The Child With Fluid and Electrolyte Imbalance
Mary A. Mondozzi, Rose Ann Urdiales Baker, Marilyn J. Hockenberry

Distribution of Body Fluids
The distribution of body fluids, or total body water (TBW), involves the presence of intracellular fluid (ICF) and extracellular fluid (ECF). Water is the major constituent of body tissues, and the TBW in an individual ranges from 45% (in late adolescence) to 75% (in term newborn) of total body weight.
The ICF refers to the fluid contained within the cells, whereas the ECF is the fluid outside the cells. The ECF is further broken down into several components: intravascular (contained within the blood vessels), interstitial (surrounding the cell; the location of most ECF), and transcellular (contained within specialized body cavities such as cerebrospinal, synovial, and pleural fluid). In the newborn about 50% of the body fluid is contained within the ECF, whereas 30% of the toddler's body fluid is contained within the ECF.
Body water is important in body function not only because of its abundance but also because it is the medium in which body solutes are dissolved and all metabolic reactions take place. Because even small alterations in fluid composition affect these metabolic processes, precise regulation of the volume and composition of the fluid is essential. In healthy individuals, body water remains singularly constant, but marked alterations in either its volume or distribution, which occur in many disease states, can produce severely damaging physiologic consequences.
Water Balance
Under normal conditions the amount of water ingested closely approximates the amount of urine excreted in a 24-hour period, and the water in food and from oxidation approximates the amount lost in feces and through evaporation. In this way, the body maintains equilibrium.
Mechanisms of Fluid Movement
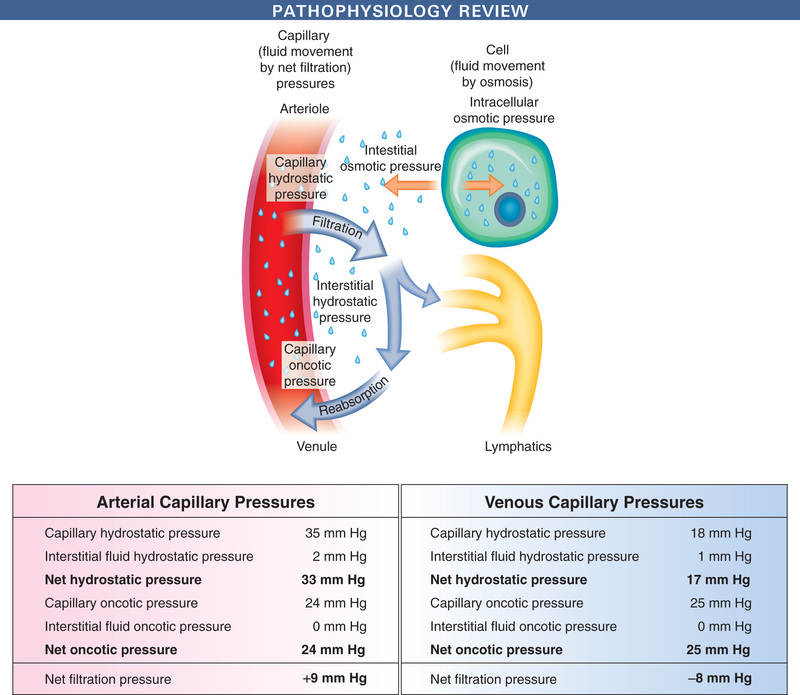
Water is retained in the body in a relatively constant amount and, with few exceptions, is freely exchangeable among all body fluid compartments. The proximity of the extravascular compartment to the cells allows for continuous change in volume and distribution of fluids, largely determined by solutes (especially sodium) and physical forces (Fig. 23.1). Transport mechanisms are the basis for all activity within the cells, and because the cells have limited ability to store materials, movement in and out of cells must be rapid. Internal control mechanisms are responsible for distribution and maintenance of fluid balance (Box 23.1).
Maintaining Water Balance
Maintenance water requirement is the volume of water needed to replace obligatory fluid loss such as that from insensible water loss (through the skin and respiratory tract), evaporative water loss, and losses through urine and stool formation. The amount and type of these losses may be altered by disease states such as fever (with increased sweating), diarrhea, gastric suction, and pooling of body fluids in a body space (often referred to as third spacing).
Nurses should be alert for altered fluid requirements in various conditions:
Basal maintenance calculations for required body water are based on the body's requirements for water in a normometabolic state, at rest; estimated fluid requirements are then increased or decreased from these parameters based on increased or decreased water losses, such as with elevated body temperature or congestive heart failure. Daily maintenance fluid requirements are listed in Table 23.1.
TABLE 23.1
| Body Weight | Amount of Fluid Per Day |
|---|---|
| 1-10 kg | 100 ml/kg |
| 11-20 kg | 1000 ml plus 50 ml/kg for each kg >10 kg |
| >20 kg | 1500 ml plus 20 ml/kg for each kg >20 kg |
*Not appropriate for neonatal use.
