Cell Injury, Cell Death, and Adaptations
Introduction to Pathology
The field of pathology is devoted to understanding the causes of disease and the changes in cells, tissues, and organs that are associated with disease and give rise to the presenting signs and symptoms in patients. There are two important terms that students will encounter throughout their study of pathology and medicine:
• Etiology refers to the underlying causes and modifying factors that are responsible for the initiation and progression of disease. It is now clear that many common diseases, such as hypertension, diabetes, and cancer, are caused by a combination of inherited genetic susceptibility and various environmental triggers. Elucidating the genetic and environmental factors underlying diseases is a major theme of modern medicine.
• Pathogenesis refers to the mechanisms of development and progression of disease, which account for the cellular and molecular changes that give rise to the specific functional and structural abnormalities that characterize any particular disease. Thus, etiology refers to why a disease arises and pathogenesis describes how a disease develops (Fig. 2.1).
Defining the etiology and pathogenesis of disease not only is essential for understanding disease but also is the basis for developing rational treatments and effective preventive measures. Thus, pathology provides the scientific foundation for the practice of medicine.
To render diagnoses and guide therapy in clinical practice, pathologists identify changes in the gross or microscopic appearance (morphology) of cells and tissues, and biochemical alterations in body fluids (such as blood and urine). Pathologists also use a variety of morphologic, molecular, and other techniques to define the biochemical, structural, and functional changes that occur in cells, tissues, and organs in response to injury. We begin, in this chapter, with a discussion of cellular abnormalities induced by a variety of internal (e.g., genetic) and external (e.g., environmental) abnormalities and stresses.
Overview of Cellular Responses to Stress and Noxious Stimuli
Cells actively interact with their environment, constantly adjusting their structure and function to accommodate changing demands and extracellular stresses. The intracellular milieu of cells is normally tightly regulated such that it remains fairly constant, a state referred to as homeostasis. As cells encounter physiologic stresses (such as increased workload in the heart) or potentially injurious conditions (such as nutrient deprivation), they can undergo adaptation, achieving a new steady state and preserving viability and function. If the adaptive capability is exceeded or if the external stress is inherently harmful or excessive, cell injury develops (Fig. 2.2). Within certain limits, injury is reversible, and cells return to their stable baseline; however, if the stress is severe, persistent, or rapid in onset, it results in irreversible injury and death of the affected cells. Cell death is one of the most crucial events in the evolution of disease in any tissue or organ. It results from diverse causes, including ischemia (lack of blood flow), infections, toxins, and immune reactions. Cell death also is a normal and essential process in embryogenesis, the development of organs, and the maintenance of tissue homeostasis.
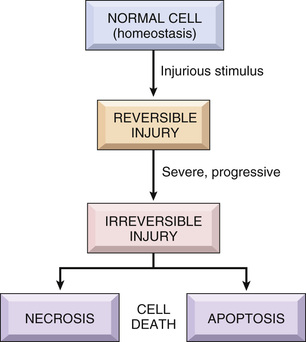
Because damage to cells is the basis of all disease, in this chapter we discuss first the causes, mechanisms, and consequences of the various forms of acute cell injury, including reversible injury and cell death. We then consider cellular adaptations to stress and conclude with two other processes that affect cells and tissues: the deposition of abnormal substances and cell aging.
Causes of Cell Injury
The causes of cell injury span a range from gross physical trauma, such as after a motor vehicle accident, to a single gene defect that results in a nonfunctional enzyme in a specific metabolic disease. Most injurious stimuli can be grouped into the following categories.
Hypoxia and ischemia. Hypoxia, which refers to oxygen deficiency, and ischemia, which means reduced blood supply, are among the most common causes of cell injury. Both deprive tissues of oxygen, and ischemia, in addition, results in a deficiency of essential nutrients and a build up of toxic metabolites. The most common cause of hypoxia is ischemia resulting from an arterial obstruction, but oxygen deficiency also can result from inadequate oxygenation of the blood, as in a variety of diseases affecting the lung, or from reduction in the oxygen-carrying capacity of the blood, as with anemia of any cause, and carbon monoxide (CO) poisoning.
Toxins. Potentially toxic agents are encountered daily in the environment; these include air pollutants, insecticides, CO, asbestos, cigarette smoke, ethanol, and drugs. Many drugs in therapeutic doses can cause cell or tissue injury in a susceptible patient or in many individuals if used excessively or inappropriately (Chapter 7). Even innocuous substances, such as glucose, salt, water and oxygen, can be toxic.
Infectious agents. All types of disease-causing pathogens, including viruses, bacteria, fungi, and protozoans, injure cells. The mechanisms of cell injury caused by these diverse agents are discussed in Chapter 9.
Immunologic reactions. Although the immune system defends the body against pathogenic microbes, immune reactions also can result in cell and tissue injury. Examples are autoimmune reactions against one's own tissues, allergic reactions against environmental substances, and excessive or chronic immune responses to microbes (Chapter 5). In all of these situations, immune responses elicit inflammatory reactions, which are often the cause of damage to cells and tissues.
Genetic abnormalities. Genetic aberrations can result in pathologic changes as conspicuous as the congenital malformations associated with Down syndrome or as subtle as the single amino acid substitution in hemoglobin giving rise to sickle cell anemia (Chapter 7). Genetic defects may cause cell injury as a consequence of deficiency of functional proteins, such as enzymes in inborn errors of metabolism, or accumulation of damaged DNA or misfolded proteins, both of which trigger cell death when they are beyond repair.
Nutritional imbalances. Protein–calorie insufficiency among impoverished populations remains a major cause of cell injury, and specific vitamin deficiencies are not uncommon even in developed countries with high standards of living (Chapter 8). Ironically, excessive dietary intake may result in obesity and also is an important underlying factor in many diseases, such as type 2 diabetes mellitus and atherosclerosis.
Physical agents. Trauma, extremes of temperature, radiation, electric shock, and sudden changes in atmospheric pressure all have wide-ranging effects on cells (Chapter 8).
Aging. Cellular senescence results in a diminished ability of cells to respond to stress and, eventually, the death of cells and of the organism. The mechanisms underlying cellular aging are discussed at the end of this chapter.
With this introduction, we proceed to a discussion of the progression and morphologic manifestations of cell injury, and then to the biochemical mechanisms in injury caused by different noxious stimuli.
Sequence of Events in Cell Injury and Cell Death
Although various injurious stimuli damage cells through diverse biochemical mechanisms, all tend to induce a stereotypic sequence of morphologic and structural alterations in most types of cells.
Reversible Cell Injury
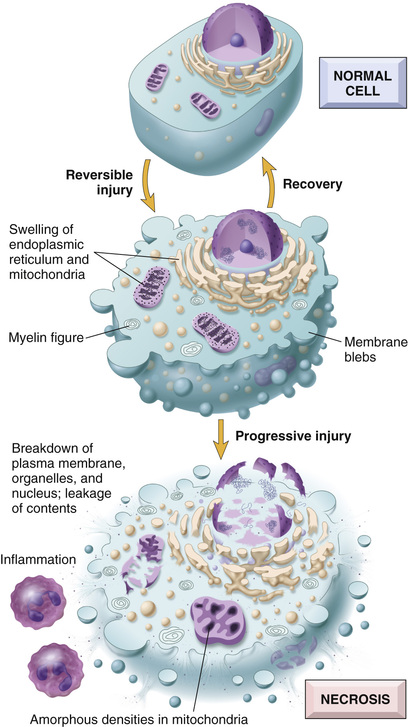
Reversible injury is the stage of cell injury at which the deranged function and morphology of the injured cells can return to normal if the damaging stimulus is removed (Fig. 2.3). In reversible injury, cells and intracellular organelles typically become swollen because they take in water as a result of the failure of energy-dependent ion pumps in the plasma membrane, leading to an inability to maintain ionic and fluid homeostasis. In some forms of injury, degenerated organelles and lipids may accumulate inside the injured cells.
In some situations, potentially injurious insults induce specific alterations in cellular organelles, such as the ER. The smooth ER is involved in the metabolism of various chemicals, and cells exposed to these chemicals show hypertrophy of the ER as an adaptive response that may have important functional consequences. For instance, many drugs, including barbiturates, which were commonly used as sedatives in the past and are still used as a treatment for some forms of epilepsy, are metabolized in the liver by the cytochrome P-450 mixed-function oxidase system found in the smooth ER. Protracted use of barbiturates leads to a state of tolerance, marked by the need to use increasing doses of the drug to achieve the same effect. This adaptation stems from hypertrophy (an increase in volume) of the smooth ER of hepatocytes and a consequent increase in P-450 enzymatic activity. P-450–mediated modification of compounds sometimes leads to their detoxification, but in other instances converts them into a dangerous toxin; one such example involves carbon tetrachloride (CCl4), discussed later. Cells adapted to one drug demonstrate an increased capacity to metabolize other compounds handled by the same system. Thus, if patients taking phenobarbital for epilepsy increase their alcohol intake, they may experience a drop in blood concentration of the anti-seizure medication to subtherapeutic levels because of smooth ER hypertrophy in response to the alcohol.
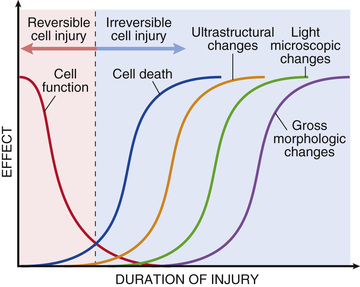
With persistent or excessive noxious exposures, injured cells pass a nebulous “point of no return” and undergo cell death. The clinical relevance of defining this transition point is obvious—if the biochemical and molecular changes that predict cell death can be identified, it may be possible to devise strategies for preventing the transition from reversible to irreversible cell injury. Although there are no definitive morphologic or biochemical correlates of irreversibility, it is consistently characterized by three phenomena: the inability to restore mitochondrial function (oxidative phosphorylation and adenosine triphosphate [ATP] generation) even after resolution of the original injury; the loss of structure and functions of the plasma membrane and intracellular membranes; and the loss of DNA and chromatin structural integrity. As discussed in more detail later, injury to lysosomal membranes results in the enzymatic dissolution of the injured cell, which is the culmination of necrosis.
Cell Death
When cells are injured they die by different mechanisms, depending on the nature and severity of the insult.
• Severe disturbances, such as loss of oxygen and nutrient supply and the actions of toxins, cause a rapid and uncontrollable form of death that has been called “accidental” cell death. The morphological manifestation of accidental cell death is necrosis (Greek, necros = death) (Table 2.1). Necrosis is the major pathway of cell death in many commonly encountered injuries, such as those resulting from ischemia, exposure to toxins, various infections, and trauma. Necrosis is traditionally considered the inevitable end result of severe damage that is beyond salvage and is not thought to be regulated by specific signals or biochemical mechanisms; in other words, necrosis happens accidentally because the injury is too severe to be repaired and many cellular constituents simply fail or fall apart.
