Anus, Rectum, and Prostate
Outline
http://evolve.elsevier.com/Jarvis/
• Bedside Assessment Summary Checklist
• Physical Examination Summary Checklist
Structure and Function
Anus and Rectum
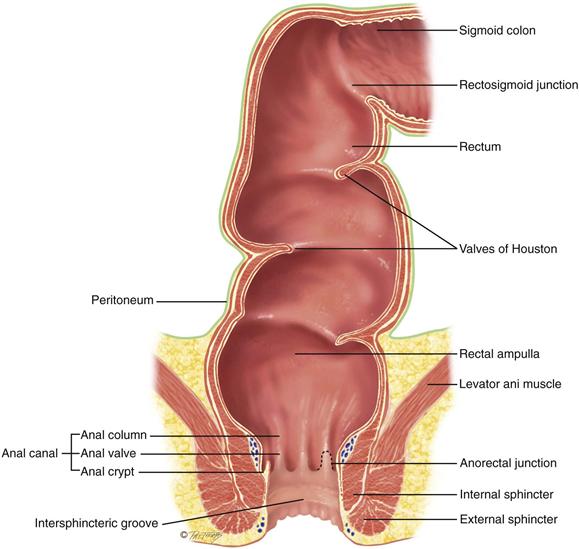
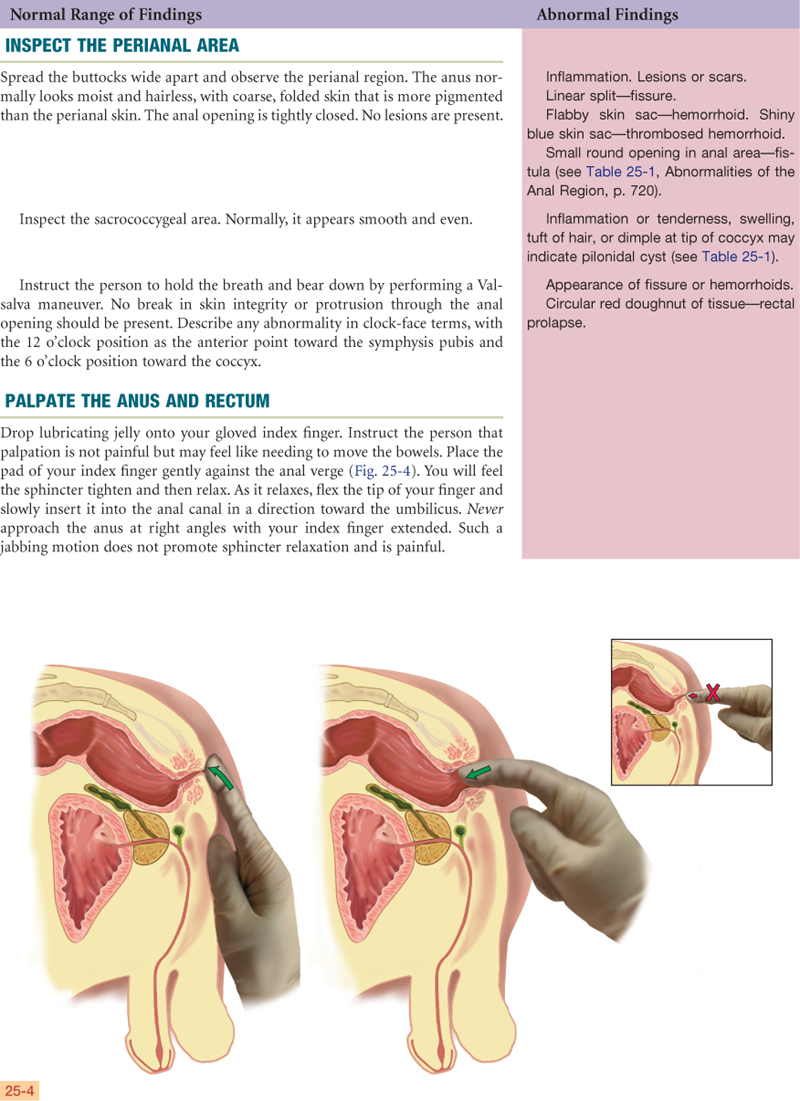
The anal canal is the outlet of the gastrointestinal (GI) tract, and it is about 3.8 cm long in the adult. It is lined with modified skin (having no hair or sebaceous glands) that merges with rectal mucosa at the anorectal junction. The canal slants forward toward the umbilicus, forming a distinct right angle with the rectum, which rests back in the hollow of the sacrum. Although the rectum contains only autonomic nerves, numerous somatic sensory nerves are present in the anal canal and external skin, so a person feels sharp pain from any trauma to the anal area.
The anal canal is surrounded by two concentric layers of muscle, the sphincters (Fig. 25-1). The internal sphincter is under involuntary control by the autonomic nervous system. The external sphincter surrounds the internal sphincter but also has a small section overriding the tip of the internal sphincter at the opening. It is under voluntary control. Except for the passing of feces and gas, the sphincters keep the anal canal tightly closed. The intersphincteric groove separates the internal and external sphincters and is palpable.
