14: Myelography and Other Central Nervous System Imaging

Anatomy

Two illustrations of the lateral view of the brain. The first one depicts the cerebellum above the brainstem and the lateral sulcus on the surface of the brain. The second one depicts the cerebrum, cerebellum and the corpus collosum below the cerebrum. The brainstem emerges from the center and consists of pons, medula oblongata, and midbrain. The spinal cord extends from the brainstem.
Brain
Spinal Cord
Meninges
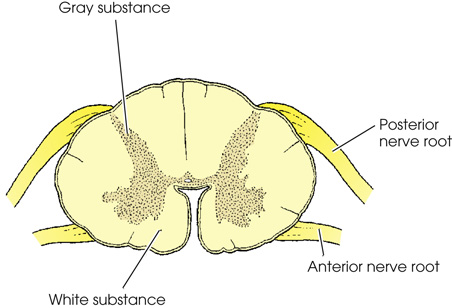
A transverse view of the spinal cord has a H shaped gray substance and a surrounding white substance. The posterior nerve root and the anterior nerve root rise from the top and the bottom portion respectively.

The lateral view of the head and upper body. The Pons lies at the base of the brain, followed by the medula oblongata. The spinal cord starts at the base of the brain and ends at the lumbar region. The Conus medullaris and Dural sac for cauda equina are at the end of the spinal cord.
Ventricular System

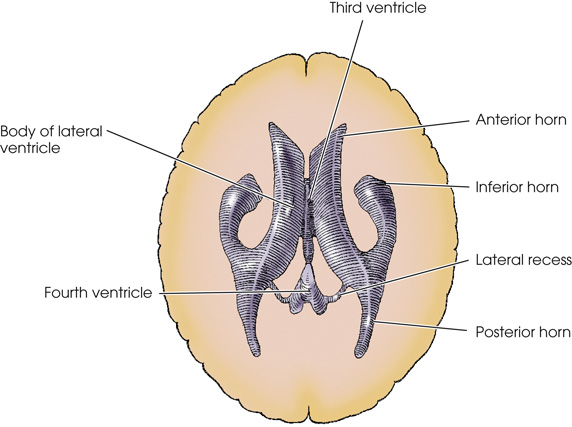
The lateral view of the cerebral ventricles of the brain. The lateral ventricle is a horn like U shaped structure at the center. It is extends into the anterior horn on the left and the posterior horn on the right. It is connected to a quadrilateral-shaped third ventricle through the intraventricular foramen. It is connected to the diamond-shaped fourth ventricle on the hind brain through the cerebral aqueduct.

The anterior view of the brain with the cerebrum ventricles. The ventricles are in the shape of m with the center between the two cerebral hemispheres. The lateral ventricle lies on the right hemisphere. The anterior horn is on the left end and the posterior horn is on the right end.

Radiography
Plain Radiographic Examination
Myelography

Contrast Media
Preparation of the Examining Room
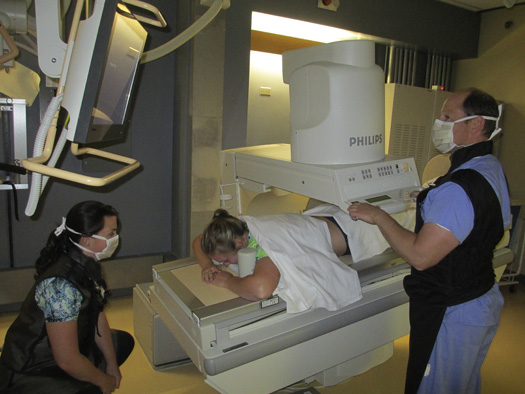
Examination Procedure

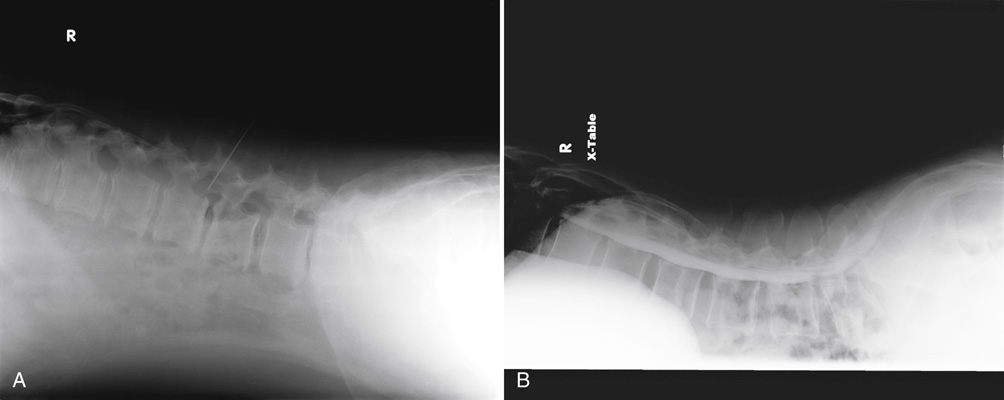
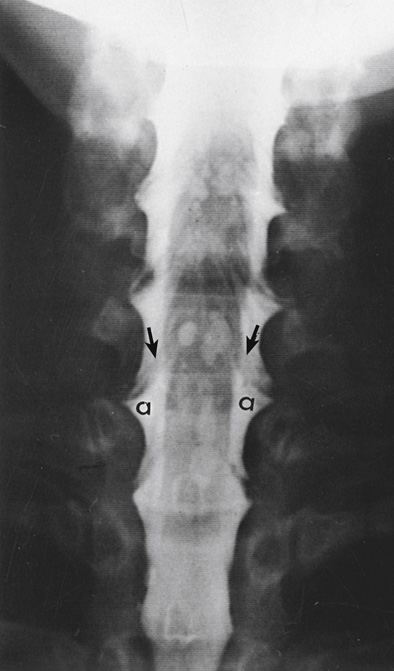

