Chapter 260 Arboviral Encephalitis outside North America
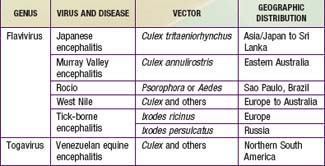
The principal causes of arboviral encephalitis outside North America are Venezuelan equine encephalitis (VEE) virus, Japanese encephalitis (JE) virus, tick-borne encephalitis (TBE), and West Nile (WN) virus (Table 260-1).
260.1 Venezuelan Equine Encephalitis
The VEE virus was isolated from an epizootic in Venezuelan horses in 1938. Human cases were first identified in 1943. Hundreds of thousands of equine and human cases have occurred over the past 70 yr. During 1971, epizootics moved through Central America and Mexico to southern Texas. After 2 decades of quiescence, epizootic disease emerged again in Venezuela and Colombia in 1995.
Etiology
VEE is an alphavirus of the family Togaviridae. VEE circulates in nature in 6 subtypes. Virus types I and III have multiple antigenic variants. Types IAB and IC have caused epizootics and human epidemics.
Epidemiology
The majority of epizootics resulting from types IAB and IC have occurred in Venezuela and Colombia. The virus resides in ill-defined sylvatic reservoirs in the South American rain forests. Known hosts include rodents and aquatic birds with transmission by Culex melaconion species. Vectors for horse-to-horse and horse-to-human transmission include Aedes taeniorhynchus and Psorophora confinnis. Epizootics move rapidly, up to several miles per day. Human cases are proportional to and follow epizootic occurrences. Viremia levels in human blood are high enough to infect mosquitoes. Because virus can be recovered from human pharyngeal swabs, and household attack rates are often as high as 50%, it is widely believed that person-to-person transmission occurs, although direct evidence is lacking. Virus types II-VI are restricted to relatively small foci; each has a unique vector-host relationship and rarely results in human infections.
Clinical Manifestations
The incubation period is 2-5 days, followed by the abrupt onset of fever, chills, headache, sore throat, myalgia, malaise, prostration, photophobia, nausea, vomiting, and diarrhea. In 5-10% of cases, there is a biphasic illness; the 2nd phase is heralded by seizures, projectile vomiting, ataxia, confusion, agitation, and mild disturbances in consciousness. There is cervical lymphadenopathy and conjunctival suffusion. Cases of meningoencephalitis may demonstrate cranial nerve palsy, motor weakness, paralysis, seizures, and coma. Microscopic examination of tissues reveals inflammatory infiltrates in lymph nodes, spleen, lung, liver, and brain. Lymph nodes show cellular depletion, necrosis of germinal centers, and lymphophagocytosis. The liver shows patchy hepatocellular degeneration, the lungs demonstrate a diffuse interstitial pneumonia with intra-alveolar hemorrhages, and the brain shows patchy cellular infiltrates.
Diagnosis
The etiologic diagnosis of VEE is established by testing an acute-phase serum collected early in the illness for the presence of virus-specific immunoglobulin (Ig) M antibodies or, alternatively, demonstrating a fourfold or greater increase in IgG antibody titers by testing paired acute and convalescent sera. The virus can also be identified by polymerase chain reaction (PCR).
Treatment
There is no specific treatment for VEE. The treatment is intensive supportive care (Chapter 62), including control of seizures (Chapter 586).
Prognosis
In patients with VE meningoencephalitis, the fatality rate ranges from 10% to 25%. Sequelae include nervousness, forgetfulness, recurrent headache, and easy fatigability.
Prevention
Several veterinary vaccines are available to protect equines. VEE virus is highly infectious in laboratory settings, and biosafety level 3 containment should be used. An experimental vaccine is available for use in laboratory workers.
Gubler DJ. The continuing spread of West Nile virus in the western hemisphere. Clin Infect Dis. 2007;45:1039-1046.
Kramer LD, Styer LM, Ebel GD. A global perspective on the epidemiology of West Nile virus. Annu Rev Entomol. 2008;53:61-81.
Siegel-Itzkovich J. Twelve die of West Nile virus in Israel. Br Med J. 2000;321:724.
Tsai TF, Popovici F, Cerescu C, et al. West Nile encephalitis epidemic in southeastern Romania. Lancet. 1998;352:767-771.
260.2 Japanese Encephalitis
Epidemics of encephalitis were reported in Japan from the late 1800s.
