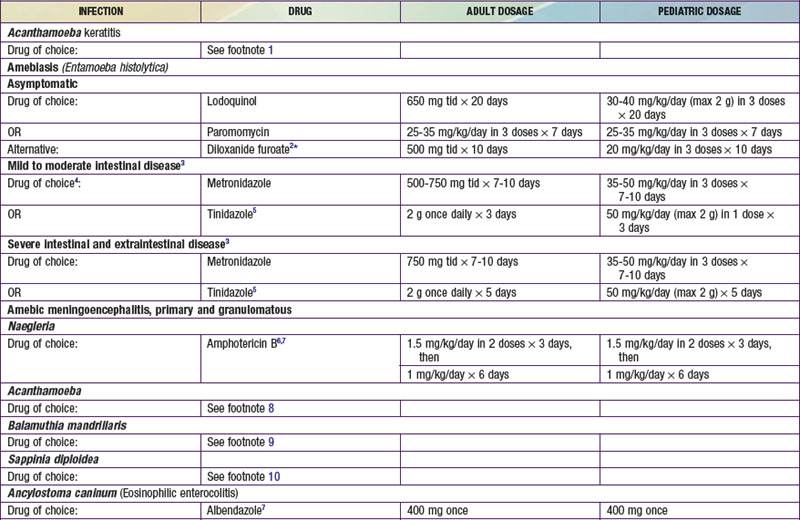
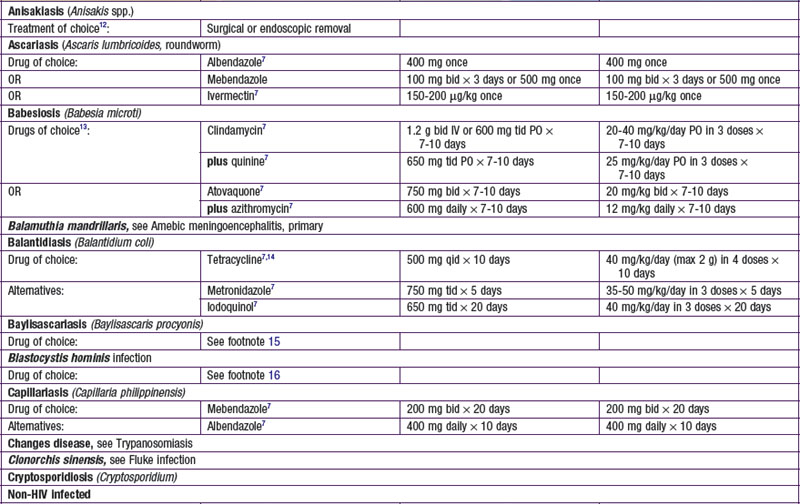
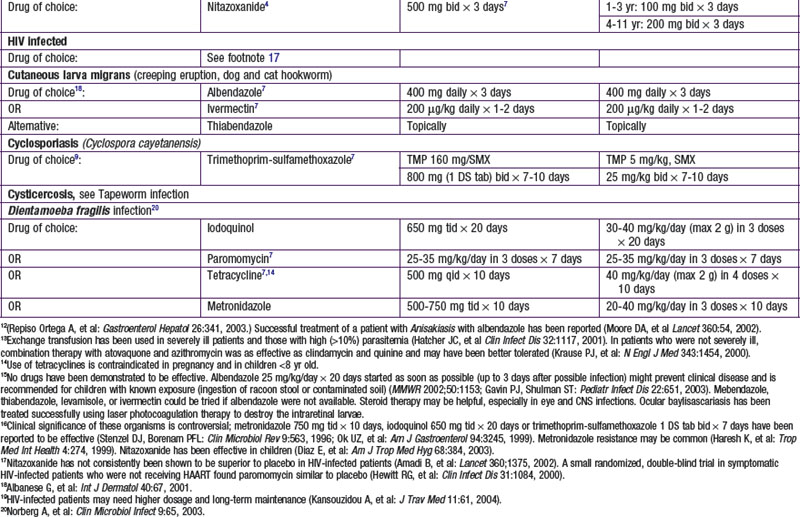
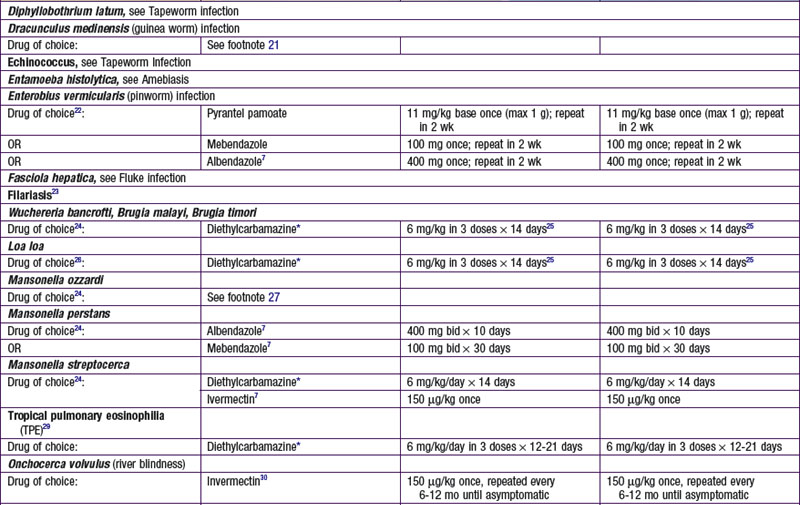
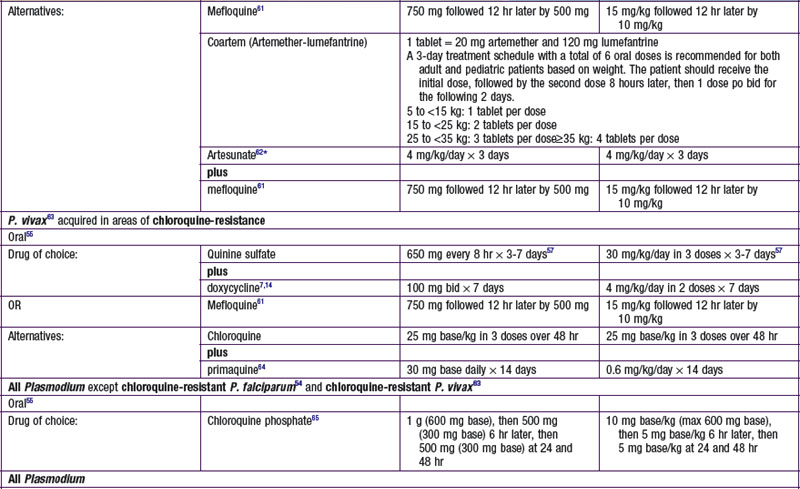
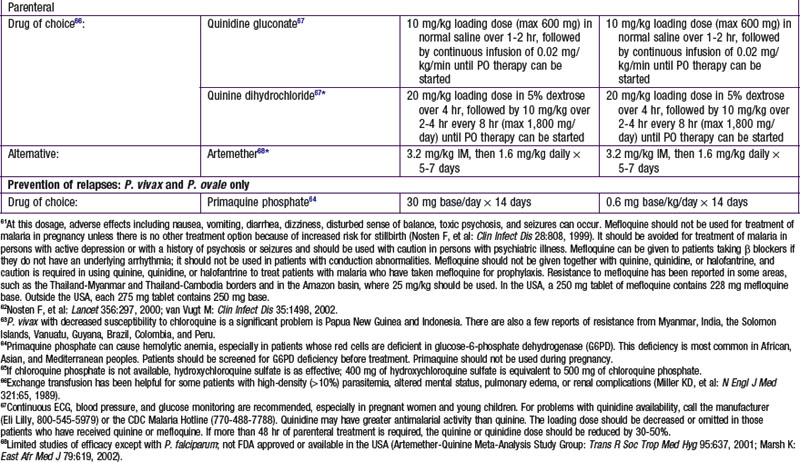
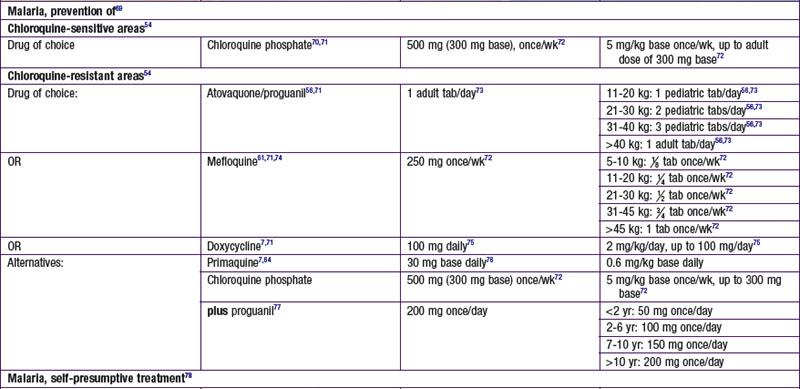
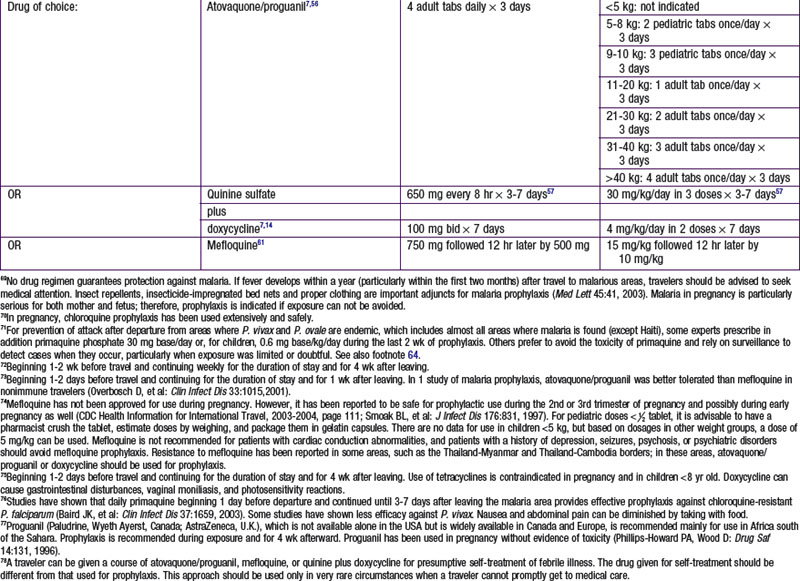
Chapter 271 Principles of Antiparasitic Therapy
Parasites are divided into 2 main groups taxonomically: protozoans, which are unicellular, and helminths, which are multicellular. Chemotherapeutic agents appropriate for 1 group may not be appropriate for the other, and not all drugs are readily available. Some drugs are available only from the manufacturer, some are not available in the USA, and some are