100
Graded Motor Imagery
Susan Watkins Stralka
• Graded motor imagery (GMI) is a sequential program and should be tailored for each patient.
• GMI should be part of a comprehensive therapeutic treatment plan.
• Patients with both musculoskeletal and neurologic conditions who cannot move or are afraid of movement may benefit from GMI.
• More evidence is needed to support the benefits of GMI.
Background
Graded motor imagery (GMI) is a technique of rehabilitation described by Moseley and Butler
1
as a way to sequentially train the brain. Recent neuroscience research using advanced and improved technologies such as functional magnetic resonance imaging (fMRI) and transcranial magnetic stimulation (TMS) demonstrate brain reorganization after the use of rehabilitation techniques such as GMI.
2,3
GMI has been shown to sequentially activate distinctly ordered stages of brain function
2
using positron emission tomography (PET), which reveals brain activation through blood flow measurements. There is a large body of literature on the differences in central nervous system (CNS) organization and function between patients with persistent or chronic pain compared with healthy control participants.
4–6
Change in cerebral organization in patients with complex regional pain syndrome (CRPS) with reorganization in central cortical somatosensory and motor networks results in altered processing of tactile and nociceptive stimuli.
7
Moseley’s interest in GMI came about after reviewing a research report of delayed reaction time in a left–right hand discrimination task that occurred in people with hand pain.
4,8
In this report, the body schema, which is a real-time cortical representation of the body in space generated by proprioceptive, somatosensory, vestibular, and other sensory inputs, was examined to see if pain had an effect on this representation, which it did.
4,8
This report stimulated Moseley’s interest in studying how a therapist can provide treatment that is less threatening than imagined movements. Other research reports found that patients with chronic unilateral pain required a longer response time (RT) with a left–right discrimination task.
9
Lundborg,
10
Merzenich and Charms,
11
and Byl and Merzenich
12
reported that the brain has the ability to reorganize, to change its input and output throughout lifetime, and this is called neuroplasticity.
These research studies provided evidence that cortical representation is subject to change. Flor and Moseley’s
13,14
investigation of the mechanism of phantom limb pain, which is caused by a body part that no longer exists, has largely contributed to our understanding of the development and maintenance of pain perceptions. These studies have shown that the brain itself plays a major role in chronic pain when it reacts to past experiences and reorganizes itself after amputation and persistent pain.
15
Mirror neurons are brain cells that fire when one acts as well as when one observes the same action performed by another; they have recently been identified as being involved in central sensitization (i.e., the development and maintenance of chronic pain).
16
These mirror neurons fire through both observation and imagining, as well as with execution of movement. Fadiga and coworkers
17
provided evidence that mirror neurons in humans fired after the observation of a movement and resulted in actual motor facilitation in brain studies. This same study demonstrated that mirror neurons fire not only when a person performs an activity but also when observing another person performing the same activity, which may assist in functional recovery. Evidence from research on limb amputation confirmed that there were changes in cortical representation from pre- to postamputation.
18
It has been noted that the somatosensory cortex that formerly received input from the amputated limb with phantom limb pain reorganizes and receives input from neighboring regions. Reorganization changes occurred only in amputees with phantom limb pain after amputation but not in amputees without pain. Ramachandran’s
15
research studies also suggested that phantom limb and phantom sensations that occurred when certain areas of the face of amputees were stimulated were linked to cortical reorganization after injury and amputation. This author suggested that this phenomenon may be maintained in some amputees because of input from peripheral nerves that were damaged and continued to send random signals to the brain.
15
These studies with amputees support past knowledge that the human brain is malleable or plastic and that “top-down training” such as mirror therapy or GMI may be helpful in dealing with phantom limb sensations or pain. Ramachandran’s research was embraced by many, and interest in the investigation and use of mirror therapy for phantom limb pain developed. Investigators asserted that phantom pain results from a mismatch between motor output and visual feedback. While observing the reflected
image of the uninvolved limb in a mirror placed on the involved side, the brain is “tricked” into thinking that the amputated limb is restored, thus reestablishing the visual feedback that would overrule this mismatch.
