Synovial Fluid Analyses, Synovial Biopsy, and Synovial Pathology
Hani S. El-Gabalawy
Analysis of synovial fluid and synovial tissue obtained from diseased joints provides important diagnostic information in specific clinical settings and is valuable in addressing a spectrum of research questions about of the pathogenesis and mechanisms of rheumatic diseases. Many peripheral joints are readily accessible to sampling of both synovial fluid effusions and synovial tissue, although the knee is the most frequently sampled joint. The techniques used to obtain and analyze synovial fluid and tissue samples are discussed in this chapter.
Synovial Fluid Analysis
Synovial Fluid in Health
Under normal conditions, a small volume of synovial fluid is present in each joint, forming a thin interface between the surfaces of the articular cartilage, and providing for friction-free movement of these surfaces. In a large joint such as the knee, the volume of synovial fluid is estimated to be less than 5 mL. Moreover, intra-articular pressure is typically subatmospheric. Compositionally, normal synovial fluid is an ultrafiltrate of plasma to which proteins and proteoglycans are added by fibroblast-like synoviocytes in the lining layer. Most of the small-molecular-weight solutes such as oxygen, carbon dioxide, lactate, urea, creatinine, and glucose diffuse freely through the fenestrated endothelium of the synovium and are normally present at levels comparable with plasma levels. Evidence for active transport of glucose has been found. The total protein concentration of normal synovial fluid is 1.3 g/dL. The concentration of individual plasma proteins is inversely proportional to the molecular size, with small proteins such as albumin present at approximately 50% of plasma levels, and large proteins such as fibrinogen, macroglobulins, and immunoglobulins present at low levels. In contrast to this selective entry on the basis of size, clearance of synovial fluid proteins through the synovial lymphatics is unrestricted by size. Hyaluronan is the major proteoglycan synthesized by synovial cells and secreted into synovial fluid. Hyaluronan is highly polymerized and reaches molecular weights exceeding one million Daltons, which gives this fluid its characteristic viscosity. The hyaluronan also acts to retain small molecules in the synovial fluid. The lubricating capacity of the synovial fluid is attributed to a glycoprotein called lubricin.1 This molecule has been fully characterized on the basis of the study of individuals with mutations of the PRG4 gene, which encodes for its production.2 These mutations result in an autosomal recessive loss-of-function disorder called the camptodactyly–arthropathy–coxa vara–pericarditis syndrome, which features a progressive, non-inflammatory arthropathy characterized by severe cartilage destruction associated with proliferation of synovial lining cells. The role of lubricin in maintaining the health of the cartilage has been further demonstrated in a murine knockout model.3
Accumulation of Synovial Effusions
Synovial fluid and its contents are cleared through the synovial lymphatics by a process that is aided by joint motion. Excess fluid can accumulate in any diarthrodial joint as a result of many processes, including non-inflammatory, inflammatory, and septic disorders. In addition, overt hemarthroses can result from both traumatic and nontraumatic disorders. The most important mechanism contributing to the accumulation of joint effusions is an increase in synovial microvascular permeability. This permeability allows for an increase in the efflux of plasma proteins, particularly larger proteins, which in turn increases osmotic pressure and contributes to the effusion. Leukocytes accumulate in the fluid after transmigration through the endothelium, stimulated by chemokines produced in the synovium. The capacity of synovial lymphatics to clear proteins, cells, and debris is rapidly exceeded, which in turn contributes to their accumulation in the synovial compartment.
Arthrocentesis
Most peripheral joints are readily accessible for diagnostic arthrocentesis, and the procedure can be performed in almost any ambulatory care setting equipped for sterile procedures. Joints that are less accessible because of their deeper location, such as the hip, may require an imaging technique that uses fluoroscopy or ultrasound to guide the needle and ensure accurate placement. Details of techniques used for arthrocentesis are described in Chapter 54. Because the ease with which joint fluid is aspirated depends on the gauge of the needle that is used, physicians should attempt arthrocentesis with a needle of adequate gauge, particularly in the larger joints. Moreover, high-suction gradients created by large syringes should be avoided, because they may actually reduce a physician's ability to successfully aspirate synovial fluid. Difficulty in aspiration of synovial fluid may stem from a number of intra-articular factors, including viscosity, the presence of debris such as rice bodies, and loculation of fluid into inaccessible areas. Instillation of a small amount of sterile saline may help to obtain enough fluid for culture in situations in which infection is highly suspected, yet direct aspiration is difficult.
Once obtained, it is important to analyze aspirated synovial fluid samples as quickly as possible to avoid spurious results. Ideally, leukocyte count and differential should be performed on fresh specimens. If the specimen cannot be analyzed quickly, and short-term storage is needed, the specimen should be kept at 4° C. An aliquot preferably should be placed in ethylenediaminetetraacetic acid (EDTA) to prevent clotting. Delays in analysis longer than 48 hours should be avoided. A simplified algorithm for analyzing synovial fluid samples is shown in Figure 53-1.
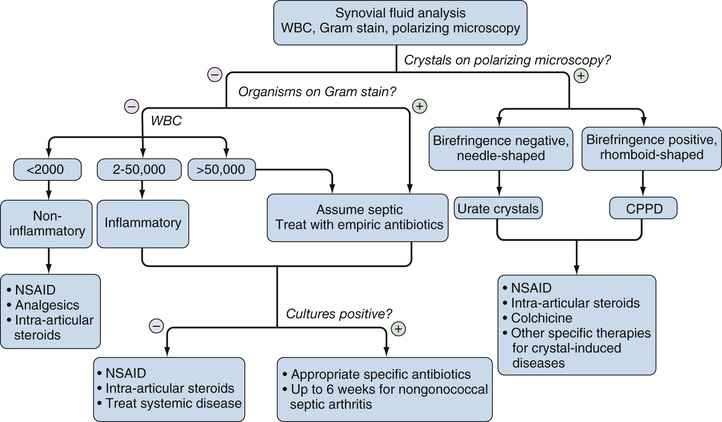
Gross Examination
The physician can get a first impression of the nature of the synovial fluid while fluid enters the syringe during the arthrocentesis procedure itself. For example, the viscosity of the fluid is apparent during this step. As has been mentioned, normal synovial fluid is highly viscous because of its hyaluronan content and forms a long string when a drop is expressed from the end of the needle. With increasing levels of inflammation associated with recruitment and activation of leukocytes in the synovial cavity, the hyaluronan is digested, resulting in loss of viscosity that is appreciated as a reduction in the “stringiness” of the fluid. Large pieces of debris such as rice bodies, thought to arise from detached ischemic synovial villi, may be visible while they are aspirated. These can cause sudden arrests in the flow of fluid into the syringe, requiring manipulation and redirection of needle placement.
Inspection of the aspirated synovial fluid can yield other important diagnostic information. For example, floridly purulent fluid will be completely opaque because of the very high number of leukocytes present, but synovial fluid that is transparent to the point at which printed text can be read through it is seen in non-inflammatory settings. Inflammatory synovial fluid of the type that would be aspirated from an individual with active rheumatoid arthritis (RA), appears cloudy and translucent. The degree of translucency depends on the intensity of the inflammatory response and the concentration of leukocytes in the sample. Synovial fluid from patients with ochronosis may have a speckled appearance, and particulate debris from joint prostheses may be visible on gross inspection.
During the arthrocentesis procedure, the physician may have an important challenge in determining whether the presence of blood in the aspirated synovial fluid indicates a hemarthrosis or, alternatively, is a result of trauma from the procedure itself. In the latter case, the blood may remain unmixed with the synovial fluid, and appear as red streaks in an otherwise yellow fluid In the case of hemarthroses, the synovial fluid is generally homogeneously bloody and does not form a clot. The causes of frank hemarthrosis are varied and include trauma, pigmented villonodular synovitis, tumors, hemophilia, and other bleeding disorders or anticoagulant therapy, Charcot joint, and, occasionally, intense inflammation from a chronic arthropathy such as RA or psoriatic arthritis.
