7 Special Studies
The pathologist’s H&E is like the clinician’s H&P (history and physical) - basic exams to be performed on every patient or specimen forming the cornerstone of diagnosis. However, the pathologist is no longer limited to the H&E; there are a wide variety of special studies available to evaluate pathologic processes, from simple histochemical stains to global gene expression patterns. Pathologists are now clinical cell biologists. Familiarity with the types of special studies available is important as the initial processing of the gross specimen may limit the types of studies that can be performed.
HISTOCHEMISTRY
Almost all histochemical stains are suitable for formalin-fixed tissues. Common stains and their uses are listed in Table 7-1. However, numerous other types of stains and modifications are used and pathologists must be aware of individual laboratory practices.
TABLE 7–1 HISTOCHEMICAL STAINS
| STAIN | COMPONENTS STAINED | POSSIBLE USES AND COMMENTS |
|---|---|---|
| AFOG (acid fuschin orange G; modified Masson’s trichrome) | Evaluation of renal biopsies | |
| Alcian blue | Sometimes used to identify mucosubstances in mesotheliomas or intestinal metaplasia. Affected by pH. Hyaluronidase digestion can be used to identify hyaluronic acid. | |
| Alcian blue/PAS | Demonstrates both acid and neutral mucins | |
| Alcian yellow | Identification of H. pylori in gastric biopsies | |
| Acid-fast bacilli stains (Fite-Faraco, Ziehl-Neelson, Kinyoun) | Identification of mycobacteria. Modifications are used to demonstrate M. leprae or Nocardia. Carnoy’s fixed tissues cannot be used and B-5 is suboptimal. Slides must be examined under oil. | |
| Alizarin red S | Calcium – orange red, polarizes | Identifies calcium in tissues |
| Bile | Bile – dark green on a yellow background | Identification of bile |
| Bodian’s | Neural tumors, identification of axons | |
| Chloroacetate esterase (Leder; CAE) | ||
| Congo red | Detection of amyloid. Immunoperoxidase studies can be used to identify specific types. Overstaining can result in false positives. | |
| Dieterle | ||
| Diff Quik (a modified Giemsa stain) | Evaluation of chronic gastritis | |
| Elastic stains (Verhoeff–van Gieson) | Identification of arteries and veins, vasculitis, invasion of lung tumors into visceral pleura, abnormal elastic fibers in elastofibromas | |
| Fibrin (see Phosphotungstic acid hematoxylin or Mallory PTAH) | To demonstrate fibrin in renal biopsies | |
| Fontana-Masson | ||
| Giemsa (May-Grunwald) | ||
| Gram (Brown-Hopps, Brown-Brenn) | Identification of bacteria, some cases of actinomycetes, Nocardia, coccidioidomycosis, blastomycosis, cryptococcosis, aspergillosis, rhinosporidiosis, and amebiasis | |
| Grimelius | Evaluation of neuroendocrine tumors (largely replaced by the use of immunohistochemistry for chromogranin) | |
| Hematoxylin and eosin (H&E) | Standard stain for the routine evaluation of tissues | |
| Iron (colloidal iron) | ||
| Jone’s silver methenamine | Basement membrane – dark maroon to black | Evaluation of renal biopsies |
| Melanin bleach | ||
| Methyl green-pyronin Y | ||
| Mucicarmine (Mayer) | Identification of adenocarcinomas, identification of Cryptococcus | |
| Oil red O | Requires frozen sections (lipids are dissolved by most fixatives or during processing). Tissue fixed in formalin can be used if tissue is frozen. | |
| Periodic acid – Schiff (PAS) | Classification of tumors with glycogen (e.g., Ewing’s/PNET, rhabdomyosarcoma, renal cell carcinoma), glomerular diseases (BM), identification of adenocarcinomas (mucin), fungal diseases (especially in argentophic areas – neutrophils and debris), spironolactone bodies in adrenal adenomas treated with this drug | |
| Periodic acid – Schiff with diastase digestion (PAS-D) | As above except glycogen that has been digested will not be stained | |
| Phosphotungstic acid hematoxylin (Mallory PTAH) | ||
| Reticular fibers (Gomor’s reticulin, Gordon and Sweets, Snook) | Bone marrow (myelophthisis), liver (fibrosis, veno-occlusive disease), carcinoma vs. sarcoma (reticular network) (but largely replaced by IHC) | |
| Silver stain (Grocott methenamine–silver nitrate – GMS) | ||
| Steiner | Evaluation of infectious diseases | |
| Sulfated Alcian blue | Identification of amyloid in cardiac biopsies | |
| Toluidine blue | Mast cell diseases, chronic cystitis | |
| Trichrome (Gomori, Masson) | Liver (fibrosis) | |
| Trichrome - modified | Evaluation of myocardial biopsies | |
| Von Kossa calcium | Demonstration of phosphate and carbonate radicals with calcium in tissues, identification of malakoplakia (Michaelis-Gutmann bodies) | |
| Warthin Starry | Infectious lesions (including syphilis, cat scratch fever, and bacillary angiomatosis) | |
| Wright’s | Blood smears |
The WebPath section of the University of Utah site (http://library.med.utah.edu/webpath) has useful descriptions of special stains and illustrative photographs.
IMMUNOPEROXIDASE STUDIES
The development of methods to detect antigens on tissue sections with antibodies was a major advance in surgical pathology. Immunohistochemical (IHC) studies are most frequently used for the following purposes:
Use of Immunohistochemistry.
A differential diagnosis is generated after examination of the H&E-stained slides. IHC is then used to gain evidence for or against diagnostic possibilities. “Trolling” cases through an immunohistochemistry laboratory by ordering numerous antibody studies without a clear reason in mind is more likely to lead to misguided diagnosis due to aberrant immunoreactivity than to provide an unexpected correct diagnosis.
Panels.
There are no absolute rules for immunoreactivity in cells and tissues. Aberrant immunoreactivity or loss of immunoreactivity is occasionally observed for all antibodies, either due to biologic variability (e.g., occasional keratin-positive melanomas) or technical factors (e.g., impure antibodies, cross-reaction with other antigens, failure to preserve antigenicity). Thus, immunohistochemical markers are used most effectively as panels of markers with interpretation based on an immunohistochemical profile.
Slides for Immunohistochemistry.
Tissue is often dislodged from normal glass slides during the treatments required for IHC. Thus, slides must be coated (e.g., with glue, poly-L-lysine, gelatin, albumin) or special commercial slides must be used. If slides are being prepared by another laboratory, the type of glass slide to be used must be specified.
Factors Affecting Immunogenicity.
There are numerous variables that can affect antigenicity. The most common are listed below. Each laboratory must optimize its procedures for each antibody used. Studies on tissues or slides not prepared in the routine fashion for a laboratory must be interpreted with caution.
Controls.
Controls are essential for the appropriate interpretation of immunohistochemical studies and ensure that all steps of this complicated procedure have been performed adequately.
Positive controls consisting of tissues known to be immunoreactive should be included each time an antibody is used for a test case. Internal positive controls should always be evaluated when present, as they control not only for the technique used but also for the antigenicity of the tissue under investigation. The immunoperoxidase table lists normal cells that are generally immunoreactive for each antibody. Some laboratories have used vimentin as a control for immunogenicity as almost all tissue should demonstrate positivity.5 Given the wide and non-specific distribution of vimentin, smooth muscle alpha actin may be more useful in this context as pericytes, vascular smooth muscle, and myoepithelial cells present in most tissues are immunoreactive.
Examples of internal controls:
Negative controls usually consist of replacing the primary antibody with non-immune animal serum diluted to the same concentration as the primary antibody. No positive reaction should be present. If multiple primary antibodies are used reactive with different target antigens, then they may serve as negative controls for each other. Although the best negative control would be to use antibody preabsorbed against the target antigen, this is rarely practical in a diagnostic laboratory. Diagnostic slides should also be evaluated for internal negative controls. Aberrant immunoreactivity of tissues that should not be positive is indicative that the immunoreactivity is nonspecific and the study should not be used for interpretation.
Evaluation of Studies
The following features must be taken into consideration when evaluating studies:
Location of Immunoreactivity
Antigens are present in specific sites. Some antigens may be present in more than one location or be extracellular.
Nonspecific positivity should be suspected when immunoreactivity is present in atypical locations:
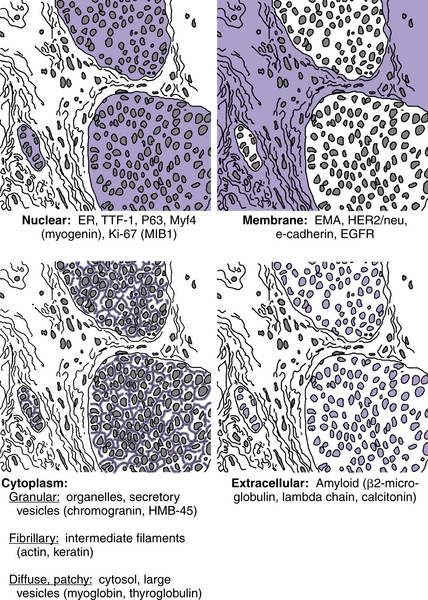
Examples of the normal location of antigens are shown in Figure 7-1.
Identification of Immunoreactive Cells
Immunoreactivity of tumor cells must be distinguished from immunoreactivity of normal entrapped cells (e.g., desmin [+] skeletal muscle cells infiltrated by tumor, S100 [+] Langerhans cells in tumors, smooth muscle alpha actin [+] blood vessels, etc.). Plasma cells have large amounts of cytoplasmic immunoglobulin and can react nonspecifically with many antibodies.
Intensity of Immunoreactivity
Some weak immunoreactivity may be present as a nonspecific finding. It is important to compare positive cells with control slides and with normally nonimmunoreactive cells to determine whether the immunoreactivity is significant.
Number of Immunoreactive Cells
In some cases, the number of positive cells may be important as a criterion for positivity or as a prognostic marker (e.g., markers of proliferation such as Ki-67, HER2/neu). In other cases, rare weakly positive cells must be distinguished from intermingled normal cells or just nonspecific immunoreactivity.
Criteria for a “Positive” Result
Specific criteria for evaluating IHC have been developed for a few antibodies (see Tables 7-12 to 7-16). However, criteria do not exist for most antibodies or are not universally used by all pathologists. The significance of immunoreactivity varies with the type of lesion, the antibody, and the specific assay. Strong positivity in the majority of cells is easily interpreted as positive. As the number of positive cells decreases, and the intensity of immunoreactivity weakens, the lower threshold of a “positive” result becomes more difficult to determine.