Maintenance fluids contain both water and electrolytes and can be estimated from the child's age, body weight, degree of activity, and body temperature. Basal metabolic rate (BMR) is derived from standard tables and adjusted for the child's activity, temperature, and disease state. For example, for afebrile patients at rest, the maintenance water requirement is approximately 100 ml for each 100 kcal expended. Children with fluid losses or other alterations require adjustment of these basic needs to accommodate abnormal losses of both water and electrolytes as a result of a disease state. For example, insensible losses increase when basal expenditure increases by fever or hypermetabolic states. Hypometabolic states, such as hypothyroidism and hypothermia, decrease the BMR.
Changes in Fluid Volume Related to Growth
The percentage of TBW varies among individuals, and in adults and older children, it is related primarily to the amount of body fat. Consequently, females, who have more body fat than males, and obese persons tend to have less water content in relation to weight.
The fetus is composed primarily of water, with little tissue substance. As the organism grows and develops, a progressive decrease occurs in TBW, with the fastest rate of decline taking place during fetal life. The changes in water content and distribution that occur with age reflect the changes that take place in the relative amounts of bone, muscle, and fat making up the body. At maturity the percentage of TBW is somewhat higher in the male than in the female and is probably a result of the differences in body composition, particularly fat and muscle content (Fig. 23.2).
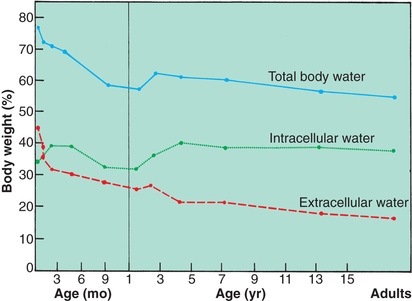
Another important aspect of growth change as it corresponds to water distribution is related to the ICF and ECF compartments. In the fetus and prematurely born infant, the largest proportion of body water is contained in the ECF compartment. As growth and development proceed, the proportion within the ECF compartment decreases as the ICF and cell solids increase. The ECF diminishes rapidly from approximately 40% of body weight at birth to less than 30% at 1 year of age. The different effects on males and females become apparent at puberty.
Water Balance in Infants
Because of several characteristics, infants and young children have a greater need for water and are more vulnerable to alterations in fluid and electrolyte balance. Compared with older children and adults, they have a greater fluid intake and output relative to size. Water and electrolyte disturbances occur more frequently and more rapidly, and children adjust less promptly to these alterations.
The fluid compartments in the infant vary significantly from those in the adult, primarily because of an expanded extracellular compartment. The ECF compartment constitutes more than half of the TBW at birth and has a greater relative content of extracellular sodium and chloride. The infant loses a considerable amount of fluid in the first few days after birth and still maintains a larger amount of ECF than the adult until about 2 to 3 years of age. This contributes to greater and more rapid water loss during this age period.
Fluid losses create compartment deficits that reflect the duration of dehydration. In general, approximately 60% of fluid is lost from the ECF, and the remaining 40% comes from the ICF. The amount of fluid lost from the ECF increases with acute illness and decreases with chronic loss.
Fluid losses may be divided into insensible, urinary, and fecal losses and vary with the patient's age. Approximately two-thirds of insensible water losses occurs through the skin, and the remaining one-third is lost through the respiratory tract. Environmental heat and humidity, skin integrity, body temperature, and respiratory rate all influence insensible fluid loss. Infants and children have a much greater tendency to become highly febrile than do adults. Fever increases insensible water loss by approximately 7 ml/kg/24 hr for each 1°F rise in temperature above 37.2°C (99°F). Fever and increased surface area relative to volume both contribute to greater insensible fluid losses in young patients.
Body Surface Area
The infant's relatively greater body surface area (BSA) allows larger quantities of fluid to be lost through the skin. It is estimated that the BSA of the premature neonate is five times more, and that of the newborn is two or three times more, than that of the older child or adult. The proportionately longer gastrointestinal tract in infancy is also a source of relatively greater fluid loss, especially from diarrhea.
Metabolic Rate
The rate of metabolism in infancy is significantly higher than in adulthood because of the larger BSA in relation to the mass of active tissue. Consequently, infants have a greater production of metabolic wastes that the kidneys must excrete. Any condition that increases metabolism causes greater heat production, with its concomitant insensible fluid loss and an increased need for water for excretion. The BMR in infants and children is higher to support cellular and tissue growth.
Kidney Function
The infant's kidneys are functionally immature at birth and are therefore inefficient in excreting waste products of metabolism. Of particular importance for fluid balance is the inability of the infant's kidneys to concentrate or dilute urine, to conserve or excrete sodium, or to acidify urine. Therefore the infant is less able to handle large quantities of solute-free water than is the older child and is more likely to become dehydrated when given concentrated formulas or overhydrated when given excessive free water or dilute formula.
