inadequate clot formation is associated with impaired wound healing and that the extrinsic coagulation pathway is critical to repair (Brissett and Hom, 2003; Monaco and Lawrence, 2003).
Inflammation.
Once the bleeding is controlled, the focus becomes control of infection and establishment of a clean wound bed. This process can be compared with the repair of a damaged home or building. Before rebuilding can begin, the damaged components must be removed. Wound “cleanup” involves breakdown of any devitalized tissue and elimination of excess bacteria by a team of white blood cells. Acute wounds, such as sutured lacerations or incisions, typically have limited devitalized tissue and low bacterial loads, so the cleanup phase usually is brief, lasting only hours to a few days (Braiman-Wiksman et al, 2007; Pradhan et al, 2009).
Within 10 to 15 minutes after injury, vasoconstriction subsides, followed by vasodilation and increased capillary permeability. Vasoactive substances released by damaged cells and by clot breakdown (histamine, prostaglandins, complement factor, thrombin) mediate this response. The dilated capillaries permit plasma and blood cells to leak into the wound bed. Clinically, this process is observed as edema, erythema, and exudate. At the same time, the damaged cells and platelets produce cytokines and growth factors that attract leukocytes (neutrophils, macrophages, lymphocytes) to the wound bed. The twin processes of chemoattraction and vasodilation result in the delivery of multiple phagocytic cells to the wound site within minutes of injury (Gill and Parks, 2008; Pradhan et al, 2009; Rodriguez et al, 2008).
Leukocyte migration out of the vessels and into the wound bed occurs via margination and diapedesis. Margination involves adherence of the leukocytes to the endothelial cells lining the capillaries in the wound bed. Integrins on the surface of the leukocytes attach to cell adhesion molecules on the surface of the endothelial cells, causing the leukocytes to “line up” against the vessel wall. Through a process known as diapedesis, the leukocytes migrate through the dilated capillaries into the wound bed (Cross and Mustoe, 2003; Monaco and Lawrence, 2003; Wilgus, 2007).
The first leukocytes to arrive in the wound space, the neutrophils, are present in the wound bed within minutes after injury and dominate the scene for the first 2 to 3 days. The primary function of neutrophils is phagocytosis of bacteria and foreign debris. Neutrophils first bind to the damaged tissue or target bacteria via cell adhesion molecules and then engulf and destroy the target molecules via intracellular enzymes and free oxygen radicals. In addition, growth factors released by the neutrophils attract additional leukocytes to the area (Cross and Mustoe, 2003).
By days 3 to 4 after injury, the neutrophils begin to spontaneously disappear as a result of apoptosis and are replaced by macrophages (activated monocytes). The macrophages continue to phagocytize bacteria and break down damaged tissues as already described. In addition, the macrophages release a large number of potent growth factors that stimulate angiogenesis, fibroblast migration and proliferation, and connective tissue synthesis.
T lymphocytes are present in the wound tissue in peak quantities between days 5 and 7 after injury. T lymphocytes contribute to the inflammatory phase of wound healing by secreting additional wound-healing cytokines and by destroying viral organisms and foreign cells. Thus significant deficiencies of T lymphocytes can delay or compromise the repair process (Monaco and Lawrence, 2003).
Although all leukocytes contribute to elimination of bacteria and establishment of a clean wound bed, macrophages contribute the most significantly to the repair process. Studies indicate that wounds can heal without neutrophils, especially if no bacterial contamination is present. However, elimination of macrophages severely compromises wound repair (Cross and Mustoe, 2003; Gabriel et al, 2010).
The result of the inflammatory phase of wound healing is a clean wound bed. In acute wounds healing by primary intention, the inflammatory phase lasts approximately 3 days. At this point, bacterial levels usually are controlled, and any devitalized tissue has been removed. Elimination of these noxious stimuli allows the wound to transition to the “rebuilding” phase. This transition involves apoptosis of the inflammatory cells, possibly due in part to the effects of antiinflammatory cytokines and increased production of growth factors that promote proliferation as opposed to inflammation (Demidova-Rice et al, 2012; Goldman, 2004). However, in wounds complicated by necrosis and/or infection, the inflammatory phase is prolonged and wound healing is delayed (Burns et al, 2003). A prolonged inflammatory phase increases the risk for wound dehiscence because approximation of the incision is totally dependent on the closure material (sutures, staples, fibrin glue) until sufficient connective tissue has been synthesized to provide tensile strength to the incision. (Tensile strength during the inflammatory phase is 0%.)
Factors affecting the duration and intensity of inflammation.
The intensity and duration of the inflammatory phase appear to be critical factors in the amount of scar tissue produced. Numerous studies support a strong link between prolonged inflammation and hyperproliferative scarring (Dubay and Franz, 2003; Pradhan et al, 2009; Rahban and Garner, 2003; Robson, 2003). Thus the clinician needs to have a clear understanding of both proinflammatory and antiinflammatory factors and the implications for wound management. As noted, local factors such as bacterial load and presence of devitalized tissue are major proinflammatory factors that should be aggressively managed through debridement and appropriate use of topical dressings and antimicrobial agents (Demidova-Rice et al, 2012). Necrotic tissue and infection trigger production of cytokines that attract excessive numbers of inflammatory cells to the wound bed; these inflammatory cells produce reactive oxygen species (ROS) that, in conjunction with proinflammatory cytokines, cause degradation of the growth factors needed for normal function of proliferative cells such as fibroblasts. Thus necrosis and infection are major impediments to wound healing.
The ECM is another important determinant; it can either promote or inhibit inflammation, depending on the types of MMPs produced and their functions. Some MMPs promote inflammation by releasing proinflammatory cytokines from the cells in the wound bed; proinflammatory cytokines attract inflammatory cells to the wound bed (Pradhan et al, 2009). Other MMPs inhibit inflammation by degrading proinflammatory cytokines or inhibiting their release. For example, tumor necrosis factor-α (TNF-α) is a proinflammatory cytokine that normally is present at high levels during the early inflammatory phase, and levels of TNF-α are controlled by two ECM proteins. One protein, a disintegrin and metalloproteinase (ADAM-17), promotes release of TNF-α. Another protein, a tissue inhibitor of metalloproteinase-3 (TIMP-3), controls ADAM-17, thereby reducing production of TNF-α. Research is ongoing into factors that determine the combination and concentration of ECM proteins and into strategies for measuring and controlling levels of various MMPs to achieve therapeutic outcomes (Gill and Parks, 2008; Macri and Clark, 2009).
