4
Pharmacology of Vasoconstrictors
Demetra Daskalos Logothetis, RDH, MS
1. Discuss the problems associated with the vasodilatory properties of local anesthetics.
2. Discuss the benefits of adding vasoconstrictors to local anesthetic solutions.
3. Name the two vasoconstrictors that are added to local anesthetics available in the United States.
4. Discuss the use of vasoconstrictors in dentistry.
5. Discuss epinephrine, including:
• Its mechanism of action
• Epinephrine dilutions
• Sodium bisulfate preservative
• The actions of epinephrine on specific systems and tissue
• Termination of action
• Maximum recommended dose
6. Discuss levonordefrin, including:
• Actions on specific systems and tissue
• Levonordefrin dilution
• Sodium bisulfate preservative
• Termination of action
• Maximum recommended dose
7. Discuss the effects, mechanisms of action, and uses of norepinephrine, phenylephrine, and felypressin.
8. Discuss the side effects and overdose of vasoconstrictors.
Introduction
Local anesthetics are vasodilators with the ester procaine having the most vasodilatory properties, compared with the amides mepivacaine and prilocaine that have the least. No matter how readily the anesthetic can penetrate the nerve and bind to the receptor sites, the local blood vessels in the area of injection will immediately begin to absorb the anesthetic by causing vasodilation of the blood vessels, leading to increased blood flow to the site of injection causing:
• An increased rate of anesthetic absorption into the bloodstream by carrying the anesthetic away from the injection site.
• A decrease in the duration of the anesthetic’s action by diffusing quickly from the site of administration.
• Higher blood levels of local anesthetics, increasing the risk of systemic toxicity.
• Increased bleeding in the area due to the increase in bloodflow.1
Vasoconstrictors are combined with local anesthetics to counteract the vasodilating properties of local anesthetics. Simply stated, vasoconstrictor drugs work by contracting the smooth muscle in blood vessels, which causes the vessels to constrict. Vasoconstrictors are important additives to the local anesthetic solution because of their ability to constrict blood vessels, thus providing the following beneficial effects:
• A decrease in the blood flow by constricting the blood vessels in the area of anesthetic administration, and the amount of anesthetic needed to produce profound anesthesia.
• An increased duration of the anesthetic’s effect by localizing the high concentration of the drug in the area of injection, within the nerve, improving the success rate and intensity of the nerve block. Using lidocaine 2% as an example, the duration of pulpal anesthesia in a plain solution (without a vasoconstrictor) is approximately 5 to 10 minutes, the duration of action dramatically increases approximately six times when a vasoconstrictor is added to 60 minutes of pulpal anesthesia.
• Slowing absorption of local anesthetic into the cardiovascular system (CVS), resulting in lower drug levels in the blood, reducing the probability of systemic toxicity. The anesthetic metabolism is able to keep pace with drug absorption, providing hemostasis at the injection site, which is particularly useful in areas of heavy bleeding. Deep scaling procedures performed by the dental hygienist involve soft tissue manipulation resulting in hemorrhage, especially with severely inflamed tissues. These procedures generally require the patient to be anesthetized, and vasoconstrictors added to the anesthetic solution counteract unwanted bleeding caused by local anesthetic drugs.
Chemistry
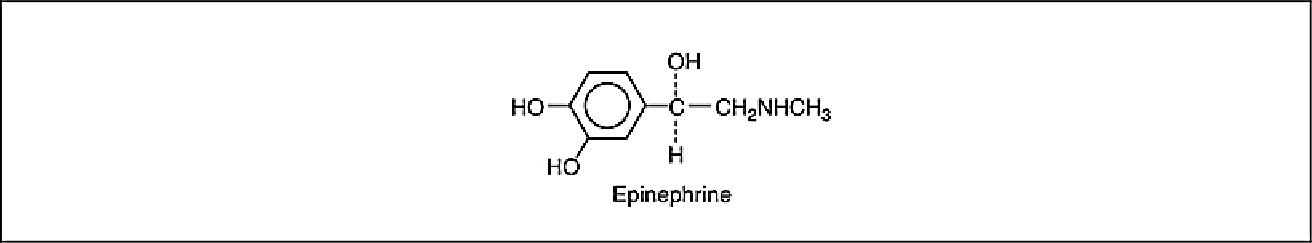
There are two vasoconstrictors that are added to local anesthetic drugs available in the United States, epinephrine and levonordefrin. Epinephrine is the most commonly used vasoconstrictor in dental local anesthetics and is referred to as the benchmark. Vasoconstrictors used in dentistry are structurally identical with the natural, nonsteroid mediators of the sympathetic nervous system (see Chapter 2), epinephrine and norepinephrine, which are secreted by the adrenal medulla (Figure 4-1). Because these drugs mimic similar effects as those caused by stimulation of the adrenergic nerves, they are referred to as sympathomimetic or adrenergic drugs. The term catecholamine is also appropriate for these agents because they are naturally occurring catecholamines of the sympathetic nervous system and have a distinct structure of a benzene ring with two hydroxyl groups. Epinephrine and norepinephrine are naturally occurring catecholamines of the sympathetic nervous system, and levonordefrin is a synthetic catecholamine. They increase heart rate, contract blood vessels, dilate air passage, and participate in the “fight-or-flight” response of the sympathetic nervous system.
