Chapter 212 Leptospira
Leptospirosis is a common and widespread zoonosis in the world and is caused by spirochetes of the genus Leptospira.
Etiology
Leptospira are aerobic spiral bacteria with a terminal hook at 1 or both ends. Pathogenic leptospires belong to a single species, Leptospira interrogans, which includes >200 distinct serovars. A single serovar can produce a variety of distinct syndromes, and a single clinical manifestation may be caused by multiple serotypes. Nonpathogenic leptospires are classified as Leptospira biflexa, with >60 serovars.
Epidemiology
Most human cases of leptospirosis occur in tropical and subtropical countries, but the distribution is worldwide. The rat is the principal source of human infection, but leptospires infect many species of domestic and feral animals, including livestock, birds, fish, dogs, cats, and wild animals, including reptiles. Infected animals excrete spirochetes in their urine for prolonged periods. Most human cases result from occupational exposure to water or soil contaminated with rat urine. Groups with a high incidence of leptospirosis include persons exposed occupationally or recreationally to contaminated soil or water, agricultural workers, veterinarians, abattoir workers, meat inspectors, rodent control workers, laboratory workers, and workers in other occupations that require contact with animals. The major animal reservoir in the USA is the dog. Transmission via animal bites and directly from person to person has been rarely reported.
Pathology and Pathogenesis
Leptospires enter humans through abrasions and cuts in the skin or through mucous membranes. After penetration, they circulate in the bloodstream to all body organs, causing endothelial lining damage of small blood vessels with secondary ischemic damage to the end organs.
Clinical Manifestations
The spectrum of human leptospirosis ranges from asymptomatic infection (most cases) to severe disease with multiorgan dysfunction and death. The onset is usually abrupt, and the illness tends to follow a biphasic course (Fig. 212-1). After an incubation period of 7-12 days, there is an initial or septicemic phase lasting 2-7 days, during which leptospires can be isolated from the blood, cerebrospinal fluid (CSF), and other tissues. This phase may be followed by a brief period of well-being before onset of a second symptomatic immune or leptospiruric phase. This phase is associated with the appearance of circulating antibody, disappearance of organisms from the blood and CSF, and appearance of signs and symptoms associated with localization of leptospires in the tissues. Despite the presence of circulating antibody, leptospires can persist in the kidney, urine, and aqueous humor. The immune phase can last for several weeks. Symptomatic infection may be anicteric or icteric.
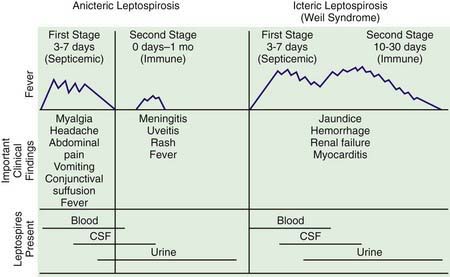
Figure 212-1 Stages of anicteric and icteric leptospirosis. Correlation between clinical findings and presence of leptospires in body fluids, CSF, cerebrospinal fluid.
(Reprinted with permission from Feigin RD, Anderson DC: Human leptospirosis, CRC Crit Rev Clin Lab Sci 5:413–467, 1975. Copyright CRC Press, Inc, Boca Raton, FL.)
Anicteric Leptospirosis
The septicemic phase of anicteric leptospirosis has an abrupt onset with flulike signs of fever, shaking chills, lethargy, severe headache, malaise, nausea, vomiting, and severe debilitating myalgia most prominent in the lower extremities, lumbosacral spine, and abdomen. Bradycardia and hypotension can occur, but circulatory collapse is uncommon. Conjunctival suffusion with photophobia and orbital pain (in the absence of chemosis and purulent exudate), generalized lymphadenopathy, and hepatosplenomegaly may also be present. A transient (<24 hr) erythematous maculopapular, urticarial, petechial, purpuric, or desquamating rash occurs in 10% of cases. Rarer manifestations include pharyngitis, pneumonitis, arthritis, carditis, cholecystitis, and orchitis. The second or immune phase can follow a brief asymptomatic interlude and is characterized by recurrence of fever and aseptic meningitis. CSF abnormalities include a modest elevation in pressure, pleocytosis with early polymorphonuclear leukocytosis followed by mononuclear predominance rarely exceeding 500 cells/mm3, normal or slightly elevated protein levels, and normal glucose values. Although 80% of infected children have abnormal CSF profiles, only 50% have meningeal manifestations. Encephalitis, cranial and peripheral neuropathies, papilledema, and paralysis are uncommon. A self-limited unilateral or bilateral uveitis can occur during this phase, rarely resulting in permanent visual impairment. Central nervous system symptoms usually resolve spontaneously within 1 wk, with almost no mortality.
