Nonopioid (Nonnarcotic) Analgesics
Learning objectives
1. Describe pain and its purpose and main components.
2. Discuss the classification of analgesic agents and the chemistry, pharmacokinetics, pharmacologic effects, adverse reactions, toxicity, drug interactions, and uses of aspirin.
3. Define the term nonsteroidal antiinflammatory drug and discuss the chemistry, pharmacokinetics, pharmacologic effects, adverse reactions, toxicity, drug interactions, uses, and several examples of these drugs.
4. Discuss the properties, pharmacologic effects, adverse reactions, drug interactions, uses, and dosing of acetaminophen.
5. Explain the disease known as gout and summarize the drugs used to treat it.
![]() http://evolve.elsevier.com/Haveles/pharmacology
http://evolve.elsevier.com/Haveles/pharmacology
Pain control is of great importance in dental practice. Pain often brings the patient to the dental office. Conversely, pain can be the factor that keeps the patient from seeking dental care at the appropriate time. Thus dental treatment is often rendered on inflamed, hypersensitive tissues of a patient who suffers from mental fatigue after enduring pain for a length of time.
The dental hygienist must be able to recognize and evaluate a patient’s need for medication to control pain. Because pain is such a complex phenomenon, the entire patient must be considered before the type of medication that may be needed is determined.
Pain
The sensation of pain is the means by which the body is made urgently aware of the presence of tissue damage. Pain represents a protective reflex for self-preservation. Just as the hand is quickly removed from a hot object, a painful dental abscess brings the patient to the dental office seeking professional assistance for its resolution. Pain is a diagnostic symptom of an underlying pathologic condition. Although the relief of pain is an immediate objective, only by treatment of the underlying cause is the ultimate resolution achieved.
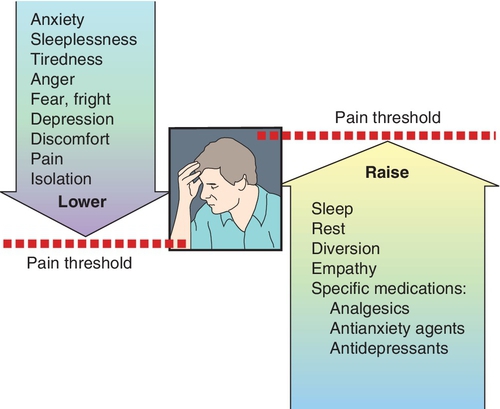
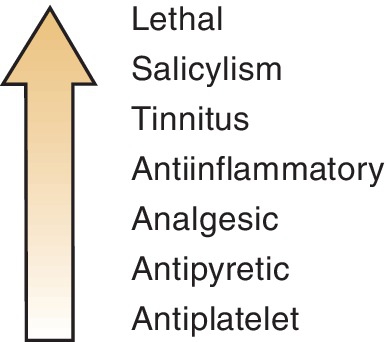
The two components of pain are perception and reaction. Perception, the physical component of pain, involves the message of pain that is carried through the nerves eventually to the cortex. Reaction, the psychological component of pain, involves the patient’s emotional response to the pain. Although individuals are surprisingly uniform in their perception of pain, they vary greatly in their reaction to it. A decrease in the pain threshold (a greater reaction to pain) has been said to be associated with emotional instability, anxiety, fatigue, youth, certain nationalities, female gender, and fear and apprehension. The pain threshold is raised by sleep, sympathy, activities, and analgesics (Figure 5-1). As a result, analgesic therapy must be selected for the individual. A level of discomfort that may not require drug treatment in one person may demand extreme therapy in another. Although some patients undergoing routine exodontia require no postoperative medication, even the strongest analgesics do not completely control postoperative extraction pain in others.
Classification
The analgesic agents can be divided into two groups, the nonopioids, also called nonnarcotic analgesics, and the opioids, also called narcotic analgesics.
An important difference between the nonopioid and the opioid analgesics (narcotic analgesics) is their sites of action. Nonopioid analgesics act primarily at the peripheral nerve endings, although their antipyretic effect is mediated centrally. Opioids act primarily within the central nervous system (CNS).
Another difference between the opioids and the nonopioid analgesic agents is their mechanism of action. The action of the nonopioid analgesic agents is related to their ability to inhibit prostaglandin synthesis (Figure 5-2). The opioids affect the response to pain by depressing the CNS (the reaction). The side effect profiles of the two groups also differ.
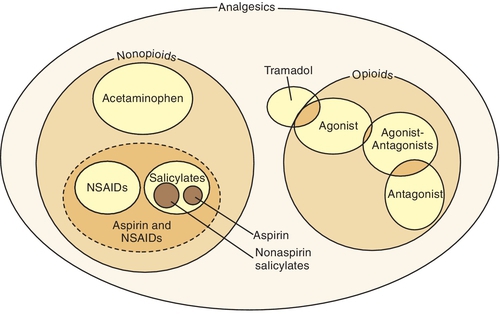
The nonopioids can be divided into the salicylates (aspirin-like group), acetaminophen, and the nonsteroidal antiinflammatory drugs (NSAIDs) (Box 5-1). Aspirin, a member of the salicylates, is discussed first.
Salicylates
Since antiquity, extracts of willow bark containing salicin have been used to reduce fever. Over the years, many other salicylates (sa-LI-si-lates) have been synthesized, but aspirin is the most useful salicylate for analgesia. Box 5-2 lists some analgesic and some topical salicylates. Because aspirin is the prototype salicylate, it is discussed.
Acetylsalicylic Acid
Chemistry
Acetylsalicylic acid (aspirin, ASA) is broken down into acetic acid (HA) and salicylic acid (SA) (Figure 5-3). Acetic acid imparts the characteristic vinegar odor to a bottle of aspirin. Therefore the degree of breakdown of aspirin can be roughly determined by smelling a bottle of aspirin tablets. (If one thinks “phew” when opening an aspirin bottle, it is time to purchase a new bottle.) In addition, salicylic acid is a strong keratolytic agent (used to remove plantar warts from the bottom of feet) and may cause additional adverse gastrointestinal effects if degraded aspirin is administered orally.
Mechanism of Action
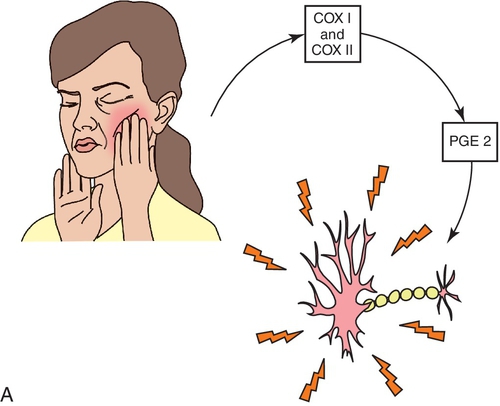
The mechanism of aspirin’s analgesic, antipyretic, antiinflammatory, and antiplatelet effects is related to its ability to inhibit prostaglandin synthesis. Aspirin inhibits the enzyme cyclo-oxygenase (COX I and COX II, prostaglandin synthase) by acetylating serine, which results in inhibition of the production of prostaglandins. Figure 5-4 shows the synthesis of the prostaglandins and leukotrienes from arachidonic acid. Prostaglandins, which are lipids that are synthesized locally by inflammatory stimuli, can sensitize the pain receptors to substances such as bradykinin. Prostaglandins can also lower the pain threshold to painful stimuli, cause inflammation and fever, and affect vascular tone and permeability, resulting in edema.
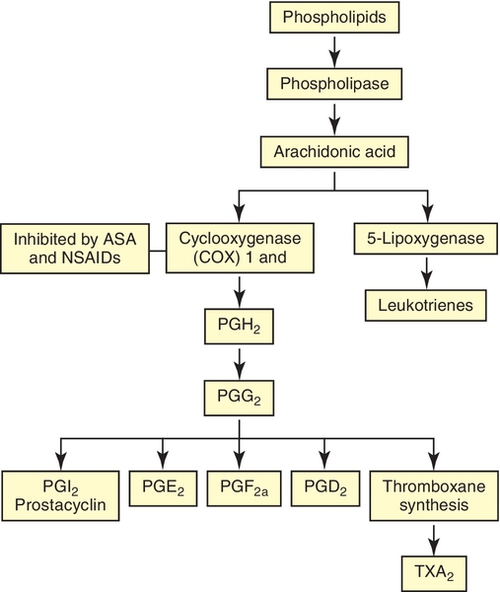
Therefore a reduction in prostaglandins results in a reduction in pain. Because aspirin blocks the synthesis of prostaglandins by nonselectively blocking cyclooxygenase, it is more effective if given before the painful stimuli are experienced. Because of this mechanism, aspirin is more effective against “throbbing” pain (caused by inflammation and common in dentistry) than against “stabbing” pain (direct effect on nerve endings).