The Use of Vasoconstrictors in Dentistry
When a vasoconstrictor is not added to the local anesthetic solution, the drug is quickly removed from the injection site into the systemic circulation, increasing the possibility of systemic toxicity and decreasing the duration of action. As discussed previously, vasoconstrictors added to local anesthetic solutions have several potentially beneficial effects that counteract these undesirable properties of local anesthetics. However, epinephrine, which is the most widely used vasoconstrictor in dentistry, is not an ideal drug. Exogenous epinephrine is absorbed from the site of injection into the circulation just like local anesthetics, and measurable levels of epinephrine in the blood can affect the heart and blood vessels by causing the sympathomimetic “fight-or-flight” response of apprehension, increased heart rate, palpitations and sweating.1 This has caused continued debate regarding the harmful influences of vasoconstrictors in some situations.1,2 The benefits of vasoconstrictor use should be carefully weighed against the risks for patients who are medically compromised by severe cardiovascular disease, high blood pressure, or hyperthyroidism. However, completely avoiding the use of vasoconstrictors in these patients can cause a lack of profound anesthesia leading to pain during the dental procedure, and subsequently stimulating a significant release of endogenous epinephrine from the adrenal gland. This endogenous release of epinephrine in response to inadequate anesthesia during the dental appointment can be much greater than the amount that reaches the circulation from an injection of anesthetic with vasoconstrictor. Figure 4-2 illustrates the blood levels of endogenous epinephrine during rest and during mild-to-severe stress. Blood levels of endogenous epinephrine release are higher than the maximum recommended dose of epinephrine, 0.04 mg per appointment, for a cardiovascularly compromised patient. (See Chapter 8 for dosing calculations of epinephrine.)
Another important consideration is the endogenous release of epinephrine by the adrenal gland for healthy patients experiencing anxiety and stress before and during the dental appointment. As shown in Figure 4-2, endogenous release of epinephrine in minor to moderate stress is increased for particularly anxious patients. This can compound the adverse effects of exogenous administration of epinephrine via the anesthetic injection containing epinephrine (Figure 4-3). Fortunately, the undesirable systemic effects of epinephrine are short-lived. The rapid inactivation of epinephrine by the reuptake of adrenergic nerves quickly reduces the vasoconstrictor’s harmful effects. According to Sisk, when epinephrine is administered intravenously, it has a half-life of 1 to 3 minutes.4
Patients with a recent myocardial infarction, coronary bypass surgery, or cerebrovascular accident within the past 6 months, and those with uncontrolled hypertension, angina, arrhythmias, or hyperthyroidism should not be given an anesthetic with a vasoconstrictor until their medical condition is controlled. Patients with a relative contraindication for vasoconstrictors can receive epinephrine-containing local anesthetic agents in the lowest possible dose, not to exceed the maximum recommended dose of 0.04 mg per appointment, using the best technique, which includes aspiration to reduce the risk of intravascular injection and injecting slowly to reduce the possibility of rapid systemic absorption. Depending upon the severity of the condition, the clinician must determine if a reduced amount of vasoconstrictor should be administered or if no vasoconstrictor should be administered at all. In general, there are only a few absolute contraindications to the use of a vasoconstrictor, and in most situations limiting the amount of vasoconstrictor a patient can receive produces the benefits of vasoconstrictor use in local anesthetics without compromising the patient. Absolute and relative contraindications to the use of vasoconstrictors are discussed in Chapter 7.