Diabetes is a clinical condition associated with prolonged inflammation. Some data suggest that leukocyte migration may be impaired in people with this condition; in addition, the leukocytes that do migrate to the wound bed frequently are dysfunctional, especially in the presence of hyperglycemia. The end result of diminished leukocyte migration and impaired leukocyte function is failure to effectively control bacterial loads. This situation results in a persistent stimulus for proinflammatory cytokines, chronic inflammation, and delayed healing (Pradhan et al, 2009). Conditions resulting in ischemia/hypoxia also produce prolonged inflammation, because oxygen and ROS are required for oxidative killing of bacteria. The inability to control bacteria that accompanies hypoxia results in chronic inflammation and failure to heal. Hyperbaric oxygen therapy can improve tissue oxygen levels, contribute to bacterial control, and reduce the production of proinflammatory cytokines, thus promoting the repair process (Rodriguez et al, 2008; Thom, 2009).
Conditions and medications that affect the function of autonomic and sensory nerves and their receptors may also affect inflammation. For example, diabetes is associated with diminished production of substance P, a neuropeptide that normally contributes to a healthy inflammatory response by supporting vasodilation and leukocyte migration. Diminished production of substance P may be one factor leading to compromised inflammation and impaired healing in the diabetic population (Pradhan et al, 2009). Preliminary research suggests β-adrenergic agents may reduce inflammation by reducing neutrophil recruitment and production of inflammatory cytokines (Pullar et al, 2008).
Proliferation.
The third phase of acute full-thickness wound healing is the proliferative phase. During this phase, the wound surface is covered with new epithelium that restores the bacterial barrier, vascular integrity is restored, and the incisional defect is mended with new connective tissue. The key components of the proliferative phase are epithelialization, neoangiogenesis, and matrix deposition/collagen synthesis. Limited contraction of the newly formed ECM also may occur.
Epithelialization.
Epithelialization of a full-thickness wound healing by primary intention begins within hours after injury and typically is complete within 24 to 48 hours. This “neoepithelium” is only a few cells thick but is sufficient to provide a closed wound surface and a bacterial barrier. This process is the basis for the Centers for Disease Control and Prevention’s recommendation that new surgical incisions be covered with a sterile dressing for the first 24 to 48 hours postoperatively. Until reepithelialization is complete, the potential for bacterial invasion exists, and the wound should be managed with sterile wound care and a cover dressing that provides a bacterial barrier (Mangram et al, 1999; Yamamoto et al, 2014). As with partial-thickness wound repair, the processes of lateral migration, vertical migration, and differentiation continue throughout the proliferative phase and gradually reestablish epidermal thickness and function (Braiman-Wiksman et al, 2007; Myers et al, 2007). In full-thickness wounds, the new epidermis is slightly thinner than the original epidermis. Because the neoepidermis is covering scar tissue rather than normal dermis, the rete pegs that normally dip into the dermis are lacking (Monaco and Lawrence, 2003).
Granulation tissue formation.
A hallmark outcome of the proliferative phase is the formation of granulation tissue, which begins as the inflammatory phase subsides, at 3 to 4 days postinjury (Braiman-Wiksman et al, 2007). Granulation tissue is composed primarily of capillary loops and newly synthesized connective tissue proteins and is often referred to as the ECM. Fibroblasts and inflammatory cells also are present in this new matrix. Neoangiogenesis and connective tissue synthesis occur simultaneously in a codependent fashion to form the new ECM that will fill the wound defect. Through angiogenesis, new capillaries are formed and joined with existing severed capillaries, thus restoring the delivery of oxygen and nutrients to the wound bed. At the same time, a new “provisional” ECM is formed through the synthesis of connective tissue proteins such as type III collagen (Figure 4-5) (Pradhan et al, 2009).

Neoangiogenesis.
Endothelial cells typically are quiescent. Neoangiogenesis requires stimulation by growth factors, which convert the quiescent cells to actively proliferating cells. Molecules in the ECM also play a critical role by influencing the response of endothelial cells to angiogenic growth factors (Cheresh and Stupack, 2008). Neoangiogenesis occurs by two mechanisms: production of new vessels by local endothelial cells and recruitment of circulating stem progenitor cells to form new vessels de novo (Thom, 2009).
The growth factors that stimulate production of new vessels are produced by the cells in the wound bed (injured endothelial cells, macrophages, fibroblasts, keratinocytes) and include vascular endothelial growth factor (VEGF), basic fibroblast growth factor, and possibly PDGF. The most important angiogenic growth factor is VEGF; in addition to stimulating local endothelial cells to proliferate and migrate, VEGF attracts stem progenitor cells to the wound bed and stimulates them to differentiate into endothelial cells (Braiman-Wiksman et al, 2007; Gill and Parks, 2008; Thom, 2009). A number of ECM factors also affect neoangiogenesis. One is the provisional matrix itself, which affects the migration of endothelial cells (Gill and Parks, 2008). Another is the level of TIMP-3, which has a very negative effect on angiogenesis. TIMP-3 binds to the receptor sites for VEGF and blocks its angiogenic effects (Gill and Parks, 2008). A third factor is the level of nitric oxide, which promotes angiogenesis by activating MMP-13 (Lizarbe et al, 2008).
Clinical factors affecting neoangiogenesis include oxygen levels within the wound bed, patient age, gender, diabetes, coronary artery disease, radiation therapy, and chemotherapy. Hypoxia acts as an initial stimulus to angiogenesis, but persistent hypoxia interferes with endothelial cell proliferation and new vessel formation. Hyperglycemia and glycolysis are associated with production of substances that are toxic to endothelial cells. Diabetes, aging, female gender, coronary artery disease, radiation therapy, and chemotherapy all interfere with angiogenesis by reducing mobilization of stem progenitor cells, thus compromising the potential for de novo development of new vessels. Aging is also associated with dysfunction and impaired mobilization of local endothelial cells, partly due to impaired expression of VEGF and other angiogenic stimuli. Of interest, some data suggest that exercise can partially reverse these age-related changes (Hoenig et al, 2008; Rodriguez et al, 2008; Thom, 2009). Therapeutic modalities that may promote angiogenesis include exogenous application of basic fibroblast growth factor and topical or systemic administration of erythropoietin (EPO). Animal studies and limited human case reports indicate potent stimulation of angiogenesis and granulation tissue formation with each of these therapies; EPO has also been associated with inhibition of excessive inflammation (Hamed et al, 2014; O’Goshi and Tagami, 2007).
