Head Injuries
Tim Andrews
- • The term 'head injury’ is a broad classification that includes injuries to the face, scalp, skull or brain. Facial and scalp injuries can be painful and distressing but brain injuries have the greatest potential for mortality and morbidity.
- • Early recognition and management of traumatic brain injuries (TBIs) can reduce mortality and morbidity.
- • TBIs are usually the result of direct trauma to the head but can also occur as a result of severe acceleration or deceleration without head strike. TBI is the leading cause of death and disability in young adults (Shum, 2007).
- • Primary TBI occurs at the time of impact due to direct neuronal damage (Dunn et al., 2015) and describes the destruction of neurons and vascular structures by the mechanical forces of impact or deceleration (Atkinson & Wilberger, 2003). Secondary TBI evolves as a result of the body's inability to maintain normal brain perfusion following the primary injury. Inflammation, haemorrhage, oedema, blood pressure and hypoxaemia all contribute to the development of a secondary injury. Unlike the primary injury, the degree of secondary injury is subject to paramedic management.
- • TBIs are classified as mild (Glasgow Coma Scale score [GCS] 13–15), moderate (GCS 9–12) or severe (GCS < 9), but a GCS score obtained early in the clinical course is not a strong predictor of severity of injury (Reith et al., 2017).
- • National statistics for the incidence of TBI are lacking, but in New South Wales (NSW) the rates of all TBI are 99.1 : 100,000 (Pozzato et al., 2019), while in Victoria, incidence of severe TBI are 4.2 : 100,000 (Beck et al., 2016).
- • Overall mortality from TBI is 5.9 : 100,000 (Pozzato et al., 2019), while for severe TBI, there was a 42.5% rate of mortality (Beck et al., 2016).
- • Males outrate females in TBI at a rate of 2.9 : 1 (Pozzato et al., 2019).
- • Indigenous Australians suffer TBI at three times the rate of non-Indigenous Australians (Farrell & Dempsey, 2016), while in NSW, Indigenous Australians suffered TBI at a rate of 1.7 times that of the state populace (Pozzato et al., 2019).
- • The elderly are overrepresented for head injuries sustained from falls. Those aged > 65 years are overrepresented in both high and low (> 1 m or < 1 m) falls.
- • Head injury from child abuse is common and is estimated to represent up to 66% of TBI cases in children (Marx et al., 2017), and children of low socioeconomic status families are more likely to suffer head injuries than children of higher socioeconomic status families (Trefan et al., 2016).
- • While survival rates from TBI in Australia and New Zealand are not readily available as a national sample, in NSW TBI resulted in death 5.3% of the time (Pozzato et al., 2019), while that number increases to 42.5% of severe TBI resulting in death, and further increases to 78% death for severe TBI if the patient is over 64 years old (Beck et al., 2016).
- • Falls were the greatest cause of death when assessing all levels of head injury, with 67.9% of all deaths in NSW fall related (Pozzato et al., 2019). This means falls with low mechanism can result in death, especially in the over-65 age group.
- • The accepted signs of brain injury (unequal pupils, hypertension, bradycardia, irregular respirations) are very late signs of severe injury and as such do not act as a diagnostic tool for mild to moderate injuries that may subsequently evolve into more serious injuries.
Introduction
The term head injury usually refers to traumatic brain injury (TBI) but in a broader sense it can refer to any injury to structures above the neck, involving scalp and skull (Pushkarna et al., 2010). These injuries can be divided into two basic categories: those involving injuries to the brain (craniocerebral) and those involving injuries to the face or scalp (craniomaxillofacial). While facial injuries can be painful, confronting and dangerous, this chapter is primarily concerned with injuries where the force applied to the skull is transmitted through to the cranium and damages the soft tissues of the brain. The anatomical structure of the skull and brain are such that severe brain injuries can evolve from relatively minor events and with little sign of external injury. The ability to assess, triage and manage TBIs poses a number of clinical reasoning challenges and so this chapter focuses on these types of injuries.
Hospital management of TBIs has undergone significant changes in the past decade and these changes are now flowing out into the out-of-hospital environment. For example, rapid sequence intubation (RSI) by paramedics is now well established in several Australian states but requires significant training and support if the clinical benefits of this complex procedure are to be realised. Some of these recent changes to TBI management also contradict longstanding out-of-hospital practices and may be difficult to integrate without organisational support and ongoing training and education.
Statistics regarding the type and causes of head injuries may be limited or incomplete as they are not likely to include patients who die at the scene (or those who decline health services). However, in most developed countries there has been a decline in mortality and morbidity from TBIs over the past two decades due to a systematic approach to trauma care, which includes safety strategies such as seatbelt legislation, drink-driving legislation and bicycle helmet use (Cameron et al., 2014).
Anatomy
In order to understand the presentation and evolution of head and brain injuries, paramedics must have an understanding of the anatomy of the skull and the soft tissues that surround and are contained within it. Functionally and structurally, the skull can be divided into two distinct areas: the cranium and the face. The cranium can be described as a vault made up of flat bones that contain the brain: frontal, temporal, parietal, occipital, sphenoid and ethmoid (Mandavia et al., 2011; see Figs 34.1–34.4). The bones are 2–6 mm thick and are at their thinnest in the temporal region and at the base of the skull. Communication between the cranium and the rest of the body occurs through the foramina located beneath the cranium.
The bones that make up the face (maxilla, mandible, zygoma, lacrimal) attach to the anterior-inferior surface of the cranium and encompass the sensory organs and airways. Some bones such as the sphenoid are part of both the cranium and the face. The bones of the cranium are flat and thickened by a cancellous (spongy) middle layer where they are exposed to the external environment, while the bones of the face tend to be thinner, more delicate and complex.
Severe injuries to the face can pose issues of airway patency (obstruction from teeth, blood, etc.) or require specialist care (e.g. eyes, teeth, jaw). If the mechanism of injury is blunt force, paramedics should always consider the potential for brain injury, even when the primary presenting injury is to the face.
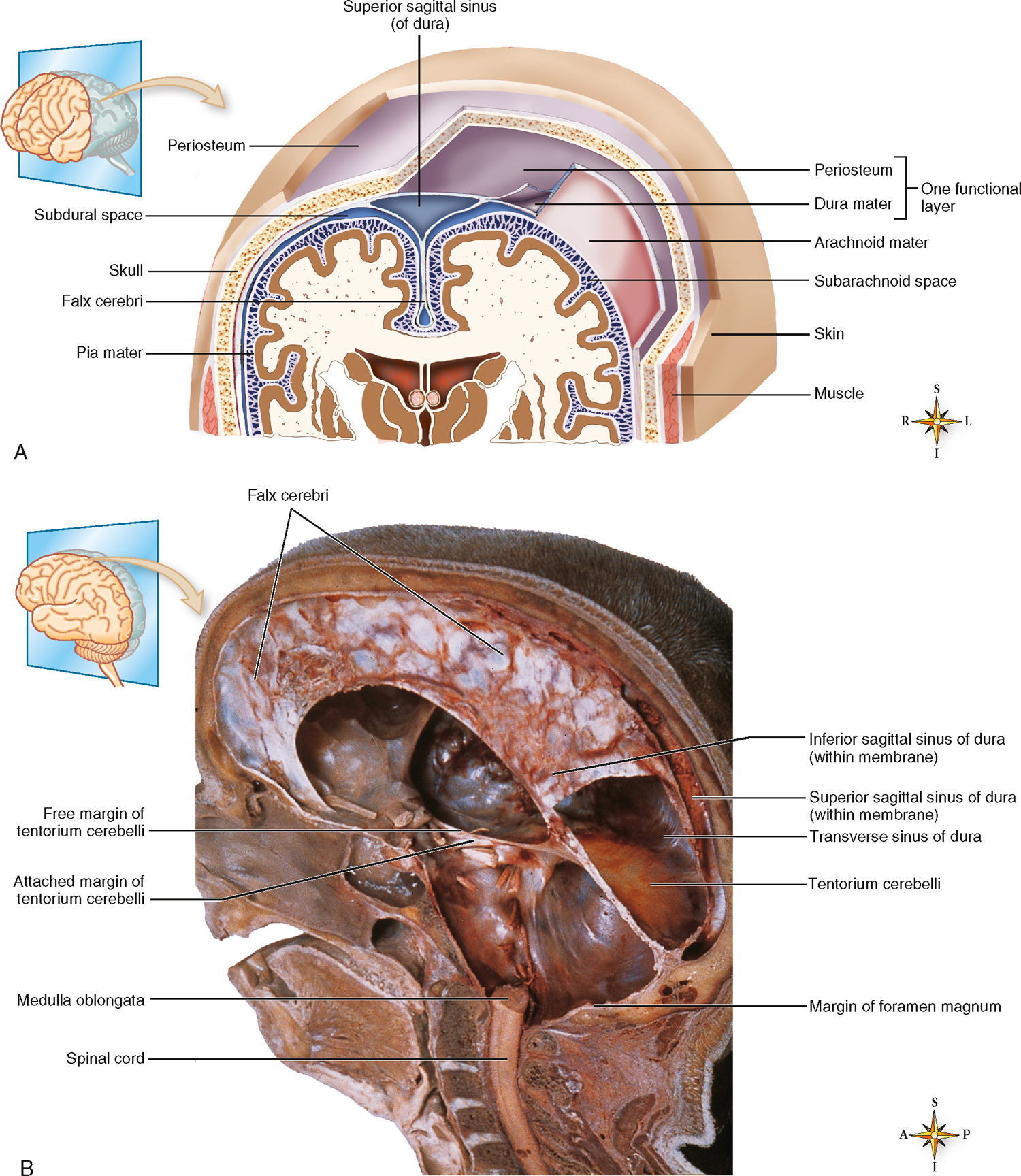
Essential to an understanding of both primary and secondary brain injuries is the knowledge that the bones of the cranium protect the brain in a structure that does not allow for expansion or contraction. Within the cranium three meningeal layers and cerebrospinal fluid (CSF) further protect the brain. Understanding the placement and structure of the meninges can provide clues to the type of injury a patient has suffered and how the injury may evolve.
