Chapter 706 Envenomations
Few experiences are more frightening for patients than being bitten or stung by a venomous animal or insect. Most bites and stings by spiders, snakes, scorpions, and other venomous animals cause little more than local pain and do not require medical attention. There are thousands of species of potentially harmful or deadly venomous creatures worldwide, and each country/region has its own array of medically important organisms.
An important concept that is commonly confused in this topic is the terms venomous and poisonous. Venom is produced in specialized glands of the host and is usually injected by means of a bite or sting. Poisonous refers to detrimental effects from consuming or touching a plant, animal, or insect. One major difference is that poison is generally found throughout the animal and venom is isolated to the specialized glands.
Another important concept is that not every bite from a venomous creature is harmful. In many cases no venom is injected, so-called dry bites. A dry bite may occur for many reasons, including failure of the venom delivery mechanism and depletion of venom. Up to 20% of pit viper, 80% of coral snake, and approximately 50% of all venomous snake bites are dry.
In the 2007 report of the American Association of Poison Control Centers, 70,833 consultations were related to bites and stings of various creatures, with approximately one third involving victims younger than 19 years. There were 3 deaths, all in adult males, 2 by snakes and 1 by a stinging insect.
General Approach to the Envenomated Child
Children may be bitten or stung as they play and explore their environment. The evaluation may be hampered by an unclear history of the circumstances and the possible offending organism, particularly with preverbal children. The overall effects of some venomous bites and stings may be relatively more severe in children than in adults, because children generally receive a similar venom load from the offending animal yet have less circulating blood volume to dilute its effects.
General Management
When faced with an envenomated child, the treating physician should anticipate a dynamic clinical syndrome that may progress with time. A high level of diligence should be maintained so that important, potentially subtle findings are not missed. As with any disease process, treatment of an envenomated child should start with assessing and managing as necessary the airway, breathing, and circulation (ABCs). Most envenomations require little more than local wound care, pain control, reassurance, and possibly observation. The severely envenomated child may need airway and respiratory protection and support (e.g., high-concentration oxygen administration and endotracheal intubation) and adequate IV access in an unaffected extremity if possible. Early hypotension tends to be related to vasodilatation and should be treated with volume expansion using appropriate infusion of physiologic saline solutions (normal saline boluses of 20 mL/kg body weight; repeated as needed up to 3 times). Shock unresponsive to volume repletion may require addition of a vasoactive pharmacologic agent such as epinephrine or dopamine (in addition to antivenom administration as appropriate—see later).
The affected body part should be immobilized in a position of function and any areas of edema should be marked, measured and monitored. If antivenom (AV) is available for the envenomation, efforts should be initiated to locate and secure an adequate amount to treat the patient (at least a starting dose). In the USA, regional poison control centers can facilitate this effort and are especially helpful if the offending species is exotic. Guidance in dosing the appropriate AV can generally be found in the package insert that accompanies the agent, although the advice in inserts for some products from developing countries may contain inaccurate and incorrect recommendations. Physicians who do not regularly treat venomous bites and stings should consult local or regional experts for assistance.
Antivenom Administration
Specific AVs are available for many venomous creatures of the world, particularly snakes, spiders, and scorpions. These products essentially impart passive immunity to the victim and should be given in cases of significant envenomation as early in the process as possible, because AV is capable of neutralizing only circulating, unbound venom components in the blood.
Antivenoms may be either in liquid form or lyophilized (requiring reconstitution prior to administration). Most AVs are given intravenously. There is no benefit to giving any AV locally at the bite site. As soon as the need for AV is established, it should be placed into solution (generally diluted in a quantity of normal saline equivalent to 20 mL/kg body weight, up to 250-1000 mL total).
As heterologous serum products, AVs carry some variable risk of inducing nonallergic or allergic anaphylactic reactions. Therefore, the patient should be closely monitored, and a physician should be present during the infusion, with access to all the appropriate equipment and medications needed to reverse such a reaction. Skin tests, often recommended by AV manufacturers, are unreliable and should be omitted.
Intravenous AV should be started slowly, and the rate gradually increased as tolerated by the patient, with a goal to administer the entire dose in approximately 1 hr.
If the victim experiences a reaction to the product, it should be temporarily stopped. Intramuscular epinephrine and intravenous antihistamines and steroids should be given. Then the AV should be restarted, possibly at a slower rate and in more dilute solution. If the reaction is severe, the decision must be made as to whether the benefits of the AV outweigh the risks of anaphylaxis on the basis of the patient’s clinical condition.