Matrix deposition/collagen synthesis.
Fibroblasts are responsible for synthesis of the connective tissue proteins that compose the provisional ECM; therefore fibroblasts are critical to the repair process. Fibroblasts migrate into the wound bed from the surrounding tissues in response to growth factors and interleukins released by degranulating platelets, activated leukocytes (neutrophils and macrophages), and keratinocytes. Migration requires up-regulation of binding sites (integrin receptors) on the cell wall, which is mediated by PDGF and TGF-β. This up-regulation of binding sites is essential because fibroblasts migrate by maintaining attachment to one binding site while extending lamellipodia in search of another site. Once the fibroblast is able to bind to a new site, it releases the original attachment and “moves” in the direction of the wound bed (Monaco and Lawrence, 2003; Myers et al, 2007).
Fibroblasts begin to appear in the wound bed toward the end of the inflammatory phase, 2 to 3 days after injury. By day 4, fibroblasts are the predominant cells in the wound matrix (Dubay and Franz, 2003). Once fibroblasts arrive at the wound site, growth factors bind to fibroblast receptor sites and trigger intracellular processes that move the fibroblasts into the reproductive phase of the cell cycle, thus stimulating proliferation of the fibroblast. Finally, the fibroblasts are converted into “wound fibroblasts” by TGF-β, a growth factor secreted by macrophages. These wound fibroblasts differ from typical dermal fibroblasts in that they exhibit decreased proliferative behavior but increased synthesis of connective tissue proteins such as collagen (Frank et al, 2002).
Collagen synthesis follows the established process for synthesis of any protein. The collagen molecule is characterized by a glycine-X-Y repeating sequence. The collagen molecule must undergo a series of intracellular modifications before the molecule is secreted into the extracellular environment and becomes part of the ECM. One of the most critical modifications is “cross-linking” of the proline and lysine molecules, a process known as hydroxylation. These cross-links are essential for the development of tensile strength. A number of factors are required for normal hydroxylation, including oxygen, ascorbic acid, iron, copper, and selected enzymes. Thus hypoxia, vitamin C deficiency, copper deficiency, and iron deficiency all can compromise hydroxylation and the development of tensile strength. High-dose corticosteroids also can impair tensile strength because corticosteroids suppress the enzymes needed for hydroxylation. Following intracellular modifications, the collagen molecule is secreted into the extracellular environment as the triple helix procollagen. It then undergoes additional steps that culminate in the formation of cross-linked fibrils. The enzyme lysyl oxidase is essential to these processes and to the development of stable collagen fibers (Monaco and Lawrence, 2003).
The early granulation tissue is a provisional matrix characterized by unstructured collagen and high levels of fibronectin. This provisional matrix also includes vibronectin, laminin, and proteoglycans such as hyaluronic acid (Braiman-Wiksman et al, 2007; Gill and Parks, 2008; Schneider et al, 2008). As the ECM matures, collagen becomes the predominant protein, representing a little over 50% of the new ECM. Proteoglycans such as hyaluronic acid are present in small amounts but serve critical functions. For example, hyaluronic acid facilitates cell migration, protects cells against free-radical and proteolytic damage, and contributes viscoelastic properties to the new matrix (Chen and Abatangelo, 1999; Monaco and Lawrence, 2003).
Wounds healing by primary intention, such as sutured incisions, require a limited amount of connective tissue to mend the defect. In these wounds, ECM production is essentially complete within 14 to 21 days (Hunt and Van Winkle, 1997; Monaco and Lawrence, 2003). Although the granulation tissue is not visible in these wounds, a “healing ridge” can be palpated just under the intact suture line by days 5 to 9. The healing ridge is produced by the newly formed connective tissue proteins. Absence of this healing ridge indicates impaired healing and increased risk for dehiscence (Hunt and Van Winkle, 1997).
Factors affecting granulation tissue formation.
Factors affecting granulation tissue formation include perfusion status, oxygen levels, nutritional status, and glucose levels/diabetes. Granulation tissue formation is very much an oxygen-dependent process. Hypoxia can act as a stimulus to fibroblast proliferation and initial collagen synthesis, but adequate levels of oxygen (30–40 mm Hg) are absolutely essential for the latter steps in collagen synthesis and for the cross-linking that provides tensile strength. Adequate oxygen levels are also required for prevention of infection. Infection rates in surgical wounds are inversely proportional to oxygen levels (Rodriguez et al, 2008). Tissue oxygenation of acute wounds is discussed in more detail in Chapters 28 and 34. Nutritional status affects granulation because granulation tissue formation requires adequate levels of protein and micronutrients such as ascorbic acid, iron, and copper. Diabetes is associated with delayed deposition of granulation tissue and insufficient production of collagen to establish a mature matrix (Braiman-Wiksman et al, 2007).
Contraction.
Contraction occurs when specialized fibroblasts known as myofibroblasts exert tractional forces on the ECM, reducing the size of the wound. Wounds healing by primary intention have limited or no contraction because the wound edges have been surgically approximated (Monaco and Lawrence, 2003).
Maturation/remodeling.
The final phase in full-thickness wound healing is the maturation, or remodeling, phase, which begins around day 21 after wounding and continues beyond 1 year. The early collagen is characterized by poorly organized fibers having limited tensile strength. At 3 weeks after injury, the healing wound exhibits only 20% of the strength of intact dermis. The provisional matrix is gradually converted to a mature and relatively acellular scar, a process that involves replacement of the type III collagen in the provisional matrix with type I collagen, the type normally found in dermal tissue. However, even the final form of collagen does not exhibit the normal basket-weave pattern of the collagen in unwounded dermis. Rather, the fibers of “repair collagen” are aligned parallel to the stress lines of the wound. The new ECM also lacks elastin, which provides the uninjured skin with elasticity; thus scar tissue is “stiff” compared with normal tissue (Monaco and Lawrence, 2003; Rodriguez et al, 2008).
