Chapter 557 Thyroid Development and Physiology
Fetal Development
The fetal thyroid bilobed shape is recognized by 7 wk of gestation, and characteristic thyroid follicle cell and colloid formation is seen by 10 wk. Thyroglobulin synthesis occurs from 4 wk, iodine trapping occurs by 8-10 wk, and thyroxine (T4) and, to a lesser extent, triiodothyronine (T3) synthesis and secretion occur from 12 wk of gestation. There is evidence that several transcription factors—TTF-1/NKX-2.1, TTF-2 (also termed FOXE1), NKX2.5, and PAX8—are important in thyroid gland morphogenesis and differentiation and possibly also in its caudal migration to its final location. These factors also bind to the promoters of thyroglobulin and thyroid peroxidase genes and so influence thyroid hormone production. Hypothalamic neurons synthesize thyrotropin-releasing hormone (TRH) by 6-8 wk, the pituitary portal vessel system begins development by 8-10 wk, and thyroid-stimulating hormone (TSH) secretion is evident by 12 wk of gestation. Maturation of the hypothalamic-pituitary-thyroid axis occurs over the 2nd half of gestation, but normal feedback relationships are not mature until approximately 3 mo of postnatal life. Another transcription factor, Pit-1, is important for differentiation and growth of thyrotrophs, along with somatotrophs and lactotrophs.
Thyroid Physiology
The main function of the thyroid gland is to synthesize T4 and T3. The only known physiologic role of iodine (or iodide [I−] in its ionized form) is in the synthesis of these hormones; the recommended dietary allowance of iodine is 30 µg/kg/24 hr for infants, 90-120 µg/24 hr for children, and 150 µg/24 hr for adolescents and adults.
The median iodine intake in the United States has decreased by approximately 50% between the 1970s (320 µg/L) and the 1990s (145 µg/L), although at present intake appears to have stabilized. Whatever the chemical form ingested, iodine eventually reaches the thyroid gland as iodide. Thyroid tissue has an avidity for iodide and is able to trap (with a gradient of 100 : 1), transport, and concentrate it in the follicular lumen for synthesis of thyroid hormone. Entry of iodide from the circulation into the thyroid is carried out by the sodium-iodide symporter.
Before trapped iodide can react with tyrosine, it must be oxidized; this reaction is catalyzed by thyroidal peroxidase. Dual oxidase maturation factor 2 (DUOXA2) is required to express DUOX2 enzymatic activity, which is required for H2O2 generation, a crucial step in iodide oxidation. The thyroid cells also elaborate a specific thyroprotein, a globulin with approximately 120 tyrosine units (thyroglobulin). Iodination of tyrosine forms monoiodotyrosine and diiodotyrosine; 2 molecules of diiodotyrosine then couple to form 1 molecule of T4, or 1 molecule of diiodotyrosine and 1 of monoiodotyrosine to form T3. Once formed, hormones are stored as thyroglobulin in the lumen of the follicle (colloid) until ready to be delivered to the body cells. Thyroglobulin is a large globular glycoprotein with a molecular weight of about 660,000. T4 and T3 are liberated from thyroglobulin by activation of proteases and peptidases.
The metabolic potency of T3 is 3 to 4 times that of T4. In adults, the thyroid produces approximately 100 µg of T4 and 20 µg of T3 daily. Only 20% of circulating T3 is secreted by the thyroid; the remainder is produced by deiodination of T4 in the liver, kidney, and other peripheral tissues by type I 5′-deiodinase. Selenocysteine is the active center of the iodothyronine deiodinases. Thus, selenium indirectly plays a role in normal growth and development. In the pituitary and brain, approximately 80% of required T3 is produced locally from T4 by a different enzyme, type II 5′-deiodinase. The level of T3 in blood is 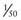 that of T4, but T3 is the physiologically active thyroid hormone.
that of T4, but T3 is the physiologically active thyroid hormone.