Pharmacokinetics
Aspirin is rapidly and almost completely absorbed from the stomach and small intestine, producing its peak effect on an empty stomach in 30 minutes (90 minutes for salicylate). The buffered tablet reaches its peak in about 20 minutes (salicylate). Before a tablet of aspirin can be absorbed, it must be dispersed and dissolved. Addition of a buffer to the tablet facilitates this process. This facilitation is borne out by the somewhat quicker peak of action and higher blood levels attained with buffered aspirin preparations. Buffered aspirin has a higher proportion of the aspirin in the ionized form, which should make absorption slower, but this is offset by the increase in the rate of dissolution, which is facilitated. This difference in absorption has not been shown to translate into a clinically significant quicker effect.
Aspirin is widely distributed into most body tissues and fluids. It is poorly bound to plasma proteins. It is hydrolyzed to salicylate in the mucosa of the gastrointestinal tract and on first pass through the liver. The half-life of unhydrolyzed aspirin is about 15 minutes. The half-life of hydrolyzed aspirin is dose-dependent. With small doses, the half-life is 2-3 hours; with higher doses, a half-life of 15-30 hours can be attained. The half-life varies with the dose because a constant amount rather than a constant percentage of the drug is metabolized per hour. This type of metabolism is called zero-order kinetics (see Chapter 2).
Pharmacologic Effects
Analgesic Effect
Aspirin’s analgesic effect has been repeatedly demonstrated in many clinical trials. In fact, new drugs are often compared in analgesic strength to aspirin. Aspirin typically relieves mild-to-moderate pain such as arthritis, headache or toothache. Aspirin’s effects by dose are illustrated in Figure 5-5.
Because of its easy accessibility and long history of use, aspirin’s worth as an analgesic is often unrecognized by the lay public.
Antipyretic Effect
The ability of aspirin to reduce fever (antipyretic effect) results from its inhibition of prostaglandin synthesis in the hypothalamus. Hypothalamic prostaglandin synthesis is caused by elevated blood values of leukocyte pyrogens induced by inflammation. Increased hypothalamic prostaglandin levels produce higher body temperature. Therefore the inhibition of hypothalamic prostaglandin synthesis results in a return to more normal body temperature. Aspirin reduces fever by inducing peripheral vasodilation and sweating. Although it reduces an elevated temperature, it has no effect on normal body temperature. In fact, in toxic doses aspirin causes hyperthermia (see the section on Adverse Reactions).
Antiinflammatory Effect
Aspirin’s antiinflammatory effect is derived from its ability to inhibit prostaglandin synthesis (COX I and COX II). The prostaglandins are potent vasodilating agents that also increase capillary permeability. Therefore aspirin causes decreased erythema and swelling of the inflamed area. This antiinflammatory action is useful in dental patients because inflammation is a significant part of most dental pain. Patients with arthritis may be given large doses of aspirin to provide symptomatic relief of pain and inflammation in the joints.
Uricosuric Effect
Although large doses (greater than 3 g/day) of aspirin can produce a uricosuric effect, small doses (less than 1 g/day) produce uric acid retention. Aspirin can also counteract the uricosuric effect of probenecid (proe-BEN-e-sid) (Benemid), which is used to treat gout. Aspirin is no longer used as a uricosuric agent because more effective agents are available to treat gout.
Antiplatelet Effect
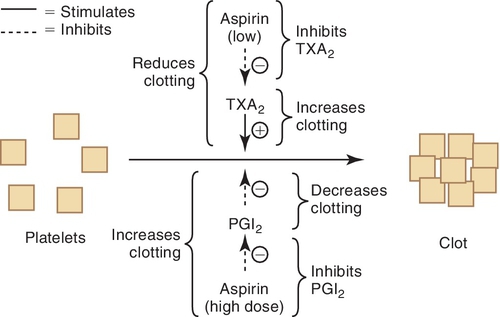
Aspirin irreversibly binds to platelets. Its antiplatelet effect has been shown to be clinically effective for secondary myocardial infarction prevention in adults, the primary prevention of coronary artery disease, and the treatment of an ischemic event or the prevention of a further ischemic event. The effect of aspirin on platelets depends on the dose taken. Depending on the dose, aspirin can inhibit either prostacyclin (inhibits platelet aggregation) or thromboxane A2 (stimulates platelet aggregation). Figure 5-6 demonstrates that inhibition of thromboxane A2 would prevent clotting because thromboxane A2 promotes clotting. Further studies are needed to determine aspirin’s usefulness and dose in preventing clotting events in different patient populations.
Adverse Reactions
In sufficiently high doses, aspirin can have a variety of undesirable effects. Some of aspirin’s side effects can be minimized but not eliminated. Precautions for and contraindications to the administration of aspirin are listed in Table 5-1.
Table 5-1
Precautions for and Contraindications to the Administration of Aspirin
| Disease or Condition | Drug Used | Possible Effects of Aspirin |
| Myocardial infarction, atrial fibrillation, valve replacement | Warfarin (Coumadin) | Increases anticoagulant effect of warfarin |
| Peptic ulcer (heartburn), gastroesophageal reflux disease (GERD) | Histamine H2-blockers (e.g., cimetidine) | Gastric irritant effect |
| Gout | Probenecid (Benemid) | Antagonizes uricosuric effect of probenecid |
| Arthritis, cancer, psoriasis | Methotrexate (MTX) | Increases toxicity of methotrexate |
| Rheumatic fever, arthritis | Large doses of aspirin | Do not add more aspirin if patient is taking large doses already |
| Hemophilia | Factor VIII | Gastric bleeding |
| Hypoprothrombinemia | Bleeding | |
| Vitamin K deficiency, alcoholism | Bleeding | |
| G6PD deficiency | Hemolysis | |
| Diabetes | Oral hypoglycemics | Hypoglycemia |
Gastrointestinal Effects
Aspirin’s most common side effect is related to the gastrointestinal tract. It may be simple dyspepsia, nausea, vomiting, or gastric bleeding. These adverse effects result from direct gastric irritation and inhibition of prostaglandins. Because prostaglandins are responsible for inhibition of gastric acid secretion and stimulation of the cytoprotective mucus in the stomach, aspirin counteracts these effects. In high doses, aspirin’s stimulation of the chemoreceptor trigger zone in the CNS can also produce nausea and vomiting. Salicylates may exacerbate preexisting ulcers, gastritis, or hiatal hernia.
Bleeding
At usual therapeutic doses, aspirin irreversibly interferes with the clotting mechanism by reducing platelet adhesiveness caused by interfering with adenosine diphosphate (ADP) release. The bleeding time is prolonged, and each platelet is affected until new platelets are formed (4-7 days). Replacement of all of the affected platelets is not required to produce normal clotting. After about 20% of the platelets have been replaced with newly formed platelets, clotting will return to normal by about 36 hours. Therefore with lower doses of aspirin, 1.5 days should be enough to obtain normal clotting. With large doses of aspirin, the half-life is prolonged. Aspirin inhibits the production of prothrombin, resulting in hypoprothrombinemia. Three other mechanisms—the local irritant effects on the stomach, the decrease in platelet stickiness, and the loss of protective mucosa—magnify adverse effects on the stomach. Salicylate-induced gastric bleeding is painless. With a small loss of blood, aspirin does not produce significant bleeding. Salicylates may exacerbate preexisting conditions such as ulcers, gastritis, hiatal hernia, and gastrointestinal esophageal reflux disease (GERD).
Reye’s Syndrome
In children and adolescents with either chickenpox or influenza, the use of aspirin has been epidemiologically associated with Reye’s (pronounced “rize”) syndrome. In place of aspirin, acetaminophen is used in pediatrics for both its analgesic and its antipyretic actions. Reye’s syndrome is associated with hepatotoxicity and encephalopathy, commonly fatal.
Hepatic and Renal Effects
Rarely, aspirin can produce hepatotoxicity. Renal papillary necrosis and interstitial nephritis leading to dialysis is associated with use of certain analgesics. It may be caused by the concomitant administration of aspirin and acetaminophen.
Pregnancy and Nursing Considerations
Although animal studies have shown that aspirin can cause birth defects, human studies have demonstrated only a slight positive correlation between long-term aspirin ingestion and congenital abnormalities. With aspirin abuse, increased risk of stillbirth, neonatal death, and decreased birth weight occur. With near-term, high-dose administration of aspirin, gestation can be prolonged, parturition delayed, and risk of hemorrhage in the newborn and mother increased. Even premature closure of the patent ductus arteriosus (hole in the fetal heart) has been reported. Although salicylates are excreted in the breast milk, usual occasional therapeutic doses of aspirin do not present a problem for the healthy nursing infant.
Hypersensitivity (Allergy)
The incidence of true aspirin allergy is less than 1% (0.2-0.4%). Many patients with “allergy to aspirin” in their charts, on questioning, actually have stomach problems rather than true allergy. In the patient’s chart, it is important to differentiate aspirin’s adverse reactions from its hypersensitivity reactions. Adequate questioning of patients who “claim” to be allergic to aspirin is needed; patients with true aspirin hypersensitivity cannot be given any of the NSAIDs because of some cross-hypersensitivity. Allergic reactions can vary from rash, wheezing, urticaria, and angioneurotic edema to anaphylactic shock. When a true aspirin allergy exists, any aspirin-containing products or NSAIDs should be avoided.
