46: The child with cerebral dysfunction
Marilyn J. Hockenberry
![]() http://evolve.elsevier.com/Perry/maternal
http://evolve.elsevier.com/Perry/maternal
The brain and increased intracranial pressure
The brain, tightly enclosed in the solid bony cranium, is well protected but highly vulnerable to pressure that may accumulate within the enclosure (Fig. 46.1). Its total volume—brain (80%), cerebrospinal fluid (CSF; 10%), and blood (10%)—must remain approximately the same at all times. A change in the proportional volume of one of these components (e.g., increase or decrease in intracranial blood) must be accompanied by a compensatory change in another (e.g., decrease or increase in CSF). In this way, the volume and pressure normally remain constant. Examples of compensatory changes are reduction in blood volume, decrease in production of CSF, increase in CSF absorption, or shrinkage of brain mass by displacement of intracellular and extracellular fluid.
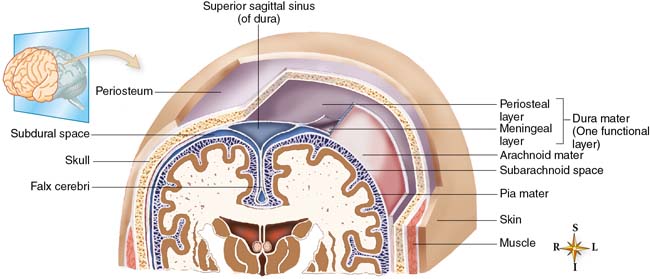
A diagram showing the coronal section of the top of the head showing the meningeal layers and surrounding structures. The clockwise labels read Superior sagittal sinus of the dura, epidural spaces, one functional layer including the periosteum and the dura mater, arachnoid, subarachnoid space, skin, muscle, corpus callosum, third ventricle, lateral ventricle, pia mater, falx cerebri, skull, subdural space, and galea aponeurotica of periosteum.
Source: (From Patton, K. T., & Thibodeau, G. A. [2010]. Anatomy and physiology [7th ed.]. St. Louis, MO: Mosby.)Children with open fontanels compensate for increased volume by skull expansion and widened sutures. However, at any age the capacity for spatial compensation is limited. An increase in intracranial pressure (ICP) may be caused by tumors or other space-occupying lesions, accumulation of fluid within the ventricular system, bleeding, or edema of cerebral tissues. Once compensation is exhausted, any further increase in volume results in a rapid rise in ICP.
The early signs and symptoms of increased ICP, such as headache, vomiting, personality changes, irritability, and fatigue, are often subtle (Box 46.1). In older children, subjective symptoms are headache, especially when arising after lying flat (e.g., on awakening in the morning) or when coughing, sneezing, or bending over, and nausea and vomiting. The child may complain of double vision or blurred vision with movement of the head. Seizures may occur. In children whose cranial sutures have not closed, there is an increase in head circumference and tense or bulging fontanels. Cranial sutures may widen. Head circumference can enlarge until the child is 5 years of age if the condition progresses slowly. As pressure increases, the pupils become progressively sluggish in reaction and eventually become fixed and dilated. The level of consciousness (LOC) progressively deteriorates from drowsiness to eventual coma. Problems related to increased ICP are discussed later in the chapter in relation to head injury and hydrocephalus. (See also Chapter 44, Brain Tumors.)
Physiologic and biochemical changes within the cerebral vasculature serve to complicate the primary causes of increased ICP. Especially in cases of trauma, blood flow often initially increases as a result of venous congestion or vasomotor paralysis. If cerebral hypoxia is associated with the cerebral dysfunction, the compensatory vasodilation caused by oxygen deficiency will tend to increase the cerebral flow. However, blood flow is reduced as ICP progressively increases, with diminished blood supply to the brain tissues. The classic responses observed in adults (widening pulse pressure, increased blood pressure) rarely occur in children or are very late signs. Periodic or irregular breathing is an ominous sign of brainstem (especially medullary) dysfunction that often precedes apnea.
Evaluation of neurologic status
Earlier chapters discuss methods to evaluate neurologic function in relation to numerous aspects of child care. The neurologic examination is an integral part of the health assessment (see Chapter 29) and newborn assessment (see Chapter 23). Chapter 49 discusses some of the tests used to differentiate neuromuscular disorders. The assessment tools and examinations in this chapter are primarily those used to assess intracranial integrity.
Assessment: General aspects
Children younger than 2 years of age require special evaluation because they are unable to respond to directions designed to elicit specific neurologic responses. Early neurologic responses in infants are primarily reflexive; these responses are gradually replaced by meaningful movement in the characteristic cephalocaudal direction of development. This evidence of progressive maturation reflects more extensive myelinization and changes in neurochemical and electrophysiologic properties.
Most information about infants and small children comes from observation of spontaneous and elicited reflex responses. As they develop increasingly complex gross and fine motor skills and communication skills, more sophisticated techniques are used to assess acquisition of developmental milestones. Delay or deviation from expected milestones helps to identify high-risk children. Persistence or reappearance of primitive reflexes indicates a pathologic condition. In evaluating the infant or young child, it is important to obtain the history of the pregnancy, delivery, respiratory status at birth, and neonatal health, including any need for intensive care hospitalization to determine the possible impact of intrauterine and extrauterine environmental influences known to affect the orderly maturation of the central nervous system (CNS). These influences include maternal infections, chemical exposure, trauma, medication, illicit drug use, and metabolic insults.
History
A family history can sometimes offer clues regarding possible genetic disorders with neurologic manifestations. A review of family members often identifies conditions that might otherwise be overlooked, especially increased number of miscarriages or siblings or relatives who died at an early age. The nurse asks questions regarding specific neurologic problems, such as intellectual and developmental disabilities, deafness, epilepsy, blindness, unusual movements, weakness, ataxia, stroke, and progressive mental deterioration. History of consanguinity is also important.
A health history provides valuable clues regarding the cause of neurologic dysfunction. A history is assessed for injury with loss of consciousness, febrile illness, an encounter with an animal or insect, ingestion of neurotoxic substances, inhalation of chemicals, past illness, and known diabetes mellitus or sickle cell disease. Sudden or progressive alterations in movement or mental abilities may provide clues for investigation. It is also important to ascertain the chronologic course of the illness.
Physical examination
Physical examination includes observation of the size and shape of the head (particularly in the infant and young child), spontaneous activity and postural reflex activity, and sensory responses. Note whether the patient is lethargic, drowsy, stuporous, alert, active, or irritable. The nurse also observes the overall tone, noting whether there is a normal flexed posture or one of extreme extension, opisthotonos, or hypotonia. Symmetry of movement is also assessed.
Facial features may suggest a specific syndrome. A high-pitched, piercing cry in an infant is often associated with CNS disorders. An abnormal respiratory cycle, such as prolonged apnea, ataxic breathing, paradoxic chest movement, and hyperventilation, may be the result of a neurologic problem.
Older children can be evaluated by the usual methods used in a neurologic examination. In addition, an estimation of the level of development provides essential information about neurologic function. This assessment is discussed throughout the book in relation to evaluation for specific disorders such as intellectual and developmental disabilities, failure to thrive, attention-deficit/hyperactivity disorder, cerebral palsy, cerebral tumors, and other physical or behavioral problems. Developmental screening tests can assess developmental progress in the young child.
Muscular activity and coordination, including ocular movements and gait, are valuable sources of information. Ocular movements, pupillary response, facial movements, and mouth functions provide clues regarding CNS involvement or impingement. (See Chapter 29 for CNS and reflex testing.) Testing reflexes, strength, and coordination and for the presence and location of tremors, twitching, tics, or other unusual movements is also an aspect of the neurologic assessment.
Altered states of consciousness
Consciousness implies awareness—the ability to respond to sensory stimuli and have subjective experiences. Consciousness has two aspects: alertness, an arousal-waking state that includes the ability to respond to stimuli, and cognition, which includes the ability to process stimuli and produce verbal and motor responses.
An altered state of consciousness usually refers to varying states of unconsciousness that may be momentary or may last for hours, days, or indefinitely. Unconsciousness is depressed cerebral function—the inability to respond to sensory stimuli and have subjective experiences. Coma is defined as a state of unconsciousness from which the patient cannot be aroused, even with powerful stimuli.
Etiology
An altered state of consciousness may be the outcome of several processes that affect the CNS. Impaired neurologic function can result from a direct or indirect cause. Some altered states, such as the diffuse changes observed in encephalitis, are directly related to cerebral insult. Others are the result of dysfunction in other organs or processes. For example, biochemical changes can impair neurologic function without morphologic findings, as in hypoglycemia.
Level of consciousness
Assessment of LOC remains the earliest indicator of improvement or deterioration in neurologic status. LOC is determined by observations of the child’s responses to the environment. Other diagnostic tests, such as motor activity, reflexes, and vital signs, are more variable and do not necessarily directly parallel the depth of the comatose state. The most consistently used terms are described in Box 46.2.
Coma assessment
Diminished alertness as a result of pathologic conditions occurs on a continuum and is designated as the comatose state, which extends from somnolence at one end to deep coma at the other. To produce coma, one of the following must occur: (1) extensive, diffuse, bilateral cerebral hemispheric destruction (the brainstem may be intact); (2) a lesion in the diencephalon; or (3) destruction of the brainstem down to the level of the lower pons.
Several scales have been devised in an attempt to standardize the description and interpretation of the degree of depressed consciousness. The most popular of these is the Glasgow Coma Scale (GCS), which consists of a three-part assessment: eye opening, verbal response, and motor response. The GCS was created to meet a clinical need to identify criteria for the consciousness level. For clinical purposes, the primary role of observation of the LOC is to detect a life-threatening complication such as cerebral edema. The GCS requires observational skills and is readily reproducible between observers.
A pediatric version of the GCS recognizes that expected verbal and motor responses must be related to the child’s age (Fig. 46.2). The pediatric coma scale does not assess verbal responses as such but records smiling, crying, and interaction. It uses a six-point motor scale that is inappropriate for children younger than 6 months of age. In children younger than 5 years of age, speech is understood to be any sound at all, even crying. Young children demonstrate orientation by identifying their parents correctly or giving their own names. When assessing LOC in young children, the nurse may find it helpful to have a parent present to help elicit a desired response. An infant or child may not respond in an unfamiliar environment or to unfamiliar voices.

A table representing the pediatric coma scale. It consists of a table on the top for neurological assessment for pupils including right and left size and reaction, eyes open including spontaneously, to speech, to pain, and none, the best motor response including obeys commands, localizes pain, flexion withdrawal, flexion abnormal, extension, and none, the best response to an auditory and visual stimulus above or equal to two years includes orientation, confused, inappropriate words, incomprehensible words, none, and endotracheal tube or trach, and below two years including smiles, listens, follows, cries, consolable, inappropriate persistent cry, agitated, restless, and no response. In the end, the coma scale total includes handgrip, muscle tone, fontanel, LOC, eye movement, and mood or affects.
Numeric values are assigned to the levels of response in each category. The sum of these numeric values provides an objective measurement of the patient’s LOC. The lower the score, the deeper the coma. A person with an unaltered LOC would score the highest, 15; a score of 8 or below is generally accepted as a definition of coma; the lowest score, 3, indicates deep coma or death.
The GCS in itself is not sufficient to determine depressed consciousness in all children. For example, because a child with quadriplegia cannot respond to commands physically, the child’s GCS can be very low but the child may be cognitively intact. Nevertheless, the GCS provides a more objective method for evaluating the state of consciousness in most cases. Severely injured children (GCS ≤ 8) may have a consistent grading of motor response, verbal response, and eye opening.
The GCS score performed during preadmission (i.e., assessment in the field), in the emergency department, and throughout the inpatient admission is universally accepted as one criterion to determine the patient’s prognosis (Braine & Cook, 2017). GCS scores of 5 or less are associated with poor outcome (Murphy, Thomas, Gertz, et al., 2017).
Irreversible coma
There is no precise diagnosis for clinical death. Different tissues undergo permanent damage after varying periods of exposure to an ongoing insult; the brain (especially the cerebrum) has become the tissue of most importance in determining the time of death. The current concept of dying is a process that takes place over a finite interval of time rather than an event that occurs spontaneously. Brain death is a clinical diagnosis based on the total cessation of brainstem and cortical brain function that causes irreversible widespread brain injury and coma. In children the most common causes are trauma, anoxic encephalopathy, infections, and cerebral neoplasms. The pronouncement of brain death requires two conditions: (1) complete cessation of clinical evidence of brain function and (2) irreversibility of the condition. It is essential to establish the absence of a reversible condition, especially a toxic and metabolic disorder, sedative-hypnotic drugs, paralytic agents, hypothermia, hypotension, and surgically remediable conditions (Nakagawa, Ashwal, Mathur, et al., 2012).
Organ transplantation has created a need to separate the process of death from the retrieval of viable tissues at a time when the brain is already dead. The clinical criteria for brain death must be met so that there is no error. Although the legal status of the concept of death varies among individual states and communities in the United States, the Task Force for the Determination of Brain Death in Children has established guidelines for the determination of brain death in children (see Nursing Care Guidelines box). (See Chapter 36, Organ or Tissue Donation and Autopsy.) At least two different attending physicians should participate in diagnosing brain death in children (Nakagawa et al., 2012).
Neurologic examination
The purpose of the neurologic examination is to establish an accurate, objective baseline of neurologic function. Therefore it is essential that the neurologic examination be documented in a descriptive and detailed fashion, thereby enhancing the ability to detect subtle changes in neurologic status over time. Descriptions of behaviors should be simple, objective, and easily interpreted—for example, “Drowsy but awake and conversationally rational/oriented” or “Sleepy but arousable with vigorous physical stimuli; pressure to nail base of right hand results in upper extremity flexion/lower extremity extension.”
Vital signs
Pulse, respiration, and blood pressure provide information on the adequacy of circulation and the possible underlying cause of altered consciousness. Autonomic activity is most intensively disturbed in deep coma and in brainstem lesions. Body temperature is often elevated; sometimes the elevation is extreme. High temperature is most often a sign of an acute infectious process or heatstroke, but it may be caused by ingestion of some drugs (especially salicylates, alcohol, and barbiturates) or by intracranial bleeding, especially subarachnoid hemorrhage. Hypothalamic involvement may cause elevated or decreased temperature. Serious infection may produce hypothermia.
