3:  Ultrasound of the Gallbladder and Biliary Tree
Ultrasound of the Gallbladder and Biliary Tree
The Gallbladder and Biliary Tree
Ultrasound is an essential first-line investigation in suspected gallbladder and biliary duct disease. It is highly sensitive, accurate, and comparatively cheap, and is the imaging modality of choice.1 Gallbladder pathologic conditions are common and asymptomatic in over 13% of the population.2
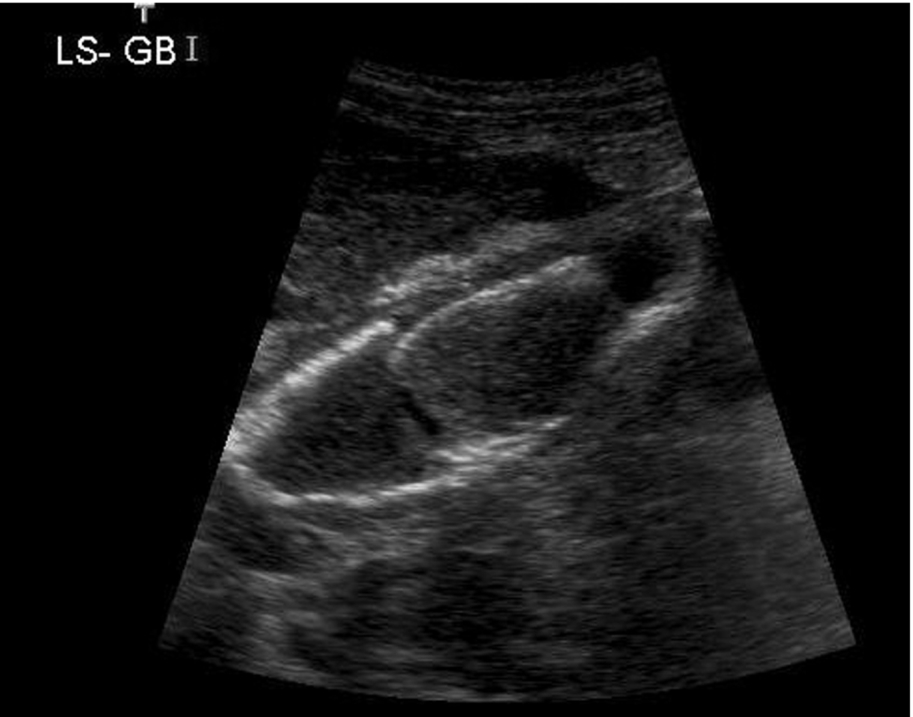
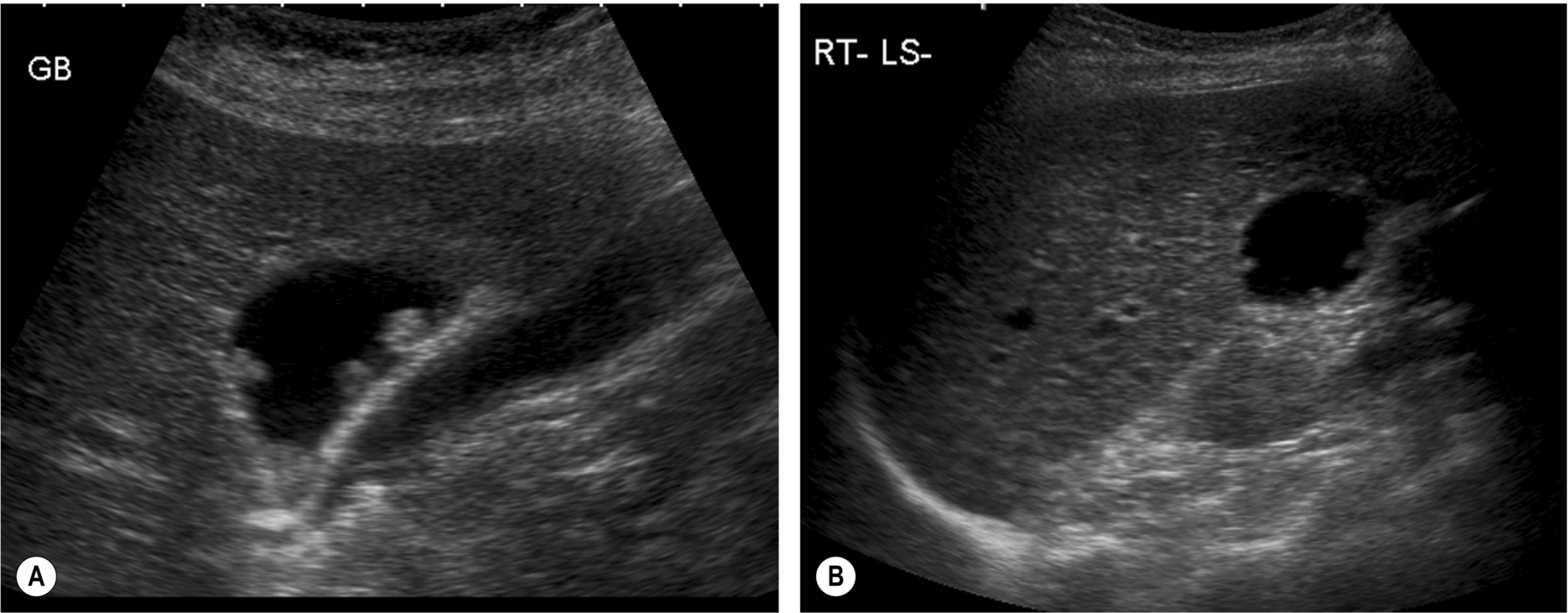
The normal gallbladder is best visualized after fasting to distend it. It should have a hyperechoic, thin wall (< 2 mm) and contain anechoic bile (Fig. 3.1). Measure the wall thickness in a longitudinal section of the gallbladder, with the calipers perpendicular to the wall itself. (A transverse section may not be perpendicular to the wall and can overestimate the thickness.)
After fasting for around 6 h, the gallbladder should be distended with bile into an elongated pear-shaped sac. The size is too variable to allow direct measurements to be of any use, but a tense, rounded shape can indicate pathological, rather than physiological dilatation.
Because the size, shape, and position of the gallbladder are infinitely variable, so are the techniques required to scan it. There are, however, several useful pointers to maximize visualization of the gallbladder:
- Use the highest frequency possible – 5.0 MHz or higher is especially useful for anterior gallbladders; in this instance, a high-frequency linear probe may be useful.
- Use a high line density to pick up tiny stones or polyps. (Reduce the sector angle and the frame rate if possible.) Make sure the focal zone is set over the back wall of the gallbladder to maximize your chances of identifying small stones.
- Alter the time gain compensation (TGC) to eliminate or minimize anterior artifacts and reverberation echoes inside the gallbladder, particularly in the near field.
- Use tissue harmonic imaging to reduce artifact within the gallbladder and sharpen the image of the wall (particularly in a large abdomen).
- Ensure any compounding software is switched off if you are unsure if the small lesion is a stone; removing the compounding software will allow posterior shadowing to be seen more clearly.
- Always scan the gallbladder in at least two planes (find the gallbladder’s long axis, incorporating the neck and fundus; sweep from side to side, then transversely from neck to fundus) and two patient positions. You will almost certainly miss pathologic conditions if you do not.
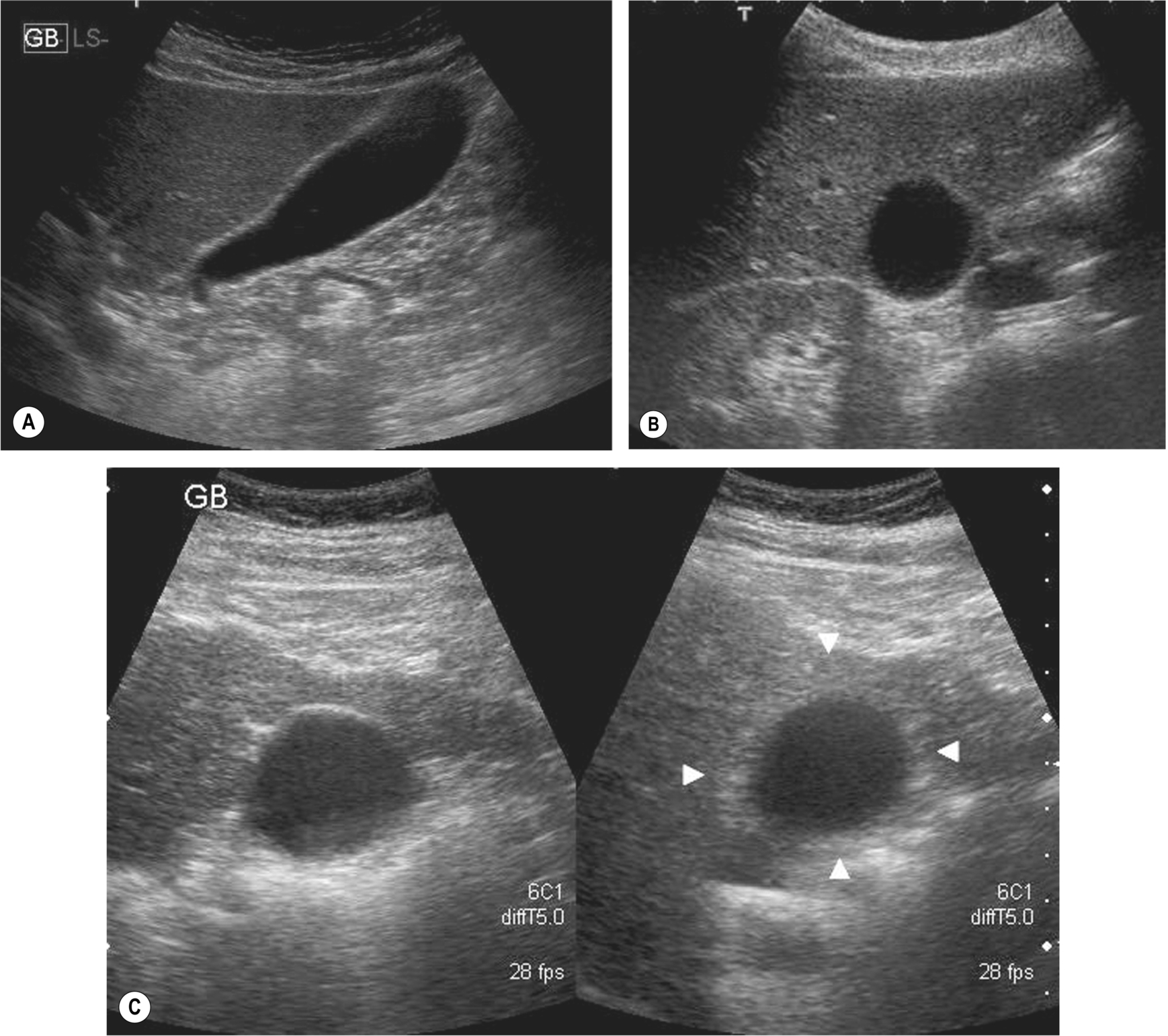
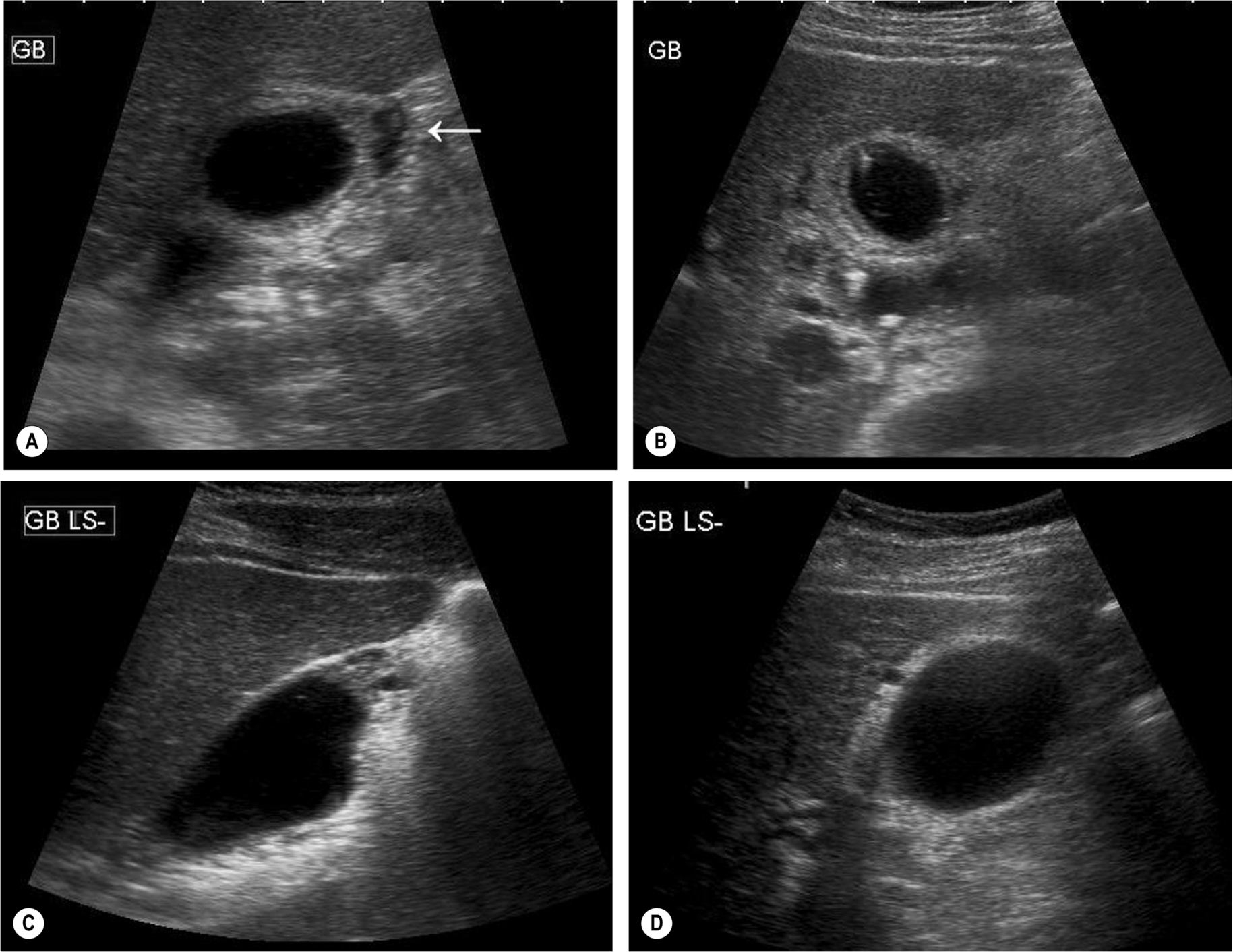
- The gallbladder may be “folded” (the so-called Phrygian cap). To be able to view this in it is entirety, it may be necessary to turn the patient into various positions (Fig. 3.2).
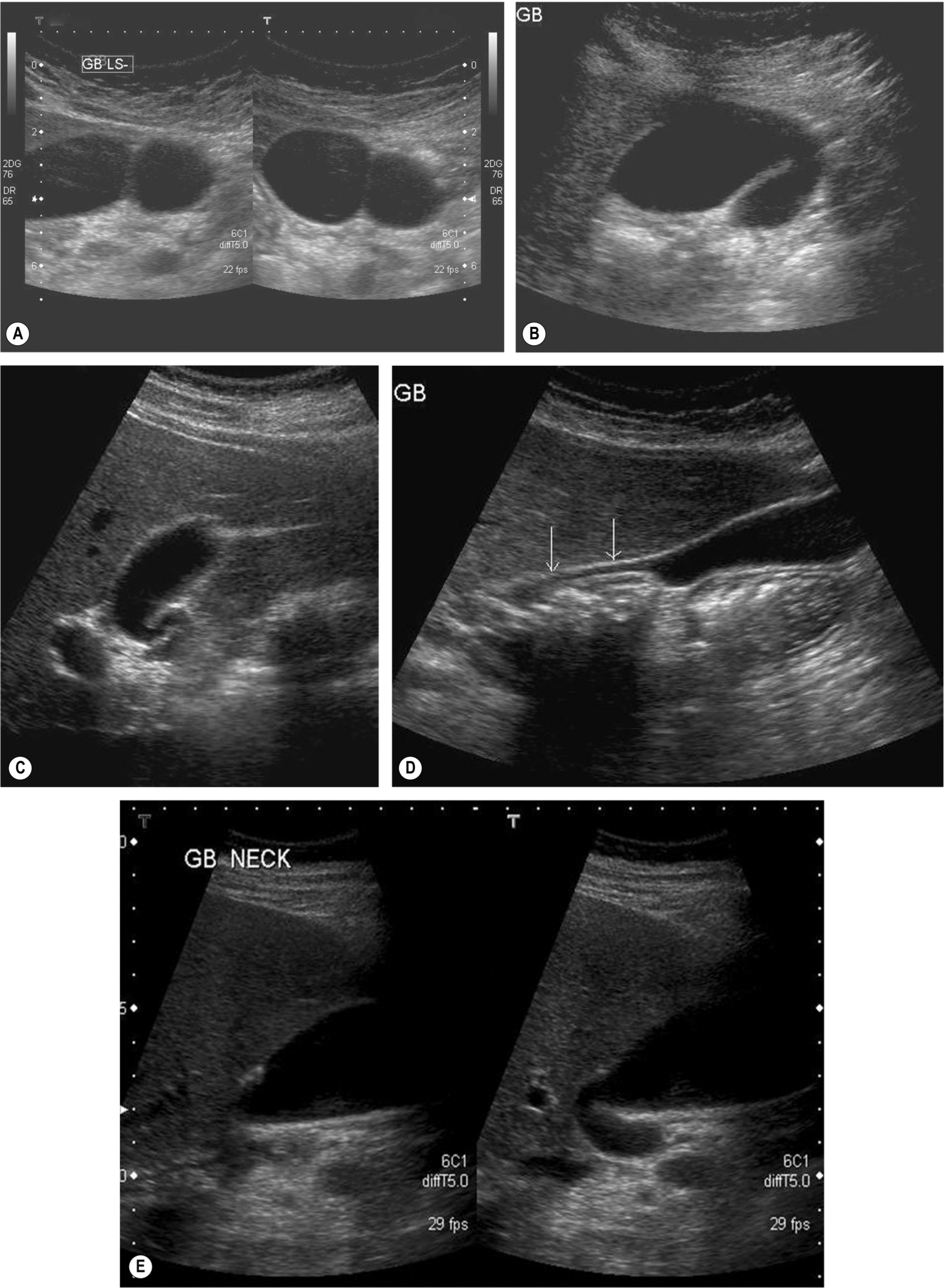
Fig. 3.2 (A) A folded gallbladder is difficult to examine with the patient supine, and the fold may mimic septation. (B) Scanning from a different angle (coronally) demonstrates the true nature of the folded gallbladder. (C) Many gallbladder necks can be tortuous, potentially hiding pathology. (D) Turning the patient decubitus, the right side is raised, unfolds the gallbladder, enabling the neck to be fully examined (arrows). (E) shows how hidden a gallbladder neck can be. - Bowel gas over the fundus can also be moved by various patient positions; consider scanning with the patient erect to try and move the bowel away from the gallbladder.
Normal Variants of the Gallbladder
The mesenteric attachment of the gallbladder to the inferior surface of the liver is variable in length. This gives rise to large variations in position; at one end of the spectrum, the gallbladder, attached only at the neck, may be fairly remote from the liver, even lying in the pelvis; at the other, the gallbladder fossa deeply invaginates the liver, and the gallbladder appears to lie “intrahepatically” enclosed on all sides by liver tissue.
The presence of a true septum in the gallbladder is rare. A folded gallbladder frequently gives the impression of a septum, but this can be distinguished by positioning the patient to unfold the gallbladder. Occasionally a gallbladder septum completely divides the lumen into two parts. True gallbladder duplication is a rare entity (Fig. 3.3), and it is important not to mistake this for a gallbladder with a pericholecystic collection in a symptomatic patient. Occasionally the gallbladder is absent altogether.
Pitfalls in Scanning the Gallbladder
If the gallbladder cannot be found:
- Check for previous surgery – evidence of laparoscopic surgery may be difficult to see in the darkened scanning room.
- Check that the patient has fasted.
- Look for an ectopic gallbladder – positioned low in the pelvis, for example.
- Check that near field artifact has not obscured an anterior gallbladder – a particular problem in very thin patients.
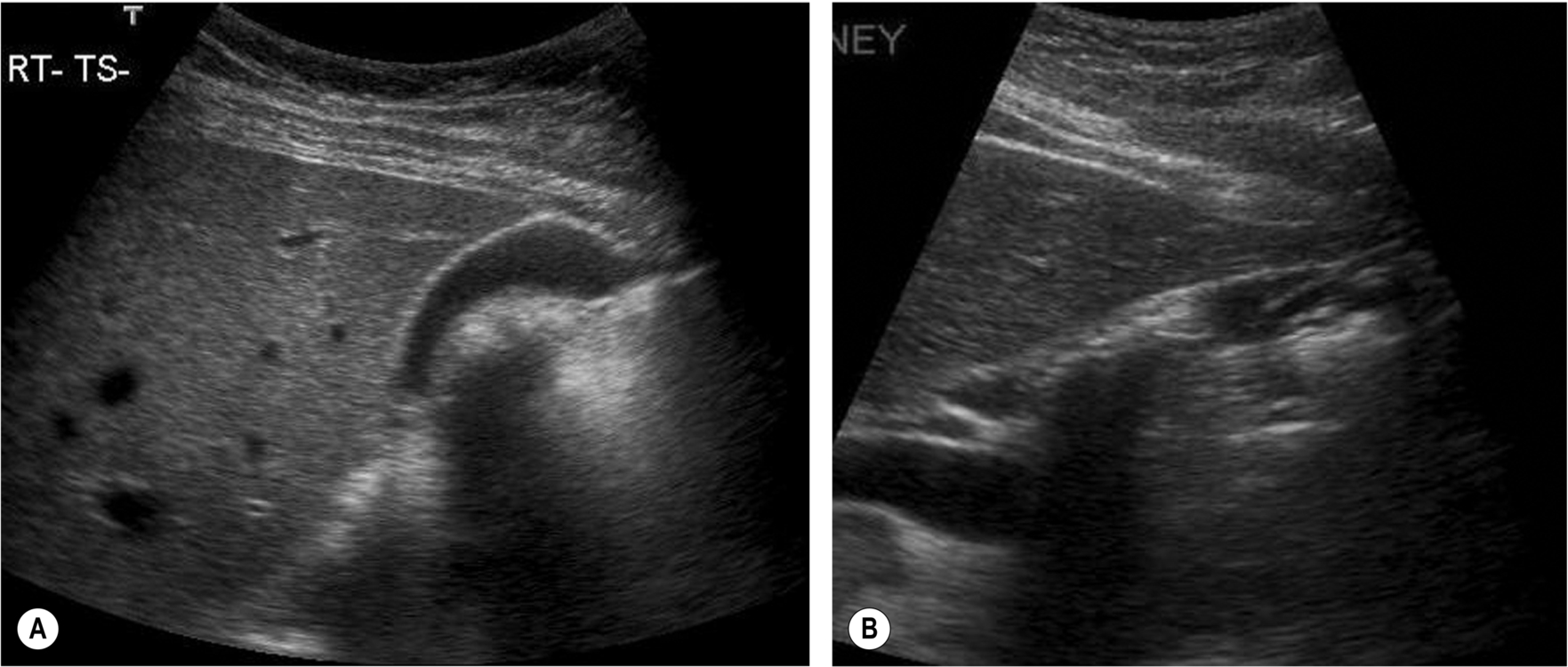
If you still cannot find it, ensure the scanner frequency and settings are optimized, find the porta hepatis, and scan just below it in the transverse section. This is the area of the gallbladder fossa, and you should see at least the anterior gallbladder wall if the gallbladder is present (Fig. 3.4).
A contracted, stone-filled gallbladder, producing heavy shadowing, can be difficult to identify because of the lack of any contrasting fluid in the lumen.
True agenesis of the gallbladder is rare.
Duodenum mimicking gallbladder pathologic conditions:
- The close proximity of the duodenum to the posterior gallbladder wall often causes it to invaginate the gallbladder. Maximize your machine settings to visualize the posterior gallbladder wall separate from the duodenum and turn the patient to cause the duodenal contents to move.
Other segments of the fluid-containing gastrointestinal tract can also cause confusion (Fig. 3.5).
Stones that do not shadow:
- This is likely to be because of poor settings, rather than any lack of stone reflectivity.
- Ensure they are stones and not polyps by positioning the patient erect and watching them move with gravity (beware – polyps on long stalks also move around).
- The stones may be smaller than the beamwidth, making the shadow difficult to display.
- Ensure the focal zone is set at the back of the gallbladder.
- Increase the line density, if possible, by reducing the field of view.
- Scan with the highest possible frequency to ensure the narrowest beamwidth.
- Reduce the TGC and/or power to ensure you have not saturated the echoes distal to the gallbladder.
Beware of the folded gallbladder! If the gallbladder is folded and the fundus lies underneath the bowel, you may miss pathologic conditions. Always try to unfold it by positioning the patient. A fold in the gallbladder may mimic a septum. Septations are comparatively rare and have been overreported in the past because of the presence of folding.
Pathology or Artifact?
Sometimes the gallbladder may contain some echoes of doubtful significance or be insufficiently distended to evaluate accurately. A rescan, after a meal followed by further fasting, can be useful. This can flush out sludge, redistending the gallbladder with clear bile. It may also help to clarify any confusing appearances of adjacent bowel loops.
The Bile Ducts
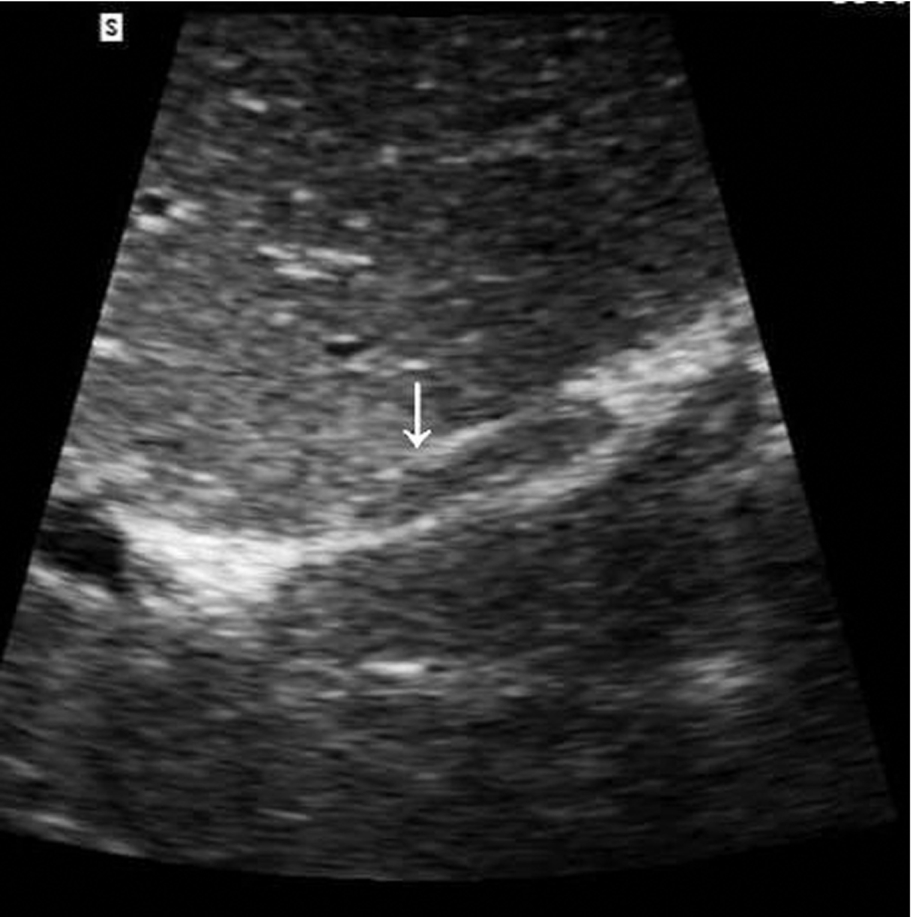
The common duct can be easily demonstrated in its intrahepatic portion just anterior and slightly to the right of the portal vein. A cross-section of the main hepatic artery can usually be seen passing between the vein and the duct (Fig. 3.6), although a small proportion of hepatic arteries lie anterior to the duct. At this point, it is usually referred to as the common duct, although it may, in fact, represent the right hepatic duct rather than the common bile duct (CBD) because we cannot tell at what point it is joined by the cystic duct.
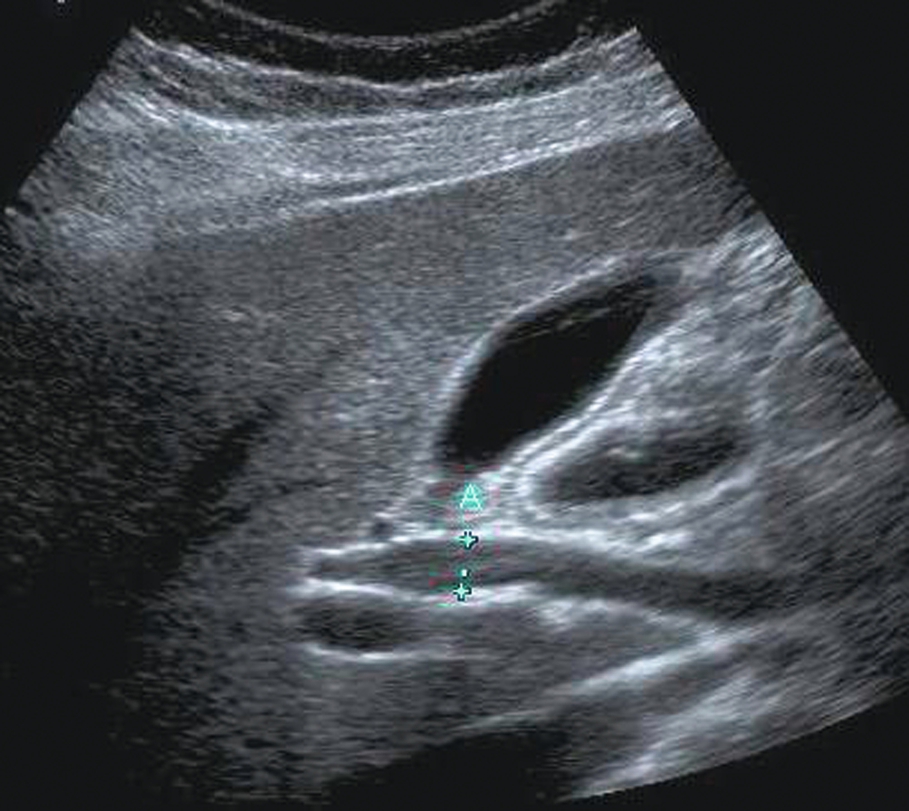
The extrahepatic portion of the duct is less easy to see as it is often obscured by overlying duodenal gas. Good visualization of the duct usually requires perseverance on the part of the operator. It is insufficient just to visualize the intrahepatic portion of the duct, as early obstruction may be present with a normal caliber intrahepatic duct and dilatation of the distal end (Fig. 3.7).
Bile Duct Measurements
The internal diameter of the common duct is usually taken as 6 mm or less. However, it is age-dependent and can be 8-9 mm in an older adult because of degeneration of the elastic fiber in the duct wall. Ensure this is not early obstruction by thoroughly examining the distal CBD or rescanning after a short time interval.
The diameter can vary quite considerably, not only between subjects but along an individual duct. The greatest measurement should be recorded in the longitudinal section. Never measure the duct in a transverse section (for example, at the head of the pancreas); it is invariably an oblique plane through the duct, which will overestimate the diameter. Intrahepatically, the duct diameter decreases. The right and left hepatic ducts are just visible, but more peripheral branches are usually too small to see.
Techniques
The main, right, and left hepatic ducts tend to lie anterior to the portal vein branches. However, as the biliary tree spreads out, the position of the duct relative to the portal branches is highly variable. Do not assume that a channel anterior to the portal vein branch is always a biliary duct. The walls of the ducts are hyperechoic and can help distinguish the ducts from vessels however, if in doubt, use color Doppler to distinguish the bile duct from the portal vein or hepatic artery. The proximal bile duct is best seen either with the patient supine, using an intercostal approach from the right, or turning the patient oblique, right side raised. This projects the duct over the portal vein, which is used as an anatomic marker.