Epinephrine (Adrenalin)
As previously discussed, epinephrine is a naturally occurring catecholamine secreted by the adrenal medulla, consisting of approximately 80% of its secretions. It is also available as a synthetic catecholamine, which is identical in structure with the natural hormone epinephrine. Epinephrine is the most widely used vasoconstrictor in dentistry and the most potent. Epinephrine is the standard by which all other vasoconstrictors are measured (Table 4-1).1
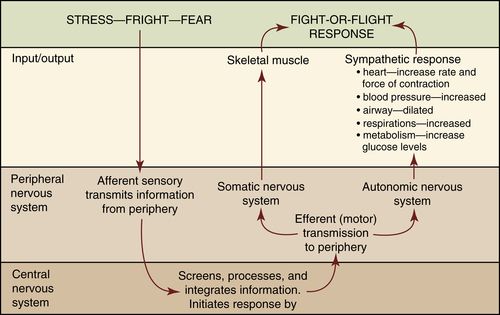
FIGURE 4-3 Nervous system response during “fight-or-flight” or stress. (From McKenry L, Tessier E, Hogan M: Mosby’s pharmacology in nursing, ed 22, St Louis, 2006, Mosby.)
TABLE 4-1
Epinephrine (Adrenalin) Dilutions and Uses in Dentistry
Concentration (Dilutions) | Anesthetic Preparations | Uses |
| 1:1000 | Epinephrine alone | Emergency treatment of anaphylaxis and acute asthma attacks |
| 1:50,000 | 2% lidocaine | Most concentrated (least diluted) Provides greatest hemostasis Provides similar pain control as other anesthetic preparations containing epinephrine |
| 1:100,000 | 2% lidocaine 4% articaine | Provides hemostasis but to a lesser degree than 1:50,000 dilution Provides similar pain control as other anesthetic preparations Most commonly used dilution |
| 1:200,000 | 4% articaine 4% prilocaine 0.5% bupivacaine | Least concentrated (most diluted) Provides hemostasis but to a lesser degree than 1:100,000 and 1:50,000 dilutions Provides similar pain control as other anesthetic/vasoconstrictor preparations Good alternative for patients with significant cardiovascular disease Good alternative for elderly patients sensitive to epinephrine |
Mechanism of Action
Epinephrine and norepinephrine cause vasoconstriction by activating adrenergic receptors located in most tissues. According to Ahlquist2 in 1948, these targeted receptor sites are divided into two major groups of adrenergic receptors, alpha (α) and beta (β), with several subtypes. Ahlquist recognized that α receptors have excitatory actions, and β receptors have inhibitory actions from catecholamines on smooth muscles. The excitatory action of α receptors by sympathomimetic drugs causes vasoconstriction of the smooth muscle in blood vessels. These α receptors have been further subcategorized into α1 and α2 depending upon differences in their location and function, α1 receptors are excitatory-postsynaptic and α2 receptors are inhibitory-postsynaptic.1,2 The inhibitory action of β receptors by sympathomimetic drugs causes smooth muscle relaxation (vasodilation and bronchodilation), and cardiac stimulation. The β receptors have also been further subcategorized into β1 found in the heart (causing cardiac stimulation) and small intestines (causing lipolysis), and β2 found in the bronchi, vascular beds, and uterus causing bronchodilation and vasodilation.1,3
Norepinephrine activates predominantly α receptors, and epinephrine activates both α and β receptors, causing vasoconstriction and vasodilation respectively. α Receptors are less sensitive to epinephrine.3 However, once the α receptor is stimulated by high levels of epinephrine, this activation will override the vasodilation caused by β receptors and subsequently will cause vasoconstriction of the smooth muscle in peripheral arterioles and veins. This is the main reason sympathomimetic agents are added to local anesthetic solutions.
