Body Fluid Analysis:
Manual Hemacytometer Counts and Differential Slide Preparation
After studying this chapter, the student should be able to:
- 1. State four factors that adversely affect manual cell counts performed using a hemacytometer.
- 2. Discuss advantages and disadvantages of each diluent used to perform body fluid cell counts.
- 3. Discuss the challenges associated with cell counting of viscous fluids—for example, synovial fluid, semen—including pretreatment options and their effects if any on cell counts.
- 4. Describe step by-step how to perform a manual cell count using a hemacytometer.
- 5. Calculate the cell count in a body fluid when provided with the necessary information.
- 6. Explain cytocentrifugation and its use in preparing slides of body fluid for differential analysis, including the equipment needed, advantages, and disadvantages.
Using A Hemacytometer
Manual methods using a hemacytometer are often used to perform cell counts on body fluids such as cerebrospinal fluid (CSF), synovial fluid, pleural fluid, pericardial fluid, and peritoneal fluid, as well as peritoneal dialysates, bronchoalveolar lavages, and semen. In health, the numbers of red blood cells (RBCs) and white blood cells (WBCs) in these body fluids are low, and other cells or cellular debris can be present. As discussed in Chapter 16, automated cell counting analyzers can produce erroneous results when the cell count is low. It is the responsibility of each laboratory to define its lower limit for cell counts (RBCs and WBCs) and to have a protocol for performing manual cell counts using a hemacytometer when cell counts are below the laboratory-defined lower limit.1
Highly viscous body fluids (e.g., synovial fluid) and fluids that fail to appropriately liquefy (e.g., semen) require pretreatment before cell counting by manual or automated methods. Note that cell counts using a clotted body fluid are inaccurate. Because it may not be possible to obtain another body fluid specimen, every effort is made to work with the healthcare provider to provide valid, useful information. This may include performing a cell count and including on the report a statement such as, “Specimen clotted; cell counts must be interpreted with caution.”
Manual cell counts using a hemacytometer are time-consuming, require advanced technical skills, have poor precision (reproducibility), and are subject to numerous errors as a result of the multiple steps involved. Therefore it is imperative that well-trained and technically proficient laboratorians perform them and that appropriate materials are used to verify the achievement of quality goals.
Diluents and Dilutions
The visual appearance of the body fluid aids in determining whether a dilution should be made for cell counting and what dilution should be prepared. Body fluids that are clear do not require a dilution, and the fluid can be loaded directly onto a hemacytometer. Fluids that are visibly cloudy or bloody must be diluted to obtain accurate cell counts. Table 17.1 is provided as a guide to dilution selection based on visual appearance. When diluents that do not lyse RBCs are used, a higher dilution may be necessary. Body fluids that are visibly clear indicate a low cell count and are evaluated undiluted; sometimes cell counts in hazy or slightly cloudy fluids can also be performed on the undiluted fluid. To enhance visualization of WBCs in fluids other than synovial fluid, the fluid can be “exposed” to glacial acetic acid (Box 17.1). Blood-tinged or bloody fluids can be diluted using diluents that (1) lyse RBCs and (2) enhance visualization of nucleated cells.
Table 17.1
| Fluid Appearance | WBC Count | RBC Count |
|---|---|---|
| Clear | Undiluted | Undiluted |
| Hazy (slightly cloudy) | 1:2a dilution | Undiluted |
| Blood-tinged | 1:2a dilution | Undiluted |
| Cloudy | 1:20 dilution | Undiluted |
| Bloody | 1:2a or 1:20 dilution | 1:200 dilution |
RBC, Red blood cell; WBC, white blood cell.
aUsing a diluent that lyses RBCs.
