Surgery for Back and Neck Pain (Including Radiculopathies)
Introduction
Pain is the most common reason for surgery on the back or neck, but pain alone is an insufficient reason to perform surgery. To understand the appropriate use of surgical procedures for back and neck pain it is necessary to understand that surgery corrects only one of two conditions. Surgical procedures can be designed to relieve compression on nerves or the spinal cord, and they can stabilize dynamically unstable motion segments (Bohannon and Gajdosik 1987, Cholewicki and McGill 1996). The key in the surgical decision-making process is not only to make a diagnosis but also to determine whether the patient has anatomical abnormalities amenable to surgical correction.
Only a small minority of patients in virtually all the disease categories that will be discussed will ever require surgery (Bell and Rothman 1984, BenDebba et al 2002). Surgery should be considered in two situations:
The majority of acute back and neck pain syndromes relent spontaneously without treatment in a reasonable period (Basmajian 1989, Bigos et al 1994). Since watchful waiting with adequate pain control is all that is required for the majority of patients, it should not be surprising that the principal classification used for both back and neck pain is temporally based rather than being focused on causes, pathologies, or treatments (BenDebba et al 2000, Long et al 1996).
New basic research, however, strongly suggests that this time-honored approach to surgery may be insufficient. Laboratory data indicate that the inflammatory processes that occur with disc herniation or injury may excite nociceptors locally and even cause significant neuronal changes in the dorsal root ganglia and substantia gelatinosa. These findings may offer an explanation for why local anti-inflammatory drugs are frequently helpful to patients with herniated lumbar and cervical discs. The implications for surgery are as yet unknown.
Acute Pain
Acute back or neck pain is of two types. The first is termed transient and generally relents within a few hours or a few days. Such patients rarely seek medical intervention. The more typical acute back or neck syndrome lasts days to weeks and may even stretch into a few months. The symptoms may be very severe and neurological deficits may be present. In the absence of unbearable pain or a significant neurological deficit, watchful waiting is all that is required. Patients are treated with adequate analgesics, placed at rest for a few days until the symptoms begin to abate, and then returned to reasonable function as quickly as possible. Virtually all agree that these guidelines will lead to spontaneous recovery for most patients. In the absence of red flags, imaging is not required, and for the majority of patients with acute back and neck syndromes, no cause is ever determined.
Persistent Pain
We have recently identified a new category that we have termed persistent pain syndrome (BenDebba et al 1997, Long et al 1996). These patients may have either back or neck pain and constitute a group who simply do not recover from the original acute pain syndrome. Even though they do not recover, they exhibit none of the characteristics of the so-called chronic pain syndrome. They remain functional, although function may be limited. Psychological abnormalities do not develop. They are refractory to the usual conservative treatments and the symptoms persist. The symptoms remained more or less constant over a period of 2–5 years in patients reported from our prospective large study.
Chronic Pain
The so-called chronic pain syndrome is characterized by chronicity of the pain complaints and is often complicated by depression, demoralization, misuse of analgesic medications, and a focus on pain and disability that is out of character with the physical impairments. These patients are quite different from those we have termed persistent, although they are indistinguishable in terms of physical causes (Long 1997a, 2002). The majority of patients undergoing surgery comes from the persistent pain category.
Chronic back pain, often with associated leg pain, is the most common medical complaint in developed countries (Bigos et al 1994). The cost associated with back pain is enormous (Kelsey 1975, 1982; Frymoyer and Cats-Baril 1991). Back pain is one of the most frequent reasons why patients see physicians and one of the most common reasons for secondary referral, and both operative and non-operative treatment of back pain ranks high in terms of total expenditure of health care dollars (Frymoyer and Cats-Baril 1991, Davis 1994, Anderson 1996).
Causes and Treatment
Despite the obvious importance of back and neck pain as a complaint, the causes are poorly understood and few treatments have been validated (Bigos et al 1994). A major issue is that the actual pathophysiological causes of the complaints are mostly unknown (Boden et al 1990, Kuslich et al 1991, Jonsson and Stromqvist 1993). It has been estimated that only about 10% of patients with acute and chronic pain complaints have definable causes that can be clearly related to the complaint of pain. It is well validated that degenerative and spondylotic changes, no matter how severe, do not correlate well with pain complaints.
There is reasonable evidence that overt instability causes pain and that elimination of the instability will reduce the pain (Cholewicki and McGill 1996). The strongest evidence is that root compression is associated with pain and neurological deficits (Weber 1994, BenDebba et al 2002). Decompression is satisfactory treatment for the majority of patients (Bohannon and Gajdosik 1987). However, these two conditions are relatively rare in the spectrum of patients with back or leg pain complaints, and for the majority the association of complaints with demonstrated structural abnormalities is tenuous at best (Weber 1994).
It is not surprising that therapies are problematic in a condition without known causes of the pain (Bell and Rothman 1984, Basmajian 1989, Bigos et al 1994). Until there is a better scientific basis for understanding spinal pain and its treatment, we must use what is known to decide on the best current therapy for these patients (Saal and Saal 1989; Saal et al 1990; Koes et al 1992a, 1992b).
To reach supportable decisions concerning the evaluation and treatment of these patients, the physician involved in their care must have an organized classification framework in which to work (Box 71-1). Knowing what the various diagnostic tests available can be expected to demonstrate is important. Rigorous evaluation of claims of therapeutic efficacy for all modalities of treatment is required. It is also important to have an equally rigorous understanding of outcome measurement to assess these claims (Waddell 1987b, BenDebba et al 2000).
The goal of this chapter is to provide a framework for the evaluation, diagnosis, choice of treatment, and assessment of treatment outcome for patients complaining of chronic low back pain with or without sciatica or neck pain and with or without a radicular component with a focus on selection criteria for surgery and outcomes of common surgical procedures.
Diagnosis of Spinal Pain
History
The evaluation should begin with a careful history that describes the severity and location of the pain and its triggers (Hakelius and Hindmarsh 1972a, 1972b; Deyo et al 1992). Physical examination is unlikely to be diagnostic but will assess the patient’s neurological and musculoskeletal abnormalities. Routine neurological examination is needed as a baseline at least (Hurme et al 1983).
During these examinations listen for danger signals such as night pain (intraspinal tumor), constant pain (cancer or infection), systemic symptoms (cancer or infection), and symptoms of other organ or systemic disease (Long et al 1996).
Also, observe the patient’s behavior during the examination. Is there much pain behavior? Are the patient’s actions consistent with the complaints? Are the results of motor examination reliable (Waddell et al 1980, Hurme and Alaranta 1987, McCombe et al 1989)?
Unlike acute pain problems, imaging is important in patients with persistent pain (Ackerman et al 1997a). Plain flexion–extension films are important. Magnetic resonance imaging (MRI) is best for most screening (Ackerman et al 1997b). Computed tomography (CT) can be used if bony pathology is suspected; with two- and three-dimensional reconstructions, fixator artifacts can be reduced (Annertz et al 1990, Hashimoto et al 1990). CT myelography is needed rarely, most commonly in patients with previous surgery (Hakelius and Hindmarsh 1972a, Wiesel et al 1984, Boden et al 1990, Deyo et al 1994).
There is no need for psychological testing unless symptoms suggestive of psychiatric disturbance are present (Waddell et al 1980, Long 1990).
In a recent study we demonstrated that expert spinal surgeons almost always made an accurate diagnosis on the basis of the history alone. The key elements are the temporal and special characteristics of the pain, as well as its severity. Other associated neurological complaints may be present, but they are not required and are not usually very serious impairments (BenDebba et al 2002, Long et al 1996).
Physical Examination
Physical examination is not nearly as important in patients with spinal pain as in many other areas of neurological practice (Deyo et al 1992, Long et al 1996). When typical, it is useful to localize an abnormality, but most patients do not have focal physical findings. The classic combinations of reflex, motor, and sensory changes described in texts are relatively rare. In a study of nearly 3000 patients, we demonstrated such classic combinations in less than 1% (BenDebba et al 2002, Long et al 1996).
Range of motion of the part of the spine at which the pain is located, the presence or absence of muscle spasm, local areas of tenderness and other local non-specific signs of abnormality, strength, sensation, and reflexes should all be assessed in the standard way. Straight-leg raising is the only physical sign of great value in patients with lumbar disc disease. It is positive in the majority of patients, and the crossed straight-leg raising test is strongly indicative of a root compression syndrome. There is no similar test for cervical disc herniation. The important function of the physical examination is to determine the presence or absence of neurological loss and the severity of these deficits (Jonsson and Stromqvist 1993).
Cervical myelopathy is of particular concern. Cervical spinal cord compression is a serious issue that usually requires surgery. When cervical pain is the complaint, careful examination for myelopathy is important. Spinal cord compression should be signaled by increased reflexes; presence of the Hoffman and Babinski signs; bilateral loss of motor power, particularly grip strength and walking stability; and patchy sensory loss without a typical sensory level.
Imaging Diagnosis
Evaluation begins with plain films of the affected area of the spine, including dynamic films. If simple disc herniation or spinal stenosis is suspected, MRI should be diagnostic. It would be the only test needed for the large majority of patients. If significant bony abnormalities are suspected, however, CT is useful. When neither a diagnostic image nor the clinical syndrome is typical, CT myelography can still be a useful study (Ackerman et al 1997b).
Electrodiagnostic Studies
Such studies are helpful in several instances, but they are rarely indicated in patients with typical disc herniation. Electromyography is most useful in differentiating peripheral neuropathies and entrapment neuropathies from obscure root compression syndromes. Neurophysiological studies can also be used to verify cauda or spinal cord compression.
Provocative Blocks
In indeterminate cases, diagnostic provocative blocks are often important (Bogduk 1994, Schwarzer et al 1994, Bogduk et al 1995). The hypothesis is the same for all: elimination of the pain component by anesthetization of a particular structure suggests that the structure is involved as a pain generator. A further part of the study is usually provocation of the patient’s individual pain syndrome by needling or by injection. The combination of the two is considered the most concordant. Structures commonly blocked include nerve roots, zygapophyseal joints, and discs. A positive result means that the patient’s pain was mimicked by the procedure and relieved during the short period of anesthetization. Placebo blocks are required to achieve high specificity and selectivity.
These blocks should never be depended on as an indication for surgery alone. Rather, they are part of the overall patient assessment and may add some information implicating a particular vertebral segment as a pain generator (Schwarzer et al 1994, 1995a). To achieve validity, the location of the needles must be verified.
Provocative discography has now been demonstrated to have predictive value for spinal arthroplasty and fusion.
Causes of Spinal Pain
Another way to categorize surgical patients with chronic back and neck pain is to list the causes that may lead to surgery. Most experts agree that the preponderance of these patients have spondylotic disease, which is at least associated with the pain problem if not yet proved to be causative (Spangfort 1972, Weber 1983, Waddell 1987b) (Box 71-2).