Stimulation of α receptors by adrenergic drugs such as epinephrine causes constriction of the smooth muscle in the blood vessels, also referred to as vasoconstriction. β2 Receptor activation by epinephrine relaxes bronchial smooth muscles, causing the bronchi of the lungs to dilate. In addition, β1 receptors have stimulatory effects that increase the rate and force of heart contractions. These stimulatory effects on β1 receptors are undesirable side effects of incorporating sympathomimetic agents such as epinephrine into local anesthetic solutions. This is of particular concern in patients with preexisting cardiovascular and thyroid disease. The risks of adding the vasoconstrictor to the local anesthetic must be weighed against the benefits, and decreasing the amount of the drug administered should be considered (discussed next). Table 4-2 lists the major systemic effects of injected sympathomimetic agents involved in the cardiovascular and respiratory systems.1,3
Epinephrine Dilutions
The concentration of vasoconstrictors in local anesthetic solutions is referred to as a ratio rather than a percentage as expressed by the local anesthetic drug. For example, a concentration of 1:100,000 means there is 1 g (or 1000 mg) of drug contained (dissolved) in a 100,000 mL solution, or 0.01 mg/mL. The most common dilutions of epinephrine combined with local anesthetics are 1:50,000 (0.02 mg/mL), 1:100,000 (0.01 mg/mL), and 1:200,000 (0.005 mg/mL). The 1:50,000 dilution is manufactured in combination with 2% lidocaine, the 1:100,000 dilution is manufactured in combination with 2% lidocaine and 4% articaine, and the 1:200,000 dilution is manufactured in combination with 4% prilocaine, 4% articaine, and 0.5% bupivacaine. Therefore, in a typical dental anesthetic cartridge containing both a local anesthetic drug and vasoconstrictor, the label will identify both drugs (Figure 4-4). Using prilocaine with epinephrine as an example, the cartridge may contain 4% prilocaine (referred to as a percentage) with 1:200,000 epinephrine (referred to as a ratio) (Figure 4-4). Table 4-3 lists the vasoconstrictor dilutions combined with dental local anesthetics used in the United States and Canada.
TABLE 4-2
Systemic Effects of Adrenergic Amines on the Cardiovascular and Respiratory Systems
| Cardiovascular System | Receptor Affected | Response |
| Heart rate | β1, β2 | Increased (may be blocked or reversed by compensatory vagal reflex activity) |
| Contractile force | β1, β2 | Increased |
| Coronary arterioles | α1, α2, β2 | Constriction/dilation (local regulatory processes largely govern blood flow) |
| Conduction velocity | β1, β2 | Increased (may be blocked or reversed by compensatory vagal reflex activity) |
| Peripheral resistance | α1, α2, β2 | Increased/decreased |
| Respiratory System | Receptor Affected | Response |
| Bronchial smooth muscle | β2 | Relaxation |
| Bronchial glands | α1, β2 | Decreased/increased |
| Pulmonary arterioles | α1, β2 | Constriction/dilation |
From Jastak, JT, Yagiela JA, Donaldson D: Local anesthesia of the oral cavity, St Louis, 1995, Saunders.
The 1:50,000 dilution represents the highest concentration, and 1:200,000 dilution represents the lowest concentration and therefore produces fewer side effects.1 Concentrations greater than 1:200,000 offer no advantage in prolonging the duration of anesthesia, reducing the plasma levels, or advancing pain control. Therefore, because a 1:50,000 offers no added benefits to most clinical situations and can produce more profound undesired sympathomimetic actions (fight or flight), there is questionable rationale for using a 1:50,000 dilution of epinephrine for pain control. However, higher concentrations of epinephrine, specifically the 1:50,000 dilution are more effective for bleeding control (hemostasis). Local anesthetics with vasoconstrictors may be infiltrated for hemostasis even when pulpal anesthesia has been obtained. Therefore, using a 1:100,000 dilution of epinephrine for obtaining pulpal anesthesia may be used in combination with a relatively small infiltrated dose of 1:50,000 dilution of epinephrine into the papilla or gingival margin to decrease bleeding to less than half of that recorded from a similar volume of 1:100,000 (Figure 4-5). This is particularly important for bleeding control during periodontal surgeries and for dental hygienists providing nonsurgical periodontal therapy.5,6 (See Chapters 12 and 13.)
