Troubleshooting in Laparoscopic Surgery
Historically, in large series, the overall incidence of laparoscopic complications in urology has been in the range of 4%. Mortality has been distinctly unusual, with a rate of 0.03% to 0.08% (Mintz 1977; Winfield et al, 1991; Chapron et al, 1998; Fahlenkamp et al, 1999; Harkki-Siren et al, 1999). However, because of the increasing complexity of laparoscopic urologic procedures (e.g., nephrectomy, prostatectomy) and the continued decrease in simpler laparoscopic procedures (e.g., varicocelectomy, bladder neck suspension, pelvic lymph node dissection), laparoscopic complications in urology appear to be on the rise. In one update from Johns Hopkins University regarding transperitoneal laparoscopic retroperitoneal procedures (e.g., renal surgery, retroperitoneal lymph node dissection), Parsons and colleagues (2004) report a 0.2% mortality rate and a 12% overall complication rate. Of concern, the most often cited injury was vascular, occurring in 2.8% of patients followed by bowel injury in 1.1% of patients (Table 9–5). The authors attribute this increased incidence to the larger number of surgeons now doing laparoscopy and moving through the learning curve, the addition of several new procedures that are substantially more challenging than past procedures (e.g., partial nephrectomy, retroperitoneal lymph node dissection), and the aging and higher risk of our population in general (Parsons et al, 2004). Of interest, the complications for pelvic laparoscopy although higher than for retroperitoneal laparoscopy are of a more minor nature. Indeed, Vallancien and colleagues (2002) noted a 22.6% overall complication rate among 1311 procedures, of which 84% were pelvic; however, there were no deaths and the most often injured organ was bowel at 1.2%; vascular injuries were cited in only 0.5% of their procedures (see Table 9–5). Another recent meta-analysis of radical prostatectomy techniques compared open, laparoscopic, and robotic complications. The overall complication rates for the different approaches were 10.3%, 15.6%, and 6.6%, respectively (Berryhill et al, 2008). Another recent series of 1500 robotic radical prostatectomy cases reported a postoperative complication rate of 4.3%. Intraoperatively, the complication rate was only 0.13% (two rectal injuries) (Patel et al, 2008). Possible reasons for the difference in the complication rates between laparoscopic and robotic prostatectomy might be that the robotic platform expedites the learning curve for the procedure, provides a more comfortable working environment for the surgeon, and allows the surgeon to work in an intuitive 3D environment with 6 degrees of freedom instrumentation.
Table 9–5 Major Complications of Transperitoneal Abdominal Surgery
| TOTAL PROCEDURES | 894* (100% ABDOMINAL) |
1311† (84% PELVIC) |
|---|---|---|
| Overall complications | 13.2% | 22.6% |
| Intraoperative/postoperative | 5.7%/7.5% | 3.6%/19% |
| Deaths | 0.2% | 0% |
| Vascular injury | 2.8% | 0.5% |
| Bowel injury | 1.1% | 1.2% |
| Adjacent organ injury | 1.1% | 0.8% |
| Conversion rate | 1.7% | 1.7% |
* Data from Parsons JK, Varkarakis I, Rha KH, et al. Complications of abdominal urologic laparoscopy: longitudinal five-year analysis. Urology 2004;63:27–32.
† Data from Vallancien G, Cathelineau X, Baumert H, et al. Complications of transperitoneal laparoscopic surgery in urology: review of 1,311 procedures at a single center. J Urol 2002;168:23–6.
Minimizing the Incidence of Complications during the Laparoscopic Learning Curve
Early in one’s experience with laparoscopic and robotic surgery it is wise to apply this minimally invasive approach to low-risk surgical candidates of normal body habitus. In addition, it is advisable and recommended by many laparoscopic organizations, as well as by hospital credentialing boards, that the neophyte minimally invasive surgeon seek training in three arenas: (1) in-depth instructional courses, including didactic, “live-case” transmissions and “hands-on” laboratory sessions; (2) preceptor training in which the surgeon-in-training views more than five or more procedures being done by an already skillful laparoscopic surgeon; and (3) a mentoring experience, during which a trained laparoscopic surgeon oversees the initial procedures performed by the surgeon-in-training (Society of American Gastrointestinal and Endoscopic Surgeons, 2003). Further training can be obtained through self-teaching using videotapes and a pelvic trainer. The latter is extremely helpful for developing one’s sense of laparoscopic proprioception and for becoming facile with laparoscopic suturing and knot tying. Data have clearly shown benefits for individuals who have taken the time to practice their laparoscopic skills using a pelvic trainer in all areas of laparoscopy (cutting, clipping, and suturing) compared with individuals who had no such training (Derossis et al, 1998). Similarly, participation in a 1-week mini-residency has been found to increase the likelihood that participants would perform more complex laparoscopic procedures (81% of participants) (Corica et al, 2006). The use of virtual reality simulators is under evaluation but further validation testing is required before these complex and often expensive trainers can be recommended strongly. A detailed discussion of laparoscopic and robotic practice and training is presented later in this chapter.
General Procedural Complications
Aside from training in the basic psychomotor skills, neophyte minimally invasive surgeons must be educated with regard to prevention, recognition, and appropriate treatment of complications. Accordingly the following section covers the myriad complications that can occur with any laparoscopic/robotic procedure. Recognition, resolution, and prevention of these various problems are discussed.
Malfunction of Equipment
A successful outcome of any laparoscopic procedure depends not only on the psychomotor technical skills of the surgeon but also on a proper working knowledge of all the equipment involved in performing these procedures. To ensure undisturbed functioning of all technology, the surgeon must be supported by well-trained staff who are capable not only of quickly recognizing any equipment malfunction but also of providing an immediate, adequate response to correct problems. In this regard, the Society of American Gastrointestinal Endoscopic Surgeons has issued a troubleshooting guide for video and electronic failure. Newer laparoscopy systems have become more simplified in some ways offering automatic settings, touch screen control, and voice command; however, these new systems can be more taxing as well because they offer a host of optional settings and capabilities. For integrated operating room systems offered by most major equipment manufacturers the surgeon and operating room staff need to receive in-depth training on the system’s operation, capabilities, and limitations. In this way, equipment failure will be minimized. Additionally, the contact information for the equipment vendor’s trouble shooting experts should be readily available.
With regard to the da Vinci Robotic System, equipment malfunction is rare. In a review of 11 institutions with a total of 8240 cases reviewed, the overall incidence of malfunction was 0.4%. Of the 34 cases with malfunction, 24 cases were canceled before the procedure, 2 cases were converted to laparoscopic, and 8 were converted to open surgery (Lavery et al, 2008).
Complications Related to Obtaining the Pneumoperitoneum
Complications Associated with Closed Access (Veress Needle Placement)
Preperitoneal Placement
Preperitoneal placement of the Veress needle may preclude successful trocar placement. If not recognized early, 1 to 2 L of CO2 may be instilled; indeed, once this much CO2 has been insufflated into the preperitoneal space, many signs indicative of correct intraperitoneal insufflation may be present (e.g., distention, tympanic sound on percussion) thereby misleading the surgeon until the first trocar is placed. The first sign of preperitoneal insufflation is that there may be a steep rise in pressure with only 500 mL of CO2; plus, if more CO2 is instilled, unequal distention of the abdomen occurs. If this early sign is missed, then the laparoscope reveals only fat after trocar placement; the intraperitoneal viscera are not seen.