25-1
The anal columns (or columns of Morgagni) are folds of mucosa. These extend vertically down from the rectum and end in the anorectal junction (also called the mucocutaneous junction, pectinate, or dentate line). This junction is not palpable, but it is visible on proctoscopy. Each anal column contains an artery and a vein. Under conditions of chronic increased venous pressure, the vein may enlarge, forming a hemorrhoid. At the lower end of each column is a small crescent fold of mucous membrane, the anal valve. The space above the anal valve (between the columns) is a small recess, the anal crypt.
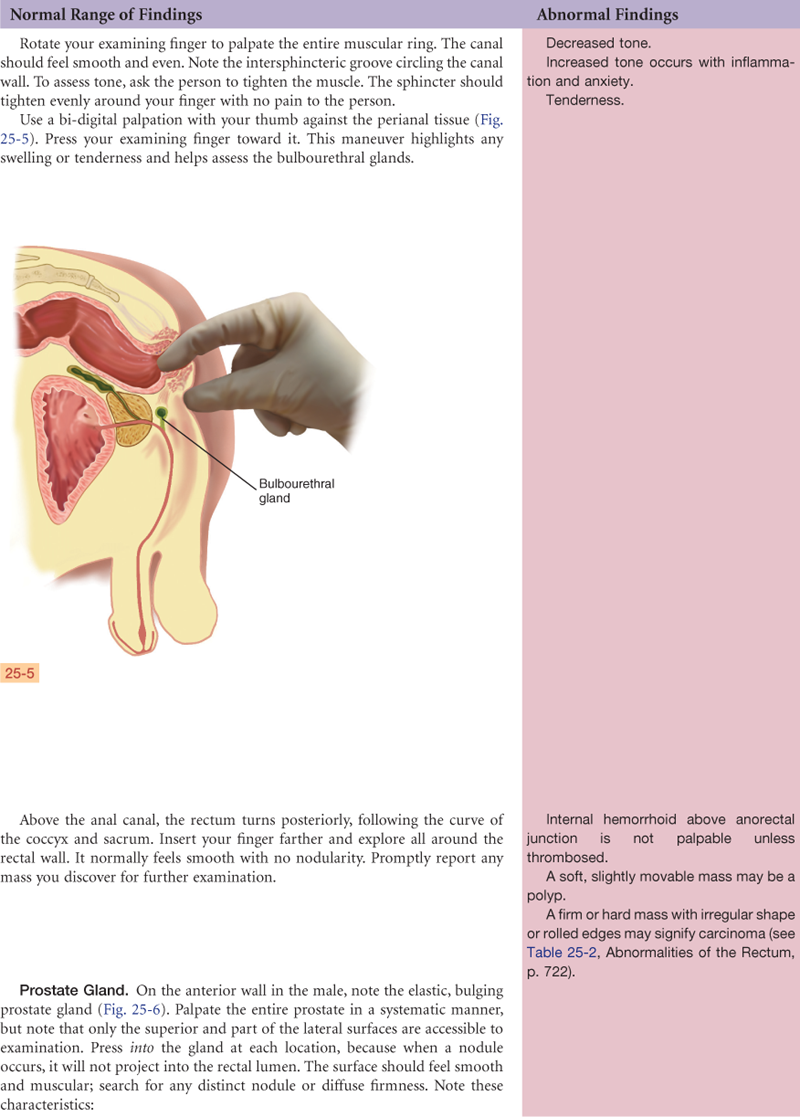
The rectum, which is 12 cm long, is the distal portion of the large intestine. It extends from the sigmoid colon, at the level of the third sacral vertebra, and ends at the anal canal. Just above the anal canal, the rectum dilates and turns posteriorly, forming the rectal ampulla. The rectal interior has three semilunar transverse folds called the valves of Houston. These cross one-half the circumference of the rectal lumen. Their function is unclear, but they may serve to hold feces as the flatus passes. The lowest is within reach of palpation, usually on the person’s left side, and must not be mistaken for an intrarectal mass.
Peritoneal Reflection
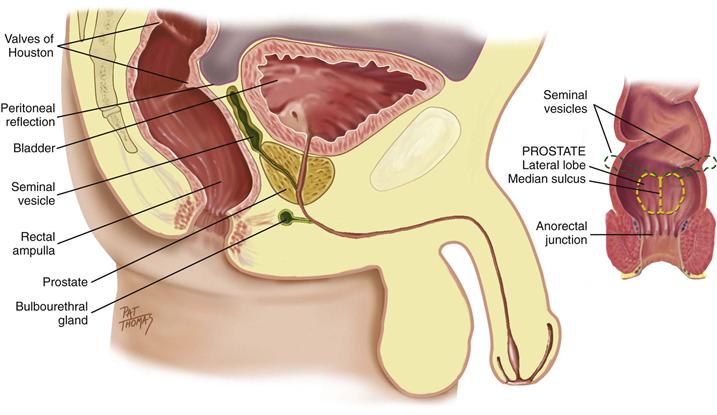
The peritoneum covers only the upper two thirds of the rectum. In the male, the anterior part of the peritoneum reflects down to within 7.5 cm of the anal opening, forming the rectovesical pouch (Fig. 25-2) and then covers the bladder. In the female, this is termed the recto-uterine pouch and extends down to within 5.5 cm of the anal opening.