Time
Alkaline phosphatase chromogens (red color) fade over time. DAB (a brown color) is more permanent. This is not a problem in evaluating current pathology specimens. However, if slides are reviewed after a period of time, some chromogens may fade and once positive results may appear to be negative.
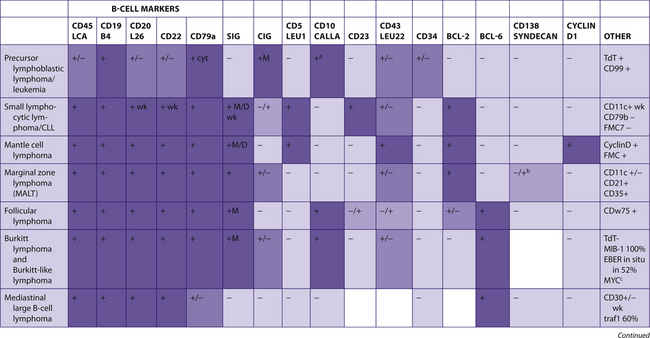
Common Panels for Immunohistochemical Studies
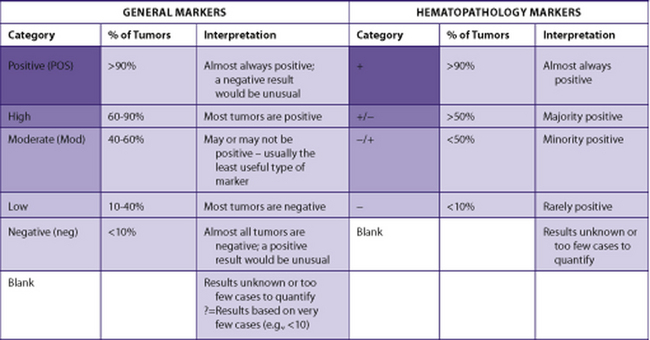
The following tables include information from the literature as well as the personal experiences of the staff at Brigham and Women’s Hospital. Because of the many differences in specific antibodies, laboratory assays, and criteria for considering a result “positive,” results may vary for different institutions. The results have been divided into five categories for general markers and four categories for hematopathology markers. Note that “%” refers to the number of tumors reported to be positive, not the number of cells positive within a tumor (Table 7-2).
TABLE 7–2 EVALUATION OF POSITIVITY OF IMMUNOHISTOCHEMICAL STUDIES
 |
The actual markers used to evaluate a case will depend upon the differential diagnosis based on the H&E appearance. In some cases, an initial panel, which is often used for typical cases, has been suggested. Not all markers listed would be used for all cases and some markers are included to indicate when they would not be useful for distinguishing the tumors listed in the table.
POSITIVE is defined as the presence of immunoreactive cells and NEGATIVE as the absence of immunoreactive cells. Unfortunately, “positive” has also been used in some studies to mean “absence of immunoreactivity” when this finding supports a diagnosis. For example, the absence of SMAD4 (DPC4) has been reported as a “positive” result for pancreatic carcinoma, as this marker is absent in the majority of these tumors. This method of reporting results becomes confusing as “positive” and “negative” are dependent on the expected diagnosis. It is preferable to report the findings (positive = immunoreactivity present; negative = immunoreactivity absent) and then interpret them as supporting, or not supporting, the diagnoses in the differential diagnosis.
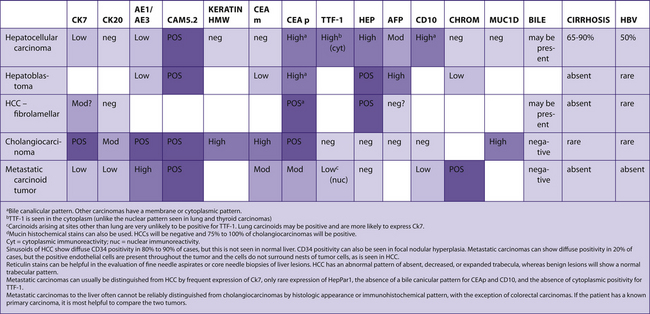
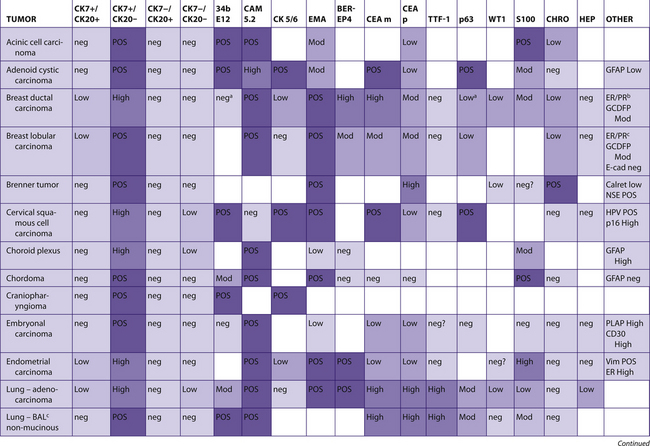
Cytokeratin 7 and cytokeratin 20 tables
The combination of these two cytokeratins have been found to be useful to divide carcinomas into four main groups (Ck7+/Ck20+, Ck7+/Ck20-, Ck7-/Ck20+, Ck7-/Ck20-).
In Tables 7-3 to 7-7, other commonly used antibodies have been included to show differences within each group. The most useful additional antibodies will depend on the specific differential diagnosis.6-8
TABLE 7–3 PREDOMINANTLY CK7+/CK20+
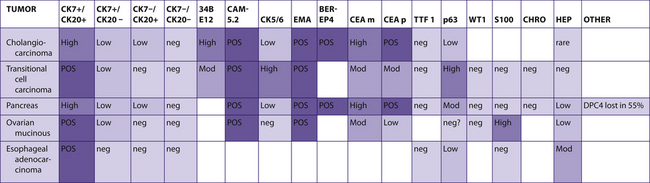 |
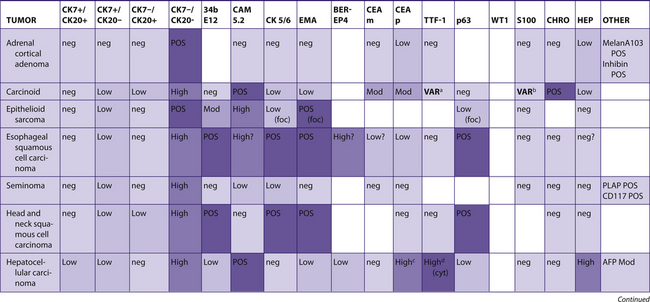
TABLE 7–4 NO DOMINANT CK7/CK20 PATTERN OR PATTERN UNKNOWN
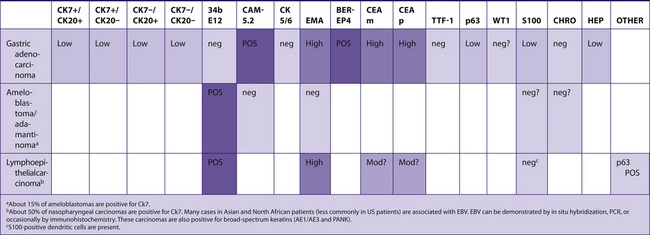 |
TABLE 7–5 PREDOMINANTLY CK7+/CK20−
 |
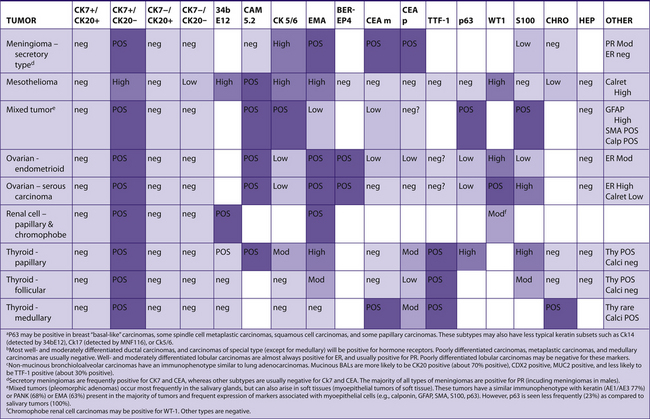 |
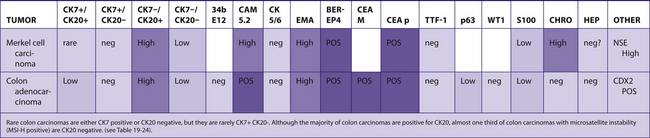 |
TABLE 7–7 PREDOMINANTLY CK7−/CK20–
 |
 |
Spindle cell lesions, soft tissue lesions, and sarcomas
See Table 7-9.
