X-ray interactions with matter
Objectives
• Explain classical interactions, including production, energy, effects on patient dose, and effects on image quality.
• Explain Compton interactions, including production, energy, effects on patient dose, and effects on image quality.
• Explain photoelectric interactions, including production, energy, effects on patient dose, and effects on image quality.
• Explain photodisintegration.
• Relate differential absorption to x-ray beam interactions with the human body and image formation.
Key terms
absorption
classical interaction
Compton electron
Compton scattering
differential absorption
main-chain scission
occupational exposure
pair production
photodisintegration
photoelectric interaction
photoelectron
radiolucent
radiopaque
secondary photons
transmission
Introduction
It is helpful for the radiographer to understand the way x-ray photons interact with matter for two important reasons (Figure 7-1). First, it allows the radiographer to minimize the physical effects of x-ray photons on the patient that result in radiation dose and biologic harm. Second, an understanding of x-ray photon–body tissue interaction allows the radiographer to better manipulate how the particular anatomic area of interest appears radiographically. Minimizing harm to the patient and producing a quality radiographic image are both integral to the role and responsibility of the radiographer. X-rays may interact with matter in five different ways, depending on their energy. This chapter discusses all five, but keep in mind that only the first three occur within the range of energy used in diagnostic radiography. The chapter concludes with a discussion of differential absorption and other terms related to how the x-ray beam interacts in general with body tissues.

CRITICAL CONCEPT 7-1
Understanding X-ray Interactions
Interactions between x-ray photons and human tissues determine how anatomic structures are imaged and the patient’s radiation dose.
Classical interactions
Classical interactions are also commonly known as coherent scattering or Thomson scattering. In this scattering event the incident x-ray photon interacts with an orbital electron of a tissue atom and changes direction. In this particular interaction the incident x-ray photon is of a rather low energy (generally less than 10 keV). When such low-energy incident photons interact with tissue atoms, they are not likely to ionize (remove orbital electrons from their shell). Instead, the atom absorbs the energy of this x-ray photon, causing excitation of the atom, and then immediately releases the energy in a new direction (Figure 7-2). Because the energy is reemitted in a new direction, it is now a scatter photon. It is of equal energy to the incident photon but travels in a new direction. Because of its low energy, most classical scatter photons are absorbed in the body through other interactions and do not contribute significantly to the image, but do add slightly to patient dose.

CRITICAL CONCEPT 7-2
Classical Interactions
Classical interactions do not involve ionization of the atom. They are scattering events that do not contribute significantly to the image but contribute slightly to patient dose.
Compton interactions
Compton scattering occurs throughout the diagnostic range, but generally involves moderate-energy x-ray photons (e.g., 20-40 keV). In this interaction, an incident x-ray photon enters a tissue atom, interacts with an orbital electron (generally a middle- or outer-shell electron), and removes it from its shell. In doing so, the incident photon loses up to one third of its energy and is usually deflected in a new direction (Figure 7-3). This interaction does three things: First, it ionizes the atom, making it unstable. Ionization in the body is significant because the atom is changed and may bond differently to other atoms, potentially causing biologic damage. If one of the “middle” orbital shells is involved, a characteristic cascade (outer-shell electrons filling inner-shell vacancies and emitting x-ray photons) also results, creating characteristic photons just as in the tube target. But here they are called secondary photons. These secondary photons are x-ray photons, but of a rather low-energy variety. Such photons generally contribute only to patient dose. Second, the ejected electron, called a Compton electron, or secondary electron, leaves the atom with enough energy to go through interactions of its own in adjacent atoms. The type of interaction the Compton electron undergoes depends on the energy it has and the type of atom it interacts with. Third, the incident photon is deflected in a new direction and is now a Compton scatter photon. It too, has enough energy to go through other interactions in the tissues or exit the patient and interact with the image receptor. The problem with Compton scatter interacting with the image receptor is that it is not following its original path through the body and strikes the image receptor in the wrong area. In so doing, it contributes no useful information to the image and only results in image fog. Because most scattered photons are still directed toward the image receptor and result in image fog, it is desirable to minimize Compton scattering as much as possible.