Persons with asthma are more likely to have a hypersensitivity reaction to aspirin, with the incidence ranging from 5% to 15%. The aspirin hypersensitivity triad, consisting of aspirin hypersensitivity, asthma, and nasal polyps—often occur together. This reaction is thought to be the result of the shunting of the products of arachidonic acid from the production of prostaglandins to the leukotrienes and is thought to be a potential mechanism for this hypersensitivity. These patients exhibit cross-hypersensitivity involving aspirin and other agents, including the NSAIDs, and they should not be given any NSAIDs.
Toxicity
An overdose of aspirin can have harmful effects and even cause death.
Symptoms
When the blood level of salicylates reaches a certain point, a toxic reaction, referred to as salicylism, occurs. It is characterized by tinnitus (ringing in the ears), headache, nausea, vomiting, dizziness, and dimness of vision. Hyperthermia and electrolyte imbalance can also occur. With higher blood levels, stimulation of respiration leads to hyperventilation, which produces respiratory alkalosis. Compensatory alkalosis results in renal loss of bicarbonate, sodium, and potassium. Both respiratory acidosis and metabolic acidosis ensue. The cause of death from aspirin poisoning is usually acidosis and electrolyte imbalance.
Prevention
Children are the primary victims of accidental poisoning. The lethal dose of aspirin for a child is 4 g, and the lethal dose of aspirin for an adult is 10-30 g. The education of parents regarding the potential for poisoning and proper storage and childproof containers for over-the-counter (OTC) aspirin have significantly reduced accidental poisonings in children.
Treatment
Treatment of aspirin poisoning includes removing excess drug in the stomach by inducing emesis or administering activated charcoal to absorb the aspirin. Other symptoms are treated symptomatically. For example, hyperthermia is treated with cooling baths or “blankets,” acidosis with sodium bicarbonate, hypokalemia with potassium, and hypoglycemia with intravenous (IV) glucose. Box 5-3 lists the patient instructions for aspirin.
Drug Interactions
The drug interactions of aspirin are listed in Table 5-1. Some of the more notable are briefly discussed in the following:
Warfarin: The drug interaction between aspirin and warfarin can result in bleeding. Warfarin (WAR-far-in), an oral anticoagulant, is highly protein bound. If aspirin is administered to a patient taking warfarin, it can displace the warfarin from its binding sites, increasing its anticoagulant effect. In addition, aspirin affects both platelets and the gastrointestinal tract. Bleeding and hemorrhage may result from these interactions.
Probenecid: Aspirin interferes with the uricosuric effect of probenecid (proe-BEN-e-sid). Aspirin has been reported to precipitate an acute attack of gout. One should avoid using aspirin in patients taking probenecid.
Methotrexate: Methotrexate (meth-oh-TREX-ate) (MTX) is an antineoplastic drug used to treat certain kinds of cancer and autoimmune diseases (arthritis, psoriasis). Aspirin can displace MTX from its protein-binding sites and can also interfere with its clearance. The displacement and interference result in an increased serum concentration and MTX toxicity such as bone marrow depression.
Sulfonylureas: Higher doses of salicylates (more than 2 g) may produce a hypoglycemic effect. One proposed mechanism involves the displacement of the sulfonylureas from their plasma protein-binding sites by aspirin. This hypoglycemia effect can also be observed with insulin.
Antihypertensives: Aspirin reduces the antihypertensive effect of many antihypertensives, including angiotensin-converting enzyme (ACE) inhibitors, β-blockers, and thiazide and loop diuretics. Several doses of aspirin over a few days are needed for this reduction to occur. Aspirin’s effect on the renal function, resulting in water and sodium retention, may contribute to this effect.
Uses
General Uses
One use of aspirin is to provide analgesia for mild-to-moderate pain. It is the analgesic against which new analgesics are measured for efficacy. Its antipyretic effect is useful in the control of fever but should be avoided in children (Reye’s syndrome). Its antiinflammatory action is used in the treatment of inflammatory conditions such as rheumatic fever and arthritis.
Low-Dose Aspirin Therapy
Because of its effect on platelet aggregation (inhibition), aspirin is used to prevent unwanted clotting. Clinical trials have shown that daily use of aspirin as a secondary preventive measure can reduce all-cause mortality by 18% and myocardial infarctions by 30% in persons with known cardiovascular disease. Aspirin can be of benefit to persons that have already had a stroke, heart attack, angina, or peripheral vascular disease as well as those who have had angioplasty or bypass surgery. Low-dose aspirin therapy (75-325 mg) is recommended for these patients unless there are contraindications to therapy.
Patients experiencing a myocardial infarction are usually advised by emergency responders to chew one low-dose aspirin at the onset of symptoms. Patients should call 9-1-1 first and wait for their instructions before taking the aspirin tablet. The aspirin tablet can be administered once the emergency responder is present, in the ambulance, or in the emergency room. Again, this measure is indicated only in patients who do not have aspirin allergies, hypersensitivity, or other contraindications.
Aspirin can also be used as preventative therapy in persons who have not experienced a heart attack or stroke but are at increased risk for these events. The U.S. Preventative Service Task Force recommends that men with no history of heart disease or stroke aged 45-79 years of age take low-dose aspirin each day to prevent a myocardial infarction and that women with no history of heart attack or stroke aged 55-79 take low-dose aspirin each day to prevent a stroke. However, the benefit of aspirin therapy must outweigh the risk of the many adverse effects associated with aspirin therapy, especially gastrointestinal bleeding.
Doses and Preparations
The usual adult dose of aspirin for the treatment of pain or fever is 325-650 mg every 4 hours. The dose for arthritis is 3000- 4000 mg per day in divided doses. For prevention of myocardial infarction, the dose is 75-325 mg/day. The dose for children 2-11 years of age is 10-15 mg/kg every 4-6 hours (not to exceed 4 g per day).
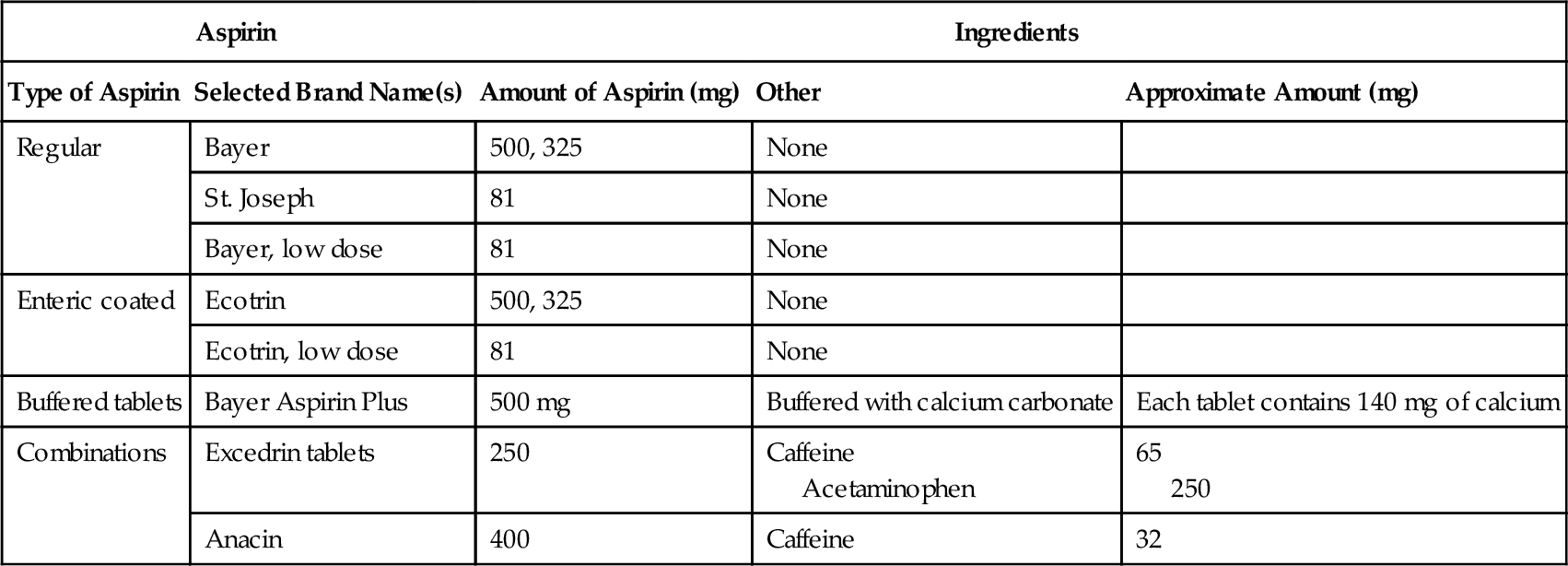
Many types of preparations containing aspirin are available by prescription and OTC (Table 5-2). Some of these types are as follows:
Table 5-2
Selected Over-the-Counter (OTC) Aspirin-Containing Products
| Aspirin | Ingredients | |||
| Type of Aspirin | Selected Brand Name(s) | Amount of Aspirin (mg) | Other | Approximate Amount (mg) |
| Regular | Bayer | 500, 325 | None | |
| St. Joseph | 81 | None | ||
| Bayer, low dose | 81 | None | ||
| Enteric coated | Ecotrin | 500, 325 | None | |
| Ecotrin, low dose | 81 | None | ||
| Buffered tablets | Bayer Aspirin Plus | 500 mg | Buffered with calcium carbonate | Each tablet contains 140 mg of calcium |
| Combinations | Excedrin tablets | 250 | Caffeine Acetaminophen | 65 250 |
| Anacin | 400 | Caffeine | 32 | |
Regular Aspirin
Single-entity forms of aspirin include the commonly used 325-mg (5-grain) tablet and the 81-mg flavored children’s tablet. Many brand name and generic products are available in all strengths.