The pulse is variable and may be rapid, slow and bounding, or feeble. Blood pressure may be normal, elevated, or very low. The Cushing reflex, or pressor response that causes a slowing of the pulse and an increase in blood pressure, is uncommon in children; when it does occur, it is a very late sign of increased ICP. Medications can also affect vital signs. For assessment purposes, actual changes in pulse and blood pressure are more important than the direction of the change.
Respirations are more often slow, deep, and irregular. Slow and deep breathing often occurs in the heavy sleep caused by sedatives, after seizures, or in cerebral infections. Slow, shallow breathing may result from sedatives or opioids. Hyperventilation (deep and rapid respirations) is usually the result of metabolic acidosis or abnormal stimulation of the respiratory center in the medulla caused by salicylate poisoning, hepatic coma, or Reye syndrome (RS). A pattern of alternating hyperventilation and breath holding during wakefulness is common in Rett syndrome.
Breathing patterns have been described with a number of terms (e.g., apneustic, cluster, ataxic, Cheyne-Stokes). However, it is better to describe what is being observed rather than placing a label on it, because the terms are often used and interpreted incorrectly. Periodic or irregular breathing is a sign of brainstem (especially medullary) dysfunction. This is an ominous sign that often precedes complete apnea. The odor of the breath may provide additional clues (e.g., the fruity and acetone odor of ketosis, the foul odor of uremia, the fetid odor of hepatic failure, or the odor of alcohol).
Skin
The skin may offer clues to the cause of unconsciousness. The body surface should be examined for injury, needle marks, petechiae, bites, and ticks. Evidence of toxic substances may be found on the hands, face, mouth, and clothing—especially in small children.
Eyes
Assess pupil size and reactivity (Fig. 46.3). Pupils either do or do not react to light. Pinpoint pupils are commonly observed in poisoning (e.g., opiate or barbiturate poisoning) or in brainstem dysfunction. Widely dilated and reactive pupils are often seen after seizures and may involve only one side. Widely dilated and fixed pupils suggest paralysis of cranial nerve (CN) III (oculomotor nerve) secondary to pressure from herniation of the brain through the tentorium. A unilateral fixed pupil usually suggests a lesion on the same side. Bilateral fixed pupils, if present for more than 5 minutes, usually imply brainstem damage. Dilated and nonreactive pupils also occur in hypothermia, anoxia, ischemia, poisoning with atropine-like substances, or prior instillation of mydriatic drugs. Some of the therapies used (e.g., barbiturates) can alter pupil size and reaction.

A six-part illustration representing the variations in pupil size with altered states of consciousness.
A) It shows the right eye fully opened and the left eye half-closed, representing the ipsilateral pupillary constriction with slight ptosis.
B) It shows both the eyes widely opened, representing bilateral small pupils.
C) It shows both the eyes opened where the left pupil is slightly larger than the right and represents the mid-position, light fixed to all stimuli.
D) It shows both the pupils fully dilated, representing the bilateral dilated and fixed pupils.
E) It shows the right eye fully opened with a small pupil and left eye half-closed with dilated pupil, representing dilated pupils, left eye abducted with ptosis.
F) It shows pinpoint pupils.
The description of eye movements should indicate whether one or both eyes are involved and how the reaction was elicited. Ask the parents if the child has strabismus, which may cause the eyes to appear misaligned.
Blinking observed at rest or in response to a sudden loud noise or bright light implies that the pontine reticular formation is intact. The corneal reflex, blinking of the eyelids when the cornea is touched with a wisp of cotton, can test the integrity of the ophthalmic division of cranial nerve (CN) V (trigeminal nerve). Posttraumatic strabismus indicates CN VI (abducens nerve) damage.
Eye movements are assessed by the doll’s head maneuver, in which the child’s head is rotated quickly to one side and then to the other. When the brainstem centers for eye movement are intact, there is conjugate (paired or working together) movement of the eyes in the direction opposite the head rotation. Absence of this response suggests dysfunction of the brainstem or CN III. Downward or lateral deviation is often observed in association with pupillary dilation in dysfunction of CN III.
The caloric test, or oculovestibular response, is elicited by irrigating the external auditory canal with 10 mL of ice water over a period of approximately 20 seconds (with the head of bed elevated at a 30-degree angle). This test normally causes movement of the eyes toward the side of stimulation. This response is lost when the pontine centers are impaired and thus provides important information in assessment of the comatose patient.
Funduscopic examination reveals additional clues. Because it takes 24 to 48 hours to develop, papilledema (e.g., optic disc swelling, indistinct margins, hemorrhages, tortuosity of vessels, absence of venous pulsations), if it develops at all, will not be evident early in the course of unconsciousness. The presence of retinal hemorrhages in children is usually the result of accidental or inflicted trauma with intracranial bleeding (usually subarachnoid or subdural hemorrhage) but is sometimes caused by infection (Minns, Jones, Tandon, et al., 2017).
Motor function
Observation of spontaneous activity, posture, and response to painful stimuli provides clues to the location and extent of cerebral dysfunction. Asymmetric movements of the limbs or the absence of movement suggests paralysis. In hemiplegia the affected limb lies in external rotation and falls uncontrollably when lifted and allowed to drop. Observations should be described rather than labeled.
In the deeper comatose states, the child has little or no spontaneous movement, and the musculature tends to be flaccid. There is considerable variability in motor behavior in lesser degrees of coma. For example, the child may be relatively immobile or restless and hyperkinetic; muscle tone may be increased or decreased. Tremors, twitching, and spasms of muscles are common observations. The patient may display purposeless plucking or tossing movements. Combative or negativistic behavior is not uncommon. Hyperactivity is more common in acute febrile and toxic states than in cases of increased ICP. Seizures are common in children and may be present in coma as a result of any cause. Any repetitive movements and movements during seizures are described.
Posturing
Primitive postural reflexes emerge as cortical control over motor function is lost in brain dysfunction. These reflexes are evident in posturing and motor movements directly related to the area of the brain involved. Posturing reflects a balance between the lower exciting and the higher inhibiting influences. Strong muscles overcome weaker ones. Flexion posturing (Fig. 46.4A) occurs with severe dysfunction of the cerebral cortex or with lesions to corticospinal tracts above the brainstem. Typical flexion posturing includes rigid flexion, with arms held tightly to the body; flexed elbows, wrists, and fingers; plantar flexed feet; legs extended and internally rotated; and possibly fine tremors or intense stiffness. Extension posturing (see Fig. 46.4B) is a sign of dysfunction at the level of the midbrain or lesions to the brainstem. It is characterized by rigid extension and pronation of the arms and legs, flexed wrists and fingers, clenched jaw, extended neck, and possibly an arched back. Unilateral extension posturing is often caused by tentorial herniation.

A two-part illustration representing different types of posturing.
A) It represents the flexion posturing showing the arms held tightly to the body, flexed elbows, wrists, and fingers, plantarflexed feet, legs extended, and internally rotated.
B) It represents the extension posturing showing rigid extension and pronation of the arms and legs, flexed wrists and fingers, clenched jaw, extended neck, and possibly an arched back.
Posturing may not be evident when the child is quiet but can usually be elicited by applying painful stimuli such as a blunt object pressed on the base of the nail. Nurses should avoid applying thumb pressure to the supraorbital region of the frontal bone (risk of orbital damage). Noxious stimuli (e.g., suctioning), turning, or touching will elicit a response. When the nurse is describing posturing, the stimulus needed to provoke the response is as important as the reaction.
Reflexes
Testing of certain reflexes, such as those present in an intact spinal cord, may be of limited value (see Chapter 46). In general, the corneal, pupillary, muscle-stretch, superficial, and plantar reflexes tend to be absent in deep coma. The state of reflexes is variable in lighter grades of unconsciousness and depends on the underlying pathologic process and the location of the lesion. The doll’s eye reflex maneuver, described previously, reflects paralysis of CN III. The absence of corneal reflexes (CN V) and the presence of a tonic neck reflex are associated with severe brain damage. The Babinski reflex, in which the lateral portion of the bottom of the foot is stroked and causes the big toe to go up, may be of value if it is found to be present consistently in children older than 1 year. A positive Babinski reflex is significant in the assessment of pyramidal tract lesions when it is unilateral and associated with other pyramidal signs. A fluctuating Babinski reflex is often observed after seizures.
Special diagnostic procedures
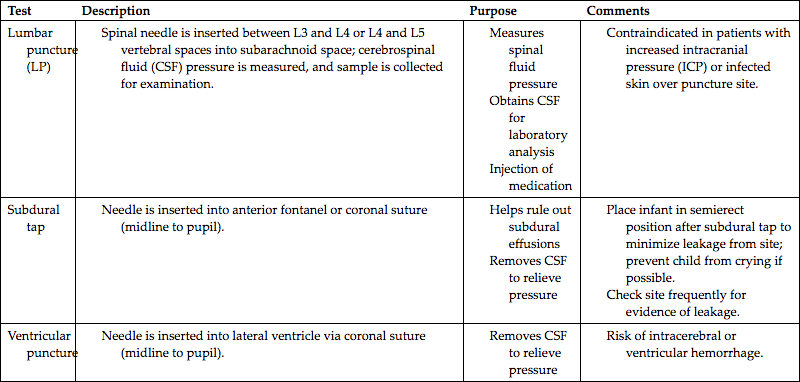
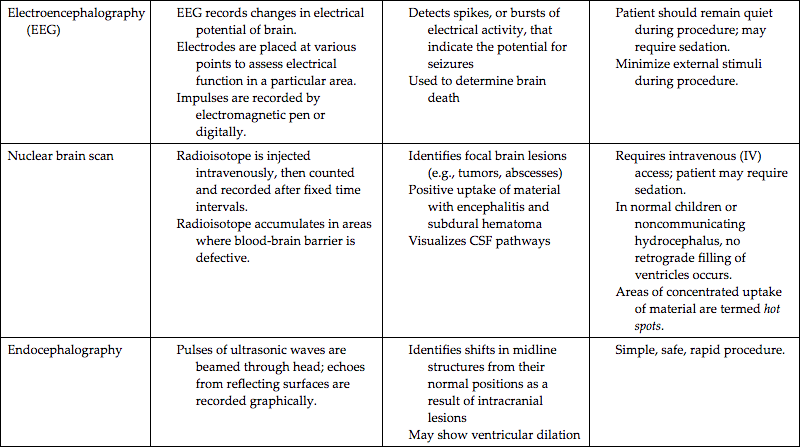
Numerous diagnostic procedures are used for assessment of cerebral function. Laboratory tests that may help to determine the cause of unconsciousness include blood glucose, urea nitrogen, and electrolyte (pH, sodium, potassium, chloride, calcium, and bicarbonate) tests; clotting studies, hematocrit, and a complete blood count; liver function tests; blood cultures if there is fever; and sometimes studies to detect lead or other toxic substances, such as drugs.
An electroencephalogram (EEG) may provide important information. For example, generalized random, slow activity suggests suppressed cortical function, and localized slow activity suggests a space-occupying lesion. A flat tracing is one of the criteria used as evidence of brain death. Examination of spinal fluid is carried out when toxic encephalopathy or infection is suspected. Lumbar puncture is delayed if intracranial hemorrhage is suspected and is contraindicated in the presence of increased ICP because of the potential for brainstem herniation.
Auditory and visual evoked potentials are sometimes used in neurologic evaluation of very young children. Brainstem auditory evoked potentials are useful for evaluating the continuity of brainstem auditory tracts and are particularly useful for detecting demyelinating disease and neoplasms of the brainstem and for distinguishing between brainstem and cortical lesions. For example, a normal evoked potential in a comatose patient suggests involvement of the cerebral hemispheres.
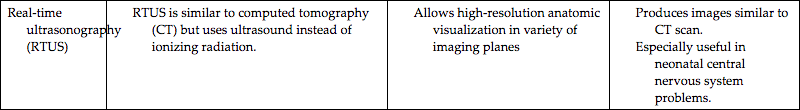
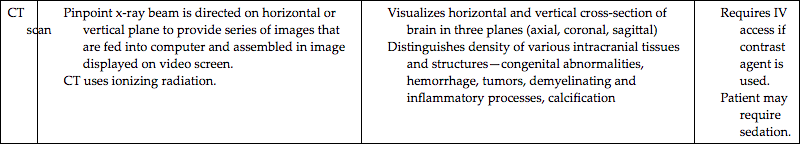
Highly sophisticated tests are carried out with specialized equipment. Two imaging techniques, computed tomography (CT) and magnetic resonance imaging (MRI), assist in diagnosis by scanning both soft tissues and solid matter. Most of these tests are listed in Table 46.1. Because these tests can be threatening to children, the nurse needs to prepare patients and their parents or guardians for the tests and provide support and reassurance during the tests. Consultation with a child life specialist can also be helpful. (See Chapter 39, Preparation for Diagnostic and Therapeutic Procedures.)





Children who are old enough to understand require careful explanation of the procedure, why it is being done, what they will experience, and how they can help. School-age children usually appreciate a more detailed description of why contrast material is injected. Because children are often frightened of needles, they and their families need to be informed of any medication or contrast medium that will be administered intravenously. Special anxiety reduction strategies may be necessary for children who have blood-injury-injection (needle) phobia (McMurtry, Taddio, Noel, et al., 2016). This phobia is the most inheritable of all phobias. The nurse should talk with parents to find out if they also have this phobia and will need help with anxiety management.
The importance of lying still for tests needs to be stressed. Children unfamiliar with the machines can be shown a picture beforehand. Although radiographic examinations are not painful, the machinery often appears so frightening that the child protests because of anxiety. This is especially true of CT and MRI, both of which require that the child’s head be placed within a special immobilizing device. Chin and cheek pads are sometimes used to prevent the slightest head movement, and straps are applied to the body to prevent a slight change in body position. The nurse can explain these events to a frightened child by comparing them to an astronaut’s preparation for a space flight. It is important to emphasize to the child that at no time is the procedure painful.
It is helpful for nurses to become acquainted with the equipment and the general environment in which the test will take place so that they can better explain the procedure to children and their families at their level of understanding. Written material describing the procedure should be available for parents and may be appropriate to share with children. Equipment is often strange and ominous to children. They need constant reassurance from a trusted companion. The nurse should not expect cooperation from a young child. Sedation may be required. Many different agents are currently used for sedation of children undergoing neurologic diagnostic procedures. (See Chapter 30, Pain Management.)