TABLE 7–9 SPINDLE CELL/SOFT TISSUE LESIONS/SARCOMAS
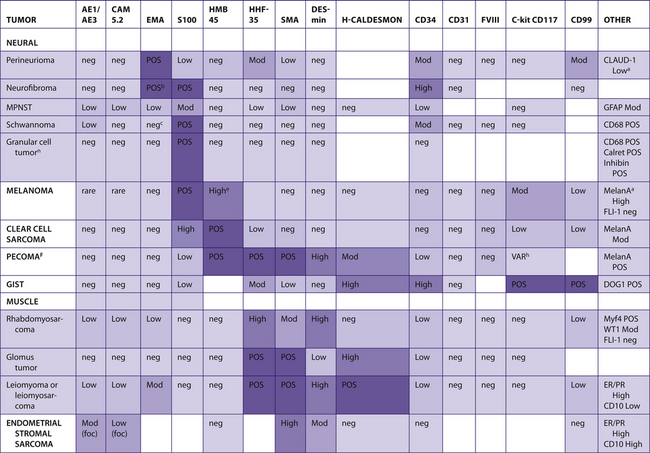 |
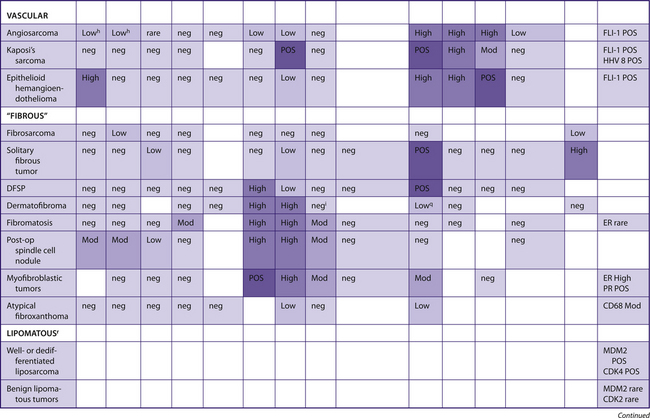 |
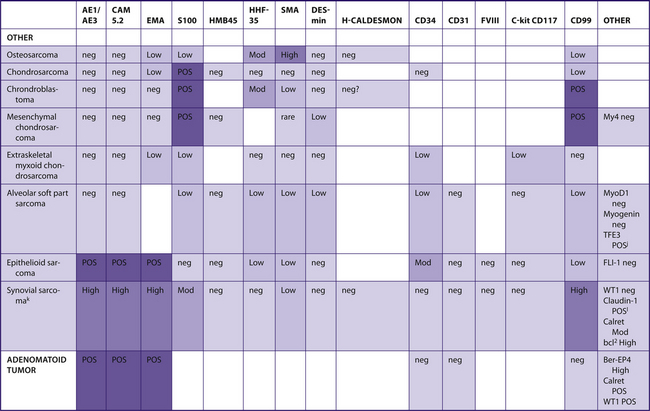 |
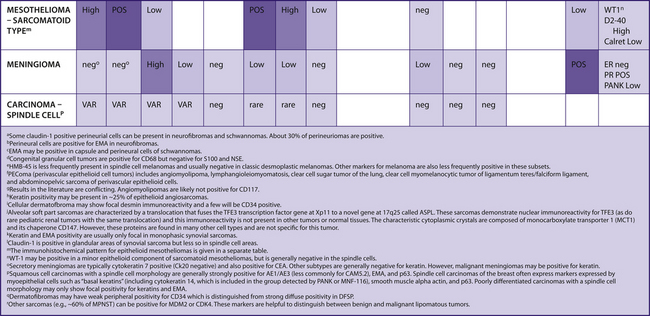 |
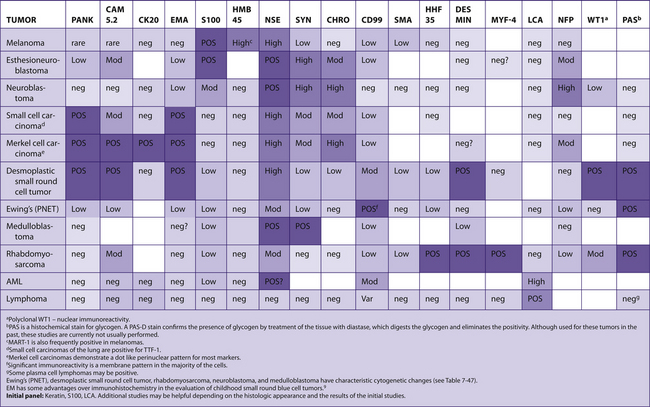
Metastatic tumors of unknown origin
Pathologists frequently receive specimens with metastatic tumors. Often, the site of origin is known to the clinician, but this information is not provided to the pathologist. A good clinical history is frequently more successful for correct classification than a battery of studies.
The Ck7/Ck20 pattern is generally helpful to narrow down the potential site of origin of carcinomas (see Tables 7-3 to 7-7). Additional studies can then be used to identify specific types of carcinoma.
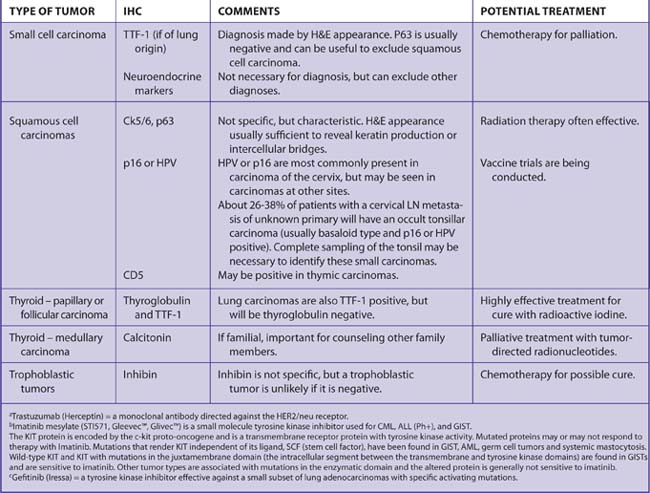
The most important tumors to identify are those with specific therapeutic treatments for cure or palliation (Table 7-10).10
Poorly differentiated tumors
See Table 7-11.
TABLE 7–11 POORLY DIFFERENTIATED TUMORS
| TYPE OF TUMOR | IMMUNOHISTOCHEMICAL MARKER | COMMENTS |
|---|---|---|
| Carcinoma | Broad spectrum keratins AE1/AE3 or PANK (MNF-116) | |
| Melanoma | S100 protein | |
| Lymphoma | Leukocyte common antigen (LCA) |
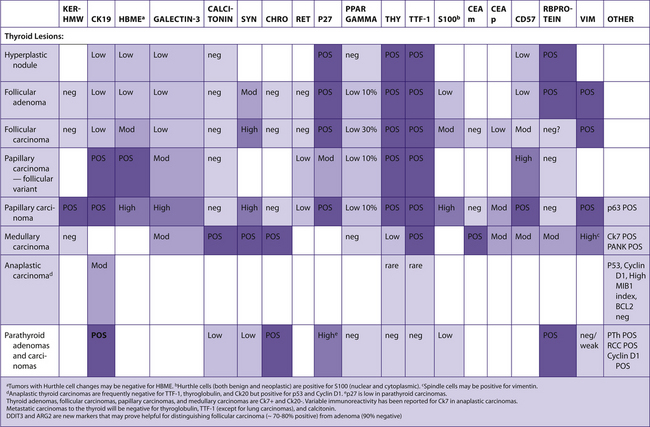
Estrogen and progesterone receptor evaluation
Hormone receptors are routinely determined on all invasive breast carcinomas and DCIS. ER and PR are weak prognostic markers and are more useful to predict the likelihood of response to hormonal therapies.
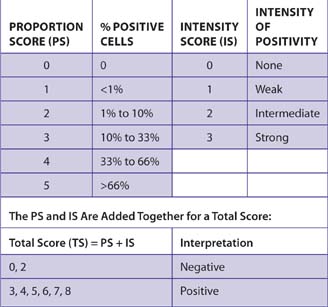
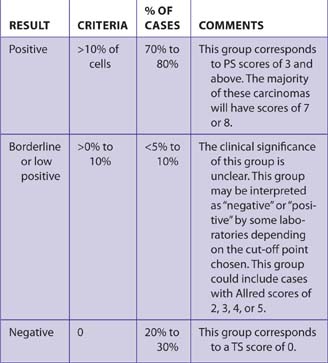
Many different methods are currently used to report the results of IHC studies for ER and PR. One method that has been used in multiple studies is the Allred score method (Table 7-12).
Patients with carcinomas that scored 3 (<1% of cells with intermediate intensity or 1% to 10% of cells with weak intensity) or above had improved disease-free survival when treated with endocrine therapy.11 Patients with carcinomas with a total score of 2 (<1% weakly positive cells) had a survival rate similar to ER-negative carcinomas (total score of 0).
About 80% of DCIS cases are positive for ER using the same method of scoring. Women with ER-positive DCIS were shown to experience fewer local recurrences, contralateral recurrences, and distant recurrences when treated with tamoxifen (NSABP B24 study).
With optimization of IHC using newer antigen retrieval methods, 99.2% of carcinomas will have scores of 0, 7, or 8.12 Therefore, many laboratories report results as positive or negative. The value of further subdividing cases by percent positive cells, H-score, or image analysis for either prognosis or to predict response to tamoxifen has not been demonstrated. Intensity of immunoreactivity is difficult to evaluate as most cases show strong reactivity with optimal assay methods and most carcinomas show considerable variability in intensity.
A possible method for reporting results is shown in Table 7-13. The same system can be used for reporting progesterone receptor results. The use of both ER and PR may be helpful for determining the likelihood of response to tamoxifen, as has been shown with data using the biochemical assay (Table 7-14). Presumably, the presence of the ER-regulated gene product PR is more predictive of an intact ER regulatory pathway.
TABLE 7–14 RESPONSE TO TAMOXIFEN
| STATUS OF CARCINOMA | % OF CARCINOMAS | % OF PATIENTS RESPONDING TO TAMOXIFEN |
|---|---|---|
| ER+ PR+ | 63% | 75% to 80% |
| ER+ PR– | 15% | 25% to 30% |
| ER– PR+ | 5% | 40% to 45% |
| ER– PR– | 17% | <10% |
Recent national guidelines for reporting ER and PR have been released and should be consulted for additional information about performing and interpreting these studies (Hammond ME, et al, American Society of Clinical Oncology/College of American Pathologists Guideline Recommendations for Immunohistochemical Testing of Estrogen and Progesterone Receptors in Breast Cancer, J Clin Oncol 2010 Apr 19).False negative results, and to a lesser extent, false positive results, can also be problems. False negative results may be due to a large number of causes including:
Most cases of false negativity can be suspected, as the normal breast tissue will also be negative. In such cases, the assay should be repeated on the same block, a different block from the same case, or blocks from another case, if available. If the normal tissue remains negative, the possibility of loss of antigenicity in the tissue can be mentioned in the report.
False positive results are quite unusual, as the antibody should not crossreact with other antigens.
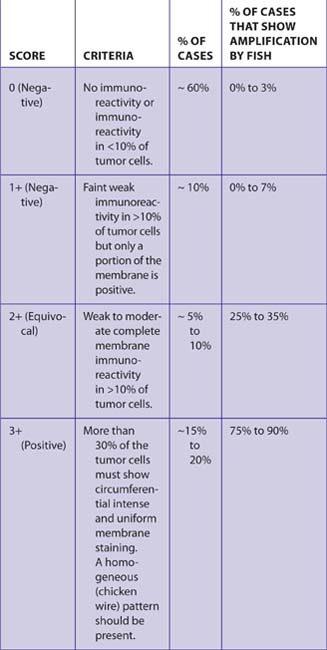
HER2/neu score
The HER2/neu immunoreactivity scoring system in Table 7-15 was recommended by an expert panel.13 Other panel suggestions:
Only membrane immunoreactivity is scored. Marked cytoplasmic immunoreactivity may make interpretation difficult. FISH studies may be preferred for such cases (Table 7-16).
| RESULT | CRITERIA | comment |
|---|---|---|
| Positive for amplification | >6.0 gene copies or >2.2 ratio | |
| Equivocal for amplification | 4.0 to 6.0 genes or 1.8 to 2.2 ratio | Guidelines suggest counting additional cells for FISH, retesting, or performing IHC |
| Negative for amplification | <4.0 genes or <1.8 ratio |
Patients with a ratio of 2.0 or greater have been eligible for Herceptin trials.