available through the Centers for Disease Control and Prevention Drug Service (telephone: 404-639-3670, weekdays; 404-639-2888, evenings, weekends, and holidays) ![]() (see Tables 271-1 and 271-2 on the Nelson Textbook of Pediatrics website at www.expertconsult.com).
(see Tables 271-1 and 271-2 on the Nelson Textbook of Pediatrics website at www.expertconsult.com).
Table 271-2 MANUFACTURERS OF DRUGS USED TO TREAT PARASITIC INFECTIONS
* Not available in the USA; may be available through a compounding pharmacy.
‡ Available under an Investigational New Drug (IND) protocol from the CDC Drug Service, Centers for Disease Control and Prevention, Atlanta, Georgia 30333; 404-639-3670 (evenings, weekends, or holidays: 404-639-2888).
§ Available in the USA only from the manufacturer.
Modifed from Drugs for parasitic infection. Med Lett August 2004, p 12. Available at www.medicalletter.org.
Selected Antiparasitic Drugs for Protozoans
Nitazoxanide
Nitazoxanide is a nitrothiazole benzamide, initially developed as a veterinary anthelmintic. Nitazoxanide inhibits pyruvate-ferredoxin oxidoreductase, which is an enzyme necessary for anaerobic energy metabolism. In humans, nitazoxanide is effective against many protozoans and helminths. In December 2002, the U.S. Food and Drug Administration (FDA) approved the use of nitazoxanide for the treatment of diarrhea caused by Cryptosporidium species in children 1-11 yr of age and by Giardia intestinalis in children ≥1 yr of age.