The next step is to evacuate the CO2 through the sidearm of the trocar and proceed with an open insertion technique. The initial incision can be widened, and the peritoneal surface can be grasped with a pair of Allis clamps and incised. A Hasson cannula is then secured in place, as previously described, and the peritoneal cavity is insufflated.
Several steps can be taken to avoid this complication. First, if the Veress needle is preperitoneal on initial insufflation, pressures are usually higher than the maximal initial allowable pressure of 10 mm. Second, if the Veress needle is preperitoneal, it cannot be easily advanced 1 cm deeper without resistance. If one has truly entered the peritoneal cavity properly, the Veress needle should be able to be moved 0.5 to 1 cm deeper without meeting any resistance (the “advancement test”).
Vascular Injuries
During initial placement of the Veress needle at the umbilicus, minor or major intra-abdominal blood vessels may be punctured by the 14-gauge needle. The first sign of intravascular entry is blood appearing in the hub of the needle. Aspiration results in additional blood filling the syringe. As long as the needle has not been manipulated, it can usually be withdrawn without excessive bleeding. An alternative site for Veress needle placement or open cannula insertion should be used at this point.
To prevent this problem it is important when using an umbilical approach to direct the Veress needle toward the hollow of the pelvis. Passing the Veress needle through a 12-mm incision, bluntly spreading the subcutaneous fat, and grasping and stabilizing the anterior fascia with a pair of Allis clamps may help prevent this problem when using an umbilical access. These maneuvers become especially important in children, who have less space between intra-abdominal structures and the abdominal wall. Lastly, for a case in which this problem occurs, it is important that the path of the initial Veress needle passage be traced on proper entry into the abdomen. The prior site of the Veress needle passage is carefully inspected at a pressure of 5 mm Hg; any site of bleeding can be expeditiously treated by the application of a surgical hemostatic agent such as thrombin-impregnated gelatin matrix or fibrin glue. Gentle pressure can be applied to the bleeding vessel. Likewise, any hemodynamic instability associated with loss of “working space” within the abdomen during the procedure should alert the surgeon to the possibility of an expanding “unseen” retroperitoneal hematoma, a distinctly unusual occurrence.
Prevention of vascular complications can be further achieved in one of two manners. First, using a nonumbilical site for Veress needle passage (i.e., just superior and medial to the iliac crest or subcostal in the midclavicular line) places no major vessels in danger. Second, the use of only blunt trocars decreases the chance of injury of the epigastric vessels by fivefold (reduced incidence from 0.83% to 0.16%) (Hashizume and Sugimachi, 1997; Thomas et al, 2003).
Visceral Injuries
During Veress needle placement, intra-abdominal organs may be punctured. The initial signs of this complication consist of aspiration of blood, urine, or bowel contents through the Veress needle or, in the case of a solid organ, high pressures on initial insufflation.
Management consists of simply removing the Veress needle. The Veress needle may then be reintroduced at a different site, or an open cannula placement can be used through a separate incision site. On entry into the abdomen, any bleeding site on the liver or spleen can be treated with an argon beam coagulator or the application of a surgical hemostatic agent (e.g., thrombin containing gelatin matrix or fibrin glue) (see earlier).
Bowel or bladder entry of the Veress needle needs no further treatment other than needle withdrawal. These problems can be prevented by placing a nasogastric tube and a transurethral indwelling bladder catheter to decompress the stomach and bladder, respectively, before Veress needle passage. Stabilization of the abdominal wall fascia with towel clips or Allis clamps at the time of Veress needle puncture may help in stabilizing the fascia. One should not lift up on the fascia because this will only increase the space between the fascia and the peritoneum while not changing the intra-abdominal space. Likewise, insufflation should never be initiated unless all of the signs for proper peritoneal entry (negative aspiration, easy irrigation of saline, negative aspiration of saline, positive drop test, and normal advancement test) have been confirmed.
Complications Related to Insufflation and Pneumoperitoneum
Bowel Insufflation
If entry into the bowel is not recognized at the time of irrigation and aspiration through the Veress needle, then the surgeon may well insufflate the small or large bowel. The first sign of this problem is asymmetrical abdominal distention. Associated signs may be passage of flatus and insufflation of only a small amount of CO2 (<2 L) before high pressures are reached.
If this complication is suspected, then the insufflation line should be disconnected; the outflow of gas will immediately confirm bowel entry. The needle can be withdrawn, and open access cannula placement should be done at a different abdominal site. Prevention of this problem is ensured if one properly performs the aspiration/irrigation/aspiration tests recommended for safe Veress needle placement and if one avoids sites of prior surgery. Similarly, initial open cannula insertion may decrease the chances of this complication.
Gas Embolism
Carbon dioxide gas has favorable solubility in blood, as opposed to air, helium, or nitrous oxide (LD50 = 1750 mL); however, use of CO2 may still result in a gas embolus. The most common cause of CO2 embolism is puncture of a blood vessel or organ with the Veress needle, followed by insufflation; this can occur only when the surgeon has ignored the previously described tests for proper entry into the peritoneal cavity. The first sign of intravascular insufflation is acute cardiovascular collapse. Other signs include dysrhythmias, tachycardia, cyanosis, and pulmonary edema. The diagnosis is usually made by the anesthesiologist based on an abrupt increase of end-tidal CO2 accompanied by a sudden decline in oxygen saturation and then a marked decrease in end-tidal CO2 (Loris, 1994). Sometimes, a “millwheel” precordial murmur can be auscultated (Keith et al, 1974). In addition, the anesthesiologist may notice foaming of a drawn blood sample owing to the presence of insufflated CO2.
The treatment is immediate cessation of insufflation and prompt desufflation of the peritoneal cavity. The patient, if at all possible, is turned into a left lateral decubitus, head-down position (i.e., right side up) in hopes of minimizing right ventricular outflow problems and forcing the air embolus to rise into the apex of the right ventricle. The patient is hyperventilated with 100% oxygen. Advancement of a central venous line into the right side of the heart with subsequent attempts to aspirate gas may rarely be helpful. The use of hyperbaric oxygen and cardiopulmonary bypass have also been reported (McGrath et al, 1989; Diakun, 1991; Abdel-Meguid and Gomella, 1996).
This devastating complication can be precluded by meticulous attention to Veress needle and initial trocar placement and performance of each of the recommended tests for intraperitoneal entry. Insufflation should never be initiated if the surgeon has even the slightest doubt about correct positioning of the Veress needle; instead, the surgeon should withdraw the Veress needle and pass it at an alternate site or immediately proceed with open cannula access.
Barotrauma
Prolonged elevated pressures (>15 mm Hg) may result in barotrauma (McGrath et al, 1989; Diakun, 1991; Abdel-Meguid and Gomella, 1996). Prolonged high pressures may be caused by insufficient and infrequent monitoring of CO2 pressure, malfunction of the insufflator, or additional pressures produced by auxiliary devices (e.g., argon beam coagulator, CO2-cooled laser). Furthermore, barotrauma may be caused by ventilation techniques using positive end-expiratory pressure resulting in rupture of a pulmonary bleb or bulla.