In >90% of carcinomas with protein overexpression, the HER2/neu gene has been amplified. In 3% to 5% of cases, protein overexpression can occur due to other mechanisms. In <5% of cases, there may be gene amplification without protein overexpression. In general, there is a 20% to 40% response to Herceptin alone in patients with cancers showing gene amplification by FISH, and <5% of patients respond if the gene is not amplified. Therefore, FISH studies may be helpful for cases with 2+ positivity or difficult to interpret cases (e.g., with variable positivity or cytoplasmic positivity).
Well- and moderately differentiated lobular carcinomas are rarely positive (<5%). However, in some cases there may be edge enhancement of individual tumor cells that may be difficult to interpret. FISH studies may be helpful.
In rare cases, DCIS overexpresses HER2/neu but the accompanying invasive carcinoma does not. This is a source of potential false positive results for IHC or FISH.
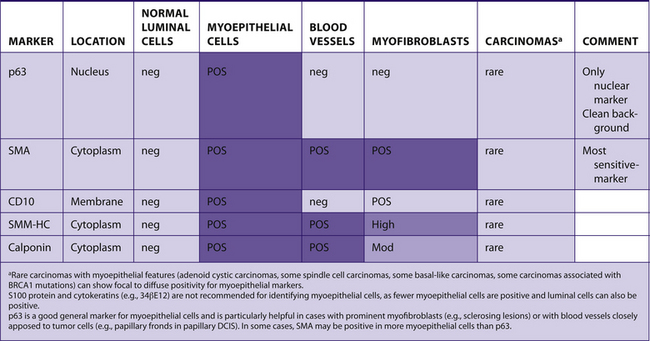
Myoepithelial markers in breast carcinoma
Myoepithelial markers can be useful for the evaluation of breast lesions (Table 7-17):
TABLE 7–17 MYOEPITHELIAL MARKERS IN BREAST CARCINOMA
 |
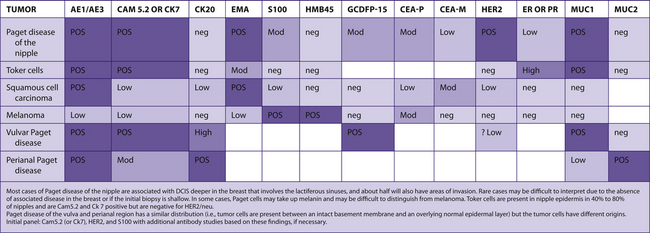
Epidermal lesions of the nipple
See Table 7-18.
TABLE 7–18 EPIDERMAL LESIONS OF THE NIPPLE AND PAGET DISEASE AT OTHER SITES
 |
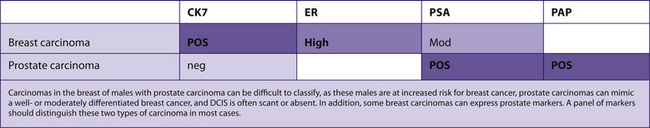
Breast carcinoma in males versus metastatic prostate carcinoma
See Table 7-19.
TABLE 7–19 BREAST CARCINOMA IN MALES VERSUS METASTATIC PROSTATE CARCINOMA
 |
Signet ring cell carcinomas of the stomach and breast (lobular carcinoma)
See Table 7-20 and Fig. 7-2.
TABLE 7–20 SIGNET RING CELL CARCINOMAS OF THE STOMACH AND BREAST (LOBULAR CARCINOMA)
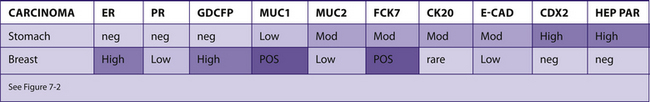 |
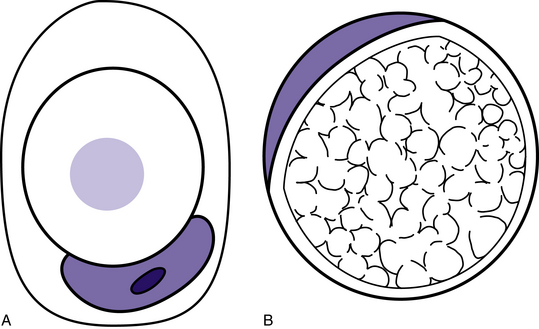
Figure 7–2 Metastatic lobular carcinoma of the breast can morphologically resemble primary signet ring cell gastric carcinomas. Both typically lack e-cadherin expression. In addition, lobular breast carcinomas can be clinically occult or can present as distant metastases many years after the initial presentation. Signet ring cells associated with breast carcinoma more commonly have a central mucin vacuole with a targetoid appearance (cell A). Gastric signet ring cells usually have many small vacuoles giving the cytoplasm a foamy appearance (cell B). These criteria are not reliable in distinguishing these two carcinomas. However, the presence of the first type of signet ring cell in a biopsy from the gastrointestinal tract should raise the possibility of metastatic breast carcinoma.14 The majority of lobular breast carcinomas will be ER positive, and this is a reliable marker to exclude gastric carcinoma. In the minority of ER-negative cases, PR, GCDFP, MUC1, CDX2, and Hep Par may be helpful markers.15
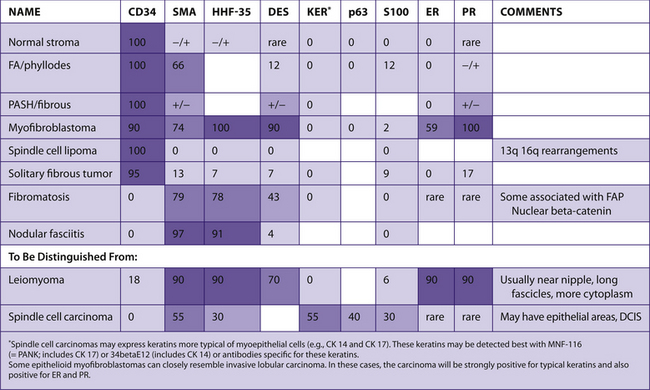
Fibroblastic/myofibroblastic lesions of the breast
See Table 7-21.
TABLE 7–21 FIBROBLASTIC/MYOFIBROBLASTIC LESIONS OF THE BREAST
 |
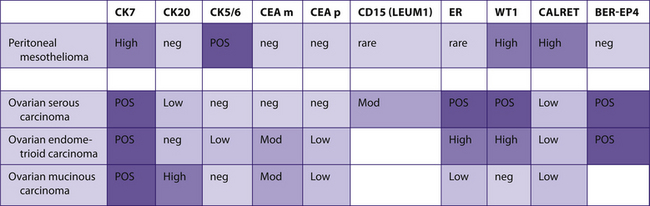
Ovarian carcinoma versus mesothelioma
See Table 7-22.
TABLE 7–22 OVARIAN CARCINOMA VERSUS MESOTHELIOMA
 |
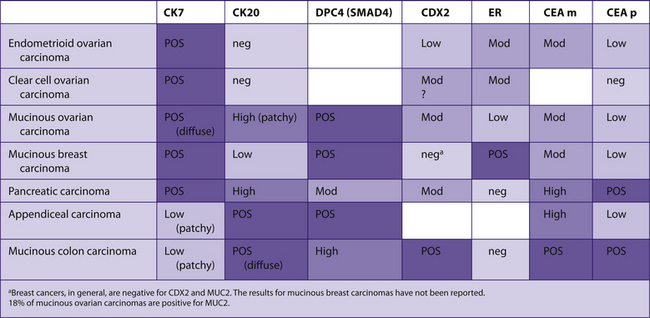
Primary ovarian carcinoma versus metastatic carcinomas
See Table 7-23.
TABLE 7–23 PRIMARY OVARIAN CARCINOMA VERSUS METASTATIC CARCINOMAS
 |
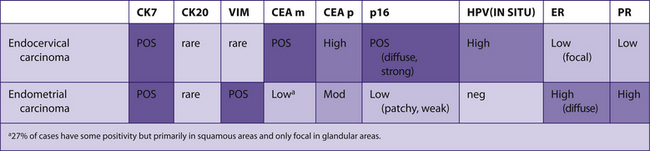
Endocervical carcinoma versus endometrial carcinoma
See Table 7-24.
TABLE 7–24 ENDOCERVICAL CARCINOMA VERSUS ENDOMETRIAL CARCINOMA
 |
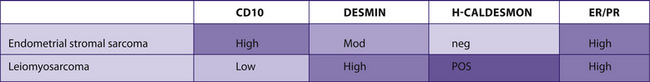
Endometrial stromal sarcoma versus leiomyosarcoma
See Table 7-25.
TABLE 7–25 ENDOMETRIAL STROMAL SARCOMA VERSUS LEIOMYOSARCOMA
 |
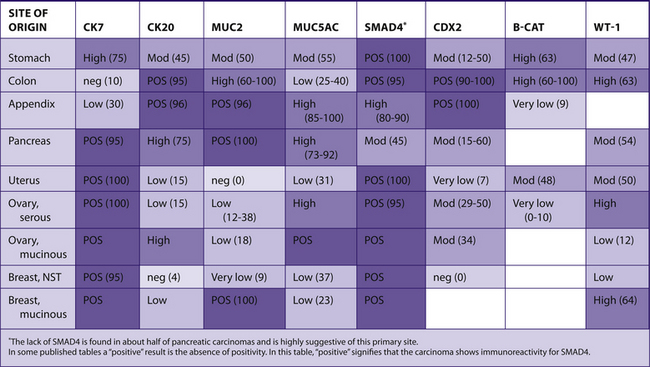
Metastatic adenocarcinomas in the abdomen
See Table 7-27.