Nitazoxanide is available as an oral suspension, which has a pink color and strawberry flavor. The bioavailability is doubled with food. The drug is well absorbed from the gastrointestinal tract. One third is excreted in urine, and two thirds is excreted in feces as the active metabolite, tizoxanide. Although in vitro metabolism studies have not demonstrated cytochrome P450 enzyme effects, no pharmacokinetic studies have been performed yet in patients with compromised renal or hepatic function. In addition, no studies have been performed in pregnant or lactating women. Common adverse effects include abdominal pain, diarrhea, and nausea. Rare side effects include anorexia, flatulence, increased appetite, fever, pruritus, and dizziness. Intriguingly, nitazoxanide has been demonstrated to have activity against both hepatitis C and rotavirus, although this use of the agent is investigational.
Tinidazole (Tindamax)
Tinidazole is a synthetic nitroimidazole with a chemical structure similar to metronidazole. It is FDA approved for treatment of trichomoniasis and for giardiasis and amebiasis in children ≥3 yr of age. Its mechanism of action against Trichomonas may be secondary to the generation of free nitro radicals by the protozoan. The mechanism of action against Giardia lamblia and Entamoeba histolytica is unknown. After oral administration, tinidazole is rapidly and completely absorbed and distributes into almost all tissues and body fluids, including crossing the blood-brain barrier and placental barrier. It is excreted via urine and feces. Hemodialysis increases clearance of drug. No studies have been performed for patients undergoing peritoneal dialysis or for patients with compromised hepatic function. Tinidazole carries a pregnancy category C classification and can be detected in breast milk. Breast-feeding should be interrupted during treatment and for 3 days after treatment.
Atovaquone/Proguanil (Malarone)
Atovaquone is a hydroxynaphthoquinone and has been used in the past predominantly against Pneumocystis pneumonia in AIDS patients. Its mechanism of action is via disruption of mitochondria membrane potential through interaction with cytochrome B. Atovaquone can effectively inhibit liver stages of all Plasmodium species.
Proguanil was approved for use in the USA in 1948 for malaria but ceased to be marketed in the 1970s. Its mechanism of action is inhibition of the parasite dihydrofolate reductase enzyme by the active form, cycloguanil. When used alone, it has poor efficacy for prophylaxis.
Proguanil acts in synergy with atovaquone on the cytochrome B enzyme in Plasmodia mitochondria. The exact mechanism of synergy is unknown. In 2000, the FDA approved atovaquone/proguanil for the prevention and treatment of acute, uncomplicated Plasmodium falciparum malaria. Atovaquone alone and in combination with proguanil is the only drug to completely inhibit the liver stage, which provides the advantage of only needing to use the drug for 7 days after departing a malaria-endemic area (compared to several weeks).
Two double-blind, randomized clinical trials assessing malaria prophylaxis demonstrated that atovaquone/proguanil was at least comparable to (and perhaps better than) chloroquine plus proguanil, and that atovaquone/proguanil was comparable to mefloquine. Atovaquone/proguanil was better tolerated than chloroquine plus proguanil and mefloquine. Atovaquone/proguanil treatment of acute uncomplicated P. falciparum infection has demonstrated higher or comparable cure rates when compared with other P. falciparum treatment drugs. Compared with other antimalaria treatment therapies, atovaquone/proguanil treatment has the highest cost.
Artemisinin Derivatives and Artemether/Lumefantrine (Coartem, Artemether, Artesunate)
Artemisinin is a sesquiterpene lactone isolated from the weed Artemisia annua. It was developed in China where it is known as qinghaosu. Artemisinins act very rapidly against Plasmodium vivax as well as chloroquine-sensitive and chloroquine-resistant P. falciparum. Artemisinins are also rapidly eliminated. Emerging resistance to artemisinins has been seen in Cambodia, but not all of Southeast Asia. Coartem is the 1st artemisinin-containing drug approved for use by the FDA. It is a fixed-dose combination of 2 novel antimalarials, artemether 20 mg and lumefantrine 120 mg. It is a highly-effective 3-day malaria treatment with cure rates of over 96% even in areas of multidrug resistance.
