Gas Diffusion
After reading this chapter, you will be able to:
- • Differentiate between diffusion and bulk gas flow.
- • Use the alveolar gas equation to perform various calculations.
- • Explain why the respiratory exchange ratio affects the calculation of alveolar oxygen pressure (PAO2).
- • Identify the factors that affect diffusion, as illustrated by Fick’s law.
- • Use Graham’s law and Henry’s law to explain the differences in oxygen (O2) and carbon dioxide (CO2) diffusion rates in the lung.
- • Explain why O2 transfer from lung to blood is perfusionlimited but carbon monoxide (CO) transfer is diffusionlimited.
- • Explain why CO (rather than O2) is the test gas normally used for measuring the diffusion capacity of the lung.
- • Explain how diffusion capacity is measured in a pulmonary function laboratory.
- • Correlate disease entities with the abnormal processes that decrease diffusion rate.
- • Explain why the diffusion capacity of the lung for carbon monoxide (DLCO) test detects oxygenation problems in the natural progression of disease before abnormalities in arterial blood oxygen pressure (PaO2) become evident.
Key Terms
diffusion
diffusion capacity of the lung for CO (DLCO)
Fick’s law
Graham’s law
Henry’s law
polycythemia
respiratory exchange ratio (R)
single-breath CO diffusion test
What is diffusion?
Diffusion is the result of high-speed random motion of gas or liquid molecules. For example, if the number of molecules present in a hypothetical area A is greater than in area B, mathematical probability dictates that more molecules will move from A to B than from B to A. This net movement of molecules from high to low concentrations is called diffusion. Diffusion continues until the molecules are evenly distributed everywhere, such that the numbers of molecules moving from A to B and from B to A are the same. This state is known as equilibrium.
The high-speed random impacts of atmospheric air molecules on solid surfaces create the atmosphere’s pressure. Air is a gas mixture; the contribution each gas makes to the atmospheric pressure is proportional to the number of its molecules present (i.e., each gas exerts its own partial pressure, as explained in Chapter 4). A gas diffuses from one point to another in a gas mixture when there are differences in its partial pressures within the mixture; the direction of diffusion is always from high to low partial pressure. When no partial pressure difference exists for any gas throughout the mixture, equilibrium is present. Individual gas partial pressure differences are called diffusion gradients. During diffusion, each gas in a mixture moves down its own diffusion gradient, from high pressure to low pressure. This means two different gases may simultaneously diffuse in opposite directions because of oppositely oriented partial pressure gradients. This occurs for oxygen (O2) and carbon dioxide (CO2) across the alveolar-capillary membrane (Fig. 7.1).
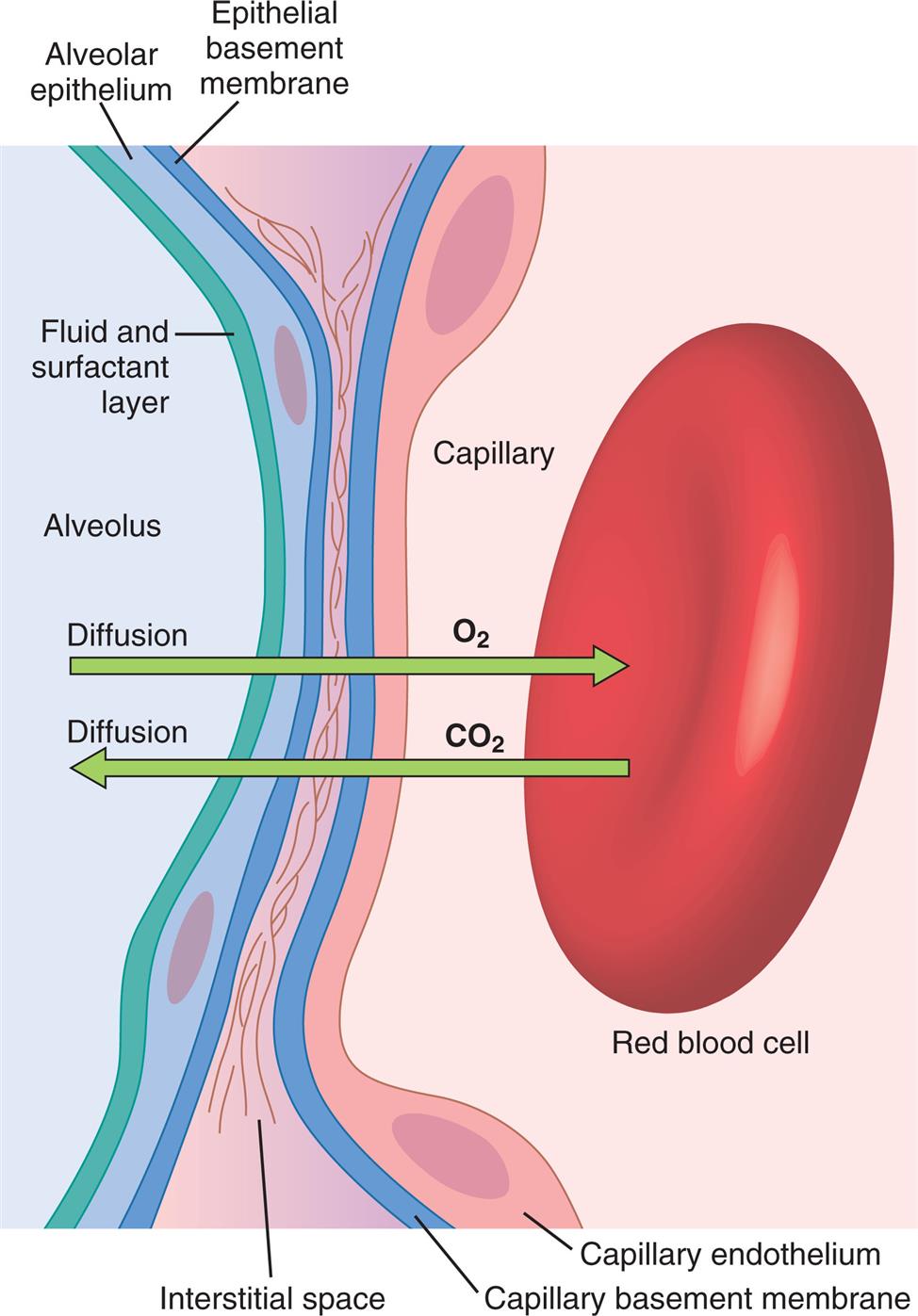
Diffusion is not the same as bulk gas flow, in which a large pressure gradient causes all molecules of all gases to move together in one direction. An example of bulk gas flow is ventilation, in which mouth and alveolar pressure differences cause all gas molecules in the mixture that is air to move together, in and out of the lungs. In terminal airways and alveoli, random molecular diffusion is the main mechanism by which gas molecules reach the alveolar membrane surface.
