22: Physiologic and behavioral adaptations of the newborn
Kathryn Rhodes Alden
![]() http://evolve.elsevier.com/Perry/maternal
http://evolve.elsevier.com/Perry/maternal
The neonatal period includes the time from birth through day 28 of life. During this time, the neonate or newborn must make many physiologic and behavioral adaptations to extrauterine life. Physiologic adjustment tasks are those that involve: (1) establishing and maintaining respirations; (2) adjusting to circulatory changes; (3) regulating temperature; (4) ingesting, retaining, and digesting nutrients; (5) eliminating waste; and (6) regulating weight. Behavioral tasks include: (1) establishing a regulated behavioral tempo independent of the parent, which involves self-regulating arousal, self-monitoring changes in state, and patterning sleep; (2) processing, storing, and organizing multiple stimuli; and (3) establishing a relationship with caregivers and the environment. The term infant usually makes these adjustments with little or no difficulty. This chapter describes the physiologic and behavioral adaptations required by the neonate for transition to extrauterine life.
Stages of transition to extrauterine life
The major adaptations associated with transition from intrauterine to extrauterine life occur during the first 6 to 8 hours after birth. The predictable series of events during transition are mediated by the sympathetic nervous system and result in changes that involve heart rate, respirations, temperature, and gastrointestinal (GI) function. This transition period represents a time of vulnerability for the newborn and warrants careful observation. To detect disorders in adaptation soon after birth, nurses must be aware of normal features of the transition period.
In their classic work on newborn adaptation to extrauterine life, Desmond, Rudolph, and Phitaksphraiwan (1966) proposed three stages of newborn transition. The stages are still considered valid. The first stage of the transition period lasts up to 30 minutes after birth and is called the first period of reactivity. The newborn’s heart rate increases rapidly to 160 to 180 beats/min but gradually falls after 30 minutes or so to a baseline rate of 100 to 120 beats/min. Respirations are irregular, with a rate between 60 and 80 breaths/min. Fine crackles can be heard on auscultation. Audible grunting, nasal flaring, and retractions of the chest also can be present, but these should cease within the first hour after birth. The infant is alert and may have spontaneous startles, tremors, crying, and head movement from side to side. Bowel sounds are audible, and meconium may be passed.
After the first period of reactivity, the newborn either sleeps or has a marked decrease in motor activity. This period of decreased responsiveness lasts from 60 to 100 minutes. During this time the infant is pink, and respirations are rapid (up to 60 breaths/min) and shallow but unlabored. Bowel sounds are audible, and peristaltic waves may be noted over the rounded abdomen.
The second period of reactivity, the third stage of transition, occurs approximately between 2 and 8 hours after birth and lasts from 10 minutes to several hours. Brief periods of tachycardia and tachypnea occur, associated with increased muscle tone, changes in skin color, and mucus production. Meconium is commonly passed at this time. Most healthy newborns experience this transition, regardless of gestational age or type of birth; very preterm infants do not because of physiologic immaturity.
Physiologic adaptations
Respiratory system
As the infant emerges from the intrauterine environment and the umbilical cord is clamped and severed, profound adaptations are necessary for survival. The most critical of these is the establishment of effective respirations. Most newborns breathe spontaneously after birth and are able to maintain adequate oxygenation. Preterm infants often encounter respiratory difficulties related to their immature lungs.
Initiation of breathing
During intrauterine life, oxygenation of the fetus occurs through transplacental gas exchange. At birth, the lungs must be established as the site of gas exchange. In utero, fetal blood was shunted away from the lungs, but when birth occurs the pulmonary vasculature must be fully perfused for this purpose. Clamping the umbilical cord causes a rise in blood pressure (BP), which increases circulation and lung perfusion.
There is no single trigger for newborn respiratory function. The initiation of respirations in the neonate is the result of a combination of chemical, mechanical, thermal, and sensory factors (Blackburn, 2018).
Chemical factors
The activation of chemoreceptors in the carotid arteries and aorta results from the relative state of hypoxia associated with labor. With each labor contraction there is a temporary decrease in uterine blood flow and transplacental gas exchange, resulting in transient fetal hypoxia and hypercarbia. Although the fetus is able to recover between contractions, there appears to be a cumulative effect that results in progressive decline in Po2, increased Pco2, and lowered blood pH. Decreased levels of oxygen and increased levels of carbon dioxide seem to have a cumulative effect that is involved in initiating neonatal breathing by stimulating the respiratory center in the medulla. Another chemical factor may also play a role; it is thought that as a result of clamping the cord, there is a drop in levels of a prostaglandin that can inhibit respirations.
Mechanical factors
Respirations in the newborn can be stimulated by changes in intrathoracic pressure resulting from compression of the chest during vaginal birth. As the infant passes through the birth canal, the chest is compressed. With birth this pressure on the chest is released, and the negative intrathoracic pressure helps to draw air into the lungs. Crying increases the distribution of air in the lungs and promotes expansion of the alveoli. The positive pressure created by crying helps to keep the alveoli open.
Thermal factors
At birth the newborn enters the extrauterine environment in which the temperature is significantly lower. The profound change in environmental temperature stimulates receptors in the skin, resulting in stimulation of the respiratory center in the medulla.
Sensory factors
Sensory stimulation occurs in a variety of ways at birth. Some of these include handling by the obstetric health care provider, suctioning the mouth and nose, and drying by the nurses. Environmental factors (lights, sounds, smells) stimulate the respiratory center.
Establishing respiration
At term the lungs hold approximately 20 mL of fluid per kilogram. Air must be substituted for the fluid that filled the fetal respiratory tract. Traditionally, it had been thought that the thoracic squeeze occurring during normal vaginal birth resulted in significant clearance of lung fluid. However, it appears that this event plays a minor role. In the days preceding labor, there is reduced production of fetal lung fluid with a corresponding decrease in alveolar fluid volume. Shortly before the onset of labor, there is a catecholamine surge that seems to promote fluid clearance from the lungs, which continues during labor. The movement of lung fluid from the air spaces occurs through active transport into the interstitium, with drainage occurring through the pulmonary circulation and lymphatic system. Retention of lung fluid can interfere with the infant’s ability to maintain adequate oxygenation, especially if other factors that compromise respirations (e.g., meconium aspiration, congenital diaphragmatic hernia, esophageal atresia with fistula, choanal atresia, congenital cardiac defect, immature alveoli) are present. Infants born by cesarean in which labor did not occur before birth can experience some lung fluid retention, although it typically clears without harmful effects on the infant. These infants are also more likely to develop transient tachypnea of the newborn (TTN) (Fraser, 2021).
The alveoli of the term infant’s lungs are lined with surfactant, a lipoprotein manufactured in type II lung cells. Lung expansion depends largely on chest wall contraction and adequate surfactant secretion. Surfactant lowers surface tension, therefore reducing the pressure required to keep the alveoli open with inspiration, and prevents total alveolar collapse on exhalation, thereby maintaining alveolar stability. The decreased surface tension results in increased lung compliance, helping to establish the functional residual capacity of the lungs (Blackburn, 2018). With absent or decreased surfactant, more pressure must be generated for inspiration, which can soon tire or exhaust preterm or sick term infants.
Breathing movements that began in utero as intermittent become continuous after birth, although the mechanism for this is not well understood. Once respirations are established, breaths are shallow and irregular, ranging from 30 to 60 breaths/min, with periods of breathing that include pauses in respirations lasting less than 20 seconds. These episodes of periodic breathing occur most often during the active (rapid eye movement [REM]) sleep cycle and decrease in frequency and duration with age. Apneic periods longer than 20 seconds are abnormal and should be evaluated.
Newborn infants are by preference nose breathers, which enhances the ability to coordinate sucking, swallowing, and breathing. The reflex response to nasal obstruction is to open the mouth to maintain an airway. This response is not present in most infants until 3 weeks after birth; therefore, cyanosis or asphyxia can occur with nasal blockage.
In most newborns, auscultation of the chest reveals loud, clear breath sounds that seem very near because little chest tissue intervenes. Breath sounds should be clear and equal bilaterally, although fine rales for the first few hours are not unusual. The ribs of the infant articulate with the spine at a horizontal rather than a downward slope; consequently, the rib cage cannot expand with inspiration as readily as that of an adult. Because neonatal respiratory function is largely a matter of diaphragmatic contraction, abdominal breathing is characteristic of newborns. The newborn infant’s chest and abdomen rise simultaneously with inspiration.
Signs of respiratory distress
Signs of respiratory distress can include nasal flaring, intercostal or subcostal retractions (in-drawing of tissue between the ribs or below the rib cage), or grunting with respirations. Suprasternal or subclavicular retractions with stridor or gasping most often represent an upper airway obstruction. Seesaw or paradoxic respirations (exaggerated rise in abdomen with respiration as the chest falls) instead of abdominal respirations are abnormal and should be reported. A respiratory rate of less than 30 or greater than 60 breaths/min with the infant at rest must be evaluated. The respiratory rate of the infant can be slowed, depressed, or absent as a result of the effects of analgesics or anesthetics administered to the mother during labor and birth. Apneic episodes can be related to events such as rapid increase in body temperature, hypothermia, hypoglycemia, or sepsis. Tachypnea can result from inadequate clearance of lung fluid, or it can be an indication of respiratory distress syndrome (RDS). Tachypnea can be the first sign of respiratory, cardiac, metabolic, or infectious illnesses (Gardner, Enzman-Hines, & Nyp, 2021).
Changes in the infant’s color can indicate respiratory distress. Normally, within the first 3 to 5 minutes after birth, the newborn’s color changes from blue to pink. Acrocyanosis, the bluish discoloration of hands and feet, is a normal finding in the first 24 hours after birth (Fig. 22.1). Transient periods of duskiness while crying are common immediately after birth; however, central cyanosis is abnormal and signifies hypoxemia. With central cyanosis, the lips and mucous membranes are bluish (circumoral cyanosis). It can be the result of inadequate delivery of oxygen to the alveoli, poor perfusion of the lungs that inhibits gas exchange, or cardiac dysfunction. Because central cyanosis is a late sign of distress, newborns usually have significant hypoxemia when cyanosis appears.

