Chapter 327 Peptic Ulcer Disease in Children
Peptic ulcer disease, the end result of inflammation due to an imbalance between cytoprotective and cytotoxic factors in the stomach and duodenum, manifests with varying degrees of gastritis or frank ulceration. The pathogenesis of peptic ulcer disease is multifactorial, but the final common pathway for the development of ulcers is the action of acid and pepsin-laden contents of the stomach on the gastric and duodenal mucosa and the inability of mucosal defense mechanisms to allay those effects. Abnormalities in the gastric and duodenal mucosa can be visualized on endoscopy, with or without histologic changes. Deep mucosal lesions that disrupt the muscularis mucosa of the gastric or duodenal wall define peptic ulcers. Gastric ulcers are generally located on the lesser curvature of the stomach, and 90% of duodenal ulcers are found in the duodenal bulb. Despite the lack of large population-based pediatric studies, rates of peptic ulcer disease in childhood appear to be low. Large pediatric centers anecdotally report an incidence of 5-7 children with gastric or duodenal ulcers per 2,500 hospital admissions each year.
Ulcers in children can be classified as primary peptic ulcers, which are chronic and more often duodenal, or secondary, which are usually more acute in onset and are more often gastric (Table 327-1). Primary ulcers are most often associated with Helicobacter pylori infection; idiopathic primary peptic ulcers account for up to 20% of duodenal ulcers in children. Secondary peptic ulcers can result from stress due to sepsis, shock, or an intracranial lesion (Cushing ulcer) or in response to a severe burn injury (Curling ulcer). Secondary ulcers are often the result of using aspirin or nonsteroidal anti-inflammatory drugs (NSAIDs); hypersecretory states like Zollinger-Ellison syndrome (Chapter 327.1), short bowel syndrome, and systemic mastocytosis are rare causes of peptic ulceration.
Table 327-1 ETIOLOGIC CLASSIFICATION OF PEPTIC ULCERS
NSAID, nonsteroidal anti-inflammatory drug.
* Requires search for other specific causes.
From Vakil N, Megraud F: Eradication therapy for Helicobacter pylori, Gastroenterology 133:985–1001, 2007.
Pathogenesis
Acid Secretion
By 3-4 yr of age, gastric acid secretion approximates adult values. Acid initially secreted by the oxyntic cells of the stomach has a pH of ∼0.8, whereas the pH of the stomach contents is 1-2. Excessive acid secretion is associated with a large parietal cell mass, hypersecretion by antral G cells, and increased vagal tone, resulting in increased or sustained acid secretion in response to meals and increased secretion during the night. Control of acid secretion is achieved through multiple different feedback mechanisms involving endocrine, paracrine, and neural pathways. The secretagogues that promote gastric acid production include acetylcholine released by the vagus nerve, histamine secreted by enterochromaffin cells, and gastrin released by the G cells of the antrum. Mediators that decrease gastric acid secretion and enhance protective mucin production include prostaglandins.
Mucosal Defense
A continuous layer of mucous gel that serves as a diffusion barrier to hydrogen ions and other chemicals covers the gastrointestinal (GI) mucosa. Mucus production and secretion are stimulated by prostaglandin E2. Underlying the mucous coat, the epithelium forms a second-line barrier, the characteristics of which are determined by the biology of the epithelial cells and their tight junctions. Another important function of epithelial cells is to secrete chemokines when threatened by microbial attack. Secretion of bicarbonate into the mucous coat, which is regulated by prostaglandins, is important for neutralization of hydrogen ions. If mucosal injury occurs, active proliferation and migration of mucosal cells occurs rapidly, driven by epithelial growth factor, transforming growth factor-α, insulin-like growth factor, gastrin, and bombesin, and covers the area of epithelial damage.
Clinical Manifestations
The presenting symptoms of peptic ulcer disease vary with the age of the patient. Hematemesis or melena is reported in up to half of the patients with peptic ulcer disease. School-aged children and adolescents more commonly present with epigastric pain and nausea, presentations generally seen in adults. Dyspepsia, epigastric abdominal pain or fullness, is seen in older children. Infants and younger children usually present with feeding difficulty, vomiting, crying episodes, hematemesis, or melena. In the neonatal period, gastric perforation can be the initial presentation.