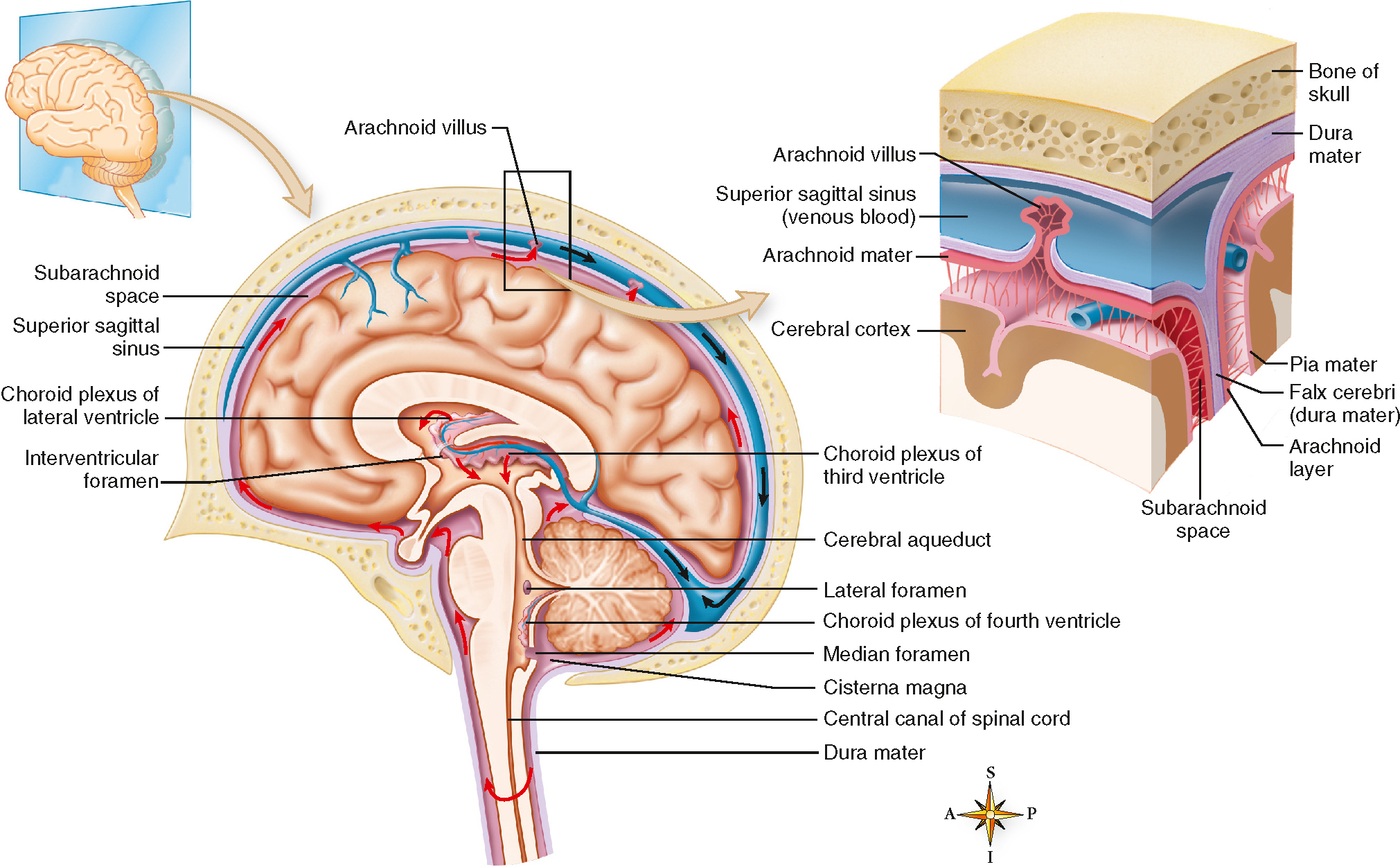
The innermost meningeal layer is the pia mater and it is attached to the external surface of the brain, the cerebral cortex (see Fig 34.5). The thin and transparent pia mater provides support to the network of blood vessels on the surface of the brain and follows the brain's surface into its sulci. Moving out, the delicate, web-like arachnoid mater is actually attached to the dura mater, the outermost meningeal layer. The space between the pia mater and the arachnoid mater is filled with CSF and is described as the subarachnoid space. The arachnoid mater actually extends small villi through the dura into the venous sinus to allow CSF to exit the subarachnoid space and return to the bloodstream. The dura mater (literally, tough mother) is a white, fibrous layer that is tightly bound to the inside of the skull bones. Importantly, the dura extends three strong and large folds between the right and left sides of the brain (falx cerebri), the right and left side of the cerebellum (falx cerebelli), and the cerebellum and the cerebral hemispheres (tentorium cerebelli). These folds anchor the brain within the skull as the head turns or nods. They also provide support for the thin-walled venous sinuses that drain blood from the brain, which is returned to the heart via the superior sagittal sinus. CSF is constantly being produced: it is exuded from capillaries lining the ventricles of the brain and returns to the bloodstream via the arachnoid villi. The normal amount of CSF in an adult is approximately 140 mL: 23 mL is contained in the ventricles of the brain and the remainder is contained in the subarachnoid space and the spinal cord (see Fig 34.6). Together, the meninges and CSF provide blood, hydration and physical protection to the brain.
Pathophysiology
Primary brain injury
Primary brain injury (PBI) occurs at the time of trauma and can be the result of direct impact, rapid acceleration/deceleration, penetrating injury or blast waves: in essence any mechanism where external forces or energy is transferred to brain tissue, resulting in injury (Abdelmalik et al., 2019). In most cases it is a result of direct trauma to the head and usually involves tissue damage to the scalp and skull. While the presence of scalp and skull injuries can indicate that PBI has occurred, the absence of external head injuries does not preclude PBI. The difficulty in detecting mild PBI in the field requires paramedics and other primary responders to understand the types of external and internal injuries that can occur as a result of direct head trauma. These include:
- • scalp lacerations
- • skull fractures
- • brain contusions
- • epidural haematomas
- • subdural haematomas
- • subarachnoid haemorrhage
- • intracerebral haemorrhage
- • diffuse axonal injury
- • penetrating head injuries.
Scalp lacerations
The scalp is tough, mobile and multilayered and is the thickest layer of skin in the body, with underlying subcutaneous tissue containing hair follicles and the rich blood supply for the scalp (Mandavia et al., 2011). It is highly vascular and the vessels fixed within the scalp are unable to retract and constrict when lacerated. This is why they bleed profusely and can result in haemorrhagic shock (Mandavia et al., 2011). Direct pressure is usually sufficient to control bleeding but the pressure may need to be prolonged. Close examination of any wound (visual and tactile) should indicate any underlying bony injury.
Linear fractures
Linear fractures (which appear straight in x-rays) make up 80% of all skull fractures (Mandavia et al., 2011). Motor vehicle crashes cause the majority of skull fractures, and 85% of these occur in males (Mandavia et al., 2011). Seatbelts, airbags and motorcycle helmets have contributed to a decrease in frequency of fractures. Other causes of linear fractures include falls and assaults. Provided they are not depressed or open, these fractures are not prone to complications, but they do indicate that significant force has been applied to the cranium, some of which will have been transmitted to the brain.
Depressed fractures
Depressed fractures describe the inward displacement of a fractured portion of bone (see Fig 34.7). This usually occurs when a large amount of force is applied to a small area (e.g. a blow with a hammer). On physical examination of the skull, the fracture is commonly not palpable because of swelling of the soft tissue. The displaced bone is likely to cause underlying brain tissue damage: one-third of depressed fractures cause a haematoma or tissue contusion. If the area of depression is small and not badly displaced, the actual amount of brain tissue damaged can be quite small: patients with this type of injury may not have any significant neurological signs soon after the injury. However, the total amount of force (or mechanism) will assist in determining whether a widespread brain injury is likely to have occurred.
Basilar fractures
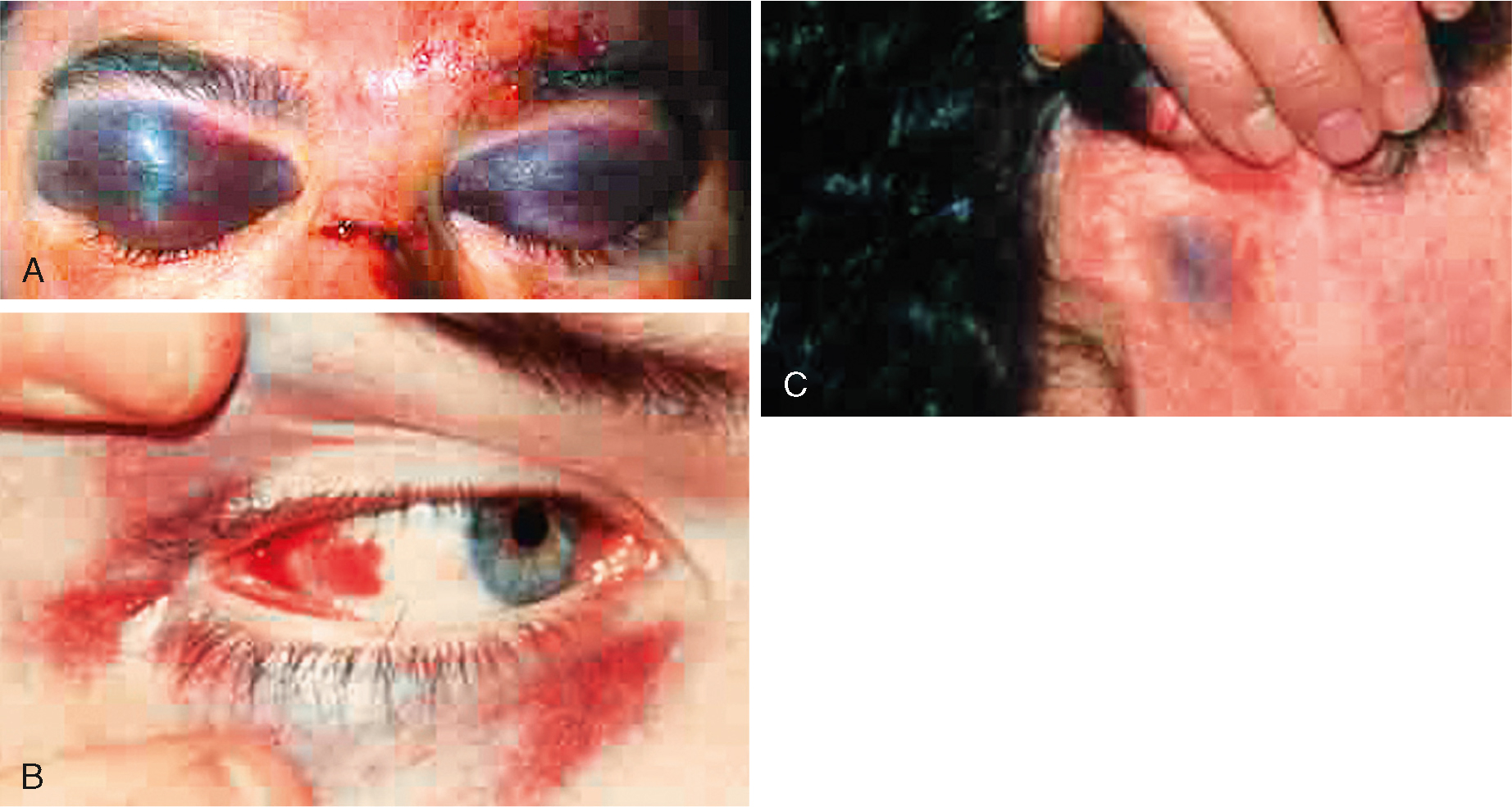
Basilar fractures to the base of the skull are usually associated with high mechanisms of injury and major trauma. They can occur because the mandible has been forced superiorly and posteriorly, fracturing the base of skull, or due to an upward force from the spinal column causing a linear fracture. These fractures are notoriously difficult to identify on x-ray and the diagnosis is often made clinically by the presence of CSF mixed with blood coming from the ears (haemotympanum), or ecchymosis beneath both eyes (raccoon eyes) or above the mastoid processes (Battle's sign; see Fig 34.8). CSF leaks are difficult to diagnose if the fluid is mixed with blood. The classically described 'bulls-eye’ sign, where after being absorbed by a cloth the CSF leaves a ring around the central blood stain, is not reliable and testing for CSF can be conducted at hospital. Treatment is generally conservative unless cranial nerve injury mandates surgical decompression. Signs of infection or meningitis are closely monitored. A persistent CSF leak beyond 2 weeks may require operative repair with a dural patch (Mandavia et al., 2011).
