The Child With Endocrine Dysfunction
Amy Barry, Erin Connelly

The Endocrine System
The endocrine system controls and regulates metabolism; this includes energy production, growth, fluid and electrolyte balance, response to stress, and sexual reproduction (Gardner, Anderson, & Nissenson, 2011). This system has three components: (1) the cell, which sends a chemical message using a hormone; (2) the target cells, or organs, which receive the chemical message; and (3) the environment through which the chemical is transported (e.g., blood, lymph, extracellular fluids) from the site of synthesis to the sites of cellular action.
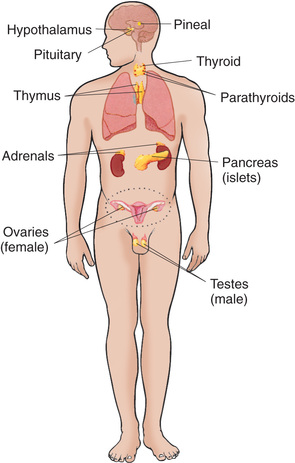
The endocrine glands, which are distributed throughout the body, are listed in Box 31.1; also listed are several additional structures sometimes considered endocrine glands, although they are not usually included.
Hormones
A hormone is a complex chemical substance produced and secreted into body fluids by a cell or group of cells that exerts a physiologic controlling effect on other cells (Garibaldi & Chemaitilly, 2016). Local hormones create their effect near the point of secretion. For example, secretin, a digestive hormone made by cells lining the duodenum, stimulates the pancreas. General hormones are released by endocrine glands into the bloodstream, where they are carried to responsive tissues (Fig. 31.1). Some of these hormones (such as thyroid hormone [TH] and growth hormone [GH]) affect most cells of the body, whereas others (such as the tropic hormones) produce their effects on specific tissues, called target tissues. These responsive, or target, tissues may be another endocrine gland, an organ, or tissue (Gardner, Anderson, & Nissenson, 2011). For example, pituitary hormones stimulate the adrenal glands and the thyroid gland to secrete adrenocorticotropic hormone (ACTH) and thyroid-stimulating hormone (TSH), respectively.
Control of Hormone Secretion
Regulation of hormonal secretion is often based on negative feedback. As a rule, endocrine glands have a tendency to oversecrete particular hormones. However, once the hormone's physiologic effect has been achieved, this information is transmitted to the producing gland, either directly or indirectly, to inhibit further secretion. If the gland undersecretes, the inhibition is stopped, and the gland increases production of the hormone again. As a result, the hormone is secreted according to the amount needed. This is the primary function of the tropic hormones.
The anterior pituitary gland, located below the hypothalamus, is often referred to as the master gland. It is primarily responsible for stimulation and inhibition of tropic hormones. Tropic (which literally means “turning”) hormones secreted by the anterior pituitary regulate the secretion of hormones from various target organs (Fig. 31.2). As blood concentrations of the target hormones reach normal levels, a negative message is sent to the anterior pituitary to inhibit release of the tropic hormone. For example, TSH responds to low levels of circulating TH. As blood levels of TH reach normal concentrations, a negative feedback message is sent to the anterior pituitary, resulting in diminished release of TSH.
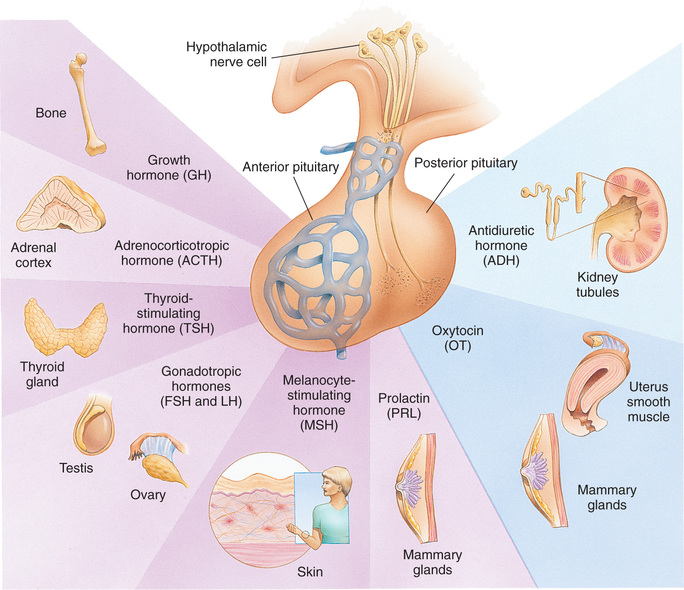
The pituitary gland is controlled by either hormonal or neuronal signals from the hypothalamus. Two types of substances are secreted from the hypothalamus: (1) releasing hormones and (2) inhibitory hormones. Both are secreted within the hypothalamus and transported by way of the pituitary portal system to the anterior pituitary, where they stimulate the secretion of tropic hormones. An example of this is the secretion of corticotropin-releasing factor (CRF) by the hypothalamus. CRF stimulates the pituitary to secrete ACTH. In this instance the anterior pituitary is the target of the hypothalamus. ACTH then stimulates the adrenals to secrete glucocorticoids, which have multiple target sites throughout the body. Pituitary hormones that lack feedback control from the product of a target tissue (e.g., GH, prolactin, and melanocyte-stimulating hormone) require hypothalamic inhibitors and stimulators for their control.
Not all hormones depend on other hormones for their release. For example, insulin is secreted in response to blood glucose concentrations. Other glandular hormones that are not under the control of the pituitary gland are glucagon, parathyroid hormone (parathormone, PTH), antidiuretic hormone (ADH), and aldosterone.
Neuroendocrine Interrelationships
Two regulatory systems maintain hemostasis: the endocrine and the autonomic nervous systems (collectively known as the neuroendocrine system) (Gardner, Anderson, & Nissenson, 2011). The autonomic nervous system consists of the sympathetic and parasympathetic systems that control nonvoluntary functions—specifically that of smooth muscle, myocardium, and glands. The parasympathetic system primarily regulates the digestive processes, whereas the sympathetic system functions to maintain homeostasis during times of stress.
The higher autonomic centers, located in the hypothalamus and limbic system, help control the functioning of both autonomic systems. Both sympathetic and parasympathetic nerve fibers secrete neurotransmitting substances: acetylcholine, released by cholinergic fibers, and norepinephrine, released by adrenergic fibers. Release of norepinephrine into the plasma produces the same effects as secretion of this substance by the adrenal medulla. Thus the interrelatedness between the two systems is demonstrated.
The neuroendocrine system acts by synthesizing and releasing various chemical substances that regulate body functions. Information is carried by means of neural impulses in the autonomic system and by the blood in the endocrine system. In general, neural responses are more rapid and localized, whereas endocrine responses are more lasting and widespread. The two systems function synergistically because neural impulses transmitted to the central nervous system (CNS) stimulate the hypothalamus to manufacture and release several releasing or inhibiting factors.
Because of the interdependent relationship of these glands, a malfunction in one gland produces effects elsewhere. Endocrine dysfunction may result from an intrinsic defect in the target gland or from a diminished or elevated level of tropic hormones. Endocrine problems occur when there is hypofunction or hyperfunction of the glands. Primary hypofunction is usually associated with a more profound deficiency of the target gland hormone because little or no hormone is secreted. In secondary dysfunction the target glands secrete some of their hormones but in smaller amounts and less rapidly.
Disorders of Pituitary Function
The pituitary gland is divided into two lobes: the anterior pituitary (adenohypophysis) and the posterior pituitary (neurohypophysis). It is regulated by hormones secreted from the hypothalamus. The anterior pituitary is responsible for secreting GH, TSH, ACTH, follicle-stimulating hormone (FSH), luteinizing hormone (LH), and prolactin. The posterior pituitary secretes ADH and oxytocin (Sathyapalan & Dixit, 2012).
Deficiencies of the anterior pituitary hormones may be due to organic defects or have an idiopathic etiology. Clinical manifestations depend on the hormones involved and the age of the patient. If the tropic hormones are involved, the resulting disorder reflects the altered stimulus of the target gland. For example, if TSH is deficient, the thyroid gland does not secrete TH and the child displays clinical signs of hypothyroidism.
An overproduction of the anterior pituitary hormones can result in gigantism (caused by excess GH production during childhood), hyperthyroidism, hypercortisolism (Cushing syndrome), and precocious puberty from excessive gonadotropins (LH and FSH). Overproduction may be caused by hyperplasia of the pituitary cells—which may eventually progress to a tumor (adenoma)—or a primary hypothalamic defect that results in an excess of the hormone's releasing factor. Although the initial clinical manifestations are a result of pituitary oversecretion, eventually pituitary insufficiency occurs, and the signs of panhypopituitarism become evident. Panhypopituitarism is defined clinically as the loss of all anterior pituitary hormones, leaving only posterior pituitary function intact (Toogood & Stewart, 2008).
Hypopituitarism
Hypopituitarism is the diminished secretion of one or more pituitary hormones. The consequences of the condition depend on the degree of dysfunction. It often leads to the following:
- • Gonadotropin deficiency (decrease in LH or FSH), where children show absence or regression of secondary sexual characteristics
- • GH deficiency, which children display stunted somatic growth
- • TSH deficiency, which produces hypothyroidism
- • Corticotropin deficiency, which results in manifestations of adrenal hypofunction
Hypopituitarism can result from any of the conditions listed in Box 31.2. The most common organic cause of pituitary undersecretion is a tumor in the pituitary or hypothalamic region. Craniopharyngiomas are tumors known to invade these regions of the brain and cause panhypopituitarism (Box 31.3). A child may experience decreased growth velocity for some time before developing any symptoms or signs of increased intracranial pressure, local compression, or the destructive effects of a tumor. Other causes of panhypopituitarism include encephalitis, radiation to the head or neck, traumatic brain injury, and congenital hypoplasia of the hypothalamic area (Rose & Auble, 2012; Sathyapalan & Dixit, 2012).
Congenital hypopituitarism can be seen in newborn infants and run in families, suggesting a genetic cause; however, the majority of cases have no genetic association (Schoenmakers, Alatzoglou, Chatterjee, et al., 2015). Neonates may have symptoms of hypoglycemia and seizure activity (Schoenmakers, Alatzoglou, Chatterjee, et al., 2015). A child with combined GH deficiency and hypothyroidism should be screened for congenital pituitary defects and genetic mutations (Pine-Twaddell, Romero, & Radovick, 2013).
Idiopathic hypopituitarism, or idiopathic pituitary growth failure, is usually related to GH deficiency, which inhibits somatic growth in all cells of the body (Amin, Mushtaq, & Alvi, 2015). Although most children with hypopituitarism are normal at birth, many show growth patterns that progressively deviate from the normal growth rate, often beginning in infancy. The chief complaint is often short stature. Of those who seek help, boys outnumber girls three to one. Idiopathic hypopituitarism can be associated with deficiencies of other pituitary hormones, such as TSH and ACTH; thus it is theorized that the disorder is probably secondary to hypothalamic deficiency.