TABLE 7–27 METASTATIC ADENOCARCINOMAS IN THE ABDOMEN
 |
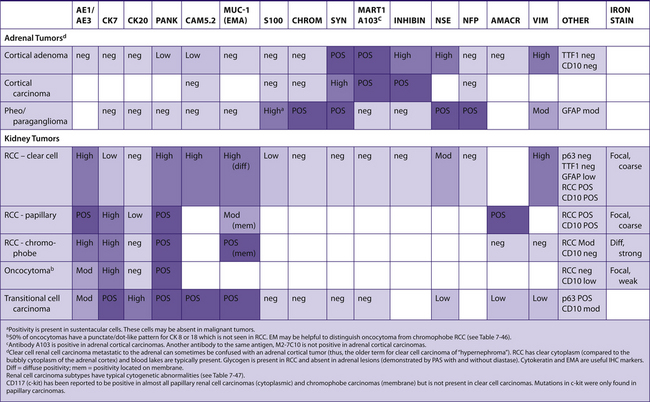
Hemangioblastoma versus metastatic renal cell carcinoma
See Table 7-29.
TABLE 7–29 HEMANGIOBLASTOMA VERSUS METASTATIC RENAL CELL CARCINOMA
 |
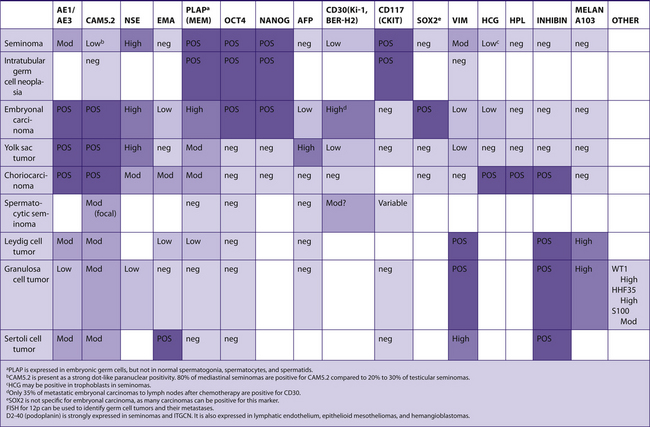
Tumors of germ cells and sex-cord stromal tumors
See Table 7-30.
TABLE 7–30 TUMORS OF GERM CELLS AND SEX-CORD STROMAL TUMORS
 |
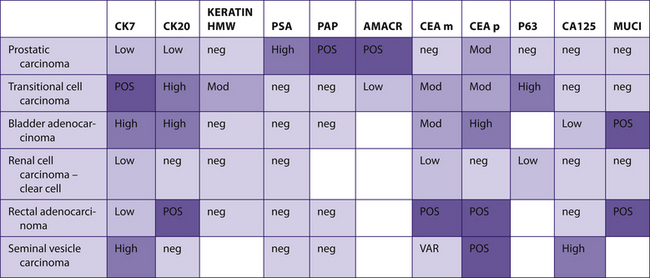
Tumors of bladder, prostatic, and renal origin
See Table 7-32.
TABLE 7–32 TUMORS OF BLADDER, PROSTATIC, AND RENAL ORIGIN
 |
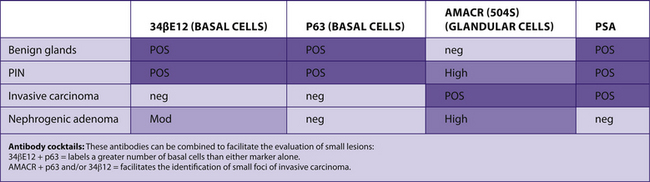
Prostate carcinoma versus other lesions
See Table 7-33.
TABLE 7–33 PROSTATE CARCINOMA VERSUS OTHER LESIONS
 |
Differential diagnosis of epithelial mesothelioma and lung adenocarcinoma
See Table 7-36.
TABLE 7–36 DIFFERENTIAL DIAGNOSIS OF EPITHELIAL MESOTHELIOMA AND LUNG ADENOCARCINOMA
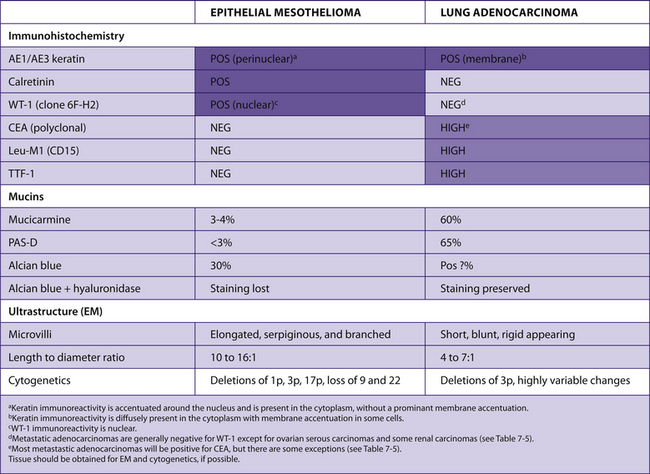 |
Initial panel: AE1/AE3, calretinin, WT-1 (clone 6F-H2), CEA, Leu-M1, and TTF-1 with additional studies ordered in difficult cases.
Other antibodies generally reported as negative in epithelial mesotheliomas and positive in lung adenocarcinomas include the following: MOC-1, B72.3, Ber-EP4, and BG-8. Cytokeratins 5/6 are reported to be positive in mesotheliomas and negative in lung carcinomas. However, in our experience, these markers have proven less useful than the ones listed earlier. The use of EMA is controversial. Strong membrane positivity is characteristic of epithelial mesothelioma, whereas cytoplasmic positivity is characteristic of adenocarcinomas.
Less is known about the immunophenotype of pure sarcomatoid mesotheliomas. The spindle cells are positive for cytokeratin, but are less frequently positive for the other markers as compared to the epithelioid cells. Tumors that can, on occasion, resemble mesotheliomas are generally negative for cytokeratins, with the notable exceptions of some cases of angiosarcoma, epithelioid hemangioendothelioma, synovial sarcoma, epithelioid sarcoma, and leiomyosarcoma (see Table 7-9).16
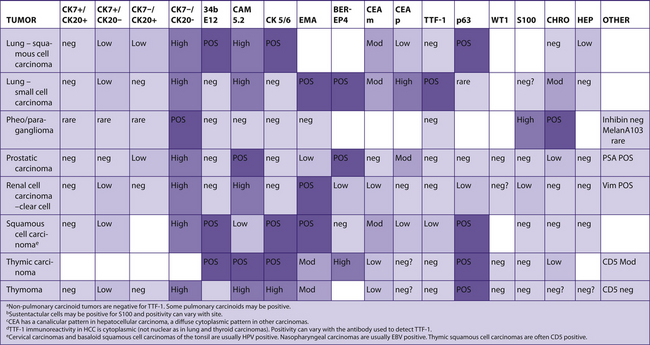
Lung carcinoma
See Tables 7-37 and 7-38.
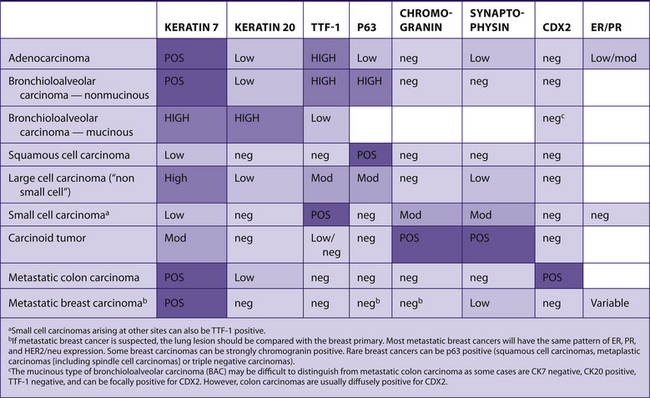 |
TABLE 7–38 DIFFERENTIAL DIAGNOSIS OF LUNG CARCINOMAS
| DIFFERENTIAL DIAGNOSIS | MOST USEFUL MARKERS |
|---|---|
| Adenocarcinoma vs. squamous cell carcinoma | Keratin 7, keratin 20, TTF-1, p63 |
| Small cell carcinoma vs. basaloid squamous cell carcinoma | P63, TTF-1 |
| Small cell carcinoma vs. carcinoid tumor | Mitoses, necrosis, amount of cytoplasm |
| Large cell neuroendocrine carcinoma vs. carcinoid tumor | Mitoses, necrosis |
| Mucinous lung carcinoma vs. metastatic colon cancer | TTF-1, CDX2 (mucinous BAC can be focally positive for CDX2) |
| Lung carcinoma vs. metastatic breast carcinoma | TTF-1, compare ER/PR/HER2 pattern in primary breast carcinoma and lung tumor |
Amyloid
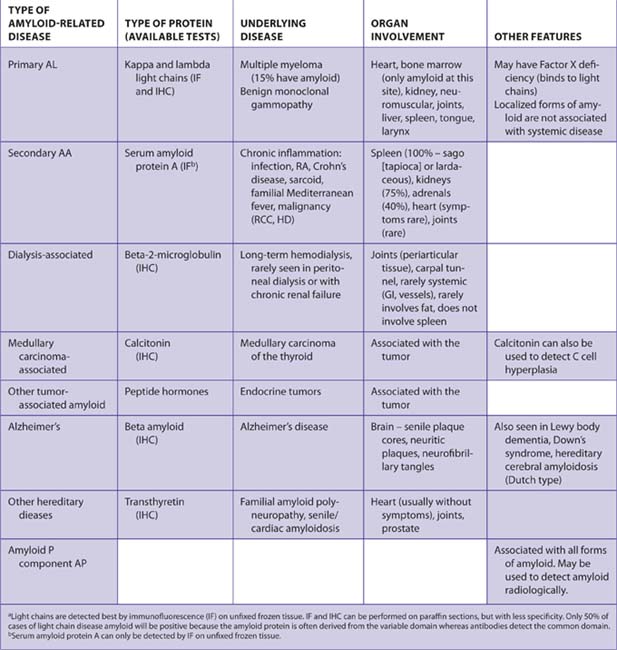
Amyloidosis (Greek for amylon = starch plus eidos = resemblance) is seen in many different clinical settings and is associated with many diseases. Pathologists can narrow down the differential diagnosis considerably to help guide clinical decision making. Finding an amyloid deposit in any tissue is similar to finding metastatic carcinoma in a lymph node – in both settings clinical information (e.g., history, physical examination, radiology studies, results of laboratory tests) is essential in arriving at the correct interpretation. A little immunohistochemistry and a lot of clinical judgment by the pathologist can help establish the cause with a greater degree of certainty.17
Finding and characterizing amyloid deposits:
Antibodies for immunohistochemistry
See Tables 7-43 and 7-44.
