6
Rh Blood Group System
On completion of this chapter, the reader should be able to:
1. Explain how the D antigen received the terminology of Rh.
2. Compare and contrast the current genetic theory of the inheritance of Rh blood group system antigens with theories proposed by Fisher-Race and Wiener.
3. Discuss the biochemistry of the Rh blood group system, including the gene products and antigen structures.
4. Translate between the Fisher-Race and Wiener terminology.
5. Express phenotyping results for the Rh blood group system antigens in the terminology currently accepted by the International Society of Blood Transfusion.
6. Predict the most probable Rh genotype given the Rh antigen typing results (phenotype).
7. Define weak D and list the genetic circumstances that can cause this phenotype.
8. Describe the appropriate application and test procedure for the weak D antigen.
9. Interpret results of the weak D test when the control is positive, and explain why this would occur.
10. Define compound antigens, and give two examples of this phenotype.
11. Distinguish the G antigen from other antigens in the Rh blood group system, and explain the significance of anti-G.
12. Explain the relationship of the RHAG blood group system to the Rhnull and Rhmod phenotypes.
13. Describe the characteristics of the Rh blood group system antibodies and their clinical significance with regard to transfusion and hemolytic disease of the fetus and newborn (HDFN).
14. Compare and contrast the antibody and antigen characteristics of the LW blood group system to the Rh blood group system.
15. Solve case studies with data from Rh phenotyping.
The Rh blood group system is highly complex, polymorphic, and the second most important blood group system after the ABO blood group system. Since the initial discovery of the D antigen in 1939, the Rh blood group system has grown to include more than 50 related antigens. This chapter focuses on the five principal antigens—D, C, E, c, and e—and their corresponding antibodies, which account for most clinical transfusion issues.
Section 1
Historical Overview of the Discovery of the D Antigen
The terms “Rh-positive” and “Rh-negative” refer to the presence or absence of the D red cell antigen; these terms are also known as “D-positive” and “D-negative.” In contrast to the ABO blood group system, the absence of the D antigen or other Rh blood group system antigens on the red cell does not typically correspond with the presence of the antibody in the plasma. In other words, individuals who phenotype as “group A, D–negative” would have anti-B in their serum but not anti-D. The production of anti-D and other Rh blood group system antibodies requires immune red cell stimulation from red cells positive for the antigen. This exposure may occur during transfusion or pregnancy.
The discovery of the Rh blood group system, as with many other blood group systems, followed the investigation of an adverse transfusion reaction or hemolytic disease of the fetus and newborn (HDFN). In 1940, Levine and Stetson linked the cause of HDFN to an antibody in the Rh blood group system.1 They named the system Rh, based on the characteristics of the maternal antibody with one reported by Landsteiner and Weiner. Landsteiner and Weiner reported an antibody made from stimulating guinea pigs and rabbits with Rhesus macaque monkey red cells.2 The Rh antibody agglutinated 85% of human red cells tested and was nonreactive with 15%. From this discovery, the population was characterized as Rh positive or Rh negative. Later experiments demonstrated that the Rh antibody made in the guinea pigs and rabbits was similar, but not identical, to the anti-Rh produced by humans. The two antibodies were different. The rhesus antibody specificity was directed toward another red cell antigen, named LW in honor of Landsteiner and Wiener. The name of the Rh blood group system had been established by then and was not changed.
Section 2
Genetics, Biochemistry, and Terminology
Genetics and Biochemistry of the Rh Blood Group System
The current theory of genetic control of Rh antigen expression was enhanced with the ability to characterize the amino acid sequences produced by genes that code for proteins on the red cell membrane. Originally postulated by Tippett,3 the Rh blood group system antigens were encoded by two closely linked genes—RHD and RHCE—on chromosome 1. RHD determines the D antigen expression on the surface of red cells. D-negative individuals have no genetic material at the site.4 An antithetical “d” antigen does not exist. Adjacent to the RHD locus, the gene RHCE determines the C, c, E, and e antigen specificities. Alleles present at this locus include RHCE, RHCe, RHcE, and RHce. The antigens CE, Ce, cE, and ce are expressed (Fig. 6.1).5 The RHCE gene codes for similar polypeptides, distinguished by two amino acid sequences as illustrated in Fig. 6.2.6 The assortment of other antigens in the Rh blood group system occurs as a result of variations of these polypeptides embedded in the cell membrane bilayer in unique configurations. The RHD gene, which codes for the D antigen, can vary by many more amino acids, creating more variability among individuals. These differences between individuals help explain why exposure to D antigen can result in a likely immune response.7 A list of commonly encountered Rh antigens is provided in Table 6.1.
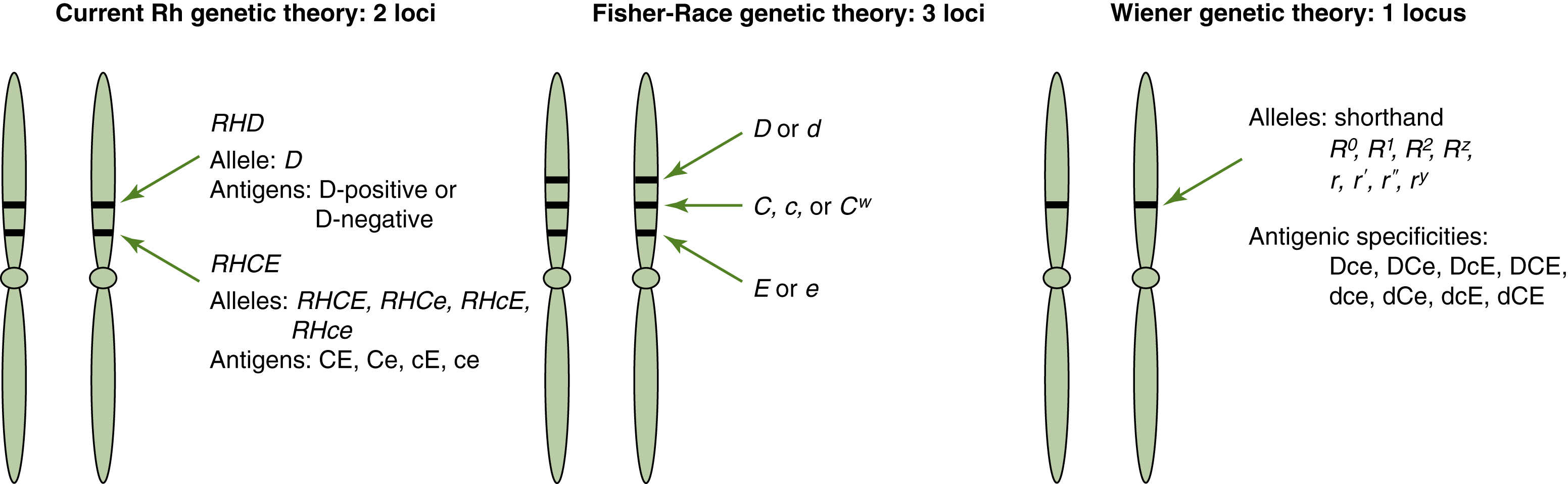
Fig. 6.1 Comparison of Rh genetic theories.
Comparison of three Rh genetic theories that have influenced the nomenclature of the Rh blood group system. Modern molecular techniques have established that the Rh blood group system antigens are determined by two genetic loci.