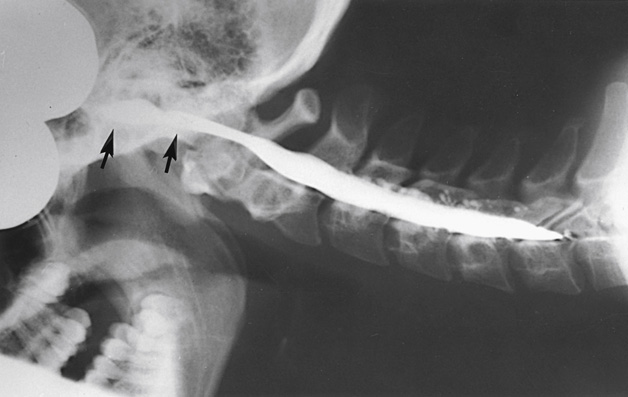
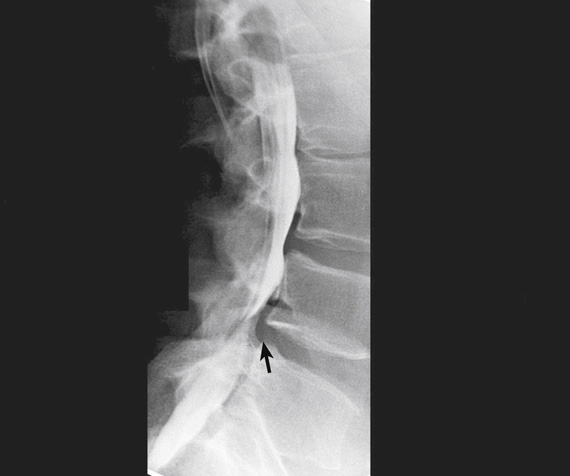
Lumbar Puncture
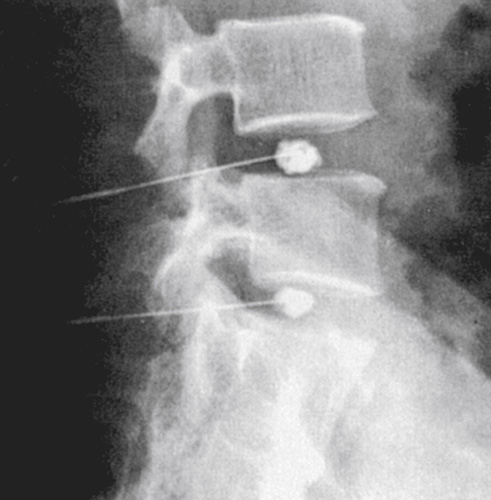
Vertebral Augmentation


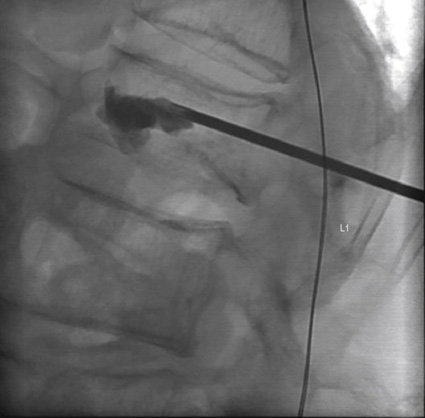

Other Neuroradiographic Procedures
Provocative Diskography
Interventional Pain Management

Definition of Terms
- arachnoid: Thin delicate membrane surrounding the brain and spinal cord.
- brain: Portion of the CNS contained within the cranium.
- cauda equina: Collection of nerves located in the spinal canal inferior to the spinal cord.
- cerebellum: Part of the brain located in the posterior cranial fossa behind the brain stem.
- cerebral aqueduct: Opening between the third and fourth ventricles.
- cerebrum: Largest uppermost portion of the brain.
- conus medullaris: Most inferior portion of the spinal cord.
- cortex: Outer surface layer of the brain.
- CSF: Cerebrospinal fluid—the fluid that flows through and protects the ventricles, subarachnoid space, brain, and spinal cord.
- dura mater: Tough outer layer of the meninges, which lines the cranial cavity and spinal canal.
- epidural space: Outside or above the dura mater.
- falx cerebri: Fold of dura mater that separates the cerebral hemispheres.
- filum terminale: Threadlike structure that extends from the distal end of the spinal cord.
- hindbrain: Portion of the brain within the posterior fossa; it includes the pons, medulla oblongata, and cerebellum.
- intrathecal injection: Injection into the subarachnoid space of the spinal canal.
- kyphoplasty: Interventional radiology procedure used to treat vertebral body compression fractures using a specialized balloon and bone cement.
- lumbar puncture: Procedure involving the insertion of a spinal needle into the subdural space to remove CSF for laboratory analysis, relieve pressure, or to inject medication (also known as spinal tap).
- pons: Oval-shaped area of the brain anterior to the medulla oblongata.
- spinal cord: Extension of the medulla oblongata that runs through the spinal canal to the upper lumbar vertebrae.
- tentorium cerebelli: Layer of dura that separates the cerebrum and cerebellum.
- vermis: Wormlike structure that connects the two cerebellar hemispheres.
- vertebroplasty: Interventional radiology procedure used to treat vertebral body compression fractures by stabilizing bone fragments with cement.