Diffusion Gradients of Respiratory Gases
Fig. 7.2 illustrates diffusion gradients between alveolar gases and blood and between blood and body tissues. Inspired air contains about 21% O2 and almost 0% CO2. Inspired oxygen partial pressure (PIO2) is almost 160 mm Hg, as the following calculation shows:
 1
1
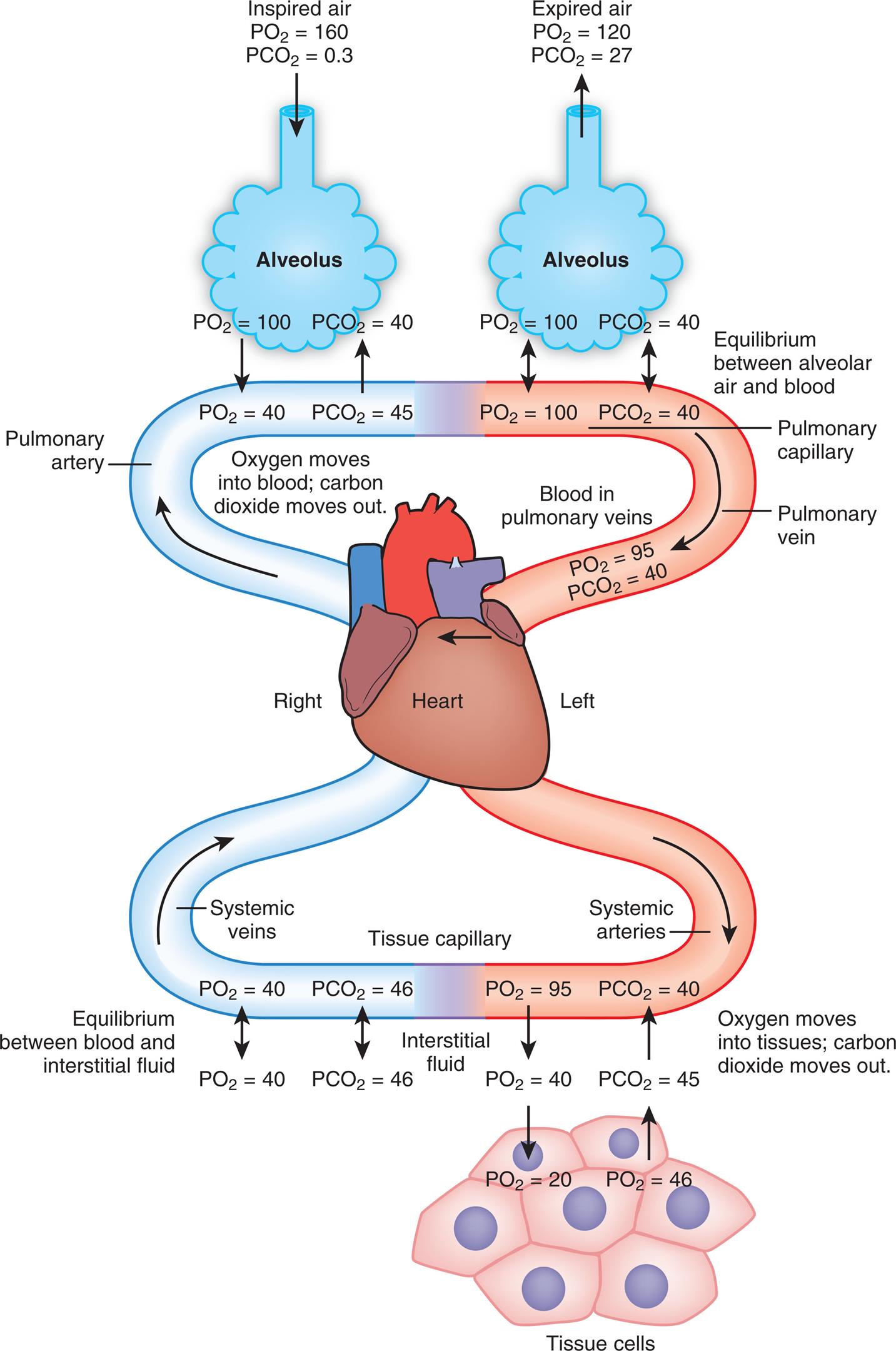
PO2 in the conducting airway gas is lower than PO2 in the atmosphere because gas in the lung is 100% saturated with water vapor. At body temperature, the partial pressure of water vapor in the lung (PH2O) is 47 mm Hg. Therefore PO2 in conducting airways is about 150 mm Hg, as the following calculation shows:
 2
2Alveolar PO2 (PAO2) is lower still because O2 diffuses out of the alveoli, lowering PAO2 to about 100 mm Hg. (Calculation of PAO2 is discussed in the next section.)
The diffusion gradient between alveolar gas and mixed venous blood is much larger for O2 than it is for CO2 (60 mm Hg vs. 6 mm Hg, respectively), as shown in Fig. 7.2. At rest, these diffusion gradients transfer about 250 mL of O2 into the blood and 200 mL of CO2 into the alveoli each minute. By the time blood leaves the alveolar capillary, the PO2 and PCO2 of the blood have reached equilibrium with alveolar gases, even during exercise when blood flows very rapidly through the capillary. PO2 of blood entering the left atrium is never as high as PO2 of blood leaving the pulmonary capillaries (see Fig. 7.2) because a small amount of deoxygenated bronchial venous blood mixes with capillary blood; this constitutes a normal anatomical shunt. Anatomical shunt is mostly responsible for the normal P(A-a)O2 (alveolar-to-arterial oxygen pressure difference). Left atrial blood then normally flows unaltered into the systemic arteries.
Alveolar Gas Equation
The sum of all gas pressures at any point in the lung is equal to 760 mm Hg at sea level. When air—which is CO2-free—is inspired and enters the alveoli, its PCO2 immediately increases to about 40 mm Hg as it mixes with the CO2 that diffuses into the alveoli from capillary blood; because the sum of all alveolar gas pressures is constant (760 mm Hg), the PAO2 must decrease by a similar amount. If the amount of O2 molecules diffusing out of alveoli into the blood each minute were exactly equal to the amount of CO2 molecules diffusing from the blood into alveoli each minute, PAO2 would be calculated by simply subtracting PACO2 (normally 40 mm Hg) from the result of eq. 2. However, O2 diffuses out of the alveolus at a greater rate than CO2 diffuses into the alveolus. At rest, pulmonary capillary blood removes about 250 mL of O2 per minute from the alveoli, but only 200 mL/min of CO2 diffuses from the blood into the alveoli. The ratio of CO2 diffusion into the alveoli ( ) to oxygen diffusion out of the alveoli (
) to oxygen diffusion out of the alveoli ( ) is called the respiratory exchange ratio (R), and its value is normally about 0.8 (
) is called the respiratory exchange ratio (R), and its value is normally about 0.8 ( ). In other words, the amount of CO2 entering the alveoli is equal to 0.8 or 80% of the amount of O2 leaving the alveoli.