Physical preparation for the diagnostic test may involve administration of a sedative. If so, children should be helped through the preparation and administration and assured that someone will remain with them (if this is possible). Children need continual support and reinforcement during procedures in which they remain conscious. Vital signs and physiologic responses to the procedure are monitored throughout. Many diagnostic procedures performed on an outpatient basis require sedation, and children need recovery time and observation. The nurse should review written instructions with parents if the child is discharged after a procedure. Children who have undergone a procedure with a general anesthetic require postanesthesia care, including positioning to prevent aspiration of secretions and frequent assessment of vital signs, oxygen saturation, and LOC. In addition, other neurologic functions such as pupillary responses, motor strength, and movement are tested at regular intervals. Any surgical wound resulting from the test is checked for bleeding, CSF leakage, and other complications. Children who undergo repeated subdural taps should have their hematocrit monitored to detect excessive blood loss from the procedure.
The child with cerebral compromise
Nursing care of the unconscious child
The unconscious child requires nursing attendance with observation, recording, and evaluation of changes in objective signs. These observations provide valuable information regarding the patient’s progress and often serve as a guide to diagnosis and treatment. Careful and detailed observations are essential for the child’s welfare. In addition, vital functions must be maintained and complications prevented through conscientious and meticulous nursing care. The outcome of unconsciousness is variable and ranges from early and complete recovery to death within a few hours or days, or persistent and permanent unconsciousness, or recovery with varying degrees of residual mental or physical disability. The outcome and recovery of the unconscious child may depend on the level of nursing care and observational skills.
Direct emergency measures toward ensuring circulation, airway, and breathing (CAB); stabilizing the spine when indicated; treating shock; and reducing ICP (if present). Delayed treatment often leads to increased damage. Therapies for specific causes of unconsciousness begin as soon as emergency measures have been implemented; in many cases they occur concurrently. Because nursing care is closely related to the medical management, both are considered here.
Continual observation of the LOC, pupillary reaction, and vital signs is essential to management of CNS disorders. Regular assessment of neurologic status and vital signs is an integral part of the nursing care of unconscious children. The frequency depends on the cause of unconsciousness, the LOC, and the progression of cerebral involvement. Intervals between observations may be as short as every 15 minutes or as long as every 2 hours. Significant alterations are reported immediately.
The temperature is measured every 2 to 4 hours, depending on the child’s condition. An elevated temperature may occur in children with CNS dysfunction; therefore a light covering may be sufficient. Vigorous efforts, such as tepid sponge baths or application of a hypothermia blanket, are needed to prevent brain damage if the rectal temperature exceeds 104°F (40°C).
The LOC is assessed periodically, including pupillary size, equality, and reaction to light. Signs of meningeal irritation, such as nuchal rigidity, need to be assessed. Assessment of LOC also includes response to vocal commands, spontaneous behavior, resistance to care, and response to painful stimuli. Note any abnormal movements, changes in muscle tone or strength, and body position. If a seizure occurs, describe the seizure, including the body areas involved from the beginning to the end of the seizure, and the duration of seizure (see Box 46.8 and Critical Thinking Case Study later in the chapter).
Pain management for the unconscious child requires astute nursing observation and management. Signs of pain include changes in behavior (e.g., increased agitation and rigidity) and alterations in vital signs and perfusion (usually an increased heart rate, respiratory rate, and blood pressure and decreased oxygen saturation). Because these findings are not specific for pain, the nurse should be alert for their appearance during times of induced or suspected pain and for their disappearance after the inciting procedure or the administration of analgesia. A pain assessment record is used to document indications of pain and the effectiveness of interventions. (See Chapter 30, Pain Assessment.) The use of opioids, such as morphine, to relieve pain is controversial because these drugs can mask signs of altered consciousness or depress respirations. However, unrelieved pain activates the stress response, which can elevate ICP. To block the stress response, some authorities advocate the use of analgesics, sedatives, and, in some cases such as head injury, paralyzing agents via continuous intravenous (IV) infusion. A commonly used combination is fentanyl, midazolam, and vecuronium (Norcuron). If there are concerns about assessing the LOC or respiratory depression, naloxone can be used to reverse the opioid effects. Regardless of the drugs used, adequate dosage and regular administration are essential to provide optimum pain relief.
Other measures to relieve discomfort include providing a quiet, dimly lit environment; limiting visitors; preventing any sudden, jarring movement, such as banging into the bed; and preventing an increase in ICP. The latter is most effectively achieved by proper positioning and prevention of straining, such as during coughing, vomiting, or defecating. (See Chapter 5, Pain Management.) Antiepileptic drugs, such as fosphenytoin (Cerebyx) or phenobarbital, may be ordered for control of seizure activity.
Respiratory management
Respiratory effectiveness is the primary concern in the care of the unconscious child, and establishment of an adequate airway is always the first priority. Carbon dioxide has a potent vasodilating effect and will increase cerebral blood flow (CBF) and ICP. Cerebral hypoxia at normal body temperature that lasts longer than 4 minutes often causes irreversible brain damage.
Children in lighter stages of coma may be able to cough and swallow, but those in deeper states of coma are unable to manage secretions, which tend to pool in the throat and pharynx. Dysfunction of CNs IX and X (glossopharyngeal and vagus nerves) places the child at risk of aspiration and cardiac arrest. Therefore position the child with the head and body to the side to prevent aspiration of secretions and empty the stomach to reduce the likelihood of vomiting. In infants, the blockage of air passages from secretions can happen in seconds. In addition, upper airway obstruction from laryngospasm is a common complication in comatose children.
An oral airway can be used for the child who is suffering a temporary loss of consciousness, such as after a contusion, seizure, or anesthesia. For children who remain unconscious for a longer time, a nasotracheal or orotracheal tube is inserted to maintain the open airway and facilitate removal of secretions. A tracheostomy is performed in cases where laryngoscopy for introduction of an endotracheal tube would be difficult or dangerous or for a child who needs long-term ventilatory support. Suctioning is used only as needed to clear the airway, exerting care to prevent increasing ICP. Respiratory status is observed and evaluated regularly. Signs of respiratory distress may indicate a need for ventilator assistance.
Mechanical ventilation is usually indicated when the respiratory center is involved. Blood gas analysis is performed regularly, and oxygen is administered when indicated. Moderately severe hypoxia and respiratory acidosis are often present, but they are not always evident from clinical manifestations. Hypoventilation often accompanies unconsciousness and may lead to respiratory alkalosis, or it may represent the body’s attempt to compensate for metabolic acidosis. Blood gas and pH determinations are essential guides for electrolyte therapy. Chest physiotherapy is carried out on a regular basis, and the child’s position is changed at least every 2 hours to prevent pulmonary complications. Regular oral hygiene is recommended to reduce the risk of ventilator-associated pneumonia (VAP) (Hua, Xie, Worthington, et al., 2016).
Intracranial pressure monitoring
The role of ICP monitoring after traumatic brain injury (TBI) is controversial. Placement of the ICP monitor often occurs in the emergency department and where older in age (Kannan, Quistberg, Wang, et al., 2017). Early placement of ICP monitors may guide assessment management of patients with intracranial hypertension or those at higher risk for developing intracranial hypertension. ICP monitoring also may assist with decision making regarding transfer to the operating room or pediatric intensive care unit (PICU). However, in a large study using two national databases of 3084 children with severe TBI, no evidence was found of a benefit from ICP monitoring on functional survival of children with severe TBI (Bennett, DeWitt, Greene, et al., 2017). Development of noninvasive ICP sensors has the potential of decreasing the need for invasive interventions in pediatric patients in the future (Harary, Dolmans, & Gormley, 2018).
Direct ventricular pressure measurement remains the gold standard of ICP monitoring. The catheter method involves introduction of a catheter into the lateral ventricle on the nondominant side, if known, or placement in the subdural space. The catheter has the advantage of providing a means of extraventricular (or continuous) drainage of CSF to reduce pressure. A drainage bag attached to the system is kept at the level of the ventricles and can be lowered to decrease ICP. This device requires full penetration of the brain, requires skill and experience with placement, and carries the risk of infection. Infection risks can be lowered by always using aseptic technique when handling the external ventricular drainage (EVD) system, manipulating the EVD as little as possible, and sterile dressing changes only weekly or when the dressing is compromised, whichever occurs first (Hepburn-Smith, Dynkevich, Spektor, et al., 2016).
With the bolt method, the end of the bolt is placed into the subarachnoid space. The bolt cannot be adequately secured in a small child’s pliant skull, although special modifications have been developed for children younger than 6 years of age. The placement of the bolt is not adjusted by anyone except the neurosurgeon who placed the device. The neurosurgeon is notified if a satisfactory waveform is not observed.
An epidural sensor can be placed between the dura and the skull through a burr hole and connected to a stopcock assembly and a transducer, which provides a readout of the pressure. Although less invasive, the epidural sensor may have inconsistent correlation of pressure readings. In infants, a fontanel transducer can be used to detect impulses from a pressure sensor and convert them to electrical energy. The electrical energy is then converted to visible waves or numeric readings on an oscilloscope. ICP measurement from the anterior fontanel is noninvasive but may prove to be inaccurate if the equipment is poorly placed or inconsistently recalibrated. Intraparenchymal pressure-monitoring devices (e.g., Camino) use fiberoptic technology and perform reliably.
ICP can be increased by direct instillation of solutions; antibiotics are administered systemically if a positive CSF culture is obtained. However, ICP monitoring rarely causes infection. CSF is a body fluid; implement Standard Precautions according to hospital policy. (See Chapter 39, Infection Control.)
Nurses caring for patients with intracranial monitoring devices must be acquainted with the system, assist with insertion, interpret the monitor readings, and be able to distinguish between danger signals and mechanical dysfunction. Because systemic blood pressure, ICP, and therefore cerebral perfusion pressure (CPP) are normally lower in children, the child’s age must be taken into account when deciding what constitutes abnormally high ICP or abnormally low CPP.
Several medical measures are available to treat increased ICP resulting from cerebral edema. These include sedation, CSF drainage, and osmotic diuretics. Osmotic diuretics may provide rapid relief of ICP in emergency situations. Although their effect is transient, lasting only approximately 6 hours, they can be lifesaving in emergencies. These substances are rapidly excreted by the kidneys and carry with them large quantities of sodium and water. Mannitol (or sometimes urea) administered intravenously is the drug most commonly used for rapid reduction of ICP. The infusion is generally given slowly but may be pushed rapidly if there is herniation or impending herniation. Because of the profound diuretic effect of the drug, an indwelling catheter is inserted to ensure bladder emptying. Arterial carbon dioxide (PaCO2) should be maintained at approximately 30 mm Hg to produce vasoconstriction, which reduces CBF, thereby decreasing ICP. Recording and analyzing the child’s volume state, plasma sodium concentration, and serum osmolarity can avert potential fluid and electrolyte problems. Administration of adrenocorticosteroids is not recommended for cerebral edema secondary to head trauma.
Nursing activities
In cases of high levels of increased ICP, nursing procedures tend to trigger reactive pressure waves in many children. For example, increased intrathoracic or abdominal pressure will be transmitted to the cranium. The goals of monitoring a child who is neurologically compromised include maintaining CPP; controlling ICP, cerebral edema, and factors that increase cerebral metabolism (e.g., fever, seizures); and maintaining hemodynamic stability. Take particular care in positioning these patients to avoid neck vein compression that may further increase ICP by interfering with venous return.
Sandbags or other support devices can help maintain correct head position. The child can be propped to one side or the other, and the use of a pressure-relieving or pressure-decreasing mattress decreases the chance of prolonged pressure to vulnerable skin areas. Frequent clinical assessment of the child cannot be replaced by an ICP monitoring device.
It is important to avoid activities that may increase ICP by causing pain or emotional stress. Clustering nursing activities together and minimizing environmental stimuli by decreasing noxious procedures help control ICP. Range of motion exercises can be carried out gently but should not be performed vigorously. Any necessary disturbing procedures should be scheduled to take advantage of therapies that reduce ICP, such as osmotherapy and sedation. Make efforts to minimize or eliminate environmental noise, including managing the number of visitors. Assessment and intervention to relieve pain are important nursing functions to decrease ICP.
Suctioning and percussion are poorly tolerated; these procedures are contraindicated unless the child has concurrent respiratory problems. Hypoxia and the Valsalva maneuver associated with cough acutely elevate ICP. Vibration, which does not increase ICP, accomplishes excellent results and should be tried first if treatment is needed. If suctioning is necessary, it should be used judiciously and preceded by hyperventilation with 100% oxygen, which can be monitored during suctioning with a pulse oxygen sensor reading to determine oxygen saturation.
Nutrition and hydration
In the unconscious child, fluids and calories are supplied initially by the IV route. The type of fluid administered depends on the patient’s general condition. Children on the ketogenic diet and with certain metabolic disorders, such as pyruvate dehydrogenase deficiency, should receive normal saline rather than fluids containing dextrose, which can cause seizures and worsen their condition. Fluid therapy requires careful monitoring and adjustment based on neurologic signs and electrolyte determinations. Often unconscious children cannot tolerate the same amounts of fluid as when they are healthy. Overhydration must be avoided to prevent fatal cerebral edema. When cerebral edema is a threat, fluids may be restricted to reduce the chance of fluid overload. Examine skin and mucous membranes for signs of dehydration. Adjustments to fluid administration are based on urinary output, serum electrolytes and osmolarity, blood pressure, and arterial filling pressure. Observation for signs of altered fluid balance related to abnormal pituitary secretions is a part of nursing care.
Provide long-term nutrition in a balanced formula given by nasogastric or gastrostomy tube. The nasogastric tube is usually taped in place, with care taken to prevent pressure on the nares. Most children have continuous feedings. When bolus feedings are used, the tube is rinsed with water after each feeding. Tubes are replaced according to institutional policy. Irritation of the nasal mucosa is prevented by alternating nares each time the nasogastric tube is replaced.
Avoid overfeeding to prevent vomiting and the associated risk of aspiration. Stomach contents are aspirated with a syringe and measured before feeding to ascertain the amount remaining in the stomach. The removed contents may be refed. If the residual volume is excessive (depending on the child’s size), consult the dietitian and physician regarding the composition and amount to determine whether changes are required to provide calories and nutrients in a smaller volume.
