3
Organization of the Nervous System II
The charm of neurology . . . lies in the way it forces us into daily contact with principles. A knowledge of the structure and function of the nervous system is necessary to explain the simplest phenomena of disease, and it can only be attained by thinking scientifically.
Henry Head
Key Terms
afferent
afferent fibers
anterior (ventral) horn cells
aqueduct of Sylvius
arachnoid granulations
arachnoid mater
autonomic nervous system
bilateral
Broca’s (expressive) aphasia
cerebrospinal fluid (CSF)
choroid plexus
circle of Willis
constructional disturbances
contralateral
cranial nerves
diaphragma sella
dura mater
efferent fibers
encephalitis
enteric nervous system
falx cerebelli
falx cerebri
foramen
foramina
hemorrhage
homeostasis
internal carotid arteries
intervertebral foramina
ipsilateral
ischemia
lesion
meninges
motor fibers
nucleus solitarius
parasympathetic divisions
peripheral nervous system (PNS)
phrenic nerves
pia mater
postganglionic
preganglionic
sensory fibers
septa
somites
spinal peripheral nerves
subarachnoid space
sympathetic divisions
tentorium cerebelli
unilateral
venous sinuses
ventricular system
vertebral artery
Wernicke’s (receptive) aphasia
x-ray
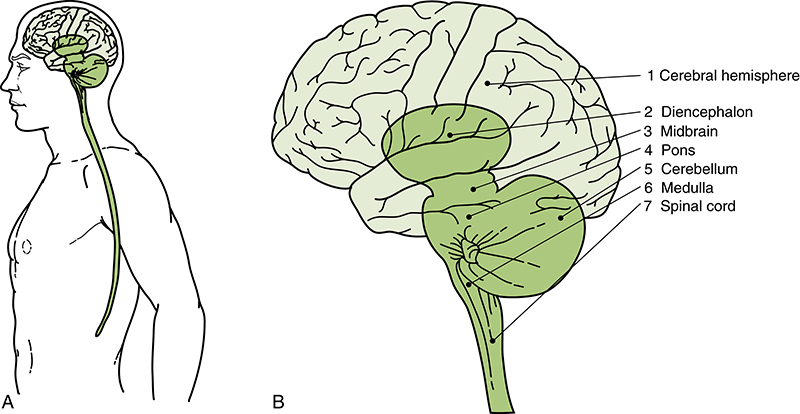
FIGURE 3-1 A, Location of the central nervous system (CNS) in the body. B, Seven major divisions of the CNS: (1) cerebral hemisphere, (2) diencephalon, (3) midbrain, (4) pons, (5) cerebellum, (6) medulla, and (7) spinal cord. The midbrain, pons, and medulla comprise the brainstem. (Reprinted from Martin, J. H. [1996]. Neuroanatomy text and atlas [2nd ed.]. New York: McGraw-Hill.)
The central nervous system (CNS) with its vast network of neurons and neural connections is the controlling influence in the human communication nervous system. Glial cells reside within the CNS and provide structure and metabolic support for the neurons. tThe CNS, however, would not be functional, or necessary, without the lower level structures reviewed in this chapter. The nervous system consists of separate peripheral and central components organized into two parts—not independent from each other, but interdependent on each other. This chapter outlines the functions of the peripheral nervous system that allow this interdependence with the CNS (Fig. 3-1).
Peripheral Nervous System
The peripheral nervous system (PNS) includes (1) the cranial nerves with their roots and rami (branches), (2) the peripheral (spinal) nerves, and (3) the peripheral parts of the autonomic nervous system. The peripheral ganglia are groups of nerve cell bodies located outside the CNS forming an enlargement on a nerve or on two or more nerves at their junction. They are primarily sensory in nature, although motor ganglia are found particularly in the autonomic nervous system.
The cranial nerves exit from the neuraxis at various levels of the brainstem and the uppermost part of the spinal cord. When we use the term peripheral nerves, we are typically referring to the spinal nerves plus their branches.
Cranial nerves V (the trigeminal nerve), VII (the facial nerve), IX (the glossopharyngeal nerve), and X (the vagus nerve) have dedicated sensory ganglia originating from the neural crest during embryonic development. They contain pseudounipolar cell bodies. These sensory ganglia are classified as the trigeminal or semilunar ganglion (cranial nerve V); the geniculate ganglion of cranial nerve VII; the superior and inferior ganglia of cranial nerve IX, or the glossopharyngeal nerve; and the jugular and nodose ganglia of cranial nerve X, or the vagus nerve. The jugular and nodose ganglia are often referred to as the superior and inferior ganglia of the vagus nerve. Cranial nerve VIII, the vestibulocochlear nerve, primarily arises from the otic placodes. Figure 3-2 illustrates the cranial nerve nuclei and their juxtaposition within the brainstem.
Spinal Nerves
Spinal peripheral nerves are described as mixed nerves, meaning they carry both sensory and motor fibers. Each spinal nerve is connected to the spinal cord by two roots: the anterior root and the posterior root. The anterior root of the spinal nerve consists of bundles of nerve fibers that transmit nerve impulses away from the CNS. These nerve fibers are called efferent fibers. Efferent fibers that go to the muscles and make them contract are also called motor fibers. The motor fibers of the spinal nerves originate from a group of cells or motor nuclei in the spinal cord called the anterior (ventral) horn cells. The anterior horn cells are the point of synapse, or connection, with the spinal nerves as they leave the neuraxis. When nerve impulses have left the neuraxis, they have reached what the great British neurophysiologist Charles Sherrington (1857-1952) called the final common pathway, or the terminal route of all neural impulses acting on the muscles.

FIGURE 3-2 An interior (ventral) view of the brainstem with particular emphasis on cranial nerves. (Reprinted from Haines, D. [2006]. Fundamental neuroscience [3rd ed.]. Philadelphia: Churchill Livingstone.)
The posterior root of the spinal nerve consists of afferent fibers that carry information about the sensations of touch, pain, temperature, and vibration into the CNS via the spinal cord. They are also called sensory fibers. The cell bodies of the sensory fibers are a swelling on the posterior root of the spinal nerve called the posterior root ganglion.
The motor and sensory roots leave and enter the spinal cord at the intervertebral foramina, where the roots unite to form a spinal nerve. At this point the motor and sensory fibers mix together.
Clinical principles have been developed based on the organization of the spinal roots that can be used when damage occurs to the spinal cord or spinal nerves. First, recall that a generalization can be made that the anterior, or ventral, half of the spinal cord is devoted to motor or efferent activity, and the posterior, or dorsal, half is devoted to sensory or afferent activity. Which motor and sensory activities are impaired depends on the specific site of lesion in the spinal cord. A lesion, or damaged area, impairs motor and/or sensory activities at and below that cord level depending on the specific site of the lesion and the particular tracks interrupted. Naturally, large lesions in the spinal cord impair both sensory and motor functions.
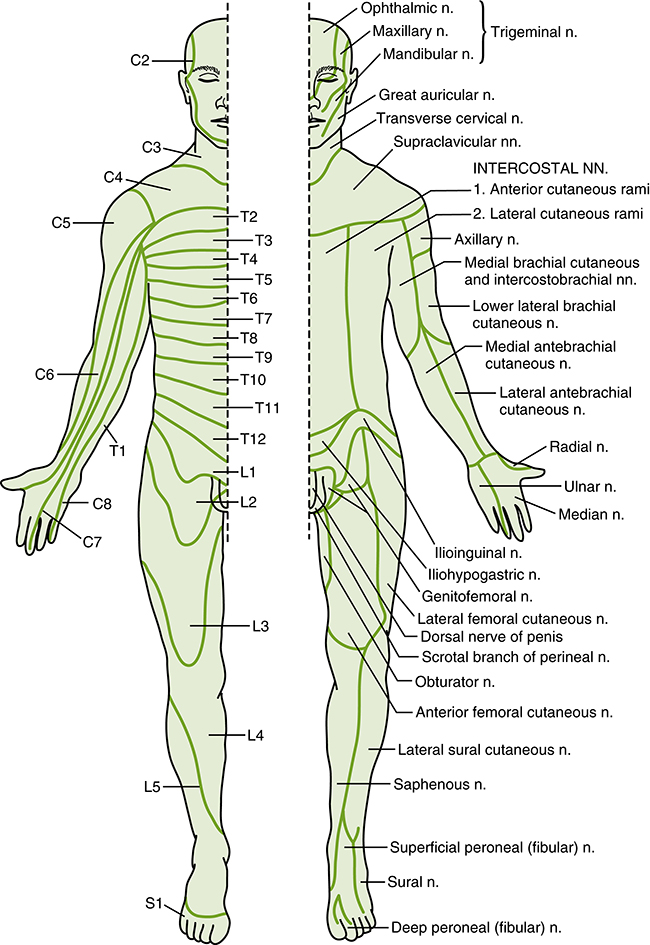
FIGURE 3-3 Dermatomal patterns (left) and peripheral nerve fields (right). (Modified from Gilman, S., & Newman, S. W. [2003]. Manter and Gatz’s essentials of clinical neuroanatomy and neurophysiology. [10th ed.]. Philadelphia: F. A. Davis.)