). In other words, the amount of CO2 entering the alveoli is equal to 0.8 or 80% of the amount of O2 leaving the alveoli.
Simply stated, when inspired gas enters the alveoli, its PO2 falls because O2 immediately diffuses into capillary blood; we need to know how much the PO2 falls, and then subtract this amount from the inspired PO2 calculated in eq. 2. Knowledge of R’s value helps us compute this amount. For example, if R is equal to its normal value of 0.8, then the milliliters of CO2 entering the alveolus is equal to 80% of the milliliters of O2 that left the alveolus. Similarly, if PACO2 is 40 mm Hg, then 40 mm Hg is equal to 80% of fall in inspired PO2 that occurred when gas entered the alveoli. In this example, if x equals the fall in inspired PO2, then 40 mm Hg = 0.8(x), which means x = 40 mm Hg/0.8, or 50 mm Hg. PAO2 would then be calculated by subtracting 50 mm Hg from inspired PO2 computed in eq. 2. In the clinical setting, PaCO2 is substituted for PACO2 because these values are generally almost equal and because PaCO2 is a traditionally measured arterial blood gas value.
A more complex version of the alveolar gas equation increases accuracy but is clinically insignificant. For the sake of completeness, a brief explanation follows: The fact that more O2 diffuses out of the alveoli than is replaced by CO2 diffusing into the alveoli causes the alveolar gas volume to shrink slightly, but since alveolar gas ultimately communicates with the atmosphere through the trachea, alveolar gas pressure must equal atmospheric gas pressure, which is 760 mm Hg at sea level. The shrinkage in alveolar volume concentrates the alveolar nitrogen molecules, which the more complex alveolar gas equation takes into account. This equation is known as the ideal alveolar gas equation because it assumes the ventilation-to-blood flow ratios of each alveolus in the lung are identical:
 3
3
In this equation PIO2 equals FiO2 (760 − 47). The bracketed part of eq. 3 is a correcting factor that takes into account the effect of R on PAO2. When R equals 1, the correction factor equals 1 and does not need to be applied.
The term 1 − FiO2 is actually equal to the inspired nitrogen concentration. When R is less than 1, the effect is to increase nitrogen concentration, causing the bracketed term of Eq. 3 to increase to values greater than 1. In eq. 3, if R = 0.8 and FiO2 = 0.21 (room air), the bracketed factor is equal to 1.1975, or 1.2.
Examination of eq. 3 shows that higher FiO2 values require progressively smaller correction factors; at 100% inspired oxygen (FiO2 = 1.0), no correction is needed. A sufficiently accurate equation for clinical use is a simplified form of eq. 3 for patients breathing an FiO2 of 0.60 or less. This is shown as follows:
 4
4
For FiO2 values greater than 0.60, a sufficiently accurate clinical equation is as follows:
 5
5
In Eqs. 4 and 5, arterial PCO2 (PaCO2) is substituted for alveolar PCO2 (PACO2) because these two values are generally almost equal, unless the lungs have a high degree of alveolar dead space. PaO2 cannot be used as an estimate of PAO2 because shunting always creates an alveolar-arterial PO2 difference.
A normal PAO2 for a person breathing room air at sea level, with a PaCO2 equal to 40 mm Hg and an R equal to 0.8, is about 100 mm Hg (using eq. 4). This is shown as follows:
 5
5
Table 7.1 summarizes respiratory gas partial pressures at sea level in dry inspired air, humidified (tracheal) air, alveolar air, and mixed-expired air. Expired gas PO2, PCO2, and PN2 differ from alveolar values because expired air contains dead space gas mixed with alveolar gas.
Laws Governing Diffusion
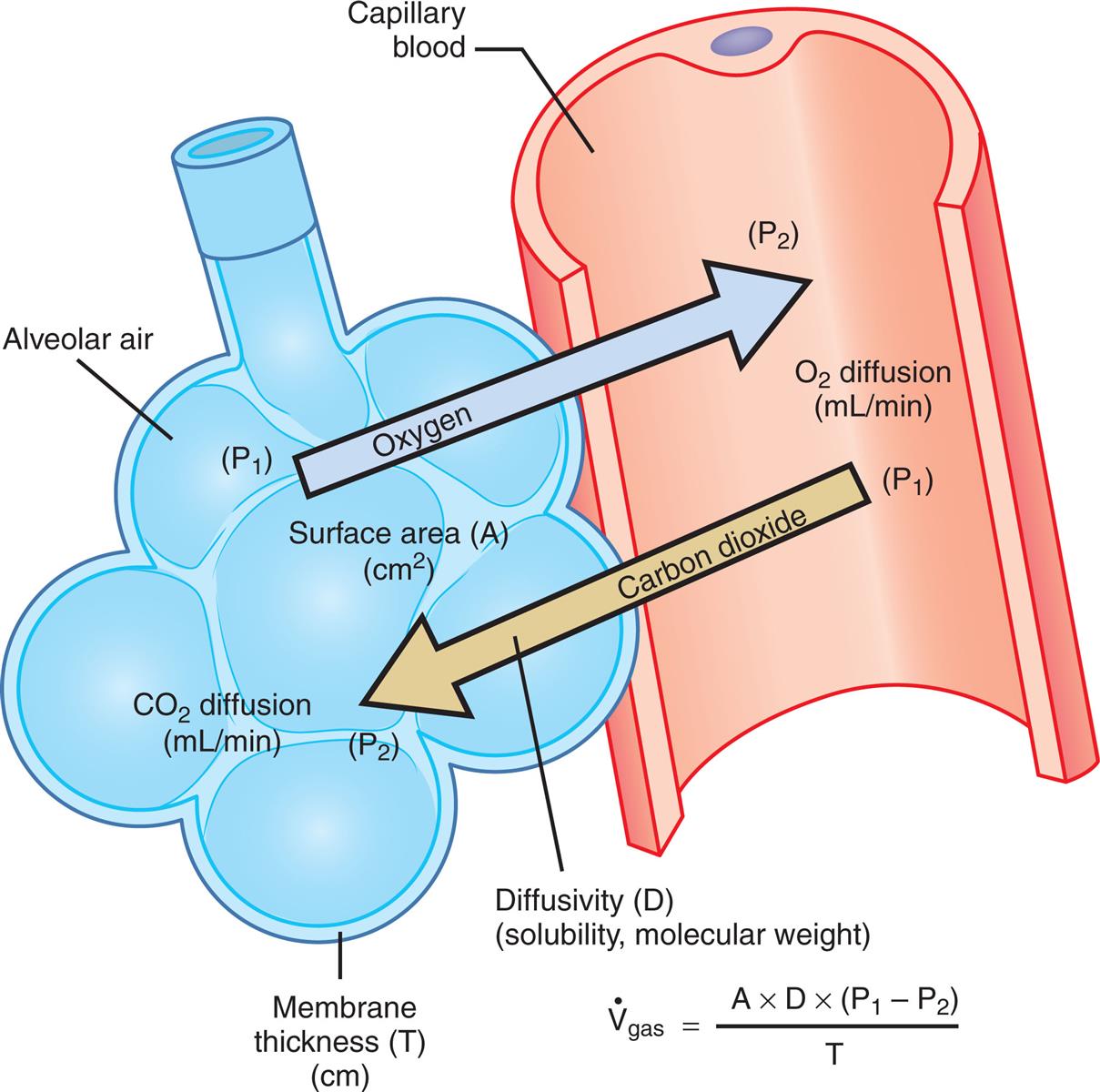
Fick’s law summarizes the factors that determine the rate of gas diffusion through the alveolar-capillary membrane. This is shown as follows:

In this equation,  represents the volume of gas diffusing through the membrane per minute (milliliters per minute), and A represents the surface area of the membrane available for diffusion (cm2).
represents the volume of gas diffusing through the membrane per minute (milliliters per minute), and A represents the surface area of the membrane available for diffusion (cm2).