25-2
Prostate
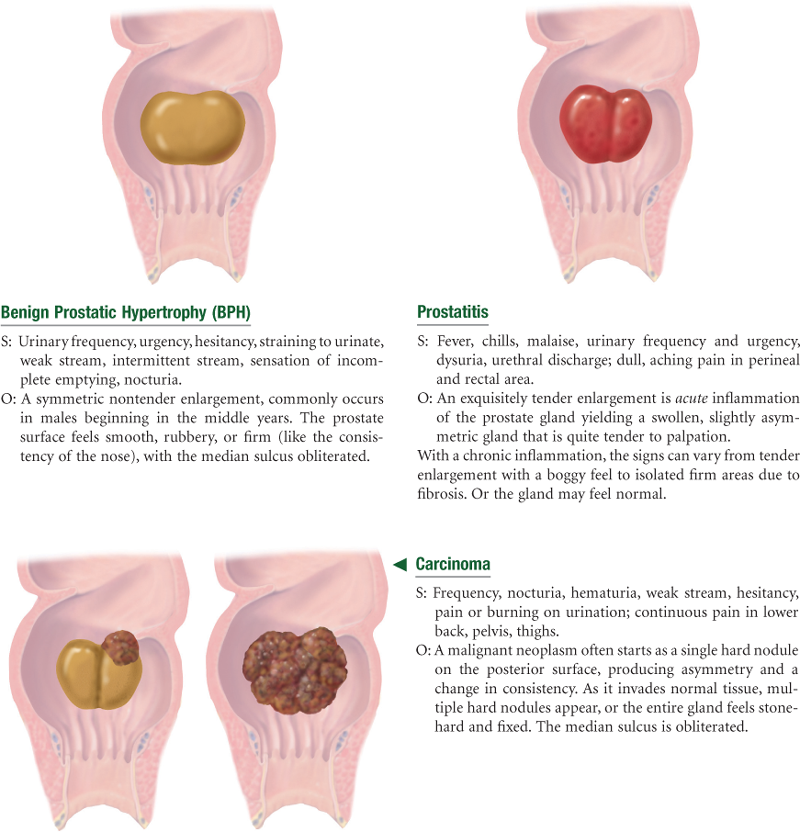
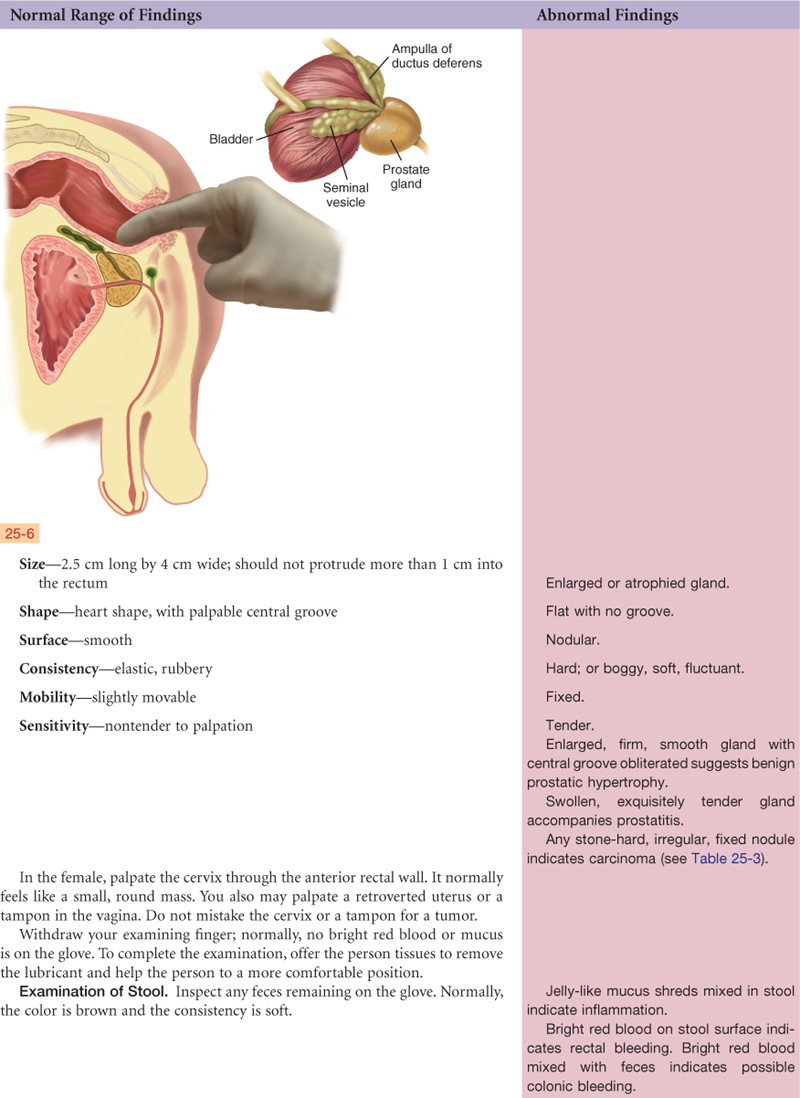
In the male, the prostate gland lies in front of the anterior wall of the rectum and 2 cm behind the symphysis pubis. It surrounds the bladder neck and the urethra and has 15 to 30 ducts that open into the urethra. The prostate secretes a thin, milky, alkaline fluid that helps sperm viability. It is a bilobed structure with a round or heart shape. It measures 2.5 cm long and 4 cm in diameter. The two lateral lobes are separated by a shallow groove called the median sulcus.