The categories of spinal pain are similar for the lumbar and cervical segments, and there is no practical need to differentiate them. Generic descriptions will suffice for both despite the obvious anatomical differences. Any such discussion must begin with the statement that at present we do not know the cause of the pain in the majority of patients who seek medical care with back or neck complaints. There are many hypotheses, mostly presented by therapists to justify their particular form of therapy. None has a solid scientific basis and therefore cannot be considered more than theoretical.
Most patients in these categories will recover spontaneously from acute episodes, and there is no indication for more than symptomatic relief initially (Bigos et al 1994, Long et al 1996).
Disease States for which Correlation with Pain is Strong
Infection in and around the spine virtually always causes pain. Discitis or osteomyelitis is notoriously painful, undoubtedly from a combination of inflammation, instability, and nerve root compression. Such patients sometimes require surgery, but the fundamental treatment is eradication of the infection (Long et al 1996, Woertgen et al 2006).
Tumors
Tumors involving the spinal canal may be painful. They often encroach on the periosteum surrounding muscles and ligaments and compress or invade peripheral nerves. Tumors are also associated with inflammation in addition to compression and destruction. Surgical procedures are often required to stabilize the spine and decompress the nervous tissue (Long 1997b).
Arthritic Conditions
There is a group of defined arthritic conditions, such as rheumatoid arthritis and ankylosing spondylitis, that are commonly associated with pain and instability and may threaten neurological function. Surgical treatment is usually directed at stabilization of the spine to prevent or reverse a significant neurological deficit.
Herniated Intervertebral Disc
One of the most common reasons for performing spinal surgery is to excise a herniated intervertebral disc, which often occurs with significant degenerative disc disease and facet arthropathy (Spangfort 1972, Weber 1994). The disc herniation causes unilateral or multiroot compression and, if in the cervical region, can compress the spinal cord and produce myelopathy. The majority causes a unitary radiculopathy that is manifested as pain in the distribution of that nerve root and an appropriate associated neurological deficit. Pain without neurological loss is common.
Disc herniation may occur at any level but is most common at L4–5 and L5–S1 in the lumbar region and at C5–6 and C6–7 in the cervical region. Free-fragment discs are pieces of herniated discs that have penetrated the posterior longitudinal ligament and are lying free in the spinal canal or foramen, usually trapping a root against a bony surface. The herniated disc that is not a free fragment is contained by the posterior longitudinal ligament. Even though it is completely herniated through the annulus, it is still restricted by the intact posterior ligament. However, most of this so-called disc herniation is associated with other evidence of spondylosis, including bone spurs, thickened ligaments, and compromised foramina. Surgical treatment may require a more complicated operation than simple disc removal (Long and McAfee 1992, Zeidman and Long 1998).
New laboratory data strongly suggest that products of the inflammatory cascade arising from herniated or injured discs may be irritative and provoke pain, but the clinical implications of these findings are uncertain. It does appear that anti-inflammatory therapy maybe helpful. There is no evidence, however, that this has any impact on the decisions for or against surgery as yet.
Patients with a truly herniated disc usually complain of an acute onset of severe pain that radiates in the distribution of a sciatic or cervical nerve. The root involved determines the location of the pain, and the diagnosis can nearly always be made from the patient’s description of the pain alone. There may be neurological findings related to the specific nerve root. These clinical syndromes are well described in standard spine texts. However, in our studies of these problems, classic physical findings were rarely present. Although these classic findings make excellent textbook descriptions, they do not occur frequently in clinical practice (Long et al 1996). The typical patient has radicular pain alone as the indication for surgery, and thus it is the severity of the pain that determines treatment, not the presence of a neurological deficit. Of course, if a significant neurological deficit is present, this argues for earlier surgery. The diagnosis of disc herniation is made reliably by MRI or CT myelography. CT will be much less sensitive alone (Spengler and Freeman 1979).
Stenosis
The most common reason for surgery on the lumbar or cervical spine is stenosis (Spivak 1998, Hilibrand and Rand 1999). Many patients have congenitally short pedicles and thus a canal of restricted size. Then, with age, degeneration of discs, and hypertrophy of ligaments, the canal is compressed even more and multiple roots and/or the spinal cord are compromised. Degenerative changes alone can cause the stenosis. Lumbar stenosis is typified by symptoms suggestive of vascular claudication. These patients cannot walk very far without their legs feeling weak, tired, and often painful. Because multiple roots are involved, multiple neurological changes occur. Complaints of bowel, bladder, and sexual dysfunction are common. The pain in most patients is worsened by activity, particularly standing or walking, and is improved by rest, especially in the flexed position. The diagnosis is made by MRI, CT, or CT myelography. When the symptoms progress to a significant level, the outcome of surgical treatment is excellent. There are no known alternative treatments.
Cervical spinal stenosis differs only in the fact that the spinal cord is compressed rather than the cauda equina (King et al 2003). Thus, signs of myelopathy with bilateral hyperreflexia, pathological signs indicating spinal cord compression, and variable degrees of sensory loss, including local radicular loss and sensory levels of spinal cord origin, may be present. These patients often describe subtle abnormalities. They may have a disturbance in gait that is difficult to demonstrate on examination and frequently have equally subtle impairment of posterior column function that makes it difficult for them to walk in the dark or when they cannot see the surface on which they are walking.
Degenerated Discs
Loss of hydration of discs occurs throughout life and is present in virtually everyone as they age. Why some of these discs are painful and the majority is not is unknown. Nevertheless, there is reasonable evidence that some patients have painful degenerated discs (Bogduk 1991). This inference is drawn from a study inaptly called discography (Bogduk 1994). Discography was originally introduced to identify degenerated discs radiographically by the injection of radiopaque material directly into the nucleus of the degenerated disc. The original supposition was that excision and replacement of these degenerated discs would provide pain relief.
Discography was reintroduced as a provocative test to identify painful segments of the spine. The goal of the test is twofold:
These studies demonstrate that some degenerated discs clearly correlate with patients’ pain descriptions when distended whereas others do not. From these studies it has again been assumed that excision of these painful discs and stabilization of that segment will relieve the pain. No studies establishing statistical correlation between the results of discography and surgery have yet been forthcoming, but the inferences are positive, and identification of painful degenerated discs with subsequent surgical treatment is common practice (Knox and Chapman 1993). Several studies have now recently correlated the positive pain response from discography with good outcome after either fusion or arthroplasty.
Instability
Instability occurs in a number of situations, including infection, tumors, and trauma. Pain may be a part of any of these syndromes, and surgery may certainly play an important role in their management, but they are fundamentally outside the scope of this chapter. The majority of patients with significant spinal instability suffers either from degenerative disc disease with all its associated spondylotic changes or from congenital abnormalities such as spondylolysis. The goal in all these situations is to eliminate the instability, but the issue is what constitutes clinically significant instability. This definition is uncertain and a number of authors have provided a spectrum of conditions that they believe must be met before instability can be diagnosed. All are arbitrary and none has been verified in a prospective manner in any clinical series (Grob et al 1995, France et al 1999).
The diagnosis of instability is generally made with imaging studies. Such studies include plain films with dynamic views, axial CT for accurate visualization of bony structures, and MRI to assess soft tissues. The instability may be dynamic, with actual movement being seen on the dynamic films. The degree of movement required to be abnormal is much debated, and the measurements commonly given in texts are arbitrary. It is uncertain how much movement is required to cause pain. Instability may also be glacial, with a change in spinal position over time being demonstrated. In addition, malalignment of the spine is demonstrated on these films and may be rotational, scoliotic, or axial. When progressive, these changes in alignment can seriously compromise function and may be painful, even when static.
Lesser degrees of instability are even more difficult to quantify. Severe facet arthropathy may be unstable and is often demonstrated on imaging studies (Bogduk 1991, Schwarzer et al 1995b).
Treatment of all these conditions is virtually always surgical stabilization if the symptoms warrant.
Back and Neck Pain as a Manifestation of Unexpected or Intercurrent Disease
In our own series, unexpected systemic disease was found in 3% of patients. Osseous metastases were the most common finding. Retroperitoneal inflammatory processes, sometimes associated with chronic gastrointestinal disease, pancreatitis, and chronic and acute renal disease, may all rarely cause back pain. The pain from any of these processes is usually local. Radiation of pain to the leg may be secondary to involvement of the lumbosacral plexus but is rarely typically sciatic. Such pain is constant, tends not to be exacerbated by activity or relieved by rest, and is often associated with other signs or symptoms of systemic disease. The leg pain is non-radicular, as are the physical findings associated with infiltration of the plexus. Signs and symptoms that suggest intercurrent disease are intractable pain, unremitting pain, neurological deficit suggestive of involvement of the lumbosacral plexus, a history of cancer or inflammatory disease, a history of any disease likely to be complicated by infection, and a history of significant trauma (Long et al 1996, BenDebba et al 1997).
Spondylitis
The most common inflammatory condition associated with chronic back pain is rheumatoid arthritis. Back pain is not usually an early characteristic of the disease, so the diagnosis is known when back pain occurs. If systemic manifestations are not prominent, it can be difficult to be certain about the diagnosis on clinical grounds. Plain films, CT, and MRI all suggest inflammatory spondylotic disease. The diagnosis is confirmed by serological testing. Treatment consists of three phases:
The principal rheumatoid spinal problem requiring surgery is cervical instability causing myelopathy, but spinal stenosis in the lumbar region does occur. The syndrome is typical neurogenic claudication. Surgery requires decompression and frequently fusion (Fig. 71-1).
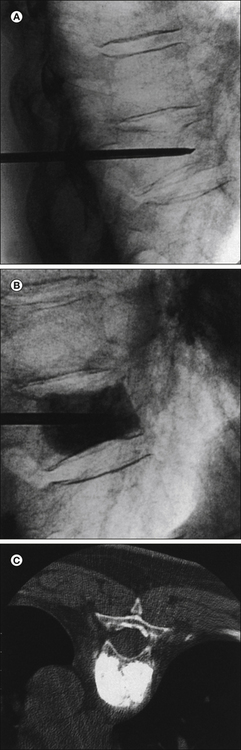
Figure 71-1 Vertebroplasty.
A, Thoracic compression fracture with a transpedicular needle in the vertebral body. B, Injection of methacrylate into the vertebral body as seen under fluoroscopy. C, The finished product on axial computed tomography after bilateral transpedicular injection. The vertebral body is now filled with methacrylate and strength is restored. Pain relief was immediate.
The second common problem is ankylosing spondylitis (Lawrence 1977), which is predominantly a disease in males. Symptoms usually begin with back pain early in life, and a progressive history of constant back pain is typical. Radicular or cord compression symptoms are uncommon. The diagnosis is made by observation of the typical “bamboo spine” appearance on plain radiography or MRI. Treatment is typically symptomatic. As spontaneous fusion progresses up the spine, the pain relents in the lumbar region, only to reappear at higher levels progressively. Fusion of painful segments is sometimes required.
Psoriatic arthritis is a rare, similar disease. The problem is more facet arthropathy with synovial proliferation (Liu et al 1990). Thus, in psoriatic arthritis, root compression syndromes are common and spinal stenosis is typical. Back pain alone is best treated symptomatically. Surgical decompression and occasional stabilization are required for root compression syndromes.