Thyroid hormones increase oxygen consumption, stimulate protein synthesis, influence growth and differentiation, and affect carbohydrate, lipid, and vitamin metabolism. The free hormones enter cells, where T4 may be converted to T3 by deiodination. Monocarboxylate transporter 8 is an active, specific thyroid hormone transporter that facilitates T4 entry into cells. Mutations of the MCT8 are associated with high T3 levels, thyroid hormone resistance, and severe X-linked psychomotor retardation. Intracellular T3 then enters the nucleus, where it binds to thyroid hormone receptors. Thyroid hormone receptors are members of the steroid hormone receptor superfamily that includes glucocorticoids, estrogen, progesterone, vitamin D, and retinoids. Four different isoforms of the thyroid hormone receptor (α1, α2 β1, and β2) are expressed in different tissues; the protein product of the formerly designated c-erb A proto-oncogene (THRA2) is the α2 thyroid hormone receptor in the brain and hypothalamus. Thyroid hormone receptors consist of a ligand-binding domain (binds T3), hinge region, and DNA-binding domain (zinc finger). Binding of T3 activates the thyroid hormone receptor response element, resulting in production of an encoded mRNA and protein synthesis and of secretion specific for the target cell. In this manner, a single hormone, T4, acting through tissue-specific thyroid hormone receptor isoforms and gene-specific thyroid response elements, can produce multiple effects in various tissues.
About 70% of the circulating T4 is firmly bound to thyroxine-binding globulin (TBG). Less-important carriers are thyroxine-binding prealbumin, called transthyretin, and albumin. Only 0.03% of T4 in serum is not bound and comprises free T4. Approximately 50% of circulating T3 is bound to TBG, and 50% is bound to albumin; 0.30% of T3 is unbound, or free, T3. Because the concentration of TBG is altered in many clinical circumstances, its status must be considered when interpreting T4 or T3 levels.
Thyroid Regulation
The thyroid is regulated by TSH, a glycoprotein produced and secreted by the anterior pituitary. This hormone activates adenylate cyclase in the thyroid gland and is important in all steps of thyroid hormone biosynthesis, from trapping of iodine to release of thyroid hormones. TSH is composed of 2 noncovalently bound subunits (chains): α and β. The α subunit is common to luteinizing hormone, follicle-stimulating hormone, and chorionic gonadotropin; the specificity of each hormone is conferred by the β subunit. TSH synthesis and release are stimulated by TSH-releasing hormone (TRH), which is synthesized in the hypothalamus and secreted into the pituitary. TRH is found in other parts of the brain besides the hypothalamus and in many other organs; aside from its endocrine function, it may be a neurotransmitter. TRH is a simple tripeptide. In states of decreased production of thyroid hormone, TSH and TRH are increased. Exogenous thyroid hormone or increased thyroid hormone synthesis inhibits TSH and TRH production. Except in the neonate, levels of TRH in serum are very low.
Further control of the level of circulating thyroid hormones occurs in the periphery. In many nonthyroidal illnesses, extrathyroidal production of T3 decreases; factors that inhibit thyroxine-type I 5′-deiodinase include fasting, chronic malnutrition, acute illness, and certain drugs. Levels of T3 may be significantly decreased, whereas levels of free T4 and TSH remain normal. Presumably, the decreased levels of T3 result in decreased rates of oxygen production, of substrate use, and of other catabolic processes.
557.1 Thyroid Hormone Studies
Serum Thyroid Hormones
Methods are available to measure all the thyroid hormones in serum: T4, free T4, T3, and free T3. A metabolically inert T3 (3,5′,3′-triiodothyronine), called reverse T3, is also present in serum. Age must be considered in interpreting results, particularly in the neonate.
Thyroglobulin is a glycoprotein dimer that is secreted through the apical surface of the thyrocyte into the colloid. Small amounts escape into the circulation and are measurable in serum. Levels increase with TSH (also called thyrotropin) stimulation and decrease with TSH suppression. Thyroglobulin levels are increased in the neonate, in patients with Graves disease and other forms of autoimmune thyroid disease, and in those with endemic goiter. The most marked elevations of thyroglobulin occur in patients with differentiated carcinoma of the thyroid. Athyreotic infants can have markedly reduced levels of thyroglobulin in serum.