The initial sign of barotrauma may be hypotension owing to decreased cardiac output secondary to an acute drop in venous return caused by compression of the vena cava. Also, a pneumothorax or pneumomediastinum may develop because of high ventilation pressures. Increased intra-abdominal pressures may exacerbate a hiatal hernia as well.
The anesthesiologist usually alerts the surgeon to the problem of excessive intra-abdominal pressure; it usually presents as an increase in ventilation pressures. The surgeon should desufflate the abdomen and, once the hemodynamic changes have been reversed, reinitiate the pneumoperitoneum at 10 mm Hg. Any malfunctioning insufflator should be replaced. Also, if one is using an argon beam coagulator or a CO2-cooled laser device, the sidearm on one port should be left open to allow high-pressure excess gas to readily escape while the device is being activated.
These problems are avoided by the alert, meticulous surgeon. The insufflator should always be checked before initiating the procedure; at maximum inflow settings, gas should flow freely at less than 2 mm Hg pressure down the opened, unconnected insufflation line, and when the line is purposely kinked by the surgeon, the insufflator-recorded pressure should rise rapidly to its preset limit (usually 15 mm Hg), the inflow of CO2 should cease, and the high pressure alarm should sound. Again, troubleshooting the insufflator should be part of the routine prelaparoscopy check in every case.
Subcutaneous Emphysema
This problem develops owing to improper placement of the Veress needle or, more commonly, to leakage of CO2 around ports. The latter situation occurs when port site incisions are too large, when the procedure is particularly lengthy, or when high intra-abdominal pressures are used. The pathognomonic sign is crepitus over the abdomen and thorax; in male patients, a pneumoscrotum may also develop.
If the problem is due to improper placement of the Veress needle, then withdrawal of the Veress needle and open insertion are recommended. If the problem develops intraoperatively, the surgeon should check for gas leakage around a port site. If this is found, the surgeon can either place a purse-string suture around the port or, preferably, change the trocar to a larger size or switch to the balloon-based Hasson type cannula, which creates a tight seal between the intra-abdominal balloon and its outer soft foam cuff. Also, the surgeon should consider reducing the insufflation pressure.
This complication is eminently avoidable if the surgeon adheres to all the diagnostic tests for proper Veress needle placement and if the port site incisions are carefully tapered to the size of the port to be placed. In this regard, it is important to place each port so that it is pointing toward the surgical field, to avoid the continued forceful redirection of the port during the procedure that results in widening of the tissue tract around the port and subsequent escape of CO2 into the surrounding subcutaneous tissues. In addition, the cannulas must be secured so they do not pull back into the abdominal wall tissues; to this end, the surgeon can use a screw-type cannula or may elect to simply use a suture to fix the sidearm of the port to the skin, thereby precluding its retraction beyond a certain point. If the former solution is chosen, the surgeon must be careful to never use a plastic retaining collar with a metal trocar; this situation greatly increases the chance of inadvertent monopolar electrosurgical injury owing to stray current along the exposed shaft of the now partially insulated metal trocar. Also, the insufflator must be tested before each procedure, as part of a preincisional routine checklist, to make sure it is functioning properly, so that when high pneumoperitoneum pressures develop, the inflow of CO2 automatically ceases.
Several studies have demonstrated that the incidence of subcutaneous emphysema is somewhat higher during retroperitoneal laparoscopy than during transperitoneal laparoscopy, albeit without any clinically significant sequelae (Wolf et al, 1995; Zhao et al, 2008). However, the risk of surgical emphysema and other CO2-related sequelae during retroperitoneoscopic and extraperitoneoscopic surgery can be effectively minimized by adopting a variety of practical measures (Gill, 1998; Ng et al, 1999). Specifically working at a lower pressure is helpful (i.e., 12 mm Hg). In one study, in the initial 20 cases with an insufflation pressure of 15 mm Hg, subcutaneous emphysema was noted in three patients (15%); however, in the subsequent 180 cases the insufflation pressure was maintained below 12 mm Hg and subcutaneous emphysema was noted in only two patients (1.1%) (Rassweiler et al, 1998a). Also, use of the balloon-based Hasson cannula to seal the initial entry site is important. Invariably, the problem resolves itself during the initial 2 to 3 postoperative days.
Pneumomediastinum, Pneumothorax, and Pneumopericardium
Gas leaking along major blood vessels through congenital defects or secondary enlargement of openings in the diaphragm may lead to pneumomediastinum, pneumopericardium, or pneumothorax (Kalhan et al, 1990; Pascual et al, 1990; See et al, 1993; Abreu et al, 2004; Zhao et al, 2008). Although a pneumomediastinum is usually not associated with specific clinical symptoms, a pneumopericardium may result in impaired cardiac function. The incidence of the latter complication is estimated to be 0.8% (Abreu et al, 2004). The diagnosis is rarely made during the procedure unless cardiac impairment occurs. Usually, the diagnosis is made on a chest radiograph taken in the recovery room. However, if there is sudden cardiac decompensation during a procedure, the same maneuvers are undertaken as described for treatment of a suspected gas embolism—interruption of the procedure and desufflation of the abdomen. If there is a strong suspicion of pericardial tamponade, pericardiocentesis is indicated.
A pneumothorax may be associated with pneumomediastinum, barotrauma, or with direct puncture of the pleural space with a trocar (Doctor and Hussain, 1973; Kalhan et al, 1990; Pascual et al, 1990). The incidence of this complication in a recent series was 1.6% to 4.0% (Abreu et al, 2004; Zhao et al, 2008). Like subcutaneous emphysema, the incidence of pneumothorax is more common in retroperitoneal procedures (Zhao et al, 2008). The earliest signs of this problem may be the development of subcutaneous emphysema, especially in the neck and chest area. More ominous signs, such as hypotension and decreased breath sounds with an increase in ventilatory pressure, are indicative of a tension pneumothorax. Although a chest radiograph will confirm the diagnosis, the development of pulmonary collapse with loss of breath sounds on one side mandates immediate decompression of the chest by passage of a 16-gauge needle into the second or third intercostal space in the midclavicular line followed by tube thoracostomy if a tension pneumothorax is suspected (See et al, 1993).
Of note, the occurrence of pulmonary complications appears to be greater with a retroperitoneal approach. Indeed, in one large series, 90% of these complications were associated with retroperitoneoscopy, whereas only 10% of these problems occurred in patients undergoing transperitoneal laparoscopy (Abreu et al, 2004).
Prevention of these problems is similar to the means to avoid subcutaneous emphysema: keep the intra-abdominal pressure at 15 mm Hg or less, make sure all port site incisions are tight around the laparoscopic cannulas, and make sure all cannulas are well seated in the peritoneal cavity. In addition, all trocars must remain below the 12th rib. While dissecting in the upper quadrants of the abdomen, especially during laparoscopic ablative renal surgery, the surgeon should be aware of the anatomic relationships of the kidneys, adrenal glands, and great vessels to the diaphragm to avoid direct injury.