The selection of an appropriate vasoconstrictor dilution, if any, should be determined considering several factors such as the length of the dental procedure, medical status of the patient, and the need for hemostasis.1 As discussed earlier, the addition of a vasoconstrictor to an anesthetic such as lidocaine can dramatically increase the pulpal anesthesia, and the clinical effectiveness of lidocaine. The medical status of the patient must be reviewed and considered when selecting a vasoconstrictor. In general, for patients with significant cardiovascular diseases ASA III and IV, patients with hyperthyroidism, sulfite allergies, and patients taking certain medications, the severity of each of these conditions must be evaluated to determine the appropriateness of a selected vasoconstrictor. The medical status of the patient in relationship to the use of vasoconstrictors will be covered thoroughly in Chapter 7. Finally, the need for hemostasis should be considered. Epinephrine is effective in decreasing blood flow during surgical procedures and nonsurgical periodontal therapy. Because epinephrine possesses both α and β actions with α receptors being less sensitive to epinephrine than β receptors, higher doses of epinephrine are needed to produce the vasoconstriction action of α receptors. Although all epinephrine dilutions provide bleeding control, a 1:50,000 dilution (being double the concentration of 1:100,000, and four times the concentration of 1:200,000) provides the most rigorous bleeding control when compared with the 1:100,000 and 1:200,000 dilutions. However, an important consideration is that as the tissue level of epinephrine begins to decline, it produces a rebound vasodilatory effect when the β2 action begins to predominate. This vasodilatory action can potentially lead to postoperative bleeding approximately 6 hours after the procedure.1
Sodium Bisulfite Preservative
Synthetic epinephrine is not very stable and must include the addition of an acidic preservative to stabilize the solution to prevent oxidation of epinephrine. Because a local anesthetic is manufactured as an acid salt, the drug is highly soluble in water and acidic, pH 5 to 6. The addition of the preservative sodium bisulfite provides a shelf life of approximately 18 months because of its antioxidant properties.1 However, there are disadvantages to the presence of this preservative. First, sodium bisulfite can further acidify the pH of the anesthetic solution to the range of 3.3 to 5.5, thus reducing the efficiency of the quaternary amine to dissociate (once injected) into the uncharged tertiary amine base necessary to penetrate the lipid-rich membrane of the nerve, and slightly slow the onset of action of the local anesthetic. This is of particular concern in an acidic tissue environment associated with an active infection (see Chapter 3). Second, according to the FDA, sulfites associated with vasoconstrictors may cause allergic-type reactions in certain susceptible persons, especially asthmatics. Patients who have a true allergy to sodium bisulfite should not receive an anesthetic/vasoconstrictor combination. However, for sulfite-containing epinephrine for injection for use in allergic emergency, the FDA states “epinephrine is the preferred treatment for serious allergic or other emergency situations even though the product contains a sulfite that may in other products cause allergic-type reactions including anaphylactic symptoms or life-threatening or less severe asthmatic episodes in susceptible persons. The alternative to using epinephrine in a life-threatening situation may not be satisfactory. The presence of a sulfite(s) in the product should not deter administration of the drug for treatment of serious allergic or other emergency situations” (CFR – Code of Federal Regulations Title 21). Cartridges that do not contain a vasoconstrictor, such as 3% mepivacaine and 4% prilocaine, do not contain sodium bisulfite and are therefore safe to be administered to patients with sulfite allergies. Because mepivacaine and prilocaine produce only minor vasodilation compared with other available anesthetics, they can still produce adequate pulpal anesthesia for short dental appointments when administering nerve blocks (see Chapter 5). People often confuse sulfites with sulfa medications; although both can cause allergic reactions in people, they are chemically unrelated.
Actions of Epinephrine on Specific Systems and Tissue
Mode of action: Epinephrine exerts its action directly on the adrenergic receptors including both α and β receptors, affecting β receptors predominantly.
Myocardium: The pharmacologic effect of epinephrine is essentially a result of its direct effect as agonist on specific α and β receptors (β1 and β2). Epinephrine increases heart rate, stroke volume, and cardiac output by stimulating β1 receptors.
Pacemaker cells: Epinephrine stimulates β1 receptors, increasing the incidence of dysrhythmias.
Coronary arteries: Epinephrine increases coronary artery flow by dilating coronary arteries.
Blood pressure: Small doses of epinephrine increase systolic pressure to a greater extent than the diastolic pressure (diastolic pressure may decrease). Higher doses of epinephrine increase diastolic pressure.
Cardiovascular system: The β1 effects of epinephrine have direct stimulation on the cardiovascular system, which leads to an overall decrease in cardiac efficiency.
Vasculature: Epinephrine constricts the α receptors that are contained in the skin and mucous membranes. The effects of epinephrine on the blood vessels of the skeletal muscles, which contain both α and β2 receptors, are dose dependent due to the predominance of β2 receptors, which are more sensitive to epinephrine than α receptors. Therefore smaller doses are affected by β2 actions and produce vasodilation, and larger doses are affected by α actions and produce vasoconstriction.