Open fractures
Open fractures describe wounds where there is direct communication between a scalp laceration and brain tissue. These are usually associated with significant mechanisms or injury (high-speed motor vehicle crashes, gunshot wounds) and a high mortality rate. Open fractures require surgical debridement and repair and in the field efforts should be made to limit further contamination of the wound.
Brain contusions
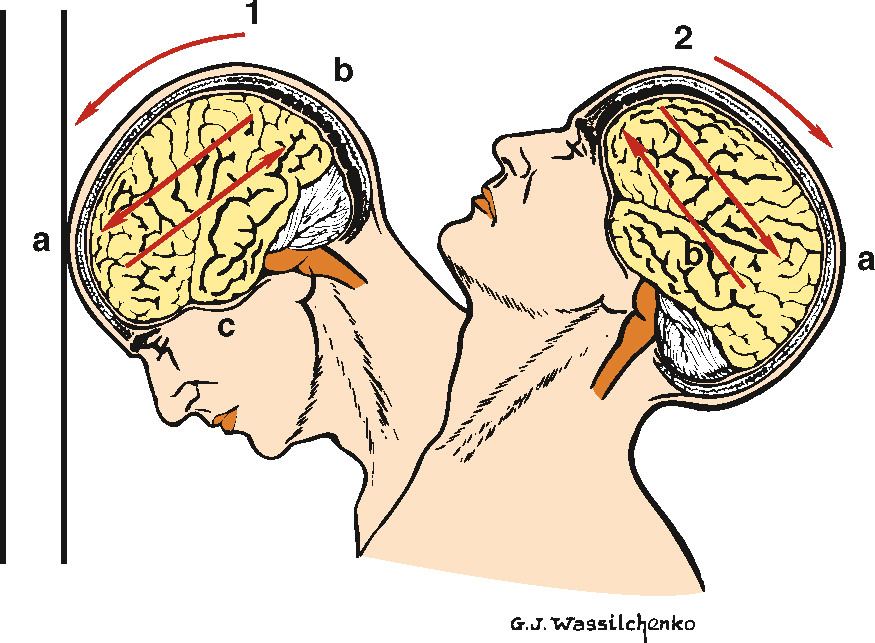
Cerebral contusion occurs when the brain parenchyma strikes fixed portions of the skull during acceleration or deceleration. The forces of compression or shearing can damage the cells and vasculature of the brain and effectively cause bruising of the brain tissue (see Fig 34.9).
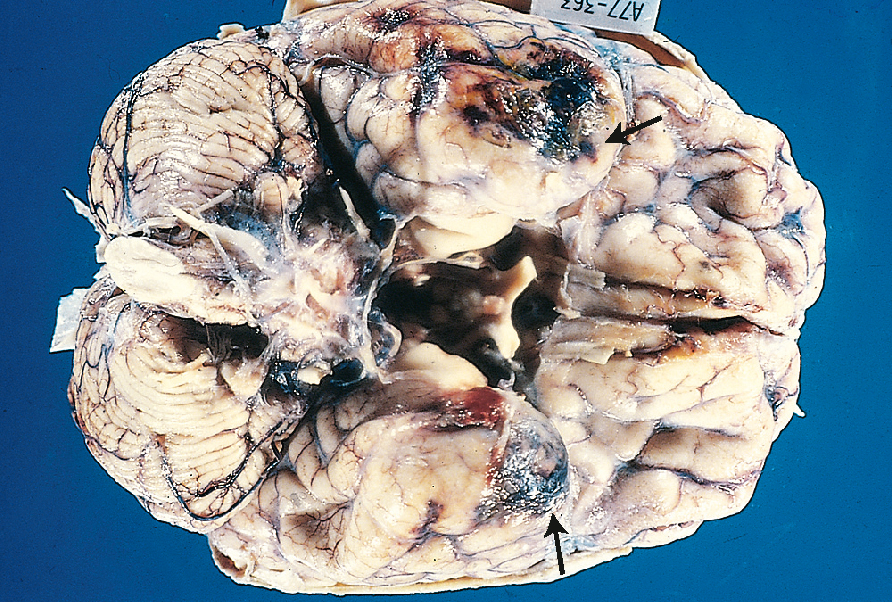
While the skull is smooth on the outer surface, within the cranial vault there are many protrusions and ridges, particularly on the base of the vault, which can generate high levels of compression or shearing when brain tissue is forced against them. Flexion and extension of the neck can result in contusions on the opposite side to where the force was originally applied (contrecoup; see Fig 34.10).
The frontal lobe striking the frontal bone and the temporal lobe striking the sphenoid bone are the two most common locations for cerebral contusions (Mandavia et al., 2011). Frontal lobe contusions are typically characterised by agitation, confusion, repetitive questioning, impaired short-term memory and aggressiveness that often requires physical and chemical restraint. These cerebral contusions, especially frontal or temporal, are often characterised by a high incidence of posttraumatic seizures and neurosurgeons often recommend routine prophylactic treatment with anticonvulsants for approximately 7 days post-injury (Mandavia et al., 2011).
Epidural haematomas
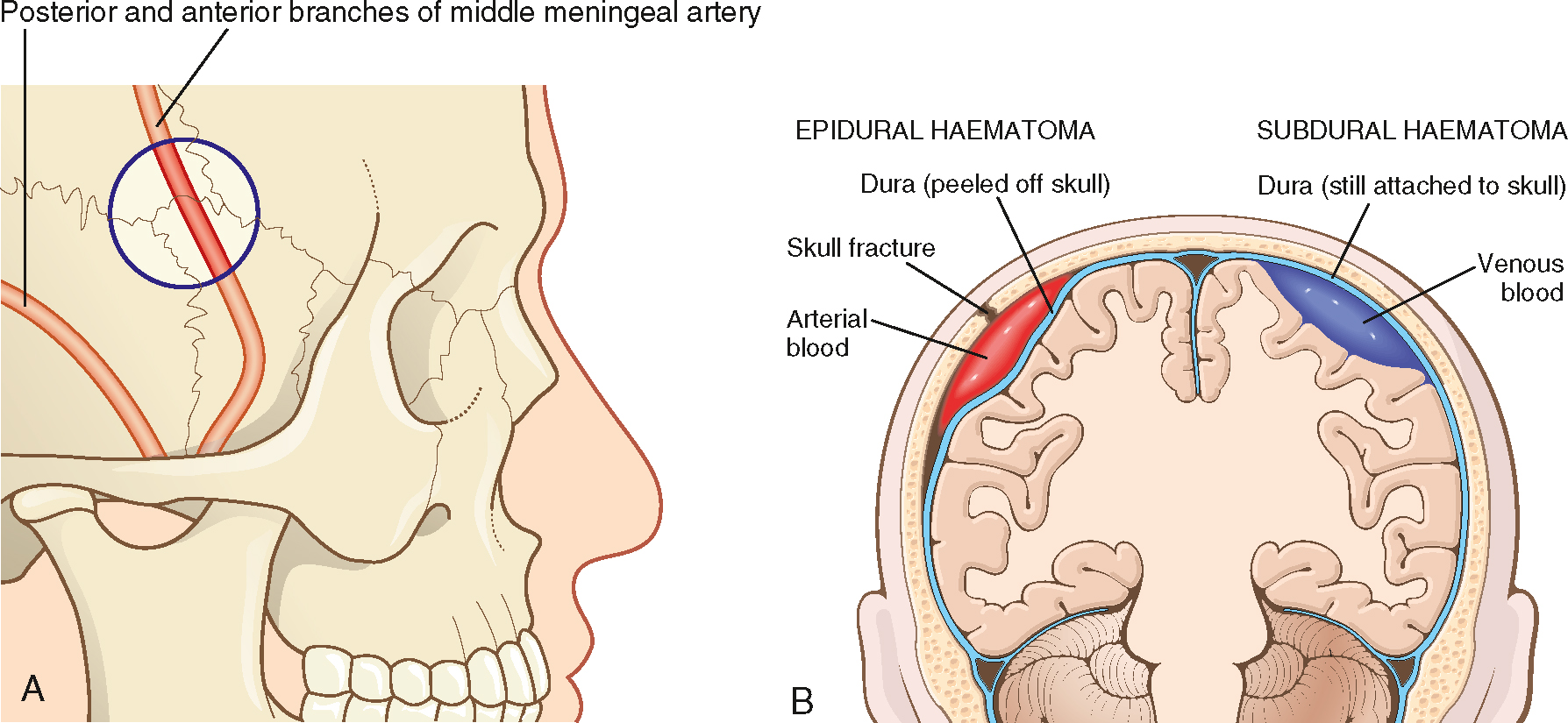
Epidural haematomas (EDHs) occur in about 2.7–4% of head injuries (Bullock et al., 2006a) and are characterised by blood collecting between the dura mater and the skull bone. EDH is most commonly due to the rupture of the middle meningeal artery, often the result of trauma to the skull (Bullock et al., 2006a).
In more than 90% of EDHs this occurs due to a fracture of the skull near the temple, as the pterion is the weakest part of the skull. Branches of the middle meningeal artery often lie in bony canals in this region and fracture will lead to rupture of the artery. These types of fractures most commonly occur following motor vehicle crashes, including with pedestrians, and sports injuries (Mandavia et al., 2011).
Because the dura is tightly adhered to the skull, EDHs can develop slowly as blood under systemic arterial pressure slowly forces its way between the dura and the bone (see Fig 34.11). EDHs represent the difficulties of assessing head injuries in the field: the force required to fracture the skull in this region is often sufficient to cause a brief loss of consciousness but is not sufficient to cause widespread primary injury. As a result, the patient will often have recovered from their initial loss of consciousness and be lucid upon presentation to clinicians; however, as the haematoma continues to increase in size, the patient's clinical progression will continue, with symptoms of headache, lethargy and confusion likely, as well as potentially seizure activity and hemiparesis, the result of an expanding haematoma compressing underlying brain tissue (see Table 34.1). The speed of this clinical progression can vary, depending on the blood vessel which has ruptured, and whether the bleed is arterial (faster progression) or venous (slower progression). However, this pattern of unconscious, lucid period and then unconscious is only reported in 47% of cases, and the overall mortality for EDH is around 10% (Bullock et al., 2006a).