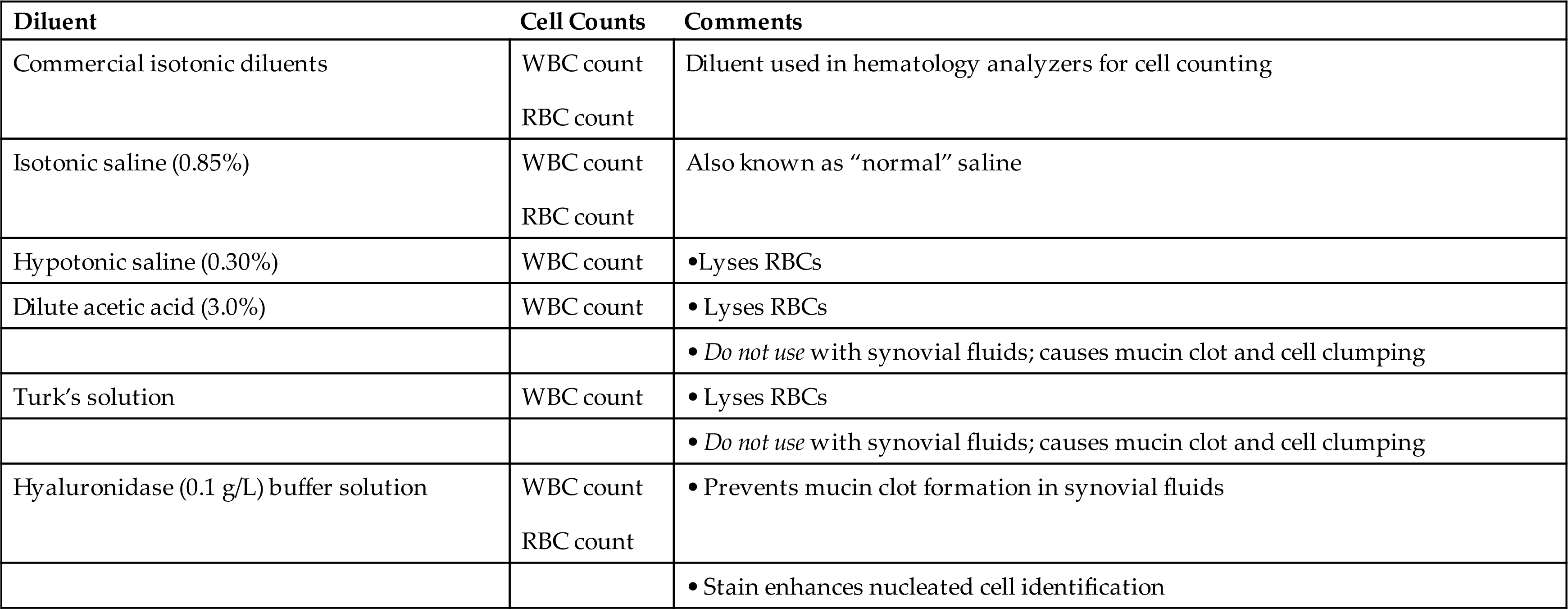
Often isotonic, particle-free commercial diluents used by the laboratory’s hematology analyzer can be used to dilute body fluids (e.g., Cellpack, Sysmex Corporation, Kobe, Japan). Laboratory-prepared isotonic solutions such as normal saline (0.85%) can also be used for RBC counts, whereas dilute acetic acid solutions are often used for the nucleated cell and WBC counts. Acetic acid in the diluent performs two functions: It lyses any RBCs present and it enhances the nuclei of WBCs. Table 17.2 summarizes diluents commonly used for body fluids when WBC and RBC counts are performed. Appendix D provides details on the preparation of these diluents.
Table 17.2
| Diluent | Cell Counts | Comments |
|---|---|---|
| Commercial isotonic diluents | Diluent used in hematology analyzers for cell counting | |
| Isotonic saline (0.85%) | Also known as “normal” saline | |
| Hypotonic saline (0.30%) | WBC count | •Lyses RBCs |
| Dilute acetic acid (3.0%) | WBC count | |
| Turk’s solution | WBC count | |
| Hyaluronidase (0.1 g/L) buffer solution | ||
RBC, Red blood cell; WBC, white blood cell.
aSee Appendix D for diluent preparation.
The diluent used depends on the body fluid being evaluated. Note that synovial fluid cannot be diluted using weak acid diluents, such as acetic acid. Because of the high hyaluronic acid and protein content of synovial fluid, acetic acid will cause a mucin clot (i.e., the coprecipitation of hyaluronic acid and protein), which interferes with accurate cell counting. Instead, synovial fluid is diluted using a commercial isotonic diluent, normal saline (0.85%), hypotonic saline (0.30%), or a hyaluronidase buffer solution. A hyaluronidase buffer solution prevents mucin clots, and when toluidine blue stain is included, it aids in the visualization and identification of cellular elements. When WBC counts are performed on synovial fluid and hypotonic saline is used as the diluent, any RBCs present are lysed and mucin formation is not initiated.
To obtain accurate cell counts, dilutions of body fluids must be made using a quantitative technique. Calibrated automatic pipettes (e.g., Pipetman, Eppendorf, Drummond, MLA) are used to prepare these dilutions manually; commercial diluting systems (e.g., Unopettes) are not available in most locations. Note that when viscous fluids, such as synovial fluid and semen, are pipetted, a positive displacement pipette must be used because an air displacement pipette cannot accurately dispense these viscous fluids. In contrast, CSF, pleural, pericardial, and peritoneal fluids and pretreated synovial fluid can be diluted using either an air or a positive displacement pipette.
Pretreatment and Dilution of Synovial Fluid Specimens
When synovial fluid is evaluated without pretreatment, the viscosity of the fluid can cause an uneven distribution of cells in the hemacytometer. Also when preparing dilutions of untreated synovial fluid, a positive displacement pipette is required to accurately prepare dilutions of the fluid. An alternative is to pretreat synovial fluid using the enzyme hyaluronidase. This enzyme eliminates the fluids’ viscosity by depolymerizing hyaluronic acid, which will also prevent mucin clot formation. Two pretreatment approaches using hyaluronidase are provided in Appendix D. Basically, hyaluronidase is added to an aliquot of synovial fluid, which is mixed well. To enhance depolymerization the sample can be briefly incubated at 37°C. Note that pretreatment with hyaluronidase does not affect crystals that can be present in synovial fluid. Additionally, some excessively viscous synovial fluids may need to be pretreated with hyaluronidase despite the use of a hyaluronidase buffer solution as the diluent.
