Chapter 272 Primary Amebic Meningoencephalitis
Naegleria, Acanthamoeba, and Balamuthia are small, free-living amebae that cause human amebic meningoencephalitis, which has 2 distinct clinical presentations. The more common is an acute, usually fatal amebic meningitis that is caused by Naegleria and occurs in previously healthy children and young adults. Granulomatous amebic meningoencephalitis is caused by Acanthamoeba, Balamuthia, and Sappinia and is a more indolent infection that is more likely to occur in immunocompromised individuals.
Etiology
Naegleria is an ameboflagellate that can exist as cysts, trophozoites, and transient flagellate forms. Temperature and environmental nutrient and ion concentrations are the major factors that determine the stage of the ameba. Trophozoites are the only stages that are invasive, although cysts are potentially infective, because they can convert to the vegetative form very quickly under the proper environmental stimuli. Although there are several species of Naegleria, only N. fowleri has been shown to be pathogenic for humans.
Acanthamoeba exists in cyst and trophozoite forms; only the trophozoite form is invasive. Cases of Acanthamoeba keratitis usually follow incidents of trivial corneal trauma followed by flushing with contaminated tap water. Infections can also occur among contact lens wearers who come in contact with contaminated water during swimming or using contact lenses cleaned or stored in contaminated tap water. Granulomatous amebic encephalitis from Acanthamoeba occurs worldwide and is associated with an immunocompromising condition such as HIV infection, diabetes mellitus, alcoholism, immunosuppressive therapy, or radiation therapy.
Balamuthia mandrillaris has been implicated as an etiology of granulomatous amebic encephalitis. Although the clinical presentation is similar to infection with Acanthamoeba, most patients have no known immunocompromising condition.
Other free-living amebae can also cause infection, as illustrated by case reports of Sappinia diploidea granulomatous encephalitis.
Epidemiology
The free-living amebae have a worldwide distribution. Naegleria species have been isolated from a variety of freshwater sources, including ponds and lakes, domestic water supplies, hot springs and spas, thermal discharge of power plants, groundwater, and occasionally from the nasal passages of healthy children. Acanthamoeba species have been isolated from soil, mushrooms and vegetables, brackish water, and seawater, as well as most of the freshwater sources for Naegleria. It can also be found in tap water, since chlorination does not kill Acanthamoeba. Balamuthia is present in soil and may be transmitted by inhalation or contamination of preexisting skin lesions.
Naegleria meningoencephalitis has been reported from every continent. Most of the cases occur during the summer months in previously healthy individuals who have a history of swimming in or contact with freshwater before their illness. Only 1-2 cases are reported in the USA per year, but 8 cases were reported in 2001-2002, and 6 cases in 2007. Most of the reports have come from the southern and southwestern states, with occasional infections occurring in the Midwest and East.
Pathogenesis
The free-living amebae enter the nasal cavity by inhalation or aspiration of dust or water contaminated with trophozoites or cysts. Naegleria gains access to the central nervous system through the olfactory epithelium and migrates via the olfactory nerve to the olfactory bulbs located in the subarachnoid space and bathed by the cerebrospinal fluid (CSF). This space is richly vascularized and is the route of spread to other areas of the central nervous system. Grossly, there is widespread cerebral edema and hyperemia of the meninges. The olfactory bulbs are necrotic, hemorrhagic, and surrounded by a purulent exudate. Microscopically, the gray matter is the most severely affected, with severe involvement in all cases. Fibrinopurulent exudate may be found throughout the cerebral hemispheres, brainstem, cerebellum, and upper portions of the spinal cord. Pockets of trophozoites may be seen in necrotic neural tissue, usually in the perivascular spaces of arteries and arterioles.
The route of invasion and penetration in cases of granulomatous amebic meningoencephalitis caused by Acanthamoeba and Balamuthia may be by direct spread through olfactory epithelium or hematogenous, from a primary focus in the skin or lungs. Pathologic examination reveals granulomatous encephalitis, with multinucleated giant cells mainly in the posterior fossa structures, basal ganglia, bases of the cerebral hemispheres, and cerebellum. Both trophozoites and cysts may be found in the central nervous system lesions, primarily located in the perivascular spaces and invading blood vessel walls. The olfactory bulbs and spinal cord are usually spared. The single case of Sappinia encephalitis followed a sinus infection, and evaluation revealed a solitary 2 cm temporal lobe mass with mild ring enhancement.
Clinical Manifestations
The incubation of Naegleria infection may be as short as 2 days or as long as 15 days. Symptoms have an acute onset and progress rapidly. Infection is characterized by a sudden onset of severe headache, fever, nausea, and vomiting, signs of meningitis, and then encephalitis. Most cases end in death within 1 wk of onset of symptoms.
Granulomatous amebic meningoencephalitis may occur weeks to months after infection. The presenting signs and symptoms are often those of single or multiple central nervous system space-occupying lesions and include hemiparesis, personality changes, seizures, and drowsiness. Altered mental status is often a prominent symptom. Headache and fever occur only sporadically, but stiff neck is seen in a majority of cases. Palsies of the cranial nerves may be present. There is also 1 report of acute hydrocephalus and fever with Balamuthia. Results of neuroimaging studies of the brain usually demonstrate multiple low-density lesions resembling infarcts or enhancing lesions of granulomas (Fig. 272-1).