Table 2.1
Features of Necrosis and Apoptosis
| Feature | Necrosis | Apoptosis |
| Cell size | Enlarged (swelling) | Reduced (shrinkage) |
| Nucleus | Pyknosis → karyorrhexis → karyolysis | Fragmentation into nucleosome-sized fragments |
| Plasma membrane | Disrupted | Intact; altered structure, especially orientation of lipids |
| Cellular contents | Enzymatic digestion; may leak out of cell | Intact; may be released in apoptotic bodies |
| Adjacent inflammation | Frequent | No |
| Physiologic or pathologic role | Invariably pathologic (culmination of irreversible cell injury) | Often physiologic means of eliminating unwanted cells; may be pathologic after some forms of cell injury, especially DNA and protein damage |
• In contrast, when the injury is less severe, or cells need to be eliminated during normal processes, they activate a precise set of molecular pathways that culminate in death. Because this kind of cell death can be manipulated by therapeutic agents or genetic mutations, it is said to be “regulated” cell death. The morphologic appearance of most types of regulated cell death is apoptosis (see Table 2.1). In some instances, regulated cell death shows features of both necrosis and apoptosis, and has been called necroptosis. The discovery of these previously unrecognized forms of cell death that were regulated by identifiable genes and signaling pathways showed that cell death can be a controlled process. The idea of regulated cell death also raises the possibility that specific molecular pathways can be targeted therapeutically to prevent the loss of cells in pathologic conditions. Apoptosis is a process that eliminates cells with a variety of intrinsic abnormalities and promotes clearance of the fragments of the dead cells without eliciting an inflammatory reaction. This “clean” form of cell suicide occurs in pathologic situations when a cell's DNA or proteins are damaged beyond repair or the cell is deprived of necessary survival signals. But unlike necrosis, which is always an indication of a pathologic process, apoptosis also occurs in healthy tissues. It serves to eliminate unwanted cells during normal development and to maintain constant cell numbers, so it is not necessarily associated with pathologic cell injury. These types of physiologic cell death are also called programmed cell death.
It is important to point out that cellular function may be lost long before cell death occurs, and that the morphologic changes of cell injury (or death) lag far behind loss of function and viability (Fig. 2.5). For example, myocardial cells become noncontractile after 1 to 2 minutes of ischemia, but may not die until 20 to 30 minutes of ischemia have elapsed. Morphologic features indicative of the death of ischemic myocytes appear by electron microscopy within 2 to 3 hours after the death of the cells, but are not evident by light microscopy until 6 to 12 hours later.
Necrosis
Necrosis is a form of cell death in which cellular membranes fall apart, and cellular enzymes leak out and ultimately digest the cell (Fig. 2.3). Necrosis elicits a local host reaction, called inflammation, that is induced by substances released from dead cells and which serves to eliminate the debris and start the subsequent repair process (Chapter 3). The enzymes responsible for digestion of the cell are derived from lysosomes and may come from the dying cells themselves or from leukocytes recruited as part of the inflammatory reaction. Necrosis often is the culmination of reversible cell injury that cannot be corrected.
The biochemical mechanisms of necrosis vary with different injurious stimuli. These mechanisms include: failure of energy generation in the form of ATP because of reduced oxygen supply or mitochondrial damage; damage to cellular membranes, including the plasma membrane and lysosomal membranes, which results in leakage of cellular contents including enzymes; irreversible damage to cellular lipids, proteins, and nucleic acids, which may be caused by reactive oxygen species (ROS); and others. These biochemical mechanisms are discussed later when we consider the individual causes of cell necrosis.
Morphologic Patterns of Tissue Necrosis
In severe pathologic conditions, large areas of a tissue or even entire orgrans may undergo necrosis. This may happen in association with marked ischemia, infections, and certain inflammatory reactions. There are several morphologically distinct patterns of tissue necrosis that may provide etiologic clues. Although the terms that describe these patterns do not reflect underlying mechanisms, such terms are commonly used and their implications are understood by pathologists and clinicians.
Leakage of intracellular proteins through the damaged cell membrane and ultimately into the circulation provides a means of detecting tissue-specific necrosis using blood or serum samples. Cardiac muscle, for example, contains a unique isoform of the enzyme creatine kinase and of the contractile protein troponin, whereas hepatic bile duct epithelium contains the enzyme alkaline phosphatase, and hepatocytes contain transaminases. Irreversible injury and cell death in these tissues elevate the serum levels of these proteins, which makes them clinically useful markers of tissue damage.
Apoptosis
Apoptosis is a pathway of cell death in which cells activate enzymes that degrade the cells' own nuclear DNA and nuclear and cytoplasmic proteins (Fig. 2.11). Fragments of the apoptotic cells then break off, giving the appearance that is responsible for the name (apoptosis, “falling off”). The plasma membrane of the apoptotic cell remains intact, but the membrane is altered in such a way that the fragments, called apoptotic bodies, become highly “edible,” leading to their rapid consumption by phagocytes. The dead cell and its fragments are cleared with little leakage of cellular contents, so apoptotic cell death does not elicit an inflammatory reaction. Thus, apoptosis differs in many respects from necrosis (Table 2.1).
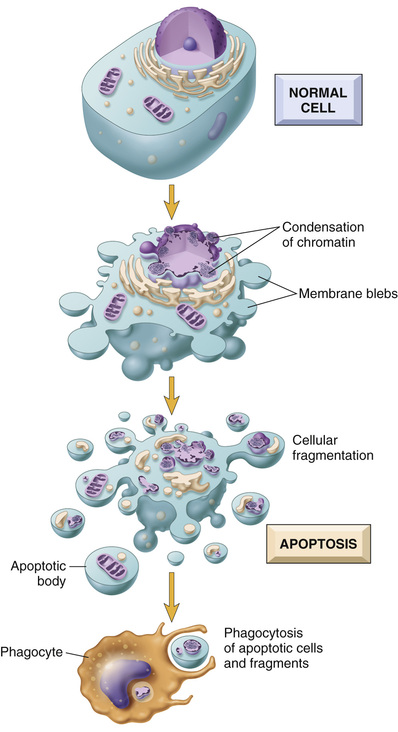
Causes of Apoptosis
Apoptosis occurs in many normal situations and serves to eliminate potentially harmful cells and cells that have outlived their usefulness (Table 2.2). It also occurs as a pathologic event when cells are damaged, especially when the damage affects the cell's DNA or proteins; thus, the irreparably damaged cell is eliminated.
• Physiologic apoptosis. During normal development of an organism, some cells die and are replaced by new ones. In mature organisms, highly proliferative and hormone-responsive tissues undergo cycles of proliferation and cell loss that are often determined by the levels of growth factors. In these situations, the cell death is always by apoptosis, ensuring that unwanted cells are eliminated without eliciting potentially harmful inflammation. In the immune system, apoptosis eliminates excess leukocytes left at the end of immune responses as well as lymphocytes that recognize self-antigens and could cause autoimmune diseases if they were not purged.
• Apoptosis in pathologic conditions. Apoptosis eliminates cells that are damaged beyond repair. This is seen when there is severe DNA damage, for example, after exposure to radiation and cytotoxic drugs. The accumulation of misfolded proteins also triggers apoptotic death; the underlying mechanisms of this cause of cell death and its significance in disease are discussed later, in the context of ER stress. Certain infectious agents, particularly some viruses, induce apoptotic death of infected cells.
Table 2.2
Physiologic and Pathologic Conditions Associated With Apoptosis
| Condition | Mechanism of Apoptosis |
| Physiologic | |
| During embryogenesis | Loss of growth factor signaling (presumed mechanism) |
| Turnover of proliferative tissues (e.g., intestinal epithelium, lymphocytes in bone marrow, and thymus) | Loss of growth factor signaling (presumed mechanism) |
| Involution of hormone-dependent tissues (e.g., endometrium) | Decreased hormone levels lead to reduced survival signals |
| Decline of leukocyte numbers at the end of immune and inflammatory responses | Loss of survival signals as stimulus for leukocyte activation is eliminated |
| Elimination of potentially harmful self-reactive lymphocytes | Strong recognition of self antigens induces apoptosis by both the mitochondrial and death receptor pathways |
| Pathologic | |
| DNA damage | Activation of proapoptotic proteins by BH3-only sensors |
| Accumulation of misfolded proteins | Activation of proapoptotic proteins by BH3-only sensors, possibly direct activation of caspases |
| Infections, especially certain viral infections | Activation of the mitochondrial pathway by viral proteins Killing of infected cells by cytotoxic T lymphocytes, which activate caspases |

Mechanisms of Apoptosis
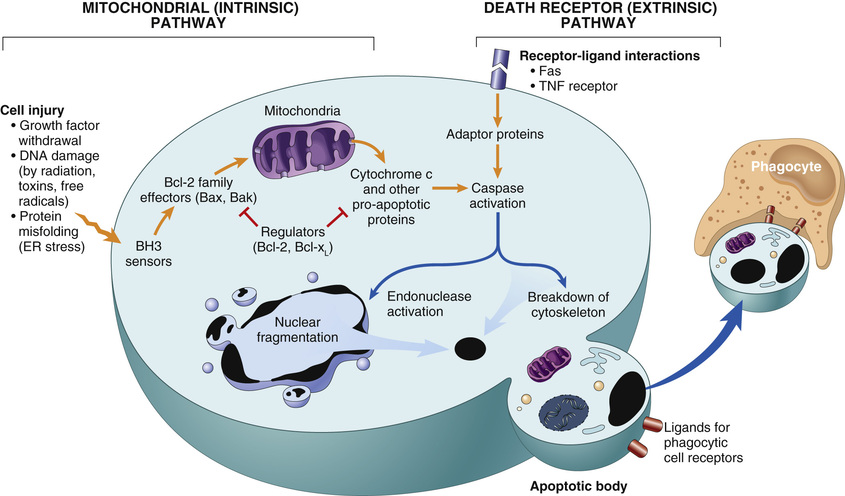
Apoptosis is regulated by biochemical pathways that control the balance of death- and survival-inducing signals and ultimately the activation of enzymes called caspases. Caspases were so named because they are cysteine proteases that cleave proteins after aspartic acid residues. Two distinct pathways converge on caspase activation: the mitochondrial pathway and the death receptor pathway (Fig. 2.12). Although these pathways can intersect, they are generally induced under different conditions, involve different molecules, and serve distinct roles in physiology and disease. The end result of apoptotic cell death is the clearance of apoptotic bodies by phagocytes.
