Chapter 409 Chylothorax
Chylothorax is a pleural collection of fluid formed by the escape of chyle from the thoracic duct or lymphatics into the thoracic cavity.
Etiology
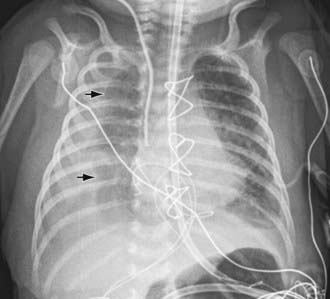
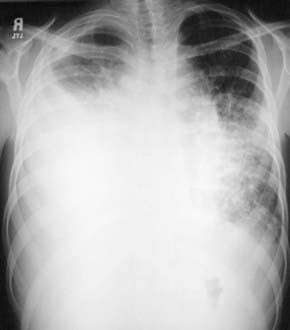
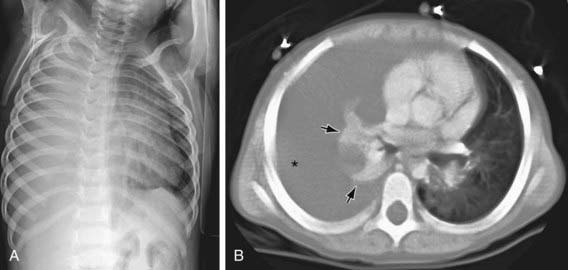
Chylothorax in children occurs most frequently because of thoracic duct injury as a complication of cardiothoracic surgery (Fig. 409-1). Other cases are associated with chest injury (Fig. 409-2) or with primary or metastatic intrathoracic malignancy (Fig. 409-3), particularly lymphoma. In newborns, rapidly increased venous pressure during delivery may lead to thoracic duct rupture. Less common causes include lymphangiomatosis; restrictive pulmonary diseases; thrombosis of the duct, superior vena cava, or subclavian vein; tuberculosis or histoplasmosis; and congenital anomalies of the lymphatic system (Fig. 409-4). Refractory chylothorax in the fetus has been associated with a missense mutation in integrin aα9. Chylothorax can occur in trauma and child abuse (Chapter 37). It is important to establish the etiology, because treatment varies with the cause. In some patients, no specific cause is identified.
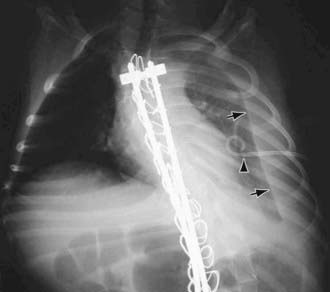
Figure 409-2 Left chylothorax (arrows) following spinal fusion with Harrington rods. It is postulated that the thoracic duct was injured during spine surgery. The pigtail chest tube (arrowhead) needed to be retracted to better drain the effusion.
Clinical Manifestations
The signs and symptoms of chylothorax are the same as those due to pleural effusion of similar size. Chyle is not irritating, so pleuritic pain is uncommon. Onset is often gradual. However, after trauma to the thoracic duct chyle may accumulate in the posterior mediastinum for days and then rupture into the pleural space with sudden onset of dyspnea, hypotension, and hypoxemia. About 50% of newborns with chylothorax present with respiratory distress in the 1st day of life. Chylothorax is rarely bilateral and usually occurs on the right side.
Laboratory Findings
Thoracentesis demonstrates a chylous effusion, a milky fluid containing fat, protein, lymphocytes, and other constituents of chyle; fluid may be yellow or bloody. In newborn infants or those who are not ingesting food, the fluid may be clear. A pseudochylous milky fluid may be present in chronic serous effusion, in which fatty material arises from degenerative changes in the fluid and not from lymph. In chylothorax, the fluid triglyceride level is >110 mg/dL, the pleural fluid:serum triglyceride ratio is >1.0, and pleural fluid:serum cholesterol ratio is <1.0; lipoprotein analysis reveals chylomicrons. The cells are primarily T lymphocytes. Chest radiographs show an effusion; CT scans show normal pleural thickness and may reveal a lymphoma as the etiology of the chylothorax. A lymphangiogram can localize the site of the leak, and lymphoscintigraphy may demonstrate abnormalities of the lymphatic trunks and peripheral lymphatics.
Complications
Repeated aspirations may be required to relieve the symptoms of pressure. Chyle reaccumulates quickly, and repeated thoracentesis may cause malnutrition with significant loss of calories, protein, and electrolytes. Immunodeficiencies, including hypogammaglobulinemia and abnormal cell-mediated immune responses, have been associated with repeated and chronic thoracenteses for chylothorax. The loss of T lymphocytes is associated with increased risk of infection in neonates; otherwise, infection is uncommon, but patients should not receive live virus vaccines. Lack of resolution of chylothorax can lead to inanition, infection, and death.
Treatment
Spontaneous recovery occurs in >50% of cases of neonatal chylothorax. Initial therapy includes enteral feedings with a low-fat or medium-chain triglyceride, high-protein diet or parenteral nutrition. Thoracentesis is repeated as needed to relieve pressure symptoms; tube thoracostomy is often performed. If there is no resolution in 1-2 wk, total parenteral nutrition is instituted; if this measure is unsuccessful, a pleuroperitoneal shunt, thoracic duct ligation, or application of fibrin glue is considered. Surgery should be considered earlier in neonates with massive chylothorax and chyle output of >50 mL/kg/day despite maximum medical therapy for 3 days. Parenteral octreotide at a dose of 80 to 100 µg/kg/day intravenously has been used to manage chylothorax, but further study is needed. Other therapeutic approaches include pressure control ventilation with positive end-expiratory pressure, talc or iodopovidone pleurodesis, and inhalation of nitric oxide. Treatment is similar for traumatic chylothorax. Chemical pleurodesis or irradiation is used in malignant chylothorax. OK432 (picibanil) has been used to treat fetal and newborn chylothorax.
Anderst JD. Chylothorax and child abuse. Pediatr Crit Care Med. 2007;8:394-396.
Bellini C, Boccardo F, Campisi C, et al. Lymphatic dysplasias in newborns and children: the role of lymphoscintigraphy. J Pediatr. 2008;152:587-589.
Cleveland K, Zook D, Harvey K, et al. Massive chylothorax in small babies. J Pediatr Surg. 2009;44:546-550.
Epaud R, Dubern B, Larroquet M, et al. Therapeutic strategies for idiopathic chylothorax. J Pediatr Surg. 2008;43:461-465.
Helin RD, Angeles ST, Bhat R. Octreotide therapy for chylothorax in infants and children: a brief review. Pediatr Crit Care Med. 2006;7:576-579.
Ma GC, Liu CS, Chang SP, et al. A recurrent ITGA9 missense mutation in human fetuses with severe chylothorax. Prenat Diagn. 2008;28:1057-1063.
Panthongviriyakul C, Bines JE. Post-operative chylothorax in children: an evidence-based management algorithm. J Paediatr Child Health. 2008;44:716-721.