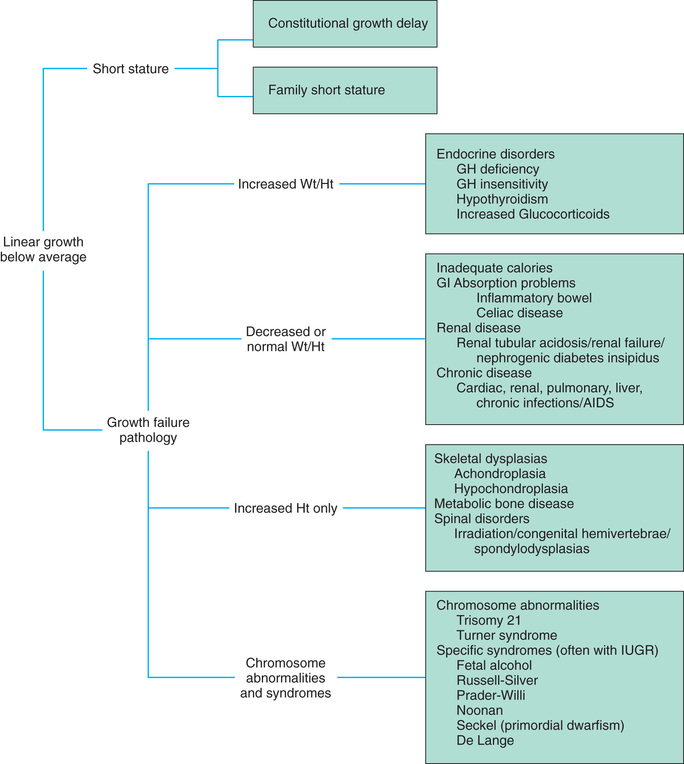
Isolated GH deficiency without other associated pituitary hormone deficiencies or a known organic cause is also seen in children (Stanley, 2012). Growth failure is defined as an absolute height of less than −2 standard deviations (SD) for age, or a linear growth velocity consistently less than −1 SD for age. When this occurs without the presence of hypothyroidism, systemic disease, or malnutrition, an abnormality of the GH–insulin-like growth factor (IGF) axis should be considered (Grimberg, Divall, Polychonakos, et al., 2016).
Not all children with short stature have GH deficiency. In most instances the cause of short stature is considered idiopathic. Idiopathic short stature (ISS) is defined as a condition in which the height of an individual is more than 2 SD below the mean height for his or her age, sex, and population group, without evidence of systemic, endocrine, nutritional, or chromosomal abnormalities (Argente, 2016). Children with ISS fall into one of three groups: those with familial short stature, those with constitutional delay of growth and puberty, and those with a yet unidentified cause of short stature. Familial short stature refers to healthy children who have ancestors with adult height in the lower percentiles and whose height during childhood is appropriate for genetic background (Fig. 31.3). Constitutional delay of growth and puberty refers to individuals with delayed linear growth and delayed skeletal and sexual maturation for age (Amin, Mushtaq, & Alvi, 2015). GH therapy in children with ISS continues to be debated frequently by pediatric endocrinologists (Murray, Dattani, & Clayton, 2016). New guidelines are available that recommend the clinical management of children with growth failure related to growth hormone deficiency and idiopathic short stature. They now include recombinant IGF-1 therapy for primary IGF-1 deficiency (Grimberg, Divall, Polychronakos, et al., 2016).
Clinical Manifestations
Children with GH deficiency often grow normally during the first year and then follow a slowed growth curve that is below the 3rd percentile. In children with a partial GH deficiency, the growth retardation is less marked than in children with complete GH deficiency. Height may be stunted more than weight because, with good nutrition, these children can become overweight or even obese. Their well-nourished appearance is an important diagnostic clue to differentiation from other disorders such as failure to thrive. Skeletal proportions are normal for the age, but these children appear younger than their chronologic age. Later in life, premature aging is common. Bone age is delayed but is closely related to height age; the degree of growth delay depends on the duration and extent of the hormonal deficiency. Because of the underdeveloped jaw, teeth may be overcrowded and malpositioned.
Children with isolated GH deficiency have normal intelligence. However, emotional problems are common, especially as they near puberty, when their smallness becomes increasingly apparent in comparison with their peers. Height discrepancy has been correlated with emotional adjustment problems and may be a predictor of the extent to which GH-delayed children or children with short stature will experience emotional distress, relationship dysfunction (friendship and later dating/marriage), educational attainment, and type of employment (Sandberg & Gardner, 2015).
Diagnostic Evaluation
Only a small number of children with delayed growth or short stature have hypopituitary dysfunction. Diagnostic evaluation is aimed at isolating organic causes, which, in addition to GH deficiency, may include brain tumor, hypothyroidism, oversecretion of cortisol, gonadal aplasia, chronic illness, nutritional inadequacy, Russell-Silver dwarfism, or hypochondroplasia.
A complete diagnostic evaluation should include a family history, a history of the child's growth patterns and previous health status, a physical examination, and a psychosocial evaluation. Specific radiographic imaging, including magnetic resonance imaging (MRI), endocrine studies, and genetic testing, may also be warranted (Stanley, 2012).
Family History
A family history is of utmost importance in relating short stature to genetic background. The midparental height is an important prognosticator of the child's ultimate adult height. Normal adult height should fall within 5 cm (2 inches) of midparental height (Ferguson, 2011). Children with constitutional delays frequently are the products of parents who experienced similar slow growth patterns and delayed sexual maturation. A small percentage of those with GH deficiency demonstrate an autosomal recessive inheritance pattern. Height and weight of siblings should be compared with the child's growth patterns at comparable age periods.
Child's History
The child's history should include a thorough prenatal history to rule out maternal disorders that may have influenced growth, such as malnutrition. Compare birth height and weight with gestational age. Children with hypopituitarism are usually of normal size and normal gestational age at birth.
Investigate the child's health history for evidence of chronic illness that may have influenced growth patterns, although a chronic illness, such as congenital heart disease, malabsorptive disorders, severe anemia, or neurologic impairments, usually is identified long before the growth problem becomes a concern. Signs and symptoms suggesting a tumor, such as visual disturbances, headache, and signs of increasing intracranial pressure, are important. With lesions involving the hypothalamus, the history may also reveal somnolence, thermodysregulation, epilepsy, and polyphagia, resulting in obesity. Because a craniopharyngioma can affect the secretion of any of the pituitary hormones, assessment for hypothyroidism, hypoadrenalism, and hypoaldosteronism should also be included.
Whenever possible, evaluate the child's growth patterns since birth, especially growth velocity, and compare them with standard measurements. The age of onset of short stature provides a significant diagnostic clue. When the clinician evaluates the results of plotting height and weight, upward or downward changes in height velocity in children older than 3 years may indicate a growth abnormality (Cheetham & Davies, 2014).
Physical Examination
Accurate measurement of height (using a calibrated stadiometer) and weight and comparison with standard growth charts are essential (Box 31.4). Multiple height measures reflect a more accurate assessment of abnormal growth patterns (Cheetham & Davies, 2014). Other measurements may include crown-to-pubis and pubis-to-heel length to compare body proportions. Sexual development should be assessed and compared with age-appropriate development. Observation of general appearance yields valuable clues, especially signs of premature aging and infantile facial features. Funduscopic examination and testing for visual acuity are important to detect evidence of ocular damage from a tumor.
Radiographic Surveys
A skeletal survey in children less than 3 years of age and radiographic examination of the hand-wrist for centers of ossification (bone age) (Box 31.5) in older children are important in evaluating growth. Epiphyseal maturation is delayed in GH deficiency but consistent with height. This is in contrast with gonadal dysplasia, such as Turner syndrome, in which bone age is near normal. A skull series may help identify abnormalities such as an abnormally small sella turcica where the pituitary is located. MRI, computed tomography (CT), radionuclear scans, or carotid angiograms may be needed to establish diagnosis and localization of brain lesions (Stanley, 2012).
Endocrine Studies
Definitive diagnosis of GH deficiency is based on absent or subnormal reserves of pituitary GH. Measuring serum IGF-1 and IGF binding protein 3 (IGFBP3) levels may assist in the decision to pursue further testing for GH deficiency. It is recommended that GH stimulation tests be reserved for children with low serum IGF-1 and IGFBP3 levels and poor growth who do not have other endocrine or nonendocrine causes for short stature (Hokken-Koelega, 2011). However, although the IGF-1 test is useful in detecting severe GH insensitivity, it may not be accurate in detecting less severe cases of idiopathic short stature (Hokken-Koelega, 2011).
GH stimulation testing involves the use of pharmacologic agents such as levodopa, clonidine, arginine, insulin, propranolol, or glucagon followed by the measurement of GH in the blood (Parks & Felner, 2016). Studies have shown that traditional GH stimulation test results can be less reliable than previously accepted, and recommendations have been made to standardize assays (Stanley, 2012). Currently, findings in a child of poor linear growth, delayed bone age, and peak levels of GH at less than 10 ng/ml in two stimulation tests are consistent with GH deficiency (Parks & Felner, 2016). New reference standards that decrease the lower limit of normal GH levels have been proposed and continue to be debated (Murray, Dattani, & Clayton, 2016).
Therapeutic Management
Treatment of GH deficiency caused by organic lesions is directed toward correction of the underlying disease process (e.g., surgical removal or irradiation of a tumor). GH therapy in children with ISS continues to be debated frequently by pediatric endocrinologists (Murray, Dattani, & Clayton, 2016). New guidelines are available that recommend the clinical management of children with growth failure related to growth hormone deficiency and idiopathic short stature. They now include recombinant IGF-1 therapy for primary IGF-1 deficiency (Grimberg, Divall, Polychronakos, et al., 2016).
For more than 20 years cadaver-derived human growth hormone (HGH) was used successfully to enhance linear growth in short children. In 1985 the U.S. Food and Drug Administration stopped use of the hormone in response to reported deaths resulting from Creutzfeldt-Jakob disease (CJD) in three former HGH recipients. Patients have been identified who received HGH and became infected with CJD, a rare and fatal neurodegenerative condition iatrogenically transmitted through human tissue (Cali, Miller, Parisi, et al., 2015). Biosynthetic GH prepared by recombinant deoxyribonucleic acid (DNA) technology (and without the risk of CJD) is now used exclusively (Grimberg, Divall, Polychronakos, et al., 2016).
In the United States the recommended dosage range of recombinant GH is 0.037 to 0.18 to 0.3 mg/kg per week, divided into six or seven daily subcutaneous doses (Parks & Felner, 2016). Maximum growth velocity occurs in the first year of treatment; with each consecutive year of treatment, growth rates decline (Parks & Felner, 2016). A meta-analysis of the GH literature concluded that, although GH may improve growth velocity in children, individuals who receive treatment remain shorter than their peers (Bryant, Baxter, Cave, et al., 2007). For children to achieve their genetic growth potential, early diagnosis of and intervention for growth disorders are essential (Argente, 2016).
The child, family, and health care team make the decision jointly to stop GH therapy. Growth rates of less than 1 inch per year and a bone age of more than 14 years in girls and more than 16 years in boys are often used as criteria to stop GH therapy (Parks & Felner, 2016). Children with other hormone deficiencies require replacement therapy to correct the specific disorders. This may involve administration of thyroid extract, cortisone, testosterone, or estrogens and progesterone. The sex hormones are usually begun during adolescence to promote normal sexual maturation.
Nursing Care Management
The principal nursing consideration is identifying children with growth problems. Although the majority of growth problems are not a result of organic causes, any delay in normal growth and sexual development poses special emotional adjustments for these children.
The nurse is a key person in helping establish a diagnosis. For example, if serial height and weight records are not available, the nurse can question parents about the child's growth compared with that of siblings, peers, or relatives. Investigating clothing sizes is often helpful in determining growth at different ages. (See Quality Patient Outcomes box.)
Because the behavioral or physical changes that suggest a tumor are insidious, they are frequently overlooked. It is important to correlate the onset of any positive findings with the initial evidence of growth retardation. For example, visual problems and headaches are not uncommon in school-age children and can coincidentally occur after a growth problem is recognized. In fact, headache may represent the emotional trauma caused by short stature rather than be a symptom of a tumor. Pursue this line of questioning cautiously to avoid alarming parents unduly about the possibility of a brain tumor.
Part of a nurse's role in helping establish a diagnosis is assisting with diagnostic tests. Preparation of the child and family is especially important if a number of tests are being performed and the child requires particular attention during GH stimulation testing. Blood samples are usually taken every 30 minutes for a 3-hour period. Children also have difficulty overcoming hypoglycemia generated by tests with insulin, so they should be carefully observed for signs of hypoglycemia. Those receiving glucagon are at risk of nausea and vomiting. Patients receiving clonidine require close blood pressure monitoring. Nursing administration of intravenous (IV) fluids may be required if hypotension is detected. The use of arginine is often well tolerated by children, but it may cause hypoglycemia in some infants and toddlers. Therefore close monitoring for hypoglycemia is necessary.