Acromegalic spondylitis is common in untreated patients. Fortunately, with the quality of therapy now available for these pituitary tumors, this problem has become very rare. The syndrome is similar to ankylosing spondylitis, with progressive pain usually beginning in the lumbar region and ceasing with spontaneous fusion. Treatment is symptomatic.
Spondylotic Low Back Pain (Idiopathic)
The majority of patients with complaints of low back pain (with or without leg pain) has associated spondylotic spinal disease (Fig. 71-2, Box 71-3). These spondylotic changes may be defined as primary loss of hydration of the lumbar discs with a subsequent change in signal characteristics and associated ligament thickening, chronic inflammatory changes in ligaments and adjacent end plates, loss of disc height, facet arthropathy and hypertrophy (with or without associated canal and foraminal stenosis), or evidence of instability. The presence of spondylosis and the complaints are at least correlated, but the correlation is far from certain (Boden et al 1990, Weber 1994). The diagnostic problem is that a substantial number of patients have spondylotic changes without any apparent related symptoms (Wiesel et al 1984, Boden et al 1990).
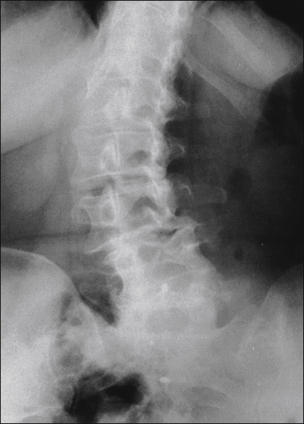
Figure 71-2 Anteroposterior plain radiograph of the lumbar spine demonstrating dramatic kyphoscoliosis involving lumbar vertebrae 1–4 and the lower thoracic spine.
These structural deformities are often associated with severe pain and respond well to surgical correction.
Careful population studies with detailed histories outlining the past and present history of these patients have not been done. Nevertheless, virtually every physician has had the experience of seeing a patient with profound spondylotic changes who has no complaints currently and has never had complaints. These observations make correlation of back pain with spondylotic changes difficult, and more evidence than the simple occurrence of spondylosis is needed to prove causation. Even so, in a sizable majority of patients with chronic back pain complaints, spondylotic changes are present and are the only abnormalities that seem to explain the pain.
The history of the problem is the most important diagnostic tool. Patients with pain of apparent spondylotic origin characteristically have more pain when standing or load-bearing. The symptoms are improved by rest in most patients. Severe back pain is often associated with non-radicular leg pain, usually diffuse and aching in the anterior or posterior aspect of the thighs. Associated true radicular pain is common, as is neurogenic claudication. Nearly all patients have pain worsened by activity and improved by rest. The pain is generally least in the morning and worsens with activity, in contradistinction to osteoarthritis and the other spondylotic problems.
Tarlov described sacral perineural cysts in the 1930s. He subsequently published a book that demonstrated the pathology of the condition and described successful surgical treatment of a few pain-provoking cysts. McKusick and colleagues described progressive dual ectasia involving the sacrum and pelvis in patients with connective tissue disorders (see, for instance, McKusick 1975). These patients consistently have pain and many have progressive neurological deficits, which is unusual in patients with Tarlov cysts. Internal meningoceles have also been described and are frequently associated with pain. All three entities have largely been ignored. Even though no epidemiological studies are available, it does appear from what has been published that Tarlov cysts of varying size occur in 1.5–2% of the population. Only a small number of these cysts are symptomatic. However, a number of reports over the past 60 years have indicated that large cysts and ectasia can produce severe pain and progressive neurological deficits. Surgical repair is a rarity, but a number of successful cases have been reported. This is a field that deserves much more attention.
Back and Neck Pain as a Manifestation of Psychiatric Disease: Implications for Surgical Therapy
There is a strong tendency among physicians and many others involved in the treatment of chronic pain to assume that all pain for which no diagnosable cause can be found is psychosomatic. Nowhere is this truer than with chronic back pain. National Low Back Pain Study data indicate that the incidence of psychopathology is no greater in patients with back pain than would be expected in any ill population (BenDebba et al 1997). Three percent of patients in this study had a psychiatric diagnosis that was thought by the examiners to be the primary cause of the complaint. However, in a population of more than 2000 patients admitted to a pain treatment program for manifestations of chronic pain syndrome, the incidence of psychopathology was much higher (Long 1990). Between 15% and 20% of these patients had a primary psychiatric diagnosis that was thought to be the origin of the pain complaint or at least an important mediator. Endogenous depression was the most common diagnosis. Somatoform disorder was the next most common diagnosis, followed by schizophrenia.
There is an important difference of opinion among pain specialists about what is the most common diagnosis in some experiences of personality disorder. Some experts find an increased incidence of a diagnosable personality disorder in patients with chronic pain syndrome. Others note an increased frequency of symptoms suggestive of a personality disorder without a truly diagnosable syndrome by criteria of the Diagnostic and Statistical Manual of Mental Disorders. Frequently, there is imprecise separation of the psychosocial symptoms following an acute pain event and those that were present before the noxious event. In addition, there is the added factor that the presence of a psychiatric diagnosis or these personality traits does not eliminate the possibility of a bona fide painful problem involving the spine.
This chapter is not the place for an exhaustive review of these psychological factors. In this context it is important only to emphasize that psychiatric disease may have pain as an important symptom. The presence of psychiatric disease does not eliminate the possibility that the patient has a diagnosable separate cause. The presence of traits suggestive of a personality disorder cannot be construed as being causative of the complaint of back pain. However, the increased incidence of co-morbid conditions in patients with these traits is important to consider when contemplating surgical therapy (Waddell et al 1980).
Selction of Patients for Surgery
Selection of patients for surgery is based on the severity of symptoms and demonstration of a correctable anatomical problem (Hurme and Alaranta 1987, Long et al 1996). Pain is the most common reason for which surgery is chosen to treat spinal problems. Therefore, the surgeon must be certain that the pain and resulting functional impairments are serious enough to warrant the magnitude of the procedure. When the patient’s history appears to be accurate, the pain complaints are consistent with the abnormalities observed, and the functional impairments are consistent with the complaint of pain, surgery based on that complaint is reasonable. Red flags include exaggerated complaints of pain, complaints that are inconsistent with the known anatomy and physiology, impairments that are inconsistent with the complaints and/or anatomical findings, and other behavioral abnormalities that suggest a psychiatric or psychosocial component of the pain syndrome (Waddell 1987a).
The presence of psychological and behavioral issues does not necessarily mean that the patient should not be treated. They do imply that these patients may need a comprehensive pain program to address all the elements of their pain syndrome as surgical treatment is being contemplated. If the psychological and other issues are left unaddressed, they are likely to complicate and retard the recovery process. This is particularly true of patients who are misusing analgesic medication (Long 1990).
The next most common reason for surgery is the presence of a significant or progressive neurological deficit. Deficits may include one or more nerve roots and/or the spinal cord. If the deficit is of sufficient magnitude (i.e., if it would be seriously impairing if permanent), surgery is typically indicated.
In general, selection of patients for surgery hinges on two factors: either the patient’s complaints are intolerable and urgent surgery is required, or enough time has elapsed and treatments used to know that the patient will not recover spontaneously (Bush et al 1992). To determine when these situations exist, it is necessary to know the natural history of the problem being treated, as well as the clinical status of the patient (Spengler and Freeman 1979, Weber 1994).
Natural History of Acute Cervical and Lumbar Spinal Pain
We have collected extensive data from a multicenter prospective examination of patients referred to spinal orthopedists or neurosurgeons for acute spinal problems and consideration for surgery that indicate that the majority of such patients improve spontaneously and never need surgery. In this study only 14% of patients referred to spine surgeons ever underwent surgery. There are equally good data from a thorough examination of patients with acute low back pain to say that spontaneous recovery is almost certain and that only a few patients with acute low back pain and/or acute sciatica, even with significant disc herniation, will ever need surgical intervention (Long et al 1996; BenDebba et al 1997, 2002).
Our data are less detailed for patients with cervical problems, but an examination of patients with acute neck pain with and without a radicular component seen in the spine services at Johns Hopkins indicates that the outcome of patients with neck pain is essentially the same as it is for those with back pain. The great majority recovers even when overt disc herniation with some evidence of radiculopathy is present. The data from our patients suggest that whenever possible, the surgeon should wait at least 1 month before considering surgery. Only intractable pain and a significant neurological deficit should change that recommendation. If improvement has not begun in 1 month, it will probably not ensue. However, if improvement has started, it is feasible to wait at least 3 months before making a surgical decision. Virtually all patients will recover in 3 months.
Some studies have indicated that patients continue to improve over a 5-year period, but this is not consistent with the findings of our study of low back pain. In our patients, the majority was stable at 2 and 5 years. A small group did continue to improve after 3–6 months, but that group was not large enough to have statistical significance. Thus, it appears that the majority of patients who suffer from acute back or neck pain with or without radicular pain and irrespective of cause will recover spontaneously without more than symptomatic treatment. Most will be fully recovered in 3 months. If recovery has not occurred in 6 months, it can be expected to be stable over the next 2–5 years, neither improving nor worsening. The proportion who did not improve in our study was just 14%, so the majority would be expected to improve. Severe pain, significant neurological deficit, or discovery of another disease process can all mean earlier intervention (Long et al 1996).
Alternatives to Surgery for Low Back Pain
Surgical procedures are of value only for the relief of demonstrated nerve root compression, correction of a fixed deformity, or stabilization of an unstable segment (Spangfort 1972, Weber 1983, Hurme and Alaranta 1987). Therapy otherwise is symptomatic (Wiesel et al 1980; Bell and Rothman 1984; Postacchini et al 1988; Koes et al 1992a, 1992b ). For patients whose symptoms have been present for 6 months or longer, there is strong new evidence that spontaneous improvement will not occur.
Our studies have also examined many forms of standard physical therapy, manipulation therapy, and a wide variety of other treatments, including acupuncture, back schools, nutritional therapies, pain treatment centers, and cognitive therapies. We were unable to determine any effect of any of these treatments as currently applied in practice (BenDebba et al 2002). Therefore, it will not suffice to simply refer patients for any of these treatments. A program based on the best available data should be individualized for each patient (Waddell 1987b).
Adequate analgesia is a first choice. Non-steroidal anti-inflammatory analgesics are standard (Basmajian 1989). Some investigators are examining the use of long-acting narcotics for the relief of pain of benign origin. Some patients will benefit from short-term bracing, especially when active, so a trial of lumbosacral support is worthwhile. There is reasonable evidence that an individualized exercise program to strengthen the paravertebral and abdominal muscles, combined with local measures to restore painless range of motion, will benefit many patients. These programs are not typically available from the usual physical medicine sources, but if such a program can be obtained, many patients will benefit. Elderly patients are particularly helped by a general physical-conditioning program as well, but any reconditioned patient can be helped. Weight loss has not been proved to be beneficial, but reduction in weight makes intuitive sense and is included in most vigorous rehabilitation efforts. Serious osteoporosis should also be treated (Koes et al 1992b).