Leukocyte Count
Analysis of leukocyte counts and cytology provide important diagnostic information regarding the cause of a synovial effusion (Table 53-1). A fresh specimen should be placed in a heparinized tube for rapid analysis and, if the fluid is particularly viscous, it may need to be diluted in normal saline before counting. Normal synovial fluid contains fewer than 180 nucleated cells/mm3, most of which originate as desquamated synovial lining cells. The leukocyte count broadly classifies synovial fluids as non-inflammatory (<2000 cells/mm3), inflammatory (2000 to 50,000 cells/mm3), and septic (>50,000 cells/mm3). These definitions provide broad guidelines to help narrow the differential diagnosis rather than representing inherent biologic properties of the fluid.
TABLE 53-1
Characteristics of Synovial Fluid
| Appearance | Viscosity | Cells per mm3 | % PMNs | Crystals | Culture | |
| Normal | Transparent | High | <200 | <10% | Negative | Negative |
| Osteoarthritis | Transparent | High | 200-2000 | <10% | Occasional calcium pyrophospate and hydroxyapatite crystals | Negative |
| Rheumatoid arthritis | Translucent | Low | 2000-50,000 | Variable | Negative | Negative |
| Psoriatic arthritis | Translucent | Low | 2000-50,000 | Variable | Negative | Negative |
| Reactive arthritis | Translucent | Low | 2000-50,000 | Variable | Negative | Negative |
| Gout | Translucent to cloudy | Low | 200->50,000 | >90% | Needle-shaped, negatively birefringent monosodium urate monohydrate crystals | Negative |
| Pseudogout | Translucent to cloudy | Low | 200-50,000 | >90% | Rhomboid, positively birefringent calcium pyrophosphate crystals | Negative |
| Bacterial arthritis | Cloudy | Variable | 2000->50,000 | >90% | Negative | Positive |
| PVNS | Hemorrhagic or brown | Low | — | — | Negative | Negative |
| Hemarthrosis | Hemorrhagic | Low | — | — | Negative | Negative |

PMNs, Polymorphonuclear neutrophils; PVNS, pigmented villonodular synovitis.
The most common causes of non-inflammatory synovial fluids are mechanical derangements of the joint and osteoarthritis. Other causes include endocrinopathies such as acromegaly and hyperparathyroidism; inherited disorders such as ochronosis, hemochromatosis (which can also present with hemarthrosis), Ehlers-Danlos syndrome, Wilson's disease, and Gaucher's disease; acquired disorders such as Paget's disease, avascular necrosis, and osteochondritis dissecans; and an uncommon condition called intermittent hydrarthrosis, in which joints become effused in a cyclic manner. At the other extreme, leukocyte counts of 50,000 to 300,000 cells/mm3 are most commonly associated with septic arthritis and should prompt the clinician to empirically treat the individual as such until this diagnosis is excluded with a high degree of certainty, which typically requires definitive culture results and, possibly, repeat aspiration. Leukocyte counts exceeding 50,000 cells/mm3 are often seen in acute crystal-induced arthritis, particularly gout. Inflammatory cell counts between 3000 and 50,000 cells/mm3 are seen in a wide spectrum of articular disorders, including many cases of septic arthritis. Thus, most patients with acute attacks of gout and pseudogout, active RA, reactive arthritis, and psoriatic arthritis, as well as patients with gonococcal arthritis and other nonpyogenic forms of septic arthritis, will typically present with synovial fluid cell counts in this range (see Table 53-1).
Synovial Fluid Cytology
Characterization of the cells present in synovial fluid is an important diagnostic step that the examiner can achieve initially by performing cytology on a wet mount of the synovial fluid. To perform the wet mount analysis, the examiner places a single drop of synovial fluid on a clean glass slide, which then is covered by a coverslip and examined under low- and high-power light microscopy. In addition to leukocytes, and in the case of traumatic taps or hemarthroses, or large numbers of erythrocytes, wet mount may reveal the presence of clumps of fibrin and crystals, cartilage and synovium fragments, and lipid droplets. These can all appear as amorphous material, and care should be taken to avoid assuming their composition without further characterization.
Characterization of synovial fluid leukocytes is best achieved by staining a dried smear of the fluid. Wright stain is most commonly used for this purpose. The phenotype and morphology of the leukocytes can then be assessed under high power by using oil immersion. Septic-range synovial fluid containing more than 50,000 cells/mm3 is almost always associated with a high preponderance of polymorphonuclear leukocytes, often greater than 90%. Monocytes and lymphocytes predominate in the synovial fluid of patients with viral arthritis, lupus, and other connective tissue diseases. Synovial fluid samples from patients with active RA, reactive arthritis, psoriatic arthritis, and acute attacks of crystal-induced arthritis typically demonstrate a preponderance of polymorphonuclear leukocytes, although fluids from patients with early-stage RA may have a low leukocyte count with primarily mononuclear cells. The presence of large numbers of “ragocytes,” which are granulocytes that have engulfed immune complexes, is associated with active RA, and their presence may indicate an unfavorable prognosis in this disease.4 Reiter's cells represent cytophagocytic mononuclear cells that have phagocytized apoptotic polymorphonuclear leukocytes. This may represent a pathway by which autolysis and release of damaging mediators from the latter cells are avoided.5 The presence of Reiter's cells is not specific for reactive arthritis, nor indeed for spondyloarthropathies in general. Occasionally, eosinophils will predominate in the synovial fluid, which may be associated with parasitic infection, urticaria, or hypereosinophilic syndrome. It has been suggested that cytocentrifugation of synovial fluid is the optimum method for performing cytopathology, although the cost-effectiveness of this technique is questionable in most clinical settings.
Wet Smear Analysis by Polarized Microscopy
A search for crystals by using polarized microscopy is particularly valuable in the diagnosis of acute monoarthritis or oligoarthritis, in which gout and pseudogout are often on the differential diagnosis. In such a clinical situation, if indeed the absence of pathogenic crystals in synovial fluid can be established, the likelihood of septic arthritis increases, prompting the initiation of intravenous antibiotics and potentially necessitating a hospital admission. Thus the rapid and accurate diagnosis of a crystal-induced process can prevent a costly and unnecessary sequence of events. It is helpful if the individual or the team that performs the arthrocentesis can also rapidly examine the specimen by polarized microscopy. For this procedure, the examiner requires a functional polarizing microscope, as well as adequate experience in the identification of crystals using this technique. This is particularly important in the case of calcium pyrophosphate crystals, which are notoriously difficult to detect.
The examiner should take care that the slide and the coverslip are free of dust, talc, and other particulate matter. Crystals present in the specimen rotate the light in such a way that they appear as bright objects in an otherwise dark field. Birefringent debris frequently are scattered throughout the slide and should not be mistaken for crystals.
The first-order red compensator is usually inserted immediately below the upper filter and serves to block out green light. Birefringent material in the specimen appears as a bright yellow or blue color in the red field generated by the first-order compensator. While birefringent crystals are rotated relative to the axis of the first-order compensator, the color changes from yellow to blue, or vice versa. Crystals that are yellow when oriented parallel to the axis of the compensator are negatively birefringent, and those that are blue are positively birefringent.