Child and Family Support
If an organic cause of the problem has been confirmed, the parents and child need an opportunity to express their thoughts and feelings. Frequently a growth problem that was present since birth is missed until adolescence, at which time the child's difference in body development becomes dramatically evident in comparison with peers. Family members may feel anger and resentment toward members of the health staff for not detecting the problem sooner. Parents may experience guilt for not seeking medical attention earlier, especially if the child has been miserable from the ridicule and criticism of peers. Appropriate emotional support from the nurse can include an affirmation of each person's justified feelings, such as anger or guilt, and emphasis on the treatment plan and prospects for improvement in the future.
Even when hormone replacement is successful, these children attain their eventual adult height at a slower rate than their peers; therefore they need assistance in setting realistic expectations regarding improvement. Psychologic counseling may be considered for children diagnosed with idiopathic short stature whether done in conjunction with GH therapy or treatment instead (Sandberg & Gardner, 2015). Both sexes need guidance toward appropriate vocational goals. Because these children appear younger than their chronologic age, others frequently relate to them in infantile or childish ways. Children having school problems need special counseling. Parents and teachers benefit from guidance directed toward setting realistic expectations for the child based on age and abilities. For example, in the home such children should have the same age-appropriate responsibilities as their siblings. As they approach adolescence, encourage them to participate in group activities with peers. If abilities and strengths are emphasized rather than physical size, such children are more likely to develop a positive self-image.
Professionals and families may find research, education, support, and advocacy from the Human Growth Foundation.* Treatment is expensive—up to $52,000 per year depending on the dosage or approximately $35,000 per inch (Ferguson, 2011). Usually the cost is partially covered by insurance if the child has a documented deficiency, and some pharmaceutical companies offer copay assistance.
Pituitary Hyperfunction
Excess GH before closure of the epiphyseal shafts results in proportional overgrowth of the long bones until the individual reaches a height of 2.4 m (8 feet) or more. Vertical growth is accompanied by rapid and increased development of muscles and viscera. Weight is increased but is usually in proportion to height. Proportional enlargement of head circumference also occurs and may result in delayed closure of the fontanels in young children. Children with a pituitary-secreting tumor may also demonstrate signs of increasing intracranial pressure, especially headache.
If oversecretion of GH occurs after epiphyseal closure, growth is in the transverse direction, producing a condition known as acromegaly. Typical facial features include the following:
- • Overgrowth of the head, lips, nose, tongue, jaw, and paranasal and mastoid sinuses
- • Separation and malocclusion of the teeth in the enlarged jaw
- • Disproportion of the face to the cerebral division of the skull
- • Increased facial hair; thickened, deeply creased skin
- • Increased tendency toward hyperglycemia and diabetes mellitus (DM)
Excessive secretion of GH by a pituitary adenoma causes most cases of acromegaly. Acromegaly can develop slowly, with patients being diagnosed as much as 10 years after their symptoms first appear. Left untreated, these patients have a higher mortality rate due to potential cardiovascular, metabolic, and pulmonary complications (Pivonello, Auriemma, Grasso et al., 2017).
Diagnostic Evaluation
Diagnosis is based on a history of excessive growth during childhood and evidence of increased levels of IGF-1 concentration. If this is elevated, a specialized GH test will be done to assess for excess secretion (Ribeiro-Oliveira & Barkan, 2012). MRI may reveal a tumor in an enlarged sella turcica, normal bone age, enlargement of bones (such as the paranasal sinuses), and evidence of joint changes. Endocrine studies to confirm excess of other hormones, specifically thyroid, cortisol, and sex hormones, should also be included in the differential diagnosis.
Therapeutic Management
If a lesion is present, surgical treatment by cryosurgery or hypophysectomy is performed to remove the tumor when possible. Transsphenoid surgery (TSS) is the most common surgical treatment for pituitary adenoma. External radiation (XRT) or radioactive implants may be used to destroy GH-secreting tissue. Medical therapy using drugs that can suppress GH may be used in conjunction with TSS and/or XRT to treat this disease (Maffezzoni, Frara, Doga, et al., 2016). Depending on the extent of surgical excision and degree of pituitary insufficiency, hormone replacement with thyroid extract, cortisone, and sex hormones may be necessary.
Nursing Care Management
The primary nursing consideration is early identification of children with excessive growth rates. Although medical treatment of acromegaly will not reduce a patient's height, it will prevent further excess growth. Nurses in ambulatory settings who are frequently involved in growth screening should refer children who demonstrate excessive linear growth for a medical evaluation. They should also observe for signs of a tumor, especially headache, and evidence of concurrent hormonal excesses, particularly the gonadotropins, which cause sexual precocity.
Children with excessive growth rates require as much emotional support as those with short stature. However, girls may suffer from the effects of excessive height much more than boys. Like boys, some girls may find their height to be an asset when pursuing sports such as basketball. Children and their parents need an opportunity to express their thoughts. A compassionate nurse can be supportive to these children, especially before adolescence when they are larger than their peers. The nurse can emphasize to a tall girl that as boys grow older, they become taller and she will not always be looking down at them.
Precocious Puberty
Manifestations of sexual development before age 9 years in boys or age 8 years in girls have traditionally been considered precocious development, and these children were recommended for further evaluation (Brito, Spinola-Castro, Kochi, et al., 2016), but guidelines on this have been debated (see Research Focus box).
Normally the hypothalamic-releasing factors stimulate secretion of the gonadotropic hormone from the anterior pituitary at the time of puberty. In the male, interstitial cell–stimulating hormone stimulates Leydig cells of the testes to secrete testosterone. In the female follicle-stimulating hormone and luteinizing hormone stimulate the ovarian follicles to secrete estrogens. This sequence of events is known as the hypothalamic-pituitary-gonadal axis. If for some reason the cycle undergoes premature activation, the child displays evidence of advanced or precocious puberty. Sex hormones affect bone growth, and premature exposure may cause short stature. Box 31.6 lists the causes of precocious puberty.
Isosexual precocious puberty is more common among girls than boys. Approximately 50% of children with precocious puberty have central precocious puberty (CPP), in which pubertal development is activated by the hypothalamic gonadotropin-releasing hormone (GnRH). This produces early maturation and development of the gonads, with secretion of sex hormones, development of secondary sexual characteristics, and sometimes production of mature sperm and ova (Garibaldi & Chemaitilly, 2016). CPP occurs more frequently in girls and is usually idiopathic, with 95% demonstrating no causative factor (Brito, Spinola-Castro, Kochi, et al., 2016). A CNS insult or structural abnormality occurs in more than 75% of boys with CPP (Garibaldi & Chemaitilly, 2016).
Peripheral precocious puberty (PPP) refers to early puberty resulting from hormone stimulation other than the hypothalamic GnRH–stimulated pituitary gonadotropin release. Clinical findings normally associated with puberty may be seen as variations in normal sexual development (Brito, Spinola-Castro, Kochi, et al., 2016). They appear without other signs of puberty and are probably caused by unusual end-organ sensitivity to prepubertal levels of estrogen or androgen. Included are premature thelarche (development of breasts in prepubertal girls), premature pubarche (premature adrenarche, early development of sexual hair), and premature menarche (isolated menses without other evidence of sexual development).
Therapeutic Management
Children with evidence of precocious puberty should be evaluated by a pediatric endocrinologist. In every case, an MRI of the brain should be performed to assess for hypothalamic brain tumor (Brito, Spinola-Castro, Kochi, et al., 2016). When there is no organic cause for CPP, patients will be monitored closely for growth. In 50% of cases, precocious pubertal development regresses or stops advancing without any treatment. CPP can be managed with monthly injections of a synthetic analog of luteinizing hormone–releasing hormone, which decreases the pituitary secretion of LH and FSH (Garibaldi & Chemaitilly, 2016). A slow-release formulation of leuprolide acetate (Lupron Depot) is given in a dosage of 0.2 to 0.3 mg/kg intramuscularly every 4 weeks. A longer-lasting preparation may be given intramuscularly every 3 months and has also been successful in the treatment of CPP in a majority of patients. Although expensive, the GnRH analog (GnRHa) histrelin has been formulated as a subdermal implant with effects lasting 12 months and may be beneficial for some patients who would like to avoid injections (Garibaldi & Chemaitilly, 2016).
After initiation of treatment, breast development regresses or does not advance, and growth returns to normal rates, enhancing predicted height. Treatment is discontinued at a chronologically appropriate time, allowing pubertal changes to resume. Despite the early sexual development, maturation of the gonads and the appearance of secondary sexual characteristics proceed in the usual order. The most difficult time for the child is usually the school years before adolescence. After puberty, physical differences from peers are no longer present. Some patients, however, do not attain adult targeted height during therapy. The use of GH to improved adult height is being investigated with children with precocious puberty and advanced bone age (Garibaldi & Chemaitilly, 2016).
Nursing Care Management
Psychologic support and guidance of the child and family are the most important aspects of management. Parents and children need anticipatory guidance, support and information resources, and reassurance. GnRH agonists are associated with side effects such as headache, emotional lability, and vasodilation causing hot flashes (Harrington & Palmert, 2016). Nurses are essential in providing medical education to the patient and family. Dress and activities for the physically precocious child should be appropriate to the chronologic age. Sexual interest is not usually advanced beyond the child's chronologic age, and parents need to understand that the child's mental age is congruent with the chronologic age.
Although the child's sexual behavior may be appropriate for the chronologic age, the nurse should emphasize to parents that the child may be fertile. Usually no form of contraception is necessary unless the child is sexually active. In this situation proper counseling is important because hormonal forms of birth control, such as estrogen pills, prematurely initiate epiphyseal closure, resulting in stunted linear growth.
Diabetes Insipidus
The principal disorder of posterior pituitary hypofunction is diabetes insipidus (DI). Also known as neurogenic DI, central diabetes insipidus results from the undersecretion of antidiuretic hormone (ADH), also known as vasopressin. This disease results in the production of large volumes of urine (polyuria), which leads to a state of uncontrolled diuresis (Di Iorgi, Napoli, Allegri, et al., 2012). Central DI is not to be confused with nephrogenic DI, a rare hereditary disorder affecting primarily males and caused by unresponsiveness of the renal tubules to the hormone. (See Chapter 24.)
Neurogenic DI may result from a number of different causes. Primary causes are familial or idiopathic; approximately 20% to 50% of the total cases are idiopathic (Di Iorgi, Allegri, Napoli, et al., 2014). More than 55 genetic mutations have been identified, which result in a deficiency of vasopressin and cause familial central DI (Di Iogri, Napoli, Allegri, et al., 2012). Secondary causes include trauma (accidental or surgical), tumors, granulomatous disease, Langerhans cell histiocytosis (LCH), autoimmune disease, infections (meningitis or encephalitis), cranial malformations, and vascular anomalies (aneurysm). Certain drugs, such as alcohol or phenytoin (diphenylhydantoin), can cause a transient polyuria. DI may be an early sign of an evolving cerebral process (Di Iogri, Napoli, Allegri, et al., 2012).
Clinical Manifestations
The cardinal signs of DI are polyuria and polydipsia. In the older child, signs can include excessive urination accompanied by insatiable thirst so intense that the child does little more than go to the toilet and drink fluids. Frequently the first sign is enuresis. In the infant the initial symptom is irritability that is relieved with feedings of water but not milk. The infant is also prone to severe dehydration, electrolyte imbalance, hyperthermia, azotemia, and potential circulatory collapse. Other symptoms such as vomiting, constipation, fever, irritability, sleep issues, failure to thrive, and growth problems may be seen.
Dehydration is usually not a serious problem in older children, who are able to drink larger quantities of water. However, any period of unconsciousness, such as after trauma or anesthesia, may be life threatening because the voluntary demand for fluid is absent. During such instances careful monitoring of urine volumes, blood concentration, and IV fluid replacement is essential to prevent dehydration.
Diagnostic Evaluation
The simplest test used to diagnose this condition is the water deprivation test, which restricts oral fluids and observes changes in urine volume and concentration. Normally, reducing fluids results in concentrated urine and diminished volume. In DI, fluid restriction has little or no effect on urine formation but causes weight loss from dehydration. Accurate results from this procedure require strict monitoring of fluid intake and urinary output, measurement of urine concentration (specific gravity or osmolality), and frequent weight checks. A weight loss between 3% and 5% indicates significant dehydration and requires termination of the fluid restriction.