The dramatic increase in the number of physicians dedicated to pain management in the United States has led to an equally dramatic increase in the number of minimally invasive injections carried out for back pain. The most frequent of these include injections of various steroids around zygapophyseal joints, transforaminal epidural injections, intralaminar and caudal epidural injections, and similar infiltration around the sacroiliac joints. Because of the proliferation of these procedures, there has been much interest in guidelines for their use and their outcomes. Although much controversy remains, it does appear that there is convincing evidence for the use of steroid injections for the treatment of acute and chronic back and leg pain secondary to disc injury, herniation, and compression. Not all agree, however, and a number of reports indicate that steroid techniques are not useful. Most experts believe that they have a role in patients with acute radicular syndromes in providing more rapid resolution of the most severe symptoms. There are still no studies that have demonstrated other clinical improvements in the natural history (Berman et al 1984, Bowman et al 1993).
A controversial treatment called prolotherapy is less well supported. Injections of hypertonic solutions are postulated to cause proliferative healing of injured ligaments. One study suggests a non-specific beneficial effect (Yelland et al 2003).
Passive physical therapy measures (e.g., heat, massage, ultrasound, transcutaneous electrical stimulation) are in common use. All are known to relieve muscle spasm, areas of myositis, and tenderness and reduce inflammation. Patients often feel much better in the short term, but there is no evidence that these measures significantly affect long-term recovery (BenDebba et al 2002).
Many reports support the immediate efficacy of manipulation. However, controlled studies do not indicate a significant effect of manipulation therapy on the long-term outcome. In our study, no effect could be demonstrated that was different from that expected with no treatment (Shekelle et al 1992, Meade et al 1995, Triano et al 1995).
Acupuncture is widely used for the treatment of pain, but the published studies do not support the use of acupuncture for back or neck pain. Meta-analysis indicates no significant effects (BenDebba et al 2002).
Surgical Procedures: Lumbar and Cervical
The Need for Urgent Surgery and Bilateral Leg Pain
A major area of concern is when is urgent surgery needed. Severe or progressive neurological loss or instability is the usual indication. The most serious are severe myelopathy or cauda equina syndrome. Common causes are hematoma, acute disc herniation, infection, and subluxation. Cauda equina syndrome rarely occurs with acute disc herniation. To make the diagnosis of cauda equina syndrome, the symptoms and signs should be bilateral.
Leg symptoms alone do not constitute cauda equina syndrome. To meet the definition, the sacral segments must be involved along with sensory loss and sphincter disturbance. There is considerable disagreement concerning the rapidity with which surgery must be performed, but current opinion indicates that better results are obtained if surgery is carried out in 48 hours or less after the onset of symptoms, and some authors recommend a 12-hour window of opportunity for the best outcomes.
Of course, any serious loss or neurological deficit that one would not like to be permanent can be an indication for surgery. If the patient has significant motor or sensory loss that is not promptly relieved by conservative measures, such as high-dose steroids, surgery offers a reasonable chance that progression of the deficit can be halted and some recovery may occur.
Indications for urgent cervical or thoracic surgery are almost entirely neurological. Of course, uncorrectable pain can occur, but it is less common than in the lumbar area. A serious neurological deficit is the most frequent reason for urgent surgery. The most pressing need for surgical decompression is when signs of myelopathy appear. Minor changes such as hyperreflexia or pathological signs do not necessarily lead to immediate surgery, but when the motor and sensory symptoms and signs are strongly suggestive of myelopathy, prompt surgery is required. The more serious the deficit, the more urgent the situation. There are no definitive data to determine a time course for intervention for the best outcome of the intervention.
One of the most common diseases of the lumbar spine is a herniated intervertebral disc. Disc herniations that require surgery are typically treated by exploration and excision of the offending disc material through what is termed hemi- or partial laminectomy. The procedure may be performed under local or general anesthesia. Typical surgical positions are prone and with the patient lying on the side. A small midline incision is made and the muscles and ligaments are elevated from the spinous processes and laminae. A small partial laminectomy allows access to the interspace, and then the goal of surgery is removal of the degenerated material that is causing compression of the nerve root. Surgeons vary in their opinion about removal of additional degenerative disc material from the interspace. Some surgeons use operating microscopes to provide better illumination and magnification. Our own study of more than 700 patients undergoing first-time lumbar disc surgery indicated that a significant number need more than simple discectomy. Varying degrees of canal and foraminal decompression were required for the majority of patients. The surgical technique must be tailored to the anatomical abnormalities to be corrected, and no stereotyped approach will benefit all patients (BenDebba et al 2002).
A far lateral disc herniation is extraforaminal. Treatment of this herniation requires a lateral paramedian approach.
The outcome of lumbar disc surgery is variously reported. Some studies have success rates in the 60–70% range. The best results reported typically occur in series in which selection and surgery have been carried out by one surgeon. Good outcomes in 90% or more of patients are common. In our own study involving eight centers, more than 90% of patients received statistically significant pain relief and maintained this relief throughout the 2 years of the study (O’Connell 1951, Spangfort 1972, Weber 1983, Wenger et al 2001, BenDebba et al 2002).
Recently, controlled trials of the outcome of surgery versus conservative measures have confirmed the findings of the National Low Back Pain Study (Weinstein et al 2006).
Spinal Stenosis
Another common problem is spinal stenosis. In these patients the goal of surgery is expansion of the spinal canal and neural foramina so that compression does not occur. The simplest way to do this is multilevel laminectomy over the affected segments together with foraminotomies where needed.
The syndrome of spinal stenosis is signified by back pain and leg pain with claudication. Patients are typically seen first with a simple disturbance in gait; that is, they have trouble walking well after ambulating for more than a short time. Pain then occurs, and the hallmark of the diagnosis is painful weakness in both legs brought on by walking. Typically, the pain in these patients is relieved immediately on cessation of walking, and they often assume a flexed position to speed the resolution of symptoms. These features differentiate it from vascular claudication, the symptoms of which take longer to resolve after cessation of activity. Sensory complaints are usually stocking-like and suggestive of metabolic peripheral neuropathy, with which the problem is often confused. The findings on neurological examination may be entirely normal at rest, but both motor and sensory loss can appear after activity.
The diagnosis is made with imaging studies. Plain radiographs are misleading, but either MRI (Fig. 71-3) or CT will make the diagnosis.
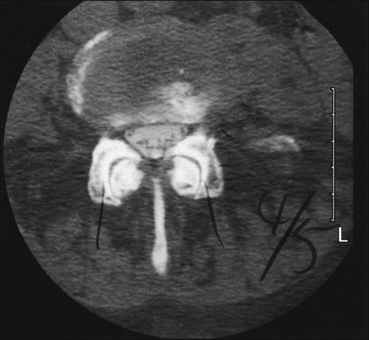
Figure 71-3 Axial magnetic resonance imaging at L4–5 demonstrating moderate canal stenosis with a trefoil appearance.
Significant foraminal stenosis and dramatic facet arthropathy are present. These changes are typical of spinal stenosis.
Treatment of spinal stenosis when the symptoms are severe is surgical decompression. The outcomes are excellent (Atlas et al 1996). The issue to be considered is when are the symptoms serious enough to warrant surgery? The majority of patients has mild to moderate incapacity and does not necessarily require surgery. When the symptoms become significantly incapacitating or when progressive important neurological deficits occur, surgery is indicated. Before that time, symptomatic treatment is warranted. Symptomatic therapies include modification of lifestyle, adequate analgesia, and an exercise activity program designed to maximize function and minimize symptoms.
Surgical treatment consists of decompressive laminectomy with foraminotomies and stabilization, if necessary. When severe back pain is an important part of the syndrome, it is probable that fusion will be required. When the syndrome is claudication, decompression alone will be adequate. It is our experience that more than 90% of patients will achieve satisfactory relief of their claudication symptoms but back pain as a complaint is more likely to persist. Even with fusion, it is our experience that no more than 60–70% of these patients achieve satisfactory relief of their back pain (Grob et al 1995, Kristof et al 2002).
Foraminal Root Compression Syndrome
A variation of the spinal stenosis problem occurs with compression of an isolated root in a spondylotic foramen. Patients have complaints similar to those of acute disc herniation, but the time course is protracted and the onset has usually been gradual. Patients complain of sciatica or femoral pain in the accepted distribution of a single root. Sometimes the root involvement is bilateral, and occasionally more than one root on one side is affected. However, the syndrome is not of claudication but of ongoing root compression; that is, the pain tends to be constant, although it is often exacerbated by activity, being upright, and axial loading. The associated reflex, motor, and sensory changes relate to the individual root. A positive straight-leg rising test is unusual and is different from the tension sign of acute disc herniation. The diagnosis is made by MRI (Fig. 71-4) and CT, which visualize the neural foramina and compression of the nerve root.
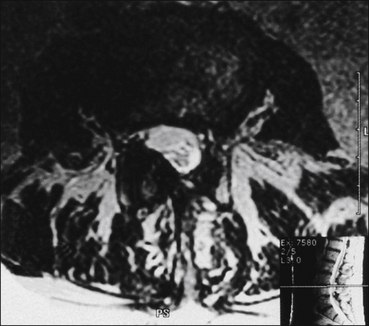
Figure 71-4 Axial magnetic resonance image demonstrating unilateral facet hypertrophy with encroachment on the spinal canal and foraminal stenosis.
The change is at L3–4 and the patient’s symptoms were right-sided anterior thigh pain.
There is little evidence that any conservative measures will benefit such patients, but if the pain is tolerable and no major neurological deficit is present, a thorough trial of an individualized exercise activity program is indicated. If the rehabilitation program is not effective and the pain is intractable or if there are associated neurological deficits, surgery should proceed. The operation consists of decompression through adequate foraminotomy. The outcome of surgery should be as good as that enjoyed for acute disc herniation (BenDebba et al 2002).
Spinal Instability and Progressive Deformity Syndromes
Progressive spinal deformities will develop in a significant number of patients with spondylotic disease. These deformities are degenerative in nature in the majority, although congenital and traumatic instability can certainly occur and will be similar in findings. Fortunately, treatment is very much the same, so the cause makes very little difference.
Degenerative Spondylolisthesis
Degenerative spondylolisthesis is the most common of the apparent instability syndromes encountered. However, the apparent movement expected because of malalignment of adjacent vertebral bodies may not be seen on flexion–extension films. It may be slowly progressive and occasionally may be fixed. Patients typically complain of back pain in the lumbar area with or without associated root signs. The diagnosis is made by plain radiographs, which should include dynamic films to be certain that the spine is moving actively.