Epidemiology
JE is a mosquito-borne viral disease of humans as well as horses, swine, and other domestic animals that causes human infections and acute disease in a vast area of Asia, northern Japan, Korea, China, Taiwan, Philippines, and the Indonesian archipelago and from Indochina through the Indian subcontinent. Culex tritaeniorhynchus summarosus, a night-biting mosquito that feeds preferentially on large domestic animals and birds but only infrequently on humans, is the principal vector of zoonotic and human JE in northern Asia. A more complex ecology prevails in southern Asia. From Taiwan to India, C. tritaeniorhynchus and members of the closely related Culex vishnui group are vectors. Before the introduction of JE vaccine, summer outbreaks of JE occurred regularly in Japan, Korea, China, Okinawa, and Taiwan. Over the past decade, there has been a pattern of steadily enlarging recurrent seasonal outbreaks in Vietnam, Thailand, Nepal, and India, with small outbreaks in the Philippines, Indonesia, and the northern tip of Queensland, Australia. Seasonal rains are accompanied by increases in mosquito populations and JE transmission. Pigs serve as amplifying host.
The annual incidence in endemic areas ranges from 1-10/10,000 population. Children <15 yr of age are principally affected, with nearly universal exposure by adulthood. The case : infection ratio for JE virus has been variously estimated at 1 : 25 to 1 : 1,000. Higher ratios have been estimated for populations indigenous to enzootic areas. JE occurs in travelers visiting Asia; therefore, a travel history in the diagnosis of encephalitis is critical.
Clinical Manifestations
After a 4- to 14-day incubation period, cases typically progress through the following 4 stages: prodromal illness (2-3 days), acute stage (3-4 days), subacute stage (7-10 days), and convalescence (4-7 wk). Onset may be characterized by abrupt onset of fever, headache, respiratory symptoms, anorexia, nausea, abdominal pain, vomiting, and sensory changes, including psychotic episodes. Grand mal seizures are seen in 10-24% of children with JE; parkinsonian-like nonintention tremor and cogwheel rigidity are seen less frequently. Particularly characteristic are rapidly changing central nervous system signs (e.g., hyperreflexia followed by hyporeflexia or plantar responses that change). The sensory status of the patient may vary from confusion through disorientation and delirium to somnolence, progressing to coma. There is usually a mild pleocytosis (100-1,000 leukocytes/mm3) in the cerebrospinal fluid, initially polymorphonuclear but in a few days predominantly lymphocytic. Albuminuria is common. Fatal cases usually progress rapidly to coma, and the patient dies within 10 days.
Diagnosis
JE should be suspected in patients reporting exposure to night-biting mosquitoes in endemic areas during the transmission season. The etiologic diagnosis of JE is established by testing an acute-phase serum collected early in the illness for the presence of virus-specific IgM antibodies or, alternatively, demonstrating a fourfold or greater increase in IgG antibody titers by testing paired acute and convalescent sera. The virus can also be identified by PCR.
Treatment
There is no specific treatment for JE. The treatment is intensive supportive care (Chapter 62), including control of seizures (Chapter 586).
Prognosis
Patient fatality rates for JE are 24-42% and are highest in children 5-9 yr of age and in adults older than 65 yr. The frequency of sequelae is 5-70% and is directly related to the age of the patient and severity of disease. Sequelae are most common in patients younger than 10 yr at the onset of disease. The more common sequelae are mental deterioration, severe emotional instability, personality changes, motor abnormalities, and speech disturbances.
Prevention
Travelers to endemic countries who plan to be in rural areas of the endemic region during the expected period of seasonal transmission and travelers in rural areas experiencing endemic transmission should receive JE vaccine. An inactivated vaccine manufactured in Japan by intracerebral injection of young mice and available throughout the world has been taken off the market owing to a high incidence of adverse events. In 2008-2009, tissue culture–based JE vaccine (IXIARO) was licensed in Europe, Australia, and the USA. In the USA, this vaccine (also called IC51) is licensed for use in older children and adults and is distributed by Novartis (Basel). For this vaccine, JE virus is grown in Vero cells, then formalin inactivated and administered intramuscularly as two 0.5-mL doses, 28 days apart. The final dose should be completed at least 1 wk prior to the patient’s expected arrival in a JE endemic area. This vaccine contains alum and protamine sulfate and has exhibited only mild adverse events. A highly efficacious live-attenuated single-dose JE vaccine developed in China for children is licensed and marketed in some Asian countries. This vaccine can be co-administered with live-attenuated measles vaccine without altering the immune responses to either vaccine. In humans, prior dengue virus infection provides partial protection from clinical JE.
Personal measures should be taken to reduce exposure to mosquito bites, especially for short-term residents in endemic areas. They consist of avoiding evening outdoor exposure, using insect repellents, covering the body with clothing, and using bed nets or house screening.
Commercial pesticides, widely used by rice farmers in Asia, are effective in reducing populations of C. tritaeniorhynchus. Fenthion, fenitrothion, and phenthoate are effectively adulticidal and larvicidal. Insecticides may be applied from portable sprayers or from helicopters or light aircraft.
Bista MB, Banerjee MK, Shin SH, et al. Efficacy of a single dose of SA 14–14–2 live-attenuated Japanese encephalitis vaccine: a case-control study. Lancet. 2001;358:791-795.