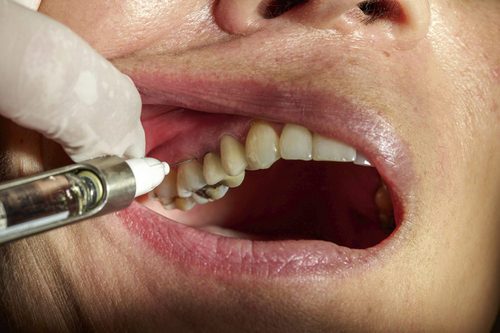
FIGURE 4-5 Small amounts of lidocaine 1:50,000 epinephrine can be infiltrated into the papilla to decrease bleeding during nonsurgical periodontal therapy after pulpal anesthesia has been obtained using a lower concentration of epinephrine such as lidocaine 1:100,000 epinephrine.
Metabolic system: Epinephrine inhibits insulin secretion, causing a rise in blood sugar and an increase in free fatty acids.
This is of particular concern in brittle diabetics taking large doses of insulin, especially if, in addition, these individuals have cardiovascular disease. Inconsistent blood sugar levels ranging from severe hypoglycemia and hyperglycemia may result. Well-controlled diabetics may receive epinephrine without precautions.
Respiratory system: Epinephrine is a potent bronchial dilator due to the β2 receptor effects. It is an invaluable drug for treating acute asthmatic attacks and anaphylactic reactions.
Central nervous system (CNS): In normal therapeutic doses, epinephrine does not stimulate the CNS. Overdose of epinephrine produces signs and symptoms of CNS stimulation, which may include anxiety, nausea, restlessness, weakness, tremor, headache, and hyperventilation.
Termination of Action
The absorption of epinephrine is retarded because of the drug’s vasoconstricting properties. It may take several hours for absorption to be completed. The effects of epinephrine following inadvertent intravascular injection becomes apparent within 1 minute and because of the body’s efficiency at removing catecholamines, the effects of epinephrine in the blood last only 5 to 10 minutes. Once absorbed, the action of epinephrine is terminated primarily by the reuptake action of adrenergic nerves, any epinephrine that escapes the reuptake action is inactivated by enzymes catechol-O-methyltransferase (COMT), and monoamine oxidase (MAO) in the blood.1,2 Only 1% of epinephrine is excreted unchanged in urine. Figure 4-6 illustrates the distribution and fate of catecholamines injected into peripheral tissues.
Maximum Recommended Dose
The American Heart Association7 and the New York Heart Association8 indicate that the lowest possible effective dose should be used when administering local anesthetics with epinephrine to either healthy or medically compromised patients. Proper aspiration on several planes and slow injection should also be done when administering local anesthetics with epinephrine. The maximum recommended dose per visit of epinephrine for a healthy patient is 0.2 mg. The maximum recommended dose per visit of epinephrine for a cardiovascularly compromised patient or a patient needing treatment modifications (i.e., a patient taking a tricyclic antidepressant) is 0.04 mg. See Chapter 7 for medical history considerations when administering epinephrine. Maximum recommended doses for vasoconstrictors and local anesthetics, as well as calculation methods, are discussed in Chapter 8.
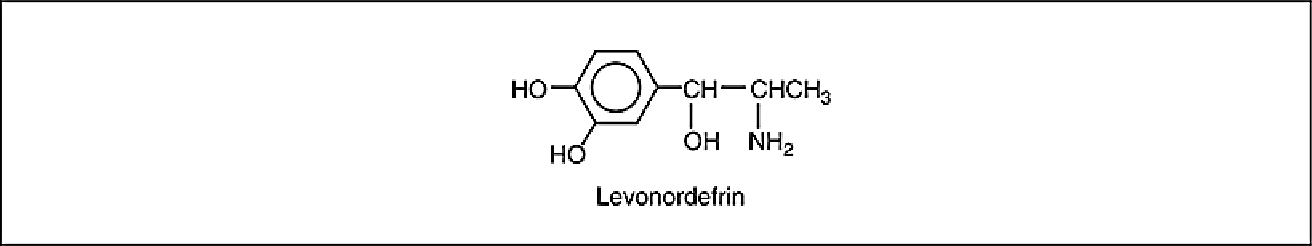
Levonordefrin (Neo-Cobefrin)
Levonordefrin is a synthetic vasoconstrictor that is approximately one sixth (15%) as potent as epinephrine; it is manufactured in a higher concentration to achieve the same effects as epinephrine 1:100,000.2 Levonordefrin is available in dentistry only with 2% mepivacaine, in a 1:20,000 dilution, which is 5 times greater than epinephrine in a dilution of 1:100,000, but because it is one sixth as potent as epinephrine, a 1:20,000 dilution of levonordefrin produces the same clinical effects as the epinephrine 1:100,000 dilution. Like epinephrine, levonordefrin contains the preservative sodium bisulfite to delay its deterioration (Table 4-4).