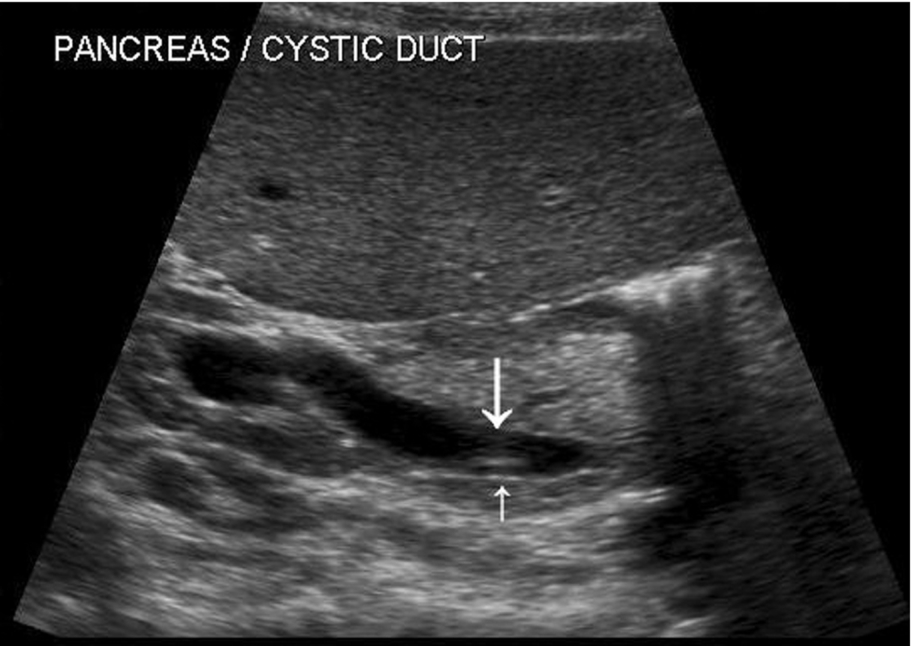
Scanning the distal duct usually requires more effort. Right oblique or decubitus positions are useful. Gentle pressure to ease the duodenal gas away from the duct can also be successful. Sometimes, filling the stomach with water (which also helps to display the pancreas) and allowing it to trickle through the duodenum does the trick. Try also identifying the duct in the pancreatic head (Fig. 3.9) and then tracing it retrogradely toward the liver. Asking the patient to take deep breaths is occasionally successful but may make matters worse by filling the stomach with air. It is definitely worth persevering with your technique, particularly in jaundiced patients.
Common Referral Patterns for Hepatobiliary Ultrasound
Jaundice
Jaundice is a frequent cause of referral for an abdominal ultrasound. Therefore, it is essential for the sonographer to have a basic understanding of the various mechanisms to maximize the diagnostic information from the ultrasound scan.
Jaundice or hyperbilirubinemia, is an elevated level of bilirubin in the blood. It is recognized by a characteristic yellow coloration of the skin and sclera of the eye, often accompanied by itching if prolonged. Bilirubin is derived from the hem portion of hemoglobin. Red blood cells are broken down in the liver into hem and globin, releasing their bilirubin which is non-soluble. This is termed unconjugated bilirubin. This is then taken up by the liver cells and converted to a water-soluble form – conjugated bilirubin, which is excreted via the biliary ducts into the duodenum to aid fat digestion.
By knowing which of these two types of bilirubin is present in the jaundiced patient, the clinician can narrow the diagnostic possibilities. The ultrasound then further refines the diagnosis (Fig. 3.10).
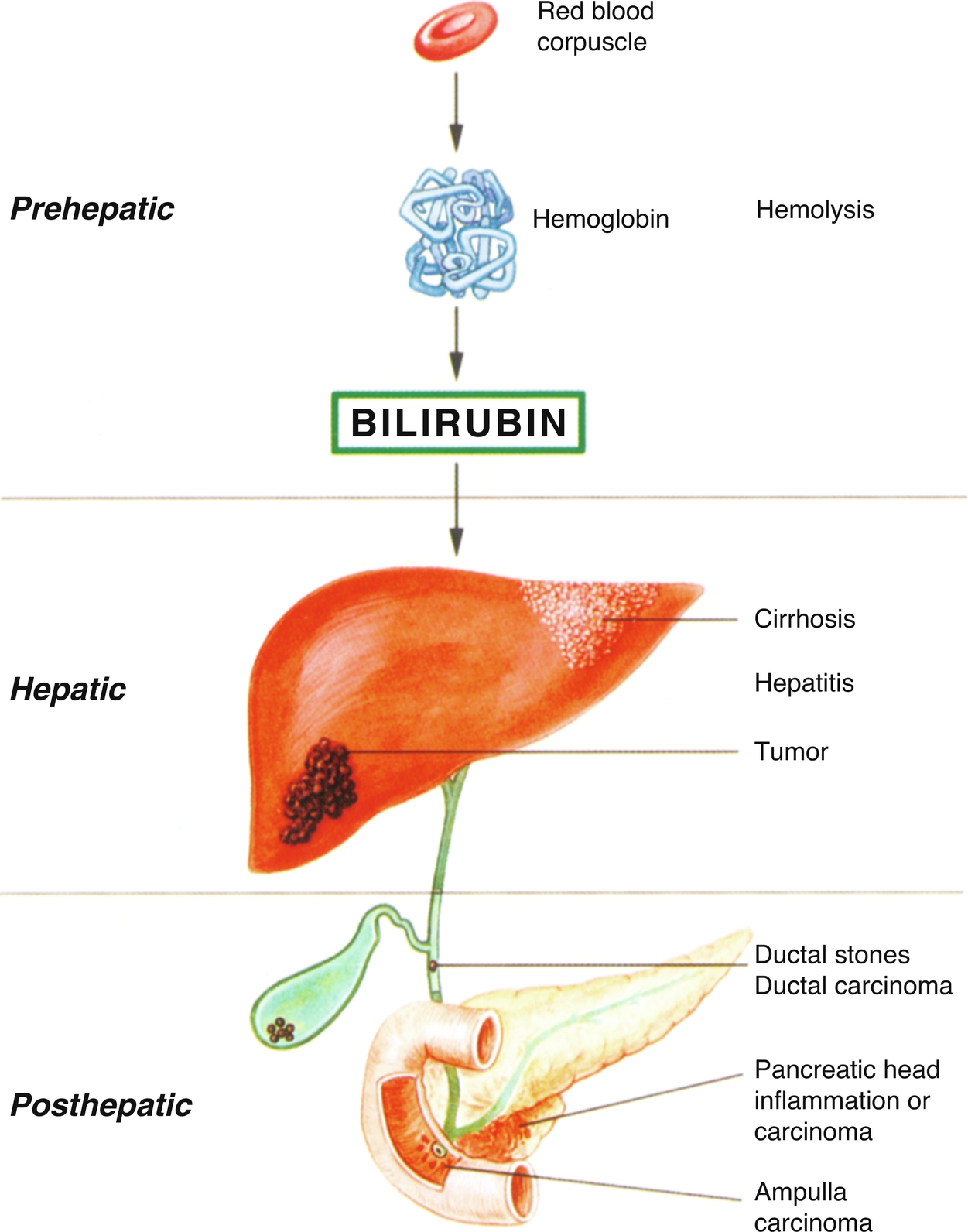
Jaundice can fall into one of two categories:
- Obstructive (sometimes called posthepatic) – in which the bile is prevented from draining out of the liver because of obstruction to the biliary duct(s).
- Non-obstructive (prehepatic or hepatic) – in which the elevated bilirubin level is because of hemolysis (the breakdown of the red blood cells) or a disturbance in the mechanism of the liver for uptake and storage of bilirubin, such as in inflammatory or metabolic liver diseases.
Abnormal Liver Function Tests
Altered or deranged liver function tests (LFTs) are another frequent cause of referral for an abdominal ultrasound. Biochemistry from a simple blood test is often a primary pointer to pathologic conditions and is invariably one of the first tests performed as it is quick and easily accessible. Raised bilirubin in combination with elevated alkaline phosphatase (Alk Phos) is suggestive of obstruction, where an isolated raised bilirubin is more likely to represent a metabolic condition. Ultrasound can be used in cases of obstructive jaundice, which demonstrates some degree of biliary duct dilatation.
Most of the other markers are highly unspecific, being associated with many types of diffuse and focal liver pathologic conditions and may not be relevant in gallbladder disease.
Gallbladder Pathologic Conditions
Cholelithiasis
The most commonly and reliably identified gallbladder pathologic condition is gallstones. More than 10% of the population of the United Kingdom have gallstones. Many of these are asymptomatic (Box 3.1), which is important to remember. When scanning a patient with abdominal pain, it should not automatically be assumed that when gallstones are present, they are responsible for the pain. It is not uncommon to find further pathologic conditions in the presence of gallstones, and a comprehensive upper abdominal survey should always be carried out. However, up to 35% of patients who have gallstones require surgery to relieve symptoms (Table 3.1).3
| Physiological | Post-Prandial |
|---|---|
| Inflammatory | Acute or chronic cholecystitis |
| Sclerosing cholangitis | |
| Crohn’s disease | |
| AIDS | |
| Adjacent inflammatory causes | Pancreatitis |
| Hepatitis | |
| Pericholecystic abscesses | |
| Non-inflammatory | Adenomyomatosis |
| Gallbladder carcinoma | |
| Focal areas of thickening because of metastases or polyps | |
| Leukemia | |
| Edema | Ascites from a variety of causes, including organ failure, lymphatic obstruction, and portal hypertension |
| Varices | Varices of the gallbladder wall in portal hypertension |
Gallstones are associated with a number of conditions. They occur when the normal ratio of components making up the bile is altered, most commonly when there is increased secretion of cholesterol in the bile. Conditions that are associated with increased cholesterol secretion and therefore the formation of cholesterol stones include obesity, diabetes, pregnancy, and estrogen therapy. The incidence of stones also rises with age, probably because the bile flow slows down.
An increased secretion of bilirubin in the bile, as in patients with cirrhosis, is associated with pigment (black or brown) stones.
Ultrasound Appearances
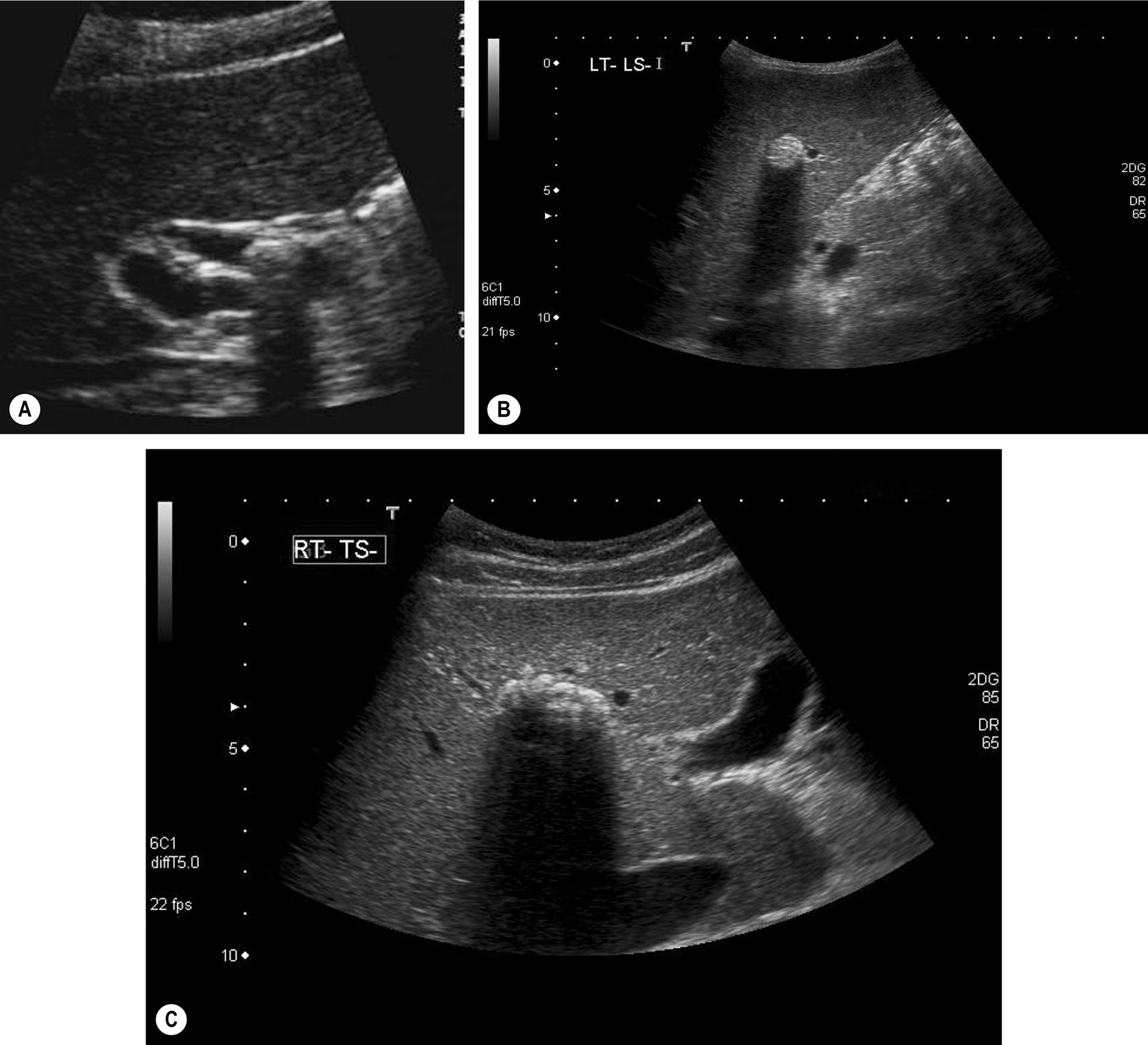
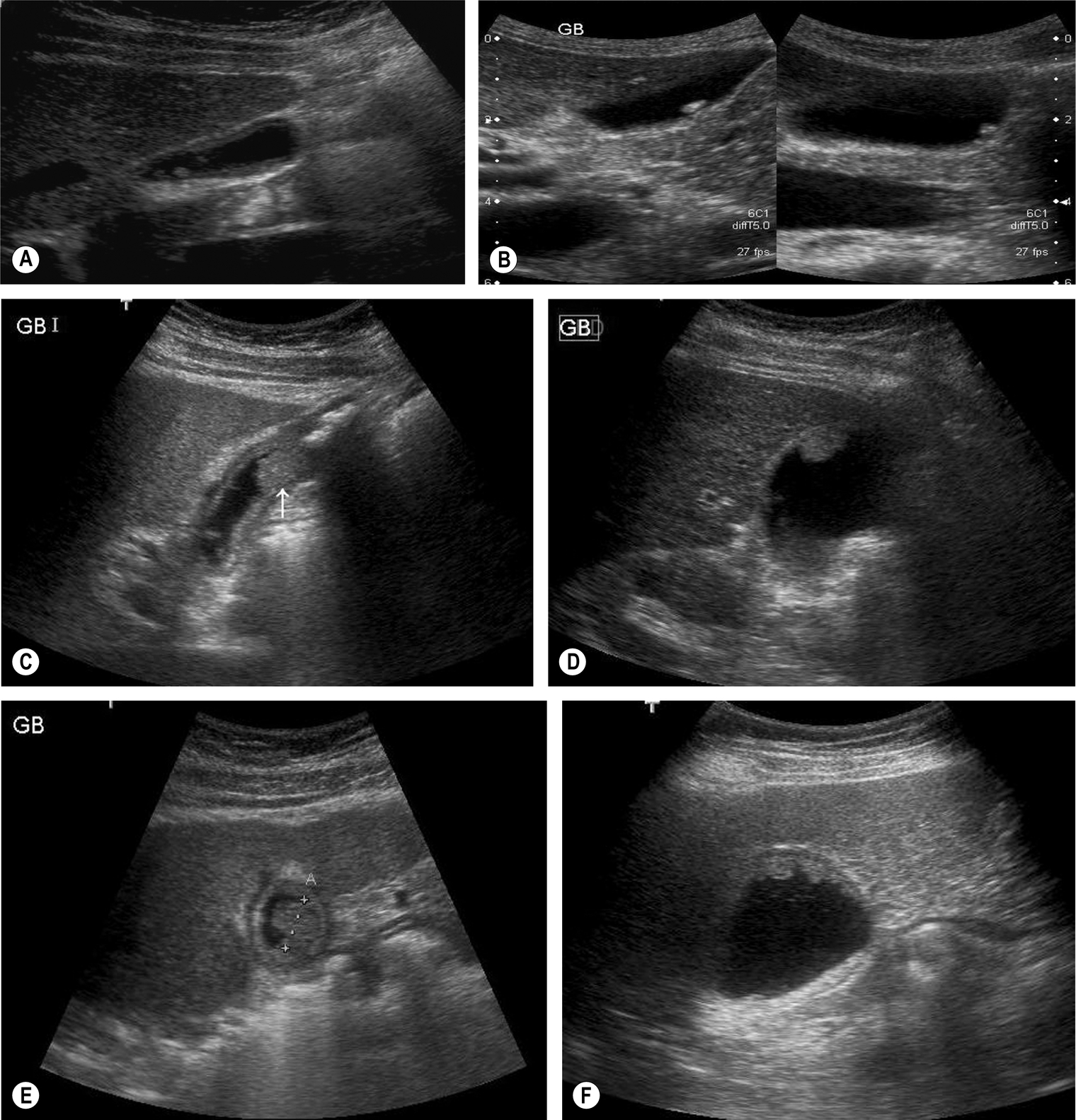
There are three classic acoustic properties associated with stones in the gallbladder; they are highly reflective, mobile, and cast a distal acoustic shadow. In most cases, all these properties are demonstrated (Figs. 3.11–3.13).
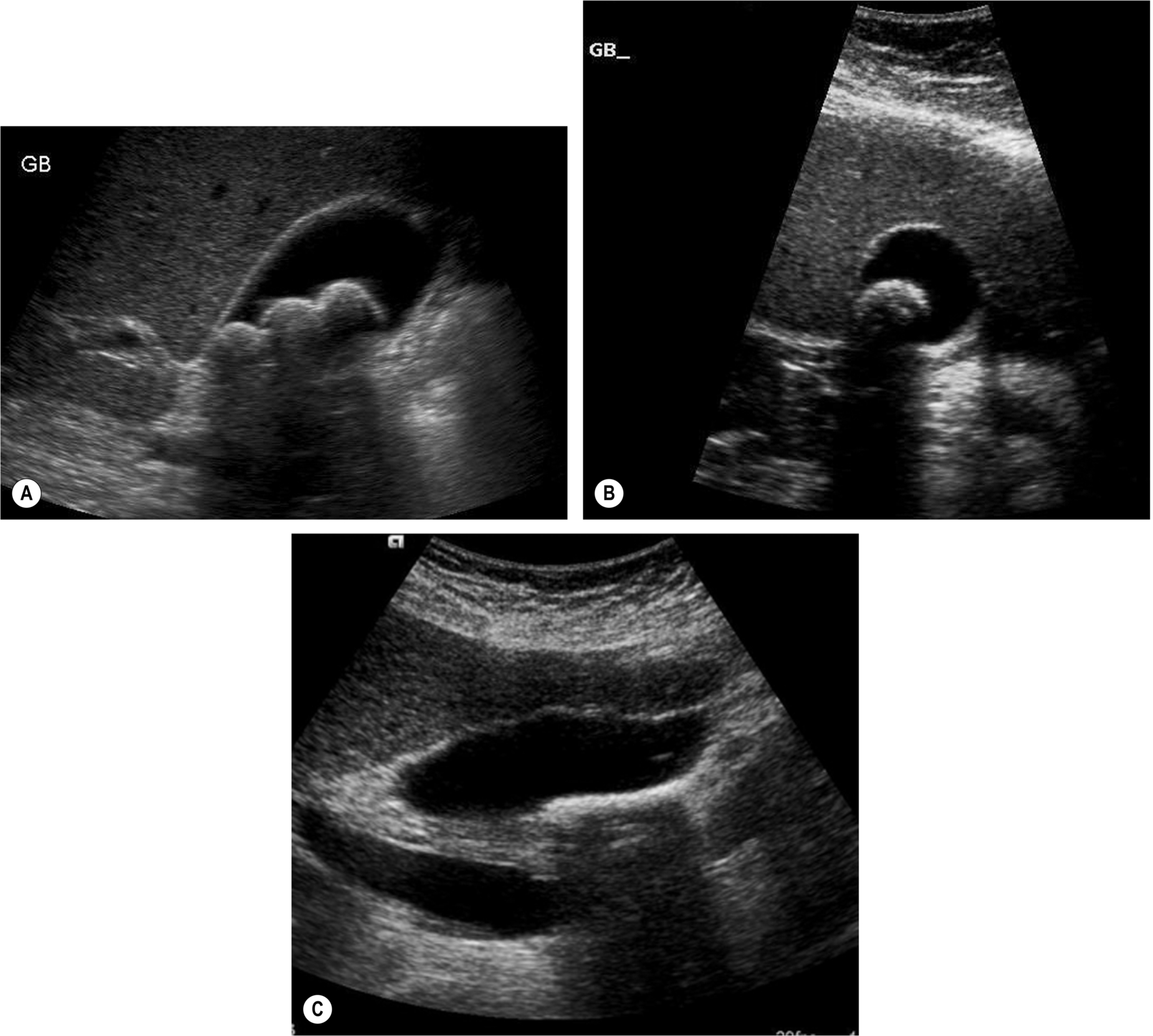
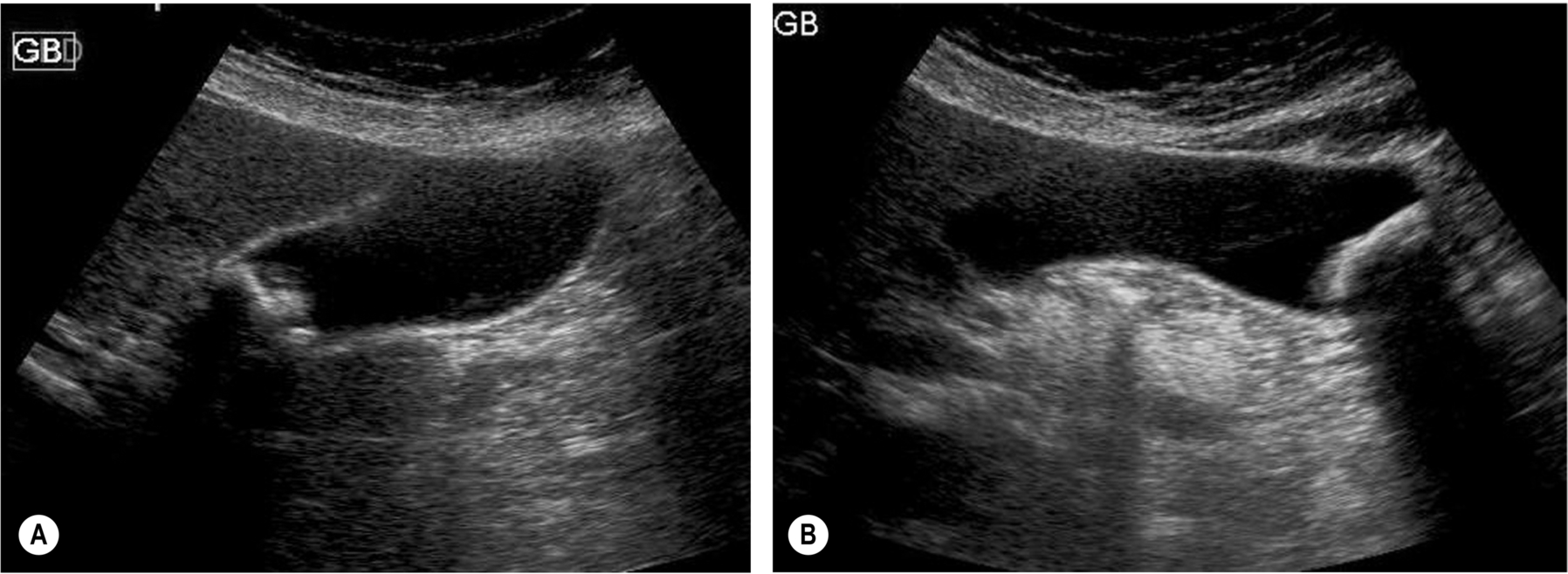
Shadowing
The ability to display a shadow posterior to a stone depends on several factors:
- 1. The reflection and absorption of sound by the stone. This is fairly consistent, regardless of the composition of the stone.
- 2. The size of the stone in relation to the beamwidth. A shadow occurs when the stone fills the width of the beam (Fig. 3.14). This will happen easily with large stones, but a small stone may occupy less space than the beam, allowing sound to continue behind it, so a shadow is not seen. Small stones must therefore be within the focal zone (narrowest point) of the beam and in the center of the beam to shadow (Fig. 3.15). Higher frequency transducers have better resolution and are therefore more likely to display fine shadows than lower frequencies.
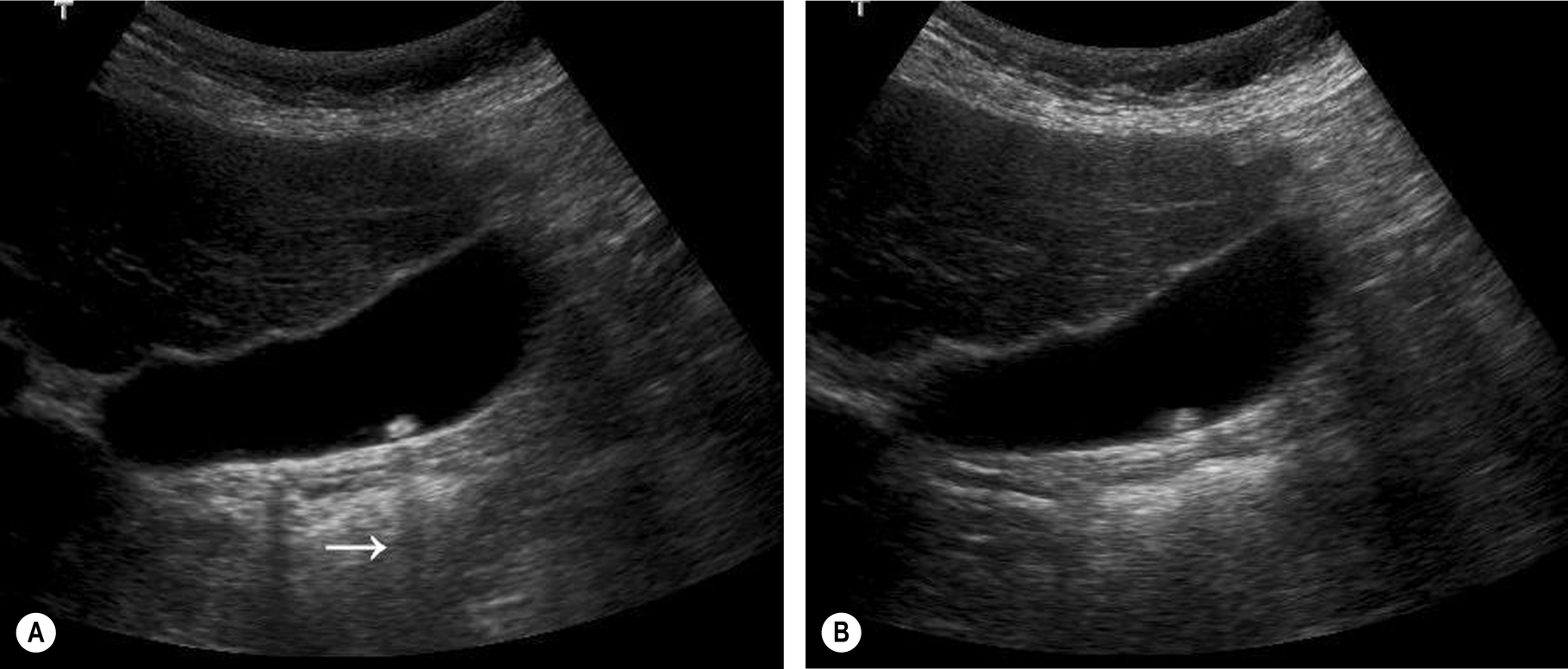
Fig. 3.14 (A) This small stone is wider than the beamwidth and casts an acoustic shadow (arrow). (B) The shadow is no longer evident when the beam focusing is moved anterior to the gallbladder as the beam is now wider than the stone. 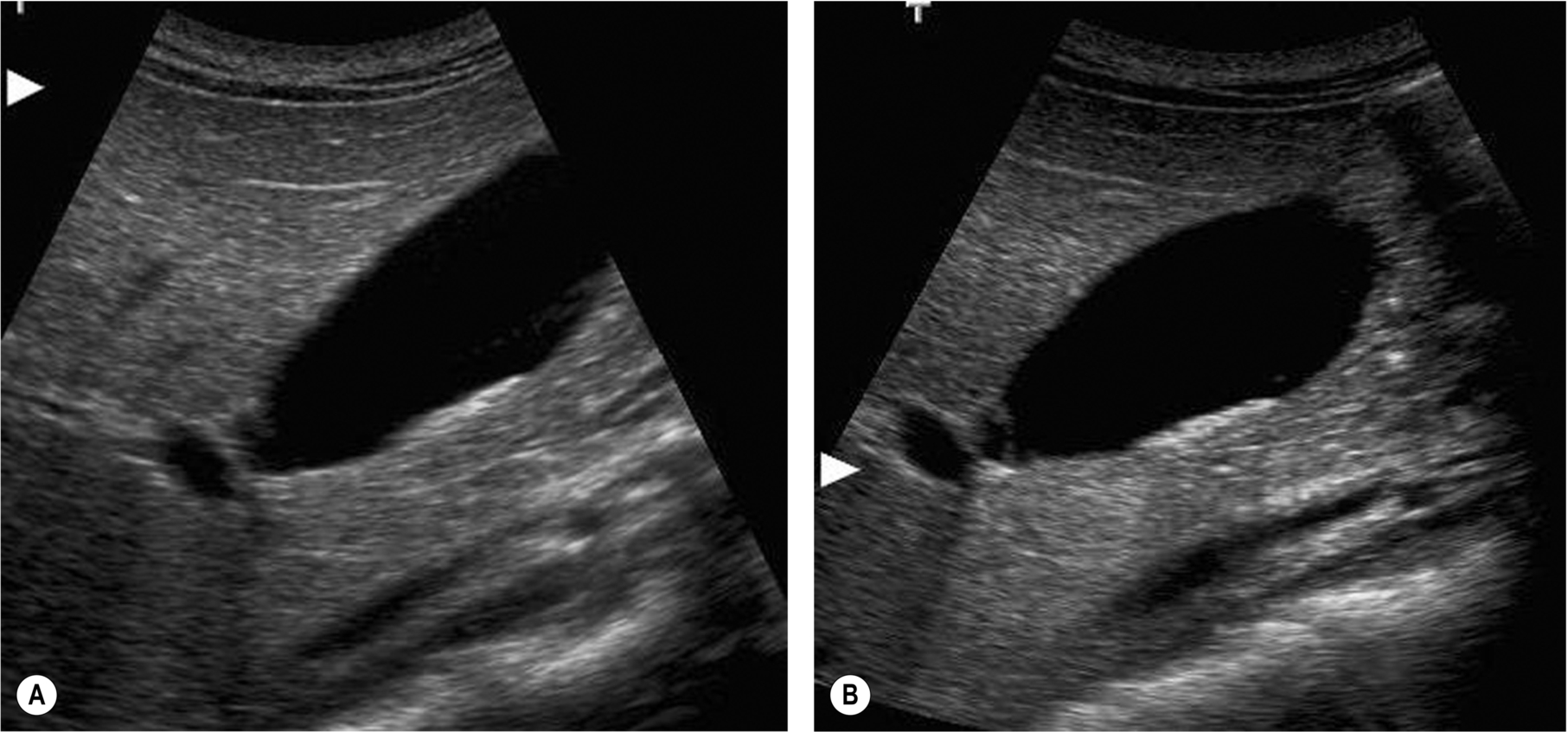
Fig. 3.15 (A) A layer of tiny stones does not shadow when the focal zone is placed incorrectly (arrow), but the shadow is easily demonstrated (B) when the focal zone is placed at the posterior wall of the gallbladder, narrowing the beam at this point. - 3. The machine settings must be compatible with demonstrating narrow bands of shadowing. The fluid-filled gallbladder often displays posterior enhancement or increased through transmission. If the echoes posterior to the gallbladder are “saturated,” this will mask fine shadows. Turn the overall gain down to display this better (Fig. 3.16). Some image processing options may reduce the contrast between the shadow and the surrounding tissue, so make sure a suitable dynamic range and image program are used.