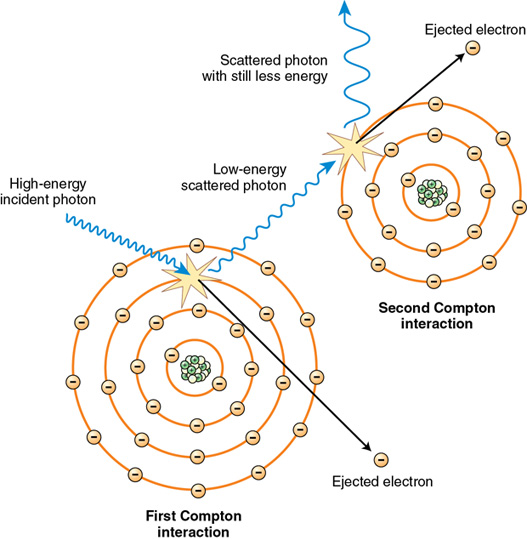
Compton scattering is one of the most prevalent interactions between x-ray photons and the human body in general diagnostic imaging and is responsible for most of the scatter that fogs the image. The probability of Compton scattering does not depend on the atomic number of atoms involved. Compton scattering may occur in both soft tissue and bone. The probability of Compton scattering is related to the energy of the photon. As x-ray photon energy increases, the probability of that photon penetrating a given tissue without interaction increases. However, with this increase in photon energy, the likelihood of Compton interactions relative to photoelectric interactions also increases.
Compton scatter photons may travel in any direction from their point of scattering. A deflection of zero degrees means no energy is transferred. Those photons scattered at 180 degrees represent maximum deflection and energy transfer. But keep in mind that the scattered photon still retains about two thirds of its energy. This is one reason the radiographer should never stand near the patient during exposure. Some Compton scatter photons exit the patient and would expose the radiographer. This is why shielding (lead aprons, lead gloves, etc.) is necessary during fluoroscopy or any procedure in which the radiographer or other health care worker may be near the patient and x-ray tube during exposure. It is important for the radiographer to remember that Compton scattering is the major source of occupational exposure.
CRITICAL CONCEPT 7-3
Compton Interactions
Compton interactions are scattering events that ionize the atom. They may contribute negatively to the radiographic image as fog and add to patient and occupational radiation dose.
Photoelectric interactions
Photoelectric interactions occur throughout the diagnostic range (e.g., 20-120 kVp) and involve inner-shell orbital electrons of tissue atoms. For photoelectric events to occur, the incident x-ray photon energy must be equal to or greater than the orbital shell binding energy. In these events the incident x-ray photon interacts with the inner-shell electron of a tissue atom and removes it from orbit. In the process, the incident x-ray photon expends all of its energy and is totally absorbed (Figure 7-4). The resulting ejected electron is called a photoelectron. The energy transfer between the incident photon and inner-shell electron is equal to the incident photon energy minus the binding energy of the orbital electron. This energy transfer constitutes the energy of the photoelectron. In soft-tissue atoms, the energy of the photoelectron is nearly equal to that of the incident x-ray photon because the binding energy of the soft tissue atom is very low and more is left over as kinetic energy for the photoelectron. In bone, the energy of the photoelectron is less because the orbital electron binding energy of bone atoms is greater and the incident x-ray photon has to expend more energy to remove it, leaving less as kinetic energy for the photoelectron. In either case, the photoelectron has enough kinetic energy to undergo interactions of its own before filling a vacancy in another atom elsewhere.
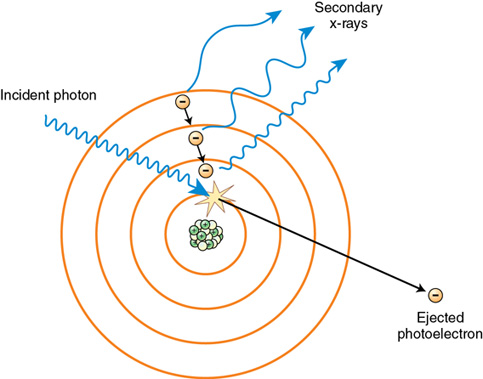
Note that this absorption that constitutes photoelectric interactions contributes significantly to patient dose accrued with each diagnostic image. Although some absorption is necessary to create an x-ray image, it is the radiographer’s responsibility to select technical factors that strike a balance between image quality (absorption and transmission of x-ray photons needed to produce a good image) and patient dose.