Fluid Requirements
As a result of these characteristics, infants ingest and excrete a greater amount of fluid per kilogram of body weight than do older children. Because electrolytes are excreted with water and the infant has limited ability for conservation, maintenance requirements include both water and electrolytes. The daily exchange of ECF in the infant is much greater than that of older children, which leaves the infant little fluid volume reserve in dehydrated states. Fluid requirements depend on hydration status, size, environmental factors, and underlying disease.
Disturbances of Fluid and Electrolyte Balance
Disturbances of fluids and their solute concentration are closely interrelated. Alterations in fluid volume affect the electrolyte component, and changes in electrolyte concentration influence fluid movement. Because intracellular water and electrolytes move to and from the ECF compartment, any imbalance in the ICF is reflected by an imbalance in the ECF. Disturbances in the ECF involve either an excess or a deficit of fluid or electrolytes. Of these, fluid loss occurs more frequently.
Sodium is the chief solute in ECF and the primary determinant of ECF volume. It is considered a unique electrolyte in that water balance determines sodium concentration; when water is lost and sodium concentration becomes elevated, compensatory mechanisms in the kidney stop ADH secretion so water is retained. The thirst mechanism (not fully functional in infants) is also stimulated so water is replaced, thus increasing the total body water content and returning sodium to a normal level (Greenbaum, 2015). Sodium depletion in diarrhea occurs in two ways: out of the body in stool and into the ICF compartment to replace potassium to maintain electrical equilibrium. Potassium is found primarily inside the cell (intracellular), but small amounts are also found in ECF.
Depletion of ECF, usually caused by gastroenteritis, is one of the most common problems encountered in infants and children. (See Chapter 25.) Until modern techniques for fluid replacement were perfected, gastroenteritis was one of the chief causes of infant mortality. Fluid and electrolyte problems related to specific diseases and their management are discussed throughout the book where appropriate. The major fluid disturbances, their usual causes, and clinical manifestations are listed in Table 23.2; the most common fluid disturbances, dehydration and edema, are elaborated further in the following sections. Problems of fluid and electrolyte disturbance always involve both water and electrolytes; therefore replacement includes administration of both, calculated on the basis of ongoing processes and laboratory serum electrolyte values.
TABLE 23.2


ADH, Antidiuretic hormone; ECG, electrocardiogram; IV, intravenous.
In conditions that involve alterations in the amount and composition of body fluid compartments, nurses consider many factors when planning management (Box 23.2). The following discussion is concerned with the general concepts of two common fluid volume disturbances, dehydration and edema, which are features of a variety of conditions. Specific disorders are discussed in Chapter 25 and elsewhere in the book where appropriate.
Dehydration
Dehydration is a common body fluid disturbance encountered in the nursing care of infants and young children; it occurs whenever the total output of fluid exceeds the total intake, regardless of the underlying cause. Dehydration is also commonly referred to as volume depletion. Although dehydration can result from lack of oral intake (especially in elevated environmental temperatures), more often it is a result of abnormal losses, such as those that occur in vomiting or diarrhea, when oral intake only partially compensates for the abnormal losses. Other significant causes of dehydration are diabetic ketoacidosis and extensive burns.
In early dehydration (during the first 2 days), fluid loss is derived from both the ECF and the ICF because the increased osmolality of the diminished ECF volume causes fluid from the ICF compartment to move into the ECF compartment. As dehydration becomes chronic, the cellular losses become greater.
Types of Dehydration
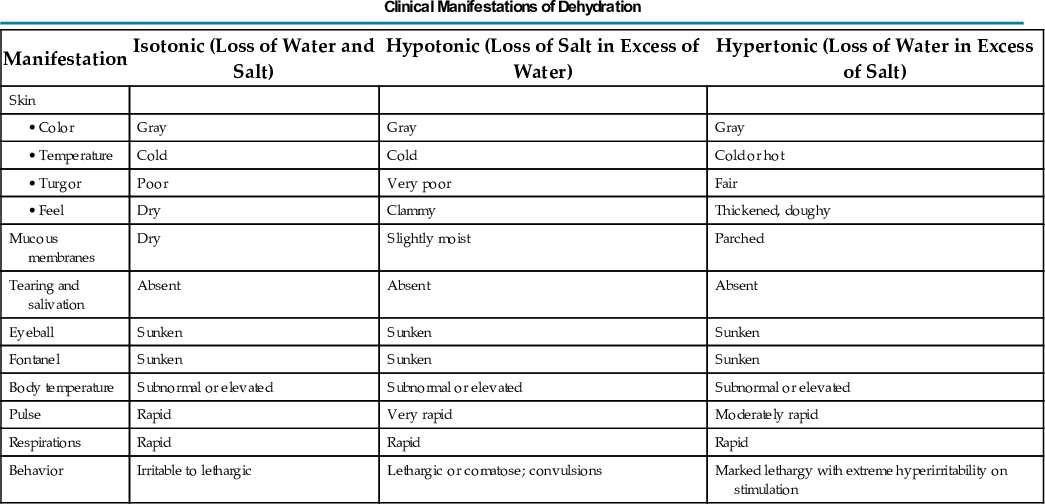
Because sodium is the primary osmotic force that controls fluid movement between the major fluid compartments, dehydration is often described according to plasma sodium concentrations (e.g., isonatremic, hyponatremic, or hypernatremic). Other osmotic forces, however, such as glucose in diabetic ketoacidosis and protein in nephrotic syndrome, may also play a dominant role. Consequently, dehydration is conventionally classified as isotonic, hypotonic, or hypertonic.