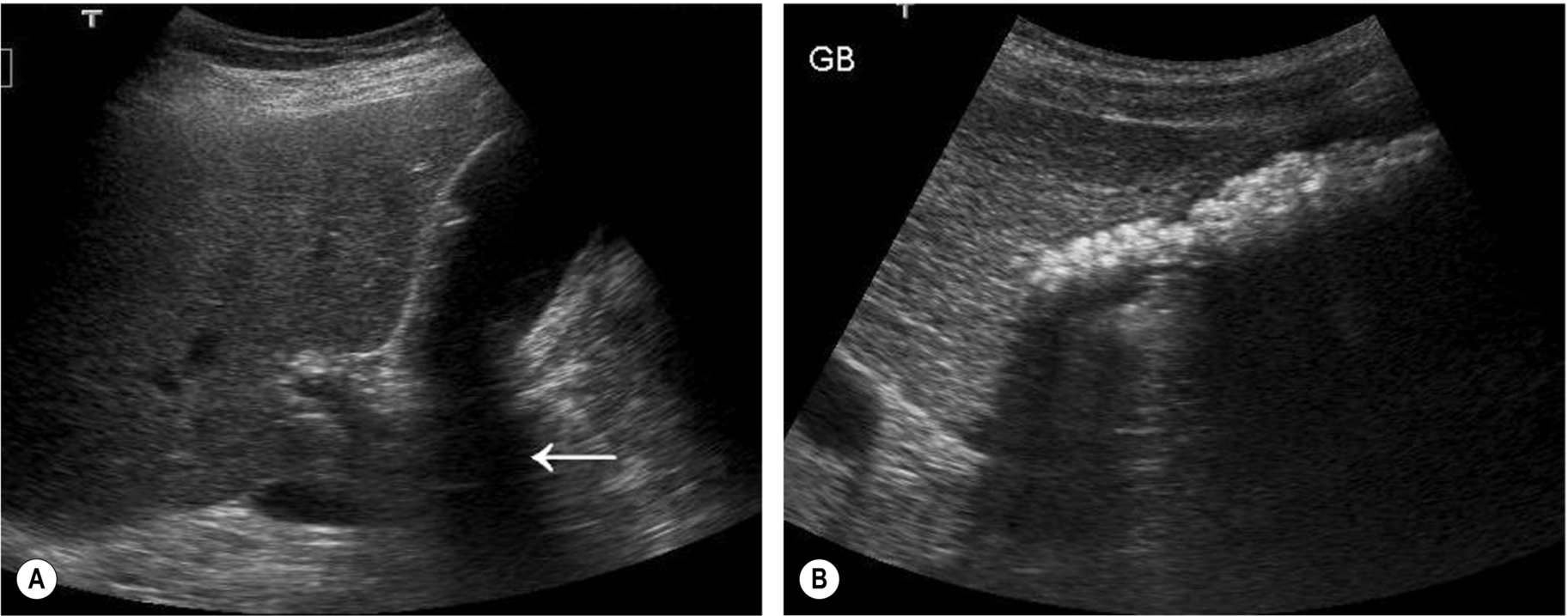
- 4. Bowel posterior to the gallbladder may cast its own shadows from gas and other contents, making the gallstone shadow difficult to demonstrate (Fig. 3.17). This is a particular problem with stones in the CBD. Try turning the patient to move the gallbladder away from the bowel. The shadow cast by gas in the duodenum, which contains reverberation, should usually be distinguishable from that cast by a gallstone, which is sharp and clean.
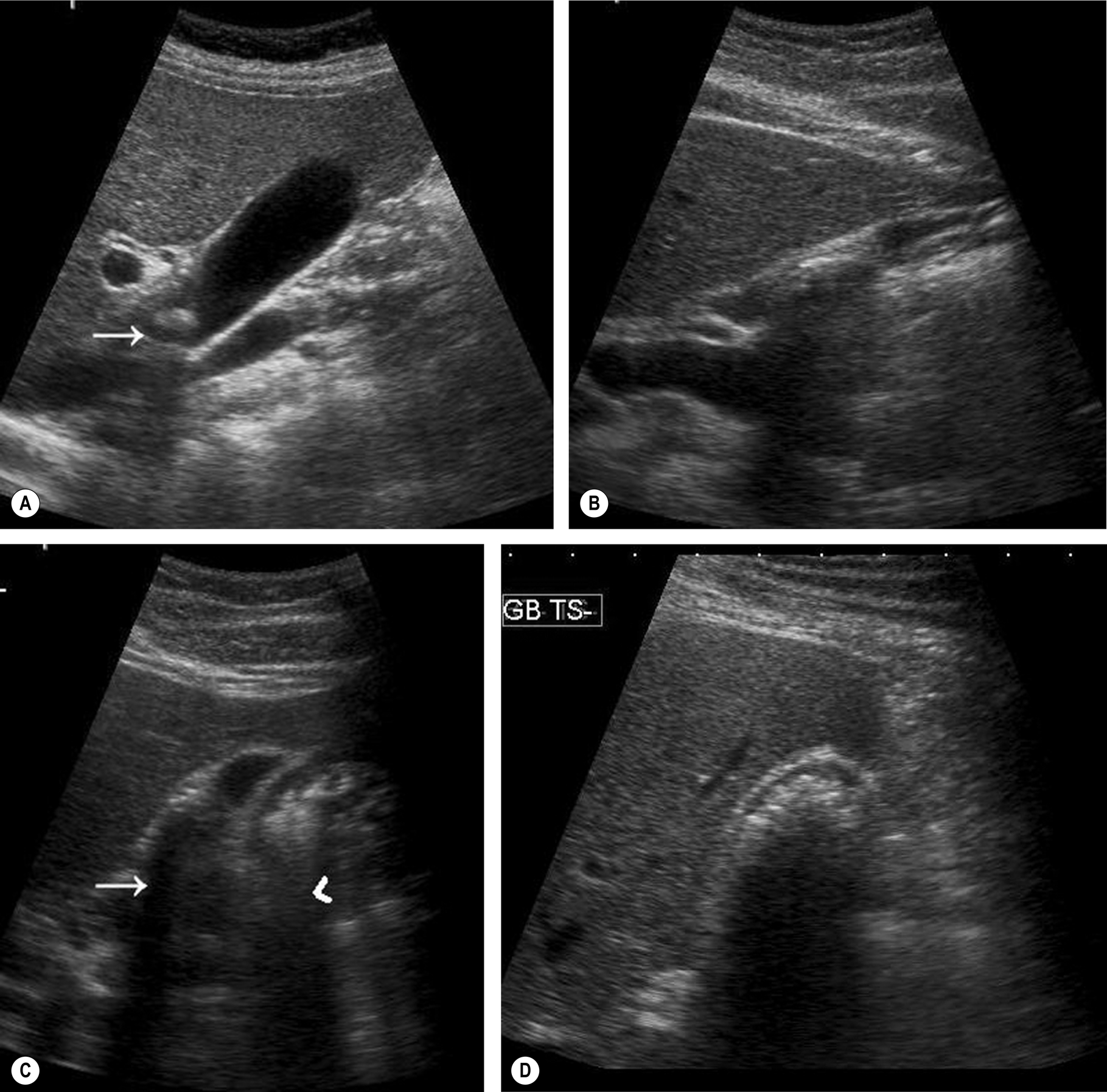
Fig. 3.17 (A) Shadowing from the stone in the neck of the gallbladder (arrow) is less obvious because of the structures lying behind the gallbladder. (B) Shadowing from the duodenum posterior to the gallbladder can obscure the shadowing from tiny stones within the gallbladder lumen. Changing patient position and angle of scanning moves the gas away from the gallbladder. (C) Shadowing from stones in the contracted gallbladder lumen (arrow) is stronger and better defined than the “dirty” shadowing from the adjacent bowel (arrowhead). (D) A contracted gallbladder full of stones casts a strong acoustic shadow from the gallbladder fossa. Note that the echoes from the anterior gallbladder wall are distinct from the echoes from the stones.
Reflectivity
The reflective nature of the stone is enhanced by its being surrounded by echo-free bile. In a contracted gallbladder, the reflectivity of the stone is often not appreciated because the hyperechoic gallbladder wall is collapsed over it.
Some stones are only poorly reflective but should still cause a distal acoustic shadow.
Mobility
Most stones are gravity-dependent, and this may be demonstrated by scanning the patient in an erect position (Fig. 3.12) when a mobile calculus will drop from the neck or body of the gallbladder to lie in the fundus. However, some stones will float, forming a reflective layer just beneath the anterior gallbladder wall with shadowing that obscures the rest of the lumen (Fig. 3.13B). When the gallbladder lumen is contracted, either because of physiological or pathological reasons, any stones present are unable to move, and this is also the case in a gallbladder packed with stones (Fig. 3.17D).
Occasionally a stone may become impacted in the neck, and movement of the patient is unable to dislodge it. Stones lodged in the gallbladder neck or cystic duct may result in a permanently contracted gallbladder, a gallbladder full of fine echoes because of inspissated (thickened) bile (Fig. 3.18), or a distended gallbladder because of a mucocele (see below).
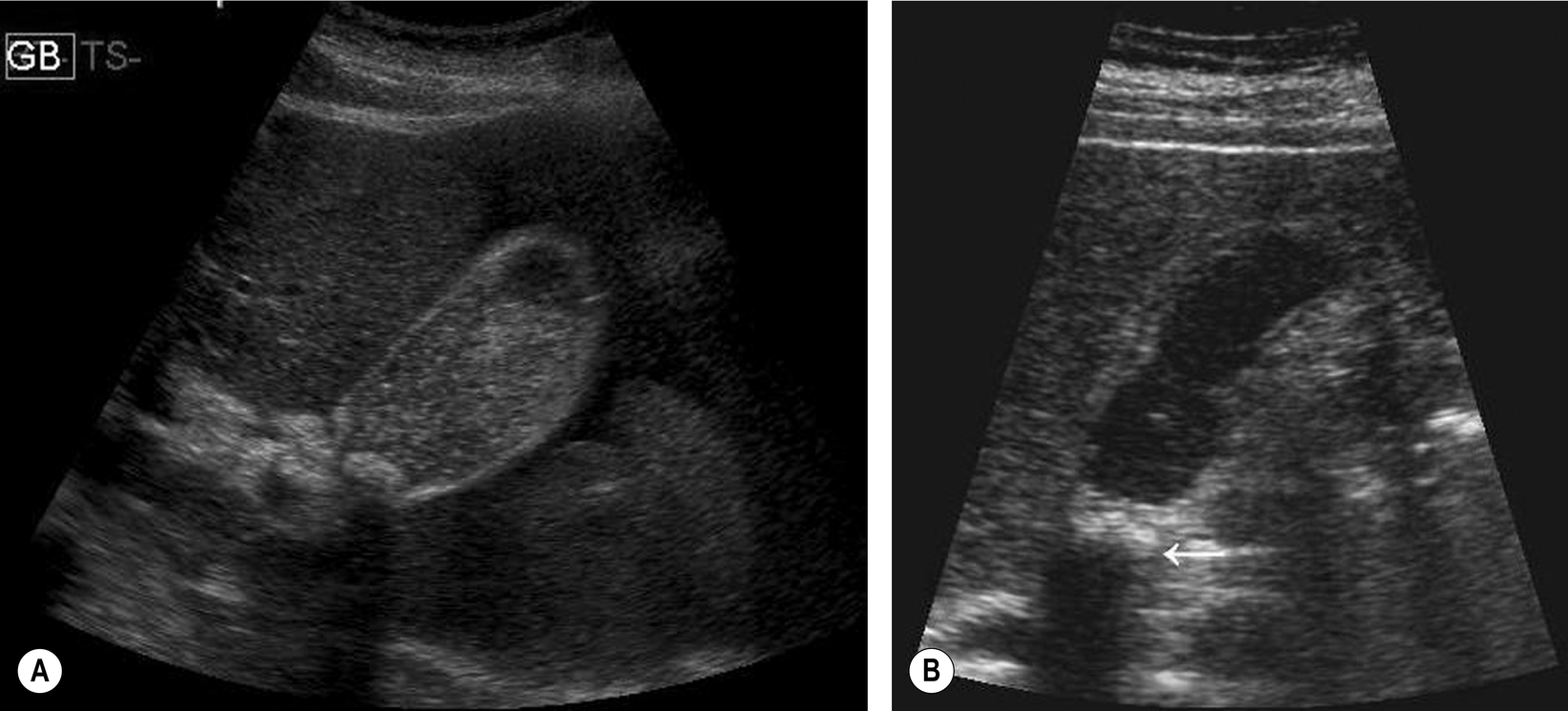
Choledocholithiasis
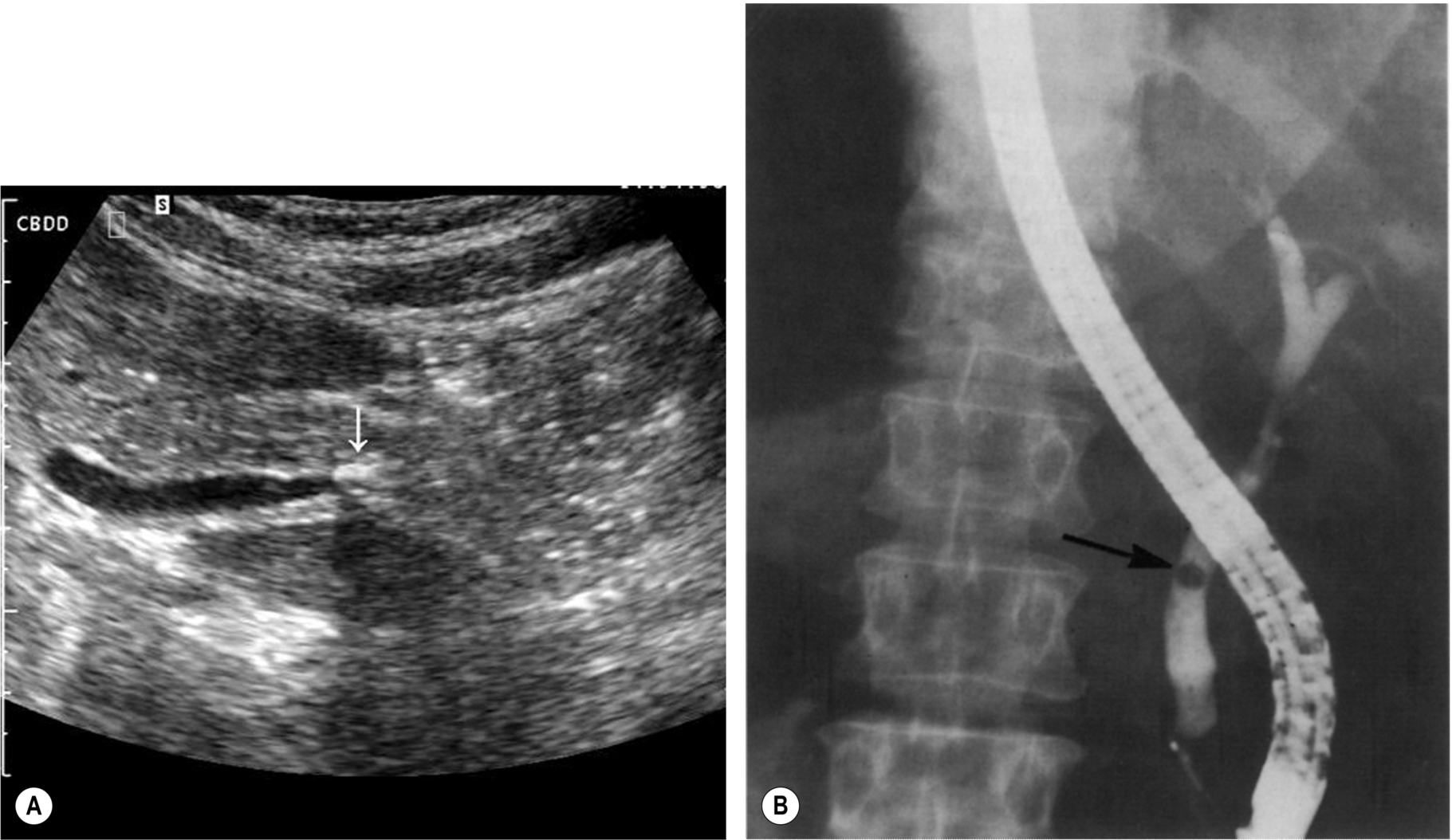
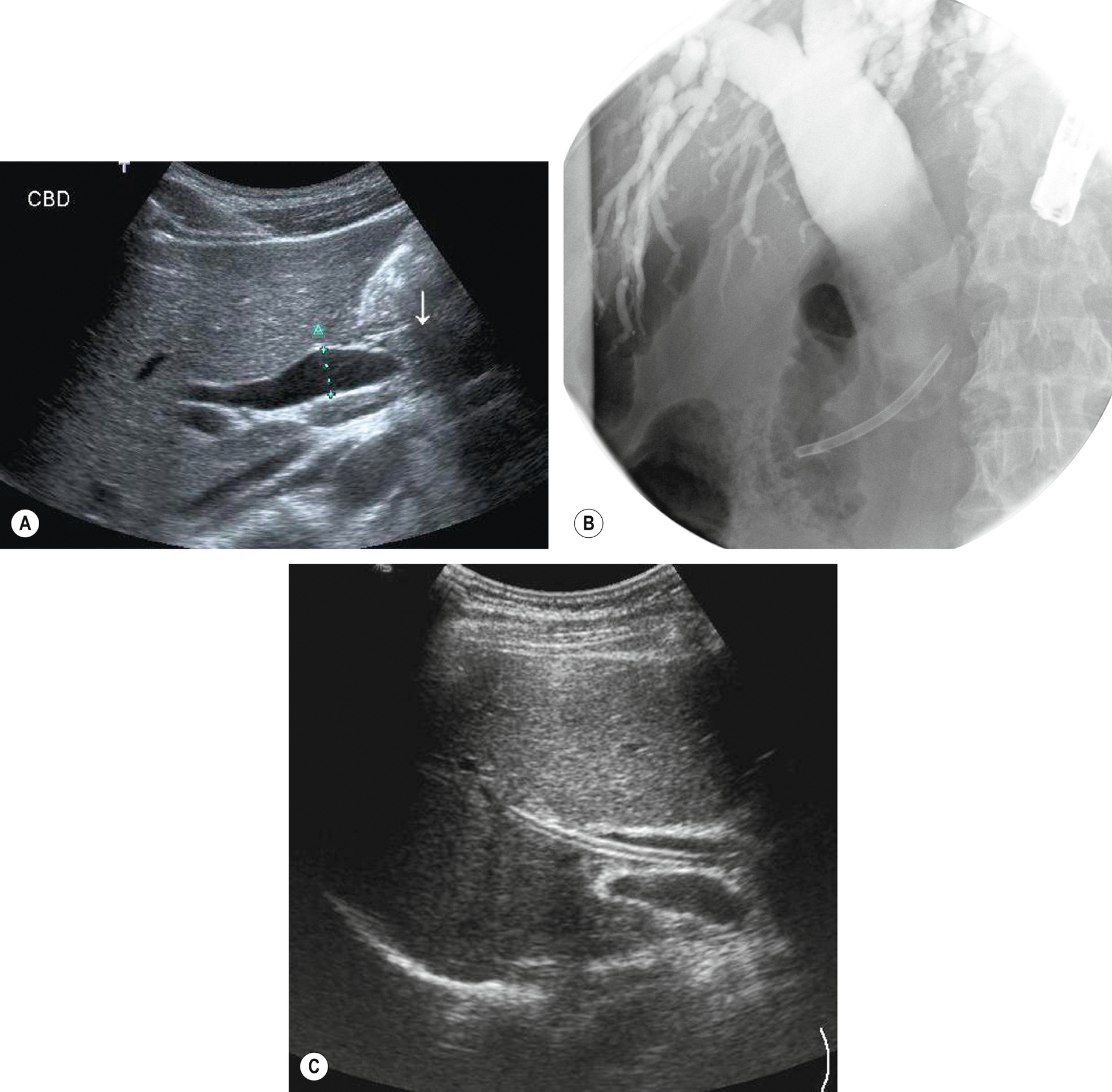
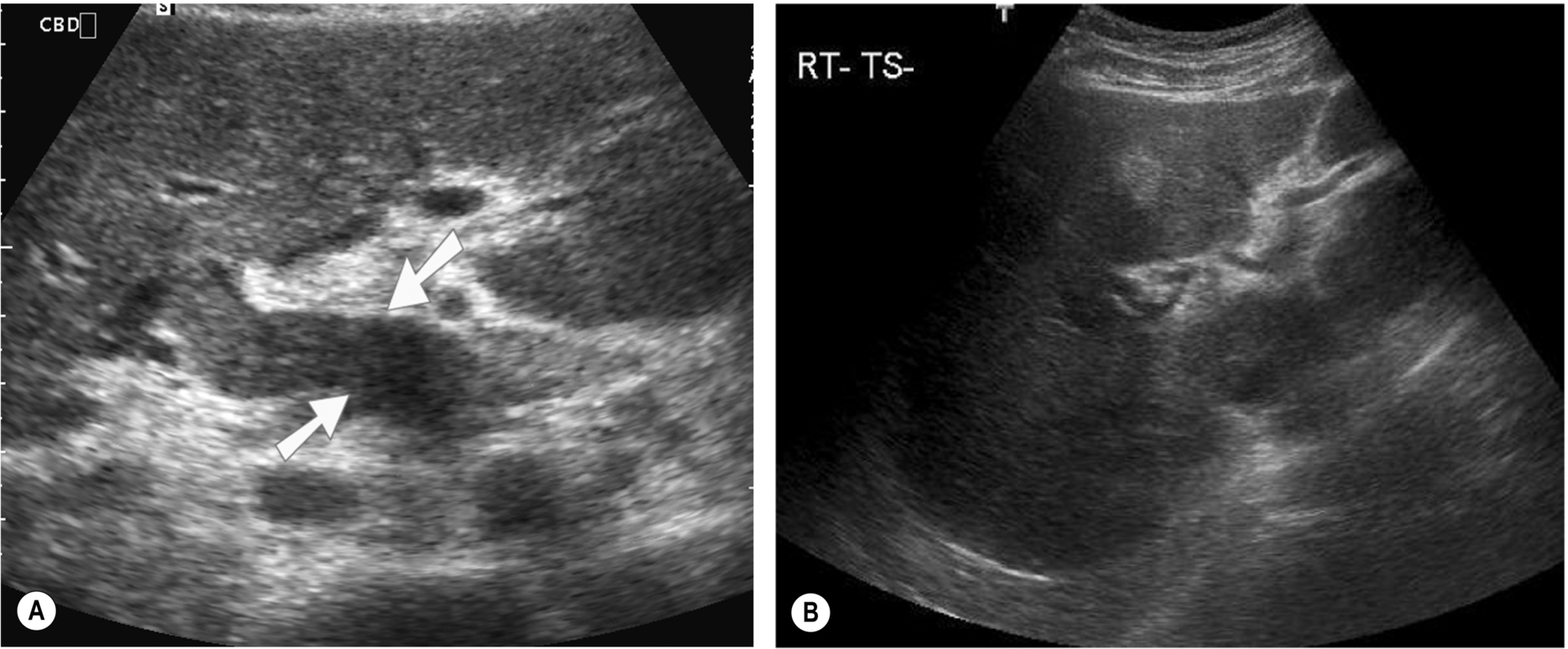
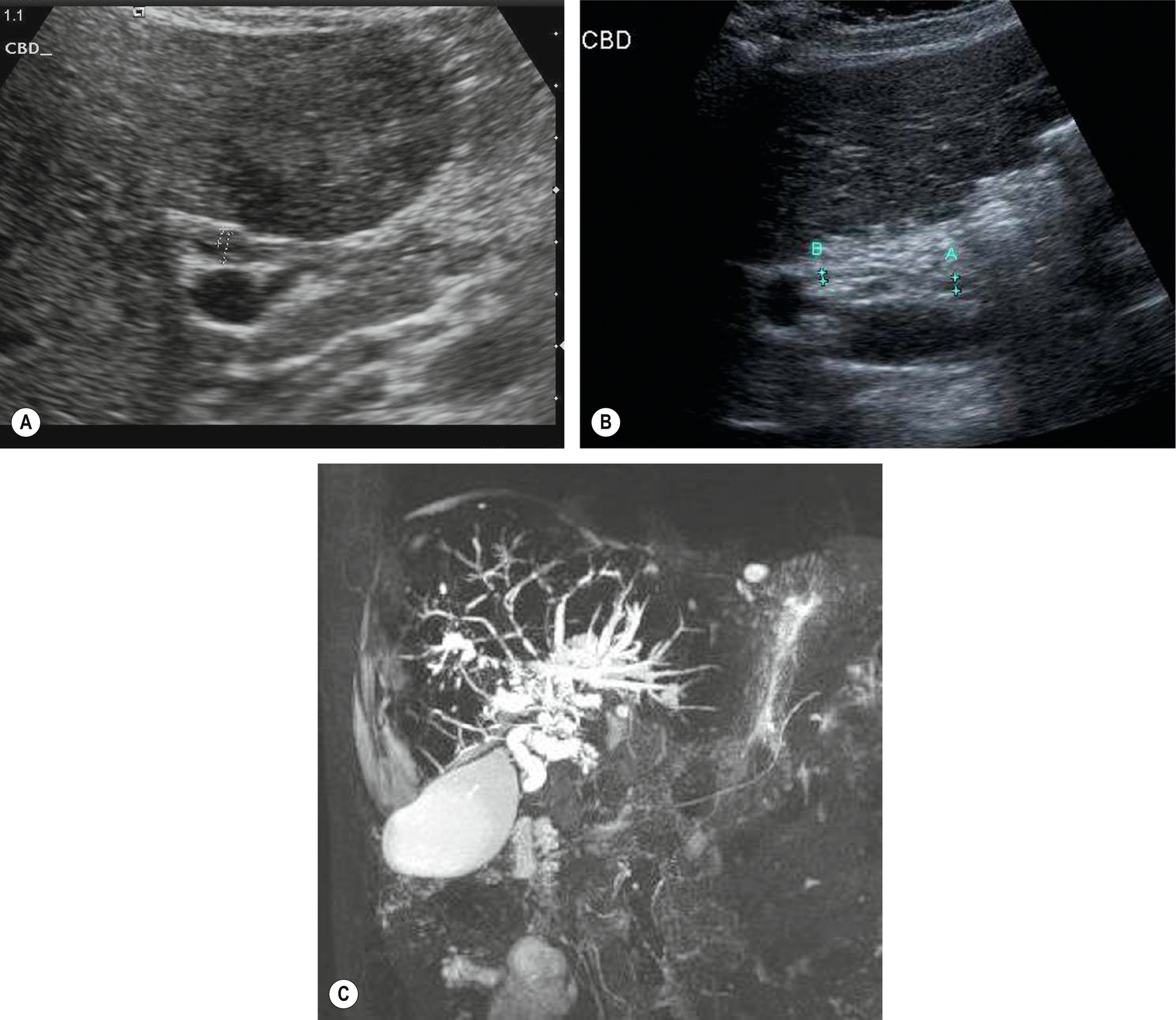
Choledocholithiasis develops in up to 20% of patients with gallstones.4 Stones may pass from the gallbladder into the common duct or may develop de novo within the duct. Stones in the CBD may obstruct the drainage of bile from the liver causing obstructive jaundice. Due to shadowing from the adjacent duodenum, ductal stones are often difficult to demonstrate, and care must be taken to visualize the lower end of the duct if possible (Fig. 3.19).
Usually, CBD stones are accompanied by stones in the gallbladder and a degree of dilatation of the CBD. In these cases, the operator can usually persevere and demonstrate the offending article at the lower end of the duct. However, the duct may be dilated but empty, the stone having recently passed. Stones may be seen to move up and down a dilated duct. This can create a ball-valve effect so that obstruction may be intermittent. It is not unusual to demonstrate a stone in the CBD without stones in the gallbladder, a phenomenon that is also well documented following cholecystectomy. This may be because of a single calculus in the gallbladder having moved into the duct, or stone formation, de novo, within the duct.
It is also important to remember that stones in the CBD may be present without duct dilatation, and attempts to image the entire common duct with ultrasound should always be made, even if it is of normal caliber at the porta (Fig. 3.20).
Other ultrasound signs to look for are shown in Box 3.2.
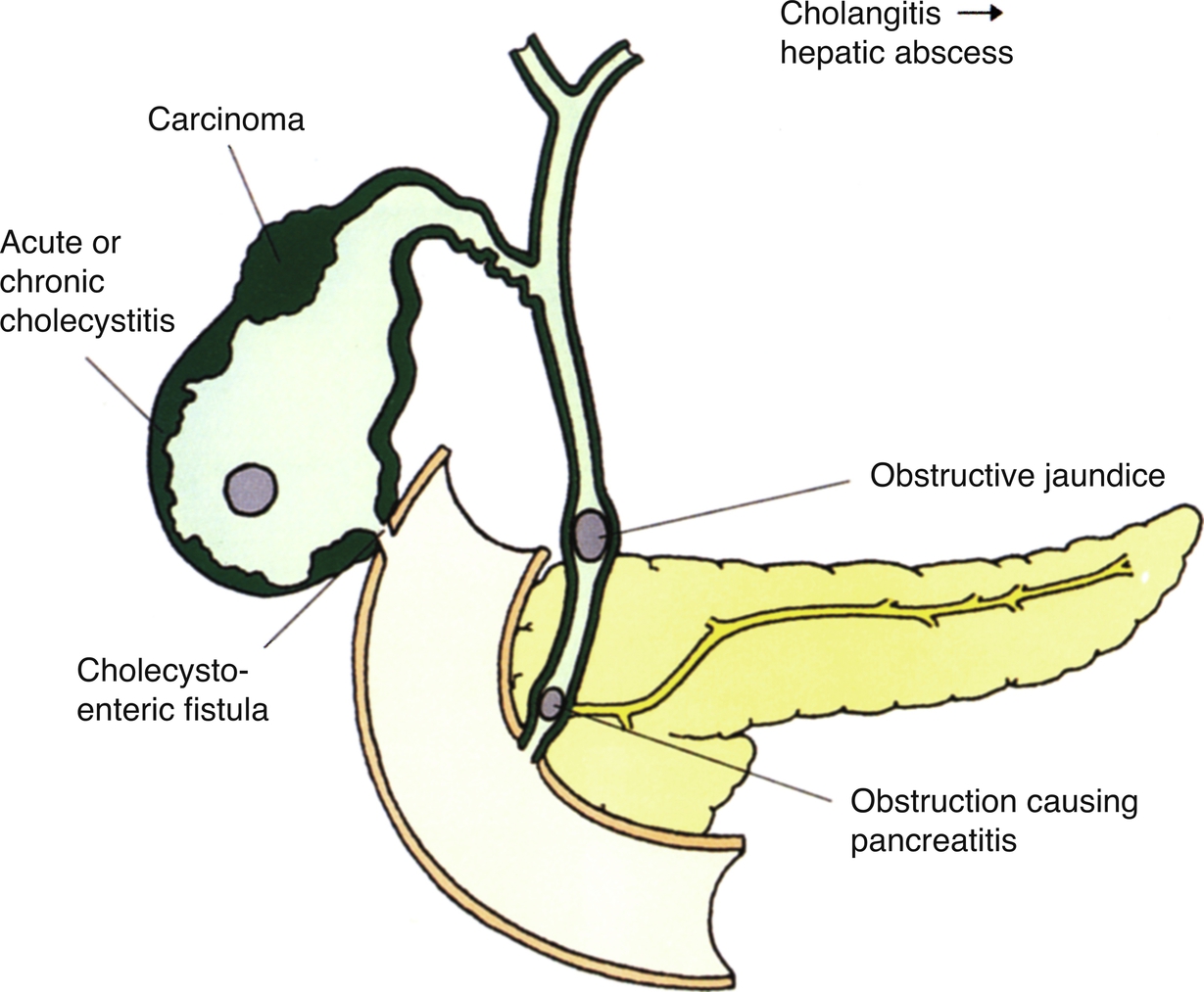
Possible complications of gallstones are outlined in Fig. 3.21. In rare cases, stones may perforate the inflamed gallbladder wall to form a fistula into the small intestine or colon. A large stone passing into the small intestine may impact the ileum, causing an intestinal obstruction.