The products of both the RHD and the RHCE genes are proteins of 416 amino acids that traverse the membrane 12 times and display short loops of amino acids on the exterior (see Fig. 6.2).7 The Rh blood group system polypeptides, in contrast to most blood group–associated proteins, carry no carbohydrate residues. Rh antigens have been detected only on red cell membranes. Specific antibodies also do not recognize Rh proteins when the proteins are separated from the membrane.8 The functions of the Rh antigens on the red cells might be related to cation transport and membrane integrity.9 The lack of Rh blood group system antigens, called Rhnull, causes a membrane abnormality that shortens red cell survival. Rhnull is discussed in further detail later in this chapter.
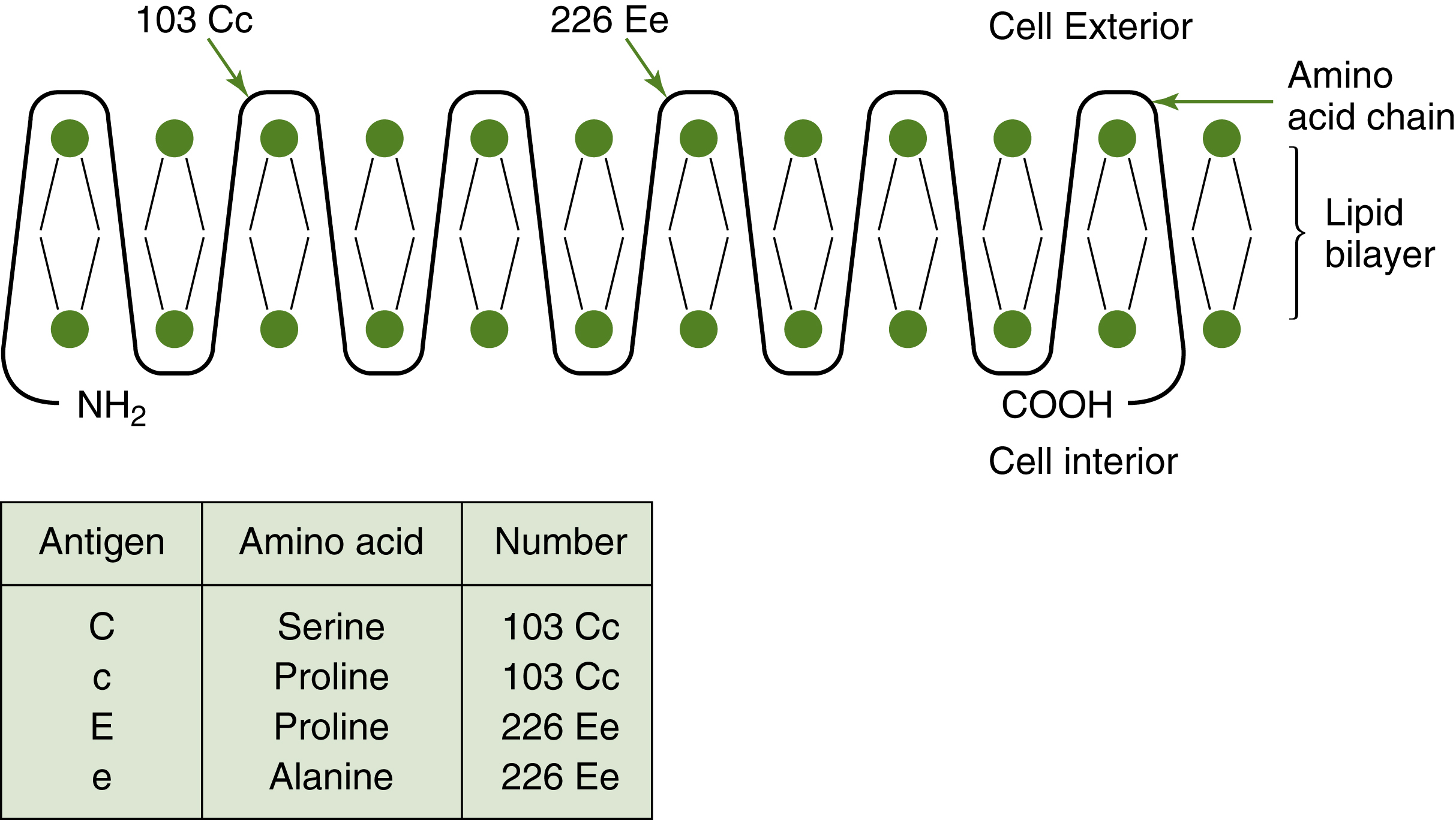
Fig. 6.2 Model of the Rh polypeptide.
Model of the differences in the amino acid sequence for the antigens produced by the RHCE gene. The basic structure is similar. Differences in the amino acid at the residue number indicated determine the serologic typing to be C or c, E or e.
TABLE 6.1
Common Antigens in the Rh Blood Group System: Equivalent Notations
| Numeric | Fisher-Race | Other Names | ISBT No. |
| Rh1 | D | Rh + | 004001 |
| Rh2 | C | 004002 | |
| Rh3 | E | 004003 | |
| Rh4 | c | 004004 | |
| Rh5 | e | 004005 | |
| Rh6 | ce | cis-ce or f | 004006 |
| Rh7 | Ce | cis-Ce | 004007 |
| Rh8 | Cw | 004008 | |
| Rh9 | Cx | 004009 | |
| Rh10 | ces | V | 004010 |
| Rh12 | G | 004012 |

Another gene, Rh-associated glycoprotein (RHAG), resides on chromosome 6.11 This gene is important to the expression of the Rh antigens. RHAG encodes a glycosylated polypeptide (protein with attached carbohydrates) with a structure very similar to the Rh proteins. The RHAG forms complexes with the Rh proteins and must be present for Rh antigen expression. By itself, the RHAG does not express any Rh antigens. The International Society of Blood Transfusion (ISBT) has assigned a blood group system to the RHAG.
Rh Terminologies
This section describes four different terminologies used in the Rh blood group system. Two terminologies, Fisher-Race and Weiner, were derived from genetic theories of Rh inheritance. These systems reflect serologic observations and inheritance theories based on family studies. Because these systems are used interchangeably in the transfusion setting, it is necessary to understand these theories well enough to “translate” from one to another. Fisher-Race terminology is used to name the antibody found in a patient specimen. Weiner terminology is used to identify red cell phenotypes on the antibody identification panel cells, etc. Two additional systems were developed because of a need for a universal language compatible with computers. The third terminology, Rosenfield, describes the presence or absence of an Rh antigen. The ISBT created the fourth terminology. Blood bank professionals must be familiar with all of these terminologies when discussing the Rh blood group system. Table 6.1 lists the equivalent notations for the more common Rh antigens.
Fisher-Race: CDE Terminology
Fisher and Race12 postulated that the Rh blood group system antigens were inherited as a gene complex or haplotype that codes for three closely linked sets of alleles. The D gene is inherited at one locus, C or c genes are inherited at the second locus, and E or e genes are inherited at the third locus. Each parent contributes one haplotype or set of Rh genes. Fig. 6.1 illustrates this concept. Each gene expresses an antigen that is given the same letter as the gene. When referring to the gene, the letter is italicized. For example, the gene that produces the C antigen is C. Each red cell antigen can be recognized by testing with a specific antibody. The original theory assumed the d allele was present when the D allele was absent. According to the Fisher-Race theory, the order of the genes on the chromosome is DCE; however, it is often written alphabetically as CDE.