If pretreated synovial fluid is clear, it can be evaluated undiluted for cell counts. When a dilution is needed, a diluent that will not cause a mucin clot is required, such as a commercial or laboratory-made isotonic diluent, hypotonic saline, or a 0.1-g/L hyaluronidase buffer solution (see Appendix D for diluent preparation).
Semen Dilution and Pretreatment of Specimens
As with synovial fluid specimens, semen is viscous even after liquefaction, and positive displacement pipettes are required to accurately prepare dilutions.2 Often, the diluent used to dilute semen for sperm counts is a solution of sodium bicarbonate, formalin (a fixative), and, optionally, a stain—trypan blue or gentian violet (see Appendix D for diluent preparation). Including a stain enhances visualization, which assists in differentiating among sperm, immature sperm (spermatids, spermatocytes), and WBCs—primarily neutrophils, monocytes, and macrophages.
Semen specimens that fail to liquefy adequately after 60 minutes require treatment before sperm count, sperm motility assessment, and chemical testing can be performed. One treatment approach involves diluting the seminal fluid using an isotonic medium followed by mechanical mixing—repeated aspiration and dispensing of the mixture using a pipette. Equal parts of semen and a medium such as Dulbecco’s phosphate-buffered saline can be used.2 An alternate approach consists of digestion using the proteolytic enzyme bromelain. Semen is diluted 1:2 using this enzyme solution (i.e., 1 part semen+1 part bromelain solution). Note that any dilutions of the sample must be accounted for when the sperm concentration is calculated.
The effects that these treatments have on sperm function, morphology, or the biochemistry of the seminal plasma are not known.2 Therefore when a semen specimen is specially treated for testing, this must be documented on the report. Note that any laboratory analyzing semen for any purpose other than postvasectomy analysis should have available the WHO Laboratory Manual for the Examination and Processing of Human Semen.2 This comprehensive and indispensable text is a vital resource for all aspects of testing when semen analysis is performed.
Hemacytometer Cell Counts
The total nucleated cell or WBC count is an important count that is requested on almost all body fluids. In contrast, little clinical value is derived from an RBC count other than to identify a traumatic puncture procedure, so it may not be performed, particularly when counts are done manually. Box 17.2 summarizes a protocol for a manual cell count using an “improved” Neubauer hemacytometer (Fig. 17.1).
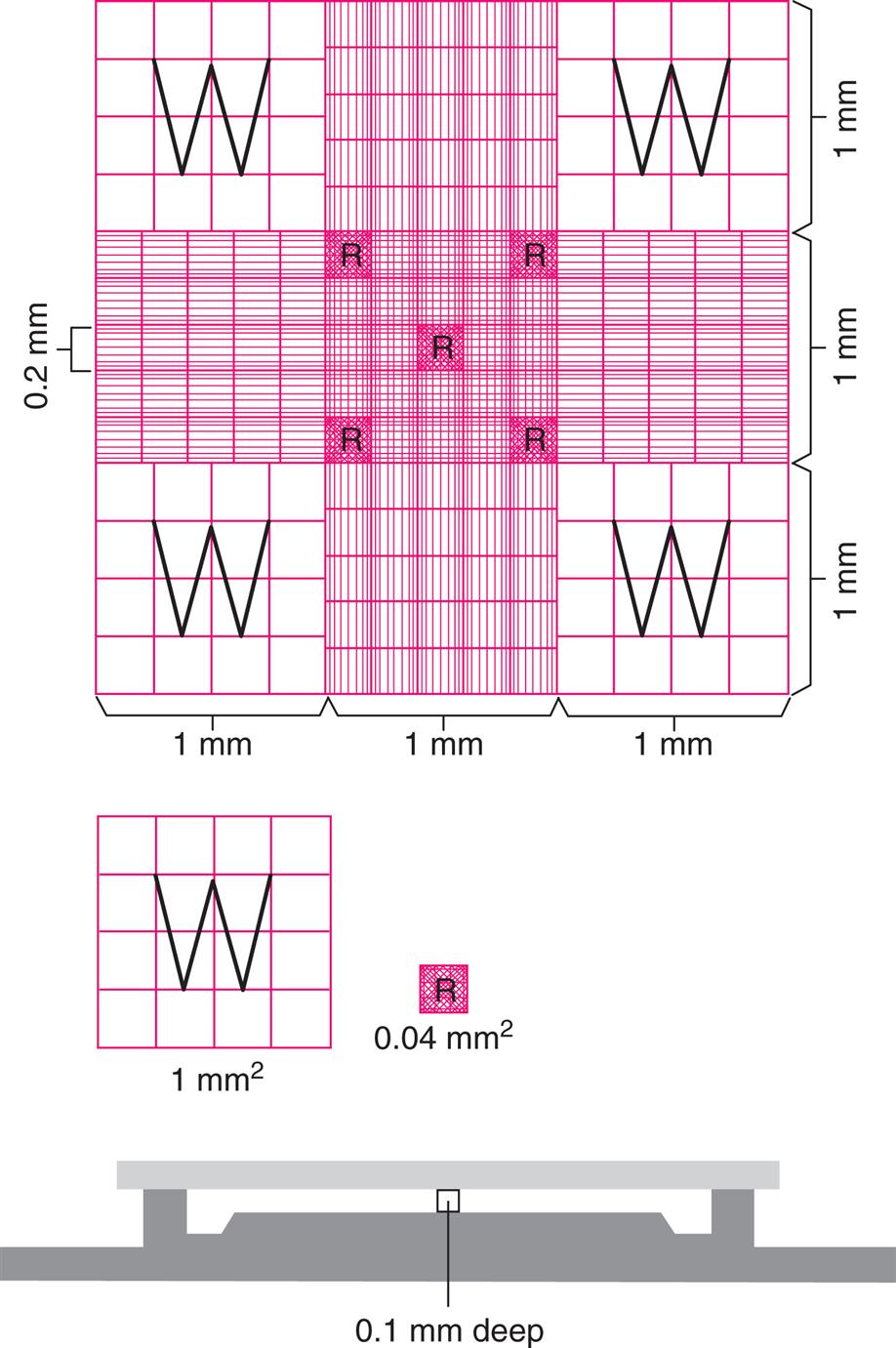