Treatment should be symptomatic unless the pain is intractable or significant nerve root compression with a neurological deficit is occurring. Symptomatic therapies include bracing, modification of lifestyle, and individualized active exercise for axial muscle strengthening, range of motion, and leg strength. Surgery should be considered when axial back pain, radicular pain, or both are severe and disabling. Most patients with progressive listhesis or slowly progressive scoliotic deformities of any kind will require the use of a fixator for stabilization and fusion. It is best to proceed when it is apparent that the symptoms are serious enough to warrant surgery, but before extremely severe deformities occur. The surgeon should be experienced in the use of a fixator and in this complex reconstructive spinal surgery. The goal of surgery is to restore or maintain lumbar lordosis in association with decompression of individual lumbar roots and stabilization of all segments required (Turner et al 1992, Zdeblick 1993, Temple et al 1994).
When fusion is required, multiple techniques are now available. The oldest technique currently in use is onlay fusion, sometimes termed in situ or simply posterior fusion. In this procedure, bone fragments are placed in and around the zygapophyseal joints over the pars and residual laminae.
A number of new fixators are available for restoration of the normal spinal contour and stabilization. The so-called XLIF is a laterally placed interbody device that corrects the spinal contour in both the sagittal and coronal planes. The X-STOP is another device that is placed between the spinous processes to reduce spinal stenosis and provide stability at a single interspace without fusion. Dynamic fixators that stabilize the spine while still allowing motion to occur have also been developed. Reports of good outcomes following the use of all these fixators are found in the literature. Although all appear to be useful, their relative indications are still being developed through experience. Individual surgeons use all three effectively.
Posterior lateral fusions involve extension of bone graft beyond the posterior aspect to expose the transverse processes and place bone between them over the segments to be fused (Dawson et al 1981). The outcome of these bone-only fusions has been studied over many years. Fusion rates between 80% and 85% are commonly reported, and the stability provides relief in the majority of patients, although the outcomes are not as predictably good as with simple disc herniation. Obviously, the disease requiring laminectomy is more extensive than herniation of a single intervertebral disc. Orthopedists and neurosurgeons differ in their use of fusion. The majority of orthopedists use fusions routinely, whereas a minority of neurosurgeons include fusion as a part of laminectomy (Fischgrund et al 1997, Epstein 1998).
The major advance of the past 20 years in lumbar fusion has been the development of several practical systems of instrumented fixation of the spine. The most common is placement of metal screws posteriorly through pedicles into the vertebral body supplemented by a posterior supporting rod connecting all the screws. The fixator system provides instant stability, and bone is backed around the system to provide fusion and long-term stability. Such operations are more extensive than simple bone fusion and require substantially more dissection and more significant intervention. In well-selected patients outcomes are excellent, with good results reported in more than 90%.
Interbody fusion is another technique that has been developed more extensively in the past 10 years. The technique of complete removal of an intervertebral disc and replacement of bone is a half century old, but in the recent past new spacers in the form of metal cages, bone screws, or artificial disc replacements have been added. These spacers may be inserted through an anterior transabdominal retroperitoneal route or posteriorly through a midline or paramedian approach. In all these techniques the disc is as thoroughly removed as possible and then a spacer supplemented by bone is inserted to maintain or restore disc height and provide immediate stability (Lin et al 1983, O’Brien et al 1986, Regan et al 1995).
It is common to use pedicle screw and intervertebral body techniques together in the lumbar region. Reported series vary greatly in terms of outcome. The majority of patients undergoing these procedures will be benefited, but obviously these more extensive operations are required for more serious disease, and overall outcomes are not as good as with the correction of simple problems. The frequent comparative criticisms heard neglect the fact that the patients being treated are not comparable and therefore comparisons between procedures are meaningless unless identical patients are being treated (O’Brien et al 1986).
Artificial discs are an attractive possibility. All the fusion techniques require mobilization of the interspace, and thus the biomechanics of the spine are significantly altered. The concept of an artificial disc that retains the force dispersion characteristics of the real disc is under development. This looks promising in both the neck and low back region, but long-term results are lacking (McAfee 2004).
Percutaneous techniques are also commonly used and have included percutaneous mechanical discectomy, percutaneous laser discectomy, and percutaneous microdiscectomy. Therapeutic efficacy has not been demonstrated for any of these techniques in well-studied reported series. Most have enjoyed short bursts of popularity and then disappeared. At one time, intradiscal chymopapain injection was used widely. The incidence of anaphylaxis and the marginal success rate have virtually eliminated this technique (Kambin 1988, 1991a, 1991b; Onik et al 1990; Revel et al 1993; Kaiser et al 2002).
Prolotherapy consists of the injection of several types of hypertonic solutions into and around ligamentous structures of the spine. The theory is that these solutions induce proliferation and thus strengthen weakened ligaments. Yelland and collaborators (2003) studied these injections in a well-designed examination of outcomes following injections. Pain relief was satisfactory in the majority of patients undergoing injections of any kind, thus suggesting a non-specific effect, which has not yet been defined. Minimally invasive operations done with limited exposure are increasingly becoming popular. Some of these operations are truly minimally invasive, whereas others are not very different from operations that have been standard for many years. The goals of these operations are the same as for the standard procedures, which require much more extensive surgical exposure. All attempt to decompress affected nerve roots or provide stability. Many of these operations appeared to be useful in experienced hands, but the outcomes reported are mostly short-term, and definitive comparisons are not yet available (Yelland et al 2003, Lehman et al 2005).
The Failed Back Syndrome
The failed back syndrome is an imprecise term that is generally used to categorize a large group of patients who have undergone one or more of these operations on the lumbar spine without benefit (Fager and Freidberg 1980). It serves no useful purpose and should be abandoned (Connolly and Long 1999). A patient who has not benefited from one or more operations needs an evaluation that if anything, is more complex than that for a patient who has not undergone surgery (Fig. 71-5). It may be possible to make a specific diagnosis of the cause of the pain with greater frequency than in most idiopathic spondylitic back pain problems (Kieffer et al 1986). The goal of evaluation should be the most precise definition of the abnormalities possible so that an individualized treatment plan can be prescribed (LaRocca 1990).
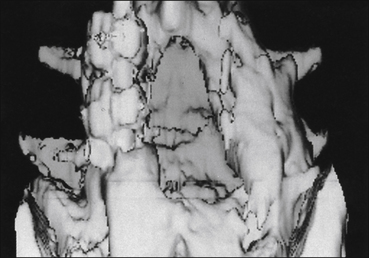
Figure 71-5 Three-dimensional reconstruction demonstrating the previous laminectomy and fusion.
These views are most useful in reconstructing the post-surgical changes and in assessing fusion stability; remember that the averaging techniques used will always overstate the solidity of the fusion.
Patients fall into broad categories within this heterogeneous group that are useful to guide evaluation and formulation of therapeutic plans:
There is another very small group of patients in whom an intercurrent diagnosis has been missed. A typical example is a patient with chronic back pain who harbors a tumor of the cauda equina, usually a schwannoma or ependymoma (Long 2000).
Whatever the proposed treatment plan, precise definition of the abnormalities that are likely to be generating the pain and that must be treated is imperative. Specifically, if any reoperative surgery is to be done, it should be planned for abnormalities that are as well defined as for first surgery. It is still true in these patients that surgery will benefit only individuals who have nerve root compression or clearly demonstrable instability (Sotiropoulos et al 1989, Srdjan 1994, Schwarzer et al 1995a).
In an extensive review of a group of patients for whom all preoperative studies were available and complete records detailed their status before failed lumbar surgery, we determined that the majority did not meet the commonly accepted criteria for lumbar surgery (Long 2000); it is therefore not surprising that surgery failed to relieve their pain. In a recent multicenter nationwide prospective study of the outcome of first-time back surgery, we determined that more than 90% of patients improved when chosen for surgery by expert spinal surgeons. By contrast, in a small group of patients rejected for surgery by these same experts, only 10% were improved by surgery carried out outside our study, and worsening of symptoms following surgery was the rule. In examining patients in whom lumbar surgery failed to relieve the symptoms, it is important to determine the original indications for the procedure if possible. Of course, it is still quite possible for such patients to have had a surgical complication that now requires correction. Even though patients did not have appropriate indications for the original surgery, they still require evaluation to determine the cause of the current problem (Cauchoix et al 1978). If the symptoms remain the same and no new abnormalities are found, it is unlikely that any surgical procedure will be beneficial. However, the original surgeons may have failed to correct an abnormality, or some complication may have occurred that will influence a decision about additional surgery. Therefore, whatever the original indications, complete evaluation of patients is required (LaRocca 1990).
The first questions relate to the pain. Is it the same or has something new occurred? Does the pain suggest a mechanical back problem only or is there a radicular component? Does the radicular pain sound neuropathic and thus suggest nerve root injury (Hanley and Shapiro 1989, Hanley et al 1991)?
Physical examination is unlikely to be helpful in the diagnosis, but it will record the patient’s current physical state.
Imaging studies begin with plain flexion–extension films. Oblique views should be included to evaluate the zygapophyseal joints. CT imaging, particularly with multidimensional reconstructions, will demonstrate bony detail, the effects of previous surgery, the status of the zygapophyseal joints, the size of the neural foramina, and such common complications as pars fracture. MRI is more useful for the examination of discs, nerve root relationships in the neural foramina, the size of the spinal canal, and inflammation (Bundschuh et al 1988, 1990; Hochhauser et al 1988; Hueftle et al 1988; Cavanagh et al 1993). If fixators are in place, three-dimensions reconstruction will eliminate the metallic artifact and provide a view of placement of the fixator. CT myelography is sometimes required, particularly when a fixator is in place.
Complications of Lumbar Surgery
The specific complications of lumbar surgery are relatively straightforward and are basically the same for all these procedures.
Leakage of spinal fluid can occur. Durotomy is a relatively frequent complication of even the most straightforward spinal surgery. Appropriate repair solves the problem, and there have been no consequences of simple durotomy in any of the surgical series yet reported. However, when repair is not successful, leakage of spinal fluid can occur and requires action to seal the leak.
Injury to neural tissue can occur. Individual nerve root injury is probably the most common event, but an occasional surgical complication results in injury to multiple roots of the cauda equina and a severe neurological deficit. Even the best surgeons occasionally have such an event take place because of the complexity of the disease and the nature of the pathology combined with the techniques available to correct the problem. With current skills, some small percentage of nerve injury cannot be avoided. The important issue when nerve injury is apparent after surgery is to determine that no correctable cause exists (Dennis et al 2009).
With either anterior or posterior approaches, injury to the bowel or the great vessels can occur. Occasionally, the anatomical arrangements are such that disc rongeurs can penetrate the annulus and injure a vessel or the bowel. With the anterior retroperitoneal approach, direct injury is possible. Both are usually recognized during surgery and repaired. Vascular injury in particular can be catastrophic, as can unrecognized injury to the bowel. These are serious operations with serious complications and patients should understand these facts when making a decision to undergo surgery (Waddell 1987a, Wetzel and LaRocca 1991, Long 2002).
A large number of complications can occur with laminectomy and fusion, including nerve root injury, pseudomeningocele, surgically induced instability, infection, and excessive scar formation (Frymoyer et al 1978, LaRocca 1990, Long 2002).