D represents the diffusion coefficient, or diffusivity, of a particular gas. It is directly proportional to the gas solubility (sol) but inversely proportional to the square root of the gas molecular weight (mw):
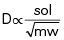
P1 − P2 represents the partial pressure difference across the membrane (i.e., diffusion pressure gradient [mm Hg]). T represents the membrane thickness, or diffusion path distance (cm).
This equation states that  increases if there are increases in the membrane surface area, gas diffusivity, or diffusion pressure gradient. The diffusion rate decreases if membrane thickness increases. Doubling the membrane surface area or diffusion pressure gradient doubles the overall diffusion rate; doubling the membrane thickness reduces the diffusion rate by half. Fig. 7.3 illustrates the factors involved in Fick’s law.
increases if there are increases in the membrane surface area, gas diffusivity, or diffusion pressure gradient. The diffusion rate decreases if membrane thickness increases. Doubling the membrane surface area or diffusion pressure gradient doubles the overall diffusion rate; doubling the membrane thickness reduces the diffusion rate by half. Fig. 7.3 illustrates the factors involved in Fick’s law.
Physical Gas Characteristics and Diffusion
O2 and CO2 diffuse through gaseous and liquid phases in the lung; the alveolar-capillary membrane (see Fig. 7.1) is a liquid barrier. Light gases diffuse more rapidly than heavier gases, and highly soluble gases diffuse through liquids more rapidly than less soluble gases. The rate of gas diffusion in the lung is inversely proportional to its molecular weight and directly proportional to its solubility; the diffusion coefficient in Fick’s law is derived from these two factors. Specifically, the gas diffusion rate is inversely proportional to the square root of its gram molecular weight (gmw) (Graham’s law). Relative rates of diffusion for O2 (molecular weight = 32) and CO2 (molecular weight = 44) in a gaseous medium are as follows:

Because O2 is a lighter molecule, it diffuses through a gas medium 1.17 times faster than CO2.
However, CO2 is much more soluble in water than O2. Henry’s law states that the amount of gas dissolving in a liquid is directly proportional to the gas partial pressure. At body temperature (37°C) and 760 mm Hg pressure, 0.592 mL of CO2 and 0.0244 mL of O2 dissolve in 1 mL of water. CO2 is about 24 times more soluble than O2, as the following equation shows:

Combining Graham’s law and Henry’s law, CO2 diffuses across the alveolar-capillary membrane about 20 times faster than O2. This is shown as follows:

For this reason, alveolar-capillary membrane defects limit O2 diffusion long before they limit CO2 diffusion. In a practical, clinical sense, the alveolar-capillary membrane never limits outward diffusion of CO2 from blood to alveoli. Discussion of diffusion in this chapter is therefore primarily focused on O2 diffusion.
Factors Affecting Oxygen Diffusion
Factors influencing the rate of O2 transfer across the alveolar-capillary membrane include (1) the partial pressure gradient across the membrane, (2) the diffusion path length, and (3) the membrane surface area. (All factors are included in Fick’s law of diffusion.)
Effects of the Partial Pressure Gradient and Capillary Blood Transit Time on Gas Equilibrium