Table 34.1
Comparison of intracranial injuries
| Type of patient | Anatomical location | CT findings | Common cause | Classic symptoms | |
| Epidural haematomas | Young, rare in elderly and those aged < 2 | Potential space between skull and dura mater | Biconvex football-shaped haematoma | Skull fracture with tear of middle meningeal artery | Immediate loss of consciousness, with lucid period prior to deterioration (20% of patients) |
| Subdural haematomas | Higher risk in elderly and alcoholic patients | Space between dura mater and arachnoid | Crescent or sickle-shaped haematoma | Acceleration/deceleration with tearing of the bridging veins | Acute: rapid loss of consciousness Chronic: altered mental state, gradual decrease in consciousness |
| Subarachnoid haematomas | Any age group after blunt trauma | Subarachnoid | Blood in the basilar cisterns, hemispheric sulci and fissures | Acceleration/deceleration with tearing of the subarachnoid vessels | Mild, moderate or severe injury and meningeal symptoms |
| Contusion/intracerebral haematoma | Any age group after blunt trauma | Usually anterior temporal or posterior frontal lobe | May be normal initially, delayed bleed | Severe or penetrating trauma, shaken baby syndrome | Symptoms range from normal to loss of consciousness |
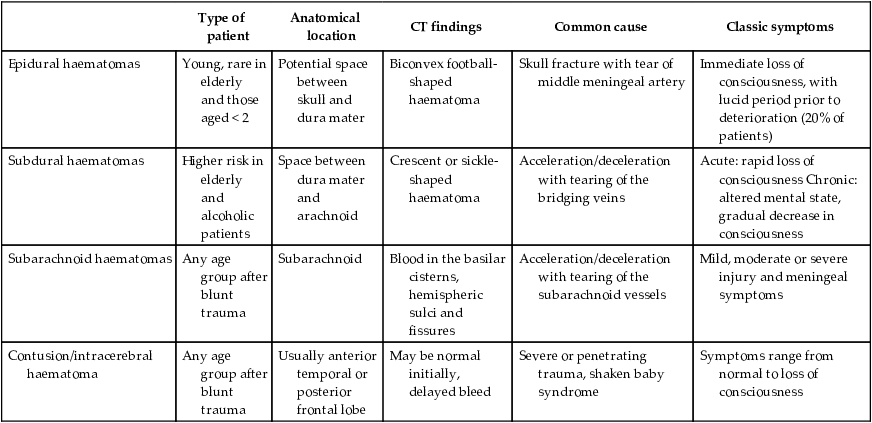
Source: Adapted from Tintinalli et al. (2016).
Definitive diagnosis is based on CT but the typical 'biconvex lens-like’ presentation may not occur early if the amount of bleeding is small (see Fig 34.12). In adults the haematoma will not cross suture lines because the dura is tightly adhered at the sutures, but this is not the case in children and the haematoma can extend across a wide area (Mandavia et al., 2011). Approximately 8% of EDHs are detected after a significant delay. Patients who are not in a coma at the time of presentation usually recover very well as there is often little underlying brain damage.
Subdural haematomas
Subdural haematoma (SDH) describes the presence of blood between the dura mater and the brain (see Fig 34.11). This most often occurs due to movement of the brain inside the skull tearing the thin-walled venous sinuses that cross this space. Deceleration with or without head strike is a common cause and those whose brain has decreased in size (e.g. due to age, alcoholism) are at greater risk as the degree of movement is potentially greater. Reports show that SDH occurs in approximately 21% of all head injuries and occurs in all age groups (Bullock et al., 2006b). Unlike EDH, skull fractures are not common with SDH. Symptoms depend on the size and location of the SDH, the rate of accumulation of blood, the degree of underlying brain injury and premorbid level of functioning. SDH is divided into three phases based on the time elapsed since injury.
- • Acute SDH: 0–24 hours (50–80% mortality)
- • Sub-acute SDH: 1–7 days (25% mortality)
- • Chronic SDH: > 7 days (20% mortality) (Mandavia et al., 2011)
On CT there may be a shift of cerebral structures across the midline and this degree of disruption can cause changes in mental state, headache, nausea and vomiting, and other symptoms of increased intracranial pressure (ICP). Because the haematoma forms under venous pressure, the development can be slow and may not present with symptoms for a number of days after the primary injury. Patients who are on anticoagulant medications are at higher risk (also because of age and increased likelihood of falls) and changes in mental state can be difficult to detect if confusion, disorientation, lethargy and depression are already present (Mandavia et al., 2011).
SDH can develop from head trauma that may have been relatively minor and forgotten by the patient. Treatment for large SDH in the acute or subacute phases is via craniotomy. Small haematomas are managed conservatively with close neurological observation. Decisions on chronic SDH depend on premorbid function, the size of the haematoma and the presence or absence of symptoms (Mandavia et al., 2011).
Subarachnoid haemorrhage
Subarachnoid haemorrhage (SAH) describes bleeding between the arachnoid membrane and the pia mater: the space normally occupied by CSF. This may occur spontaneously as a result of a cerebral aneurysm (see Ch 26) but is more commonly associated with head trauma (Parrillo & Dellinger, 2019). The vessels most often involved are those arteries that contribute to the circle of Willis at the base of the brain, but other vessels may be involved. Determining the relationship between SAH and trauma can be difficult as patients who suffer a spontaneous SAH and subsequent loss of consciousness while operating a vehicle are likely to present to paramedics and emergency responders following some sort of collision. Similarly, the altered level of consciousness following a collapse from a SAH can be difficult to separate from a headstrike unless the patient complained of symptoms prior to collapsing. Outcomes from SAH are dependent on the extent of the haemorrhage.
Intracerebral haemorrhage
The compressive and shearing forces that occur when the brain collides with the skull can cause bleeding in the brain parenchyma. Intracerebral haemorrhages describe collections of more than 5 mL of blood in the brain tissue. Symptoms depend on the size and location of the haematoma and also the cause. Low-velocity penetrating wounds (e.g. from knives) may produce a localised haematoma without significant surrounding tissue damage, while high-velocity wounds from gun shots can produce small haematomas with extensive surrounding tissue damage.
Diffuse axonal injury
Diffuse axonal injury (DAI) is the widespread damage to axons in the brain, particularly the brain neural tracts, corpus callosum and brainstem, and its diagnosis is an independent factor in increased mortality. DAI can exist without any external trauma being evident, and is more often associated with high-energy trauma such as traffic crashes or non-accidental injury in infants. Importantly, motorcyclists are at an increased risk of DAI because while the helmet worn may prevent visible skull damage, the acceleration/deceleration forces that occur during a motorcycle crash may lead to axonal injury. DAI can be clinically diagnosed by coma lasting greater than 6 hours, after excluding swelling or brain lesions, and it is commonly associated with loss of brain function and persistent vegetative states. Studies have shown that over time, return to normal, or near-normal, cognitive function can occur; however, 25% of patients with DAI will die. The globalised swelling and inflammation, caused by axonal damage, leads to increased ICP, which in turn can inhibit brain perfusion, which further worsens inflammation and swelling.
Penetrating head injuries
Penetrating head injuries involving gunshot wounds frequently result in death or profound disability. When a bullet enters the skull it produces multiple, high-velocity fragments from both bullet fragmentation and skull fragmentation (Mandavia et al., 2011). As the projectile enters the brain, it can produce a cavity up to four times larger than the bullet, transferring a large amount of kinetic energy which leads to the majority of brain destruction. The initial GCS can be a reliable predictor of outcome in gunshot wounds to the brain, with a GCS < 5 having a 100% mortality rate; however, patients with a GCS of 9 or higher and reactive pupils only have a mortality rate of 25% (Tintinalli et al., 2016).
Penetrating injuries from knives usually have less kinetic energy and therefore a better prognosis (Tintinalli et al., 2016). Small penetrating wounds can be missed because they are covered with matted hair, and blades may be broken off and embedded in the cranium. Patients with exposed brain matter, gunshot wounds that cross the midline of the brain, severe coagulopathy or early transtentorial herniation invariably die of their injuries but can still be considered as potential organ donors (Mandavia et al., 2011).
Concussion
The term 'concussion’ is often used to describe mild TBI. The traditional definition usually includes the phrases 'fully reversible brain injury’ and 'lack of abnormalities on radiological examination’. These criteria are now being challenged as the frequency of long-term symptoms from apparently minor brain injuries becomes more widely understood. The term 'post-concussion syndrome’ (PCS) is often applied to these patients (Brown & Edwards, 2014), but which symptoms are included, their severity and duration are not widely agreed upon (Rees, 2003).
Concussion typically refers to the disruption of higher cortical functions for a short period that resolves without treatment. The short loss or alteration of consciousness occurs when the forces acting to compress or shear brain tissue interrupt the function of the reticular activating system. This usually returns quickly but the widespread effects of the forces persist and develop.
The clinical indicators for concussion have been narrowed down to the following four criteria (Carney et al., 2014):
- 1. observed and documented disorientation and confusion immediately after the event
- 2. impaired balance within 1 day after injury
- 3. slower reaction time within 2 days after injury
- 4. impaired verbal learning and memory within two days after injury.
The pathophysiology of concussion is not unlike that of DAI; however, the severity is significantly reduced. Concussion is often the result of sporting injury, and the range of symptoms present can vary depending on the patient and the force which has occurred. Symptoms can include cognitive impairment, impairment of balance as well as emotional symptoms. This can result in clinical presentations such as:
- • delayed verbal responses or an inability to focus or maintain attention
- • headache, nausea and blurred vision
- • retrograde or anterograde amnesia, especially repeat questioning after the event
- • disproportionate emotional responses (crying over seemingly small issues)
- • seizure activity has been reported in only approximately 5% of all patients with concussion; of those 5%, it is estimated that 25% will have a seizure in the first hour post-injury and 50% will have a seizure within 24 hours (Evans & Whitlow, 2019).
The presence of these signs and symptoms is not definitive as there is still no definitive test for concussion; however, these clinical signs and symptoms will result in the patient being deemed likely to have sustained a concussion (Carney et al., 2014). Equally as important is to recognise possible signs and symptoms that do not correlate with concussion, including focal limb weakness, hemiparesis, pupillary abnormalities or Horner syndrome (Evans & Whitlow, 2019).
Adequate assessment and testing of patients with concussion is essential in the out-of-hospital setting, with a number of validated tests for concussion available. The majority of these tests rely on a baseline being established prior to the event, and while paramedics and other healthcare professionals may not have these baseline readings available to them, the format and symptoms assessed can help the clinician to perform repeatable and comparable tests to assess the patient's clinical trajectory.
- • Standardised Assessment for Concussion: This was developed to be a standardised tool for assessment of concussion, and assesses areas including orientation, immediate memory, concentration, delayed recall, neurological screening and exertional manoeuvres (McCrea et al., 1997).
- • Westmead Post-Traumatic Amnesia Scale: This test has been shown to be an easy, repeatable and rapid (reportedly less than 1 minute) test for concussion. It has a good correlation to more detailed neuropsychological testing and was found to be more valid than the GCS when predicting patients with concussion (Shores et al., 2008).
Secondary brain injury
The injury caused by PBI is determined by a number of factors including patient age and mechanism of injury. Clinicians, especially in the out-of-hospital setting, can affect the degree to which secondary brain injury (SBI) occurs and reduce SBI through effective management and goal-directed therapies. Following the initial impact injury, there are a number of unavoidable clinical and biochemical responses that, if not managed, can escalate a survivable primary injury into a fatal event. SBI should be considered an injury that evolves as a result of the body's inability to maintain normal brain function following the primary injury. Management of SBI relies on the maintenance of homeostasis, in particular maintaining ventilation and perfusion.
Avoid secondary brain injury by preventing the following:
- • hypocapnia or hypercapnia by maintaining CO2 levels within the range of 30–35 mmHg
- • hypoxia, maintaining SpO2 > 90%
- • hypotension—targeting blood pressures over 100 mmHg; similarly, exceedingly high blood pressures should also be avoided
- • hyperthermia—targeting normothermia and prevention of fevers
- • hypoglycaemia, ensuring blood glucose readings are > 4 mmol/L
- • seizure management—seizure activity is high in oxygen and glucose demand; prompt seizure management is essential.
Paramedics have a crucial role in the prevention of SBI. While procedures such as RSI show improved outcomes for patients with TBI (Bernard, 2006), timely and holistic care should be the cornerstone of all management.
Hypoxic-ischaemic brain injury
The brain is metabolically extremely active and, despite contributing less than 5% of total body mass, it requires 20% of the body's total oxygen requirements and 15% of total cardiac output (Tintinalli et al., 2016). Hypoxia (PaO2 < 40 mmHg) within the first 24 hours significantly increases the risk of death in patients with TBI (Ó Briain et al., 2018); therefore, maintenance of SpO2 > 90% is crucial.