16
According to Rock and Victor
19,20
in 1964, the brain has been shown to prioritize visual input over proprioceptive input, so when the unaffected limb moves in front of the mirror, it appears as though the affected limb is functioning normally when watching the image in the mirror. Flor
13
and Moseley and Butler’s
1
research findings demonstrated through fMRI that the duration and extent of injury correlated with remapping of the brain after chronic pain, stroke, dystonia, phantom limb pain, and CRPS involving specific sensory and motor cortical networks.
13
Ramachandran and Moseley reported that the changes in the cortical map are thought to be part of “maladaptive neuroplasticity “ that develops after injury.
1,18
A number of studies have emphasized the importance of correct left–right identification, which reflects an intact body schema. Central abnormalities include disruption of sensory and motor cortical processing and disrupted body schema causing sensory disturbances, dysfunctional motor control, and spreading of symptoms.
21,22
With recent neuroscience advances in the understanding of brain activity and its relationship to clinical symptoms, a new roadmap for treating patients with persistent pain symptoms and loss of motor control has evolved. The importance of identifying and treating both “bottom-up” or peripheral symptoms and “top-down” symptoms (i.e., brain training) has opened a new avenue for consideration with therapy patients. GMI is called a “brain-based treatment” or a “top-down treatment” because the intervention has been shown to turn on specific brain areas or sequentially activate cortical premotor and motor networks in a graded manner.
1
It has been termed a graded exposure process with pacing to avoid trigging a protective response such as pain. Graded exposure requires appropriate patient education regarding pain and activity and allows for a reduction of pain with gradual improvement toward movement.
Graded Motor Imagery Phases
Phase 1: Laterality Training or Left–Right Identification
The first stage of the GMI training is laterality or left–right recognition; the difference between identifying left from right is dependent on an intact body schema. Laterality identification assesses how quickly and accurately the patient identifies whether an image of a hand, arm, or other body part is the left or right. Laterality cards are commercially available flash cards, each containing a photo of a left or right hand or arm. The patient is asked to quickly identify whether the image is of a left or right hand or arm (Fig. 100.1). This identification is done first in the clinic, and then the patient is sent home with pictures or magazines to use for home training. Cards may be purchased or fabricated. Laterality training is progressed by increasing the number of cards, improving the time or increasing the difficulty of the pictures.
23
Laterality training is the first phase in the GMI training because it is thought that until the patient has an accurate cortical representation of her or his body, it is counterproductive to move forward with cortical retraining.
23
The average RT of identifying the hand cards is greater than 80% accuracy and less than 2 ± 0.05 seconds
1
(Table 100.1). The patient should perform left–right training multiple times a day and for short periods at a time. According to Moseley and Acerra,
24
by disengaging the primary motor cortex, it promotes inhibition and helps with desmudging in the sensory cortex. Laterality training activates premotor cortices and reestablishes left and right concepts in the brain, which helps to decrease the firing of painful neurotags.
25
(Neurotags are formed by neurons that have learned to link with each other in the interpretation of a single input.) Phase I of GMI is usually performed at home by the patient for at least a 2-week period but varies from patient to patient. Additional timeframes are discussed later in the chapter. Left–right identification cards include the hands, feet, knees, backs, and shoulders. There are computer-driven laterality recognition software programs called Recognize and Orientate.
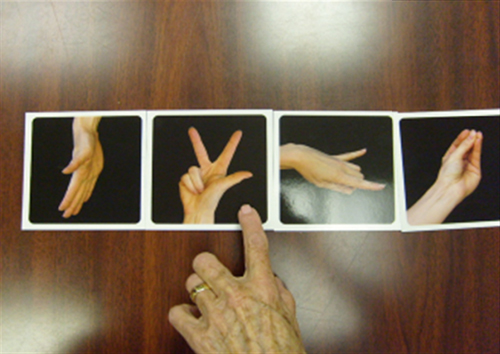
TABLE 100.1
Adapted from Mosely GL, Butler DS, Beames TB, Giles TJ. The Graded Motor Imagery Handbook. Adelaide, Australia: NOI Group; 2012.