At a resting cardiac output, a red blood cell (RBC) spends about 0.75 second traveling through the pulmonary capillary. Normally, the equilibrium between alveolar gas and capillary blood PO2 occurs within the first 0.25 second, or about one-third of the distance through the capillary (Fig. 7.4, dark curved line). The time to reach equilibrium may be prolonged if alveolar-capillary membranes are thickened (light curved line in Fig. 7.4). Even so, equilibrium is practically always achieved at rest. The rate of diffusion is rapid at first, when the partial pressure gradient across the alveolar-capillary membrane is greatest. The diffusion rate then continuously slows as the partial pressure gradient diminishes (notice the shape of the PO2 curve [dark line] in Fig. 7.4) until diffusion completely ceases at equilibrium. Thus during the last two-thirds of travel through the pulmonary capillary, no net diffusion occurs.
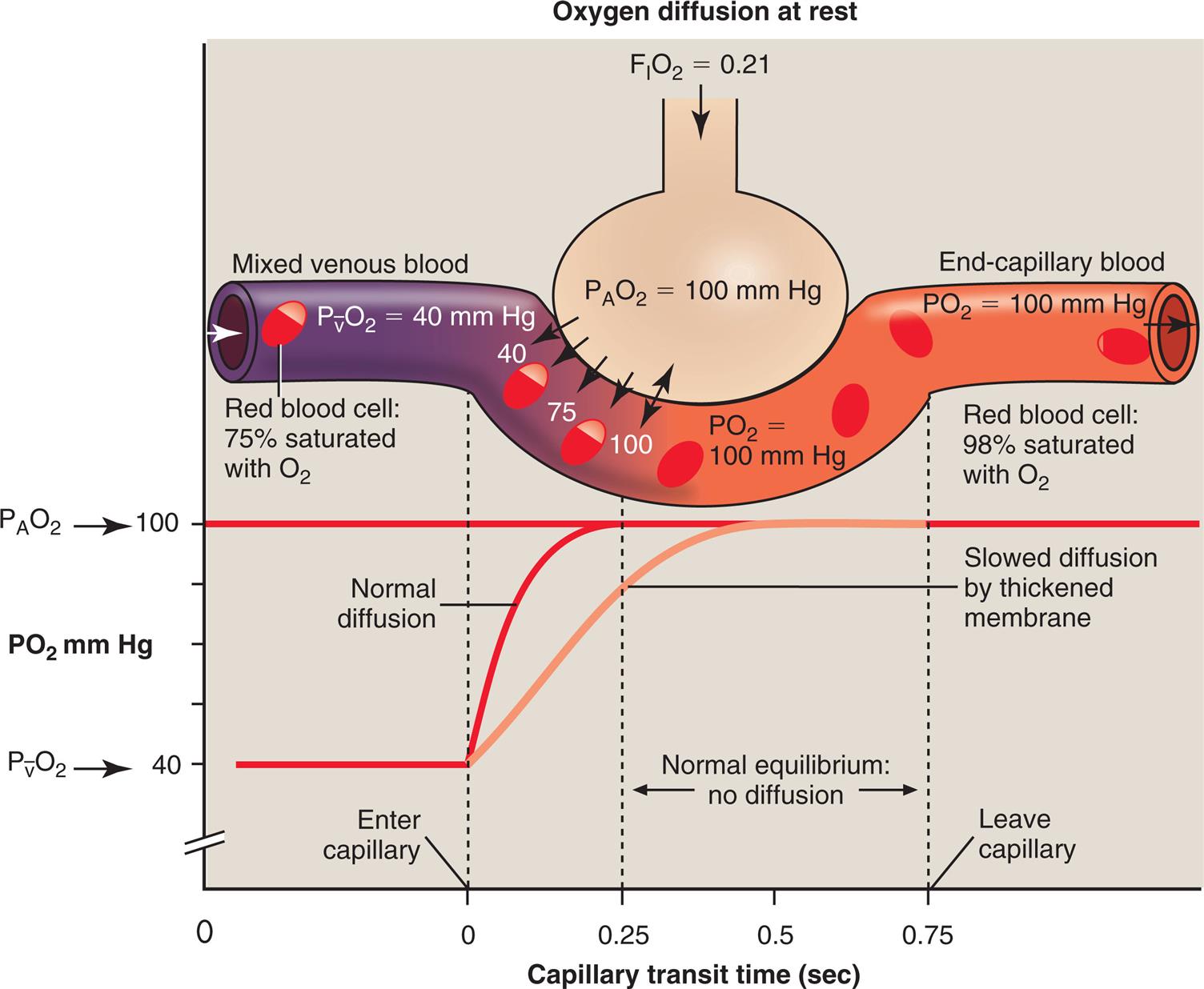
Even when capillary transit time is shortened to 0.25 second by a high blood flow rate during exercise, complete O2 equilibrium normally still occurs between alveolar gas and end-capillary blood; it simply occurs at the end of the capillary path. In such a situation, the number of O2 molecules transferred into the blood each minute increases greatly, because the greater rate of blood flow takes up more O2 molecules from the alveoli each minute. Another reason exercise increases the number of O2 molecules transferred each minute is that previously nonperfused capillaries are recruited, which increases the surface area for diffusion.
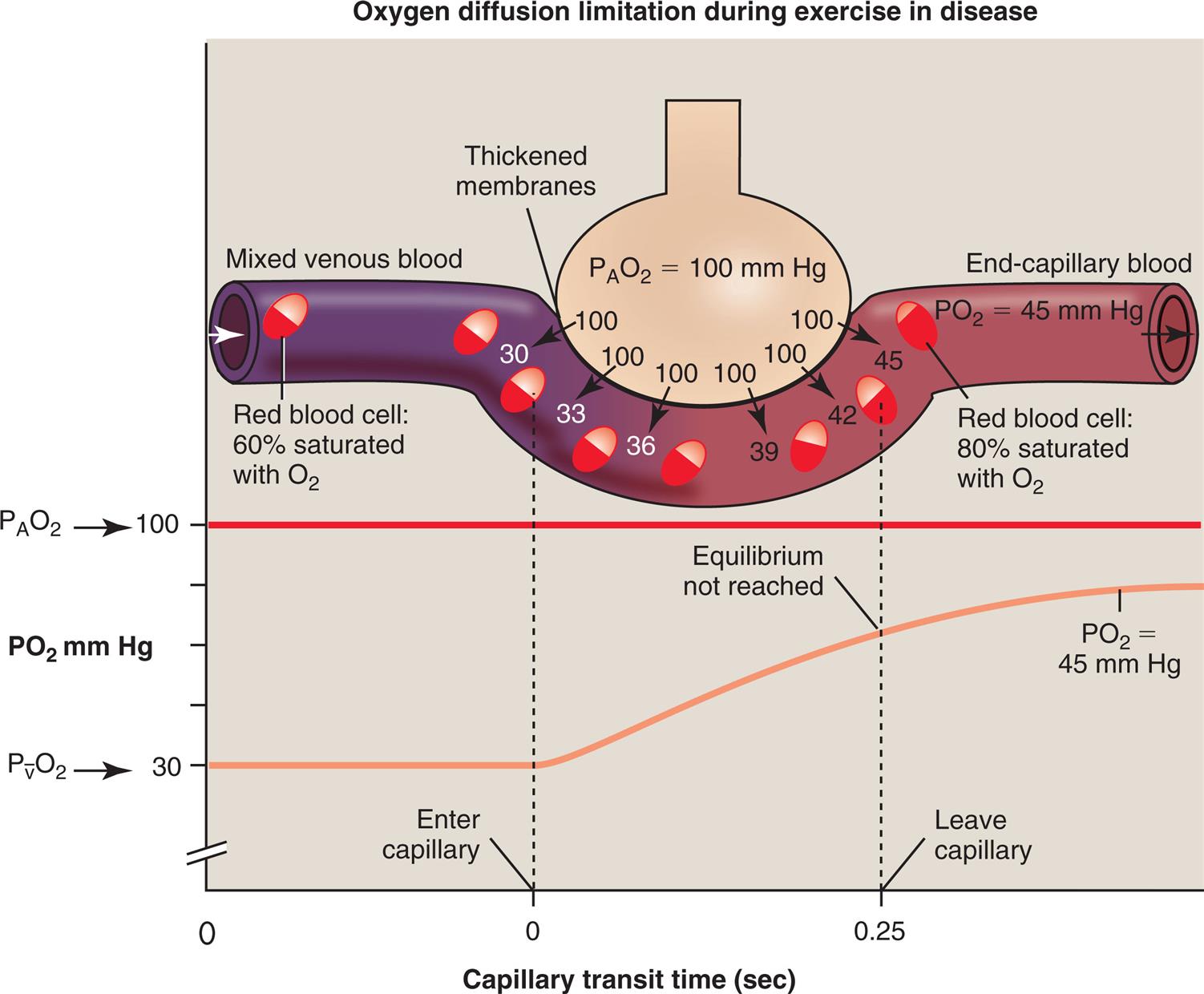
If a healthy person exercises vigorously at a high altitude, where atmospheric PO2 is very low, the O2 diffusion gradient may be so low that O2 cannot diffuse across the alveolar-capillary membrane fast enough to establish equilibrium during the shortened capillary transit time. Alternatively, if disease thickens the alveolar-capillary membrane, the diffusion rate across the membrane may be slowed enough to prevent complete O2 equilibrium by the time blood leaves the capillary during exercise. In the clinical setting, this situation rarely occurs at rest, even in severe lung disease.1 However, exercise immediately exposes the diffusion abnormality problem because it shortens the blood’s transit time through the capillary, making it likely that alveolar and capillary O2 equilibrium never occur (Fig. 7.5). Patients with thickened alveolar-capillary membranes, as seen in pulmonary fibrosis, are most likely to show evidence of O2 diffusion impairment during exercise.2
Perfusion and Diffusion Limitations to Oxygen Transfer
Fig. 7.4 shows that diffusion normally stops when O2 equilibrium is reached between the alveolus and capillary, which occurs long before the blood travels the length of the capillary. If blood flow rate (perfusion) increases, more O2 leaves the lung each minute because the O2-saturated blood moves out of the capillary more quickly, which means deoxygenated blood enters more quickly to take up more O2. Therefore the O2 diffusion rate through the alveolar-capillary membrane is normally perfusion limited (see Fig. 7.4); that is, a change in blood flow rate alters the number of O2 molecules that cross the alveolar-capillary membrane each minute. If O2 equilibrium between the alveolus and capillary never occurs because of thickened membranes, O2 transfer is truly diffusion limited. In such an instance, it is the alveolar-capillary membrane, not blood flow rate, that influences the O2 transfer rate (see Fig. 7.5).