The classic symptom of peptic ulceration, epigastric pain alleviated by the ingestion of food, is present only in a minority of children. Many pediatric patients present with poorly localized abdominal pain, which may be periumbilical. The vast majority of patients with periumbilical or epigastric pain or discomfort do not have a peptic ulcer, but rather a functional GI disorder, such as irritable bowel syndrome or nonulcer (functional) dyspepsia. Patients with peptic ulceration rarely present with acute abdominal pain from perforation or symptoms and signs of pancreatitis from a posterior penetrating ulcer. Occasionally, bright red blood per rectum may be seen if the rate of bleeding is brisk and the intestinal transit time is short. Vomiting can be a sign of gastric outlet obstruction.
The pain is often described as dull or aching, rather than sharp or burning, as in adults. It can last from minutes to hours; patients have frequent exacerbations and remissions lasting from weeks to months. Nocturnal pain waking the child is common in older children. A history of typical ulcer pain with prompt relief after taking antacids is found in <33% of children. Rarely, in patients with acute or chronic blood loss, penetration of the ulcer into the abdominal cavity or adjacent organs produces shock, anemia, peritonitis, or pancreatitis. If inflammation and edema are extensive, acute or chronic gastric outlet obstruction can occur.
Diagnosis
Esophagogastroduodenoscopy is the method of choice to establish the diagnosis of peptic ulcer disease. It can be safely performed in all ages by experienced pediatric gastroenterologists. Endoscopy allows the direct visualization of esophagus, stomach, and duodenum, identifying the specific lesions. Biopsy specimens must be obtained from the esophagus, stomach, and duodenum for histologic assessment as well as to screen for the presence of H. pylori infection. Endoscopy also provides the opportunity for hemostatic therapy including injection and the use of a heater probe or electrocoagulation if necessary. Fecal enzyme immunoassay tests for H. pylori are available and have varying utility in children.
Primary Ulcers
Helicobacter Pylori Gastritis
H. pylori is among the most common bacterial infections in humans. H. pylori is a gram-negative, S-shaped rod that produces urease, catalase, and oxidase, which might play a role in the pathogenesis of peptic ulcer disease. The mechanism of acquisition and transmission of H. pylori is unclear, although the most likely mode of transmission is fecal-oral or oral-oral. Viable H. pylori organisms can be cultured from the stool or vomitus of infected patients. Risk factors such as low socioeconomic status in childhood or affected family members also influence the prevalence. All children infected with H. pylori develop histologic chronic active gastritis but are often asymptomatic. In children, H. pylori infection can manifest with abdominal pain or vomiting and, less often, refractory iron deficiency anemia or growth retardation. H. pylori can be associated, though rarely, with chronic autoimmune thrombocytopenia. Chronic colonization with H. pylori can predispose children to a significantly increased risk of developing a duodenal ulcer, gastric cancer such as adenocarcinoma, or MALT (mucosa-associated lymphoid tissue) lymphomas. The relative risk of gastric carcinoma is 2.3-8.7 times greater in infected adults as compared to uninfected subjects. H. pylori is classified by the World Health Organization as a group I carcinogen.
Anemia, idiopathic thrombocytopenic purpura, short stature, and sudden infant death syndrome (SIDS) have also been reported as extragastric manifestations of H. pylori infection. In one published study, H. pylori infection has been correlated with cases of SIDS, but there is no evidence to suggest that H. pylori plays a role in the pathogenesis of SIDS.
The diagnosis of H. pylori infection is made histologically by demonstrating the organism in the biopsy specimens (Fig. 327-1). Although serologic assays using validated immunoglobulin G (IgG) antibody detection may be helpful for screening children for the presence of H. pylori, they do not help predict active infection or assess the success of antimicrobial eradication therapy. 13C-urea breath tests and stool antigen tests are also noninvasive methods of detecting H. pylori infection. Nonetheless, for children with suspected H. pylori infection, an initial upper endoscopy is recommended to evaluate and confirm H. pylori disease. The range of endoscopic findings in children with H. pylori infection varies from being grossly normal to the presence of nonspecific gastritis with prominent rugal folds, nodularity (Fig. 327-2), or ulcers. Because the antral mucosa appears to be endoscopically normal in a significant number of children with primary H. pylori gastritis, gastric biopsies should always be obtained from the body and antrum of the stomach regardless of the endoscopic appearance. If H. pylori is identified, even in a child with no symptoms, eradication therapy should be offered (Tables 327-2 and 327-3).
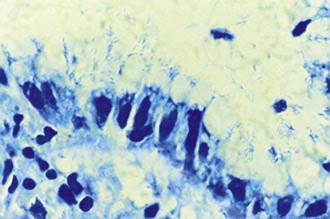
Figure 327-1 Appearance of Helicobacter pylori on the gastric mucosal surface with Giemsa stain (high-power view).