Isotonic (isosmotic or isonatremic) dehydration occurs in conditions in which electrolyte and water deficits are present in approximately balanced proportions. This is the primary form of dehydration occurring in children. The observable fluid losses are not necessarily isotonic, but losses from other avenues make adjustments so that the sum of all losses, or the net loss, is isotonic. Because no osmotic force is present to cause a redistribution of water between the ICF and ECF, the major loss is sustained from the ECF compartment. This significantly reduces the plasma volume and thus the circulating blood volume, with its effect on the skin, muscles, and kidneys. Shock is the greatest threat to life in isotonic dehydration, and the child with isotonic dehydration displays symptoms characteristic of hypovolemic shock. Plasma sodium remains within normal limits, between 130 and 150 mEq/L (Friedman, Beck DeGroot, et al., 2015; Huether, 2014).
Hypotonic (hyposmotic or hyponatremic) dehydration occurs when the electrolyte deficit exceeds the water deficit. Because ICF is more concentrated than ECF in hypotonic dehydration, water transfers from the ECF to the ICF to establish osmotic equilibrium. This movement further increases the ECF volume loss, and shock is a frequent result. Because there is a greater proportional loss of ECF in hypotonic dehydration, the physical signs tend to be more severe with smaller fluid losses than in isotonic or hypertonic dehydration. Plasma sodium concentrations are typically less than 130 mEq/L (Huether, 2014).
Hypertonic (hyperosmotic or hypernatremic) dehydration results from water loss in excess of electrolyte loss and is usually caused by a proportionately larger loss of water or a larger intake of electrolytes. This type of dehydration is the most dangerous and requires much more specific fluid therapy. This sometimes occurs in infants with diarrhea who are given fluids by mouth that contain large amounts of solute or in children receiving high-protein nasogastric tube feedings that place an excessive solute load on the kidneys. In hypertonic dehydration, fluid shifts from the lesser concentration of the ICF to the ECF. Plasma sodium concentration is greater than 150 mEq/L (Huether, 2014).
Because the ECF volume is proportionately larger, hypertonic dehydration consists of a greater degree of water loss for the same intensity of physical signs. Shock is less apparent in hypotonic dehydration. However, neurologic disturbances, such as seizures, are more likely to occur. Cerebral changes are serious and may result in permanent damage. These include disturbance of consciousness, poor ability to focus attention, lethargy, increased muscle tone with hyperreflexia, and hyperirritability to stimuli (e.g., tactile, auditory, bright lights).
Degree of Dehydration
A determination of the type and degree of dehydration is necessary to develop an effective plan of therapy. The degree of dehydration has been described as a percentage of body weight dehydrated: mild—less than 3% in older children or less than 5% in infants; moderate—5% to 10% in infants and 3% to 6% in older children; and severe—more than 10% in infants and more than 6% in older children (Carson, Mudd, & Madati, 2016; Greenbaum, 2015). Water constitutes only 60% to 70% of the infant's weight. However, adipose tissue contains little water and is highly variable in individual infants and children. A more accurate means of describing dehydration is to reflect acute fluid loss (time frame of ≥48 hours) in milliliters per kilogram of body weight. For example, a loss of 50 ml/kg is considered to be a mild fluid loss, whereas a loss of 100 ml/kg produces severe dehydration.
Weight is the most important determinant of the percent of total body fluid loss in infants and younger children. However, often the preillness weight is unknown. Other predictors of fluid loss include a changing level of consciousness (irritability to lethargy), altered response to stimuli, decreased skin elasticity and turgor, prolonged capillary refill (>2 seconds), increased heart rate, and sunken eyes and fontanels.
Clinical signs provide clues to the extent of dehydration (Table 23.3). The earliest detectable sign is usually tachycardia, followed by dry skin and mucous membranes, sunken fontanels, signs of circulatory failure (coolness and mottling of extremities), loss of skin elasticity, and prolonged capillary filling time (see Table 23.4 for clinical manifestations of dehydration and Fig. 23.3 for signs of dehydration). There is evidence that the clinical signs of abnormal capillary refill, abnormal skin turgor, and abnormal respiratory pattern are the most useful in predicting dehydration of 5% or more in children (Carson, Mudd, & Madati, 2017).