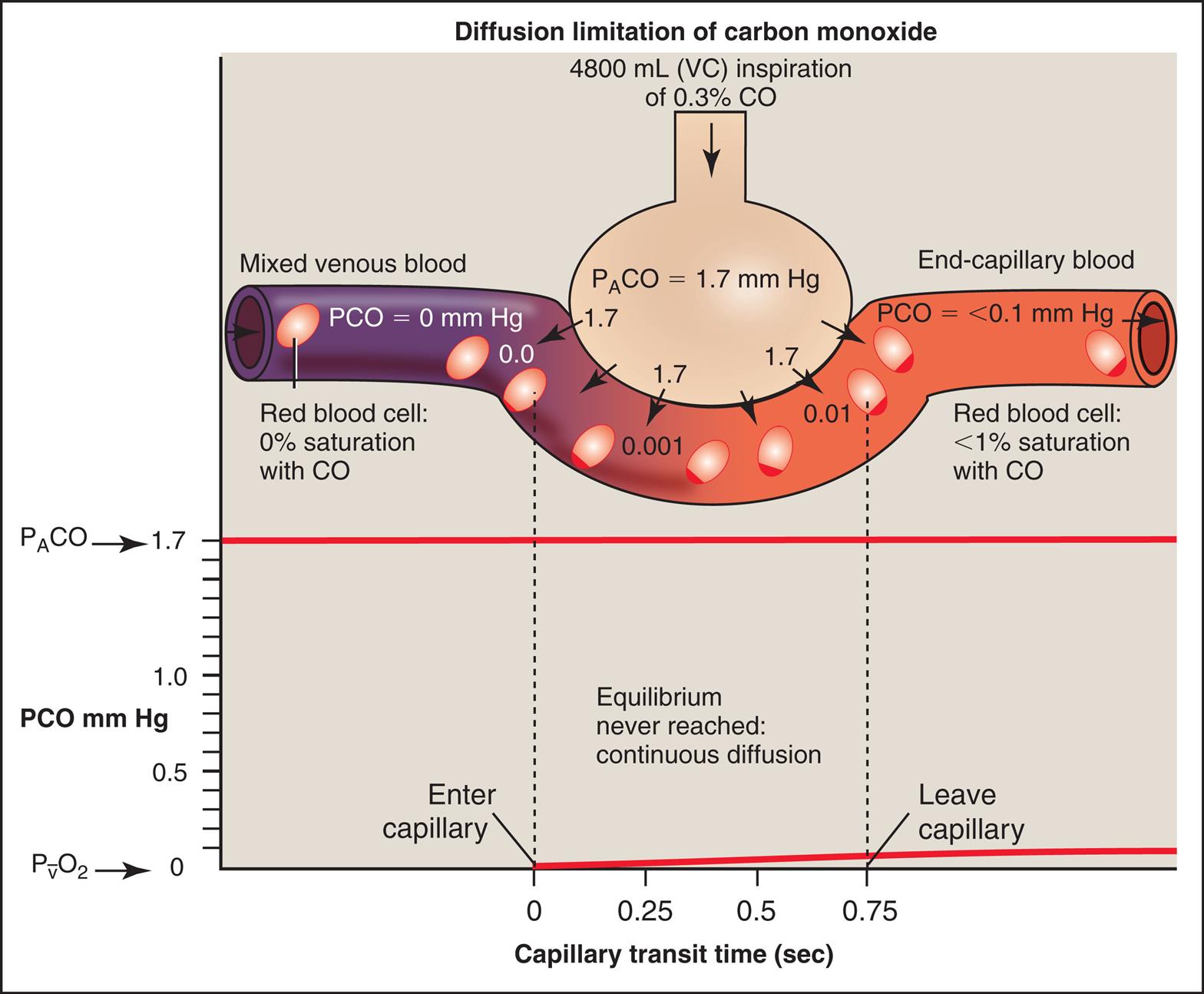
If one wishes to evaluate the extent to which the alveolar-capillary membrane itself impedes the diffusion rate (milliliters per minute of O2 transfer per millimeters of mercury pressure gradient), one should not measure the O2 diffusion rate because it is affected by blood flow rate. Instead, one should measure the diffusion rate of a gas that never reaches equilibrium between alveolar gas and capillary blood, even in resting conditions. In other words, the blood’s capacity for this gas should be so great that the gas cannot diffuse across the membrane fast enough to saturate the blood to capacity before it leaves the capillary, regardless of blood flow rate. For such a gas, the only factor that limits diffusion is the resistance of the alveolar-capillary membrane itself. Carbon monoxide (CO) is the ideal gas for this kind of measurement because blood can absorb it at a much greater rate than CO can diffuse across the alveolar-capillary membrane, even under resting conditions. For this reason, CO is always diffusion limited. Fig. 7.6 illustrates the diffusion-limited characteristics of CO. All events that are illustrated in Fig. 7.6 occur at rest; an increased blood flow would not result in greater CO uptake because CO equilibrium never occurs between alveolar gas and capillary blood, which means CO is already diffusing through the alveolar-capillary membrane at its maximum rate under resting conditions. For this reason, CO is commonly used in pulmonary function laboratories to evaluate the lung’s true diffusion capacity.