AV can also cause delayed immunoglobulin G and M–mediated serum sickness in some patients. Serum sickness occurs 1-2 weeks after AV administration, manifesting as fever, myalgias, arthralgias, urticaria, and potential renal and neurologic involvement. It is easily treated with oral steroids, antihistamines, and acetaminophen.
General Wound Care
As with all other breaks in the skin, bites and stings require basic wound care, including copious tap water or normal saline irrigation under pressure when possible. Tetanus immunization should be updated as needed. Blisters should be left intact to act as natural bandages and help prevent infection. Exposed tissue should be covered with wet to dry dressings. Necrotic wounds, such as those that might follow some snake and spider bites, should be judiciously debrided, with removal of only clearly necrotic tissue. Reconstructive surgery with skin grafts or muscle/tendon grafts may be necessary. In general, prophylactic antibiotics are not necessary except, perhaps, in cases in which an ill-informed “rescuer” cut into the bite and applied mouth suction. Antibiotics should generally be reserved for signs of established secondary infection.
Snake Bites
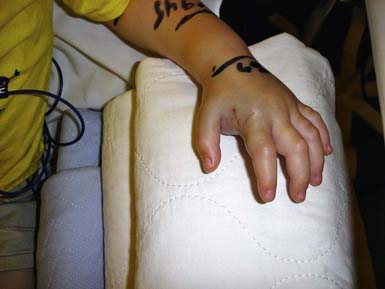
Most snake bites are inflicted by nonvenomous species and are of no more consequence than a potentially contaminated puncture wound (Fig. 706-1). Venomous snakes, however, kill many tens of thousands of people in the world each year. The precise number is difficult to ascertain, because the toll in human suffering is far greatest in developing nations. Developed nations, with established medical care systems, have relatively few fatalities each year.
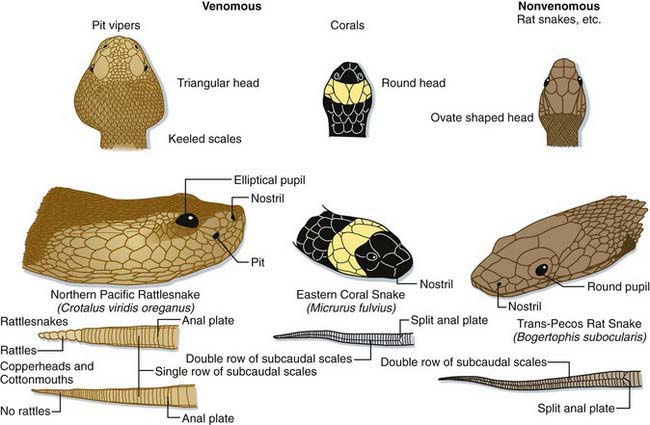
Figure 706-1 Anatomic comparison of pit vipers, coral snakes, and nonvenomous snakes of the USA. (Note: the northern Pacific rattlesnake is now classified as Crotalus oreganus oreganus.)
(Modified from Adams JG, et al, editors: Emergency medicine, Philadelphia, 2008, Saunders/Elsevier. © Elsevier, drawing by Marlin Sawyer.)
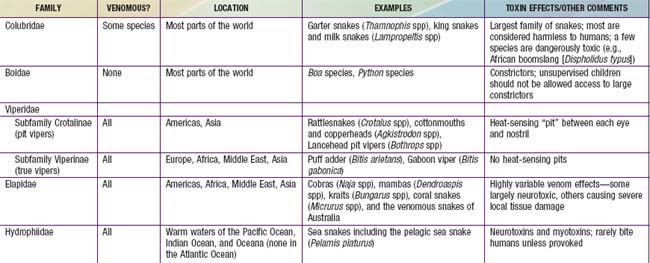
Most of the world’s medically significant venomous snakes belong to one of two families—Viperidae and Elapidae (Table 706-1). In developing nations, most snake envenomations occur in agricultural workers who inadvertently contact snakes while in the fields. Many victims of snake envenomation in developed nations are adolescent or young adult males, frequently intoxicated, who are attempting to handle or catch the snake. Bites are located on an extremity in over 95% of cases. In the USA, approximately 98% of venomous snake bites are inflicted by pit vipers (family Viperidae; subfamily Crotalinae). A small fraction of bites are caused by coral snakes (family Elapidae) in the South and Southwest, and by exotic snakes that have been imported.