Complications during Open Access (Hasson Technique)
Potential problems associated with open access are similar to, albeit less frequent than, problems associated with a closed Veress needle pneumoperitoneum. The biggest major risk in this regard is injury to underlying viscera while traversing the peritoneum. In a densely scarred abdomen, the bowel may be adherent to the underside of the abdominal wall and hence may still be injured. If a bowel injury is recognized early, it can often be repaired through the same incision that was made for insertion of the Hasson cannula. Although vascular injury with this approach is distinctly rare, the surgeon must realize that even with open access this devastating complication can occur (Hanney et al, 1999).
The only minor risk in using the open access technique is failure of the surgeon to obtain secure transfascial retaining sutures on either side of the cannula. If this is not done, the bulb of the Hasson cannula will not be seated tightly into the incision, thereby resulting in significant leakage of gas and subsequent subcutaneous emphysema. One way to prevent this problem is to also place a purse-string suture in the fascia to better secure it to the hub struts of the Hasson cannula. In addition, some Hasson type cannulas have built-in retention systems. The Bluntport (US Surgical, Norwalk, CT) has a retention balloon that can be inflated in the peritoneal cavity and then drawn up tightly against the peritoneal side of the abdominal wall; an outer foam sealing ring can then be advanced down the extra-abdominal shaft of the cannula, thereby sandwiching the abdominal wall between the inflated balloon and the foam seal, effectively precluding any leakage of gas during the case. This is especially effective when doing retroperitoneoscopic procedures. Another self-sealing trocar, the Anchorport (SurgiQuest, Orange, CT) has an elastic self-sealing trocar shaft that automatically forms to the patient’s body wall when the inner cannula is removed, creating a tight seal.
Complications Related to Initial “Blind” Placement of the First Trocar after Obtaining a Veress Needle Pneumoperitoneum
With the advent of nonbladed trocars (several of which also have clear tips for direct visualization of individual abdominal wall layers during port placement), the likelihood of catastrophic injuries to vital structures has been markedly reduced (Thomas et al, 2003). Accordingly, the authors no longer consider the use of bladed trocars to be clinically appropriate and these trocars have been removed from our operating rooms.
Injury to Gastrointestinal Organs
Perforation of the small or large intestine during passage of the primary port is the most common cause of trocar-induced injury of gastrointestinal organs. Other organs (e.g., stomach) are affected much less frequently. Given the lateral positioning of the spleen and liver, injury of these organs with the passage of the primary trocar is distinctly unusual. The first sign that one has entered the bowel depends on whether the injury is through one wall or both walls (“through-and-through” injury) of the bowel. In the former instance, as soon as the laparoscope is introduced the surgeon sees the mucosal folds of the interior of the bowel. However, with a through-and-through injury the diagnosis is not made until the first secondary trocar is passed; at that time the surgeon should routinely pass the laparoscope through the secondary port to inspect the puncture site of the initial port. The trocar will be seen passing completely through both walls of the bowel. If the surgeon fails to perform this maneuver routinely this injury will not be noted until the end of the case when the trocars are being removed, thereby resulting in a broader injury and a prolonged time of intraperitoneal contamination. A missed bowel injury of this nature leads to peritonitis, when diagnosed intraoperatively, and possible death, when discovered only in the postoperative period.
In the case of a one-wall injury of the bowel, the surgeon can elect to leave the trocar in place and pass a second trocar in another location using an open access technique. On inspection of the abdomen the site of injury to the bowel will be immediately apparent because the initial trocar will still be residing in the bowel. At this time, the surgeon may elect to open and repair the bowel or, if skilled in laparoscopy, may place two more ports and proceed to close the bowel using laparoscopic suturing or stapling techniques. An intraoperative consultation with a general surgeon should be obtained regardless of whether the urologist performs the repair; from a medicolegal and quality of care standpoint, involvement of the general surgeon at the time of the acute event facilitates subsequent care should further complications arise while ensuring the best possible repair of the injury at the time of the acute event.
When the injury to the bowel is a through-and-through injury, the safest path is to open and proceed with repair; alternatively, if particularly skilled, one may consider laparoscopic repair. In either case, the abdomen should be irrigated with 4 to 5 L of saline containing an antibiotic solution and the patient must be placed on broad-spectrum, triple-drug antibiotic coverage.
In an effort to preclude bowel injury, a mechanical and antibiotic bowel preparation is recommended in patients with a history of extensive prior abdominal surgery. For those patients with a low risk of bowel injury, a simple mechanical preparation to “empty” the bowels should suffice. Commonly this is accomplished with magnesium citrate the day before the procedure; in addition, the patient can be placed on a clear liquid diet for 1 to 2 days preoperatively.
Perforation of the stomach is distinctly rare; however, to best preclude this problem patients should refrain from oral intake for 12 hours before surgery. To decompress the stomach, a nasogastric or orogastric tube should be placed before puncture of the abdomen with the Veress needle. The management of this complication is the same as for injury to the bowel with primary closure and general surgery consultation. In all of these cases a nasogastric tube is left indwelling until the output to gravity is minimal.
Injury to Intra-Abdominal Vessels
Major vascular injury is a rare but serious complication, occurring in 0.11% to 2% of cases (Hanney et al, 1995; Geers and Holden, 1996; Usal et al, 1998; Lin and Grow, 1999; Vallancien et al. 2002; Parsons et al, 2004) (Fig. 9–27). It is far more common in procedures related to the retroperitoneum, as opposed to pelvic laparoscopy. The aorta and common iliac arteries are most frequently involved. The inferior vena cava is less affected because of its lateral location in relation to the aorta; likewise, the common iliac vein is rarely involved given its posterior position in relation to the common iliac artery. Rarely, in a patient with adhesions or prior surgery, intestinal mesenteric vessels servicing a “fixed” loop of bowel may be injured. In addition, the epigastric vessels are at risk for injury during trocar placement.
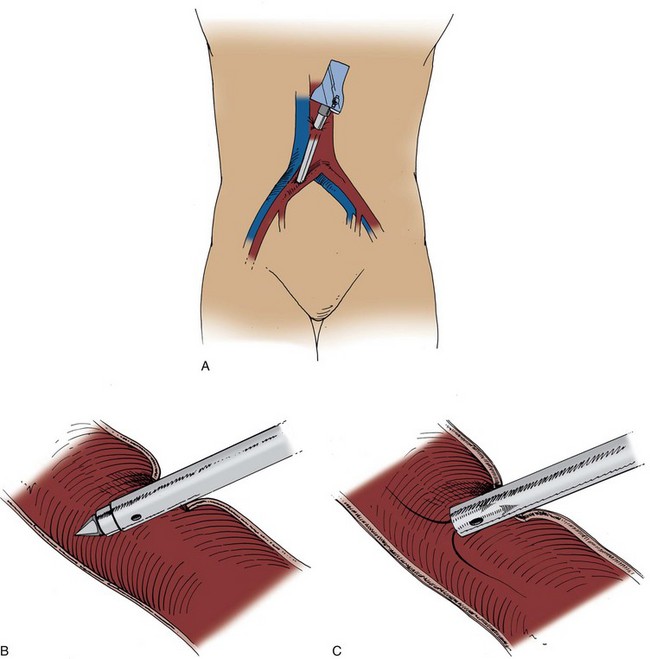
Figure 9–27 A and B, The right common iliac artery has been punctured by a trocar. C, As soon as the obturator is removed, blood fills the cannula.