• The mitochondrial (intrinsic) pathway seems to be responsible for apoptosis in most physiologic and pathologic situations. Mitochondria contain several proteins that are capable of inducing apoptosis, including cytochrome c. When mitochondrial membranes become permeable, cytochrome c leaks out into the cytoplasm, triggering caspase activation and apoptotic death. A family of more than 20 proteins, the prototype of which is Bcl-2, controls the permeability of mitochondria. In healthy cells, Bcl-2 and the related protein Bcl-xL, which are produced in response to growth factors and other stimuli, maintain the integrity of mitochondrial membranes, in large part by holding two proapoptotic members of the family, Bax and Bak, in check. When cells are deprived of growth factors and survival signals, or are exposed to agents that damage DNA, or accumulate unacceptable amounts of misfolded proteins, a number of sensors are activated. These sensors are called BH3 proteins because they contan the third domain seen in Bcl-family proteins. They in turn shift this delicate, life-sustaining balance in favor of pro-apoptotic Bak and Bax. As a result, Bak and Bax dimerize, insert into the mitochondrial membrane, and form channels through which cytochrome c and other mitochondrial proteins escape into the cytosol. After cytochrome c enters the cytosol, it, together with certain cofactors, activates caspase-9. The net result is the activation of a caspase cascade, ultimately leading to nuclear fragmentation and formation of apoptotic bodies.
• The death receptor (extrinsic) pathway of apoptosis. Many cells express surface molecules, called death receptors, that trigger apoptosis. Most of these are members of the tumor necrosis factor (TNF) receptor family, which contain in their cytoplasmic regions a conserved “death domain,” so named because it mediates interaction with other proteins involved in cell death. The prototypic death receptors are the type I TNF receptor and Fas (CD95). Fas ligand (FasL) is a membrane protein expressed mainly on activated T lymphocytes. When these T cells recognize Fas-expressing targets, Fas molecules are crosslinked by FasL and bind adaptor proteins via the death domain. These then recruit and activate caspase-8, which, in turn, activates downstream caspases. The death receptor pathway is involved in the elimination of self-reactive lymphocytes and in the killing of target cells by some cytotoxic T lymphocytes (CTLs) that express FasL.
In either pathway, after caspase-9 or caspase-8 is activated, it cleaves and thereby activates additional caspases that cleave numerous targets and ultimately activate enzymes that degrade the cells' proteins and nucleus. The end result is the characteristic cellular fragmentation of apoptosis.
• Clearance of apoptotic cells. Apoptotic cells and their fragments entice phagocytes by producing a number of “eat-me” signals. For instance, in normal cells, phosphatidylserine is present on the inner leaflet of the plasma membrane, but in apoptotic cells this phospholipid “flips” to the outer leaflet, where it is recognized by tissue macrophages, leading to phagocytosis of the apoptotic cells. Cells that are dying by apoptosis also secrete soluble factors that recruit phagocytes. The plasma membrane alterations and secreted proteins facilitate prompt clearance of the dead cells before the cells undergo membrane damage and release their contents (which can induce inflammation). Numerous macrophage receptors have been shown to be involved in the binding and engulfment of apoptotic cells. The phagocytosis of apoptotic cells is so efficient that dead cells disappear without leaving a trace, and inflammation is virtually absent.
Other Pathways of Cell Death
In addition to necrosis and apoptosis, two other patterns of cell death have been described that have unusual features. Although the importance of these pathways in disease remains to be established, they are the subjects of considerable current research, and it is useful to be aware of the basic concepts.
• Necroptosis. This form of cell death is initiated by engagement of TNF receptors as well as other, poorly defined triggers. Unlike the extrinsic pathway of apoptosis, which also is downstream of TNF receptors, in necroptosis, kinases called receptor-interacting protein (RIP) kinases are activated, initiating a series of events that result in the dissolution of the cell, much like necrosis. The name necroptosis implies that there are features of both necrosis and apoptosis. Some infections are believed to kill cells by this pathway, and it has been hypothesized to play a role in ischemic injury and other pathologic situations, especially those associated with inflammatory reactions in which the cytokine TNF is produced. However, when and why it occurs and how significant it is in human diseases is not well understood.
• Pyroptosis. This form of cell death is associated with activation of a cytosolic danger-sensing protein complex called the inflammasome (Chapter 5). The net result of inflammasome activation is the activation of caspases, some of which induce the production of cytokines that induce inflammation, often manifested by fever, and others trigger apoptosis. Thus, apoptosis and inflammation coexist. The name pyroptosis stems from the association of apoptosis with fever (Greek, pyro = fire). It is thought to be one mechanism by which some infectious microbes cause the death of infected cells. Its role in other pathologic situations is unknown.
Autophagy
Autophagy (“self-eating”) refers to lysosomal digestion of the cell's own components. It is a survival mechanism in times of nutrient deprivation, so that the starved cell can live by eating its own contents and recycling these contents to provide nutrients and energy. In this process, intracellular organelles and portions of cytosol are first sequestered within an ER-derived autophagic vacuole, whose formation is initiated by cytosolic proteins that sense nutrient deprivation (Fig. 2.14). The vacuole fuses with lysosomes to form an autophagolysosome, in which lysosomal enzymes digest the cellular components. In some circumstances, autophagy may be associated with atrophy of tissues (discussed later) and may represent an adaptation that helps cells survive lean times. If, however, the starved cell can no longer cope by devouring its contents, autophagy may eventually lead to apoptotic cell death.
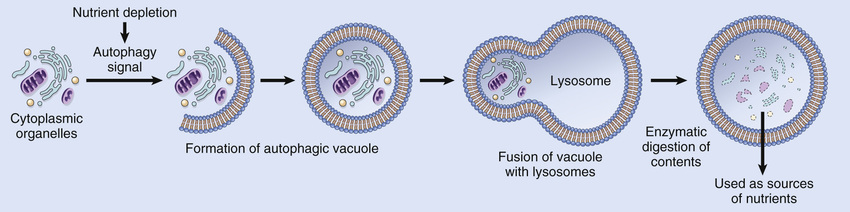
Extensive autophagy is seen in ischemic injury and some types of myopathies. Polymorphisms in a gene involved in autophagy have been associated with inflammatory bowel disease, but the mechanistic link between autophagy and intestinal inflammation is not known. The role of autophagy in cancer is discussed in Chapter 6. Thus, a once little-appreciated survival pathway in cells may prove to have wide-ranging roles in human disease.
Mechanisms of Cell Injury and Cell Death
Before discussing individual mechanisms of cell injury and death, some general principles should be emphasized.
• The cellular response to injurious stimuli depends on the type of injury, its duration, and its severity. Thus, low doses of toxins or a brief period of ischemia may lead to reversible cell injury, whereas larger toxin doses or longer ischemic times may result in irreversible injury and cell death.
• The consequences of an injurious stimulus also depend on the type, status, adaptability, and genetic makeup of the injured cell. The same injury has vastly different outcomes depending on the cell type. For instance, striated skeletal muscle in the leg tolerates complete ischemia for 2 to 3 hours without irreversible injury, whereas cardiac muscle dies after only 20 to 30 minutes of ischemia. The nutritional (or hormonal) status also can be important; understandably, a glycogen-replete hepatocyte will survive ischemia better than one that has just burned its last glucose molecule. Genetically determined diversity in metabolic pathways can contribute to differences in responses to injurious stimuli. For instance, when exposed to the same dose of a toxin, individuals who inherit variants in genes encoding cytochrome P-450 may catabolize the toxin at different rates, leading to different outcomes. Much effort is now directed toward understanding the role of genetic polymorphisms in responses to drugs and toxins, a field of study called pharmacogenomics. In fact, genetic variations influence susceptibility to many complex diseases as well as responsiveness to various therapeutic agents. Using the genetic makeup of the individual patient to guide therapy is one example of “precision medicine.”
• Cell injury usually results from functional and biochemical abnormalities in one or more of a limited number of essential cellular components (Fig. 2.15). As we discuss in more detail later, different external insults and endogenous perturbations typically affect different cellular organelles and biochemical pathways. For instance, deprivation of oxygen and nutrients (as in hypoxia and ischemia) primarily impairs energy-dependent cellular functions, culminating in necrosis, whereas damage to proteins and DNA triggers apoptosis. However, it should be emphasized that the very same injurious agent may trigger multiple and overlapping biochemical pathways. Not surprisingly, therefore, it has proved difficult to prevent cell injury by targeting an individual pathway.
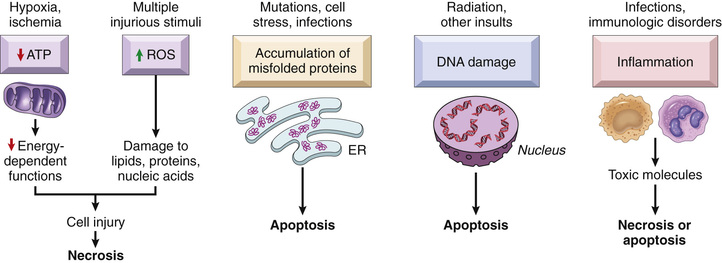
As we discussed in the beginning of this chapter and have alluded to throughout, there are numerous and diverse causes of cell injury and cell death. Similarly, there are many biochemical pathways that can initiate the sequence of events that lead to cell injury and culminate in cell death. Some of these pathways are recognized to play important roles in human diseases. In the following section, we organize our discussion of the mechanisms of cell injury along its major causes and pathways, and discuss the principal biochemical alterations in each. For the sake of clarity and simplicity, we emphasize the unique mechanisms in each pathway, but it is important to point out that any initiating trigger may activate one or more of these mechanisms, and several mechanisms may be active simultaneously.
Hypoxia and Ischemia
Deficiency of oxygen leads to failure of many energy-dependent metabolic pathways, and ultimately to death of cells by necrosis. Most cellular ATP is produced from adenosine diphosphate (ADP) by oxidative phosphorylation during reduction of oxygen in the electron transport system of mitochondria. High-energy phosphate in the form of ATP is required for membrane transport, protein synthesis, lipogenesis, and the deacylation-reacylation reactions necessary for phospholipid turnover. It is estimated that in total, the cells of a healthy human burn 50 to 75 kg of ATP every day! Not surprisingly, therefore, cells deprived of oxygen are at risk of suffering catastrophic failure of many essential functions. Oxygen deprivation is one of the most frequent causes of cell injury and necrosis in clinical medicine.