Venoms and Effects
Snake venoms are complex mixtures of proteins including large enzymes that cause local tissue destruction and low molecular weight polypeptides that have the more lethal systemic effects. The symptoms and severity of an envenomation vary according to the type of snake, the amount of venom injected, and the location of the bite. The fear caused by a snake bite can result in nausea, vomiting, diarrhea, cold/clammy skin, and even syncope regardless of whether or not venom was injected. In general, viper venoms can have deleterious effects on almost any organ system. Most viper bites cause significant local pain, swelling, ecchymosis, and variable necrosis of the bitten extremity (Fig. 706-2). The pain and swelling typically begin quickly after the bite and progress over hours to days. Serious envenomations may result in a consumptive coagulopathy, hypotension, and respiratory distress. In contrast, venoms from the Elapidae tend to be more neurotoxic with little or no local tissue damage. These bites cause variable local pain and the onset of systemic effects can be delayed for hours. Manifestations of neurotoxicity generally begin with cranial nerve palsies such as ptosis, dysarthria, and dysphagia and may progress to respiratory failure and complete paralysis. There are exceptions; some members of the Elapidae family cause little or no neurotoxicity but rather severe tissue necrosis (e.g., African spitting cobras). Some vipers cause significant neurotoxicity (e.g., some populations of the Mohave rattlesnake [Crotalus scutulatus]). Physicians should proactively learn the important species in their regions, including how the species can be identified, the expected effects of their venoms, and proper approaches to management.
Management
Prehospital care should focus on immobilization and rapid transport to the emergency department. Constrictive clothing, jewelry, and watches should be removed, and the injured body part should be immobilized in a position of function at the level of the heart. All proposed field treatments for snake bites, such as tourniquets, ice, electric-shock, incision, and suction, have proven problematic, with most being ineffective and deleterious.
At the hospital, attention is directed to the ABCs and supportive care as needed. An effort should be made to identify the offending snake and then to secure the appropriate antivenom. Intravenous access should be established in an unaffected extremity, and standard laboratory specimens should be obtained, including those for a complete blood count (CBC), coagulation studies, fibrinogen concentration, and serum chemistry analysis including total creatine kinase. A blood sample should be sent for typing and screening, although blood products are rarely actually required in management of snake bite. Samples collected later in the clinical course may be hard to crossmatch because venom and AV can interfere with the testing. Tourniquets placed in the field by laypeople should be cautiously removed after venous access is obtained, to watch for and treat adverse effects that may follow a sudden bolus of acidotic, hyperkalemic blood mixed with venom into the systemic circulation. The bitten extremity should be marked at 2 or more sites proximal to the bite, and the circumferences at these locations should be assessed every 15 min to monitor for progressive edema—indicative of ongoing venom effects.
AVs are relatively specific for the snake species against which they are designed to protect. There is no benefit to administering an AV for an unrelated species, and doing so certainly involves unacceptable risk (e.g., anaphylaxis) and expense. If it is determined that the child requires AV, a search for the appropriate product should begin as soon as possible—checking the hospital pharmacy, regional poison control center, and perhaps local zoos and museums that keep captive snakes.
Indications for administering antivenom can be found in Table 706-2.
Table 706-2 INDICATIONS FOR SNAKE ANTIVENOM ADMINISTRATION
| Evidence of systemic toxicity: | |
| Hemodynamic or respiratory instability | Hypotension, respiratory distress |
| Hemotoxicity | Clinically significant bleeding or abnormal coagulation studies |
| Neurotoxicity | Any evidence of toxicity: usually beginning with cranial nerve abnormalities and progressing to descending paralysis including the diaphragm |
| Evidence of local toxicity | Progressive soft tissue swelling |
Occasionally the sharp, recurved teeth of snakes, including nonvenomous snakes, are left behind in wounds; they should be identified (using soft tissue radiographs or ultrasound as needed) and removed.
Spider Bites
More than 20,000 venomous spiders have been identified, but most lack either potent venom or fangs long enough to penetrate human skin, and are therefore of no medical significance. No spiders can be considered truly deadly, meaning that an untreated bite in a human would be expected to cause death. The spiders of medical significance can be broadly divided into 2 major groups: those that cause a neurotoxic syndrome and those that cause tissue necrosis. In the USA, the only significant morbidities are caused by one genus of spider from each group: Lactrodectus (the widow spiders) and Loxosceles (the fiddleback or recluse spiders).