If this test is positive, the child should be given a test dose of injected aqueous vasopressin (Pitressin), which should alleviate the polyuria and polydipsia. Unresponsiveness to exogenous vasopressin usually indicates nephrogenic DI but a rise in urine osmolality after vasopressin administration indicates central DI (Arima, Azuma, Morishita, et al., 2016).
An important diagnostic consideration is differentiating DI from other causes of polyuria and polydipsia, especially diabetes mellitus. MRI to look for secondary causes of central DI such as a tumor or central brain anomaly is essential (Di Iorgi, Napoli, Allegri, et al., 2012). Often kidney function tests, urine osmolality tests, blood electrolyte levels, and specific endocrine studies are sent to isolate associated problems (Breault & Majzoub, 2016). In rare instances a psychologic consultation may be warranted to confirm the possibility of compulsive water drinking related to psychogenic causes.
Therapeutic Management
The usual treatment is hormone replacement using DDAVP, which is a synthetic analog of the endogenous hormone arginine vasopressin (AVP). DDAVP can be given orally, intranasally, or parenterally. The intranasal and oral forms of DDAVP are most commonly used in children. Oral DDAVP has few complications and is easier to give, which likely increases compliance (Di Iorgi, Napoli, Allegri, et al., 2012).
The medication is usually administered twice daily—at bedtime to allow the child to sleep through the night and in the morning to allow fewer interruptions in the school day. Some “breakthrough” urination is allowed during the evening hours as a precaution against overmedication. The signs of overmedication are similar to manifestations associated with the syndrome of inappropriate ADH (SIADH). (See the next section.)
Nursing Care Management
The initial objective is identification of the disorder. Because an early sign may be sudden enuresis in a child who is toilet trained, excessive thirst with bed-wetting is an indication for further investigation. Another clue is persistent irritability and crying in an infant that is relieved only by bottle-feedings of water. After head trauma or certain neurosurgical procedures, the development of DI can be anticipated; therefore closely monitor these patients. (See Quality Patient Outcomes box.)
Assessment includes measurement of body weight, serum electrolytes, blood urea nitrogen (BUN), hematocrit, and urine specific gravity taken before surgery and every other day after the procedure. Fluid intake and output should be carefully measured and recorded. Alert patients are able to adjust intake to urine losses, but unconscious or very young patients require closer fluid observation. In children who are not toilet trained, collection of urine specimens may require application of a urine-collecting device.
After confirmation of the diagnosis, parents need a thorough explanation regarding the condition, with specific clarification that DI is a different condition from DM. They must realize that treatment is lifelong. If children are to receive DDAVP, ideally two caregivers should learn the correct procedure for preparation and administration of the drug. Once children are old enough, encourage them to assume full responsibility for their care.
For emergency purposes, these children should wear medical alert identification. School personnel need to be aware of the problem so they can grant children unrestricted use of the lavatory. Failure to permit this may result in embarrassing accidents that often result in a child's unwillingness to attend school. Medication may need to be kept at school depending on dosing schedules.
Syndrome of Inappropriate Antidiuretic Hormone
Oversecretion of the posterior pituitary antidiuretic hormone (ADH) causes the disorder known as syndrome of inappropriate antidiuretic hormone (SIADH). This disorder occurs with increased frequency in a variety of conditions that disrupt CNS function such as infection, tumor, or surgery. It can also be the side effect of a variety of medications. SIADH is the most common cause of hyponatremia in hospitalized patients (Cuesta, Garrahy, & Thompson, 2016).
Excess ADH causes free water to be reabsorbed from the kidneys. As increased free water circulates, serum osmolality goes down, and urine osmolality inappropriately increases. Clinical signs of SIADH are directly related to fluid retention and hyponatremia. When hyponatremia occurs acutely, swelling of the brain occurs (Giuliani & Peri, 2014). When serum sodium levels are diminished to 120 mEq/L, affected children may display anorexia, nausea, vomiting, stomach cramps, irritability, and personality changes. With progressive hyponatremia, more serious neurologic signs, such as stupor and seizures, may occur.
Fluid restriction is the immediate management of choice. Subsequent management depends on the cause and severity. Fluids may be restricted in anticipation of SIADH development postoperatively. Some children may be treated with oral sodium replacement. Severe SIADH may require hypertonic saline infusion that is only given in the hospital setting under close supervision (Cuesta, Garrahy, & Thompson, 2016).
Nursing Care Management
The recognition of SIADH symptoms is the primary nursing goal. Close attention to measurements of intake and output, weight, and monitoring for the development of neurologic symptoms is essential, especially in patients at risk in the intensive care setting. (See Quality Patient Outcomes box.)
Seizure precautions are implemented in children at high risk for SIADH. The child and family need education and support regarding the rationale for fluid restrictions. The rare child with chronic SIADH is placed on long-term ADH-antagonizing medication. The patient and family will need instruction regarding medication administration.
Disorders of Thyroid Function
The thyroid gland secretes two types of hormones: thyroid hormone (TH) and calcitonin. TH is made up of the hormones thyroxine (T4) and triiodothyronine (T3). The anterior pituitary hormone TSH controls the secretion of TH. TSH is regulated by the hypothalamic hormone thyrotropin-releasing factor (TRF) as a negative feedback response. Hypothyroidism or hyperthyroidism may result from a defect in the thyroid or from a disturbance in the secretion of TSH or TRF. Because the functions of T3 and T4 are qualitatively the same, the term thyroid hormone (TH) is used throughout the discussion (Box 31.7).
The synthesis of TH depends on available sources of dietary iodine and tyrosine. The thyroid is the only endocrine gland capable of storing excess amounts of hormones for release. During circulation T4 and T3 are bound to carrier proteins (thyroxine-binding globulin). They must be unbound before they are able to exert their metabolic effect.
The main physiologic action of TH is to regulate metabolism and control the processes of growth and tissue differentiation, as outlined in Box 31.7. Unlike GH, TH is involved in many more diverse activities that influence the growth and development of body tissues. Therefore a deficiency of TH exerts a more profound effect on growth than that seen in GH deficiency.
Calcitonin helps maintain blood calcium levels by decreasing the calcium concentration. It inhibits skeletal demineralization and promotes calcium deposition in the bone. This effect is the opposite of parathyroid hormone (PTH).
Juvenile Hypothyroidism
Hypothyroidism is one of the most common endocrine problems of childhood. It may be either congenital (see Chapter 8) or acquired and represents a deficiency in secretion of TH (Parks & Felner, 2016). Hypothyroidism from dietary insufficiency of iodine is now rare in the United States because iodized salt is a readily available source of the nutrient.
Beyond infancy, a number of defects may cause primary hypothyroidism. For example, a congenital hypoplastic thyroid gland may provide sufficient amounts of TH during the first year or two but be inadequate when rapid body growth increases demands on the gland. A partial or complete thyroidectomy for cancer or thyrotoxicosis can leave insufficient thyroid tissue to furnish hormones for body requirements. Radiotherapy for Hodgkin disease or other malignancies may lead to hypothyroidism (Metzger, Krasin, Choi, et al., 2016). Infectious processes may cause hypothyroidism. It can also occur when dietary iodine is deficient.
Low levels of circulating thyroid hormones and raised levels of TSH at birth characterize congenital hypothyroidism. If left untreated, congenital hypothyroidism causes mental retardation. Improvements in newborn screening have led to earlier detection and prevention of cognitive dysfunction (Hanley, Lord, & Bauer, 2016).
Thyromegaly (enlarged thyroid gland) and growth deceleration are seen in children with hypothyroidism. Growth and development are less impaired when hypothyroidism is acquired at a later age. Because brain growth is nearly complete by 2 to 3 years of age, intellectual disability and neurologic sequelae are not associated with juvenile hypothyroidism. Clinical manifestations may include dry skin, puffiness around the eyes, sparse hair, constipation, sleepiness, lethargy, and mental decline. Growth failure, delayed puberty, and excessive weight gain can also be seen.
Therapy is TH replacement, the same as for hypothyroidism in the infant. In children with severe symptoms, the restoration thyroid function is achieved more gradually with administration of increasing amounts of L-thyroxine over a period of 4 to 8 weeks. This is done to avoid symptoms of hyperthyroidism. Researchers have found that children treated early continue to have mild delays in reading, comprehension, and arithmetic but should catch up over time (Pardo Campos, Musso, Keselman, et al., 2017). Adolescents may demonstrate problems with memory, attention, and visuospatial processing.
Nursing Care Management
The importance of early recognition of congenital hypothyroidism in the infant is discussed in Chapter 8. Growth deceleration in a child whose growth has previously been normal should alert the observer to the possibility of hypothyroidism. Treatment is daily oral TH replacement. The importance of daily compliance and the need for periodic monitoring of serum thyroid levels should be stressed to patients and their families.
Goiter
A goiter is an enlargement or hypertrophy of the thyroid gland. It may occur with deficient (hypothyroid), excessive (hyperthyroid), or normal (euthyroid) TH secretion. It can be congenital or acquired. Congenital disease usually occurs as a result of maternal administration of antithyroid drugs or iodides during pregnancy. Acquired disease can result from increased secretion of pituitary TSH in response to decreased circulating levels of TH or from infiltrative neoplastic or inflammatory processes. In areas where dietary iodine (essential for TH production) is deficient, goiter can be endemic.
Enlargement of the thyroid gland may be mild and noticeable only when there is an increased demand for TH (e.g., during periods of rapid growth). Where iodine deficiency is severe, a large percentage of the population display goiters. Enlargement of the thyroid at birth can be sufficient to cause severe respiratory distress. Sporadic goiter is usually caused by lymphocytic thyroiditis, and intrinsic biochemical defects in synthesis of the hormones are associated with goiters. TH replacement is necessary to treat the hypothyroidism and reverse the TSH effect on the gland.
Nursing Care Management
Large goiters are identified by their obvious appearance. Smaller nodules may be evident only on palpation. Nurses in ambulatory settings need to be aware of the possibility of goiters and report such findings. Benign enlargement of the thyroid gland may occur during adolescence and should not be confused with pathologic states. Nodules are rarely caused by a cancerous tumor but always require evaluation. Include questions regarding exposure to radiation in the assessment.
Immediate surgery to remove part of the gland may be lifesaving in infants born with a goiter. When thyroid replacement is necessary, parents have the same needs regarding its administration as discussed for the parents of children who have hypothyroidism. (See Chapter 8.)
Chronic Lymphocytic Thyroiditis
Chronic lymphocytic thyroiditis (also known as Hashimoto disease) is the most common cause of thyroid disease in children and adolescents and accounts for the largest percentage of juvenile hypothyroidism (Hanley, Lord, & Bauer, 2016). It accounts for many of the enlarged thyroid glands formerly designated as thyroid hyperplasia of adolescence or adolescent goiter. Although lymphocytic thyroiditis can occur during the first 3 years of life, it occurs more frequently after age 6, with peak incidence occurring during adolescence. Some children may have subclinical hypothyroidism, but the presence of a goiter and elevated thyroglobulin antibody with progressive increase in both thyroid peroxidase antibody and TSH are predictive factors for development of overt hypothyroidism (Hanley, Lord, & Bauer, 2016).
Pathophysiology
There is a strong genetic predisposition to the development of lymphocytic thyroiditis. In families this disease is closely related to other thyroid disorders (e.g., Graves disease, idiopathic hypothyroidism, idiopathic myxedema) and autoimmune disorders (e.g., pernicious anemia, Addison disease, type 1 DM, and hypoparathyroidism).
Thyroid cell damage and death are autoimmune mediated by T cells and cytokines. Lymphocytes infiltrate the thyroid gland and cause inflammation. Eventually, thyroid cells are replaced with fibrous tissue. Several antithyroid antibodies have been recognized in patients with thyroiditis and these antibodies are the best marker to determine a diagnosis (Caturegli, De Remiqis, & Rose, 2014). The identification of genes involved in this disease has led to improved diagnostic testing and may lead to new treatments in the future (Tomer, 2014).