Fixators have their own set of problems. Fusions may fail (pseudarthrosis), hardware may loosen and even extrude, and sometimes hardware is inappropriately placed. It is important to examine all patients who have undergone fusion to assess the competency of bony fusion, the location of a fixator and its stability, and the appropriateness of fixator placement (Raugstad et al 1982, Steinmann and Herkowitz 1992, Yuan et al 1994).
There are other problems related to surgery that are not direct or even immediate complications. Fixation of one or more spinal segments may lead to degeneration of the segment above. Sometimes this is acute, but more typically it is a chronic problem. The symptoms are those of instability and stenosis above the fusion.
A large number of patients have obviously thick scar that may deform the thecal sac following surgery. The significance of these scars is uncertain. There is no evidence that operating on those that are not compressive will benefit the patient (Bundschuh et al 1988, 1990).
Donor Site Pain
A commonly overlooked problem is pain in the donor site over the hip where bone was harvested for grafting (Spangfort 1972). Many patients complain of seriously disabling pain in the donor site. The cause is unknown and the problem is extremely difficult to treat. The diagnosis is usually made by point tenderness over the donor site, although it may spread to the hip and thigh. Local injection virtually always relieves the pain. No consistently effective treatments have been described. The problem does seem to ameliorate with time. As autograft use declines, this problem is disappearing. Persistent complaints can be relieved by carefully controlled local injections of 1%–2.5% phenol in local anesthetic.
Arachnoiditis
Arachnoiditis is an unusual problem if viewed as a clinical syndrome, not a radiological finding. Many patients who have undergone spinal surgery with or without myelography demonstrate root adhesions and adherence of nerve roots to the dural sac (Fig. 71-6). Most of these findings have no obvious clinical significance, but some do seem to be associated with symptoms. However, chronic adhesive arachnoiditis is rarely progressive and needs to be considered in some patients. Symptoms of this rare syndrome are diffuse lower extremity pain, often neuropathic in character, and slowly progressive loss of function.
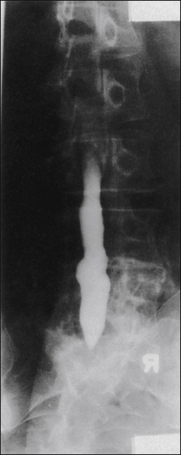
Figure 71-6 Anteroposterior view of a myelogram carried out with a water-based agent and demonstrating the changes typical of epidural fibrosis and arachnoiditis.
The thecal sac is deformed laterally by an epidural scar from L3 to L5. No root sheaths are seen; roots are adherent to the dura, and no internal structures within the thecal sac are evident. This is a typical form of arachnoiditis. The patient’s complaints were of migratory pain involving different nerve roots at different times.
MRI may suggest arachnoiditis, but myelography is required to be certain of the degree of arachnoiditis, particularly when spinal stenosis is present. Arachnoiditis was most commonly associated with infection when first described and then was described as a complication of myelography and surgery. Arachnoiditis is now seen as a complication of surgery alone in patients who have not had myelography. When this rare progressive problem is present, there is little question of the relationship between the myelographic findings of arachnoiditis and the symptomatology. However, in the majority of patients, the relationship between arachnoiditis and symptoms is still uncertain. Simply finding arachnoiditis does not imply that this pathological process is the cause of the patient’s complaints (Fitt and Stevens 1995, Long 1992).
Cervical Surgery
The most common cervical disease is degenerative changes within one or more cervical discs without overt disc herniation. Nerve root compression is usually secondary to collapse of the foramen and osteophytes. Standard treatment is anterior cervical fusion. This procedure has been used for nearly 50 years but has recently undergone metamorphosis. Techniques commonly used now include an allograft for disc replacement and immediate stabilization of the spine with an anterior cervical metallic plate. Anterior fusion is carried out in the supine position through an anterior incision that is usually made horizontally for cosmetic reasons. The discs to be removed are exposed and removed totally, along with removal of the cartilaginous plates as well. When needed, the procedure can be expanded to include total corpectomy. This is typically required only for spinal cord decompression. A disc spacer is inserted and may be artificial or treated cadaver bone. Spacers are shaped to restore lost disc height and the normal lordotic curve. The spacers are then stabilized with a plate placed anterior to the vertebral bodies and screwed in place. The operation is successful for root compression, axial neck pain, and spinal cord compression. Good outcomes in more than 90% of patients are routinely reported. Common complications include hoarseness and impairment of swallowing. Catastrophic complications include injury to the carotid artery and the spinal cord.
Isolated herniation of a cervical nucleus pulposus does occur but is much less common than the typical root compression from osteophytes. A truly herniated disc may be removed via the anterior approach or may be treated by posterior excision following foraminotomy. The posterior approach is typically used for lateral disc herniation, and the anterior approach is superior for herniations with medial extension. The posterior operations are carried out through a midline incision under general anesthesia with the patient in a prone or sitting position. Partial laminectomy and foraminotomy are used to expose the root. When the disc can be removed easily, it should be excised. However, some of these discs are difficult to mobilize, and foraminotomy alone is usually adequate to solve the problem. Outcomes of this surgery are excellent, with success rates higher than 95%.
Cervical spinal stenosis is the next most common problem (King et al 2003, Yamazaki et al 2003). Patients have varying degrees of myelopathy. Surgical treatment may consist of multilevel anterior fusion or posterior laminectomy with or without fusion. My personal practice is to perform the anterior procedure when the compression is anterior and the disease involves three or more segments. When there is compression at more than three levels, I prefer posterior laminectomy with multilevel foraminotomies and fixation with lateral metal plates and fusion if the spine is unstable. This is particularly true with a substantial component of posterior compression (Zdeblick and Bohlman 1989).
Recently, the use of fixating plates has greatly improved the fusion rate with multilevel fusions. We completed an examination of three- and four-level fusions in patients with spinal stenosis and concluded that with fixation, fusion rates comparable to those for one or two levels can be achieved. Multilevel anterior fusion has a greater incidence of hoarseness and swallowing difficulties, but excellent results can now be achieved with the newer fusion techniques.
The decision to carry out an anterior or posterior operation is usually a matter of choice by the surgeon. There are some considerations that make the posterior approach a better choice. If the patient is dependent on voice, the posterior procedure is superior. If the principal compression is posterior, the posterior procedure should be used and is usually preferable for compression syndromes with four or more levels involved. These indications are relative, however, and either technique is typically useful in most patients.
These cervical procedures are carried out in a variety of ways and in many combinations: there may be more or less bone removed up to multilevel corpectomy, several kinds of disc spacers and fixations devices can be used, and operations and devices may be used together. These are all relatively minor variations on the fundamental themes, and the basic principles remain the same; that is, the goals of surgery will include decompression of compressed neural tissue and stabilization of unstable spinal segments (Uematsu et al 1998). The choices of how to accomplish these goals are many and varied, and different surgeons will choose different approaches. At present, superiority of one over another in the general situation has not been validated (Teramoto et al 1994, Eleraki et al 1999, Kaiser et al 2002).
Artificial cervical discs are increasingly becoming popular. The major theoretical advantage is that retaining mobility in a space should reduce the incidence of transition syndrome above and below a fused area. The outcome of anterior cervical fusion is extremely good, and anterior disc replacement is unlikely to provide better outcomes, but it may certainly reduce the incidence of late complications.
Complications of Cervical Surgery
Surgery in the face of a significant myelopathy is dangerous, and patients must understand this before choosing surgery (Bhardwaj et al 2001). A compressed spinal cord is susceptible to mechanical manipulation, fluctuations in blood pressure that affect central perfusion pressure, and mechanical trauma during surgery. Serious neurological deficits can follow perfectly executed anesthesia and surgery because of the susceptibility of the damaged spinal cord to either mechanical or vascular injury.
Even though these events are uncommon, it is important that patients understand the possibility for spinal cord injury. In patients without spinal cord compression, injury to the cord itself is extremely rare. However, injury to the nerves being operated on or compromised nerves at other levels does occur. The typical nerve injury involves C5, but all are susceptible during either the anterior or the posterior approach. Most recover. When a deficit does occur, it is important to ascertain the cause as promptly as possible and institute treatment of any injury that is remediable. Unfortunately, in the majority of patients nothing is found to clearly explain the cause of a problem, and thus there will be no effective treatment (Long 2003).
Surgery for Tumors and Infection
Spinal pain from cancer is a common event that may involve direct bone invasion or may relate to pathological fractures. The surgical techniques used for spondylotic disease are generally satisfactory to deal with neoplastic disease but must often be extremely extensive to provide adequate stability.
Surgery for infection is also commonly carried out with pain as the major factor. The pain may be secondary to local inflammation, neurocompression, or bony collapse with instability. Surgery has the twin goals of eradication of the infection and elimination of pain through the provision of stability. The techniques described are generally applicable. Surgery for infection is similar to surgery for tumor, with radical bone excision and immediate fixation. The current availability of antibiotics that are nearly always effective for the treatment of discitis or osteomyelitis means that use of a metal fixator and fusion can be carried out as a primary or secondary procedure following the removal of infected bone and disc and drainage of abscesses. Local infections sometimes complicate spinal surgery through spondylotic disease. The principles of treatment are the same as for primary infection. The principal symptom of infection is pain that is typically local and may have a radicular character. Patients may or may not demonstrate evidence of systemic febrile disease. The diagnosis is made by imaging, and treatment is eradication of the infection with antibiotics, with surgery being reserved for refractory infection, drainage of abscess, decompression of neural structures, and provision of stability.
Management of spinal epidural abscess is undergoing change. For many years, discovery of an epidural abscess with or without symptoms was considered to be a strong indication for prompt surgical evacuation. Now, even with some neurological symptoms, treatment with antibiotics is frequently initiated first. Surgery proceeds only when antibiotics fail to improve the patients. Most surgeons still believe that if a patient has a significant neurological deficit, prompt surgery is the best way to treat the problem, although recovery certainly cannot be guaranteed. Timing of surgery is a matter of debate. The majority of authors favors a window of 24 hours for the best outcome, but the data are far from definitive.
Surgery for Spinal Hematoma
Another area of significant contention is the timing of surgery for spinal hematoma. As the use of anticoagulation for a number of diseases increases, so does the risk for spontaneous or post-traumatic spinal hematoma.
Postoperative hematoma has always been managed by prompt surgery as soon as the symptoms become serious and the hematoma is diagnosed. The matter of when one should operate on spontaneous or post-traumatic hematoma has been studied better. The most definitive meta-analysis currently available suggests a window of opportunity of 36–48 hours following the onset of symptoms and signs of hematoma. It is important to remember that being outside this window of opportunity does not preclude a good outcome for the patient.
Recovery is still possible even when surgery has been delayed outside the optimum time frame. The most important issue with both hematoma and infection is to recognize the problem and institute a management plan that allows surgical decompression as soon as it is obvious that the spinal cord compression is serious and/or progressive. The best data suggest that outcomes are improved if surgery occurs within a 24- to 48-hour window, with the best meta-analysis favoring 48 hours.
The references for this chapter can be found at www.expertconsult.com.