(From Campbell DI, Thomas JE: Heliobacter pylori infection in paediatric practice, Arch Dis Child Edu Pract Ed 90:ep25–ep30, 2005.)
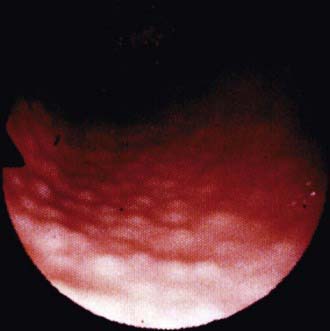
Figure 327-2 Endoscopic view of lymphoid modular hyperplasia of the gastric antrum.
(From Campbell DI, Thomas JE: Heliobacter pylori infection in paediatric practice, Arch Dis Child Edu Pract Ed 90:ep25–ep30, 2005.)
Table 327-2 RECOMMENDED ERADICATION THERAPIES FOR H. PYLORI–ASSOCIATED DISEASE IN CHILDREN
| MEDICATIONS | DOSE | DURATION OF TREATMENT |
|---|---|---|
| Amoxicillin | 50 mg/kg/day in 2 divided doses | 14 days |
| Clarithromycin | 15 mg/kg/day in 2 divided doses | 14 days |
| Proton pump inhibitor | 1 mg/kg/day in 2 divided doses | 1 mo |
| or | ||
| Amoxicillin | 50 mg/kg/day in 2 divided doses | 14 days |
| Metronidazole | 20 mg/kg/day in 2 divided doses | 14 days |
| Proton pump inhibitor | 1 mg/kg/day in 2 divided doses | 1 mo |
| or | ||
| Clarithromycin | 15 mg/kg/day in 2 divided doses | 14 days |
| Metronidazole | 20 mg/kg/day in 2 divided doses | 14 days |
| Proton pump inhibitor | 1 mg/kg/day in 2 divided doses | 1 mo |
Adapted from Gold BD, Colletti RB, Abbott M, et al: Medical position statement: The North American Society for Pediatric Gastroenterology and Nutrition. Helicobacter pylori infection in children: recommendations for diagnosis and treatment, J Pediatr Gastroenterol Nutr 31:490–497, 2000.
Table 327-3 ANTISECRETORY THERAPY WITH PEDIATRIC DOSAGES
| MEDICATION | PEDIATRIC DOSE | HOW SUPPLIED |
|---|---|---|
| H2 RECEPTOR ANTAGONISTS | ||
| Cimetidine | 20-40 mg/kg/day Divided 2 to 4 × a day |
Syrup: 300 mg/mL Tablets: 200, 300, 400, 800 mg |
| Ranitidine | 4-10 mg/kg/day Divided 2 or 3 × a day |
Syrup: 75 mg/5 mL Tablets: 75, 150, 300 mg |
| Famotidine | 1-2 mg/kg/day Divided twice a day |
Syrup: 40 mg/5 mL Tablets: 20, 40 mg |
| Nizatidine | 10 mg/kg/day Divided twice a day |
|
| PROTON PUMP INHIBITORS | ||
| Omeprazole | 1.0-3.3 mg/kg/day <20 kg: 10 mg/day >20 kg: 20 mg/day Approved for use in those >2 yr old |
Capsules: 10, 20, 40 mg |
| Lansoprazole | 0.8-4 mg/kg/day <30 kg: 15 mg/day >30 kg: 30 mg/day Approved for use in those >1 yr old |
Capsules: 15, 30 mg Powder packet: 15, 30 mg Solu-tab: 15, 30 mg |
| Rabeprazole | Adult dose: 20 mg/day | Tablet: 20 mg |
| Pantoprazole | Adult dose: 40 mg/day | Tablet: 40 mg |
| CYTOPROTECTIVE AGENTS | ||
| Sucralfate | 40-80 mg/kg/day | Suspension: 1,000 mg/5 mL Tablet: 1,000 mg |
Idiopathic Ulcers
H. pylori–negative duodenal ulcers in children who have no history of taking NSAIDs represent 15-20% of pediatric duodenal ulcers. These patients do not have nodularity in the gastric antrum or histologic evidence of gastritis. In idiopathic ulcers, acid suppression alone is the preferred effective treatment. Either proton pump inhibitors (PPIs) or H2 receptor antagonists may be used. Idiopathic ulcers have a high recurrence rate after discontinuing antisecretory therapy. These children should be followed closely, and if symptoms recur, antisecretory therapy should be restarted. In such cases, if the child is older than 1 yr, PPIs are preferred for maintenance therapy, because they have been shown to be superior to H2 receptor antagonists in preventing recurrent ulcers.