TSH levels in serum are an extremely sensitive indicator of primary hypothyroidism. A 3rd generation of assays (chemiluminescent assays) that can measure complete suppression of TSH below the normal range is standard. After the neonatal period, normal levels of TSH are less than 6 µU/mL. These sensitive TSH assays obviate the need for TRH stimulation in the diagnosis of most patients with thyroid disorders.
Fetal and Newborn Thyroid
Fetal serum T4 increases progressively from mid-gestation to approximately 11.5 µg/dL at term. Fetal levels of T3 are low before 20 wk and then gradually increase to about 45 ng/dL at term. Reverse T3 levels (inactive form of T3), however, are high in the fetus (250 ng/dL at 30 wk) and decrease to 150 ng/dL at term. Serum levels of TSH gradually increase to 10 mU/L at term. Approximately one third of maternal T4 crosses the placenta to the fetus. Maternal T4 plays a role in fetal development, especially that of the brain, before the synthesis of fetal thyroid hormones begins. The fetus of a hypothyroid mother may be at risk for neurologic damage, and a hypothyroid fetus may be partially protected by maternal T4 until delivery. The amount of T4 that crosses the placenta is not sufficient to interfere with a diagnosis of congenital hypothyroidism in the neonate.
At birth, there is an acute release of TSH; peak serum concentrations reach 60 mU/L in 30 min in full-term infants. A rapid decline occurs in the ensuing 24 hr and a more gradual decline within the next 5 days to <10 µU/mL. The acute increase in TSH produces a dramatic increase in levels of T4 to approximately 16 µg/dL and of T3 to approximately 300 ng/dL in ~4 hr. This T3 seems largely derived from increased peripheral conversion of T4 to T3. T4 levels gradually decrease during the 1st 2 wk of life to 12 µg/dL. T3 levels then decline during the 1st wk of life to levels below 200 ng/mL. Serum free T4 levels are 0.9-2.3 ng/dL in infancy and decline to 0.7-1.8 ng/dL in childhood. Serum free T3 concentrations are approximately 540 pg/dL in infancy and decline to 210-440 pg/dL in childhood. Reverse T3 levels are maintained for 2 wk (200 ng/dL) and decrease by 4 wk to around 50 ng/dL. In preterm infants, changes in thyroid function after birth are qualitatively similar to but quantitatively smaller than full-term infants. Serum T4 and T3 levels are proportional to gestational age and birth weight.
Serum Thyroxine-Binding Globulin
The thyroid hormones are transported in plasma bound to TBG, a glycoprotein synthesized in the liver. Estimation of TBG levels is occasionally necessary because TBG is increased or decreased in a variety of clinical situations, with effects on the level of total thyroxine. TBG binds about 70% of T4 and 50% of T3. TBG levels increase in pregnancy, in the newborn period, and with administration of estrogens (oral contraceptives), perphenazine, or methadone, and they decrease with androgens, anabolic steroids, glucocorticoids, and L-asparaginase. These effects are the results of modulation of hepatic synthesis of TBG. TBG levels may be markedly decreased owing to decreased production with liver disease or loss in the gut with protein-losing enteropathies or urine, as in the congenital nephrotic syndrome. Decreased or increased levels of TBG also occur as genetic traits (Chapter 558).
Some drugs, in particular phenytoin (diphenylhydantoin), carbamezepine, furosemide, aspirin, and heparin, also inhibit binding of T4 and T3 to TBG. In addition, phenytoin and carbamezepine cause abnormalities of thyroid function tests by another mechanism. They stimulate hepatic cytochrome P-450 degradation of T4 and accelerate transport of T4 into tissues.