The two seminal vesicles project like rabbit ears above the prostate. The seminal vesicles secrete a fluid that is rich in fructose, which nourishes the sperm and contains prostaglandins. The two bulbourethral (Cowper) glands are each the size of a pea and are located inferior to the prostate on either side of the urethra (also see Fig. 25-5). They secrete a clear, viscid mucus.
Regional Structures
In the female, the uterine cervix lies in front of the anterior rectal wall and may be palpated through it.
The combined length of the anal canal and the rectum is about 16 cm in the adult. The average length of the examining finger is from 6 cm to 10 cm, bringing many rectal structures within reach.
The sigmoid colon is named from its S-shaped course in the pelvic cavity. It extends from the iliac flexure of the descending colon and ends at the rectum. It is 40 cm long and is accessible to examination only through the colonoscope. The flexible fiberoptic scope in current use provides a view of the entire mucosal surface of the sigmoid, as well as the colon.
 Developmental Competence
Developmental Competence
The first stool passed by the newborn is dark green meconium and occurs within 24 to 48 hours of birth, indicating anal patency. From that time on, the infant usually has a stool after each feeding. This response to eating is a wave of peristalsis called the gastrocolic reflex. It continues throughout life, although children and adults usually produce no more than one or two stools per day.
The infant passes stools by reflex. Voluntary control of the external anal sphincter cannot occur until the nerves supplying the area have become fully myelinated, usually around 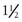 to 2 years of age. Toilet training usually starts after age 2 years.
to 2 years of age. Toilet training usually starts after age 2 years.
At male puberty, the prostate gland undergoes a very rapid increase to more than twice its prepubertal size. During young adulthood, its size remains fairly constant.
The prostate gland commonly starts to enlarge during the middle adult years. This benign prostatic hypertrophy (BPH) is present in 1 of 10 males at the age of 40 years and grows larger with age. It is thought that the hypertrophy is caused by hormonal imbalance that leads to the proliferation of benign adenomas. These gradually impede urine output because they obstruct the urethra.
 Culture And genetics
Culture And genetics
Prostate cancer (PC) is more common in North America and northwestern Europe and is less common in Central and South America, Africa, and Asia. The incidence of PC is higher for African-American men than for men of other racial groups; African-American men are more likely to be diagnosed at an advanced stage of the disease, and mortality rates are two times higher for African-American men than for white men.2
What are the risk factors? Family history is positively associated; men with one first-degree relative (father or brother) are 2 to 3 times more likely to develop PC.2 Genetic factors contribute some risk: men with BRCA2 mutations have increased risk for developing a more aggressive form of PC and at a younger age. Environmental factors may account for more risk than genetics because migration studies show men of Asian and African heritage living in the United States have a higher risk for PC than their counterparts living in Asia and Africa.2,9 Environmental factors include diet, a potentially modifiable risk factor. Diets high in red meat and processed meat, animal and saturated fats, and dairy products may increase risk,2 whereas diets high in fiber, fruits, and vegetables may lower risk.23