As discussed later in this text, in early embryologic development, paired structures called somites are formed on the embryo. These somites differentiate into nonneural tissue (e.g., muscle, bone, and connective tissue). This somitic differentiation results in segmentally distributed zones called dermatomes. The dermatome region of each somite gives rise to a myotome, which is a muscle-forming body, and a skin plate, which is involved in formation of dermal tissue. The sensory component of each spinal nerve is distributed to a dermatome. Figure 3-3 shows the segmental distribution of cutaneous innervation, showing on the left side of the figure which area of skin is supplied by which spinal nerve. The peripheral nerve field illustration on the right refers to an area of skin that is supplied by a particular peripheral nerve in that spinal nerve’s distribution. Myotomes generally follow the same distribution as the dermatome, although there is more overlap of control. Box 3-1 lists the general myotome distribution for muscle innervation of upper and lower limbs by spinal nerves.
If there is a high spinal cord injury or lesion at the level of the cervical vertebrae, speech production may be affected because the respiratory muscles are controlled by spinal nerves exiting from the intervertebral foramina of the cervical and thoracic regions. The phrenic nerves, which supply motor signals to the diaphragm, originate mainly from C4, but contributions are also from C3 and C5. If respiration is stopped, death may follow with a lesion above the third, fourth, and fifth cervical nerves. Spinal cord injuries involving the caudal portion of the cord do not affect speech production but are of interest to the speech-language pathologist, who may work with spinal cord–injured patients on language or other related problems. These injuries are instructive in understanding the effect of lesions at various levels of the nervous system. Injuries in the spinal cord may produce partial or complete loss of function at the level of the lesion. Function is also completely or partially impaired below the level of the lesion. Spinal cord injuries must be considered very serious because they impair functions beyond those directly controlled at the lesion point.
Cranial Nerves
The cranial nerves, in contrast to the spinal nerves, are of more significance to the speech pathologist because most of the cranial nerves have some relation to the speech, language, and hearing process and seven of the 12 nerves are directly related to speech production and hearing.
On dissection, the 12 pairs of cranial nerves look like thin, gray-white cords. They consist of nerve fiber bundles surrounded by connective tissue. Like the spinal nerves, they are relatively unprotected and may be damaged by trauma. The cranial nerves leave the brain and pass through foramina of the skull to reach the sense organs or muscles of the head and neck with which they are associated. Some are associated with special senses such as vision, olfaction, and hearing. Cranial nerves innervate the muscles of the jaw, face, pharynx, larynx, tongue, and neck.
Unlike the spinal nerves, which attach to the cord at regular intervals, the cranial nerves are attached to the brain at irregular intervals. They do not all have dorsal (sensory) and ventral (motor) roots. Some have motor functions, some have sensory functions, and some have mixed functions. Their origin, distribution, brain and brainstem connections, functions, and evolution are complicated. (The cranial nerves are discussed in detail in Chapter 7.) They are traditionally designated by Roman numerals: cranial nerve I, olfactory; cranial nerve II, optic; cranial nerve III, oculomotor; cranial nerve IV, trochlear; cranial nerve V, trigeminal; cranial nerve VI, abducens; cranial nerve VII, facial; cranial nerve VIII, acoustic-vestibular; cranial nerve IX, glossopharyngeal; cranial nerve X, vagus; cranial nerve XI, spinal accessory; and cranial nerve XII, hypoglossal.
The cranial nerves from the brainstem are explicitly illustrated in Figure 3-2 and further outlined in Figure 3-4 regarding their location in the anterior (ventral) view and the anterolateral (ventrolateral) view of the brainstem.
Autonomic Nervous System
The innervation of involuntary structures such as the heart, smooth muscles, and glands is accomplished through the autonomic nervous system. Although this system has primarily indirect effects on speech, language, and hearing, the speech-language pathology and audiology student must be familiar with its contribution to total body function to understand how involuntary but vital functions such as hormonal secretions, visual reflexes, and blood pressure are controlled within the nervous system.

FIGURE 3-4 An anterolateral (ventrolateral) view of the brainstem with special emphasis on cranial nerves. Note the position and relations of the foramen of Luschka. (Reprinted from Haines, D. [2006]. Fundamental neuroscience [3rd ed.]. Philadelphia: Churchill Livingstone.)
The autonomic nervous system is distributed throughout both the CNS and the PNS. The enteric nervous system, which is formed by neuronal plexus in the gastrointestinal tract, is considered a division of the autonomic nervous system. Enteric functioning has a direct effect on the deglutition and digestion of food.
Aside from the enteric system that deals directly with swallowing and digestion, the major divisions of the autonomic nervous system are the sympathetic and parasympathetic divisions, which have almost antagonistic functions. The sympathetic system is the body’s alerting system, sometimes referred to as the fight-or-flight system. This part of the autonomic nervous system is responsible for such preparatory measures as accelerating the heart rate, causing constriction of the peripheral blood vessels, raising the blood pressure, and redistributing the blood so that it leaves the skin and intestines to be used in the brain, heart, and skeletal muscles if needed. It serves to raise the eyelids and dilate the pupils. The sympathetic part also decreases peristalsis (the propelling contractions of the intestine) and closes the sphincters.
The parasympathetic part of the autonomic nervous system has an almost opposite calming effect on bodily function. It serves to conserve and restore energy by slowing the heart rate, increasing intestinal peristalsis, and opening the sphincters. As a result of parasympathetic action, other functions, such as increased salivation and increased secretion of the glands of the gastrointestinal tract, may take place.
The autonomic nervous system is composed of both efferent (conducting away from the CNS) and afferent (conducting toward the CNS) nerve fibers. Several similarities and differences exist between the neural control of skeletal muscle and visceral effectors such as smooth muscle. Lower motor neurons function as the final common pathway linking the CNS to skeletal muscle fibers. Similarly, the sympathetic and parasympathetic outflows serve as the final, but often dual, common neural pathway from the CNS to visceral effectors. However, unlike the somatic nervous system, the peripheral visceral motor pathway consists of two neurons. The first is the preganglionic neuron, which has its cell body in either the brainstem or the spinal cord. Its axons project as a thinly myelinated preganglionic fiber to an autonomic ganglion. The second, the postganglionic neuron, has its cell body in the ganglion and sends unmyelinated axon (postganglionic fiber) to visceral effector cells such as smooth muscle. Typically, parasympathetic ganglia are close to the effector tissue and sympathetic ganglia are close to the CNS. Consequently, parasympathetic pathways typically have long preganglionic fibers and short postganglionic fibers, whereas sympathetic pathways more often have short preganglionic and long postganglionic fibers.
The autonomic system also provides neural control of smooth muscle, cardiac muscle, glandular secretory cells, or a combination of these. For example, the gut wall is composed of smooth muscle and glandular epithelium. The sympathetic and parasympathetic systems have overlapping and, as stated previously, antagonistic influences on those viscera located in body cavities and on some structures of the head such as the iris. Visceral targets are also present in the body wall and limbs. These are found in skeletal muscle (blood vessels) and in the skin (blood vessels and sweat glands). Visceral structures of the body wall and extremities are generally regulated by the sympathetic division alone. Thus the sympathetic outflow has a global distribution in that it innervates visceral structures in all parts of the body, whereas the parasympathetic outflow serves only the head and body cavities.
Rarely is autonomic activity solely sympathetic or parasympathetic. Both parts work together in the autonomic nervous system along with the endocrine system to maintain the stability of the body’s internal environment or homeostasis. The endocrine system is a group of glands and other structures that release internal secretions called hormones into the circulatory system. These hormones influence metabolism and other body processes. The endocrine system includes such organs as the pancreas, pineal gland, pituitary gland, gonads, thyroid, and adrenal glands. These work more slowly than the autonomic nervous system.