Unlike muscle tissue, the integrity of brain cell membranes is limited and even a short period of poor perfusion can cause increased permeability, swelling (see Box 34.1) and apoptosis. Brain tissue is also unique because, being enclosed in a bony vault, any increase in volume within the skull causes a corresponding increase in pressure and a subsequent decrease in the perfusion of brain tissue.
The release of blood or cellular contents into the interstitial space as a result of the primary injury triggers a typical inflammatory response (see Ch 4). This results in vasodilation and tissue oedema at the site of the injury. This increase in cerebral volume increases the ICP and forces some of the CSF surrounding the brain out of the cranial vault. If the degree of swelling exceeds the amount of CSF that can be transferred out of the skull, pressure in the intracranial space will start to rise beyond the normal limit of 10–15 mmHg. Normal ICP is the pressure that blood must overcome if it is to enter the cranial vault and perfuse the brain. Cerebral blood flow is largely dependent on cerebral perfusion pressure (CPP), which can be calculated as mean arterial pressure (MAP) less ICP (Cameron et al., 2014).
The body's ability to autoregulate cerebral perfusion (by means of heart rate, vasoconstriction and vasodilation) usually ensures that the brain receives an adequate supply of blood, even when MAP ranges from 50 to 150 mmHg. To maintain CPP when ICP rises, MAP must also rise. In the case of severe head injury, the swelling that occurs (from bleeding and inflammation) after the primary injury may raise the ICP beyond the point at which the body can raise the MAP and maintain cerebral perfusion. Injured brain tissue and the release of inflammatory mediators can also limit the brain's ability to control local flow and perfusion can be inadequate despite extremely high systolic and diastolic pressures. Cerebral autoregulation has been shown to become impaired in one-third of all patients with a severe TBI (Rajajee, 2018a, 2018b), therefore maintaining systolic blood pressure over 100 mmHg for patients 50–69 years of age, and a systolic of > 110 mmHg for patients 15–49 years or over 70 years is recommended to ameliorate this possible impact on autoregulation (Carney et al., 2016). While future research may develop medications that limit the inflammatory response and/or the resistance to damage of individual brain cells, the only current strategy to limit secondary head injury is to maintain cerebral perfusion in the period after injury.
Although the mechanism of moderate to severe primary head injuries can vary, understanding the physiological stages that contribute to the secondary injury can assist paramedics in identifying patients who are likely to deteriorate or who are deteriorating. The potential for apnoea in the critical phase is the opportunity to stop a survivable injury progressing to a fatal event.
- • Critical phase. This commences at the time of injury and extends for up to 10 minutes. A traumatic loss of consciousness usually invokes some degree of apnoea (Atkinson & Wilberger, 2003). If this persists, cerebral hypoxia can become severe and generate a widespread inflammatory response regardless of the severity of the primary injury. A loss of consciousness can also allow for positional occlusion of the airway that will generate hypoxia even if the ventilatory reflex returns. Manually maintaining a patent airway and assisting ventilations during this period are essential to limit the evolution of a secondary injury. Cerebral hypoxia will generate a sympathetic response that increases both heart rate and blood pressure (see Ch 3), but the degree of sympathetic response in head injury exceeds that usually associated with hypoxia or hypercarbia and it is likely there is a direct mechanism between brain injury and the sudden release of catecholamines (Atkinson & Wilberger, 2003). The surge can cause blood pressure to rise sufficiently to cause neurogenic pulmonary oedema.
- • Exponential phase. Extending for up to 24 hours, the secondary injury becomes established during this phase as the inflammatory cascade provokes vasodilation and fluid to shift from the intravascular space. Tissue that was ischaemic from the primary injury becomes necrotic and previously uninjured tissue experiences ischaemia as the brain swells and ICP rises.
- • Plateau phase. After 24 hours ICP should be stabilised but may still be raised. While the acute inflammatory cascade has subsided, the necrotic tissue is still evolving and can cause further bleeding. Up to 75% of deaths from head injury in hospitalised patients occur during this phase.
- • Resolution phase. In this phase maximally swollen necrotic tissue begins to resolve but cerebral oedema is still present. There is a continued risk of intracerebral haemorrhage (Atkinson & Wilberger, 2003). This phase can extend for weeks but the long-term sequelae may persist for months or years.
It is vital at this point to reinforce the variability of head injuries. An isolated epidural haematoma under a linear fracture may appear dramatic and can restrict perfusion to a portion of the brain, but the amount of tissue damage may not be sufficient to cause the cascade of inflammatory mediators that leads to widespread brain swelling and extensive secondary injury. The factors that influence the development of secondary injury are highly individual, with the degree of injury, the location of bleeding and the extent of the inflammatory response all contributing.
The most important factor in reducing SBI is to maintain adequate perfusion and oxygenation: brain cells exposed to prolonged ischaemia will swell or rupture, contributing to both oedema and the inflammatory process. Even short periods of systemic hypoxia (PaO2, 40 mmHg) or hypotension (systolic BP, 100/110 mmHg) increase the likelihood of a poor outcome (Carney et al., 2016). The combination of both hypotension and hypoxia, for a little as 5 minutes, can result in poorer outcomes (Stein et al., 2010).
 ASSESS
ASSESS
The variety of mechanisms and patterns of head injury, the heterogeneity of responses and the unpredictable evolution of SBI create significant diagnostic challenges for paramedics when confronted with patients who have suffered a head injury. While the traditional division of mild, moderate and severe head injuries appears to offer a degree of initial classification, the diagnostic challenge for paramedics who encounter patients shortly after their primary injury is to identify which patients are likely to progress from mild to a more severe injury.
Patient history
Determining the mechanism of injury contextualises decision-making and planning in all traumatic injuries and should always be the starting point of the assessment. Obtaining a detailed history of the traumatic event is an ideal starting point when assessing patients with head injury. While the force of a fist or knee to the head in a game of football is not comparable to the large impact of a motor vehicle collision, the area of impact and immediate effects should be taken into consideration. Such an injury is unlikely to cause massive secondary injuries, but this should be considered a possibility in the minutes following the impact.
As detailed in the history, this patient suffered a brief loss of consciousness, which is indicative of a primary head injury, and based on his GCS of 14, his head injury can be categorised as mild (Evans & Whitlow, 2019). However, there are no keys as to whether or not this patient will develop an SBI based on mechanism or GCS alone.
Longer periods of loss of consciousness are associated with greater severity of injury but, importantly, a TBI can also occur without loss of consciousness. Brief tonic–clonic seizures ('concussive convulsions’) are also not uncommon immediately following loss of consciousness, but similarly there is a lack of evidence as to whether they are predictive of injury severity. Where a patient is involved in a contact sport there is the possibility that the current injury is not the only TBI to have occurred recently and this must be considered. Second impact syndrome (SIS) describes the sudden development of an SBI following a relatively minor impact in a patient whose brain is still recovering from an earlier injury (McCrory et al., 2012).
Posttraumatic amnesia or confusion can complicate gaining a specific and general medical history. Recent alcohol and drug use are also complicating factors, but any alteration in consciousness following trauma should be considered as a brain injury and not the result of alcohol or illicit drug use.
Neurological exam
Conscious state
A number of tools can be used to assess the patient's level of consciousness and severity of TBI. The GCS score (see Box 34.2 and Table 34.2) is by far the most common and it offers a widely understood tool that allows serial measurements of the patient's conscious state. The GCS is not without its limitations as it was originally designed for non-traumatic injuries, clinicians are notoriously poor at calculating scores and the three categories were never intended to be added together and are less accurate as a sum than when considered individually (Green, 2011). Despite the GCS authors advising against using the scale to predict severity or outcome of TBI (Teasdale & Jennett, 1978), it has become the standard tool to separate mild, moderate and severe TBI. A wide variety of clinical presentations occur with moderate head injury, but an important clinical scenario in the spectrum of moderate head injury is that of the 'talk and deteriorate’ patient. These patients have a GCS of ≥ 13 on presentation but deteriorate to a GCS of ≤ 8 within 48 hours (Marx et al., 2017). A GCS score < 9 is an indication for advanced airway management in some regions, but possibly the greatest utility of the GCS in the out-of-hospital setting is that it sets a reproducible baseline and a deterioration of 2 points or more should be considered significant (NZGG, 2006). Any GCS score should be considered a reliable indicator of TBI only after hypotension, hypoxia, hypoglycaemia and hypercapnia have been corrected.
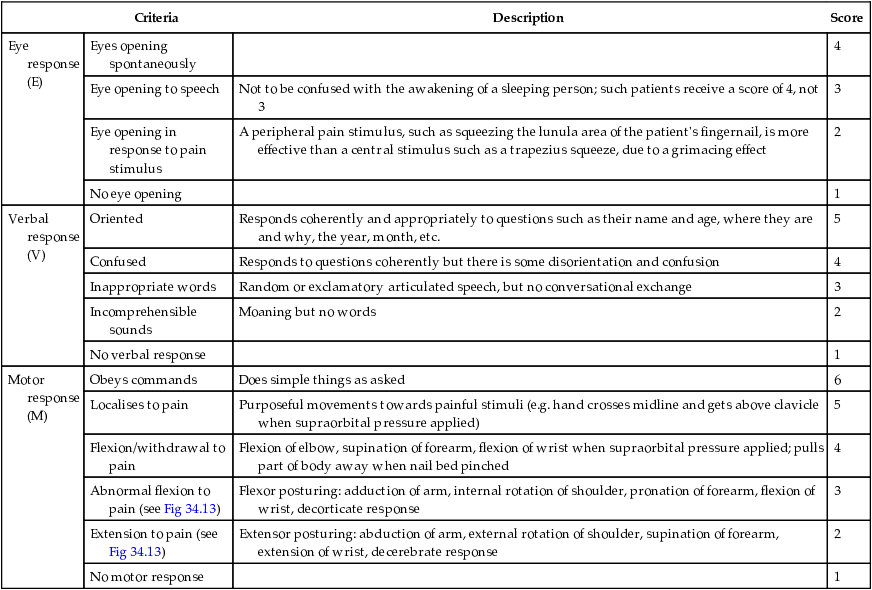
Table 34.2
| Criteria | Description | Score | |
| Eye response (E) | Eyes opening spontaneously | 4 | |
| Eye opening to speech | Not to be confused with the awakening of a sleeping person; such patients receive a score of 4, not 3 | 3 | |
| Eye opening in response to pain stimulus | A peripheral pain stimulus, such as squeezing the lunula area of the patient's fingernail, is more effective than a central stimulus such as a trapezius squeeze, due to a grimacing effect | 2 | |
| No eye opening | 1 | ||
| Verbal response (V) | Oriented | Responds coherently and appropriately to questions such as their name and age, where they are and why, the year, month, etc. | 5 |
| Confused | Responds to questions coherently but there is some disorientation and confusion | 4 | |
| Inappropriate words | Random or exclamatory articulated speech, but no conversational exchange | 3 | |
| Incomprehensible sounds | Moaning but no words | 2 | |
| No verbal response | 1 | ||
| Motor response (M) | Obeys commands | Does simple things as asked | 6 |
| Localises to pain | Purposeful movements towards painful stimuli (e.g. hand crosses midline and gets above clavicle when supraorbital pressure applied) | 5 | |
| Flexion/withdrawal to pain | Flexion of elbow, supination of forearm, flexion of wrist when supraorbital pressure applied; pulls part of body away when nail bed pinched | 4 | |
| Abnormal flexion to pain (see Fig 34.13) | Flexor posturing: adduction of arm, internal rotation of shoulder, pronation of forearm, flexion of wrist, decorticate response | 3 | |
| Extension to pain (see Fig 34.13) | Extensor posturing: abduction of arm, external rotation of shoulder, supination of forearm, extension of wrist, decerebrate response | 2 | |
| No motor response | 1 |
There are a number of validated tests for concussion available that aren't simply the GCS. The majority of these tests rely on a baseline being established prior to the event, and while paramedics and other healthcare professionals may not have these baseline readings available to them, the format and symptoms assessed can help the clinician to perform repeatable and comparable tests to assess the patient's clinical trajectory.