Selected Antiparasitic Drugs for Helminths
Albendazole (Albenza)
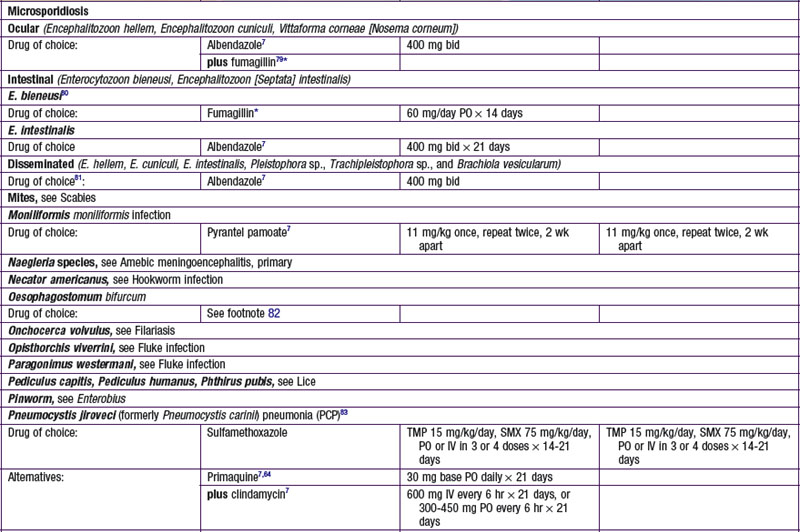
Albendazole is a benzimidazole carbamate structurally related to mebendazole and has similar anthelmintic activity. Its absorption from the gastrointestinal tract is poor but improved with a concomitant high-fat meal. Albendazole sulfoxide, the principal metabolite with anthelmintic activity, has a plasma half-life of 8.5 hr. It is widely distributed in the body, including the bile and cerebrospinal fluid. It is eliminated by bile. Albendazole is FDA approved for treatment of neurocysticercosis and hydatid diseases (Echinococcus granulosus). It is not FDA approved but is used for Ancylostoma caninum, ascariasis, Chinese liver fluke, cutaneous larva migrans, pinworms, filariasis, gnathostomiasis, hookworms, microsporidiosis, and visceral larva migrans. Albendazole is generally well tolerated. Common adverse effects include headache, nausea, vomiting, and abdominal pain. Serious adverse effects include elevated liver enzymes and leukopenia, which have occurred in a few patients with treatment of hydatid disease. Rare adverse effects include acute renal failure, pancytopenia, granulocytopenia, and thrombocytopenia.
Ivermectin (Stromectol, Mectizan)
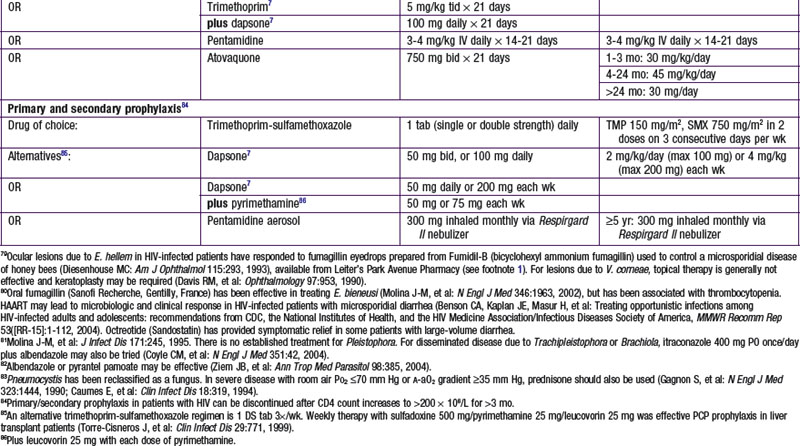
Ivermectin is a semisynthetic derivative of 1 of the avermectins, which is a group of macrocyclic lactones produced by Streptomyces avermitilis. After oral administration, ivermectin has peak plasma concentrations after about 4 hr and a plasma elimination half-life of about 12 hr. It is excreted as metabolites over a 2-wk period via feces. It is FDA approved for treatment of onchocerciasis and intestinal strongyloidiasis. It may have some effect in treating cutaneous larva migrans, intestinal nematode infections, loiasis, lymphatic filariasis, Mansonella infections, and scabies. Combination therapies of ivermectin with albendazole or diethylcarbamazine are being used to treat lymphatic filariasis. Common adverse events include dizziness, headache, pruritus, and gastrointestinal effects. Serious adverse events include Mazzotti reactions, including arthralgia, synovitis, enlarged lymph nodes, rash, and fever secondary to microfilaria death in patients with onchocerciasis.