Enteric-Coated Aspirin
Aspirin can be formulated with a coating that dissolves in the intestine rather than in the stomach. The advantage of such enteric-coated aspirin is that gastric symptoms are reduced. The disadvantage is that these products can be erratically absorbed, resulting in unreliable blood levels. The onset of action is too long to make them useful for acute dental pain. They have limited use in treatment of chronic arthritis when gastric irritation is a problem. They can be used when daily aspirin is used for clot prevention.
Combinations
With Buffer
Although claimed to have fewer gastrointestinal side effects, buffered tableted preparations have never been shown to do so. They are absorbed at a slightly quicker rate. The liquid buffered preparations do produce less gastrointestinal irritation, but they contain sodium, which is relatively contraindicated in patients with high blood pressure.
With Another Analgesic
Aspirin can be combined with an opioid analgesic or acetaminophen. Caffeine is part of this combination. Mixing aspirin with an opioid can allow a decrease in the amount of the opioid in the product and therefore reduce its side effects.
With Sedatives
Adding a sedative to aspirin can make it more effective if anxiety is a substantial component of the pain. Prescribing a separate antianxiety agent would give the prescriber more control, however, and is preferred.
With Caffeine
Caffeine potentiates the analgesic effect of aspirin and other analgesics. The addition of 130 mg of caffeine is equivalent to increasing the dose of the analgesic by one third or more. Most proprietary preparations contain about half this much caffeine. (However, one can always take two tablets of most analgesics.)
Nonacetylated Salicylates
Common Agents
Sodium, choline, magnesium salicylate and salicylamide, and salsalate are other salicylates. These drugs do not interfere with platelet aggregation, are rarely associated with gastrointestinal (GI) bleeding, and are well tolerated by persons with asthma. Also, there is no cross-sensitivity with aspirin. However, there are no well-controlled studies demonstrating the comparative efficacy of nonacetylated salicylates for treating chronic pain. Their efficacy as analgesic agents and the appropriate doses for analgesia must be determined. Magnesium is contraindicated in patients with renal disease, and sodium in those with cardiovascular disease. Salicylamide is a weak analgesic. Salsalate is made up of the combination of two salicylic acids.
Diflunisal
Diflunisal (dye-FLOO-ni-sal) (Dolobid) is a nonacetylated salicylate classified as an NSAID. Its peak action occurs 2-3 hours after ingestion, and its half-life is 8-12 hours in the normal patient. It is as effective as the other NSAIDs in the treatment of pain. Like other NSAIDs, diflunisal can be administered before a dental procedure to delay the onset of postsurgical pain. Because of its long half-life, it is dosed only two or three times daily. The general comments relating to the NSAIDs also apply to diflunisal. Its antipyretic effect is not clinically useful.
Nonsteroidal antiinflammatory drugs
NSAIDs have important applications in dentistry. Their mechanism of action and many of their pharmacologic effects and adverse reactions resemble those of aspirin. Many prescribers agree that the NSAIDs are the most useful drug group for the treatment of dental pain. The availability of OTC NSAIDs gives the dental health hygienist several products that can be recommended for purchase. Whether a prescription should be written for an NSAID or an OTC NSAID recommended depends on appraisal of the patient’s attitudes.
Chemical Classification
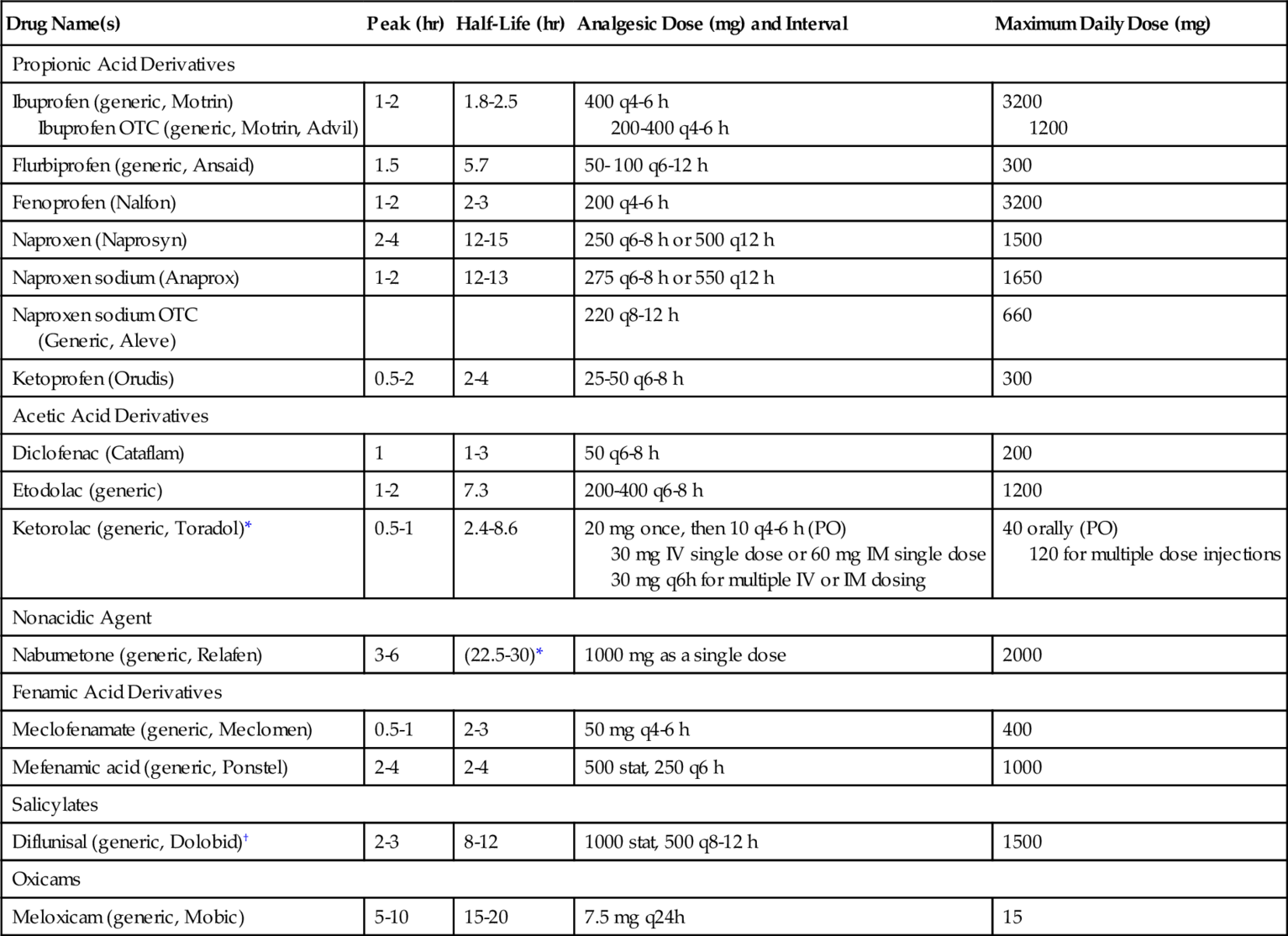
NSAIDs are divided into several chemical derivatives: propionic acids, acetic acids, fenamates, pyrazolones, oxicams, and others. Table 5-3 lists selected NSAIDs by chemical classification, pharmacokinetic parameters, analgesic dose, and dosing interval.