A newborn shows bluish discolouration on hands and feet. A pair of gloved hands holds the newborn with one hand supporting the neck and the other hand under the hips. Another gloved hand is shown at the back of the newborn for support.
Source: (Courtesy Barbara Wilson, West Jordan, UT.)Infants who experience mild TTN often have signs of respiratory distress during the first 1 to 2 hours after birth as they transition to extrauterine life. Tachypnea with rates up to 100 breaths/min can be present along with intermittent grunting, nasal flaring, and mild retractions. Supplemental oxygen may be needed. TTN usually resolves in 48 to 72 hours (Blackburn, 2018).
In neonates with more serious respiratory problems, symptoms of distress are more pronounced and tend to last beyond the first 2 hours after birth. Respiratory rates can exceed 120 breaths/min. Moderate to severe retractions, grunting, pallor, and central cyanosis can occur. The respiratory symptoms can be accompanied by hypotension, temperature instability, hypoglycemia, acidosis, and signs of cardiac problems. Common respiratory complications affecting neonates include RDS, meconium aspiration, pneumonia, and persistent pulmonary hypertension of the newborn (PPHN). Congenital defects such as anomalies of the great vessels, diaphragmatic hernia, or chest wall defects can cause severe respiratory problems. Blood incompatibilities such as hydrops fetalis can result in respiratory compromise (Gardner et al., 2021) (see Chapter 25).
Cardiovascular system
The cardiovascular system changes significantly after birth. The infant’s first breaths, combined with increased alveolar capillary distention, inflate the lungs and reduce pulmonary vascular resistance to pulmonary blood flow from the pulmonary arteries. Pulmonary artery pressure drops, and pressure in the right atrium declines. Increased pulmonary blood flow from the left side of the heart increases pressure in the left atrium, which causes a functional closure of the foramen ovale. During the first few days of life, crying can temporarily reverse the flow through the foramen ovale and lead to mild cyanosis. Soon after birth, cardiac output nearly doubles and blood flow increases to the lungs, heart, kidneys, and GI tract.
In utero, fetal Po2 is 20 to 30 mm Hg. After birth, when the Po2 level in the arterial blood approximates 50 mm Hg, the ductus arteriosus constricts in response to increased oxygenation. Circulating prostaglandin E2 (PGE2) levels also have an important role in closing the ductus arteriosus. In term infants, it functionally closes within the first 24 hours after birth; permanent (anatomic) closure usually occurs within 2 to 3 months, and the ductus arteriosus becomes a ligament. The ductus arteriosus can reopen in response to low oxygen levels in association with hypoxia, asphyxia, prolonged crying, or pathologic problems. With auscultation of the chest, a patent ductus arteriosus can be detected as a heart murmur (Blackburn, 2018).
When the cord is clamped and severed, the umbilical arteries, umbilical vein, and ductus venosus are functionally closed; they are converted into ligaments within 2 to 3 months. The hypogastric arteries also occlude and become ligaments.
Heart rate and sounds
The heart rate for a term newborn ranges from 120 to 160 beats/min, with brief fluctuations greater and less than these values usually noted during sleeping and waking states. The range of the heart rate in the term infant is approximately 80 to 100 beats/min during deep sleep and can increase to 180 beats/min or higher when the infant cries. A heart rate that is either high (more than 160 beats/min) or low (fewer than 100 beats/min) should be reevaluated within 30 minutes to 1 hour or when the activity of the infant changes.
The apical impulse (point of maximal impulse [PMI]) in the newborn is at the fourth intercostal space and to the left of the midclavicular line. The PMI is often visible and easily palpable because of the thin chest wall; this is also called precordial activity.
Irregular heart rate or sinus dysrhythmia is common in the first few hours of life but thereafter may need to be evaluated. Heart sounds during the neonatal period are of higher pitch, shorter duration, and greater intensity than during adult life. The first sound (S1) is typically louder and duller than the second sound (S2), which is sharp. The third and fourth heart sounds are not audible in newborns. Most heart murmurs heard during the neonatal period have no pathologic significance, and more than one-half of the murmurs disappear by 6 months of age. However, the presence of a murmur and accompanying signs such as poor feeding, apnea, cyanosis, or pallor is considered abnormal and should be investigated. There can be significant cardiac defects without a murmur or other symptoms. This reinforces the importance of ongoing assessment.
Blood pressure
The primary factors affecting BP are gestational age, postconceptional age, and birth weight (Flynn, 2021). BP values rise as these variables increase. Cuff size, state of alertness, and movement also affect BP measurement. Nurses and health care providers can compare infant BP measurements with available nomograms and tables to determine if values are within expected parameters. The mean arterial pressure (MAP) should be nearly equivalent to the weeks of gestation. For example, an infant born at 40 weeks of gestation should have a MAP of at least 40. The BP increases predictably over the first 5 days of life and then levels off, with minor variations noted during the first month of life. A drop in systolic BP (approximately 15 mm Hg) in the first hour of life is common. Expected values for BP (systolic/diastolic) in a term infant are:
Blood volume
Blood volume in the term newborn ranges from 80 to 100 mL/kg of body weight. In the preterm infant, the range is 90 to 105 mL/kg (Diehl-Jones & Fraser, 2021). The preterm infant has a relatively greater blood volume than the term newborn. This occurs because the preterm infant has a proportionately greater plasma volume, not a greater red blood cell (RBC) mass.
Delayed clamping of the umbilical cord changes the circulatory dynamics of the newborn. Delayed cord clamping (DCC) expands the blood volume from the so-called placental transfusion of blood to the newborn by as much as 100 mL, depending on the length of time to cord clamping and cutting. DCC has been associated with increased blood volume and BP and reduced risk for intraventricular hemorrhage and necrotizing enterocolitis. These benefits are most important for preterm infants. Polycythemia that occurs with delayed clamping is usually not harmful, although there can be an increased risk for hyperbilirubinemia that requires phototherapy. The American College of Obstetricians and Gynecologists (ACOG) recommends delayed cord clamping for at least 30 to 60 seconds for term and preterm infants unless there is a specific need for immediate clamping due to neonatal or maternal factors (ACOG, 2020) (see Chapter 16).
Signs of cardiovascular problems
Variations in vital signs can be indicative of cardiovascular problems. Persistent tachycardia (more than 160 beats/min) can be associated with anemia, hypovolemia, hyperthermia, or sepsis. Persistent bradycardia (less than 80 beats/min) can be a sign of a congenital heart block or hypoxemia. Unequal or absent pulses, bounding pulses, and decreased or elevated BP can indicate cardiovascular problems.
The newborn’s skin color can reflect cardiovascular problems. Pallor in the immediate postbirth period is often a sign of underlying problems such as anemia or marked peripheral vasoconstriction as a result of intrapartum asphyxia or sepsis. Cyanosis other than in the hands or feet, with or without increased work of breathing, can indicate respiratory and/or cardiac problems. The presence of jaundice can indicate ABO or Rh factor incompatibility problems (see Chapter 25).
Congenital heart defects are the most common types of congenital malformations (Centers for Disease Control and Prevention [CDC], 2020) (see Chapter 25). Although the more serious defects such as tetralogy of Fallot are likely to have clinical manifestations such as cyanosis, dyspnea, and hypoxia, others such as small ventricular septal defects can be asymptomatic. The prenatal history can provide information regarding risk factors for congenital heart defects, alerting the nurse to observe for symptoms. Maternal illness such as rubella, metabolic disease such as diabetes, and maternal drug ingestion are associated with an increased risk for cardiac defects.
Hematologic system
Red blood cells
Because fetal circulation is less efficient at oxygen exchange than the lungs, the fetus needs additional RBCs for transport of oxygen in utero. At birth the average levels of RBCs, hemoglobin, and hematocrit are higher than those in the adult; these levels fall slowly over the first month. At birth, the RBC count ranges from 4.6 to 5.2 million/mm3 (Blackburn, 2018). The term newborn can have a hemoglobin concentration of 14 to 24 g/dL at birth, decreasing gradually to 12 to 20 g/dL during the first 2 weeks (Pagana, Pagana, & Pagana, 2019). Hematocrit at birth ranges from 51% to 56%, increases slightly in the first few hours or days as fluid shifts from intravascular to interstitial spaces (Blackburn), and by 8 weeks is between 39% and 59% (Pagana et al.). Polycythemia (central venous hematocrit greater than 65%) can occur in term and preterm infants as a result of DCC, maternal hypertension or diabetes, or intrauterine growth restriction.
The source of the sample is a significant factor in levels of RBCs, hemoglobin, and hematocrit. Capillary blood yields higher values than venous blood.
The timing of blood sampling is also significant; the slight rise in RBCs after birth is followed by a substantial drop. At birth the infant’s blood contains an average of 70% to 80% fetal hemoglobin; however, because of the shorter life span of the cells containing fetal hemoglobin, the percentage falls rapidly, so that by the age of 6 to 12 months, levels of fetal hemoglobin are less than 2% of the total hemoglobin (Chou, 2020). Iron stores generally are sufficient to sustain normal RBC production for approximately 4 months in the term infant, at which time a transient physiologic anemia can occur.
Leukocytes
Leukocytosis, with a white blood cell (WBC) count ranging from 9000 to 30,000/mm3, is normal at birth (Pagana et al., 2019). The number of WBCs increases up to 24,000/mm3 during the first day after birth. The initial high WBC count of the newborn decreases rapidly, and a stable level of 12,000/mm3 is normally maintained during the neonatal period (Blackburn, 2018). Newborns are susceptible to infection. Leukocytes, especially the polymorphonuclear neutrophils, are limited in their ability to recognize foreign protein and localize and fight infection early in life (Benjamin & Maheshwari, 2020). Sepsis can be accompanied by a corresponding rise in neutrophils; however, some infants initially have clinical signs of sepsis without a significant elevation in WBCs.
Platelets
Platelets appear to be activated during the birth process and demonstrate improved aggregation in the first hours after birth. The platelet count ranges between 150,000 and 300,000/mm3 and is essentially the same in newborns as in adults. Levels of vitamin K-dependent clotting factors II, VII, IX, and X at birth are about 50% of adult levels and increase slowly after birth. Levels of factors V, VIII, and XIII at birth resemble adult ranges (Letterio, Pateva, Petrosiute, et al., 2020).
Blood groups
The infant’s blood group is determined genetically and established early in fetal life. However, during the neonatal period the strength of the agglutinogens present in the RBC membrane gradually increases. Cord blood samples can be used to identify the infant’s blood type and Rh status.
Thermogenic system
Next to establishing respirations and effective extrauterine circulation, heat regulation is most critical to the newborn’s survival. During the first 12 hours after birth, the neonate attempts to achieve thermal balance in adjusting to the extrauterine environmental temperature. Thermoregulation is the maintenance of balance between heat loss and heat production. Newborns attempt to stabilize their core body temperatures within a narrow range. Hypothermia from excessive heat loss is a common and potentially serious problem.
Anatomic and physiologic characteristics of neonates place them at risk for heat loss. Newborns have a thin layer of subcutaneous fat. The blood vessels are close to the surface of the skin. Newborns have larger body surface-to-body weight (mass) ratios than children and adults. Changes in environmental temperature alter the temperature of the blood, thereby influencing temperature regulation centers in the hypothalamus (Blackburn, 2018).
Heat loss
The body temperature of newborn infants depends on the heat transfer between the infant and the external environment. Factors that influence heat loss to the environment include the temperature and humidity of the air, the flow and velocity of the air, and the temperature of surfaces in contact with and around the infant. The goal of care is to provide a neutral thermal environment for the newborn in which heat balance is maintained. The neutral thermal environment is the ideal environmental temperature that allows the newborn to maintain a normal body temperature to minimize oxygen and glucose consumption. Heat loss in the newborn occurs by four modes:
- 1. Convection is the flow of heat from the body surface to cooler ambient air. Because of heat loss by convection, the ambient temperature in newborn care areas should range between 22°C and 26°C (72°F to 78°F) and the humidity between 30% and 60% (American Academy of Pediatrics [AAP] & ACOG, 2017). Newborns in open bassinets are wrapped to protect them from the cold. A cap may be worn to decrease heat loss from the infant’s head.
- 2. Radiation is the loss of heat from the body surface to a cooler solid surface not in direct contact but in relative proximity. To prevent this type of loss, bassinets and examining tables are placed away from outside windows, and care is taken to avoid direct air drafts.
- 3. Evaporation is the loss of heat that occurs when a liquid is converted to a vapor. In the newborn, heat loss by evaporation occurs as a result of moisture vaporization from the skin. This heat loss is intensified by failing to completely dry the newborn after birth or with bathing. Evaporative heat loss, as a component of insensible water loss, is the most significant cause of heat loss in the first few days of life.
- 4. Conduction is the loss of heat from the body surface to cooler surfaces in direct contact. During the initial assessment, the newborn is placed on a prewarmed bed under a radiant warmer to minimize heat loss. The scales used for weighing the newborn should have a protective cover to minimize conductive heat loss.
Heat loss must be controlled to protect the infant. Control of such modes of heat loss is the basis of caregiving policies and techniques. Drying the infant quickly after birth is essential to prevent hypothermia. Skin-to-skin contact with the mother is an effective means of reducing conductive and radiant heat loss and enhancing newborn temperature control and maternal-infant interaction. The naked newborn is placed on the mother’s bare chest and covered with a warm blanket; a cap may be placed on the infant’s head to help conserve heat (Fig. 22.2). Alternatively, the neonate is placed under a radiant warmer to reduce heat loss and promote thermoregulation.

A mother lying on a bed with a newborn baby on top of her chest and both are covered by sheets till the woman’s upper arm.
Source: (Courtesy Ashley Martin, Denver, NC.)Thermogenesis
In response to cold, the neonate attempts to generate heat (thermogenesis) by increasing muscle activity. Cold infants may cry and appear restless. Because of vasoconstriction the skin can feel cool to touch, and acrocyanosis can be present. There is an increase in cellular metabolic activity, primarily in the brain, heart, and liver; this also increases oxygen and glucose consumption.
In an effort to conserve heat, term newborns assume a position of flexion that helps to guard against heat loss because it diminishes the amount of body surface exposed to the environment. Infants also can reduce the loss of internal heat through the body surface by constricting peripheral blood vessels.
Adults are able to produce heat through shivering; however, the shivering mechanism of heat production is rarely operable in the newborn unless there is prolonged cold exposure (Blackburn, 2018). Newborns produce heat through nonshivering thermogenesis, triggered when the skin temperature decreases to less than 95°F to 96.8°F (35°C to 36°C). This is accomplished primarily by metabolism of brown fat, which is unique to the newborn; and secondarily by increased metabolic activity in the brain, heart, and liver. Brown fat is located in superficial deposits in the interscapular region and axillae and in deep deposits at the thoracic inlet, along the vertebral column, and around the kidneys (Fig. 22.3). Brown fat has a richer vascular and nerve supply than ordinary fat. Heat produced by intense lipid metabolic activity in brown fat can warm the newborn by increasing heat production as much as 100%. Reserves of brown fat, usually present for several weeks after birth, are rapidly depleted with cold stress. The amount of brown fat reserve increases with the weeks of gestation. A full-term newborn has greater stores than a preterm infant (Brand & Shippey, 2021).

An illustration shows the anterior and posterior views of a newborn. The anterior view shows brown fat located in the neck, thoracic inlet, around the heart, and along the vertebral column. The posterior view shows brown fat located in interscapular region and around the kidneys.
Source: (Modified from Murray, S. S., & McKinney, E. S. [2010]. Foundations of maternal-newborn and women’s health nursing [6th ed.]. St. Louis: Elsevier.)Hypothermia and cold stress
When the neonate’s temperature drops, in response to norepinephrine release, vasoconstriction occurs as a mechanism to conserve heat. The infant can appear pale and mottled; the skin feels cool, especially on the extremities. If the hypothermia is not corrected, it will progress to cold stress, which imposes metabolic and physiologic demands on all infants, regardless of gestational age and condition. The respiratory rate increases in response to the increased need for oxygen. In the cold-stressed infant, oxygen consumption and energy are diverted from maintaining normal brain and cardiac function and growth to thermogenesis for survival. If the infant cannot maintain an adequate oxygen tension, pulmonary vasoconstriction follows and jeopardizes pulmonary perfusion. As a consequence, the Po2 is decreased and the blood pH drops. Surfactant synthesis can be altered. These changes can prompt a transient respiratory distress or aggravate existing RDS. Moreover, decreased pulmonary perfusion and oxygen tension can maintain or reopen the right-to-left shunt across the ductus arteriosus.
The basal metabolic rate increases with cold stress. If cold stress is protracted, anaerobic glycolysis occurs, resulting in increased production of acids. Metabolic acidosis develops, and, if a defect in respiratory function is present, respiratory acidosis also develops (Fig. 22.4). Excessive fatty acids can displace the bilirubin from the albumin-binding sites and exacerbate hyperbilirubinemia. Hypoglycemia is another metabolic consequence of cold stress. The process of anaerobic glycolysis can deplete existing stores. If the infant is sufficiently stressed and low glucose stores are not replaced, hypoglycemia, which can be asymptomatic in the newborn, can develop (Gardner & Cammack, 2021).