In Vivo Radionuclide Studies
Markedly improved direct tests of thyroid function have made radioiodine uptake studies less necessary. The iodine trapping or concentrating mechanism of the thyroid can be evaluated by measuring the uptake of radioactive isotope 123I (half-life, 13 hr). The technology allows doses of radioiodine (0.1-0.5 mCi) that are only a fraction of those formerly used with 131I. Technetium (99mTc) is a particularly useful radioisotope for children because in contrast to iodine, it is trapped but not organified by the thyroid and has a half-life of only 6 hr. Thyroid scanning may be indicated to assess the presence of thyroid tissue in questions of thyroid agenesis and to detect ectopic thyroid tissue, and thyroid uptake may be indicated to evaluate possible “hot” thyroid nodules. These studies should be performed with 99mTc pertechnetate or 123I because they have the advantages of lower radiation exposure and high-quality scintigrams. Use of 131I in children should be limited to those known to have thyroid cancer.
Thyroid Ultrasonographic Studies
Thyroid ultrasound examinations can determine the location, size, and shape of the thyroid gland, and they can assess the solid or cystic nature of nodules. Ultrasound is not as reliable as radionuclide studies in evaluating infants with suspected thyroid dysgenesis, particularly ectopic glands. Ultrasound examinations are useful in identifying normal thyroid gland position in children with suspected thyroglossal duct cysts. In children with autoimmune thyroiditis, ultrasound reveals scattered hypoechogenicity. Ultrasound examinations are more accurate than physical examination in estimating goiter size and assessing thyroid nodules.
DeBoer MD, LaFranchi SH. Pediatric thyroid testing issues. Pediatr Endocrinol Rev. 2007;5:570-577.
Brown RS, Huang SA, Fisher DA. The maturation of thyroid function in the perinatal period and during childhood. In: Braverman LE, Utiger RD, editors. Werner & Ingbar’s the thyroid: a fundamental and clinical text. ed 9. Philadelphia: Lippincott Williams & Wilkins; 2005:1013-1028.
Fisher DA. Physiological variations in thyroid hormones: physiological and pathophysiological considerations. Clin Chem. 1996;42:135-139.
Kalomenou I, Alevizaki M, Ladopoulos C, et al. Thyroid volume and echostructure in schoolchildren living in an iodine-replete area: relation to age, pubertal stage, and body mass index. Thyroid. 2007;17:875-881.
LaFranchi SH, Haddow JE, Hollowell JG. Is thyroid inadequacy during gestation a risk factor for adverse pregnancy and developmental outcomes? Thyroid. 2005;15:60-71.
Williams Fl, Mires GJ, Barnett C, et al. Transient hypothyroxinemia in preterm infants: the role of cord sera thyroid hormone levels adjusted for prenatal and intrapartum factors. J Clin Endocrinol Metab. 2005;90:4599-4606.
Brown RS, Huang SA, Fisher DA. The maturation of thyroid function in the perinatal period and during childhood. In: Braverman LE, Utiger RD, editors. Werner & Ingbar’s the thyroid: a fundamental and clinical text. ed 9. Philadelphia: Lippincott Williams & Wilkins; 2005:1013-1028.
Caldwell KL, Jones R, Hollowell JG. Urinary iodine concentration: United States National Health and Nutrition Examination Survey 2001–2002. Thyroid. 2005;15:692-699.
Carrasco N, Kopp P. Thyroid synthesis and secretion. In: Braverman LE, Utiger RD, editors. Werner & Ingbar’s the thyroid: a fundamental and clinical text. ed 9. Philadelphia: Lippincott Williams & Wilkins; 2005:37-76.
De Felice M, Di Lauro F. The development of the thyroid gland: what we know and what we would like to know. Curr Opin Endocrinol Metab. 2005;12:4-9.
Friesema ECH, Grueters A, Biebermann H, et al. Association between mutations in a thyroid hormone transporter and severe X-linked psychomotor retardation. Lancet. 2004;364:1435-1437.
Heuer H, Visser TJ. Minireview: pathophysiological importance of thyroid hormone transporters. Endocrinol. 2009;150:1078-1083.
Santisteban P. Development and anatomy of the hypothalamic-pituitary-thyroid axis. In: Braverman LE, Utiger RD, editors. Werner & Ingbar’s the thyroid: a fundamental and clinical text. ed 9. Philadelphia: Lippincott Williams & Wilkins; 2005:8-25.