The integration of the autonomic activity with endocrine and somatic responses, allowing homeostasis to be maintained, is regulated by the hypothalamus. Evidence exists for a network of central neuronal circuits that includes the hypothalamus and the insula, the amygdala, and an area in the midbrain called the periaqueductal gray matter. These structures receive input from the nucleus solitarius, a prominent nucleus of the medulla that receives input from all visceral organs. Input is also received from other nuclei in the brainstem and spinal cord. This network is referred to as the central autonomic network and is probably responsible for adjustments to basic cardiovascular and respiratory functions as they relate to a range of body activities, such as food intake, emotional behavior, and mental activity.13
As stated earlier, the autonomic nervous system is of importance to the speech-language pathologist because of its indirect effect on communication functioning. A good example of the power of the autonomic nervous system is the sweaty palms, dry mouth, blushing, and upset stomach some people experience before delivering a speech. Those indirect effects may make a great deal of difference in how well one communicates.
Protection and Nourishment of the Brain
The brain and the spinal cord, which make up part of the CNS, PNS, and autonomic nervous system and house most of their mechanisms, must be protected and nourished to continue to function. Following is a discussion of the protection and nourishment of these structures.
Meninges
The spinal cord and brain are the major coordinating and integrating structures for all physical and mental activities of the body and fortunately are well protected. The brain and spinal cord are covered by layers of tissue called the meninges. Within certain layers of these meninges is a cushioning layer of fluid called cerebrospinal fluid. The meninges are composed of three membranes; moving from the outermost to the innermost covering, they are known as the dura mater (“tough mother” in Latin), the arachnoid mater, and the pia mater.
The dura mater of the spinal cord is continuous with that of the brain through the opening in the skull called the foramen magnum. It consists of two layers that are closely united except where, in certain spots, they separate. In some parts of the dura mater of the brain they separate to form the venous sinuses (Fig. 3-5). In other parts of the brain, the inner layer also separates from the outer layer to reflect inward and form partitions, described as complex folds that divide the contents of the cranial cavity into different cerebral subdivisions. These infoldings or septa of the dura join with those formed in the opposite hemisphere to create three different double-layered partitions: the falx cerebri, the tentorium cerebelli, and the falx cerebelli (Fig. 3-6).

FIGURE 3-5 The relation of the meninges to the brain and spinal cord and to their surrounding bony structures. The dura is represented in light green, the arachnoid in dark green. (Reprinted from Haines, D. E. [2000]. Neuroanatomy: An atlas of structures, sections, and systems [5th ed.]. Baltimore: Lippincott Williams & Wilkins.)
The falx cerebri develops first as two portions that become a single continuous structure later. It reflects downward between the hemispheres, attaching anteriorly and laterally to points on the skull and posteriorly to another partition, the tentorium cerebelli (also known simply as the tentorium). The falx cerebelli is located below the tentorium cerebelli on the middle of the occipital bone. This small dural infolding extends into the space between the cerebellar hemispheres, attaching to the occipital crest of the skull and the posterior portion of the tentorium. Part of it encloses the occipital venous sinuses. The tentorium cerebelli is the second largest of the dural folds. It attaches to bony walls of the skull in the temporal, occipital, and parietal regions. The tentorium cerebelli is the infolding found between the cerebral hemispheres above and the cerebellar hemispheres below. It is only found in mammals and birds, being absent in fish, amphibians, and reptiles.11 There is another very small infolding of the dura, the diaphragma sella, which forms the roof of the sella turcica, the structure that encloses the pituitary gland. There is a tiny hole in the center of it, allowing the stalk (or infundibulum) of the pituitary to pass through. The anterior and posterior intercavernous sinuses are found in their respective edges of the diaphragma sella. These infoldings are illustrated in Figures 3-6 and 3-7 They are pictured as they appear on a magnetic resonance imaging scan.
When you consider the placement of these sturdy partitions being under and between the cerebral hemispheres as well as over and between the cerebellar hemispheres, it is fairly easy to understand how the dural infoldings brace the brain against rotary displacement.
There is no subdural space between the dura mater and the next layer of the meninges. When there is subdural space identified, it means that tissue damage has occurred to the deepest layer, creating a space.
Because there is no normal subdural space, the next meningeal layer can be found immediately below the dura. This second layer is the arachnoid mater. This membrane bridges over the sulci or folds of the brain. In some areas it projects into the venous sinuses to form arachnoid villi. The arachnoid villi aggregate to form the arachnoid granulations. The arachnoid granulations are from where the cerebrospinal fluid diffuses into the bloodstream.
Beneath the arachnoid mater layer is a space called the subarachnoid space, which is filled with cerebrospinal fluid. All cerebral arteries and veins, as well as the cranial nerves, pass through this space. This is why you often hear of subarachnoid hemorrhage or bleed because damage to the brain has resulted in artery or vein tears and blood is released into this subarachnoid space.

FIGURE 3-6 Veins, meninges, and dural sinuses. (Reprinted from Drake, R., Vogl, W., & Mitchell [2006]. Gray’s anatomy for students [2nd ed.]. Philadelphia: Churchill Livingstone.)
The third meningeal layer, the pia mater, closely adheres to the surface of the brain, covering the gyri (ridges) and going down into the sulci. The pia mater also fuses with the ependyma (a cellular membrane lining the ventricles) to form the choroid plexus of the ventricles. Figure 3-8 illustrates the meningeal layers.
Ventricular System
The ventricular system of the brain has three parts: the lateral ventricles, the third ventricle, and the fourth ventricle. These actually are small cavities within the brain joined to each other by small ducts and canals. Each ventricle contains a tuftlike structure called the choroid plexus, which mainly is concerned with the production of cerebrospinal fluid.
Figure 3-9 is an example of a midsagittal view of the brain showing the third and fourth ventricles in relation to the cerebral aqueduct and nearby structures.
The lateral ventricles are paired, one in each hemisphere. Each is a C-shaped cavity and can be divided into a body, located in the parietal lobe, and anterior, posterior, and inferior or temporal horns, extending into the frontal, occipital, and temporal lobes, respectively. The lateral ventricle is connected to the third ventricle by an opening called the intraventricular foramen, or the foramen of Munro. The choroid plexus of the lateral ventricle projects into the cavity on its medial aspect (see Fig. 3-9).
The third ventricle is a small slit between the thalami. It also is connected to the fourth ventricle through the cerebral aqueduct or the aqueduct of Sylvius. The choroid plexus is situated above the roof of the ventricle.
The fourth ventricle sits anterior to the cerebellum and posterior to the pons and the superior half of the medulla. It is continuous superiorly with the cerebral aqueduct and the central canal below. The fourth ventricle has a tent-shaped roof, two lateral walls, and a floor (see Fig. 3-9). It contains three small openings: the two lateral foramina of Luschka and the median foramen of Magendie. Through these openings the cerebrospinal fluid enters the subarachnoid space. The ventricular system serves as a pathway for the circulation of the cerebrospinal fluid. The choroid plexus of the ventricles appears to secrete cerebrospinal fluid actively, although some of the fluid may originate as tissue fluid formed in the brain substance.

FIGURE 3-7 Axial (A), coronal (B), and sagittal (C) T1-weighted magnetic resonance images showing the relations of the falx cerebri (A and B) and the tentorium cerebelli (B and C). Note the positions of the right and left supratentorial compartments and the infratentorial compartment in relation to these large dural reflections in all three planes. (Reprinted from Haines, D. [2006]. Fundamental neuroscience [3rd ed.]. Philadelphia: Churchill Livingstone.)
Cerebrospinal Fluid
The brain and the spinal cord are suspended in a clear, colorless fluid called cerebrospinal fluid (CSF), which serves as a cushion between the CNS and the surrounding bones, thereby protecting the brain against direct trauma. This fluid aids in regulation of intracranial pressure, nourishment of the nervous tissue, and removal of waste products.
The path of circulation of the CSF is illustrated in Figure 3-10. It flows from the lateral ventricles into the third ventricle, to the fourth ventricle, and into the subarachnoid space. It then travels to reach the inferior surface of the cerebrum and moves superiorly over the lateral aspect of each hemisphere. Some of it moves into the subarachnoid space around the spinal cord.
The CSF produced by the choroid plexus passes through the ventricular system to exit the fourth ventricle through the foramina of Luschka and Magendie. At this point, the CSF enters the subarachnoid space, which is continuous around the brain and spinal cord. The CSF in this subarachnoid space provides the buoyancy necessary to prevent the weight of the brain from crushing nerve roots and blood vessels against the internal surface of the skull. The weight of the brain, approximately 1400 g in air, is reduced to 45 g when it is suspended in CSF. Consequently, the tethers formed by delicate connective tissue strands traversing the subarachnoid space, the arachnoid trabeculae (see Fig. 3-8), are adequate to maintain the brain in a stable position.