An illustration shows a newborn lying sideways. A flowchart reads as follows: Cold stress increases oxygen consumption that leads to increased respiratory rate. This in turn affects pulmonary vasoconstriction that decreases oxygen uptake by lungs; and peripheral vasoconstriction that decreases oxygen to tissues. This then increases anaerobic glycolysis that decreases PO2 and pH resulting in metabolic acidosis.
Source: (Modified from Murray, S. S., & McKinney, E. S. [2010]. Foundations of maternal-newborn and women’s health nursing [6th ed.]. St. Louis: Elsevier.)Hyperthermia
Although less frequently than hypothermia, hyperthermia can occur and must be corrected. A body temperature greater than 37.5°C (99.5°F) is considered to be abnormally high and is typically caused by excess heat production related to sepsis or a decrease in heat loss. Hyperthermia can result from the inappropriate use of external heat sources such as radiant warmers, phototherapy, sunlight, increased environmental temperature, and the use of excessive clothing or blankets. The clinical appearance of the infant who is hyperthermic often indicates the causative mechanism. Infants who are overheated because of environmental factors such as being swaddled in too many blankets exhibit signs of heat-losing mechanisms: skin vessels dilate, skin appears flushed, hands and feet are warm to touch, and the infant assumes a posture of extension. The newborn who is hyperthermic because of sepsis appears stressed: vessels in the skin are constricted, color is pale, and hands and feet are cool. Hyperthermia develops more rapidly in a newborn than in an adult because of the relatively larger surface area of an infant. Sweat glands do not function well. Hyperthermia can cause neurologic injury and increased risk for seizures; severe cases can result in heat stroke and death (Brand & Shippey, 2021; Gardner & Cammack, 2021).
Renal system
At term, the kidneys occupy a large portion of the posterior abdominal wall. The bladder lies close to the anterior abdominal wall and is both an abdominal and a pelvic organ. In the newborn, almost all palpable masses in the abdomen are renal in origin.
At birth, a small quantity (approximately 40 mL) of urine is usually present in the bladder of a full-term infant. Many newborns void at the time of birth, although this is easily missed and may not be recorded. During the first few days, term infants generally excrete 15 to 60 mL/kg/day of urine; output gradually increases over the first month (Blackburn, 2018). The frequency of voiding varies from 2 to 6 times per day during the first and second days of life and increases during the subsequent 24 hours. After day 4, approximately six to eight voidings per day of pale straw-colored urine indicate adequate fluid intake.
During the first few days after birth, urine specific gravity for full-term newborns ranges from 1.008 to 1.012 (Dell, 2020). Newborns have limited capacity to concentrate urine; urine osmolality is about half that of a 2-year-old (Vogt & Springel, 2020). Normal urine during early infancy is usually straw colored and almost odorless. Sometimes pink-tinged uric acid crystals or “brick dust” appear on the diaper. Uric acid crystals are normal during the first week but thereafter can be a sign of inadequate intake. Loss of fluid through urine, feces, lungs, increased metabolic rate, and limited fluid intake can result in a 5% to 10% loss of the birth weight over the first 3 to 5 days. Excessive weight loss can be related to feeding difficulties or other issues. The neonate should regain the birth weight within 10 to 14 days, depending on the feeding method (breastfeeding, breast milk feeding, or infant formula).
Fluid and electrolyte balance
In the term neonate, approximately 75% of body weight consists of total body water (extracellular and intracellular). A reduction in extracellular fluid occurs with diuresis during the first few days after birth. The weight loss experienced by most newborns during the first few days after birth is caused primarily by extracellular water loss (Dell, 2020).
The daily fluid requirement for neonates weighing more than 1500 g is 60 to 80 mL/kg during the first 2 days of life. From 3 to 7 days the requirement is 100 to 150 mL/kg/day, and from 8 to 30 days it is 120 to 180 mL/kg/day (Dell, 2020).
At birth, the glomerular filtration rate (GFR) of a newborn is significantly lower than in the adult. This results in a decreased ability to remove nitrogenous and other waste products from the blood. The GFR rapidly increases during the 2 to 4 weeks after birth as a result of postnatal physiologic changes, including decreased renal vascular resistance, increased renal blood flow, and increased filtration pressure. The GFR gradually rises to adult levels by 2 years of age (Blackburn, 2018; Vogt & Springel, 2020).
Sodium reabsorption is decreased as a result of a lowered sodium- or potassium-activated adenosine triphosphatase activity. The decreased ability to excrete excess sodium results in hypotonic urine compared with plasma, leading to a higher concentration of sodium, phosphates, chloride, and organic acids and a lower concentration of bicarbonate ions.
Tubular reabsorption of glucose in the term neonate is similar to that of an adult. Although the renal threshold for glucose is lower, newborns do not typically exhibit glycosuria.
Because of a lower renal threshold for bicarbonate and a limited capacity for reabsorption, the neonate’s serum bicarbonate and plasma pH levels are lower. Buffering capacity is decreased. This reduces the newborn’s ability to cope with events (e.g., cold stress) that produce acidosis (Blackburn, 2018).
Signs of renal system problems
The renal system has a wide range of functions. Dysfunction resulting from physiologic abnormalities can range from the lack of a steady stream of urine to gross anomalies such as hypospadias and exstrophy of the bladder, which can be identified easily at birth. Enlarged or cystic kidneys can be identified as masses during abdominal palpation. Some kidney anomalies also can be detected by ultrasound examination during pregnancy (see Chapter 25).
Gastrointestinal system
The full-term newborn is capable of swallowing, digesting, metabolizing, and absorbing proteins and simple carbohydrates and emulsifying fats. With the exception of pancreatic amylase, the characteristic digestive enzymes are present even in low birth weight neonates.
In the adequately hydrated infant, the mucous membrane of the mouth is moist and pink; the hard and soft palates are intact. The presence of moderate to large amounts of mucus is common in the first few hours after birth. Small whitish areas (Epstein pearls) may be found on the gum margins and at the juncture of the hard and soft palates. The cheeks are full because of well-developed sucking pads. These, like the labial tubercles (sucking calluses) on the upper lip, disappear at approximately 12 months of age when the sucking period is over.
Feeding behavior is related to gestational age and is influenced by neuromuscular maturity, maternal medications during labor and birth, and the type of initial feeding. Feeding requires that the neonate is able to coordinate sucking, swallowing, and breathing. Sucking is a reflex behavior that begins in utero as early as 15 to 16 weeks. By 28 weeks, some infants can coordinate sucking and swallowing. By 32 to 34 weeks, most are able to coordinate sucking, swallowing, and breathing; these abilities are well developed by 36 to 38 weeks (Blackburn, 2018). Sucking takes place in small bursts of 3 or 4 and up to 8 to 10 sucks at a time, with a brief pause between bursts. The neonate is unable to move food from the lips to the pharynx; therefore placing the nipple (breast or bottle) well inside the baby’s mouth is necessary. Peristaltic activity in the esophagus is uncoordinated in the first few days of life. It quickly becomes a coordinated pattern in healthy full-term infants, and they swallow easily.
Teeth begin developing in utero, with enamel formation continuing until approximately 10 years of age. Tooth development is influenced by neonatal or infant illnesses and medications and by maternal illnesses or medications taken by the mother during pregnancy. The fluoride level in the water supply also influences tooth development. Occasionally an infant may be born with one or more teeth. These natal teeth have poorly formed roots and, as they loosen, place the infant at risk for aspiration; for this reason natal teeth are often extracted.
The mucosal barrier in the intestines is not fully mature until 4 to 6 months of age, which allows antigens and other macromolecules such as bacteria to be transported across the intestinal wall into the systemic circulation. This increases the risk for allergies and infection (Blackburn, 2018).
Intestinal flora, or gut microbiota, are established within the first week after birth, and normal intestinal flora help to synthesize vitamin K, folate, and biotin. Traditionally, it was thought that the fetus grows and develops in a sterile environment. Research on the human microbiome suggests that the pregnant woman and the developing fetus coexist with a variety of commensal and symbiotic microbes that have important influences on the health of both the mother and her infant. Research evidence of microbial presence in amniotic fluid, placenta, and meconium indicates that the fetus is exposed to microbes during pregnancy. The mode of birth (vaginal or cesarean) seems to play a major role in the microbial colonization of the neonate. Infants born vaginally appear to be initially colonized by the maternal vaginal microbes, whereas infants born by cesarean are first colonized by maternal skin microbes. This initial colonization plays a major role in establishing intestinal flora; research is ongoing to identify the implications for the child’s future health. The microbiome of the infant is also influenced by diet, antibiotics, and environmental factors (Neu, 2018).
Breastfeeding is important in establishing the intestinal microbiome of the newborn. Human milk contains a variety of microbes that appear to originate in the mother’s GI tract. Oligosaccharides in human milk may have a prebiotic function that facilitates the growth of beneficial bacteria in the neonatal GI tract (Neu, 2018).
The capacity of the newborn stomach varies widely, depending on the size of the infant, from less than 10 mL on day 1 to nearly 30 mL on day 3 and expanding to 60 mL on day 7. After birth, the newborn stomach becomes increasingly more compliant and relaxed to accommodate larger volumes. Several factors such as time and volume of feedings or type and temperature of food can affect the emptying time.
The normal intermittent relaxation of the lower esophageal sphincter results in involuntary backflow of stomach contents into the esophagus, known as gastroesophageal reflux (GER). As a result, newborns are prone to regurgitation, “spitting,” and vomiting, especially during the first 3 months. GER can be minimized by avoiding overfeeding, burping, and positioning the infant with the head slightly elevated.
In some infants, GER is severe enough to cause dysphagia, esophagitis, and aspiration. This is known as gastroesophageal reflux disease (GERD). Treatment may include medications to reduce gastric acidity such as antacids, histamine-blocking agents, or proton pump inhibitors, and medication to increase gastric motility. In severe cases, surgical treatment may be considered (Richards & Goldin, 2018).
Digestion
The infant’s ability to digest carbohydrates, fats, and proteins is regulated by the presence of certain enzymes. Most of these enzymes are functional at birth except for pancreatic amylase and lipase. Amylase is produced by the salivary glands after approximately 3 months of age and by the pancreas at approximately 6 months of age. This enzyme is necessary to convert starch into maltose and occurs in high amounts in colostrum. The other exception is lipase, also secreted by the pancreas; it is necessary for the digestion of fat. The normal newborn is capable of digesting simple carbohydrates and proteins but has a limited ability to digest fats. Mammary lipase in human milk aids in digestion of fats by the neonate.
Lactase levels in newborns are higher than in older infants. This enzyme is necessary for digestion of lactose, the major carbohydrate in human milk and commercial infant formula.
Stools
Meconium fills the lower intestine at birth. It is formed during fetal life from the amniotic fluid and its constituents, intestinal secretions (including bilirubin), and cells (shed from the mucosa). Meconium is greenish black and viscous and contains occult blood. Most healthy term infants pass meconium within the first 12 to 24 hours of life, and almost all do so by 48 hours (Fig. 22.5). The number of stools passed varies during the first week, being most numerous between the third and sixth days. Newborns fed early pass stools sooner. The colostrum consumed by breastfed neonates during the first 2 to 3 days after birth promotes stooling. Progressive changes in the stooling pattern indicate a properly functioning GI tract (Box 22.1).

Close-up of perineal area of a newborn with her both legs held up together by a hand shows dark green stool with sticky opalescent fluid coming out of her vagina.
Source: (Courtesy Kathryn Alden, Stanley, NC.)Feeding behaviors
Variations occur among infants regarding interest in food, signs of hunger, and amount ingested at one time. The amount the infant consumes at any feeding depends on gestational and chronologic age, weight, hunger level, and alertness. When put to breast, some infants feed immediately, whereas others require a longer learning period. Random hand-to-mouth movement and sucking of fingers are well developed at birth and intensify when the infant is hungry. Caregivers should be alert and responsive to these hunger cues.
Signs of gastrointestinal problems
The time, color, and character of the infant’s first stool should be noted. Failure to pass meconium can indicate bowel obstruction related to conditions such as an inborn error of metabolism (e.g., cystic fibrosis) or a congenital disorder (e.g., Hirschsprung disease or an imperforate anus). An active rectal “wink” reflex (contraction of the anal sphincter muscle in response to touch) is a sign of good sphincter tone. Passage of meconium from the vagina or urinary meatus is a sign of a possible fistulous tract from the rectum.
Fullness of the abdomen above the umbilicus can be caused by hepatomegaly, duodenal atresia, or distention. Abdominal distention at birth usually indicates a serious disorder such as a ruptured viscus (from abdominal wall defects) or tumors. Distention that occurs later can be the result of overfeeding or can be a sign of a GI disorder. A scaphoid (sunken) abdomen, with bowel sounds heard in the chest and signs of respiratory distress, indicates a diaphragmatic hernia. Fullness below the umbilicus can indicate a distended bladder.
Some infants are intolerant of certain commercial infant formulas. If an infant is allergic or unable to digest a formula, the stools can become very soft with a high water content that is signaled by a distinct water ring around the stool on the diaper. Forceful ejection of stool and a water ring around the stool are signs of diarrhea. Care must be taken to avoid misinterpreting transitional stools for diarrhea. The loss of fluid in diarrhea can rapidly lead to fluid and electrolyte imbalance.
The amount and frequency of regurgitation, “spitting,” or vomiting after feedings should be documented. Color change, gagging, and projectile (very forceful) vomiting occur in association with esophageal and tracheoesophageal anomalies. Vomiting in large amounts, especially if it is projectile, can be a sign of pyloric stenosis. Bilious (green) emesis is suggestive of intestinal obstruction or malrotation of the bowel.
Hepatic system
In the newborn, the liver can be palpated approximately 1 to 2 cm below the right costal margin. The infant’s liver plays an important role in iron storage, glucose and fatty acid metabolism, bilirubin synthesis, and coagulation. Although the liver is relatively immature at birth, healthy term infants do not typically experience problems.
Iron storage
The fetal liver, which serves as the site for production of hemoglobin after birth, begins storing iron in utero. The infant’s iron store is proportional to total body hemoglobin content and length of gestation. At birth, the term infant has an iron store sufficient to last approximately 4 months. Iron stores of preterm and small-for-gestational-age infants are often lower and are depleted sooner than in healthy term infants. Although both breast milk and cow’s milk contain iron, the bioavailability of iron in breast milk (lactoferrin) is far superior.
Glucose homeostasis
The liver is responsible for regulation of blood glucose levels. In utero, the glucose concentration in the umbilical vein is approximately 80% of the maternal level. At birth, the newborn is removed from the maternal glucose supply, resulting in an initial drop in blood glucose from fetal levels of 70 to 90 mg/dL to levels of 55 to 60 mg/dL between 30 and 90 minutes after birth. During this time, glucagon levels increase while insulin levels decrease and the limited hepatic glycogen stores are mobilized. The initiation of feedings helps to stabilize blood glucose levels as milk lactose is metabolized. Glucose production also occurs through glycogenolysis and gluconeogenesis. Glucose levels rise gradually and stabilize by the second or third day at levels greater than 60 mg/dL (Garg & Devaskar, 2020).
Glucose levels are not routinely assessed in newborns unless there are risk factors or symptoms of hypoglycemia. Risk factors include small or large for gestational age, preterm, and infant of a diabetic mother. The hypoglycemic infant can be asymptomatic or can display the classic symptoms of jitteriness, lethargy, apnea, feeding problems, or seizures. Hypoglycemia in the initial newborn period is most often transient and easily corrected through feeding. Persistent or recurrent hypoglycemia necessitates intravenous glucose therapy and possible pharmacologic intervention.
Fatty acid metabolism
Fatty acid metabolism is an additional source of energy for the neonate in the initial hours after birth. Catecholamine release increases the rate of lipolysis, which produces fatty acids for oxidation and ketone body synthesis. Hepatic ketogenesis is increased in term newborns for the first 3 days.
Bilirubin synthesis
The liver is responsible for the conjugation of bilirubin, which results from the breakdown of RBCs. When RBCs reach the end of their life span, their membranes rupture, and hemoglobin is released. The hemoglobin is phagocytosed by macrophages; it then splits into heme and globin. The heme is broken down by the reticuloendothelial cells, converted to bilirubin, and released in an unconjugated form. The unconjugated (indirect) bilirubin is relatively insoluble and almost entirely bound to circulating albumin, a plasma protein. Bilirubin that is not bound to albumin, or free bilirubin, can easily cross the blood-brain barrier and cause neurotoxicity (acute bilirubin encephalopathy or kernicterus [see later discussion]).
The unconjugated bilirubin must be conjugated so it becomes soluble and excretable. In the liver, the unbound bilirubin is conjugated with glucuronic acid in the presence of the enzyme glucuronyl transferase. The conjugated form of bilirubin (direct bilirubin) is soluble and excreted from liver cells as a constituent of bile. Along with other components of bile, direct bilirubin is excreted into the biliary tract system that carries the bile into the duodenum. Bilirubin is converted to urobilinogen and stercobilinogen within the duodenum through the action of the bacterial flora. Urobilinogen is excreted in urine and feces; stercobilinogen is excreted in the feces (Fig. 22.6). The effectiveness of bilirubin excretion through the feces depends on the stooling pattern of the newborn and the substances in the intestine that break down conjugated bilirubin. In the newborn intestine, the enzyme β-glucuronidase is able to convert conjugated bilirubin into the unconjugated form, which is subsequently reabsorbed by the intestinal mucosa and transported to the liver; this is called enterohepatic circulation. Feeding is important in reducing serum bilirubin levels because it stimulates peristalsis and produces more rapid passage of meconium, thus diminishing the amount of reabsorption of unconjugated bilirubin. Feeding also introduces bacteria to aid in the reduction of bilirubin to urobilinogen. Colostrum, a natural laxative, facilitates the passage of meconium in breastfed infants.