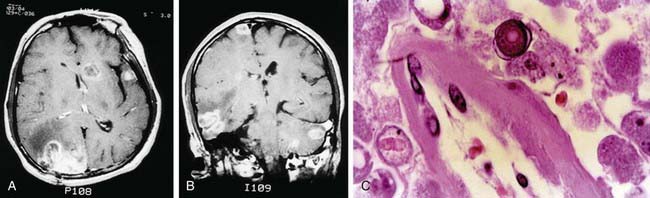
Figure 272-1 A and B, MRIs of the brain of a patient with Balamuthia mandrillaris granulomatous amebic encephalitis. Multiple enhancing lesions are seen in the right hemisphere, left cerebellum, midbrain, and brainstem. C, Photomicrograph of the brain lesion from the same patient showing perivascular amebic trophozoites. A round amebic cyst with a characteristic double wall is seen in the top center (hematoxylin and eosin, original magnification ×100).
(From Deol I, Robledo L, Meza A, et al: Encephalitis due to a free-living amoeba [Balamuthia mandrillaris]: case report with literature review, Surg Neurol 53:611–616, 2000.)
Diagnosis
The CSF in Naegleria infection may mimic that of herpes simplex encephalitis early in the disease and that of acute bacterial meningitis later in the disease, with a neutrophilic pleocytosis, elevated protein level, and hypoglycorrhachia. The amebae, which may be motile, may be seen on a wet mount of the CSF but are often mistaken for lymphocytes. A hanging drop examination of CSF and a strong clinical suspicion early in the course of disease affords the best chance for early treatment and cure. Naegleria can be grown on agar enriched with gram-negative bacteria, on which they feed.
The CSF findings of granulomatous meningoencephalitis resemble those of aseptic meningitis. The isolation and identification of Acanthamoeba from brain tissue and CSF are the best methods of diagnosis. Acanthamoeba may be cultured from the same agar used for growing Naegleria, but Balamuthia must be grown on mammalian cell cultures. Pediatric cases of Balamuthia meningoencephalitis have been diagnosed antemortem by brain biopsy as well as postmortem. Immunofluorescence staining of brain tissue can differentiate Acanthamoeba and Balamuthia.
Treatment
Naegleria infection is nearly always fatal, and early recognition and early treatment are crucial to successful therapy. There are several reports of treatment survivors, most of whom recovered fully. Naegleria infections have been successfully treated with regimens of amphotericin B, rifampin, and fluconazole or ketoconazole; amphotericin B, rifampin, and chloramphenicol; and amphotericin B alone. The early use of dexamethasone may be considered, since steroid treatment was utilized in the few cases of survivors (as well as nonsurvivors). The optimal duration of treatment is unknown.
The optimal therapy for granulomatous amebic meningoencephalitis is also uncertain. Strains of Acanthamoeba isolated from fatal cases are usually susceptible in vitro to pentamidine, ketoconazole, flucytosine, and less so to amphotericin B. One patient was successfully treated with sulfadiazine and fluconazole, and another was successfully treated with intravenous pentamidine followed by oral itraconazole. Acanthamoeba keratitis responds to long courses of topical propamidine–polymyxin B sulfate or topical polyhexamethylene biguanide or chlorhexidine gluconate, and antifungal azoles plus topical steroids. Limited success has been demonstrated in Balamuthia infection with systemic azole therapy combined with flucytosine. More recently, the combination of flucytosine, pentamidine, fluconazole, sulfadiazine, a macrolide, and phenothiazines resulted in the survival of 2 patients with Balamuthia meningoencephalitis, although both were left with some neuromotor and cognitive impairment. Corticosteroids prior to initiating effective therapy appear to have a detrimental effect, contributing to rapid progression of disease.
Bennett WM, Nespral JF, Rosson MW, et al. Use of organs for transplantation from a donor with primary meningoencephalitis due to Naegleria fowleri. Am J Transplant. 2008;8:1334-1335.
Centers for Disease Control and Prevention. Balamuthia amebic encephalitis—California, 1999–2007. MMWR Morb Mortal Wkly Rep. 2008;57:768-771.
Centers for Disease Control and Prevention. Primary amebic meningoencephalitis—Arizona, Florida, and Texas, 2007. MMWR Morb Mortal Wkly Rep. 2008;57:573-577.
Deetz TR, Sawyer MH, Billman G, et al. Successful treatment of Balamuthia amoebic encephalitis: presentation of 2 cases. Clin Infect Dis. 2003;37:1304-1312.
Gelman BB, Popov V, Chaljub G, et al. Neuropathological and ultrastructural features of amebic encephalitis caused by Sappinia diploidea. J Neuropathol Exp Neurol. 2003;62:990-998.
Radford CF, Minassian DC, Dart J. Acanthamoeba keratitis in England and Wales: incidence, outcome, and risk factors. Br J Ophthalmol. 2002;86:536-542.
Schuster FL, Visvesvara GS. Opportunistic amoebae: challenges in prophylaxis and treatment. Drug Resist Updat. 2004;7:41-51.
Schuster FL, Visvesvara GS. Free-living amoebae as opportunistic and non-opportunistic pathogens of humans and animals. Int J Parasitol. 2004;34:1001-1027.
Vargas-Zepeda J, Gomez-Alcala AV, Vasquez-Morales JA, et al. Successful treatment of Naegleria fowleri meningoencephalitis by using intravenous amphotericin B, fluconazole and rifampicin. Arch Med Res. 2005;36:83-86.