Altered pituitary secretion
An altered ability to handle fluid loads is attributed in part to the syndrome of inappropriate antidiuretic hormone secretion (SIADH) and diabetes insipidus (DI) resulting from hypothalamic dysfunction. SIADH often accompanies CNS conditions such as head injury, meningitis, encephalitis, brain abscess, brain tumor, and subarachnoid hemorrhage. In the child with SIADH, scant quantities of urine are excreted, electrolyte analysis reveals hyponatremia and hypoosmolality, and manifestations of overhydration are evident. It is important to evaluate all parameters because the reduced urinary output might be erroneously interpreted as a sign of dehydration. The treatment of SIADH consists of fluid restriction until serum electrolytes and osmolality return to normal levels. If fluid restriction is not completely ineffective, medications such as sodium chloride and diuretics may be used.
DI may occur after intracranial trauma. In DI there is increased urinary volume and the accompanying danger of dehydration. Adequate replacement of fluids is essential, and observation of electrolyte balance is necessary to detect signs of hypernatremia and hyperosmolality. Exogenous vasopressin may be administered.
Medications
The cause of unconsciousness determines specific drug therapies. Children with infectious processes are given antibiotics appropriate to the disease and the infecting organism. Corticosteroids are prescribed for inflammatory conditions and edema. Cerebral edema is an indication for osmotic diuretics. Antiepileptic medications are prescribed for seizure activity. Sedation in the combative child provides amnesic and anxiolytic properties in conjunction with a paralytic agent. This combination decreases ICP and allows treatment of cerebral edema. Usual drugs include morphine and midazolam. Midazolam is attractive because of its short half-life.
Deep coma induced by the administration of barbiturates is controversial in the management of ICP. Barbiturates are currently reserved for the reduction of increased ICP when all else has failed. Barbiturates decrease the cerebral metabolic rate for oxygen and protect the brain during times of reduced CPP. Barbiturate coma requires extensive monitoring. EEG monitoring can assess depth of coma, record EEG background abnormalities that can help to predict outcome, and evaluate any seizure activity. Cardiovascular and respiratory support and ICP monitoring are needed to assess response to therapy. Paralyzing agents such as vecuronium may be needed to aid in performing diagnostic tests, improving effectiveness of therapy, and reducing the risks of secondary complications. Elevation of ICP or heart rate in patients who are being given paralyzing agents or are under sedation may indicate the need for another dose of either or both medications or the need for pain medication.
Thermoregulation
Hyperthermia often accompanies cerebral dysfunction; if it is present, the nurse implements measures to reduce the temperature to prevent brain damage from hyperthermia and to reduce metabolic demands generated by the increased body temperature. Antipyretics are the method of choice for fever reduction; cooling devices are used for hyperthermia. (See Chapter 39, Controlling Elevated Temperatures.) Laboratory tests and other methods help to determine the cause, if any, of the hyperthermia. Treatment with hypothermia and barbiturates increases the risk of iatrogenic complications.
Elimination
A urinary catheter is usually inserted in the acute phase, but diapers may be used and weighed to record urinary output. The child who previously had bowel and bladder control is generally incontinent. If the child remains comatose for a long period, the indwelling catheter may be removed and periodic bladder emptying accomplished by intermittent catheterization. Stool softeners are usually sufficient to maintain bowel function, but suppositories or enemas may be needed occasionally for adequate elimination and to prevent fecal impaction. The passage of liquid stool after a period of no bowel activity is usually a sign of impaction. To avoid this preventable problem, daily recording of bowel activity is essential.
Hygienic care
Routine measures for cleansing and maintaining skin integrity are an integral part of nursing care of the unconscious child. Skinfolds require special attention to prevent excoriation. The child who is unable to move is prone to develop tissue breakdown and necrosis; the child is placed on a resilient appliance (e.g., alternating-pressure or water-filled mattress) to prevent pressure on prominent areas of the body. The goal is prevention by regular change of position and inspection of vulnerable areas (e.g., the ankle, heels, trochanter, sacrum, and shoulder). Unconscious children undergo numerous invasive procedures, and the skin sites used for these procedures require special assessment and intervention to promote healing and prevent infection. Keep bed linens and any clothing dry and free of wrinkles. Rubbing the back and extremities with lotion stimulates circulation and helps prevent drying of the skin. However, to prevent further tissue damage, do not massage reddened and nonblanching skin. (See Chapter 39, Maintaining Healthy Skin.) If the child requires surgery or radiography, the nurse checks all dressings, bony sites, catheters, and IV access lines before and after the procedure.
Oral care is performed at least twice daily because the mouth tends to become dry or coated with mucus. The teeth are carefully brushed with a soft toothbrush or cleaned with gauze saturated with saline. Commercially prepared cleansing devices, such as Toothettes, are convenient for cleansing the mouth and teeth. Lips are coated with ointment to protect them from drying, cracking, or blistering.
The unconscious child is also prone to eye irritation. The corneal reflexes are absent; therefore the eyes are easily irritated or damaged by linens, dust, or other substances that may come in contact with them. Excessive dryness results from incomplete closure of the lids and/or decreased secretions, especially if the child is undergoing osmotherapy to reduce or prevent brain edema.
Keep the child’s hair combed, and secure to prevent tangling. Keep the scalp clean with dry or wet shampoos as needed. The child’s head may need to be shaved for tests or surgical procedures. The family may want the hair to be saved.
Positioning and exercise
The unconscious child is positioned to minimize ICP and to prevent aspiration of saliva, nasogastric secretions, and vomitus. The head of the bed is elevated, and the child is placed in a side-lying or semiprone position. A small, firm pillow is placed under the head, and the uppermost limbs are flexed and supported with pillows. The weight of the body should not rest on the dependent arm. In the semiprone position the child lies with the dependent arm at the side behind the body; the opposite side is supported on pillows, and the uppermost arm and leg are flexed and resting on the pillows. This position prevents undue pressure on the dependent extremities. The dependent position of the face encourages drainage of secretions and prevents the flaccid tongue from obstructing the airway.
Normal range-of-motion exercises help maintain function and prevent contractures of joints. Perform exercises gently to minimize increasing ICP and with full range of motion. Place a small, rolled pad in the palms to help maintain proper positioning of fingers. Splinting may be needed to prevent severe contractures of the wrists, knees, or ankles.
Stimulation
Sensory stimulation is as important in the care of the unconscious child as it is in the care of the alert child. For the temporarily unconscious or semiconscious child, sensory stimulation helps arouse the child to the conscious state and orient the child in terms of time and place. Auditory and tactile stimulation are especially valuable. Tactile stimulation is not appropriate for a child in whom it may elicit an undesirable response. However, for other children tactile contact often has a relaxing and calming effect. When the child’s condition permits, holding or rocking the child is soothing and provides the body contact needed by young children.
Hearing is often intact in a state of coma. Hearing is the last sense to be lost and the first one to be regained; speak to the child as any other child. Conversation around the child should not include thoughtless or derogatory remarks. Soft music is often used to provide auditory stimulation. Singing the child’s favorite songs or reading a favorite story is a strategy used to maintain the child’s contact with a familiar world. Playing songs or favorite stories recorded in the parents’ voices can provide a continuous source of familiar stimulation.
Family support
Helping the parents of an unconscious child cope with the situation is especially difficult. They may demonstrate all of the guilt, fear, hostility, and anxiety of any parent of a seriously ill child (see Chapter 36). In addition, these parents face the uncertain outcome of the cerebral dysfunction. The fear of death, cognitive impairment, or permanent physical disability is present. Nursing intervention with parents depends on the nature of the pathologic condition, the parents’ coping skills, and the parent-child relationship before injury or illness.
Awakening from a coma is a gradual process; however, some children regain consciousness within a short time. If there is little or no residual effect, the child is discharged home fairly soon. The parents need the most intensive nursing intervention during the period of crisis and uncertainty. During the recovery phase the nurse gives them information, clarifies it as needed, and encourages them to become involved in the child’s care. Often the child’s hospitalization is brief; however, some children require extended hospitalization for intensive therapy and rehabilitation. The parents of children who die require support and guidance to cope with the reality of the death and to resolve their grief (see Chapter 36).
Probably the most difficult situations are those that involve children who never regain consciousness. Unlike losing a child through death, these children lack finality, which often leaves the parents in a state of suspended grief. Like parents of dying children, parents of comatose children search for any signs of hope. Well-meaning friends and relatives relate instances of miraculous recoveries. The parents seek confirmation and support for such possibilities and assign erroneous meanings to any sign in the child that might be interpreted as evidence of recovery (e.g., reflexive muscle contractions).
At these times nurses need to respond with compassion and honesty. They can acknowledge that miraculous recoveries do occur but are rare. The important message is to maintain open communication with the family.
Like parents who lose a child through death, the parents of a child who is unconscious attempt to construct a representation of the child. They bring items that belong to the child, such as favorite toys or music. This may be interpreted as an attempt to provide stimulation for the child in the hope of eliciting a response, to let the hospital staff know the child as the unique individual he or she was, and to reconstitute an image of the child “lost” to them and for whom they mourn. The nurses’ recognition and understanding of these behaviors and coping mechanisms are important to support the parents in their grief process.
In addition to the process of grieving for the “lost” child, the parents may face difficult decisions. When the child’s brain is so severely damaged that vital functions must be maintained by artificial means, the parents must make the final decision whether to remove the life support systems and allow a natural death. After parents are provided with information about what allowing a natural death and removal from life support mean, the parents may turn to both the provider and the nurses with their questions and concerns. Nurses play a critical role in assisting families in participating in their child’s care to the greatest extent possible and in planning the child’s death when that is the inevitable outcome of the neurologic disorder (Bloomer, Endacott, Copnell, et al., 2016).
Head injury
Head injury is a pathologic process involving the scalp, skull, meninges, or brain as a result of mechanical force. Unintentional injuries are the number-one health risk for children and the leading cause of death in children older than 1 year of age (Chen, Shi, Stanley, et al., 2017). However, children younger than 1 year of age have a significantly higher rate of severe head injury (Chen et al., 2017). In 2013 approximately 660,000 children 0 to 14 years old experienced a TBI and 17,900 of those children were hospitalized; 1484 children died as a result of their brain injury (Taylor, Bell, Breiding, et al., 2017).
Etiology
The most common causes of head injury in children are falls, being struck by or striking an object with one’s head, and motor vehicle accidents, in that order (Centers for Disease Control and Prevention, 2017a). Assaults are the leading cause of death from TBI in children 4 years of age or younger (Taylor et al., 2017). Neurologic injury accounts for the highest mortality rate, with boys usually affected twice as often as girls. There are a number of head trauma strategies, including safety gates on stairs, restricting sleeping in the top bunk to children older than 6 years of age, seat belts and car seat use, and helmets during recreational activities such as biking and skiing. Furthermore, preventing child abuse is necessary and possible.
Pathophysiology
The pathology of brain injury is directly related to the force of impact. Intracranial contents (brain, blood, CSF) are damaged because the force is too great to be absorbed by the skull and musculoligamentous support of the head. Although nervous tissue is delicate, it usually requires a severe blow to cause significant damage.
A child’s response to head injury is different from that of adults. The larger head size in proportion to body size and insufficient musculoskeletal support render the very young child particularly vulnerable to acceleration-deceleration injuries.
Primary head injuries are those that occur at the time of trauma and include skull fractures, contusions, intracranial hematomas, and diffuse injuries. Subsequent complications include hypoxic brain injury, increased ICP, and cerebral edema. The predominant feature of a child’s brain injury is the diffuse amount of swelling that occurs. Hypoxia and hypercapnia threaten the energy requirements of the brain and increase CBF. The added volume across the blood-brain barrier and the loss of autoregulation exacerbate cerebral edema. Pressure inside the skull that is greater than arterial pressure results in inadequate perfusion. Because the cranium of very young children has the ability to expand and the thin skull is more compliant, they may tolerate increases in ICP better than older children and adults.
Physical forces act on the head through acceleration, deceleration, or deformation. Acceleration or deceleration is more descriptive of the circumstances responsible for most head injuries. When the stationary head receives a blow, the sudden acceleration causes deformation of the skull and mass movement of the brain. Continued movement of the intracranial contents allows the brain to strike parts of the skull (e.g., the sharp edges of the sphenoid or the irregular surface of the anterior fossa) or the edges of the tentorium.
Although the brain volume remains unchanged, significant distortion and cavitation occur as the brain changes shape in response to the force transmitted from the impact to the skull. This deformation can cause bruising at the point of impact (coup) or at a distance as the brain collides with the unyielding surfaces opposite or far removed from the point of impact (contrecoup) (Fig. 46.5). Thus a blow to the occipital region can cause severe injury to the frontal and temporal areas of the brain.

An illustration showing the mechanical distortion of the cranium during injury. The different points on the skull are marked A through E, where A is the point of injury and represents the preinjury contour of the skull, B represents the immediate postinjury contour of the skull, C represents the torn subdural vessels, D represents the shearing forces inside the brain, and E represents the trauma from the contact with the floor of the cranium.
Source: (Redrawn from Grubb, R. L., & Coxe, W. S. [1974]. Central nervous system trauma: Cranial. In S. G. Eliasson, A. L. Presky, & W. B. Hardin [Eds.], Neurological pathophysiology. New York, NY: Oxford University Press.)When a moving head strikes a stationary surface, such as during a fall, sudden deceleration occurs and causes the greatest cerebral injury at the point of impact. Deceleration is responsible for most severe brainstem injuries.
Children with an acceleration-deceleration injury demonstrate diffuse generalized cerebral swelling produced by increased blood volume or by a redistribution of cerebral blood volume (cerebral hyperemia) rather than by the increased water content (edema).
Another effect of brain movement is shearing forces, which are caused by unequal movement or different rates of acceleration at various levels of the brain. A shearing force may tear small arteries that travel from the cerebral surfaces through the meninges to the dural sinuses and cause subdural hemorrhages. Shearing or stretching effects can also be transmitted to nerve fibers. Maximum stress from the shearing force occurs at the interface between structures of different density so that the gray matter (cell body) rapidly accelerates, whereas the white matter (axons) tends to lag behind. Although maximum shearing forces are at the cerebral surface and extend toward the center of rotation within the brain, the most serious effects are often in the area of the brainstem. Severe compression of the skull can cause the brain to be forced through the tentorial opening and produce irreparable damage to the brainstem.
