Chapter 208 Hansen Disease (Mycobacterium leprae)
Hansen disease (leprosy) is a chronic disease resulting from infection with Mycobacterium leprae and moderated by the ensuing host response. The respiratory mucosa, skin, and peripheral nervous system are most prominently affected, with occasional testicular and ocular involvement. Humans were long believed to be the sole host of M. leprae, but naturally acquired infection has been documented in armadillos in the southeastern USA, and experimental infection has been established in primates, nude mice, and armadillos.
The sequelae of leprosy include chronic skin lesions, madarosis, sensory neuropathy resulting in the loss of digits or limbs, and paresis secondary to motor nerve dysfunction. The highly visible nature of these debilities led to the historical stigmatization of the “leper.” The psychological and sociologic sequelae of this stigma can be as debilitating as the disease itself and can result in delays in seeking medical attention. To combat this prejudice, the term leprosy patient has replaced the word leper, and Hansen disease, after Armauer Hansen who identified M. leprae as the cause of leprosy, is the accepted designation.
Etiology
M. leprae is an acid-fast bacillus of the family Mycobacteriaceae. It is an obligate intracellular pathogen with a generation time of 14 days. M. leprae grows optimally at 27-30°C and cannot be cultured in vitro. The incubation period of leprosy in humans ranges from 3 mo to 20 yr (average 3-5 yr). The rare occurrence of leprosy in infants as young as 3 mo of age suggests that in utero transmission can occur or that very short incubation periods are possible in certain situations.
Epidemiology
Since the introduction of multidrug therapy by the World Health Organization (WHO) in 1982, there has been a steady decline in the prevalence of leprosy. In 1991, the WHO called for the elimination of leprosy as defined by a worldwide prevalence of <1/10,000 population. That goal was achieved by the year 2000 in all but 15 of the 122 countries where leprosy was considered a public health problem in 1985. Those countries are mainly in Asia, Africa, and South America. In 1985, there were 10-12 million cases of leprosy worldwide, and in 2004, the prevalence was 457,792 cases. In 1998, 750,000 new cases were detected, and in 2008, there were 218,605 new cases registered for treatment. By 2008, 80% of the world’s leprosy patients resided in India, Brazil, or Indonesia. Approximately 150 cases are reported annually in the USA, of which 85% are in immigrants. Small numbers of endemic cases are reported from Texas, Hawaii, and Louisiana.
Human-to-human transmission is responsible for an overwhelming majority of secondary cases, with the highest risk among family members and other close contacts. Leprosy occurs at all ages, but infections in infants are extremely rare. The incidence rates peak during childhood and early adulthood in endemic areas. HIV infection has not been documented to alter the risk for leprosy in areas of high prevalence for both pathogens.
Pathogenesis
Possible modes of transmission include contact with desquamated infected epidermis, ingestion of infected breast milk, and bites of mosquitoes or other vectors. However, at present the basis for most infections appears to be transmission from untreated lepromatous patients following prolonged contact with infected nasal secretions containing a high bacterial load. Skin testing and serologic studies suggest that up to 90% of infected persons develop immunity without ever manifesting clinical disease. Studies in endemic areas using polymerase chain reaction (PCR) show widespread presence of the organism in nasal secretions from asymptomatic persons.
M. leprae appears to be transported hematogenously from the nasal mucosa to skin and peripheral nerves. Using the armadillo model of neuritis, M. leprae organisms have been shown to colonize the perineural space and gain access to the interstitium of the endoneural space. Once there, organisms are available for phagocytosis by Schwann cells and interstitial macrophages surrounding peripheral nerve axons. Intracellular replication of M. leprae follows, with varying degrees depending on the host cellular immune response. M. leprae attachment to and ingestion by Schwann cells has been shown to depend on receptors on the lamin-2 glycoprotein in the basal lamina and the α-dystroglycan complex in the Schwann cell basement membrane. M. leprae–specific phenolic glycolipid-1 appears to be the ligand mediating this binding.