Of the men who have PC, those more likely to have an advanced disease at diagnosis include men who were uninsured or Medicaid-insured and men of racial/ethnic minority groups.24 This has implications for screening and culturally appropriate health education. Screening guidelines were updated in 20102 and state the ages at which men should receive health information about benefits and risks of PC tests. Men at average risk for PC should receive information at 50 years; at higher risk (African Americans and positive family history) at 45 years; and at very high risk (multiple family members with PC) at 40 years. Screening includes the blood test for prostate-specific antigen (PSA) and a physical examination (i.e., a digital rectal exam [DRE]).32
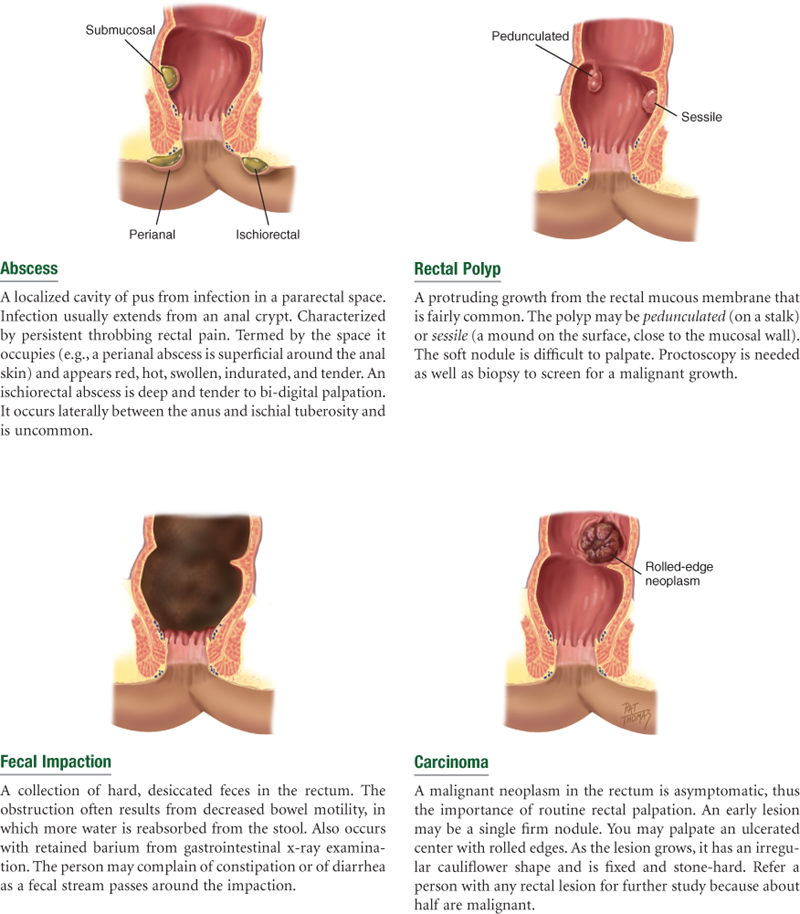
Colorectal cancer has a racial variation as well. The incidence rates are almost 20% higher for African-American women and men than for whites, and the mortality rates are almost 50% higher for African Americans than for whites.10 Because colorectal cancer can largely be prevented by removal of adenomatous polyps, guidelines for average-risk adults start at age 50 years and include health teaching about options for screening. Options include a colonoscopy every 10 years with bowel preparation and conscious sedation, and an annual guaiac-based fecal occult blood test or fecal immunochemical test.22
Subjective Data
3. Rectal bleeding, blood in the stool
4. Medications (laxatives, stool softeners, iron)
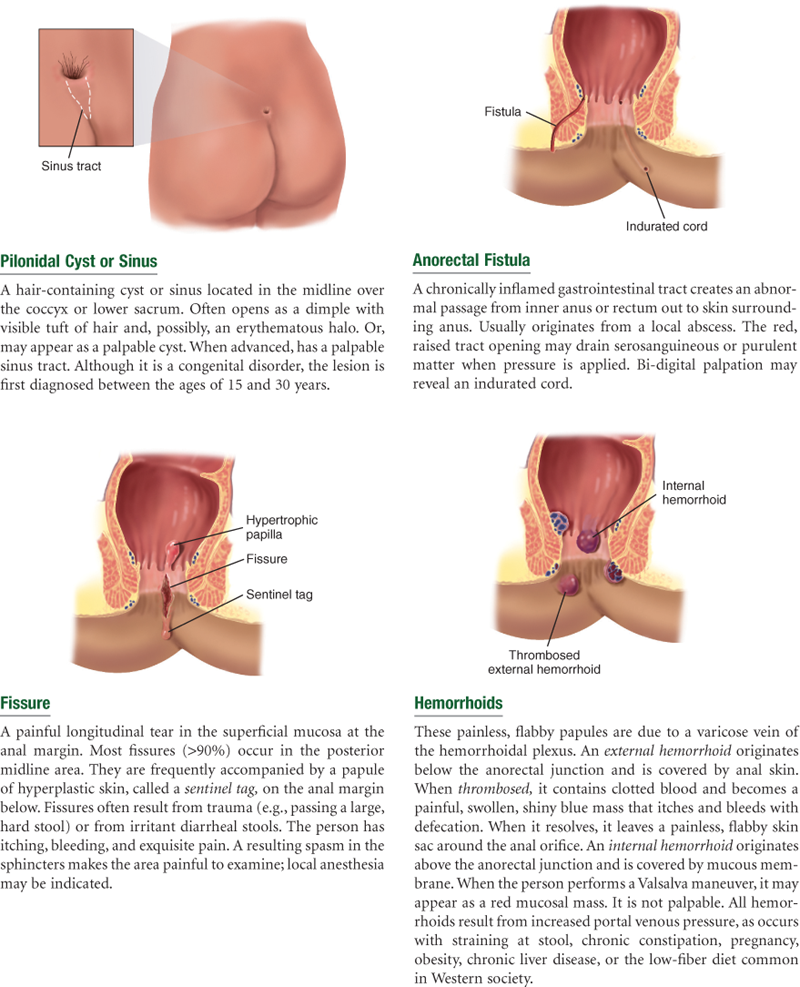
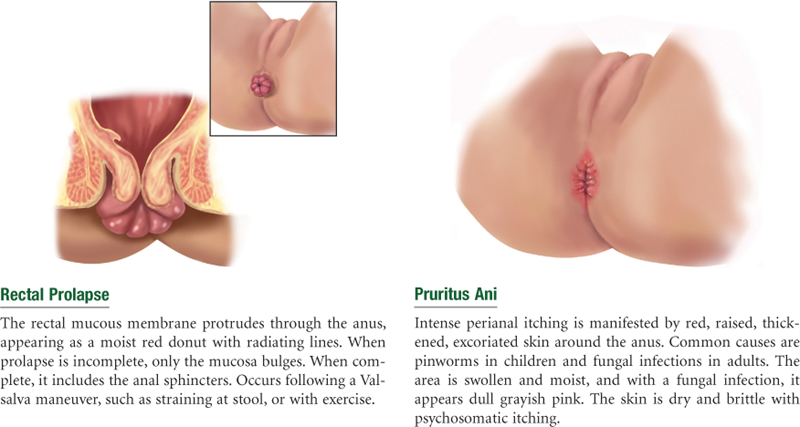
5. Rectal conditions (pruritus, hemorrhoids, fissure, fistula)
7. Self-care behaviors (diet of high-fiber foods, most recent examinations)
| Examiner Asks | Rationale |
| 1.Usual bowel routine. Bowels move regularly? How often? Usual color? Hard or soft? •Any straining at stool, incomplete evacuation, urge to have bowel movement but nothing comes? •Eat breakfast? (This increases colon motility and prompts a bowel movement in many.) •Pain while passing a bowel movement? | Assess usual bowel routine. Constipation is ≤3 stools/week and is a common concern among aging adults. Dyschezia. Pain due to a local condition (hemorrhoid, fissure) or constipation. |
| 2.Change in bowel habits. Any change in usual bowel habits? Loose stools or diarrhea? When did this start? Is the diarrhea associated with nausea and vomiting, abdominal pain, something you ate recently? | Diarrhea occurs with gastroenteritis, colitis, irritable colon syndrome. |
| •Eaten at a restaurant recently? Anyone else in your group or family have the same symptoms? | Consider food poisoning. |
| •Traveled to a foreign country during the past 6 months? | Consider parasitic infection. |
| •Stools have a hard consistency? When did this start? | Constipation. |