(From Clayman RV, McDougall EM, editors. Laparoscopic urology. St. Louis: Quality Medical Publishing; 1993.)
The first sign of a major vascular complication is the onset of sudden hypotension and associated tachycardia. If the trocar has not been moved, then, as the obturator is withdrawn, the diagnosis is made immediately based on whether there is a pulsatile (arterial) or nonpulsatile (venous) profuse return of blood from the trocar sheath. If the trocar has been displaced from the injured vessel, then, depending on the vessel injured, the surgeon will see blood rapidly accumulating in the abdominal cavity, a mesenteric hematoma, blood dripping from the trocar entry site, or, rarely, blood that preferentially accumulates retroperitoneally, in which case, the space within the peritoneal cavity will appear to be markedly reduced and actively decreasing because of the expanding retroperitoneal hematoma.
The response to injury to a major arterial structure must be rapid. A vascular or trauma surgeon should be called to the room. If blood is coming through the trocar, then the trocar should be closed and left in place. An emergency laparotomy is performed, and the trocar is followed to its point of entry into the vessel. Controlling sutures can be placed on either side of the trocar or a Satinsky clamp can be placed to isolate the area of injury, so that, as the trocar is withdrawn, the wound can be rapidly controlled. Alternatively, the procedure can be converted to a hand-assisted approach and the surgeon can then use the intra-abdominal hand to control the bleeding vessel.
If the injury is discovered at the time of passage of the laparoscope (i.e., the trocar is no longer residing in the vessel), then the sheath and laparoscope can be swung up to the underside of the abdominal wall and an immediate cutdown can be done on top of the laparoscope and sheath, thereby providing for a rapid and safe laparotomy. The site of injury must be rapidly located and controlled. Again, the aid of a vascular or trauma surgeon in this case is quite helpful.
The best way to handle this complication is to avoid it completely. In this regard, knowledge of the exact location and possible anatomic variations of major intra-abdominal blood vessels is mandatory. The CT scan should be reviewed before passage of any trocars to look for vena caval or other abnormalities of the great vessels. Because of limited intraperitoneal space, special care must be given to trocar placement in children and very thin adults. Strict adherence to laparoscopic guidelines, such as ensuring that all the safety signs of passage of a Veress needle are present before proceeding with trocar passage, obtaining an adequate pneumoperitoneum before trocar passage (intra-abdominal pressure may be raised to 25 mm Hg temporarily for placement of the primary trocar), passing the initial trocar under direct endoscopic control (i.e., clear plastic port), and avoiding initial trocar passage through an abdominal scar, are important in helping to prevent this problem. Similarly, avoidance of the epigastric vessels is ensured if trocars are either placed in the midline or at least 6 cm lateral to the midline.
With regard to hemorrhage, when the situation is recognized but controlled, which occurs more commonly with venal caval than arterial injuries, the surgeon has the ability to “prepare” for subsequent conversion. In that regard, a second suction unit should be obtained, the vascular or trauma surgeon should be in attendance, and the anesthesiologist has time to make sure the patient is well hydrated and that blood is in the room for transfusion. In addition, before conversion, the use of a 4 × 8-inch laparoscopic pad, which can be introduced through a 12-mm port may be helpful (personal communication: I. Gill); however, if the bleeding is profuse then the surgeon should have available ample amounts of large gauze rolls (e.g., Kerlix) for packing the area as soon as the abdomen is opened.
Furthermore, it is helpful to consider having a “hemorrhage” tray available in the operating room at all times (Table 9–6). This laparoscopic tray should contain a Satinsky clamp, a 10-mm suction tip for large clot evacuation, an Endo Stitch device with 4-0 Vicryl suture, a Lapra-Ty clip applier and a rack of Lapra-Ty clips (6 clips per rack), two laparoscopic needle holders, and 4-0 vascular suture. With this tray available, some injuries to major venous structures (e.g., inferior vena cava) can be successfully resolved laparoscopically.
Table 9–6 Contents of Hemorrhage Tray for Laparoscopic Surgery
Injury to the Urinary Tract
Urinary tract injuries during laparoscopy are most commonly associated with trocar passage. The incidence of this problem varies widely as reported in the gynecologic literature: 0.02% to 8.3% range (Ostrzenski and Ostrzenska, 1998; Lin and Grow, 1999; Soong et al, 2007). Usually, these injuries occur to the bladder at the time of initial trocar placement. Chances of this problem occurring have been greatly reduced by the introduction of blunt trocars.
Trocar injuries of the urinary tract have reportedly affected only the bladder. The initial sign of this problem is pneumaturia or macroscopic hematuria. The diagnosis is confirmed by retrograde intravesical instillation of indigo carmine diluted with saline; this allows the surgeon to rapidly identify the cystotomy site. The injury can be repaired laparoscopically, either with laparoscopic suturing techniques or by use of the laparoscopic tissue stapler; extensive defects may require open surgical repair (Ostrzenski and Ostrzenska, 1998). These injuries should always be closed and not left to heal “on their own” with prolonged Foley catheter drainage.
Prevention of this problem is simple. Preoperative placement of a urethral catheter to drain the bladder is recommended for all major laparoscopic urologic cases. Not only does it largely preclude bladder injury, but it also provides the necessary means for monitoring urine output during major laparoscopic urologic procedures.
Complications Related to Placement of Secondary Trocars
Bleeding at the Cannula Site
Blood dripping from the port entry site and onto the underlying abdominal viscera is the first sign of an injured abdominal wall vessel. The exact site of hemorrhage is determined by cantilevering the trocar into each of the four quadrants and noting which positioning of the trocar tamponades the bleeding.
Definitive therapy for this problem can be undertaken in one of three ways. The simplest method, albeit the most costly, is the insertion of curved electrosurgical scissors or forceps through another port; this instrument can be articulated up into the port site to electrocoagulate the bleeding site.
The least expensive method is to suture the area of hemorrhage. This can be accomplished by inserting a straight Keith needle with a 0-0 absorbable suture from the outside of the abdomen at one side of the affected quadrant and then grasping the needle with laparoscopic forceps and pushing it back out of the abdomen at the opposite side of the affected quadrant until it can be recovered on the surface of the abdomen (Fig. 9–28 on the Expert Consult website![]() ). This broad suture is then tied over a gauze 4 × 4-inch bolster on the abdominal surface; the port can be used throughout the procedure. Alternatively, various port closure devices, in particular the Carter-Thomason device, may be used to similarly pass a suture to control the bleeding (Ortega, 1996); at the end of the procedure, a device of this nature should be used to definitively close the port site and occlude the injured vessel.
). This broad suture is then tied over a gauze 4 × 4-inch bolster on the abdominal surface; the port can be used throughout the procedure. Alternatively, various port closure devices, in particular the Carter-Thomason device, may be used to similarly pass a suture to control the bleeding (Ortega, 1996); at the end of the procedure, a device of this nature should be used to definitively close the port site and occlude the injured vessel.