A GCS value of 8 or less in pediatric patients indicates severe injury and requires aggressive therapeutic management (Hartman & Cheifetz, 2020). Three out of four children with a score of 3 or 4 will be severely disabled, be in a persistent vegetative state, or die within a year of their injury (Fulkerson, White, Rees, et al., 2015). A number of studies indicate that the Simplified Motor Scale (SMS) is equivalent to the GCS in predictive power but that the GCS is better for prognosticating death (Singh, Murad, Prokop, et al., 2013).
Concussion
The most common and mildest TBI is concussion, an alteration in mental status with or without loss of consciousness that occurs immediately after a head injury (McCrea, Nelson, & Guskiewicz, 2017). Direct head trauma and “whiplash” seen with rapid acceleration and deceleration of the head are the most frequent causes in children. Sports-related activities are responsible for the majority of concussions (Mullally, 2017).
The hallmarks of a concussion are confusion and amnesia. These are often not preceded by loss of consciousness and may occur immediately after the injury or several minutes later. The belief that loss of consciousness is the hallmark of concussion is a common misconception. A study of 182 adolescent athletes who sustained a concussion found that only 22% lost consciousness, whereas 34% experienced amnesia (Meehan, Mannix, Stracciolini, et al., 2013).
The pathogenesis of concussion is still unclear, but it may be a result of shearing forces that cause stretching, compression, and tearing of nerve fibers, particularly in the area of the central brainstem, the seat of the reticular activating system. It has also been suggested that the anatomic alterations of nerve fibers cause the release of large quantities of acetylcholine into the CSF and a reduction in oxygen consumption with increased lactate production.
Contusion and laceration
The terms contusion and laceration are used to describe visible bruising and tearing of cerebral tissue. Contusions represent petechial hemorrhages or localized bruising along the superficial aspects of the brain at the site of impact (coup injury) or a lesion remote from the site of direct trauma (contrecoup injury). In serious accidents there may be multiple sites of injury.
The major areas of the brain susceptible to contusion or laceration are the occipital, frontal, and temporal lobes. In addition, the irregular surfaces of the anterior and middle fossae at the base of the skull are capable of producing bruises or lacerations on forceful impact. Contusions may cause focal disturbances in strength, sensation, or visual awareness. The degree of brain damage in the contused areas varies according to the extent of vascular injury. Signs vary from mild, transient weakness of a limb to prolonged unconsciousness and paralysis. However, the signs and symptoms may be clinically indistinguishable from those of concussion.
Infants who are roughly shaken, referred to as shaken baby syndrome or abusive head trauma, can sustain profound neurologic impairment, seizures, retinal hemorrhages (usually bilateral), and intracranial subarachnoid or subdural hemorrhages (Joyce & Huecker, 2019).
Cerebral lacerations are generally associated with penetrating or depressed skull fractures. However, they may occur without fracture in small children. When brain tissue is actually torn, with bleeding into and around the tear, more severe and prolonged unconsciousness and paralysis usually occur, leaving permanent scarring and some degree of disability.
Fractures
Skull fractures result from a direct blow or injury to the skull and are often associated with intracranial injury. Many of the falls that resulted in a skull fracture in children younger than 2 years of age involved short distances of less than 3 feet, such as falls from a caregiver’s arms (Burrows, Trefan, Houston, et al., 2015).
The types of skull fractures that occur are linear, comminuted, depressed, open, basilar, and growing fractures. As a rule, the faster the blow, the greater the likelihood of a depressed fracture; a low-velocity impact tends to produce a linear fracture.
Linear skull fractures are a single fracture line that starts at the point of maximum impact and spreads; however, they do not cross suture lines. Linear skull fractures constitute the majority of childhood skull fractures and typically occur in the parietal bone. Most linear skull fractures are associated with an overlying scalp hematoma, particularly in infants younger than 2 years of age and in the parietal or temporal region (Burns, Grool, Klassen, et al., 2016). Scalp hematomas, in turn, are associated with the presence of intracranial injury whether there is a linear fracture or not (Burns et al., 2016).
Comminuted fractures consist of multiple associated linear fractures. They usually result from intense impact, often from repeated blows against an object or ejection from a car at a high rate of speed. They may suggest child abuse.
Depressed fractures are those in which the bone is locally broken, usually into several irregular fragments that are pushed inward. The greater the depression, the higher the risk of a tear in the dura or cortical laceration. Depressed skull fractures may be associated with direct underlying parenchymal damage and should be suspected when a child’s head appears misshapen. Surgery may be needed to elevate the depressed bone fragment if there is an associated intracranial hematoma and if the depression is greater than 1 cm (0.4 inch).
Basilar fractures involve the bones at the base of the skull in either the posterior or the anterior region. The bones involved are the ethmoid, sphenoid, temporal, or occipital bones. These fractures usually result in a dural tear. Because of the proximity of the fracture line to structures surrounding the brainstem, a basal skull fracture is a serious head injury. Basilar fractures often involve frontal bone fractures. This can result in clinical features such as leakage of CSF from the nose (CSF rhinorrhea) or ear (CSF otorrhea), blood behind the tympanic membrane (hemotympanum), subcutaneous bleeding over the mastoid process that is located posterior to the ear, and subcutaneous bleeding around the orbit (Bonfield, Naran, Adetayo, et al., 2014). Meningitis, although rare, is always a potential risk with CSF leakage.
Open fractures result in a communication between the skull and the scalp or the mucosa of the upper respiratory tract. The risk of CNS infection is increased with open fractures. Compound fractures consist of a skin laceration overlying the bone fracture. Open fractures that involve the paranasal sinuses or middle ear may lead to leakage of CSF (rhinorrhea or otorrhea). Prophylactic antibiotics are recommended to prevent osteomyelitis.
Growing skull fracture is an unusual complication of head trauma. The fracture is accompanied by an underlying tear in the dura or brain injury that fails to heal properly. A leptomeningeal cyst, dilated ventricles, or herniated brain may result and cause growth of the original fracture. Most growing skull fractures occur before 30 months of age and occur in the parietal bone (Vezina, Al-Halabi, Shash, et al., 2017). Physical examination usually shows a swelling scalp and skull defect. Clinical neurologic symptoms may be delayed for months to years after the initial skull fracture and include headache, seizures, hemiparesis, and learning and intellectual disabilities (Vezina et al., 2017).
Complications
The major complications of trauma to the head are hemorrhage, infection, edema, and herniation through the brainstem. Infection is always a hazard in open injuries. Edema is related to tissue trauma. Vascular rupture may occur even in minor head injuries, causing hemorrhage between the skull and cerebral surfaces. Compression of the underlying brain produces effects that can be rapidly fatal or insidiously progressive.
Epidural hematoma
Epidural (extradural) hematoma is a hemorrhage into the space between the dura and the skull. As the hematoma enlarges, the dura is stripped from the skull; this accumulation of blood results in a mass effect on the brain, forcing the underlying brain contents downward as it expands (Fig. 46.6A). Because bleeding is generally arterial, brain compression occurs rapidly. The lower incidence of epidural hematoma in childhood is attributed to the fact that the middle meningeal artery is not embedded in the skull’s bone surface until approximately 2 years old. Therefore a temporal bone fracture is less likely to lacerate the artery. Neuroimaging studies in 210 infants and young children with isolated mild TBI showed skull fractures with extra-axial hemorrhage/no midline shift (30%), nondisplaced skull fractures (28%), and intracranial hemorrhage without fractures/midline shift (19%) (Noje, Jackson, Nasr, et al., 2019).

A) A diagram showing the coronal section of the brain illustrating the epidural or the extradural hematoma and compression of the temporal lobe through tentorial hiatus shows the accumulation of blood between the skull and the inner membranes. The labels from top to bottom read: Epidural hematoma and tentorial herniation.
B) A diagram showing the coronal section of the brain illustrating the subdural hematoma, where the blood is accumulated between the skull and inner brain membranes producing a compression on the brain. The labels from top to bottom read: subdural hematoma and tentorial herniation.
Child abuse accounts for a significant number of cases of epidural hematomas in infants and children, whereas motor vehicle accidents account for most epidural hematomas in adolescents.
Because bleeding is generally arterial, brain compression occurs rapidly. Most often the expanding hematoma is located in the parietal and temporal regions (Teichert, Rosales, Lopes, et al., 2012), which forces the medial portion of the temporal lobe under the edge of the tentorium, where it places pressure on nerves and blood vessels. Pressure on the arterial supply and venous return to the reticular formation causes loss of consciousness; pressure on CN III produces dilation and (later) fixation of the ipsilateral pupil. Pressure on the fibers of the pyramidal tract is evidenced by contralateral weakness or paralysis and increased deep tendon reflexes. Extreme pressure may cause brain herniation and death. Expanding epidural hemorrhages may be better tolerated in young children with open sutures that allow for expansion of the skull. In addition, young children have larger subarachnoid and extracellular spaces, which provide space for the expanding hematoma without compression on the brain parenchyma.
The classic clinical picture of an epidural hemorrhage is a lucid interval of minutes to hours followed by rapidly altered mental status, then loss of consciousness or coma due to blood accumulation in the epidural space and compression of the brain. The child may be seen with varying degrees of impaired consciousness, depending on the severity of the traumatic injury. Common symptoms in a child with no neurologic deficit are irritability, headache, and vomiting. In infants younger than 24 months of age, common symptoms are scalp swelling, irritability, and lethargy. They may also have seizures, reduced oral intake, and increasing head circumference (Sellin, Moreno, Ryan, et al., 2017).
An epidural hematoma can be detected by an initial CT scan. If the severity of the child’s symptoms is not recognized, herniation and death will result. Cushing triad (systemic hypertension, bradycardia, and respiratory depression) is a late sign of impending brainstem herniation.
Subdural hematoma
A subdural hematoma is a hemorrhage between the dura and the arachnoid membrane that overlies the brain and the subarachnoid space. The hemorrhage may be from two sources: (1) tearing of the veins that bridge the subdural space and (2) hemorrhage from the cortex of the brain caused by direct brain trauma (see Fig. 46.6B). Subdural hematomas are much more common than epidural hematomas in infants and children.
Unlike epidural hemorrhage, which develops inwardly against the less-resistent brain tissue, subdural hemorrhage tends to develop more slowly and spreads thinly and widely, crossing cranial sutures, until it is limited by the dural barriers: the falx and tentorium. The small subdural space and the dura, which is firmly attached to the skull in this area, are highly vulnerable to increased ICP.
Subdural hematoma is fairly common in infants. Most often it is the result of assaults or violent shaking. The caregiver’s response to infant crying, often perceived as inconsolable, is an important risk factor (Barr, 2014). In neonates, subdural hematoma can be a consequence of labor and delivery. Subdural hemorrhage can cause either acute or chronic subdural hematoma. Acute subdural hematoma may be associated with contusions or lacerations and develops within minutes or hours of injury. Chronic subdural hematoma is more common. The clinical course and manifestations vary depending on the damage sustained by the brain and the child’s age.
Presenting signs of acute hematoma include irritability, vomiting, increased head circumference, bulging anterior fontanel (in the infant), lethargy, coma, or seizures. In infants with open fontanels, large amounts of intracranial blood may accumulate, causing hemorrhagic shock or fever before there are any changes in the neurologic examination. Retinal hemorrhages and skull and skeletal fractures are suggestive of physical abuse. An infant who has an altered LOC and in whom the CT scan shows subarachnoid hemorrhage or subdural hematoma may have been physically abused. A child with a GCS of 12 or less or a decrease in GCS score by 2 or more points requires emergency consultation with the neurosurgeon (Huang, Bi, Abd-El-Barr, et al., 2016).
Closely observe older children for signs of neurologic deterioration, including altered mental status, vomiting, lethargy, and signs of increased ICP. Hemiparesis, hemiplegia, and anisocoria (unequal pupils) are signs of brainstem compression and require emergency treatment targeted at decreasing ICP. The surgical management of subdural hematomas depends on the physical examination, size of the hematoma, and presence of other abnormalities on the CT scan. Not all children require surgery or are candidates for surgery. Various surgical options to treat subdural hematomas include transfontanel percutaneous aspiration, subdural drains, placement of burr hole, or craniotomy (Huang et al., 2016).
Other hemorrhagic lesions
A subarachnoid hemorrhage is bleeding within the subarachnoid space, which is normally filled with CSF. Nontraumatic intracranial hemorrhages are rare in children. The most common causes of spontaneous intracranial hemorrhage in children are arteriovenous malformations and fistulas and brain tumors (Ding, Starke, Kano, et al., 2017). Sudden onset of a severe headache, headaches occurring out of sleep, first-time seizure, and abnormal neurologic examination are symptoms that require evaluation including neuroimaging (Blume, 2017).
Cerebral edema
Some degree of brain edema is expected after craniocerebral trauma and often accompanies any of the previously mentioned disorders. Cerebral edema peaks at 24 to 72 hours after injury and may account for changes in a child’s neurologic status. Cerebral edema associated with TBI may be a result of two different mechanisms: cytotoxic edema or vasogenic edema. Cytotoxic edema is a result of direct cell injury and is caused by intracellular swelling. In many cases the brain cells are irreversibly damaged. Vasogenic edema is due to increased permeability of capillary endothelial cells, resulting in increased intracellular fluid. In vasogenic edema the nerve cells are not primarily injured. Either mechanism can result in increased ICP as a result of increased intracranial volume and changes in CBF as a result of loss of autoregulation and/or hypercapnia or hypoxia. Children at risk for deterioration can be identified by abnormalities seen on noncontrast CT scans.
Sequelae of traumatic brain injury
Postconcussion syndrome (PCS) is a sequela to brain injury with or without loss of consciousness. Concussions usually resolve in 1 to 3 weeks without complications. Up to a third of children may have ongoing somatic, behavioral, cognitive, and psychological symptoms, including headaches, visual and balance problems, difficulty concentrating, irritability, and changes in their sleep patterns (Morgan, Zuckerman, Lee, et al., 2015). The pathophysiology of these symptoms is unclear. When these symptoms continue for more than 4 weeks after the concussion, the term postconcussion syndrome (PCS) is used (Zemek, Barrowman, Freedman, et al., 2016). Risk factors for PCS in youth athletes include a personal or family history of mood disorders and other psychiatric illnesses and migraines (Morgan et al., 2015). Previously, concussion treatment guidelines recommended cognitive and physical rest as a path to recovery. However, recent studies have found that early participation in physical activity is significantly likely to prevent the development of PCS (Grool, Aglipay, Momoli, et al., 2016).