References
Ackerman S.J., Steinberg E.P., Bryan R.N., et al. Persistent low back pain in patients suspected of having herniated nucleus pulposus: radiologic predictors of functional outcome—implications for treatment selection. Radiology. 1997;203:815–822.
Ackerman S.J., Steinberg E.P., Bryan R.N., et al. Patient characteristics associated with diagnostic imaging evaluation of persistent low back problems. Spine. 1997;22:1634–1640.
Anderson G.B.J. The epidemiology of spinal disorders. In: Frymoyer J.W., ed. The adult spine, principles and practice. ed 2. Philadelphia: Lippincott; 1996:277–288.
Annertz M., Holtas S., Cronqvist S., et al. Isthmic lumbar spondylolisthesis with sciatica: MR imaging vs myelography. Acta Radiologica. 1990;31:449–453.
Atlas S.J., Deyo R.A., Keller R.B., et al. The Maine Lumbar Spine Study: Part III. 1-year outcomes of surgical and nonsurgical management of lumbar spinal stenosis. Spine. 1996;21:1787–1795.
Basmajian J.V. Acute back pain and spasm: a controlled multicenter trial of combined analgesic and antispasm agents. Spine. 1989;14:438–439.
Bell G.R., Rothman R.H. The conservative treatment of sciatica. Spine. 1984;9:54–56.
BenDebba M., Torgerson W.S., Boyd R.J., et al. Persistent low back pain and sciatica in the United States: treatment outcomes. Journal of Spinal Disorders and Techniques. 2002;15:2–15.
BenDebba M., Torgerson W.S., Long D.M. Personality traits, pain duration and severity, functional impairment, and psychological distress in patients with persistent low back pain. Pain. 1997;72:115–125.
BenDebba M., Torgerson W.S., Long D.M. A validated, practical classification procedure for many persistent low back pain patients. Pain. 2000;87:89–97.
Berman A.T., Garbarino J.L., Jr., Fisher S.M., et al. The effects of epidural injection of local anesthetics and corticosteroids on patients with lumbosciatic pain. Clinical Orthopedics. 1984;188:144–151.
Bhardwaj A., Long D.M., Ducker T.B., et al. Neurologic deficits after cervical laminectomy in the prone position. Journal of Neurosurgical Anesthesiology. 2001;13:314–319.
Bigos S.J., Bowyer O., Braen G., et al. Acute low back problems in adults. Clinical Practice Guideline No 14. AHCPR Publication No 95(0642). US Department of Health and Human Services. Rockville: MD; 1994.
Boden S.D., Davis D.O., Dina T.S., et al. Abnormal magnetic-resonance scans of the lumbar spine in asymptomatic subjects: a prospective investigation. Journal of Bone and Joint Surgery. American Volume. 1990;72:403–408.
Bogduk N. The lumbar disc and low back pain. Neurosurgery Clinics of North America. 1991;2:791–806.
Bogduk N. Diskography. American Pain Society Journal. 1994;3:149–154.
Bogduk N., Aprill C., Derby R. Selective nerve root blocks. In: Wilson D.J., ed. Interventional radiology of the musculoskeletal system. London: Edward Arnold; 1995:121–132.
Bohannon R.W., Gajdosik R.L. Spinal nerve root compression: some clinical implications. A review of the literature. Physical Therapy. 1987;67:376–382.
Bowman S.J., Wedderburn L., Whaley A., et al. Outcome assessment after epidural corticosteroid injection for low back pain and sciatica. Spine. 1993;18:1345–1350.
Bundschuh C.V., Modic M.T., Ross J.S., et al. Epidural fibrosis and recurrent disk herniation in the lumbar spine: MR imaging assessment. AJR. American Journal of Roentgenology. 1988;150:923–932.
Bundschuh C.V., Stein L., Slusser J.H., et al. Distinguishing between scar and recurrent herniated disk in postoperative patients: value of contrast-enhanced and MR imaging. AJNR. American Journal of Neuroradiology. 1990;11:949–958.
Bush K., Cowan N., Katz D.E., et al. The natural history of sciatica associated with disc pathology: a prospective study with clinical and independent radiologic follow-up. Spine. 1992;17:1205–1212.
Cauchoix J., Ficat C., Girard B. Repeat surgery after disc excision. Spine. 1978;3:256–259.
Cavanagh S., Stevens J., Johnson J.R. High-resolution MRI in the investigation of recurrent pain after lumbar discectomy. Journal of Bone and Joint Surgery. British Volume. 1993;75:524–528.
Cholewicki J., McGill S.M. Mechanical stability of the in vivo lumbar spine: implications for injury and chronic low back pain. Clinical Biomechanics. 1996;11:1–15.
Connolly E.S., Long D.M., Spinal reoperations. Benzel E.C., ed. Spine surgery: techniques, complication avoidance, and management, vol 2. New York: Churchill Livingstone; 1999:843–847.
Cramer E.D., Maher C.P., Pertigrew D.B., et al. Major neurologic deficit immediately after adult spinal surgery. Incidence and etiology over 10 years at a single training institution. Journal of Spinal Disorders & Techniques. 2009;22:565–570.
Davis H. Increasing rates of cervical and lumbar spine surgery in the United States, 1979–1990. Spine. 1994;19:1117–1124.
Dawson E.G., Lotysch M., III., Urist M.R. Intertransverse process lumbar arthrodesis with autogenous bone graft. Clinical Orthopedics. 1981;154:90–96.
Deyo R.A., Haselkorn J., Hoffman R., et al. Designing studies of diagnostic tests for low back pain or radiculopathy. Spine. 1994;19(Suppl 18):2057S–2065S.
Deyo R.A., Rainville J., Kent D.L. What can the history and physical examination tell us about low back pain? JAMA: Journal of the American Medical Association. 1992;268:760–765.
Eleraki M.A., Llanos C., Sonntag V.K. Cervical corpectomy: report of 185 cases and review of the literature. Journal of Neurosurgery. 1999;90(Suppl 1):35–41.
Epstein N.E. Decompression in the surgical management of degenerative spondylolisthesis: advantages of a conservative approach in 290 patients. Journal of Spinal Disorders. 1998;11:116–123.
Fager C.A., Freidberg S.R. Analysis of failures and poor results of lumbar spine surgery. Spine. 1980;5:87–94.
Fischgrund J.S., Mackay M., Herkowitz H.N., et al. Degenerative lumbar spondylolisthesis with spinal stenosis: a prospective randomized study comparing decompressive laminectomy and arthrodesis with and without spinal instrumentation. Spine. 1997;22:2807–2812.
Fitt G.J., Stevens J.M. Postoperative arachnoiditis diagnosed by high resolution fast spin-echo MRI of the lumbar spine. Neuroradiology. 1995;37:139–145.
France J.C., Yaszemski M.J., Lauerman W.C., et al. A randomized prospective study of posterolateral lumbar fusion: outcomes with and without pedicle screw instrumentation. Spine. 1999;24:553–560.
Frymoyer J.W., Cats-Baril W.L. An overview of the incidences and costs of low back pain. Orthopedic Clinics of North America. 1991;22:263–271.
Frymoyer J.W., Matteri R.E., Hanley E.N., et al. Failed lumbar disc surgery requiring second operation: a long-term follow-up study. Spine. 1978;3:7–11.
Grob D., Humke T., Dvorak J. Degenerative lumbar spinal stenosis: decompression with and without arthrodesis. Journal of Bone and Joint Surgery. American Volume. 1995;77:1036–1041.
Hakelius A., Hindmarsh J. The comparative reliability of preoperative diagnostic methods in lumbar disc surgery. Acta Orthopaedica Scandinavica. 1972;43:234–238.
Hakelius A., Hindmarsh J. The significance of neurological signs and myelographic findings in the diagnosis of lumbar root compression. Acta Orthopaedica Scandinavica. 1972;43:239–246.
Hanley E.N., Jr., Phillips E.D., Kostuik J.P., Who should be fused?. Frymoyer J.W., Ducker T.B., Hadler N.M., et al, eds. The adult spine: principles and practice, vol 2. New York: Raven Press; 1991:1893–1917.
Hanley E.N., Jr., Shapiro D.E. The development of low-back pain after excision of a lumbar disc. Journal of Bone and Joint Surgery. American Volume. 1989;71:719–721.
Hashimoto K., Akahori O., Kitano K., et al. Magnetic resonance imaging of lumbar disc herniation: comparison with myelography. Spine. 1990;15:1166–1169.
Hilibrand A.S., Rand N. Degenerative lumbar stenosis: diagnosis and management. Journal of the American Academy of Orthopaedic Surgeons. 1999;7:239–249.
Hochhauser L., Kieffer S.A., Cacayorin E.D., et al. Recurrent postdiskectomy low back pain: MR-surgical correlation. AJR. American Journal of Roentgenology. 1988;151:755–760.
Hueftle M.G., Modic M.T., Ross J.S., et al. Lumbar spine: postoperative MR imaging with Gd-DTPA. Radiology. 1988;167:817–824.
Hurme M., Alaranta H. Factors predicting the result of surgery for lumbar intervertebral disc herniation. Spine. 1987;12:933–938.
Hurme M., Alaranta H., Torma T., et al. Operated lumbar disc herniation: epidemiological aspects. Annales de Chirurgie et Gynaecologiae. 1983;72:33–36.
Jonsson B., Stromqvist B. Symptoms and signs in degeneration of the lumbar spine: a prospective, consecutive study of 300 operated patients. Journal of Bone and Joint Surgery. British Volume. 1993;75:381–385.
Kaiser M.G., Haid R.W., Subach B.R., et al. Anterior cervical arthrodesis after discectomy and fusion with cortical allograft. Neurosurgery. 2002;50:229–238.
Kambin P. Percutaneous lumbar discectomy: current practice. Surgical Rounds for Orthopedics. 1988;2:31–35.
Kambin P. Arthroscopic microdiskectomy. Seminars in Orthopaedics. 1991;6:97–108.
Kambin P. Arthroscopic microdiscectomy laser nucleolysis. Medicine (Baltimore). 1991;87:548–549.
Kelsey J.L. An epidemiological study of acute herniated lumbar intervertebral discs. Rheumatological Rehabilitation. 1975;14:144–159.
Kelsey J.L. Idiopathic low back pain: magnitude of the problem. In: White A.A., Gordon S.L., eds. American Academy of Orthopaedic Surgeons Symposium on Idiopathic Low Back Pain. St Louis: Mosby; 1982:5–8.
Kieffer S.A., Witwer G.A., Cacayorin E.D., et al. Recurrent post-discectomy pain: CT surgical correlation. Acta Radiologica. 1986;369(Suppl):719–722.
King J.T., McGinnis K.A., Roberts M.S. Quality of life assessment with the medical outcomes study short form-36 among patients with cervical spondylotic myelopathy. Neurosurgery. 2003;52:113–121.
Knox B.D., Chapman T.M. Anterior lumbar interbody fusion for discogram concordant pain. Journal of Spinal Discordance. 1993;6:242–244.