Biliary Reflux and Gallstone Pancreatitis
A stone may become lodged in the distal CBD near the ampulla. If the main pancreatic duct joins the CBD proximal to this, bile and pancreatic fluid may reflux up the pancreatic duct, causing inflammation and severe pain. Reflux up the CBD may also result in ascending cholangitis, particularly if the obstruction is prolonged or repetitive. Cholangitis may result in dilated bile ducts with mural irregularity on ultrasound, but magnetic resonance cholangiopancreatography (MRCP) is usually superior in demonstrating intrahepatic ductal changes of this nature.
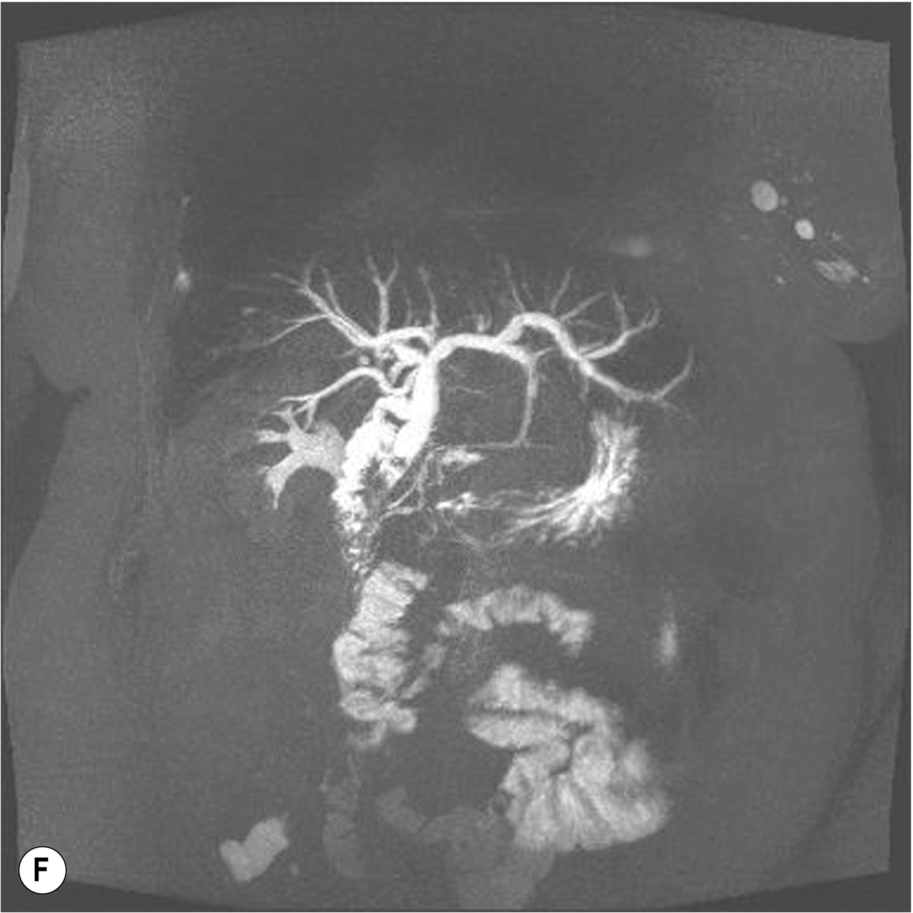
Bile reflux is also associated with anomalous cystic duct insertion (Fig. 3.22A, B), which is more readily recognized on MRCP than ultrasound.
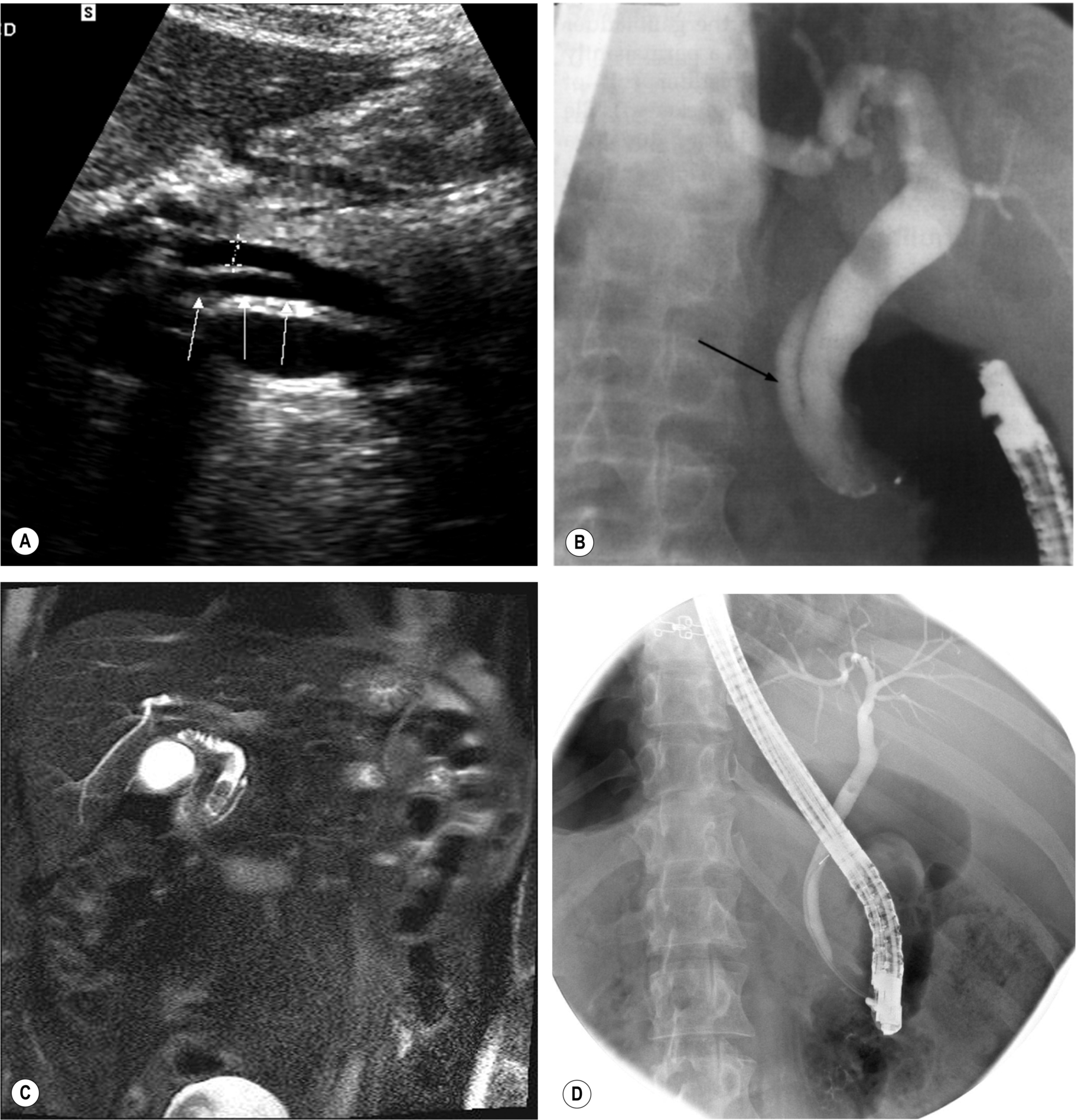
Further Management of Gallstones
MRCP and endoscopic retrograde cholangiopancreatography (ERCP) demonstrate stones in the duct with greater accuracy than ultrasound, particularly at the lower end of the CBD, which may be obscured by duodenal gas on ultrasound (Fig. 3.22C, D).5 ERCP is invasive, carrying a small risk of morbidity or rarely mortality because of perforation, infection, or pancreatitis but has the advantage of providing the therapeutic option of sphincterotomy and stone removal. This is the modality of choice when stones are known to exist in the duct (for example, following MRCP) and has supplanted surgical removal in many cases.6
Laparoscopic cholecystectomy is the preferred method of treatment for symptomatic gallbladder disease in an elective setting. Acute cholecystitis is also increasingly managed by early laparoscopic surgery, with a slightly higher rate of conversion to open surgery than elective cases.7 Laparoscopic ultrasound may be used as a suitable alternative to operative cholangiography to examine the common duct for residual stones during surgery.8 It compares well to cholangiography, with a sensitivity and specificity of 96% and 100%, and avoids any radiation dose, but has been slow to be adopted in the United Kingdom, as it requires specialized equipment and training.9 Both ultrasound and cholescintigraphy are used in monitoring postoperative biliary leaks or hematoma (Fig. 3.23).
Other less common options include dissolution therapy and extracorporeal shock wave lithotripsy. However, these treatments are often only partially successful, require careful patient selection, and also run a significant risk of stone recurrence.10
Enlargement of the Gallbladder
Due to the enormous variation in size and shape of the normal gallbladder, it is impossible to diagnose pathological enlargement by simply using measurements. Three-dimensional techniques may prove useful in assessing gallbladder volume,11 but this is a technique that is only likely to be clinically useful in a minority of patients with impaired gallbladder emptying.
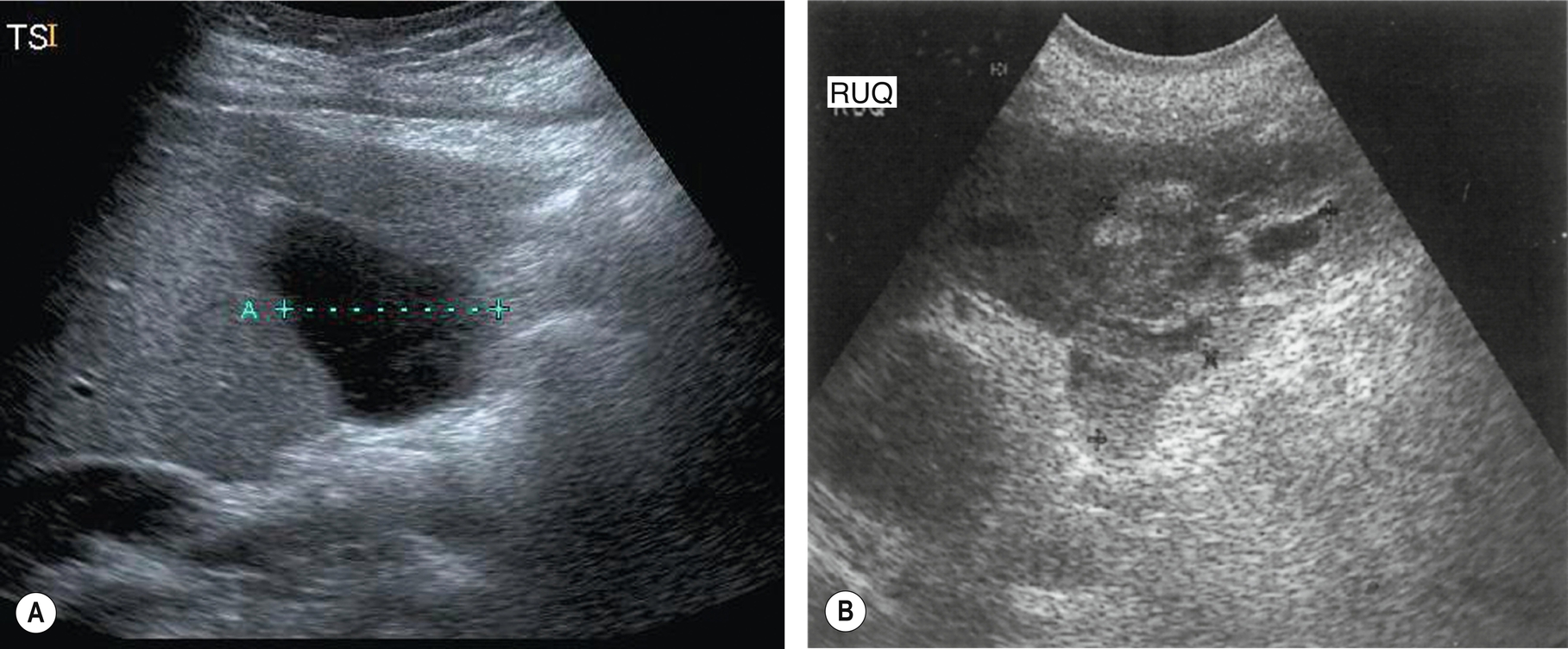
An enlarged gallbladder is frequently referred to as hydropic. It may be because of obstruction of the cystic duct (see below) or associated with numerous disease processes such as diabetes, primary sclerosing cholangitis (PSC), leptospirosis, or in response to some types of drugs. A pathologically dilated gallbladder, as opposed to one which is physiologically dilated, usually assumes a more rounded, tense appearance (Fig. 3.24).
Mucocele of the Gallbladder
If the cystic duct is obstructed – usually by a stone that has failed to pass through to the CBD – the normal flow of bile from the gallbladder is interrupted. Chronic cystic duct obstruction causes the bile to be replaced by mucous secreted by the lining of the gallbladder, resulting in a mucocele. The biliary ducts remain normal in caliber.
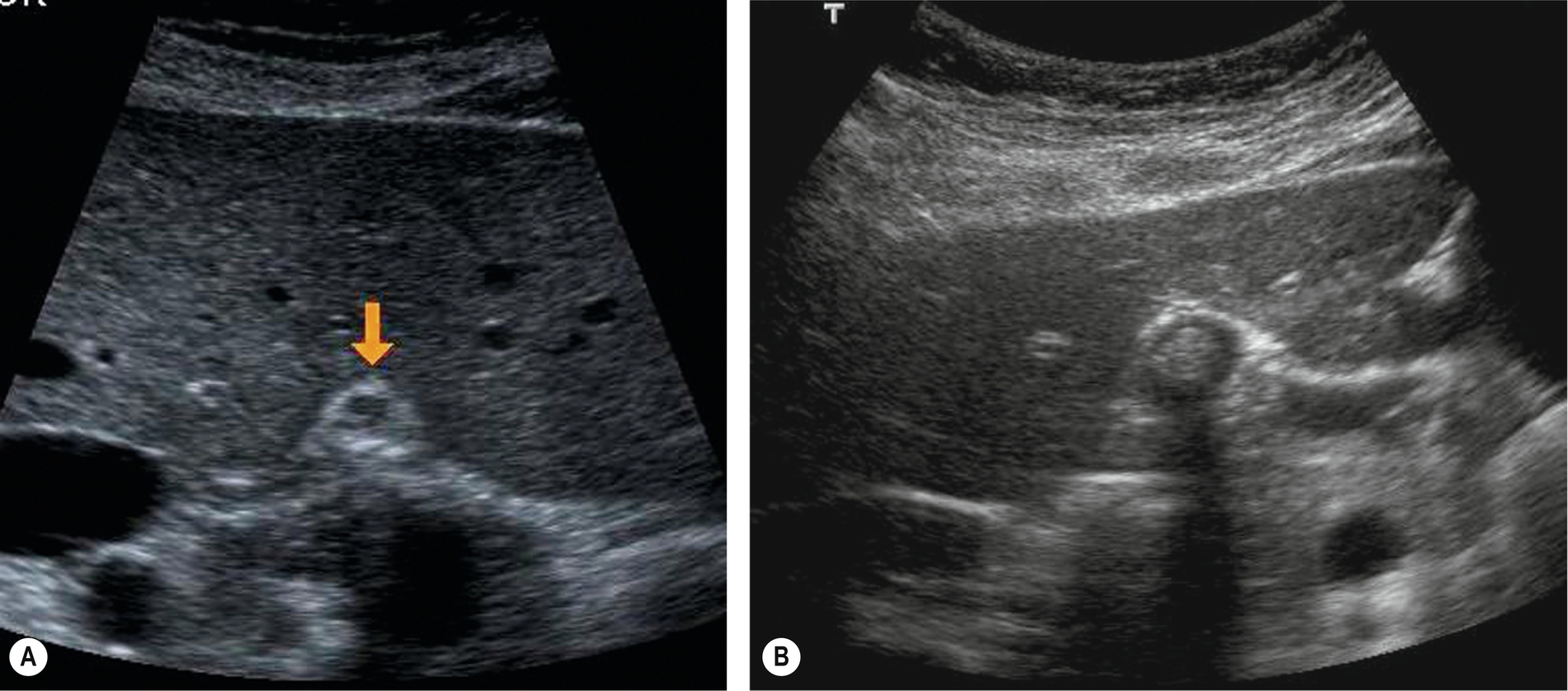
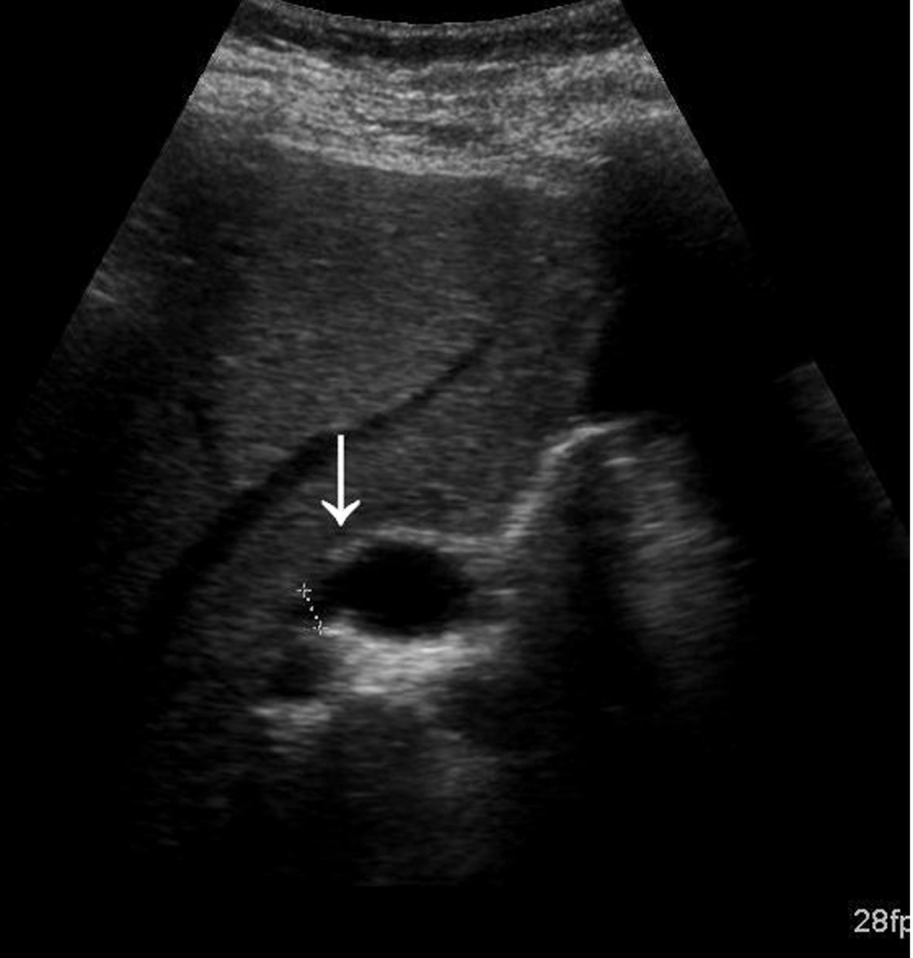
If the gallbladder is dilated in the absence of duct dilatation, do a careful search for an obstructing lesion at the neck; a stone in the cystic duct is more difficult to identify on ultrasound as it is not surrounded by echo-free bile (Fig. 3.18).
Mirizzi’s Syndrome
Mirizzi’s syndrome is a rare cause of biliary obstruction in which compression of the biliary tree is caused by a stone in the adjacent cystic duct. This usually happens in combination with a surrounding inflammatory process which compresses and obstructs the adjacent common hepatic duct, causing distal biliary duct dilatation. This is associated with a low insertion of the cystic duct into the common hepatic duct. Occasionally a fistula forms between the hepatic duct and the gallbladder because of erosion of the duct wall by the stone. Ultimately this may lead to gallstone ileus – small bowel obstruction resulting from migration of a large stone through the cholecystoenteric fistula. If the condition is not promptly diagnosed, recurring cholangitis leading to secondary biliary cirrhosis may result.
On ultrasound, the gallbladder is typically contracted and contains debris. A stone impacted at the neck may be demonstrated together with dilatation of the intrahepatic ducts with a normal caliber lower common duct (Fig. 3.24). The diagnosis is difficult, as it is frequently impossible to rule out carcinoma. Computed tomography (CT) or magnetic resonance imaging (MRI) may assist in this distinction, and ERCP is still considered the “gold standard” especially as it can offer therapeutic stone removal and/or stent placement.12 Endoscopic or intraductal ultrasounds, if available, have improved the diagnostic accuracy of suspected cases.13 Although rare, it is an important diagnosis as cholecystectomy in these cases has a higher rate of operative and postoperative complications.14
The Contracted or Small Gallbladder
Post-Prandial
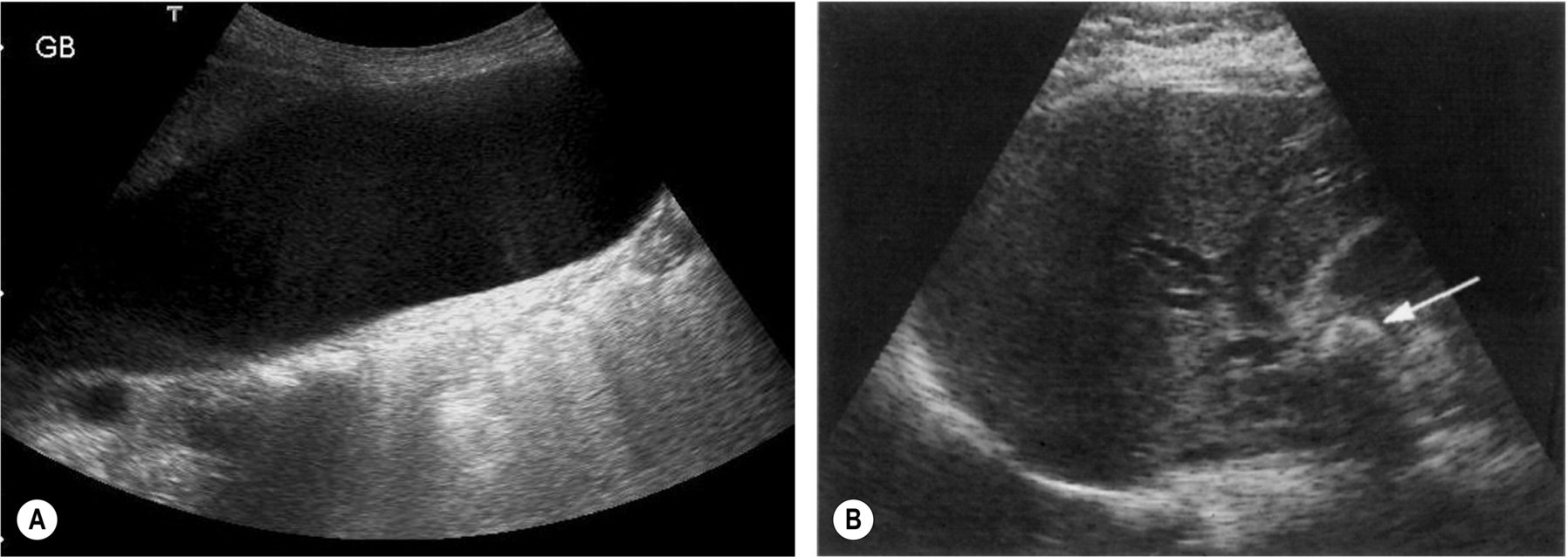
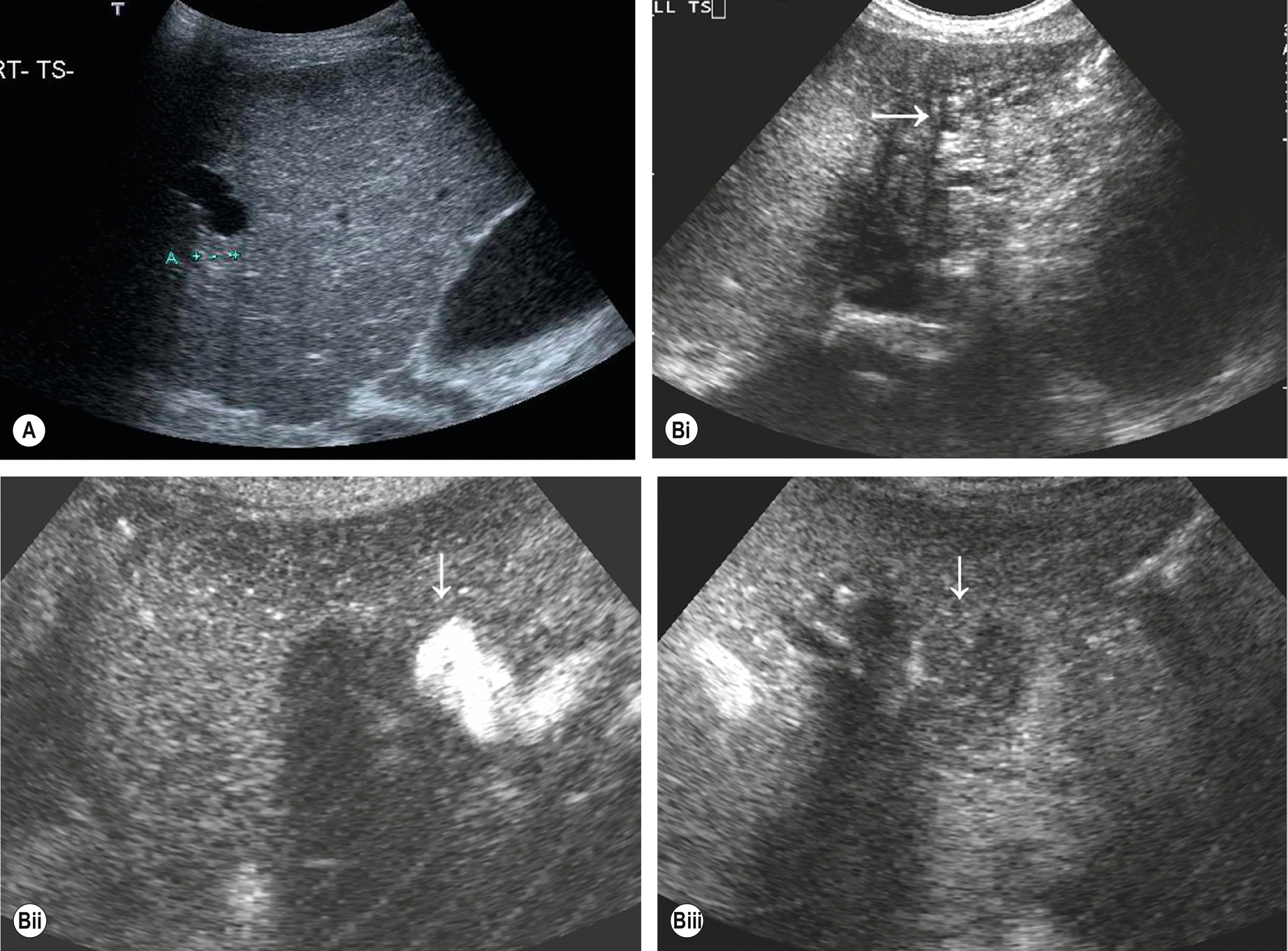
The most likely cause of a contracted gallbladder is physiological, following a meal. (This may still occur despite instructions to fast, and it is always worth enquiring when and what the patient has last eaten or drunk.) The normal gallbladder wall is thickened when contracted, and this must not be confused with a pathological process (Fig. 3.25).
Pathological Causes of a Small Gallbladder
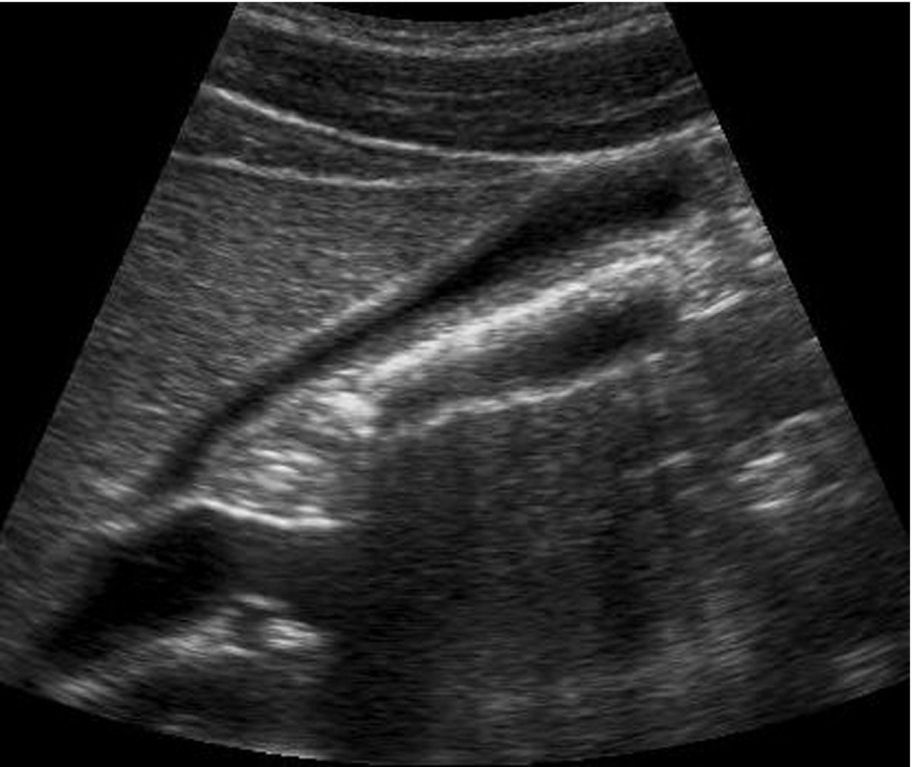
Most pathologically contracted gallbladders contain stones. When the gallbladder cannot be identified, try scanning transversely through the gallbladder fossa, just caudal to the porta hepatis. Strong shadowing alerts the sonographer to the possibility of a contracted gallbladder full of stones.
The reflective surface of the stones and distal shadowing are apparent, and the anterior gallbladder wall can be demonstrated with correct focusing and good technique (Figs. 3.17D, 3.26). Do not confuse the appearances of a previous cholecystectomy when bowel in the gallbladder fossa casts a shadow for a contracted stone-filled gallbladder.