Cells subjected to the stress of hypoxia that do not immediately die activate compensatory mechanisms that are induced by transcription factors of the hypoxia-inducible factor 1 (HIF-1) family. HIF-1 simulates the synthesis of several proteins that help the cell to survive in the face of low oxygen. Some of these proteins, such as vascular endothelial growth factor (VEGF), stimulate the growth of new vessels and thus attempt to increase blood flow and the supply of oxygen. Other proteins induced by HIF-1 cause adaptive changes in cellular metabolism by stimulating the uptake of glucose and glycolysis and dampening mitochondrial oxidative phosphorylation. Anaerobic glycolysis can generate ATP in the absence of oxygen using glucose derived either from the circulation or from the hydrolysis of intracellular glycogen. Understandably, normal tissues with a greater glycolytic capacity because of the presence of glycogen (e.g., the liver and striated muscle) are more likely to survive hypoxia and decreased oxidative phosphorylation than tissues with limited glucose stores (e.g., the brain). Although it seems counterintuitive, rapidly proliferating normal cells and cancer cells rely on aerobic glycolysis to produce much of their energy, a phenomenon referred to as the Warburg effect. The reason for this is that although glycolysis yields less ATP per molecule of glucose burned than oxidative phosphorylation, metabolites generated by glycolysis and the TCA cycle serve as precursors for the synthesis of cellular constituents (e.g., proteins, lipids, and nucleic acids) that are needed for cell growth and division. Alterations in cellular metabolism are frequently seen in cancer cells, so they are discussed in more detail in Chapter 6.
Persistent or severe hypoxia and ischemia ultimately lead to failure of ATP generation and depletion of ATP in cells. Loss of this critical energy store has deleterious effects on many cellular systems (Fig. 2.16).
• Reduced activity of plasma membrane ATP-dependent sodium pumps, resulting in intracellular accumulation of sodium and efflux of potassium. The net gain of solute is accompanied by isoosmotic gain of water, causing cell swelling and dilation of the ER.
• The compensatory increase in anaerobic glycolysis leads to lactic acid accumulation, decreased intracellular pH, and decreased activity of many cellular enzymes.
• Prolonged or worsening depletion of ATP causes structural disruption of the protein synthetic apparatus, manifested as detachment of ribosomes from the rough ER (RER) and dissociation of polysomes into monosomes, with a consequent reduction in protein synthesis.
• It also has been suggested that hypoxia per se increases the accumulation of ROS. Whether this is true is a matter of debate; however, there is ample evidence that hypoxia predisposes cells to ROS-mediated damage if blood flow (and oxygen delivery) is reestablished, a phenomenon called reperfusion injury (described later).
• Ultimately, there is irreversible damage to mitochondrial and lysosomal membranes, and the cell undergoes necrosis. Membrane damage is a late event in cell injury caused by diverse mechanisms, and is discussed later. Although necrosis is the principal form of cell death caused by hypoxia, apoptosis by the mitochondrial pathway is also thought to contribute.
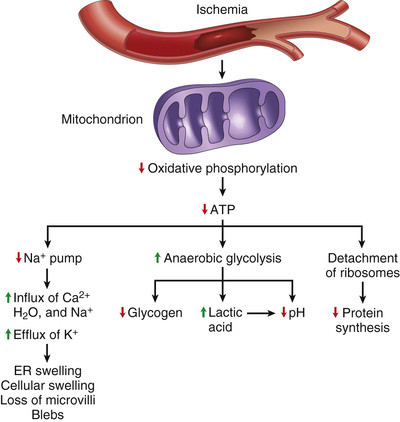
The functional consequences of hypoxia and ischemia depend on the severity and duration of the deficit. For instance, the heart muscle ceases to contract within 60 seconds of coronary occlusion. If hypoxia continues, worsening ATP depletion causes further deterioration, undergoing the sequence of changes illustrated in Fig. 2.3 and described earlier.
Ischemia-Reperfusion Injury
Under certain circumstances, the restoration of blood flow to ischemic but viable tissues results, paradoxically, in increased cell injury. This is the reverse of the expected outcome of the restoration of blood flow, which normally results in the recovery of reversibly injured cells. This so-called “ischemia-reperfusion injury” is a clinically important process that may contribute significantly to tissue damage, especially after myocardial and cerebral ischemia.
Several mechanisms may account for the exacerbation of cell injury resulting from reperfusion of ischemic tissues:
• New damage may be initiated during reoxygenation by increased generation of ROS (described in more detail below). Some of the ROS may be generated by injured cells with damaged mitochondria that cannot carry out the complete reduction of oxygen, and at the same time cellular anti-oxidant defense mechanisms may be compromised by ischemia, exacerbating the situation. ROS generated by infiltrating leukocytes also may contribute to the damage of vulnerable injured cells.
• The inflammation that is induced by ischemic injury may increase with reperfusion because it enhances the influx of leukocytes and plasma proteins. The products of activated leukocytes may cause additional tissue injury (Chapter 3). Activation of the complement system also may contribute to ischemia-reperfusion injury. Complement proteins may bind to the injured tissues, or to antibodies that are deposited in the tissues, and subsequent complement activation generates byproducts that exacerbate the cell injury and inflammation.
Oxidative Stress
Oxidative stress refers to cellular abnormalities that are induced by ROS, which belong to a group of molecules known as free radicals. Free radical-mediated cell injury is seen in many circumstances, including chemical and radiation injury, hypoxia, cellular aging, tissue injury caused by inflammatory cells, and ischemia-reperfusion injury. In all these cases, cell death may be by necrosis, apoptosis, or the mixed pattern of necroptosis.
Free radicals are chemical species with a single unpaired electron in an outer orbit. Such chemical states are extremely unstable, and free radicals readily react with inorganic and organic molecules; when generated in cells, they avidly attack nucleic acids as well as a variety of cellular proteins and lipids. In addition, free radicals initiate reactions in which molecules that react with the free radicals are themselves converted into other types of free radicals, thereby propagating the chain of damage.
Generation and Removal of Reactive Oxygen Species
The accumulation of ROS is determined by their rates of production and removal (Fig. 2.17). ROS are produced by two major pathways.
• ROS are produced normally in small amounts in all cells during the reduction-oxidation (redox) reactions that occur during mitochondrial respiration and energy generation. In this process, molecular oxygen is reduced in mitochondria to generate water by the sequential addition of four electrons. This reaction is imperfect, however, and small amounts of highly reactive but short-lived toxic intermediates are generated when oxygen is only partially reduced. These intermediates include superoxide ( ), which is converted to hydrogen peroxide (H2O2) spontaneously and by the action of the enzyme superoxide dismutase (SOD). H2O2 is more stable than
), which is converted to hydrogen peroxide (H2O2) spontaneously and by the action of the enzyme superoxide dismutase (SOD). H2O2 is more stable than  and can cross biologic membranes. In the presence of metals, such as Fe2+, H2O2 is converted to the highly reactive hydroxyl radical
and can cross biologic membranes. In the presence of metals, such as Fe2+, H2O2 is converted to the highly reactive hydroxyl radical  by the Fenton reaction. The properties and pathologic effects of the major ROS are summarized in Table 2.3.
by the Fenton reaction. The properties and pathologic effects of the major ROS are summarized in Table 2.3.
• ROS are produced in phagocytic leukocytes, mainly neutrophils and macrophages, as a weapon for destroying ingested microbes and other substances during inflammation and host defense (Chapter 3). The ROS are generated in the phagosomes and phagolysosomes of leukocytes by a process that is similar to mitochondrial respiration and is called the respiratory burst (or oxidative burst). In this process, a phagosome membrane enzyme catalyzes the generation of superoxide, which is converted to H2O2. H2O2 is in turn converted to a highly reactive compound, hypochlorite (the major component of household bleach), by the enzyme myeloperoxidase, which is present in leukocytes. The role of ROS in inflammation is described in Chapter 3.
• Nitric oxide (NO) is another reactive free radical produced in macrophages and other leukocytes. It can react with  to form a highly reactive compound, peroxynitrite, which also participates in cell injury.
to form a highly reactive compound, peroxynitrite, which also participates in cell injury.
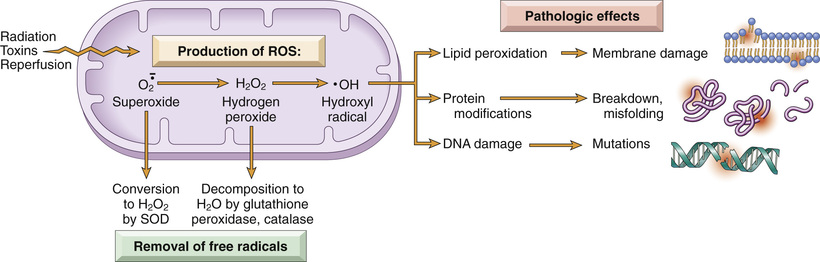
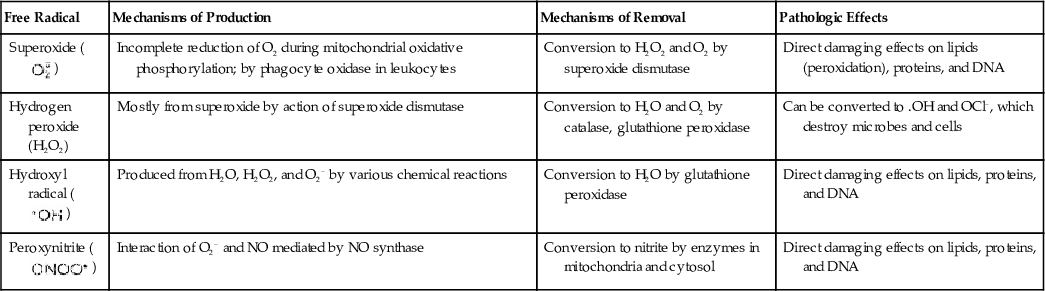
Table 2.3
Principal Free Radicals Involved in Cell Injury
| Free Radical | Mechanisms of Production | Mechanisms of Removal | Pathologic Effects |
Superoxide ( ) ) | Incomplete reduction of O2 during mitochondrial oxidative phosphorylation; by phagocyte oxidase in leukocytes | Conversion to H2O2 and O2 by superoxide dismutase | Direct damaging effects on lipids (peroxidation), proteins, and DNA |
| Hydrogen peroxide (H2O2) | Mostly from superoxide by action of superoxide dismutase | Conversion to H2O and O2 by catalase, glutathione peroxidase | Can be converted to .OH and OCl−, which destroy microbes and cells |
Hydroxyl radical ( ) ) | Produced from H2O, H2O2, and O2− by various chemical reactions | Conversion to H2O by glutathione peroxidase | Direct damaging effects on lipids, proteins, and DNA |
Peroxynitrite ( ) ) | Interaction of O2− and NO mediated by NO synthase | Conversion to nitrite by enzymes in mitochondria and cytosol | Direct damaging effects on lipids, proteins, and DNA |
The generation of free radicals is increased under several circumstances:
• The absorption of radiant energy (e.g., ultraviolet (UV) light, x-rays). Ionizing radiation can hydrolyze water into hydroxyl (•OH) and hydrogen (H•) free radicals.