TABLE 7–44 ALTERNATIVE NAMES FOR ANTIGENS
| LOOKING FOR? | FIND IT UNDER: |
|---|---|
| 1D5 | Estrogen receptor (G) |
| 6F/3D | Beta-amyloid |
| 12E7 | CD99 (G, H) |
| 34βE12 | Keratins (G) |
| 38.13 | CD77(H) |
| 70 kD NF | Neurofilaments (G) |
| 200 kD NF | Neurofilaments (G) |
| 903 | Keratins--34βE12 (G) |
| A (blood group antigen) | Blood group antigens (G) |
| A (Ig heavy chain alpha) | Heavy chain immunoglobulins (H) |
| A32 antigen | CD146 (G) |
| A103 | MELAN-A (G) |
| AAT | Alpha 1-antitrypsin (G, H) |
| ACH | Alpha-1 antichymotrypsin (H) |
| AE1/AE3 | Keratins (G) |
| AFP | Alpha-fetoprotein (G) |
| Alpha 1-antitrypsin | Alpha 1-antitrypsin (G, H) |
| Alpha 1-antichymotrypsin | Alpha 1-antichymotrypsin (H) |
| Alpha 1-fetoprotein | Alpha fetoprotein (G) |
| Alpha fetoprotein | Alpha fetoprotein (G) |
| Alpha-methylacyl-CoA racemase | AMACR (G) |
| Alpha smooth muscle actin | Alpha smooth muscle actin (G) |
| AMACR | AMACR (G) |
| Amyloid | Beta-amyloid (G) |
| Androgen receptor | Androgen receptor (G) |
| Apolipoprotein J | Clusterin (H) |
| AR | Androgen receptor (G) |
| B (blood group antigen) | Blood group antigens (G) |
| B1 | CD20 (H) |
| B2 | CD21 (H) |
| B4 | CD19 (H) |
| B72.3 | B72.3 (G) |
| bcl-1 | Cyclin D1 (H) |
| bcl-2 | bcl-2 (H, G) |
| B-cell specific activator protein | BSAP (H) |
| BER-EP4 | BER-EP4 (G) |
| BERH2 | CD30 (G, H) |
| Beta-amyloid | Beta-amyloid (G) |
| Beta-catenin | Beta-catenin (G) |
| Beta-2 microglobulin | Beta-2 microglobulin (G) |
| BG8 | BG8 (G) |
| B-HCG | Human chorionic gonadotropin (G) |
| BLA.36 | CD77 (H) |
| BL-CAM | CD22 (H) |
| Blood group antigens | Blood group antigens (G, H) |
| BR-2 | Gross cystic disease fluid protein-15 (G) |
| BRST-2 | Gross cystic disease fluid protein-15 (G) |
| C3b/C4bR | CD35 (H) |
| C5b-9 | C5b-9 (G) |
| c-kit | CD117 (G) |
| CA 15-3 | Epithelial membrane antigen (G, H) |
| CA 19-9 | CA 19-9 (G) |
| CA 27.28 | Epithelial membrane antigen (G, H) |
| CA 72-4 | B72.3 (G) |
| CA125 | CA125 (G) |
| CA19-9 | CA19-9 (G) |
| Calcitonin | Calcitonin (G), Hormones (G) |
| Caldesmon | Caldesmon (G) |
| Calgranulin | MAC 387 (G) |
| CALLA | CD10 (G, H) |
| CALP | Calponin (G) |
| Calponin | Calponin (G) |
| Calprotectin | MAC 387 (G) |
| Calretinin | Calretinin (G) |
| CAM5.2 | Keratins (G) |
| Carbohydrate antigen 19-9 | CA19-9 (G) |
| Carcinoembryonic antigen | Carcinoembryonic antigen (G) |
| CD1a | CD1a (H) |
| CD2 | CD2 (H) |
| CD3 | CD3 (H) |
| CD4 | CD4 (H) |
| CD5 | CD5 (G, H) |
| CD7 | CD7 (H) |
| CD8 | CD8 (H) |
| CD10 | CD10 (G, H) |
| CD11b | CD11b (H) |
| CD11c | CD11c (H) |
| CD13 | CD13 (H) |
| CD15 | CD15 (G, H) |
| CD16 | CD16 (H) |
| CD19 | CD19 (H) |
| CD20 | CD20 (H) |
| CD21 | CD21 (H) |
| CD22 | CD22 (H) |
| CD23 | CD23 (H) |
| CD25 | CD25 (H) |
| CD30 | CD30 (G, H) |
| CD31 | CD31 (G) |
| CD33 | CD33 (H) |
| CD34 | CD34 (G, H) |
| CD35 | CD35 (H) |
| CD38 | CD38 (H) |
| CD43 | CD43 (H) |
| CD44v3 | CD44v3 (G) |
| CD45 | CD45 (H) |
| CD45RA | CD45RA (H) |
| CD45Ro | CD45Ro (H) |
| CD56 | CD56 (H) |
| CD57 | CD57 (G) |
| CD61 | CD61 (H) |
| CD68 | CD68 (G, H) |
| CD74 | CD74 (H) |
| CDw75 | CDw75 (H) |
| CD77 | CD77 (H) |
| CD79a | CD79a (H) |
| CD79b | CD79b (H) |
| CD95 | CD95 (H) |
| CD99 | CD99 (G, H) |
| CD117 | CD117 (G) |
| CD141 | CD141 (G) |
| CDX | CDX (G) |
| CDKN2 | p16 (G) |
| CDP | Gross cystic disease fluid protein-15 (G) |
| CEA | Carcinoembryonic antigen (G) |
| c-erbB2 | HER-2/neu (G) |
| Chromogranin A | Chromogranin A (G) |
| c-kit | CD117 (G) |
| CLA | CD45 (H) or HECA-452 (H) |
| CLDN1 | Claudin (G) |
| Clusterin | Clusterin (H) |
| Collagen IV | Collagen IV (G) |
| Common acute leukemia antigen | CD10 (G, H) |
| Complement lysis inhibitor | Clusterin (H) |
| CR1 | CD35 (H) |
| Cyclin D1 | Cyclin D1 (H) |
| Cystic fibrosis antigen | MAC 387 (G) |
| D (Ig heavy chain delta) | Heavy chain immunoglobulins (H) |
| DBA.44 | DBA.44 (H) |
| Desmin | Desmin (G) |
| DF3 | Epithelial membrane antigen (G, H) |
| DPB | CD45RA (H) |
| E2 antigen | CD99 (G, H) |
| EBERS | Epstein-Barr virus (G, H) |
| EBNA | Epstein-Barr virus (G, H) |
| E-cadherin | E-cadherin (G) |
| EGFR | EGFR (G) |
| EM ACT | HHF-35 (G) |
| EMA | Epithelial membrane antigen (G) |
| E-MEL | HMB-45 (G) |
| Endothelial cell antigen | HECA-452 (H) |
| Ep-CAM | BER-EP4 (G) |
| Epidermal growth factor receptor | EGFR (G) |
| Epithelial membrane antigen | Epithelial membrane antigen (G, H) |
| Epithelial specific antigen | BER-EP4 (G) |
| Epstein-Barr virus | Epstein-Barr virus (G, H) |
| ER | Estrogen receptor (G) |
| erbB2 | HER-2/neu (G) |
| ESA | BER-EP4 (G) |
| Estrogen receptor | Estrogen receptor (G) |
| Ewing’s sarcoma marker | CD99 (G, H) |
| Factor VIII related antigen | Factor VIII (G) |
| FVIII:RAg | Factor VIII (G) |
| Factor XIIIa | Factor XIIIa (G) |
| Fascin | Fascin (H) |
| Fast myosin | Myosin heavy chain (G) |
| Fibronectin | Fibronectin (G) |
| Fli-1 | Fli-1 (G) |
| FMC7 | FMC7 (H) |
| FMC 29 | CD99 (G, H) |
| Friend leukemia integrin-site 1 | Fli-1 (G) |
| FVIII:g | Factor VIII (G) |
| G (Ig heavy chain gamma) | Heavy chain immunoglobulins (H) |
| Gal-3 | Galectin-3 (G) |
| Galectin-3 | Galectin-3 (G) |
| Gastrin | Hormones (G) |
| GCDFP | Gross cystic disease fluid protein-15 (G) |
| GFAP | Glial fibriallary acidic protein (G) |
| Glial fibrillary acidic protein | Glial fibriallary acidic protein (G) |
| Glucagon | Hormones (G) |
| Glucose transporter 1 | GLUT-1 (G) |
| GLUT-1 | GLUT-1 (G) |
| GPIIIa | CD61 (H) |
| gp80 | Clusterin (H) |
| gp200 | RCC (G) |
| GPA | Glycophorin A (H) |
| Granzyme B | Granzyme B (H) |
| Gross cystic disease fluid disease-15 | Gross cystic disease fluid protein-15 (G) |
| H (blood group antigen) | Blood group antigens (G) |
| H222 | Estrogen receptor (G) |
| Hb | Hemoglobin (H) |
| HBME-1 | HBME-1 (G) |
| h-caldesmon | Caldesmon (G) |
| H-CAM | CD44v3 (G) |
| HCG | Human chorionic gonadotropin (G) |
| HCL | DBA.44 (H) |
| HBME-1 | HBME-1 (G) |
| Heavy chain immunoglobulins | Heavy chain immunoglobulins (H) |
| HECA-452 | HECA-452 (H) |
| Hematopoietic progenitor cell, class 1 | CD34 |
| Hemoglobin | Hemoglobin (H) |