Remodeling involves the dual processes of synthesis and degradation of the ECM and is regulated by fibroblasts and ECM proteins (MMPs). The new collagen that is formed is more orderly and provides more tensile strength to the wound; tensile strength improves steadily during the 2 to 3 months following initial injury and repair. However, the tensile strength of the remodeled ECM (scar tissue) is never more than 80% of the tensile strength in nonwounded tissue (Hunt and Van Winkle, 1997).
An imbalance between the dual processes of matrix synthesis and matrix breakdown can complicate wound healing. For example, hypertrophic scarring and keloid formation are believed to be caused in part by an excess of matrix synthesis compared with matrix degradation (Rahban and Garner, 2003; Wilgus, 2007). In contrast, hypoxia, malnutrition, or excess levels of MMPs can interfere with synthesis and deposition of new matrix proteins, resulting in wound breakdown (Monaco and Lawrence, 2003).
Full-thickness wound repair: Wounds healing by secondary intention
Although full-thickness wounds healing by secondary intention (e.g., pressure ulcers) proceed through the same phases as full-thickness wounds healing by primary intention, key differences do exist within each phase. These differences are summarized in Table 4-2 and discussed here.
TABLE 4-2
| Primary Intention | Secondary Intention | |
| Examples | Laceration
Surgical incision | Chronic wound (e.g., pressure ulcer, venous ulcer)
Wound dehiscence |
| Hemostasis | Bleeding
Platelets rupture and release growth factors Coagulation pathways (intrinsic and extrinsic) activated | No bleeding
No coagulation Hemostasis absent |
| Inflammatory phase | Any necrotic tissue removed
Bacterial balance restored Duration usually limited to 1–3 days unless complicated by infection | Necrotic tissue removed
Bacterial balance restored Duration prolonged until nonviable tissue eliminated and bacteria controlled; also prolonged by presence of excess proinflammatory cells |
| Proliferative phase | Epithelialization (first event in proliferative phase)
Synthesis of connective tissue Duration typically 14–21 days | |
| Maturation (remodeling phase) | Collagen synthesis and lysis
Tensile strength partially reestablished | Collagen synthesis and lysis
Tensile strength partially reestablished |
Absence of hemostasis.
The absence of hemostasis has a tremendous impact on the healing trajectory of wounds. Bleeding and hemostasis do not occur in wounds healing by secondary intention. Failure to bleed and clot compromises the repair process because the wound-healing sequence of events is normally initiated by clot breakdown and the subsequent release of growth factors. One theorized benefit of surgical debridement of chronic wounds is to trigger clot formation and the release of growth factors so that the repair process is reactivated.
Prolonged inflammatory phase.
The inflammatory phase frequently is prolonged. Because the goal of this phase is to establish a clean wound bed and to obtain bacterial balance, the wound will remain in this phase until necrotic tissue has been eliminated and bacterial loads have been controlled. Wounds healing by secondary intention frequently are characterized by large amounts of devitalized tissue and heavy bacterial loads. Thus the inflammatory phase generally lasts considerably longer than the 3 days that are typical in the approximated incision.
Prolonged proliferative phase.
The proliferative phase is prolonged, and the sequence of events is different. In wounds healing by primary intention, epithelialization occurs first, followed by angiogenesis and formation of a limited volume of connective tissue proteins (ECM); contraction does not occur or is very limited. These processes usually are complete within 14 to 21 days. In secondary-intention wound healing, the proliferative phase begins with granulation tissue formation (to fill in the soft tissue defect), followed by contraction (to minimize the defect); epithelialization is the final phase.
The volume of granulation tissue required to fill the defect (and the time required for this phase of repair) is determined by the size of the wound and the degree to which contraction is able to reduce the size of the defect. Because the wound bed is visible, the clinician is able to assess progress in healing. Healthy granulation tissue presents as a red, vascular, granular wound bed as a result of the numerous capillary loops in combination with the newly synthesized ECM proteins (see Plate 3).
Increased amount of contraction.
Contraction is much more important in secondary-intention wound healing than in closed wounds because contraction reduces the size of the soft tissue defect and thus reduces the amount of granulation tissue required. The rate of contraction for open wounds averages 0.6 to 0.7 mm/day (Gabbiani, 2003; Monaco and Lawrence, 2003). The degree to which a specific wound will contract is determined partly by the mobility of the surrounding tissue. For example, the tissue surrounding sacral and abdominal wounds is quite mobile and can contract easily. In contrast, the tissue surrounding a wound on the extremity or overlying a bony prominence has limited potential for contraction. Contraction is considered undesirable in some wounds because it can cause cosmetic deformities or flexion contractures of joints.
Contraction is mediated by myofibroblasts, modified fibroblasts that contain actin and myosin monofilaments and smooth muscle proteins. Differentiation of fibroblasts into myofibroblasts is stimulated by growth factors such as TGF-β1 and PDGF. Substances within the ECM itself are also thought to contribute to the development of myofibroblasts. Intracellular actin filaments and extracellular fibronectin work jointly to establish a contractile force that compresses and “shrinks” the ECM, thus pulling the wound edges toward each other (Demidova-Rice et al, 2012; Gabbiani 2003).
Delayed epithelialization.
Because full-thickness wounds involve loss of the deep dermis and epidermal appendages (along with their epithelial lining), epithelialization in these wounds proceeds from the periphery of the wound inward in a centripetal fashion. Epithelial migration requires an open, proliferative wound edge. Closed, nonproliferative wound edges, also known as epibole, are sometimes seen in open wounds healing by secondary intention, probably due to premature keratinization of the wound edges (Figure 4-6 and Plate 4). In these wounds, an open edge must be reestablished, by either surgical excision or chemical cauterization, before epithelial migration can occur (Schultz, Sibbald, Falanga, 2003).
Prolonged remodeling.
The remodeling process for wounds healing by secondary intention is essentially the same as that for wounds healing by primary intention. Clinicians and caregivers must remain acutely aware that newly “healed” wounds initially lack tensile strength, and measures should be initiated to minimize stress on the remodeling wound until tensile strength has developed, which occurs 2 to 3 months after closure. For example, the patient with a newly healed pressure ulcer should remain on a therapeutic support surface and should minimize time spent lying on the involved surface.
What makes a chronic wound chronic?
An acute wound in a relatively healthy host will heal fairly quickly because of a cascade of growth factors, cytokines, and matrix proteins that tend to keep the acute wound on the “healing track.” In clinical practice, however, chronic wounds such as pressure ulcers, vascular ulcers, and neuropathic wounds behave much differently and may be extremely slow to heal. In order to intervene effectively, the clinician must be knowledgeable regarding the various factors contributing to delayed healing.