Identification of crystals in synovial fluid is greatly facilitated by detailed examination of the specimen, under both low and high power, by using the approach previously described. A combination of morphology and birefringence serves to identify the crystals. Monosodium urate (MSU) crystals, as shown in Figure 53-2, are the easiest to identify because the crystal load is typically high during an acute attack of gout. A good degree of concordance between laboratories in the identification of MSU crystals has been shown.6-8 These crystals appear as strongly negatively birefringent needle-shaped objects, many of which are intra-cellular, having been phagocytized by synovial fluid leukocytes. In contrast, calcium pyrophosphate dihydrate (CPPD) crystals seen during attacks of pseudogout tend to be smaller, rhomboid-shaped objects that are weakly positively birefringent, as shown in Figure 53-3. Because the CPPD crystal load during an attack of pseudogout tends to be relatively low, and because CPPD crystals are only weakly birefringent, it is important to examine all areas of the specimen on the microscope slide, and possibly to prepare a second wet mount to exclude or confirm this diagnosis. Concordance between laboratories in the recognition of CPPD is substantially lower than in the case of MSU crystals.6-8 A particularly challenging situation arises when intra-cellular crystals cannot be identified, yet birefringent extra-cellular objects resembling crystals are seen scattered throughout the slide. This may be caused by powder from gloves or dirt on the slides.
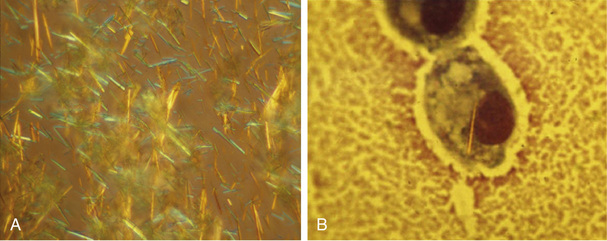
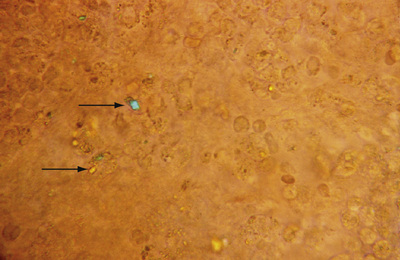
As with other analyses on the synovial fluid, the examiner should perform wet mount preparation and analysis as quickly as possible, although identification of crystals can still be successful after prolonged storage of specimens. The crystal load decreases substantially while the acute inflammatory attack subsides, thus making a specific diagnosis more difficult while the attack begins to subside. Urate crystals have been detected in synovial fluid between attacks of gout.
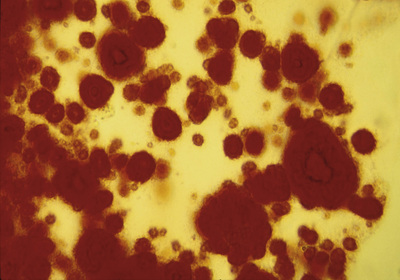
Deposits of hydroxyapatite or basic calcium phosphate are present within the joint and in periarticular locations such as around the shoulder area, and are associated with osteoarthritis. These crystals have been implicated in a particularly destructive syndrome that has been named Milwaukee shoulder.9 Hydroxyapatite can be detected in synovial fluid, but because these crystals are generally nonbirefringent, it is not possible to detect them by polarized microscopy. A useful and rapid method with which to detect hydroxyapatite and other calcium-containing crystals such as octacalcium and tricalcium phosphate is to stain the fluid with alizarin red S stain and look for clumps of crystals under routine light microscopy (Figure 53-4). These crystals have also been identified by using electron microscopy, although this method is rarely available to the practicing clinician.
Synovial cholesterol crystals appear as flat, platelike structures with notched corners (Figure 53-5), and lipid crystals have the appearance of Maltese crosses. Both can be strongly birefringent, both negatively and positively. Corticosteroid crystals can be highly birefringent and mimic urate or CPPD crystals. Large amounts of lipid in the synovial fluid can be visible on gross examination. The significance of these crystals in synovial fluid is unclear, but it is unlikely that they are pathogenic in most cases.

Detection of Microorganisms by Gram Stain, Culture, and Polymerase Chain Reaction Analysis of Synovial Fluid
A wide spectrum of organisms can cause septic arthritis, although the most common pathogens are Gram-positive bacteria such as staphylococci and streptococci. Because septic arthritis causes rapid destruction of the joint, and because it can spread hematogenously to other areas and is associated with significant mortality, it is imperative that a specific diagnosis be made as quickly as possible, and that empiric therapy with broad-spectrum antibiotics be started until this diagnosis can be confirmed or excluded.
A Gram stain performed on fresh synovial fluid will identify an organism in an estimated 50% of cases of septic arthritis,10 with the highest sensitivity for Gram-positive organisms. Moreover, the specificity of a positive Gram stain approaches 100%. Clearly, this indicates that the positive predictive value for the Gram stain is very high, and that the negative predictive value is substantially lower. The gold standard for diagnosing septic arthritis is still bacteriologic culture, which has a sensitivity of 75% to 95% and a specificity of 90% in cases of nongonococcal septic arthritis.11,12 The use of blood culture bottles further increases the yield of positive synovial cultures.13 Bacteriologic cultures are the only studies that provide a guide for specific anti-microbial therapy. Because the sensitivity of bacteriologic cultures declines dramatically after antibiotic therapy is instituted, it is important that the clinician perform arthrocentesis before any antibiotics are administered. Cultures should be performed even when uric acid or other crystals are demonstrated in the synovial fluid because gout and septic arthritis can coexist.14 In the case of gonococcal arthritis, the sensitivity of bacteriologic culture, even if performed on a sample collected by using appropriate media, is low, with an estimate of less than 10%.
Polymerase chain reaction (PCR) carries a high degree of sensitivity and specificity for the detection of microorganisms in synovial fluid and tissue, even in individuals who are culture negative.15 Most bacteria can be detected on the basis of amplifying specific sequences in their ribosomal RNA (16S rRNA). PCR is now the procedure of choice for diagnosing gonococcal arthritis,16,17 and is a highly sensitive and specific method of detecting tuberculous arthritis, although as discussed later, analysis of synovial tissue is better than analysis of synovial fluid for making this diagnosis.18,19 PCR is also a method of verifying the successful elimination of the offending organism in cases of septic arthritis.20,21
The sensitivity and specificity of PCR in detecting synovial microorganisms should be balanced against the biologic significance of a positive test. Contaminants are easily detected by using this method, and highly stringent conditions for sample collection are required to prevent false-positive tests. Moreover, PCR studies of synovial fluid and tissue from a spectrum of chronic forms of arthritis, including RA, osteoarthritis, reactive arthritis, and undifferentiated arthritis, have indicated the presence of microorganisms in a significant number of specimens.22,23 The biologic significance of these findings and the potential role of bacterial DNA or cell wall fragments in the pathogenesis of these arthropathies remain unclear.
Biochemical Analysis of Synovial Fluid
A number of widely available biochemical tests may add to the diagnostic impression of aspirated synovial fluid samples, although lack of specificity of these biochemical analyses tends to limit their value.12,24 Testing for synovial fluid glucose, protein, and lactate dehydrogenase (LDH) has long been included in routine practice, and values obtained should be compared with serum values. Samples from septic arthritis typically exhibit very low glucose, low pH, and high lactate levels; these levels are indicative of a switch to anaerobic metabolism. Highly inflammatory synovial fluids from RA exhibit a similar profile, along with high protein and LDH levels. Levels of pressure of oxygen (pO2) in the blood are often in the hypoxic range in RA synovial fluids and are correlated with increased lactate and levels of pressure of carbon dioxide (pCO2) in the blood.25,26 A prospective study conducted to evaluate these tests in a spectrum of inflammatory and non-inflammatory disorders demonstrated considerable variability in each diagnostic category, which limits their clinical utility.24
Serologic testing of synovial fluid to detect rheumatoid factor, anti-nuclear antibodies, and complement levels has been suggested as a method that can be used to confirm a diagnosis of RA or other connective tissue diseases. In particular, RA synovial fluids may be positive for rheumatoid factor even when serum is not,27 and complement levels are typically low as a result of consumption by immune complexes. These findings are of insufficient sensitivity and specificity to be of value on a routine clinical basis.