Once inside the Schwann cell, M. leprae replicates slowly over years. At some stage, specific T cells recognize the presence of mycobacterial antigens within the cells and initiate a chronic inflammatory reaction. Genetic susceptibility to leprosy has been linked to the natural resistance associated with macrophage protein-1, which is present only in certain racial groups. Factors that can lead to the development of localized vs disseminated disease are the degree of expression of Toll-like receptors on monocytes and the production of interferon-γ (IFN-γ) by T cells.
Once M. leprae colonizes the surface of nerves and infects endoneural macrophages and Schwann cells, several mechanisms of skin and nerve injury can occur, depending on the host immune response. One end of the spectrum is tuberculoid leprosy (TL), in which there is a vigorous and specific cell-mediated immune response to M. leprae antigens. In tissue biopsies there are tightly organized granulomas composed of epithelioid cells and lymphocytes, but bacilli are scant or absent. Macrophages, when present, do not contain intracellular organisms. Caseation is rare. Heavy cellular infiltration is found in the dermis, with destruction of cutaneous nerve fibers.
At the other end of the spectrum is lepromatous leprosy (LL), in which there is total and specific anergy to M. leprae both by skin testing and by in vitro assays of cell-mediated immunity. Large amounts of circulating and tissue-based antibody to mycobacterial antigens is present, but it affords no protective immunity. Bacilli are found in enormous numbers in the skin, nasal mucosa, and peripheral nerves. There is continual bacillemia as well as bacillary invasion of all major organs except the central nervous system. Tissue granulomas are poorly formed and are composed chiefly of loose aggregates of foamy histiocytes. Macrophages teeming with undigested bacilli are common. There is extensive, symmetric involvement of peripheral nerves, although the cutaneous nerve endings are usually spared.
An M. leprae-specific suppressor T-cell population is found in the circulation of patients with LL, with increased numbers of suppressor T cells found in the skin granulomas. T cells from lepromatous patients also produce less interleukin 2 and less IFN-γ after stimulation with M. leprae antigens than do T cells from tuberculoid patients or normal controls.
Borderline or dimorphous leprosy is subdivided into 3 subclasses that lie between the tuberculoid and lepromatous poles: borderline tuberculoid, borderline, and borderline lepromatous.
Clinical Manifestations
The manifestations of Hansen disease reflect the host response to the infection.
Indeterminate Leprosy
Indeterminate leprosy (IL) is the earliest clinically detectable form of leprosy. Although it is diagnosed in only 10-20% of infected patients, most patients with advanced leprosy have passed through this stage. Usually there is a single hypopigmented macule 2-4 cm in diameter with a poorly defined border and without erythema or induration. Anesthesia is minimal or absent, particularly if the lesion is on the face. Tissue samples can show granulomas, but bacilli are rarely demonstrable. The diagnosis is usually made by exclusion of other skin disorders in contacts of leprosy patients, especially in children. The lesions heal spontaneously in 50-75% of patients with IL and progress to 1 of the classic forms in the remainder.
Tuberculoid Leprosy
In tuberculoid leprosy, there is usually a single, large (often >10 cm in diameter) lesion with a well-demarcated, elevated erythematous rim (Fig. 208-1). The interior of the lesion is flat, atrophic, hypopigmented, occasionally scaly, and anesthetic. Rarely, there are as many as 4 lesions. The closest superficial nerve is often impressively thickened. The ulnar, posterior tibial, and great auricular nerves are most commonly affected. Periodic examination of all leprosy patients and their contacts should include palpation of these nerves. Without therapy, the skin lesion tends to enlarge slowly, but documented instances of spontaneous resolution exist. The coloration of the rim slowly fades with therapy and the induration resolves, resulting in a flat lesion with central hypopigmentation and a ring of postinflammatory hyperpigmentation. Loss of hair follicles, sweat glands, cutaneous nerve receptors, and sensation in the central portion of the lesion is irreversible. Marked improvement should be apparent within 1-2 mo after initiating therapy, but complete resolution can take up to 8-12 mo.