Over the past decade, extensive research analyzing the cellular, biochemical, and molecular components of acute and chronic wounds has significantly expanded the understanding of the detailed complexities of normal wound healing and the pathophysiologic mechanisms of chronic wounds. Box 4-1 summarizes the characteristics of a chronic wound.
Underlying pathology
The nature of the injury differs between acute and chronic wounds. Acute wounds usually begin with a sudden, solitary insult and proceed to heal in an orderly manner. In contrast, chronic wounds are commonly caused by an underlying pathologic process, such as vascular insufficiency, that produces repeated and prolonged insults to the tissues. Failure to correct or control the underlying pathology can result in a persistent cycle of injury that causes repetitive tissue damage. In contrast, correction of the underlying pathology frequently can shift the wound to a healing pathway (Goldman, 2004).
Increased levels of inflammatory substances
Chronic wounds frequently are complicated by impediments to healing, such as ischemia, necrotic tissue, heavy bacterial loads, and high levels of proinflammatory MMPs. These factors prolong the inflammatory phase of wound healing by continuing to recruit macrophages and neutrophils into the wound bed. In addition, high levels of proinflammatory MMPs are associated with ongoing destruction of the ECM (Demidova-Rice et al, 2012; Gill and Parks, 2008; Goldman, 2004). Studies indicate that the levels of inflammatory substances in chronic wounds are 100 times higher than the levels in acute wounds (Berg Vande and Robson, 2003). Fortunately, this cycle can be interrupted by elimination of the noxious stimuli, that is, by debridement of necrotic tissue and control of the bacterial burden (Goldman, 2004).
Low levels of growth factors
In addition to high levels of inflammatory proteases, low levels of growth factors commonly characterize the environment of chronic wounds. Normal levels of growth factors are critical to repair, because a “threshold level” of growth factors is required to move target cells out of the quiescent G0 phase and into the reproductive cycle (Berg Vande and Robson, 2003). The low levels of growth factors commonly found in chronic wounds may be the result of inadequate production by the cells of the wound bed (or insufficient numbers of “producer” cells). Additional potential causes include rapid breakdown of growth factors by the high levels of MMPs or “binding” of the growth factors by the ECM (Berg Vande and Robson, 2003; Steed, 2003). Although this imbalance between inflammatory and proliferative stimuli usually is thought to be the cause of impaired healing, some investigators suggest that the imbalance instead may be a result of chronicity. When the wound begins to heal, the ratio of inflammatory to proliferative stimuli normalizes (Goldman, 2004; Henry and Garner, 2003).
Cellular and ECM abnormalities.
Current evidence suggests distinct phenotypic differences in the fibroblasts and keratinocytes found in chronic wounds compared with those found in acute wounds. Specifically both fibroblasts and keratinocytes exhibit reduced proliferative behavior and reduced motility, possibly due in part to abnormalities in the ECM (Demidova-Rice et al, 2012). Additional differences noted in the fibroblasts and keratinocytes of chronic wounds include reduced numbers of growth factor receptor sites and increased cellular senescence (that is, decreased ability to proliferate) (Telgenhoff and Shroot, 2005). Because all phases of wound healing are controlled primarily by growth factors, reduced binding sites or reduced responsiveness would significantly alter the cells’ ability to contribute to wound healing. Cellular senescence may occur in the elderly as a normal component of aging and may contribute to the delayed healing commonly seen in this population. In addition, the high levels of ROS common to the chronic wound environment can cause DNA damage and loss of the cells’ ability to replicate normally, even in younger individuals. Fortunately, this type of cellular senescence is reversible through optimal wound management (Berg Vande and Robson, 2003; Pittman, 2007; Telgenhoff and Shroot, 2005).
Miscellaneous host conditions
Additional “host factors” that trigger chronicity in a wound include ischemia, malnutrition, and comorbidities such as diabetes (Steed, 2003). Malnutrition is a particularly common contributor to wound chronicity. Fibroblasts that lack the requisite raw ingredients cannot synthesize connective tissue proteins. The importance of nutritional status is reflected in studies documenting consistently impaired wound healing in patients whose albumin levels were lower than 2.0 g/L (Burns et al, 2003). In addition, a study on wound fluid as a predictor of healing found the only constituents to reliably predict healing were total protein and albumin, that is, albumin levels greater than 2.0 g/L (James et al, 2000; Schultz et al, 2003).
Pale, pink tissue that is smooth rather than granular is indicative of delayed healing and a compromised ability to synthesize collagen and other connective tissue proteins (see Plate 5).
Denervation
Denervation is another potential cause of failure to heal. Sensory nerves secrete neuropeptides (e.g., substance P) that are highly chemotactic for inflammatory cells. Therefore denervated wounds are subject to impaired inflammation and compromised healing. Denervation may be one of the factors contributing to chronicity of pressure ulcers in patients with spinal cord injury and neuropathic ulcers in diabetic patients (Macri and Clark, 2009; Richards et al, 1997).
In summary, differences in the healing trajectory for acute and chronic wounds stem from the nature of the injury, the cellular events that follow injury, and miscellaneous host factors. In general, healing wounds are characterized by high mitotic activity, therapeutic levels of inflammatory cytokines, low levels of proteases, and mitotically competent cells. In contrast, chronic wounds exhibit low mitotic activity, excessive levels of inflammatory cytokines, high levels of proteases, and senescent or mitotically incompetent cells.
Mediators of wound healing
In order for healing to occur normally, the critical cells must be recruited to the wound bed (at the appropriate time), stimulated to reproduce, and then directed to carry out essential functions, such as neoangiogenesis, connective tissue synthesis, and reepithelialization. This complex process is controlled and coordinated by an equally complex array of regulatory substances; key elements include bioactive molecules, matrix proteins, and the matrix itself. The effect of these regulatory factors is further influenced by “host” factors, such as cell receptor sites, cellular senescence, availability of nutrients, and cofactors required for phagocytosis and collagen synthesis, and comorbid conditions, such as hypoxia and diabetes. This section provides a brief overview of regulatory factors, which are discussed further in Chapter 20.
