Chapter 510 Rapidly Progressive (Crescentic) Glomerulonephritis
“Rapidly progressive” describes the clinical course of several forms of glomerulonephritis (RPGN) whose unifying feature is the histopathologic finding of crescents in the majority of glomeruli (Fig. 510-1). Therefore the terms rapidly progressive glomerulonephritis (RPGN) and crescentic glomerulonephritis (CGN) are synonymous. The natural history of most forms of CGN is rapid and relentless progression to end-stage renal failure.
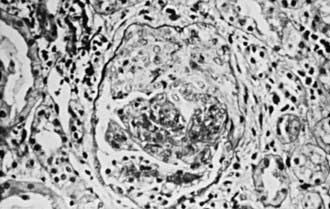
Figure 510-1 Light micrograph of a biopsy specimen from a child with Henoch-Schönlein purpura glomerulonephritis demonstrating a crescent overlying the glomerulus (×180).
Classification
CGN can be a severe manifestation of essentially every defined primary and secondary GN, but particular forms of GN are more likely to manifest as RPGN or evolve into CGN (Table 510-1). If no underlying cause is identified by systemic features, serologic testing, or histologic examination, the disease is classified as idiopathic CGN. The incidence of specific etiologies of CGN in children varies widely in reports from different centers; certain common themes are shared in all such reports. Patients with systemic vasculitis appear to be particularly prone to develop CGN. Patients with Henoch-Schönlein purpura (HSP), antineutrophil cytoplasmic antibody (ANCA)–mediated GN (microscopic polyangiitis and Wegener granulomatosis), and systemic lupus erythematosus (SLE) account for the majority of patients with CGN. Poststreptococcal GN rarely progresses to CGN, but because it is the most common form of GN in childhood it accounts for a significant percentage of patients with CGN in most reports. Membranoproliferative GN and idiopathic cases make up most of the remaining cases of CGN. Immunoglobulin (Ig)A nephropathy, a common GN, only rarely is rapidly progressive. Goodpasture disease often has rapidly progressive GN as a component of the syndrome, but its rarity in childhood results in its making up only a small percentage of children with CGN.
Table 510-1 CLASSIFICATION OF RAPIDLY PROGRESSIVE (CRESCENTIC) GLOMERULONEPHRITIS
ANTI-GBM ANTIBODY-MEDIATED RPGN
RPGN ASSOCIATED WITH GRANULAR IMMUNE DEPOSITS
RPGN WITHOUT GLOMERULAR IMMUNE DEPOSITS
RPGN, rapidly progressive (crescentic) glomerulonephritis; GBM, glomerular basement membrane.
Modified from Kliegman RM, Greenbaum LA, Lye PS: Practical strategies in pediatric diagnosis and therapy, ed 2, Philadelphia, 2004, Saunders, p 426.
Pathology and Pathogenesis
The hallmark of CGN is the histopathologic finding of crescents in glomeruli (see Fig. 510-1). Crescent formation, through proliferation of parietal epithelial cells in Bowman’s space, may be the final pathway of any severe inflammatory glomerular injury. Fibrin deposition and macrophage infiltration in the same areas suggest prominent involvement in the pathogenesis of the epithelial cell proliferation. Fibrous crescents, in which proliferative cellular crescents are replaced by collagen, are a late finding. The immunofluorescence findings, as well as the pattern of any deposits by electron microscopy (EM) can delineate the underlying glomerulopathy in CGN secondary to lupus, HSP nephritis, MPGN, postinfectious GN, IgA nephropathy or Goodpasture disease. Rare or absent findings by immunofluorescence and EM typify pauci-immune GN (Wegener disease and microscopic polyangiitis) and idiopathic crescentic GN.
Clinical Manifestations
Most children present with acute nephritis (hematuria, some degree of renal insufficiency, and hypertension) and usually have concomitant proteinuria, often with nephrotic syndrome. Occasional patients present late in the course of disease with oliguric renal failure. Extrarenal manifestations, such as pulmonary involvement, joint symptoms, or skin lesions, can help lead to the diagnosis of the underlying systemic disease causing the CGN.
Diagnosis and Differential Diagnosis
The diagnosis of CGN is made by biopsy. Delineation of the underlying etiology is reached by a combination of additional biopsy findings (described earlier), extrarenal symptoms and signs, and serologic testing, including evaluation of antinuclear and anti-DNA antibodies, serum complement levels, and ANCA. If the patient has no extrarenal manifestations and a negative serologic evaluation, and if the biopsy has no immune or EM deposits, the diagnosis is idiopathic, rapidly progressive CGN.
Prognosis and Treatment
Although the outcome is not uniformly positive, children with rapidly progressive poststreptococcal GN and CGN can spontaneously recover. The natural course of the disease is far more severe in the setting of other etiologies, including the idiopathic category, and progression to end-stage renal failure within weeks to months from onset is common. Having a majority of fibrous crescents on the renal biopsy portends a poor prognosis, because the disease usually has progressed to irreversible injury. Although there are few controlled data, the consensus of most nephrologists is that the combination of high-dose corticosteroids and cyclophosphamide may be effective in preventing progressive renal failure in patients with SLE, HSP nephritis, Wegener granulomatosis, and IgA nephropathy if given early in the course when cellular crescents predominate. Although such therapy can also be effective in the other diseases causing RPGN, renal outcomes in those settings are less favorable. Progression to end-stage renal disease often occurs despite aggressive immunosuppressive therapy. In combination with immunosuppression, plasmapheresis has been reported to benefit patients with Goodpasture disease. However, the possible benefits of plasmapheresis in other forms of RPGN are unclear.
Bagga A. Crescentic glomerulonephritis. In: Avner ED, Harmon WE, Niaudet P, et al, editors. Pediatric nephrology. ed 6. Heidelburg, Germany: Springer-Verlag; 2009:815-828.
Jindal KK. Management of idiopathic crescentic and diffuse proliferative glomerulonephritis: evidence-based recommendations. Kidney Int. 1999;70:S33-S40.
Lionaki S, Jennette JC, Falk RJ. Anti-neutrophil cytoplasmic (ANCA) and anti-glomerular basement membrane (GBM) autoantibodies in necrotizing and crescentic glomerulonephritis. Semin Immunopathol. 2007;29:459-474.
Pedchenko V, Bondar O, Fogo AB, et al. Molecular architecture of the Good-pasture autoantigen in anti-GBM nephritis. N Engl J Med. 2010;363(4):343-354.