In photoelectric interactions, as with Compton interactions, the tissue atom is ionized. In the case of photoelectric interactions, the inner-shell vacancy makes the atom unstable; to regain stability a characteristic cascade occurs, producing secondary x-ray photons. This cascade is the same phenomenon that occurs with Compton interactions that produce secondary photons. Again, these secondary photons are of low energy and are absorbed by the body in other photoelectric events. Note that the absorption of these secondary photons also contributes to patient dose.
CRITICAL CONCEPT 7-4
Photoelectric Interactions
Photoelectric events result in the total absorption of the incident photon. For this to occur, the incident photon energy must be equal to or greater than the orbital shell binding energy. In this process the atom is ionized, a characteristic cascade producing secondary photons results, and an ejected photoelectron exits the atom with enough energy to undergo many more interactions.
The probability of photoelectric interaction depends on the energy of the incident photons and the atomic number of the tissue atoms with which they interact. For photoelectric interactions to occur, the incident x-ray photon energy must be greater than or equal to the inner-shell binding energy of the tissue atoms involved. The greatest number of photoelectric interactions occurs when the incident x-ray photon energy is equal to or slightly greater than the inner-shell binding energy of the tissue atom. As the incident photon energy begins to exceed the inner-shell binding energy of the tissue atom, the chances of photoelectric interaction begin to decline and the chances increase that it will penetrate the tissue being examined. This function is a cubic relationship. That is, the probability of a photoelectric event is inversely proportional to the third power of the x-ray energy. What this means to the radiographer is that if a kVp range is too high for the anatomic part of interest, less absorption takes place, and some absorption is necessary for image formation.
The probability of photoelectric events is directly proportional to the third power of the atomic number of the absorber. What this cubic relationship means to the radiographer is that when he or she makes small changes in the kVp setting or there are small changes in the atomic number of the tissue (due to anatomic variations or a pathologic condition), large changes in the probability of photoelectric events will result. With tissues, the higher the atomic number of the tissue atom, the greater the number of photoelectric events. Such atoms are more complex; that is, they have more electrons and stronger binding energies and are more likely to absorb the incident x-ray photon. This is why bone shows up as lighter shades on the radiographic image. In bone, more photons are absorbed, which means fewer photons are exposing the image receptor, resulting in the lighter shades of the image.
THEORY TO PRACTICE 7-1
If more photoelectric events are needed to make a particular structure visible on a radiographic image (when, for example, the tissues to be examined do not have high–atomic number atoms), contrast agents such as barium or iodine are added. These agents have high atomic numbers and thereby increase the number of photoelectric events in these tissues. Protective shielding is another way of using photoelectric interactions. Lead has a very high atomic number and is used as a shielding material because the odds are great that photons will be absorbed by it.
CRITICAL CONCEPT 7-5
Photoelectric Probability
Photoelectric probability depends on the energy of the incident photon and the atomic number of the tissue being irradiated. The energy must be equal to or greater than the orbital shell binding energy, and the greater the atomic number of the tissue atom, the greater the probability of photoelectric interactions.
Pair production
Pair production occurs only with very high–energy photons of 1.02 MeV or greater. The interaction occurs when the incident x-ray photon has enough energy to escape interaction with the orbital electrons and interact with the nucleus of the tissue atom. In this interaction, two particles are produced: a positron (positively charged electron) and an electron (Figure 7-5). For these particles to exist, they must each have energy of 0.51 MeV (the energy equivalent of an electron). If the photon has energy greater than 1.02 MeV, it is shared between the two as kinetic energy.
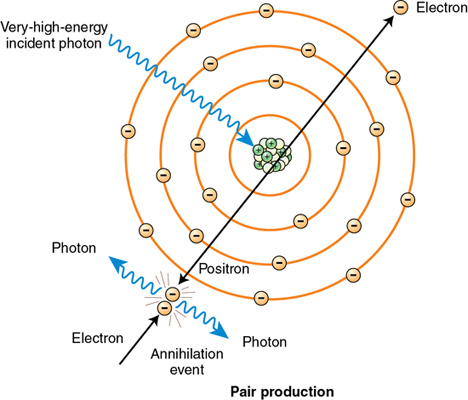
Both particles travel out of the atom. The electron undergoes many interactions before coming to rest in another atom. The positron is an “unnatural particle” and, as such, travels until it strikes an electron, causing an annihilation event. In this annihilation event, the positron and the electron it interacts with are destroyed and their energy is converted into two x-ray photons that radiate out of the atom. Pair production does not occur in radiography because the energy levels required exceed the range used in diagnostic x-ray production.