Note that the dimensions are not the same in different types of counting chambers. For example, the depth in a Neubauer chamber is 0.1 mm whereas in a Fuch-Rosenthal chamber the depth is 0.2 mm. Using the correct dimensions is crucial because the resultant calculations (i.e., cell count) are dependent on them.
Before filling a hemacytometer chamber with undiluted fluid or preparing a dilution, the specimen must be adequately mixed to evenly disperse cells. Body fluid specimens can be mixed for 2 to 5 minutes on an automated rocking mixer or by inverting the tube 10 to 15 times by hand.1 Note that synovial fluid specimens should be mixed for 5 to 10 minutes. When using nondisposable hemacytometers, they should be cleaned before use by flooding with 70% alcohol and wiping dry with lens paper. This cleans the chambers and removes dust, which can interfere with placement of the coverslip.
The number of squares counted in each chamber of the hemacytometer depends on the total number of cells present. Accuracy in cell counts is directly related to cell numbers; the more cells counted, the better the accuracy. Therefore dilutions should be avoided if possible. When necessary, a minimum volume of 50 μL should be used to avoid pipetting errors associated with smaller volumes.2 Dilutions should not be excessive and should result in an adequate number of cells for counting. Ideally, a minimum of 100 cells should be counted, but this is not feasible in many body fluids (e.g., CSF). To compensate for fluids with extremely low cell counts, additional squares of the hemacytometer grid can be counted. With fluids that have a high cell count, fewer squares can be counted. Note that regardless of the variation used, calculations must be properly adjusted for the volume of body fluid actually counted.
To ensure detection of potential errors during the preparation of body fluids for manual cell counts, dilutions should be performed and analyzed in duplicate. In other words, two separate dilutions of the body fluid are prepared and loaded into a hemacytometer—each dilution is loaded into one chamber. The cell counts obtained for each chamber are compared and must agree with the criteria established by the laboratory, usually 20% or less. If the counts between chambers are unacceptable (exceed 20%), the body fluid dilutions and counts must be repeated. Note that counting the same chamber twice or comparing two different chambers filled using the same dilution is not true replication and will not detect errors in pipetting, dilution, and mixing.
Calculations
When using a hemacytometer, regardless of the number of squares counted or the dilution used, the number of cells per microliter (or cubic millimeter) of fluid is determined using the following formula.