Table 5-3
Nonselective Nonsteroidal Antiinflammatory Drugs: Peak, Half-Life, and Analgesic and Maximum Doses
| Drug Name(s) | Peak (hr) | Half-Life (hr) | Analgesic Dose (mg) and Interval | Maximum Daily Dose (mg) |
| Propionic Acid Derivatives | ||||
| Ibuprofen (generic, Motrin) Ibuprofen OTC (generic, Motrin, Advil) | 1-2 | 1.8-2.5 | 400 q4-6 h 200-400 q4-6 h | 3200 1200 |
| Flurbiprofen (generic, Ansaid) | 1.5 | 5.7 | 50- 100 q6-12 h | 300 |
| Fenoprofen (Nalfon) | 1-2 | 2-3 | 200 q4-6 h | 3200 |
| Naproxen (Naprosyn) | 2-4 | 12-15 | 250 q6-8 h or 500 q12 h | 1500 |
| Naproxen sodium (Anaprox) | 1-2 | 12-13 | 275 q6-8 h or 550 q12 h | 1650 |
| Naproxen sodium OTC (Generic, Aleve) | 220 q8-12 h | 660 | ||
| Ketoprofen (Orudis) | 0.5-2 | 2-4 | 25-50 q6-8 h | 300 |
| Acetic Acid Derivatives | ||||
| Diclofenac (Cataflam) | 1 | 1-3 | 50 q6-8 h | 200 |
| Etodolac (generic) | 1-2 | 7.3 | 200-400 q6-8 h | 1200 |
| Ketorolac (generic, Toradol)* | 0.5-1 | 2.4-8.6 | 20 mg once, then 10 q4-6 h (PO) 30 mg IV single dose or 60 mg IM single dose 30 mg q6h for multiple IV or IM dosing | 40 orally (PO) 120 for multiple dose injections |
| Nonacidic Agent | ||||
| Nabumetone (generic, Relafen) | 3-6 | (22.5-30)* | 1000 mg as a single dose | 2000 |
| Fenamic Acid Derivatives | ||||
| Meclofenamate (generic, Meclomen) | 0.5-1 | 2-3 | 50 mg q4-6 h | 400 |
| Mefenamic acid (generic, Ponstel) | 2-4 | 2-4 | 500 stat, 250 q6 h | 1000 |
| Salicylates | ||||
| Diflunisal (generic, Dolobid)† | 2-3 | 8-12 | 1000 stat, 500 q8-12 h | 1500 |
| Oxicams | ||||
| Meloxicam (generic, Mobic) | 5-10 | 15-20 | 7.5 mg q24h | 15 |
OTC, over-the-counter.
Stat, immediately.
* For short-term (< 5 days) treatment following the use of the parenteral form.
† Salicylate.
Mechanism of Action
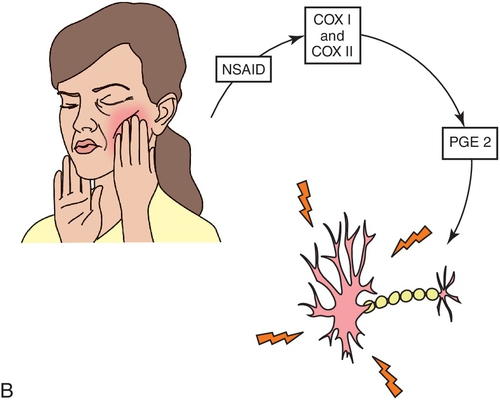
Like aspirin, NSAIDs inhibit the enzyme COX (prostaglandin synthase), resulting in a reduction in the formation of prostaglandin precursors and thromboxanes from arachidonic acid (see Figure 5-4). Many of the actions and the adverse reactions of the NSAIDs result from their inhibition of prostaglandin synthesis.
All of the currently available NSAIDs inhibit both COX I and COX II. COX I is a widely distributed constitutive (present at all times) enzyme responsible for the adverse reactions of the NSAIDs, such as stomach problems, reduced renal function, fluid retention, and reduced platelet adhesiveness. COX II is an inducible enzyme that is synthesized only when inflammation occurs. COX II is also expressed in the kidneys, where it helps maintain perfusion.
Pharmacokinetics
Most NSAIDs peak in about 1-2 hours. The effect of food on absorption of the NSAIDs approved to treat pain—ibuprofen, the naproxens, and diflunisal—is to reduce the rate but not the extent of their absorption. Oral antacids have no effect on absorption of the NSAIDs except for diflunisal (antacids reduce absorption). These agents are metabolized in the liver and excreted by the kidney. The half-lives of the individual agents are listed in Table 5-3.
Pharmacologic Effects
The analgesic, antipyretic, and antiinflammatory actions of the NSAIDs result from the same mechanism as aspirin’s inhibition of prostaglandin synthesis, the inhibition of COX. NSAIDs are useful for treating dysmenorrhea (painful menstruation) because an excess of prostaglandins in the uterine wall produces painful contractions. In the treatment of gout, the action of the NSAIDs is related to their analgesic and antiinflammatory actions but is independent of their effect on serum uric acid.
Adverse Reactions
Gastrointestinal Effects
Gastrointestinal irritation, pain, and bleeding problems leading to tarry stools can occur with all NSAIDs. The prostaglandins stimulate the production of cytoprotective mucus that protects the stomach against gastric acid secretion. Prostaglandin inhibitors, such as NSAIDs, can interfere with the normal protective mechanisms in the stomach and increase acid secretion, causing symptoms or even an ulceration or perforation. A prostaglandin, misoprostol (mye-soe-PROST-ole) or prostaglandin E2 (Cytotec, PGE2), is available to prevent NSAID-induced ulcers.
Central Nervous System Effects
The dose-dependent CNS side effects of NSAIDs include sedation, dizziness, confusion, mental depression, headache, vertigo, and convulsions. Because of the CNS effects of the NSAIDs, patients taking them should be cautioned about driving an automobile. These agents are not addicting, tolerance does not develop, and no withdrawal syndrome can be induced.
Blood Clotting
The NSAIDs reversibly inhibit platelet aggregation because they inhibit thromboxane A2 production. In contrast to that of aspirin, an NSAID’s effect remains only as long as the drug is present in the blood: 1 day for ibuprofen, 4 days for naproxen, and 2 weeks for oxaprozin.
Cardiovascular Effects
There have been reports of an increased risk of serious cardiovascular events such as myocardial infarction and stroke in patients taking some NSAIDs. The risks appear to be highest with diclofenac and lowest with naproxen. The risk appears to increase with higher doses of celecoxib but otherwise appears to be similar to that with nonselective NSAIDs.
Renal Effects
All NSAIDs, including celecoxib, inhibit renal prostaglandins, decrease renal blood flow, cause fluid retention, and may cause hypertension and renal failure in some patients, especially the elderly. Renal effects of the NSAIDs include renal failure, cystitis, and an increased incidence of urinary tract infections. The NSAIDs have little effect on the patient with normal kidney function; however, in those with renal disease, decreases in both renal blood flow and glomerular filtration rate can occur. NSAIDs have precipitated renal insufficiency. In patients with decreased renal function, peripheral edema with fluid retention has been noted.
Oral Effects
Oral manifestations of NSAIDs reported include ulcerative stomatitis, gingival ulcerations, and dry mouth.
Other Effects
Other adverse effects associated with the NSAIDs are muscle weakness, ringing in the ears, hepatitis, hematologic problems, and blurred vision. Celecoxib has been associated with cholestatic jaundice, which may be related to sulfonamide allergy. Celecoxib is contraindicated in persons with sulfonamide allergies.
Hypersensitivity Reactions
Like aspirin, the NSAIDs can induce a wide range of hypersensitivity reactions, including hives or itching, angioneurotic edema, chills and fever, Stevens-Johnson syndrome, exfoliative dermatitis, and epidermal necrolysis. Anaphylactoid reactions including bronchospasm (wheezing) have been reported.
Pregnancy and Nursing Considerations
Like aspirin, the NSAIDs given late in pregnancy can prolong gestation, delay parturition, and produce dystocia or premature closure of the ductus arteriosus. The uterine prostaglandins are responsible for parturition and closure of the ductus arteriosus. Fenoprofen, ibuprofen, and naproxen have not been shown to be teratogenic in animal studies (U.S. Food and Drug Administration [FDA] pregnancy category B). Diflunisal, tolmetin, and mefenamic acid have been shown to be teratogenic in animals (FDA pregnancy category C).
Ibuprofen has not been detected in breast milk, whereas fenoprofen and mefenamic acid are present in small quantities. Small amounts of both naproxen (1% of serum) and diflunisal (5% of serum) are excreted in breast milk. Ibuprofen is the drug of choice for treating a nursing woman.
Drug Interactions
The drug interactions of the NSAIDs are summarized in Table 5-4. Interactions for each NSAID continue to be under investigation for their clinical significance and presence. Lithium toxicity has been reported in patients taking lithium for bipolar affective disorders who also take NSAIDs. These agents may increase the effect of digoxin, a drug used for congestive heart failure. Digoxin’s narrow therapeutic index is one reason for caution. NSAIDs have been shown to reduce the effect of agents used as antihypertensives, such as diuretics, angiotensin-converting enzyme inhibitors (ACE) inhibitors, and β-blockers. Probenecid can raise serum levels of NSAIDs. These agents can increase the toxicity of cyclosporin and MTX. Before patients are given NSAIDs, drug interactions should be checked.