Bioactive molecules
Bioactive molecules include growth factors and cytokines. They are produced by the cells in the wound bed (e.g., platelets, neutrophils, macrophages, and fibroblasts) and act as “directors” of cell function and thus of the repair process. They do so by attracting the needed cells to the wound bed, stimulating them to proliferate, and then directing the cells to carry out specific aspects of repair. For example, TNF-α is a proinflammatory cytokine that attracts neutrophils and macrophages to the wound bed and is present in high concentrations during the inflammatory phase, whereas VEGF is a growth factor that supports proliferation and migration of endothelial cells and therefore is important during angiogenesis (Gill and Parks, 2008; Macri and Clark, 2009; Thom, 2009).
Matrix proteins
Growth factors and cytokines act as “controllers” for the cells critical to the repair process. However, it is increasingly apparent that proteins within the ECM control the levels and function of growth factors and cytokines. Key categories of matrix proteins include MMPs, ADAMs, and tissue inhibitors of metalloproteinase (TIMPs). Matrix metalloproteinases and ADAMs can “up-regulate” the levels of growth factors and cytokines by cleaving them from the cell and thus activating them; however, they also can “down-regulate” the levels of growth factors and cytokines by degrading them or by inhibiting their release. Tissue inhibitors of metalloproteinase control the activity of MMPs by binding to them, thus blocking their effects (Gill and Parks, 2008).
Extracellular matrix
The ECM serves as a scaffold for migrating cells and as a repository for growth factors. It also influences the response of the cells to growth factors. For example, keratinocyte proliferation requires both attachment to the ECM and stimulation by growth factors. Cell migration is another aspect of repair that is dependent on both matrix proteins and integrins within the matrix itself. Matrix proteins promote migration by degrading the bonds between the migrating cells and the underlying wound bed, and the matrix promotes reattachment through the expression of binding sites (integrins) within the matrix. The matrix also supports cell-to-cell communication. The ECM needs to be porous and pliable in order to support the movement of cells, nutrients, and growth factors through the wound environment (Cheresh and Stupack, 2008; Schultz and Wysocki, 2009). Dermal matrix dressings (see Chapter 19) may be beneficial to support cell migration and granulation tissue formation, in select wounds (Nataraj et al, 2007).
Host factors
A number of host factors influence cells’ response to growth factors, cytokines, and matrix proteins. One of these factors is the type of receptors on the cell wall. This is an important factor because all cells within the wound bed are exposed to the same mix of regulatory substances, but only select cells respond. In addition, different cells may exhibit different responses to the same regulatory substance because regulatory substances exert their effects primarily through binding with cell receptors. Therefore only cells with the specific receptor sites respond to the regulatory substance, and the effects of receptor binding vary based on cell type. For example, PDGF stimulates migration of some cells and mitosis in others, but it has no effect on other cells (Martin, 1997; Witte and Barbul, 1997). Furthermore, fibroblasts and keratinocytes in elderly individuals have a decreased number of receptor sites, which may explain why elderly patients tend to exhibit a diminished response to some regulatory substances (Ashcroft et al, 2002).
Other factors that impact an individual’s ability to heal normally are systemic factors such as nutritional status, perfusion, oxygenation, and comorbidities such as diabetes. These factors are addressed briefly in this chapter and in greater depth in other chapters in this text.
Extremes of repair: Scarless healing versus excessive scarring
This chapter has focused on what is currently considered “normal” repair for full-thickness wounds, that is, formation of granulation tissue (scar) to mend the defect, with a covering of new epithelium. This form of repair represents an intermediate point between “ideal” repair, also referred to as scarless healing, and “abnormal” repair. Hypertrophic and keloid scars are examples of abnormal repair or excessive scarring. As we learn more about the factors that lead to “normal” and excessive scarring, we hope to be able to optimize repair and minimize scarring for all patients.
Scarless healing
As mentioned earlier in this chapter, the early-gestation fetus typically heals without scarring, an ability that is lost at 22 to 24 weeks of gestation (Samuels and Tan, 1999). Features and characteristics of early-gestation healing are listed in Box 4-2. One significant difference is a markedly diminished inflammatory response. Multiple studies using various models have demonstrated that inflammation is minimal or essentially absent in early-gestation (scarless) healing. This finding is consistent with other studies showing a strong link between the intensity and duration of the inflammatory response and the subsequent development of scar tissue (Wilgus, 2007). The specific factors thought to contribute to this very minimal inflammatory response include the following: (1) Fetal platelets release much lower levels of proinflammatory growth factors and cytokines, most specifically the profibrotic isoforms of TGF-β. TGF-β is responsible for attracting inflammatory cells to the wound bed, for promoting angiogenesis and fibroblast activity, and for remodeling of the ECM; (2) Marked reduction in migration of inflammatory cells (neutrophils, macrophages, and T cells); this is partially due to the lower levels of proinflammatory growth factors and partially due to the fact that the fetal immune system is poorly developed; (3) Marked reduction in angiogenic stimuli linked to scar formation (VEGF, TGF-β1, PDGF, and prostaglandin E2) (Walraven et al, 2014; Wilgus, 2007).
Another difference in early-gestation healing is the rate at which epithelial resurfacing occurs. Rapid up-regulation of the adhesion molecules within the ECM allows cells to move laterally, which promotes early keratinocyte migration (Wilgus, 2007).
Most importantly, a number of differences in fetal healing contribute to production of a new ECM that retains the characteristics of unwounded tissue. One contributing factor may be the high levels of hyaluronic acid and hyaluronic-acid-stimulating activity factor. This factor is significant because hyaluronic acid supports rapid cell migration. In addition, high levels of MMPs compared with TIMPs favor degradation of the ECM, which is thought to help prevent overproduction of collagen. Finally, fetal fibroblasts retain the ability to produce new matrix proteins that retain the basket-weave configuration characteristic of normal dermis (Wilgus, 2007).
In summary, early-gestation fetal repair is characterized by a significantly reduced inflammatory response and a rapid and balanced proliferative phase that restores the dermal architecture without scarring (see Box 4-2).
Excessive scarring
Excessive or hyperproliferative scarring is a complication of acute full-thickness wound healing that presently is not well understood. The two types of hyperproliferative scarring are hypertrophic scars and keloid scars (see Plate 7, C and D). Both types appear raised, are red or pink, and are pruritic. Hypertrophic scars are confined to the original incisional or scar area, whereas keloids expand beyond the incision into the surrounding tissue. This expansion, sometimes described as a mushroom effect, is the result of continued proliferation of connective tissue proteins that may continue indefinitely (Atiyeh, 2005; Slemp and Kirschner, 2006).