Actions of Levonordefrin on Specific Systems and Tissue
Mode of action: Levonordefrin is a selective α2 agonist (75%) and produces vasoconstriction in low systemic concentrations; it has much less β activity (only 25%) compared with epinephrine that has 50% α and 50% β activity.1
Myocardium: The pharmacologic effect of levonordefrin on the myocardium is essentially the same as epinephrine by increasing cardiac output and heart rate, but to a lesser degree.
Pacemaker cells: Same as epinephrine by increasing dysrhythmias, but to a lesser degree.
Coronary arteries: Increases coronary artery flow by dilating coronary arteries similar to epinephrine, but to a lesser degree.
Blood pressure: Increases systolic pressure to a greater extent than the diastolic (diastolic pressure may decrease). Higher doses of levonordefrin increase diastolic pressure.
TABLE 4-4
Levonordefrin (Neo-Cobefrin) Dilution and Uses in Dentistry
Concentration (Dilution) | Anesthetic Preparations | Uses |
| 1:20,000 | 2% mepivacaine | Similar pain control as epinephrine dilutions Hemostasis is less effective than epinephrine dilutions |
Vasculature: Effects on the vasculature are similar to epinephrine by providing α constriction of the skin and mucous membranes, but to a lesser degree.
Metabolic system: Levonordefrin inhibits insulin secretion, causing a rise in blood sugar and an increase in free fatty acids similar to epinephrine, but to a lesser degree.
Respiratory system: Levonordefrin provides some bronchodilation but significantly less than epinephrine.
Central nervous system (CNS): In normal therapeutic doses, levonordefrin does not stimulate the CNS. In an overdose, it is not a potent stimulant when compared with epinephrine.
Hemostasis: Levonordefrin provides hemostasis, but significantly less effective than epinephrine.
Termination of Action
Levonordefrin is terminated by reuptake by adrenergic nerves and escaped levonordefrin is inactivated by COMT. Levonordefrin is not terminated by MAO.2
Maximum Recommended Dose
The maximum recommended dose per visit of levonordefrin for a healthy patient is 1.0 mg. The maximum recommended dose per visit of levonordefrin for a cardiovascularly compromised patient or a patient needing treatment modifications is 0.2 mg. Some sources indicate that a cardiac dose is not necessary for levonordefrin, and that for all patients the maximum dose is 1.0 mg or 11.1 cartridges.1 This author and others recommend that the need for a cardiac dose of levonordefrin should be determined on an individual patient basis, utilizing physician consultations if necessary.7,9,10 Levonordefrin should be avoided in patients taking tricyclic antidepressants to avoid hypertensive crises.1,9 See Chapter 7 for medical history considerations when administering levonordefrin, and Chapter 8 for maximum recommended dose and dosing methods.7,9,10
Other Vasocontrictors
Norepinephrine (Levarterenol)
Norepinephrine is a naturally occurring catecholamine. Twenty percent of its production is from the adrenal medulla, but it is also available as a synthetic catecholamine. The usual dilution used in dentistry is 1:30,000. Norepinephrine has the same action as levonordefrin on α receptors. It almost exclusively activates α receptors (90%) and minimally activates β receptors (10%).1 Because of this, norepinephrine produces intense peripheral vasoconstriction with a possible dramatic increase in blood pressure. Therefore its use in dentistry is not recommended. The intense vasoconstriction/hemostasis caused by norepinephrine is likely to produce tissue necrosis, especially on the palate (Figure 4-7).
Phenylephrine (Neo-Synephrine)
Phenylephrine is a synthetic sympathomimetic amine that exerts its action predominantly on α receptors (95%), with very little β effects on the heart. It is a very weak vasoconstrictor (only 5%) as potent as epinephrine and is formulated at much higher concentrations than epinephrine (1:2500 dilution). It is not used in dentistry, but is used in combination with local anesthetics for the management of hypotension and for nasal decongestants and ophthalmic solutions.