Synovial Fluid Analysis in Arthritis Research
The ease with which synovial fluid is aspirated from inflamed joints has allowed a wide spectrum of research studies to be conducted on this biologic material. In research settings, cells in synovial fluid samples are typically separated by centrifugation, and cellular and noncellular components of the fluid are analyzed separately. Detailed analysis of the phenotype and functional properties of synovial fluid leukocytes has been particularly informative in RA and reactive arthritis research, in which immunophenotyping of lymphocyte subpopulations has provided important clues to the pathogenesis of these diseases. In the case of reactive arthritis, in which triggering organisms are often identified, the proliferative and cytokine responses of synovial fluid lymphocytes to antigens derived from Chlamydia, Yersinia, and other pathogens have been elucidated.28,29 Synovial fluid T cells from reactive arthritis patients are biased toward production of T helper (Th)2 cytokines such as interleukin (IL)-10 and IL-4, whereas synovial fluid T cells from patients with RA are Th1 biased and exhibit defects in Th2 differentiation.30-32
Analysis of the noncellular portion of synovial fluid has provided important information regarding a spectrum of soluble molecules, including cytokines and growth factors,33 extra-cellular matrix proteins, autoantibodies, and therapeutic drug levels. Moreover, broad-based proteomic studies of synovial fluid with the use of fractionation techniques and mass spectrometry are beginning to provide novel approaches to understanding pathogenesis and prognosis in arthropathies such as RA.34
Synovial Biopsy
Sampling of synovial tissue is a direct approach to defining the pathologic processes that cause swollen, painful joints.. In clinical settings, it can be particularly valuable in evaluating an undiagnosed persistent monoarthritis when other investigations, including synovial fluid analysis, have failed to provide a specific diagnosis. In research settings, analysis of synovial tissue samples has dramatically improved our understanding of the pathogenetic mechanisms underlying RA, spondyloarthropathies, and other chronic articular disorders. More recently, synovial biopsy has been explored as a method for defining the target tissue response to therapeutic agents, particularly targeted biologic therapies.
Blind Percutaneous Synovial Biopsy
Percutaneous needle biopsy is most commonly performed according to the method originally described by Parker and Pearson,35,36 utilizing a biopsy needle that now carries their name. Percutaneous synovial biopsy is most often performed on the knee joint, although the technique can readily be adapted for use in other joints such as the wrist, elbow, ankle, or shoulder. A modification of the original Parker-Pearson needle has facilitated synovial biopsy of small hand joints such as metacarpophalangeal and proximal interphalangeal joints.37 The technique for Parker-Pearson synovial biopsy uses a 14 gauge needle with a lateral aperture just proximal to the inserted end of the needle. This lateral opening features a sharp cutting edge for severing trapped synovial tissue that is captured by applying suction with a 3 to 5 mL syringe. With this approach, multiple 1 to 3 mm samples are obtained by angling the trocar in several directions. This also serves to minimize the sampling error involved. Synovial samples are typically pink and are easily removed with a slight twisting motion. Because of the blind nature of the procedure, samples of fat, muscle, or fibrous tissue may be obtained and need to be separated from true synovial samples.
Percutaneous synovial biopsy is easily performed in most ambulatory care settings with the use of relatively inexpensive equipment, and recently ultrasound guidance has improved the yield. The overall morbidity of the procedure is low and is comparable with that of arthrocentesis, with perhaps a slightly higher rate of hemarthrosis. The risk of hemarthrosis can be minimized if the patient does not bear weight for a few hours after the procedure. The main disadvantage of the procedure is its blind nature. In comparison with visually guided arthroscopy, samples derived from the interface between synovium and adjacent cartilage are underrepresented when the blind procedure is used.38,39 As discussed later, this drawback is particularly relevant to a number of research questions.
Arthroscopically Guided Synovial Biopsy
Arthroscopy is widely used by orthopedic specialists for the diagnosis and treatment of a variety of articular disorders, particularly mechanical derangements of intra-articular structures such as cruciate ligaments and menisci. During the past two decades, the arthroscopic procedure has been adapted for diagnostic synovial biopsies in settings that do not require a fully equipped operating theater and general anesthetic. In most cases, intra-articular local anesthesia suffices for the procedure, although conscious sedation may be required in some individuals. The procedure is well tolerated and is associated with low morbidity, although the risks to the patient of hemarthrosis and infection after the procedure are slightly higher than that of percutaneous needle biopsy. The patient should be instructed to minimize weight bearing for 24 to 48 hours after the procedure.
The primary advantage of arthroscopy is its ability to visually guide the biopsy procedure. This permits macroscopic evaluation of the synovium and sampling of areas that appear to be severely affected by the pathologic process, and it allows for sampling of the interface between inflamed synovium and adjacent cartilage. This interface is an area of particular interest for understanding the pathogenesis of destructive arthropathies such as RA.38 As with samples obtained by percutaneous synovial biopsy, individual samples are allocated for specific laboratory studies depending on the clinical or research question under investigation.
Processing Synovial Tissue Samples
In all cases, an adequate number of individual synovial specimens need to be allocated for routine light microscopy with the use of formalin fixation and paraffin embedding. This provides the highest-quality sections for hematoxylin and eosin (H&E) histologic analysis, and it allows the most accurate delineation of pathologic processes within tissue. Although formalin-fixed sections can be used in some cases for immunohistology, formalin fixation alters the conformation of many protein antigens, making them inaccessible for specific identification by immunohistology. Many of the molecular markers used to analyze diseased synovium, including cell surface markers, cytokines, adhesion molecules, and proteases, require that tissue samples be snap frozen in a suitable mounting medium, such as optimal cutting temperature compound, and then sectioned with the use of a cryostat. The sections can be processed by using antigen-specific monoclonal or polyclonal antibodies, and color development achieved by one of several immunofluorescence or immunoperoxidase methods. Typically, a nuclear counterstain is also used to assist in orientation of the tissue—hematoxylin in the case of immunoperoxidase studies. If only formalin-fixed, paraffin-embedded tissue is available, an alternative method for detecting antigens that are sensitive to formalin fixation is antigen retrieval.
Several antigen retrieval methods are available, including enzymatic and thermal methods,40 which have been used to successfully retrieve a spectrum of antigens from archival synovial tissue samples for immunohistologic studies, although the quality of the tissue sections often deteriorates after antigen retrieval. A number of double-staining immunohistology techniques have been developed for simultaneous evaluation of the expression of two markers in the same tissue section, although these techniques are labor intensive and often require considerable experimentation to generate good stains.41 Formalin fixation dissolves crystals, and if this is a diagnostic consideration, the specimen should be fixed in ethanol.