Hypertrophic scars are characterized by increased vasculature, increased numbers of white blood cells and fibroblasts, and a thickened epidermal layer (Atiyeh, 2005). The collagen fibers in hypertrophic scars are organized and oriented parallel to the epidermal layer; however, they contain an abundance of myofibroblasts, which leads to contracture formation. One positive feature of hypertrophic scars (compared with keloids) is their potential to eventually regress during the remodeling phase (Slemp and Kirschner, 2006).
Keloids are a much more serious problem than hypertrophic scars. Keloids may continue to expand, creating both functional and cosmetic deficits. In addition, scars that initially appear to be “normal” may deviate into a pattern of keloid growth over time. Biochemical analysis of keloid scars reveals increased numbers of receptor sites for growth factors and major abnormalities in the behavior of fibroblasts and keratinocytes, which may partially explain the excessive production of ECM. Most importantly, the balance between synthesis and degradation of connective tissue proteins is lost. This loss of balance is theorized to result from abnormalities in cell-to-cell communication and immune function, failure of apoptosis (spontaneous cell death), and the effects of hypoxia and oxygen free radicals. Keloid scars are characterized by disorganized collagen bundles; however, these scars do not contain myofibroblasts and are not associated with contracture formation (Atiyeh, 2005; Hahn et al, 2013; Slemp and Kirschner, 2006).
Risk factors for hyperproliferative scarring include a strong family history (particularly important with keloid formation), age 10 to 30 years, and darkly pigmented skin. In the United States, individuals of African American descent have a 5% to 15% increased risk compared with Caucasians (Atiyeh, 2005; Slemp and Kirschner, 2006). Exposure to mechanical stretch may be a trigger for keloid production (Ogawa et al, 2012).
Strategies designed to prevent or correct hyperproliferative scarring have provided inconsistent and often suboptimal results. Surgical excision, intralesional steroids (either single or combination steroids), and topical silicone sheeting are the most commonly prescribed therapies. Table 4-3 lists current treatment options. Research is ongoing, with the goals of accurately identifying the cellular and biochemical abnormalities that produce these scars and designing therapies to prevent and correct them (Kieran et al, 2013; Sayed and Bayat, 2013; Yamamoto and Kiyosawa, 2012).
TABLE 4-3
| Treatment | Indications | Considerations |
| Surgical excision | Hypertrophic scars
Keloid scars | Recurrence rare with hypertrophic
Recurrence common (45%–100%) with keloid if surgical excision is done without adjunctive therapies |
| Radiation (as adjunct to surgical excision) | Hypertrophic scars
Keloid scars | Promotion of fibroblast apoptosis (death)
Low recurrence rates (1%–35%) Theoretical risk of radiation-induced malignancy (no data) |
| Corticosteroid therapy (systemic), intralesional injections (single or combined agents) | Hypertrophic scars
Keloid scars | Reduces scar overgrowth, pruritus, contractures
Pain of repeated injections may limit patient adherence Adverse effects: skin atrophy, depigmentation, telangiectasias |
| Silicone gel, sheeting | Hypertrophic scars
Keloid scars | Increases wound hydration
Decreases fibroblast activity Down-regulates fibrogenic isoforms of TGF-β Worn 12–24 hr/day for at least 2–3 months Risk of maceration and skin breakdown with gel (gel use limited to areas where sheeting will not conform) |
| Laser (CDL, PDL) | Hypertrophic scars
Keloid scars | Stimulates regression of keloid
Reduces pruritus with hypertrophic (PDL) Variable results if used independently; best if used in conjunction with steroids or silicone gel CDL: recurrence rate as high as 50% PDL: Works best with steroids + silicone No comparative studies of laser, surgery, and adjunctive therapy |
| Retinoids (topical) | Hypertrophic scars
Keloid scars | Suppresses collagen synthesis
Increases epithelial cell turnover May be applied topically both preoperatively/postoperatively 80% of lesions have favorable short-term outcomes |
| Intralesional cryotherapy | Hypertrophic scars
Keloid scars | Scar regression through reduction of hyperproliferative response |
| Compression garments | Hypertrophic scars (primary indication)
Keloid scars | Hypoxic and thermal effects in compressed areas reduce fibroblast activity
Must be worn 8–24 hr/day for first 6 months Must fit correctly |
| Antiproliferative agents (5-fluorouracil, bleomycin) | Hypertrophic scars
Keloid scars | Induce scar regression
5-Fluorouracil more effective with hypertrophic Provide short-term suppression of small keloids Few small studies support use of bleomycin topically Side effects: pulmonary fibrosis, fever, rash, hyperpigmentation |
| Intralesional injections of verapamil | Keloid scars | Inhibit inflammation
Stimulate production of collagenase and other enzymes to degrade excess extracellular matrix Used for both prevention and treatment |
| Immunotherapy (immunomodulators, immunosuppressants, antibody therapy [e.g., imiquimod, tacrolimus, sirolimus, tumor necrosis factor-α, interferons, interleukins]) | Keloid scars | Suppress fibroblast activity
Promote fibroblast apoptosis Help reduce inflammation and regulate cellular activity Significant side effects depending on agent and dose |
| Suture ligature | Keloid scars | Deprives tissue of nutrients and oxygen, thus promoting tissue death
Keloid must be amenable to suture ligature Requires weekly office visits and daily maintenance of local wounds |
Data from Al-Attar et al, 2006; Berman, 2007; D’Andrea, 2002; Franz et al, 2007; Funayama et al, 2003; Gupta and Kumar, 2001; Har-Shai, 2003; Kieran et al, 2013; Parikh, 2008; Stashower, 2006; Syed and Bayat, 2013; Xia et al, 2004.
CDL, Carbon dioxide laser; PDL, pulsed dye laser; TGF, transforming growth factor.
Factors affecting the repair process
By observing a number of similar wounds and tracking their time to healing, it is possible to construct a curve that represents the healing “trajectory” for that type of wound. For example, epithelialization of a surgical wound healing by primary intention typically is complete by 48 hours, although difficult to palpate; a healing ridge develops by days 5 to 9; and initial collagen deposition should be complete by postoperative day 21. Interestingly, it also is possible to plot a “healing trajectory” for neuropathic ulcers, venous ulcers, and pressure ulcers. As described in Chapter 6, studies indicate that the usual “time to healing” is similar for all patients with a particular type of wound (Steed, 2003). Multiple factors affect the time to healing for a specific wound; it is critical for the clinician to identify and correct factors that are known to delay healing. Factors that can delay the healing process and shift the trajectory to the right are outlined in Table 4-4.