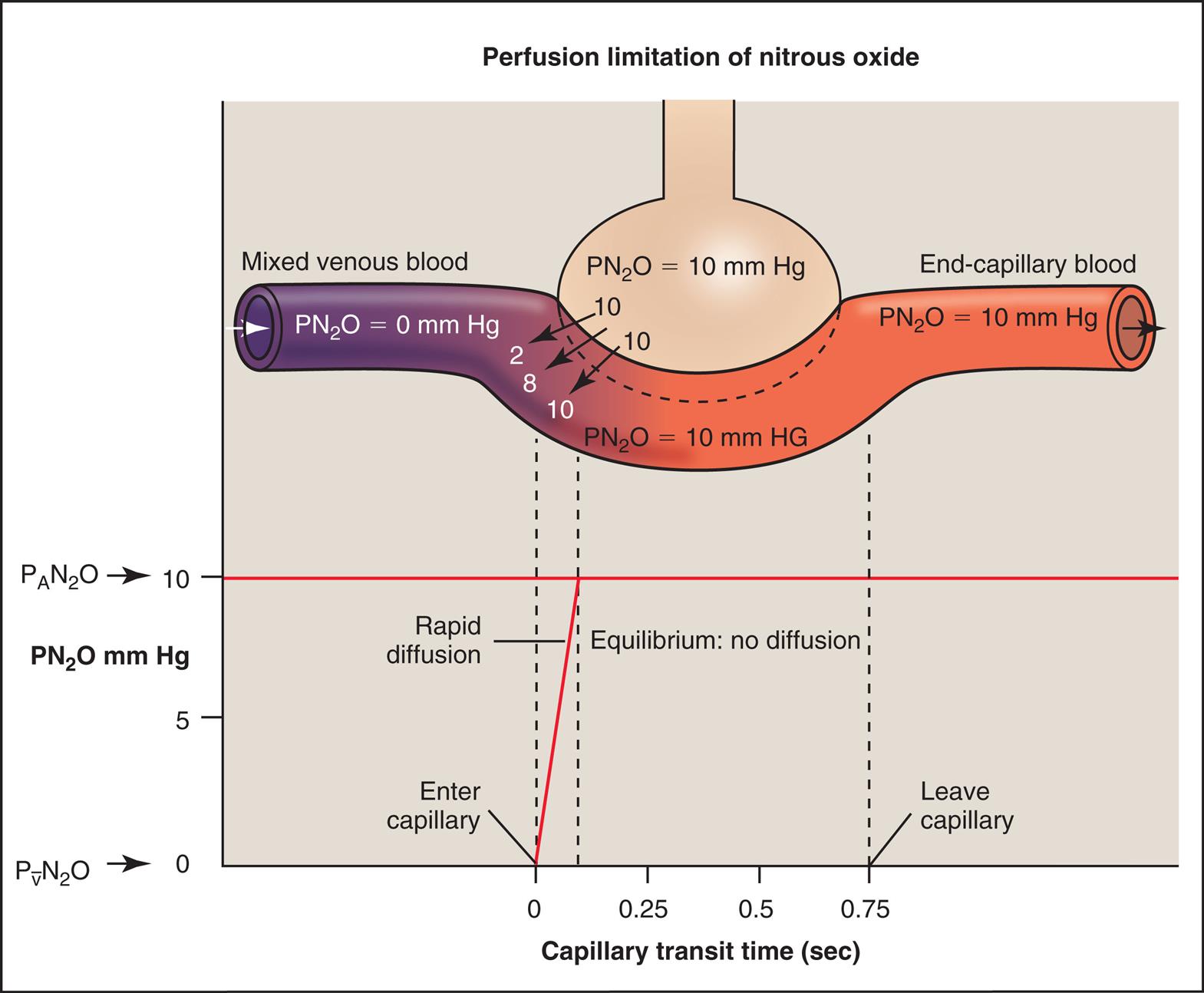
Hemoglobin in the RBC chemically binds with and takes up O2 and CO, in effect making these gases much more soluble in whole blood than they are in the alveolar-capillary membrane interstitial fluid or the blood plasma. In contrast, a gas such as nitrous oxide (N2O), which is highly soluble in the alveolar-capillary membrane and plasma, does not chemically bind with hemoglobin; therefore its solubility in whole blood is much less than that of CO or O2. If N2O is inhaled, the pulmonary capillary blood reaches its maximum capacity for N2O almost instantly (Fig. 7.7). N2O partial pressures across the alveolar-capillary membrane reach equilibrium in the first one-twentieth of the distance along the capillary. An increased blood flow in this situation would cause the N2O-saturated blood to exit the capillary sooner, allowing more mixed venous blood to enter the capillary and take up N2O from the alveolus. Thus diffusion of N2O in the lung is strictly perfusion limited because equilibrium between capillary blood and alveolar gas occurs almost instantly. This characteristic makes N2O an ideal test gas to measure pulmonary blood flow.
In the healthy lung, O2 diffusion is normally perfusion limited because its pressure equilibrates across the alveolar-capillary membrane long before blood leaves the capillary, even under exercise conditions; increased blood flow increases the amount of O2 taken up from the alveolus. However, in the unhealthy lung with thickened alveolar-capillary membranes, exercise may increase blood flow enough that O2 becomes diffusion limited—that is, the thickened membrane may prevent alveolar-capillary PO2 equilibrium from ever occurring.
Diffusion Path Length
The distance for diffusion includes the entire path length from alveolar gas to the hemoglobin in the RBC. It was originally thought that the alveolar-capillary membrane was the only important rate-limiting barrier to diffusion; it is now known that the RBC membrane and the rate at which O2 combines with hemoglobin limit the lung’s O2 uptake to the same extent as the alveolar-capillary membrane itself.3 The total diffusion path distance is normally less than 0.1 μ and includes the following elements (see Fig. 7.1): (1) the surfactant layer that lines the alveolar surface, (2) the alveolar epithelium, (3) the basement membrane of the alveolar epithelium, (4) the extremely thin interstitial space, (5) the basement membrane of the capillary endothelium, (6) the capillary endothelium, (7) the plasma, (8) the RBC membrane, and (9) the intracellular fluid bathing the hemoglobin molecule.
Various abnormal conditions can increase the diffusion path length, including the following: (1) fibrotic thickening of alveolar and capillary walls; (2) interstitial edema fluid, separating alveolar and capillary membranes; (3) fluid in the alveoli; (4) interstitial fibrotic processes that thicken the interstitial space; and (5) dilated, engorged capillaries, which allow RBCs to flow side by side. However, abnormalities that increase the distance for diffusion are rarely the cause of decreased arterial PO2 at rest. The major cause of resting hypoxemia in these patients is a mismatch between ventilation and blood flow.1 That is, the processes that increase diffusion distance generally also decrease lung compliance, which decreases ventilation in these areas. Blood perfusing these underventilated areas tends to be inadequately oxygenated, which ultimately lowers the PaO2.
Diffusion Surface Area
Diffusion surface area is the total area of contact between ventilated alveoli and perfused capillaries. A decrease in the number of open, perfused capillaries or in the number of open, ventilated alveoli decreases the diffusion surface area and the lung’s diffusion capacity. In such instances, diffusion capacity is decreased, although the diffusion path length may be normal at all points. For example, emphysema destroys alveolar walls and their associated capillaries, which decreases the diffusion surface area. Although the diffusion path length is not thickened at any point, overall diffusion rate is decreased because much of the functional gas-exchange surface area is lost.