| HepPar-1 | HepPar-1 (G) |
| Hepatocyte paraffin-1 | HepPar-1 (G) |
| HER-2/neu | HER-2/neu (G) |
| HHF-35 | HHF-35 (G) |
| HHV8 | HHV8 (H) |
| HLA-DR | HLA-DR (H) |
| HMB-45 | HMB-45 (G) |
| HMFG | Epithelial membrane antigen (G, H) |
| hMLH1 | hMLH1 (G) |
| hMSH2 | hMLH1 (G) |
| HNK-1 | CD57 (G) |
| HP1 | HepPar-1 (G) |
| HPCA-1 | CD34 (G, H) |
| HPL | Human placental lactogen (G) |
| HuLy-m6 | CD99 (G, H) |
| Human chorionic gonadotropin | Human chorionic gonadotropin (G) |
| Human herpes virus 8 | HHV8 (G, H) |
| Human mutL homologue 1 | hMLH1 (G) |
| Human mutS homologue 2 | hMLH1 (G) |
| Human placental lactogen | Human placental lactogen (G) |
| IL-2 receptor | CD25 (H) |
| Inhibin-alpha subunit | Inhibin-alpha subunit (G) |
| Insulin | Hormones (G) |
| J5 | CD10 (G, H) |
| JOVI 1 | TCR (H) |
| K (Ig lighit chain kappa) | Light chain immunoglobulins (H) |
| Keratin 5/6 | Keratins (G) |
| Keratin 7 | Keratins (G) |
| Keratin 20 | Keratins (G) |
| Keratins | Keratins (G) |
| Ki-1 | CD30 (G, H) |
| Ki-67 | Ki-67 (G) |
| kip2 | p57 (G) |
| Kit | CD117 (G) |
| KP-1 | CD68 (G, H) |
| L (Ig light chain lambda) | Light chain immunoglobulins (H) |
| L1 antigen | MAC 387 (G) |
| L26 | CD20 (H) |
| L60 | CD43 (H) |
| Laminin | Laminin (G) |
| LCA | CD45 (H) |
| Leu 1 | CD5 (H) |
| Leu 2 | CD8 (H) |
| Leu 3 | CD4 (H) |
| Leu 5a + b | CD2 (H) |
| Leu 7 | CD57 (G, H) |
| Leu 9 | CD7 (H) |
| Leu16 | CD20 (H) |
| Leu 22 | CD43 (H) |
| Leukocyte common antigen | CD45 (H) |
| Leu-M1 | CD15 (G, H) |
| Light chain immunoglobulins | Light chain immunoglobulins (H) |
| LFA-2 | CD2 (H) |
| LMP-1 | Epstein-Barr virus (G, H) |
| LN1 | CDw75 (H) |
| LN2 | CD74 (H) |
| Lysozyme | Lysozyme (H, G) |
| M (Ig heavy chain mu) | Heavy chain immunoglobulins (H) |
| Mac-1 | CD11b (H) |
| MAC 387 | MAC 387 (G) |
| Mac-M | CD68 (G, H) |
| MART 1 | MELAN A (G) |
| Mast cell tryptase | Mast cell tryptase (H) |
| mb-1 | CD79a (H) |
| MCAM | CD146 (G) |
| ME491 | CD63 (G) |
| MELAN-A | MELAN-A (G) |
| Melanoma antigen recognized by T cells | MELAN-A (G) |
| Melanoma-associated antigen | CD63 (G) |
| Melanoma cell adhesion molecule | CD146 (G) |
| Melanoma-specific antigen | HMB-45 (G) |
| MELCAM (or Mel-CAM) | CD146 (G) |
| MIB-1 | Ki-67 (G) |
| MIC-2 | CD99 (G, H) |
| MLH1 | hMLH1 |
| MN-4 | CD146 (G) |
| MNF-116 | Keratin--Pan-K (G) |
| MPO | Myeloperoxidase (H) |
| MRF4 | Myf-4 (G) |
| MSA | HHF-35 (G) |
| MSH2 or MSH6 | hMLH1 |
| MTS1 | p16 (G) |
| MUC1 | Epithelial membrane antigen (G, H) |
| MUC18 | CD146 (G) |
| Muscle common actin | HHF-35 (G) |
| Muscle specific actin | HHF-35 (G) |
| My 7 | CD13 (H) |
| My 9 | CD33 (H) |
| Myeloperoxidase | Myeloperoxidase (H) |
| Myf-4 | Myf-4 (G) |
| MyoD1 | MyoD1 (G) |
| Myogenin | Myf-4 (G) |
| Myoglobin | Myoglobin (G) |
| Myosin heavy chain | Myosin heavy chain (G) |
| NCAM | CD56 (H) |
| Neprilysin | CD10 (G, H) |
| NEU N | NEU N (G) |
| Neurofilaments | Neurofilaments (G) |
| Neuron specific enolase | Neuron specific enolase (G) |
| NFP | Neurofilaments (G) |
| NKI-betab | HMB-45 (G) |
| NKI/C3 | CD63 (G) |
| NSE | Neuron specific enolase (G) |
| O13 | CD99 (G, H) |
| OC125 | CA125 (G) |
| Oct2 | Oct2 (H) |
| Octomer transcription factor | Oct2 (H) |
| p16 | p16 (G) |
| p27kip1 | p27kip1 (G) |
| p53 | p53 (G) |
| p57 | p57 (G) |
| p63 | p63 (G) |
| P504S | AMACR (G) |
| PAN-K | Keratins (G) |
| PAP | Prostate acid phosphatase (G) |
| PECAM-1 | CD31 (G) |
| PEM | Epithelial membrane antigen (G, H) |
| Perforin | Perforin (H) |
| PGM1 | CD68 (G, H) |
| PgR | Progesterone receptor (G) |
| PK antigen | CD77 (H) |
| Placental alkaline phosphatase | Placental alkaline phosphatase (G) |
| PLAP | Placental alkaline phosphatase (G) |
| Platelet glycoprotein IIIa | CD61 (H) |
| PMS2 | hMLH1 |
| Podoplanin | D2-40 |
| PR | Progesterone receptor (G) |
| PRAD1 | Cyclin D1 (H) |
| PrAP | Prostate acid phosphatase (G) |
| Prealbumin | Prealbumin (G) |
| Progesterone receptor | Progesterone receptor (G) |
| Prostate acid phosphatase | Prostate acid phosphatase (G) |
| Prostate specific antigen | Prostate specific antigen (G) |
| PSA | Prostate specific antigen (G) |
| QBEnd10 | CD34 (G, H) |
| Renal cell carcinoma marker | RCC (G) |
| ret | ret (G) |
| RCC | RCC (G) |
| rT3 | CD2 (H) |
| S-100 | S-100 (G) |
| S-Endo-1 | CD146 (G) |
| SGP-2 | Clusterin (H) |
| SMA | Alpha smooth muscle actin (G) |
| SM-ACT | Alpha smooth muscle actin (G) |
| Smad4 | DPC4 (G) |
| SM-MHC | Myosin heavy chain (G) |
| Somatostatin | Hormones (G) |
| SP40 | Clusterin (H) |
| Stem cell factor receptor | CD117 (G) |
| Synaptophysin | Synaptophysin (G) |
| Syndecan-1 | CD138 (H) |
| Synuclein-1 | Synuclein-1 (G) |
| T3 | CD3 (H) |
| T4 | CD4 (H) |
| T6 | CD1a (H) |
| T8 | CD8 (H) |
| T11 | CD2 (H) |
| T64 | Clusterin (H) |
| TAG-72 | B72.3 (G) |
| Tau | Tau (G) |
| T cell antigen receptor | TCR (H) |
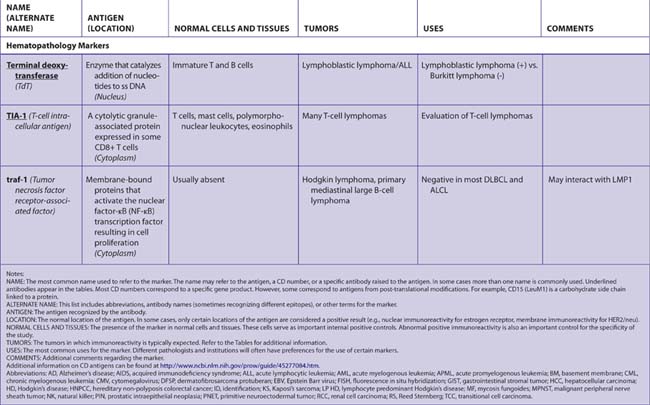
| T cell intracellular antigen | TIA-1 (H) |
| TCR | TCR (H) |
| TdT | Terminal deoxytransferase (H) |
| TE | CD2 (H) |
| Terminal deoxytransferase | Terminal deoxytransferase (H) |
| TH | CD4 (H) |
| Thrombomodulin | CD141 (G) |
| Thyroglobulin | Thyroglobulin (G) |
| Thyroid transcription factor 1 | TTF-1 (G) |
| TIA-1 | TIA-1 (H) |
| TM | CD141 (G) |
| traf-1 | traf-1 (H) |
| Transthyretin | Prealbumin (G) |
| TRPM2 | Clusterin (H) |
| TTF-1 | TTF-1 (G) |
| TTR | Prealbumin (G) |
| Tumor-associated glycoprotein 72 | B72.3 (G) |
| Tumor necrosis factor receptor–associated factor | traf-1 (H) |
| UCHL-1 | CD45Ro (H) |
| UEA 1 | Ulex (G) |
| Ulex | Ulex (G) |
| Vimentin | Vimentin (G) |
| von Willebrand’s factor | Factor VIII (G) |
| VWF | Factor VIII (G) |
| Wilms’ tumor 1 protein | WT1 (G) |
| WT1 | WT1 (G) |
G, general markers; H, hematopathology markers.
Results
The results of studies are incorporated into the surgical pathology report. The following information is included:
ELECTRON MICROSCOPY
Indications for EM Studies
TABLE 7–46 CELLS, TUMORS, AND STRUCTURES WITH CHARACTERISTIC FINDINGS BY ELECTRON MICROSCOPY
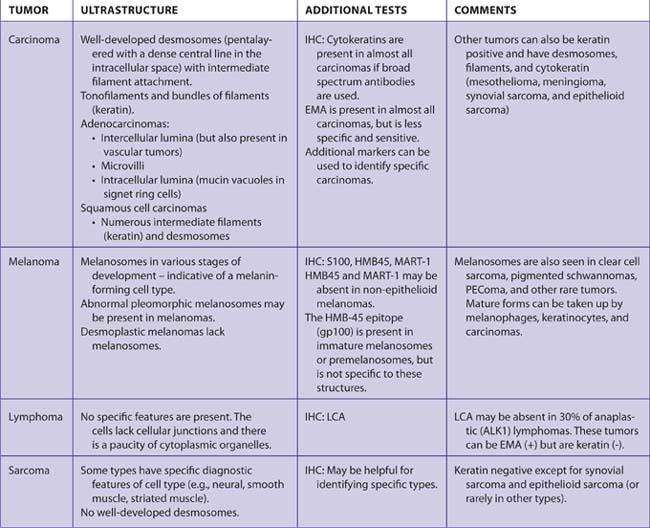
See Tables 7-8, 7-9, 7-22, and 7-47 for additional information.