Phase 2: Motor Imagery or Mental Imagery Rehearsal
Motor imagery has been around for many years, and it was a common practice with athletes before performing their sports (Fig. 100.2). During the past decade, there has been a rapid growth in the use of motor imagery in rehabilitation and psychology.
26,27
With motor imagery, the patient imagines his or her involved limb in a certain position. The patient is asked to imagine pain-free movement of that limb without increasing symptoms. Imagined hand movements are thought to activate the cortex in a similar manner to executed movements. Parsons
28
has shown that a motor imagery program, performed sequentially, activates cortical mechanisms associated with movement without evoking pain. When there is no pain, the patient then imagines the limb moving or imagines performing an activity with the limb. It is important to pace this phase by first having the patient imagine a static position before imagining movement of the limb. This phase of imagined movement is self-generated and activates areas of the brain similar to those of actual movement.
2
Other methods of using motor imagery involve having the patient view pictures of limb motions on the television or on video clips and then imagine performance of the observed motion(s) without pain. This is a conscious access to neurosignatures or movement memories representing intention, preparation, carrying out, and eventual movement, an approach that has been used in sports with healthy people.
27
The rationale for imagined movements is based on the findings that people with ongoing arm pain may experience pain just by thinking about movement. A Cochrane review on mental imagery revealed that mental imagery and physical practice more effectively improved upper extremity function than physical activity alone.
29
Even
though information from several research studies suggests that mental imagery may be very helpful and is a promising technique, more studies are needed to confirm its efficacy.
Phase 3: Mirror Therapy or Mirror Visual Feedback
Mirror therapy involves looking into a mirror and seeing the reflected image of the uninvolved limb in the mirror, which has been placed on the involved side, in front of the involved limb. The reflected image appears to be that of the involved limb (Fig. 100.3). The first step is to have the patient look at the image in the mirror and see the illusion of the involved hand. This creates the illusion that the injured hand is now without pain. It is important for the patient to be comfortable, with both hands relaxed. The patient is then asked what hand he or she sees in the mirror. When the reflected image in the mirror has been identified by the patient as the involved hand, movement of the uninvolved hand in front of the mirror can begin. If there are no increased symptoms while looking at the image of movement in the mirror, then bimanual movement may begin. The patient should move both hands in a bilateral synchronous manner to feel the movement at the same time as she or he is observing the reflection of the normal limb movement.
30
According to McCabe and coworkers,
30
mirror therapy is thought to provide strong positive sensory feedback to the motor cortex that not all movements need to be painful. Another theory notes that mirror therapy may work because of increased attention to the limb, causing improved ownership
25
; however, the exact mechanisms of mirror therapy are still not fully understood. McCabe and coworkers
30
reported using mirror therapy alone in treating patients with CRPS; this treatment was effective in patients with early CRPS. Others report use of all three phases of GMI because this is a sequential order that does not overwhelm the sensitized nervous system and exacerbate symptoms. Moseley and Butler
1
suggest that the entire three-phase approach be performed sequentially. This is a graded approach to cortical activation using techniques that activate cortical regions affiliated with movement preparation (priming) and then slowly staging movement execution of sight.
29
Laqueux and associates
31
reported on seven patients with CRPS who were treated with a modified GMI program and who had lessened pain as reflected on the short form McGill pain Questionnaire (P = .046), grip force improvement (P = .042), and global impression of change (P = .015) but not improved function on the DASH (Disabilities of the Arm, Shoulder and Hand) questionnaire.
All three phases of GMI use the principle of graded exposure; some studies have demonstrated that premature exposure to sensory or motor stimuli in patients with CRPS and limb pain leads to activation of painful neurotags.
1
The phasing in from left–right identification to motor imagery and then followed by mirror therapy follows the principles of graded exposure and is less likely to set off a painful neurotag or central sensitization. The goal in pain management with GMI is to achieve movement with decreased pain without causing increased symptoms. There is research backing the graded exposure theory that reveals improvement in cortical representation or reorganization in the brains neural network.