TABLE 23.3
| LEVEL OF DEHYDRATION | |||
|---|---|---|---|
| Clinical Signs | Mild | Moderate | Severe |
| Weight loss—infants | 3%-5% | 6%-9% | ≤10% |
| Weight loss—children | 3%-4% | 6%-8% | 10% |
| Pulse | Normal | Slightly increased | Very increased |
| Respiratory rate | Normal | Slight tachypnea (rapid) | Hyperpnea (deep and rapid) |
| Blood pressure | Normal | Normal to orthostatic (>10 mm Hg change) | Orthostatic to shock |
| Behavior | Normal | Irritable, more thirsty | Hyperirritable to lethargic |
| Thirst | Slight | Moderate | Intense |
| Mucous membranes* | Normal (moist) | Dry | Parched |
| Tears | Present | Decreased | Absent, sunken eyes |
| Anterior fontanel | Normal | Normal to sunken | Sunken |
| External jugular vein | Visible when supine | Not visible except with supraclavicular pressure | Not visible even with supraclavicular pressure |
| Skin* | Capillary refill >2 sec | Slowed capillary refill (2-4 sec [decreased turgor]) | Very delayed capillary refill (>4 sec) and tenting; skin cool, acrocyanotic or mottled |
| Urine | Decreased | Oliguria | Oliguria or anuria |
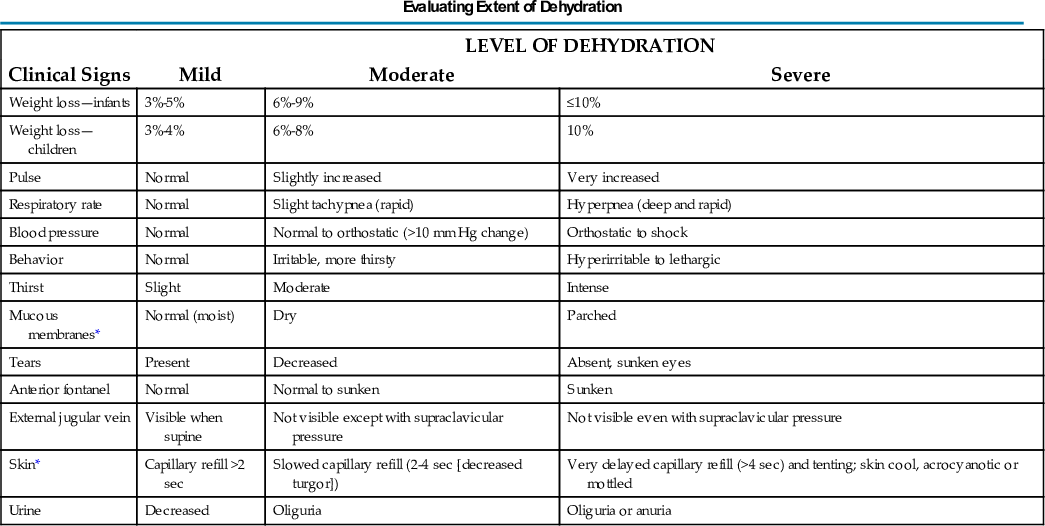
*These signs are less prominent in patients who have hypernatremia.
Data from Jospe, N., & Forbes, G. (1996). Fluids and electrolytes—clinical aspects. Pediatrics in Review, 17(11), 395-403; and Steiner, M. J., DeWalt, D. A., & Byerly, J. S. (2004). Is this child dehydrated? Journal of the American Medical Association, 291(22), 2746-2754.
TABLE 23.4
Compensatory mechanisms attempt to maintain fluid volume by adjusting to these losses. Interstitial fluid moves into the vascular compartment to maintain the blood volume in response to hemoconcentration and hypovolemia, and vasoconstriction of peripheral arterioles helps maintain pumping pressure. When fluid losses exceed the body's ability to sustain blood volume and blood pressure, circulation is seriously compromised and the blood pressure falls. This results in tissue hypoxia with accumulation of lactic acid, pyruvate, and other acid metabolites, which contribute to the development of metabolic acidosis.
Renal compensation is impaired by reduced blood flow through the kidneys, and little urine is formed. Increased serum osmolality stimulates the secretion of antidiuretic hormone (ADH) to conserve fluid and initiates the renin-angiotensin mechanisms in the kidney, causing further vasoconstriction. Aldosterone is released to promote sodium retention and conserve water in the kidneys. If dehydration increases in severity, urine formation is greatly diminished and metabolites and hydrogen ions that are normally excreted by this route are retained.
Shock, a common manifestation of severe depletion of ECF volume, is preceded by tachycardia and signs of poor perfusion and tissue oxygenation (by pulse oximeter readings). Peripheral circulation is poor as a result of reduced blood volume; therefore the skin is cool and mottled with decreased capillary filling. Impaired kidney circulation often leads to oliguria and azotemia. Although low blood pressure may accompany other symptoms of shock, in infants and young children it is usually a late sign and may herald the onset of cardiovascular collapse (see p. 967, Congestive Heart Failure).