Secondary Ulcers
Aspirin and Other Nonsteroidal Anti-inflammatory Drugs
Nonsteroidal anti-inflammatory drugs (NSAIDs) produce mucosal injury by direct local irritation and by inhibiting cyclooxygenase and prostaglandin formation. Prostaglandins enhance mucosal resistance to injury; therefore, a decrease in prostaglandin production increases the risk of mucosal injury. The severe erosive gastropathy produced by NSAIDs can ultimately result in bleeding ulcers or gastric perforations. The location of these ulcers is more commonly in the stomach than in the duodenum, and usually in the antrum.
“Stress” Ulceration
Stress ulceration usually occurs within 24 hr of onset of a critical illness in which physiologic stress is present. In many cases, the patients bleed from gastric erosions, rather than ulcers. Approximately 25% of the critically ill children in a pediatric intensive care unit (PICU) have macroscopic evidence of gastric bleeding. Preterm and term infants in the neonatal intensive care unit can also develop gastric mucosal lesions and can present with upper GI bleeding or perforated ulcers. Although prophylactic measures to prevent stress ulcers in children are not standardized, drugs that inhibit gastric acid production (see later) are often used in the PICU to reduce the rate of gastric erosions or ulcers.
Treatment
The management of acute hemorrhage includes serial monitoring of pulse, blood pressure, and hematocrit to insure hemodynamic stability and avoid significant anemia. Normal saline can be used to resuscitate a patient who has poor intravascular volume status. This can be followed by packed red blood cell transfusions for significant symptomatic anemia. The patient’s blood should be typed and cross matched, and a large-bore catheter should be placed for fluid or blood replacement. A nasogastric tube should be placed to determine if the bleeding has stopped. Significant anemia can occur after fluid resuscitation due to equilibration or continued blood loss (which can also cause shock). Fortunately, most acute peptic ulcer bleeding stops spontaneously.
Patients with suspected peptic ulcer hemorrhage should receive high-dose intravenous PPI therapy, which lowers the risk of rebleeding. Some centers also use octreotide, which lowers splanchnic blood flow and gastric acid production.
Once the patient is hemodynamically stable, endoscopy may be indicated to identify the source of bleeding and treat a potential bleeding site. Methods used for vessel hemostasis include pressure, laser, thermal, or electric coagulation; clips; bands; and injections (epinephrine, saline).
Ulcer therapy has two goals: ulcer healing and elimination of the primary cause. Other important considerations are relief of symptoms and prevention of complications. The first-line drugs for the treatment of gastritis and peptic ulcer disease in children are H2 receptor antagonists and PPIs (see Table 327-3). PPIs are more potent in ulcer healing. Cytoprotective agents can also be used as adjunct therapy if mucosal lesions are present. Antibiotics in combination with a PPI must be used for the treatment of H. pylori-associated ulcers (see Table 327-2).
H2 receptor antagonists (cimetidine, ranitidine, famotidine, nizatidine) competitively inhibit the binding of histamine at the H2 subtype receptor of the gastric parietal cell. PPIs block the gastric parietal cell H+/K+ATPase pump in a dose-dependent fashion, reducing basal and stimulated gastric acid secretion. At least 5 PPIs are available in the United States: omeprazole, lansoprazole, pantoprazole, esomeprazole, and rabeprazole. Although not all of them are approved for use in children, they are well tolerated with only minor adverse effects, such as diarrhea (1-4%), headache (1-3%), and nausea (1%). When one considers therapeutic efficacy, the evidence suggests that all PPIs have comparable efficacy in treatment of peptic ulcer disease using standard doses and are superior to H2-receptor antagonists. PPIs have their greatest effect when given before a meal.
Treatment of H. Pylori–Related Peptic Ulcer Disease
In pediatrics, antibiotics and bismuth salts have been used in combination with PPIs to treat H. pylori infection (see Table 327-2). Eradication rates in children range from 68% to 92% when the dual or triple therapy is used for 4-6 wk. The ulcer healing rate ranges from 91% to 100%. Triple therapy yields a higher cure rate than dual therapy. The optimal regimen for the eradication of H. pylori infection in children has yet to be established, but the use of a PPI in combination with clarithromycin and amoxicillin or metronidazole for 2 wk is a well-tolerated and recommended triple therapy (see Table 327-2). Although children <5 yr of age can become reinfected, the most common reason for treatment failure is poor compliance or antibiotic resistance. H. pylori has become more resistant to clarithromycin or metronidazole due to the extensive use of these antibiotics for other infections. In the case of resistant H. pylori infection, sequential treatment or rescue therapy with different antibiotics is acceptable options. The sequential treatment regimen is a 10-day treatment consisting of a PPI and amoxicillin (both twice daily) administered for the first 5 days followed by triple therapy consisting of a PPI, clarithromycin, and metronidazole for the remaining 5 days. Levofloxacin, rifabutin, or furazolidone can be used with amoxicillin and bismuth as a rescue therapy depending on the age of the patient. Fidaxomicin has equivalent efficacy to vancomycin in adults. Knowledge of the community’s H. pylori resistance pattern to clarithromycin or metronidazole might help chose the initial or rescue therapy.