Posttraumatic headaches, one of the most common symptoms after mild TBI, may occur within 1 week to 3 months after a mild TBI. They occur in 25% to 75% of individuals and are most commonly classified as migraines (Kuczynski, Crawford, Bodell, et al., 2013). Posttraumatic headaches are treated based on the primary headache type: migraine or tension/chronic headache (Kacperski & Arthur, 2016).
Posttraumatic seizures occur in a number of children who survive a head injury, often within 24 hours, but they also can occur sometime after the injury (Rumalla, Smith, Letchuman, et al., 2018). Risk factors for seizures include preexisting comorbidities, shaken infant syndrome, subdural hematoma, closed-type injury, and changes in LOC (Rumalla et al., 2018).
Hydrocephalus may develop after subarachnoid hemorrhage or infection. Normal pressure hydrocephalus can be a complication of TBI. In infants, signs and symptoms include rapidly increasing head circumference, irritability, refusal to feed, and sleepiness. The clinical signs and symptoms in children include changes in personality, developmental regression, ataxia, and incontinence. These signs are also seen during posttraumatic amnesia, making early recognition of this syndrome difficult. Focal deficits, including optic atrophy, CN palsies, motor deficits, DI, or aphasia, may be seen. The type of residual effect depends on the location and nature of the trauma.
Diagnostic evaluation
A detailed health history, both past and present, is essential in evaluating the child with head trauma. Certain disorders such as drug allergies, hemophilia, diabetes mellitus, or epilepsy may produce similar symptoms. Even a minor traumatic injury can aggravate a preexisting disease process, thereby producing neurologic signs out of proportion to the injury.
After a minor injury, initial unconsciousness (if present) is brief. The child ordinarily exhibits a transient period of confusion, somnolence, and listlessness; this period is most often accompanied by irritability, pallor, and one episode of vomiting. A severe head injury requires immediate evaluation and treatment. Because head injuries are often accompanied by injuries in other areas (e.g., spine, viscera, extremities), the examination is performed with care to avoid further damage. Box 46.3 lists manifestations of head injury.
Initial assessment
Priorities in the initial phase in the care of a child with a head injury include assessment of the CAB; neurologic examination focusing on mental status, papillary responses, and motor responses; and assessment for spinal cord injury. The assessment is carried out quickly in relation to vital signs (see Emergency Treatment box).
Ocular signs such as fixed, dilated, and unequal pupils; fixed and constricted pupils; and pupils that are poorly reactive or unreactive to light and accommodation indicate increased ICP or brainstem involvement. It is important to remain with the patient who demonstrates fixed and dilated pupils because these are ominous signs often associated with impending respiratory arrest. Dilated, nonpulsating blood vessels indicate increased ICP before the appearance of papilledema. Retinal hemorrhages often occur with acute head injuries, specifically with shaken baby syndrome.
Funduscopic examination should be performed routinely to detect retinal hemorrhages in a child with CNS trauma. Vestibuloocular symptoms such as diplopia, dizziness, motion sensitivity, eye-tracking and eye-focusing problems, photosensitivity, and visual inattention may develop (Ellis, Cordingley, Vis, et al., 2015). Transient vision loss may occur after mild head trauma but may not be obvious in children unless this diagnosis is evaluated. Theories of possible causes are vasospasm or localized cerebral edema.
Less urgent but important assessments include examination of the scalp for lacerations, widely separated sutures, and the size and tension of fontanels, which indicate intracranial hemorrhage or rapidly developing cerebral edema. Scalp lacerations may require surgical intervention. A significant amount of blood loss can occur from scalp lacerations. CT scan may be necessary to evaluate possible skull fractures and acute intracranial hemorrhage (Ryan, Jaju, Ciolino, et al., 2016).
A documented accurate assessment of clinical signs provides baseline information. Serial evaluations, preferably by a single observer, help detect changes in neurologic status. Alterations in mental status, evidenced by increased difficulty in rousing the child, mounting agitation, development of focal neurologic signs, or marked changes in vital signs, usually indicate extension or progression of the basic pathologic process.
Evaluation of reflexes provides information about cerebral and pyramidal involvement, although transient abnormalities of the primitive reflexes and Babinski sign may be present in children with mild head trauma. Conscious, cooperative children are examined for cerebellar signs such as ataxia and dysmetria. Children may display unsteadiness, clumsiness, or tremor with intentional movement after head injury. Temperature may be moderately elevated for 1 or 2 days after an initial mild hypothermia after injury. A persistent fever may indicate subarachnoid hemorrhage or infection.
Special tests
After a thorough clinical examination, a variety of diagnostic tests are helpful in providing a more definitive diagnosis of the type and extent of the trauma. A hematocrit and urinalysis are typically done. Serum electrolytes and glucose may also be measured in children with severe head injuries; hyperglycemia and disseminated intravascular coagulation are associated with a poor prognosis. The severity of a head injury may not be apparent on clinical examination of the child but detectable on a CT scan. Whenever the child has a history consistent with a serious head injury (as with an unrestrained occupant in a severe motor vehicle accident or a fall from higher than their own height), it is important to perform a scan even if the child initially appears alert and oriented. All children with head injuries who have any alteration of consciousness, headache, vomiting, skull fracture, seizure, or predisposing medical condition should undergo a diagnostic evaluation that includes CT scanning.
MRI may be done to further assess cerebral edema or other structural brain abnormalities. A neurodevelopmental assessment after early head injury may be useful in documenting cognitive impairment. Skull radiographs are of little benefit in diagnosing skull fractures. Other radiographic tests may be indicated, depending on the severity or cause of the trauma. Electroencephalography is not helpful for diagnosis of a head injury but is useful for defining seizures and looking for subclinical seizures, which can impair consciousness (Gainza-Lein, Sanchez-Fernandez, & Loddenkemper, 2017). Lumbar puncture is rarely used for craniocerebral trauma and is contraindicated in the presence of increased ICP because of the possibility of herniation.
Therapeutic management
Most children with mild TBI who have not lost consciousness can be cared for and observed at home after careful examination reveals no serious intracranial injury. The nurse should give parents both verbal and written instructions of signs and symptoms that warrant concern and the need for reevaluation. These include persistent or worsening headaches, vomiting, change in mental status or behavior, unsteady gait, or seizure. The child should have a physical examination 1 or 2 days after the injury. The manifestations of epidural hematoma in children do not generally appear until 24 hours or more after injury.
Maintaining contact with parents for continued observation and reevaluation of the child, when indicated, facilitates early diagnosis and treatment of possible complications from head injury, such as hematoma, cerebral edema, and posttraumatic seizures. Children are generally hospitalized for 24 to 48 hours of observation if their family lives far from medical facilities or lacks transportation or a telephone, which would provide access to immediate help. Other circumstances, such as language or other communication barriers or even emotional trauma, may hinder learning and make it difficult for families to feel confident caring for their child at home.
Children with severe injuries, those who have lost consciousness for more than a few minutes, and those with prolonged and continued seizures or other focal or diffuse neurologic signs must be hospitalized until their condition is stable and their neurologic signs have diminished. The child is maintained on nothing-by-mouth (NPO) status or restricted to clear liquids (if able to take fluids by mouth) until it is determined that vomiting will not occur. IV fluids are indicated in the child who is comatose, displays dulled sensorium, or is persistently vomiting.
The volume of IV fluid is carefully monitored to minimize the possibility of overhydration in case of SIADH and cerebral edema. However, damage to the hypothalamus or pituitary gland may produce DI, with its accompanying hypertonicity and dehydration. Fluid balance is closely monitored by daily weight, strict intake and output measurement, and serum osmolality (to detect early signs of water retention).
Sedating drugs are usually withheld in the acute phase. Headache is usually controlled with acetaminophen, although opioids may be needed. Antiepileptics are used for seizure control. Antibiotics are administered if there are lacerations or penetrating injuries. Prophylactic tetanus toxoid is given as appropriate. Cerebral edema is managed as described for the unconscious child. Hyperthermia is controlled with tepid sponges or a hypothermia blanket.
Surgical therapy
Approximately 10% to 30% of pediatric head traumas will result in skull fractures. Because of the greater capacity of a child’s skull fracture to heal, conservative nonsurgical management is often adequate. Children hit in the head or who have TBI as a result of a motor vehicle accident are more likely to require surgical intervention, especially if the frontal bones have been fractured (Bonfield et al., 2014).
Scalp lacerations are sutured after careful examination of underlying bone. The use of topical lidocaine, adrenaline, and tetracaine (LAT) or lidocaine, epinephrine, and tetracaine (LET) provides noninvasive, effective anesthesia for suturing, particularly when combined with consultation from and the bedside presence of a child life specialist (Martin, 2017).
Depressed fractures require surgical reduction and removal of bone fragments. Torn dura is also sutured. A skull fracture depressed more than the thickness of the skull or an intracranial hematoma that causes more than a 5-mm (0.2-inch) midline shift is an indication for surgery. Direct pressure should never be applied to a depressed skull fracture. Parents should be advised that painful hardware and wound infections may need further surgical intervention. Parents and other caregivers must be taught the importance of meticulous hand washing after surgical repair of a skull fracture.
Prognosis
The outcome of craniocerebral trauma depends on the extent of injury and complications. Neurologic, cognitive, emotional, and behavioral symptoms can result in significant impairment. They may not present until the child is older and preparing to reach certain developmental milestones (Babikian, Merkley, Savage, et al., 2015). These symptoms can become chronic and include epilepsy, attention-deficit/hyperactivity disorder, and learning or psychiatric disorders. Children with learning and behavior problems before their head trauma are more likely to suffer these consequences (Beauchamp & Anderson, 2013). More than 90% of children with concussions or simple linear fractures recover without symptoms after the initial period.
Children may be more vulnerable than adults to long-term cognitive and behavioral dysfunction after diffuse brain injury. Contrary to what was previously thought about “brain plasticity,” evidence now indicates that children’s brains may be especially vulnerable to early injury due to their ongoing maturation processes, which can be disrupted by head trauma (Babikian et al., 2015). Parents of children who have suffered TBI should be advised to seek evaluation and treatment sooner rather than later if any of these symptoms present. TBI is recognized as a disability that may qualify a child for special education services under the Individuals with Disabilities Education Act (IDEA) of 1990.
True coma (i.e., not obeying commands, eyes closed, and not speaking) usually does not last for more than 2 weeks. A child’s eventual outcome can range from brain death to a persistent vegetative state to complete recovery. However, even the best recovery may be associated with personality changes, including mood lability and loss of confidence, impaired short-term memory, headaches, and subtle cognitive impairments. In general, 90% of the long-term neurologic outcome has been achieved within 6 months to 1 year after the injury.
Nursing care management
The hospitalized child requires careful neurologic assessment and evaluation repeated as frequently as every 15 minutes to establish a correct diagnosis, identify signs and symptoms of increased ICP, determine clinical management, and prevent many complications. The goals of nursing management of the child with a head injury are to maintain adequate ventilation, oxygenation, and circulation; to monitor and treat increased ICP; to minimize cerebral oxygen requirements; and to support the child and family during recovery. (See Quality Patient Outcomes box.)
The child is placed on bed rest, usually with the head of the bed elevated slightly and the head in midline position. Appropriate safety measures, such as side rails kept up and seizure precautions, are implemented. If the child is extremely restless, hard surfaces may be padded and restraints used to prevent further injury. Individualize care according to the child’s specific needs.
A key nursing role is to provide sedation and analgesia for the child. The conflict between the need to promote the child’s comfort and relieve anxiety versus the need to assess for neurologic changes presents a dilemma. Both goals can be achieved with close observation of the child’s LOC and response to analgesics (using a pain assessment record) and effective communication with the provider. Decreasing restlessness after administration of an analgesic most likely reflects pain control rather than a decreasing LOC. (See Chapter 5, Pain Assessment and Pain Management.)
Children may be restless and irritable, but more often their reaction is to fall asleep when left undisturbed. A quiet environment can help reduce restlessness and irritability. Bright lights are irritating. This often makes checking the ocular responses more difficult and aggravating to the child.
Frequent examinations of vital signs, neurologic signs, and LOC are extremely important nursing observations. When possible, they should be performed by a single observer to better detect subtle changes that may indicate worsening of neurologic status. Pupils are checked for size, symmetry, reaction to light, and accommodation. Unless there is brainstem involvement, vital signs generally return to normal after the initial changes seen after injury.
The most important nursing observation is assessment of the child’s LOC. In the progression of an injury, alterations in consciousness appear earlier than alterations in vital signs or focal neurologic signs (see evaluation of responsiveness later in the chapter). Frequent examinations of alertness are fatiguing to the child; the child often desires to fall asleep, which may be confused with depressed consciousness. It is not uncommon to observe ocular divergence through the partially closed eyelids.
Observations of position and movement provide additional information. Note any abnormal posturing and whether it occurs continuously or intermittently. Questions nurses might ask include the following:
- • Are the child’s hand grips strong and equal in strength?
- • Are there any signs of extension or flexion posturing?
- • What is the child’s response to auditory and physical stimulation?
- • Is movement purposeful, random, or absent?
- • Are movement and sensation equal on both sides or restricted to one side only?
The child may report a headache or other discomfort. The child who is too young to describe a headache may be fussy and resist being handled. The child who suffers from vertigo often vigorously resists being moved from a position of comfort. Forcible movement causes the child to vomit and display spontaneous nystagmus. Seizures are relatively common in children at the time of head injury and may be of any type. Carefully observe any seizure activity and describe it in detail. Children in postictal states are more lethargic, with sluggish pupils.
Document drainage from any orifice. Bleeding from the ear suggests the possibility of a basal skull fracture. Clear nasal drainage is suggestive of an anterior basal skull fracture. Observe the amount and characteristics of the drainage.
Head trauma is often accompanied by other undetected injuries; therefore any bruises, lacerations, or evidence of internal injuries or fractures of the extremities are noted and reported. Associated injuries are evaluated and treated appropriately.
The child with a normal LOC is usually allowed clear liquids unless fluid is restricted. If the child has an IV infusion, it is maintained as prescribed. The diet is advanced to that appropriate for the child’s age as soon as the condition permits. Intake and output are measured and recorded with attention to the development of constipation. Any incontinence of bowel or bladder is noted for the child who has been toilet trained.