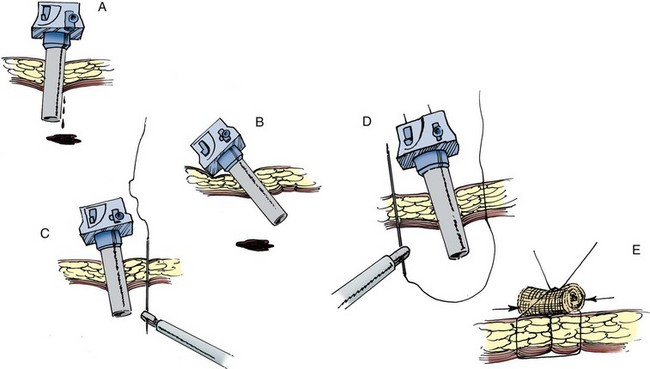
Figure 9–28 A, Bleeding at the cannula site. B, Cannula can be cantilevered into each of the four different quadrants to identify the source of bleeding. C and D, Straight Keith needle may be used to traverse the site of bleeding. E, Suture is tied down over a gauze bolster.
(From Clayman RV, McDougall EM, editors. Laparoscopic urology. St. Louis: Quality Medical Publishing; 1993.)
This problem can often be avoided by routinely transilluminating the abdominal wall, especially in the thin patient, before trocar placement so large surface vessels can be avoided. In addition, the routine spreading of the subcutaneous tissues of the proposed port site with a blunt clamp (e.g., Kelly clamp) may be helpful. Also, the use of only blunt trocars reduces the chance of vascular wall injury nearly 10-fold. Finally, careful laparoscopic inspection of the peritoneal surface before each secondary port site placement is helpful to identify the area of the inferior epigastric vessels as well as any overlying peritoneal vessels, which can then be avoided; in this regard, port sites should be placed either in the midline or at least 6 cm lateral to the midline (Hashizume and Sugimachi, 1997).
Trocar Position-Related Problems
Three potential problems may occur when the secondary trocars are not properly positioned: “crossing swords,” “striking handles,” and “rollover.” The problem of “crossing swords” is due to the trocars being placed too close to one another; as a result, the intra-abdominal portions of two trocars cross each other so that the two cannot easily be used to deliver instruments to the same surgical site (Fig. 9–29). Similarly, the problem of “striking handles” is also due to trocars being placed too close to one another; as a result, the upper portions of the trocars strike one another on the abdominal surface, again precluding delivery of instruments to a specific surgical site. “Rollover” is a variant of the crossing swords problem, but it occurs between the laparoscope and an instrument. Instead of running parallel to the surgical site, the primary cannula holding the laparoscope and one of the instrument-holding secondary ports are pointed toward each other; as the instrument is advanced toward the surgical site, it strikes and is deflected by the larger laparoscope, thereby rolling over the laparoscope and hence suddenly moving out of the field of view. Because of the large size of most laparoscopes and the much smaller size of the working instruments, it is commonly the surgeon who is most sensitive to this problem.
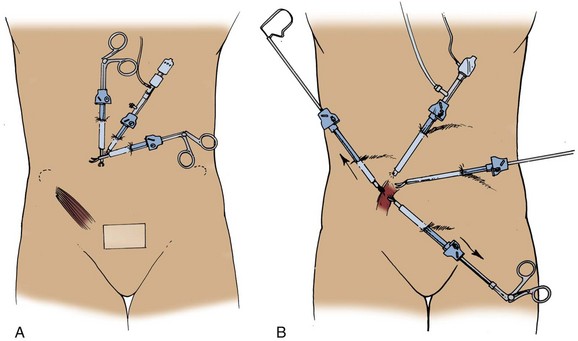
Figure 9–29 A, The cannulas have been placed too close to each other; hence, the intra-abdominal portions of the cannulas are also too close to each other, thereby impairing use of instruments passed through the ports. B, Correct spacing of the trocars eliminates this problem.
(From Clayman RV, McDougall EM, editors. Laparoscopic urology. St. Louis: Quality Medical Publishing; 1993.)
By definition, these problems are more likely to occur during laparoendoscopic single-site surgery. Because the ports are purposely placed in close proximity to one another the surgeon must use advanced techniques or special equipment such as articulating instrumentation or using instruments with different shaft lengths or a low-profile 5-mm laparoscope to overcome these pitfalls. Similarly, when using the da Vinci Robotic System the ports should be placed at least 8 to 10 cm away from one another to avoid robotic arm collision. This can sometimes be challenging on thin patients with limited abdominal wall space.
Usually these problems are a minor annoyance, and the surgeon and assistant need to experience the problem only once to adjust for it. Specifically, to avoid the problem of “striking handles,” the sheaths can be withdrawn a bit from the abdomen, thereby increasing the space between the handles of the instruments. The problems of “crossing swords” and “rollover” can be remedied by moving the handles of the crossing trocars closer to one another, thereby moving the tips of the trocars farther apart. When this is done to correct a “rollover” the surgical site may be displaced into one corner of the monitor; however, the desired delivery of the instrument to the surgical site can then be accomplished. A 30-degree laparoscopic lens can usually allow the laparoscope to be placed parallel to the instrument it is rolling over and then rotated to maintain the operative site image and eliminate the rollover. Alternatively, the handle of the instrument causing the rollover can be moved toward the extra-abdominal end of the laparoscope, thereby “separating” them intra-abdominally.
The best way to handle these situations is to properly place and direct each trocar at the beginning of the case. For some procedures, such as pyeloplasty, this may be accomplished by placing all the trocars on the same line (i.e., midline) so they are all working parallel to each other, whereas for procedures such as nephrectomy the goal is to place the trocars so that they surround the surgical site, forming a diamond pattern within which the kidney lies. Regardless, each trocar needs to be inserted such that it “points” to the pathology; this precludes the problem of having to redirect the trocar throughout the case, adding to surgeon fatigue and unnecessary trauma to the peritoneum and abdominal wall musculature. Also, the surgeon should avoid advancing sheaths too far into the abdomen. This can be accomplished by selecting the proper length of trocar for each patient. Lastly, if trocar interactions become particularly vexing during a procedure, the surgeon should not hesitate to place an additional 5-mm secondary trocar in a more conducive site to eliminate the problem.
Complications Related to General Anesthesia Unique to Laparoscopy
Cardiac Arrhythmias and Cardiac Arrest
Cardiac arrhythmias are frequently seen during anesthesia in laparoscopic procedures. The most common arrhythmia is sinus tachycardia; bradyarrhythmias (e.g., atrioventricular dissociation, nodal rhythm, sinus bradycardia) may develop independently or in combination with tachycardia during the same procedure (Myles, 1991). Conditions leading to development of arrhythmias are CO2 insufflation, hypercapnia, increased vagal tone owing to traction on pelvic or peritoneal structures, Trendelenburg position, anesthetic drugs (especially halothane in combination with spontaneous ventilation), preoperative patient anxiety, endobronchial intubation, and gas embolism (Harris et al, 1984; Myles, 1991). In rare cases, asystolic cardiac arrest and cardiovascular collapse may develop (Shifren et al, 1992).