• The enzymatic metabolism of exogenous chemicals (e.g., carbon tetrachloride—see later)
• Inflammation, in which free radicals are produced by leukocytes (Chapter 3)
Cells have developed mechanisms to remove free radicals and thereby minimize their injurious effects. Free radicals are inherently unstable and decay spontaneously. There also are nonenzymatic and enzymatic systems, sometimes called free radical scavengers, serving to inactivate free radicals (Fig. 2.17).
• The rate of decay of superoxide is significantly increased by the action of superoxide dismutase (SOD).
• Glutathione (GSH) peroxidases are a family of enzymes whose major function is to protect cells from oxidative damage. The most abundant member of this family, GSH peroxidase 1, is found in the cytoplasm of all cells. It catalyzes the breakdown of H2O2 by the reaction 2GSH + H2O2 → GS-SG + 2H2O. The intracellular ratio of oxidized GSH to reduced GSH is a reflection of this enzyme's activity and thus of the cell's ability to catabolize free radicals.
• Catalase, present in peroxisomes, catalyzes the decomposition of hydrogen peroxide (2H2O2 → O2 + 2H2O). It is one of the most active enzymes known, capable of degrading millions of molecules of H2O2 per second.
• Endogenous or exogenous anti-oxidants (e.g., vitamins E, A, and C and β-carotene) may either block the formation of free radicals or scavenge them after they have formed.
Cell Injury Caused by Reactive Oxygen Species
ROS causes cell injury by damaging multiple components of cells (Fig. 2.17):
• Lipid peroxidation of membranes. Double bonds in membrane polyunsaturated lipids are vulnerable to attack by oxygen-derived free radicals. The lipid–radical interactions yield peroxides, which are themselves unstable and reactive, and an autocatalytic chain reaction ensues. Damage to plasma membranes as well as mitochondrial and lysosomal membranes can have devastating consequences, as discussed earlier in the context of ischemia and hypoxia.
• Crosslinking and other changes in proteins. Free radicals promote sulfhydryl-mediated protein crosslinking, resulting in enhanced degradation or loss of enzymatic activity. Free radical reactions also may directly cause polypeptide fragmentation. Damaged proteins may fail to fold properly, triggering the unfolded protein response, described later.
• DNA damage. Free radical reactions with thymine residues in nuclear and mitochondrial DNA produce single-strand breaks. Such DNA damage has been implicated in apoptotic cell death, aging, and malignant transformation of cells.
• In addition to the role of ROS in cell injury and the killing of microbes, low concentrations of ROS are involved in numerous signaling pathways in cells and thus in many physiologic reactions. Therefore, these molecules are produced normally but, to avoid their harmful effects, their intracellular concentrations are tightly regulated in healthy cells.
Cell Injury Caused by Toxins
Toxins, including environmental chemicals and substances produced by infectious pathogens, induce cell injury that culminates primarily in necrotic cell death. Different types of toxins induce cell injury by two general mechanism:
• Direct-acting toxins. Some toxins act directly by combining with a critical molecular component or cellular organelle. For example, in mercuric chloride poisoning (as may occur from ingestion of contaminated seafood) (Chapter 8), mercury binds to the sulfhydryl groups of various cell membrane proteins, causing inhibition of ATP-dependent transport and increased membrane permeability. Many anti-neoplastic chemotherapeutic agents also induce cell damage by direct cytotoxic effects. Also included in this class are toxins made by microorganisms (described in Chapter 9). These often cause damage by targeting host cell molecules that are needed for essential functions, such as protein synthesis and ion transport.
• Latent toxins. Many toxic chemicals are not intrinsically active but must first be converted to reactive metabolites, which then act on target cells. Understandably, such toxins typically affect the cells in which they are activated. This is usually accomplished by cytochrome P-450 in the smooth ER of the liver and other organs. Although the metabolites might cause membrane damage and cell injury by direct covalent binding to protein and lipids, the most important mechanism of cell injury involves the formation of free radicals. Carbon tetrachloride (CCl4)—once widely used in the dry cleaning industry but now banned—and the analgesic acetaminophen belong in this category. The effect of CCl4 is still instructive as an example of chemical injury. CCl4 is converted to a toxic free radical, principally in the liver, and this free radical is the cause of cell injury, mainly by membrane phospholipid peroxidation. In less than 30 minutes after exposure to CCl4, there is sufficient damage to the ER membranes of hepatocytes to cause a decline in the synthesis of enzymes and plasma proteins; within 2 hours, swelling of the smooth ER and dissociation of ribosomes from the RER have occurred. There also is decreased synthesis of apoproteins that form complexes with triglycerides and thereby facilitate triglyceride secretion; this defect results in the accumulation of lipids in hepatocytes and other cells and the “fatty liver” of CCl4 poisoning. Mitochondrial injury follows, and subsequently diminished ATP stores result in defective ion transport and progressive cell swelling; the plasma membranes are further damaged by fatty aldehydes produced by lipid peroxidation in the ER. The end result can be cell death.
Endoplasmic Reticulum Stress
The accumulation of misfolded proteins in a cell can stress compensatory pathways in the ER and lead to cell death by apoptosis. During normal protein synthesis, chaperones in the ER control the proper folding of newly synthesized proteins, and misfolded polypeptides are ubiquitinated and targeted for proteolysis. If unfolded or misfolded proteins accumulate in the ER, they first induce a protective cellular response that is called the unfolded protein response (Fig. 2.18). This adaptive response activates signaling pathways that increase the production of chaperones and decrease protein translation, thus reducing the levels of misfolded proteins in the cell. When a large amount of misfolded protein accumulates and cannot be handled by the adaptive response, the signals that are generated result in activation of proapoptotic sensors of the BH3-only family as well as direct activation of caspases, leading to apoptosis by the mitochondrial (intrinsic) pathway.
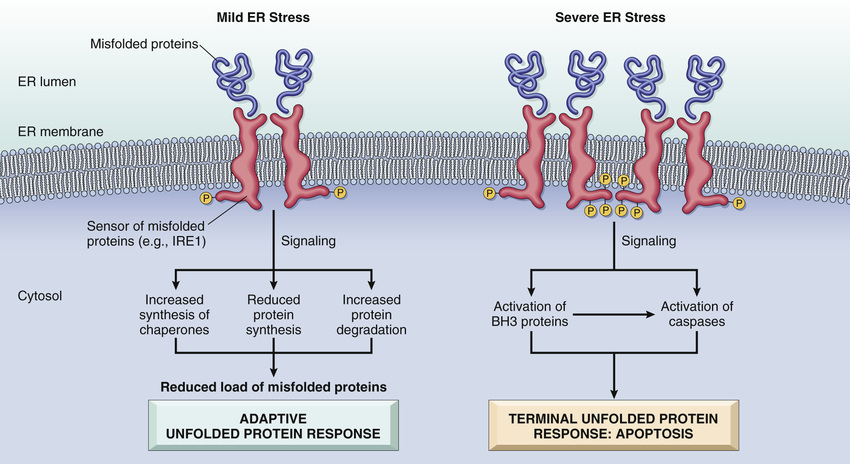
Intracellular accumulation of misfolded proteins may be caused by abnormalities that increase the production of misfolded proteins or reduce the ability to eliminate them. This may result from gene mutations that lead to the production of proteins that cannot fold properly; aging, which is associated with a decreased capacity to correct misfolding; infections, especially viral infections, when large amounts of microbial proteins are synthesized within cells, more than the cell can handle; increased demand for secretory proteins such as insulin in insulin-resistant states; and changes in intracellular pH and redox state. Protein misfolding is thought to be the fundamental cellular abnormality in several neurodegenerative diseases (Chapter 23). Deprivation of glucose and oxygen, as in ischemia and hypoxia, also may increase the burden of misfolded proteins.
Protein misfolding within cells may cause disease by creating a deficiency of an essential protein or by inducing apoptosis (Table 2.4).
• Misfolded proteins often lose their activity and are rapidly degraded, both of which can contribute to a loss of function. If this function is essential, cellular injury ensues. One important disease in which this occurs is cystic fibrosis, which is caused by inherited mutations in a membrane transport protein that prevent its normal folding.
• Cell death as a result of protein misfolding is recognized as a feature of a number of diseases, including the neurodegenerative disorders Alzheimer disease, Huntington disease, and Parkinson disease, and may underlie type 2 diabetes as well (Table 2.4).
Table 2.4
Diseases Caused by Misfolded Proteinsa
| Disease | Affected Protein | Pathogenesis |
| Diseases Caused by Mutant Proteins That Are Degraded, Leading to Their Deficiency | ||
| Cystic fibrosis | CFTR | Loss of CFTR leads to defects in chloride transport and death of affected cells |
| Familial hypercholesterolemia | LDL receptor | Loss of LDL receptor leads to hypercholesterolemia |
| Tay-Sachs disease | Hexosaminidase β subunit | Lack of the lysosomal enzyme leads to storage of GM2 gangliosides in neurons |
| Diseases Caused by Misfolded Proteins That Result in ER Stress-Induced Cell Loss | ||
| Retinitis pigmentosa | Rhodopsin | Abnormal folding of rhodopsin causes photoreceptor loss and cell death, resulting in blindness |
| Creutzfeldt-Jacob disease | Prions | Abnormal folding of PrPsc causes neuronal cell death |
| Alzheimer disease | Aβ peptide | Abnormal folding of Aβ peptide causes aggregation within neurons and apoptosis |
| Diseases Caused by Misfolded Proteins That Result From Both ER Stress-Induced Cell Loss and Functional Deficiency of the Protein | ||
| Alpha-1-anti-trypsin deficiency | α-1 anti-trypsin | Storage of nonfunctional protein in hepatocytes causes apoptosis; absence of enzymatic activity in lungs causes destruction of elastic tissue giving rise to emphysema |

As discussed later in the chapter, improperly folded proteins can also accumulate in extracellular tissues, as in amyloidosis.
DNA Damage
Exposure of cells to radiation or chemotherapeutic agents, intracellular generation of ROS, and acquisition of mutations may all induce DNA damage, which if severe may trigger apoptotic death. Damage to DNA is sensed by intracellular sentinel proteins, which transmit signals that lead to the accumulation of p53 protein. p53 first arrests the cell cycle (at the G1 phase) to allow the DNA to be repaired before it is replicated (Chapter 6). However, if the damage is too great to be repaired successfully, p53 triggers apoptosis, mainly by stimulating BH3-only sensor proteins that ultimately activate Bax and Bak, proapoptotic members of the Bcl-2 family. When p53 is mutated or absent (as it is in certain cancers), cells with damaged DNA that would otherwise undergo apoptosis survive. In such cells, the DNA damage may result in mutations or DNA rearrangements (e.g., translocations) that lead to neoplastic transformation (Chapter 6).