 Equation 17.1
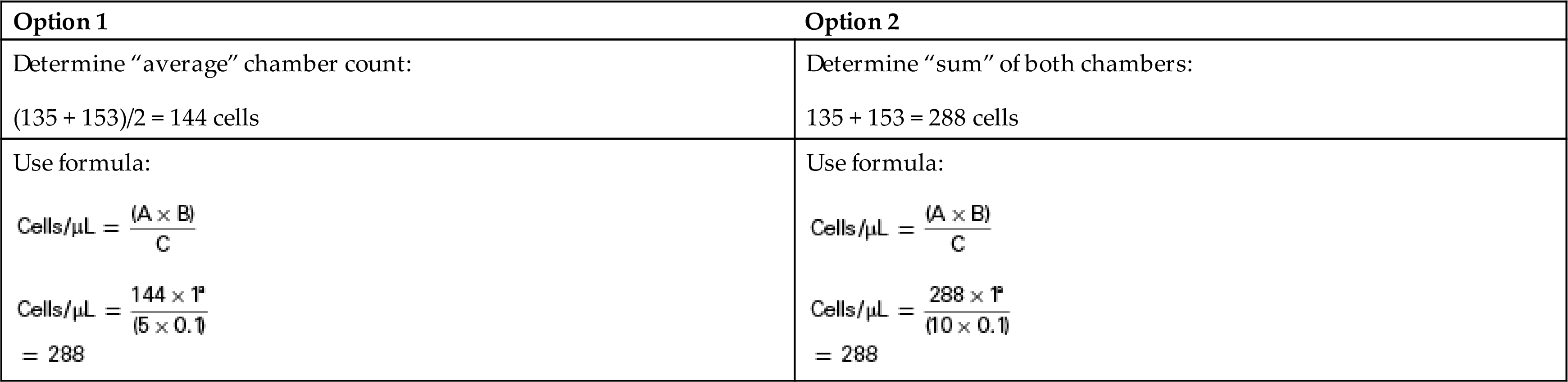
Equation 17.1There are two approaches to using this formula, and both will produce the same result. One approach is to count both chambers of the hemacytometer, compare the counts to ensure that precision criteria are met, average the count (if acceptable), and then use the formula to calculate the number of cells per microliter. An alternate approach is to count each chamber, compare the counts to ensure that precision criteria are met, and if acceptable, determine the sum of both chambers and use the formula to calculate the number of cells per microliter; see the example provided in Box 17.3.
Depending on the approach used, (A) is either (1) the average number of cells counted in a chamber (Option 1, Box 17.3) or the sum of the number of cells counted in both chambers (Option 2, Box 17.3) of the hemacytometer. This value (A) is multiplied by the dilution factor (B), which accounts for any dilution made of the fluid. If the fluid is analyzed without diluting, this factor is 1. The cells were distributed in the actual volume of fluid evaluated (C). This volume (mm3 = μL) is determined by multiplying the area counted (mm2) by the depth between the coverslip and the chamber, which is standardized at 0.1 mm. Disposable hemacytometers have a fixed (immovable) coverslip, which maintains the depth of 0.1 mm. However, when using a nondisposable hemacytometer with a removable coverslip, if the chamber is overfilled or underfilled, the depth is not the assumed value of 0.1 mm, which will cause erroneous results. The concentration of cells per microliter can easily be converted to the cell number per liter of fluid as follows.
 Equation 17.2
Equation 17.2
Hemacytometer Calculation Examples
Following are some examples using a Neubauer hemacytometer to determine cell counts of body fluids using undiluted and diluted fluid. The calculations used follow Option 2 described in Box 17.3. Note that an additional dilution factor is required when a fluid is “pretreated” before dilution for cell counting, which can occur with synovial fluid or semen (see Example C).
Example A: Using Undiluted Body Fluid
A well-mixed undiluted CSF specimen is loaded on a hemacytometer, and the WBCs in five large squares (4 “W”+Center) are counted in each chamber (i.e., 5 mm2). The following results are obtained.