A flowchart shows steps involved in formation and removal of bilirubin as follows:
• Red blood cell contains hemoglobin.
• Hemoglobin contains heme and globin.
• Heme breaks down into iron and bilirubin with plasma protein.
• Unconjugated bilirubin and glucuronic acid are converted to conjugated bilirubin glucuronide by the action of liver glucuronyl transferase.
• Which is excreted through feces or urine.
When levels of unconjugated bilirubin exceed the ability of the liver to conjugate it, plasma levels of bilirubin increase and jaundice appears. Jaundice, the visible yellowish color of the skin and sclera, is likely to appear when the total serum bilirubin (TSB) level exceeds 6 to 7 mg/dL. Jaundice is generally noticeable first in the head, especially in the sclera and mucous membranes, and progresses gradually to the thorax, abdomen, and extremities. The degree of jaundice is determined by serum total bilirubin measurements (Kamath-Rayne, Froese, & Thilo, 2021).
The newborn is at risk for hyperbilirubinemia because of distinctive aspects of normal neonatal physiology. The higher RBC mass at birth and shorter life span of neonatal RBCs create the need for greater bilirubin synthesis. The ability of the liver to conjugate bilirubin is reduced during the first few days after birth; it can metabolize and excrete only approximately two-thirds of the circulating bilirubin. In addition, there are fewer bilirubin binding sites because newborns have lower serum albumin levels. In the intestines, conjugated bilirubin becomes unconjugated and recirculated through the enterohepatic circulation, which increases serum bilirubin levels.
Traditionally, newborn jaundice has been categorized as either physiologic or pathologic (nonphysiologic), depending primarily on the time it appears and on serum bilirubin levels. Controversy surrounds the definitions of normal or physiologic ranges of TSB. TSB levels in newborns are affected by variables such as gestational age, chronologic age, weight, race, nutritional status, mode of feeding, and presence of extravasated blood (e.g., cephalhematoma or severe bruising) (Blackburn, 2018). The time of onset of jaundice is a key factor in evaluating its cause and determining if treatment is needed.
Among the factors that increase the risk for hyperbilirubinemia, preterm birth is the most significant. Prematurity affects liver and brain metabolism and albumin binding sites, placing preterm and late preterm infants at greater risk for hyperbilirubinemia. Infants of Asian, Native-American, and Inuit ethnicity have higher bilirubin levels. Breastfeeding infants are at greater risk for hyperbilirubinemia (see later discussion) (Watchko, 2018). Risk factors for severe hyperbilirubinemia are listed in Box 22.2.
Physiologic jaundice
Physiologic or nonpathologic jaundice (unconjugated hyperbilirubinemia) occurs in approximately 60% of term newborns. It appears after 24 hours of age and usually resolves without treatment.
In normal full-term newborns, TSB levels progressively increase from 2 mg/dL in cord blood to an average peak of 5 to 6 mg/dL by 72 to 96 hours of life. From that point, TSB levels gradually decrease to a plateau of approximately 3 mg/dL by 1 week of age, reaching normal adult levels of 2 mg/dL or less by 2 weeks of age. This pattern varies according to racial group, method of feeding (breast milk vs. formula), and gestational age (Kamath-Rayne et al., 2021).
Pathologic jaundice
Bilirubin can accumulate to hazardous levels and lead to a pathologic condition. Pathologic or nonphysiologic jaundice is unconjugated hyperbilirubinemia that is either pathologic in origin or severe enough to warrant further evaluation and treatment. Jaundice is usually considered pathologic or nonphysiologic if it appears within 24 hours after birth, TSB levels increase by more than 0.2 mg/dL/h, TSB is greater than the 95th percentile for age in hours, direct serum bilirubin levels exceed 1.5 to 2 mg/dL, or clinical jaundice lasts for more than 2 weeks (Kamath-Rayne et al., 2021). High levels of unconjugated bilirubin are usually caused by excessive production of bilirubin through hemolysis; the most frequent cause is hemolytic disease of the newborn due to maternal/newborn blood group incompatibility (Rh, ABO, or minor blood groups). Other factors contributing to increased hemolysis include enclosed hemorrhage (e.g., cephalhematoma, excessive bruising), polycythemia, delayed passage of meconium, and delayed feeding. It can also be caused by glucose-6-phosphate dehydrogenase (G6PD) deficiency, a genetic disorder that is most prevalent among infants with genetic heritage from Asia, Africa, the Middle East, and the Mediterranean region (Watchko, 2018). Unconjugated hyperbilirubinemia can be the result of altered hepatic clearance of bilirubin related to immaturity, metabolic disorders such as Crigler-Najjar disease, asphyxia, sepsis, and congenital anomalies such as biliary atresia (Blackburn, 2018; Watchko, 2018).
If increased levels of unconjugated bilirubin are left untreated, neurotoxicity can result as bilirubin is transferred into the brain cells. Acute bilirubin encephalopathy refers to the acute manifestations of bilirubin toxicity that occur during the first weeks after birth. This can include a range of symptoms such as lethargy, hypotonia, irritability, seizures, coma, and death. Kernicterus refers to the irreversible, long-term consequences of bilirubin toxicity such as hypotonia, delayed motor skills, hearing loss, cerebral palsy, and gaze abnormalities (Blackburn, 2018) (see Chapter 25).
Jaundice related to breastfeeding
Two forms of breastfeeding-related jaundice are recognized: breastfeeding-associated jaundice and breast milk jaundice. These typically occur in otherwise healthy infants. Both types can occur in the same infant and are not easily differentiated (Kamath-Rayne et al., 2021).
Breastfeeding-associated jaundice (also known as early-onset jaundice, breastfeeding jaundice, suboptimal intake jaundice, or starvation jaundice) occurs during the first week of life. Breastfeeding does not cause the jaundice; rather it is a lack of effective breastfeeding that contributes to the hyperbilirubinemia. If the infant is not feeding effectively, there is less caloric and fluid intake, ongoing weight loss, and possible dehydration. Hepatic clearance of bilirubin is reduced. With less intake, there are fewer stools. As a result, bilirubin is reabsorbed from the intestine back into the bloodstream and must be conjugated again so it can be excreted (Blackburn, 2018; Noble & Rosen-Carole, 2022).
Breast milk jaundice (late-onset jaundice) usually occurs at 5 to 10 days of age. Infants are typically feeding well and gaining weight appropriately. Rising levels of unconjugated bilirubin peak during the second week and gradually diminish. Despite high levels of bilirubin that can persist for 3 to 12 weeks, these infants have no signs of hemolysis or liver dysfunction. The etiology of breast milk jaundice is uncertain. However, it seems to be related to factors in the breast milk (e.g., pregnanediol, fatty acids, and β-glucuronidase) that either inhibit the conjugation of bilirubin or decrease the excretion of bilirubin (Blackburn, 2018; Noble & Rosen-Carole, 2022). (See Chapter 24 for a discussion of these conditions in relation to newborn nutrition.)
Coagulation
The liver plays an important role in blood coagulation. Coagulation factors, which are synthesized in the liver, are activated by vitamin K. The lack of intestinal bacteria needed to synthesize vitamin K results in transient blood coagulation deficiency between the second and fifth days of life. The levels of coagulation factors slowly increase to reach adult levels by 9 months of age. The administration of intramuscular vitamin K shortly after birth helps to prevent vitamin K deficiency bleeding (VKDB), which can occur suddenly and can be catastrophic (Letterio et al., 2020). Any bleeding problems noted in the newborn should be reported immediately and tests for clotting ordered.
Drug metabolism
The immaturity of the liver and depressed liver enzyme systems at birth result in slower biotransformation and elimination of drugs. This can result in slower drug clearance, increased serum levels, and longer half-lives (Blackburn, 2018).
Signs of hepatic system problems
Hypoglycemia and hyperbilirubinemia are the most common liver-related problems experienced by newborns. In most cases the problems are transient and require little if any treatment. Preterm infants are at increased risk for hepatic system problems because of the immaturity of the liver.
The hematologic status of all newborns should be assessed for anemia. For the first week of life, neonates are at risk for bleeding until the coagulation factors are well established. Male newborns who are circumcised prior to discharge from the birth facility must be monitored carefully for bleeding.
Immune system
Beginning early in gestation, the immune system of the fetus is developing the capacity to respond to foreign antigens. The development of the immune system is necessary to equip the neonate to meet the numerous environmental challenges (e.g., microorganisms) associated with life in the extrauterine world. Compared with adults, the immune response at birth is reduced, leading to increased susceptibility to pathogens.
Neonatal levels of circulating immunoglobins are low in comparison with adult levels. Most of the circulating antibodies in the newborn are immunoglobulin G (IgG) antibodies that were transported across the placenta from the maternal circulation. This transfer of antibodies from the mother begins as early as 14 weeks of gestation and is greatest during the third trimester. By term, the IgG levels in the cord blood of the infant are higher than those in maternal blood. The passive immunity afforded the infant through the placental transfer of IgG usually provides sufficient antimicrobial protection during the first 3 months of life. Production of adult concentrations of IgG is reached by 4 to 6 years of age (Benjamin & Maheshwari 2020).
The fetus is capable of producing IgM by the eighth week of gestation, and low levels (less than 10% of adult levels) are present at term. IgM is important for immunity to blood-borne infections and is the major immunoglobulin synthesized during the first month. By 2 years of age, IgM reaches adult levels. The production of IgA, IgD, and IgE is much more gradual, and maximal levels are not attained until early childhood (Benjamin & Maheshwari 2020).
The membrane-protective IgA is missing from the respiratory and urinary tracts, and, unless the newborn is breastfed, it also is absent from the GI tract. The secretory IgA in human milk acts locally in the intestines to neutralize bacterial and viral pathogens. It can also lessen the risk for allergy and food intolerance through modulation of exposure to foreign milk protein antigens.
Other components of breast milk strengthen the neonate’s immune system. Antimicrobial factors such as oligosaccharides, lysozyme, and lactoferrin aid in microbial clearance. Infants who are breastfed have enhanced antibody responses to vaccines. Long-term effects of breastmilk on the immune system are demonstrated by lower risk for immune-mediated conditions such as allergies, inflammatory bowel disease, and type I diabetes mellitus.
The WBCs of the newborn display a delayed response to invading bacteria. Neutrophil levels are low, and therefore their key functions of phagocytosis, chemotaxis, and intracellular killing are limited. The influx of phagocytic cells to areas of inflammation is somewhat slowed, although the ability of these cells to attack and destroy bacteria is equivalent to that of adults. B cells and T cells are present in the newborn, although their function is immature (Benjamin & Maheshwari, 2020).
Risk for infection
All newborns, and preterm newborns especially, are at high risk for infection during the first several months of life. During this period, infection is one of the leading causes of morbidity and mortality. The newborn cannot limit the invading pathogen to the portal of entry because of the generalized hypofunctioning of the inflammatory and immune mechanisms.
Early signs of infection must be recognized so prompt diagnosis and treatment can occur. Temperature instability or hypothermia can be symptomatic of serious infection; newborns do not typically exhibit fever, although hyperthermia can occur (temperature greater than 38°C [100.4°F]). Lethargy, irritability, poor feeding, vomiting or diarrhea, decreased reflexes, and pale or mottled skin color are some of the clinical signs that suggest infection. Respiratory symptoms such as apnea, tachypnea, grunting, or retracting can be associated with infection such as pneumonia. Any unusual discharge from the infant’s eyes, nose, mouth, or other orifice must be investigated. If a rash appears, it must be evaluated closely; many normal rashes in the newborn are not associated with any infection. Infants must be protected from infections by the use of proper hand hygiene (see Chapter 35).
The greatest risk factor for neonatal infection is prematurity, because of immaturity of the immune system. Other risk factors include prelabor rupture of membranes, chorioamnionitis, maternal fever, antenatal or intrapartal asphyxia, invasive procedures, stress, and congenital anomalies.
Integumentary system
All skin structures are present at birth. The epidermis and dermis are loosely bound and extremely thin. After 35 weeks of gestation, the skin is covered by vernix caseosa (a cheeselike, whitish substance) that is fused with the epidermis and serves as a protective covering. Vernix caseosa is a complex substance that contains sebaceous gland secretions. It has emollient and antimicrobial properties and prevents fluid loss through the skin; it also has antioxidant properties. Removal of the vernix is followed by desquamation of the epidermis in most infants. There is evidence that leaving residual vernix intact after birth has positive benefits for neonatal skin such as decreasing the skin pH, decreasing skin erythema, and improving skin hydration (Association of Women’s Health, Obstetric and Neonatal Nurses [AWHONN], 2018; Narendran, 2020).
The skin of a term infant is erythematous (red) for a few hours after birth, and then it fades to its normal color. The skin often appears blotchy or mottled, especially over the extremities. The hands and feet appear slightly cyanotic (acrocyanosis); this is caused by vasomotor instability and capillary stasis. Acrocyanosis is common during the first 48 hours and appears intermittently over the first 7 to 10 days, especially with exposure to cold (see Fig. 22.1).
The healthy term infant usually has a plump appearance because of large amounts of subcutaneous tissue and extracellular water content. Subcutaneous fat accumulated during the last trimester acts as insulation. Fine lanugo hair may be noted over the face, shoulders, and back. Edema of the face and ecchymosis (bruising) or petechiae may be noted as a result of face presentation, forceps-assisted birth, or vacuum extraction.
Creases are located on the palms of the hands and the soles of the feet. The simian line, a single palmar crease, is often seen in Asian infants and infants with Down syndrome. The soles of the feet should be inspected for the number of creases during the first few hours after birth; as the skin dries, more creases appear. More creases correlate with a greater maturity rating. Preterm newborns have few, if any, creases.
Sweat glands
Distended, small, white sebaceous glands noticeable on the newborn face are known as milia (Fig. 22.7). Although sweat glands are present at birth, term infants usually do not sweat for the first 24 hours. By day 3, sweating begins on the face, then progresses to the palms. Infants can sweat as a function of body or environmental temperature; there can also be emotional sweating from crying or pain (Narendran, 2020).