Inflammation
A common cause of injury to cells and tissues is the inflammatory reaction that is elicited by pathogens, necrotic cells, and dysregulated immune responses, as in autoimmune diseases and allergies. In all these situations, inflammatory cells, including neutrophils, macrophages, lymphocytes, and other leukocytes, secrete products that evolved to destroy microbes but also may damage host tissues. These injurious immune reactions are classified under hypersensitivity. Their mechanisms and significance are discussed in Chapter 5.
Common Events in Cell Injury From Diverse Causes
In the previous discussion, we addressed the mechanisms of cell injury according to the initiating cause, and highlighted the principal pathways of injury that are triggered in different pathophysiologic situations. Some abnormalities characterize cell injury regardless of the cause, and are thus seen in a variety of pathologic situations. Two of these changes are described next.
Mitochondrial Dysfunction
Mitochondria may be viewed as “mini-factories” that produce life-sustaining energy in the form of ATP. Not surprisingly, therefore, they also are critical players in cell injury and death. Mitochondria are sensitive to many types of injurious stimuli, including hypoxia, chemical toxins, and radiation (Fig. 2.19). Mitochondrial changes occur in necrosis and apoptosis. They may result in several biochemical abnormalities:
• Failure of oxidative phosphorylation leads to progressive depletion of ATP, culminating in necrosis of the cell, as described earlier.
• Abnormal oxidative phosphorylation also leads to the formation of ROS, which have many deleterious effects, as already described.
• Damage to mitochondria is often associated with the formation of a high-conductance channel in the mitochondrial membrane, called the mitochondrial permeability transition pore. The opening of this channel leads to the loss of mitochondrial membrane potential and pH changes, further compromising oxidative phosphorylation.
• Mitochondria also contain proteins such as cytochrome c that, when released into the cytoplasm, tell the cell there is internal injury and activate a pathway of apoptosis, as discussed earlier.
Defects in Membrane Permeability
Increased membrane permeability leading ultimately to overt membrane damage is a feature of most forms of cell injury that culminate in necrosis. The most important sites of membrane damage during cell injury are the mitochondrial membrane, the plasma membrane, and membranes of lysosomes. As noted earlier, increased permeability of the plasma membrane and lysosomal membranes is not a feature of apoptosis.
• Mitochondrial membrane damage. As discussed earlier, damage to mitochondrial membranes results in decreased production of ATP, with many deleterious effects culminating in necrosis.
• Plasma membrane damage. Plasma membrane damage leads to loss of osmotic balance and influx of fluids and ions, as well as loss of cellular contents. The cells may also leak metabolites that are vital for the reconstitution of ATP, thus further depleting energy stores.
• Injury to lysosomal membranes results in leakage of their enzymes into the cytoplasm and activation of the acid hydrolases in the acidic intracellular pH of the injured (e.g., ischemic) cell. Activation of these enzymes leads to enzymatic digestion of cell components, and the cells die by necrosis.
We have now concluded the discussion of cell injury and cell death. As we have seen, these processes are the root cause of many common diseases. We end this chapter with brief considerations of three other processes: cellular adaptations to stresses; intracellular accumulations of various substances and extracellular deposition of calcium, both of which are often associated with cell injury; and aging.
Cellular Adaptations to Stress
Adaptations are reversible changes in the number, size, phenotype, metabolic activity, or functions of cells in response to changes in their environment. Physiologic adaptations usually represent responses of cells to normal stimulation by hormones or endogenous chemical mediators (e.g., the hormone-induced enlargement of the breast and uterus during pregnancy), or to the demands of mechanical stress (in the case of bones and muscles). Pathologic adaptations are responses to stress that allow cells to modulate their structure and function and thus escape injury, but at the expense of normal function, such as squamous metaplasia of bronchial epithelium in smokers. Physiologic and pathologic adaptations can take several distinct forms, as described in the following text.
Hypertrophy
Hypertrophy is an increase in the size of cells resulting in an increase in the size of the organ. In contrast, hyperplasia (discussed next) is an increase in cell number. Stated another way, in pure hypertrophy there are no new cells, just bigger cells containing increased amounts of structural proteins and organelles. Hyperplasia is an adaptive response in cells capable of replication, whereas hypertrophy occurs when cells have a limited capacity to divide. Hypertrophy and hyperplasia also can occur together, and obviously both result in an enlarged organ.
Hypertrophy can be physiologic or pathologic and is caused either by increased functional demand or by growth factor or hormonal stimulation.
• The massive physiologic enlargement of the uterus during pregnancy occurs as a consequence of estrogen-stimulated smooth muscle hypertrophy and smooth muscle hyperplasia (Fig. 2.20). In contrast, in response to increased workload the striated muscle cells in both the skeletal muscle and the heart undergo only hypertrophy because adult muscle cells have a limited capacity to divide. Therefore, the chiseled physique of the avid weightlifter stems solely from the hypertrophy of individual skeletal muscles.
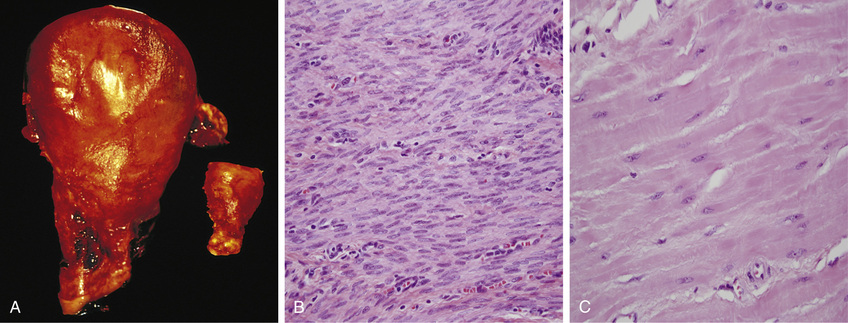
• An example of pathologic hypertrophy is the cardiac enlargement that occurs with hypertension or aortic valve disease (Fig. 2.21). The differences between normal, adapted, and irreversibly injured cells are illustrated by the responses of the heart to different types of stress. Myocardium subjected to a persistently increased workload, as in hypertension or with a narrowed (stenotic) valve, adapts by undergoing hypertrophy to generate the required higher contractile force. If, on the other hand, the myocardium is subjected to reduced blood flow (ischemia) due to an occluded coronary artery, the muscle cells may undergo injury.
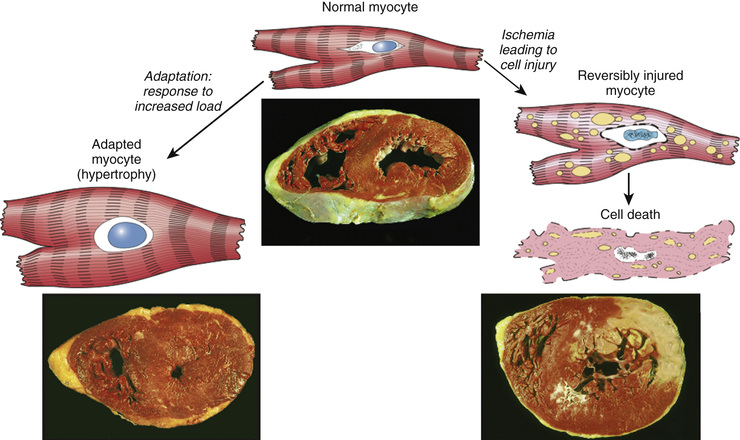
The mechanisms driving cardiac hypertrophy involve at least two types of signals: mechanical triggers, such as stretch, and soluble mediators that stimulate cell growth, such as growth factors and adrenergic hormones. These stimuli turn on signal transduction pathways that lead to the induction of a number of genes, which in turn stimulate synthesis of many cellular proteins, including growth factors and structural proteins. The result is the synthesis of more proteins and myofilaments per cell, which increases the force generated with each contraction, enabling the cell to meet increased work demands. There may also be a switch of contractile proteins from adult to fetal or neonatal forms. For example, during muscle hypertrophy, the α-myosin heavy chain is replaced by the fetal β form of the myosin heavy chain, which produces slower, more energetically economical contraction.
An adaptation to stress such as hypertrophy can progress to functionally significant cell injury if the stress is not relieved. Whatever the cause of hypertrophy, a limit is reached beyond which the enlargement of muscle mass can no longer compensate for the increased burden. When this happens in the heart, several degenerative changes occur in the myocardial fibers, of which the most important are fragmentation and loss of myofibrillar contractile elements. Why hypertrophy progresses to these regressive changes is incompletely understood. There may be finite limits on the abilities of the vasculature to adequately supply the enlarged fibers, the mitochondria to supply ATP, or the biosynthetic machinery to provide sufficient contractile proteins or other cytoskeletal elements. The net result of these degenerative changes is ventricular dilation and ultimately cardiac failure.
Hyperplasia
Hyperplasia is an increase in the number of cells in an organ that stems from increased proliferation, either of differentiated cells or, in some instances, less differentiated progenitor cells. As discussed earlier, hyperplasia takes place if the tissue contains cell populations capable of replication; it may occur concurrently with hypertrophy and often in response to the same stimuli.
Hyperplasia can be physiologic or pathologic; in both situations, cellular proliferation is stimulated by growth factors that are produced by a variety of cell types.
• The two types of physiologic hyperplasia are (1) hormonal hyperplasia, exemplified by the proliferation of the glandular epithelium of the female breast at puberty and during pregnancy, and (2) compensatory hyperplasia, in which residual tissue grows after removal or loss of part of an organ. For example, when part of a liver is resected, mitotic activity in the remaining cells begins as early as 12 hours later, eventually restoring the liver to its normal size. The stimuli for hyperplasia in this setting are polypeptide growth factors produced by uninjured hepatocytes as well as nonparenchymal cells in the liver (Chapter 3). After restoration of the liver mass, various growth inhibitors turn off cell proliferation.
• Most forms of pathologic hyperplasia are caused by excessive hormonal or growth factor stimulation. For example, after a normal menstrual period there is a burst of uterine epithelial proliferation that is normally tightly regulated by the stimulatory effects of pituitary hormones and ovarian estrogen and the inhibitory effects of progesterone. A disturbance in this balance leading to increased estrogenic stimulation causes endometrial hyperplasia, which is a common cause of abnormal menstrual bleeding. Benign prostatic hyperplasia is another common example of pathologic hyperplasia induced in responses to hormonal stimulation by androgens. Stimulation by growth factors also is involved in the hyperplasia that is associated with certain viral infections; for example, papillomaviruses cause skin warts and mucosal lesions that are composed of masses of hyperplastic epithelium. Here the growth factors may be encoded by viral genes or by the genes of the infected host cells.