Neurotoxic Spiders
The major neurotoxic spiders are the widow spiders (Latrodectus spp), the funnel web spiders (Atrax and Hydranyche spp—found in Australia), and the banana spiders (Phoneutria spp—indigenous to Latin America).
Venoms and Effects
Neurotoxic spiders all possess venoms that act at neural synapses, both at neuromuscular junctions and at autonomic nervous system junctions. All of the widow spiders (Latrodectus spp, including the well-studied black widow [Latrodectus mactans; Fig. 706-3)] and the Australian red-back spider [Latrodectus hasseltii]) possess very similar venoms, with the most important neurotoxin being α-latrotoxin. The neurotoxin of the Sydney funnel web spider (Atrax robustus) is robustoxin.

Figure 706-3 This “black widow” spider, Latrodectus mactans, was photographed during a study of migrant labor camp disease vectors. Though spiders are “mostly” harmless to man, two genera, Latrodectus spp, the “black widow” spiders, and Loxosceles spp, including the “brown recluse” spider, Loxosceles reclusa, produce bites that are poisonous to humans.
(From The Centers for Disease Control and Prevention Public Health Image Library, Image #5449.)
Bites by the neurotoxic spiders tend to be very painful, and the offending spider is often seen. Systemic effects may include hypertension, tachycardia, bradycardia, hypersalivation, diaphoresis, and diffuse muscle spasms.
Management
Management of neurotoxic spider envenomation centers on sound supportive care. Several Latrodectus AVs are available, and each appears to be effective regardless of which species of widow spider was responsible for the bite. There is also an AV for the Sydney funnel web spider, the only species of funnel web that has caused human fatalities (none since the introduction of the AV), and a polyspecific AV for the banana spider in South America. These AVs should be used in significant bites with potentially serious systemic effects. The package insert for the appropriate product is used to guide therapy.
In the USA, Latrodectus AV is administered to reverse serious systemic effects of widow spider envenomation. One vial is given either intramuscularly (IM) or IV. Efficacy is usually noted within 1 hr of administration, reversal of systemic toxicity and relief of pain being noted. Occasionally, a second vial is necessary. There have been deaths related to acute nonallergic anaphylactic reactions to the U.S. AV, so its administration should be undertaken with due caution and close monitoring.
If AV is to be withheld or is not available, generous doses of opioid analgesics and benzodiazepines may be used to ease symptoms (though this may require up to 72 hr of therapy).
Disposition
Most neurotoxic spider bite victims, even those requiring AV, can be discharged from the emergency department if they have a satisfactory response to therapy. Parents should be warned to bring their child back for any recurrence of venom effects. Children with more severe cases should be admitted for 24 hr of monitoring.
Necrotizing Spiders
Venoms and Effects
Although many spiders may cause a small amount of local tissue damage after their bites, the spiders most notorious for their dermonecrotic potential are the violin or recluse spiders of the genus Loxosceles. The best known member of this genus is the brown recluse (Loxosceles reclusa) (Fig. 706-4), found in the midwestern and southern portions of the USA. The venom of Loxosceles spiders contains a phospholipase enzyme, sphingomyelinase D, which attacks cell membranes and can cause local tissue damage that can occasionally be severe. The bite of this spider is generally painless and initially goes unnoticed. A few hours after the bite, pain related to focal ischemia begins at the site. Within a day, the site may have a central clear or blood-filled vesicle with surrounding ecchymosis and a rim of pale ischemia. The lesion may gradually expand over a period of days to weeks until necrotic tissue sloughs and healing begins.

Figure 706-4 Male recluse spider (Loxosceles sp). Note the distinct violin-shaped marking on the dorsum of the cephalothorax.
(Courtesy of Michael Cardwell/Extreme Wildlife Photography.)
Rare cases of systemic loxoscelism appear to be more common in young children than adults. They present with systemic toxicity, including fever, chills, nausea, malaise, diffuse macular rash, and petechiae, and may experience hemolysis, coagulopathy, and/or renal failure.