TABLE 4-4
| Factor | Effects on Repair Process | Clinical Implications |
| Perfusion/oxygenation
Hypoxia Adequate oxygen levels | Initiates new vessel development, promoting fibroblast proliferation
Critical for cellular production of ATP, bacterial killing, collagen synthesis, development of tensile strength Critical oxygen levels ≥30–40 mm Hg (higher for bacterial killing) | Hypovolemia, hypotension, vasoconstriction, vascular impairment, edema, hypoxia all deleterious to repair
Intervene to promote perfusion and oxygenation: warmth, hydration, pain control, management of edema, maintenance or restoration of blood flow Supplemental oxygen may be beneficial See Chapter 28 |
| Excessive bioburden | Causes direct tissue damage and breakdown of growth factors; inhibits keratinocyte migration, collagen synthesis, and development of tensile strength
Bacteria compete with fibroblasts for oxygen and nutrients, thus delaying repair Inflammatory response to high bacterial loads keeps wound “stuck” in inflammatory phase and prevents progression to proliferative phase of repair | Debride necrotic tissue to eliminate environment favorable to bacterial growth
Monitor wound for signs and symptoms of invasive infection (cellulitis or osteomyelitis); treat any invasive infection with systemic antibiotics Monitor for signs and symptoms of critical colonization or biofilm formation; remove biofilm mechanically and treat critical colonization topically (with antimicrobial agents) |
| Smoking/tobacco use | Byproducts (nicotine, carbon monoxide, hydrogen cyanide) reduce oxygenation, impair immune response, reduce fibroblast activity, increase platelet adhesion and thrombus formation
Smoking associated with significantly higher infection rates | Counsel patients on negative effects of tobacco use
Offer comprehensive program for smoking cessation: support groups, nicotine replacement, medications (No increase in wound infection with nicotine patch) See Chapter 28 |
| Nutritional status | Adequate nutritional status critical for collagen synthesis, tensile strength, immune function
Critical nutrients include micronutrients (vitamins, minerals), key amino acids (glutamine, l-arginine), adequate protein | Nutritional assessment and support are critical aspects of effective wound care program and must include attention to micronutrients and calorie and protein intake
See Chapter 27 |
| Diabetes mellitus | Associated with | Tight Glycemic control (glucose levels <150 mg/dl) associated with a reduced incidence of postoperative wound infections
See Chapter 14 |
| Obesity | Adipose tissue poorly vascularized
Large volumes of adipose tissue put additional stress on incisional lines, increasing risk of dehiscence Associated with higher incidence of infection, seroma formation, wound dehiscence | Monitor intake to ensure adequate intake of protein and micronutrients and appropriate caloric intake
Incisional support (binders) beneficial in reducing risk of dehiscence See Chapter 35 |
| Medications | Chemotherapeutic agents impair production of white blood cells and fibroblasts, interfering with both inflammatory and proliferative phases of repair and increasing risk of infection
Corticosteroids suppress inflammation and reduce proliferation of keratinocytes and fibroblasts, thereby impairing both granulation and epithelial resurfacing; impact is time and dose dependent, with impairment limited primarily to patients receiving steroids in doses >40 mg/day and for at least 30 days before wounding High doses of NSAIDs may impair healing | Delay chemotherapy when possible to permit healing (delay in healing most significant when chemotherapy given within first weeks following surgery/injury)
Limited data suggest that topical vitamin A may partially reverse negative effects of corticosteroids (applied to clean wound bed before dressing application). Systemic administration is not recommended at present due to concerns that this could block the intended effects of the steroids Recommended dose range is 25,000–100,000 international units per day (note this recommendation based on limited and anecdotal data) For patient with impaired healing taking high-dose NSAIDs, collaborate with prescribing provider to reduce dose if possible |
| Advanced age | Diminished proliferation of cells critical to repair
Hormonal changes Increased number of senescent cells Diminished production of growth factors and possible reduction in receptor sites for growth factors Multiple existing comorbidities | Correct any reversible comorbidities
Optimize nutritional status Provide evidence-based wound care |
| Immunosuppression | Increased susceptibility to infection
Compromises body’s ability to manifest signs of infection | Provide meticulous wound care to minimize risk of infection
Monitor wound closely for muted signs of infection (faint erythema, mild increase in exudate, pain) and treat appropriately |
| Stress | Increased production of corticosteroids | Implement strategies to minimize stress: pain control, environmental management, patient education and counseling |
| Cellular senescence | Impairs healing due to reduced ability of cells involved in repair to replicate normally
Associated with aging and with chronic wound environment, specifically high levels of ROS | Control bioburden and inflammation/levels of ROS to avoid damage to cellular DNA and “induced senescence” |
| Other | Malignancy
Multisystem failure Failure to maintain clean, moist wound bed | Provide holistic wound management that addresses comorbidities
Implement topical therapy to eliminate necrotic tissue and heavy bacterial loads Keep wound surface clean and moist |
Data from Anstead, 1998; Burns et al, 2003; Ehrlich and Hunt, 1968; Ehrlich and Hunt, 1969; Greenhalgh, 2003; Hardman and Ashcroft, 2008; Howard et al, 2013; Manassa et al, 2003; Pittman, 2007; Sorensen et al, 2003; Telgenhoff and Shroot, 2005; Wang et al, 2013; Whitney, 2003; Wicke et al, 2009; Wientjes, 2002; Williams and Barbul, 2003; Wilson and Clark, 2003.
ATP, Adenosine triphosphate; NSAID, nonsteroidal antiinflammatory drug; ROS, reactive oxygen species.
Summary
Wound healing is a complex series of events. Normally wound healing is initiated by an injury that leads to clot formation and platelet degranulation, is controlled by a myriad of cytokines and growth factors, and is affected significantly by systemic factors such as perfusion, nutritional status, and steroid levels. Effective management of any wound requires an understanding of the normal repair process and the factors that may interfere with normal repair. This understanding provides the foundation for comprehensive assessment of the wound and of the patient and for selection of interventions designed to optimize healing.