1,13,17
Moseley and Butler
1
stated that GMI helps prevent adverse responses by avoiding nonproductive neural pathways and reinforcing the development of productive pathways. Many have just looked at mirror therapy rather than all three phases of GMI,
30
and other studies have looked at the entire GMI process.
1
It is important to emphasize that GMI is just one part of treating patients with persistent chronic pain and should be combined with other therapies.
Pain and Graded Motor Imagery
Within the past 2 decades, progress has been made in the understanding of the changes in the CNS when there is persistent pain. According to Melzack’s
32
neuromatrix theory published in 1996, a combination of cortical mechanisms, when activated, produces danger for the brain and pain.
The brain considers multiple inputs when interpreting whether the incoming stimuli are dangerous to the body. The brain then evaluates all inputs and decides if protective action is needed, which determines the response. Researchers have determined that pain disrupts the body schema and the internal cortical representation of the involved limb in the homunculus, thus causing proprioceptive and sensorimotor dysfunctions, such as dystonia. Melzack
32
states that pain is produced by the brain after a neural signature has been activated and the brain concludes that the body is in danger.
1,31
Thus, a neurotag is formed by neurons that have learned to link with each other in the interpretation of a single input and therefore produce a programmed output. With GMI, we want to turn off painful neurotags and turn on painless neurotags. GMI works on the underlying mechanism that neural networks, which are normally involved in movement planning and execution, are also equally active during perception, perceptual reorganization, and imagined movements.
27
In recent years, it has become increasingly clear that pain includes both peripheral and central features.
23
The management of hands and upper extremities after musculoskeletal injuries has traditionally focused on the tissue, joint or bone involvement, and the
signs and symptoms originating from the physiological impairments. The peripheral identification of symptoms works for acute injuries, but when pain persists and symptoms increase, the therapist should consider that there is CNS plastic reorganization.
33
This involvement of the CNS is called central sensitization (central sensitivity) and, after acute injury or immobilization, should be addressed at the initial visit. This amplification of neural signaling within the CNS elicits pain and hypersensitivity and is considered a form of maladaptive plasticity.
11,34
The early indications for the use of GMI as described by Moseley
34
were for CRPS, phantom limb, and stroke. These three conditions involve cortical abnormalities or changes, and Moseley suggested that sequential cortical organization treatment without setting off a painful neurotag was necessary. In previous research, Moseley found that early movement of the involved limb in patients with CRPS evoked intolerable pain, and activating neural networks first, without movement, would reduce pain and swelling in patients with CRPS.
1,24
Moseley’s research with 13 patients with chronic CRPS compared a motor imagery program with ongoing therapy without GMI. The GMI program consisted of 2 weeks each of hand laterality recognition tasks or left–right identification and imagined hand movements for another 2 weeks followed by mirror therapy. The results showed that not starting with limb movement but instead starting with left–right recognition is effective for patients with CRPS.
25
A 1995 research study by Flor and associates
6
proved that there is neuronal activation in the somatosensory cortical area representing the amputated limb during the patient’s experience of phantom pain. Flor and associates
6
proposed that mirror therapy would reverse the cortical reorganization seen in patients with phantom limb pain and would alleviate the pain; this theory was supported through brain imaging. There is a relationship between the cortical reorganization and pain: as the cortical changes reverse and return to normal, the pain reduces, correlating with the maladaptive reorganization.
6,13
This illustrates that pain is a complex and dynamic process that is unique to each patient with pain. Therefore, incorporating GMI (i.e., retraining the brain) is necessary when treating patients with persistent pain.
Stroke and Graded Motor Imagery
Recovery after stroke depends on the remaining cortical sensorimotor networks for functional recovery and the reorganization processes. Neural reorganization depends on the information provided by sensorimotor efferent–afferent feedback loops.
35
When a functional area or system of the brain is completely damaged, recovery is achieved by other areas of the brain that are recruited to take over the functions of the area damaged by stroke.