Felypressin
Felypressin is a synthetic hormone analogue of vasopressin.1 It is a direct stimulator of vascular smooth muscles.3 Felypressin causes few side effects because it has little or no direct effect on the myocardium or adrenergic nerve transmission. Therefore it may be safely administered to patients with uncontrolled hyperthyroidism, or patients taking tricyclic antidepressants or MAO inhibitors. Felypressin is available in Great Britain and other countries usually in combination with 3% prilocaine. It is favorably compared with epinephrine in anesthesia for restorative dentistry.1,6
Side Effects and Overdose of Vasoconstrictors
Overdose of vasoconstrictors from accidental intravenous injection or by administering more than the maximum recommended dose produces overstimulation of adrenergic receptors that can produce signs and symptoms normally observed from CNS stimulation. Table 4-5 lists the typical overdose responses to epinephrine.
With high plasma levels of epinephrine, dysrhythmias, ventricular fibrillation, dramatic increase in heart rate, and possible cardiac arrest are possible, more so in patients with increased susceptibility to the adverse cardiovascular effects of adrenergic drugs. Because the body is very efficient at removing vasoconstrictors, these adverse effects only last about 5 to 10 minutes. Even so, for patients with severe conditions such as unstable angina, recent myocardial infarction (within 6 months), recent coronary bypass surgery (within 6 months), uncontrolled hypertension, uncontrolled hyperthyroidism, uncontrolled dysrhythmias, or congestive heart failure, the risk of using a vasoconstrictor may outweigh the benefits. The patient’s physician should be consulted before using vasoconstrictors on these patients. These conditions, as well as drug interactions, are discussed in Chapter 7. For all patients, only the minimal effective dose should be administered, not to exceed the maximum recommended dose.1,6 (See Table 4-6 for recommended doses for vasoconstrictors.)
TABLE 4-5
Overdose Responses to Vasoconstrictors
| Tension | Increased heart rate |
| Anxiety | Increased blood pressure |
| Apprehension | Throbbing headache |
| Nervousness | Hyperventilation |
| Tremors |
From Jastak T, Yagiela J, Donaldson D: Local anesthesia of the oral cavity, St Louis, 1995, Saunders.
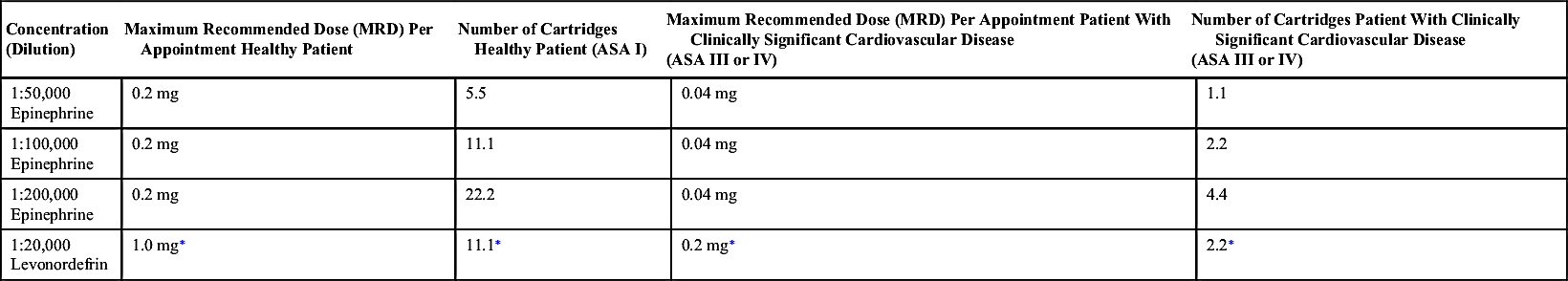
TABLE 4-6
Recommended Maximum Dosages of Vasoconstrictors
Concentration (Dilution) | Maximum Recommended Dose (MRD) Per Appointment Healthy Patient | Number of Cartridges Healthy Patient (ASA I) | Maximum Recommended Dose (MRD) Per Appointment Patient With Clinically Significant Cardiovascular Disease (ASA III or IV) | Number of Cartridges Patient With Clinically Significant Cardiovascular Disease (ASA III or IV) |
1:50,000 Epinephrine | 0.2 mg | 5.5 | 0.04 mg | 1.1 |
1:100,000 Epinephrine | 0.2 mg | 11.1 | 0.04 mg | 2.2 |
1:200,000 Epinephrine | 0.2 mg | 22.2 | 0.04 mg | 4.4 |
1:20,000 Levonordefrin | 1.0 mg∗ | 11.1∗ | 0.2 mg∗ | 2.2∗ |