The sensitivity and specificity of molecular DNA and RNA techniques provide unprecedented opportunities to explore the pathogenesis of synovial disorders. Although these studies can be carried out on very small quantities of tissue, examiners need to take great care in handling and processing tissue samples to prevent degradation of the nucleic acids, particularly with RNA when RNAase enzymes are ubiquitous, and can rapidly degrade the small quantity of RNA present in a tissue sample. As discussed later, the search for microbial DNA and RNA has been of particular interest in attempts to understand the cause and pathogenesis of reactive arthritis, RA, and other forms of chronic synovitis of unknown cause. Techniques used to analyze human gene expression in small tissue samples have rapidly progressed. This has enabled the detection and quantitation of multiple mRNA transcripts in very small quantities of biopsy material, in many cases without the need for amplification.42,43
Synovial Pathology
Synovial Membrane in Healthy Individuals
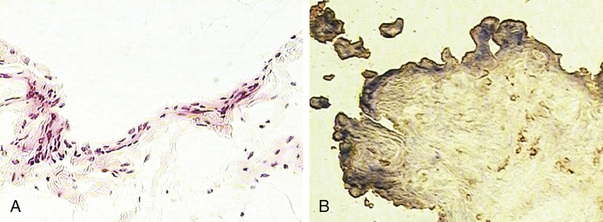
A detailed description of the composition of normal synovium is provided in Chapter 2. Histologically, the normal synovial lining layer is one to three cells thick and is composed of closely associated macrophage-like (type A) and fibroblast-like synoviocytes (type B) that are not separated from the underlying stroma by a basement membrane, as is the case with a true epithelium. In many areas, visible gaps in this lining layer allow small molecules to easily diffuse through the extra-cellular matrix into the synovial fluid. The two types of lining layer synoviocytes are distinct and can be differentiated on the basis of ultrastructural and immunohistologic features. Macrophage-like synoviocytes are myeloid in origin, as they exhibit the morphologic characteristics of phagocytic cells and express macrophage markers such as CD68, CD14, and FcγRIIIa. Fibroblast-like synoviocytes are synthetic cells of mesenchymal origin that are the primary source of hyaluronan and other proteoglycans found in normal synovial fluid. They express CD55 (decay-accelerating factor [DAF]), high levels of vascular cell adhesion molecule (VCAM)-1, and the enzyme uridine diphosphoglucose dehydrogenase (UDPGD), which is involved in the synthesis of hyaluronan and has been detected by cytochemical methods (Figure 53-6). Fibroblast-like synoviocytes have also been shown to uniquely express cadherin-11, a specialized adhesion molecule that is involved in homotypic aggregation of these cells and that contributes to maintaining the integrity of the synovial lining layer.44 Quantitatively, most of the cells in the normal synovial lining layer are type B cells. The underlying stroma features a rich network of capillaries with fenestrated endothelium in the immediate sublining area that serve to maintain the health and viability of adjacent cartilage. Larger arterioles and venules can be found deeper in the synovial stoma. The synovial microvasculature is surrounded by loose connective tissue, which also incorporates the synovial lymphatics that serve to drain this tissue. The synovium of completely asymptomatic individuals not uncommonly exhibits a modest infiltrate of T lymphocytes that are occasionally organized in perivascular aggregates, although B cells were not seen.45
Synovial Histopathology in the Evaluation of Monoarthritis
Pathologic analysis of synovial tissue samples can be of considerable value in certain clinical settings. However, the histopathologic interpretation of synovial biopsy specimens is often nondiagnostic and lacks specificity.46 Pathologic analysis of synovial samples from patients with undiagnosed monoarthritis may be of particular value. The presence of large numbers of neutrophils in the synovial tissue stroma is highly suggestive of septic arthritis, and in such cases Gram stain may reveal bacteria in the tissue. Because septic arthritis is usually acute in onset, synovial biopsy is rarely required, and the diagnosis can be made by analyzing synovial fluid as described previously. Gonococcal arthritis may require synovial biopsy for diagnosis (Figure 53-7). A mononuclear cell infiltrate, on the other hand, is more consistent with a chronic inflammatory process and has a wide differential diagnosis, as described previously. The presence of granulomas supports a diagnosis of tuberculous arthritis or sarcoidosis, both of which cause chronic monoarthritis. The synovial granulomas of tuberculosis (TB) may be caseating or noncaseating, and staining of the tissue for acid-fast bacilli, culture, and molecular probing can yield a definitive diagnosis in an estimated 50% of cases. Similarly, a spectrum of fungal infections can be diagnosed by using similar approaches, but special stains such as a Gomori stain may be required. The diagnosis of sarcoid arthropathy is suspected in synovial specimens with noncaseating granulomas in cases where mycobacterial or fungal infection has been excluded.
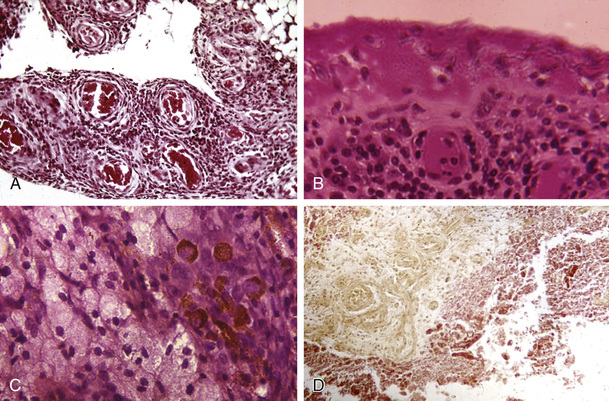
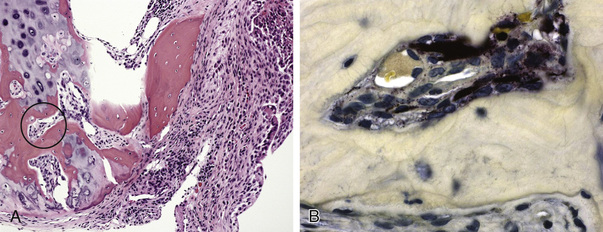
Pigmented villonodular synovitis is an important consideration in individuals with chronic monoarthritis of a large joint such as the knee or hip. This disorder has a characteristic MRI appearance caused by hemosiderin deposits in the synovium and large cystic lesions in adjacent bone. Histopathologic analysis of the synovium can confirm this diagnosis and demonstrates a diffusely hypervascular proliferative lesion with mononuclear cells of the monocyte/macrophage lineage, foamy multinucleated cells resembling osteoclasts, and hemosiderin deposits47 (see Figure 53-7). Synovial sarcomas are rare tumors that must be diagnosed on the basis of synovial pathology.
Synovial Histopathology in the Evaluation of Polyarthritis
In current clinical practice, the availability of well-validated diagnostic criteria and specific serologic tests, combined with a relative lack of specificity in synovial histopathologic features, limits the clinical utility of synovial pathology in the differential diagnosis of oligoarthritis and polyarthritis. On the other hand, analysis of synovial tissue samples obtained in the context of research studies from patients with RA and various spondyloarthropathies has dramatically enhanced our understanding of the cellular and molecular mechanisms of these disorders. This is reflected in a large body of literature published during the past three decades.38,48
RA synovium has been the most extensively studied histopathologically, and a detailed discussion of RA synovitis can be found in Chapter 69. The two characteristic features seen in RA synovitis are hyperplasia of the lining layer and infiltration of the sublining stroma with mononuclear cells (Figure 53-8). The surface of the lining layer is often covered with fibrin deposits generated from activation of the fibrinolytic system in inflammatory synovial fluid. Occasionally, the synovial lining layer is completely denuded and is replaced by a dense fibrin cap. In highly inflamed tissues, fibrin deposits extend deeply into the sublining stroma, which may be edematous because of the marked increase in vascular permeability. The earliest synovial changes in RA appear to feature microvascular abnormalities,49 and mononuclear cell infiltrates have been detected in asymptomatic joints of patients with RA.50,51 These features are nonspecific and are seen in the synovium of acutely inflamed joints from a spectrum of disorders, including reactive arthritis and psoriatic arthritis.
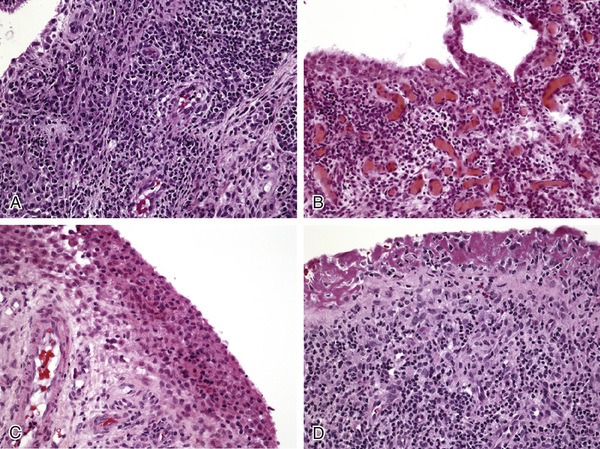
In RA, the mononuclear cell infiltrate in the sublining stroma can be diffuse but more commonly is arranged in perivascular aggregates that resemble lymphoid follicles (see Figure 53-8). Although the presence of lymphoid aggregates in the synovial membrane is typical of RA, this histopathologic feature is by no means unique to RA synovitis.52-55 Lymphoid follicles are typically located near vessels with tall endothelium, which are termed high endothelial venules; these vessels specialize in the recruitment of lymphocytes (Figure 53-9). Multinucleated giant cells are occasionally seen in RA synovium (Figure 53-10), and some tissues demonstrate granuloma formation. Finally, synovial tissue obtained at the time of joint arthroplasty often exhibits extensive fibrosis and may be indistinguishable from arthroplasty samples obtained from patients with osteoarthritis.