The role of the anesthesiologist throughout the laparoscopic procedure is of paramount importance. Continuous monitoring of cardiovascular (electrocardiogram, arterial blood pressure), and pulmonary (capnometry, in-line oxygen, airway pressures and tidal volume, frequent arterial blood gas analyses) parameters is essential. Invasive cardiac monitoring should be instituted in patients with heart disease (using a Swan-Ganz catheter) or in high-risk (i.e., American Society of Anesthesiologists type 3 or 4) patients when prolonged and complicated laparoscopic procedures are expected, especially because a central venous pressure line cannot be relied on for accurate readings during laparoscopy.
Because hypercarbia is one of the most common underlying causes of cardiac arrhythmias it is essential to monitor and control this problem. Overall, hypercapnia can be corrected rapidly by adjustment of ventilatory rate and tidal volume, use of positive end-expiratory pressure as needed, and reduction of intra-abdominal pressure to 10 mm Hg. The surgeon can also desufflate the abdomen for 5 to 10 minutes to allow the anesthesiologist to “catch up” and correct the hypercarbia; pneumoperitoneum can then be reinitiated at a lower pressure (5 to 10 mm Hg).
In rare cases, if the hypercarbia cannot be controlled by these maneuvers, helium should be substituted for CO2 as the insufflant; however, this is a rare event and one that can usually be predicted before the procedure. For patients with preoperatively known severe pulmonary compromise, it is prudent to have a tank of helium and the proper helium yoke available in the operating room so the insufflant can be easily switched. Alternatively, argon gas can be used in the acute situation because it is readily available in many operating rooms owing to the argon beam coagulator (Badger et al, 2008).
In the event of cardiac arrest, the surgeon should immediately desufflate the abdomen and provide cardiac massage (compressions) while the anesthesiologist administers 100% oxygen and appropriate drug therapy. If a CO2 embolus is suspected, additional maneuvers, such as immediately turning the patient to a left lateral decubitus, head-down position and possibly attempting to aspirate the embolus, may be performed.
Preventive measures include avoidance of excessive intra-abdominal pressures (>25 mm Hg) over a prolonged period of time and avoidance of certain anesthetic agents or situations (e.g., halothane and spontaneous ventilation). Premedication with atropine may prevent excessive vagal stimulation (Wolf and Monk, 1996).
Changes in Blood Pressure
Hypertension may be caused by inadequate general anesthesia, elevated intra-abdominal pressures, or hypercarbia. Hypotension may be the result of hypoxia, pneumothorax, pneumomediastinum, gas embolus, or hemorrhage (Abdel-Meguid and Gomella, 1996).
Intermittent or continuous noninvasive blood pressure measurements or invasive monitoring of intra-arterial (e.g., radial artery) pressure is part of all laparoscopic procedures. In the event of a marked change in blood pressure, one of the aforementioned conditions must be ruled out. The initial response of the surgeon, provided that there is neither active bleeding nor evidence of retroperitoneal hemorrhage, should be to desufflate the abdomen. In addition to desufflation, therapy specific to an underlying laparoscopic cause may include CO2 elimination by increased ventilation, attempts to increase oxygen saturation, treatment of an underlying pneumothorax, pneumomediastinum, or gas embolus, and pharmacologic (vasodilators or vasoconstrictors) therapy.
Aspiration of Gastric Contents
Aspiration of gastric contents may occur more frequently in patients with a hiatal hernia, significant obesity, diabetes with a history of gastroparesis, or any form of gastric outlet obstruction (Hanley, 1992). The combination of elevated intra-abdominal pressures from the pneumoperitoneum, morbid obesity, and use of the Trendelenburg position increases the likelihood of this complication (Abdel-Meguid and Gomella, 1996).
The diagnosis is easily made because the problem usually occurs during intubation, with associated coughing. The response to suspected aspiration depends on the intubation status of the patient. If the patient is not intubated, the head should be turned sideways and all gastric secretions should be vigorously suctioned (Hagberg and Boin, 1999). If the endotracheal tube is already in place, it should be left in situ and aggressive suctioning should be initiated. If aspiration of gastric contents occurs postoperatively, reintubation with mechanical ventilation and positive end-expiratory pressure may be indicated (Hagberg and Boin, 1999). Neither corticosteroid nor broad-coverage antibiotic administration is indicated (Tasch, 1999).
To prevent this problem in high-risk patients, oral or intravenous administration of 10 mg of metoclopramide is recommended. This medication may decrease the incidence of aspiration by increasing the tone of the lower esophageal sphincter. In addition, in patients with known gastroesophageal reflux, H2 blockers reduce gastric acidity and attendant morbidity if aspiration of gastric contents should occur (Hanley, 1992; Abdel-Meguid and Gomella, 1996). Also, in patients with known gastroesophageal reflux or other predisposing factors for gastric aspiration, a cuffed endotracheal tube should always be placed. Lastly, among these high-risk patients, administration of atropine should be avoided because it decreases the tone of the lower esophageal sphincter (Duffey, 1979).
Hypothermia
The patient’s body core temperature may drop during prolonged laparoscopic procedures, especially if there is leakage of insufflant around the port sites. The CO2 that is used is typically neither warm nor humidified. The resulting decrease in temperature may be 0.3° C for each 50 L of CO2 insufflated (Ott, 1991a). The ambient operating room temperature may exacerbate this effect.
The clinical effects of hypothermia are well described. Core body temperatures around the 35° C level may result in (1) increased bleeding tendency owing to impaired platelet function, reduced activity of coagulation factors in the coagulation cascade, and enhanced fibrinolysis; (2) increased adrenergic response with vasoconstriction and increased arterial blood pressure; (3) prolonged recovery time due to increased blood gas solubility; (4) twofold to threefold increase in the incidence of early postoperative myocardial ischemia in high-risk patients; and (5) impaired wound healing and increased susceptibility to wound infections (Rosenberg and Frank, 1999).
In general there are no specific anesthetic symptoms that can be appreciated intraoperatively except for cardiac arrhythmias. In particular, atrial fibrillation may occur in extreme cases of hypothermia (body core temperature around 30° C). In such a case, if the patient arrests, cardiac resuscitative efforts should be prolonged because the patient must be warmed for resuscitative efforts to have their proper impact. The problem is combated by use of warm intravenous fluid and application of active warming systems.
In almost all cases, hypothermia can be avoided. Adjuncts to support the patient’s body temperature include intravenous fluid warming, active warming by forced-air systems, circulating warm-water mattresses, and radiant heaters (Rosenberg and Frank, 1999). In addition, warming and humidifying CO2 to physiologic levels, especially when a prolonged laparoscopic procedure is anticipated, is helpful (Ott, 1991b). However, warming the insufflant alone, should be avoided, because this causes a drying of the intraperitoneal tissues and has been associated with increased postoperative patient discomfort (Slim et al, 1999).