Table 5-4
Selected Drug Interactions of the Nonsteroidal Antiinflammatory Drugs
| Drug | Potential Outcome |
| Lithium | Increased effect of lithium |
| Methotrexate (MTX) | Increased effect of MTX leads to bone marrow toxicity |
| Diuretics | Reduced antihypertensive effect |
| Angiotensin-converting enzyme (ACE) inhibitors | Reduced antihypertensive effect |
| β-Blockers | Reduced antihypertensive effect |
| Digoxin | Increased digoxin effect |
Contraindications and Cautions
The contraindications to and cautions for using an NSAID (Table 5-5) are related to their adverse reactions. Patients with asthma, cardiovascular or renal diseases with fluid retention, coagulopathies, peptic ulcer, and ulcerative colitis should be given NSAIDs cautiously, if at all.
Table 5-5
Contraindications to and Cautions for the Use of Aspirin and Nonsteroidal Antiinflammatory Drugs (NSAIDs)
| Drugs | Disease | Comments |
| Aspirin: Small doses | Prevent clotting, heart disease | May use NSAIDs, continue aspirin |
| Aspirin: High doses | Rheumatic fever | Aspirin lowers blood level of NSAIDs |
| Lithium | Bipolar (manic) disorder | NSAIDs reduce lithium clearance; potential lithium toxicity |
| H2-blockers Proton pump inhibitors | Peptic ulcer, gastroesophageal reflux disease | Gastric bleeding, esophagitis |
| Angiotensin-converting enzyme (ACE) inhibitors, Angiotensin receptor blockers (ARBs) | Renal disease | Can decrease kidney function, especially in the elderly |
| Diuretics | Renal disease | Can decrease kidney function, especially in the elderly, especially if added to ACE inhibitors and ARBs |
| Methotrexate (MTX) | Rheumatoid arthritis, psoriasis, cancer | Potentiates MTX toxicity, bone marrow suppression* |
| Vitamin K deficiency | Alcoholism, liver disease | Bleeding |
| Warfarin | Myocardial infarction, atrial fibrillation, prosthetic heart valve | Aspirin contraindicated; bleeding; use NSAIDs with caution; increases anticoagulant effect of warfarin |
| Factor VIII | Hemophilia | Gastric bleeding |
| Probenecid* | Gout | Probenecid inhibits excretion of NSAIDs |
| Colchicine | Gout | More gastrointestinal adverse reactions |
| Allopurinol | Gout | No contraindications |
| None | Pregnancy | Use should be avoided during pregnancy, especially the third trimester |
| None | Glucose-6-phosphate dehydrogenase (G6PD) deficiency | Hemolysis |
| None | Hypoprothrombinemia | Bleeding |
| Acute viral illness | Dehydration | Decreased renal function |
* Once-a-week dosing as for autoimmune diseases can be used with caution.
All NSAIDs have been associated with the development of acute kidney injury. This risk is higher in those patients who are hypovolemic, who have a viral illness, or who may be taking ACE inhibitors or angiotensin receptor blockers alone or in combination with diuretics. For this reason, renal function should be monitored in patients at risk for renal disease and NSAID use should be avoided in these patients.
All NSAIDs contain block box warnings for the following events:
Cardiovascular disease: All NSAIDs may increase the risk of serious cardiovascular (CV) thrombotic events, myocardial infarction, and stroke, all of which may be fatal. The risk may increase with a longer duration of use, and in patients with cardiovascular disease or risk factors for cardiovascular disease.
Gastrointestinal effects: All NSAIDs increase the risk for serious GI events, including bleeding, ulceration, and perforation of the stomach or intestine, which may be fatal. Severe gastrointestinal events can occur at any time and without warning, and elderly patients are at a higher risk.
Patients also at higher risk for adverse reactions to NSAIDs include those with renal function impairment or a history of previous hypersensitivity to aspirin or other NSAIDs and geriatric patients, who are more prone to adverse hepatic or renal reactions. Box 5-4 lists the patient instructions for NSAIDs.
Therapeutic Uses
Medical
Depending on the specific NSAID and the clinical trials that have been conducted, medical use of NSAIDs involves many conditions. Osteoarthritis, rheumatoid arthritis, gouty arthritis, fever, dysmenorrhea, and pain are indications for the NSAIDs. Accepted unlabeled indications for which NSAIDs are often prescribed include bursitis and tendonitis.
Dental
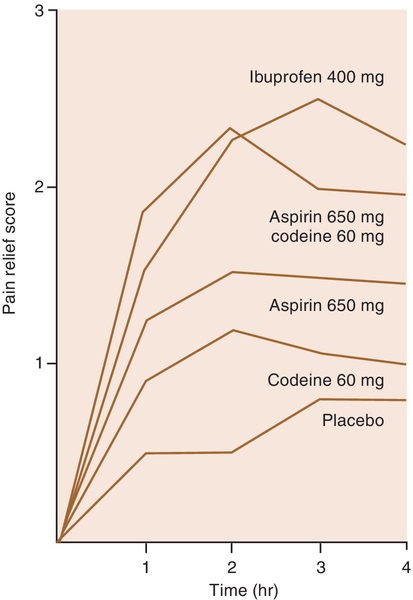
NSAIDs are useful in the management of dental pain. Many studies that have compared the analgesic efficacy of the NSAIDs with that of the opioid analgesics find that they are equivalent in many clinical situations. For example, usual analgesic doses of NSAIDs have been shown to be as effective as 650 mg of aspirin or acetaminophen plus 60 mg of codeine and even as effective as the intermediate-strength opioid combinations (oxycodone plus aspirin or acetaminophen). In usual prescription doses, NSAIDs can be shown to be statistically significantly better than codeine alone, aspirin, acetaminophen, or placebo. All NSAIDs are equally efficacious at equianalgesic doses. Figure 5-7 shows the pain relief over time of several commonly used analgesics. Their relative effectiveness is discussed in the following paragraphs.
Specific Nonsteroidal Antiinflammatory Drugs
Ibuprofen
Ibuprofen (eye-byoo-PRO-fen) (Advil, Motrin), the oldest member of the NSAIDs, has the most clinical experience. It is rapidly absorbed orally, and food decreases its rate but not its extent of absorption; antacids have no effect. The half-life is about 2 hours. Its onset of action is about half an hour, and its duration of action is 4-6 hours. It undergoes hepatic metabolism and is excreted by the kidney. It is an effective analgesic and has been studied in many dental situations. Ibuprofen is the drug of choice for treatment of dental pain when an NSAID is indicated. Only in rare cases or if new information becomes available are other NSAIDs indicated. When a longer-acting agent is desired for patient convenience, the naproxens can be used.
The usual prescription analgesic dose of ibuprofen is 400-800 mg every 4-6 hours (maximum dose: 3200 mg/day) and the usual OTC analgesic dose is 200-400 mg every 4-6 hours (maximum dose: 1200 mg/day). The higher dosage range may have more antiinflammatory effects. Most studies can easily demonstrate that 400 mg of ibuprofen is better than any usual therapeutic dose of codeine.
Ibuprofen is available OTC in 200-mg tablets and by prescription in 400-, 600-, and 800-mg tablets. Side effects, such as CNS effects, are dose dependent, so they occur more often at the higher end of the dose range. Ibuprofen is also available OTC in suspension form for pediatric use and is often used its fever reducing effects.
Naproxen Sodium
Naproxen (na-PROX-en) (Naprosyn), naproxen sodium (Anaprox), and naproxen sodium OTC (Aleve) are propionic acid NSAIDs that have slightly longer half-lives than ibuprofen and can be dosed on an 8-12-hour schedule. They should also be given with a loading dose (see Table 5-3). Aleve is dosed 220 mg every 8-12 hours, not to exceed 2 tablets in the 8-12-hour period. The patient may take 2 tablets for the first dose for a total of 3 tablets in the first 24-hour time period. The pharmacologic effects, adverse reactions, and efficacy of naproxen are similar to those of ibuprofen. In addition to tablets, this product is available in suspension form. One should note that lithium blood levels rise when naproxen is given because it inhibits lithium clearance.
Other Nonsteroidal Antiinflammatory Drugs
Other NSAIDs (see Table 5-3), such as fenoprofen, ketorolac, and diflunisal, may be used for patients whose pain does not respond to either ibuprofen or naproxen sodium. Certain patients need an agent with which they are not familiar. “Shoppers” looking for scheduled drugs show better response to an unknown drug’s name. Prescribing one of the new NSAIDs whose name has not yet become familiar may be effective.
Ketorolac (kee-TOE-role-ak) (Toradol) is a newer NSAID. The use of this agent increased because it was being heavily advertised to dental professionals. It is equivalent in efficacy to the other NSAIDs; however, unlike other NSAIDs, it is available parenterally. One should make sure that before a new agent is prescribed, it has some documented clinical advantage.