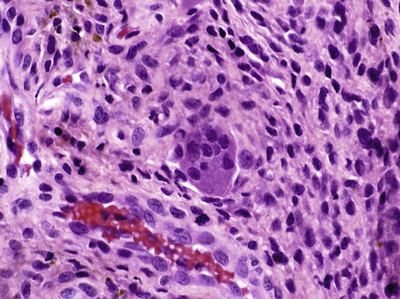
The synovial histopathology of psoriatic arthritis, ankylosing spondylitis, and reactive arthritis has been compared with that of RA.56,57 In all cases, a similar spectrum of inflammatory cell populations has been identified, but several subtle and potentially important differences have been observed. Overall, synovial histologic and immunohistologic features of psoriatic arthritis, both oligo- and polyarticular, resemble those of other spondyloarthropathies to a greater extent than RA (see Synovial Immunohistology section below).57 Comparative studies have suggested that synovial lesions in psoriatic arthritis are more vascular than those of RA, with more tortuosity of the synovial microvasculature.58,59 This is evident both macroscopically and microscopically. Moreover, lymphoid aggregates of various sizes were identified in 25 of 27 synovial tissue samples from patients with psoriatic arthritis, and 13 of 27 had large organized aggregates with all of the features of ectopic lymphoid neogenesis that have been associated with RA synovitis.52 Studies of synovium from the peripheral joints of ankylosing spondylitis patients have revealed intense infiltrates of lymphocytes, plasma cells, and lymphocytic aggregates.60,61 Comparisons made between the synovial lesions seen in patients with reactive arthritis and those seen in patients with early-stage RA of similar disease duration suggest that reactive arthritis synovia are less infiltrated with B lymphocytes, plasma cells, and macrophages.62,63 It should also be noted that synovium from patients with osteoarthritis often features the presence of lymphocyte aggregates, although these tend to be small and less well developed than those seen in RA.54
The synovium of lupus patients showed synovial hyperplasia, inflammatory infiltrates, vascular proliferation, edema and congestion, fibrinoid necrosis and intimal fibrous hyperplasia of blood vessels, and superficial fibrin deposits, although these changes were quantitatively modest compared with those of RA.64 In early scleroderma, the lining layer was seen to be thin with deposits of fibrin and stromal lymphocytes and plasma cells,65 and similar changes were seen in patients with dermatomyositis and polymyositis66 (see Figure 53-7). A recent study comparing the immunopathologic features of early untreated Behçet's disease with those of psoriatic arthritis noted that although a similar degree of inflammation was seen in the two disorders, synovitis in Behçet's disease demonstrated higher numbers of neutrophils and T cells than were seen in psoriatic synovitis.67
In patients with chronic crystal arthropathies, large deposits of birefringent material can be detected in the synovium.68 Amyloid arthropathy can be diagnosed by demonstrating amyloid deposits in the synovium with Congo red staining (see Figure 53-7). The synovium in ochronosis contains brownish shards of cartilage.69 Multicentric reticulohistiocytosis can be diagnosed pathologically by the presence of large foamy cells and multinucleated cells in the synovium. In arthritis of hemochromatosis, the synovium exhibits brown hemosiderin deposits in the lining cells, and CPPD crystals can also be observed.70
Synovial Immunohistology
Sampling Error and Quantitative Analysis
Immunohistology utilizes specific monoclonal or polyclonal antibodies with well-defined molecular targets and is an effective tool for analyzing the cellular and molecular features of the synovium. While the field has progressed during the past two decades, it has become clear that algorithms for generating reproducible quantitative data from immunohistologically stained sections are required. Moreover, approaches are needed for minimizing the sampling bias that is inherent in biopsy-based studies.71 Studies have suggested that if six or more individual specimens from different parts of the joint are examined, variance is reduced to less than 10% for T cell and activation markers.72 Furthermore, synovial inflammatory features are similar in areas adjacent to and distant from the pannus cartilage junction, with the possible exception of macrophage numbers, which tend to be higher in adjacent areas.73,74
Various methods have been proposed by which quantitative data for immunohistologically stained synovial tissue sections can be generated.75,76 The easiest and least costly method is to generate semiquantitative scores of staining intensity (e.g., on a 0 to 3 scale) from multiple areas of the tissue, and on the basis of these to obtain an average score for the entire tissue. The reliability and reproducibility of this method are increased if two observers score the tissue sections independently and a final average of the scores is generated. Computer-assisted image analysis involves capturing images from multiple areas of the tissue samples to which color-specific quantitative software algorithms are then applied. This method generates the greatest quantity of reproducible data but requires expensive equipment and certain levels of operator skill. Furthermore, differences in the background staining intensity of individual sections can make this type of analysis technically difficult.
Synovial Lining Cell Layer
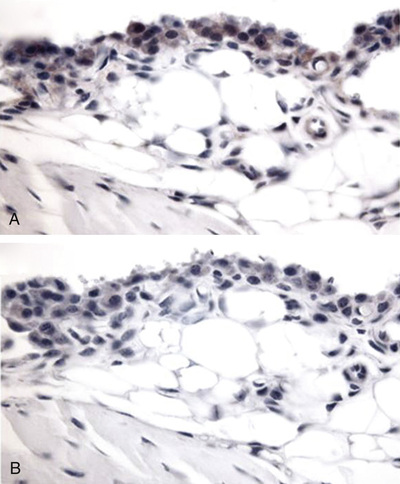
Compared with normal synovium, the lining layer in RA and other forms of chronic inflammatory arthritis is often hyperplastic, resulting from an increase in both type A and type B cells, as indicated by an increase in CD68 and CD55 staining, respectively (Figure 53-11). It is assumed that macrophage-like synoviocytes are recruited from the blood and then migrate through the synovial stroma and ultimately are retained in the lining layer in close association with fibroblast-like synoviocytes. It is currently proposed that the increase in fibroblast-like synoviocytes may be related more to defects in apoptosis than to recruitment or local proliferation. Expression of several families of adhesion molecules by both types of lining cells results in their close association and modulates their activation status. These include β1 and β2 integrins and their respective immunoglobulin supergene family ligands, particularly intercellular adhesion molecule (ICAM)-1 and VCAM-1.77-79 Cadherin-11 expressed by fibroblast-like cells likely plays a key role in the adhesive interactions that sustain the lining layer hyperplasia.44 This adhesion molecule is widely expressed in the lining layer of normal cells, as is shown in Figure 53-12. The relationship between fibroblast-like synoviocytes in the lining layer and other populations of mesenchymal cells in the sublining stroma remains uncertain. Immunohistology indicates that expression of CD55, VCAM-1, and cadherin-11 is primarily seen in the lining cell layer with minimal evidence of expression in sublining fibroblast populations. Similarly, our understanding of the relationship between lining layer macrophage-like cells and sublining macrophages is incomplete, and both express widely used macrophage markers such as CD68 and CD14. Seminal work from Edwards and associates has suggested that macrophage-like lining cells preferentially express FcγRIIIa receptors, which may serve to localize immune complexes to the synovium.80
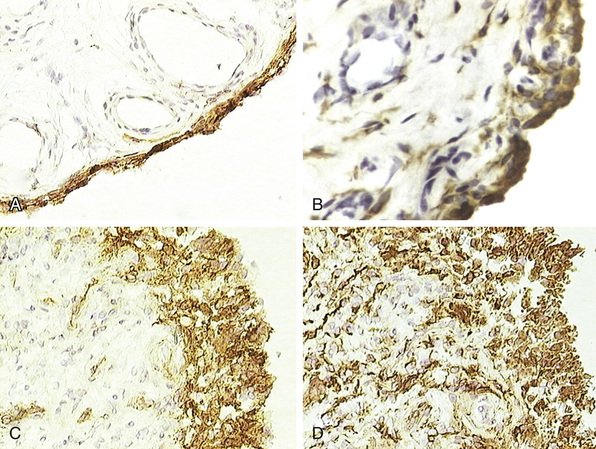
Functionally, the lining cell layer in chronic inflammatory arthropathies such as RA and psoriatic arthritis typically has the appearance of being activated. Human leukocyte antigen (HLA)-DR is highly expressed, particularly by macrophage-like cells, which may suggest a role for these cells in antigen presentation.81 Several studies have indicated that cells in the lining layer are the principal source of cartilage-degrading proteases, particularly matrix metalloproteinase (MMP)-1 and MMP-382,83 (Figure 53-13). The lining layer is generally less hyperplastic in spondyloarthropathies such as psoriatic arthritis and reactive arthritis compared with RA.57,61,84 Less is known about the functional state of the lining cells in these disorders, although it is likely that differences compared with RA are quantitative rather than qualitative.