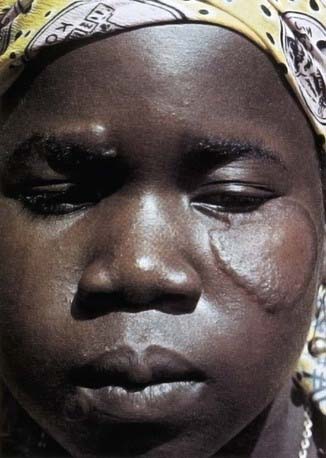
Figure 208-1 A patient with tuberculoid leprosy showing a lesion with a raised border and flattened center.
There is an entity of pure neural tuberculoid leprosy, which manifests as either pure sensory or combined sensory and motor nerve dysfunction with prominent nerve thickening but no cutaneous lesions. Histopathology is mandatory to establish this diagnosis. Nerve trunk size varies widely, and overdiagnosis of “enlarged” nerves is common among inexperienced histologists. Nodular or fusiform nerve thickening has greater diagnostic value than a palpable nerve that is smooth and symmetric.
Borderline Leprosy
The clinical and histologic criteria for the 3 subdivisions of borderline leprosy are less well defined than are those of the 2 polar categories. In contrast to the tuberculoid and lepromatous patterns, those in the borderline divisions are unstable. Host or bacterial factors can result in immunologic downgrading of the clinical condition toward the lepromatous pattern or upgrading it toward the tuberculoid pattern. Therapy is the most common cause of upgrading reactions; downgrading can be seen in conditions that compromise host immunity (pregnancy). Clinical characteristics of the 3 generally accepted borderline subclasses are as follows.
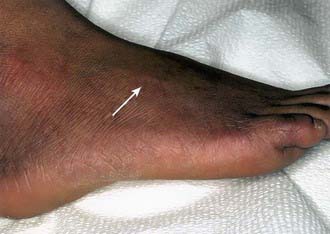
In the borderline tuberculoid (BT) pattern, the lesions are greater in number but smaller in size than in TT. There may be small satellite lesions around older lesions, and the margins of the BT lesions are less distinct. There is usually thickening of 2 or more superficial nerves (Fig. 208-2).
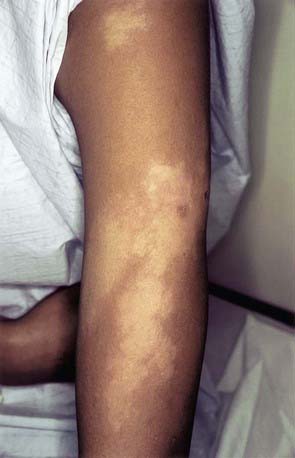
In the borderline (BB) pattern, the lesions are more numerous and more heterogeneous in appearance. They can become confluent, and plaques may be present. The borders are poorly defined, and the erythematous rim fades into the surrounding skin (Fig. 208-3). There may be anesthesia, but hypesthesia is more common. Mild to moderate nerve thickening is common, but severe muscle wasting and neuropathy are unusual.
In the borderline lepromatous (BL) pattern, a large number of asymmetrically distributed lesions are heterogeneous in appearance. Macules, papules, plaques, and nodules can all coexist. Individual lesions are small unless confluent. Anesthesia is mild, and superficial nerve trunks are spared. The initial response to therapy is often dramatic, and nodules and plaques flatten within 2-3 mo. With continued therapy, the lesions become macular and almost invisible.