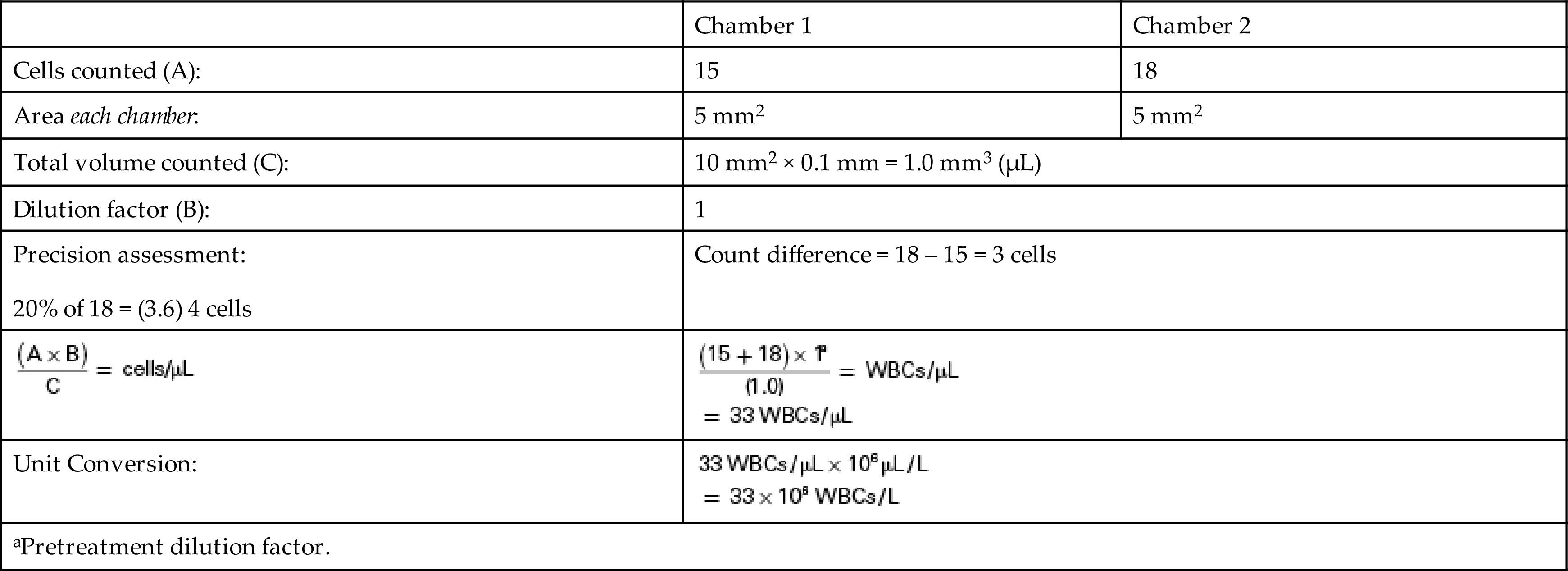
Note that the counts from both chambers agree within precision criteria of less than or equal to 20%, which indicates that the fluid was well mixed and equivalently dispensed in both chambers. The difference in cell number between side 1 and side 2 (18 – 15) is 3 cells, which is less than 20% (3.6 cells = 18 × 0.20) and acceptable.
Example B: Using Diluted Body Fluid
A well-mixed, hazy-appearing synovial fluid specimen is diluted 1:2 in duplicate using hypotonic saline. Both dilutions are loaded on a hemacytometer—one dilution on each side. The WBCs in five large squares (4 “W”+Center) are counted in each chamber (i.e., 5 mm2). The following results are obtained.

Note that the counts from both chambers do not agree within 20%. The difference in cell number between side 1 and side 2 (58 – 44) is 14 cells, which exceeds 20% (11.6 cells = 58 × 0.20), which indicates that the steps used to prepare both dilutions (mixing, pipetting, and diluting) were not equivalent. The dilutions and the cell counts should be repeated.
Example C: Sperm Count Using Diluted Semen
A semen specimen was pretreated 1:2 using a bromelain enzyme preparation to get it to liquefy. For the sperm count, the fluid is diluted 1:20, in duplicate. Both dilutions are loaded on a hemacytometer—one dilution on each side. The five “R” squares in the large central square (i.e., four small corner+center squares) of the hemacytometer are counted in each chamber (i.e., 0.2 mm2). The following results are obtained. Note that sperm counts are reported as the number of sperm per milliliter (mL), which requires the use of a different unit conversion factor.
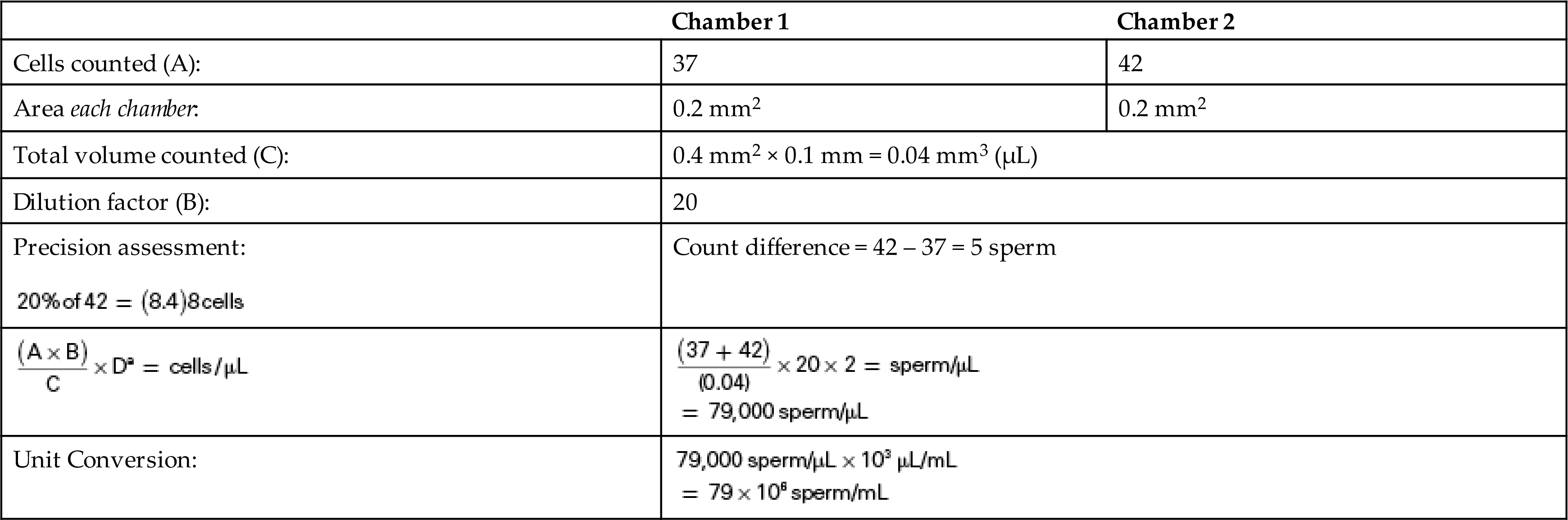
The dilution factor (D) accounts for the dilution made when the fluid was pretreated to get it to liquefy using the bromelain enzyme solution. In this example, the semen counts from both chambers agree within 20%. The difference in sperm number between side 1 and side 2 (42 – 37) is 5 sperm. This value is less than 20% (8.4 sperm = 42 × 0.20), which indicates that the steps used to prepare both dilutions (mixing, pipetting, and diluting) were equivalent.