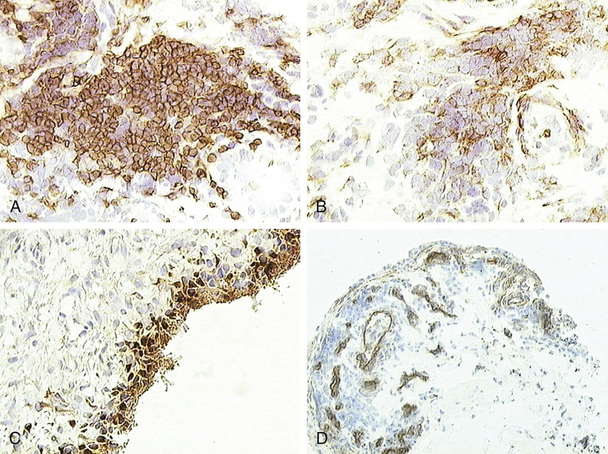
Synovial Lymphocytes and Plasma Cells
A predominance of CD3+ T cells, is found in the synovial tissues of patients with RA and spondyloarthropathies, and the CD4/CD8 ratio is 4 : 1 or greater in the lymphocytic aggregates but is lower in more diffuse infiltrates. Moreover, the CD4 cells in the aggregates also express CD27,85 which facilitates B cell help. Considerable attention has been paid to whether the infiltrating T cells in RA and other arthropathies are primarily Th1 (interferon [IFN]-γ producing) or Th2 (IL-4 producing) biased, but the data in this area have been inconsistent. Until recently, it was suggested that T cells in synovium from patients with RA are more Th1 biased compared with those in synovium from patients with spondyloarthropathies, with a higher Th1/Th2 cytokine ratio.86 Identification of a third subset of Th cells that express IL-17 and play a central role in chronic inflammatory disorders has necessitated a revision in the role that T cells play in synovitis.87,88 The presence of IL-17, IL-1β, and tumor necrosis factor (TNF) in RA synovium was found to be predictive of progressive damage.89 Furthermore, a subset of CD4 T cells expressing CD25 and the gene FoxP3, so-called regulatory T cells (Tregs), are now known to play a regulatory role in antigen-specific T cell expansion. Although Tregs are readily detected in the joints of patients with RA and other inflammatory arthropathies, their suppressor function appears to be defective in this microenvironment.90-93 It has been suggested that CD8 T cells are needed to maintain the structure of ectopic lymphoid-like structures in RA synovium, even though the numbers of these T cells typically are substantially lower than the numbers of CD4+ cells.94
B cells are identified by expression of CD19 and CD20 and are particularly abundant in tissues that exhibit large lymphoid aggregates with germinal centers. B cells typically are found in close association with CD4+ T cells in these aggregates (see Figure 53-9). Experiments in severe combined immunodeficiency (SCID) mice suggest that B cells may be critical for maintaining the microarchitecture of synovial lymphoid follicles and for T cell activation.95 Memory B cells are efficient antigen-presenting cells, and rheumatoid factor–producing B cells are well suited for capturing a wide spectrum of antigens in immune complexes.
The areas surrounding the lymphoid aggregates are often densely infiltrated with sheets of CD38+ plasma cells. Analysis of V gene variants and rearrangements in B cells and plasma cells in both RA and reactive arthritis synovium indicates that plasma cells from a particular aggregate are clonally related, suggesting that their terminal differentiation occurred in the synovial microenvironment.96 Synovial plasma cells actively synthesize immunoglobulin, some of which result in the production of autoantibodies such as anticitrulline antibodies, which recognize local citrullinated antigens.97-99 As stated previously, plasma cell infiltrates are also seen in psoriatic arthritis, ankylosing spondylitis, and reactive arthritis synovium, although a systematic analysis of synovial samples from patients with early arthritis has suggested that their presence is most suggestive of RA.62 One study found that intra-cellular citrullinated proteins were detected in RA but not in spondylarthropathy synovium.57 In contrast, another study found that the presence of citrullinated proteins was not specific for RA synovitis.100
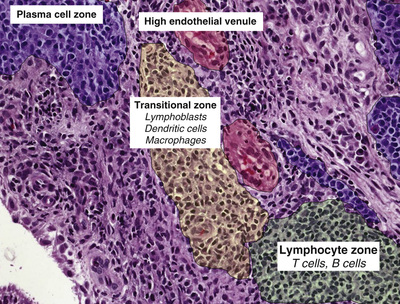
The areas immediately adjacent to the dense lymphoid aggregates, which comprise primarily CD4+ T cells and B cells, have been called transitional zones101,102 (see Figure 53-9). These areas feature a lower CD4/CD8 ratio and appear to be particularly active immunologically. Transitional areas are rich in macrophages and interdigitating dendritic cells, both of which are highly efficient antigen-presenting cells. Lymphoblasts, in particular CD8+ T cells, are seen to be present in close proximity to antigen-presenting cells.