Assessment for unusual behavior can be made only in relation to the child’s typical behavior. For example, urinary incontinence during sleep would be of no consequence in a child who routinely wets the bed but would be highly significant for one who is always dry. Parents are invaluable resources in evaluating objective behaviors of their children. Information obtained from parents at or shortly after admission is essential in evaluating the child’s behavior (e.g., the ease with which the child is roused normally, the usual sleeping position, how much the child sleeps during the day, the child’s motor activities [rolling over, sitting up, climbing], hearing and visual acuity, appetite, and manner of eating [spoon, bottle, cup]). Documentation of the child’s baseline developmental and behavioral level is crucial. There is less concern about a child who falls asleep several times during the day if this is consistent with the child’s usual behavior.
When the child is discharged, advise the parents of probable posttraumatic symptoms they may observe, such as behavioral changes, sleep disturbances, phobias, and seizures. Parents should be taught seizure first aid. They should understand observations they need to make and when and how to contact the provider or health facility in case the child develops any unusual signs or symptoms. Emphasize the importance of follow-up evaluation.
Family support
The emotional and educational support of the family presents a challenge. Witnessing the parents’ grief and helplessness on seeing their child in an intensive care unit connected to monitoring equipment and in an altered state evokes empathy. The nurse can encourage the family to be involved in the child’s care, to bring in familiar belongings, or to make a recording of familiar voices and sounds. Parents may need a demonstration on how to touch or cuddle their child and may want to talk about their grief. The nurse listens attentively, reinforces what is being done to assist the child, and directs parents toward signs and symptoms of recovery to instill hope without promises. Honesty and kindness, along with consistent and competent care, help families through this difficult time.
Rehabilitation
Rehabilitation and management of the child with permanent brain injury are essential aspects of care. Rehabilitation begins as soon as possible and usually involves the family and a rehabilitation team. The nurse makes a careful assessment of the child’s capabilities and limitations and implements appropriate interventions to maximize the residual capacities. The Brain Injury Association of America* provides information and listings of rehabilitation services and support groups throughout the country.
The child with a disability resulting from head trauma requires functional assessment of his or her physical, cognitive, emotional, and social levels. The child has experienced separation, pain, sensory deprivation and overload, changes in circadian cycle, and fear of the unknown. Recovery and transition require new coping strategies at the same time that regressive and acting-out behaviors may start. Parents and children need honest communication for decision making.
Rehabilitation is recommended when the child is making progress and no longer requires acute care hospitalization but continues to require daily therapies to return to his or her premorbid functional level. Children are more likely to continue in outpatient rehabilitation if they have had an inpatient assessment of their rehabilitation needs (Jimenez, Symons, Wang, et al., 2016). The Rancho Los Amigos Scale provides a systematic assessment of the progress that a child with a severe head injury may achieve.
Pediatric rehabilitation focuses on the child’s strengths and needs. The rehabilitation team should include physical medicine; rehabilitation nursing; nutritional counseling; physical, occupational, and speech therapy; special education; and psychological, neuropsychological, child life, and social services support. Before the child’s transfer, the hospital team should provide a detailed care plan of the child’s needs and abilities, especially communication skills, and a description of the child’s usual schedule, nursing care interventions, and the family’s concerns and needs. To augment the care plan, a video introducing the child and family and showing any unique aspects of their care can be sent to the rehabilitation center.
Prevention
Preventive strategies are underused in almost all cases of accidental childhood injury. Head injuries occur in the most serious accidents—especially motor vehicle accidents, sports, and falls.
Strides are being made in the prevention of secondary brain tissue damage after the initial head injury in children. This secondary injury is caused by altered CBF that results in ischemia, hypoxia, and eventually the death of brain cells (Popernack, Gray, & Reuter-Rice, 2015). Studies are ongoing in both humans and animals using therapeutic hypothermia, glucagon, blood pressure medications, and antioxidants (Toth, Szarka, Farkas, et al., 2016). The roles of calcium, oxyradicals, and prostaglandins are being investigated.
However, the greatest benefit lies in prevention of head injuries. Nurses can exert a valuable influence on behalf of children through education. Accidents that are preventable occur because unnecessary risks go unchecked. Inadequate supervision combined with a child’s natural sense of curiosity and exploration can lead to lethal results. Nurses are in the unique position of influencing caregivers in terms of growth and development. The use of car seats; seat belts in strollers and feeding chairs; and helmets for biking, skateboarding, and other sports has been shown to reduce both the number and the severity of head injuries in children (Gaw, Chounthirath, & Smith, 2017).
Studies of ways to prevent abusive head trauma are ongoing. Promising interventions include teaching parents about infant crying and ways to cope with it (Lopes & Williams, 2016). Infants who have been hospitalized after birth have an increased risk of being abused after discharge. Neonatal nurses have an important role to play in teaching parents of these children about child abuse prevention.
Public education coupled with legislative support can aid in the prevention of childhood injuries. Research has shown that increased household income can prevent abusive head trauma (Klevens, Schmidt, Luo, et al., 2017).
Submersion injury
Drowning is one of the major causes of unintentional injury-related death in children ages 1 to 19 years. In children ages 1 to 4 years, it is the leading cause of unintentional injury-related death (Gilchrist & Parker, 2014). The term near-drowning is no longer used; instead, the term submersion injury should be used until the time of drowning-related death.
Most cases of submersion injury are accidental, usually involving children who are helpless in water, such as inadequately attended children in or near swimming pools or infants in bathtubs; small children who fall into ponds, streams, and flooded excavations, usually near home; occupants of pleasure boats who fail to wear life preservers; children who have diving accidents; and children who are able to swim but overestimate their endurance. Accidental submersion injury occurs predominantly in males, toddlers, and African Americans (Gilchrist & Parker, 2014).
Submersion injury can take place in any body of water. Children less than 1 year of age are most likely to have a submersion injury in a bathtub, whereas top-heavy toddlers fall headfirst into a pail of water and are unable to free themselves (Xu, 2014). Preschoolers are at risk for injury in swimming pools, and school-age children and adolescents are most commonly at risk in natural bodies of water such as lakes, ponds, and rivers (Xu, 2014). The suction created at the outlet of pools, hot tubs, or whirlpool spas is strong enough to trap even larger children underwater. Submersion injury as a form of fatal child abuse also occurs. Homicidal submersion injuries are not witnessed, usually occurring in the home, and the victims are either infants or toddlers.
Pathophysiology
Physiologically, most organ systems are affected, especially the pulmonary, cardiovascular, and neurologic systems. The major pulmonary changes that occur in submersion injury are directly related to the length of submersion (regardless of the type and amount of fluid aspirated), the victim’s physiologic response, and the development and degree of immersion hypothermia. Cerebral hypoxia is a major component of morbidity and mortality in these individuals. Therefore early and aggressive resuscitation is imperative.
Physiologic factors in submersion injuries are hypothermia, aspiration, and hypoxia. The temperature of the liquid plays an important role. Cold water decreases metabolic demands and activates the diving reflex, which causes blood to be shunted away from the periphery to vital organs (i.e., the brain and heart). Hypothermia occurs rapidly in infants and children, partly because of their large surface area relative to size and partly as a result of the cold water itself. Profound hypothermia is usually evidence of lengthy submersion. Prolonged submersion in cold liquids can impair cognition, coordination, and muscle strength that ultimately result in loss of consciousness, decreased cardiac output, and cardiac arrest (Thomas & Caglar, 2020). Submersion in cold water had previously been thought to be somewhat neuroprotective, but it is not (Quan, Mack, & Schiff, 2014).
Submerged children struggle initially to stay above water, and often breath-holding leads to air hunger. Reflex inspiration eventually occurs, which leads to aspiration (Thomas & Caglar, 2020). Fluid is quickly absorbed in the pulmonary circulation, resulting in pulmonary edema, atelectasis, and airway spasm. Hypoxia is the primary problem because it results in global cell damage, with different cells tolerating variable lengths of anoxia. Neurons, especially cerebral cells, sustain irreversible damage after 4 to 6 minutes of submersion. The heart and lungs can survive up to 30 minutes. Regardless of the amount of water aspirated, the victim suffers arterial hypoxemia (resulting from atelectasis and shunting of blood through the nonventilated alveoli), combined respiratory acidosis (resulting from retained carbon dioxide), and metabolic acidosis (caused by buildup of acid metabolites because of anaerobic metabolism). Although electrolyte imbalances are contributing factors, they are not the major causes of morbidity and mortality. The pathologic events are directly related to the duration of submersion. Approximately 10% of submersion injury victims die without aspirating fluid but succumb from acute asphyxia as a result of prolonged reflex laryngospasm.
Aspiration of fluid occurs in most submersion injuries. The aspirated fluid results in pulmonary edema, atelectasis, airway spasm, and pneumonitis, which aggravates hypoxia. Submersion in salt water is associated with better outcomes than submersion in fresh water, although duration of the submersion is the main factor that predicts outcome (Quan, Bierens, Lis, et al., 2016).
Clinical manifestations
Clinical manifestations are directly related to the duration of loss of consciousness and neurologic status after rescue and resuscitation.
Therapeutic management
With rapid treatment, some children can be saved. Resuscitative measures should begin at the scene, and the victim should be transported to the hospital with maximum ventilatory and circulatory support. In the hospital, intensive pulmonary care is implemented and continued according to the patient’s needs.
In general, management of the victim with a submersion injury is based on the degree of cerebral insult. The first priority is to restore oxygen delivery to the cells and prevent further hypoxic damage. A spontaneously breathing child does well in an oxygen-enriched atmosphere; the more severely affected child requires endotracheal intubation and mechanical ventilation. Blood gases and pH are monitored at frequent intervals as a guide to oxygen, fluid, and electrolyte therapies. Rewarming the hypothermic patient is initiated. Seizures may occur due to hypoxia and cerebral edema. Seizures result in increased cerebral oxygen consumption. Therefore it is imperative to aggressively control seizure activity. In addition, blood glucose should be monitored; both hypoglycemia and hyperglycemia are harmful to the brain.
All children who have a submersion injury should be hospitalized for observation. Although some children do not appear to have sustained adverse effects from the event, respiratory compromise or cerebral edema may occur within 24 hours after the incident. In the acute recovery period, fever should be prevented, although prophylactic antibiotics are not recommended. Aspiration pneumonia is a common complication that occurs approximately 48 to 72 hours after the episode. Bronchospasm, alveolar-capillary membrane damage, atelectasis, abscess formation, and acute respiratory distress syndrome are other complications that occur after aspiration of fluid.
Prognosis
Children who have submersion injuries usually have a good outcome with no or mild neurologic sequelae, no severe neurologic disabilities, and rarely morbidity (Thomas & Caglar, 2020). The best predictors of a good outcome are length of submersion less than 5 minutes and the presence of sinus rhythm, reactive pupils, and neurologic responsiveness at the scene. The worst outcomes are for children submerged for more than 10 minutes and unresponsive to advanced life support within 25 minutes. All children without spontaneous, purposeful movement and normal brainstem function 24 hours after sustaining a submersion injury suffered severe neurologic deficits or death (Thomas & Caglar, 2020).
Nursing care management
Nursing care depends on the child’s condition. A child who survives may need intensive respiratory nursing care with attention to vital signs, mechanical ventilation or tracheostomy, blood gas determination, chest physiotherapy, and IV infusion. Often the child has sustained a hypoxic insult and requires the same care as an unconscious child.
A difficult aspect in the care of the child who sustained a submersion injury is helping the parents cope with the grief, guilt, and anger reactions. Given the magnitude of the event, parents need repeated assurance that everything possible is being done to treat their child.
Nurses often have difficulty relating to the parents if obvious neglect has precipitated the accident and subsequent problems; it is important for those who care for these children and their families to assess their own feelings about the situation, in addition to assessing the family’s coping abilities and resources. Caring for victims of a submersion injury and their families requires the nurse to be sensitive to the needs of the child and the family and to recognize his or her own reactions and emotions.
Prevention
Most submersion injuries are preventable. The most common cause of submersion injury in infants and young children is inadequate adult supervision, including a momentary lapse of supervision. Parents are often unaware that they must be within arm’s reach and constantly supervising without being distracted (Thomas & Caglar, 2020). Children with known risk factors such as epilepsy and autism require eyes-on surveillance. In general, children are not developmentally ready for formal swimming lessons until their fourth birthday. All parents and swimming pool owners should be familiar with basic cardiopulmonary resuscitation (CPR) because rapid, basic CPR is one of the keys to improving outcomes (Tobin, Ramos, Pu, et al., 2017). Water safety and survival training should be required for all school-age children. Pool covers and fencing on all sides and the presence of lifeguards can prevent accidents.
Nurses can be active advocates in their communities. Nurses are also in a position to emphasize the importance of adequate adult supervision when children are around any body of water and should include the necessity of the adult not engaging in distracting activities.
Intracranial infections
The nervous system is subject to infection by the same organisms that affect other organs of the body. However, the nervous system is limited in the ways in which it responds to injury. Laboratory studies are needed to identify the causative agent. The inflammatory process can affect the meninges (meningitis) or brain (encephalitis).
Meningitis can be caused by a variety of organisms, but the three main types are (1) bacterial, or pyogenic, caused by pus-forming bacteria, especially meningococci and pneumococci organisms; (2) viral, or aseptic, caused by a wide variety of viral agents; and (3) tuberculous, caused by the tuberculin bacillus. The majority of children with acute febrile encephalopathy have either bacterial meningitis or viral meningitis as the underlying cause.
Bacterial meningitis
Bacterial meningitis is an acute inflammation of the meninges and CSF. The advent of antimicrobial therapy has had a marked effect on the course and prognosis. The introduction of conjugate vaccines against Haemophilus influenzae type b (Hib vaccine) in 1990 and Streptococcus pneumoniae (pneumococcus) in 2000 has led to dramatic changes in the epidemiology of bacterial meningitis (see Evidence-Based Practice box). Currently, Hib infection has been virtually eradicated among young children in areas where the Hib vaccine is administered routinely. By 2013 there were fewer than 40 cases of Hib disease in children younger than 5 years old (Centers for Disease Control and Prevention, 2016). Before the vaccine, Hib was responsible for almost half of all cases of bacterial meningitis, but now it is the least likely pathogen to cause meningitis (Castelblanco, Lee, & Hasbun, 2014). Since the introduction of widespread vaccination for S. pneumoniae, the incidence of pneumococcal meningitis in children in the United States has decreased, but it remains the most common cause of meningitis in children ages 3 months to 11 years (Castelblanco et al., 2014). It is also the most likely to result in death (Heckenberg, Brouwer, & Van de Beek, 2014).