FIGURE 3-8 Relation of the brain to the dura, arachnoid, and pia maters. (Redrawn from Bullock, T. H., Orkland, R., & Grinnel, A. [1977]. Introduction to the nervous system. San Francisco: W. H. Freeman.)
The movement of the CSF through the ventricular system and the subarachnoid space is influenced by two major factors. A subtle pressure gradient exists between the points of production of CSF (the choroid plexus in the cerebral ventricles) and the points of transfer into the venous system (arachnoid villi). Because CSF is not compressible, it tends to move along this gradient. It also moves into the subarachnoid space by purely mechanical means, including gentle movements of the brain on its arachnoid trabecular tethers during normal activities and the pulsations of the numerous arteries found in the subarachnoid space.
Blockage of the CSF movement or a failure of the absorption mechanism results in the accumulation of fluid in the ventricles or around the brain tissue (Fig. 3-11). This results in hydrocephalus and is characterized by an increase in CSF volume enlargement of one or more of the ventricles, and usually an increase in CSF pressure.

FIGURE 3-9 The brain ventricles and the cerebrospinal fluid. This is a transparent view, looking from the left side of the brain. The two lateral ventricles communicate with the third ventricle, which in turn communicates with the fourth ventricle. (Modified from Boron, W., & Boulpaep, E. [2009]. Medical Physiology [2nd ed.]. St. Louis: Elsevier.)

FIGURE 3-11 Comparison of normal and hydrocephalic brains in sagittal (A), axial (B), and coronal (C) planes as seen on magnetic resonance images. (Reprinted from Haines, D. [2006]. Fundamental neuroscience [3rd ed.]. Philadelphia: Churchill Livingstone.)
The CSF is important in medical diagnostic procedures. The pressure of the fluid can be measured; if it is abnormally high, intracranial tumor, hemorrhage, hydrocephalus, meningitis, or encephalitis may be suspected. Chemical and cell studies may be made on CSF that is drawn out of the nervous system through a procedure called a lumbar puncture or spinal tap. This route also may be used to inject drugs to combat infection or induce anesthesia. Circulation of the CSF is illustrated in Figure 3-10.
Blood Supply of the Brain
The blood serves the brain much as food serves the body; it nourishes it by supplying its most important element, oxygen. The brain uses approximately 20% of the blood in the body at any given time and requires approximately 25% of the oxygen of the body to function maximally. Initially blood is delivered to the brain through four main arteries. Two large internal carotid arteries are on either side of the neck; these are a result of bifurcation, or splitting, of the common carotid artery from the heart. The other two main arteries supplying the brain are the vertebral arteries (Fig. 3-12).
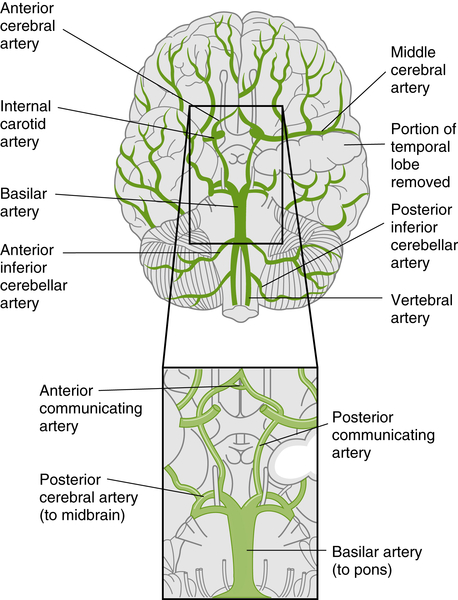
FIGURE 3-12 The major arteries of the brain. Ventral view: the enlargement of the boxed area showing the circle of Willis. (From Aitken, L., Marshall, A., & Chaboyer, W. [2012]. ACCCN’s Critical Care Nursing [2nd ed.]. Mosby Australia: Sydney.)
Internal Carotid Arteries and Their Branches
The internal carotid arteries ascend in the neck and pass through the base of the skull at the carotid canal of the temporal bone. Each artery then runs horizontally forward and perforates the dura mater. After entering the subarachnoid space, the artery turns posteriorly and, at the medial end of the lateral sulcus, divides into the anterior and middle cerebral arteries. Other cerebral arteries are also given off by the internal carotid artery. The ophthalmic artery supplies the eye, the frontal area of the scalp, the dorsum of the nose, and the ethmoid and frontal sinuses. The posterior communicating artery runs posteriorly above the oculomotor nerve and joins the posterior cerebral artery, forming part of the circle of Willis. The anterior communicating artery joins the two anterior cerebral arteries together in the circle of Willis (Fig. 3-13).

FIGURE 3-13 The cerebral arterial circle (circle of Willis; A) The circle of Willis. (From Nolte, J. [2010]. Essentials of the Human Brain. St. Louis: Mosby.) (B) The large cerebral arteries: anterior, middle, and posterior. The area supplied by the anterior cerebral artery is green, the area supplied by the middle cerebral artery is pink, and the area supplied by the posterior cerebral artery is yellow. (From Lundy-Eckman, L. [2013]. Neuroscience [4th ed.]. Saunders: St. Louis.)
Through these cortical branches, the internal carotid artery provides the blood supply to a large portion of the cerebral hemisphere. The anterior cerebral artery supplies the medial surface of the cortex as far back as the parietal-temporal-occipital sulcus. It also supplies the so-called leg areas of the motor strip. Branches of this artery supply a small portion of the caudate nucleus, lentiform nucleus, and internal capsule. Following is a summary of the internal carotid artery branches (see Fig. 3-12):

FIGURE 3-14 Middle cerebral artery. (From Kenyon, K., & Kenyon, J. [2010]. The Physiotherapist’s Pocket Book [2nd ed.]. St. Louis.)
• The ophthalmic artery gives rise to the central artery of the retina; damage to this artery (including ophthalmic aneurysms) causes ipsilateral visual loss.
• The posterior communicating artery joins the posterior cerebral artery and the anterior choroidal artery and follows along the optic tract.
• The anterior cerebral artery passes superiorly over the optic chiasm and is joined by its counterpart the anterior communicating artery; it supplies blood to the hypothalamus and the optic chiasm.
• The middle cerebral artery usually is the larger of the two terminal branches of the internal carotid artery (Fig. 3-14).
The middle cerebral artery is the largest branch of the internal carotid. Its branches supply the entire lateral surface of the hemisphere except for the small area of the motor strip supplied by the anterior cerebral artery, the occipital pole, and the inferolateral surface of the hemisphere, which is supplied by the posterior cerebral artery. The middle cerebral artery’s central branches also provide the primary blood supply to the lentiform and caudate nuclei and the internal capsule.
Vertebral Artery and Its Branches
The vertebral artery passes through the foramina in the upper six cervical vertebrae and enters the skull through the foramen magnum. It passes upward and forward along the medulla and at the lower border of the pons and joins the vertebral artery from the opposite side to form the basilar artery. Before the formulation of the basilar artery, several branches are given off, including the following:
• The meningeal branches, which supply the bone and dura of the posterior cranial fossa
• The posterior spinal artery, which supplies the posterior third of the spinal cord
• The anterior spinal artery, which supplies the anterior two thirds of the spinal cord
• The posterior inferior cerebellar artery, which supplies part of the cerebellum, the medulla, and the choroid plexus of the fourth ventricle
• The medullary arteries, which are distributed to the medulla
After the basilar artery is formed by the union of the opposite vertebral arteries, it ascends and then divides at the upper border of the pons into the two posterior cerebral arteries. These arteries supply the inferolateral surface of the temporal lobe and the lateral and medial surfaces of the occipital lobe (i.e., the visual cortex). They also supply parts of the thalamus and other internal structures (see Fig. 3-13).
Other branches of the basilar artery include the following (see Fig. 3-12):
• The pontine arteries, which enter the pons
• The labyrinthine artery, which supplies the internal ear
• The anterior inferior cerebellar artery, which supplies the anterior and inferior parts of the cerebellum
• The superior cerebellar artery, which supplies the superior portion of the cerebellum
Circle of Willis
The circle of Willis, or the circulus arteriosus, is formed by the anastomosis of the two internal carotid arteries with the two vertebral arteries. The anterior communicating, anterior cerebral, internal carotid, posterior communicating, posterior cerebral, and basilar arteries are all part of the circle of Willis (see Fig. 3-13). This formation of arteries allows distribution of the blood entering from the internal carotid artery or vertebral artery to any part of both hemispheres. Cortical and central branches arise from the circle and further supply the brain.