Lepromatous Leprosy
In lepromatous leprosy, the lesions are innumerable, often confluent, and symmetric. Initially there may be only vague macules or even uniform, diffuse skin infiltrations without discernible lesions. As the disease progresses, the lesions become increasingly papular and nodular, so that with the diffuse thickening and infiltration of the skin, the characteristic leonine facies accompanied by loss of the eyebrows and distortion of the earlobes becomes apparent. Anesthesia of the lesions is less severe than in TT, but a symmetric peripheral sensory neuropathy usually develops late in the course of the disease. Testicular infiltration leading to azoospermia, infertility, and gynecomastia is common in adults but not in children. Bacilli are demonstrable in most internal organs other than the central nervous system, but tissue damage or interference with function is uncommon. Glomerulonephritis, when it occurs, is believed to be secondary to immune complex deposition rather than to infection per se. The initial response to therapy may be encouraging but is often followed by a 2-5 yr period of very slow improvement. In true LL, the specific anergy to the leprosy bacillus persists despite therapy, thus making the patient theoretically susceptible to relapse if even a single viable bacillus remains at the end of therapy.
Reactional States
Acute clinical exacerbations are common in leprosy and are believed to reflect abrupt changes in the host-parasite immunologic balance. Although these reactional states do occur in the absence of therapy, they are especially common during the initial years of treatment. Approximately 30% of patients receiving effective chemotherapy can develop reactions. Unless the patient is adequately treated, these reactions result in crippling deformities. Two major variants are recognized.
Type 1 (reversal) reactions are observed predominantly in borderline leprosy and result from a sudden increase in effective cell-mediated immunity in response to M. leprae antigens in dermis and Schwann cells. Acute tenderness and swelling at the site of existing cutaneous and neural lesions and the development of new lesions are the major manifestations. Existing or new skin lesions often ulcerate to leave hideous scars. The acute neuritis can manifest either as a severe painful episode or as insidious and painless loss of function that if not treated immediately can lead to irreversible nerve injury with anesthesia, facial paralysis, claw hand, and footdrop. Reversal reactions constitute perhaps the only medical emergency related to leprosy. Patients should be instructed to contact their physicians immediately if signs of a reaction appear.
Type 2 (erythema nodosum leprosum) reactions (ENL) occur in lepromatous and borderline lepromatous cases as a systemic inflammatory response to deposition of extravascular immune complexes of antibody and M. leprae antigen. The hallmarks of this syndrome are tender, red dermal papules or nodules that clinically resemble erythema nodosum. These develop in a few hours and last only a few days. High fever, migratory polyarthralgia, painful swelling of lymph nodes or spleen, orchitis, iridocyclitis, and, rarely, nephritis can occur. Leukocytosis and albuminuria may be present. Circulating and tissue-based immune complexes are often present and might explain the resemblance to other immune complex disorders, but the underlying mechanism appears to involve the activation of a helper T-cell subset. There is a strong tendency to recurrence, which occurs in 45% of patients, and a risk for amyloidosis and renal failure if treatment is inadequate.
Diagnosis
The critical factor in the diagnosis of leprosy is to consider this in the differential diagnosis of a skin disorder in anyone who has resided in an endemic leprosy region. Anesthetic skin lesions with or without thickened peripheral nerves are virtually pathognomonic of leprosy. A full-thickness skin biopsy from an active lesion (stained with both a standard histologic stain and an acid-fast stain such as Fite-Faraco) is the optimal procedure for confirmation of the diagnosis and accurate classification of the disease. Acid-fast bacilli are rarely found in patients with indeterminate or tuberculoid disease, but the presence of granulomas and lymphocytic infiltration of nerves in anesthetic skin lesions confirms the diagnosis. For purposes of assigning patients to the appropriate WHO multidrug regimen, slit skin smears are assessed to determine whether patients have paucibacillary infection (≤5 skin lesions and no bacilli on skin smears) or multibacillary infection (≥6 skin lesions and bacilli on skin smears). The bacterial index can range from 0 (no bacilli in 100 oil-immersion fields) to 6+ (>1,000 bacilli per field). PCR detects M. leprae only when biopsy specimens are also positive for acid-fast bacilli.