Method
Ultrastructural details of tissues are lost rapidly. Therefore, fresh tissue must be fixed rapidly and well for EM. Tissues are usually fixed in special fixatives for EM to preserve lipids and glycogen (e.g., 2% paraformaldehyde and 2.5% glutaraldehyde in 0.1 M cacodylate buffer, pH 7.4).
Note: If tissue from a small biopsy is found to be nondiagnostic on H&E, any tissue saved for EM should be retrieved for examination by light microscopy.
Results
A separate electron microscopy report is usually issued. The results should be incorporated into the final diagnosis.19
SNAP FROZEN TISSUE
Frozen tissue is useful for staining (some antibodies only detect antigens in frozen tissue), enzyme studies (muscle biopsies), and to save tissue for DNA or RNA studies.
Indications
All specimens with a question of a lymphoproliferative disorder, sarcomas, unusual tumors, muscle biopsies.
IMMUNOFLUORESCENCE
Like immunoperoxidase studies, immunofluorescence (IF) detects antigens in tissues. However, because amplification of the signal is not used, it is better suited for precise localization of antigen/antibody complexes in tissues or for determining the deposition pattern of immune complexes (e.g., linear vs. granular). Thus, it is most useful for the investigation of diseases related to immune complex deposition such as glomerular diseases and bullous diseases of the skin.
Tissue for IF may be snap frozen (see instructions earlier) or stored in special fixatives for IF. If the specimen is not frozen, special care must be taken to ensure that the biopsy is kept moist in a sealed container.
Immunofluorescence of Skin Lesions
MOLECULAR GENETIC PATHOLOGY
Molecular genetic pathology is the newest subspeciality in pathology with board certification. Molecular diagnostics incorporates many types of techniques for the investigation of genetic alterations in cells and viruses (e.g., Southern blotting, PCR analysis, FISH). It has applications in three main areas:
These studies are especially helpful for hematolymphoid proliferations that are difficult to classify because of the frequent and characteristic rearrangements that occur in many of these disorders. Unlike cytogenetics, the cells need not be viable. However, it is preferable that the nucleic acids are relatively intact. Southern blot and RNA-based PCR (RT-PCR) assays are best performed on fresh or frozen tissues. Formalin-fixed, paraffin-embedded tissue is amenable to DNA-based PCR assays. Some fixatives (e.g., Bouin’s) cause extensive breakage of DNA and may preclude genetic analysis of the tissue.
Indications
CYTOGENETICS
Cytogenetic studies have been demonstrated to be useful in several areas important to pathology:
Cells may be cultured to perform complete karyotype analysis or tissues can be analyzed for specific chromosomal alterations by fluorescence in situ hybridization (FISH).
FISH studies can be performed on cultured cells, cytology specimens, touch preparations, and paraffin-embedded tissues.
Indications
Method for Submitting Tissue
Tissue for karyotyping must be fresh, viable, and relatively sterile. However, tissue may be submitted even if it has not been handled under strictly sterile conditions (contamination is not usually a problem). If specimens are to be held overnight, the tissue should be minced (into 0.1 cm cubes) in a sterile specimen container, covered with culture medium, and held overnight in the refrigerator. Fluids may also be submitted for analysis (especially pleural effusions with a suspicion of mesothelioma).
Results
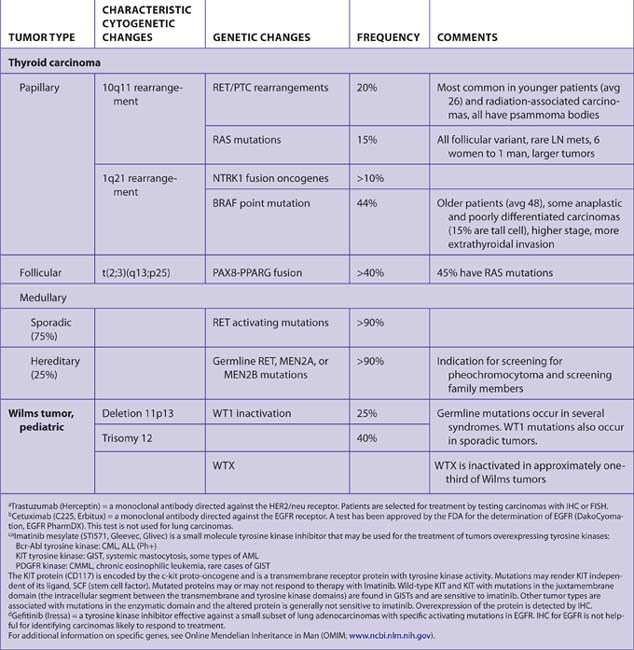
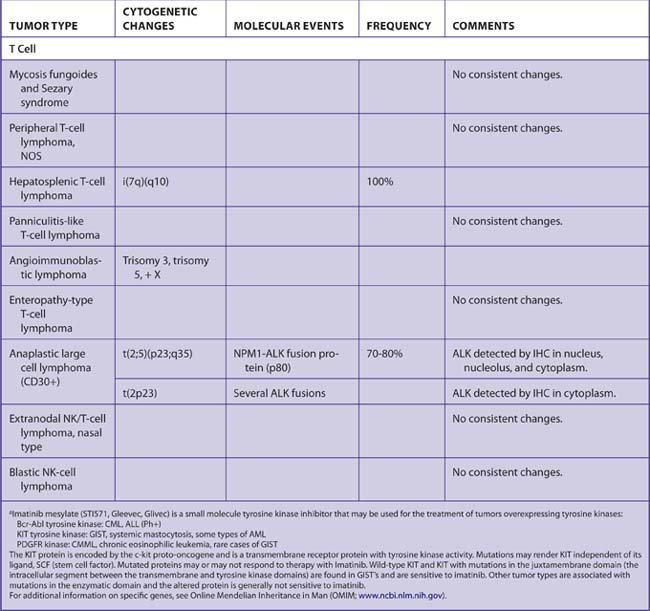
The results of the cytogenetic analysis should be incorporated into the final diagnosis or reported separately (Tables 7-47 and 7-48).
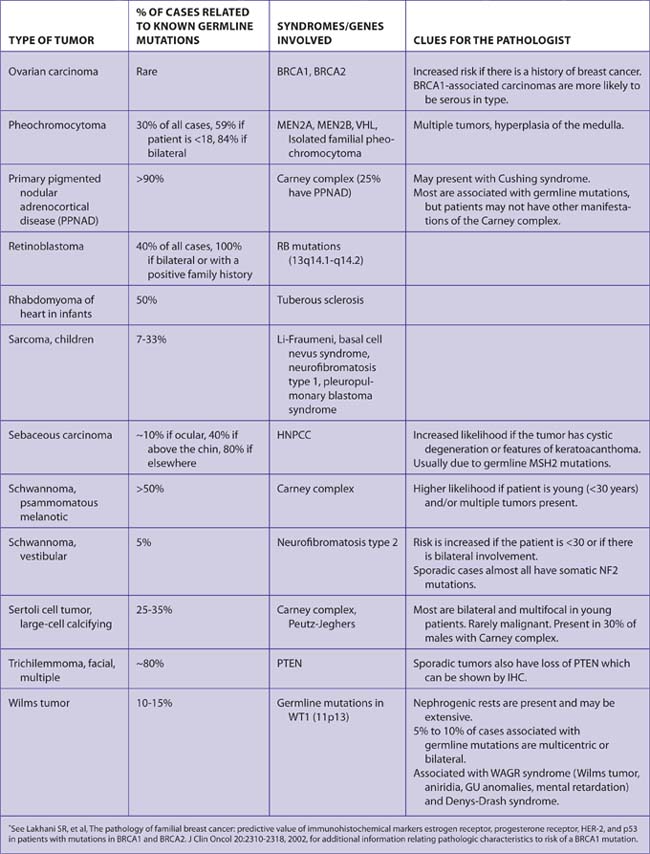
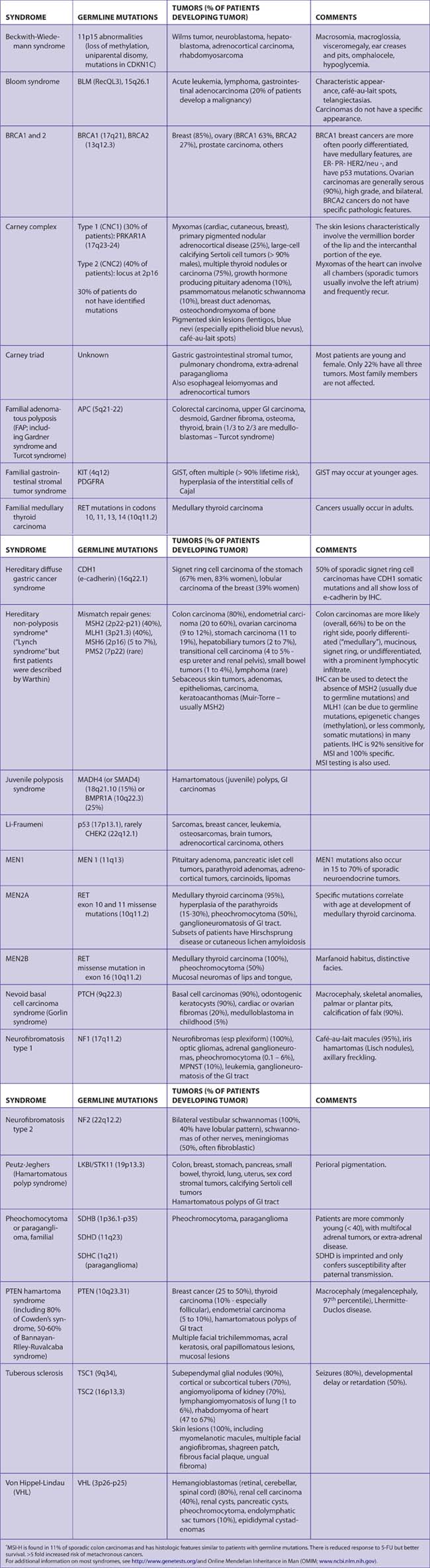
Tumors and Diseases Associated with Germline Mutations (Tables 7-49 and 7-50)
The following features are suggestive of hereditary susceptibility to cancer:
Pathologists can aid in the detection of hereditary carcinomas by being aware of the types and pathologic characteristics of carcinomas associated with these syndromes. Patients with germline mutations are important to identify in order to:
Although the sporadic forms of cancers, in general, far outnumber cases associated with germline mutations, in some cases the appearance or site of a carcinoma is highly suggestive of a known syndrome and further investigation may be warranted.