Management
For necrotizing spider bites, management of the wound involves sound supportive care, including intermittent local ice therapy for the first 72 hr and administration of antibiotics if there is any question of secondary bacterial infection. Daily wound cleansings, combined with splinting of the bitten area, should be performed until the wound is healed.
Nothing has been definitively proven effective in limiting the extent of necrosis following a spider bite. There is no role for steroids in managing necrotic arachnidism. Dapsone, though used anecdotally in managing Loxosceles bites in adults, is not approved for use in children and should not be prescribed.
Children appearing systemically unwell should be admitted and undergo laboratory evaluation (CBC, coagulation studies, and urinalysis). Systemic loxoscelism is managed with intravenous hydration, management of renal failure as needed, and a brief course of systemic steroids to stabilize red blood cell membranes. Though there are documented fatalities following bites by the South American violin spider (Loxosceles laeta), there has never been a definitively proven fatality following a brown recluse bite in the USA. No AV is commercially available in the USA for management of necrotizing spider bites such as those from Loxosceles species.
Disposition
Victims with potentially necrotic bites should be monitored for a few days with daily wound checks. Local, intermittent cooling therapy should be continued for approximately 72 hr. Any child with a probable necrotizing spider bite and evidence of systemic involvement should be admitted to be watched for hemolysis and coagulopathy.
Scorpion Stings
Of the more than 1,200 species of scorpions worldwide, only a few cause more than a painful sting. In the USA, there is one medically significant scorpion, the bark scorpion (Centruroides sculpturatus [formerly Centruroides exilicauda]). Though this scorpion has caused death in children in the past, such an outcome is exceedingly rare. It is found only in Arizona and small areas of immediately surrounding states. In other regions of the world, especially Latin America, Africa, the Middle East, and Asia, a number of scorpions regularly cause fatalities, particularly in small children.
Venoms and Effects
The major components of important scorpion venoms are neurotoxins that alter neural membrane ionic channels, causing autonomic and cardiovascular dysfunction through the release of acetylcholine and catecholamines. Manifestations of scorpion stings in children vary from mild to severe and may include pain, paresthesias, roving eye movements, cranial nerve dysfunction, opisthotonos/emprosthotonos, seizures, hypertensive crisis, cardiovascular collapse, and respiratory failure.
Management
Most stings require only pain control and respond well to ice, immobilization, and analgesics. Management of severe stings should begin with the ABCs. Opioid analgesics may have some synergy with scorpion neurotoxins and should be used with caution. Benzodiazepines may be more useful, especially for severe muscle spasms or sedation of the agitated child. Approximately 20 different scorpion AVs are available worldwide, but their use is controversial, due to variable efficacy and the risk of potential nonallergic anaphylaxis. Practitioners should be familiar with the local standard of care for treating the stings of their indigenous scorpions and should consult a local or regional expert for assistance as necessary. In the USA, there are currently no commercially available AVs for the bark scorpion, but clinical experience is growing with the experimental use of a Mexican antivenom (Alacramyn) in certain Arizona hospitals. The practitioner should contact the Arizona Poison Control for details and assistance (520-626-6016). In some regions of the world, prazosin is used in severe scorpion stings to ameliorate acute cardiovascular effects.
Disposition
The child with evidence of systemic toxicity following a scorpion sting should be admitted for at least 24 hr of monitoring (including cardiac monitoring) and, if envenomation is severe, should be monitored in a pediatric intensive care unit. In the absence of systemic toxicity and with adequate pain control, children older than 1 yr can be discharged home with a responsible adult.
Hymenoptera Stings
The insect order Hymenoptera includes the stinging ants, bees, and wasps, which are characterized by the presence of a modified ovipositor (stinger) at the end of the abdomen through which venom is injected. Various members of the order can be found throughout the world.
Venoms and Effects
Hymenoptera venoms, mixtures of proteins and vasoactive substances, are not very potent. Most stings result in only local pain, redness, and swelling, followed by itching and resolution. Some patients experience a large local reaction in which swelling progresses beyond the sting site, possibly involving the entire extremity. Approximately 0.4-0.8% of children are at risk for acute, life-threatening reactions due to hymenoptera venom sensitivity. Each year, an estimated 50-150 people in the USA die of allergic anaphylaxis caused by hymenoptera stings (Chapter 64). Rare cases of delayed serum sickness can follow hymenoptera stings. Finally, with the spread of Africanized honey bees (Apis mellifera scutellata), massive stinging episodes resulting in systemic venom toxicity (hypotension, respiratory failure, shock, hemolysis, and renal failure) appear to be increasing in Latin America and the southwestern U.S. states.