Wiener: Rh-Hr Terminology
In contrast to the Fisher-Race theory, Wiener13 postulated that alleles at one gene locus were responsible for expression of the Rh blood group system antigens on red cells. Each parent contributes one RH gene. The inherited form of the gene may be identical (homozygous) to or different (heterozygous) from each parent. According to Wiener, eight alleles exist at the RH gene locus: R0, R1, R2, Rz, r, r′, r″, and ry. The gene encodes a structure on the red cell called an agglutinogen, identified by its parts or factors. These factors are identified with the same antisera that agglutinate the D, C, c, E, and e antigens mentioned earlier in the Fisher-Race nomenclature. The difference between the Wiener and Fisher-Race theories is the inheritance of the Rh blood group system on a single gene locus rather than three separate genes. The antigen complex or agglutinogen comprises factors that are identifiable as separate antigens (Table 6.2). For example, in Wiener terminology, the R1 gene codes for the Rh1 agglutinogen, which is made up of factors Rh0, rh′, and hr″ that correspond to D, C, and e antigens, respectively. The r gene codes for the rh agglutinogen, made up of factors hr′ and hr″ that correspond to c and e antigens, respectively. The longhand factor notations of Rh0, rh′, hr′, rh″, and hr″ that correspond to D, C, c, E, and e antigens, respectively, are outdated and rarely used.
Wiener terminology can be easily translated to Fisher-Race terminology when the following points are kept in mind: R is the same as D; r indicates no D antigen; the number 1 and the character ′ denote C; and the number 2 and the character ″ denote E (Table 6.3). For example, in Wiener nomenclature, c, D, and E factors or antigens would be written as R2. Although most workers prefer the Fisher-Race terminology to Wiener terminology, it is often easier to describe a phenotype as R2R2 than as D+, C−, c+, E+, e−. Subscripts refer to antigens, whereas italics and superscripts indicate genes.
Rosenfield: Numeric Terminology
Both the Wiener and the Fisher-Race terminologies are based on genetic concepts. The Rosenfield system was developed to communicate phenotypic information more suited for computerized data entry; it does not address genetic information.14 In this system, each antigen is given a number that corresponds to the order of its assignment in the Rh blood group system. A red cell’s phenotype is expressed with Rh followed by a colon and the numbers corresponding to the tested antigens. If a red cell sample is negative for the antigen tested, a minus sign is written before the number. For example, red cells that tested D+, C+, E−, c+, e+ would be written as Rh:1,2,−3,4,5. Table 6.1 compares Fisher-Race and numeric terminology.
International Society of Blood Transfusion: Standardized Numeric Terminology
The ISBT, in an effort to standardize blood group system nomenclature, assigned a six-digit number to each blood group specificity.15 The first three numbers represent the system, and the remaining three represent the antigen specificity. The assigned number of the Rh blood group system is 004, and the remaining three numbers correspond to the Rosenfield system. For example, the ISBT number for the C antigen is 004002. An ISBT “symbol,” or alphanumeric designation similar to the Rosenfield terminology, is used to refer to a specific antigen. The term Rh is written in uppercase letters, and the antigen number immediately follows the system designation. The ISBT symbol for C is RH2. A partial list of Rh antigens that includes the ISBT numeric designation is given in Table 6.1.
Determining the Genotype from the Phenotype
The term phenotype refers to the test results obtained with specific antisera, and the term genotype refers to the genetic makeup of an individual. The genotype cannot be determined without family studies or molecular testing but can be inferred from the phenotype based on the frequency of genes in a population. Five antisera, used to determine the Rh blood group system phenotypes, include anti-D, anti-C, anti-c, anti-E, and anti-e. Agglutination with the antisera indicates that the antigen is present on the red cell; no agglutination indicates the absence of the antigen.
TABLE 6.4
Order of Frequency of the Common Rh Blood Group System Haplotypes
| White | Black | Rare (Both Races) | |
| CDe (R1) |  | cDe (R0) | Ce (r′) |
| ce (r) | ce (r) | cE (r″) | |
| cDE (R2) | CDe (R1) | CE (ry) | |
| cDe (R0) | cDE (R2) | CDE (RZ) |
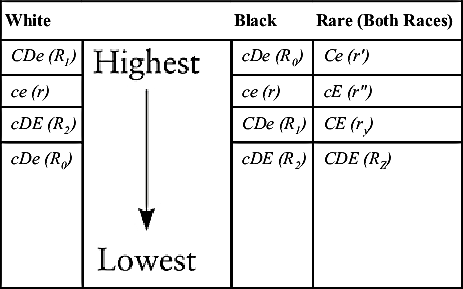
TABLE 6.5
| Results with Antisera | Genotype | Genotype Frequency (%) | |||||||
| Anti-D | Anti-C | Anti-E | Anti-c | Anti-e | Phenotype | CDE | Rh-hr | White | Black |
| + | + | − | + | + | CcDe | CDe/ce | R1r | 31 | 9 |
| CDe/cDe | R1R0 | 3 | 15 | ||||||
| Ce/cDe | r′R0 | <1 | 2 | ||||||
| + | + | − | − | + | CDe | CDe/CDe | R1R1 | 18 | 3 |
| CDe/Ce | R1r′ | 2 | <1 | ||||||
| + | − | + | + | + | cDEe | cDE/ce | R2r | 10 | 6 |
| cDE/cDe | R2R0 | 1 | 10 | ||||||
| + | − | + | + | − | cDE | cDE/cDE | R2R2 | 2 | 1 |
| cDE/cE | R2r″ | <1 | <1 | ||||||
| + | + | + | + | + | CcDEe | CDe/cDE | R1R2 | 12 | 4 |
| CDe/cE | R1r″ | 1 | <1 | ||||||
| Ce/cDE | r′R2 | 1 | <1 | ||||||
| + | − | − | + | + | cDe | cDe/ce | R0r | 3 | 23 |
| cDe/cDe | R0R0 | <1 | 19 | ||||||
| − | − | − | + | + | ce | ce/ce | rr | 15 | 7 |
| − | + | − | + | + | Cce | Ce/ce | r′r | <1 | <1 |
| − | − | + | + | + | cEe | cE/ce | r″r | <1 | <1 |
| − | + | + | + | + | CcEe | Ce/cE | r′r″ | <1 | <1 |

When the phenotype is known, the most probable genotype can be determined by knowing the most common Rh blood group system genes for the race of the person being tested (Table 6.4). In the white population, the four most common genes encountered, in order of frequency from highest to lowest, are CDe (R1), ce (r), cDE (R2), and cDe (R0). In the black population, the order of gene frequency from highest to lowest is cDe (R0), ce (r), CDe (R1), and cDE (R2). The genes Ce (r′), cE (r″), CDE (Rz), and CE (ry) are not commonly found in either race. If a red cell specimen were phenotyped as D+, C+, E−, c+, e+, the phenotype would be CcDe. When inferring the genotype from the white population, the combination CDe/ce or R1r would be the most probable genotype. In the black population, the most probable genotype would be CDe/cDe or R1R0 because the R0 allele is more common than the r allele. Table 6.5 lists phenotypes determined by reactions with specific antisera and the most probable genotype based on gene frequency in the population.
Pedigree diagrams illustrate inheritance patterns. In Fig. 6.3, the inheritance of the Rh blood group system is diagrammed to illustrate the concept that the Rh blood group system is inherited as a haplotype. Because the RHD and RHCE loci are close on chromosome 1, it is easy to follow the inheritance of the gene complex using Wiener terminology. A Punnett square, which can predict phenotypes and genotypes, can also be used to illustrate the probability of being D-positive or D-negative (Fig. 6.4).