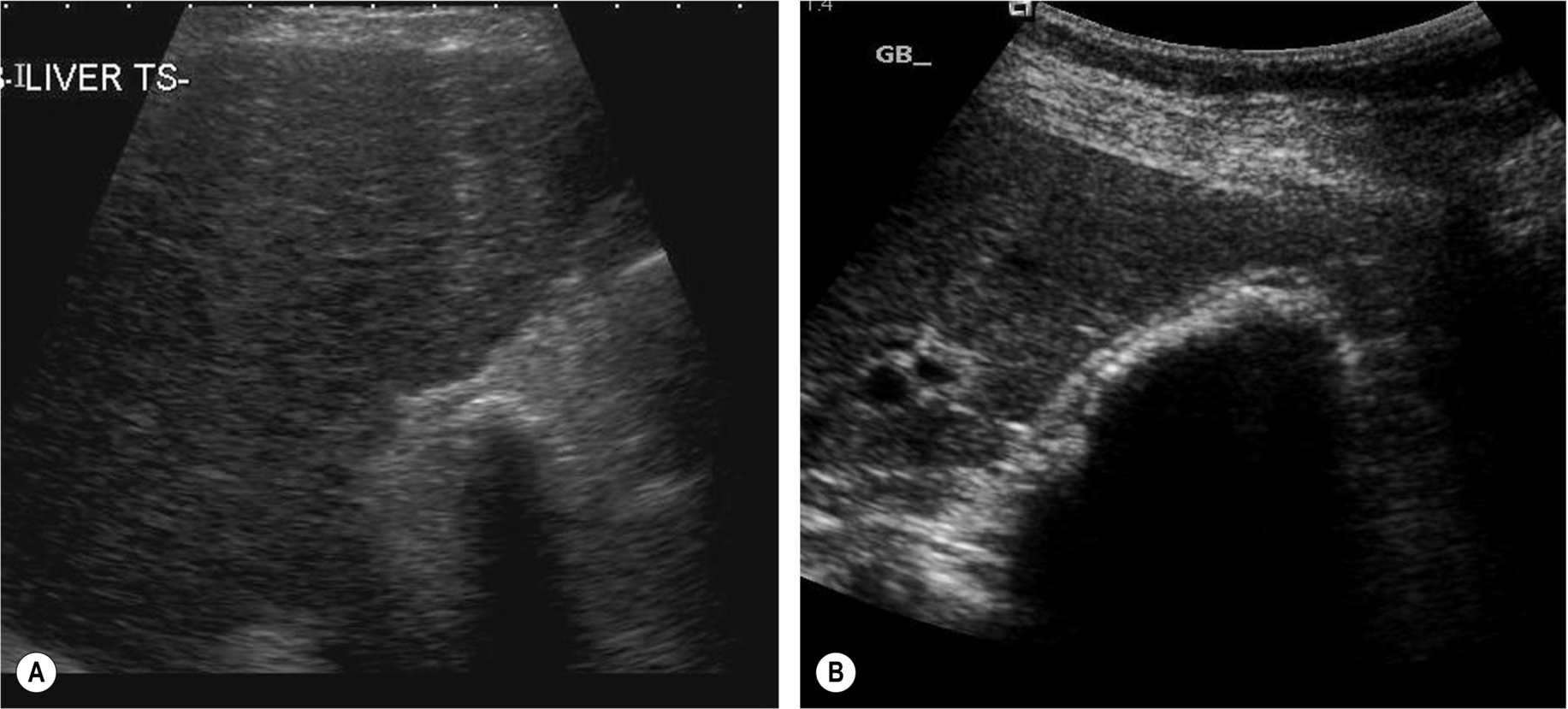
A less common cause of a small gallbladder is the microgallbladder associated with cystic fibrosis (Fig. 3.27). The gallbladder itself is abnormally small, rather than just contracted. Cystic fibrosis also carries an increased incidence of gallstones because of the altered composition of the bile and bile stasis, and the wall might be thickened and fibrosed from cholecystitis.
Porcelain Gallbladder
When the gallbladder wall becomes calcified, the resulting appearance is of a solid reflective structure, causing a distal shadow in the gallbladder fossa (Fig. 3.28). (This can be distinguished from a gallbladder full of stones where the wall can usually be seen anterior to the shadowing [Fig. 3.17D].)
A porcelain gallbladder probably results from a gallbladder mucocele – a long-standing obstruction of the cystic duct, usually from a stone. The bile inside the non-functioning gallbladder is gradually replaced by watery fluid. The wall becomes fibrotic and thickened, and ultimately calcifies.
There is an association between porcelain gallbladder and gallbladder carcinoma; therefore, a prophylactic cholecystectomy is usually performed to preempt malignant development.15
Shadowing from the calcified anterior gallbladder wall can obscure the gallbladder contents and can mimic bowel in the gallbladder fossa. A plain X-ray also clearly demonstrates the porcelain gallbladder.
Hyperplastic Conditions of the Gallbladder Wall
Adenomyomatosis
This is a common, non-inflammatory, hyperplastic condition that causes gallbladder wall thickening. It occurs in around 5% of cholecystectomy specimens and may be mistaken for chronic cholecystitis on ultrasound.
The epithelium that lines the gallbladder wall undergoes hyperplastic change – extending diverticula into the adjacent muscular layer of the wall. These diverticula or sinuses (known as Rokitansky-Aschoff sinuses) are visible within the wall as fluid-filled spaces (Fig. 3.29), which can bulge eccentrically into the lumen and may contain echogenic material or even (normally pigment) stones.
The wall thickening may be focal or diffuse, and the sinuses may be little more than hypoechoic “spots” in the thickened wall or may become quite large cavities in some cases.16 Deposits of crystals in the gallbladder wall frequently result in distinctive “comet-tail” artifacts because of rapid small reverberations of the sound.17
Focal adenomyomatosis most often occurs at the fundus (Fig. 3.29C) and may be difficult to distinguish from carcinoma. [18F]2-fluoro-2-deoxy-D-glucose positron emission tomography (FDG-PET) may be useful in the diagnosis of problem cases.18 Often asymptomatic, it may present with biliary colic, although it is unclear whether this is caused by co-existent stones. Its distinctive appearance allows the diagnosis to be made easily, whether or not stones are present.
Cholecystectomy is performed in symptomatic patients – usually those who also have stones. Although essentially a benign condition, a few cases of associated malignant transformation have been reported, usually in patients with associated anomalous insertion of the pancreatic duct.19
Polyps
Gallbladder polyps are common, usually asymptomatic lesions which are incidental findings in up to 5% of the population. Occasionally they are the cause of biliary colic. They are reflective structures projecting into the gallbladder lumen, which do not cast an acoustic shadow. Unless on a long stalk, they will remain fixed on varying the patient position and are therefore usually distinguishable from stones (Fig. 3.30).
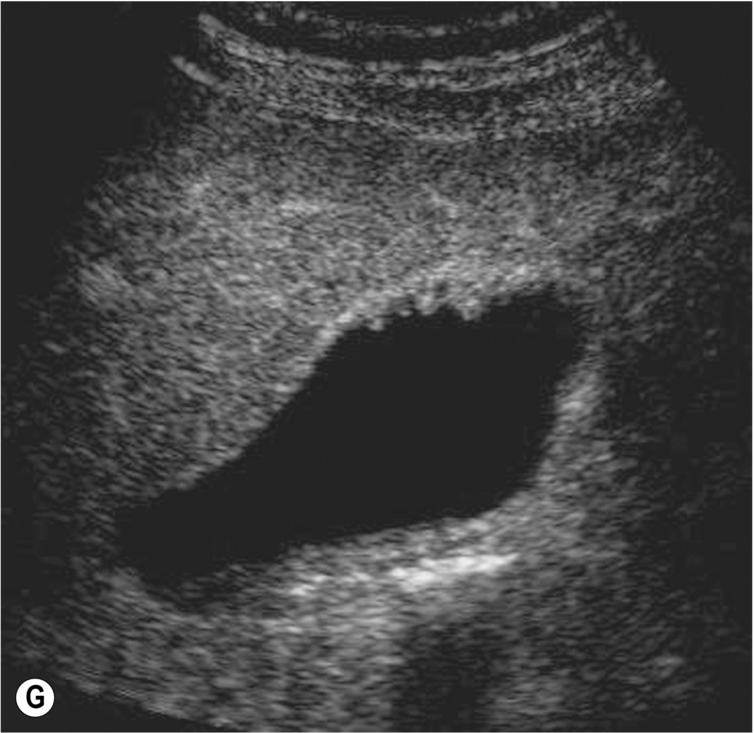
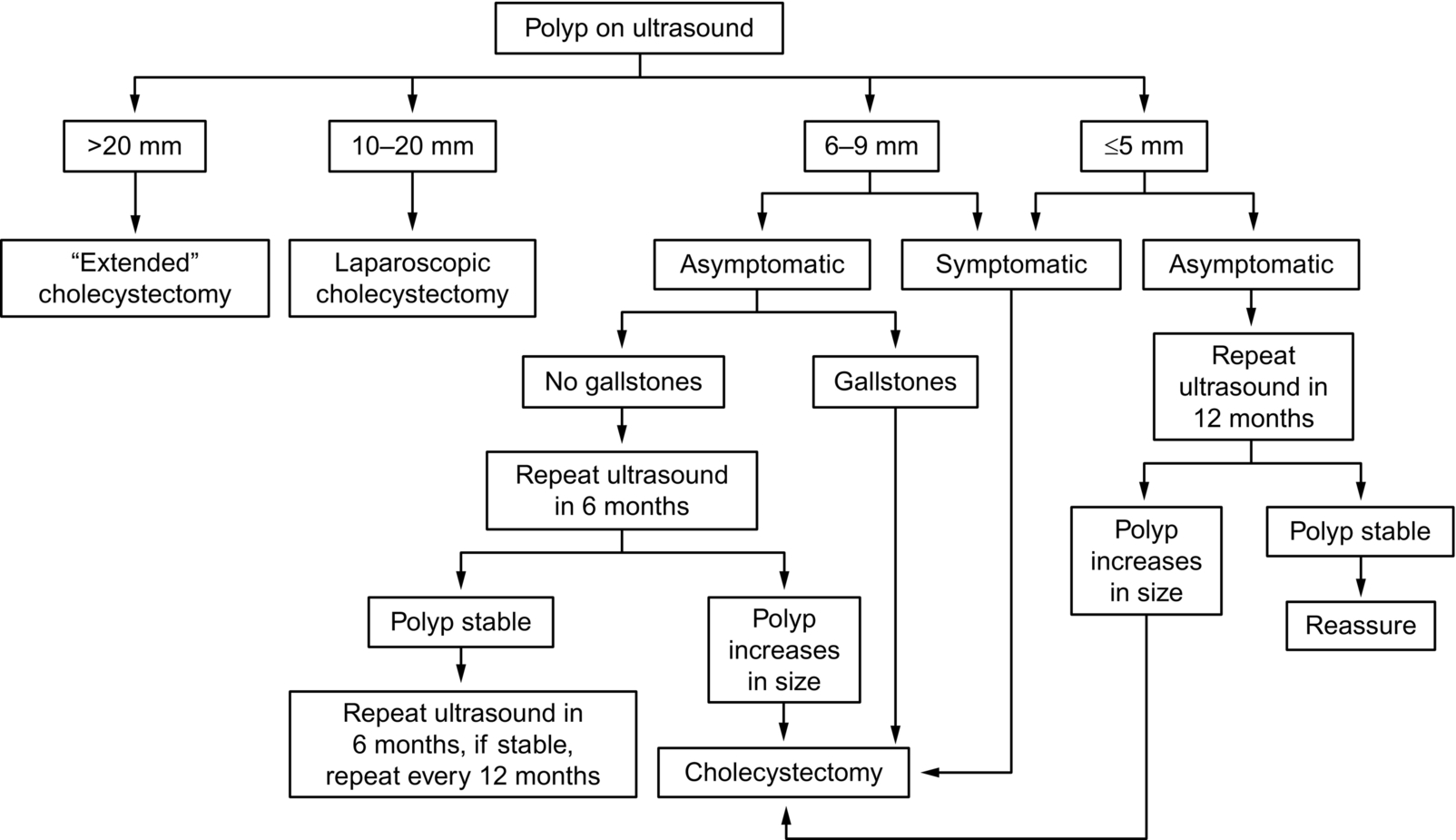
Several types of polyps exist, the most common being cholesterol polyps, which account for around 60% of gallbladder polyps. Others are adenomyomatosis and inflammatory polyps, both of which are also usually benign. True adenomas are less common. There is an association between larger adenomatous gallbladder polyps (> 10 mm) and subsequent carcinoma, especially in patients over 50 years of age. Therefore, the finding of a solitary polyp poses a diagnostic dilemma, and cholecystectomy is often advised (Fig. 3.30D, E). Endoscopic ultrasound (EUS) may provide further information in such cases,20 as it provides high-resolution images of tiny polyps and the layers of the gallbladder wall, and there is some evidence that there is a correlation between EUS appearances and gallbladder polyp histology. Generally, gallbladders in asymptomatic patients with polyps of > 1 cm should undergo rescanning to identify subsequent polyp growth and increased risk of carcinoma, and many clinicians advocate cholecystectomy to preempt malignant transformation.21 In particular, gallbladder polyps in patients with primary sclerosing cholangitis (PSC) have a much greater likelihood of malignancy (40%–60%).22 Smaller polyps of less than 1 cm in diameter may also be safely monitored with ultrasound.23
There is some evidence to suggest that ultrasound contrast agents are useful in demonstrating tumor vascularity in potentially malignant polyps, such as adenomatous polyps.24 However, so far, this technique has proved non-specific, as some benign cholesterol polyps also demonstrate increased vascularity (Fig. 3.30F, G).
Algorithms are being developed for guidance on the management of gallbladder polyps, with collaboration with primary care, hepatobiliary surgeons, and radiology departments. You should make yourself aware of your local departmental policies and guidelines on the management of gallbladder polyps.
Other guidelines also consider the risk factors that the patient may have included being > 50 years old, if they have other conditions such as PSC, and if the patient is of Indian ethnicity. Factors such as sessile (broad-based) polyps are also considered a risk factor (Diagram 3.1). These factors, polyp size and whether the patient is symptomatic then develop a pathway of surgery or surveillance, with small stable polyps only followed up for a maximum of five years. Due to the variations in guidance, it is important that departments develop local protocols to ensure continuity.
Cholesterolosis
Also known as the “strawberry gallbladder,” this gets its name because of the multiple tiny nodules on the surface of the gallbladder mucosal lining. These nodules result from a buildup of lipids in the gallbladder wall and are often not visible on ultrasound. However, in some cases, multiple polyps also form on the inner surface, projecting into the lumen, which are clearly visible on ultrasound (Fig. 3.31). Cholesterolosis may be asymptomatic or may be accompanied by stones and consequently require surgery to alleviate symptoms of biliary colic. The finding of multiple gallbladder polyps should trigger ultrasound follow-up in the asymptomatic patient (see above).
Inflammatory Gallbladder Disease
Cholecystitis is usually associated with gallstones; the frictional action of stones on the gallbladder wall causes some degree of inflammation in almost all cases. The inner mucosa of the wall is injured, allowing the access of enteric bacteria. The inflammatory process may be long-standing and chronic, acute, or a combination of acute inflammation on a chronic background.
Acute Cholecystitis
Acute inflammation of the gallbladder presents with severe right upper quadrant (RUQ) pain localized to the gallbladder area. The pain can be elicited by (gently!) pressing the gallbladder with the ultrasound transducer – a positive ultrasound Murphy’s sign. (This sign, although a useful pointer to acute inflammation, is not specific and can be frequently elicited in other conditions, such as chronic inflammatory cases.)
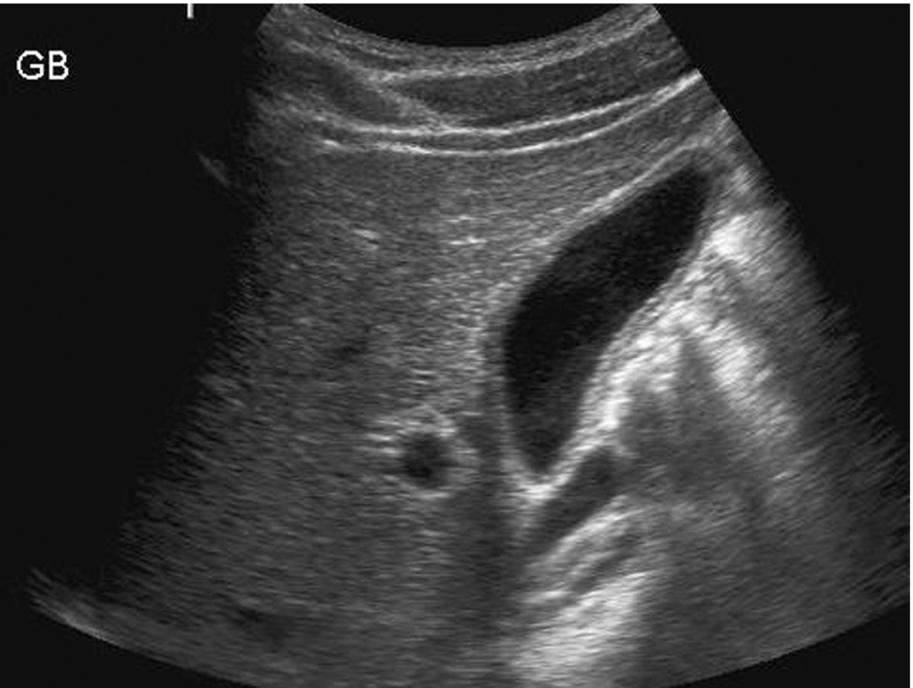
On ultrasound, the gallbladder wall is thickened – greater than 2 mm. This is not in itself a specific sign (Table 3.1), but characteristically the thickening in acute cholecystitis is symmetrical, affecting the entire wall, and there is an echo-poor “halo” around the gallbladder as a result of edematous changes (Fig. 3.32). However, this is not invariable, and focal thickening may be present, or the wall may be uniformly hyperechoic in some cases.
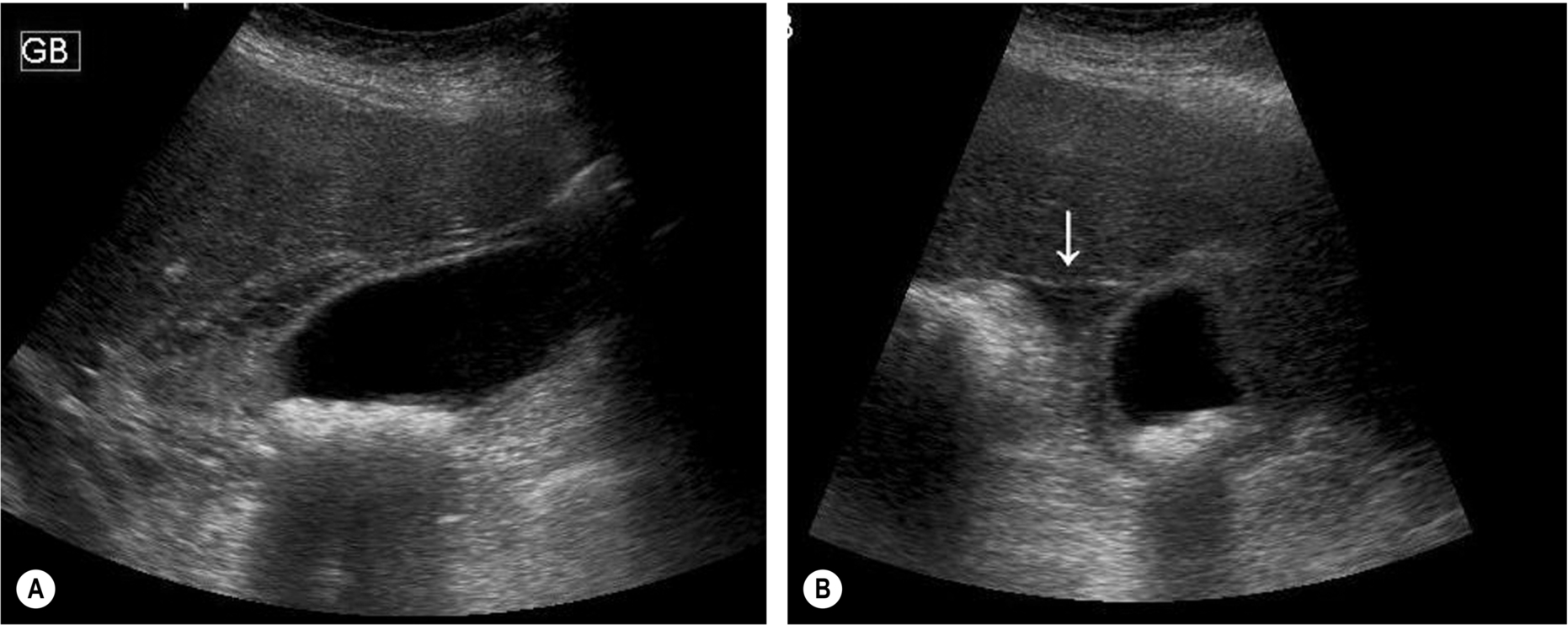
Pericholecystic fluid may also be present, and the inflammatory process may spread to the adjacent liver. Complications may occur if the acute inflammation progresses (see below) because of infection, the formation of pericholecystic abscesses, and peritonitis.
Further Management of Acute Cholecystitis
In uncomplicated acute cholecystitis, analgesia to settle the patient in the short term is followed by laparoscopic removal of the gallbladder. Open surgery is reserved for more complex cases.25,26
If unsuitable for immediate surgery, for example, in cases complicated by peritonitis, the patient is managed with antibiotics and/or percutaneous drainage of pericholecystic fluid or infected bile from the gallbladder usually under ultrasound guidance. This allows the patient’s symptoms to settle and reduces morbidity from the subsequent elective operation. Ultrasound is also useful in guiding bedside cholecystostomy or abscess drainage prior to elective surgical treatment.27
The gallbladder may appear thick-walled in the presence of adjacent inflammatory processes, such as hepatitis (Fig. 3.33). Ascites will often cause a normal gallbladder wall to look edematous and thickened (Fig. 3.34).
Chronic Cholecystitis
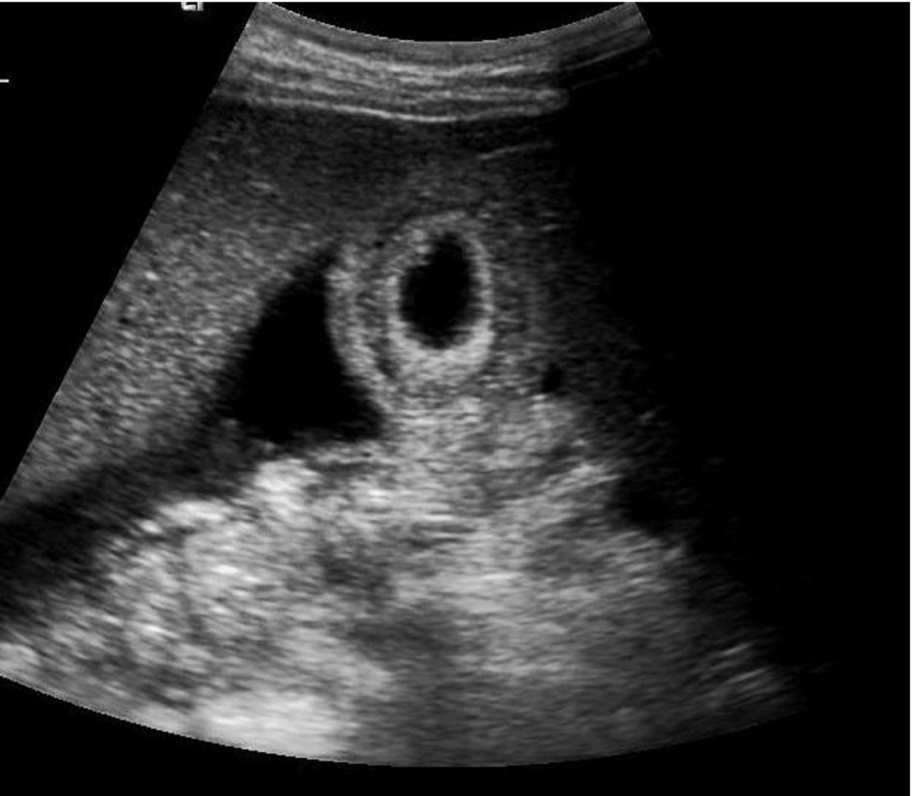
Usually associated with gallstones, chronic cholecystitis presents with lower grade, recurring RUQ pain. The action of stones on the wall causes it to become fibrosed and irregularly thickened, frequently appearing hyperechoic (Fig. 3.35). The gallbladder may be shrunken and contracted, having little or no recognizable lumen around the stones.
Chronic cholecystitis may be complicated by episodes of acute inflammation on a background of the chronic condition.
Most gallbladders that contain stones show at least some histological degree of chronic cholecystitis, even if wall thickening is not apparent on ultrasound.
Acalculous Cholecystitis
Inflammation of the gallbladder without stones is relatively uncommon, accounting for around 10% of cases of acute cholecystitis.28 A thickened, tender gallbladder wall in the absence of any other obvious cause of thickening may be because of acalculous cholecystitis. Traditionally this condition is associated with patients who are already hospitalized and have been fasting, including post-trauma patients, those recovering from surgical procedures, and patients with diabetes. It is brought about by bile stasis leading to a distended gallbladder and subsequently decreased blood flow to the gallbladder. This, especially in the weakened postoperative state, can lead to infection. However, it also occurs in otherwise healthy patients with no history of acute illness or trauma.28
Because no stones are present, the diagnosis is more difficult and may be delayed. Patients with acalculous cholecystitis are more likely to have severe pain and fever by the time the diagnosis is made, increasing the incidence of complications such as perforation.
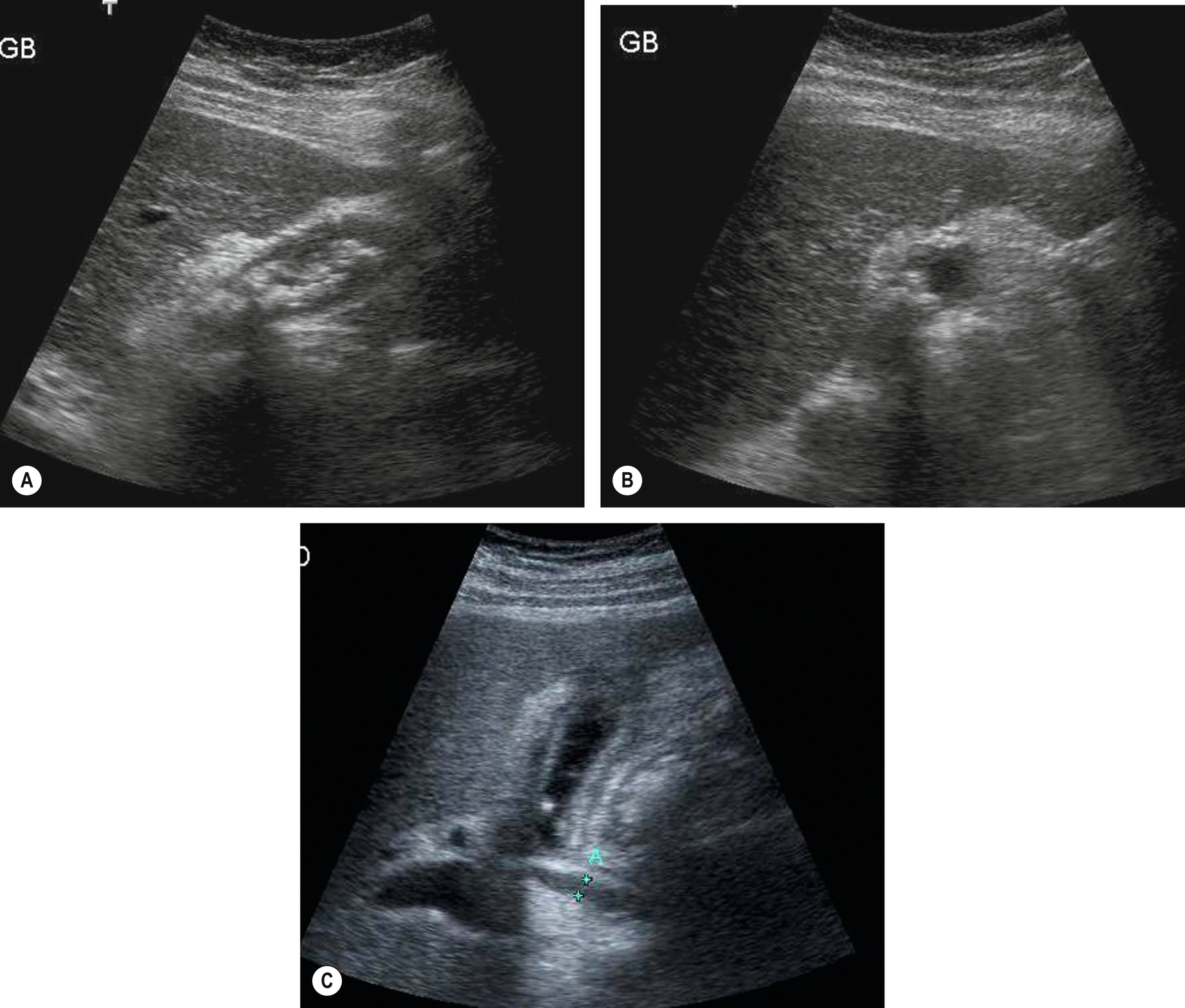
The wall may appear normal on ultrasound in the early stages but progressively thickens (Fig. 3.36). Biliary sludge is usually present, and a pericholecystic abscess may develop in the later stages. A positive Murphy’s sign may help to focus on the diagnosis, but in unconscious patients, the diagnosis is a particularly difficult one.
In patients already critically ill with their presenting disease or following surgery, ultrasound plays a role in guiding percutaneous cholecystostomy at the bedside to relieve the symptoms.29
Chronic acalculous cholecystitis implies a recurrent presentation with typical symptoms of biliary colic but no evident stones on ultrasound. Patients may also demonstrate a low ejection fraction during a cholecystokinin-stimulated hepatic iminodiacetic acid scan. The symptoms are relieved by elective laparoscopic cholecystectomy in most patients, with similar results to those for gallstone disease (although some are found to have a biliary pathologic condition at surgery which might explain the symptoms – such as polyps, cholesterolosis, or biliary crystals/tiny stones, in addition to chronic inflammation).30
Complications of Cholecystitis
Acute on Chronic Cholecystitis
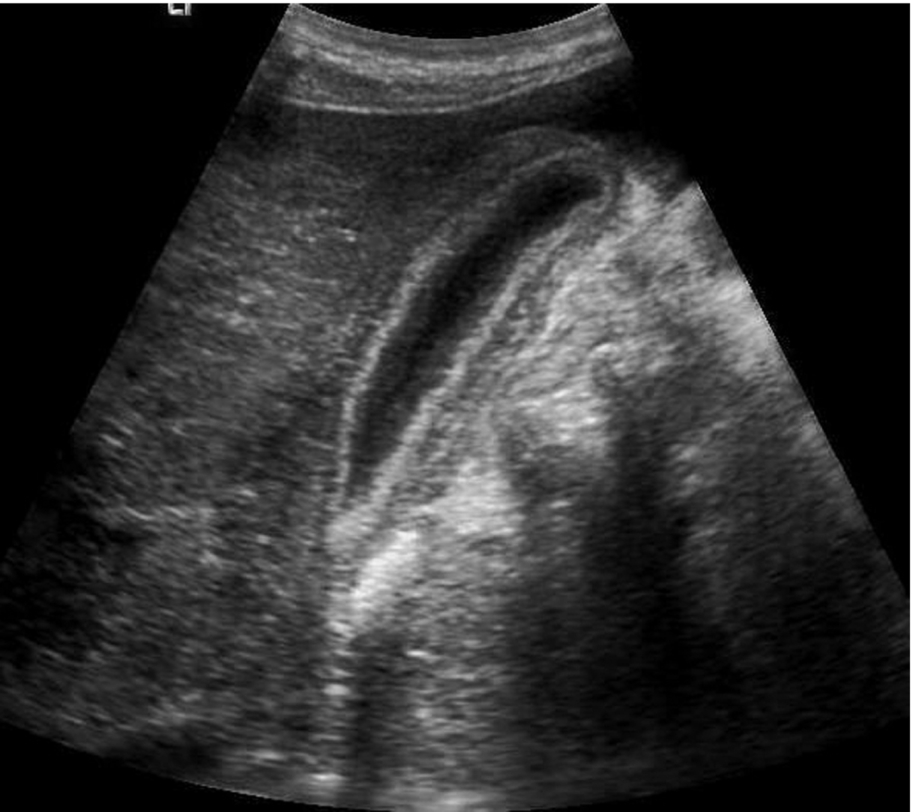
Patients with a long-standing history of chronic cholecystitis may experience (sometimes repeated) attacks of acute inflammation. The gallbladder wall is thickened, as for chronic inflammation, and may become focally thickened with both hypo- and hyperechoic regions. Stones are usually present (Fig. 3.37).