Ketorolac is an NSAID indicated for the short-term (up to 5 days) management of moderately severe acute pain that requires analgesia at the opioid level. It is contraindicated as a prophylactic analgesic before any major surgery, when hemostasis is critical because of the increased risk of bleeding. Oral ketorolac is indicated only as continuation therapy to IV or intramuscular (IM) ketorolac (injectable must be used before the tablets are prescribed).
Cyclo-Oxygenase II–Specific Agents
COX II-specific inhibitors selectively decrease the inflammatory effects of COX II while leaving the protective effects of COX I largely in place, leading to fewer adverse reactions than the older, nonselective NSAIDs. Celecoxib (Celebrex), a COX II-specific inhibitor, is indicated for arthritis. Celecoxib was thought to offer an advantage over nonselective NSAIDs because it was believed to be less irritating to the stomach. However, it has been found to be associated with a significantly higher incidence of serious gastrointestinal adverse effects. Clinically, celecoxib is equivalent to nonselective NSAIDs. Because it offers no therapeutic advantage over nonselective NSAIDs it has no real use in dentistry.
Acetaminophen
Acetaminophen (a-seet-a-MEE-noe-fen) (paracetamol, N-acetyl p-aminophenol; Tylenol; APAP) is the only member of the p-aminophenols currently available for clinical use. Its exact mechanism and site of action are unknown. However, acetaminophen is thought to produce analgesia by elevating the pain threshold. Its potential mechanism of action may involve inhibiting the nitric oxide pathway, which is mediated by a variety of neurotransmitter receptors. Its antipyretic effects may be the result of inhibition of endogenous pyrogens on the heat-regulating centers of the brain by blockage of the formation and release of prostaglandins in the CNS.
Pharmacokinetics
Acetaminophen is rapidly and completely absorbed from the gastrointestinal tract, achieving a peak plasma level in 1-3 hours. After therapeutic doses, it is excreted with a half-life of 1-4 hours. Acetaminophen is metabolized by the liver microsomal enzymes to the glucuronide conjugate, the sulfuric acid conjugate, and cysteine. When large doses are ingested, an intermediate metabolite is produced that is thought to be hepatotoxic and possibly nephrotoxic. Acetaminophen and aspirin are equally efficacious (kill the same degree of pain) and equally potent (same doses in milligrams needed for effect) as analgesics and antipyretics.
Pharmacologic Effects
The analgesic and antipyretic effects of acetaminophen are approximately the same potency (on a milligram-for-milligram basis) as aspirin. This means that acetaminophen and aspirin are equally efficacious, and because virtually the same doses are used for each agent, they are equally potent. However, acetaminophen does not possess any clinically significant antiinflammatory effect. Therefore it is less useful in the treatment of arthritis or any other type of inflammatory pain. Differences in degree of prostaglandin synthesis inhibition at different sites may account for this difference in action.
Therapeutic doses of acetaminophen have no effect on the cardiovascular or respiratory system. In contrast to aspirin, acetaminophen does not produce gastric bleeding or affect platelet adhesiveness or uric acid excretion.
Adverse Reactions
The principal toxic effects of acetaminophen are hepatic necrosis and nephrotoxicity.
Hepatic Effects
The toxic metabolite of acetaminophen that contributes to hepatic necrosis is N-acetyl-p-benzoquinoneimine. The minimum toxic dose of acetaminophen for a single ingestion is 7.5 to 10 g in adults and 150 mg/kg in children. The acute ingestion of more than 12 g of acetaminophen in single ingestion is considered a toxic dose and can poses a high risk for liver damage. In children, the acute ingestion of 250 mg/kg poses a high risk for liver damage, and the acute ingestion of 350 mg/kg can cause severe hepatotoxicity if not immediately treated. More often than not, children experience more cases of accidental overdose with acetaminophen. This occurs because the wrong dose form is used (e.g., infant drops are given to older children or adult doses are given to children). Infant drops are concentrated, and doses for toddlers to 11-year-old children are not the same as those for infants. Parents may give the infant liquid to the older child and pour it in the measuring cup, not realizing that they have overdosed their child. Also, normal doses over extended periods can lead to toxicity. Symptoms during the first 2 days after intoxication are minor. Nausea, vomiting, anorexia, and abdominal pain may occur. Liver injury becomes manifest on the second to third day, with alterations in plasma enzyme levels (elevations of transaminase and lactic hydrogenase), elevated bilirubin value, and prolongation of prothrombin time. Hepatotoxicity may progress to encephalopathy, coma, and death. If the patient recovers, no residual hepatic abnormalities persist. Patients with hepatic disease, such as those with a history of hepatitis, should avoid acetaminophen.
Most recently, the FDA has recommended tighter dose controls and warnings with acetaminophen use, in the hope of preventing even more cases of accidental liver toxicity. The FDA advisory committee is recommending that people receive no more than 650 milligrams (2 regular-strength tablets [325 mg each] every 4-6 hours, and no more than 10 tablets in one day (24-hour time period) or 2 extra-strength tablets (500 mg each) every 6 hours, and no more than 6 tablets in 1 day. Doses of 1000 mg four times per day can, after only 1 or 2 days, lead to liver toxicity. This change would include decreasing the amount of acetaminophen that is often used in combination with opioid analgesics.
Alcohol stimulates the oxidizing enzymes that metabolize acetaminophen to its toxic metabolite. Depending on the amount of alcoholic beverages ingested, the maximum dose of acetaminophen varies. The normal maximum dose of acetaminophen (4 g) may be used in patients who usually do not drink. The dose should be restricted to 2 g if a patient is a moderate drinker (less than three alcohol beverages daily). People who consume three or more alcohol beverages every day should not take acetaminophen (Table 5-6).
Table 5-6
Maximum Acetaminophen (APAP) Dose Related To Alcohol Use
| Alcohol Habitually Consumed | Maximum Daily Dose of APAP (g) |
| None | 4 |
| ≥ 2 | 2 |
| ≥ 3 | None |
Treatment of Toxicity
The treatment of acetaminophen overdose toxicity should begin with gastric lavage if the drug has recently been ingested. The administration of activated charcoal should follow. The administration of sulfhydryl groups in the form of oral N-acetylcysteine reduces or even prevents liver damage if given soon enough after ingestion.
Nephrotoxicity
Nephrotoxicity has been associated with long-term consumption of acetaminophen. The primary lesion appears to be a papillary necrosis with secondary interstitial nephritis. Although no single agent can be identified, prolonged consumption of analgesics can lead to kidney disease. Because analgesics are used in dental practice on a short-term basis, the possibility of nephrotoxicity does not present a significant problem in dental therapy. Concurrent chronic use of the combination of acetaminophen and aspirin or NSAIDs increases the risk of analgesic nephropathy, renal papillary necrosis, end-stage renal disease, and cancer of the kidney or urinary bladder.
Skin Reactions
In August 2013 the FDA issued a safety announcement regarding acetaminophen because of the risk of three rare, but potentially fatal, skin reactions: Stevens-Johnson syndrome, toxic epidermal necrolysis, and acute generalized exanthematous pustulosis. This warning advised that anyone who experiences a skin reaction, such as rash or blister, while taking acetaminophen should stop taking it and seek immediate medical care. This warning was made on the basis of a review of the medical literature and reports of adverse reactions to the FDA Adverse Event Reporting System database.
Drug Interactions
Acetaminophen is remarkably free of drug interactions at its usual therapeutic doses. The hepatotoxicity of acetaminophen can be potentiated by administration of agents that induce hepatic microsomal enzymes, such as barbiturates, carbamazepine, phenytoin, and rifampin. Long-term ingestion of large doses of alcohol can increase the toxicity of acetaminophen.
Uses
Acetaminophen is used as an analgesic and antipyretic. It is especially useful in patients who have aspirin hypersensitivity or in whom aspirin-induced gastric irritation would present a problem. In young children, the use of acetaminophen as an antipyretic has replaced that of aspirin because of aspirin’s association with Reye’s syndrome. It is not known to what degree the long-term use of therapeutic doses of acetaminophen might produce renal lesions. It has a greater propensity for causing hepatic necrosis when a large acute dose (overdose) is ingested. Box 5-5 lists the patient instructions for acetaminophen.
Doses and Preparations
Acetaminophen is available in many different dose forms. The usual adult dose is 2 325-mg (regular-strength) tablets every 4-6 hours, 2 650-mg (extra-strength) tablets every 6 hours, or 2 500 mg caplets (extra-strength) every 6 hours. No more than 10 regular-strength tablets or 6 extra-strength tablets should be ingested by adults in 24 hours. Various suspensions and meltaway and chewable tablets that are convenient for administration to children are available. Recently, acetaminophen infant and children’s dose forms have been reformulated so that both are now available in an oral suspension in a concentration of 160 mg/5 ml (teaspoonful). This was done in an effort to reduce the incidence of overdose. The infant suspension comes with a calibrated syringe, and the oral suspension with a calibrated measuring cup. The infant suspension should be given only on the advice of the child’s prescribing practitioner. The children’s oral suspension is for those children older than 2 years. The meltaway tablets are available as 80-mg and 160-mg tablets (junior-strength for children 6-11 years of age). Acetaminophen should not be administered to a child more than 5 times in 24 hours. The dosing of acetaminophen in children can be determined using Table 5-7.