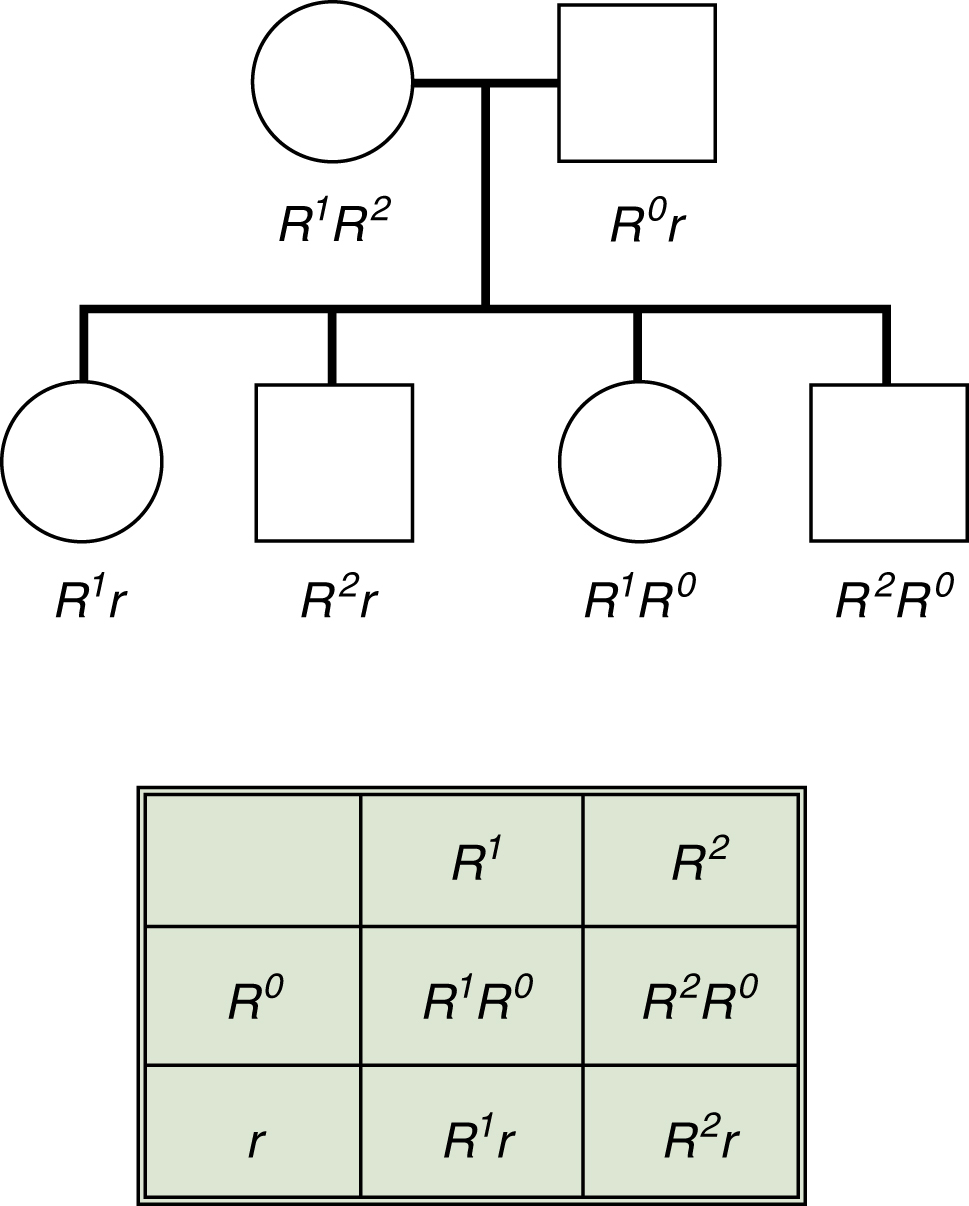
Fig. 6.3 Inheritance of Rh haplotypes.
Because the genes coding for the Rh blood group system are very close on the chromosome, Rh antigens are inherited as haplotypes. This inheritance is illustrated in a pedigree chart and in a Punnett square.
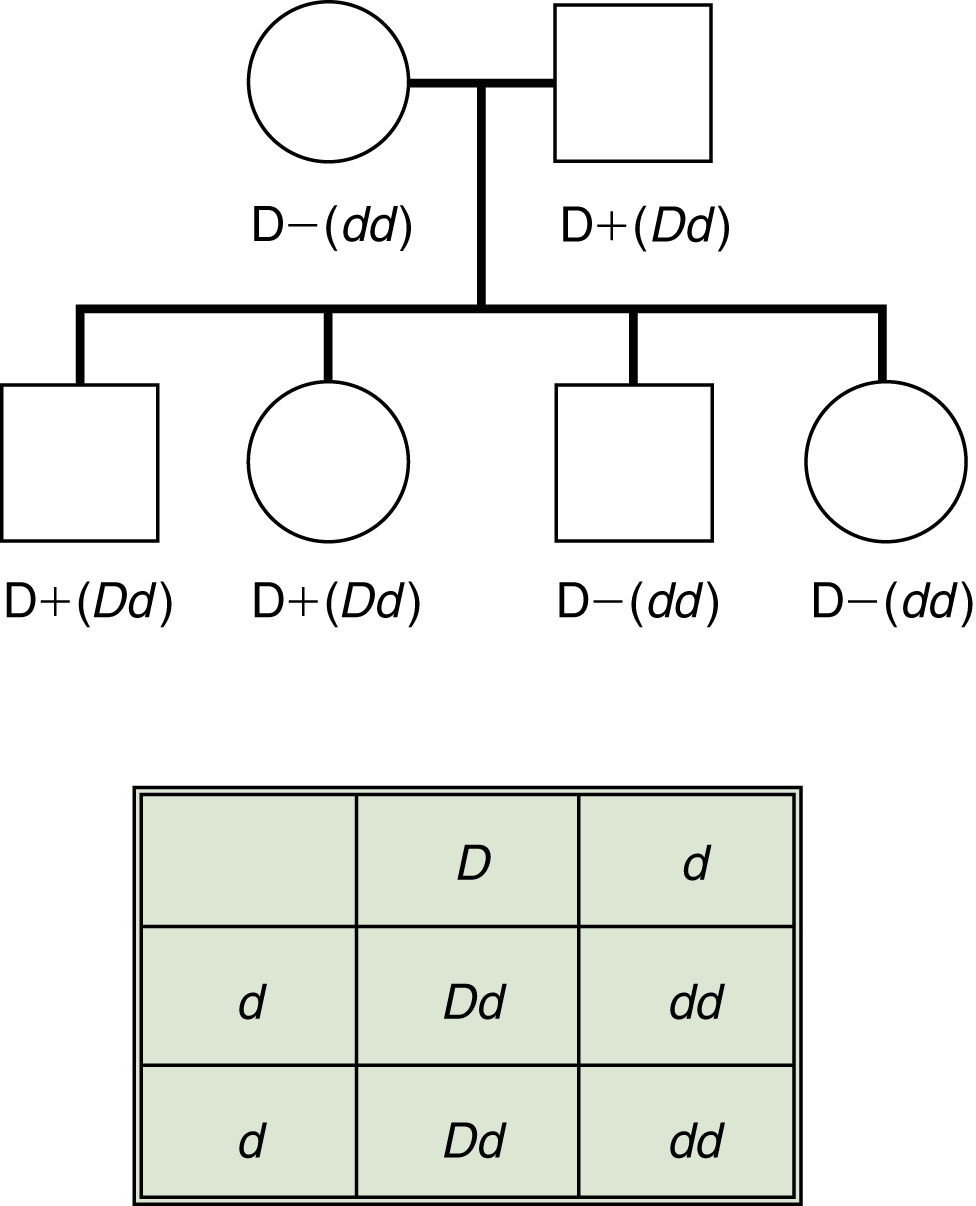
Fig. 6.4 Inheritance of the D antigen.