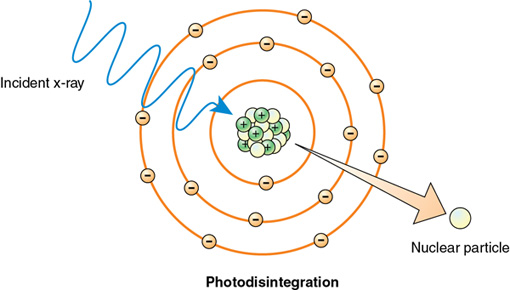
Photodisintegration
The last type of interaction between x-rays and matter is called photodisintegration. Photons with extremely high energies of more than 10 MeV may strike the nucleus of the atom and make it unstable. In photodisintegration the nucleus of the atom involved regains stability by ejecting a nuclear particle such as a proton, neutron, or alpha particle (Figure 7-6). Like pair production, photodisintegration does not occur in radiography because the energy levels required far exceed the kVp range used in diagnostic x-ray production.
Differential absorption
Differential absorption is the difference between the x-ray photons that are absorbed photoelectrically and those that penetrate the body (Figure 7-7). It is called differential because different body structures absorb x-ray photons to different extents. Anatomic structures such as bone are denser and absorb more x-ray photons than structures filled with air such as the lungs.
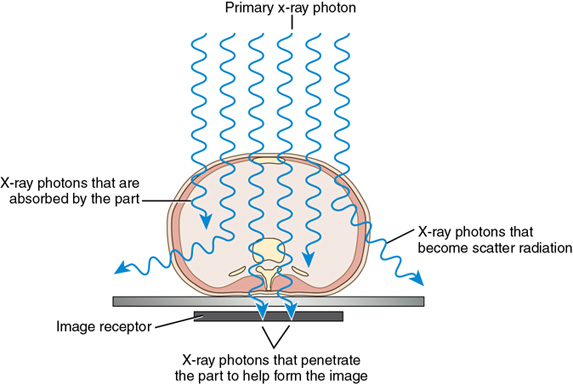
MAKE THE IMAGING CONNECTION 7-1
The process of image formation is the result of differential absorption of the x-ray beam as it interacts with anatomic tissue. Differential absorption of the primary beam creates an image that structurally represents the anatomic area of interest.
CRITICAL CONCEPT 7-6
Differential Absorption
Differential absorption is the difference between x-ray photons that are absorbed photoelectrically and those that penetrate the body. Denser tissue such as bone has greater absorption.
When radiographers speak broadly about differential absorption—how the x-ray beam interacts with the tissues of the body—they may speak of transmission versus absorption. Transmission refers to those x-ray photons that pass through the body and reach the image receptor. It is desirable for some of the x-ray photons to pass through the body area of interest or no image would result. X-ray photons reaching the image receptor create the dark (less bright) shades of the image. Absorption refers to those photons that are attenuated by the body and do not reach the image receptor. Absorption has the opposite effect on the image as penetration. Recall that these photons are absorbed photoelectrically and will not reach the image receptor. This “lack” of exposure to the image receptor results in the lighter (brighter) shades of the image. It is also desirable to have some absorption; otherwise, the image would be uniformly dark.
Again, absorption depends on the density of body tissues through which the x-ray photons are passing. Denser tissue, such as bone, increases the probability of x-ray photons being absorbed in photoelectric interactions. The result is that fewer x-ray photons pass through these areas of the body to reach the image receptor, and those structures appear lighter. Body structures that readily absorb x-rays are called radiopaque. Less dense structures have a much lower probability of absorption and are said to be radiolucent.
Throughout this chapter x-ray interactions with matter have been discussed at the atomic level where they occur. It is important for the radiographer to understand these atomic-level interactions, but it is also important to relate these transfers of energy to a macrolevel where radiation dose and damage is more apparent. Although this subject will be covered thoroughly in the radiography student’s radiobiology course, a brief discussion is offered here to complete the concepts and subject of this chapter.