Clinical Manifestations
Enlargement of the thyroid gland is often noted during routine examination. Parents may notice it when the youngster swallows. In most children the entire gland is enlarged symmetrically (but may be asymmetric) and is firm, freely movable, and nontender. There may signs of moderate tracheal compression (sense of fullness, hoarseness, and dysphagia). However, it is extremely rare for a nontoxic diffuse goiter to enlarge enough to cause tracheal compression. Most children are euthyroid, but some display symptoms of hypothyroidism. Others have signs suggestive of hyperthyroidism, such as nervousness, irritability, tachycardia, increased sweating, or hyperactivity.
Diagnostic Evaluation
Thyroid function tests are usually normal, although TSH levels may be slightly or moderately elevated. With progressive disease the T4 decreases, followed by a decrease in T3 levels and an increase in TSH. A variety of abnormalities in radioactive iodine uptake may be noted. The majority of children have serum antibody titers to thyroid antigens, but fewer children have a positive red blood cell hemagglutination test result. When both tests are used, almost all children with thyroid autoimmunity are detected. However, levels in children are lower than in adults; therefore repeated measurements may be needed in questionable cases because titers may increase later in the disease.
Therapeutic Management
In many cases the goiter is transient and asymptomatic and regresses spontaneously within a year or two. Therapy of a nontoxic diffuse goiter is usually simple, uncomplicated, and effective. Oral administration of TH decreases the size of the gland significantly and provides the feedback needed to suppress TSH stimulation, and the hyperplastic thyroid gland gradually regresses in size. Surgery is contraindicated in this disorder. Evaluate untreated patients periodically.
Nursing Care Management
Nursing care consists of identifying the youngster with thyroid enlargement, reassuring the child that the condition is probably only temporary, and reinforcing instructions for thyroid therapy.
Hyperthyroidism
Graves disease (GD) is the most common cause of hyperthyroidism in children. This disease runs in families and is autoimmune. Most cases of GD in children occur in adolescence, with a peak incidence at 12 to 14 years of age. Transient GD may be present at birth in children of thyrotoxic mothers. The incidence is higher in girls than in boys (Leger & Carel, 2013).
The hyperthyroidism of GD is caused by the generation of autoantibodies to the TSH receptor. This leads to excess secretion of TH. Currently, there is no cure for GD. Choice of treatment continues to be debated among pediatric endocrinologists due to the risk of side effects associated with each option (Leger & Carel, 2013).
Clinical Manifestations
Signs and symptoms of hyperthyroidism develop gradually, with an interval between onset and diagnosis of approximately 6 to 12 months. Clinical features include irritability, hyperactivity, short attention span, tremors, insomnia, and emotional lability. Gradual weight loss despite a voracious appetite occurs in half the cases. Linear growth and bone age are usually accelerated. Muscle weakness often occurs. Hyperactivity of the gastrointestinal tract may cause vomiting and frequent stooling. Cardiac manifestations can include rapid pulse at rest, widened pulse pressure, systolic murmur, and cardiomegaly. Dyspnea may occur during slight exertion, such as climbing stairs. The skin is warm, flushed, and moist. Heat intolerance may be severe and is accompanied by diaphoresis. The hair is unusually fine and unable to hold a wave.
Exophthalmos (protruding eyeballs), observed in many children, is accompanied by a wide-eyed staring expression, increased blinking, lid lag, lack of convergence, and absence of wrinkling of the forehead when looking upward. If exophthalmos progresses, the eyelid may not completely cover the cornea. Visual disturbances may include blurred vision and loss of visual acuity. Eye disease associated with hyperthyroidism can develop before or after the clinical diagnosis.
Diagnostic Evaluation
The diagnosis of GD is established on the basis of increased levels of T4 and T3. TSH is suppressed to unmeasurable levels. Other tests are rarely indicated.
Therapeutic Management
Therapy for hyperthyroidism is controversial, but the end goal is the same: to decrease circulating TH. The three available treatments for children are antithyroid drugs, subtotal thyroidectomy, and ablation with radioiodine (131I iodide, RAI) (Hanley, Lord, & Bauer, 2016). Each therapy has advantages and disadvantages.
When affected children exhibit signs and symptoms of hyperthyroidism (e.g., increased weight loss, pulse pressure, and blood pressure), their activity should be limited to schoolwork only. Vigorous exercise is restricted until thyroid levels are decreased to normal or near-normal values. The American Thyroid Association* has an extensive website with information relation to prevention, treatment, and cure of thyroid disease.
Drug Therapy
Antithyroid drug (ATD) therapy with carbimazole, methimazole, or propylthiouracil interferes with the biosynthesis of TH. Most centers use this as first-line treatment for hyperthyroidism. Generally, some improvement is noted within the first 2 weeks, with evidence of decreased nervousness, less fatigue, increased strength, a lowered pulse, and weight gain. In many children an initial treatment course of 1 to 2 years is followed by a complete remission of the disorder. Others require 2 to 4 years before remission is achieved (Leger & Carel, 2013).
ATD disadvantages include drug reactions (e.g., rash, hives, joint pain, nausea, vomiting) and chronic dependency on the drug. Remission is not achieved in many patients, and alternative treatments are required. The most serious side effect of ATDs is agranulocytosis (severe leukopenia), which generally occurs within the initial weeks or months of therapy. It is usually accompanied by a sore throat and fever. Treatment involves immediate discontinuation of the drug and the administration of antibiotics and glucocorticoids until symptoms resolve.
Thyroidectomy
Surgical treatment involves surgical ablation of the thyroid (thyroidectomy). Although this approach has the advantage of being a long-lasting form of therapy, it has a number of serious disadvantages. Hypothyroidism occurs along with the need for thyroxine therapy. Infrequently, recurrent laryngeal nerve palsy, permanent hypoparathyroidism, keloid formation, and surgical morbidity and mortality do occur. Surgery in most centers is reserved for children who fail ATD therapy or who are prone to recurrence.
Radioiodine Therapy
Radioiodine may be a therapy of choice in young patients with GD who relapse after medical treatment (Hanley, Lord, & Bauer, 2016). Low-dose RAI is administered to children to treat hyperthyroidism without resulting in hypothyroidism. The relapse rate is high in this case and may require multiple doses throughout life. RAI should be avoided in very young children due to the increased risk of cancer. More research on RAI in children is needed due to concerns over its potential long-term side effects, including thyroid cancer, hyperparathyroidism, and high mortality rates (Leger & Carel, 2013).
Thyrotoxicosis
Thyrotoxicosis (thyroid “crisis” or thyroid “storm”) may occur from sudden release of TH. Although thyrotoxicosis is unusual in children, it can be life threatening. Clinical signs of thyroid storm are acute onset of severe irritability and restlessness, vomiting, diarrhea, hyperthermia, hypertension, severe tachycardia, and prostration. There may be rapid progression to delirium, coma, and death. A crisis may be precipitated by acute infection, surgical emergency, or discontinuation of antithyroid therapy. In addition to ATD therapy, the administration of beta blockers is used to control symptoms until normal thyroid function is achieved (Leger & Carel, 2013). Therapy is usually required for 2 to 3 weeks.
Nursing Care Management
Because the clinical manifestations of GD often appear gradually, the goiter and ophthalmic changes may not be noticed. Excessive activity may be attributed to behavioral problems. Nurses in ambulatory settings, particularly schools, need to be alert to signs that suggest this disorder. Weight loss despite an excellent appetite, inattention, hyperactivity, unexplained fatigue, sleepiness, and difficulty with fine motor skills may be seen. Exophthalmos, infrequent blinking, and impairment of convergence are common presenting signs (Huang & LaFranchi, 2016).
Nursing care focuses on treating physical symptoms before a response to drug therapy is achieved. These children need a quiet, unstimulating environment that is conducive to rest. (See Quality Patient Outcomes box.)
Until hyperthyroidism is controlled, symptoms often interfere with daily life. Emotional lability is often manifested by sudden episodes of crying or elation. Such behavior, coupled with irritability, disrupts interpersonal relationships, creating difficulties within and outside the home. The nurse can help parents understand the medical reason for behavior changes and offer ways to minimize them. A consultation with the child's teachers is important to provide education and suggest ways of helping the child adjust at school.
Heat intolerance may cause a child to dress differently. The use of light cotton clothing in the home, good ventilation, air conditioning or fans, frequent baths, and adequate hydration is helpful in providing comfort. Although the need for calories is increased, these should be provided in wholesome foods rather than “junk” foods.
Nurses should know the side effects of ATD therapy, including urticarial rash, fever, arthritis, or arthralgia. There may be enlargement of the salivary and cervical lymph glands, a diminished sense of taste, hepatitis, and edema of the lower extremities. Parents should also be aware of the signs of hypothyroidism, which can occur from overdose of the drugs. The most common indications are lethargy and somnolence.
Surgical Care
If surgery is anticipated, iodine is administered for a few weeks before the procedure. Because oral iodine preparations are unpalatable, they should be mixed with a strong-tasting fruit juice, such as grape or punch flavors, and be given through a straw. Compliance with iodine therapy is essential to avoid the danger of thyroid crisis after sudden discontinuation.
Psychologic preparation of children for thyroidectomy is similar to that for any other surgical procedure. (See Chapter 22.) However, the fear of having one's throat cut is unique to thyroidectomy. The nurse should explain that the throat is not cut, only the skin, to remove the gland. Showing children a picture of the anatomic location of the thyroid around the trachea is often helpful. Children should be prepared for the dressing around the neck and the possibility of an endotracheal or “breathing” tube after surgery.
Postoperative care involves positioning with the neck slightly flexed to avoid strain on the sutures and observation for bleeding and complications. The children learn to support the neck in this position when they sit up. Damage to the laryngeal nerve is evidenced by severe stridor or hoarseness, although some hoarseness is expected. Laryngospasm, a spasmodic contraction of the larynx, can be a life-threatening complication of thyroidectomy. Signs of laryngospasm are stridor, hoarseness, and a feeling of tightness in the throat. Place a tracheostomy set near the bed for emergency use. The nurse should observe for signs of hypoparathyroidism, which causes hypocalcemia, in the immediate postoperative period.
Disorders of Parathyroid Function
The parathyroid glands secrete parathyroid hormone (PTH). Along with vitamin D and calcitonin, PTH regulates the homeostasis of serum calcium concentrations (Lal & Clark, 2011). The effect of PTH on calcium is opposite that of calcitonin. Box 31.8 lists the principal effects of PTH on its target sites.
PTH and vitamin D work together to maintain serum calcium levels within a narrow normal range and mineralization of bone. Secretion of PTH is controlled by a negative feedback system involving the serum calcium ion concentration. Low ionized calcium levels stimulate PTH secretion, causing absorption of calcium by the target tissues; high ionized calcium concentrations suppress PTH.
Hypoparathyroidism
Hypoparathyroidism is a spectrum of disorders that result in deficient PTH. Congenital hypoparathyroidism may be caused by a specific defect in the synthesis or cellular processing of PTH or by aplasia or hypoplasia of the gland (Lal & Clark, 2011).
Hypoparathyroidism can occur secondary to other causes. Postoperative hypoparathyroidism may follow thyroidectomy with acute or gradual onset. It may be transient or permanent. Two forms of transient hypoparathyroidism may be present in the newborn, both of which are the result of a relative PTH deficiency. One type is caused by maternal hyperparathyroidism. A more common form appears almost exclusively in infants fed a milk formula with a high phosphate-to-calcium ratio.
Clinical Manifestations
Symptoms vary from none to significant morbidity if treatment is not initiated. Mild deficiency may be identified through laboratory studies. Muscle cramps are an early symptom, progressing to numbness, stiffness, and tingling in the hands and feet. A positive Chvostek or Trousseau sign or laryngeal spasms may be present. Convulsions with loss of consciousness may occur. These episodes may be preceded by abdominal discomfort, tonic rigidity, head retraction, and cyanosis. Headaches and vomiting with increased intracranial pressure and papilledema may occur and may suggest a brain tumor (Doyle, 2016a).