| 3.Rectal bleeding, blood in the stool. Ever had black or bloody stools? When did you first notice blood in the stools? What is the color, bright red or dark red-black? How much blood: spotting on the toilet paper or outright passing of blood with the stool? Do the bloody stools have a particular smell? | Melena. Black stools may be tarry due to occult blood (melena) from GI bleeding or nontarry from ingestion of iron medications. Red blood in stools occurs with GI bleeding or local bleeding around the anus and with colon and rectal cancer. |
| •Ever had clay-colored stools? •Ever had mucus or pus in stool? | Clay color indicates absent bile pigment. |
| •Frothy stool? | Steatorrhea is excessive fat in the stool as in malabsorption of fat. |
| •Need to pass gas frequently? | Flatulence. |
| 4.Medications. What medications do you take—prescription and over-the-counter? Laxatives or stool softeners? Which ones? How often? Iron pills? Do you ever use enemas to move your bowels? How often? | |
| 5.Rectal conditions. Any problems in rectal area: itching, pain or burning, hemorrhoids? How do you treat these? Any hemorrhoid preparations? Ever had a fissure or fistula? How was this treated? | Pruritus. |
| •Ever had a problem controlling your bowels? | Fecal incontinence. Mucoid discharge and soiled underwear occur with prolapsed hemorrhoids. |
| 6.Family history. Any family history: polyps or cancer in colon or rectum, inflammatory bowel disease, prostate cancer? | Risk factors for colon cancer, rectal cancer, prostate cancer. |
| 7.Self-care behaviors. What is the usual amount of high-fiber foods in your daily diet: cereals, apples or other fruits, vegetables, whole-grain breads? How many glasses of water do you drink each day? | High-fiber foods of the soluble type (beans, prunes, barley, carrots, broccoli, cabbage) lower cholesterol, whereas insoluble fiber foods (cereals, wheat germ) reduce risk for colon cancer. Also, fiber foods fight obesity, stabilize blood sugar, and help some GI disorders. |
| •Date of last: digital rectal examination, stool blood test, colonoscopy, (for men) prostate-specific antigen blood test. | Early detection for cancer: DRE performed annually after age 50 years; fecal occult blood test annually after age 50 years; sigmoidoscopy every 5 years or colonoscopy every 10 years after age 50 years; PSA blood test annually for men older than 50 years, except Black men beginning at age 45 years.2 |
| Additional History for Infants and Children | |
| 1.Have you ever noticed any irritation in your child’s anal area: redness, raised skin, frequent itching? | In children, pinworms are a common cause of intense itching and irritated anal skin. |
| 2.How are your child’s bowel movements? Frequency? Any problems? Any pain or straining with BM? | Assess usual stooling pattern. Constipation is a decrease in BM frequency, with difficult passing of very hard, dry stools. Encopresis is persistent passing of stools into clothing in a child older than 4 years, at which age continence would be expected. |
Objective Data
| Preparation | Equipment needed |
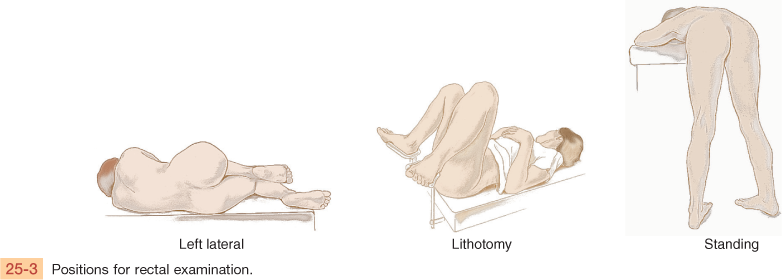