Predicting the probability of D-positive offspring from a D-negative mother (dd) and a heterozygous (Dd) father. The d gene does not exist and is used only for illustrative purposes. From this mating, 50% of the children could be D-positive.
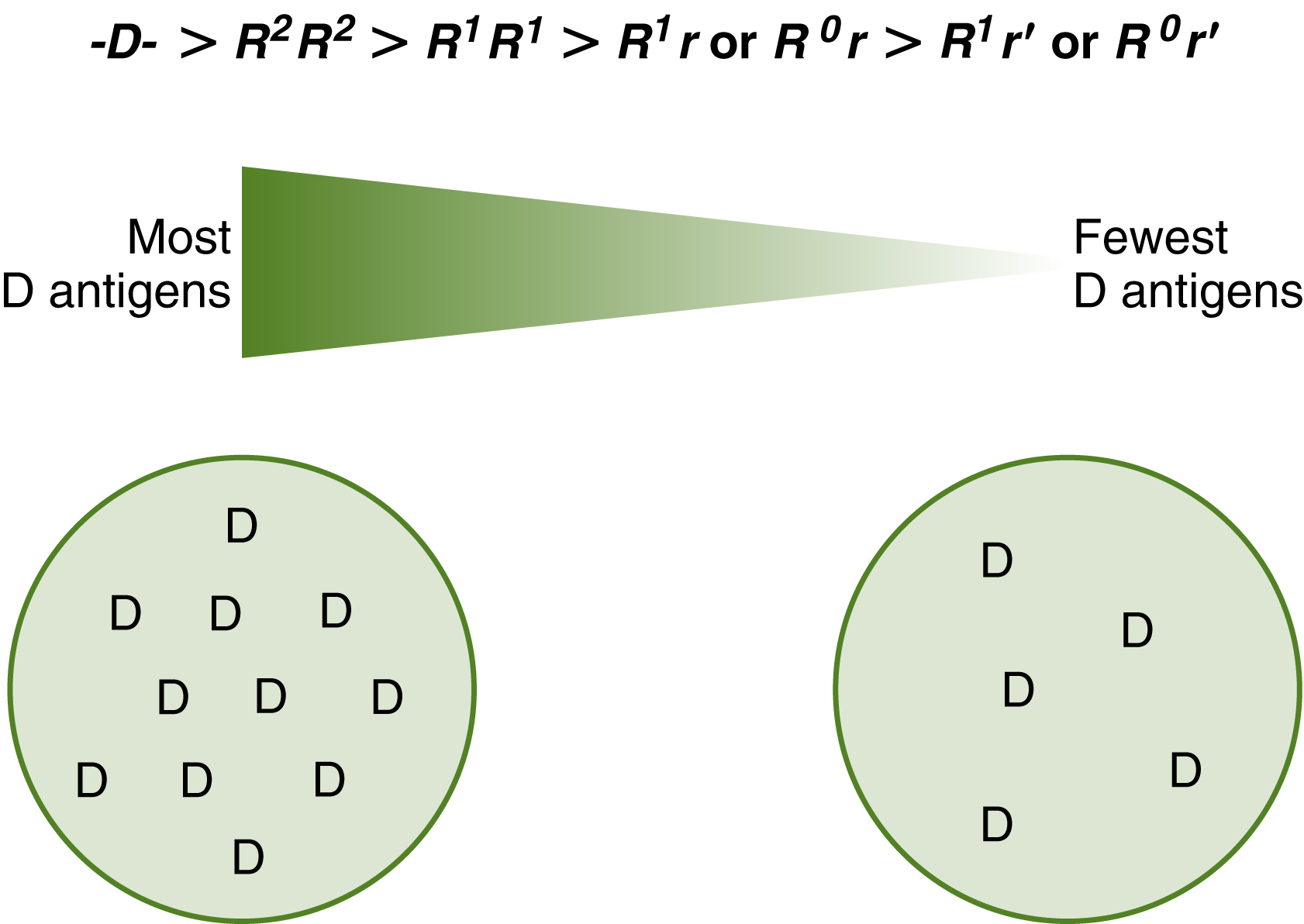
Fig. 6.5 D antigen concentration.
The D antigen concentration varies with the antigens inherited at the RHCE gene. The D-deletion phenotype has the most D antigen sites. The C gene weakens the D antigen expression if inherited on the opposite chromosome. R2R2 cells show a higher D expression than R1R1 cells. When anti-D reacts with R2R2 cells, the red cells would typically demonstrate a stronger pattern of agglutination.
Section 3
Antigens of the Rh Blood Group System
D Antigen
The D antigen is the most immunogenic antigen in the Rh blood group system. Immunogenicity refers to the ability of an antigen to elicit an immune response. As high as 70% to 85% of D-negative people receiving a D-positive red blood cell (RBC) transfusion have been reported to produce an antibody with anti-D specificity.16,17 Other reports have placed a 50% to 70% probability of immunization.18 For that reason, a D-negative patient should receive D-negative RBC units. Fig. 6.5 illustrates the variation of the D antigen concentration in different phenotypes. Scan the QR codes for more information.
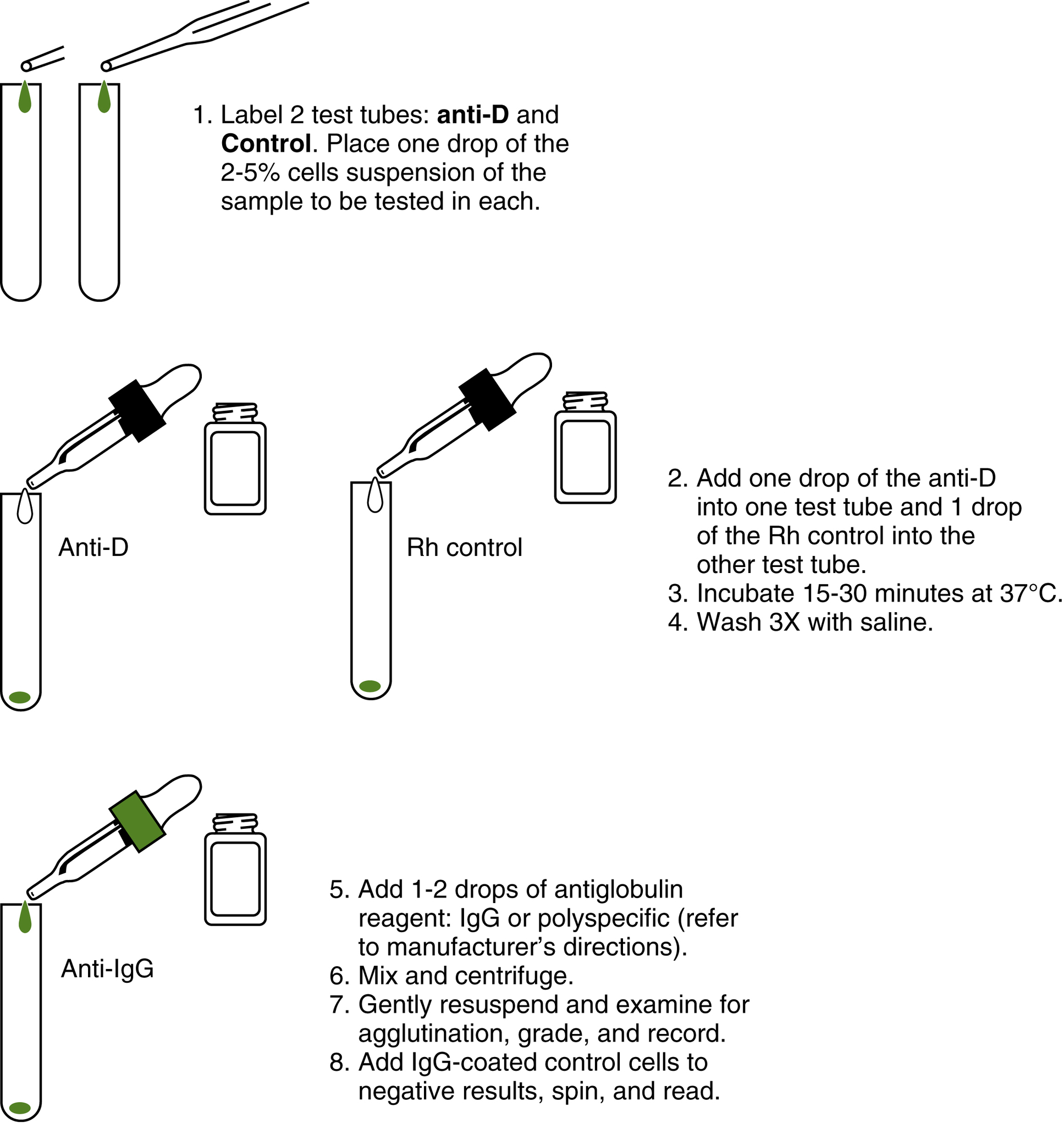
Weak D
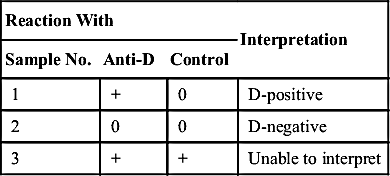
Most red cells can be phenotyped for the D antigen directly with anti-D commercial reagents. Although the antibody to D antigen is typically of the IgG class, reagent manufacturers have developed monoclonal anti-D antibodies for concurrent use with anti-A and anti-B testing. When the D antigen is weakly expressed on the red cell, its detection may require the indirect antiglobulin test (IAT) (Fig. 6.6). Red cells that are positive for D only by the IAT are referred to as weak D. Recently the serologic weak D definition has been modified to reactivity of RBCs with anti-D reagent giving no or weak (≤2+) reactivity in initial testing with moderate or strong reactivity with antihuman globulin (AHG). Table 6.6 shows the interpretation of this test, which must always include a control. The Rh or D control is a reagent made by manufacturers that consists of all additives except the D antibody. It is used to determine whether agglutination by anti-D at immediate-spin is false positive, which could be due to the reagent additives, such as albumin. The Rh or D control tested at the antiglobulin phase determines whether patient cells are already coated with IgG antibodies before testing. Reagent manufacturers specify the use of controls in their package inserts, and it is important to become familiar with these guidelines. Chapter 3 discusses Rh reagents in detail. If the control is positive, additional serologic techniques may be required.
Older terminology classified weak D antigens as Du. The IAT used to determine whether a weak form of D is present is still sometimes referred to as the Du test, although this is incorrect terminology.19 Newer monoclonal antibody reagents for Rh blood group system antigens have enhanced the ability to detect the weaker D antigens without additional IAT testing.
TABLE 6.6
Weak D Test: Interpretation with Control Results
| Reaction With | Interpretation | ||
| Sample No. | Anti-D | Control | |
| 1 | + | 0 | D-positive |
| 2 | 0 | 0 | D-negative |
| 3 | + | + | Unable to interpret |
Weaker D expression can result from several different genetic circumstances outlined briefly in the following section. They are weak D due to genetics, position effect, and partial D. Only the weak D due to genetics requires detection of the D antigen by the IAT.
Weak D: Genetic