Measuring Diffusion Capacity
General Principles
A strictly diffusion-limited test gas such as CO is used to measure the diffusion capacity of the lung. As clinically performed, the single-breath CO diffusion test measures the amount (in milliliters) of CO that diffuses across the alveolar-capillary membranes during a 10-second breath holding period after first inhaling a known concentration of CO. The average diffusion pressure gradient also must be calculated during the breath-holding period, that is, the mean PACO − mean capillary PCO. (Partial pressure of CO in the capillary blood is denoted by the symbol PćCO). The theoretical formula for the diffusion capacity of the lung for CO (DLCO) is as follows:

Normal Values
Normal DLCO values in healthy individuals vary considerably, depending on age, body size, body position, and other factors, as explained in subsequent sections. The average DLCO in a healthy young man under resting conditions is 21 mL/min/mm Hg.4 The diffusion capacity of the lung for oxygen (DLO2) is obtained by multiplying DLCO by 1.23. A mean DLCO of 21 mL/min/mm Hg yields a normal DLO2 of about 26 mL/min/mm Hg. It may seem odd that DLO2 is greater than DLCO, considering the much greater affinity of hemoglobin for CO than O2. This peculiarity is explained by the fact that O2 is more soluble than CO in the alveolar-capillary membrane and the plasma, and therefore it diffuses more rapidly. The fact that hemoglobin has 210 times greater affinity for CO than for O2 simply means that at a given partial pressure (PCO or PO2), the hemoglobin carries more CO than O2. This fact is unrelated to the O2 and CO diffusion rate across the alveolar-capillary membrane, which is determined by molecular weight and solubility coefficients.
Factors Affecting Measured Carbon Monoxide Diffusion in the Lung
Body Size
DLCO increases with body size. Larger lungs have a greater absolute DLCO than smaller lungs because they have a greater gas-exchange surface area.
Age
DLCO increases as the lungs develop from infancy to adulthood. In people older than 20 years, DLCO decreases gradually as the lung ages and structural changes reduce the functional gas-exchange surface area. A ten-year age increase is associated with a decline of about 1.32 mL/min/mm Hg in DLCO.
Lung Volume
DLCO is significantly greater at high lung volumes than low volumes. This may reflect increased pulmonary blood volume at high lung volumes (the greater subatmospheric intrapleural pressure near total lung capacity [TLC] draws more blood into the pulmonary circulation) or an increased functional surface area for diffusion. Dividing DLCO by the lung volume at which it was measured (DLCO per unit of lung volume [DL/VL]) eliminates the effect of volume on DLCO. For example, the abnormally large lung volume of a patient with emphysema may have a total DLCO in the normal range, obscuring the true decrease in surface area per unit of lung volume. The calculated DL/VL in this instance yields an abnormally low value, revealing the loss of diffusion capacity.
Exercise
DLCO increases markedly with exercise as higher blood pressure recruits previously collapsed capillaries; this greatly increases the surface area for diffusion. Increased cardiac output means capillary transit time decreases, and more O2 can be transferred into the blood each minute. During exercise (in contrast to rest), blood stays in the capillary only long enough to reach equilibrium with alveolar gas. Increased cardiac output dilates capillaries, exposing more hemoglobin to alveolar gas and allowing greater O2 uptake per minute.
Body Position
DLCO is 15% to 20% greater in supine than in standing positions.1 This increase probably occurs because gravity decreases pulmonary blood volume in the standing position—especially in the lung apices.
Alveolar Partial Pressure of Oxygen and Partial Pressure of Carbon Dioxide
Increased PAO2 is associated with decreased DLCO because in the blood O2 competes with CO for binding sites on the hemoglobin molecule. In other words, the rate of CO uptake by hemoglobin is hindered by increased arterial PO2 values but is enhanced by decreased arterial PO2 values. People with severe hypoxia have higher DLCO values because of this phenomenon and because hypoxia increases cardiac output, pulmonary blood flow, and capillary recruitment and distention. This phenomenon also explains why an abnormally elevated PACO2 increases DLCO; an elevated PACO2 means hypoventilation is present, which means PAO2 must be low.
Alveolar Partial Pressure of Carbon Monoxide
Heavy smokers already have CO in their mixed venous blood. The presence of CO in mixed venous blood lowers the diffusion pressure gradient across the alveolar-capillary membrane and slows the diffusion rate.
Hemoglobin Concentration
The blood of patients with anemia has abnormally low hemoglobin content and low CO-carrying capacity. Less CO can be taken up per minute during the DLCO test. The opposite is true for polycythemia (increased RBCs and hemoglobin concentration).
Pulmonary Diseases
Box 7.1 lists conditions that decrease diffusion capacity. DLCO is useful in differentiating emphysema from other obstructive diseases that are not associated with destroyed alveolar architecture, such as chronic bronchitis and asthma. Asthma is often associated with an increased DLCO. An explanation may be that high inspiratory resistance created by narrowed airways increases negative intrathoracic pressure during maximal inspiration of the test gas, which increases the gas diffusion pressure gradient and the blood flow in lung apices.5
Clinical Use of Carbon Monoxide Diffusion Capacity Test Results
The DLCO test assesses the extent to which the alveolar-capillary membrane is a barrier to gas diffusion. Because O2 equilibrium normally occurs before blood traverses one-third of the pulmonary capillary distance, a 50% reduction in DLCO can exist without affecting end-capillary and arterial PO2. In this sense, the DLCO test is more sensitive to potential O2 transfer problems (i.e., as disease progresses, DLCO becomes abnormal before PaO2) than the PaO2 obtained in arterial blood gas analysis. Impairment of diffusion across the alveolar-capillary membrane is only one mechanism that can cause arterial hypoxemia. Many factors decrease PaO2 without affecting the diffusion path length or membrane surface area, such as ventilation–blood flow mismatches. The DLCO test can help clarify the mechanism of arterial hypoxemia; if DLCO is normal, diffusion impairment cannot be a contributing factor.
Points to Remember
- • Gas always diffuses from an area of high partial pressure to an area of low partial pressure.
- • Diffusion ceases when partial pressure equilibrium across the alveolar-capillary membrane occurs; this normally occurs for O2 long before blood flows through the length of the capillary but never occurs for CO diffusion.
- • Gas transfer from alveolus to blood is diffusion limited if blood is able to take up the gas faster than the gas can diffuse through the alveolar-capillary membrane.
- • Gas transfer from alveolus to blood is perfusion limited if its pressures in the alveolus and blood equalize so quickly that diffusion ceases before blood leaves the capillary.
- • Diffusion impairments rarely cause hypoxemia at rest because gas equilibrium between alveolus and blood virtually always occurs; exercise unmasks the impairment because blood flows so fast that there is not enough time for equilibrium to occur.
- • Diffusion rate is affected by membrane functional surface area, physical gas characteristics, partial pressure gradient for diffusion, and membrane thickness (diffusion path length).
- • Light and soluble gases diffuse faster than heavy insoluble gases; because of its much greater solubility, CO2 diffuses across the lung 20 times faster than O2.
- • Diseases affect diffusion capacity chiefly by increasing the membrane thickness or decreasing the functional surface area.
- • DLCO can be significantly reduced before any decrease in arterial PO2 occurs.