The bloodstreams from the internal carotid artery and vertebral artery on both sides come together at a certain point in the posterior communicating artery. At that point the pressure is equal, and they do not mix. Should, however, the internal carotid artery or the vertebral artery be occluded or blocked, the blood will pass forward or backward across that point to compensate for the reduced flow. The circle of Willis also allows blood to flow across the midline of the brain if an artery on one side is occluded. The circle of Willis thereby serves a safety valve function for the brain, allowing collateral circulation (or flow of blood through an alternate route) to take place if the flow is reduced to one area. The state of a person’s collateral circulation helps determine the outcome after a vascular insult, such as a stroke, occurs and affects blood flow to the brain.
General Principles of Neurologic Organization
Certain fundamental principles of neurologic organization are particularly crucial to the understanding and diagnosis of communication disorders. The following principles will also be built on in later chapters.
Contralateral Motor Control
The first principle to remember is that major movement patterns in human beings have contralateral neurologic control in the brain. The arms and legs are represented in the motor strip of the cerebral cortex in a contralateral fashion. In other words, the cerebral hemisphere on one side of the body controls movements of the arm and leg on the other side of the body. This contralateral motor control is brought about by the crossing of the major voluntary motor pathway at the level of the lower brainstem. Auditory and visual sensory systems also have some contralateral organization, a fact that will become clinically important (see Chapters 5 and 6). The cranial nerves, as discussed subsequently, have mixed control so that you have to know which cranial nerve and/or muscle group considered in order to know the innervation pattern.
If a patient sent to the speech-language pathologist has a severe language disorder and some paralysis of the right arm and leg, these symptoms suggest that the brain lesion causing this motor deficit is probably in the left cerebral hemisphere (Fig. 3-15). The severe language disturbance accompanying the right limb paralysis serves as a confirming sign of left-sided brain lesion because in the majority of the population language dominance is located in the left hemisphere. Why the nervous system provides contralateral motor control of the limbs is not completely known, but the fact illustrates that knowledge of principles of neurologic organization can be used to posit the location of causative lesions seen in neurology and speech pathology.
Ipsilateral Motor Control
If a lesion occurs in the nervous system below the crossing of the major descending motor pathways, the effect is observed below the level of the lesion on the same side of the body where the lesion occurs. In many spinal cord injuries, paralysis and sensory loss occur below the point of injury. Thus a second important principle is to determine whether effects of lesions are ipsilateral or contralateral. An illustration of how the tongue might deviate ipsilateral to a lesion is shown in Figure 3-16. In this figure, the tongue is paralyzed on the left side from surgery to remove a lymph node from the left side of the neck, resulting in damage to the ipsilateral hypoglossal nerve fibers.
Bilateral Speech Motor Control
For the most part, the midline muscles of the body in the head, neck, and trunk tend to be represented bilaterally, and the nerve fibers supplying these regions, with certain exceptions, descend from both cerebral hemispheres. This bilateral neural control provides smooth, symmetric movement for those muscles used in speaking: the lips, tongue, soft palate, jaw, abdominal muscles, and diaphragm. The principle of bilateral control of speech muscles suggests that serious involvement of the speech muscles usually results from diseases that affect bilateral neurologic mechanisms. With unilateral damage to the nervous system, effects on speech are generally less serious, and compensatory mechanisms are made available from the other side of the midline speech system.
Representation of the body is found in an inverted fashion on the motor areas of the cerebral cortex. Pathways concerned with movements of the lower limbs originate in the upper parts of the motor strip, whereas movements of the head and neck originate at the lower end of the motor strip, just above the sylvian fissure. The area surrounding the left sylvian fissure contains major areas for language processing. The anatomic relations of motor speech areas and language suggest that speech and language disturbances may commonly coexist because of the close proximity of their control areas on the cortex.
An example of bilateral damage effects of motor speech lesions may be seen in spastic dysarthria. When damage to the upper motor neurons occurs, spasticity results. With spasticity, affected muscles (in this case, those dealing with articulation, respiration, and phonation) show an increased resistance to passive movements or movement manipulation. The more rapidly the individual tries to move the articulators (or even an upper or lower extremity, as in cerebral palsy) the greater is the resistance to the movement. In spastic dysarthria, bilateral damage to the upper motor neurons usually is caused by stroke, head trauma, or a degenerative disease (see Chapter 8). The bilateral damage is within both direct activation pathways (corticobulbar and corticospinal tracts) and indirect pathways (extrapyramidal tract).

FIGURE 3-16 Paralysis of the tongue on the left side. Removal of a lymph node from the left side of the neck (A, arrow) inadvertently resulted in damage to peripheral fibers of the hypoglossal nerve on that side. The tongue deviates to the left (side of the lesion) on protrusion (B). (Reprinted from Haines, D. [2006]. Fundamental neuroscience [3rd ed.]. Philadelphia: Churchill Livingstone.)
Unilateral Language Mechanisms
An impressive facet of cerebral asymmetry is that language mechanisms, for the most part, are unilaterally controlled in the brain, in contrast to the bilateral speech muscle mechanism. Handedness usually appears between 18 and 48 months of age. Among the adult population, more than 95% of right-handed people have their language mechanisms in the left cerebral hemisphere. Language dominance the world over is primarily in the left brain. Left-handed people are more variable. Some are right brained for language; others have bilateral representation of language. Figure 3-17 illustrates the left hemisphere and its relation to the motor and sensory (receptive) language areas. The obvious clinical principle suggested by these facts is that major language disturbance is a neurologic sign of left cerebral injury and that the left hemisphere has special anatomic properties for language.
Scheme of Cortical Organization
Students and clinicians should have in mind a general scheme of organization of the cortex because it is the site of most language functions. Although any such scheme is oversimplified and exaggerated, it nevertheless provides a crude but workable framework for conceptualizing functional localization. Later chapters in this text detail the specifics of cortical localization.
The right and left hemispheres may be designated as nonverbal and verbal, and the anterior and posterior portions may be characterized as motor and sensory areas. The central sulcus divides the cerebral hemispheres into anterior and posterior regions. Figure 3-18 provides a lateral view of the cerebral hemisphere, showing lobes and the sensory and motor cortices. In human beings, approximately half of the volume of the cerebral cortex is taken up by the frontal cortex. The frontal lobe contains the primary motor cortex, the premotor cortex, and Broca’s area, the primary motor speech association area. In the anterior portion of the frontal lobes are the prefrontal areas, which are generally concerned with behavioral control of both cognitive and emotional functions.
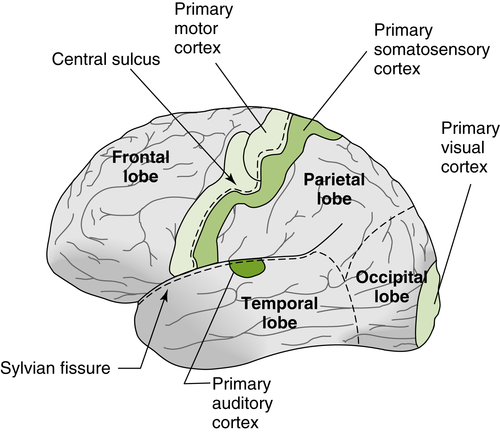
FIGURE 3-18 Lateral view of the cerebral hemisphere. Dashed lines demarcate major lobes. (Reprinted from Castro, A., Merchut, M. P., Neafsey, E. J., & Wurster, R. D. [2002]. Neuroscience: An outline approach. Philadelphia: Mosby.)
Castro et al.5 described lesions in the prefrontal cortex that may produce an “internal agnosia or the inability to communicate with one’s limbic system as though suffering from an impairment of the ‘sixth sense’ that distinguishes appropriate from inappropriate behaviors and right from wrong.” Mental shifts become difficult, and perseveration and rigidity are observed, as are a lack of self-awareness and a tendency toward concreteness. In brief, the frontal lobe appears to excel in the control, integration, and regulation of emotional and cognitive behavior. Cortical areas of the left hemisphere that mediate the processing of language are shown in Figure 3-19. Lesions in the inferior frontal lobe may result in Broca’s, or expressive, aphasia, whereas damage to the angular gyrus, the supramarginal gyrus, and the superior temporal gyrus may result in Wernicke’s, or receptive, aphasia.
In contrast, the posterior cortex appears to dominate the control, integration, and regulation of sensory behavior. The defects arising from the posterior cortex are related to the specific sensory association areas implicated by a lesion.