Some RHD genes code for a weaker expression of the D antigen. This quantitative variation in the RHD gene is more common in blacks and is often part of the cDe (R0) haplotype. An IAT using anti-D is usually required to detect this form of D antigen.
Weak D: Position Effect
Weaker expression of the D antigen can be found when the Ce (r′) gene is inherited in trans to the RHD gene (Fig. 6.7). Genes inherited in trans are inherited on opposite chromosomes. The Ce (r′) gene paired with a CDe (R1) or a cDe (R0) gene weakens the expression of the D antigen. Weak D antigen due to position effect is usually detected with monoclonal anti-D reagents because of the increased sensitivity of monoclonal reagents. The occurrence of the Ce (r′) gene is less than 2%.
Weak D: Partial D
Although rare, some individuals who are positive for the D antigen can make an alloantibody that appears to be anti-D after exposure to D-positive red cells. Investigation of this phenomenon revealed that some D-positive cells could be missing parts of the D antigen complex. When these individuals are exposed to the “whole D” antigen, they can make an antibody to the part they are missing. In the past, the partial D phenotype was termed D variant or D mosaic. Lomas et al.20 established nine partial D phenotypes, which are classified by their parts or epitopes. The nine-epitope model was later expanded to accommodate new reaction patterns with different monoclonal anti-D reagents.21 Red cells of most partial D phenotypes react as strongly with monoclonal antibody anti-D reagents as red cells composed of the complete D antigen. For this reason, partial D phenotypes are infrequently detected. A partial D phenotype should be suspected if a D-positive individual makes an antibody that reacts with D-positive cells but is nonreactive with his or her own cells.19 In addition, the partial D phenotype should be suspected if two different manufacturers’ monoclonal anti-D reagents are used and the interpretation as D-positive or D-negative does not agree. In this circumstance, the clone used to manufacture the reagent may vary in its ability to detect all the epitopes or parts of the D antigen. The types of weak D are summarized in Table 6.7.
Significance of Testing for Weak D
The AABB Standards requires testing for weak D on donor red cells that do not directly agglutinate with anti-D reagents using a method designed to detect weak expression of D.22 There is no requirement for a test that uses an IAT.19 Weak D–positive units are labeled D-positive and should be transfused only to D-positive recipients. A D control or an autocontrol is an important part of the weak D test because it verifies that a positive result is not due to red cells already coated with antibodies. If red cells are coated with IgG antibodies before testing with anti-D at the antiglobulin phase, the test is invalid and additional procedures are required to determine the D antigen status of the donor. Scan the QR code for more information.
Testing for weak D on potential transfusion recipient samples is not required. Many facilities perform only the direct test for the D antigen and do not complete the antiglobulin procedure if the reaction is negative. This policy may be most cost effective in terms of time and reagents because most D-negative individuals do not test positive for the weak D antigen. Patients are classified as D-negative and transfused with D-negative blood.
Some facilities test for weak D on recipient samples. If a weak D phenotype is detected, D-positive blood is provided. Although unlikely, a patient with a weak D antigen because of the partial D phenotype can theoretically make anti-D. The partial D phenotype is uncommon and with current monoclonal antibody reagents usually does not require the antiglobulin test for detection.