A negative result on any one of the following questions is considered a positive test for cognitive impairment after a head injury.
- • What is your name?
- • What is the name of this place?
- • Why are you here?
- • What month are we in?
- • What year are we in?
- • What town/suburb are you in?
- • How old are you?
- • What is your date of birth?
- • What time of day is it (morning/afternoon/night)?
Posttraumatic amnesia
An inability to recall events leading up to a head injury is referred to as retrograde amnesia, while memory loss following the event is differentiated as antegrade amnesia. The presence of these symptoms is important to ascertain during patient assessment and can be assessed easily through standardised, repeatable questioning. These symptoms may also be self-evident by the patient repeatedly asking the same questions of healthcare clinicians.
Cranial nerve assessment
A neurological examination of a patient with TBI requires the assessment of cranial nerve function; particularly nerves III to VII (Evans & Whitlow, 2019). Changes to pupil size, reactivity and response to light are all signs of TBI (Tintinalli et al., 2016) but they are a result of significant brain swelling and rarely occur in patients with a high GCS. As such they are rarely predictive of a developing injury, but can indicate TBI in a patient who is found unconscious. While drugs can influence pupil presentation, this usually occurs symmetrically and one-sided changes in an unconscious patient who is not hypoxic, hypotensive or hypoglycaemic strongly suggest TBI.
While there is no out-of-hospital clinical intervention derived directly from pupil asymmetry, early assessment against normal values (see Table 34.3) will act as a baseline for deterioration. In an unresponsive patient, a single fixed and dilated pupil may indicate an intracranial haematoma with uncal herniation that requires rapid operative decompression in hospital (Tintinalli et al., 2016). Pupillary asymmetry, loss of the light reflex or a dilated pupil suggests herniation of the brain. Before they become fixed and dilated, pupils may take on an ovaloid appearance as a result of compression of the third cranial nerve. Bilateral fixed and dilated pupils suggest very high ICP with poor brain perfusion and possibly bilateral uncal herniation (Tintinalli et al., 2016). Checking of the pupils also provides the opportunity to detect subconjunctival haemorrhage, periorbital oedema and ocular movements: all potential indicators of orbital or basilar fractures. Like all clinical assessments, periodically re-assessing pupillary response is important.
Table 34.3
| Asymmetrical pupils | Differ more than 1 mm in size |
| Dilated pupils | Greater than or equal to 4 mm in adults |
| Fixed pupils | Less than 1 mm change in response to bright light |
Assessment of oculovestibular (cold caloric) and ocular cephalic (doll's eyes) responses should not occur until the cervical spine is fully cleared of injury and this will not happen at the scene if the patient is unconscious.
Assessment of facial muscles (cranial nerves V and VII) is also important in patients with a head injury. Facial nerve palsy (VII) is the most commonly affected nerve in blunt trauma (Cools & Carneiro, 2018), and assessment of these nerves includes face sensation and movement.
Physical exam
Although the signs of a basilar fracture may not appear for hours after an injury they should be checked, at least to set a baseline. Widening the examination of the face, checking for swelling, pain and misalignment of teeth, can indicate facial fractures (see Fig 34.14). Similarly, examining the scalp for lacerations or fractures should be mandatory.
In addition to vital signs, a blood glucose level measurement should be standard for all patients presenting in an altered conscious state.
Initial assessment summary
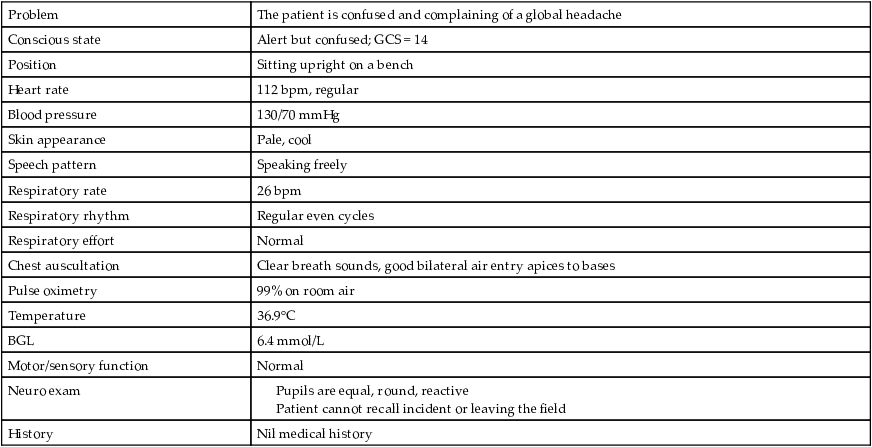
| Problem | The patient is confused and complaining of a global headache |
| Conscious state | Alert but confused; GCS = 14 |
| Position | Sitting upright on a bench |
| Heart rate | 112 bpm, regular |
| Blood pressure | 130/70 mmHg |
| Skin appearance | Pale, cool |
| Speech pattern | Speaking freely |
| Respiratory rate | 26 bpm |
| Respiratory rhythm | Regular even cycles |
| Respiratory effort | Normal |
| Chest auscultation | Clear breath sounds, good bilateral air entry apices to bases |
| Pulse oximetry | 99% on room air |
| Temperature | 36.9°C |
| BGL | 6.4 mmol/L |
| Motor/sensory function | Normal |
| Neuro exam | |
| History | Nil medical history |
D: There is no danger to the patient or crew.
A: The patient is conscious with no current airway obstruction.
B: Respiratory function is currently normal.
C: Heart rate is slightly elevated but consistent with recent activity. There is a sufficient blood pressure.
 CONFIRM
CONFIRM
The essential part of the clinical reasoning process is to seek to confirm your initial hypothesis by finding clinical signs that should occur with your provisional diagnosis. You should also seek to challenge your diagnosis by exploring findings that do not fit your hypothesis: don't just ignore them because they don't fit.
Mild TBI is typically described as a GCS of ≥ 13 following head trauma that may or may not include a brief loss of consciousness (see Table 34.4). While this patient clearly fits that category, paramedics are often required to assess patients only minutes after the trauma and before a secondary injury has time to evolve.
Table 34.4
Signs and symptoms associated with mild TBI
| Cognitive symptoms | Physical signs and symptoms | Behavioural changes |
| Attention difficulties | Headaches | Irritability |
| Concentration problems | Dizziness | Depression |
| Amnesia and perseveration | Insomnia | Anxiety |
| Short-term and long-term memory problems | Fatigue | Sleep disturbances |
| Orientation problems | Uneven gait | Emotional lability |
| Altered processing speed | Nausea, vomiting | Loss of initiative |
| Altered reaction time | Blurred vision | Loneliness and helplessness |
| Calculation difficulties and problems with executive function | Seizures | Problems related to job, relationship, home or school management |
Source: Tintinalli et al. (2016).
In reality a significant range of presentations fit within the mild category and they can assist in determining the severity and likelihood of an evolving secondary injury. Patients who present with a GCS of ≥ 13 usually lose points due to either confusion and/or drowsiness (i.e. the motor responses are rarely altered). One of the limitations of the GCS is that there are degrees of both drowsiness and confusion but they are all scored equally. A patient who quickly opens their eyes to voice and then interacts (even if confused) is neurologically quite different from a patient who requires a firm direction to open their eyes and then returns quickly to a ‘sleeping’ presentation. Patients who are persistently drowsy may fit the mild category but should be treated with suspicion and, depending on local transport times and resources, it can be worth treating them as if they fit the moderate category. The ‘disproportionate’ step between a GCS of 13 and a GCS of 14 is reflected in a number of agencies expanding the moderate criteria to include a GCS of 13.
The presence of a persistent global headache in this patient is a reason for concern and if any other neurological signs such as vomiting or photophobia were to develop they should be considered as deterioration, even if the GCS score remains unchanged (see Table 34.5).
Table 34.5
Criteria for classifying the severity of TBI
| Definitions of the severity of TBI vary across organisations and the literature. The most commonly used measures are the GCS and any loss of consciousness. | |||
| Severity of TBI | GCS on initial assessment | Duration of posttraumatic amnesia | Loss of consciousness |
| Mild | 13–15 | < 24 hours | Nil |
| Moderate | 9–12 | 1–6 days | Brief (< 5 min) |
| Severe | 3–8 | > 7 days | > 5 min |

Source: Reith et al., 2017
In this case the patient is not drowsy, is talking freely and shows no signs of agitation. For the moment the condition can be described as mild TBI.
 TREAT
TREAT
Emergency management
Principles of management for mild TBI are outlined in Box 34.3.
Cervical spine care
Cervical spine injury may be associated with blunt head trauma, especially when high forces are involved, but it is rare for spinal injuries to present asymptomatically following trauma (Sanchez et al., 2005). In many ambulance services, cervical spine immobilisation using a stiff collar remains a routine component of managing any head injury. However, questions regarding both the efficacy and the safety of cervical collars with and without full spinal immobilisation (Sundstrøm et al., 2013; Theodore et al., 2013) have led to a number of services introducing spinal injury clearance procedures to reduce the routine application of collars to asymptomatic patients (see Box 34.4). In this case, the patient meets the clearance criteria and no collar is applied.
Positioning
Head injuries have traditionally been managed with the head slightly elevated. There is a lack of strong evidence to support this practice, but it remains common when cervical collars and spinal immobilisation are not applied. Vomiting is a symptom of head injury and is one of the reasons why full spinal immobilisation is not favoured by all clinicians in the setting of isolated head injury. Although having the head raised may have no impact on reducing ICP, it will allow for adequate venous draining from the head, and in the non-intubated patient, will allow the patient better airway patency if vomiting occurs.
Prophylactic antiemetics
There is a significant decrease in the rates of nausea experienced by trauma patients, regardless if they are experiencing symptoms of nausea to begin with when routinely treated with antiemetics. Prophylactic antiemetics decreased the rates of nausea from 61% (no antiemetic given) to 5% (antiemetic given) (Easton et al., 2012). In patients who are spinally immobilised, the use of antiemetics is important to reduce the risk of aspiration.
Pain relief
Headache is a common symptom that may be associated with either primary or secondary injury due to raised ICP. Certain opiate based analgesic medications present a risk of sedating and mimicking the extension of the brain injury (NZGG, 2006). In mild TBI the use of oral analgesics such as paracetamol may be appropriate. If narcotic analgesia is used, it should be administered conservatively and titrated carefully.
Steroids
The role that inflammation plays in the evolution of SBI has led to a number of trials of anti-inflammatory medications during the acute phase of injury. While glucocorticoids were considered a mainstay of treatment for TBI, more recent studies have shown no benefit, and in some cases harm. Methylprednisolone has been shown to increase mortality for patients with TBI and increased ICP (Carney et al., 2016).