Table 5-7
Pediatric Acetaminophen Dosing Chart*
| Weight (lb) | Age (yr) | mg | Suspension† 160 mg/5 ml (no. tsp) |
| 24 | < 2 | Consult | Consult with a physician |
| 25-35 | 2-3 | 160 | 5 ml (1 tsp) |
| 36-47 | 4-5 | 240 | 7.5 ml (1.5 tsp) |
| 48-59 | 6-8 | 320 | 10 ml (2 tsp) |
| 60-71 | 9-10 | 400 | 12.5 ml (2.5 tsp) |
| 72-95 | 11 | 480 | 15 ml (3 tsp) |
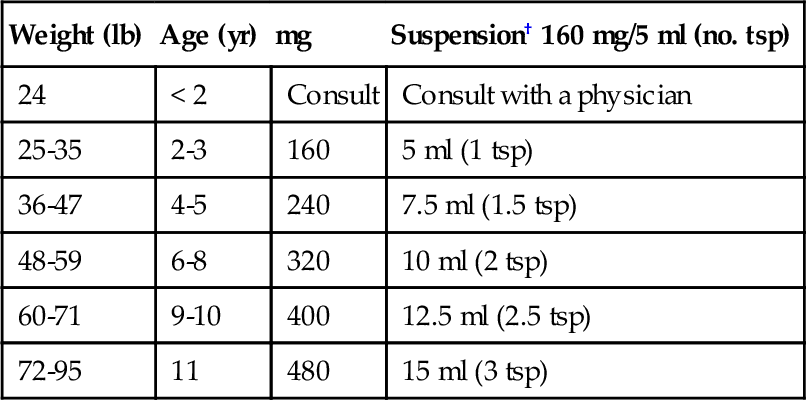
* Obtain the child’s weight in pounds; check weight column and determine applicable row; read the dose (mg) column to determine dose; identify preparation parent has or will purchase; determine the volume or number of tablets needed for the dose and product.
† CAUTION: Preparations with different concentrations available; number of teaspoonfuls given only for this concentration; infants’ concentrated drops use much less volume.
Drugs used to treat gout
Gout is an inherited disease occurring primarily in men, with an onset that usually involves one joint, often the big toe or knee. Both hyperuricemia and urate crystals, or tophi, may be found in the joints or other tissues. The excess uric acid may be the result of excessive production or reduced excretion of uric acid (two types of gout). The disease responds to colchicine.
Both the NSAIDs and colchicine are used to treat acute attacks of gout. Other agents, such as probenecid and allopurinol (al-oh-PURE-i-nole), are available to prevent gout. These are briefly mentioned here, although they are not analgesics per se.
Colchicine
Colchicine (KOL-chi-seen) has only one indication: the treatment of an acute attack of gout. It is so specific in its action on gouty attacks that it is sometimes used to diagnose the disease. Colchicine is taken hourly at the onset of the attack or until side effects, such as nausea and vomiting, are intolerable. Its mechanism is complex, but it appears to inhibit the chemotactic property of leukocytosis and interfere with the inflammatory response to urate crystals. Colchicine possesses many side effects, but gastrointestinal toxicity, including nausea, vomiting, and diarrhea, occurs often (in up to 80% of people). Bone marrow depression and hypersensitivity have also been reported.
Allopurinol
Allopurinol (Zyloprim) is a xanthine oxidase inhibitor that inhibits the synthesis of uric acid. It is used to prevent excessive uric acid from forming. It is also used in patients undergoing either chemotherapy or irradiation for malignancy, because the death of many cells causes a release of large amounts of uric acid precursors. The side effects associated with allopurinol include hepatotoxicity of a hypersensitivity type. If a pruritic rash should occur, the drug should be promptly discontinued because fatalities have been reported in patients with this reaction. This drug is not indicated for asymptomatic hyperuricemia.
Probenecid
The other approach to prevention of gout is to increase the excretion of uric acid by the administration of a uricosuric agent such as probenecid (Benemid). By blocking the tubular reabsorption of filtered urate, probenecid prevents new tophi and mobilizes those present. Increasing frequency or severity of acute gouty attacks is an indication for administration of a uricosuric agent.
Gastrointestinal side effects and hypersensitivity may occur with probenecid use. Headaches and sore gums have also been reported. Concurrent administration of aspirin can interfere with the uricosuric action of probenecid. Diabetic tests using the copper sulfate urine test (Clinitest) may have false-positive results. Occasionally, probenecid and colchicine are combined, with the colchicine preventing acute attacks and the probenecid enhancing the excretion of uric acid. Probably a more rational approach is to administer each drug separately as needed. Maintenance of adequate urinary output (at least 2 L) is important to minimize the precipitation of uric acid in the urinary tract.
Probenecid increases the plasma levels of NSAIDs and penicillin. In the latter case, this effect can be used therapeutically (see discussion of penicillin in Chapter 7). For prevention of acute gout, either probenecid or allopurinol can be used. Acute gout normally is treated with NSAIDs and colchicine.
Other Drugs
For mild-to-moderate pain associated with gout, the drug of choice is either acetaminophen or aspirin in adults. Aspirin provides an antiinflammatory effect but is contraindicated in children and adolescents. If both aspirin and acetaminophen provide inadequate pain relief, then ibuprofen can be used. Its analgesic efficacy parallels that of many products combining nonopioids with opioids, such as aspirin with codeine (Empirin #3).
Dental hygiene considerations
• If nonopioid analgesics are necessary, the dental hygienist should conduct a thorough medication/health history in order to determine whether the patient has any contraindications to or potential drug interactions.
• Information regarding salicylates, NSAIDS, and acetaminophen should include warnings to not exceed the manufacturer’s recommended daily dose over a 24-hour period.
• The dental hygienist should encourage patients to check the OTC labels for any overlapping ingredients. Often, these products contain ibuprofen, aspirin, acetaminophen, or any combination of the three with antihistamines and decongestants.
• The dental hygienist should also be aware of the fact that many opioid analgesics are combined with nonopioid analgesics. Remind the patient to not supplement with an OTC analgesic if a combination nonopioid/opioid analgesic is prescribed.
• Warnings about significant side effects associated with OTC nonopioid analgesics (such as bleeding) should be given to the patient along with instructions to call the dental practice if an adverse reaction occurs.
• NSAIDs should be avoided in persons with asthma.
• If patients complain of gastrointestinal adverse effects, they may require a semisupine chair position during dental treatment.
Academic skills assessment
1. What is the rationale for using acetaminophen in a patient with an ulcer?
2. What dose and duration of therapy should be recommended for an adult male patient?
3. What are the adverse reactions to acetaminophen?
4. Are nephrotoxicity and hepatotoxicity associated only with toxic doses of acetaminophen?
5. What would increase the risk of development of nephrotoxicity with acetaminophen?
6. What are the pharmacologic effects of acetaminophen?
7. Compare and contrast acetaminophen with aspirin in terms of pharmacologic and therapeutic effects.
8. Does acetaminophen have any potential drug interactions? If so, what are they and how can they be avoided?
9. Are there any dental concerns associated with taking one baby aspirin each day?
10. Should a patient taking high blood pressure medication take a drug like ibuprofen? Why or why not?
11. Compare and contrast the OTC NSAIDs.
12. When would a prescription NSAID be appropriate?
13. Are there interactions between NSAIDs and antihypertensive drugs?
14. The dentist recommends a short course of OTC ibuprofen. What should a patient be told about this drug?
15. Can aspirin be used in children less than age 18? Why or why not?
16. Why is it especially important to use the correct dose form of acetaminophen in children?
Clinical case study
James Smith is 45 years old and is new to your practice. This is your first meeting with him, and you would like to ask him some questions regarding his medication/health history. During the course of your conversation you learn that he has a history of coronary heart disease and is currently taking a baby aspirin each day. He takes acetaminophen for general aches and pains. He also likes to have a glass of wine with dinner each night and doesn’t mind a few beers when he is watching football. During the course of his examination you and the dentist find two cavities, which are filled that day. Mr. Smith is experiencing some mild pain after the procedure.
1. What is the rationale for using acetaminophen instead of an NSAID instead to treat Mr. Smith’s pain?
2. What dose and duration of therapy should be recommended for Mr. Smith?
3. At what doses does hepatotoxicity occur with acetaminophen?
4. How can acetaminophen toxicity be avoided in Mr. Smith?
5. Compare and contrast acetaminophen to aspirin in terms of pharmacology, adverse effects, and therapeutic effects.
6. What is the role of aspirin in the prevention of heart attack or stroke?
7. Are any dental concerns associated with low-dose aspirin therapy?
8. Can Mr. Smith take a drug like ibuprofen?
9. What should be said to Mr. Smith during a counseling session regarding acetaminophen?