An important point is that in all of these situations, the hyperplastic process remains controlled; if the signals that initiate it abate, the hyperplasia disappears. It is this responsiveness to normal regulatory control mechanisms that distinguishes pathologic hyperplasias from cancer, in which the growth control mechanisms become permanently dysregulated or ineffective (Chapter 6). Nevertheless, in many cases, pathologic hyperplasia constitutes a fertile soil in which cancers may eventually arise. For example, patients with hyperplasia of the endometrium are at increased risk of developing endometrial cancer (Chapter 19).
Atrophy
Atrophy is shrinkage in the size of cells by the loss of cell substance. When a sufficient number of cells are involved, the entire tissue or organ is reduced in size, or atrophic (Fig. 2.22). Although atrophic cells may have diminished function, they are not dead.
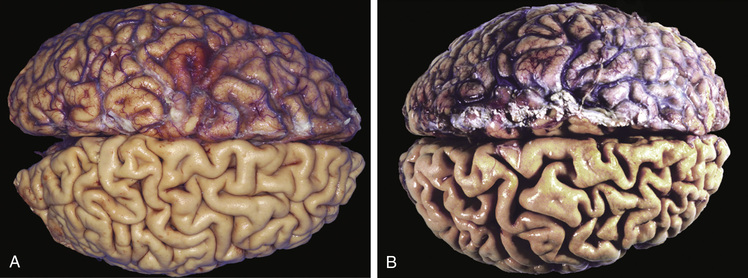
Causes of atrophy include a decreased workload (e.g., immobilization of a limb to permit healing of a fracture), loss of innervation, diminished blood supply, inadequate nutrition, loss of endocrine stimulation, and aging (senile atrophy). Although some of these stimuli are physiologic (e.g., the loss of hormone stimulation in menopause) and others are pathologic (e.g., denervation), the fundamental cellular changes are similar. They represent a retreat by the cell to a smaller size at which survival is still possible; a new equilibrium is achieved between cell size and diminished blood supply, nutrition, or trophic stimulation.
Cellular atrophy results from a combination of decreased protein synthesis and increased protein degradation.
• Protein synthesis decreases because of reduced metabolic activity.
• The degradation of cellular proteins occurs mainly by the ubiquitin-proteasome pathway. Nutrient deficiency and disuse may activate ubiquitin ligases, which attach multiple copies of the small peptide ubiquitin to cellular proteins and target them for degradation in proteasomes. This pathway is also thought to be responsible for the accelerated proteolysis seen in a variety of catabolic conditions, including the cachexia associated with cancer.
• In many situations, atrophy also is associated with autophagy, with resulting increases in the number of autophagic vacuoles. As discussed previously, autophagy is the process in which the starved cell eats its own organelles in an attempt to survive.
Metaplasia
Metaplasia is a change in which one adult cell type (epithelial or mesenchymal) is replaced by another adult cell type. In this type of cellular adaptation, a cell type sensitive to a particular stress is replaced by another cell type better able to withstand the adverse environment. Metaplasia is thought to arise by the reprogramming of stem cells to differentiate along a new pathway rather than a phenotypic change (transdifferentiation) of already differentiated cells.
Epithelial metaplasia is exemplified by the change that occurs in the respiratory epithelium of habitual cigarette smokers, in whom the normal ciliated columnar epithelial cells of the trachea and bronchi often are replaced by stratified squamous epithelial cells (Fig. 2.23). The rugged stratified squamous epithelium may be able to survive the noxious chemicals in cigarette smoke that the more fragile specialized epithelium would not tolerate. Although the metaplastic squamous epithelium has survival advantages, important protective mechanisms are lost, such as mucus secretion and ciliary clearance of particulate matter. Epithelial metaplasia is therefore a double-edged sword. Because vitamin A is essential for normal epithelial differentiation, its deficiency also may induce squamous metaplasia in the respiratory epithelium.
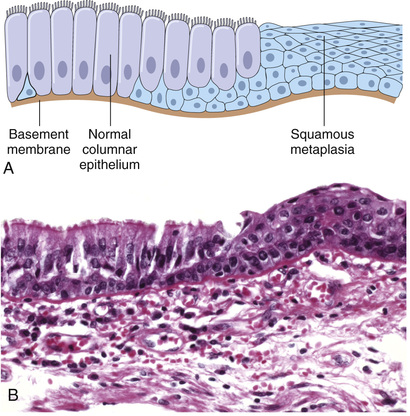
Metaplasia need not always occur in the direction of columnar to squamous epithelium; in chronic gastric reflux, the normal stratified squamous epithelium of the lower esophagus may undergo metaplastic transformation to gastric or intestinal-type columnar epithelium. Metaplasia also may occur in mesenchymal cells, but in these situations it is generally a reaction to some pathologic alteration and not an adaptive response to stress. For example, bone is occasionally formed in soft tissues, particularly in foci of injury.
The influences that induce metaplastic change in an epithelium, if persistent, may predispose to malignant transformation. In fact, squamous metaplasia of the respiratory epithelium often coexists with lung cancers composed of malignant squamous cells. It is thought that cigarette smoking initially causes squamous metaplasia, and cancers arise later in some of these altered foci.
Intracellular Accumulations
Under some circumstances, cells may accumulate abnormal amounts of various substances, which may be harmless or may cause varying degrees of injury. The substance may be located in the cytoplasm, within organelles (typically lysosomes), or in the nucleus, and it may be synthesized by the affected cells or it may be produced elsewhere.
The main pathways of abnormal intracellular accumulations are inadequate removal and degradation or excessive production of an endogenous substance, or deposition of an abnormal exogenous material (Fig. 2.24). Selected examples of each are described as follows.
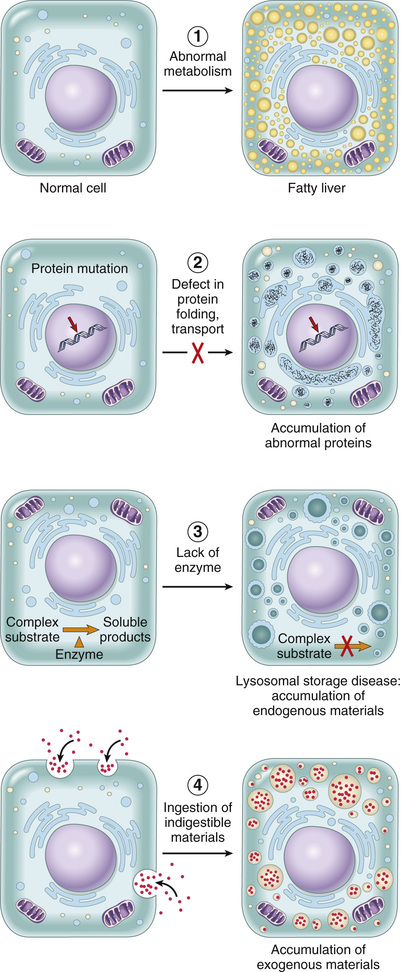
Fatty Change. Fatty change, also called steatosis, refers to any accumulation of triglycerides within parenchymal cells. It is most often seen in the liver, since this is the major organ involved in fat metabolism, but also may occur in heart, skeletal muscle, kidney, and other organs. Steatosis may be caused by toxins, protein malnutrition, diabetes mellitus, obesity, or anoxia. Alcohol abuse and diabetes associated with obesity are the most common causes of fatty change in the liver (fatty liver) in industrialized nations. This process is discussed in more detail in Chapter 16.
Cholesterol and Cholesteryl Esters. Cellular cholesterol metabolism is tightly regulated to ensure normal generation of cell membranes (in which cholesterol is a key component) without significant intracellular accumulation. However, phagocytic cells may become overloaded with lipid (triglycerides, cholesterol, and cholesteryl esters) in several different pathologic processes, mostly characterized by increased intake or decreased catabolism of lipids. Of these, atherosclerosis is the most important. The role of lipid and cholesterol deposition in the pathogenesis of atherosclerosis is discussed in Chapter 10.
Proteins. Morphologically visible protein accumulations are less common than lipid accumulations; they may occur when excesses are presented to the cells or if the cells synthesize excessive amounts. In the kidney, for example, trace amounts of albumin filtered through the glomerulus are normally reabsorbed by pinocytosis in the proximal convoluted tubules. However, in disorders with heavy protein leakage across the glomerular filter (e.g., nephrotic syndrome), much more of the protein is reabsorbed, and vesicles containing this protein accumulate, giving the histologic appearance of pink, hyaline cytoplasmic droplets. The process is reversible: if the proteinuria abates, the protein droplets are metabolized and disappear. Another example is the marked accumulation of newly synthesized immunoglobulins that may occur in the RER of some plasma cells, forming rounded, eosinophilic Russell bodies. Other examples of protein aggregation are discussed elsewhere in this book (e.g., “alcoholic hyaline” in the liver in Chapter 16; neurofibrillary tangles in neurons in Chapter 23).
Glycogen. Excessive intracellular deposits of glycogen are associated with abnormalities in the metabolism of either glucose or glycogen. In poorly controlled diabetes mellitus, the prime example of abnormal glucose metabolism, glycogen accumulates in renal tubular epithelium, cardiac myocytes, and β cells of the islets of Langerhans. Glycogen also accumulates within cells in a group of related genetic disorders collectively referred to as glycogen storage diseases, or glycogenoses (Chapter 7).
Pigments. Pigments are colored substances that are either exogenous, coming from outside the body, such as carbon, or are endogenous, synthesized within the body itself, such as lipofuscin, melanin, and certain derivatives of hemoglobin.
The most common exogenous pigment is carbon, a ubiquitous air pollutant of urban life. When inhaled, it is phagocytosed by alveolar macrophages and transported through lymphatic channels to the regional tracheobronchial lymph nodes. Aggregates of the pigment blacken the draining lymph nodes and pulmonary parenchyma (anthracosis) (Chapter 13).
• Lipofuscin, or “wear-and-tear pigment,” is an insoluble brownish-yellow granular intracellular material that accumulates in a variety of tissues (particularly the heart, liver, and brain) with aging or atrophy. Lipofuscin represents complexes of lipid and protein that are produced by the free radical–catalyzed peroxidation of polyunsaturated lipids of subcellular membranes. It is not injurious to the cell but is a marker of past free radical injury. The brown pigment (Fig. 2.25), when present in large amounts, imparts an appearance to the tissue that is called brown atrophy.