36–38
Priming, as described by Stoykov and Madhaan,
39
is a change in behavior based on previous stimuli. The general theory about priming is that the brain that has been primed by prior activation is generally more responsive to accompanying or subsequent training. It has been suggested that GMI may be a priming technique. Studies on brain mapping have investigated motor priming as a tool for inducing neuroplasticity and enhancing the effects of rehabilitation. Other research has studied priming as a way to facilitate motor learning. In a 2011 article, Pomeroy and coworkers
40
categorized priming as a restorative intervention that reduces impairment by targeting underlying neural mechanisms in neurologic disorders such as stroke. Evidence has shown that GMI, as a targeted intervention designed to improve sensorimotor recovery in upper and lower extremities, to increase the functional use of the extremities, and to work in conjunction with conventional therapies directly influences brain changes or brain reorganization. However, current evidence has also shown that motor imagery may provide additional benefits to conventional therapies for neurologic injuries. More randomized studies are needed to assess the benefits of motor imagery for cortical insults in both acute and chronic conditions. Further information on this subject is beyond the scope of this chapter and can be reviewed in an article by Muratori and coworkers
41
in the Journal of Hand Therapy. GMI seems to be a feasible tool to consider when used with conventional stroke therapies.
39
Implications for Upper Extremity and Hand Rehabilitation
Symptoms after injury to the hand and upper extremity most often are related to acute musculoskeletal consequences and can be addressed by determining the tissue involved and developing the appropriate treatment strategies. After soft tissue, nerve, and bone injury of the hand, neuroscientific evidence over the years has shown that the primary sensory and motor areas of the cortex can change as a consequence of injury. In a 2017 randomized controlled study after distal radius fractures, 36 participants were randomly selected for the GMI (n = 17) or the control group (n = 19). The GMI group received traditional therapy with GMI, and the control group received traditional therapy for 8 weeks. The GMI group had improved range of motion as well as improved function on the DASH outcome measure with less pain.
42
Patients who have been immobilized may develop persistent pain and abnormal sensations such as allodynia and hyperalgesia. Allodynia and hyperalgesia are symptoms of CNS involvement called central sensitization.
34
Treating these symptoms by only addressing the peripheral changes is not always adequate. Strategies targeting brain changes are necessary, and the use of GMI is indicated. Individual case studies and a few small sample population studies reveal improvement in sensorimotor recovery after stroke using mirror therapy alone or using GMI. Polli and coworkers
43
used GMI in a nonrandomized controlled study of the clinical effect of GMI in 28 stroke patients (14 experimental and 14 control participants). The Wolf Motor Function Test and the 66-point motor section of the Fugl-Meyer Assessment were used to assess the outcomes of treatment for both groups. The experimental group treated with GMI had better outcome scores than the control group treated with conventional therapy.
43
Conclusion
The cortical representation of the body and the cortical reorganization that occurs after injury and immobilization and the functional consequences should be considered in all patients.
33
GMI is not a preset program but a guideline for progressing patients starting with nonmovement and progressing into movement therapies as the symptoms decline. As with other therapy treatments, GMI must be individually tailored for each patient and should not be used as a single treatment choice but incorporated into the overall treatment program. GMI is one of the treatments to arise from the paradigm shift in neuroplasticity.
35
Moseley and Butler
1
in their 2012 Graded Motor Imagery Handbook stress the importance of educating the patient in the updated neuroscience of pain before starting GMI. This knowledge allows the patient to be in control and not a passive recipient of treatment. For GMI to be successful, the therapist must use clinical reasoning regarding the timing and progression of GMI. At this time, few studies exist that identify the parameters of GMI, such as time frames and frequency. In general, brief sessions offered throughout the day are recommended (e.g., 15 minutes at a time for at least five to eight sessions daily).
In summary, GMI can be useful for upper extremity injuries in patients who have pain or do not want to move, patients who have kinesophobia (i.e., fear of movement), and patients who lack sensory motor control. Further research is needed to define parameters for the use of GMI for specific diagnostic groups. Although this treatment technique is in need of further research support, evidence is mounting advocating its benefits.