Differential Diagnosis
Many diseases endemic in developing countries can mimic the appearance of leprosy, including secondary syphilis, cutaneous leishmaniasis, yaws, and cutaneous fungal infections. None of these other disorders involves paresthesia and anesthesia localized to the skin lesions or causes thickening of peripheral nerves. The presence of nerve thickening with skin lesions also differentiates leprosy from primary neurologic disease. IL can occur with minimal anesthesia, no nerve thickening, and equivocal histopathology, suggesting a superficial fungal infection, particularly tinea versicolor. The diagnosis of IL should be considered one of exclusion and is rarely established in anyone other than a close contact of a known lepromatous patient.
Treatment
Physicians in the USA considering the diagnosis or treatment of leprosy should obtain consultation and assistance in patient management from the National Hansen’s Disease Programs (800-642-2477; www.hrsa.gov/hansens/), which maintains an active physician referral list of physicians in all parts of the USA.
Only 3 antimycobacterial agents have proven to be consistently effective in the treatment of leprosy. Dapsone is the cornerstone of therapy because of its low cost, minimal toxicity, and wide availability. Secondary resistance develops when it is used as the sole agent. Dermatitis, hepatitis, and methemoglobinemia are the most common side effects; granulocytopenia is rare but potentially fatal. Dose-related hemolytic anemia, which can be severe, is seen in patients with glucose-6-phosphate dehydrogenase deficiency, methemoglobin reductase deficiency, or hemoglobin M. Pregnancy studies have not shown an increased risk for fetal abnormalities.
Rifampin is the most rapidly mycobactericidal drug for M. leprae and achieves excellent levels inside cells, where most leprosy bacilli reside. Resistance develops if rifampin is used as a single agent. The widespread use of rifampin has been limited by cost more than by toxicity. Hepatitis is the most common adverse effect that necessitates discontinuance.
Clofazimine, a phenazine dye with both antimycobacterial and anti-inflammatory activity, has been particularly useful in decreasing the incidence of reactional states. The pharmacokinetics are poorly understood, but the half-life is several days. The drug is avidly taken up by epithelial cells, an attribute that may be important for its activity but also results in cutaneous hyperpigmentation, ichthyosis, xerosis, and enteritis. The intense reddish-brown discoloration of the skin is a cosmetic deterrent to use and often results in discontinuation or poor compliance.
Minocycline, some 2nd-generation quinolones, and macrolide derivatives such as clarithromycin have shown promise in experimental models, but limited human treatment data exist.
Multidrug therapy has been very successful, with a high cure rate and a relapse rate of 1%/yr following a full course of therapy. For adults with multibacillary leprosy (all BL and LL patients), therapy is recommended for 12 mo to include rifampin (600 mg once monthly PO, directly observed), dapsone (100 mg once daily PO, self-administered), and clofazimine (300 mg once monthly, directly observed, and 50 mg once daily PO, self-administered). For adults with paucibacillary leprosy (all IL, TT, and most BT patients), therapy is recommended for 6 mo with rifampin (600 mg once monthly PO, directly observed) and dapsone 100 mg once daily PO, self-administered). Patients who experience relapse are retreated with the same regimens. To date there have been no patients treated with WHO-recommended multidrug therapy who have experienced secondary rifampin resistance, which has occurred in persons treated with rifampin monotherapy or multidrug therapy not recommended by WHO.
Patients treated in the USA may be advised to receive regimens that vary from WHO recommendations. Adults with paucibacillary disease receive dapsone (100 mg daily) and rifampin (600 mg daily) for 12 mo; those with multibacillary disease receive dapsone (100 mg daily), rifampin (600 mg daily), and clofazimine (50 mg daily) for 24 mo. Daily pediatric doses are dapsone 1 mg/kg, rifampin 10 mg/kg, and clofazimine 1 mg/kg, not to exceed the recommended adult doses.