TABLE 6.7
| Types of Weak D Expression | Detected by | Can Make Anti-D |
| Genetic, reduced D antigen | Weak D test | No |
| Ce in trans to RHD (example: R0r′) | Monoclonal reagents | No |
| Partial D | Most monoclonal reagents and/or weak D test | Yes, antibody to the missing epitope |
TABLE 6.8
Summary of Less Common Rh Blood Group System Antigens and Antibodies
| Antigen | Antigen Characteristics | Antibody Characteristics |
| ce or f | cis-product antigen; present when c and e are inherited as a haplotype | Rare antibody; can cause HTR and HDFN; c− or e− blood is f− |
| Ce or Rh7 | cis-product antigen; present when C and e are inherited as a haplotype | Anti-Ce is often made by D+ patients who make anti-C |
| Cw | Low-frequency antigen, found in 2% of whites and rarely in blacks; most Cw+ are also C+ | Can be naturally occurring; immune examples can cause HDFN and HTR |
| Cx | Low (<0.01%) occurrence; Cx+ is C+ | Rare, can cause mild HTR and HDFN |
| V or Ces | Found in 30% of blacks and <1% of whites | Often found with other antibodies; can cause HTR but not HDFN |
| G | Present on most D+ and all C+ cells | Antibody appears to be anti-D and anti-C; can cause HDFN and HTR |
| Rh29 or total Rh | Present on all red cells except Rhnull cells | Anti-Rh29 is made by Rhnull individuals (amorph and regulator) |
| RH:17 | Present on all red cells except -D- cells (D deletion) | Antibody made by individuals who are -D- |
| hrs | e-like antigens (e variants) produced by all Rh genes that make e; antigen hrs is associated with weak e antigen typing | Antibodies found when an e+ person makes an apparent anti-e |
Weak D testing is also performed on prenatal evaluations and Rh immune globulin (RhIG) workups. The decision to give RhIG to weak D–positive women during prenatal evaluations varies among institutions. It is unknown if prophylaxis would be successful in the case of a mother with a partial D phenotype with a D-positive infant. This clinical situation is discussed further in the chapter on HDFN (Chapter 12). Some debate about these policies exists; however, the important element in weak D testing by the IAT is that the interpretation is correct and uses proper controls to detect cells already sensitized with IgG antibodies.
Other Rh Blood Group System Antigens
Antigens in the Rh blood group system other than D, C, E, c, and e are alternative forms or variations produced by the RHD and RHCE genes. These antigens, corresponding antibodies, and clinical relevance are outlined in the following section. Table 6.8 provides a summary of less common antigens and antibody characteristics. Scan the QR code for a quiz on the blood group system.
Compound Antigens
Examples of compound antigens in the Rh blood group system include the following:
• Rh6 (ce or f)
• Rh7 (Ce)
• Rh27 (cE)
• Rh22 (CE)
A compound antigen is the additional antigen product formed when certain genes code for an additional protein. For example, when c and e are inherited as RHce, another epitope called “f” is expressed in addition to the c and e antigens. This epitope can also elicit its own immune response. The f antigen would not be present on the red cell if the person’s genotype was DCe/DcE even though the red cells would type positive for both the c and the e antigens. In this case the c and e alleles were inherited from the RHCe and RHcE genes, and f would not be formed. Compound antigens, previously referred to as cis products, indicate that the antigens were coded from a haplotype rather than a single gene coding for a single protein.19 Table 6.9 summarizes the compound antigen combinations.
Antibodies to compound antigens are infrequently encountered. If they were identified, locating antigen-negative units would require the use of common Rh antisera, such as anti-E, anti-C, anti-c, and anti-e. For example, if anti-f were identified, RBC units that are c-negative or e-negative would also be negative for the f antigen. When RBCs are required, units that are negative for one of the antigens creating the compound antigen can be safely transfused.
G Antigen
The G antigen is located on red cells that possess C or D antigens. Red cells that are negative for both the D and the C antigen are negative for the G antigen. Because an antibody to G reacts with red cells that are either D-positive or C-positive, the specificity appears to be anti-D and anti-C. Antibodies to G appear as an anti-D plus an anti-C; however, the specificities cannot be separated. In other words, anti-G antibodies mimic the reactions observed with anti-D and anti-C antibodies. In some cases, a D-negative person may receive D−, C+ red cells and appear to produce anti-C as well as anti-D. The antibody produced in this case was most likely anti-G. Distinguishing anti-G, anti-D, and anti-C antibodies requires adsorption and elution procedures.18 Extensive testing to identify anti-G is not usually necessary. Individuals making anti-G or what appears to be anti-D or anti-C should receive red cells that are negative for both D and C antigens. Rare cells exist that are negative for D and positive for G (rG). G is not a compound antigen; G is present when D or C is inherited. Scan the QR code for more information on G antigen.
Unusual Phenotypes
Unusual phenotypes in the Rh blood group system are rarely encountered in routine blood bank testing. Unusual phenotypes include cells that have diminished or undetectable Rh blood group system antigen expression. Understanding the inheritance patterns and cell characteristics of unusual phenotypes provides insight into the genetics and biochemistry of the system. Null phenotypes, found in many blood group systems, have led to an understanding of the role of the antigen on the red cell. Serologically, null phenotypes have provided the mechanism to categorize blood group systems.
TABLE 6.9
Compound Antigens on Rh Proteins
| Compound Antigen | Rh Protein | Fisher-Race/Wiener Notation |
| ce or f | Rhce | Dce (R0) or ce (r) |
| Ce or Rh7 | RhCe | DCe (R1) or Ce (r′) |
| cE or Rh27 | RhcE | DcE (R2) or cE (r″) |
| CE or Rh22 | RhCE | DCE (Rz) or CE (ry) |
D-Deletion Phenotype
Rare Rh phenotypes demonstrate no reactions when the red cells are tested with anti-E, anti-e, anti-C, or anti-c. Genetic material was deleted or rendered nonfunctional at the RHCE site. Red cells that lack C/c or E/e antigens may demonstrate stronger D antigen activity (see Fig. 6.5). Individuals who have the “D-deletion” phenotype may produce an antibody that reacts as a single specificity (anti-Rh17) or separable specificities such as anti-e and anti-C. An individual who produces anti-Rh17 would require D-deleted RBC units if transfusions are necessary. The D-deletion phenotype is written as -D- or D—.
Rhnull Phenotype
The Rhnull phenotype appears to have no Rh antigens and can be produced from two distinct genetic mechanisms. Cells that type as Rhnull have membrane abnormalities that shorten their survival and cause hemolytic anemia of varying severity.23 Antibodies produced by immunized individuals who lack all Rh antigens may be directed to “total-Rh” (Rh29) or to an individual Rh-antigen specificity. If an anti-Rh29 is detected, Rhnull cells are needed for transfusion. Donations from siblings, autologous donations, and donations from the rare donor registry could be potential sources of compatible RBC units.
The inheritance of the Rhnull phenotype can result from a regulator gene or an amorph gene. A regulator gene, RHAG (Rh associated glycoprotein), is inherited on chromosome 6 and codes for the thirtieth-named blood group system. Although the RHAG blood group system does not carry any Rh system antigens, its presence is essential for the expression of the Rh system antigens. RHAG mutations are associated with the absence of expression of Rh antigens.7 In the regulator type Rhnull, the Rh genes are inherited but are not expressed. The amorph Rhnull phenotype is less well understood. The RHD gene is absent, and there is a lack of expression of the RHCE gene, causing neither protein to be produced.24
Rhmod Phenotype
The Rhmod phenotype is similar to the regulator Rhnull. In this phenotype, red cells lack most of their Rh antigen expression because of the inheritance of a modified RHAG gene. Hemolytic anemia is also a characteristic of this phenotype.
Section 4
Rh Antibodies
General Characteristics
Rh blood group system antibodies are usually made by exposure to Rh antigens through transfusion or pregnancy. Antibodies to Rh blood group system antigens show similar serologic characteristics. Most antibodies are IgG (IgG1) and bind at 37° C; agglutination is observed by the IAT. Enhancement with high-protein, low-ionic-strength saline (LISS), proteolytic enzymes, and polyethylene glycol (PEG) potentiators is useful in identification procedures. Some Rh antibodies may also be IgM (anti-E) or found in individuals who never underwent transfusion or were never pregnant (anti-CW). Stronger reactivity with homozygous antigen expression (dosage) is characteristic of antibodies to C, c, E, and e, although this is not typical of anti-D. Anti-D is typically stronger with R2R2 red cells because these cells have more D antigen sites. Rh antibodies are not associated with complement activation detectable by hemolysis in tube testing or the use of polyspecific AHG reagent.