Icteric Leptospirosis (Weil Syndrome)
Weil syndrome is a rare (<10% of cases) severe form of leptospirosis seen more commonly in adults (>30 yr) than in children. The initial manifestations are similar to those described for anicteric leptospirosis. The immune phase, however, is characterized by jaundice, renal failure, thrombocytopenia, and, in fulminant cases, hemorrhage and cardiovascular collapse. Hepatic involvement leads to right upper quadrant pain, hepatomegaly, direct and indirect hyperbilirubinemia, and modestly elevated serum levels of hepatic enzymes. Liver function usually returns to normal after recovery. All patients have abnormal findings on urinalysis (hematuria, proteinuria, and casts), and azotemia is common, often associated with oliguria or anuria. Acute kidney failure occurs in 16-40% of cases and is the principal cause of death. Abnormal electrocardiograms are present in 90% of cases, but congestive heart failure is uncommon. Transient thrombocytopenia occurs in >50% of cases. Rarely, hemorrhagic manifestations occur, including epistaxis, hemoptysis, and pulmonary, gastrointestinal, and adrenal hemorrhage. The mortality rate is 5-15%.
Diagnosis
Leptospirosis should be considered in the differential diagnosis of acute flulike febrile illnesses with a history of direct contact with animals or with soil or water contaminated with animal urine. This disease may be difficult to distinguish clinically from dengue or malaria.
Use of Warthin-Starry silver staining, polymerase chain reaction, and immunofluorescent and immunohistochemical methods permits diagnosis of leptospirosis from infected tissue or body fluids. The diagnosis is most often made by serologic testing and less often by isolation of the infecting organism from clinical specimens. Serologic tests for Leptospira include genus-specific and serogroup-specific tests. The reference method is the microscopic agglutination test, a serogroup-specific assay using live antigen suspension of leptospiral serovars and dark-field microscopy for agglutination. A 4-fold or greater increase in titer in paired sera confirms the diagnosis. Agglutinins usually appear by the 12th day of illness and reach a maximum titer by the 3rd wk. Low titers can persist for years. Approximately 10% of infected persons do not have detectable agglutinins, presumably because available antisera do not identify all Leptospira serotypes. Enzyme-linked immunosorbent assay (ELISA) methods, including an immunoglobulin M-specific dot-ELISA test, are also available. Phase-contrast and dark-field microscopy are insensitive for spirochete detection.
Unlike other pathogenic spirochetes, leptospires can be recovered from the blood or CSF during the first 10 days of illness and from urine after the 2nd week by repeated culture of small inoculum (i.e., 1 drop of blood or CSF in 5 mL of medium) on commercially available media-containing rabbit serum or bovine serum albumin and long-chain fatty acids. However, the inoculum in clinical specimens is small, and growth can take up to 13 wk.
Treatment
Despite in vitro sensitivity of Leptospira to penicillin and tetracycline, the effectiveness of these antibiotics in treating human leptospirosis is unclear due to the naturally high spontaneous recovery rates. Some studies suggest that initiation of treatment before the 7th day shortens the clinical course and decreases the severity of the infection; thus treatment with penicillin or tetracycline (in children ≥9 yr of age) should be instituted early when the diagnosis is suspected. Parenteral penicillin G (6-8 million U/m2/day divided every 4 hr IV for 7 days) is recommended, with tetracycline (10-20 mg/kg/day divided every 6 hr PO or IV for 7 days) as an alternative for patients allergic to penicillin. Oral amoxicillin is an alternative therapy for children <9 yr of age.
Prevention
Prevention of human leptospirosis infection is facilitated by instituting rodent control measures and avoiding contaminated water and soil. Immunization of livestock and family pets is recommended as a means of eliminating animal reservoirs. Attempts at a human vaccine have been challenging. Protective clothing should be worn by persons at risk for occupational exposure. Leptospirosis has been prevented in American soldiers stationed in the tropics by administering doxycycline (200 mg PO once a week) as prophylaxis. This approach may be similarly effective for travelers to highly endemic areas for short periods.
Bharti AR, Nally JE, Ricaldi JN, et al. Leptospirosis: a zoonotic disease of global importance. Lancet Infect Dis. 2003;3:757-771.
Centers for Disease Control and Prevention. Leptospirosis after flooding of a university campus—Hawaii, 2004. MMWR Morb Mortal Wkly Rep. 2006;55:125-127.
Heath CWJr, Alexander AD, Galton MM. Leptospirosis in the United States. Analysis of 483 cases in man, 1949–1961. N Engl J Med. 1965;273:857-864.
Jackson LA, Kaufmann AF, Adams WG, et al. Outbreak of leptospirosis associated with swimming. Pediatr Infect Dis J. 1993;12:48-54.
Libraty DH, Myint KSA, Murray CK, et al. A comparative study of leptospirosis and dengue in Thai children. PLoS Negl Trop Dis. 2007;1(3):e111.
Meites E, Jay MT, Deresinski S, et al. Reemerging leptospirosis, California. Emerg Infect Dis. 2004;10:406-412.
Sharma R, Tuteja U, Khushiramani R, et al. Application of rapid dot–ELISA for antibody detection of leptospirosis. J Med Microbiol. 2007;56:873-874.
Wang Z, Jin L, Wegrzyn A. Leptospirosis vaccines. Microb Cell Fact. 2007;6:39.
Wong ML, Kaplan S, Dunkle LM, et al. Leptospirosis: a childhood disease. J Pediatr. 1977;90:532-537.
Wuthiekanun V, Sirisukkarn N, Daengsupa P. Clinical diagnosis and geographic distribution of leptospirosis, Thailand. Emerg Infect Dis. 2007;13:124-126.