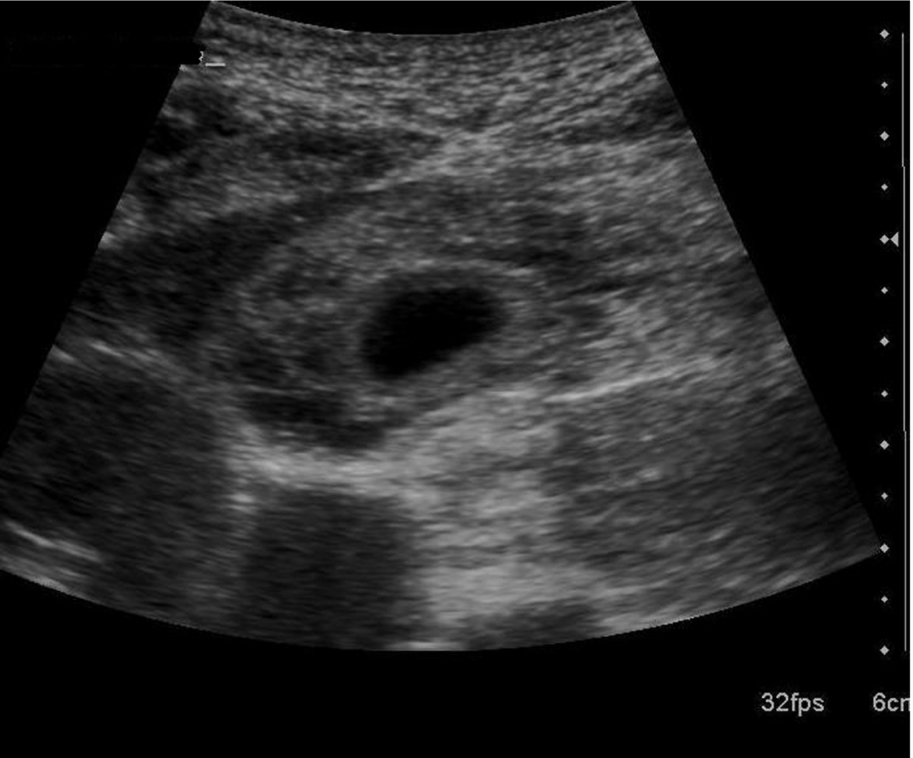
Gangrenous Cholecystitis
In a small percentage of patients, acute gallbladder inflammation progresses to gangrenous cholecystitis. Areas of necrosis develop within the gallbladder wall. The wall itself may bleed, and small abscesses form (Fig. 3.38). This severe complication of the inflammatory process requires immediate cholecystectomy.
The gallbladder wall is friable and may rupture, causing a pericholecystic collection and possibly peritonitis. Inflammatory spread may be seen in the adjacent liver tissue as a hypoechoic, ill-defined area. Loops of the adjacent bowel may become adherent to the necrotic wall, forming a cholecystoenteric fistula. This condition is rare but tends to be associated with older patients, often with a background of diabetes or cardiovascular disease, and carries increased morbidity and mortality.31
The wall is asymmetrically thickened, and areas of abscess formation may be demonstrated. The damaged inner mucosa sloughs off, forming the appearance of membranes in the gallbladder lumen. The gallbladder frequently contains infected debris. The presence of a bile leak may also be demonstrated with hepatobiliary scintigraphy, using technetium99M, which is useful in identifying a bile collection that may otherwise be obscured by bowel on ultrasound.
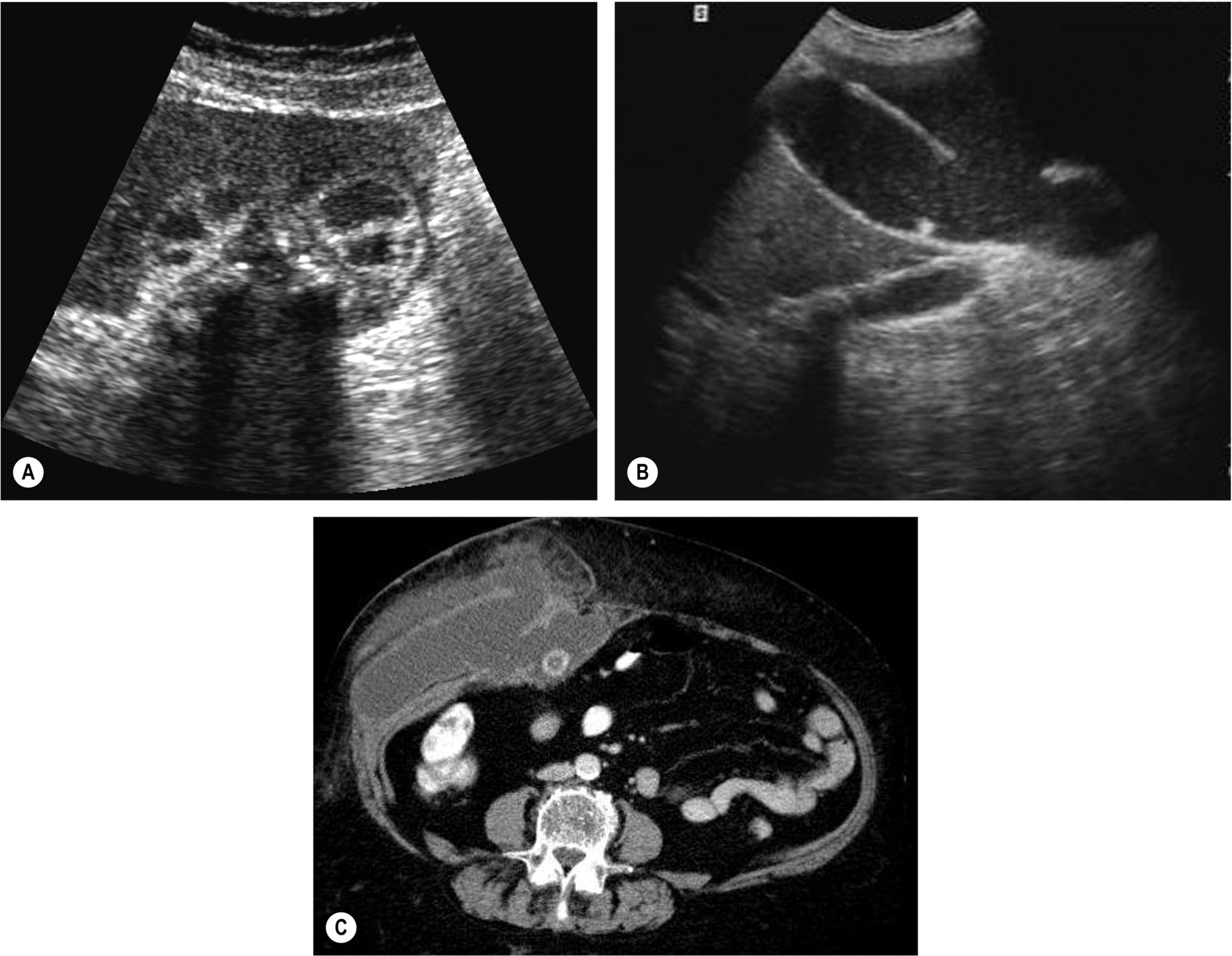
Emphysematous Cholecystitis
This is a form of acute gangrenous cholecystitis in which the inflamed gallbladder may become infected, particularly in diabetic patients with gas-forming organisms. Both the lumen and the wall of the gallbladder may contain air, which is highly reflective, but which casts a “noisy,” less definite shadow than that from stones. Discrete gas bubbles have been reported on ultrasound within the gallbladder wall32 and may also extend into the intrahepatic biliary ducts.33 The air rises to the anterior part of the gallbladder, obscuring the features behind it (Fig. 3.39). This effect may mimic air-filled bowel on ultrasound.
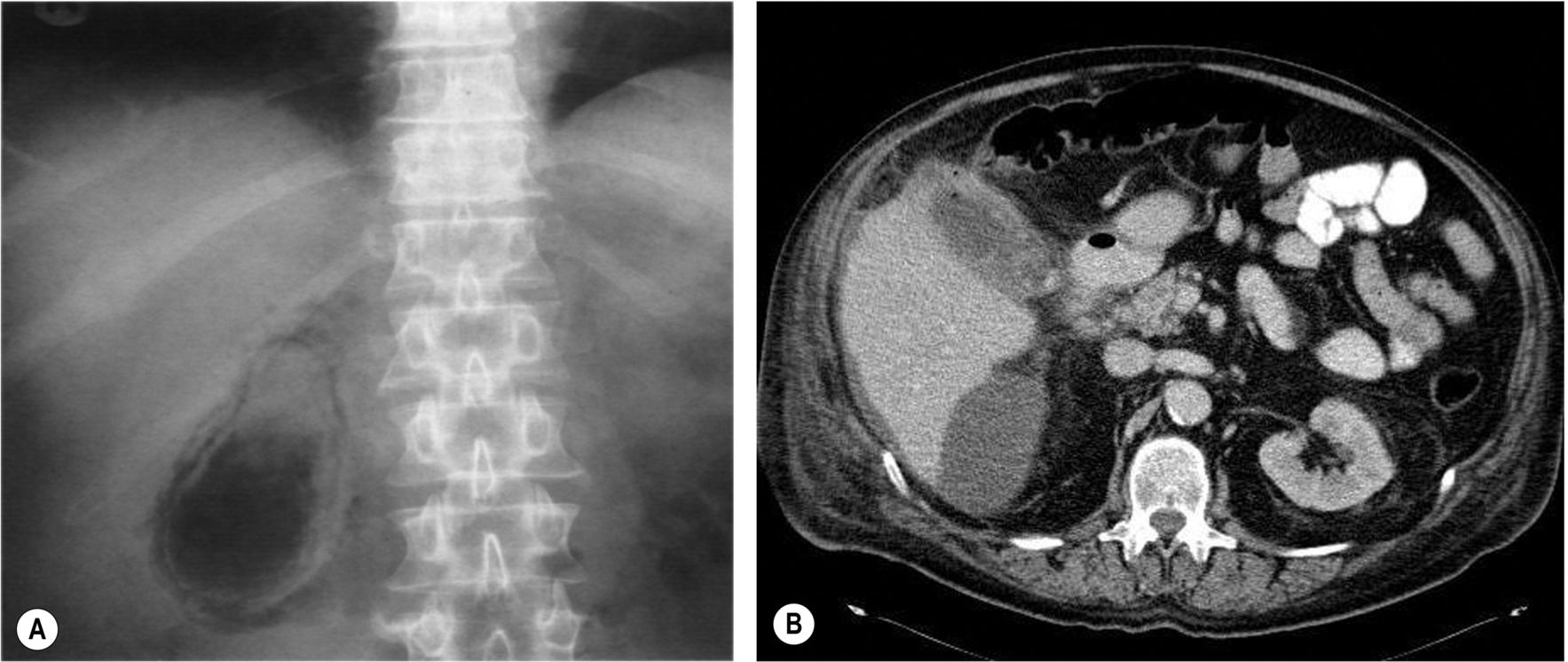
Emphysematous cholecystitis has traditionally had a much higher mortality rate than other forms of cholecystitis, requiring immediate cholecystectomy. However, improvements in ultrasound resolution and the early clinical recognition of this condition suggest that it is now being diagnosed earlier and may be managed more conservatively. The gas in the gallbladder may be confirmed on a plain X-ray (Fig. 3.40), but ultrasound is more sensitive in demonstrating the earlier stages.
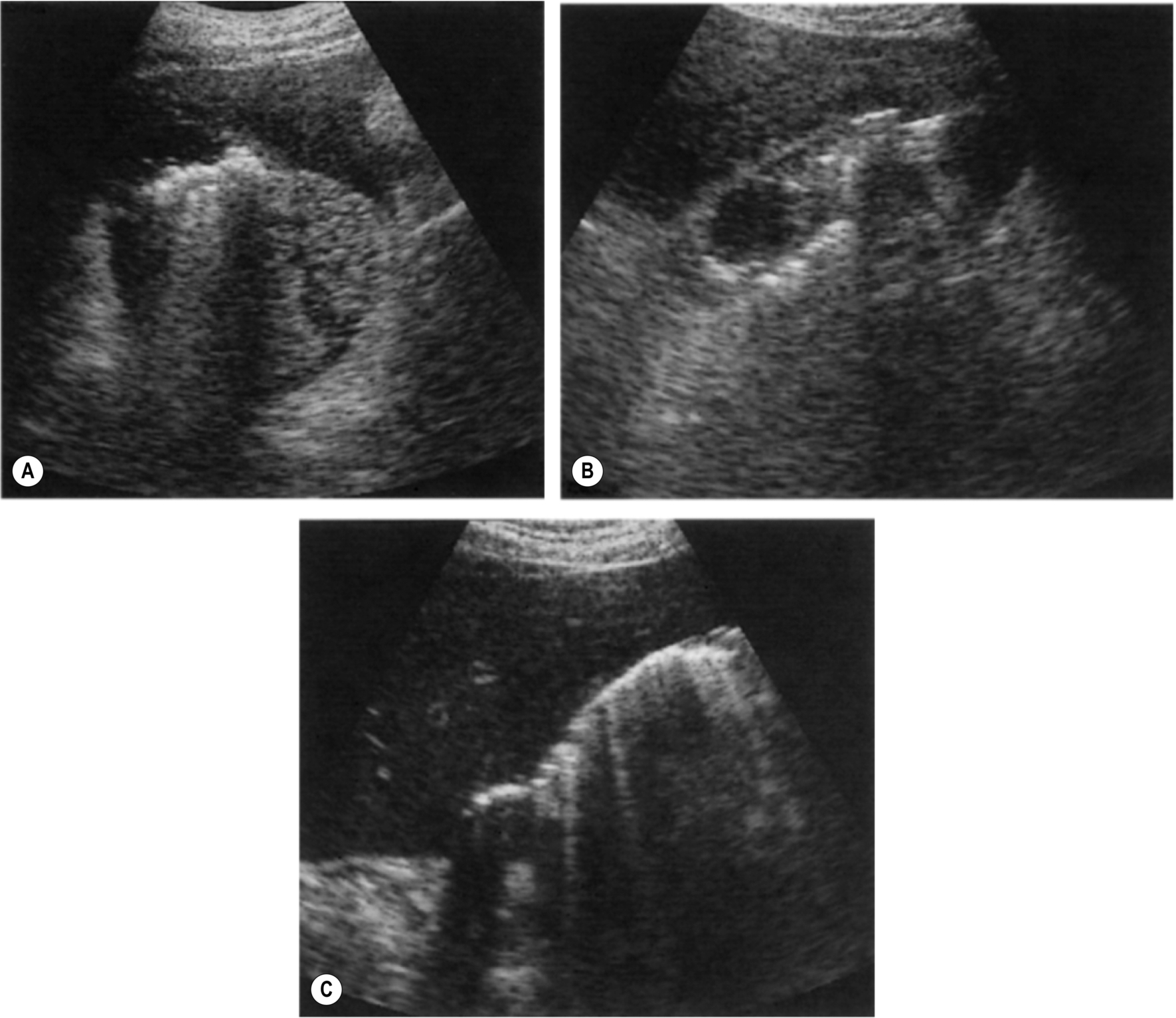
Gallbladder Empyema
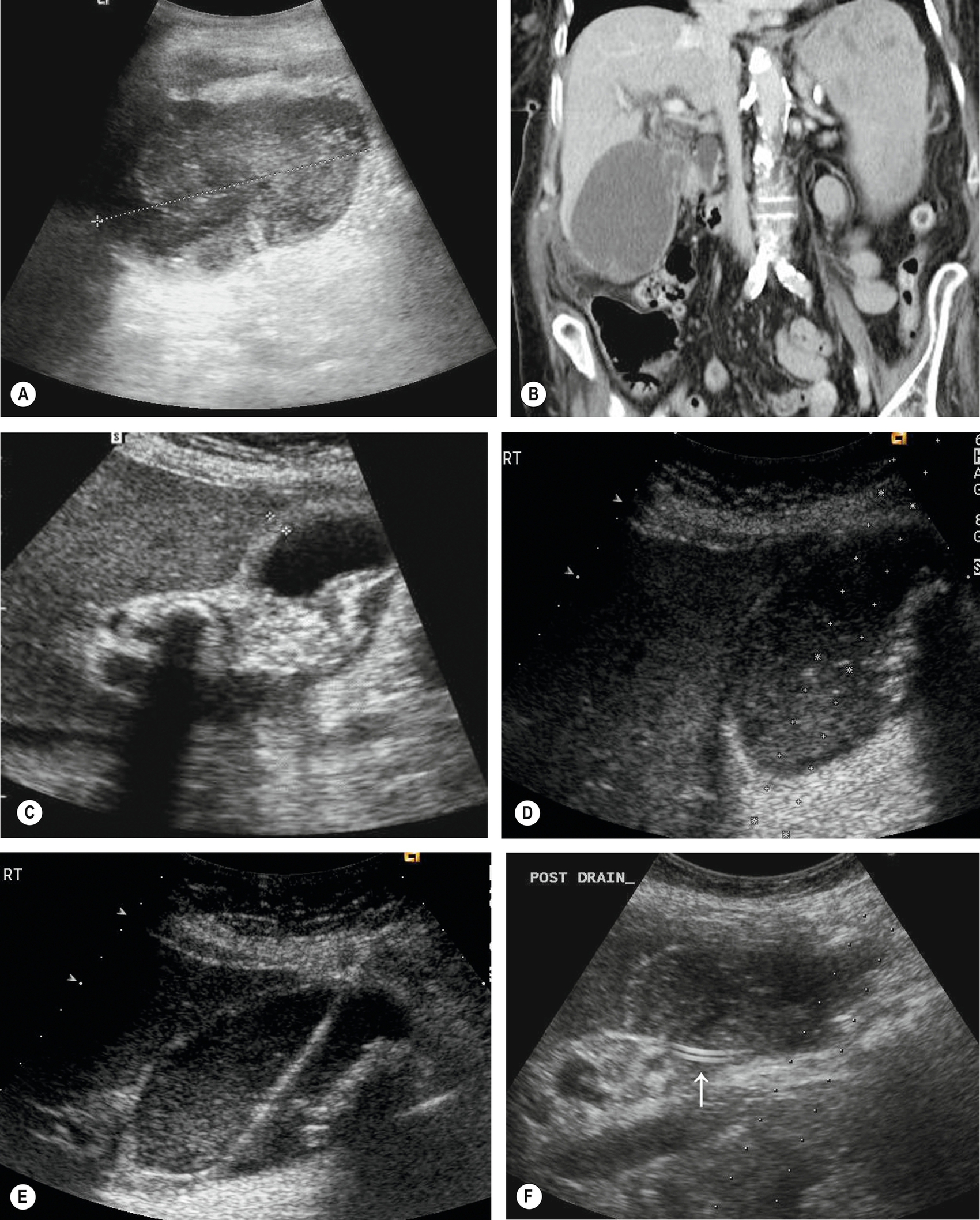
Empyema is a complication of cholecystitis in which the gallbladder becomes infected behind an obstructed cystic duct. Fine echoes caused by pus are present in the bile (Fig. 3.41). These patients may initially present with the usual features of acute cholecystitis but may then progress to become very ill with fever and acute pain. Localized perforation of the gallbladder wall may cause leakage of infected bile to form a pericholecystic gallbladder collection with subsequent peritonitis. Ultrasound may be used to guide bedside drainage (Fig. 3.41D–F) to allow the patient’s symptoms to settle before surgery is attempted.34
Biliary Tree Dilatation
Obstructive Jaundice and Biliary Duct Dilation
Obstruction of the biliary tree most commonly occurs around the distal (lower) end of the common duct and almost always results in dilatation of the bile ducts within the liver. In a few cases, it is possible to have an obstruction that fails to dilate the biliary tree, and it is also possible for the biliary tree to be dilated but not obstructed. However, most cases of biliary duct dilatation are caused by obstruction of the common duct, the most common causes being stones or a neoplasm of the bile duct, ampulla, or head of the pancreas. Up to 20% of patients with gallbladder stones will develop choledocholithiasis.4
The patient with obstructive jaundice may present with upper abdominal pain, abnormal LFTs (see Chapter 2), and if the obstruction is not intermittent, the sclera of the eye and the skin adopt a yellow tinge.
Assessment of the Level of Obstruction
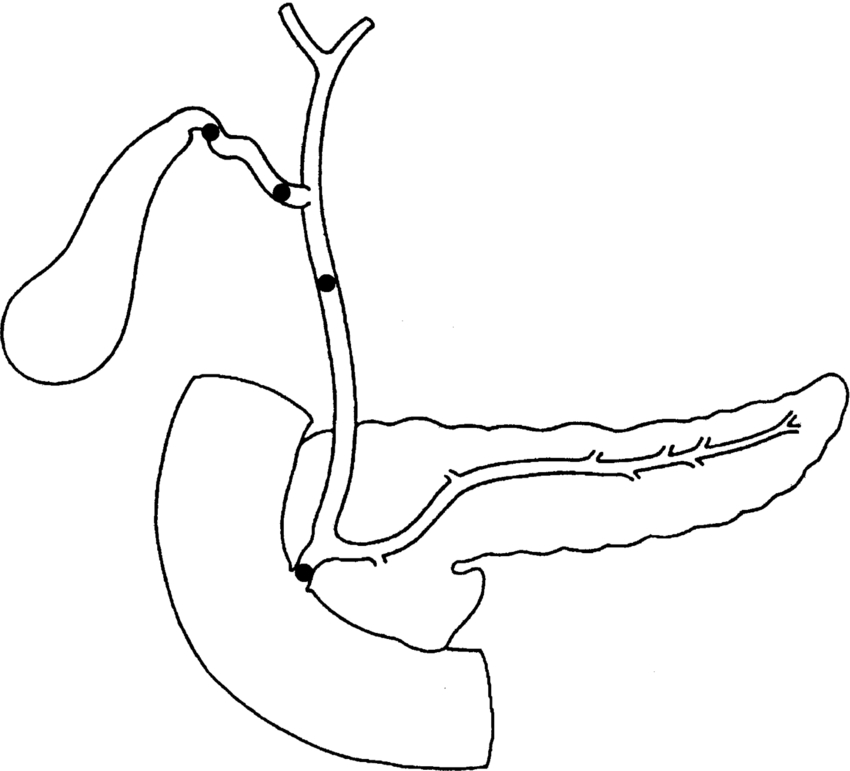
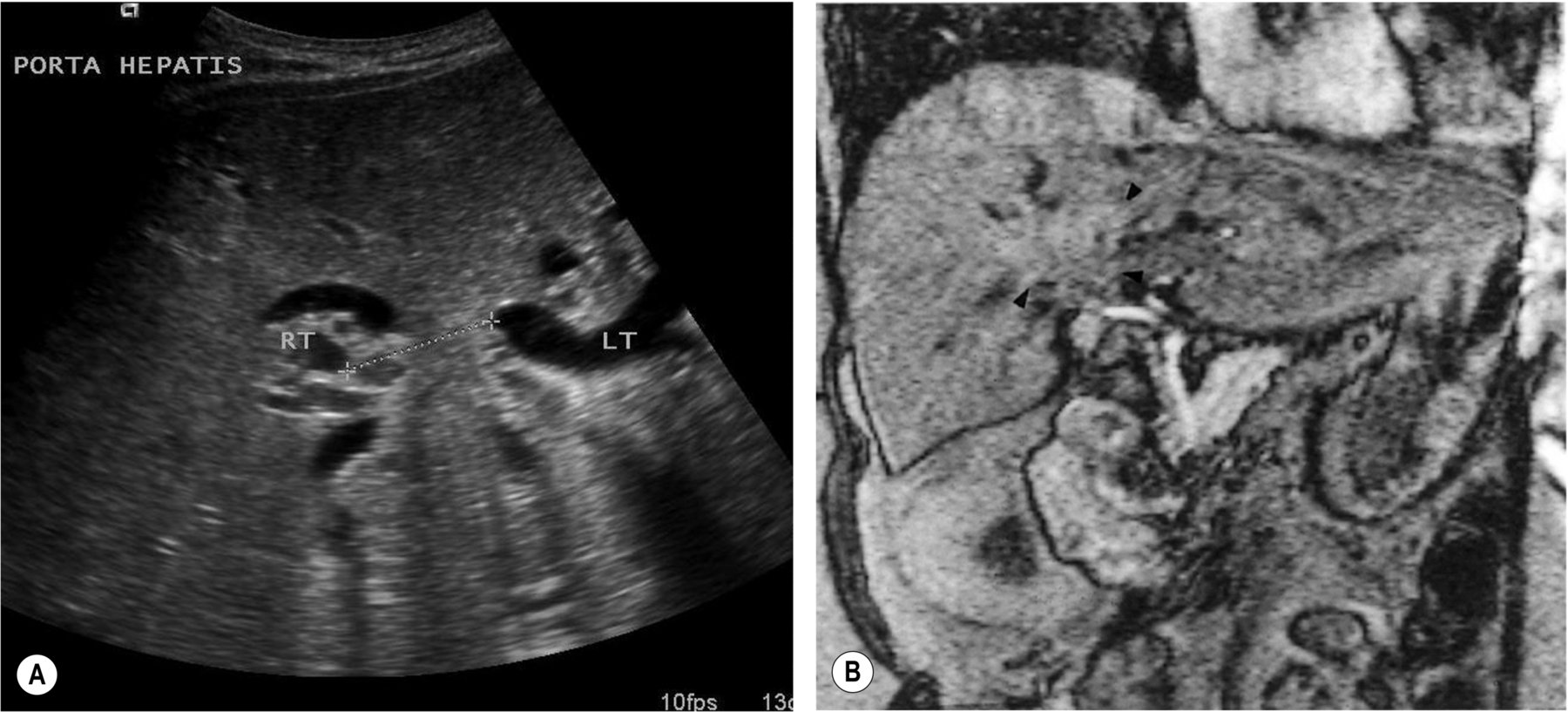
It is possible for the sonographer to work out where the obstructing lesion is situated by observing which parts of the biliary tree are dilated (Fig. 3.42):
- • Dilatation of the CBD (i.e., that portion of the duct below the cystic duct insertion) implies obstruction at its lower end.
- • Dilatation of both biliary and pancreatic ducts implies obstruction distally at the head of the pancreas or ampulla of Vater. This is more likely to be because of carcinoma of the head of the pancreas, ampulla, or acute pancreatitis than a stone. However, it is possible for a stone to be lodged just distal to the confluence of the biliary and pancreatic ducts.
- • Dilatation of the gallbladder alone (i.e., without ductal dilatation) is usually caused by obstruction at the neck or cystic duct (Fig. 3.18).
To assess whether the gallbladder is pathologically dilated may be difficult on ultrasound. The dilated gallbladder will have a rounded, bulging shape because of the increase in pressure inside it.
A gallbladder whose wall has become fibrosed from chronic cholecystitis because of stones will often lose the ability to distend so that the biliary ducts can look grossly dilated despite the gallbladder remaining “normal” in size or contracted.
Early Ductal Obstruction
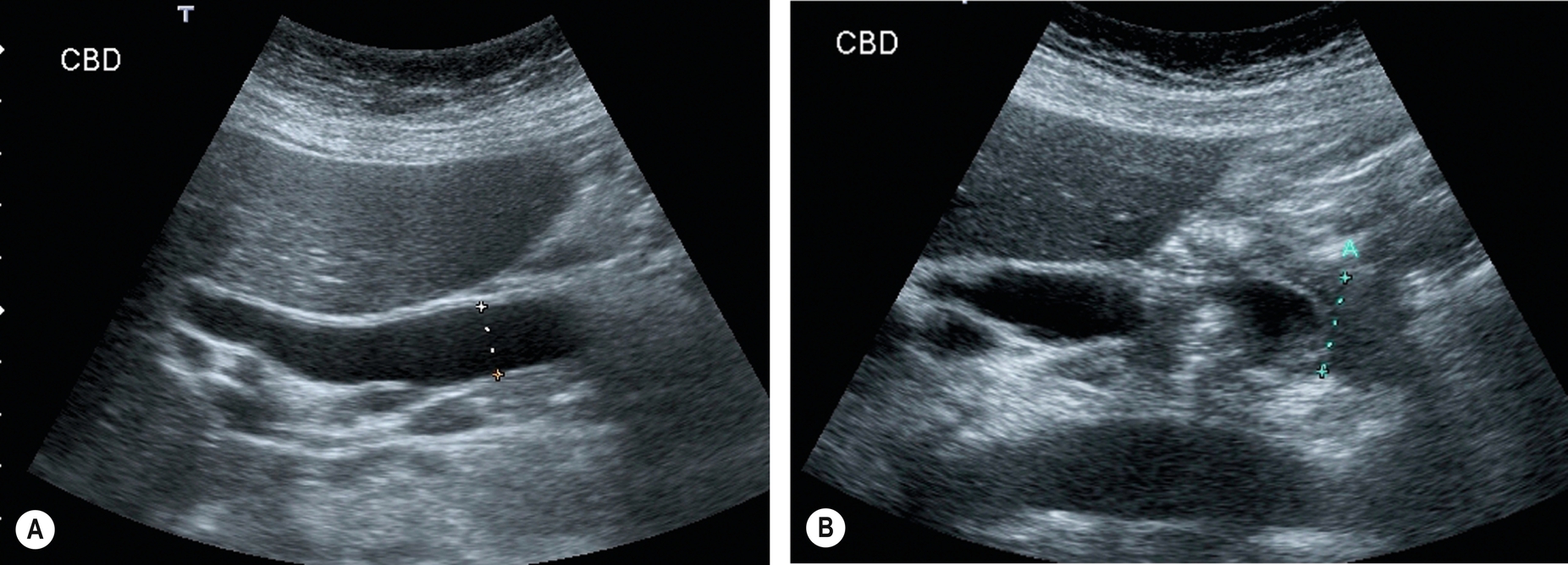
Beware of very early common duct obstruction before the duct becomes obviously dilated, the duct may be mildly dilated at the lower end, just proximal to a stone, but normal in its intrahepatic portion. Likewise, intermittent obstruction by a small stone at the lower end of the duct may be non-dilated when the scan is performed (Fig. 3.20).
A significant ultrasound feature in the absence of any other identifiable findings is that of thickening of the wall of the bile duct. This not only represents an inflammatory process in the duct wall, which may be found in patients with small stones in a non-dilated duct but is also associated with sclerosing cholangitis.35
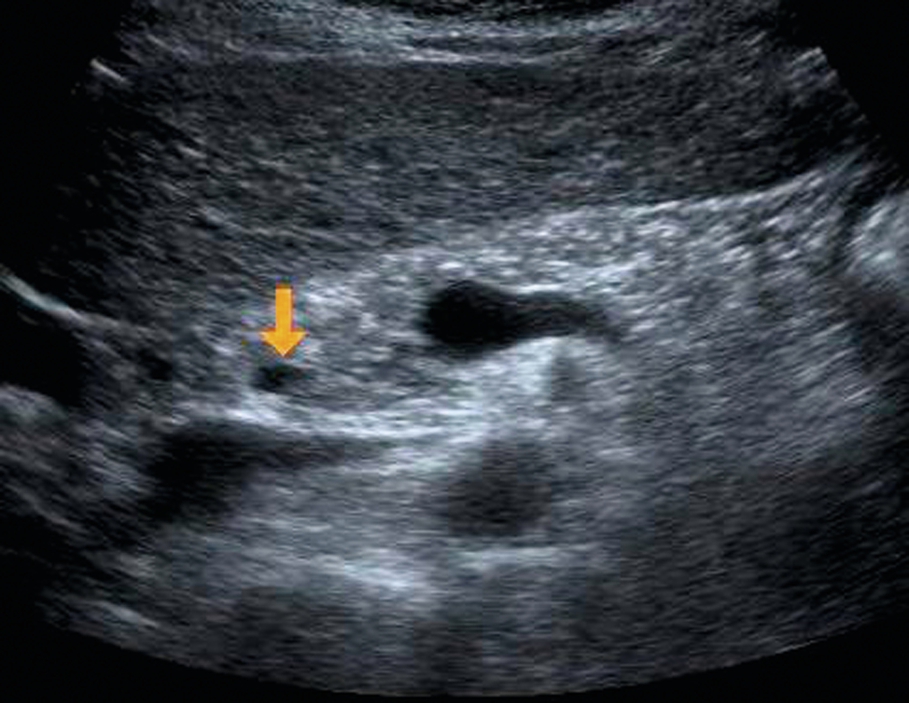
It is sometimes technically difficult in some patients (particularly those with diffuse liver disease) to determine whether a tubular structure on ultrasound represents a dilated duct or a blood vessel. Color Doppler will differentiate the dilated bile duct from a branch of the hepatic artery or portal vein (Fig. 3.43), provided that the vein is not thrombosed, of course.