Koes B.W., Bouter L.M., van Mameren H., et al. A blinded randomized clinical trial of manual therapy and physiotherapy for chronic back and neck complaints: physical outcome measures. Journal of Manipulative and Physiological Therapeutics. 1992;15:16–23.
Koes B.W., Bouter L.M., van Mameren H., et al. The effectiveness of manual therapy, physiotherapy, and treatment by the general practitioner for nonspecific back and neck complaints: a randomized clinical trial. Spine. 1992;17:28–35.
Kristof R.A., Alliashkevich A.F., Schuster M., et al. Degenerative lumbar spondylolisthesis (induced radicular compression): nonfusion-related decompression in selected patients without hypermobility on flexion–extension radiographs. Journal of Neurosurgery. 2002;97:281–286.
Kuslich S.D., Ulstrom C.L., Michael C.J. The tissue origin of low back pain and sciatica: a report of pain response to tissue stimulation during operations on the lumbar spine using local anesthesia. Orthopedic Clinics of North America. 1991;22:181–187.
LaRocca H. Failed lumbar surgery: principles of management. In: Weinstein J.N., Wiesel S.W., eds. The lumbar spine. Philadelphia: Saunders; 1990:872–881.
Lawrence J., ed. Rheumatism in populations. London: Heinemann, 1977.
Lehman R.A., Jr., Vaccaro A.R., Bertagnoli R., et al. Standard and minimally invasive approaches to the spine. Orthopedic Clinics of North America. 2005;36:281–292.
Lin P.M., Cautilli R.A., Joyce M.F. Posterior lumbar interbody fusion. Clinical Orthopedics. 1983;180:154–168.
Liu S.S., Williams K.D., Drayer B.P., et al. Synovial cysts of the lumbosacral spine: diagnosis by MR imaging. AJR. American Journal of Roentgenology. 1990;154:163–166.
Long D.M. A review of psychological considerations in the neurosurgical management of chronic pain: a neurosurgeon’s perspective. Neurosurgery Quarterly. 1990;1:185–195.
Long D.M. Chronic adhesive spinal arachnoiditis: pathogenesis, prognosis, and treatment. Neurosurgery Quarterly. 1992;2:296–319.
Long D.M., Failed back syndrome. Kostuik J.P., ed. Failed spinal surgery: state of the art reviews, vol 11. Philadelphia: Hanley & Belfus; 1997:439–452.
Long D.M. Intradural lesions of the spine. In: An H.S., ed. Principles and techniques of spine surgery. Baltimore: Williams & Wilkins; 1997:551–566.
Long D.M. Reoperation for the failed back syndrome. Summary of a thirty year experience. In: Brock M., Schwarz W., Wille C., eds. First Interdisciplinary World Congress on Spinal Surgery and Related Disciplines. Monduzzi Editore, Milan: International Proceedings Division; 2000:9–15.
Long D.M. Failed back syndrome: etiology, assessment, and treatment. In: Burchiel K., ed. Surgical management of pain. New York: Thieme; 2002:354–364.
Long D.M. 2003 Perioperative care in spinal operations. MedLink Neurology Online. 2003.
Long D.M., McAfee P.C., eds. Atlas of spinal surgery. Baltimore: Williams & Wilkins, 1992.
Long D.M., BenDebba M., Torgerson W.S., et al. Persistent back pain and sciatica in the United States: patient characteristics. Journal of Spinal Disorders. 1996;9:40–58.
McAfee P.C. The indications for lumbar and cervical disc replacement. Spine Journal. 2004;4(suppl 6):177S–181S.
McCombe P.F., Fairbank J.C., Cockersole B.C., et al. Reproducibility of physical signs in low-back pain. Spine. 1989;14:908–918.
McKusick V.A. The classification of heritable disorders of connective tissue. Birth Defects Orig Artic Ser. 1975;11(6):1–9.
Meade T.W., Dyer S., Browne W., et al. Randomised comparison of chiropractic and hospital outpatient management for low back pain: results from extended follow up. British Medical Journal. 1995;311:349–351.
O’Brien J.P., Dawson M.H., Heard C.W., et al. Simultaneous combined anterior and posterior fusion: a surgical solution for failed spinal surgery with a brief review of the first 150 patients. Clinical Orthopedics. 1986;203:191–195.
O’Connell J.E.A. Protrusions of the lumbar intervertebral discs: a clinical review based on five hundred cases treated by excision of the protrusion. Journal of Bone and Joint Surgery. British Volume. 1951;33:8–30.
Onik G., Mooney V., Maroon J.C., et al. Automated percutaneous discectomy: a prospective multi-institutional study. Neurosurgery. 1990;26:228–233.
Postacchini F., Facchini M., Palieri P. Efficacy of various forms of conservative treatment in low back pain: a comparative study. Neuro-orthopedics. 1988;6:28–35.
Raugstad T.S., Harbo K., Oogberg A., et al. Anterior interbody fusion of the lumbar spine. Acta Orthopaedica Scandinavica. 1982;53:561–565.
Regan J.J., Guyer R.D., McAfee P., et al. Early clinical results of laparoscopic fusion of the L-5 to S-1 disc space. Orthopedic Transactions. 1995;19:776–777.
Revel M., Payan C., Vallee C., et al. Automated percutaneous lumbar discectomy versus chemonucleolysis in the treatment of sciatica: a randomized multicenter trial. Spine. 1993;18:1–7.
Saal J.A., Saal J.S. Nonoperative treatment of herniated lumbar intervertebral disc with radiculopathy: an outcome study. Spine. 1989;14:431–437.
Saal J.A., Saal J.S., Herzog R.J. The natural history of lumbar intervertebral disc extrusions treated nonoperatively. Spine. 1990;15:683–686.
Schwarzer A.C., Derby R., Aprill C.N., et al. The value of the provocation response in lumbar zygapophyseal joint injections. Clinical Journal of Pain. 1994;10:309–313.
Schwarzer A.C., Aprill C.N., Derby R., et al. The prevalence and clinical features of internal disc disruption in patients with chronic low back pain. Spine. 1995;20:1878–1883.
Schwarzer A.C., Wang S.C., O’Driscoll D., et al. The ability of computed tomography to identify a painful zygapophysial joint in patients with chronic low back pain. Spine. 1995;20:907–912.
Shekelle P.G., Adams A.H., Chassin M.R., et al. Spinal manipulation for low-back pain. Annals of Internal Medicine. 1992;117:590–598.
Sotiropoulos S., Chafetz N.I., Lang P., et al. Differentiation between postoperative scar and recurrent disk herniation: prospective comparison of MR, CT, and contrast-enhanced CT. AJNR. American Journal of Neuroradiology. 1989;10:639–643.
Spangfort E.V. The lumbar disc herniation: a computer-aided analysis of 2,504 operations. Acta Orthopaedica Scandinavica. 1972;142(Suppl):1–95.
Spengler D.M., Freeman C.W. Patient selection for lumbar discectomy: an objective approach. Spine. 1979;4:129–134.
Spivak J.M. Degenerative lumbar spinal stenosis. Journal of Bone and Joint Surgery. American Volume. 1998;80:1053–1066.
Srdjan M. The role of surgery for nonradicular low-back pain. Current Opinion in Orthopedics. 1994;5:37–42.
Steinmann J.C., Herkowitz H.N. Pseudarthrosis of the spine. Clinical Orthopedics. 1992;284:80–90.
Temple H.T., Kruse R.W., van Dam B.E. Lumbar and lumbosacral fusion using Steffee instrumentation. Spine. 1994;19:537–541.
Teramoto T., Ohmori K., Takatsu T., et al. Long-term results of the anterior cervical spondylodesis. Neurosurgery. 1994;35:64–68.
Triano J.J., McGregor M., Hondras M.A., et al. Manipulative therapy versus education programs in chronic low back pain. Spine. 1995;20:948–955.
Turner J.A., Ersek M., Herron L., et al. Patient outcomes after lumbar spinal fusions. JAMA: Journal of the American Medical Association. 1992;268:907–911.
Uematsu Y., Tokuhashi Y., Matsuzaki H. Radiculopathy after laminoplasty of the cervical spine. Spine. 1998;23:2057–2062.
Waddell G. Failures of disc surgery and repeat surgery. Acta Orthopaedica Belgica. 1987;53:300–302.
Waddell G. A new clinical model for the treatment of low-back pain. Spine. 1987;12:632–644.
Waddell G., McCulloch J.A., Kummel E., et al. Nonorganic physical signs in low back pain. Spine. 1980;5:117–125.
Weber H. Lumbar disc herniation: a controlled, prospective study with ten years of observation. Spine. 1983;8:131–140.
Weber H. The natural history of disc herniation and the influence of intervention. Spine. 1994;19:2234–2238.
Weinstein J.N., Tosteson T.D., Lurie J.D., et al. Surgical VS nonoperative treatment for lumbar disk herniation. The Spine Patient Outcomes Research Trial (SPORT): a randomized trial. JAMA: Journal of the American Medical Association. 2006;296:2441–2450.
Wenger M., Mariani L., Kalbarczyk A., et al. Long-term outcome of 104 patients after lumbar sequestrectomy according to Williams. Neurosurgery. 2001;49:329–335.
Wetzel F.T., LaRocca H. The failed posterior lumbar interbody fusion. Spine. 1991;167:839–845.
Wiesel S.W., Cuckler J.M., Deluca F., et al. Acute low-back pain: an objective analysis of conservative therapy. Spine. 1980;5:324–330.
Wiesel S.W., Tsourmas N., Feffer H.L., et al. A study of computer-assisted tomography: I. The incidence of positive CAT scans in an asymptomatic group of patients. Spine. 1984;9:549–551.
Woertgen C., Rothoerl R.D., Englert C., et al. Pyogenic spinal infections and outcome according to the 36-Item Short Form Health Survey. Journal of Neurosurgery. Spine. 2006;4:441–446.
Yamazaki T., Yanaka K., Sato H., et al. Cervical spondylotic myelopathy: surgical results and factors affecting outcome with special reference to age differences. Neurosurgery. 2003;52:122–126.
Yelland J.M., Glasziou P.P., Bogduck N., et al. Prolotherapy injections, saline injections, and exercises for chronic low-back pain: a randomized trial. Spine Journal. 2003;3:253–254.
Yuan H.A., Garfin S.R., Dickman C.A., et al. A historical cohort study of pedicle screw fixation in thoracic, lumbar, and sacral spinal fusions. Spine. 1994;19(Suppl 20):2279S–2296S.
Zdeblick T.A. A prospective, randomized study of lumbar fusion: preliminary results. Spine. 1993;18:983–991.
Zdeblick T.A., Bohlman H.H. Cervical kyphosis and myelopathy. Treatment by anterior corpectomy and strut-grafting. Journal of Bone and Joint Surgery. American Volume. 1989;71:170–182.
Zeidman S.M., Long D.M. Does a herniated lumbar disk with nerve root entrapment require surgical intervention: are alternative treatments as effective? Current Pain and Headache Reports. 1998;2:115–119.