Etiology
A variety of bacterial agents can cause bacterial meningitis. Since the introduction of vaccinations against most common causes of community-acquired pathogens, the incidence of bacterial meningitis has declined precipitously. The leading causes of neonatal meningitis are group B streptococcus (GBS) and Escherichia coli (Ku, Boggess, & Cohen-Wolkowiez, 2015).
Meningococcal meningitis is the only type readily transmitted by droplet infection from nasopharyngeal secretions and so has the potential to occur in outbreaks (Vetter, Baxter, Denizer, et al., 2016). Before the development of a vaccine, it occurred predominantly in school-age children and adolescents; currently it is most common in children younger than 12 months old, with a secondary peak in incidence in 16 to 23 year olds (Centers for Disease Control and Prevention, 2017b). Meningitis caused by pneumococcal and meningococcal infections can occur at any time but is more common in late winter and early spring.
Maternal factors, such as premature rupture of fetal membranes and maternal infection during the last week of pregnancy, are major causes of neonatal meningitis. It is a devastating disease with significant morbidity and mortality. Vaccination of pregnant women is being explored as a way to protect infants from meningitis (Jones, Munoz, Spiegel, et al., 2016). Children who survive neonatal meningitis are 10 times more likely to have moderate to severe disabilities than those who have not had meningitis (Ku et al., 2015). The incidence of early-onset GBS meningitis has been reduced by more than 70% with the adoption of antenatal screening and administration of intrapartum prophylactic antibiotics (Ku et al., 2015).
Risk factors for children developing meningitis include lack of immunization to the specific pathogen; recent exposure to someone with invasive Neisseria meningitidis or Hib disease; penetrating head trauma; cochlear implant devices; and anatomic defects such as midline facial defects, inner ear fistulas, or recent placement of a ventricular shunt (Swanson, 2015).
Pathophysiology
The most common route of infection is vascular dissemination from a focus of infection elsewhere. For example, organisms from the nasopharynx invade the underlying blood vessels, cross the blood-brain barrier, and multiply in the CSF. Invasion by direct extension from infections in the paranasal and mastoid sinuses is less common. Organisms also gain entry by direct implantation after penetrating wounds, skull fractures that provide an opening into the skin or sinuses, lumbar puncture or surgical procedures, anatomic abnormalities such as spina bifida, or foreign bodies such as an internal ventricular shunt or an external ventricular device. Once implanted, the organisms spread into the CSF, by which the infection spreads throughout the subarachnoid space.
The infective process is like that seen in any bacterial infection: inflammation, exudation, white blood cell accumulation, and varying degrees of tissue damage. The brain becomes hyperemic and edematous, and the entire surface of the brain is covered by a layer of purulent exudate that varies with the type of organism. For example, meningococcal exudate is most marked over the parietal, occipital, and cerebellar regions; the thick, fibrinous exudate of pneumococcal infection is confined chiefly to the surface of the brain, particularly the anterior lobes; and the exudate of streptococcal infections is similar to that of pneumococcal infections but thinner. As infection extends to the ventricles, thick pus, fibrin, or adhesions may occlude the narrow passages and obstruct the flow of CSF.
Clinical manifestations
The clinical manifestations of acute bacterial meningitis depend to a large extent on the child’s age. The type of organism, the effectiveness of therapy for antecedent illness, and whether it occurs as an isolated entity or as a complication of another illness or injury also influence the clinical manifestation (Box 46.4).
Children and adolescents
The onset of illness may be abrupt and rapid, may develop progressively over one or several days, and may be preceded by a febrile illness. Most children with meningitis are seen with fever, chills, headache, and vomiting that are associated with or quickly followed by alterations in sensorium; however, some may present only with lethargy and irritability (Weinberg & Thompson-Stone, 2018). The child is extremely irritable and agitated and may develop seizures, photophobia, confusion, hallucinations, aggressive behavior, drowsiness, stupor, or coma.
The child resists flexion of the neck (nuchal rigidity). Kernig and Brudzinski signs are positive. Reflex responses are variable, although they show hyperactivity. (See Chapter 29, Reflexes.) The skin may be cold and cyanotic with poor peripheral perfusion.
Other signs and symptoms that are specific to individual organisms may appear. Petechial or purpuric rashes occur in 50% of cases and indicate a meningococcal infection (meningococcemia), especially when the eruption is associated with a septic shock-like state. Joint involvement is seen in meningococcal and H. influenzae infection. A chronically draining ear commonly accompanies pneumococcal meningitis. E. coli infection may be associated with a congenital dermal sinus that communicates with the subarachnoid space.
Infants and young children
Between 3 months and 2 years of age, the illness is characterized by fever or hypothermia, poor feeding, vomiting, marked irritability, restlessness, seizures, and a bulging or tense fontanel, which are often accompanied by a high-pitched cry.
Complications
The incidence of complications from acute bacterial meningitis has been significantly reduced with early diagnosis and vigorous antimicrobial therapy. If infection extends to the ventricles, thick pus, fibrin, or adhesions may occlude the narrow passages, thereby obstructing the flow of CSF and causing obstructive hydrocephalus. Subdural effusions often occur, and thrombosis may occur in meningeal veins or venous sinuses. Destructive changes may take place in the cerebral cortex, and brain abscesses may form by direct extension of the infection or by vascular dissemination. Extension of the infection to the areas of the CNs or compression necrosis from increased pressure may cause deafness, blindness, or weakness or paralysis of facial or other muscles of the head and neck.
One of the most dramatic and serious complications usually associated with meningococcal infections is meningococcal sepsis, or meningococcemia. When the onset is severe, sudden, and rapid, it is known as the Waterhouse-Friderichsen syndrome. The syndrome is characterized by overwhelming septic shock, disseminated intravascular coagulation, massive bilateral adrenal hemorrhage, and purpura. Meningococcemia requires immediate emergency treatment, hospitalization, and intensive care because of the serious sequelae that can quickly develop (Weinberg & Thompson-Stone, 2018).
Other acute complications of meningitis include SIADH, subdural effusions, seizures, cerebral edema and herniation, and hydrocephalus. Obstruction to the flow of CSF occurs during the acute phase of illness by clumping of purulent material in the drainage channels and during the chronic phase of illness by adhesive arachnoiditis or fibrotic obstruction through any of the ventricular foramina. Postmeningitic complications in neonates include ventriculitis, which results in cystic, walled-off areas of the brain with fluid accumulation and pressure.
Extension of the inflammation to CNs or compression and destruction of the nerves from ICP can produce permanent impairment of vision or hearing and other nerve palsies. CN VIII damage is usually followed by permanent deafness, the most common permanent neurologic sequela of bacterial meningitis (Weinberg & Thompson-Stone, 2018). Other long-term complications include cerebral palsy, cognitive impairments, learning disorders, attention-deficit/hyperactivity disorder, and seizures.
Hemiparesis and quadriparesis may result from damage caused by arteritis or thrombosis or other mechanisms. Behavioral changes occur in some children. Evidence indicates that psychometric and behavioral defects may be a significant concomitant sign of meningitis in childhood, although it is difficult to determine the degree to which meningitis affects the intelligence of young children. Meningitis in the neonatal period is more likely to cause lifelong impairments, including moderate to severe developmental delay, blindness, deafness, and epilepsy (Swanson, 2015).
Diagnostic evaluation
A lumbar puncture is the definitive diagnostic test. The fluid pressure is measured, and samples are obtained for culture, Gram stain, blood cell count, and determination of glucose and protein levels. The findings are usually diagnostic. Culture and sensitivity are needed to identify the causative organism. Spinal fluid pressure is usually elevated, but interpretation is often difficult when the child is crying. Sedation with fentanyl and midazolam can alleviate the child’s pain and fear associated with this procedure (see Atraumatic Care box). If there is evidence or suspicion of increased ICP, a CT scan of the head may be warranted before the procedure (Weinberg & Thompson-Stone, 2018).
The patient generally has an elevated white blood cell count, often predominantly polymorphonuclear leukocytes. The glucose level is reduced, generally in proportion to the duration and severity of the infection. The relationship between the CSF glucose and serum glucose levels is important in evaluating the glucose content of CSF; therefore a serum glucose sample is drawn approximately a half-hour before the lumbar puncture. Protein concentration is usually increased.
Blood culture is advisable for all children suspected of having meningitis if antibiotics are started before obtaining CSF. Blood culture will occasionally be positive when CSF culture is negative. Nose and throat cultures may provide helpful information in some cases.
Therapeutic management
Acute bacterial meningitis is a medical emergency that requires early recognition and immediate therapy to prevent death and avoid residual disabilities. The initial therapeutic management includes the following:
The child is usually moved to an intensive care unit for close observation. An IV infusion is started to facilitate administration of antimicrobial agents, fluids, antiepileptic drugs, and blood, if needed. The child is placed in respiratory isolation.
Drugs
Until the causative organism is identified, empiric therapy is administered. After identification of the organism, antimicrobial agents are adjusted accordingly. Signs of gastrointestinal hemorrhage or secondary infection may complicate steroid administration. Antibiotic treatment with cephalosporins demonstrates superiority for promptly sterilizing the CSF and reducing the incidence of severe hearing impairment. With increasing prevalence of antibiotic-resistant S. pneumoniae, vancomycin should be given until antibiotic susceptibility test results are available.
Nonspecific measures
Maintaining hydration is a prime concern. The patient’s condition determines whether IV fluids are needed and the type and amount of fluid. The optimum hydration involves correction of any fluid deficits and electrolyte abnormalities, followed by fluid restriction until normal serum sodium levels and no signs of increased ICP are present. If needed, measures to decrease ICP are implemented (see earlier in the chapter); however, long-term fluid restriction is not the standard of care because a lack of fluid volume can reduce blood pressure and CPP, causing CNS ischemia (Janowski & Hunstad, 2020c).
Complications, such as subdural effusion in infants and disseminated intravascular coagulation syndrome, are treated appropriately. Shock is managed by restoration of circulating blood volume and maintenance of electrolyte balance. Seizures can occur during the first few days of treatment. These are controlled with the appropriate antiepileptic drug.
Hearing loss is common. The patient should undergo auditory evaluation shortly after discharge so that audiology and speech and communication therapies can begin as soon as possible.
Lumbar puncture is carried out as needed to determine the effectiveness of therapy. The patient is evaluated neurologically during the convalescent period.
Prognosis
Less than 10% of cases of bacterial meningitis are fatal; the highest mortality rate is seen in pneumococcal meningitis and in infants younger than the age of 6 months (Janowski & Hunstad, 2020c). Prognosis is dependent in large part on the length of time between onset of illness and initiation of antibiotic therapy, rapidity of diagnosis after onset, type of organism, prolonged or complicated seizures, low CSF glucose concentration, and adequacy of therapy. Up to half of those who recover from meningitis will have some neurodevelopmental sequelae ranging from mild behavioral and learning problems to profound hearing impairment, intractable epilepsy, and significant intellectual disability (Janowski & Hunstad, 2020c).
The sequelae of bacterial meningitis occur most often when the disease occurs in the first 2 months of life and least often in children with meningococcal meningitis. The residual deficits in infants are primarily a result of communicating hydrocephalus and the greater effects of cerebritis on the immature brain. In older children the residual effects are related to the inflammatory process itself or result from vasculitis associated with the disease. Bacterial meningitis continues to cause substantial morbidity in infants and children.
Prevention
Vaccination is the foundation of prevention of CNS infections. Vaccines are available for pneumococci; types A, C, Y, and W-135 meningococci; and Hib. Routine meningococcal conjugate vaccination of children is recommended at age 11 to 12 years, with a booster at 16 years, but can be given to children 2 months to 10 years of age if they are considered at high risk (e.g., asplenia, foreign travel to high-risk areas, or present during outbreaks). Routine vaccinations for Hib and pneumococcal conjugate vaccines are recommended for all children beginning at 2 months of age.
Nursing care management
Nurses should take the necessary precautions to protect themselves and others from possible infection. Teach parents proper hand washing technique, and remind them as needed.
Keep the room as quiet as possible and environmental stimuli at a minimum because most children with meningitis are sensitive to noise, bright lights, and other external stimuli. Help the family limit the number and frequency of visitors until the child feels better. Most children are more comfortable without a pillow under their head but with the head of the bed slightly elevated. Use pillows alongside a child in a side-lying position and between the child’s knees for comfort in cases of nuchal rigidity. Avoid actions that cause pain or increase discomfort, such as lifting the child’s head. Evaluating the child for pain and implementing appropriate relief measures are important ongoing interventions. Measures are used to ensure safety because the child is often restless, disoriented, and subject to seizures. Prevention of falls is essential.
The nursing care of the child with meningitis is determined by the child’s symptoms and treatment (see Box 46.4). Observation of vital signs, neurologic signs, LOC, urinary output, and other pertinent data is carried out at frequent intervals. The child who is unconscious is managed as described previously, and all children are observed carefully for signs of the complications just described, especially increased ICP, shock, and respiratory distress. Frequent assessment of the open fontanels is needed in the infant because subdural effusions and obstructive hydrocephalus can develop as a complication of meningitis.
Administration of fluids and nourishment is determined by the child’s status. The child who is not alert and oriented is given nothing by mouth. Other children are allowed clear liquids initially and, if tolerated, progress to a diet suitable for their age. Careful monitoring and recording of intake and output are needed to determine deviations that might indicate impending shock or increasing fluid accumulation, such as cerebral edema or subdural effusion.
One of the most challenging issues in the nursing care of children with meningitis is maintaining IV infusion for the length of time needed to provide adequate antimicrobial therapy (usually 10 days). Because continuous IV fluids are usually not necessary, an intermittent infusion device is used. In some cases children who are recovering uneventfully are sent home with the device, and the parents are taught IV drug administration. (See Quality Patient Outcomes box.)
Family support
The sudden nature of the illness makes emotional support of the child and parents extremely important (see Family-Centered Care box). Parents are upset and concerned about their child’s condition and often feel guilty for not having suspected the seriousness of the illness sooner. They need reassurance that the natural onset of meningitis is sudden and that they acted responsibly in seeking medical assistance when they did. The nurse encourages the parents to openly discuss their feelings to minimize blame and guilt. Some parents will benefit from referral to a hospital chaplain, social worker, psychologist, or psychiatrist. The nurse keeps parents informed of the child’s progress and of all procedures, results, and treatments. In the event that the child’s condition worsens, the parents need the same psychological care as other parents facing the possible death of their child (see Chapter 36).