Gatchalian S, Yao Y, Zhou B, et al. Comparison of the immunogenicity and safety of measles vaccine administered alone or with live-attenuated Japanese encephalitis SA 14–14–2 vaccine in Philippine infants. Vaccine. 2008;26:2234-2241.
Innis BL, Nisalak A, Nimmannitya S, et al. An enzyme-linked immunosorbent assay to characterize dengue infections where dengue and Japanese encephalitis co-circulate. Am J Trop Med Hyg. 1989;40:418-427.
Jones T. IC-51, an injectable vaccine for the prevention of Japanese encephalitis virus infection. Curr Opin Mol Ther. 2009;11:90-96.
Schuller E, Jilma B, Voicu V, et al. Long-term immunogenicity of the new Vero cell-derived, inactivated Japanese encephalitis virus vaccine IC-51: six and 12 month results of a multicenter follow-up phase 3 study. Vaccine. 2008;26:4382-4386.
Tandan JB, Ohrr H, Sohn YM, et al. Single dose of SA 14–14–2 vaccine provides long-term protection against Japanese encephalitis: a case-control study in Nepalese children 5 years after immunization. Vaccine. 2007;25:5041-5045.
Tauber E, Kollaritshe H, von Sonnennburg F, et al. Randomized, double blind, placebo-controlled phase 3 trial of the safety and tolerability of IC51, an inactivated Japanese encephalitis vaccine. J Infect Dis. 2008;198:493-499.
260.3 Tick-Borne Encephalitis
TBE was identified by Russian scientists in 1937 and was subsequently shown to be widespread in Europe, where it was identified as the cause of milk-borne encephalitis.
Epidemiology
TBE refers to neurotropic tick-transmitted flaviviral infections occurring across the Eurasian land mass. In the Far East, the disease is called Russian spring-summer encephalitis; the milder, often biphasic form in Europe is simply called tick-borne encephalitis. TBE is found in all countries of Europe except Portugal and the Benelux countries. The incidence is particularly high in Austria, Poland, Hungary, Czech Republic, Slovakia, former Yugoslavia, and Russia. The incidence tends to be very focal. Seroprevalence is as high as 50% in farm and forestry workers. The majority of cases occur in adults, but even young children may be infected while playing in the woods or on picnics or camping trips. The seasonal distribution of cases is midsummer in southern Europe, with a longer season in Scandinavia and the Russian Far East. TBE can be excreted from the milk of goats, sheep, or cows. Before World War II, when milk was consumed unpasteurized, milk-borne cases of TBE were common.
Viruses are transmitted principally by hard ticks of Ixodes ricinus in Europe and Ixodes persulcatus in the Far East. Viral circulation is maintained by a combination of transmission from ticks to birds, rodents, and larger mammals and transtadial transmission from larval to nymphal and adult stages. In some parts of Europe and Russia, ticks feed actively during the spring and early fall, giving rise to the name “spring-summer encephalitis.”
Clinical Manifestations
After an incubation period of 7-14 days, the European form begins as an acute nonspecific febrile illness that is followed in 5-30% of cases by meningoencephalitis. The Far Eastern variety more often results in encephalitis with higher case fatality and sequelae rates. The 1st phase of illness is characterized by fever, headache, myalgia, malaise, nausea, and vomiting for 2-7 days. Fever disappears and after 2-8 days may return accompanied by vomiting, photophobia, and signs of meningeal irritation in children and more severe encephalitic signs in adults. This phase rarely lasts more than 1 wk.
Diagnosis
The diagnosis of TBE should be suspected in any patient reporting a tick bite in an endemic area during the transmission season. The etiologic diagnosis of TBE is established by testing an acute-phase serum collected early in the illness for the presence of virus-specific IgM antibodies or, alternatively, demonstrating a fourfold or greater increase in IgG antibody titers by testing paired acute and convalescent sera. The virus can also be identified by PCR.
Treatment
There is no specific treatment for TBE. The treatment is intensive supportive care (Chapter 62), including control of seizures (Chapter 586).
Prognosis
The main risk for fatal outcome is advanced age; the fatality rate in adults is about 1%, but sequelae in children are rare. Transient unilateral paralysis of an upper extremity is a common finding in adults. Common sequelae include chronic fatigue, headache, sleep disorders, and emotional disturbances.
Prevention
Specific Ig has been given to persons with seasonal tick bite exposure, although efficacy of this preventive therapy is not well studied. Effective inactivated vaccines for human use, made from virus grown in tissue culture, are licensed in Russia and Europe. They are administered in a 3-dose series.
Kluger G, Schottler A, Waldvogel K, et al. Tickborne encephalitis despite specific immunoglobulin prophylaxis. Lancet. 1995;346:1502.
McNeil JG, Lednar WM, Stansfield SK, et al. Central European tick-borne encephalitis: assessment of risk for persons in the armed forces and vacationers. J Infect Dis. 1985;152:650-651.