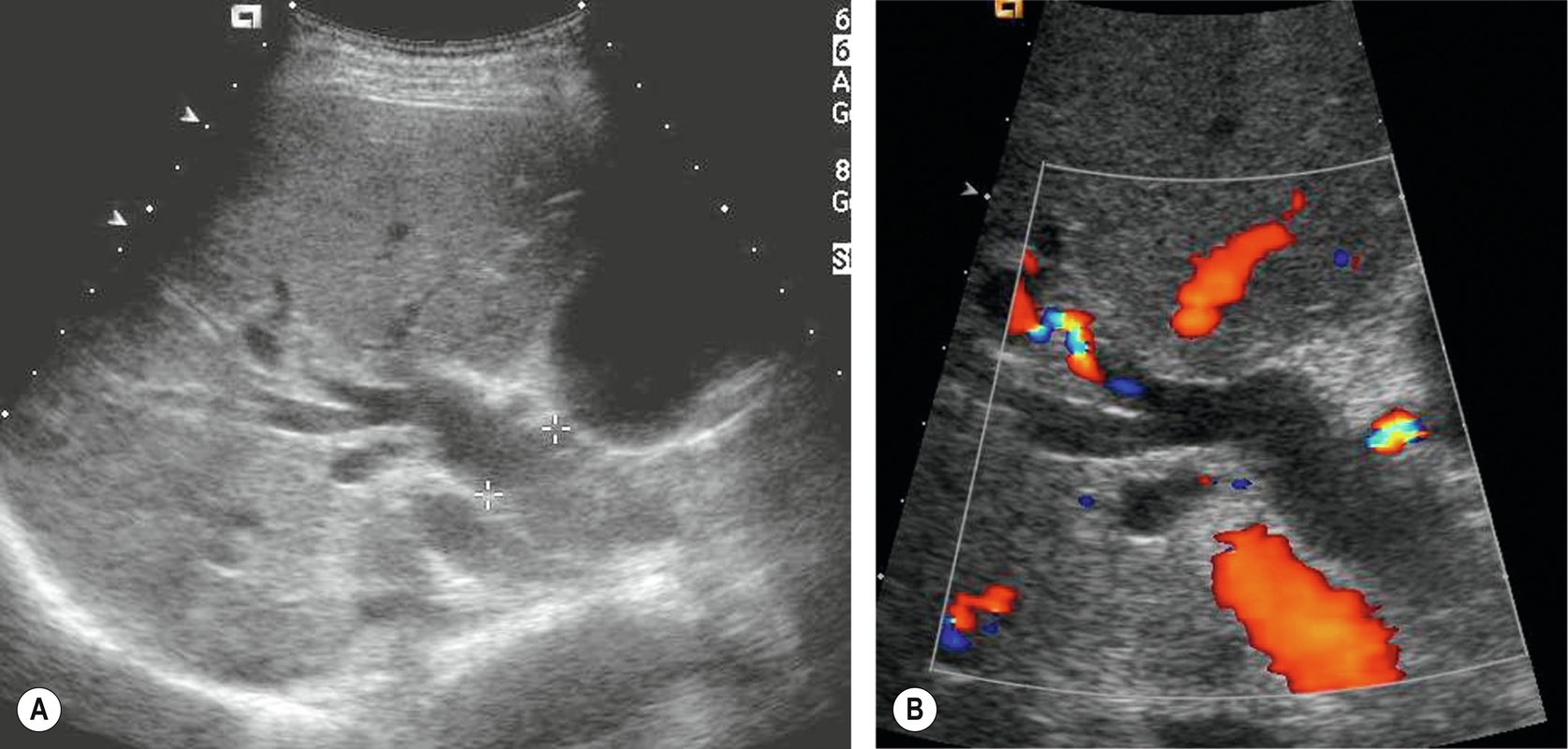
It is not useful to use color Doppler routinely to locate the CBD; the angle of insonation is frequently perpendicular to the beam, so the vein may display no color signal, confusing the operator further. In addition, the application of color Doppler reduces the line density and image resolution – exactly the opposite of what you require when searching for a small duct.
Diagnosis and Assessment of the Cause of Obstruction
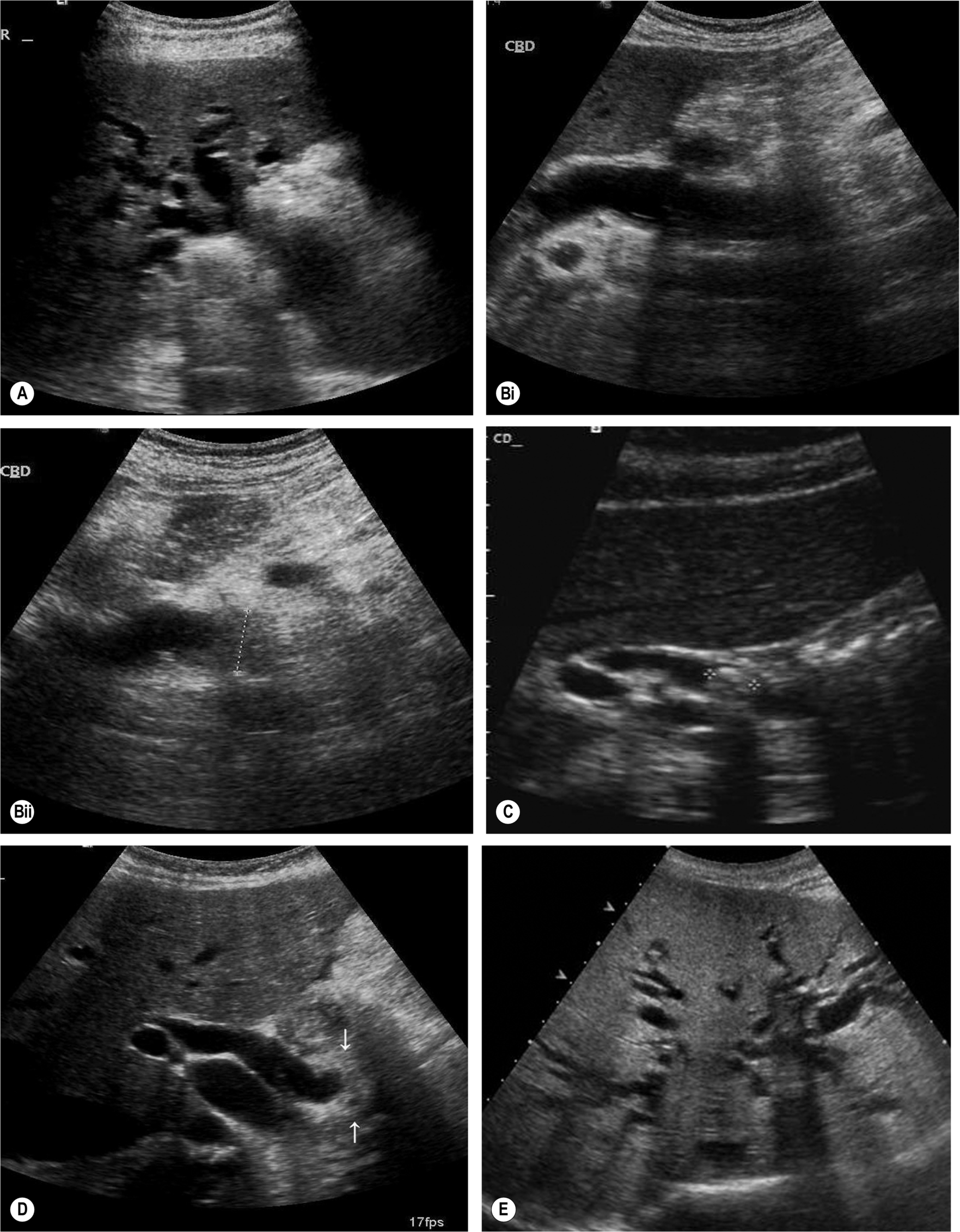
The numerous causes of biliary dilatation are summarized in Table 3.2. Frequently, ultrasound diagnoses the obstruction but does not identify the cause. This is a case for perseverance by the operator, as the lower end of the CBD is visible in most cases once the overlying duodenum has been moved away (Figs. 3.19, 3.20, 3.44). However, ultrasound is not generally regarded as a reliable tool for identifying ductal stones and is frequently unable to diagnose ductal strictures, especially those from benign causes. In the absence of a definite diagnosis of stones, MRCP is an effective, non-invasive technique in the diagnosis of CBD stones and strictures (Fig. 3.44F),5,36 avoiding the need for the more invasive ERCP.
| Intrinsic | Stones |
| Carcinoma of the ampulla of Vater | |
| Cholangiocarcinoma | |
| Stricture (associated with chronic pancreatitis) | |
| Biliary atresia/choledochal cyst | |
| Post liver transplantation bile duct stenosis (usually anastomotic) | |
| Parasites | |
| Age-related or post-surgical mild CBD dilatation | |
| Extrinsic | Carcinoma of the head of the pancreas |
| Acute pancreatitis | |
| Lymphadenopathy at the porta hepatis | |
| Other masses at the porta, e.g., hepatic artery aneurysm, gastrointestinal tract mass | |
| Intrahepatic tumors (obstructed distal segments) | |
| Diffuse hepatic conditions | Sclerosing cholangitis |
| Caroli’s disease |
Although diagnostically highly accurate, the use of ERCP is usually reserved for occasions when therapeutic stone removal or stent insertion may be required, as it is an invasive procedure with attendant risks of pancreatitis, cholangitis, and rarely, perforation of the bile duct or duodenum.37,38 Complications occur in up to 8% of patients undergoing ERCP, with a mortality rate of up to 0.5%,39 so the technique should ideally be reserved for those requiring therapeutic intervention.
EUS also has accuracy in detecting CBD stones comparable with ERCP while avoiding the risks associated with radiation and biliary instrumentation. It is not yet as readily available as ERCP and requires specialist skills and equipment with a significant learning curve.40 CT and MRI are useful for staging purposes if the obstructing lesion is malignant. Cholangiocarcinomas spread to the lymph nodes and the liver – and small liver deposits are particularly difficult to recognize on ultrasound if the intrahepatic biliary ducts are dilated.
In hepatobiliary scintigraphy, technetium99M-labeled derivatives of iminodiacetic acid are excreted in the bile and may help to demonstrate sites of obstruction, e.g., in the cystic duct – or abnormal accumulations of bile, e.g., choledochal cysts. The technique is also useful in demonstrating postoperative bile leaks.41
Courvoisier’s law, to which there are numerous exceptions, states that if the gallbladder is dilated in a jaundiced patient, then the cause is not because of a stone in the common duct. The reasoning behind this is that gallstones cause a degree of wall fibrosis from chronic cholecystitis, which would prevent it from distending. There are many exceptions to this “law,” which include the formation of stones in the duct, without gallbladder stones, and obstruction by a pancreatic stone at the ampulla.
- • Do not assume that obstructive jaundice in a patient with gallstones is because of a stone in the CBD. Jaundice may be attributable to other causes.
- • Do not assume that obstructive jaundice cannot be because of a stone in the CBD if the gallbladder does not contain stones. A solitary stone can be passed into the duct from the gallbladder, or stones can form within the duct.
Management of Biliary Obstruction
Management of biliary obstruction obviously depends on the cause and the severity of the condition. Radiological imaging may be used for both diagnostic and therapeutic purposes. The first line of investigation tends to be ultrasound. Patients may then be directed either for further imaging, such as MRCP or CT, to clarify the diagnosis or proceed straight to therapeutic intervention. Removal of stones in the CBD may be performed by ERCP with sphincterotomy. Elective, laparoscopic cholecystectomy may occur if gallstones are present in the gallbladder.
Laparoscopic ultrasound is a useful adjunct to the surgical exploration of the biliary tree, and its accuracy in experienced hands equals that of X-ray cholangiography. Its potential has been slow to be realized in many centers, as it requires training and has a steep learning curve. However, the lack of ionizing radiation, together with its accuracy in experienced hands, has allowed it to replace fluoroscopic cholangiography in some centers.42
EUS can also be used to examine the CBD, avoiding the need for laparoscopic exploration of the duct. It is highly accurate in diagnosing ductal stones, particularly in experienced hands43 and may be more sensitive than MRCP in the detection of small ductal stones (depending on the MRCP slice thickness).44 However, its invasive nature means it is usually reserved for cases when MRCP is negative in patients where the probability of stones or an obstructing lesion remains high.
The treatment of malignant obstruction is determined by the stage of the disease. Accurate staging is best performed using CT and/or MRI. If surgical removal of the obstructing lesion is not a suitable option because of local or distant spread or other comorbidities, palliative stenting may be performed endoscopically to relieve the obstruction and decompress the ducts (Fig. 3.45). The patency of the stent may be subsequently monitored with ultrasound by assessing the degree of duct dilatation.
Clinical suspicion of early obstruction should be raised if the serum alkaline phosphatase is elevated (often more sensitive in the early stages than a raised serum bilirubin). In the presence of ductal dilatation on ultrasound, further imaging, such as CT, MRCP, or EUS, may then refine the diagnosis.
Intrahepatic Tumors Causing Biliary Obstruction
Focal masses that cause segmental intrahepatic duct dilatation are usually intrinsic to the duct itself, e.g., cholangiocarcinoma. It is also possible for a focal intrahepatic mass, whether benign or malignant, to compress an adjacent biliary duct, causing subsequent obstruction of that segment. However, this is not a common cause of biliary dilatation and occurs most usually with hepatocellular carcinomas.45 Most liver metastases deform, rather than compress, adjacent structures, and biliary obstruction only occurs if the metastases are very large and/or invade the biliary tree. A hepatocellular carcinoma (HCC) or metastatic deposit at the porta hepatis may obstruct the common duct by squeezing it against adjacent extrahepatic structures.
Benign intrahepatic lesions rarely cause ductal dilatation, but occasionally their sheer size obstructs the biliary tree.
Choledochal Cysts
Most commonly found in children, this is associated with biliary atresia, in which the distal “blind” end of the duct dilates into a rounded, cystic mass in response to raised intrahepatic pressure. Choledochal cysts in adults are rare and tend to be asymptomatic unless associated with stones or other biliary diseases. They tend to be associated with an anomalous insertion of the CBD into the pancreatic duct, which allows pancreatic enzymes to reflux into the bile duct resulting in inflammation and weakening of the duct wall, forming a choledochal cyst (Fig. 3.46).46
The dilatation is less common because of a non-obstructive cause in which the biliary ducts become ectatic and can form diverticula. This may be because of a focal stricture of the duct, which causes reflux and a localized enlargement of the duct proximal to the stricture (see also “Caroli’s Disease” below).
Complications of choledochal cysts include cholangitis, formation of stones, and progression of the condition to secondary biliary cirrhosis, which may be associated with portal hypertension.
It may be difficult to differentiate a choledochal cyst, particularly if solitary, from other causes of hepatic cysts. The connection between the choledochal cyst and the adjacent biliary duct may be demonstrated with careful scanning, but further imaging, including MRCP, is frequently required.
Cholangitis
Cholangitis is an inflammation of the biliary ducts. It most commonly occurs secondary to obstruction, where stasis of bile in the biliary tree may lead to infection and acute inflammation of the ducts. Other causes of cholangitis include strictures or stenosis of ducts, post-ERCP, or duct manipulation, AIDS cholangiopathy, parasitic infections such as recurrent pyogenic cholangitis (oriental cholangiohepatitis), which is endemic in Southeast Asia, and sclerosing cholangitis (see also “Primary Sclerosing Cholangitis” below).
Duct dilatation may be present, depending on the cause of the cholangitis. In severe cases, the duct walls appear thickened and irregular, and debris can be seen in the larger ducts (Fig. 3.47). Care should be taken to differentiate ductal wall thickening from tumor invasion, and further imaging may be necessary to exclude malignancy.
Bacterial cholangitis is the most common form because of ascending bacterial infection from the duodenum or portal vein. Bacterial cholangitis is also associated with biliary enteric anastomoses and may be complicated by abscesses if the infection is progressive and untreated. Small abscesses may be difficult to diagnose on ultrasound, as they are frequently isoechoic and ill-defined in the early stages, and biliary dilatation makes evaluation of the hepatic parenchyma notoriously difficult.
Contrast CT may identify small abscesses not visible on ultrasound, and MRCP or ERCP demonstrates mural changes in the ducts.
Biliary Dilatation Without Jaundice
Post-Surgical CBD Dilatation
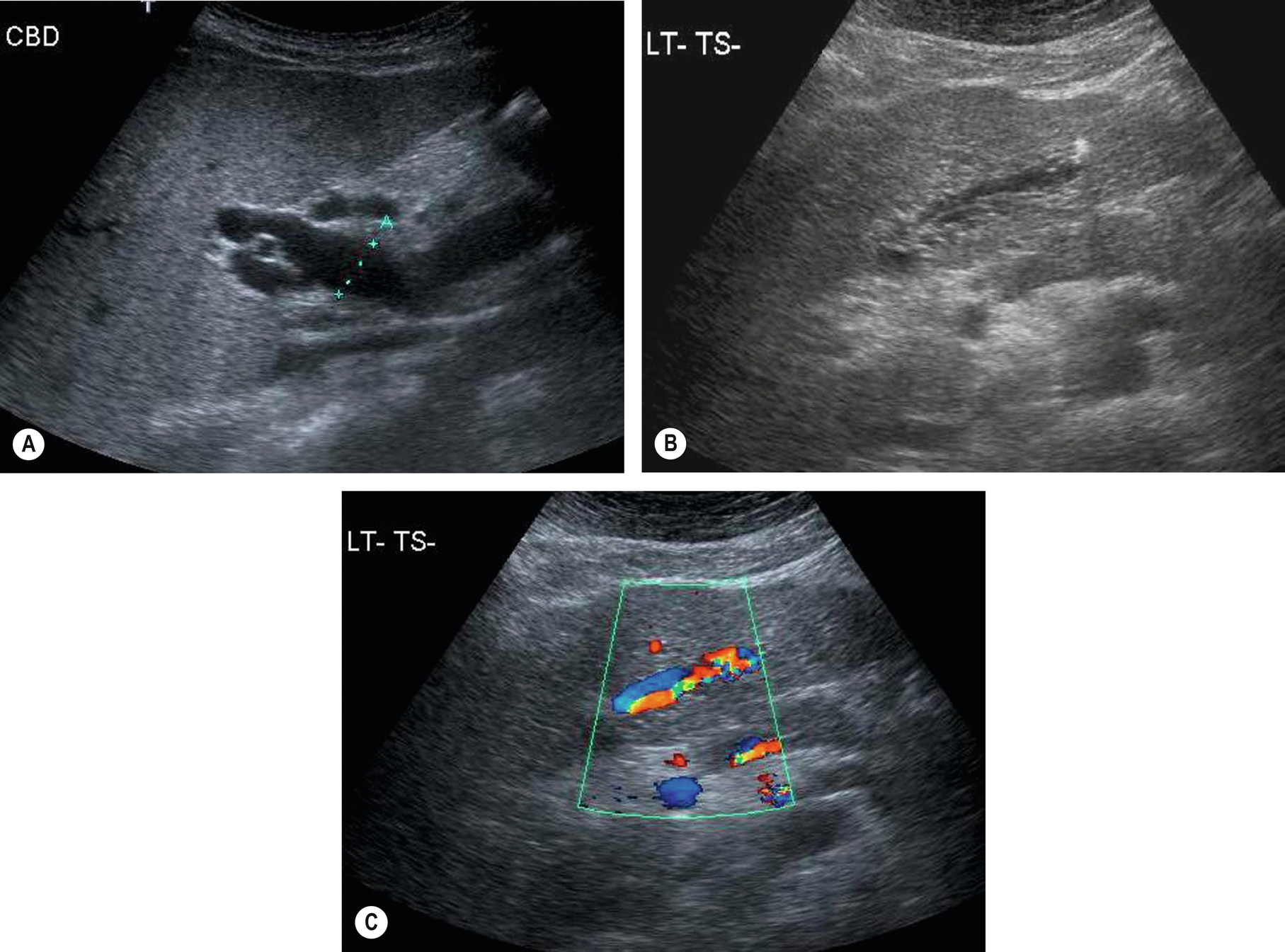
In patients who have had cholecystectomy associated with previous dilatation of the CBD, it is common to find a persistent (but non-significant) mild dilatation of the duct postoperatively. The serum alkaline phosphatase and bilirubin levels should be normal in the absence of a pathologic condition. Because stones may be found in the duct postoperatively, it is important to differentiate non-obstructive from truly obstructive dilatation in a symptomatic patient (Fig. 3.48A). If in doubt, the patient may be rescanned at a suitable interval to assess any increase in ductal diameter.
Focal obstruction
Intrahepatic tumors, such as cholangiocarcinoma, may obstruct a segment of the biliary tree while the remainder of the liver and biliary tree appears normal. Focal duct dilatation should trigger the operator to examine the proximal area of dilatation for a possible mass. Such tumors may be present before jaundice is clinically apparent.
Pitfalls
Patients with cirrhosis and portal hypertension may have dilated hepatic arteries, which can mimic the appearances of dilated ducts (Fig. 3.48B, C). Color or power Doppler will readily differentiate between these, as the bile duct lacks a Doppler signal. However, care must be taken to use an appropriate angle of insonation to display the Doppler signal in the blood vessels, as both duct and patent vessels may display no color if perpendicular to the sound beam.
Pneumobilia (air in the ducts) casts a distal acoustic shadow and may obscure ductal dilatation.
Obstruction Without Biliary Dilatation
Early Obstruction
It is possible to scan a patient at the time of recent onset of obstruction from a stone before the ducts have had time to dilate, leading to a false-negative diagnosis. If clinical suspicion persists, a rescan is frequently useful in these cases.
Occasionally, stones have a ball-valve effect in the duct, causing an intermittent obstruction which may not demonstrate ductal dilatation on the ultrasound scan.
Fibrosis of the Duct Walls
Several chronic pathological conditions cause the walls of the ducts to become fibrotic and stiff. These include PSC (see below), hepatitis, and other chronic hepatic diseases leading to cirrhosis. The liver itself becomes rigid, and this prevents biliary dilatation. In such cases, the lack of dilated bile ducts does not necessarily imply an absence of obstruction.
Other Biliary Diseases
Primary Sclerosing Cholangitis (PSC)
PSC is a rare form of chronic, cholestatic hepatobiliary disease in which the walls of the bile ducts become inflamed, causing narrowing. It predominantly affects young men (with a 2:1 male to female ratio) and is characterized by multiple biliary strictures and bead-like dilatations of the ducts. The cause of PSC remains unclear, but it is associated with inflammatory bowel disorders or may be idiopathic.
Clinical features include jaundice, itching, and fatigue. Twenty-five percent of patients also have gallstones, complicating the diagnosis; 70% of affected patients also have ulcerative colitis. Progressive, gradual fibrosis causes strictures and eventually obliterates the biliary tree. Untreated, this leads to hepatic failure. PSC has a strong association with cholangiocarcinoma, and it is this, rather than hepatic failure, which may lead to death. In the absence of malignancy, however, hepatic transplantation has a 70%-90% five-year survival rate,47 although recurrence of the disease is a recognized complication.48
Ultrasound appearances
The ultrasound appearances in PSC may be normal, particularly in the early stages, or may demonstrate a coarse, hyperechoic liver texture. Ductal strictures may cause downstream dilatation in some segments (Fig. 3.49), and in some cases, there is marked biliary dilatation. However, in most patients, the biliary ducts are prevented from dilatation by the surrounding fibrosis and so appear unremarkable on ultrasound. MRCP is superior at demonstrating intrahepatic ductal strictures (Fig. 3.50). Mural thickening, particularly in the CBD, may be demonstrated with careful scanning (Fig. 3.50A),49 and may be the only ultrasound sign. Ultrasound also demonstrates the effects of portal hypertension in advanced disease. The gallbladder may also have a thickened wall and can be dilated.50
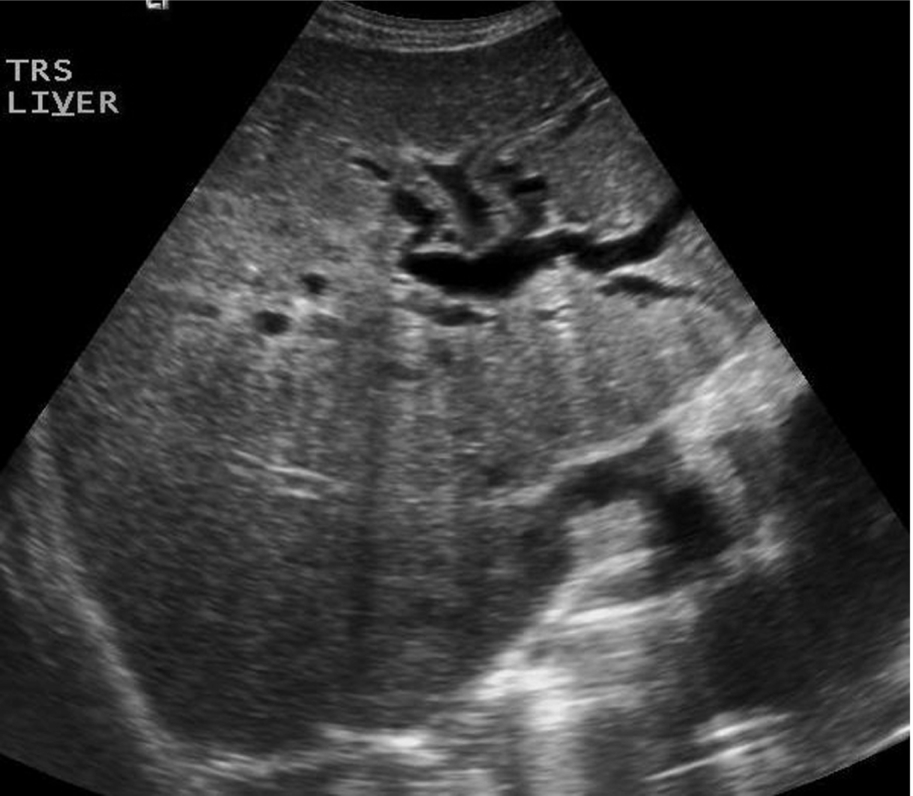
Due to the association between PSC and cholangiocarcinoma, which may be multifocal, a careful search must be done for mass lesions. Because the ultrasound appearances may be those of a coarse, nodular liver texture, it is difficult to identify small cholangiocarcinomas. Ultrasound contrast may help to highlight suspected lesions (Fig. 3.51), but MRI is the next investigation of choice to stage the disease. The diagnosis is an important one because the patient’s prognosis and management are affected by the presence of cholangiocarcinoma. If no masses are identified, the prognosis is favorable, and patients may benefit from the endoscopic removal of stones to relieve symptoms, endoscopic stenting of main duct strictures to relieve jaundice, and subsequent liver transplantation to preempt the formation of carcinoma. However, if carcinoma is already present, five-year survival falls to 10%.
Primary Biliary Cirrhosis (PBC)
PBC is another example of a cholestatic disease that gradually obliterates the biliary tree and ultimately leads to cirrhosis. It is discussed more fully in Chapter 2.
Caroli’s Disease (Congenital Intrahepatic Biliary Dilatation)
This is a rare, congenital condition where the bile ducts are irregularly dilated with diverticula-like projections. These diverticula may become infected and may separate off from the biliary duct, forming choledochal cysts (Fig. 3.52). In most cases, the entire hepatobiliary system is affected to some degree. Sufferers may present in early childhood, with symptoms of portal hypertension,51 or may remain well until adulthood, presenting with cholangitis. It is generally thought to be an autosomal recessive, inherited condition, and the prognosis is poor. Medical control of associated portal hypertension with varices can improve the quality of life.
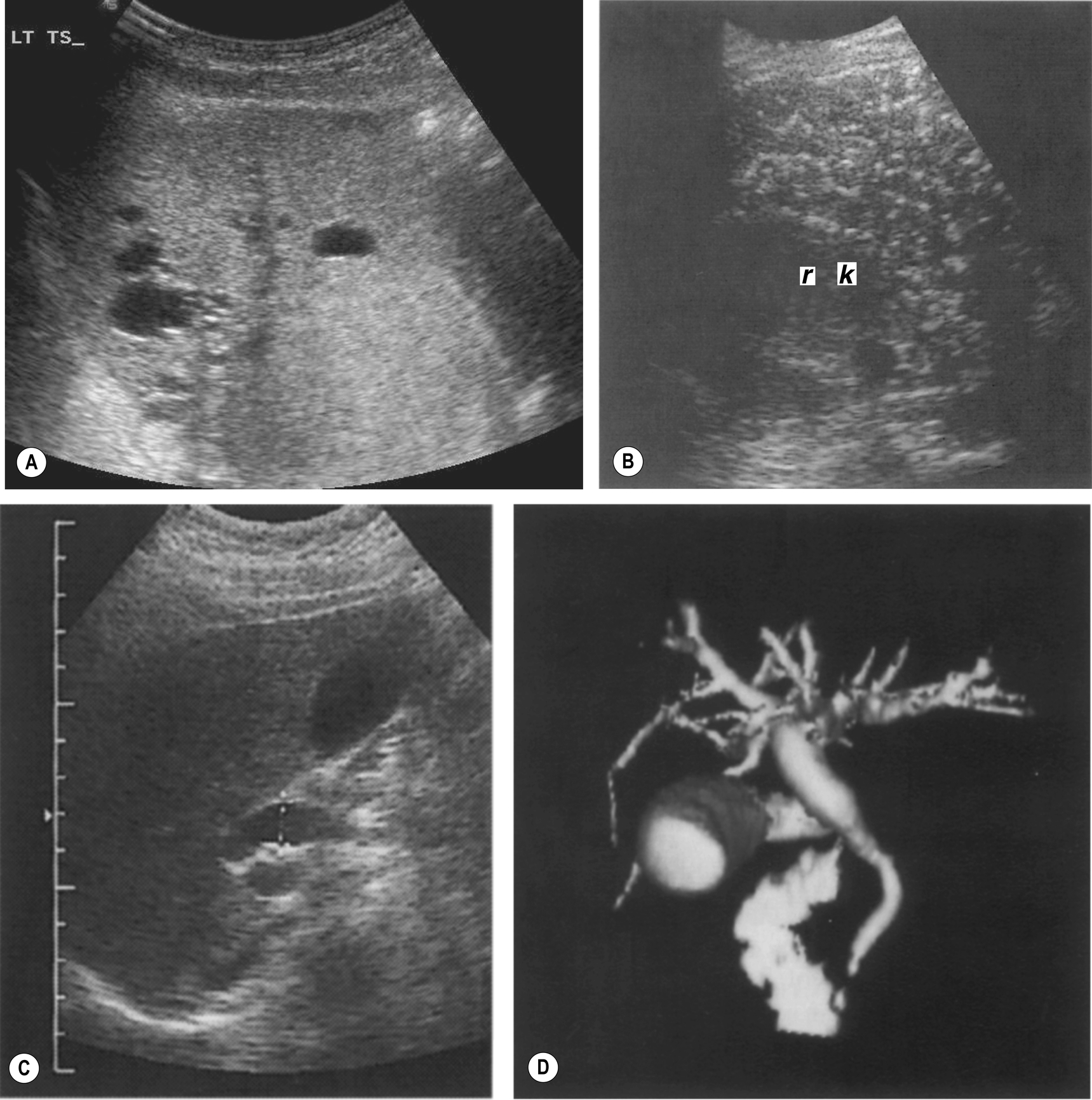
In a few cases, the disease is confined to one or two segments of the liver, in which case a cure can be effected with hepatic resection.52 The extrahepatic biliary tree is often unaffected.
The ultrasound appearances are usually widespread intrahepatic duct dilatation, with saccular and fusiform biliary ectasia. The diagnosis is often unclear because it is also associated with biliary stone formation. The dilatation is also associated with cholangitis, and signs of infection may be present in the form of debris within the ducts or even abscesses. Sometimes frank choledochal cysts can be located.
Advanced disease is associated with portal hypertension and, in some cases, cholangiocarcinoma.53 The presence of multiple cystic spaces on imaging must not be confused with multi- or polycystic disease, and the identification of communication between the cysts and the biliary tree with CT or MRI is an important factor in making the diagnosis.54
Parasites
Parasitic organisms, such as the Ascaris worm and liver fluke, are extremely rare in the United Kingdom. However, they are a common cause of biliary colic in Africa, the Far East, and South America. Hyperechoic linear structures in the gallbladder lumen or dilated ducts should raise the sonographer’s suspicion in patients native to or those who have visited these countries.55 Impacted worms in the biliary ducts may also mimic other ductal masses, and they are a rare cause of obstructive biliary dilatation (Fig. 3.53).