| Perform a rectal examination on all adults and particularly those in middle and late years. Help the person assume one of the following positions (Fig. 25-3): Examine the male in the left lateral decubitus or standing position. Instruct the standing male to point his toes together; this relaxes the regional muscles, making it easier to spread the buttocks. Place the female in the lithotomy position if examining genitalia as well; use the left lateral decubitus position for the rectal area alone. |
Penlight Lubricating jelly Glove Guaiac test container |

 Documentation and Critical Thinking
Documentation and Critical Thinking
Sample Charting
Subjective
Has one BM daily, soft, brown, no pain, no change in bowel routine. On no medications. Has no history of pruritus, hemorrhoids, fissure, or fistula. Diet includes one to two servings daily each of fresh fruits and vegetables but no whole-grain cereals or breads.
Objective
No fissure, hemorrhoids, fistula, or skin lesions in perianal area. Sphincter tone good, no prolapse. Rectal walls smooth, no masses or tenderness. Prostate not enlarged, no masses or tenderness. Stool brown, Hematest negative.
Assessment
Rectal structures intact, no palpable lesions
Focused Assessment: Clinical Case Study
C.M. is a 62-year-old white male with chronic obstructive pulmonary disease for 15 years, who today has “diarrhea for 3 days.”
Subjective
7 days PTA—C.M. seen at this agency for acute respiratory infection that was diagnosed as acute bronchitis and treated with oral ampicillin. Took medication as directed.
3 days PTA—symptoms of respiratory infection improved. Ingesting usual diet. Onset of four to five loose, unformed, brown stools a day. No abdominal pain or cramping. No nausea.
Now—diarrhea continues. No blood or mucus noticed in stool. No new foods or restaurant food in past 3 days. Wife not ill.
Objective
Vital signs: 37° C-88-18. BP 142/82.
Respiratory: Respirations unlabored. Barrel chest. Hyperresonant to percussion. Lung sounds clear but diminished. No crackles or rhonchi today.
Abdomen: Flat. Bowel sounds present. No organomegaly or tenderness to palpation.
Rectal: No lesions in perianal area. Sphincter tone good. Rectal walls smooth, no mass or tenderness. Prostate smooth and firm, no median sulcus palpable, no masses or tenderness. Stool brown, Hematest negative.
Assessment
Diarrhea R/T effects of antibiotic medication
Abnormal Findings FOR ADVANCED PRACTICE
Summary Checklist: Anus, Rectum, and Prostate Examination
![]() For a PDA-downloadable version, go to http://evolve.elsevier.com/Jarvis/.
For a PDA-downloadable version, go to http://evolve.elsevier.com/Jarvis/.
Bibliography
1. American Cancer Society. Cancer facts and figures 2010. Atlanta: Author; 2010.
2. American Cancer Society. Cancer facts and figures 2010 (Special section: Prostate cancer, pp 23-36). Atlanta: Author; 2010.
3. Arras-Boyd RE, Boyd RE, Gaehle K. Reaching men at highest risk for undetected prostate cancer. International Journal of Men’s Health. 2009;8(2):116–128.
4. Ayanian JZ. Racial disparities in outcomes of colorectal cancer screening: biology or barriers to optimal care? Journal of the National Cancer Institute. 2010;102(8):511–513.
5. Berry J, Bumpers K, Ogunlade V, et al. Examining racial disparities in colorectal cancer care. Journal of Psychosocial Oncology. 2009;27(1):59–83.
6. Bouras EP, Tangalos EG. Chronic constipation in the elderly. Gastroenterology Clinics of North America. 2009;38(3):463–480.