Surgical Therapy
Since the discovery of H. pylori and the availability of modern medical management, peptic ulcer disease requiring surgical treatment has become extremely rare. The indications for surgery remain uncontrolled bleeding, perforation, and obstruction. Since the introduction of H2 receptor antagonists, the recognition and treatment of H. pylori, and the use of PPIs, the incidence of surgery for bleeding and perforation has decreased dramatically.
Calvet X, Lario S, Ramierez-Lazaro M, et al. Comparative accuracy of 3 monoclonal stool tests for diagnosis of Helicobacter pylori infection among patients with dyspepsia. Clin Infect Dis. 2010;50:323-328.
Campbell DI, Thomas JE. Heliobacter pylori infection in paediatric practice. Arch Dis Child Edu Pract Ed. 2005;90:ep25-ep30.
Chan FKL. Proton-pump inhibitors in peptic ulcer disease. Lancet. 2008;372:1198-1200.
Czinn SJ. Heliobacter pylori infection: detection, investigation, and management. J Pediatr. 2005;146:S21-S26.
Drumm B, Day AS, Gold B, et al. Helicobacter pylori and peptic ulcer: Working Group Report of the Second World Congress of Pediatric Gastroenterology, Hepatology, and Nutrition. J Pediatr Gastroenterol Nutr. 2004;39:S626-S631.
Gatta L, Vakil N, Leandro G, et al. Sequential therapy or triple therapy for Helicobacter pylori infection: Systematic review and meta-analysis of randomized controlled trials in adults and children. Am J Gastroenterol. 2009;104:3069-3079.
Gralnek IM, Barkun AN, Bardou M. Management of acute bleeding from a peptic ulcer. N Engl J Med. 2008;359:928-936.
Jafri NS, Hornung CA, Howden CW. Meta-analysis: sequential therapy appears superior to standard therapy for Helicobacter pylori infection in patients naïve to treatment. Ann Intern Med. 2008;148:923-931.
Kato S, Sherman PM. What is new related to Heliocobacter pylori infection in children and teenagers? Arch Pediatr Adolesc Med. 2005;159:415-421.
Lee BH, Kim N. Quadruple or triple therapy to eradicate H pylori. Lancet. 2011;377:877-878.
Malfertheiner P, Chan FKL, McCall KEL. Peptic ulcer disease. Lancet. 2009;374:1449-1458.
The Medical Letter. Treatment of Clostridium difficile infection. Med Lett. 2011;53(1358):14-15.
Sabbi T, De Angelis P, Colistro F, et al. Efficacy of noninvasive tests in the diagnosis of Helicobacter pylori infection in pediatric patients. Arch Pediatr Adolesc Med. 2005;159:238-241.
Shah R. Dyspepsia and Helicobacter pylori. BMJ. 2007;334:41-43.
Stray-Pedersen A, et al. Helicobacter pylori antigen in stool is associated with SIDS and sudden infant deaths due to infectious diseases. Pediatr Res. 2008;64:405-410.
Subramanian V, Hawkey CJ. Assessing bleeds clinically: what’s the score? Lancet. 2009;373:5-7.
327.1 Zollinger-Ellison Syndrome
Zollinger-Ellison syndrome is a rare syndrome characterized by refractory, severe peptic ulcer disease caused by gastric hypersecretion due to the autonomous secretion of gastrin by a neuroendocrine tumor, a gastrinoma. Clinical presentations are similar to those of peptic ulcer disease with the addition of diarrhea. The diagnosis is suspected by the presence of recurrent, multiple, or atypically located ulcers. More than 98% of patients have elevated fasting gastrin levels. Zollinger-Ellison syndrome is common in patients with multiple endocrine neoplasia (MEN1) and rare with neurofibromatosis and tuberous sclerosis. Prompt and effective management of increased gastric acid secretion is essential in the management. PPIs are the drug of choice due to their long duration of action and potency. H2 receptor antagonists are also effective, but higher doses are required than those used in peptic ulcer disease.