Transport
If the patient is conscious, has no neurological symptoms, neck pain or nausea, and requires only oral analgesia, the necessity to transport the patient to hospital is often raised by patients, their family and even ambulance crews. The unpredictability of head injuries usually dictates a conservative approach and transport under clinical supervision, but in rural areas with limited resources there may be guidelines that suggest alternative pathways (see Table 34.6). In this instance, a prolonged period of unconsciousness (> 1 minute) and alterations to the patient's mental status, even without abnormal neurological findings, mean this patient should be transported to hospital for observation and possible imaging (Evans & Whitlow, 2019). Additionally, hospital assessment at the time of injury may allow the patient and carers to be educated about possible symptoms and their subsequent management.
Table 34.6
Sample guidelines for the transport of patients with isolated TBI
| Urgent ambulance transport to ED | ED assessment needed but ambulance not a requirement | Assessment at hospital not required |
|
• LOC < 5 min but now fully resolved |

Source: Adapted from ACC (2007).
 EVALUATE
EVALUATE
Evaluating the effect of any clinical management intervention can provide clues to the accuracy of the initial diagnosis. Some conditions respond rapidly to treatment so patients should be expected to improve if the diagnosis and treatment were appropriate. A failure to improve in this situation should trigger the clinician to reconsider the diagnosis.
Excluding hypoxia, hypoglycaemia and hypotension, the out-of-hospital management of mild traumatic head injuries can only act to limit the progression of an SBI. While this is essential in the long term, it means that patients will show little improvement during transport to hospital. Any deterioration must be considered to indicate a rapid extension of the injury, and ensuring hypoxia and hypotension do not occur is essential. Isolated head injuries in adults do not have the potential to cause hypovolaemia and the development of hypotension after a traumatic head injury should trigger suspicion that an uncontrolled haemorrhage is present in another area.
Ongoing management
Unless the patient deteriorates, ongoing management of mild TBI is conservative pain relief and regular observations (30 min to 4-hourly) to monitor for deterioration. In cases where the patient has a GCS of 15 and no loss of consciousness or amnesia, this could be conducted in the waiting room at hospital or under supervision at home. Observation for 24 hours is recommended in all cases of mild TBI, and any deterioration of symptoms can prompt further neurological examination (Evans & Whitlow, 2019).
Investigations
Diagnostic studies of head injuries are aimed at identifying the cause and the potential for progression of brain trauma. Magnetic resonance imaging (MRI) and particularly CT have revolutionised the assessment of head injuries and are integral to selecting management pathways. The specific criteria that direct patients towards CT or MRI may vary between centres.
While CT scans in patients with mild TBI are often normal, they are recommended as there is a small subgroup in which abnormalities will be revealed, making the examination worthwhile for the broader patient population. There is an estimated prevalence of 5% abnormal CT findings in mild TBI patients with a GCS of 15, and up to 30% of patients with a GCS of 13 (Borg et al., 2004).
A patient with a moderate or severe TBI will have an early CT as the preferential imaging modality as this can identify skull fractures, cerebral oedema as well as intracranial hematomas (Rajajee, 2018a, 2018b) and these scans can help determine specific management requirements such as depressive craniotomy.
Posttraumatic seizures, open or depressed skull fractures, basilar fractures, acute deterioration, loss of consciousness, amnesia and a high mechanism of injury are common indicators for CT investigation. Adults with head injuries who are over the age of 65 and/or who are on anticoagulant medications are required to have a CT scan in some centres (Tintinalli et al., 2016).
Skull x-rays are no longer routinely performed as they lack sensitivity for fractures, do not reveal soft-tissue damage and expose injured tissue to unnecessary radiation. Skull radiography should be performed only if CT is not available, and a negative finding may provide a false sense of security.
The insertion of probes to monitor ICP in severe TBI remains a standard practice in many advanced trauma centres and is a strong recommendation from the Brain Trauma Foundation (Carney et al., 2016). Probes can be inserted into the ventricles, the parenchyma or the subarachnoid or extradural spaces. The most invasive intraventricular probes are also the most accurate and enable drainage of CSF to lower pressure, but they carry the highest risk of infection and bleeding. The insertion of ICP monitoring will help monitor rises in ICP, and alterations to CPP, allowing intensivists to ameliorate these changes and ensure brain perfusion is maintained at all stages of treatment.
Other tests that may be used include cerebral electro-encephalogram (EEG), transcranial Doppler studies and infrared spectroscopy for region cerebral oxygenation. Positron emission tomography (PET) is also used to diagnose the cause of increased ICP (Brown & Edwards, 2014). Lumbar puncture is not performed with increased ICP, due to the risk of cerebral herniation with a sudden release of pressure in the skull from the area above the lumbar puncture (Brown & Edwards, 2014).
Hospital admission
The following criteria are associated with hospital admission after assessment:
- • GCS ≤ 13
- • abnormalities on CT (e.g. any intracranial haematoma, mass effector midline shift)
- • seizures
- • neurological deficit
- • ongoing emesis
- • abnormal bleeding parameters from underlying bleeding diathesis or oral anticoagulation (Evans & Whitlow, 2019).
Hospital discharge
After presentation at the hospital the following criteria are associated with safe discharge:
- • 6 hours of observations or vital signs in the normal range, or 4 hours if discharged into the care of a responsible third party
- • normal clinical examination
- • no vomiting
- • not intoxicated
- • suitable adult assistance available at home
- • monitoring of older people for declining neurological function, as they recover more slowly
- • instructions on the avoidance of agents with any platelet effects (e.g. aspirin, NSAIDs)
- • avoidance of contact sports for at least 1 week
- • instructions for carers about possible complications
- • appropriate follow-up (Dunn et al., 2015).
Head injuries across the lifespan
Paediatric patients
The size and mass of a child's head in relation to its body is greater than in adults. They are therefore commonly thrust forwards or fall head-first, and the impact is often to the head (Mandavia et al., 2011). Children are also vulnerable to TBI following assaults from adults, especially the injury known as shaken baby syndrome. In one study from the United Kingdom, the most common cause of head injury in children was related to low falls (< 1 m or < 5 stairs) and accounted for 32.1% of all presentations, which was greater than all sporting injuries combined (13.7%) (Trefan et al., 2016). There was also a noted positive correlation with socioeconomic status; children who presented to an ED with head injuries were more likely to have come from a poor socioeconomic status household (55.1 per 100,000) compared to the highest socioeconomic status household (39.7 per 100,000) (Trefan et al., 2016).
The child's skull is more compliant than an adult's, yet skull fractures are relatively common in children and cerebral contusion and SDH are common injuries. EDH is relatively rare in young children because of the tight adherence of the dura to the skull. Another injury that occurs almost exclusively in children with head trauma is transient cortical blindness (Mandavia et al., 2011). The incidence is unknown, but is thought to be a secondary phase of spasm induced by trauma. This occurs most often in children younger than 1 year.
In infant concussion syndrome, patients present with a broad range of symptoms from nonspecific complaints to seizure or coma. Classic findings include retinal haemorrhages, subdural haematomas, subarachnoid blood and no signs of external trauma (Mandavia et al., 2011). Children with cerebral concussion often present differently from adults. The infant concussion syndrome consists of transient appearance of pallor, diaphoresis, vomiting, tachycardia and somnolence and weakness, often occurring from minor head trauma such as falling off a change table. Usually the diagnosis is made by CT scan. Children may also present with lethargy, irritability, seizures, vomiting, poor feeding and periods of apnoea (Tintinalli et al., 2016). All these symptoms should raise the suspicion of a more serious head injury. It is important when assessing the child that you allow them time to adapt and become comfortable with your presence and if the child is sleeping they should be gently woken.
In infants, linear skull fractures often extend from one suture to another and result in the development of a cephalohaematoma. Other key points when assessing children with head injury include the following.
- • Children are more likely to experience cerebral oedema than adults.
- • Conducting a neurological assessment in children can be difficult due to their limited language skills.
- • Children have an increased incidence of posttraumatic seizures after severe head injuries.
- • Child abuse must be considered in all children with unexplained injuries, or injuries not consistent with the history provided.
- • Child abuse must be suspected in the case of intracerebral injury or skull fracture in infants or children.
- • Until the cranial sutures close, children's skulls are more distendable than those of adults and young children may sustain a lower increase in ICP after head trauma than adults with comparable mechanisms of injury.
- • Very young children (< 1 year) have a higher mortality after head trauma than older children with the same level of injury. Many factors contribute to this. Medical attention is often delayed in children with non-accidental injuries because of limited language and comprehension, and accurate formal neurological examination in young children is difficult. Medical personnel are often reluctant to initiate invasive procedures such as IV access for sedation in CT scanning. The GCS is difficult to apply to children younger than 5 years of age. Modified scales have been developed but none has been rigorously validated.
- • Children with severe or moderate head injuries are clinically similar to adults although with increased incidents of posttraumatic seizure after severe head injury. Children who sustain minor head injuries often have more pronounced physical signs than adults despite apparently trivial trauma: children may appear pale and lethargic, vomit and complain of headaches and dizziness.
- • Many children experience a brief impact seizure at the time of a relatively minor head injury; by the time the child is evaluated, they have returned to baseline neurological function.
- • There is a close association between SDH and child abuse and they are often associated with rib and long bone fractures. Approximately 10% of SDH cases are not accidental. Shaken babies have a reduced sucking reflex, a bulging anterior fontanelle, asymmetrical motor function and reflex responses, seizures, apnoea, bradycardia or vomiting. Shaken babies show no, or few, external signs of trauma and retinal haemorrhages.
Older patients
Elderly people are at an increased risk of falls and subsequent TBI. Falls account for 85% of TBI in patients over 65 and mortality rates increased dramatically the older the patients are over 65 years of age (Hawley et al., 2017). Altered mental status, seizures and focal neurological deficits should not be ascribed to intoxication, dementia or other chronic conditions if there is a history or evidence of head trauma. Patients taking anticoagulant medications should be considered at high risk of developing a secondary head injury. Atrophy of brain tissue provides a larger space for brain swelling to occur before compression starts to limit perfusion: signs of raised ICP in the elderly may take longer to develop.
 ASSESS
ASSESS
1529 hrs Safety: The crew position their vehicle to provide protection from traffic.
1529 hrs Primary survey: The patient is conscious and talking but appears confused. His speech is delayed and at times he uses inappropriate words. The patient's motorbike helmet is still in situ.
1530 hrs Pertinent hx: Bystanders state that the patient was riding his motorbike, weaving in and out of traffic. He was looking over his shoulder at the traffic when a car merged out from a side road. He swerved just before impact but his front wheel hit the bonnet of the car and he landed on the road, approximately 15–20 m away. He appeared unconscious for up to a minute. He woke up just before the crew arrived and appeared confused and unable to move.
The patient appears sweaty and confused. He is wearing jeans and a leather riding jacket. His motorcycle helmet has damage to the right side where it appears to have hit the road. The helmet is safely removed while maintaining in-line neck stabilisation. He has no other obvious injuries.
The patient is loaded onto the stretcher via spine board and is taken to the ambulance to continue the assessment.
1532 hrs Vital signs survey: Conscious state: Eyes open, appears alert = 4; inappropriate verbal response = 3; localises to pain = 5: GCS = 12.