Close-up of face of a newborn shows small, white, circular bumps present closely on the chin and rarely on the rest of the face.
Source: (Courtesy Ashley Martin, Denver, NC.)Desquamation
Desquamation (peeling) of the skin of the term infant does not occur until a few days after birth. Large, generalized areas of skin desquamation present at birth can be an indication of postmaturity.
Congenital dermal melanocytosis
Congenital dermal melanocytosis or slate gray nevi (formerly known as Mongolian spots) bluish black areas of pigmentation, can appear over any part of the exterior surface of the body, including the extremities. They are most common on the back and buttocks (Fig. 22.8). These pigmented areas occur most frequently in newborns whose ethnic origins are Latin America, Asia, Africa, or the Mediterranean area, but can occur in White infants. (Blackburn, 2018). Slate gray nevi fade gradually over months or years.
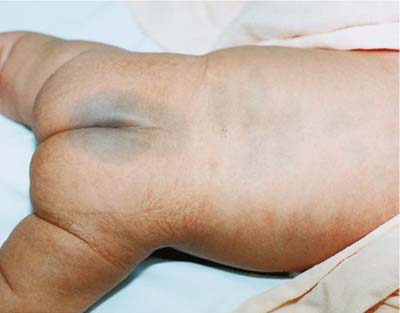
A close-up shows the posterior view of an infant’s lower body with bluish skin on the lower back and around the anus.
The presence of slate gray nevi on the newborn should be documented carefully in the medical record. These normal skin pigmentations can be mistaken for bruises once the infant is discharged, and this can raise suspicion of physical abuse.
Nevi
Nevus simplex, also known as salmon patches, telangiectatic nevi, “stork bites,” or “angel kisses,” is the result of a superficial capillary defect and occurs in up to 80% of newborns. They are usually small, flat, and pink and are easily blanched (Fig. 22.9). The most common sites are the upper eyelids, nose, upper lip, and nape of the neck. They have no clinical significance and require no treatment. Facial lesions usually fade between the first and second years of life, whereas neck lesions can be visible into adulthood (Narendran, 2020).
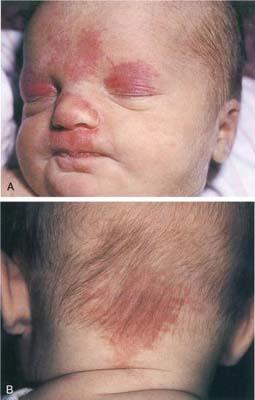
A) Close-up of face of a newborn shows flat, pink patches at the center of the forehead, both upper lids, from nose bridge descending to side of the nose, and between the nose and the upper lip.
B) Close-up of an infant’s posterior head shows a flat, pink patches on the lower back of the head.
Source: (From Eichenfield, L. F., Frieden, I. J., & Esterly, N. B. [Eds.]. [2008]. Neonatal dermatology [2nd ed.]. Philadelphia: Saunders.)A port-wine stain, or nevus flammeus, is usually visible at birth and is due to an asymmetric postcapillary venule malformation. It is usually pink and flat at birth but darkens with time, becoming red or purple and pebbly in consistency. True port-wine stains do not blanch on pressure or disappear. They are found most commonly on the face and neck (Narendran, 2020).
Infantile hemangioma
Infantile hemangiomas are the most common type of soft tissue tumors occurring in infants. They consist of dilated newly formed capillaries occupying the entire dermal and subdermal layers with associated connective tissue hypertrophy. The typical lesion is a superficial, raised, sharply demarcated, bright or dark red rough-surfaced swelling that may be present at birth or may appear during the early weeks after birth. Common sites are the scalp, face, back, and anterior chest. Some hemangiomas lie deeper in the skin as soft masses with bluish skin discoloration. Most superficial lesions reach maximum growth in approximately 6 months and then begin a slow process of involution. Deeper hemangiomas often continue to grow for a year or more (Narendran, 2020).
Erythema toxicum
Erythema toxicum, a transient rash, is also called erythema neonatorum, newborn rash, or flea bite dermatitis. It first appears in term neonates during the first 24 to 72 hours after birth and can last up to 3 weeks of age. It has lesions in different stages: erythematous macules, papules, and small vesicles (Fig. 22.10). The lesions can appear suddenly anywhere on the body. The rash is thought to be an inflammatory response. Eosinophils, which help to decrease inflammation, are found in the vesicles. Although the appearance is alarming, the rash has no clinical significance and requires no treatment (Narendran, 2020).

Close-up of an infant’s back show small red to pink rashes with pus heads at some places scattered on the skin.
Source: (From Long, K. A., & Martin, K. L. [2020]. In R. M. Kliegman, J. W. St. Geme, III, N. J. Blum, et al. [Eds.]. Nelson textbook of pediatrics [21st ed.]. Philadelphia: Elsevier.)Signs of integumentary problems
Close observation of the newborn’s skin color can lead to early detection of potential problems. Any pallor, plethora (deep purplish color from increased circulating RBCs), petechiae, central cyanosis, or jaundice is noted and documented. The skin is examined for signs of birth injuries such as forceps marks and lesions related to fetal monitoring. Bruises or petechiae can be present on the head, neck, and face of an infant born with a nuchal cord (cord around the neck) or who had a face presentation at birth. Bruising can increase the risk for hyperbilirubinemia. Petechiae can be present if increased pressure was applied to an area. Petechiae scattered over the infant’s body should be reported to the health care provider because petechiae can indicate underlying problems such as low platelet count or infection.
Unilateral or bilateral periauricular papillomas (skin tags) occur fairly frequently. Their occurrence is usually a family trait and of no consequence.
Reproductive system
Female
An increase in estrogen during pregnancy followed by a drop after birth causes female newborns to have mucoid vaginal discharge (Fig. 22.5) and even some slight bloody spotting. In term neonates, the labia majora and minora cover the vestibule (Fig. 22.11A). In preterm infants, the clitoris is prominent and the labia majora are small and widely separated. Vaginal or hymenal tags are common findings and have no clinical significance. Vernix caseosa can be present between the labia and should not be forcibly removed during bathing.

Two-part image show female and male external genitalia in A and B.
Source: (A, Courtesy Kathryn Alden, Stanley, NC; B, Courtesy Marjorie Pyle, RNC, Lifecircle, Costa Mesa, CA.)If the girl was born from a breech position, the labia can be edematous and bruised. The edema and bruising resolve in a few days; no treatment is necessary.
Male
In the uncircumcised newborn, the foreskin or prepuce completely covers the glans. The foreskin adheres to the glans and is not fully retractable for 3 to 4 years. The position of the urethra should be at the tip of the penis. With hypospadias, the urethral opening is located in an abnormal position, at any point on the ventral surface of the penis surface from the glans to the perineum. If the urethral opening is located on the dorsal surface of the penis, it is known as epispadias; this is less common and is often associated with exstrophy of the bladder (Elder, 2020). A common finding in newborn males are small, white, firm lesions called epithelial pearls at the tip of the prepuce.
By 28 to 36 weeks of gestation, the testes can be palpated in the inguinal canal and a few rugae appear on the scrotum. At 36 to 40 weeks of gestation, the testes are palpable in the upper scrotum and rugae appear on the anterior portion. After 40 weeks, the testes can be palpated in the scrotum and rugae cover the scrotal sac. The postterm neonate has deep rugae and a pendulous scrotum. Undescended testes (cryptorchidism) occur in approximately 4% of term newborn males and in 21% of preterm males; in most cases the testes gradually descend without intervention. Risk factors for cryptorchidism include preterm birth, low birth weight, maternal obesity, and cesarean birth (Sorbara & Wherrett, 2020).
The scrotum is usually more deeply pigmented than the rest of the skin (see Fig. 22.11B), particularly in darker-skinned infants. A bluish discoloration of the scrotum suggests testicular torsion, which needs immediate attention. If the male infant is born in a breech presentation, the scrotum can be very edematous and bruised (Fig. 22.12). The swelling and discoloration subside within a few days.

A newborn lying sideways while crying and shows dark discolouration on the pelvis and scrotum caused by bruising.
Source: (From O’Doherty, N. (1986). Neonatology: Micro atlas of the newborn. Nutley, NJ: Hoffman-LaRoche.)Hydrocele, caused by an accumulation of fluid around the testes, can be present. Hydroceles can be easily transilluminated with a light and usually resolve without treatment (Fig. 22.13).

Close-up of external genitalia of a newborn male show enlarged scrotum and the skin of the newborn shows yellow undertone.
Source: (From Poenaru, D. [2012]. Abdominal wall problems. In C. A. Gleason, & S. U. Devaskar [Eds.], Avery’s diseases of the newborn [9th ed.]. Philadelphia: Saunders.)Swelling of breast tissue
Swelling of the breast tissue in term infants of both sexes is caused by the hyperestrogenism of pregnancy. In a few infants a thin discharge (“witch’s milk”) can be seen. This finding has no clinical significance, requires no treatment, and subsides within a few days as the maternal hormones are eliminated from the infant’s body.
The nipples should be symmetric on the chest. Breast tissue and areola size increase with gestation. The areola appears slightly elevated at 34 weeks of gestation. By 36 weeks, a breast bud of 1 to 2 mm is palpable; this increases to 12 mm by 42 weeks.
Signs of reproductive system problems
The infant is inspected closely for ambiguous genitalia and other abnormalities. Normally in a female infant the urethral opening is located behind the clitoris. Any deviation from this can incorrectly suggest that the clitoris is a small penis, which can occur in conditions such as adrenal hyperplasia. Nearly all female infants are born with hymenal tags; absence of such tags can indicate vaginal agenesis. Fecal discharge from the vagina indicates a rectovaginal fistula. Any of these findings must be reported to the neonatal or pediatric health care provider for further evaluation.
Hypospadias, undescended testes, or other abnormalities of the male genitalia must be reported. Circumcision is contraindicated in the presence of hypospadias because the foreskin is used in repair of this anomaly.
Inguinal hernias can be present and become more obvious when the infant cries. They are common, especially in Black neonates, and usually require no treatment because they resolve with time.
Skeletal system
The infant’s skeletal system undergoes rapid development during the first year of life. At birth, more cartilage is present than ossified bone.
Head and skull
Because of cephalocaudal (head-to-rump) development, the newborn looks somewhat out of proportion. The head at term is approximately one-fourth of the total body length. The arms are slightly longer than the legs. In the newborn, the legs are approximately one-third of the total body length. As growth proceeds, the midpoint in head-to-toe measurements gradually descends from the level of the umbilicus at birth to the level of the symphysis pubis at maturity.
The face appears small in relation to the skull. The skull appears large and heavy. Cranial size and shape can be distorted by molding (the shaping of the fetal head by overlapping of the cranial bones to facilitate movement through the birth canal during labor) (Fig. 22.14).
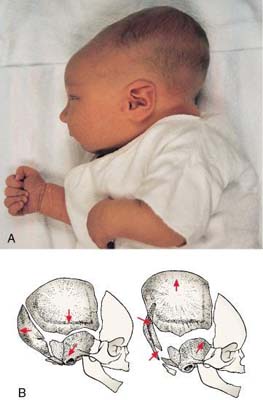
A two-part image. A: A close-up showing the lateral view of an infant lying on bed. The skull appears extended backward and very large as compared to the face. B: Diagram of the lateral view of an infant skull with cranial bones close to each other. An arrow in the occipital bone points outward; an arrow in the parietal bone points downward; and an arrow in the temporal bone also points outward. C: Diagram of the cranial bones far apart from each other. Two arrows in the occipital bone point inward; an arrow in the parietal bone points outward; and an arrow in the temporal bone also points inward.
Source: (A, Courtesy Kim Molloy, Knoxville, IA.)Caput succedaneum is a generalized, easily identifiable edematous area of the scalp, most often on the occiput (Fig. 22.15A). With vertex presentation, the sustained pressure of the presenting part against the cervix results in compression of local vessels, slowing venous return. The slower venous return causes an increase in tissue fluids within the skin of the scalp, and edema develops. This edematous area, present at birth, extends across suture lines of the skull and usually disappears spontaneously within 3 to 4 days. Infants who are born with the assistance of vacuum extraction usually have a caput in the area where the cup was applied. Bruising of the scalp is often seen in the presence of caput succedaneum.

Illustration A shows edema in between scalp and periosteum surrounding the cranium. Illustration B shows a bulge on the right side of the head with hemorrhage in between cranium and periosteum, surrounded by the scalp. Illustration C also shows a bulge on the right side of the head with subgaleal hemorrhage in between periosteum and galea. The periosteum surrounds bone and dura mater. The galea is surrounded by connective tissue and skin.
An enlarged view of the scalp shows following layers from top to bottom: Skin, connective tissue, aponeurosis (galea), loose connective tissue, and periosteum. The bone, dura mater, and brain are also labelled.
Source: (A and B, From Seidel, H., Ball, J., Dains, J., & Benedict, G. [2006]. Mosby’s guide to physical examination [6th ed.]. St. Louis: Mosby.)Cephalhematoma is a collection of blood between a skull bone and its periosteum; it does not cross a cranial suture line (see Fig. 22.15B). A cephalhematoma is firmer and better defined than a caput. Often caput succedaneum and cephalhematoma occur simultaneously. A cephalhematoma usually resolves in 2 to 8 weeks. As the hematoma resolves, hemolysis of RBCs occurs, and hyperbilirubinemia can result.
Subgaleal hemorrhage is bleeding into the subgaleal compartment (see Fig. 22.15C). The subgaleal compartment is a potential space that contains loosely arranged connective tissue; it is located beneath the galea aponeurosis, the tendinous sheath that connects the frontal and occipital muscles and forms the inner surface of the scalp. Subgaleal hemorrhage is the result of traction or application of shearing forces to the scalp, commonly associated with difficult operative vaginal birth, especially vacuum extraction. The scalp is pulled away from the bony calvarium; the vessels are torn, and blood collects in the subgaleal space. Blood loss can be severe, resulting in hypovolemic shock, disseminated intravascular coagulation (DIC), and death (Prazad, Rajpal, Mangurten, et al., 2020).
Early detection of the hemorrhage is vital; serial head circumference measurements and inspection of the back of the neck for increasing edema and a firm mass are essential. A boggy scalp, pallor, tachycardia, and increasing head circumference can be early signs of a subgaleal hemorrhage. Computed tomography or magnetic resonance imaging is useful in confirming the diagnosis. Replacement of lost blood and clotting factors is required in acute cases of hemorrhage. Another possible early sign of subgaleal hemorrhage is a forward and lateral positioning of the newborn’s ears because the hematoma extends posteriorly. Monitoring the infant for changes in level of consciousness and decreases in hematocrit is also key to early recognition and management. An increase in serum bilirubin level may be seen as a result of the degradation of blood cells within the hematoma (Prazad et al., 2020).
Spine
The bones in the vertebral column of the newborn form two primary curvatures—one in the thoracic region and one in the sacral region. Both are forward, concave curvatures. As the infant gains head control at approximately 3 months of age, a secondary curvature appears in the cervical region. The newborn’s spine appears straight and can be flexed easily. The newborn can lift the head and turn it from side to side when prone. The vertebrae should appear straight and flat. If a pilonidal dimple is noted, further inspection is required to determine whether a sinus is present. A pilonidal dimple, especially with a sinus and nevus pilosis (hairy nevus), can be associated with spina bifida.
Extremities
The infant’s extremities should be symmetric and of equal length. Fingers and toes should be equal in number (five fingers on each hand and five toes on each foot) and should have nails present. Digits may be missing (oligodactyly). Extra digits (polydactyly) are sometimes found on the hands or feet. Fingers or toes may be fused (syndactyly).
The infant is examined for developmental dysplasia of the hips (DDH). In newborns with DDH, the affected hip is unlikely to be dislocated at birth; instead it is easily dislocatable after birth. Postnatal factors determine whether the hip dislocates, subluxates, or remains stable. DDH occurs more often in breech presentations (Fig. 22.16), first-born infants, female infants, and in infants with a family history of DDH (Son-Hing & Thompson, 2020; White, Bouchard, & Goldberg, 2018).

Close-up of lower body of a newborn shows one leg flexed straight up and other leg bent. The perineal and hip area shows purple discoloration along with dark purple discoloration of vulva and opalescent discharge from the vagina.
Source: (Courtesy Cheryl Briggs, RNC, Annapolis, MD.)Signs of DDH are asymmetric gluteal and thigh skinfolds, uneven knee levels, a positive Ortolani test, and a positive Barlow test. The hips are inspected for symmetry. Gluteal and thigh skinfolds should be equal and symmetric, and legs should be of equal length (Fig. 22.17A). The level of the knees in flexion should be equal (see Fig. 22.17C). Hip integrity is assessed by using the Barlow test and the Ortolani maneuver. For the Barlow test, the examiner places the middle finger over the greater trochanter and the thumb along the midthigh. The hip is flexed to 90 degrees and adducted, followed by gentle downward pushing of the femoral head. If the hip can be dislocated with this maneuver, the femoral head moves out of the acetabulum, and the examiner feels a “clunk.” The hip is then checked to determine if the femoral head can be returned into the acetabulum using the Ortolani maneuver. As the hip is abducted and upward leverage is applied, a dislocated hip returns to the acetabulum with a clunk that is felt by the examiner (see Fig. 22.17B and D).