Diagnostic Evaluation
To initiate a therapeutic plan, several factors must be determined:
- • The degree of dehydration based on physical assessment
- • The type of dehydration based on the pathophysiology of the specific illness responsible for the dehydrated state
- • Specific physical signs other than general signs
- • Initial plasma sodium concentrations
- • Serum bicarbonate concentration
- • Any associated electrolyte (especially serum potassium) and acid-base imbalances (as indicated)
Initial and regular, ongoing evaluations assess the patient's progress toward equilibrium and the effectiveness of therapy.
In the examination of an infant or younger child, one of the most important determinants of the extent of dehydration is body weight because this can assist in determining the percentage of total body fluid lost; however, because the preillness weight is often unknown, clinical manifestations must be evaluated (see Research Focus box). Important clinical manifestations include changing sensorium (irritability to lethargy); decreased response to stimuli; integumentary changes (decreased elasticity and turgor); prolonged capillary refill; increased heart rate; sunken eyes; and, in infants, sunken fontanels. Using multiple predictors increases the sensitivity of assessing the fluid deficit, and studies have shown a reasonably high degree of agreement between experienced observers in assessment of the level of dehydration. Objective signs of dehydration are present at a fluid deficit of less than 5%.
Therapeutic Management
Medical management is directed at correcting the fluid imbalance and treating the underlying cause. When the child is alert, awake, and not in danger, correction of dehydration may be attempted with oral fluid administration. Most cases of dehydration are mild and can be managed at home by this method. Several commercial rehydration fluids are available for use (see Table 25.3). Oral rehydration management consists of replacement of fluid loss over 4 to 6 hours, replacement of continuing losses, and provision for maintenance fluid requirements. In general, the mildly dehydrated child may be given 50 ml/kg of oral rehydration solution (ORS), whereas the child with moderate dehydration may be given 100 ml/kg of ORS. The child with fluid losses from diarrhea may be given an additional 10 ml/kg for each stool (Greenbaum, 2015). Amounts and rates are determined from body weight and severity of dehydration and are increased if rehydration is incomplete or if excess losses continue, until the child is well hydrated and the basic problem is under control.
The child may not be thirsty even though dehydrated and may refuse oral fluids initially for fear of continued emesis (if occurring) or because of decreased strength, oral stomatitis, or thrush. In such children rehydration may proceed by administering 2 to 5 ml of ORS by a syringe or small medication cup every 2 to 3 minutes until the child is able to tolerate larger amounts; if the child has emesis, administering small amounts (5 ml) of ORS every 5 minutes or so may help overcome fluid deficit, and the emesis will often lessen over time (Hendrickson, Zaremba, Wey, et al., 2017). Evidence indicates that oral administration of ondansetron (Zofran) to children with acute gastroenteritis and vomiting reduces emesis and increases time to oral rehydration, thus preventing intravenous (IV) therapy (Carter & Fedorowicz, 2012; Hendrickson, Zaremba, Wey, et al., 2017). Oral rehydration therapy (ORT) is effective for treating mild or moderate dehydration in children, is less expensive, and involves fewer complications than therapy (Carson, Mudd, Madati, 2016; Kleinman & Greer, 2014). ORSs enhance and promote the reabsorption of sodium and water. These solutions greatly reduce vomiting and the need for IV infusions (Hendrickson, Zaremba, Wey, et al., 2017). ORSs, including lower-osmolarity ORS (224 mmol/L), are available in the United States as commercially prepared solutions and are successful in treating the majority of infants with dehydration. See the Quality Patient Outcomes box. (See Diarrhea, Chapter 25, for a complete discussion of fluid replacement therapy for dehydration.)
Parenteral Fluid Therapy
Parenteral fluid therapy is initiated whenever the child is unable to ingest sufficient amounts of fluid and electrolytes to meet ongoing daily physiologic losses, replace previous deficits, and replace ongoing abnormal losses. Patients who usually require IV fluids are those with severe dehydration, those with uncontrollable vomiting, those who are unable to drink for any reason (e.g., extreme fatigue, coma), or those with severe gastric distention.
Because dehydration (volume depletion) constitutes a great threat to life, the first priority is the restoration of circulation by rapid expansion of the ECF volume to treat or prevent shock. IV administration of fluid begins immediately, although the exact nature of the dehydration and the serum electrolyte values are not known. The solution selected is based on what is known regarding the probable type and cause of the dehydration. This usually involves an isotonic solution such as 0.9% sodium chloride or lactated Ringer, both of which are close to the body's serum osmolality of 285 to 300 mOsm/kg and do not contain dextrose (which is contraindicated in the early treatment stages of rehydration, but especially in diabetic ketoacidosis).