Children with long-standing hypoparathyroidism often have dry, scaly, coarse skin and horizontal lines in the nails. Mucocutaneous eruptions caused by Candida organisms are common (Doyle, 2016a). Dental and enamel hypoplasia often occurs. Cataracts develop in patients with untreated disease. Because hypoparathyroidism results in decreased bone resorption and inactive osteoclastic activity, skeletal remodeling and bone turnover are diminished. This may be the pathophysiology of poor growth that is associated with hypoparathyroidism in children (Waller, 2011).
Diagnostic Evaluation
The diagnosis of hypoparathyroidism is made on the basis of clinical manifestations associated with decreased serum calcium and increased serum phosphorus. Levels of plasma PTH are low in idiopathic hypoparathyroidism but high in pseudohypoparathyroidism. End-organ responsiveness is tested by the administration of PTH with measurement of urinary cyclic adenosine monophosphate. Kidney function tests are included in the differential diagnosis to rule out kidney disease. Although bone radiographs are usually normal, they may demonstrate increased bone density and suppressed growth.
Therapeutic Management
The objective of treatment is to maintain normal serum calcium and phosphate levels with minimum complications. Acute or severe tetany is corrected immediately by IV and oral administration of calcium gluconate and follow-up doses as necessary to achieve normal calcium levels. When diagnosis is confirmed, vitamin D therapy is begun. Vitamin D therapy is somewhat difficult to regulate because the drug has a prolonged onset and a long half-life. Some authorities advocate beginning with a lower dose with stepwise increases and careful monitoring of serum calcium until stable levels are achieved. Others prefer rapid induction with higher doses and rapid reduction to lower maintenance levels.
Long-term management administration of massive doses of vitamin D and oral calcium may be useful in maintaining adequate serum calcium levels. Blood calcium and phosphorus are monitored frequently until the levels have stabilized; they are then monitored monthly and less often until the child is seen at 6-month intervals. Renal function, blood pressure, and serum vitamin D levels are measured every 6 months. Serum magnesium levels are measured every 3 to 6 months to permit detection of hypomagnesemia, which may raise the requirement for vitamin D.
Nursing Care Management
The initial objective is recognition of hypocalcemia. Unexplained convulsions, irritability (especially to external stimuli), gastrointestinal symptoms (e.g., diarrhea, vomiting, cramping), and positive signs of tetany should lead the nurse to suspect this disorder. Initial nursing care includes institution of seizure and safety precautions and observation for signs of laryngospasm such as stridor, hoarseness, and a feeling of tightness in the throat. A tracheostomy set and injectable calcium gluconate should be located near the bedside for emergency use. The administration of calcium gluconate requires precautions against extravasation of the drug.
After initiation of treatment, the nurse discusses with the parents the need for continuous daily administration of calcium salts and vitamin D. Because vitamin D toxicity can be a serious consequence of therapy, parents should watch for signs that include weakness, fatigue, lassitude, headache, nausea, vomiting, and diarrhea. Polyuria, polydipsia, and nocturia are signs of early renal impairment.
Hyperparathyroidism
Hyperparathyroidism is rare in childhood but can be primary or secondary. The most common cause of primary hyperparathyroidism is adenoma of the gland (Doyle, 2016b). Primary hyperparathyroidism is rarely seen in children, but when it occurs, it is often due to a single parathyroid adenoma (Pashtan, Grogan, Kaplan, et al., 2013). The most common causes of secondary hyperparathyroidism are chronic renal disease, renal osteodystrophy, and congenital anomalies of the urinary tract. The common symptom of hyperparathyroidism is hypercalcemia. Box 31.9 lists the manifestations of hyperparathyroidism.
Diagnostic Evaluation
Blood studies to identify elevated calcium and decreased phosphorus levels are routinely performed. Measurement of PTH, as well as several tests to isolate the cause of the hypercalcemia, such as renal function studies, should be included. If parathyroid adenoma is suspected, imaging using ultrasound and a sestamibi nuclear substraction study are recommended (Pashtan, Grogan, Kaplan, et al., 2013). Other procedures used to substantiate the physiologic consequences of the disorder include electrocardiography and radiographic bone surveys.
Therapeutic Management
Treatment depends on the cause of hyperparathyroidism. The treatment of primary hyperparathyroidism is initially medication, but if this has no effect, surgical removal of the tumor or radioactive iodine is used (Huang & LaFranci, 2016). Parathyroidectomy may cause recurrent laryngeal nerve damage, voice impairment, and hypoparathyroidism, but the incidence of these complications is low if the surgeon has extensive experience (Huang & LaFranci, 2016). Treatment of secondary hyperparathyroidism is directed at the underlying contributing cause, which subsequently restores the serum calcium balance. However, in some instances, such as in chronic renal failure, the underlying disorder is irreversible. In this case treatment is aimed at raising serum calcium levels to inhibit the stimulatory effect of low levels on the parathyroid. This includes oral administration of calcium salts, high doses of vitamin D to enhance calcium absorption, a low-phosphorus diet, and administration of a phosphorus-mobilizing aluminum hydroxide to reduce phosphate absorption.
Nursing Care Management
The initial nursing objective is recognition of hyperparathyroidism. Because secondary hyperparathyroidism is a consequence of chronic renal failure, the nurse is always alert to signs that suggest this complication, especially bone pain and fractures. Because urinary symptoms are the earliest indication, assessment of other body systems for evidence of high calcium levels is indicated when polyuria and polydipsia coexist. Clues to the possibility of hyperparathyroidism include change in behavior, especially inactivity; unexplained gastrointestinal symptoms; and cardiac irregularities.
Much of the initial nursing care is related to the physical symptoms and prevention of complications. To minimize renal calculi formation, hydration is essential. Encourage the child to drink fruit juices that maintain a low urinary pH, such as cranberry or apple juice, because acidity of body fluids promotes calcium absorption. All urine should be strained for evidence of renal casts. Children with renal rickets (osteodystrophy) may wear braces to minimize skeletal deformities. These should be worn as prescribed. If the child is confined to bed, the nurse consults with the physical therapist regarding proper use of orthopedic appliances.
Vital signs are taken frequently, and the pulse should be counted for 1 full minute to detect irregularities. Report a decrease in pulse rate because it may signal severe bradycardia and cardiac arrest. The diet needs supervision to ensure compliance with low-phosphate foods, particularly dairy products.
If parathyroidectomy is anticipated, care is similar to that discussed for the child with hyperthyroidism. Because hypocalcemia is a potential complication, observing for signs of tetany, instituting seizure precautions, and having calcium gluconate available for emergency use are part of the nursing care.
Disorders of Adrenal Function
Adrenal Hormones
The adrenal glands consist of two distinct portions: the cortex, or outer section, and the medulla, or inner core. The adrenal cortex secretes hormones, called steroids, that are essential to life. The adrenal medulla, the inner core, produces the catecholamines epinephrine and norepinephrine. These three chemicals are also produced by the sympathetic nervous system, so if the adrenal supply fails, the body will continue to function.
Adrenal Cortex
The cortex secretes three groups of hormones that are classified according to their biologic activity: (1) glucocorticoids (cortisol, corticosterone), (2) mineralocorticoids (aldosterone), and (3) sex steroids (androgens, estrogens, and progestins). The glucocorticoids and mineralocorticoids affect metabolism and stress. The sex steroids influence sexual development but are not essential because the gonads secrete the major supply of these hormones.
Glucocorticoids
The most important glucocorticoids in humans are cortisol and corticosterone, the principal effects of which are listed in Box 31.10. Normally the hypothalamus secretes CRF, which causes the pituitary gland to produce ACTH, which stimulates the adrenal glands to produce glucocorticoids (primarily cortisol). Cortisol is the switch that controls this feedback. When blood levels of cortisol are low, the system turns on. When blood levels of cortisol rise, the system turns off.
In times of stress the anterior pituitary is stimulated by CRF from the hypothalamus, which causes the release of increased amounts of ACTH. Stressful stimuli capable of provoking this response include trauma; anesthesia; surgical intervention; sepsis; acute anoxia; hypothermia; hypoglycemia; and emotional states, especially panic, anxiety, or anger.
Body rhythms also regulate secretion of the glucocorticoids. Blood levels of cortisol demonstrate a typical diurnal or circadian pattern. In individuals who follow a regular routine of nighttime sleeping, cortisol levels are highest in the early morning hours before waking up and lowest in the evening hours before bedtime.
Mineralocorticoids
The most important mineralocorticoid is aldosterone. Like cortisol, it promotes sodium retention and potassium excretion in the renal tubules. Aldosterone is significantly more potent than that of the glucocorticoids in maintaining extracellular fluid volume, acid-base balance, and normal potassium levels.
Aldosterone synthesis is regulated primarily by the renin-angiotensin system of the kidney. The juxtaglomerular cells of the kidney respond to decreased arterial pressure or blood volume and to decreased sodium concentrations by secreting the enzyme renin into the blood. Renin in turn converts angiotensinogen to angiotensin I and then to angiotensin II. Increased levels of angiotensin stimulate the adrenal cortex to secrete aldosterone, which preserves sodium, thereby retaining water. The renin-angiotensin mechanism also results in increased blood pressure.
Sex Steroids
Except for the first few days of life, the sex hormones are normally secreted in only minimum amounts until adolescence, at which time they play a role in pubertal changes. Their actions are the same as those of the gonadal hormones on internal and external sexual structures and skeletal growth.
Adrenal Medulla
The adrenal medulla secretes the catecholamines epinephrine and norepinephrine. Both hormones have essentially the same effects on different organs as those caused by direct sympathetic stimulation, except the hormonal effects last several times longer. Their major actions are listed in Box 31.11.
Although the catecholamines evoke similar responses from target sites, there are some important differences. Epinephrine has a greater effect on cardiac activity than norepinephrine, but it causes only weak constriction of the blood vessels of muscles in comparison with the effect of norepinephrine. As a result, norepinephrine elevates blood pressure, whereas epinephrine increases cardiac output. Another important difference is their effect on metabolism. Epinephrine increases the metabolic rate to a much greater extent than norepinephrine. These differences in action have been attributed to the catecholamines' effects on α- or β-adrenergic receptors. Norepinephrine can only affect those effector cells that contain α-receptors, which are mostly excitatory (constriction and contraction). Epinephrine, however, can affect both α- and β-receptors, and β-receptors are mostly inhibitory (dilation and relaxation). Control of secretion of catecholamines, primarily in response to physiologic or emotional stress, is through the hypothalamus. Also, stimulation of the sympathetic nervous system results in the release of epinephrine and norepinephrine from the sympathetic nerves and adrenal medulla. Both systems support each other, and one can be substituted for the other. For this reason no condition is attributable to hypofunction of the adrenal medulla. Even in bilateral adrenalectomy, catecholamine replacement is not necessary because the sympathetic release of these chemicals is sufficient to meet all the physiologic functions required to cope with stressful events.
Pheochromocytoma is a rare tumor characterized by secretion of catecholamines. The tumor most commonly arises from the chromaffin cells of the adrenal medulla but may occur wherever these cells are found, such as along the paraganglia of the aorta or thoracolumbar sympathetic chain. In children, they are frequently bilateral or multiple and are generally benign. Often there is a familial transmission of the condition as an autosomal dominant trait (White, 2016a).
The clinical manifestations of pheochromocytoma are caused by an increased production of catecholamines, producing hypertension, tachycardia, headache, decreased gastrointestinal activity and resulting constipation, increased metabolism with anorexia, weight loss, hyperglycemia, polyuria, polydipsia, hyperventilation, nervousness, heat intolerance, and diaphoresis. In severe cases, signs of congestive heart failure are evident.
Acute Adrenocortical Insufficiency
The acute form of adrenocortical insufficiency (adrenal crisis) may have a number of causes during childhood. Although a rare disorder, some of the more common etiologic factors include hemorrhage into the gland from trauma, which may be caused by a prolonged, difficult labor and rapidly progressing infections, such as meningococcemia, which result in hemorrhage and necrosis (Waterhouse-Friderichsen syndrome). Abrupt withdrawal of exogenous sources of cortisone or failure to increase endogenous supplies during stressor congenital adrenogenital hyperplasia of the salt-losing type can also cause adrenal crisis.