The occipital lobe, as previously noted, contains the primary visual cortex and visual association areas. Deficits in the primary cortex result in blind spots in the visual field, and total destruction of the cortex produces complete blindness. Visual imperception and agnosias (see Chapter 5) are associated with the visual association areas.

FIGURE 3-19 Cortical areas that mediate the processing of language. Lesions in the pars orbitalis and pars triangularis of the inferior frontal lobe result in Broca’s aphasia, whereas damage in the supramarginal and angular gyri and adjacent superior temporal gyrus results in Wernicke’s aphasia.
The left parietal lobe is associated with constructional disturbances and visuospatial defects. Disorders of recognition, called agnosias, are common. The inferior parietal lobe is concerned with language association tasks, and lesions there cause defects in reading and writing. Contralateral neglect is associated with hemispheric lesions of the nondominant parietal association cortex. Such patients usually neglect stimuli on their left, as illustrated in Figure 3-20. In extreme cases the patient does not recognize the left side of his or her own body, a condition termed asomatognosia. An example is when a patient has a left neglect and ignores the left side when getting dressed or grooming.
The temporal lobe on the left is concerned with hearing and related functions. It contains the primary auditory and auditory association areas. Auditory memory storage and complex auditory perception are among the functions of the temporal lobe. An area known as the speech zone surrounds the sylvian fissure and appears to contain the major components of the language mechanism. Damage in the speech zone produces Wernicke’s aphasia (see Fig. 3-19).
With a clinical knowledge of primary sensory and related association areas and behavioral correlates to these areas, the speech-language pathologist is able to infer the approximate location of a lesion from the patient’s behavioral symptoms and to recognize the well-known speech-language syndromes associated with cortical dysfunction. The general clinical principle is that specific cortical deficits can be associated with specific behavioral syndromes. A major exception to this is difficulty with word-finding or naming which is typically a non-localizable symptom.
Neurodiagnostic Studies in Speech and Language
The structures of the brain believed to be critical for speech and language function have been established by what is called the clinicopathologic method in neurology. Developed into a powerful technique by the great French neurologist Jean Charcot, this method establishes a relation between the site of a lesion and the behavioral functions that are lost or modified. The underlying assumption is that the area of lesion is related to the lost or disordered function. This simple logic is important in clinical neurology; it forms the basis of neurologic diagnosis and is the foundation of the historically traditional neurologic examination. Some of these neurobehavioral signs have been discussed in Chapter 2 and in this chapter.
Fortunately for the evolution of medical science and clinical treatment, the field of neurology has been advanced through the years by technology that has vastly clarified the actual sites of lesions and made diagnoses more valid and reliable, now through relatively noninvasive means. Objective neurodiagnostic tests, such as computed tomography (CT), magnetic resonance imaging (MRI), electroencephalography (EEG), and evoked related potentials (ERPs) as well as other clinical neurodiagnostic tests, have established the value of the clinicopathologic method in medicine yet dramatically enhanced the identification and treatment of neurologic disorders, including those which impact communication. Speech-language pathologists (SLPs) are expected to be somewhat familiar with these types of studies and use the reported information (if available) to plan diagnostic and treatment procedures. Use of the information provided by these studies can also help the SLP to identify inconsistencies between the behaviorally based diagnostic findings and the findings of the reported study. Speech and language function is quite vulnerable to neurologic changes; the results of the imaging or electrophysiologic study may not be consistent, perhaps warranting more exploration by one or both. Therefore, as we come to the end of our overview of the communicative nervous system, a brief discussion of some of the more commonly used neurodiagnostic methodologies that the SLP might find reported in the medical record or research articles seems fitting to include at this point.
Static Brain Imaging
CT and MRI were the foundation tools of early neurologic imaging, permitting study of the structure of the human brain with a degree of detail that is occasionally comparable with the detail revealed by postmortem examination. In fact, MRI, which generates fine cross sections of brain structure without penetrating radiation, may even go beyond postmortem examination because it allows views of multiple slices of the brain.
The CT scan yields a three-dimensional representation of the brain (Fig. 3-21), unlike the conventional radiograph, which provides a two-dimensional projection of a three-dimensional object. On a radiograph, the body appears on x-ray films as overlapping structures that are sometimes difficult to distinguish. The CT scanner uses an x-ray beam that is passed through the brain from one side of the head, and the radiation not absorbed by the intervening tissue is absorbed by a series of detectors revolving around the subject’s head. The data from the radiation detectors allow a calculation of the density of tissue in a particular slice of brain. A computer then reconstructs a two-dimensional cross-sectional picture of the brain observed by the camera. Several cross sections may be printed corresponding to different planes through the head. Contrast substances are sometimes injected in the patient to increase the density of damaged tissue. This enhancement technique allows clearer visualization and more accurate diagnosis. Spiral CT, in which the x-ray tube rotates continuously around the patients while the table moves, was developed in the early 1990s along with a computational method that would eliminate the motion artifact this introduced. Spiral CT allowed much more flexible and rapid study. Because CT studies are quicker and less expensive to perform than MRI, the technology is more widely available in general medical settings. CT is frequently used in the emergency department and other acute settings. It is more sensitive to skull fracture and can detect the presence of foreign bodies, such as glass and metal, that are radiopaque. It can reveal the presence of blood in the linings and parenchyma of the brain and, therefore, is the first study of choice in a vascular event of unknown etiology so that hemorrhage could be ruled out first. Because MRI uses a magnetic field, CT is the study that would be chosen for patients with pacemakers, defibrillators, and other such devices.
MRI, probably the most widely used diagnostic imaging technique in neurology, generates cross-sectional images by using radio waves, a strong magnetic field, and gradient coils to detect the distribution of water molecules in living tissue (Figs. 3-22 and 3-23). The technique allows accurate assessment of brain tissue densities, and an excellent pictorial image can be generated by the computer. The explanation of the technology is beyond the scope of this text but it is helpful to know what “tissue weighting,” which you will see as T1, T2, or proton density (PDW), does for an image. These parameters determine the contrast between tissues, allowing certain tissues to be more easily seen in an image. A T1-weighted image highlights tissues such as fat (like white matter) and proteins, whereas CSF will appear dark. A T2-weighted image makes CSF lighter and fat appears darker, which may make this weighting more useful for identifying pathologies. If the contrast between these T1 tissues and CSF is not what is of interest, a PDW will be used, which will highlight differences in the proton densities of the two tissues with the denser tissue emphasized in the image and gray matter appearing brighter than white matter.12
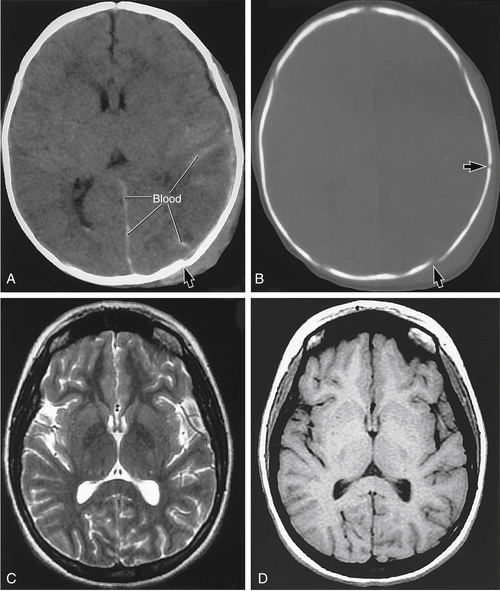
FIGURE 3-21 Computed tomography (CT) scans of a 2-month-old infant with shaken baby syndrome (A and B) and magnetic resonance (MR) images of a normal 20-year-old woman (C and D). On the CT study, note that the brain detail is less than on the MR images but that the presence of blood (A, in the interhemispheric fissure between the hemisphere and the brain substance) is obvious. In the same patient, the bone window (B) clearly illustrates the outline of the skull but also clearly shows skull fractures (arrows in A and B). In this infant, the ventricles on the left are largely compressed, and the gyri have largely disappeared because of pressure from bleeding into the hemisphere. The pressure results in the effacement of the sulci and gyri on the left side. In the T2-weighted image (C), cerebrospinal fluid is white, internal brain structures are seen in excellent detail, and vessels are obvious. In the T1-weighted image (D), cerebrospinal fluid is dark and internal structures of the brain are somewhat less obvious. (Reprinted from Haines, D. [2006]. Fundamental neuroscience [3rd ed.]. Philadelphia: Churchill Livingstone.)