Praziquantel (Biltricide)
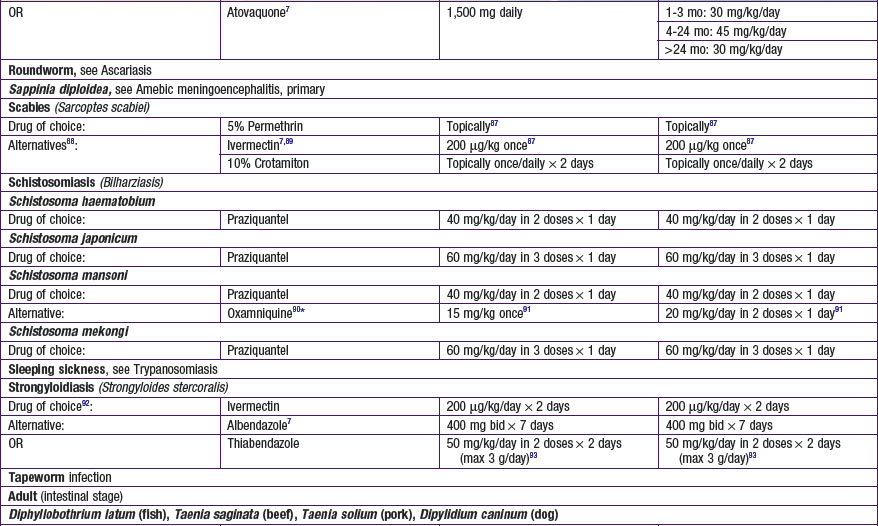
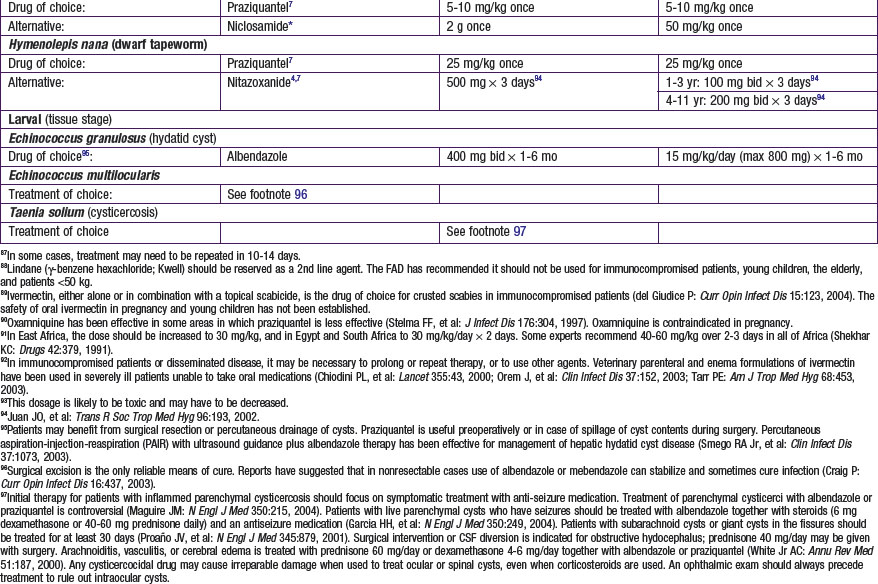
Praziquantel achieves its antiparasitic activity via the pyrazino isoquinoline ring system and was originally synthesized as a potential tranquillizer. After oral administration, praziquantel is rapidly absorbed with peak levels in 1-2 hr and plasma half-life of about 1-3 hr. Elimination via the urine and feces is >80% complete after 24 hr. Praziquantel is metabolized in the liver by the microsomal cytochrome P450 (especially 2B1 and 3A). Bioavailability of praziquantel is increased with concomitant administration of agents that inhibit cytochrome P450. Praziquantel is FDA approved for treatment of Chinese liver fluke, Southeast Asian liver fluke, and schistosomiasis. It is used for treatment of intestinal flukes, North American liver fluke, Nanophyetus salmincola, lung fluke, and tapeworm infections but is not FDA approved for these indications. Adverse effects can be seen in 30-60% of patients, although most are mild and disappear within 24 hr. Common adverse effects include headache, abdominal pain, dizziness, and malaise. Serious but rare adverse effects include arrhythmias, heart block, and convulsions.