Parenteral rehydration therapy has three phases. The initial therapy is used to expand ECF volume quickly and to improve circulatory and renal function. During initial therapy, an isotonic electrolyte solution is used at a rate of 20 ml/kg, given as an IV bolus over 5 to 20 minutes and repeated as necessary after assessment of the child's response to therapy. In a meta-analysis of 10 randomized clinical trials, isotonic fluids were found safer than hypotonic fluids in preventing severe hyponatremia following administration (Wang, Xu, & Xiao, 2014). Subsequent therapy is used to replace deficits, meet maintenance water and electrolyte requirements, and catch up with ongoing losses. Water and sodium requirements for the deficit, maintenance, and ongoing losses are calculated at 8-hour intervals, taking into consideration the amount of fluids given with the initial boluses and the amount administered during the first 24-hour period. With improved circulation during this phase, water and electrolyte deficits can be evaluated, and acid-base status can be corrected either directly through the administration of fluids or indirectly through improved renal function. Potassium is withheld until kidney function is restored and assessed and circulation has improved.
The final phase of therapy allows the patient to return to normal and begin oral feedings, with a gradual correction of total body deficits. The potassium loss in ICF is replaced slowly by way of the ECF. The body fat and protein stores are replaced through diet. If the child is unable to eat or if feeding aggravates a chronic condition, IV maintenance fluids are provided.
Although the initial phase of fluid replacement is rapid in both isotonic and hypotonic dehydration, it is contraindicated in hypertonic dehydration because of the risk of water intoxication, especially in the brain cells, specifically the central pontine cells. Central pontine myelinolysis may occur with an overcorrection of fluid deficit and an overly rapid correction of serum sodium concentration (Greenbaum, 2015). There is an apparent lag time for sodium to reach a steady state when diffusing in and out of brain cells, whereas water diffuses almost instantaneously. Consequently, rapid administration of fluid will cause equally rapid diffusion of water into the dehydrated brain cells, causing marked cerebral edema. Because ECF volume is maintained relatively well in hypertonic as opposed to the other types of dehydration, shock is not a usual manifestation.
Water Intoxication
Water intoxication, or water overload, is observed less often than dehydration. However, it is important that nurses and others who care for children be alert to this possibility in certain situations. Children who ingest excessive amounts of electrolyte-free water develop a concurrent decrease in serum sodium accompanied by central nervous system (CNS) symptoms. There is a large urinary output, and because water moves into the brain more rapidly than sodium moves out, the child may also exhibit irritability, somnolence, headache, vomiting, diarrhea, or generalized seizures. The affected child usually appears well hydrated but may be edematous or even dehydrated.
Fluid intoxication can occur during acute IV water overloading, too-rapid dialysis, tap water enemas, feeding of incorrectly mixed infant formula, or excess water ingestion, or with too rapid reduction of glucose levels in diabetic ketoacidosis (Greenbaum, 2015). Patients with CNS infections occasionally retain excessive amounts of water. Administration of inappropriate hypotonic solutions (e.g., 0.45% sodium chloride) may cause a rapid reduction in sodium and result in symptoms of water overload.
Infants are especially vulnerable to fluid overload. Their thirst mechanism is not well developed; therefore they are unable to “turn off” fluid intake appropriately. A decreased glomerular filtration rate does not allow for repeated excretion of a water load, and ADH levels may not be maximally reduced. Consequently, infants are unable to excrete a water overload effectively.
Administration of inappropriately prepared formula is one of the more common causes of water intoxication in infants (Greenbaum, 2015). Families who cannot afford to buy enough formula may dilute the formula to increase the volume or even substitute water for the formula. A family may run out of formula and dilute the remaining amount to make it last until they are able to purchase more. In addition, water is sometimes used for pacification. Water intoxication can also occur in infants who receive overly vigorous hydration during a febrile illness.
A number of clinicians have reported water intoxication in infants after swimming lessons, in water births (Byard & Zuccollo, 2010), with excessive enema administration, and with gastric lavage (Manz, 2007). Although they hold their breath, some infants apparently swallow a large amount of water during repeated submersion. Anticipatory guidance to parents should include a discussion of swimming instruction and advice to stop a lesson if the child swallows unusual amounts of water or exhibit any symptoms of hyponatremia.
Edema
Edema represents an abnormal accumulation of fluid within the interstitial tissue and subsequent tissue expansion and develops when a defect in the normal cardiovascular circulation or a failure in the lymphatic drainage to remove the increased amounts occurs. The processes responsible for fluid removal include venous hydrostatic pressure, oncotic pressure of intravascular and interstitial spaces, an intact semipermeable capillary wall, tissue tension, and lymphatic flow.