Preparation of Slides for Differential
Slides should be prepared as soon as possible after fluid collection. Delays will result in loss of morphologic cellular detail, as well as antigenic reactivity.3 When preparing slides of body fluids for the WBC differential, cytocentrifugation is the preferred technique.1 Cytocentrifugation optimizes cell recovery, concentrates the cells in a limited area on the microscope slide, and creates a monolayer that optimizes microscopic viewing. In addition, this method is fast and easy to perform. Note that wedge smears (push smears) should not be used because of their inferior ability to preserve intact cells.1
Several other techniques can be used to concentrate body fluids. The easiest and least expensive technique is simple centrifugation of the body fluid, but cell recovery varies, and cells can become damaged and distorted at high-speed centrifugation. Although sedimentation methods preserve cellular morphology, cell recovery is not good. Filtration techniques using commercial filters (e.g., from manufacturers Millipore Corp., Nucleopore, and Gelman Instrument Co.) also have excellent cellular recovery (≈90%). These techniques are time-consuming, and after the cells have been concentrated a suitable smear must be prepared, which requires significant technical skill; hence, cytocentrifugation predominates in laboratories for preparing smears from body fluids.
Before slide preparation, body fluid specimens must be gently mixed. Only fresh body fluid specimens should be used to prepare cytocentrifuge slides. If there has been a significant delay since collection (i.e., longer than 4 hours for CSF), erroneous differential counts can occur because of cellular degeneration.1
Cytocentrifugation
Numerous cytocentrifuges are commercially available and require the use of specially designed assemblies for each sample (Fig. 17.2). An assembly consists of a microscope slide, filter paper with a circular opening, and a chamber that holds the fluid specimen (Fig. 17.3).


When the body fluid is clear, usually 5 drops (≈0.25 mL) of body fluid is added directly to the chamber. However, depending on the nucleated cell count, as few as 2 drops or as many as 10 may be used. Table 17.3 provides a guideline based on the nucleated cell count for the volume of body fluid to use when preparing a slide by cytocentrifugation. When a dilution is needed, normal saline is most often used; however, some laboratories use a diluent that lyses RBCs for bloody samples (e.g., hypotonic saline). Appropriate dilutions will reduce distortions associated with the overcrowding of cells and ensure a monolayer of cells for viewing. Note that laboratories must establish their own dilution protocols because the appropriate dilution depends on the amount of sample used, as well as the duration and speed of cytocentrifugation.
