Chapter 316 Eosinophilic Esophagitis and Non-GERD Esophagitis
Eosinophilic Esophagitis
The esophageal epithelium in eosinophilic esophagitis (EoE) is infiltrated by eosinophils, typically in a density exceeding 15 per high-power field (hpf). Presenting symptoms include vomiting, feeding problems, chest or epigastric pain, and dysphagia with occasional food impactions or strictures. Most patients are male. The mean age at diagnosis is 7 yr (range 1-17 yr), and the duration of symptoms is 3 yr. Most patients have other atopic diseases and associated food allergies; laboratory abnormalities can include peripheral eosinophilia and elevated immunoglobulin E (IgE) levels. Endoscopically, the esophagus presents a granular, furrowed, ringed, or exudative appearance (Fig. 316-1); esophageal histology reveals eosinophilia, with cut-points for diagnosis variably chosen at 15-20/hpf. Up to 30% children with EoE have grossly normal esophageal mucosa. EoE is differentiated from gastroesophageal reflux disease (GERD) by its general lack of erosive esophagitis, by its greater eosinophil density, and by its refractoriness to antireflux therapies. Gastroesophageal reflux disease may be an important coexisting diagnosis. Evaluation of EoE should include a thorough search for food and environmental allergies via skin prick (IgE mediated) and patch (non–IgE mediated) tests.
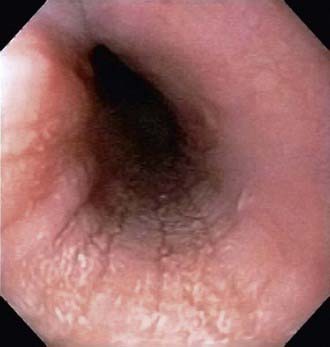
Figure 316-1 Endoscopic image of eosinophilic esophagitis with characteristic mucosal appearance of furrowing and white specks.
Treatment involves dietary restrictions that take one of 3 forms: elimination diets guided by circumstantial evidence and food allergy test results; “six food elimination diet” removing the major food allergens (milk, soy, wheat, egg, peanuts and tree nuts, seafood); and elemental diet composed exclusively of an amino acid–based formula. Successful clinical and histologic remission is observed in 70-98% patients. Topical and systemic corticosteroids have been used successfully for nonresponders and for nonallergic (“primary”) EoE, with symptomatic and histologic remission rates reaching 90%. Therapies under investigation include anti–interleukin 5 (anti-IL-5) antibody (mepolizumab, reslizumab). Little is yet known about its natural history, but it seems that EoE is a chronic remitting and relapsing disorder with a potential for complications such as stricture formation.
Infective Esophagitis
Uncommon, and most often affecting immunocompromised children, infective esophagitis is caused by fungal agents, such as Candida and Torulopsis glabrata; viral agents, such as herpes simplex, cytomegalovirus, HIV, and varicella zoster; and, rarely, bacterial infections, including diphtheria and tuberculosis. The typical presenting signs and symptoms are odynophagia, dysphagia, and retrosternal pain; there may also be fever, nausea, and vomiting. Esophageal candidiasis manifests as concurrent oropharyngeal infection in 11% of patients, and it can affect immunocompetent and immunocompromised children. Esophageal viral infections can also manifest in immunocompetent hosts as an acute febrile illness. Infectious esophagitis, like other forms of esophageal inflammation, occasionally progresses to esophageal stricture. Diagnosis of infectious esophagitis is made by endoscopy (ulcerations, exudates) and histopathologic examination; adding polymerase chain reaction, tissue-viral culture, and immunocytochemistry enhances the diagnostic sensitivity and precision. Treatment is with appropriate antimicrobial agents, analgesics, and antacids.
“Pill” Esophagitis
This acute injury is produced by contact with a damaging agent. Medications implicated in “pill” esophagitis include tetracycline, potassium chloride, ferrous sulfate, nonsteroidal anti-inflammatory medications, and alendronate. Most often the offending tablet is ingested at bedtime with inadequate water. This practice often produces acute discomfort followed by progressive retrosternal pain, odynophagia, and dysphagia. Endoscopy shows a focal lesion often localized to one of the anatomic narrowed regions of the esophagus or to an unsuspected pathologic narrowing. Treatment is supportive; lacking much evidence, antacids, topical anesthetics, and bland or liquid diets are often used.
Abid S, Mumtaz K, Jafri W, et al. Pill induced esophagitis: endoscopic features and clinical outcomes. Endoscopy. 2005;37:740-744.
Atkins D, Kramer R, Capocelli K, et al. Eosinophilic esophagitis; the newest esophageal inflammatory disease. Nat Rev Gastroenterol Hepatol. 2009;6:267-278.
Canalejo E, Duran FG, Cabello N, et al. Herpes esophagitis in healthy adults and adolescents. Medicine. 2010;89(4):204-210.
Franciosi JP, Liacouras CA. Eosinophilic esophagitis. Immunol Allergy Clin N Am. 2009;29:19-27.
Kliemann DA, Pasqualotto AC, Falavigna M, et al. Candida esophagitis: species distribution and risk factors for infection. Rev Inst Med Trop Sao Paulo. 2008;50:261-263.
Lee B, Caddy G. A rare cause of dysphagia: herpes simplex esophagitis. World J Gastroenterol. 2007;13:2756-2757.
Lee JJ, Baker RD, Khan AR, Baker SS. Childhood esophagitis: then and now. J Pediatr Gastroenterol Nutr. 2009;48:37-40.
Pentiuk S, Putnam PE, Collinc MH, Rothenberg ME. Dissociation between symptoms and histological severity in pediatric eosinophilic esophagitis. J Pediatr Gastroenterol Nutr. 2009;48:152-160.
Spergel JM, Brown-Whitehorn TF, Beausoleil JL, et al. 14 years of eosinophilic esophagitis: clinical features and prognosis. J Pediatr Gastroenterol Nutr. 2009;48:30-36.