Mechanisms of Edema Formation
A defect of any of the homeostatic mechanisms maintaining fluid balance can cause accumulation of interstitial fluid. Disequilibrium results from anything that (1) alters the retention of sodium, such as renal disease or hormonal influences; (2) affects the formation or destruction of plasma proteins, such as starvation or liver disease; or (3) alters membrane permeability, such as minimal change nephrotic syndrome or trauma.
Edema may be localized to a small or large area, such as that occurring in urticaria, infection, and pulmonary congestion, or it can be generalized, as in the hypoproteinemia of the nephrotic syndrome and starvation. A severe, generalized accumulation of great amounts of fluid in all body tissues is termed anasarca.
Increased Venous Pressure
The colloidal osmotic pressure of the plasma proteins draws fluid back into the vascular system as long as this force is greater than the venous hydrostatic pressure. However, when the venous pressure increases, fluid tends to be retained in the interstitial spaces. This can occur when an individual remains in the same position for a long time, such as swollen ankles and feet after standing or sitting for long periods. Constrictive dressings or restraints applied too tightly to extremities will obstruct venous return, increase venous and capillary pressure, and cause edema. The most graphic pathologic illustrations are pulmonary edema caused by pulmonary circulation overload in cardiac defects with a left-to-right shunt and ascites caused by portal hypertension. Edema from any cause is increased in dependent areas because of this added factor of increased venous hydrostatic pressure and the gravitational effects in these areas.
Capillary Permeability
Damage to capillary walls or alteration in their permeability permits exudation of plasma protein into the interstitial space. Most often this occurs as local edema, such as that manifested in inflammatory and hypersensitivity reactions. Capillary damage from burns allows extensive exudation of protein-rich fluid into the interstitial spaces to compound edema formation.
Diminished Plasma Proteins
A fall in plasma protein levels hampers the osmotic pull back into the vessels. Consequently, fluid remains in the interstitial spaces. Although other factors play a role, such as hydrostatic pressure of both the arterial vascular system and the tissues and sodium concentration, significantly low protein levels (<4.5 mg/dl) are associated with edema. Examples of this are the massive albumin losses of the minimal change nephrotic syndrome, diminished serum protein from insufficient dietary protein, and (sometimes) hemodilution of plasma proteins from IV fluid administration in chronic dehydration.
Lymphatic Obstruction
Obstruction of lymph flow creates edema high in protein content. This occurs infrequently in childhood but can result from trauma to the lymphatic glands or from removal of lymph nodes.
Tissue Tension
Tissue hydrostatic pressure is ordinarily of little consequence. However, it plays a significant role in determining distribution of edema fluid in certain pathologic conditions. Loose tissues allow a greater amount of fluid accumulation than tissues that are tightly bound by dense fibrous bands in which tissue pressure rapidly increases to limit further extravasation of fluid. Edema appears earlier and more readily in loose structures such as those in the periorbital and genital tissues. The alveolar structure of lung tissue is probably a contributing factor in pulmonary edema, as well as in increased hydrostatic pressure in the pulmonary vessels.
Other Factors in Edema Formation
Any factor that causes sodium retention by the kidneys will produce or augment edema formation. This includes stimulation of the renin-angiotensin-aldosterone mechanisms for sodium reabsorption created by the diminished plasma volume in edema, which resulted from primary causes. The salt-retaining property of steroids is responsible for the edema associated with their administration.
Several types of edema exist, all of which can provide a palpable swelling of the interstitial space that is either localized or generalized. These include the following:
- • Peripheral edema, or localized or generalized palpable swelling of the interstitial space
- • Ascites, or the accumulation of fluid in the abdominal cavity (usually associated with renal or liver abnormalities)
- • Pulmonary edema, which occurs when interstitial volume increases
- • Cerebral edema, which is a particularly threatening form of edema caused by trauma, infection, or other etiologic factors, including vascular overload or injudicious IV administration of hypotonic solutions
- • Overall fluid gain, especially seen in patients with kidney disease
Assessment
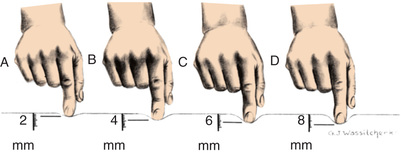
Generalized edema resulting from any of the previously listed types is manifested by swelling in the extremities, face, perineum, and torso. Loss of normal skin creases may be assessed. Daily weights are more sensitive indicators of water gain or loss and should be obtained. Abdominal girth measurement changes may also be an indicator of edema in children. Pitting edema may occur and can be assessed by pressing the fingertip against a bony prominence for 5 seconds. If the tissue rebounds immediately on removing the finger, the patient does not have pitting edema. A quick way to determine the severity is to measure the degree of pitting edema (Fig. 23.4).
Therapeutic Management
The primary goal in the management of edema is treatment of the underlying disease process, which is discussed elsewhere in relation to the specific disorder. However, an essential aspect in the management of any fluid overload is early recognition, in which nurses play a vital role. The management of edema is discussed throughout the text with specific conditions. See the Quality Patient Outcomes box.