Table 17.3
| Nucleated Cell Count, cells/μL | Drops of Body Fluid |
|---|---|
| 0–100 | 10 |
| 100–500 | 5–6 |
| 500–1000 | 3–4 |
| >1000 | 2 |
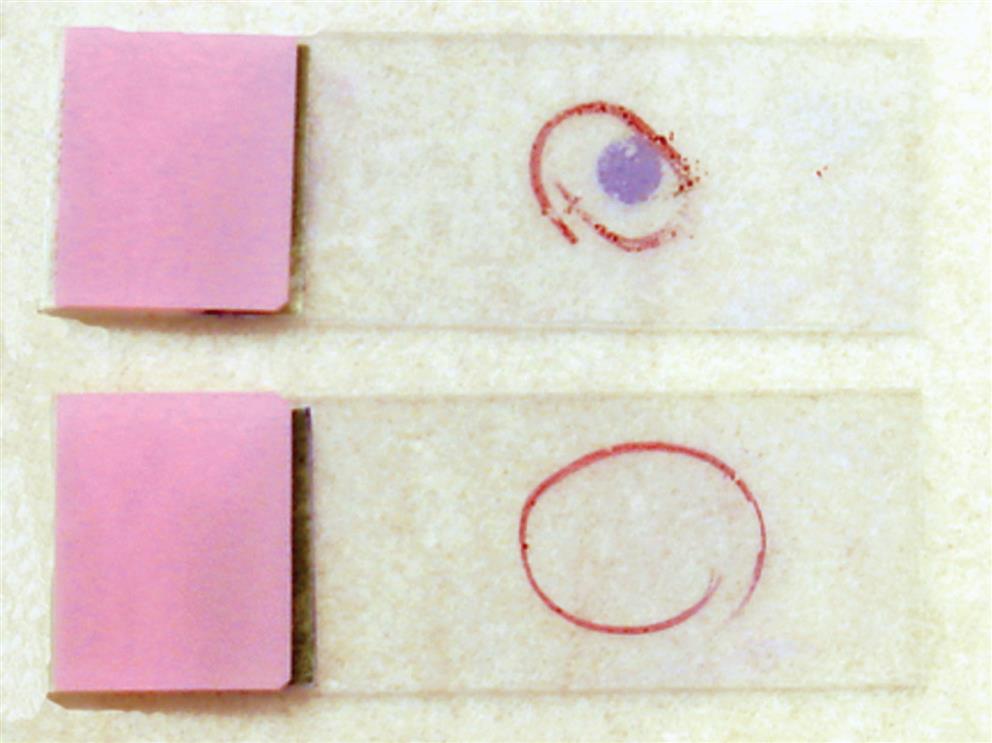
Each assembly is placed onto the rotor of the cytocentrifuge, and when the instrument is activated, centrifugal force pulls the body fluid from the sample chamber to the microscope slide. The cells adhere to the glass slide while the liquid is absorbed by the surrounding absorbent filter paper. During centrifugation the cells concentrate as a monolayer in the open circular area of the filter to form a “cell button” on the microscope slide. The centrifugal force is low (e.g., 600–800 × g) to minimize cell distortion; the time is long enough to ensure adequate drying of the cell button. Microscope slides specifically designed for cytocentrifugation are available, and they have a white circular ring that surrounds the cell button area. This assists the microscopist when the body fluid sample contains few cells that are not visually apparent macroscopically on the slide. Another option is to use a wax pencil to mark the backside of the glass microscope slide, indicating the region of the cell button (Fig. 17.4).
During cytocentrifugation, some cells are lost to the filter paper, but this loss is not selective (i.e., all cells are equally affected); consequently, the remaining cell distribution in the cell button is accurate and representative. Several predictable cellular distortions that may be observed are listed in Box 17.4. Most are associated with high cell counts, the cytocentrifugation process (speed and time), or a time delay when an older specimen is used (i.e., not fresh).3 To reduce these artifacts, laboratories should determine the optimal speed and time of cytocentrifugation for their instrument, use fresh specimens, and prepare appropriate dilutions of fluids with high cell counts.
For specimens that have a low protein content (e.g., CSF), adding a drop of 22% albumin to the sample chamber before adding the body fluid enhances adherence of cells to the glass slide and reduces cell distortion (smudging) or disintegration.1,3
Slide Preparation and Review
Slide preparations are stained using Wright or Wright-Giemsa stain performed manually or automatically using a slide stainer. The hand-drawn or premarked circle on the microscope slide indicates the location of the cell button (see Fig. 17.4).
Adjust the microscope to low-power (×100) magnification and thoroughly scan the entire cell button looking for cell clumps, which are characteristic of malignancies. Note that malignant cells can be present in low numbers, and even a single malignant cell is clinically significant. However, remember that not all cell clumps are composed of malignant cells.
Using oil immersion magnification (e.g., ×50 or ×1000), the nucleated cell or WBC differential is performed using any representative area of the cell button. A systematic approach to viewing should be used (similar to that used with blood smears) to prevent erroneous repeat counting of the same cells. Ideally, 100 to 300 cells should be evaluated.
Study Questions
- 1. Which of the following statements is not associated with the performance of cell counts using a manual hemacytometer?
- 2. Which of the following diluents will cause synovial fluid to form a mucin clot?
- 3. Which of the following diluents should be used when an RBC count is requested?
- 4. An air displacement pipette cannot accurately dispense
- 5. Which of the following actions will adversely affect the cell count obtained using a hemacytometer?
- 6. In the pretreatment of a synovial fluid with hyaluronidase, a 1:10 dilution is made, after which a WBC count is performed using a 1:20 dilution of this fluid. The WBCs in the four large corner squares (“W”) and the center square are counted in each chamber (i.e., 5 mm2). Both sides of the hemacytometer were evaluated with 37 cells and 43 cells counted in chamber 1 and chamber 2, respectively. What is the average cell count that should be reported?
- 7. A WBC count is performed using a 1:2 dilution of CSF, and the four large corner squares (“W”) and the center square are counted in each chamber (i.e., 5 mm2). Both sides of the hemacytometer were evaluated with 31 cells and 23 cells counted in chamber 1 and chamber 2, respectively. What should be done next?
- 8. Distortions observed on cytocentrifuge slide preparations have been associated with