Management
Children with typical local reactions can be treated with application of cold compresses and with analgesics and antihistamines as needed. Children with large local reactions should also receive a 5-day course of oral corticosteroids and a prescription for an epinephrine autoinjection kit (and instructions in its use) prior to discharge. Patients presenting with urticaria, angioedema, wheezing, or hypotension should be treated aggressively for an immediate hypersensitivity reaction with intramuscular epinephrine (0.01 mg/kg, up to 0.3-0.5 mg of 1 : 1000 formulation), airway management as needed, oxygen, intravenous fluids, antihistamines, and corticosteroids. Children suffering massive stinging episodes should undergo treatment similar to that for allergic anaphylaxis.
Disposition
Children with local reactions (limited or large local) can be discharged with continued care as outlined previously and instructions for wound precautions. More difficult disposition decisions are involved for children with systemic manifestations. Children with only diffuse urticaria, who are stable after a period of observation, can be discharged in the care of a responsible adult to continue antihistamines and steroids and to carry an epinephrine self-administration kit. These children seem to be at little risk for progressing to systemic anaphylaxis with future stings. Children suffering more than simple hives (e.g., wheezing, evidence of laryngeal edema or cardiovascular instability) should be admitted for 24 hr of observation and should receive a referral to an allergist for testing for hymenoptera venom sensitivity and possible immunotherapy. Immunotherapy reduces the risk of systemic anaphylaxis from future stings in high-risk patients from somewhere between 30 and 60% to less than 5%.
Marine Envenomation
The most commonly encountered venomous marine creatures are the jellyfish (Cnidaria), stingrays (Chondricthyes), and members of the family Scorpaenidae—the lionfish, scorpionfish, and stonefish. Although most injuries occur when a child ventures into the animal’s natural environment, lionfish (Pterois spp.) are commonly kept in private aquariums and children may be stung if they attempt to handle these beautiful fish.
Venoms and Effects
All jellyfish have unique stinging cells called nematocysts. These cells contain a highly folded tubule that everts on contact and injects venom. The venom is antigenic and can be dermonecrotic, hemolytic, cardiotoxic, or neuropathic, depending on the species. The nematocysts can sting even after the tentacle is severed from the body and after the jellyfish is dead. The Pacific box jellyfish (Chironex fleckeri) of Australia, with its cardiotoxic venom, is known to cause rapidly fatal stings. Although fatal anaphylaxis to jellyfish stings has been reported in coastal waters of the USA, these events are rare. For clinicians in the Americas, the primary concern with jellyfish stings is localized pain that may be associated with paresthesias or pruritus. Rarely, jellyfish victims may have systemic symptoms such as nausea, vomiting, headache, and chills.
Stingrays have a sharp, retro-serrated spine and associated venom gland at the base of the tail. Stings tend to occur when the victim steps on the animal hidden in the surf. Injuries involve jagged lacerations from the spine, and the venom has vasoconstrictive properties that can result in tissue necrosis and poor wound healing. Stingray envenomations are noteworthy for immediate and intense pain at the site of injury that lasts 24-48 h. Some patients experience nausea, vomiting, and muscle cramps, and, rarely, hypotension or seizures.
The Scorpaenidae have venomous dorsal, pelvic, and anal spines that become erect when threatened. The glands associated with these spines contain venoms that result in direct myotoxicity leading to paralysis of cardiac, involuntary, and skeletal muscles. Envenomation causes immediate pain that may persist for hours or days. Victims may experience intense local tissue destruction in which superinfections are common. Systemic symptoms include vomiting, abdominal pain, headache, delirium, seizures, and respiratory failure.
Management
Treatment of jellyfish stings begins in the ocean. The involved skin should be quickly rinsed in seawater (fresh water may stimulate further nematocyst firing). Dousing the sting site with vinegar or rubbing alcohol can inhibit nematocyst discharge. Visible tentacle fragments should be removed with a gloved hand or forceps, and microscopic fragments may be removed by gently shaving the affected area. Folk remedies such as rubbing the sting with sand and applying urine are not helpful and cause more irritation. Meat tenderizer is not effective. Antihistamines and corticosteroids are indicated for swelling and urticaria. Antibiotics are not needed.