FIGURE 3-22 The relation of imaging planes to the brain. The diagram shows the usual orientation of a patient in an MRI machine and the planes of the four scans (T1-weighted images) that are shown. A and B, Coronal scans; C and D, axial scans. (Reprinted from Haines, D. [2006]. Fundamental neuroscience [3rd ed.]. Philadelphia: Churchill Livingstone.)

FIGURE 3-23 MRI of the brain in the median sagittal plane (A) and in the sagittal plane but off the midline (B). The frontal lobe is to the left, and the occipital lobe is to the right. (Reprinted from Haines, D. [2006]. Fundamental neuroscience [3rd ed.]. Philadelphia: Churchill Livingstone.)
Generally, MRI is more sensitive to abnormalities than CT. However, it is significantly more expensive to generate the image. Damasio and Damasio7 pointed out that the analysis of CT and MRI images is sometimes difficult in that the number of brain slices provided for viewing may vary from institution to institution and from patient to patient. The number of slices may even vary in the same patient as scanning devices are improved over time.
Diffusion-weighted imaging (DWI) is another way of enhancing the usefulness of MRI in some cases. DWI allows the imaging of molecular motion or diffusion of water protons within tissue. DWI which highlights reduced diffusion has been found to be very useful to rapidly identify acute cerebral ischemia or loss of blood flow to an area. MRI study using DWI can be positive for reduced diffusion in these cases as early as 30 minutes after onset.12 Figure 3-24 shows the usefulness of DWI in identifying axonal shearing which has been typically difficult to verify in traumatic injury.
Another advance in imaging to better see the white and gray matter in the brain is through voxel-based morphometry (VBM). This is primarily a research tool in which statistical methods originally applied to positron emission tomography (PET) scans are used on MRI scans. A voxel is the basic unit of computed tomography measurement, and the term comes from a combination of the words volume and pixel. It is essentially an imaged “slab” of tissue that has been divided into small volume elements called voxels with each voxel having an x, y, and z dimension on which statistical and other manipulations can be studied.1 VBM has been used to study changes in gray matter volume in normal development, aging, and disease.16 It also has been employed in anatomic studies of speech and language disorders.15 Comparing the images of MRI, functional MRI (fMRI), and VBM has made it possible for advances in VBM for future research and clinical assessment.
Dynamic or Functional Brain Imaging
CT and MRI are unable to detect certain forms of cellular and subcellular brain pathology directly. Dynamic neuroimaging procedures that use emission tomography (PET and single-photon emission computed tomography [SPECT]) are helpful in cases in which imaging of brain structures alone is not decisive. For instance, in some cases of early dementia, CT and MRI scans appear normal, but language and neuropsychological testing reveals serious cerebral dysfunction.
Functional MRI (fMRI) uses an MRI scanner to measure regional blood flow, cerebral blood volume, and the change in blood oxygenation. fMRI study results in inferences about the location of brain activity. The most common fMRI study is a BOLD study, which stands for blood oxygenation level–dependent contrast. This study tracks the hemodynamic response to neural activity. This means that changes in blood flow and blood oxygenation are traced. fMRI has begun to be a popular technology for research by SLPs in collaboration with neurology and radiology. Brain activity in such areas as acquired language disorders, autism, and other communication disorders have been investigated with BOLD MRI.6,8,10
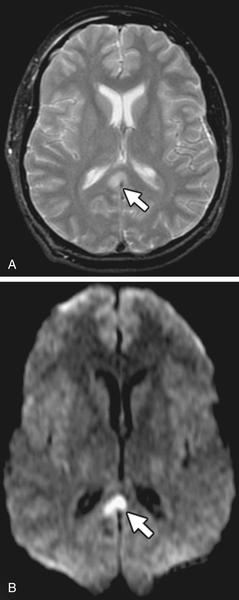
FIGURE 3-24 Axonal shearing injury.
A, Axial T2-weighted image shows a subtle area of reduced diffusion within the right paramedian corpus callosum (arrow). B, Axial diffusion-weighted image more clearly demonstrates abnormal reduced diffusion within the callosal lesion (arrow). (From Nadich, T.P., Krayenbuhl, N., Kollias, S., et al. [2013]. White Matter. In T.P. Nadich, M. Castillo, S. Cha, & Smirniotopoulos (Eds.), Imaging of the brain, Philadelphia: Elsevier/Saunders.
Positron emission tomograpy (PET) is a visual technique in which the subject is given a radioactively labeled form of glucose, which is metabolized by the brain. The radioactivity is later recorded by a special detector. Unlike CT and MRI scans, a PET scan measures metabolic activity in different brain areas. More active areas metabolize more glucose, focusing more radioactivity in these areas. Thus regional three-dimensional quantification of glucose and oxygen metabolism or blood flow in the human brain is achieved. This technique is advantageous in that glucose metabolism is a more direct measure of the function of neural tissue than is cerebral blood flow, particularly in patients whose regulatory vascular mechanisms are affected by cerebral injury or disease. PET scan studies have been used to research higher mental functions during different cognitive and language tasks and appear to offer an excellent tool for the study of language in the human brain. This technology is expensive because it requires a cyclotron or atomic accelerator. Because of the color limitations of this text, an example PET scan cannot be displayed, but many can be found through a search of web images. One interesting example is found from the Berkeley Labs site (www.lbl.gov).
Single-photon emission computed tomography (SPECT) uses the mechanism of CT scan reconstruction, but instead of detecting x-rays, the instrument detects single photons emitted from an external tracer. Radioactive compounds that emit gamma rays are injected into the subject. As these biochemicals reach the brain, emissions are picked up that are converted into patterns of metabolism or blood flow in three-dimensional cross sections of the brain. SPECT has somewhat better temporal resolution than PET but its spatial resolution is less than PET or MRI and it is more invasive. The equipment is less expensive because a cyclotron is not required and it therefore may be used at small medical centers.
Measures of Neural Connectivity
Diffusion tensor imaging (DTI) is the preferred method used to study white matter as is VBM for gray matter. DTI is a recently developed technique that is an extended version of DWI. It can measure neuronal activity and, in particular, the white matter neuronal cell networks (including the directionality of the actual tracts). Diffusion of water can be described as isotropic which means that diffusion occurs in the same degree in all directions or anisotropic, meaning that diffusion pattern is dependent on the direction being viewed. Because diffusion of water molecules occurs in the brain much more easily along lines that are parallel to axon bundles rather than perpendicular to them, the diffusion is anisotropic. Therefore this diffusion in the brain tissue has to be assessed in multiple directions with the resulting images then combined into an isotropic map, imaging the integrity of white matter tracts. DTI considers three factors in constructing the final image. One of these factors is the fractional anisotropy (FA); this results in essentially a gray scale representing the degree of variation of fiber direction at any point. The other two factors considered are (1) the mean diffusivity (MD), or overall displacement of the molecules in that voxel, and (2) the direction of greatest water mobility or the principal eigenvector (PE). FA values have been established for the normal brain, which will allow differentiation between gray and white matter. Intensity (FA) and directionality (PE) data can be combined into spectacular images called DTI color maps. In these maps, red signifies diffusion in a left-right direction, blue predominantly in a superior-inferior direction, and green represents diffusion in the anterior-posterior plane. Diffusion tensor tractography uses the data from DTI to construct a 3-D image of axon bundles. The Human Connectome Project is a large, National Institutes of Health–funded research project to map the human brain. Breathtaking images made from DTI studies can be found at the project’s website gallery.18
A promising recent functional imaging technique that has been put to use both clinically and in research protocols is that of optical imaging, which uses the technology called near infrared spectroscopy (NIRS)14 to assess response to brain activation. It is similar to fMRI in its objective, but unlike fMRI, which measures changes in deoxygenated hemoglobin, NIRS can monitor changes in deoxygenated and oxygenated hemoglobin plus changes in localized blood volume. Optical imaging is based on measuring the light absorption of different tissue properties through use of various infrared sources and detectors. It is noninvasive and can be done during motor, sensory, and cognitive tasks. Zeller et al.17 used optical imaging in a visual-spatial task with patients with Alzheimer’s disease and found decreased activation in the parietal lobe compared with control subjects.
Measures of Timing of Neural Activity
It is well known that neurons emit small currents of electrical activity. In 1924 Hans Berger developed the first method to capture this activity. Other methods evolved from his early invention of the electroencephalogram (EEG).
Electroencephalography
An older diagnostic technique, electroencephalography (EEG), has been used for decades to diagnose lesions of the brain and help clarify their nature. EEG measures voltage fluctuations resulting from ionic currents between neurons in the brain. It measures these changes with the use of noninvasive scalp electrodes. EEG is used to study seizure activity and diagnose epilepsy and its subtypes. The temporal resolution of EEG is excellent, but the limitation is that it is not correlated with any specific brain activity.