Therapy for reactional states can be complicated and generally requires expert consultation. Management depends on maintenance of antimycobacterial drugs, effective and prolonged anti-inflammatory therapy, and adequate analgesia and physical support during the phase of active neuritis. Mild ENL might respond to nonsteroidal anti-inflammatory agents. Thalidomide is the drug of choice for most of the systemic symptoms associated with ENL reactions. Thalidomide accelerates the degradation of messenger RNA encoding TNF-α, resulting in its decreased production by monocytes and macrophages. More-severe ENL usually requires corticosteroid therapy (prednisolone 1 mg/kg/day) but often relapses when the drug is discontinued. Thalidomide is absolutely contraindicated in women of childbearing age; otherwise it is much safer than corticosteroids for chronic use. The major adverse effect is fatigue. Pediatric dosages have not been established. Clofazimine 300 mg/day tapering to ≤100 mg/day for 12 mo is also useful in managing chronic ENL. Type 1 reversal reactions are optimally treated with high-dose corticosteroids (prednisolone 40-60 mg/day PO for adults; 1 mg/kg/day PO for children) tapered over several months. Alternate-day regimens may be effective in patients with frequent relapses requiring prolonged treatment.
Prognosis
The prognosis for arresting progression of tissue and nerve damage is good, but recovery of lost sensory and motor function is variable and generally incomplete. Hyperpigmentation, hypopigmentation, and loss of hair follicles or sweat glands persist. Intercurrent reactional states, poor compliance, and emergence of drug resistance can all lead to clinical exacerbations or relapses, necessitating close follow-up of patients. Much of the chronic debility results from repeated trauma to anesthetic digits and limbs. Careful counseling of patients and consultation with physical and occupational therapy services are essential for an optimal outcome.
Prevention
Two approaches are advocated for interrupting leprosy transmission in endemic areas. The 1st approach is based on the high risk for infection among household and other close contacts of leprosy patients, especially with multibacillary disease. It includes periodic examination of contacts and early treatment at the 1st evidence of leprosy. Contacts 15-19 yr of age or >30 yr of age are at greatest risk for secondary disease. The 2nd approach is the use of bacille Calmette-Guérin (BCG) vaccination. One dose of BCG appears to be 50% protective against leprosy; a 2nd dose increases the protective benefit.
The viability of M. leprae in skin biopsies decreases sharply within 3 wk of initiating therapy with dapsone and rifampin. This rapid decrease in infectivity combined with the high probability that family members have already had prolonged exposure to the patient before the diagnosis makes physical isolation of leprosy patients unnecessary.
Boggild AK, Keystone JS, Kain KC. Leprosy: a primer for Canadian physicians. CMAJ. 2004;170:71-78.
Britton WJ, Lockwood DNJ. Leprosy. Lancet. 2004;363:1209-1219.
Cambau E, Bonnafous P, Perani E, et al. Molecular detection of rifampin and ofloxacin resistance for patients who experience relapse of multibacillary leprosy. Clin Infect Dis. 2002;34:39-45.
Hartzell JD, Zapor M, Peng S, et al. Leprosy: a case series and review. South Med J. 2004;97:1252-1256.
Lawn SD, Lockwood DNJ. Leprosy after starting antiretroviral treatment. BMJ. 2007;334:217-218.
Moet FJ, Pahan D, Schurng RP, et al. Physical distance, genetic relationship, age, and leprosy classification are independent risk factors for leprosy in contacts of patients with leprosy. J Infect Dis. 2006;193:346-353.
Moschella SL. An update on the diagnosis and treatment of leprosy. J Am Acad Dermatol. 2004;51:417-426.
Ooi WW, Moschella SL. Update on leprosy in immigrants in the United States: status in the year 2000. Clin Infect Dis. 2001;32:930-937.
Ooi WW, Srinivasan J. Leprosy and the peripheral nervous system: basic and clinical aspects. Muscle Nerve. 2004;30:393-409.
World Health Organization. Global leprosy situation. Weekly Epidemiol Record. 2008;33:293-300.
Zhang FR, Huang W, Chen SM, et al. Genomewide association study of leprosy. N Engl J Med. 2009;361:2609-2618.