When an R1R1 individual makes an anti-E, anti-c often may be present, although possibly weak or undetectable. Because of this association, some clinical laboratories provide both c-negative and E-negative blood when anti-E is identified. It is recommended that more sensitive methods to detect anti-c be used when anti-E is present.
Clinical Considerations
Transfusion Reactions
Antibodies to Rh blood group system antigens can cause hemolytic transfusion reactions. Although antibodies often remain detectable for many years, their reactivity in agglutination procedures can decrease to undetectable levels. Exposure to the antigen when the antibody has formed produces a rapid secondary immune response. Antigen-negative RBCs should be transfused if antibodies to Rh blood group system antigens are identified or have been previously noted in the patient’s history. It is important to check previous records of patients who may be transfused for a history of red cell antibodies that may have developed from previous transfusions or pregnancies.
Hemolytic Disease of the Fetus and Newborn
HDFN was initially observed in infants of D-negative women with D-positive mates. First pregnancies were usually unaffected. Infants from subsequent pregnancies were often stillborn or severely anemic and jaundiced. The initial pregnancy stimulated the mother to produce anti-D from the exposure to D-positive cells that occurred during birth when the infant’s and mother’s circulations mixed. Because maternal anti-D antibodies can cross the placenta, fetal red cells in subsequent pregnancies were destroyed by the mother’s antibody. RhIG protects D-negative mothers against the production of anti-D after delivery. Anti-C, anti-c, anti-E, and anti-e are not protected by RhIG and can cause HDFN. An important aspect of prevention of HDFN is antibody screening early in pregnancy and the determination of the D antigen status of mothers to ascertain RhIG candidacy.
Section 5
LW Blood Group System
Relationship to the Rh Blood Group System
The LW blood group system is presented here because of its phenotypic relationship to the Rh blood group system. The antigens and antibodies are similar in serologic properties but are not genetically related. As discussed earlier, the LW antibody, made by guinea pigs and rabbits that were immunized with red cells from rhesus macaque monkeys in early experiments, is similar to the anti-D antibody. Anti-LW reacts strongly with D-positive cells and weakly with D-negative cells. Rhnull cells are negative for LW antigens as well. The theory suggesting a precursor relationship between the Rh blood group system and LW antigens has been discounted, although the membrane biochemistry is still being studied.26 A summary of LW system antigens and antibodies appears in Table 6.10.
The LW locus is mapped to chromosome 19. The LW system alleles are LWa, LWb, and LW. LW(a+b−) is the most common phenotype in the population because the LWa gene is of high frequency. The LW gene is an amorph, and inheriting two LW genes produces the rare LW(a−b−) phenotype. Antibodies to the LW system are clinically significant and rare.
Chapter Summary
The major concepts of the Rh blood group system regarding inheritance theories, nomenclature, antigens, and antibodies are summarized in the following table.
| Summary of Rh Blood Group System Antigens | |
| Biochemical Composition | Polypeptides with no carbohydrate residues |
| Gene Products | 416 amino acids that traverse the membrane 12 times |
| Current Genetic Theory | Two genes, RHD and RHCE; alleles include RHCE, RHCe, RHcE, and RHce |
| Fisher-Race Theory | Three genes; alleles include D/d, C/c, and E/e |
| Wiener Theory | One gene; alleles include R0, R1, R2, Rz, r, r′, r″, and ry |
| Rosenfield/Numeric Terminology | Numeric D = Rh1, C = Rh2, E = Rh3, c = Rh4, e = Rh5 |
| ISBT: Standard Numeric | Rh blood group system is 004; each antigen has a number as shown in Rosenfield system |
| Genetic Loci | Chromosome 1 |
| Weak D | D antigen negative or agglutinated with some anti-D on immediate-spin and detected by IAT |
| Compound Antigens | ce or f, Ce or Rh7, CE or Rh22, and cE or Rh27 |
| G Antigen | Present whenever the D or C antigen is on the red blood cell |
| Rhnull Phenotype | Results from an amorph gene or RHAG regulator gene; no Rh antigens, abnormal red blood cell membrane |
| Summary of Rh Blood Group System Antibodies | |
| Antibody Production | Red blood cell stimulation through transfusion or pregnancy |
| Immunoglobulin Class | IgG; usually IgG1 and IgG3 |
| In vitro Reactions | Binds at 37° C; agglutination observed using IAT |
| Enhancement | LISS, proteolytic enzymes, albumin, and PEG |
| Complement Binding | No |
| Clinical Significance | Yes; can cause delayed transfusion reactions and HDFN |
| Dosage | Yes; stronger reactions with homozygous expression |

Critical Thinking Exercises
Exercise 6.1
A transfusion recipient from an outside facility needs four units of R2R2 RBCs for transfusion. From this request, determine the following:
1. What Rh blood group system antigens are present on R2R2 RBCs?
2. What Rh blood group system antigens are not present on R2R2 RBCs?
3. What is the frequency of finding compatible R2R2 RBC units?
4. Write R2R2 donor units in Fisher-Race and Rosenfield terminology.
Exercise 6.2
The following reactions were obtained by testing red cells from a male donor with Rh blood group system antisera:
| Rh Antisera | Donor RBC Reaction |
| Anti-D | + |
| Anti-C | + |
| Anti-E | 0 |
| Table Continued | |
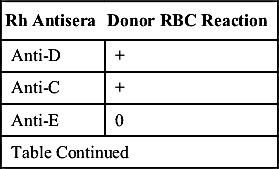
| Rh Antisera | Donor RBC Reaction |
| Anti-c | 0 |
| Anti-e | + |
| Rh control | 0 |
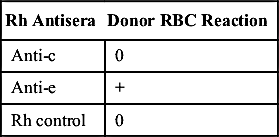
1. What is the Rh phenotype?
2. Determine the most probable Rh genotype if the donor is white.
3. If the donor were black, would the most probable Rh genotype be different in this case?
4. What Rh antibodies could this donor make after transfusion?
5. Is this Rh phenotype rare or common?
Exercise 6.3
A 65-year-old patient with cancer was tested. The following table shows the results.
1. Is the interpretation of the patient’s blood type correct?
2. What test is recommended to solve the Rh typing discrepancy?
3. Should a weak D test be performed if the patient has a positive direct antiglobulin test?
4. If the patient needs a transfusion before the resolution of the discrepancy, what blood type should this patient receive?
Exercise 6.4
The following results were obtained from a first-time blood donor:
1. Interpret the blood donor’s ABO and D phenotyping.
2. Discuss the validity of the testing.
3. Why is it unnecessary to perform an Rh control with the immediate-spin anti-D test?
4. If the transfusion service did not perform weak D testing on recipients, what type of blood would this patient receive for an RBC transfusion?
Exercise 6.5
A 25-year-old man received five units of group O, D-negative RBCs in the emergency room after a serious car accident. His blood type, which was determined from a sample collected before transfusion, was group O, D-negative. A sample was resubmitted 2 weeks after the accident for pretransfusion testing before orthopedic surgery. The antibody screen was positive, and the antibodies identified were anti-D and anti-C.
1. What are possible explanations for the antibodies identified?
2. What additional testing should be performed to explain the problem?
3. If the blood type of the units he received was correct, what is the probable Rh phenotype of the units that he received?
4. What antigen or antigens should be negative if he needs RBC transfusions in the future?
5. Based on your knowledge of Rh antigen frequency, will it be difficult to obtain these units?
Exercise 6.6
Based on your knowledge about factors relating to immunogenicity, why are the Rh antigens (especially D antigen) considered highly immunogenic?