Treatment of stingray and Scorpaenidae stings is similar. These toxins are heat labile, and immersion in hot water (approximately 42°C) for 30-60 min denatures the protein constituents and decreases pain significantly. The wounds should be thoroughly cleaned and explored with use of local or regional anesthesia to rule out retention of spine or integument fragments. Stingray spines are radiopaque and may be seen on plain films of the wounded area. Lacerations should be treated with delayed primary closure or allowed to heal by secondary intention. Systemic analgesia should be provided as needed. Due to the risk of secondary bacterial infection, there should be a low threshold for administering prophylactic antibiotics to cover Staphylococcus, Streptococcus, and Vibrio species, and wounds should be rechecked daily for a few days.
Disposition
After wound care, most victims can be discharged home with responsible adults. If there are significant systemic effects after pain control is achieved, the child should be admitted for monitoring and further care as needed.
Bites and stings by venomous creatures are common occurrences in children, but they uncommonly cause major morbidity or mortality. The majority of such injuries do very well with sound supportive care. In serious cases, aggressive management of the ABCs combined with specific interventions (such as AVs when available) maximize the potential for an optimal outcome. A low threshold should be maintained for consulting specialists in envenomation medicine as needed.
Auerbach PS. Envenomation by aquatic invertebrates. In: Auerbach PS, editor. Wilderness medicine. ed 5. St Louis: Mosby; 2007:1691-1729.
Auerbach PS. Envenomation by aquatic vertebrates. In: Auerbach PS, editor. Wilderness medicine. ed 5. St Louis: Mosby; 2007:1730-1749.
Bawaskar HS, Bawaskar PM. Efficacy and safety of scorpion antivenom plus prazosin compared with prazosin alone for venomous scorpion (Mesobuthus tamulus) sting: randomised open label clinical trial. BMJ. 2010;341:c7136.
Boyer LV, Theodorou AA, Berg RA, et al. Antivenom for critically ill children with neurotoxicity from scorpion stings. N Engl J Med. 2009;360:2090-2098.
Bronstein AC, Spyker DA, Cantilena LRJr, et al. 2007 Annual Report of the American Association of Poison Control Centers’ National Poison Data System (NPDS): 25th Annual Report. Clin Toxicol (Phila). 2008;46:927-1057.
Gold BS, Dart RC, Barish RA, et al. Bites of venomous snakes. N Engl J Med. 2002;347:347-356.
Gutierrez JM, Lomonte B, Leon G, et al. Trends in snakebite envenomation therapy: scientific, technological and public health considerations. Curr Pharm Design. 2007;13:2935-2950.
McDade J, Aygun B, Ware RE. Brown recluse spider (Loxosceles reclusa) envenomation leading to acute hemolytic anemia in six adolescents. J Pediatr. 2010;156:155-157.
Mills EJ, Ford N. Research into scorpion stings. BMJ. 2011;342:c7369.
Moffitt JE, Golden DBK, Reisman RE, et al. Stinging insect hypersensitivity: a practice parameter update. J Allergy Clin Immunol. 2004;114:869-886.
Morocco A, Morocco A. Sea urchin envenomation. Clin Toxicol. 2005;43:119-120.
Norris RL, Bush SP. Bites by venomous reptiles in the Americas. In: Auerbach PS, editor. Wilderness medicine. ed 5. St Louis: Mosby; 2007:1051-1085.
Simpson ID, Norris RL. The global snakebite crisis—a public health issue misunderstood, not neglected. Wild Environ Med. 2009;20:43-46.
Singer AJ, Dagum AB, Singer AJ, et al. Current management of acute cutaneous wounds. N Engl J Med. 2008;359:1037-1046.
Swanson DL, Vetter RS, Swanson DL, et al. Bites of brown recluse spiders and suspected necrotic arachnidism. N Engl J Med. 2005;352:700-707.
Warrell DA. Snake bite. Lancet. 2010;375:77-86.
Williams D, Gutierez JM, Harrison R, et al. The global snake bite initiative: an antidote for snake bite. Lancet. 2010;375:89-91.
World Health Organization. Snake antivenom immunoglobulins (website). www.who.int/bloodproducts/snakeantivenoms. Accessed December 3, 2010
World Health Organization. Venomous snakes distribution and species risk categories (website). http://apps.who.int/bloodproducts/snakeantivenoms/database/. Accessed December 3, 2010