Clinical Manifestations
Early symptoms of adrenocortical insufficiency include increased irritability, headache, diffuse abdominal pain, weakness, nausea and vomiting, and diarrhea. Generalized hemorrhagic manifestations are present in Waterhouse-Friderichsen syndrome. Abnormal serum electrolyte levels include hyponatremia and hyperkalemia. Fever increases as the condition worsens and is accompanied by signs of CNS involvement, such as nuchal rigidity, convulsions, stupor, and coma. The child is in a shocklike state with a weak, rapid pulse; decreased blood pressure; shallow respirations; cold, clammy skin; and cyanosis. Circulatory collapse is the terminal event.
In the newborn, adrenal crisis is accompanied by extreme hyperpyrexia (high temperature), tachypnea, cyanosis, and seizures. Usually there is no evidence of infection or purpura. However, hemorrhage into the adrenal gland may be evident as a palpable retroperitoneal mass.
Diagnostic Evaluation
There is no rapid, definitive test for confirmation of acute adrenocortical insufficiency. Samples for measurement of plasma cortisol and ACTH levels should be sent for analysis, but this is too time consuming to be practical for initial diagnosis. Therefore diagnosis is usually made based on clinical presentation, especially when hemorrhagic manifestations and signs of circulatory collapse despite adequate antibiotic therapy accompany a fulminating sepsis. Serum electrolytes can be helpful in narrowing the diagnosis as well. Because there is no real danger in administering a cortisol preparation for a short period, it is important to institute treatment immediately. Improvement with cortisol therapy confirms the diagnosis.
Therapeutic Management
Treatment involves replacement of cortisol, replacement of body fluids to combat dehydration and hypovolemia, administration of glucose solutions to correct hypoglycemia, and specific antibiotic therapy in the presence of infection. Initially, IV hydrocortisone (Solu-Cortef) is administered. Normal saline containing 5% glucose is given parenterally to replace lost fluid, electrolytes, and glucose. If hemorrhage has been severe, whole blood may be replaced. In the event that these measures do not reverse the circulatory collapse, vasopressors are used for immediate vasoconstriction and elevation of blood pressure.
After the child's condition has been stabilized, oral doses of cortisone, fluids, and salt are given, similar to the regimen used for chronic adrenal insufficiency. To maintain sodium retention, aldosterone is replaced by synthetic salt-retaining steroids.
Nursing Care Management
Because of the abrupt onset and potentially fatal outcome of this condition, prompt recognition is essential. Vital signs and blood pressure are taken every 15 minutes. Seizure precautions are instituted. The nurse should monitor the child's response to fluid and cortisol replacement. Rapid administration of fluids can precipitate cardiac failure and overdosage with cortisol may cause hypotension and a sudden fall in temperature.
When the acute phase is over and the hypovolemia has been corrected, the child is given oral fluids in small quantities. Rapid ingestion of oral fluids may induce vomiting, which increases dehydration. Therefore the nurse should plan a gradual schedule for reintroducing liquids. (See Quality Patient Outcomes box.)
The sudden, severe nature of this disorder necessitates a great deal of emotional support for the child and family. The child may be placed in an intensive care unit where the surroundings are strange and frightening. Despite the need for emergency intervention, the nurse must be sensitive to the family's psychologic needs and prepare them for each procedure, even if this is as brief as a statement such as “The IV infusion is necessary to replace fluid that the child is losing.” Because recovery within 24 hours is often dramatic, the nurse should keep the parents apprised of the child's condition, emphasizing signs of improvement, such as a lowered temperature and normal blood pressure.
If treatment needs to be continued past the acute stage, parents require the same preparation as in the case of children with chronic adrenal insufficiency. Preparation for discharge should begin as soon as possible after the child's condition has stabilized.
Chronic Adrenocortical Insufficiency (Addison Disease)
Chronic adrenocortical insufficiency is rare in children. Causes include infection, a destructive lesion of the adrenal gland, and autoimmune processes, but the cause may also be idiopathic. Because 90% of adrenal tissue must be nonfunctional before signs of insufficiency are manifested, onset of symptoms is often gradual. However, during periods of stress, when demands for additional cortisol are increased, symptoms of acute insufficiency may appear in a previously well child (Box 31.12).
Definitive diagnosis is based on measurements of functional cortisol reserve. The fasting serum cortisol and urinary 17-hydroxycorticosteroid levels are low and fail to rise, and plasma ACTH levels are elevated with corticotropin (ACTH) stimulation, the definitive test for the disease.
Therapeutic Management
Treatment involves replacement of glucocorticoids (cortisol) and mineralocorticoids (aldosterone). Some children are able to be maintained solely on oral supplements of cortisol (cortisone or hydrocortisone preparations) with a liberal intake of salt. During stressful situations (e.g., fever, infection, emotional upset, or surgery), the dosage must be tripled to accommodate the body's increased need for glucocorticoids. Failure to meet this requirement will precipitate an acute crisis. Overdosage produces appearance of cushingoid signs.
Children with more severe states of chronic adrenal insufficiency require mineralocorticoid replacement to maintain fluid and electrolyte balance. Other forms of therapy include monthly injections of desoxycorticosterone acetate or implantation of desoxycorticosterone acetate pellets subcutaneously every 9 to 12 months.
Nursing Care Management
After the disorder is diagnosed, parents need guidance concerning drug therapy. They must be aware of the continuous need for cortisol replacement. Sudden termination of the drug because of inadequate supplies or inability to ingest the oral form because of vomiting places the child in danger of an acute adrenal crisis. Parents should always have a spare supply of medication. Ideally, families will have a prefilled syringe of hydrocortisone and have training to administer this drug during a crisis. Unnecessary administration of cortisone will not harm the child, but if it is needed, it may be lifesaving. Any evidence of acute insufficiency is reported to the practitioner immediately.
Parents also need to be aware of side effects of the drugs. Undesirable side effects of cortisone include gastric irritation, which is minimized by ingestion with food or the use of an antacid; increased excitability and sleeplessness; weight gain, which may require dietary management to prevent obesity; and occasionally behavioral changes, including depression or euphoria. Parents should be aware of signs of overdose and report these to the practitioner. In addition, the drug has a bitter taste, which creates a challenge in its administration.
Because the body cannot supply endogenous sources of cortical hormones during times of stress, the home environment should be stable and relatively unstressful. Parents need to be aware that during periods of emotional or physical crisis, the child requires additional hormone replacement. The child should wear a medical identification bracelet, to notify medical personnel during emergency care.
Cushing Syndrome
Cushing syndrome is a characteristic group of manifestations caused by excessive circulating free cortisol. It can result from a variety of causes, which generally fall into one of five categories (Table 31.1).
TABLE 31.1
Cushing syndrome is uncommon in children. When seen, it is often caused by excessive or prolonged steroid therapy that produces a cushingoid appearance (Fig. 31.4). This condition is reversible after the steroids are gradually discontinued. Abrupt withdrawal will precipitate acute adrenal insufficiency. Gradual withdrawal of exogenous supplies is necessary to allow the anterior pituitary an opportunity to secrete increasing amounts of ACTH to stimulate the adrenals to produce cortisol.
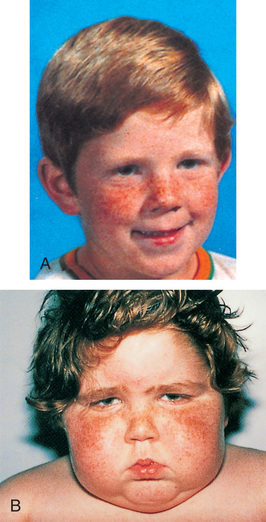
Clinical Manifestations
Because the actions of cortisol are widespread, clinical manifestations are equally profound and diverse. The symptoms that produce changes in physical appearance occur early in the disorder and are of considerable concern to school-age and older children (Fig. 31.5). The physiologic disturbances, such as hyperglycemia, susceptibility to infection, hypertension, and hypokalemia, may have life-threatening consequences unless recognized early and treated successfully. Children with short stature may be responding to increased cortisol levels, resulting in Cushing syndrome. Cortisol inhibits the action of GH.
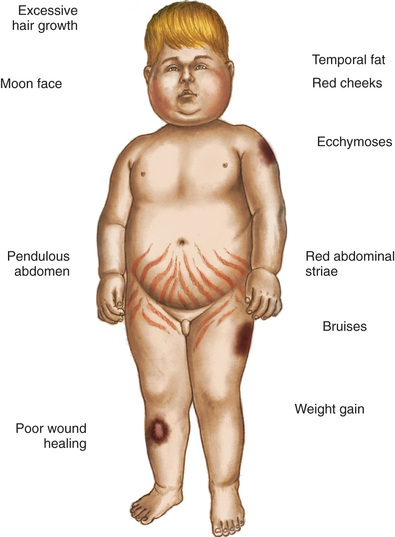
Diagnostic Evaluation
Several tests are helpful in confirming Cushing syndrome. Serum cortisol levels should be measured at midnight and in the morning along with corticotropin hormone, urinary free cortisol, fasting blood glucose levels for hyperglycemia, serum electrolyte levels for hypokalemia and alkalosis, and 24-hour urinary levels of elevated 17-hydroxycorticoids and 17-ketosteroids (Lowitz & Keil, 2015). Imaging of the pituitary and adrenal glands to assess for tumors, bone density studies for evidence of osteoporosis, and skull radiographs to determine enlargement of the sella turcica may also aid in the diagnosis. Another procedure used to establish a more definitive diagnosis is the dexamethasone (cortisone) suppression test (Ceccato & Boscaro, 2016). Administration of an exogenous supply of cortisone normally suppresses ACTH production. However, in individuals with Cushing syndrome, cortisol levels remain elevated. This test is helpful in differentiating between children who are obese and those who appear to have cushingoid features.
Therapeutic Management
Treatment depends on the cause. In most cases, surgical intervention involves bilateral adrenalectomy and postoperative replacement of the cortical hormones (the therapy for this is the same as that outlined for chronic adrenocortical insufficiency). If a pituitary tumor is found, surgical extirpation or irradiation may be chosen. In either of these instances, treatment of panhypopituitarism with replacement of GH, TH, ADH, gonadotropins, and steroids may be necessary for an indefinite period (Lau, Rutledge, & Aghi, 2015).
Nursing Care Management
Nursing care also depends on the cause. When cushingoid features are caused by steroid therapy, the effects may be lessened with administration of the drug early in the morning and on an alternate-day basis. Giving the drug early in the day maintains the normal diurnal pattern of cortisol secretion. If given during the evening, it is more likely to produce symptoms because endogenous cortisol levels are already low, and the additional supply exerts more pronounced effects. An alternate-day schedule allows the anterior pituitary an opportunity to maintain more normal hypothalamic–pituitary–adrenal control mechanisms.
If an organic cause is found, nursing care is related to the treatment regimen. Although a bilateral adrenalectomy permanently solves one condition, it reciprocally produces another syndrome. Before surgery, parents need to be adequately informed of the operative benefits and disadvantages. Postoperative teaching regarding drug replacement is the same as discussed in the previous section.
Anorexia and nausea and vomiting are common and may be improved with the use of nasogastric decompression. Muscle and joint pain may be severe, requiring use of analgesics. The psychologic depression can be profound and may not improve for months. Parents should be aware of the physiologic reasons behind these symptoms in order to be supportive of the child.
Congenital Adrenal Hyperplasia
Congenital adrenal hyperplasia (CAH) is a family of disorders caused by decreased enzyme activity required for cortisol production in the adrenal cortex. The adrenal gland produces excessive amounts of cortisol precursors and androgens to compensate. There are seven types of biochemical defects with the most common defect being 21-hydroxylase deficiency, which constitutes more than 90% of all cases of CAH (El-Maouche, Arlt, & Merke, 2017). This deficiency is an autosomal recessive disorder that results in improper steroid hormone synthesis (Mendes, Vaz Matos, Ribeiro, et al., 2015).