Natural killer cells can be identified by cell surface markers, expression of granzymes, and functional assays. Several studies have suggested an expansion of subsets of natural killer cells in RA synovial tissue and synovial fluids.103-105 Mast cells are abundant in RA synovium and co-localize with inflammatory mediators and proteases in the synovial microenvironment.106,107
Synovial Sublining Macrophages and Dendritic Cells
Macrophages are present in the sublining areas of healthy and chronically inflamed synovium and are particularly abundant in the sublining stroma of RA synovium. Indeed, when markers such as CD68 and CD14 are used to study highly inflamed tissues, no clear distinction can be made between the sublining macrophage population and the macrophage-like synoviocytes present in the hyperplastic lining layer, although expression of complement receptor for C3b and iC3b was shown to be unique for lining macrophages.108 Studies that used various macrophage markers suggest that recently migrated macrophages in perivascular areas express CD163 brightly, in addition to expressing CD68 and CD14, whereas macrophages in large lymphocytic aggregates and in the lining layer are less likely to express CD163. CD163+ macrophages, which have recently been called M2 macrophages, were found to be more abundant in spondylarthropathy than in RA synovium.57 The functional correlates of these phenotypic differences remain unclear.109-111 M1 macrophages, which produce TNF and IL-1β, are more abundant in RA and are under-represented in psoriatic arthritis and other spondyloarthropathies in which M2 macrophages are more abundant.111 Furthermore, the number of macrophages in RA synovium, primarily of the M1 subset, correlates well with the destructive potential of the synovitis, as evidenced by erosive radiographic damage.112-114 This may reflect the highly activated status of these cells, which serve as the principal source of synovial TNF and IL-1β. A body of evidence has suggested that populations of synovial macrophages serve as osteoclast precursors that mature in the synovial microenvironment and then directly mediate erosive damage to adjacent bone.115,116
Mature dendritic cells are the most efficient and potent of the antigen-presenting cells, and are found abundantly in inflamed synovium in close contact with T lymphocytes.117,118 Two major subsets of dendritic cells have been described: myeloid dendritic cells (mDCs) and plasmacytoid dendritic cells (pDCs). They can be identified by immunohistology as stellate cells with dendrites that express high levels of HLA-DR and co-stimulatory molecules such as CD80, CD83, and CD86. mDCs express CD11c and CD1c, and pDCs express CD304.119 One study suggested that compared with psoriatic synovitis, RA synovium is particularly enriched in pDCs.119 Detailed studies that have examined the expression of chemokines involved in dendritic cell migration and recruitment suggest that a substantial proportion of dendritic cells in the synovium arrive in an immature state and subsequently undergo maturation within the synovial microenvironment in a T cell–rich area.117,118 Follicular dendritic cells in the germinal centers of large lymphocytic aggregates express the markers CD16, FDC, and VCAM-1.
Synovial Microvasculature, Endothelium, and Stromal Mesenchymal Cells
The stromal elements in RA are often expanded in parallel with the inflammatory cell infiltration. The microvasculature appears to be markedly increased, particularly in the deep sublining areas, and this expansion is presumed to relate to local stimulation of angiogenesis (see Figure 53-13). Morphometric studies have suggested that the number of vessels immediately adjacent to the lining layer is actually reduced compared with normal tissue.120 This situation, combined with the metabolic demands of this tissue, may actually produce a relatively ischemic and hypoxic environment, which is reflected in the biochemical properties of RA synovial fluid.121 Immunohistologic studies have indicated that the molecular consequences of hypoxia, particularly expression of hypoxia-inducible factor-1α (HIF-1α), a key regulator of the cellular hypoxic response, are increased in RA synovitis.122,123 Studies that have directly measured synovial tissue pO2 by using arthroscopic probes have confirmed the hypoxic nature of RA synovitis.124-126 The synovial endothelium in RA and other inflammatory arthropathies is activated by pro-inflammatory mediators in the microenvironment to express adhesion molecules such as E-selectin, ICAM-1, and VCAM-1, which are involved in the recruitment of inflammatory cells.127
Synovium-Cartilage-Bone Interface
The interface between inflamed synovium and adjacent cartilage and bone in RA and other chronic arthropathies is a site of particular interest because much of the articular damage occurs in these areas. In RA, this destructive synovial tissue is called pannus, which may spread to cover most of the surface of the cartilage and invade the bone in bare areas at the joint margin (Figure 53-14). Pannus has been pathologically characterized primarily from samples obtained at the time of joint arthroplasty, although arthroscopic studies at earlier stages of disease have attempted to characterize synovial samples adjacent to this area. Immunohistology suggests that synovial macrophages and fibroblasts are abundant at the pannus-cartilage interface, and that high levels of proteases are expressed by these cells. At the interface between pannus and bone, substantial numbers of multinucleated osteoclasts can be identified morphologically and by specific markers such as calcitonin receptors, cathepsin K, and staining for tartrate-resistant acid phosphatase128 (see Figure 53-14). Moreover, expression of receptor activator of nuclear factor-κB (NF-κB) ligand (RANKL), a key cytokine in osteoclastogenesis, was prominent in these areas.129
Synovial Biopsy and Pathology as Research Tools for Clinical Biomarker Development
Multiple academic rheumatology centers have used arthroscopic or needle synovial biopsy and quantitative immunohistology as research tools with which to develop a better understanding of disease pathogenesis. In recent years, there has been a focus on the early clinical and pre-clinical stages of RA and other forms of chronic inflammatory arthritis, in the hope of devising interventions that could achieve long-term remissions and even disease prevention. In the case of RA, it is now well established that circulating autoantibodies such as RF and anti-CCP precede clinical disease onset, often by several years. 130 What is less clear is the stage where the synovium becomes engaged in the immune-inflammatory process. To address this question, synovial biopsy studies were undertaken in autoantibody-positive individuals at high risk for RA, but without clinical evidence of synovitis.131,132 These studies failed to demonstrate significant inflammatory changes in these pre-clinical synovial samples, although there were subtle T cell infiltrates in the synovium of individuals who subsequently developed RA. These findings suggest that it is unlikely that a prolonged sub-clinical synovitis stage precedes clinical detectable joint inflammation in RA.
In individuals with established inflammatory arthritis, it has been hypothesized that analysis of the synovial lesions could provide informative biomarkers for predicting the impact of therapeutic interventions in RA and other forms of inflammatory arthritis. Such studies may be particularly valuable in predicting the effects of the expanding arsenal of targeted biologic therapies, in which the molecular target and the biologic basis of the mechanism of action are well defined.133-146 To test this hypothesis, a recent study developed a classification paradigm for RA synovial phenotypes based on gene expression profiling and immunohistology, and found that these synovial phenotypes could be used as a source of biomarkers that correlated with response to biologic therapeutics.142 Using a statistical classification algorithm derived from the synovial gene expression profile and concordant immunohistologic pattern of synovitis, the study identified four distinct phenotypes: lymphoid, myeloid, low inflammatory, and fibroid. These phenotypes, in particular the lymphoid versus myeloid patterns, served to predict response to infliximab treatment in a post-hoc analysis of a synovial biopsy-based clinical trial, with good responders significantly more likely to express the myeloid, M1 macrophage-dominated phenotype.137,138 Representative soluble biomarkers were identified from the gene sets expressed by the myeloid versus lymphoid phenotype (the latter dominated by T and B lymphocyte-associated genes). These biomarkers were sICAM-1 for the myeloid phenotype and CXCL13 for the lymphoid phenotype. The serum levels of these biomarkers were found to be correlated with the clinical response to either adalimumab, a TNF inhibitor, or tocilizumab, an IL-6 receptor inhibitor, in a randomized clinical trial.143 These results, derived from group level analyses, are encouraging, but it is too early to know how effective this approach will be in predicting response to therapeutic strategies on an individual level. They do demonstrate that synovial tissue analysis is a potentially important source of clinically relevant biomarkers.
This appealing proposition is currently hindered by several important considerations. First, the arthroscopic equipment, expertise, and infrastructure needed to undertake these studies remain limited to a small number of centers. Second, considerable concern has arisen regarding the issue of sampling bias in these studies, particularly because serial biopsies are compared in the same individual. As has been discussed, various approaches are used to minimize this bias, including systematic sampling of the same areas of the joint, computerized image analysis of multiple representative tissue samples, quantification of an adequate number of microscopic fields, and utilization of quantitative PCR and proteomic techniques to assess overall levels of specific molecules.
Conclusion
Analysis of synovial fluid and tissue samples provides valuable diagnostic information in specific clinical settings. In cases in which septic or crystal-induced arthritis is suspected, as in acute monoarthritis, synovial fluid analysis is critical for the diagnosis. In cases of undiagnosed chronic monoarthritis, synovial biopsy may provide definitive evidence of conditions such as TB, sarcoidosis, and pigmented villonodular synovitis.
Systematic analysis of synovial tissue in RA and other forms of inflammatory arthritis, particularly with the use of immunohistology, has provided a wealth of information concerning the cellular and molecular mechanisms that sustain synovial lesions. Research protocols are currently exploring the utility of synovial biopsy in predicting response to anti-rheumatic therapies.