• Melanin is an endogenous, brown-black pigment that is synthesized by melanocytes located in the epidermis and acts as a screen against harmful UV radiation. Although melanocytes are the only source of melanin, adjacent basal keratinocytes in the skin can accumulate the pigment (e.g., in freckles), as can dermal macrophages.
• Hemosiderin is a hemoglobin-derived granular pigment that is golden yellow to brown and accumulates in tissues when there is a local or systemic excess of iron. Iron is normally stored within cells in association with the protein apoferritin, forming ferritin micelles. Hemosiderin pigment represents large aggregates of these ferritin micelles, readily visualized by light and electron microscopy; the iron can be unambiguously identified by the Prussian blue histochemical reaction (Fig. 2.26). Although hemosiderin accumulation is usually pathologic, small amounts of this pigment are normal in the mononuclear phagocytes of the bone marrow, spleen, and liver, where aging red cells are normally degraded. Excessive deposition of hemosiderin, called hemosiderosis, and more extensive accumulations of iron seen in hereditary hemochromatosis are described in Chapter 16.
Pathologic Calcification
Pathologic calcification, a common process in a wide variety of disease states, is the result of an abnormal deposition of calcium salts, together with smaller amounts of iron, magnesium, and other minerals. It can occur in two ways.
• Dystrophic calcification. In this form, calcium metabolism is normal but it deposits in injured or dead tissue, such as areas of necrosis of any type. It is virtually ubiquitous in the arterial lesions of advanced atherosclerosis (Chapter 10). Although dystrophic calcification may be an incidental finding indicating insignificant past cell injury, it also may be a cause of organ dysfunction. For example, calcification can develop in aging or damaged heart valves, resulting in severely compromised valve motion. Dystrophic calcification of the aortic valves is an important cause of aortic stenosis in elderly persons (Chapter 11).
Dystrophic calcification is initiated by the extracellular deposition of crystalline calcium phosphate in membrane-bound vesicles, which may be derived from injured cells, or the intracellular deposition of calcium in the mitochondria of dying cells. It is thought that the extracellular calcium is concentrated in vesicles by its affinity for membrane phospholipids, whereas phosphates accumulate as a result of the action of membrane-bound phosphatases. The crystals are then propagated, forming larger deposits.
• Metastatic calcification. This form is associated with hypercalcemia and can occur in normal tissues. The major causes of hypercalcemia are (1) increased secretion of parathyroid hormone, due to either primary parathyroid tumors or production of parathyroid hormone–related protein by other malignant tumors; (2) destruction of bone due to the effects of accelerated turnover (e.g., Paget disease), immobilization, or tumors (increased bone catabolism associated with multiple myeloma, leukemia, or diffuse skeletal metastases); (3) vitamin D–related disorders including vitamin D intoxication and sarcoidosis (in which macrophages activate a vitamin D precursor); and (4) renal failure, in which phosphate retention leads to secondary hyperparathyroidism.
Cellular Aging
Individuals age because their cells age. Although public attention on the aging process has traditionally focused on its cosmetic manifestations, aging has important health consequences, because age is one of the strongest independent risk factors for many chronic diseases, such as cancer, Alzheimer disease, and ischemic heart disease. Perhaps one of the most striking discoveries about cellular aging is that it is not simply a consequence of cells' “running out of steam,” but in fact is regulated by a limited number of genes and signaling pathways that are evolutionarily conserved from yeast to mammals.
Cellular aging is the result of a progressive decline in the life span and functional activity of cells. Several abnormalities contribute to the aging of cells (Fig. 2.27):
• Accumulation of mutations in DNA. A variety of metabolic insults over time may result in damage to nuclear and mitochondrial DNA. ROS induced by toxins and radiation exposure contribute to DNA damage associated with aging. Although most DNA damage is repaired by DNA repair enzymes, some persists and accumulates as cells age, especially if repair mechanisms become inefficient over time. Accumulation of mutations in nuclear and mitochondrial DNA ultimately compromise the functional activities and survival of cells.
• Decreased cellular replication. Normal cells (other than stem cells) have a limited capacity for replication, and after a fixed number of divisions, they become arrested in a terminally nondividing state, known as replicative senescence. Aging is associated with progressive replicative senescence of cells. Cells from children have the capacity to undergo more rounds of replication than do cells from older people. In contrast, cells from patients with Werner syndrome, a rare disease characterized by premature aging, have a markedly reduced in vitro life span.
Replicative senescence occurs in aging cells because of progressive shortening of telomeres, which ultimately results in cell cycle arrest. Telomeres are short repeated sequences of DNA present at the ends of chromosomes that are important for ensuring the complete replication of chromosome ends and for protecting the ends from fusion and degradation. Telomeric DNA also binds proteins that shield it, preventing activation of a DNA damage response. When somatic cells replicate, a small section of the telomere is not duplicated, and telomeres become progressively shortened. As they shorten, the ends of chromosomes cannot be protected and are sensed in cells as broken DNA, which signals cell cycle arrest. Telomere length is maintained by nucleotide addition mediated by an enzyme called telomerase. Telomerase is a specialized RNA-protein complex that uses its own RNA as a template for adding nucleotides to the ends of chromosomes. Telomerase is expressed in germ cells and is present at low levels in stem cells, but absent in most somatic cells (Fig. 2.28). Therefore, as mature somatic cells age, their telomeres become shorter and they exit the cell cycle, resulting in an inability to generate new cells to replace damaged ones. Conversely, in immortalized cancer cells, telomerase is usually reactivated and telomere length is stabilized, allowing the cells to proliferate indefinitely. This is discussed more fully in Chapter 6. Telomere shortening also may decrease the regenerative capacity of stem cells, further contributing to cellular aging. Despite such alluring observations, however, the relationship of telomerase activity and telomere length to aging has yet to be fully established. Abnormalities in telomere maintenance have been implicated in many diseases, such as aplastic anemia and other cytopenias (thought to be caused by a failure of hematopoietic stem cells), premature greying of hair, skin pigment and nail abnormalities, pulmonary and liver fibrosis, and others. These disorders are sometimes considered the prototypic “telomeropathies.”
• Defective protein homeostasis. With the passage of time, cells are unable to maintain normal protein homeostasis, because of increased turnover and decreased synthesis caused by reduced translation of proteins and defective activity of chaperones (which promote normal protein folding) and proteasomes (which destroy misfolded proteins). The resultant decrease in intracellular proteins can have many deleterious effects on cell survival, replication, and functions. The concomitant accumulation of misfolded proteins exacerbates the loss of functional proteins and can trigger apoptosis.
• There has been great interest in defining biochemical signaling pathways that counteract the aging process, not only because of their obvious therapeutic potential (the search for the “elixir of youth”) but also because elucidating these pathways might tell us about the mechanisms that cause aging. Calorie restriction has been found to slow down aging and prolong life in every species tested from flies to mice. It is now thought that calorie restriction alters signaling pathways that influence aging (Fig. 2.27). Among the biochemical alterations associated with calorie restriction that have been described as playing a role in counteracting the aging process is reduced activation of insulin-like growth factor receptor signaling, which involves a downstream network of kinases and transcription factors. Reduced IGF-1 signaling leads to lower rates of cell growth and metabolism and possibly reduced errors in DNA replication, better DNA repair and improve protein homeostasis. Calorie restriction also serves to improve immunity. All of these inhibit aging.
• Persistent inflammation. As individuals age, the accumulation of damaged cells, lipids, and other endogenous substances may activate the inflammasome pathway (Chapter 5), resulting in persistent low-level inflammation. Inflammation in turn induces chronic diseases, such as atherosclerosis and type 2 diabetes. Cytokines produced during inflammatory reactions may themselves induce cellular alterations that exacerbate aging, and chronic metabolic disorders may accelerate the aging process.
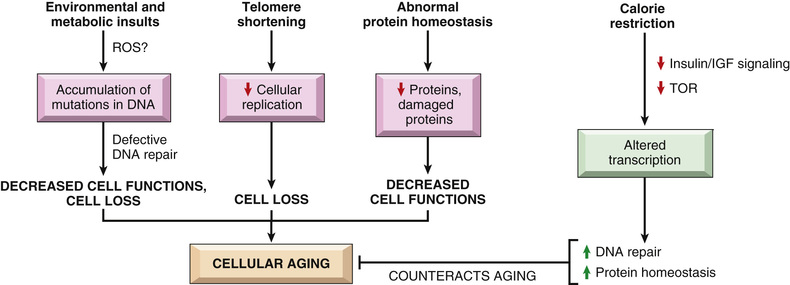
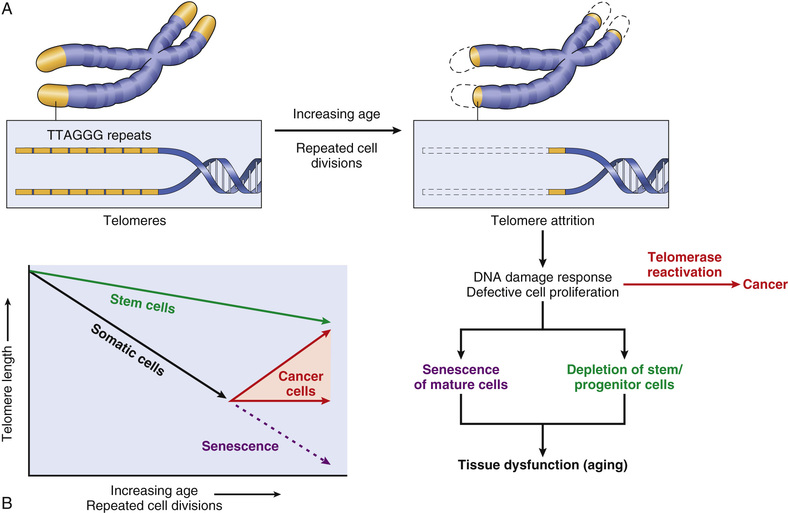
Clinical observations and epidemiologic studies have shown that physical activity and, as mentioned previously, calorie restriction slow the aging process, whereas stresses, perhaps acting via increased production of glucocorticoids, accelerate aging. Alas, the precise mechanisms underlying these effects remain to be defined, and for now we all remain vulnerable to the ravages of age.
It should be apparent that the various forms of cellular derangements and adaptations described in this chapter cover a wide spectrum, ranging from reversible and irreversible forms of acute cell injury, to adaptations in cell size, growth, and function, to largely unavoidable consequences of aging. Reference is made to these many different alterations throughout this book, because all instances of organ injury and ultimately all cases of clinical disease arise from derangements in cell structure and function.