Clinical Manifestations
Excessive androgens cause masculinization of the urogenital system at approximately the tenth week of fetal development. The most pronounced abnormalities occur in girls, who are born with varying degrees of ambiguous genitalia. Masculinization of external genitalia causes the clitoris to enlarge so that it appears as a small phallus. Fusion of the labia produces a saclike structure resembling the scrotum without testes. However, no abnormal changes occur in the internal sexual organs, although the vaginal orifice is usually closed by the fused labia. The label ambiguous genitalia should be applied to any infant with hypospadias or micropenis and no palpable gonads, and a diagnostic evaluation for CAH should be contemplated (Carroll, Aron, Findling, et al., 2011).
Increased pigmentation of skin creases and genitalia caused by increased ACTH may be a subtle sign of adrenal insufficiency. A salt-wasting crisis frequently occurs, usually within the first few weeks of life (White, 2016b). Infants fail to gain weight, and hyponatremia and hyperkalemia may be significant. Cardiac arrest can occur.
Untreated CAH results in early sexual maturation, with enlargement of the external sexual organs; development of axillary, pubic, and facial hair; deepening of the voice; acne; and a marked increase in musculature with changes toward an adult male physique. However, in contrast to precocious puberty, breasts do not develop in girls, and they remain amenorrheic and infertile. In boys, the testes remain small, and spermatogenesis does not occur. In both sexes, linear growth is accelerated, and epiphyseal closure is premature, resulting in short stature by the end of puberty.
Diagnostic Evaluation
Clinical diagnosis is initially based on congenital abnormalities that lead to difficulty in assigning sex to the newborn and on signs and symptoms of adrenal insufficiency. Newborn screening is currently done in all 50 states by measurement of the cortisol precursor 17-hydroxyprogesterone. Definitive diagnosis is confirmed by evidence of increased 17-ketosteroid levels in most types of CAH (El-Maouche, Arlt, & Merke, 2017). In complete 21-hydroxylase deficiency, blood electrolytes demonstrate loss of sodium and chloride and elevation of potassium. In older children, bone age is advanced and linear growth is increased. DNA analysis for positive sex determination and to rule out any other genetic abnormality (e.g., Turner syndrome) is always done in any case of ambiguous genitalia.
Another test that can be used to visualize the presence of pelvic structures is ultrasonography, a noninvasive, painless imaging technique that does not require anesthesia or sedation. It is especially useful in CAH because it readily identifies the absence or presence of female reproductive organs or male testes in a newborn or child with ambiguous genitalia. Because ultrasonography yields immediate results, it has the advantage of determining the child's gender long before the more complex laboratory results for chromosome analysis or steroid levels are available.
Therapeutic Management
After diagnosis is confirmed, medical management includes administration of glucocorticoids to suppress the abnormally high secretions of ACTH and adrenal androgens. If cortisone is begun early enough, it is very effective. Cortisone depresses the secretion of ACTH by the anterior pituitary, which in turn inhibits the secretion of adrenocorticosteroids, which stems the progressive virilization. The signs and symptoms of masculinization in girls gradually disappear, and excessive early linear growth is slowed. Puberty occurs normally at the appropriate age.
The recommended oral dosage is divided to simulate the normal diurnal pattern of ACTH secretion. Because these children are unable to produce cortisol in response to stress, it is necessary to increase the dosage during episodes of infection, fever, surgery, or other stresses. Acute emergencies require immediate IV or intramuscular administration. Emergency situations include bacterial and viral infections, vomiting, surgery, fractures, major injuries, and sometimes insect stings.
Children with the salt-losing type of CAH require aldosterone replacement, as outlined under chronic adrenal insufficiency, and supplementary dietary salt. Frequent laboratory tests are conducted to assess the effects on electrolytes, hormonal profiles, and renin levels. The frequency of testing is individualized to the child.
Gender assignment and surgical intervention in the newborn with ambiguous genitalia is complex and controversial. It is a significant stress for families, who need support and education from a multidisciplinary team of experienced specialists. Factors that influence gender assignment include genetic diagnosis, genital appearance, surgical options, fertility, and family and cultural preferences. Generally, genetically female (46XX) infants should be raised as girls. Early reconstructive surgery should be considered only in the case of severe virilization (El-Maouche, Arlt, & Merke, 2017). Emphasis is on functional rather than cosmetic outcomes, and surgery can often be delayed. Reports concerning sexual satisfaction after partial clitoridectomy indicate that the capacity for orgasm and sexual gratification is not necessarily impaired. Male infants may require phallic reconstruction by an experienced surgeon.
Unfortunately, not all children with CAH are diagnosed at birth and raised in accordance with their genetic sex. Particularly in the case of affected females, masculinization of the external genitalia may have led to sex assignment as a male. In males, diagnosis is usually delayed until early childhood, when signs of virilism appear. In these situations, it is advisable to continue rearing the child as a male in accordance with assigned sex and phenotype. Hormone replacement may be required to permit linear growth and to initiate male pubertal changes. Surgery is usually indicated to remove the female organs and reconstruct the phallus for satisfactory sexual relations. These individuals are not fertile.
Nursing Care Management
Of major importance is recognition of ambiguous genitalia and diagnostic confirmation in newborns. Parents need assistance in understanding and accepting the condition and time to grieve for the loss of perfection in their newborn child. As soon as the sex is determined, parents should be informed of the findings and encouraged to choose an appropriate name, and the child should be identified as a male or female with no reference to ambiguous sex.
In general, rearing a genetically female child as a girl is preferred because of the success of surgical intervention and the satisfactory results with hormones in reversing virilism and providing a prospect of normal puberty and the ability to conceive. This is in contrast to the choice of rearing the child as a boy, in which case the child is sterile and may never be able to function satisfactorily in heterosexual relationships. If the parents persist in their decision to assign a male sex to a genetically female child, a psychologic consultation should be requested to explore their motivations and ensure their understanding of the future consequences for the child.
Nursing care management regarding cortisol and aldosterone replacement is the same as that discussed for chronic adrenocortical insufficiency. Because infants are especially prone to dehydration and salt-losing crises, parents need to be aware of signs of dehydration and the urgency of immediate medical intervention to stabilize the child's condition. Parents should have injectable hydrocortisone available and know how to prepare and administer the intramuscular injection (see Chapter 19). Parents, and later the child, need to understand that the medical regimen must be a lifelong commitment; therefore provide them with the education and counseling that is most likely to ensure informed and willing compliance.
In the unfortunate situation in which the sex is erroneously assigned and the correct sex determined later, parents need a great deal of help in understanding the reason for the incorrect sex identification and the options for sex reassignment or medical-surgical intervention.
Parents should be referred for genetic counseling before they conceive another child because CAH is an autosomal recessive disorder. Prenatal diagnosis and treatment are available.
Hyperaldosteronism
Excessive secretion of aldosterone may be caused by an adrenal tumor or, in some types of adrenogenital syndromes, result from enzymatic deficiency. The signs and symptoms are caused by increased sodium levels, water retention, and potassium loss. Hypervolemia causes hypertension and resultant headaches. Hypokalemia results in muscular weakness, paresthesia, episodes of paralysis, and tetany and may be responsible for polyuria and consequent polydipsia.
The clinical diagnosis is suspected when there are findings of hypertension, hypokalemia, and polyuria that fail to respond to ADH administration. Renin and angiotensin titers are abnormally low. Urinary levels of 17-hydroxycorticosteroids and 17-ketosteroids are normal in primary hyperaldosteronism caused by an aldosterone-secreting tumor but are usually abnormal in adrenogenital syndrome.
Therapeutic Management
Temporary treatment of the disorder involves replacement of potassium and administration of spironolactone (Aldactone), a diuretic that blocks the effects of aldosterone, thereby promoting excretion of sodium and water, while preserving potassium. Definitive treatment is similar to that for chronic adrenocortical insufficiency.
Nursing Care Management
An important nursing consideration is recognition of the syndrome, particularly in children with high blood pressure. Other clues include bed-wetting, excessive thirst, and unexplained weakness. After the diagnosis, nursing care is related to the treatment regimen. If diuretics are used, they should be administered in the morning to avoid accidents during the night. Children need unrestricted restroom privileges at school. Potassium supplements should be mixed with fruit juice such as grape juice to increase their acceptability, and potassium-rich foods should be encouraged. Parents need to be aware of the signs of hypokalemia and hyperkalemia. After an adrenalectomy, nursing care is similar to that for chronic adrenocortical insufficiency.
Pheochromocytoma
Pheochromocytoma is a rare tumor characterized by secretion of catecholamines. The tumor most commonly arises from the chromaffin cells of the adrenal medulla but may occur wherever these cells are found, such as along the paraganglia of the aorta or thoracolumbar sympathetic chain. In children, they are frequently bilateral or multiple and are generally benign. Often there is a familial transmission of the condition as an autosomal dominant trait (White, 2016a).
Clinical Manifestations
The clinical manifestations of pheochromocytoma are caused by an increased production of catecholamines, producing hypertension, tachycardia, headache, decreased gastrointestinal activity with resulting constipation, increased metabolism with anorexia, weight loss, hyperglycemia, polyuria, polydipsia, hyperventilation, nervousness, heat intolerance, and diaphoresis. In severe cases, signs of congestive heart failure are evident.
Diagnostic Evaluation
The clinical manifestations mimic those of other disorders, such as hyperthyroidism or DM. Tests specific to these conditions may be performed as part of the differential diagnosis. In a small number of instances a palpable tumor suggests the diagnosis. Usually the tumor is identified by CT scan or MRI. Definitive tests include 24-hour measurement of urinary levels of the catecholamine metabolites (Young, 2016).
Therapeutic Management
Definitive treatment consists of surgical removal of the tumor. In children, the tumors may be bilateral, requiring a bilateral adrenalectomy and lifelong glucocorticoid and mineralocorticoid therapy. The major complications that can occur during surgery are severe hypertension, tachyarrhythmias, and hypotension. The first two are caused by excessive release of catecholamines during manipulation of the tumor, and the latter results from catecholamine withdrawal and hypovolemic shock.
Preoperative medication to inhibit the effects of catecholamines is begun 1 to 3 weeks before surgery to prevent these complications. The major group of drugs used is the α-adrenergic blocking agents with or without β-adrenergic blocking agents. The most commonly used α-adrenergic blocker is phenoxybenzamine (Dibenzyline). To control catecholamine release after α-adrenergic blockage has been achieved, the child is given β-adrenergic blocking agents.
Success of therapy is judged by lowering of blood pressure to normal, absence of hypertensive attacks (e.g., flushing or blanching, fainting, headache, palpitations, tachycardia, nausea and vomiting, profuse sweating), heat tolerance, decrease in perspiration, and disappearance of hyperglycemia.
Nursing Care Management
An initial nursing objective is identification of children with this disorder. Children with hypertension and hypertensive attacks should be assessed for pheochromocytoma. Because of behavioral changes (nervousness, excitability, overactivity, and even psychosis), increased cardiac and respiratory activity may appear to be related to an acute anxiety attack. Therefore a careful history of the onset of symptoms and association with stressful events is helpful in distinguishing between an organic and a psychologic cause for the symptoms.
Preoperative nursing care involves frequent monitoring of vital signs and observation for evidence of hypertensive attacks and congestive heart failure. Therapeutic effects are evidenced by normal vital signs and absence of glycosuria. Note daily blood glucose levels, urine acetone, and any signs of hyperglycemia and report immediately.
The environment is made conducive to rest and free of emotional stress. This requires adequate preparation during hospital admission and before surgery. Parents are encouraged to room-in with their child and to participate in care. Play activities need to be tailored to the child's energy level without being overly strenuous or challenging because these can increase metabolic rate and promote frustration and anxiety.
After surgery, the child is observed for signs of shock from removal of excess catecholamines. If a bilateral adrenalectomy was performed, the nursing interventions are those discussed for chronic adrenocortical insufficiency.