Illustration A shows anterior and posterior views of an infant with left leg slightly shorten. An arrow in both views point to the skin folds on the inner thigh of the left leg. Illustration B through D shows a pair of hands holding an infant’s legs. Part B shows one hand tilting the bent right leg toward right to touch the floor. Part C shows the hands holding the bent knees together. A dashed line shows the left knee shorter than right knee. Part D shows the hand moving femoral head in and out of acetabulum in both legs.
Source: (From Hockenberry, M. J., Wilson, D., & Rodgers, C. C. [2017]. Wong’s essentials of pediatric nursing [10th ed.]. St. Louis: Elsevier.)Signs of skeletal problems
Abnormalities of the skeletal system can be congenital, developmental, drug induced, or the result of intrapartum or postnatal factors. Signs of DDH, additional digits or webbing of digits, and any other abnormality should be documented and reported to the neonatal or pediatric health care provider.
A fractured clavicle often occurs in large infants and in those who had a difficult birth (e.g., shoulder dystocia). Unequal movement of the upper extremities or crepitus over the clavicular area can indicate fracture.
The newborn’s feet can appear to be abnormally positioned. This can indicate congenital deformity or can be related to fetal positioning in utero. For example, clubfoot (talipes equinovarus), a deformity in which the foot turns inward and is fixed in a plantar-flexion position, is a congenital condition that warrants attention. If the foot is turned inward in the plantar-flexion position but can be moved into the normal position, it is likely caused by fetal positioning and should gradually resolve.
Neuromuscular system
The neuromuscular system is almost completely developed at birth. The term newborn is a responsive and reactive being with remarkable capacity for social interaction and self-organization.
Growth of the brain after birth follows a predictable pattern of rapid growth during infancy and early childhood; it becomes more gradual during the remainder of the first decade and minimal during adolescence. By the end of the first year, the cerebellum ends its growth spurt, which began at approximately 30 weeks of gestation.
The brain requires glucose as a source of energy and a relatively large supply of oxygen for adequate metabolism. The necessity for glucose requires careful assessment of neonates who are at risk for hypoglycemia (e.g., infants of mothers who have diabetes; infants who are macrosomic or small for gestational age; and newborns who experienced prolonged birth, hypoxia, or preterm birth).
Spontaneous motor activity can be seen as transient tremors of the mouth and chin, especially during crying episodes, and of the extremities, notably the arms and hands. Transient tremors are normal and can be observed in nearly every newborn. They most often involve the mouth and chin or the arms and hands. These tremors should not be present when the infant is quiet and should not persist beyond 1 month of age. Persistent tremors or tremors involving the total body can indicate pathologic conditions. Normal tremors, tremors (jitteriness) of hypoglycemia, and seizure activity must be differentiated so corrective care can be instituted as necessary.
To differentiate between tremors or jitteriness and seizure activity, the nurse can consider the following signs (Hall & Reavey, 2021):
- • Tremors or jitteriness are easily elicited by motions or voice and cease with gentle restraint of the body part, whereas seizure activity continues.
- • Passive flexion and repositioning of the tremulous extremity reduces or stops the movement.
- • Seizure activity is associated with ocular changes (eyes deviating or staring) and autonomic changes (apnea, tachycardia, pupil changes, increased salivation); these signs are not associated with jitteriness or tremors.
The posture of the term newborn demonstrates flexion of the arms at the elbows and the legs at the knees. Hips are abducted and partially flexed. Intermittent fisting of the hands is common.
Muscle tone and strength are directly related. The infant with normal tone and strength exhibits some resistance to passive movement such as when being pulled to sit or when the arm or leg is extended by the examiner. The hypotonic neonate shows little resistance and can feel like a “rag doll.” Hypertonia is evidenced by increased resistance to passive movement.
Although neuromuscular control is very limited, it can be noted. If newborns are placed face down on a firm surface, they will turn their heads to the side. They attempt to hold their heads in line with their bodies if they are raised by their arms. Various reflexes serve to promote safety and adequate food intake.
Newborn reflexes
The newborn has many primitive reflexes. The times at which these reflexes appear and disappear reflect the maturity and intactness of the developing nervous system. The most common reflexes found in the normal term newborn are described in Table 22.1.
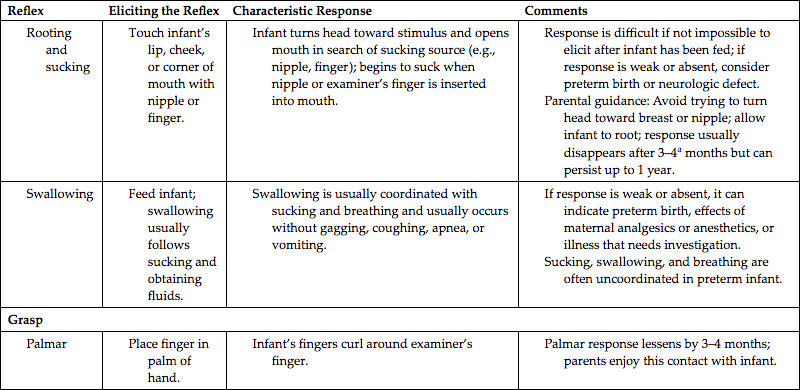
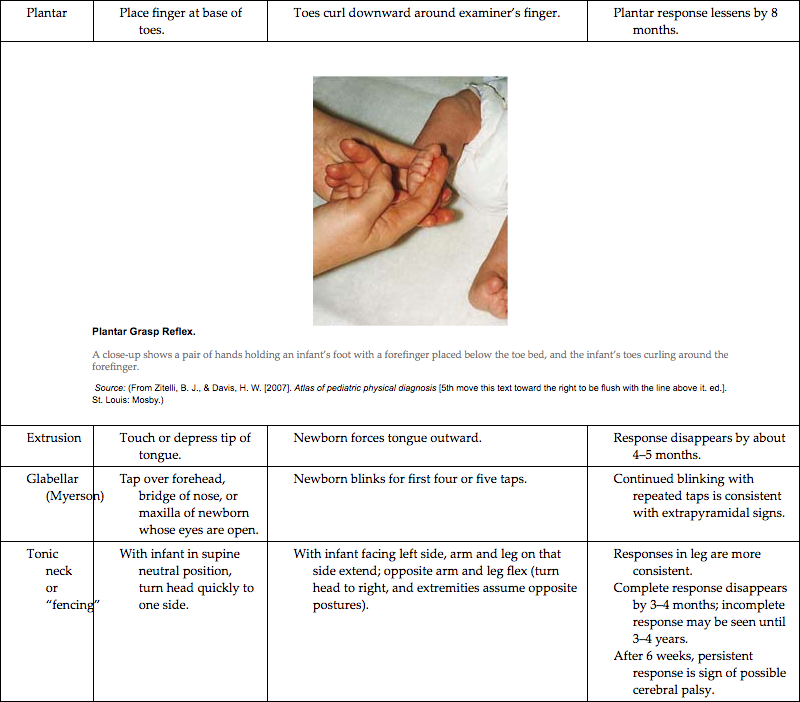
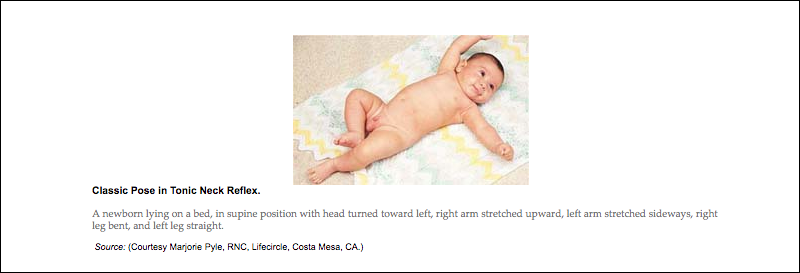
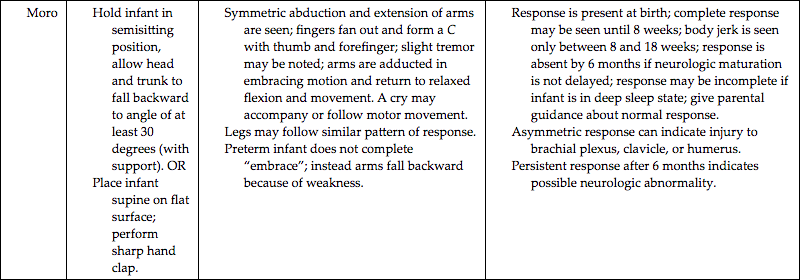
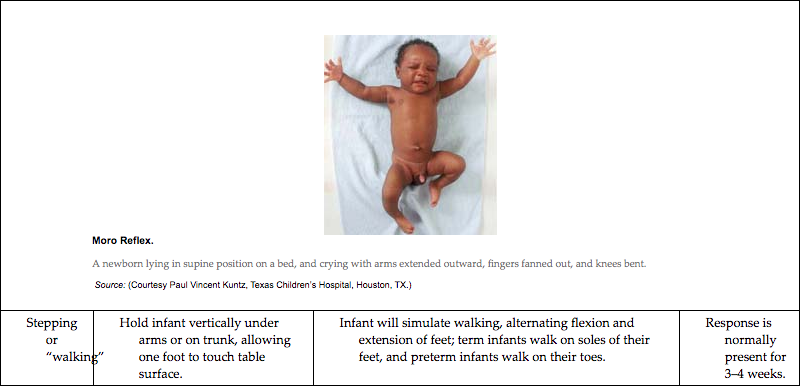
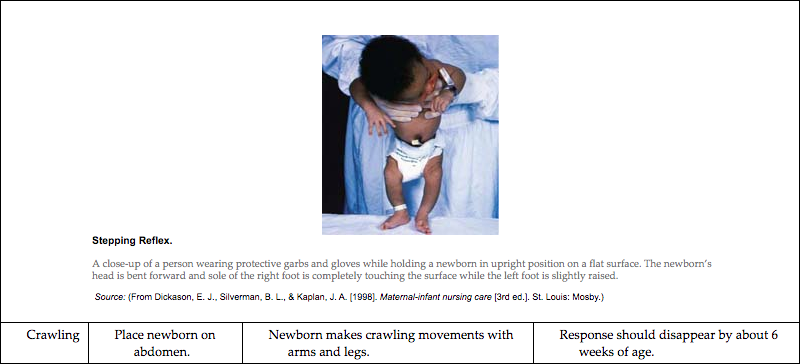
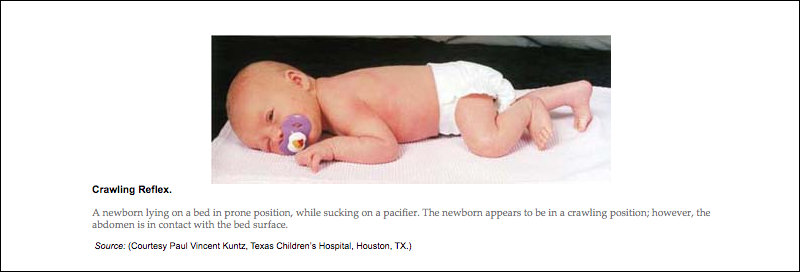
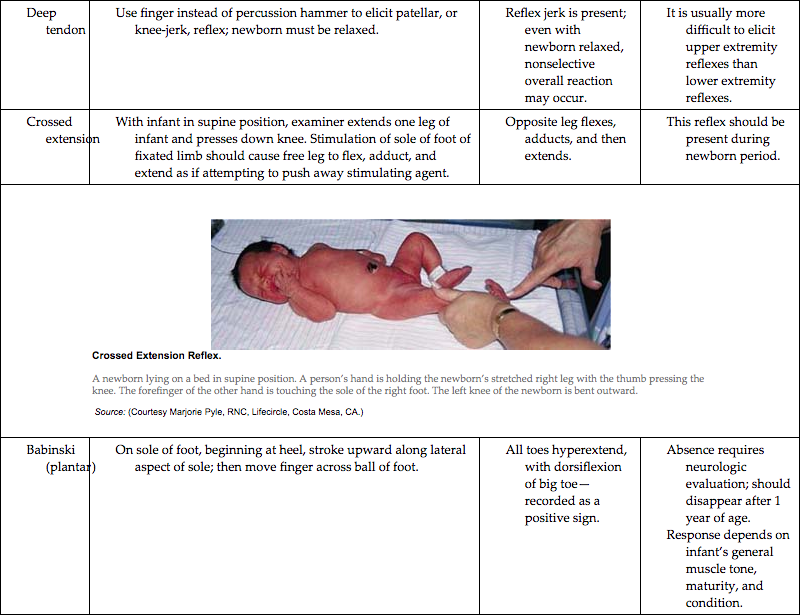
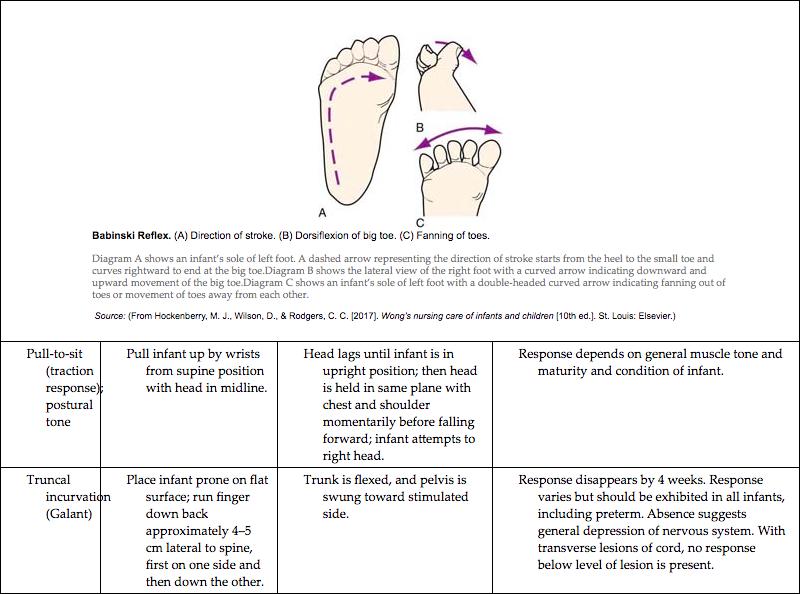
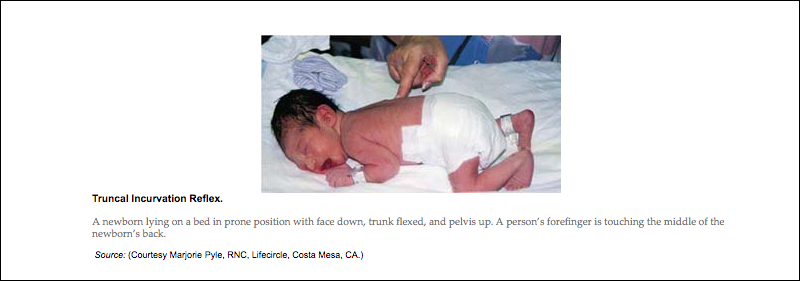
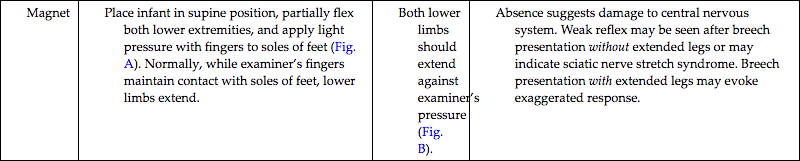
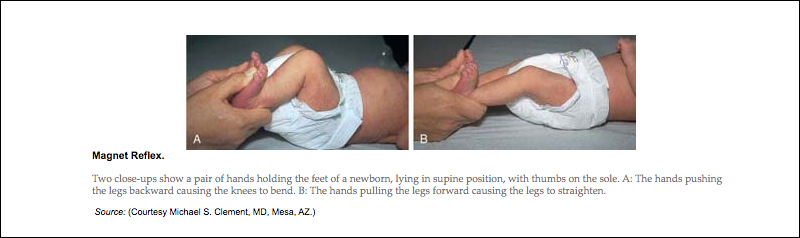