Another current use of EEG technology is the prediction of outcomes from patients with coma-related injuries. According to Boccagni and colleagues,3 standard EEG has been proven to be a predictor of recovery of cognitive functioning in patients after coma caused by cerebral anoxia.
Evoked Potentials
Measurement of evoked potentials is a derivative of EEG. Rather than measuring spontaneous potentials detected from nervous system activity, this study reveals specific electrical potentials evoked and time-locked to the presentation of a known stimulus. The stimuli used are usually sensory (visual, auditory, somatosensory). If a stimulus is repeated enough times and each repetition produces a circumscribed electrical response, computer averaging can establish the onset of a response and its termination. Measurement called event-related potentials (ERPs) involves using an averaging computer to separate the electrical activity surrounding more complex processing events from the ongoing electrical background activity of the brain. It may be a response to internal or external stimulation of the nervous system. ERPs are often used in cognitive research. Although this technique has been used widely by some speech and psychological researchers, it is not without problems.4 Both its validity and reliability can be questioned. Whether an electrical potential that occurs after a stimulus is of cerebral origin or is brought about by a motor act is not always certain. When a language stimulus evokes a cerebral potential, brain activity is not always present, and the absence of an electrical potential to a language stimulus does not mean no electrical activity occurred. Many electrical currents simply do not reach the surface electrodes; some are too small and erratic. In addition, the waveforms derived from stimulation are highly complex; therefore determining which section of the waveform that was generated in response to the language stimulus has psychological meaning is sometimes difficult.
Despite these limitations, event-related potential has the capacity to measure events in the brain millisecond by millisecond. A research paradigm frequently used in language studies yields a readiness potential. The subject is asked to repeat a word or phrase or speak freely with pauses of 3 or 4 seconds between portions of an utterance. The continuous EEG recording that precedes the onset of speech is analyzed by averaging waveforms across several utterances to discover what is termed the readiness potential.
Audiology has made good use of evoked potentials with a study known as an auditory brainstem response (ABR) or brainstem auditory evoked response (BAER). For this study, electrodes are placed on the scalp and electrical activity is recorded in response to click stimuli. The recordings from an ABR are displayed in vertical waves and waves I through V can be used to evaluate the integrity of the brainstem’s response following the response from the cranial nerve to the upper brainstem.
Magnetoencephalography
Those electrical currents inside the head also produce magnetic field oscillations around active neurons. The strength and orientation of these magnetic fields can be detected from above the scalp by use of a magnetometer (MEG), which marks spontaneous or event-related activity (event-related magnetic fields or ERFs). The MEG locates the sources of the neural activity and the locations are superimposed on anatomic images such as MRI. MEG has been used to map the brain prior to surgical intervention, such as for epilepsy, and can be a useful research tool for mapping language.9
Vascular Imaging
Noncontrast CT, contrast-enhanced CT, and MRI are the most frequently used studies at this time to identify the presence and extent of vascular infarction or disease. For the acute stroke, the most important task of the physician is to exclude hemorrhage. Hemorrhage would be a contraindication to any procedure introducing blood thinners or to the use of thrombolytic drugs or “clot busters” to perfuse an area of the brain deprived of blood flow by the presence of a thrombus or an embolus. The noncontrast CT is the most frequently used study for this purpose if the patient is seen early in the progression of the stroke. If it is available and there are no contraindications MRI is somewhat superior to CT for the acute-stage studies. Contrast-enhanced CT presents more risk to the patient but may better identify lesions.
It may be important for the physician to know the patency of the vascular system of a patient with suspected neurologic disease or injury. Once it is determined by a functional study that there is reduced blood flow to an area, it will be necessary to determine where the vascular perfusion has been interrupted or which vasculature is at risk of future occlusion, hemorrhage, or spasm.
Transcranial Doppler
A transcranial Doppler study is a noninvasive procedure done by passing inaudible low frequency sound waves to the base of the brain. This is done by placing the probes over areas where thin bones are located: above the cheekbone, beneath the jaw, at the orbit of the eyes, or at the back of the head. It depends on where the best signal is found. The Doppler analyzes reflected sound waves coming from the major blood vessels. It can reveal disruption of blood flow and identify the affected vessels. Doppler studies of the carotid arteries are often done after a transient ischemic attack (TIA) to assess the patency of the major vessel from the heart to the brain.
Cerebral Angiography and Magnetic Resonance Angiography
Conventional cerebral angiography is an invasive procedure because it involves the injection of a contrast media into the carotid artery. Once this dye is injected, a radiographic study is done while it works its way through the circulation paths of the brain, providing a radiographic picture of the vascular distribution to the brain. The risks of this procedure, especially in the acute stages of possible vascular compromise, are obvious. A plaque on the wall on the artery could be dislodged by the catheter carrying the dye resulting in an embolism, causing further deprivation of blood to the brain. In addition, allergic reaction to the contrast is possible. Therefore conventional angiography is used now only in selected patients and has been replaced, especially in the case of cerebrovascular accident, with magnetic resonance angiography (MRA).2
Summary of Neuroimaging
As pointed out in the beginning of this section, it is clinically useful for the practicing SLP to have knowledge of different types of neuroimaging studies that may be used by the physicians in the care of their patient. This familiarity will also be quite useful in reading research in speech-language pathology and audiology. Increasing collaboration is seen between SLPs and medical researchers using the various neuroimaging methods to understand normal brain function underlying communication and to identify brain differences related to acquired or developmental problems with communication. In addition to the techniques discussed in this section, additional neuroimaging methods are briefly described in Box 3-2.
Overview of the Human Communication Nervous System
The organization of the human communication nervous system is fundamental to understand and recall. In the first three chapters of this text, you have been introduced to the history of the interaction between neurology and speech-language pathology, to structural anatomy and functionality of the main components of the central and peripheral nervous systems, and to some typical methods of imaging those structures in static images and in functional studies. From this point on, we further explore various aspects of this complex nervous system of ours. We will look at how neurons communicate with each other, briefly explore sensory systems intimately involved in communication (touch and vision), and then, in more depth, explore the structures and their function that are primarily responsible for our miraculous ability to hear, speak, and process information in order to communicate with each other efficiently and effectively. The outline in Appendix 3-1 is a summary figure of the levels and an outline of the most important structures of the CNS. You may wish to quiz yourself at this point and, if necessary, review the preliminary information from this and the previous chapter on these structures. We go into more depth about them and the disorders associated with breakdown in their various systems in later chapters, and at times you will be reminded to review some of the information in these chapters.
Appendix 3-1
A vast amount of information is provided in Chapters 2 and 3. Figure 3-25 is a summary drawing of the levels of the CNS to organize the information in Chapter 2. Referring to this figure while reading subsequent chapters will be helpful. An outline of the most important structures discussed in these chapters is also provided for review and organization. For each item, ask the following questions:
• What is it?
• Where is it?
• What does it do?
When appropriate, attempt to label drawings of some of the various structures for which illustrations were used. The effort put into doing this will be rewarded in understanding subsequent chapters.
I. The human nervous system
A. Central nervous system
1. Brain
B. Cerebral hemispheres
1. Four lobes
2. Fissures
3. Sulci
4. Gyri
5. Association cortex
6. Connecting fibers
C. Basal ganglia
1. Corpus striatum
(a) Caudate nucleus
(b) Lentiform nucleus: putamen, globus pallidus
2. Claustrum
E. Cerebellum
F. Brainstem
1. Medulla oblongata
(a) Pyramids
(b) Olives
(c) Peduncles
2. Pons
3. Mesencephalon
(a) Tectum
(b) Colliculi
4. Spinal cord
II. Spinal nerves
III. Peripheral nerves
IV. Five regions
A. Meninges
V. Dura mater
VI. Arachnoid mater
VII. Pia mater
A. Ventricles
VIII. Choroid plexus
IX. CSF
A. Blood supply
X. Internal carotid artery and its branch
XI. Vertebral artery and its branches
XII. Circle of Willis
A. PNS
1. Spinal peripheral nerves
XIII. Anterior horn cell
XIV. Efferent fibers
XV. Afferent fibers
Cranial nerves
XVI. Twelve pairs
A. Autonomic nervous system
1. Parasympathetic division
2. Sympathetic division
3. Enteric division (deglutition and peristalsis)
XVII. Clinical principles of neurologic organization
A. Contralateral motor control
B. Ipsilateral motor control
C. Bilateral speech motor control
D. Unilateral language mechanisms
E. Scheme of cortical organization