7. Chan JA, Meyerhardt JA, Niedzwiecki D, et al. Association of family history with cancer recurrence and survival among patients with stage III colon cancer. Journal of the American Medical Association. 2008;229(21):2515–2523.
8. Daly JM, Merchant ML, Levy BT. Colorectal cancer screening. American Journal of Nursing. 2009;109(10):60–62.
9. Delongchamps NB, Singh A, Haas GP. Epidemiology of prostate cancer in Africa: another step in the understanding of the disease? Current Problems in Cancer. 2007;31(3):226–236.
9a. Dunivan GC, Heymen S, Palsson OS, et al. Fecal incontinence in primary care: Prevalence, diagnosis, and health care utilization. American Journal of Obstetrics and Gynecology. 2010;202:493.e1–493.e6.
10. Edwards BK, Ward E, Kohler BA, et al. Annual report to the nation on the status of cancer, 1975-2006, featuring colorectal cancer trends and impact of interventions (risk factors, screening, and treatment) to reduce future rates. Cancer. 2010;116(3):544–573.
11. Fried RG. Performing digital rectal examination can detect cancers. American Family Physician. 2010;31(9):1073.
12. Gray M, Sims T. Prostate cancer: prevention and management of the localized disease. Nurse Practitioner. 2006;31(9):14–31.
13. Greene MD. Diagnosis and management of HPV-related anal dysplasia. Nurse Practitioner. 2009;34(5):45–51.
14. Guittet L, Bouvier V, Mariotte N, et al. Comparison of a guaiac and an immunochemical faecal occult blood test for the detection of colonic lesions according to lesion type and location. British Journal of Cancer. 2009;100(8):1230–1235.
15. Holcomb SS. Colorectal cancer: new screening guideline. Nurse Practitioner. 2008;33(9):13–18.
16. Imperiale TF, Ransohoff DF, Itzkowitz SH, et al. Fecal DNA versus fecal occult blood for colorectal-cancer screening in an average-risk population. New England Journal of Medicine. 2004;351(26):2704–2714.
17. Jones RA, Steeves R, Williams I. How African American men decide whether or not to get prostate cancer screening. Cancer Nursing. 2009;32(2):166–172.
18. Knight D. Health care screening for men who have sex with men. American Family Physician. 2004;69(9):2149–2156.
19. Kong AP, Stamos MJ. Anorectal complications: office diagnosis and treatment, Part 1. Consultant. 2005;45(7):731–734.
20. Kong AP, Stamos MJ. Anorectal complications: office diagnosis and treatment, Part 2. Consultant. 2005;45(7):735–738.
21. Lee TH, Kantoff PW, McNaughton-Collins MF. Screening for prostate cancer. New England Journal of Medicine. 2009;360(13):e18.
22. Levin B, Lieberman DA, McFarland B, et al. Screening and surveillance for the early detection of colorectal cancer and adenomatous polyps, 2008: a joint guideline from the American Cancer Society, the U.S Multi-Society Task force on Colorectal Cancer, and the American College of Radiology. CA: A Cancer Journal for Clinicians. 2008;58(3):130–156.
23. Lewis JE, Soler-Vilá H, Clark PE, et al. Intake of plant foods and associated nutrients in prostate cancer risk. Nutrition and Cancer. 2009;61(2):216–224.
24. Marlow NM, Halpern MT, Pavluck AL, et al. Disparities associated with advanced prostate cancer stage at diagnosis. Journal of Health Care for the Poor and Underserved. 2010;21(1):112–131.
25. Moses KA, Paciorek AT, Penson DF, et al. Impact of ethnicity on primary treatment choice and mortality in men with prostate cancer: data from CaPSURE. Journal of Clinical Oncology. 2010;28(6):1069–1074.
26. Peters DP. Colon cancer screening: recommendations and barriers to patient participation. Nurse Practitioner. 2008;33(12):14–21.
26a. Philichi L. Pediatric constipation and encopresis. Gastroenterology Nursing. 2008;31(2):121–130.
27. Sanford KW, McPherson RA. Fecal occult blood testing. Clinics in Laboratory Medicine. 2009;29(3):523–541.
28. Walia R, Mahajan L, Steffen R. Recent advances in chronic constipation. Current Opinion in Pediatrics. 2009;21(5):661–666.
29. Wallace M, Bailey DE, Brion J. Shedding light on prostate cancer. Nurse Practitioner. 2009;34(10):25–34.
30. Ward-Smith P. Screening and preventing prostate cancer: implementing the evidence. Urologic Nursing. 2009;29(6):437–443.
31. Whitlock EP, Lin JS, Liles E, et al. Screening for colorectal cancer: a targeted, updated systematic review for the U.S Preventive Services Task Force. Annals of Internal Medicine. 2008;149(9):638–658.
32. Wolf AM, Wender RC, Etzioni RB, et al. American Cancer Society guideline for the early detection of prostate cancer: update 2010. CA: A Cancer Journal for Clinicians. 2010;60(2):70–98.