Perfusion status: HR 126 bpm; sinus tachycardia; BP 150/90 mmHg; skin cool, pale, clammy.
Respiratory status: RR 22 bpm, good air entry, L = R, no adventitious sounds, normal work of breathing, mumbling constantly; SpO2 98% on room air.
Medical hx: The patient does not answer the crew's questions but is not aggressive or obstructive: he simply does not respond. He is not wearing a medical bracelet. A police member has made contact with the patient's family by phone and inform the crew that the patient has asthma but no allergies.
1535 hrs Secondary survey: The patient has abrasions to his right elbow and shoulder, and a haematoma over his right forehead but no obvious underlying bony damage. The skin is red and haemorrhaging slightly.
Neuro exam: Pupils are equal, round and reactive. Spontaneous movement of all limbs. No obvious facial palsy or altered sensation.
1539 hrs: The patient starts to vomit and the crew maintain in-line alignment and roll the ensure airway patency.
1540 hrs: The patient is now unresponsive to the crew. His GCS is as follows: Eyes open but catatonic = 1, verbal incomprehensible (when painful stimuli is applied the patient groans) = 2, motor withdraws both hands when painful stimuli is applied = 4, GCS = 7.
Despite this rapid decline in conscious state, the patient's HR, BP and SpO2 remain as per above.
 CONFIRM
CONFIRM
In many cases paramedics are presented with a collection of signs and symptoms that do not appear to describe a particular condition. A critical step in determining a treatment plan in this situation is to consider what other conditions could explain the patient's presentation.
What else could it be?
Drugs/alcohol
The patient's behaviour prior to the incident combined with his risk-taking behaviour (weaving in and out of traffic at speed) suggests that he may possibly be affected by drugs or alcohol. However, his neuro observations (pupils) and vital signs (hypertensive and tachycardic) are not consistent with this, and in the setting of trauma any altered conscious state should be considered to be a TBI.
Hyperthermia
Profound heat stress can produce confusion and hyponatraemia, which can lead to cerebral oedema and subsequently reduced perfusion. The day is cool and the patient's tympanic temperature is already decreasing to 35.2. Ensuring the patient remains normothermic in the setting of trauma is a key component of good holistic trauma care.
 TREAT
TREAT
As bleeding into the fixed space of the cranium continues, it raises ICP. Whether due to direct insult or in response to poor cerebral perfusion, a profound sympathetic response often occurs shortly after the primary injury, producing a rise in both heart rate and blood pressure (Atkinson & Wilberger, 2003). The primary aim of TBI management is to limit any rise in ICP and minimise any SBI, but ensure homeostasis. Unfortunately, the maximal physical exertions that can occur with cerebral irritation directly raise ICP and also increase oxygen and glucose consumption.
In the hospital setting treatment involves administering sedation and a muscle relaxant and maintaining the patient's ventilation by inserting an endotracheal tube. This treatment has been proven to reduce the extent of secondary injury and improve patient outcomes, but the combination of a sedative and a muscle relaxant can have potentially severe consequences if the clinician is not able to intubate and adequately ventilate the patient. Until recently paramedics in some areas used repeated doses of sedatives (benzodiazepines) and analgesic agents (opioids) to lower the patient's level of consciousness in order to achieve intubation. Unfortunately, the combination of activating the gag reflex during laryngoscopy (raising ICP) and the vasodilatory effects of the drugs (lowering MAP) had the net effect of lowering cerebral perfusion and led to poor patient outcomes regardless of whether endotracheal tube insertion was successful (Bernard, 2006; see Box 34.5). A number of states have subsequently trained paramedics to adopt the in-hospital practice of administering a small dose of sedation followed immediately by a rapid-acting muscle relaxant (suxamethonium, rocuronium) that acts to temporarily paralyse the patient. This procedure enables intubation to be achieved without the gag reflex causing a rise in ICP or the induction agents causing a significant fall in blood pressure. Short-acting paralysis also allows a better airway view during laryngoscopy, increasing intubation success rates. IV fluid administration and maintaining paralysis and sedation after intubation ensures hypoxia and hypotension are avoided.
The administration of benzodiazepines during the intubation process may reduce the likelihood of post-injury seizures, but there is no evidence that treating seizures prophylactically in head-injured patients improves their outcomes (see Box 34.6). Inversely, the avoidance of large doses of benzodiazepines during the induction process may prevent decreases in blood pressure. The use of anaesthetic agents, such as ketamine, afford clinicians a safer induction process, less impact on perfusion and increased maintenance of the patient's own airway tone.
RSI allows for the safe and effective management of patients with head injuries who would otherwise be 'wrestled’ to hospital, putting both themselves and the crew in danger. The process of RSI is potentially dangerous, as the patient can become hypoxic during the procedure as the short-term neuromuscular blockade removes the ability to breathe. The intent to avoid hypoxia (through endotracheal intubation) can in fact cause hypoxia if the crew are not adequately trained and prepared.
The administration of sedation alone (usually a benzodiazepine) to transport the patient is also likely to risk both hypoxia and hypotension as the drug suppresses conscious state, respiratory drive and vasomotor tone. For patients with TBI and agitation/cerebral irritation, judicious analgesia is desirable over benzodiazepines, remembering that the brain injury suffered will also be painful.
Long delays to definitive care may require remote area doctors to perform urgent craniotomy or burr holes in patients with head injuries to evacuate an expanding mass lesion such as EDH or SDH. This may be required in remote communities where patients have clinical signs of intracranial herniation or witnessed deterioration and surgical evacuation may be lifesaving. The indications are a deteriorating neurological state and an inability to access neurosurgical assistance within 2 hours; pupillary dilation must be present and the site of the extradural collection is often indicated by bogginess over the scalp fracture. Approximately 75% of extradural haematomas are temporal; the landmarks are above the midpoint of the zygomatic arch and two fingers’ breadth anterior to the external auditory canal (Dunn et al., 2015). Mannitol creates an osmotic draw of fluid from the interstitial space into the vascular space and may be used to reduce ICP. It can temporarily improve cerebral blood flow, CPP and brain metabolism (Tintinalli et al., 2016).
1545 hrs: The patient is now extending to painful stimuli and his verbal responses are incomprehensible.
Conscious state: Eyes open but catatonic = 1; incomprehensible verbal response = 2; extends abnormally to pain = 2: GCS = 5.
Perfusion status: HR 126 bpm; sinus tachycardia; BP 160/90 mmHg; skin cool, pale, clammy.
Respiratory status: RR 24 bpm, good air entry, L = R, no adventitious sounds, normal work of breathing, SpO2 98% on room air.
The crew consider transporting the patient without RSI but transport time exceeds 40 minutes. They request a second crew to assist in RSI.
1547 hrs: The crew insert an IV line and commence 10 mL/kg crystalloid for hydration.
Despite this patient's high blood pressure, his head injury has led to poor cerebral perfusion, and the administration of any sedation is likely to lower blood pressure and reduce cerebral perfusion further. Infusing 10 mL/kg prior to intubation will protect against transient hypotension.
1549 hrs: The crew commence oxygen therapy via a bag valve mask.
Although the patient is not currently hypoxic, replacing the atmospheric nitrogen in the lungs with oxygen will delay any desaturation that will start the moment the muscle relaxant stops the patient ventilating. It is important that the patient is on 100% oxygen for at least 3 minutes prior to RSI to completely denitrogenate their lungs. The addition of nasal cannula will assist in apnoeic oxygenation of the patient, further limiting the risk of hypoxia (Weingart, 2010).
1552 hrs: The crew undertake RSI, using a combination anaesthetic/analgesic combined with a short-term neuromuscular blockade.
1555 hrs: Post RSI care is important in minimising or preventing SBI, ensuring the patient's EtCO2 remains in the range 30–35 mmHg, SpO2 > 95% and BP 120 mmHg. Timely transport to hospital is also critical, so undertaking care while moving is important.
 EVALUATE
EVALUATE
Evaluating the effect of any clinical management intervention can provide clues to the accuracy of the initial diagnosis. Some conditions respond rapidly to treatment so patients should be expected to improve if the diagnosis and treatment were appropriate. A failure to improve in this situation should trigger the clinician to reconsider the diagnosis.
As the paramedics are not treating the underlying cause of the patient's condition there should be no improvement in his condition. In fact, the primary aim of evaluation here is to maintain sufficient levels of sedation, muscle relaxants, ventilation and fluid support to ensure that there is no change.
The crew intubate the patient and perform post intubation checks to ensure tube placement is confirmed. Waveform capnography is the only absolute confirmation that can be used to confirm tube placement (DAS, 2015). Once tube placement is confirmed and the endotracheal tube is secured, commencement of ongoing sedation and long-term paralysis is important to reduce noxious stimuli which can increase ICP.
Close measurement of vital signs is the key to limiting SBI in this patient.
- • Ventilation parameters need to be set, targeting EtCO2 levels in the range 30–35 mmHg, and oxygen levels above 95%. Often in the out-of-hospital setting an FiO2 of 1.0 is utilised.
- • Perfusion maintenance should be over 100 mmHg, most often through fluid administration. The use of pressor therapy is controversial in TBI.
- • Holistic patient care involves ensuring the patient remains warm and is postured appropriately (slight head elevation in patients without a spinal injury not only helps venous draining from the cranial vault but also allows for better lung oxygenation and perfusion). Periodic measurement of blood glucose and temperature is required.
- • It is important that transport to hospital is always forefront in the mind of out-of-hospital clinicians, and limiting any delays that occur will ensure the patient is taken to the appropriate hospital for definitive care.
Future research
The most effective treatment for TBI is prevention. Driver and passenger airbags and age-appropriate child restraints have reduced the number of deaths from TBI, as have helmets for motorcyclists, cyclists and other sportspeople. Long-term recovery can be improved by investigating patients shortly after their injury: early brain imaging might enable accurate prediction of the severity of the injury for the benefit of the patient's family, the community and funding agencies.
Attempts using medications to reduce the inflammatory response and provide a form of neuroprotection have not proved successful but there are a number of trials in which TBI patients are aggressively cooled after intubation. Therapeutic hypothermia has been demonstrated to provide some cerebral benefit post cardiac arrest (Bernard et al., 2002) but the studies involving the process in TBI are yet to prove conclusive (Sandestig et al., 2014).
Hyperglycaemia may be caused by the post-injury surge of catecholamines and therefore can occur in patients who normally maintain good glucose control (i.e. non-diabetics; Atkinson & Wilberger, 2003). A high BGL is associated with poorer outcomes, and the next stage of out-of-hospital treatment of TBI may be restoration of normal BGL. This will require care and training, as patients who have been sedated and intubated may not show overt signs of hypoglycaemia following treatment.
TBIs that are apparently similar in mechanism and severity can produce vastly different outcomes between individuals and this may be partially explained by recent research that has identified the apolipoprotein E gene as directing at least part of the pathological response to neural injury (Sun & Jiang, 2008).
Summary
'Head injury’ is a broad term that includes injuries to the scalp and face but is used clinically to describe a TBI. The injury caused at the time that the force is applied will result in tissue damage that, if sufficiently severe and widespread, can cause a cascade of responses that lead to poor cerebral perfusion. If not corrected this SBI can cause profound morbidity and death. Recognition of the mechanism of injury, a detailed neurological examination and monitoring for deterioration are key in identifying the progression of a secondary injury. Hypoxia and hypotension increase the development of an SBI and must be actively managed if they are present.