| Reflex | Eliciting the Reflex | Characteristic Response | Comments |
|---|---|---|---|
Rooting and sucking |
Touch infant’s lip, cheek, or corner of mouth with nipple or finger. |
Infant turns head toward stimulus and opens mouth in search of sucking source (e.g., nipple, finger); begins to suck when nipple or examiner’s finger is inserted into mouth. |
Response is difficult if not impossible to elicit after infant has been fed; if response is weak or absent, consider preterm birth or neurologic defect. Parental guidance: Avoid trying to turn head toward breast or nipple; allow infant to root; response usually disappears after 3–4a months but can persist up to 1 year. |
Swallowing |
Feed infant; swallowing usually follows sucking and obtaining fluids. |
Swallowing is usually coordinated with sucking and breathing and usually occurs without gagging, coughing, apnea, or vomiting. |
If response is weak or absent, it can indicate preterm birth, effects of maternal analgesics or anesthetics, or illness that needs investigation. Sucking, swallowing, and breathing are often uncoordinated in preterm infant. |
| Grasp | |||
Palmar |
Place finger in palm of hand. |
Infant’s fingers curl around examiner’s finger. |
Palmar response lessens by 3–4 months; parents enjoy this contact with infant. |
Plantar |
Place finger at base of toes. |
Toes curl downward around examiner’s finger. |
Plantar response lessens by 8 months. |

A close-up shows a pair of hands holding an infant’s foot with a forefinger placed below the toe bed, and the infant’s toes curling around the forefinger. Source: (From Zitelli, B. J., & Davis, H. W. [2007]. Atlas of pediatric physical diagnosis [5th move this text toward the right to be flush with the line above it. ed.]. St. Louis: Mosby.) |
|||
Extrusion |
Touch or depress tip of tongue. |
Newborn forces tongue outward. |
Response disappears by about 4–5 months. |
Glabellar (Myerson) |
Tap over forehead, bridge of nose, or maxilla of newborn whose eyes are open. |
Newborn blinks for first four or five taps. |
Continued blinking with repeated taps is consistent with extrapyramidal signs. |
Tonic neck or “fencing” |
With infant in supine neutral position, turn head quickly to one side. |
With infant facing left side, arm and leg on that side extend; opposite arm and leg flex (turn head to right, and extremities assume opposite postures). |
Responses in leg are more consistent. Complete response disappears by 3–4 months; incomplete response may be seen until 3–4 years. After 6 weeks, persistent response is sign of possible cerebral palsy. |
Moro |
Hold infant in semisitting position, allow head and trunk to fall backward to angle of at least 30 degrees (with support). OR Place infant supine on flat surface; perform sharp hand clap. |
Symmetric abduction and extension of arms are seen; fingers fan out and form a C with thumb and forefinger; slight tremor may be noted; arms are adducted in embracing motion and return to relaxed flexion and movement. A cry may accompany or follow motor movement. Legs may follow similar pattern of response. Preterm infant does not complete “embrace”; instead arms fall backward because of weakness. |
Response is present at birth; complete response may be seen until 8 weeks; body jerk is seen only between 8 and 18 weeks; response is absent by 6 months if neurologic maturation is not delayed; response may be incomplete if infant is in deep sleep state; give parental guidance about normal response. Asymmetric response can indicate injury to brachial plexus, clavicle, or humerus. Persistent response after 6 months indicates possible neurologic abnormality. |
Stepping or “walking” |
Hold infant vertically under arms or on trunk, allowing one foot to touch table surface. |
Infant will simulate walking, alternating flexion and extension of feet; term infants walk on soles of their feet, and preterm infants walk on their toes. |
Response is normally present for 3–4 weeks. |

A close-up of a person wearing protective garbs and gloves while holding a newborn in upright position on a flat surface. The newborn’s head is bent forward and sole of the right foot is completely touching the surface while the left foot is slightly raised. Source: (From Dickason, E. J., Silverman, B. L., & Kaplan, J. A. [1998]. Maternal-infant nursing care [3rd ed.]. St. Louis: Mosby.) |
|||
Crawling |
Place newborn on abdomen. |
Newborn makes crawling movements with arms and legs. |
Response should disappear by about 6 weeks of age. |
Deep tendon |
Use finger instead of percussion hammer to elicit patellar, or knee-jerk, reflex; newborn must be relaxed. |
Reflex jerk is present; even with newborn relaxed, nonselective overall reaction may occur. |
It is usually more difficult to elicit upper extremity reflexes than lower extremity reflexes. |
Crossed extension |
With infant in supine position, examiner extends one leg of infant and presses down knee. Stimulation of sole of foot of fixated limb should cause free leg to flex, adduct, and extend as if attempting to push away stimulating agent. |
Opposite leg flexes, adducts, and then extends. |
This reflex should be present during newborn period. |

A newborn lying on a bed in supine position. A person’s hand is holding the newborn’s stretched right leg with the thumb pressing the knee. The forefinger of the other hand is touching the sole of the right foot. The left knee of the newborn is bent outward. Source: (Courtesy Marjorie Pyle, RNC, Lifecircle, Costa Mesa, CA.) |
|||
Babinski (plantar) |
On sole of foot, beginning at heel, stroke upward along lateral aspect of sole; then move finger across ball of foot. |
All toes hyperextend, with dorsiflexion of big toe—recorded as a positive sign. |
Absence requires neurologic evaluation; should disappear after 1 year of age. Response depends on infant’s general muscle tone, maturity, and condition. |

Diagram A shows an infant’s sole of left foot. A dashed arrow representing the direction of stroke starts from the heel to the small toe and curves rightward to end at the big toe.Diagram B shows the lateral view of the right foot with a curved arrow indicating downward and upward movement of the big toe.Diagram C shows an infant’s sole of left foot with a double-headed curved arrow indicating fanning out of toes or movement of toes away from each other. Source: (From Hockenberry, M. J., Wilson, D., & Rodgers, C. C. [2017]. Wong’s nursing care of infants and children [10th ed.]. St. Louis: Elsevier.) |
|||
Pull-to-sit (traction response); postural tone |
Pull infant up by wrists from supine position with head in midline. |
Head lags until infant is in upright position; then head is held in same plane with chest and shoulder momentarily before falling forward; infant attempts to right head. |
Response depends on general muscle tone and maturity and condition of infant. |
Truncal incurvation (Galant) |
Place infant prone on flat surface; run finger down back approximately 4–5 cm lateral to spine, first on one side and then down the other. |
Trunk is flexed, and pelvis is swung toward stimulated side. |
Response disappears by 4 weeks. Response varies but should be exhibited in all infants, including preterm. Absence suggests general depression of nervous system. With transverse lesions of cord, no response below level of lesion is present. |
Magnet |
Place infant in supine position, partially flex both lower extremities, and apply light pressure with fingers to soles of feet (Fig. A). Normally, while examiner’s fingers maintain contact with soles of feet, lower limbs extend. |
Both lower limbs should extend against examiner’s pressure (Fig. B). |
Absence suggests damage to central nervous system. Weak reflex may be seen after breech presentation without extended legs or may indicate sciatic nerve stretch syndrome. Breech presentation with extended legs may evoke exaggerated response. |