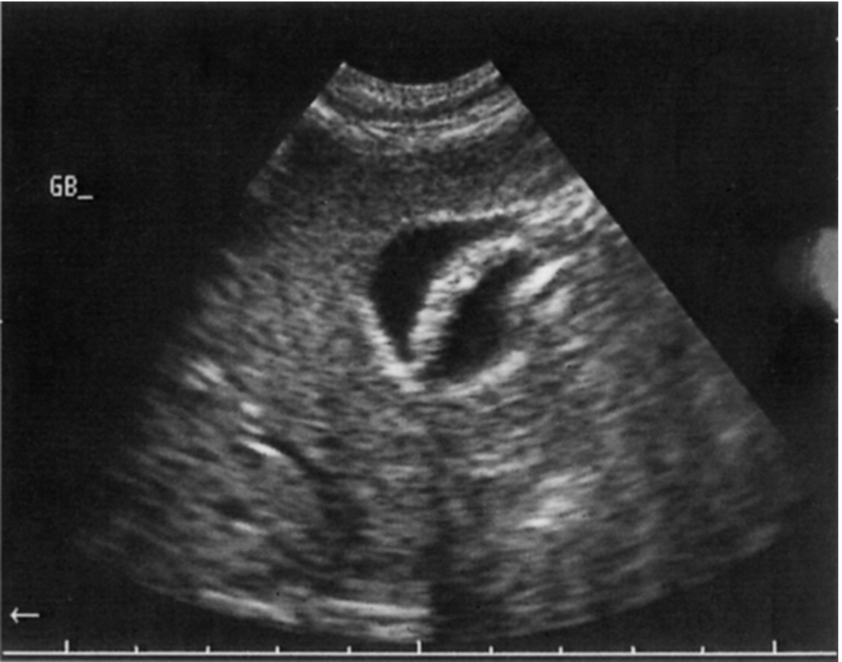
Patients may present with acute cholangitis or abdominal pain and vomiting. Endoscopic management is frequently highly effective.56
Echogenic Bile
Biliary Stasis and Bile Crystals
Fine echoes in the bile within the gallbladder are not uncommon on an ultrasound scan. This is common because of the inspissation of bile following prolonged starvation – for example, following surgery (Fig. 3.54). These appearances disappear after a normal diet is resumed, and the gallbladder has emptied and refilled. Echogenic bile occurs when the solutes in the bile precipitate, often because of hypomotility of the gallbladder, and can commonly be seen following bone marrow transplantation and in patients who have undergone prolonged periods (4–6 weeks) of total parenteral nutrition.57
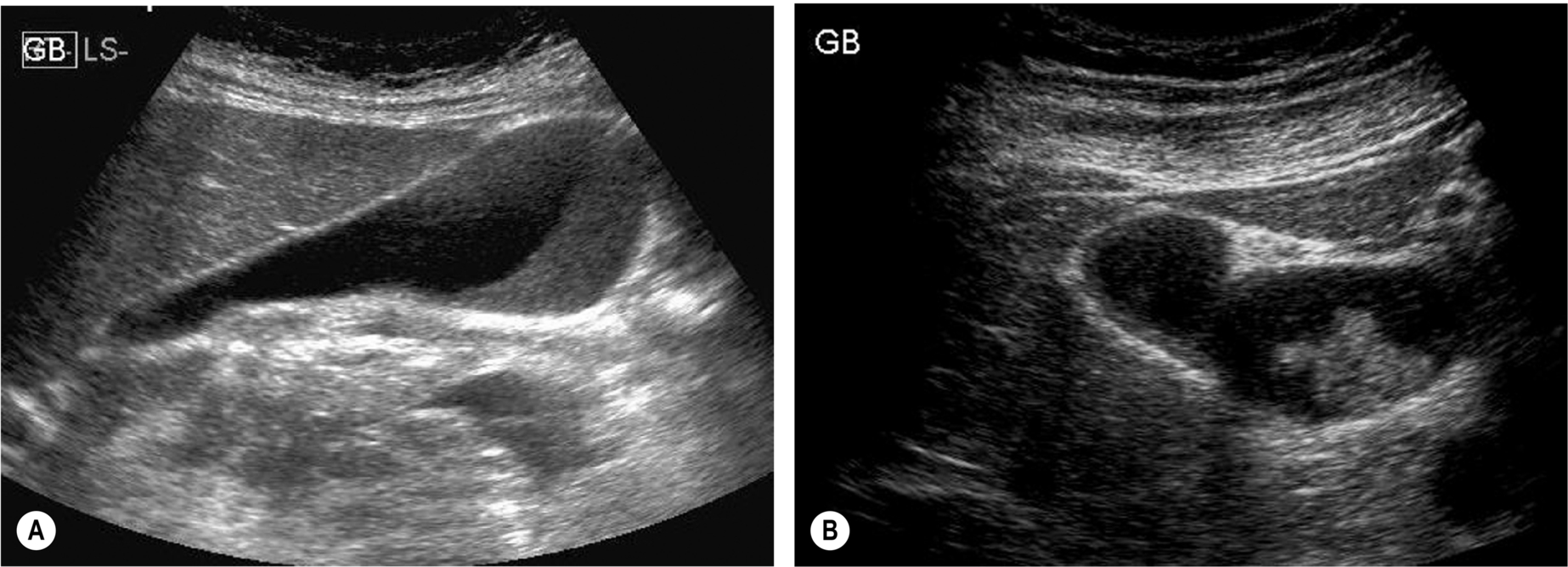
Prolonged biliary stasis may lead to inflammation and/or infection, particularly in postoperative patients and those in immunosuppression. Its clinical course varies from complete resolution to progression to gallstones. However, following the resumption of oral feeding, the gallbladder may contract and empty the sludge into the biliary tree causing biliary colic, acute pancreatitis, and/or acute cholecystitis.58 For this reason, cholecystectomy may be considered in symptomatic patients with biliary sludge.
The fine echoes may form a gravity-dependent layer and may clump together, forming “sludge” balls (Fig. 3.54B). To avoid misdiagnosing sludge balls as polyps, turn the patient to disperse the echoes or rescan after the patient has resumed a normal diet.
Biliary stasis is associated with an increased risk of stone formation.59 Occasionally, echogenic bile persists even with normal gallbladder function (Fig. 3.55). It is likely that there is a spectrum of biliary disease in which gallbladder dysmotility and subsequent saturation of the bile leads to the formation of crystals in the bile and also in the gallbladder wall. Some particles become quite large, forming microlithiasis, which is likely to be an intermediate stage in stone formation.60 Pain and biliary colic may be present prior to stone formation. Biliary crystals or “microlithiasis” (usually calcium bilirubinate granules) have a strong association with acute pancreatitis,61 and their presence in patients who do not have gallstones may therefore be highly significant.
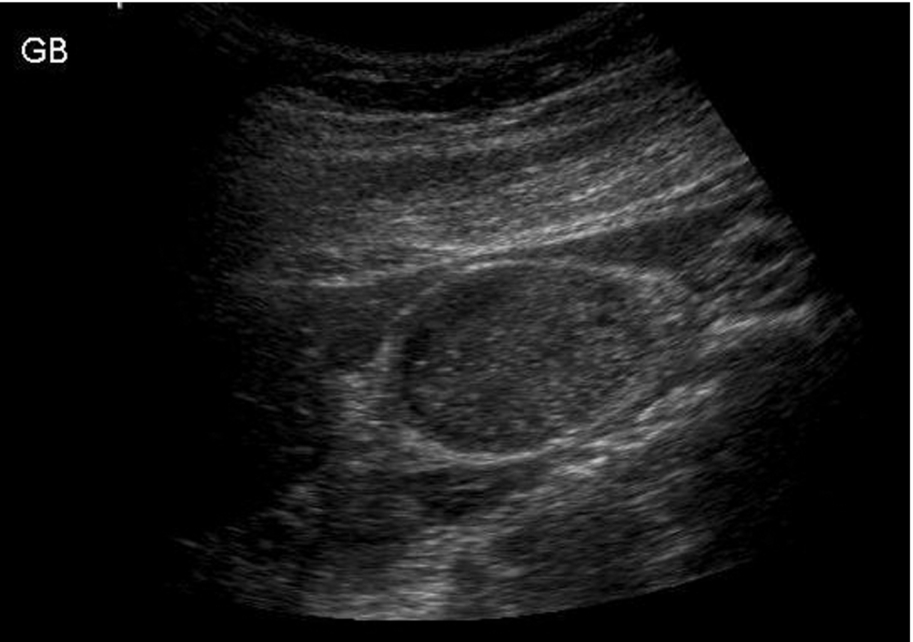
Obstructive Causes of Biliary Stasis
Pathological bile stasis in the gallbladder is because of obstruction of the cystic duct (from a stone, for example) and may be demonstrated in a normal-sized or dilated gallbladder. The bile becomes viscous and hyperechoic. The biliary ducts remain normal in caliber. Eventually, the bile turns watery and appears echo-free on ultrasound; this is known as a mucocele (see above).
Bile stasis within the ducts occurs either because of prolonged and/or repetitive obstruction or a cholestatic disease such as PBC (Chapter 2) or PSC. This can lead to cholangitis.
Haemobilia
Blood in the gallbladder can result from gastrointestinal bleeding or other damage to the gallbladder or bile duct wall – for example, iatrogenic trauma from an endoscopic procedure or biopsy – or it may be associated with ruptured hepatic artery aneurysm.
The appearances depend upon the stage of evolution of the bleeding. Fresh blood appears as fine, low-level echoes. Blood clots appear as solid, non-shadowing structures, and there may be hyperechoic, linear strands.62 A history of trauma may allow the sonographer to differentiate haemobilia from other causes of echogenic bile, particularly those associated with gallbladder inflammation, and there may be other evidence of abdominal trauma on ultrasound – such as a hemoperitoneum.
Pneumobilia
Air in the biliary tree is usually iatrogenic and frequently seen following procedures such as ERCP, sphincterotomy, or biliary surgery. Although it does not usually persist, the air can remain in the biliary tree post-procedure for months, even years, and is not significant.
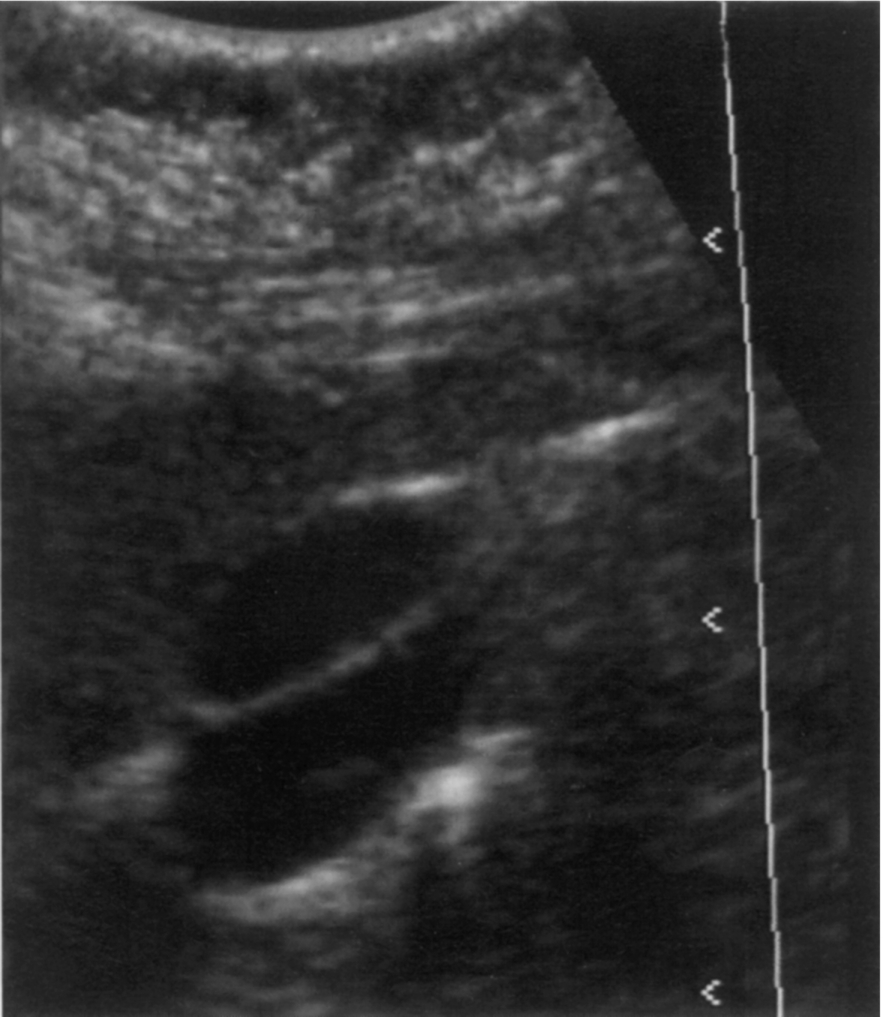
It is characterized by highly reflective linear echoes (Fig. 3.56), which follow the course of the biliary ducts. The air usually casts a shadow that is different from that of stones – often having reverberative artifacts and being much less well-defined or clear. This shadowing obscures the lumen of the duct and can make an evaluation of the hepatic parenchyma difficult.
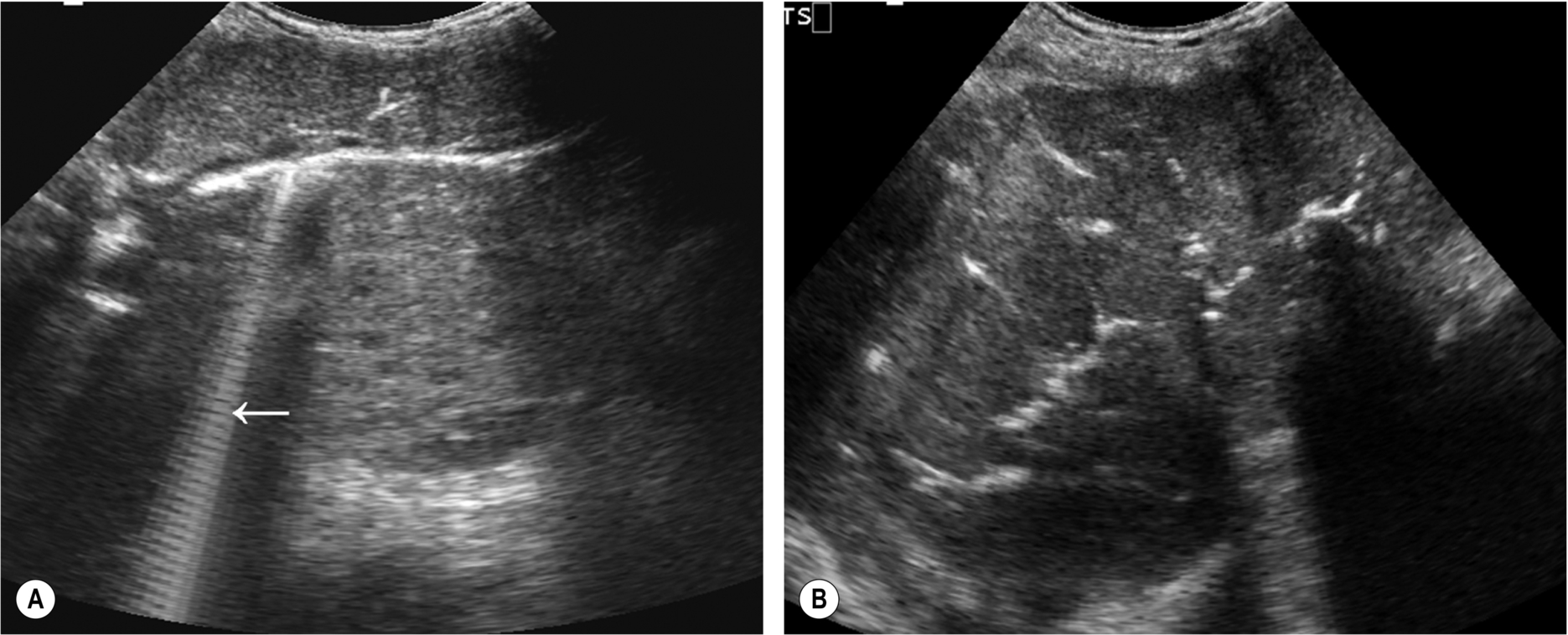
Pneumobilia may also be present in emphysematous cholecystitis, an uncommon complication of cholecystitis, in which gas-forming bacteria are present in the gallbladder (see above) or in cases where a necrotic gallbladder has formed a cholecystoenteric fistula. Rarely, multiple biliary stones form within the ducts throughout the liver, which can be confused with the appearance of air in the ducts.
Malignant Biliary Disease
Primary Gallbladder Carcinoma
Cancer of the gallbladder is usually associated with gallstones and a history of cholecystitis. Most often, the gallbladder lumen is occupied by a solid mass that may have the appearance of a large polyp. The wall appears thickened and irregular, and shadowing from the stones may obscure it posteriorly. A bile-filled lumen may be absent, further complicating the ultrasound diagnosis (Fig. 3.57). In a porcelain gallbladder (calcification of the gallbladder wall), which is associated with gallbladder carcinoma, the shadowing may obscure any lesion in the lumen, making detection almost impossible.
Particular risk factors for gallbladder carcinoma include large stones, polyps of over 1 cm in size, porcelain gallbladder, and occasionally, choledochal cyst because of the anomalous junction of the pancreatobiliary ducts.15
The carcinoma itself is frequently asymptomatic in the early stages, and patients tend to present with symptoms relating to the stones. It tends to be an aggressive lesion that quickly metastasizes to the liver and portal nodes and has a very poor prognosis, with a curative surgical resection rate of around 15%–20%. Ultrasound may also demonstrate local spread into the adjacent liver, but further staging with CT is invariably necessary.63
Cholangiocarcinoma
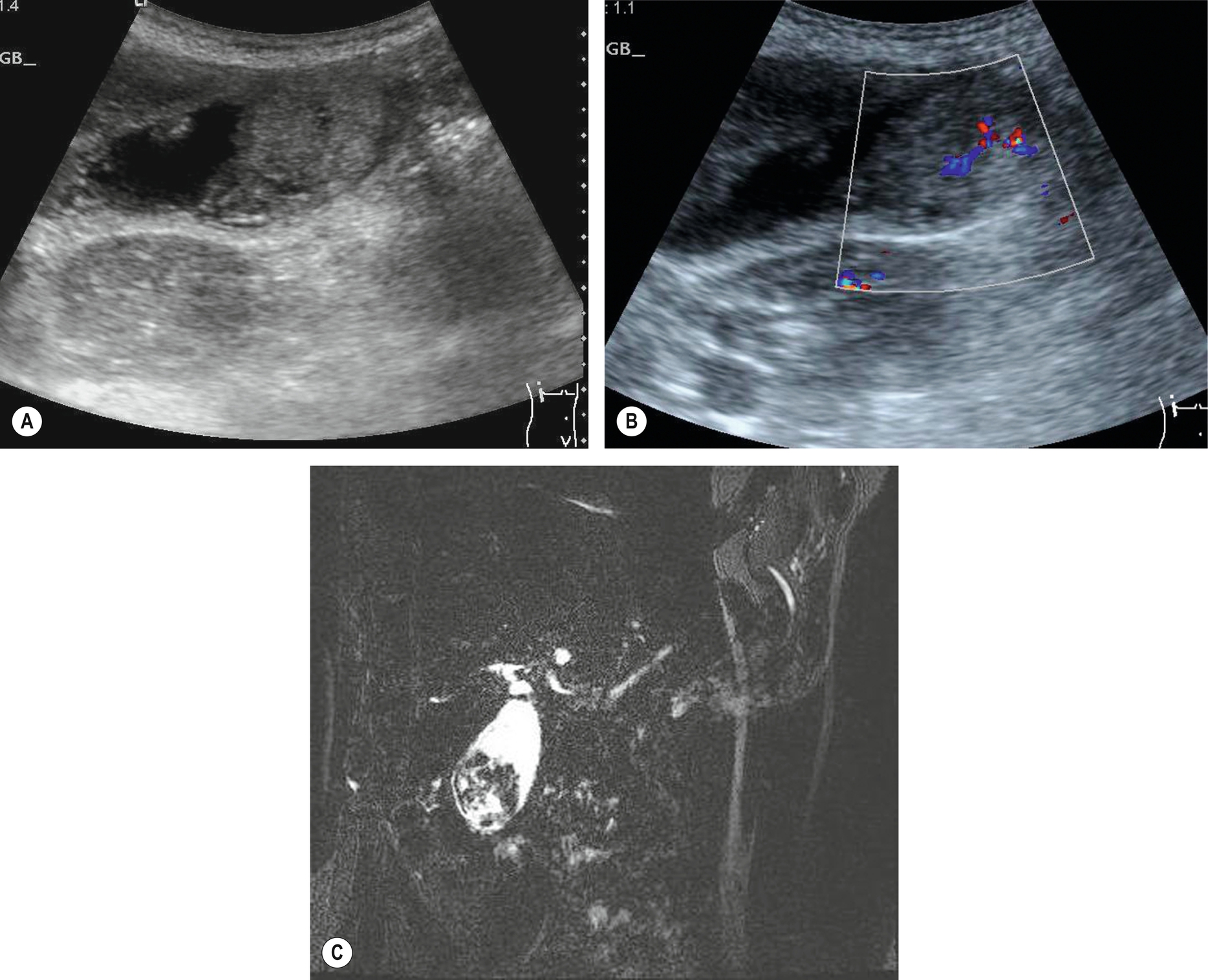
This is a malignant lesion arising in the wall of the bile duct. It is readily recognizable from an ultrasound point of view when it occurs in and obstructs the common duct, as the subsequent dilatation outlines the proximal part of the tumor with bile (Fig. 3.58). However, tumors occurring intrahepatically tend to be isoechoic and notoriously difficult to detect. Cholangiocarcinoma may occur at any level along the biliary tree and is frequently multifocal.
A cholangiocarcinoma is referred to as a Klatskin tumor when it involves the confluence of the right and left hepatic ducts. These lesions are often difficult to detect on both ultrasound and CT, and the only clue may be the proximal dilatation of the biliary ducts (Fig. 3.59). Contrast-enhanced ultrasound (CEUS) is helpful in localizing a suspected lesion because of arterial hypervascularity followed by hypoenhancement in the sinusoidal phase (Fig. 3.51).
Although rare, the incidence of cholangiocarcinoma seems to be increasing, and it is strongly associated with PSC (see above).64
Multifocal cholangiocarcinoma may spread to the surrounding liver tissue and have a poor prognosis. It may be almost impossible to identify these lesions in a liver with a texture already altered by diffuse disease before they become large. A pattern of dilated ducts distal to the lesion is a good clue (Figs. 3.60, 3.61).
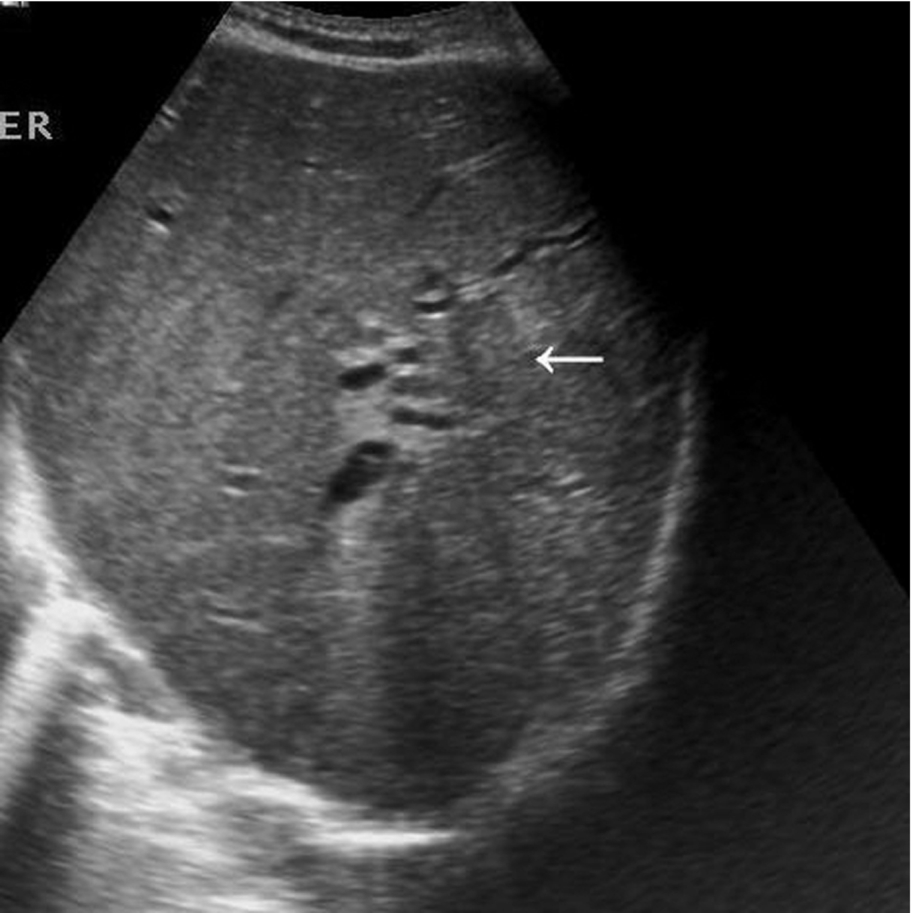
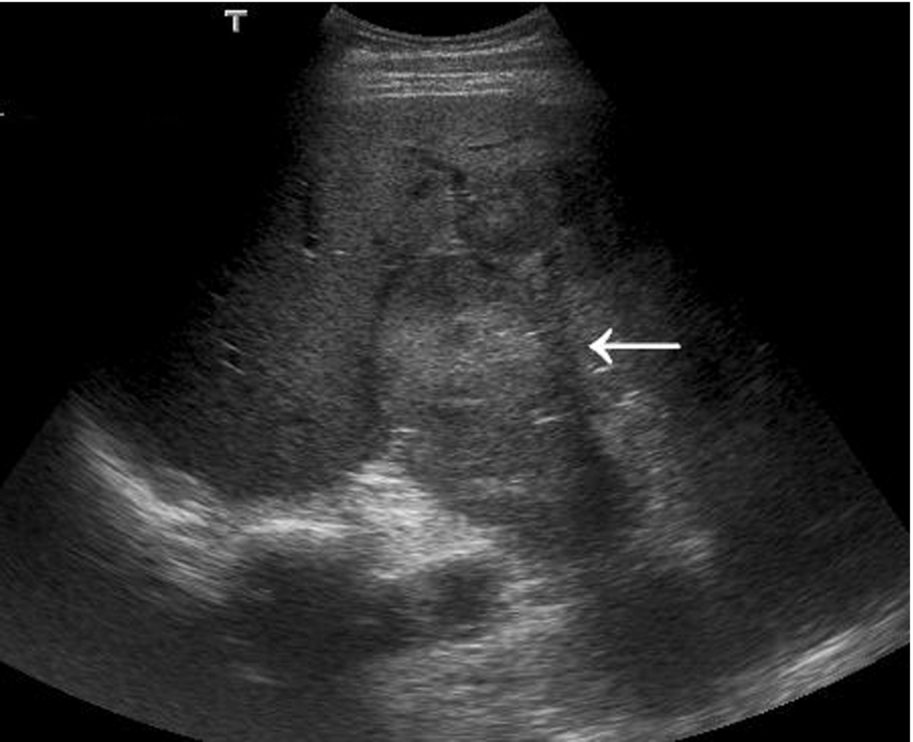
Management of the Patient with Cholangiocarcinoma
These patients have a poor prognosis, as the lesions usually present with jaundice because of invasion and obstruction of the duct. They spread to surrounding tissues, including the portal vein and lymph nodes, metastasize to the liver, and can be multifocal, particularly with PSC.
Staging of the disease is performed with CT or MRI. EUS can outline invasion into the biliary duct, and laparoscopic ultrasound can pick up peritoneal or local spread.
Surgical resection of the tumor is becoming more successful in patients with single lesions,65 and liver transplantation in patients with PSC has a good prognosis, provided the tumor bulk is low. Palliation is frequently the only feasible option, and the insertion of a stent, either percutaneously or endoscopically, to bypass the obstructing lesion and assist drainage of the liver will relieve the symptoms and often allows the patient to return home for some months.
Other treatment options, such as chemotherapy, have limited success. Despite improvements in treatment, only a minority of patients survive beyond 12 months after the initial diagnosis.66
Gallbladder Metastases
Metastases from other primaries may occasionally be deposited within the gallbladder wall (Fig. 3.62), usually as a late presentation of the disease process. Often, other metastatic deposits, for example, in the liver and lymph nodes, may raise suspicion of gallbladder metastases in an irregularly thickened gallbladder wall.
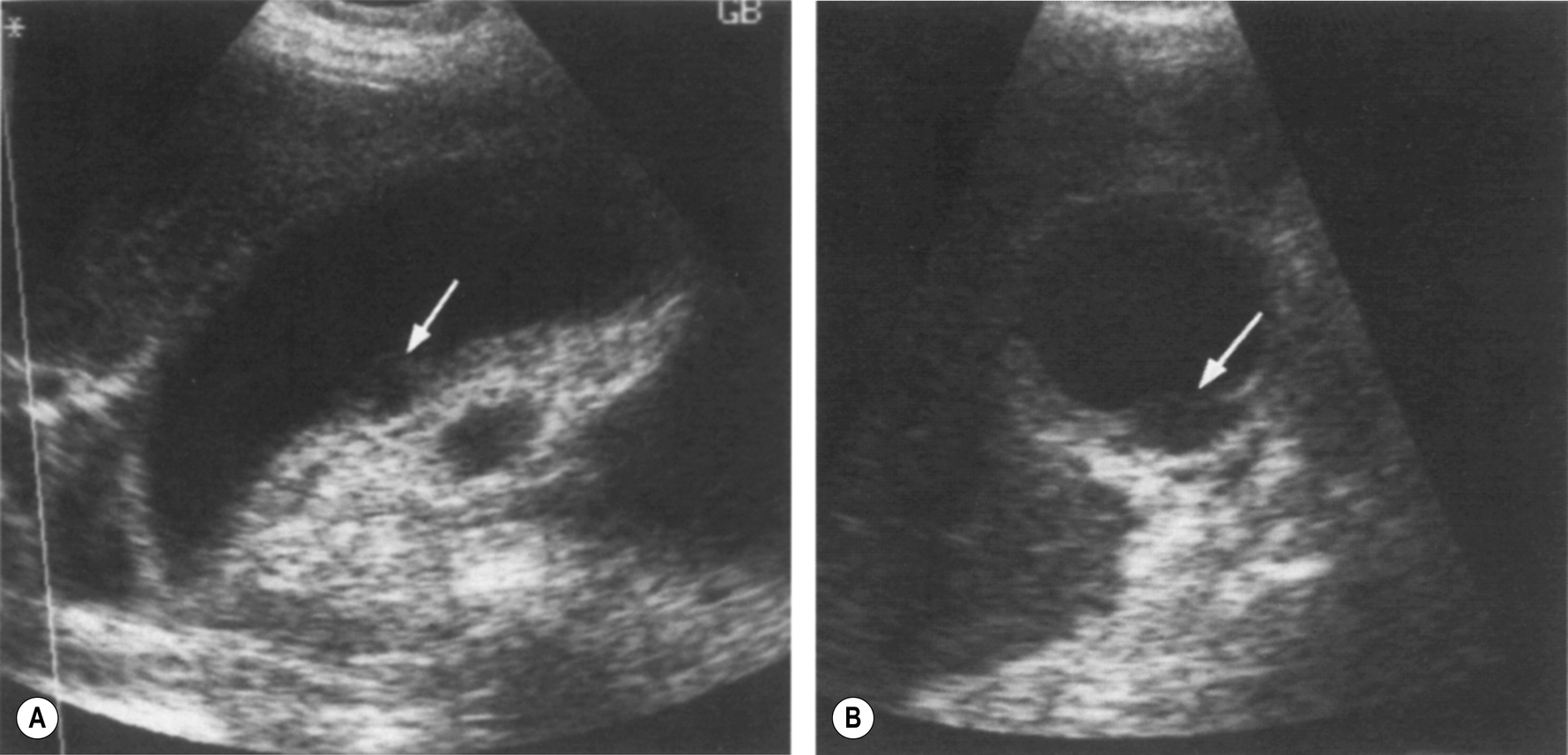
The ultrasound appearances are of focal thickening and polyp-like lesions in the wall of the gallbladder. This may mimic primary gallbladder carcinoma, but knowledge of a previously diagnosed primary, for example, melanoma, lung, or breast carcinoma, will point toward the diagnosis.