Macromolecules are large molecules made up of thousands of atoms. When an x-ray photon interacts with one of these atoms as previously described, the energy transfer may manifest as a change to the structure of the macromolecule. The three most common effects are main-chain scission, cross-linking, and point lesions (Figure 7-8). Main-chain scission refers to a breakage of the major structure, the framework if you will, of the macromolecule itself. Cross-linking is the result of the formation of “limbs” as a result of irradiation (although these exist naturally in some macromolecules) that “stick” to adjacent parts of the macromolecule or neighboring molecules, creating unnatural framework. Point lesions are the result of damage to a single chemical bond. Think of these as a “wound” to the macromolecule that may cause a malfunction of the macromolecule and damage to the cell overall. The most sensitive of molecules is DNA. Damage similar to that previously described may occur in DNA as a result of radiation exposure and manifest as a range of responses from minor damage that is reversible to malignant response and permanent damage. Finally, because the human body is about 80% water, irradiation of water (interactions between x-ray photons and water molecules) can create harmful free radicals that then indirectly damage molecules and cells.

THEORY TO PRACTICE 7-2
It is important to remember that absorption equates to patient dose. Because of the strong inverse dependence of photoelectric absorption on x-ray energy, patient dose increases as kVp decreases. When there is too much absorption resulting from a kVp setting that is too low, the patient experiences an increased radiation dose. Patient dose is a result of the radiation absorbed by the body, not the radiation that passes through it.
CRITICAL CONCEPT 7-7
Differential Absorption and Clinical Practice
In the clinical setting, some absorption and some penetration of x-ray photons through the anatomic area of interest are necessary for image production. But keep in mind that absorption equates to patient dose and it is the radiographer’s responsibility to maintain a balance of absorption and penetration so that the risk of exposure outweighs the biologic harm to the tissues and so that the resulting image benefits the patient.
Summary
• Classical interactions are scattering events in which the atom involved is not ionized. They do not significantly affect the radiographic image but contribute slightly to patient dose.
• Compton interactions are scattering events in which the atom is ionized and a characteristic cascade may result. The incident photon is deflected in a new direction, becoming a scatter photon, and this photon and the ejected Compton electron both have sufficient energy to undergo many other interactions.
• Compton interactions are one of the most prevalent interactions and may contribute negatively to the radiographic image as fog and add to patient and occupational radiation dose.
• Photoelectric events result in the total absorption of the incident photon. For this to occur, the incident photon energy must be equal to or greater than the orbital shell binding energy.
• Photoelectric events result in ionization of the atom, a characteristic cascade producing secondary photons, and an ejected photoelectron capable of many more interactions.
• Photoelectric probability depends on the energy of the incident photon and the atomic number of the tissue being irradiated.
• Pair production involves very high–energy photon interactions with the nucleus of the tissue atom producing a positron and an electron. This event occurs outside of the energy range of diagnostic radiography.
• Photodisintegration is a very high–energy photon interaction with the nucleus of a tissue atom, resulting in the nucleus emitting a nuclear particle to regain stability.
• Differential absorption is the difference between x-ray photons that are absorbed photoelectrically and those that penetrate the body.
• Some absorption and some transmission of x-ray photons through the anatomic area of interest are necessary for image production. But keep in mind that absorption equates to patient dose and a balance of absorption and penetration must be kept so that the benefit of exposure outweighs the biologic harm to the tissues.
• X-ray photons interact at an atomic level (classical, Compton, photoelectric, pair production, and photodisintegration), but the damage they may cause is manifest at the macro level through changes or damage sustained by the macromolecules. This may occur directly when x-ray photons interact with atoms composing the macromolecule or indirectly through irradiation of water that creates harmful free radicals that then damage macromolecular structures.
Critical thinking questions
1. How would a radiographer use his/her knowledge of x-ray interactions with human tissue to describe how an image is formed to a patient?
2. Imagine you are testifying before Congress on the need for regulation of professionals who dispense ionizing radiation for medical imaging and a committee member wants to know what happens inside the body when it is exposed to x-rays. Use material from this chapter to formulate a response that a layperson would understand.
Review questions
1. Which of the following is a major source of occupational exposure?
2. Which interaction, within the diagnostic range, does not involve the removal of an orbital electron?
3. Which interaction requires 1.02 MeV of energy?
4. A photon of 10 MeV colliding with a nucleus will likely result in what type of interaction?
5. Which technique will produce the greatest number of photodisintegration events in an average abdomen?
6. Which of the following events will not occur in the diagnostic range of x-ray energies?
7. Positive contrast media is administered to increase what type of interactions?
8. Which of the following contributes most to image fog?
9. Which interaction in the diagnostic range involves the total absorption of the incident photon?
10. When the kV selected is equal to or slightly greater than the inner-shell binding energy of a target tissue atom, which interaction predominates?