Two close-ups show a pair of hands holding the feet of a newborn, lying in supine position, with thumbs on the sole. A: The hands pushing the legs backward causing the knees to bend. B: The hands pulling the legs forward causing the legs to straighten. Source: (Courtesy Michael S. Clement, MD, Mesa, AZ.) |
|||
Additional newborn responses: yawn, stretch, burp, hiccup, sneeze |
These are spontaneous behaviors. |
Responses can be slightly depressed temporarily because of maternal analgesia or anesthesia, fetal hypoxia, or infection. |
Parental guidance: Most of these behaviors are pleasurable to parents. Parents need to be assured that behaviors are normal. Sneeze is usually a response to mucus in the nose and not an indicator of upper respiratory infection. No treatment is needed for hiccups; sucking may help. In a preterm infant, these are signs of neurodevelopmental immaturity and physiologic stress. |
aAll durations for persistence of reflexes are based on time elapsed after 40 weeks of gestation (i.e., if newborn was born at 36 weeks of gestation, add 1 month to all time limits given).
Behavioral adaptations
The healthy infant must accomplish behavioral and biologic tasks to develop normally. Behavioral characteristics form the basis of the social capabilities of the infant. Newborns progress through a hierarchy of developmental challenges as they adapt to their environment and caregivers. They must first be able to regulate their physiologic or autonomic system, including involuntary physiologic functions such as heart rate, respiration, and temperature. The next level is motor organization, in which infants regulate or control their motor behavior. This includes controlling random movements, improving muscle tone, and reducing excessive activity. The third level of behavior is state regulation, which refers to the ability to modulate the state of consciousness. The infant develops predictable sleep and wake states and is able to react to stress through self-regulation or communicating with the caregiver by crying and then being consoled. Finally, the infant reaches the fourth level of attention and social interaction. He or she is able to attend to visual and auditory stimulation, stay alert for long periods, and engage in social interaction (Brazelton & Nugent, 2011).
This progression in behavior is the basis for the Brazelton Neonatal Behavioral Assessment Scale (NBAS) (Brazelton & Nugent, 2011). The NBAS is an interactive examination that assesses the infant’s response to 28 areas organized according to the clusters in Box 22.3. It is generally used as a research or diagnostic tool and requires special training. The NBAS helps the practitioner to identify where the infant falls along the continuum of behaviors and determine the type of support needed.
Sleep-wake states
Healthy newborns differ in their activity levels, feeding patterns, sleeping patterns, and responsiveness. Parents’ reactions to their newborns are often determined by these differences. Showing parents the unique characteristics of their infant helps them to develop a more positive perception of the infant and promotes increased interaction between infant and parent. Infant responses to environmental stimuli and to their caregivers depend on the infant’s state or state of consciousness.
In the early newborn period, infants tend to alternate periods of sleep and wakefulness that resemble their fetal inactivity and activity patterns. Variations in the state of consciousness of infants are called sleep-wake states. The six states form a continuum from deep sleep to extreme irritability (Fig. 22.18): two sleep states (deep sleep and light sleep) and four wake states (drowsy, quiet alert, active alert, and crying) (Brazelton & Nugent, 2011). Each state has specific characteristics and state-related behaviors. The optimal state of arousal is the quiet alert state. During this state, infants smile, vocalize, move in synchrony with speech, watch their parents’ faces, and respond to people talking to them. They respond to internal and external environmental factors by controlling sensory input and regulating the sleep-wake states; the ability to make smooth transitions between states is called state modulation. The ability to regulate sleep-wake states is essential in the infant’s neurobehavioral development. Term infants are better able than preterm infants to cope with external or internal factors that affect the sleep-wake patterns.
A multi-part image labelled A through F showing close-ups of a newborn, reflecting the different sleep-wake states. A: A newborn sleeping in the crib. B: A newborn with eyes closed and held in the hands of a person. C: A newborn lying in the crib and yawning. D: A newborn lying on the bed with eyes open. E: A newborn held in the arms of a person with eyes wide open. E: A newborn lying in the crib and crying.
Source: (Courtesy Marjorie Pyle, RNC, Lifecircle, Costa Mesa, CA.)Infants use purposeful behavior to maintain the optimal arousal state as follows: (1) actively withdrawing by increasing physical distance, (2) rejecting by pushing away with hands and feet, (3) decreasing sensitivity by falling asleep or breaking eye contact by turning the head, or (4) using signaling behaviors such as fussing and crying. These behaviors permit infants to quiet themselves and reinstate readiness to interact.
The first 6 weeks of life involve a steady decrease in the proportion of active REM sleep to total sleep. A steady increase in the proportion of quiet sleep to total sleep also occurs. Periods of wakefulness increase. For the first few weeks, the wakeful periods seem dictated by hunger, but soon a need for socializing appears. The newborn sleeps approximately 16 to 19 hours/day, with periods of wakefulness gradually increasing. By the fourth week of life, some infants stay awake from one feeding to the next (Gardner & Goldson, 2021).
Infants demonstrate neurobehavioral cues that can guide parents and other caregivers in providing care and social interaction. Cues that indicate stability and engagement include alertness, eye-to-eye contact, facial gaze, smiling, vocalization, smooth movements, and flexion of arms and legs. Signs of distress or disengagement include facial grimace or worried expression, splaying of fingers, gaze aversion, staring, regurgitation or vomiting, and jitteriness (Blackburn, 2018).
Other factors influencing newborn behavior
Gestational age
Gestational age and level of central nervous system (CNS) maturity affect infant behavior. In the preterm neonate with an immature CNS, the entire body responds to a pinprick of the foot, although the response may not be observed by an untrained observer. The more mature infant withdraws only the foot. CNS immaturity is reflected in reflex development, sleep-wake states, and ability (or lack thereof) to regulate or modulate a smooth transition between different states. Preterm infants have brief periods of alertness but have difficulty maintaining alertness without becoming overstimulated, which leads to autonomic instability unless intervention is implemented. Premature or sick infants show signs of fatigue or physiologic stress sooner than full-term healthy infants.
Time
The time elapsed since birth affects the behavior of infants as they attempt to become organized initially. Time elapsed since the previous feeding and time of day also can influence infants’ responses.
Stimuli
Environmental events and stimuli affect the infant’s behavioral responses. The newborn responds to animate and inanimate stimuli. Nurses in intensive care nurseries observe that infants respond to loud noises, bright lights, monitor alarms, and tension in the unit. If a mother is tense, nervous, or uncomfortable while feeding her infant, the infant may sense her tension and demonstrate difficulty feeding.
Maternal medication
No conclusive evidence exists regarding the effects of maternal analgesia or anesthesia during labor on neonatal behavior. Researchers who have studied the effects of epidural medications on breastfeeding behaviors have been unable to show a cause-and-effect relationship. In the postpartum period, there is concern about the effects of maternal opioid use on breastfeeding infants; transfer of opioids through breast milk can cause central nervous system depression in the infant resulting in sedation and respiratory depression (American College of Obstetricians and Gynecologists, 2018; Martin, Vickers, Landau, et al., 2018). Any medication taken by the breastfeeding mother has the potential for transfer to the infant through breastmilk. A resource on the safety of medications in breastfeeding mothers is LactMed: Drugs and Lactation Database, available from the National Library of Medicine (https://www.ncbi.nlm.nih.gov/books/NBK501922/).
Sensory behaviors
From birth, infants possess sensory capabilities that indicate a state of readiness for social interaction. They effectively use behavioral responses in establishing their first dialogues. These responses, coupled with the newborns’ “baby appearance” (e.g., facial proportions of forehead, eyes larger than the lower portion of the face) and their small size and helplessness, rouse feelings of wanting to hold, protect, and interact with them.
Vision
At birth the eye is structurally incomplete, and the muscles are immature. The process of accommodation is not present but improves over the first 3 months of life. The pupils react to light, the blink reflex is stimulated easily, and the corneal reflex is activated by light touch. Term newborns can see objects as far away as 50 cm (2.5 feet). The clearest visual distance is 17 to 20 cm (8 to 12 inches), which is approximately the distance between the mother’s and infant’s faces during breastfeeding or cuddling. Newborns seem to have a preference for faces and can recognize the mother’s face. This facilitates interaction and promotes bonding. They will engage the mother or caregiver with eye contact. Newborns can imitate facial expressions and motions such as protruding the tongue. Newborns prefer complex patterns over nonpatterned stimuli. They prefer black and white, possibly because of the greater contrast. By 2 to 3 months, color discrimination occurs (Gardner & Goldson, 2021).
Hearing
Term newborns can hear and differentiate among various sounds. They will turn toward a sound and attempt to locate the source. The neonate recognizes and responds readily to the mother’s voice and shows a preference for high-pitched intonation. Newborns respond to rhythmic sounds. They are accustomed to hearing the regular rhythm of the mother’s heartbeat, which was a constant sound during intrauterine life. As a result, they respond by relaxing and ceasing to fuss and cry if a regular heartbeat simulator is placed nearby; a lullaby can have the same effect. Hearing is integral to bonding and attachment and may be more important than vision (Gardner & Goldson, 2021).
Routine hearing screening is recommended for all newborns before hospital discharge. See Chapter 23 for a discussion about newborn hearing screening.
Smell
Newborns have a highly developed sense of smell and can detect and discriminate distinct odors. It has been shown that preterm infants as early as 28 weeks are capable of reacting to odors. They react to strong odors such as alcohol or vinegar by turning their heads away but are attracted to sweet smells. By the fifth day of life, newborn infants can recognize their mother’s smell. Breastfed infants are able to smell breast milk and can differentiate their mothers from other lactating women.
Taste
Young infants are particularly oriented toward the use of their mouths, both for meeting their nutritional needs for rapid growth and for releasing tension through sucking. The early development of circumoral sensation, muscle activity, and taste would seem to be preparation for survival in the extrauterine environment. The newborn can distinguish among tastes and has a preference for sweet solutions (Gardner & Goldson, 2021).
Touch
The infant is responsive to touch on all parts of the body. The face (especially the mouth), hands, and soles of the feet seem to be the most sensitive. Reflexes can be elicited by stroking the infant. The newborn’s responses to touch suggest that this sensory system is well prepared to receive and process tactile messages (Gardner & Goldson, 2021). Early skin-to-skin contact with the mother promotes tactile interaction and stimulation. Touch and motion are essential to normal growth and development. However, each infant is unique, and variations can be seen in newborns’ responses to touch. Birth trauma or stress and depressant drugs taken by the mother can decrease the infant’s sensitivity to touch or painful stimuli.
Response to environmental stimuli
Temperament
Each neonate has a unique repertoire of behaviors that are influenced by various factors including temperament, sensory threshold, ability to habituate, and consolability. Temperament refers to individual variations in the reaction pattern of newborns. Newborns possess individual characteristics that affect selective responses to various stimuli present in the internal and external environments. Some infants appear to be quiet by nature and can remain still for extended periods. Their movements may be smooth and relaxed most of the time, and they have little difficulty settling down for feeding. Other infants are more active and seem to be in constant motion; they seem to be excited and interested in exploring the faces and sounds around them. These infants often need help to settle; containment (swaddling), physical contact, and boundaries surrounding the infant in the crib can facilitate a quiet alert state.
Habituation
Habituation is a protective mechanism that allows the infant to become accustomed to environmental stimuli. It is a psychologic and physiologic phenomenon in which the response to a constant or repetitive stimulus is decreased. In the term newborn, this can be demonstrated in several ways. Shining a bright light into a newborn’s eyes causes a startle or squinting the first two or three times. The third or fourth flash elicits a diminished response, and by the fifth or sixth flash the infant ceases to respond (Brazelton & Nugent, 2011). The same response pattern holds true for the sounds of a rattle or stroking the bottom of the foot.
The ability to habituate allows the healthy term newborn to select stimuli that promote continued learning about the social world, thus avoiding overload. The intrauterine environment seems to have programmed the newborn to be especially responsive to human voices, soft lights, soft sounds, and sweet tastes.
The newborn quickly learns the sounds in the home environment and is able to sleep in their midst. The selective responses of the newborn indicate cerebral organization capable of memory and making choices. The ability to habituate depends on the state of consciousness, hunger, fatigue, and temperament. These factors also affect consolability, cuddliness, irritability, and crying.
Consolability
Newborns vary in the ability to console themselves or be consoled. In the crying state, most newborns initiate one of several ways to reduce their distress. Hand-to-mouth movements with or without sucking and being alert to voices, noises, or visual stimuli are common. Some infants are consoled only if they are held and rocked (Brazelton & Nugent, 2011).
Cuddliness
Cuddliness is especially important to parents because they often gauge their ability to care for the child by the child’s responses to their actions. The degree to which newborns relax and mold into the contours of the person holding them varies. One extreme is the infant who always resists being held with thrashing and stiffening of the body. This is in contrast to the infant who immediately relaxes when held and molds to the body of the person. Less extreme behavior is demonstrated by infants who are passive when held and those who gradually mold after being held for a while (Brazelton & Nugent, 2011).
Irritability
Some newborns cry longer and harder than others. For some, the sensory threshold seems low. They are easily upset by unusual noises, hunger, wetness, or new experiences and thus respond intensely. Others with a high sensory threshold require a great deal more stimulation and variation to reach the active, alert state.
Crying
Crying is the language an infant uses most often to communicate needs. It can signal hunger, discomfort, pain, desire for attention, or fussiness. Infants may cry in response to environmental stimuli such as cold, being overstimulated, or being held by multiple persons. Responsiveness of the caregiver to the crying creates trust as the infant learns to associate the caregiver with comfort.
The amount and tone of crying vary based on gestational age, weight, and the reason for the cry (e.g., hunger, pain). A high-pitched cry can be a sign of a neurologic disorder. Some mothers state that they learn to distinguish among the cries. The breastfeeding mother’s body responds physiologically to infant crying by stimulating the milk-ejection reflex (“let-down”).
The duration of crying also varies greatly in each infant; newborns may cry for as little as 5 minutes or as much as 2 hours or more per day. The amount of crying peaks in the second month and then decreases. There is a diurnal rhythm of crying, with more crying occurring in the evening hours.
References
American Academy of Pediatrics and American College of Obstetricians and Gynecologists. Guidelines for perinatal care 8th ed. 2017; Author Washington, D.C.
American College of Obstetricians and Gynecologists. Committee opinion no. 742: Postpartum pain management Obstetrics and Gynecology, 1, 2018, reaffirmed 2020;132: e35-e43.
American College of Obstetricians and Gynecologists. Committee opinion no. 814: Delayed umbilical cord clamping after birth Obstetrics and Gynecology, 6, 2020;136: e100-e106.
Association of Women’s Health & Obstetric and Neonatal Nurses. Neonatal skin care 4th ed. 2018; Author Washington, D.C.
Benjamin J.T. & Maheshwari A. Developmental immunology R.J, Martin A.A, Fanaroff M.C. & Walsh. Fanaroff & Martin’s neonatal-perinatal medicine: Diseases of the fetus and infant 11th ed. 2020; Elsevier Philadelphia.
Blackburn S.T. Maternal, fetal, and neonatal physiology 5th ed. 2018; Elsevier St. Louis.
Brand M.C. & Shippey H.A. Thermoregulation M.T, Verklan M, Walden S. & Forest. Core curriculum for neonatal intensive care nursing 6th ed. 2021; Elsevier St. Louis.
Brazelton T. & Nugent J. Neonatal behavioral assessment scale 4th ed. 2011; MacKeith London, UK.
Centers for Disease Control and Prevention. (2020). Congenital heart defects: Data and statistics. Retrieved from http://www.cdc.gov/ncbddd/heartdefects/data.html.
Chou S.T. Development of the hematopoietic system R.M, Kliegman J.W, St. Geme N.J. & Blum et al. Nelson textbook of pediatrics 21st ed. 2020; Elsevier Philadelphia.
Dell K.M. Fluids, electrolytes, and acid-base homeostasis R.J, Martin A.A, Fanaroff M.C. & Walsh. Fanaroff & Martin’s neonatal-perinatal medicine: Diseases of the fetus and infant 11th ed. 2020; Elsevier Philadelphia.
Desmond M, Rudolph A. & Phitaksphraiwan P. The transitional care nursery: A mechanism for preventive medicine in the newborn Pediatric Clinics of North America, 3, 1966;13: 651-668.
Diehl-Jones W. & Fraser D. Hematologic disorders M.T, Verklan M, Walden S. & Forest. Core curriculum for neonatal intensive care nursing 6th ed. 2021; Elsevier St. Louis.
Elder J.S. Anomalies of the penis and urethra R.M, Kliegman J.W, St. Geme N.J. & Blum et al. Nelson textbook of pediatrics 21st ed. 2020; Elsevier Philadelphia.
Flynn, J. G. (2021). Etiology, clinical features, and diagnosis of neonatal hypertension. In J. A. Garcia-Prats & T. K. Mattoo (Eds.), UpToDate. Retrieved from http://www.uptodate.com/home.
Fraser D. Respiratory distress M.T, Verklan M, Walden S. & Forest. AWHONN’s Core curriculum for neonatal intensive care nursing 6th ed. 2021; Elsevier St. Louis.
Gardner S.L. & Cammack B.H. Heat balance S.L, Gardner B.S, Carter M. & Enzman-Hines et al. Merenstein & Gardner’s handbook of neonatal intensive care: An interprofessional approach 9th ed. 2021; Elsevier St. Louis.
Gardner S.L, Enzman-Hines M. & Nyp M. Respiratory diseases S.L, Gardner B.S, Carter M. & Enzman-Hines et al. Merenstein & Gardner’s handbook of neonatal intensive care: An interprofessional approach 9th ed. 2021; Elsevier St. Louis.
Gardner S.L. & Goldson E. The neonate and the environment: Impact on development S.L, Gardner B.S, Carter M. & Enzman-Hines et al. Merenstein & Gardner’s handbook of neonatal intensive care: An interprofessional approach 9th ed. 2021; Elsevier St. Louis.
Garg M. & Devaskar S.U. Disorders of carbohydrate metabolism in the neonate R.J, Martin A.A, Fanaroff M.C. & Walsh. Fanaroff & Martin’s neonatal-perinatal medicine: Diseases of the fetus and infant 11th ed. 2020; Elsevier Philadelphia.
Hall A.S. & Reavey D.A. Neurologic disorders S.L, Gardner B.S, Carter M. & Enzman-Hines et al. Merenstein & Gardner’s handbook of neonatal intensive care: An interprofessional approach 9th ed. 2021; Elsevier St. Louis.
Kamath-Rayne B.D, Froese P.A. & Thilo E.H. Neonatal hyperbilirubinemia S.L, Gardner B.S, Carter M. & Enzman-Hines et al. Merenstein & Gardner’s handbook of neonatal intensive care: An interprofessional approach 9th ed. 2021; Elsevier St. Louis.
Lawrence R.A. & Lawrence R.M. Breastfeeding: A guide for the medical profession 9th ed. 2022; Elsevier Philadelphia.
Letterio J, Pateva I, Petrosiute A., et al. Hematologic and oncologic problems in the fetus and neonate R.J, Martin A.A, Fanaroff M.C. & Walsh. Fanaroff & Martin’s neonatal-perinatal medicine: Diseases of the fetus and infant 11th ed. 2020; Elsevier Philadelphia.
Martin R, Vickers B, Landau R., et al. Academy of Breastfeeding Medicine clinical protocol no. 28: Peripartum analgesia and anesthesia for the breastfeeding mother Breastfeeding Medicine, 3, 2018;13: 1-8.
Moses, S. (2020). Blood pressure. Family practice notebook. Retrieved from Fpnotebook.com.
Narendran V. The skin of the neonate R.J, Martin A.A, Fanaroff M.C. & Walsh. Fanaroff & Martin’s neonatal-perinatal medicine: Diseases of the fetus and infant 11th ed. 2020; Elsevier Philadelphia.
Neu J. The intestinal microbiome C.A, Gleason S.E. & Juul. Avery’s diseases of the newborn 10th ed. 2018; Elsevier Philadelphia.
Noble L. & Rosen-Carole C. Breastfeeding infants with problems R.A, Lawrence R.M. & Lawrence. Breastfeeding: A guide for the medical profession 9th ed. 2022; Elsevier Philadelphia.
Pagana K.D, Pagana T.J. & Pagana T.N. Mosby’s diagnostic and laboratory test reference 14th ed. 2019; Elsevier St. Louis.
Prazad P.A, Rajpal M.N, Mangurten H.H., et al. Birth injuries R.J, Martin A.A, Fanaroff M.C. & Walsh. Fanaroff & Martin’s neonatal-perinatal medicine: Diseases of the fetus and infant 11th ed. 2020; Elsevier Philadelphia.
Richards M.K. & Goldin A.B. Neonatal gastroesophageal reflux C.A, Gleason S.E. & Juul. Avery’s diseases of the newborn 10th ed. 2018; Elsevier Philadelphia.
Son-Hing J.P. & Thompson G.H. Congenital abnormalities of the upper and lower extremities and spine R.J, Martin A.A, Fanaroff M.C. & Walsh. Fanaroff & Martin’s neonatal-perinatal medicine: Diseases of the fetus and infant 11th ed. 2020; Elsevier Philadelphia.
Sorbara J.C. & Wherrett D.K. Disorders of sex development R.J, Martin A.A, Fanaroff M.C. & Walsh. Fanaroff & Martin’s neonatal-perinatal medicine: Diseases of the fetus and infant 11th ed. 2020; Elsevier Philadelphia.
Vogt B.A. & Springel T. The kidney and urinary tract of the neonate R.J, Martin A.A, Fanaroff M.C. & Walsh. Fanaroff & Martin’s neonatal-perinatal medicine: Diseases of the fetus and infant 11th ed. 2020; Elsevier Philadelphia.
Watchko J.F. Neonatal indirect hyperbilirubinemia and kernicterus C.A, Gleason S.E. & Juul. Avery’s diseases of the newborn 10th ed. 2018; Elsevier Philadelphia.
White K.K, Bouchard M. & Goldberg M.J. Common neonatal orthopedic conditions C.A, Gleason S.E. & Juul. Avery’s diseases of the newborn 10th ed. 2018; Elsevier Philadelphia.