Complications Related to the Surgical Procedure
Bowel Injury: Electrosurgical Etiology
Electrosurgically induced thermal injury may occur because of one of four mechanisms: inappropriate direct activation, coupling to another instrument, capacitive coupling, and insulation failure. Active electrode trauma by unintended activation causes direct bowel or other organ injury; it may occur when the instrument is left unobserved within the peritoneal cavity or when electrode activation is carried out by someone other than the primary surgeon. Furthermore, active electrode trauma may be seen when coagulation extends beyond the intended site and reaches other adjacent structures (e.g., bowel, blood vessels, nerves, ureter); this is more commonly seen when high electrocoagulation settings (i.e., >30 watts) are used instead of blended or pure cutting current. Direct coupling may occur when the active electrosurgical instrument touches another instrument that is in direct contact with other tissue (e.g., bowel). If this happens outside the field of view provided by the laparoscope, it may remain unnoticed by the surgical team. Injury owing to capacitive coupling occurs when the surrounding charge, which is intrinsic to all activated monopolar electrodes, is not allowed to conduct back to and disperse through the abdominal wall (Zucker et al, 1995; Munro, 1997). This condition may develop when a metal cannula is anchored to the skin with a nonconductive plastic grip, which, as previously noted, should never be done (Fig. 9–30). As a result, the electrical field, which builds up around the activated electrosurgical instrument and is conducted to the metal trocar through which it has been placed, cannot then be conducted to the abdominal wall because the plastic retainer acts as an insulator. This may lead to a high power density along the portion of the metal cannula that is inside the abdomen; the electrical charge built up on the cannula can then travel to other tissues in contact with the cannula. Similarly, capacitive coupling may constitute a risk when electrosurgical probes are used through operating laparoscopes, which are, in turn, inserted through plastic sheaths. The metal shaft of the laparoscope then becomes a repository for electrical current and may discharge this energy to any tissue in contact with the laparoscope. In general, the side-arm operating laparoscope is rarely used during urologic procedures. The risk of this complication is also increased when older generators with high-voltage output and/or electrodes with thicker diameters are used, especially in coagulation rather than cutting mode (Munro, 1997). Lastly, insulation breakdown may allow current to escape along the shaft of the instrument, thereby harming tissues that are otherwise outside the field of view of the laparoscope. Insulation breakdown along the shaft of the instrument may be a result of repeated use, resterilization, or mechanical damage to the instrument during repeated insertion through a trocar. In this situation, the small break in insulation results in an area of very high power density that then discharges to the nearest soft tissue.
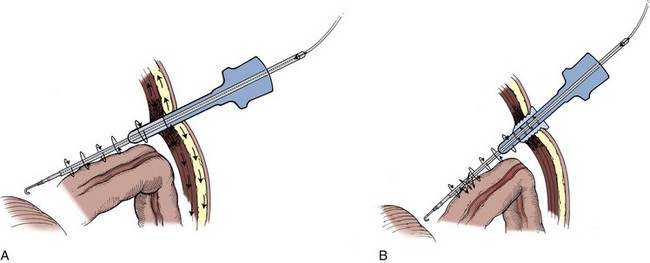
Figure 9–30 Capacitive coupling. A, Charge surrounding the activated monopolar electrode is conducted back to the all-metal cannula and dispersed by the abdominal wall. B, The electrosurgical instrument is being used through a metal cannula that has been anchored to the skin with a nonconductive plastic grip; accordingly, the electrical field cannot be conducted to the abdominal wall because the plastic retainer acts as an insulator; a stronger electrical charge is thus conducted to any other tissue in contact with the cannula.
Intraoperatively, thermal injuries of the bowel may present as whitish spots on the serosal lining. In severe cases, the muscularis mucosae or the intestinal lumen may be seen. However, in many patients, the event of thermal injury of the bowel is not realized at the time of the procedure. Postoperatively, the patient with unrecognized bowel trauma may not develop fever, nausea, or signs of peritonitis for many days; indeed, the full extent of the bowel necrosis may take up to 18 days to fully develop (Abdel-Meguid and Gomella, 1996). Therefore, the problem often does not become manifest until the patient has actually been discharged from the hospital.
Accordingly, bowel injury must be ruled out for any patient who develops a fever beyond postoperative day 1 or who complains of increasing abdominal discomfort. Abdominal radiographs are notoriously inaccurate because the CO2 from the laparoscopy may remain as “free air” for upwards of 9 days after the procedure; however, an ileus pattern is usually present. The more sensitive test is an abdominal CT scan with oral contrast accompanied by delayed films, usually 6 or more hours after the initial oral contrast load. Laboratory values may be remarkable for leukocytosis with an associated left shift (i.e., increased percentage of neutrophils); in some patients, the latter occurs in the face of a normal or even low leukocyte count, making the “left shift” a more reliable sign than the absolute white cell count.
Minor postoperative thermal injuries of the bowel, discovered late in the postoperative period (i.e., >5 to 7 days postoperatively) may be managed conservatively, aided by administration of antibiotics and an elemental diet. Indeed, a closed fistula may develop that will heal with this approach. However, if the patient does not respond rapidly or develops worsening peritonitis, open surgical exploration is mandatory. Thermal injury caused by monopolar cautery often results in tissue damage that extends beyond the visible area of necrosis. With this in mind, the surgeon should perform a bowel resection with a safety margin of 6 cm on either side before completing an end-to-end anastomosis (Abdel-Meguid and Gomella, 1996).
Thermal injury caused by bipolar electrosurgery is more confined to the visible area of damage. These injuries only occur due to direct firing of the instrument on the bowel. If the injury is small, it can be managed by simple excision of the defect and closure of the bowel wall. Bipolar injuries that involve more than half of the circumference of the bowel should be treated by excision of the affected segment of the bowel followed by end-to-end anastomosis (Abdel-Meguid and Gomella, 1996).
The goal of every laparoscopic surgeon is to never experience a thermal complication. To this end there are several actions the surgeon can take to lessen the risks. First, electrosurgical instruments must be carefully inspected before use for any “breaks” in the insulation; if these are found, the instrument must be sent out to be recoated. Next, electrosurgical instruments should never be left untended within the abdomen; when not in use they must be removed from the abdomen. Also, control of electrode activation should be performed only by the primary surgeon. The foot pedal should be placed so that only the surgeon can depress it. To facilitate surgeon-only activation of the instrument, some newer laparoscopic instruments such as the 5-mm LigaSure (ValleyLab, Boulder, CO) are designed with ergonomic thumb-operated mechanisms that only the operative surgeon can activate. Also, isolation of the area to be cauterized from the surrounding tissues (vessels, nerves, ureter), as well as use of bipolar electrocautery, reduces the risk of thermal injury to other tissues. In addition, the electrosurgical device should never be activated unless the entire extent of the metal portion of the instrument is in view; this includes not only the active tip of the instrument but also any exposed, uncoated metal joints that may lie just behind the tip of the instrument. In this manner both inadvertent direct injury to adjacent tissue and direct coupling to another instrument can be avoided. Problems of capacitive coupling can be precluded by not creating a situation in which a mixture of conducting and nonconducting elements are used by the surgeon (e.g., metal trocars combined with plastic retainers, electrosurgical devices through uncoated operating laparoscopes passed through plastic trocars). In addition, use of modern generators and small-diameter electrodes significantly decreases the risk of capacitive coupling (Munro, 1997), as does greater use of blended or pure cutting current. The high voltages needed for pure coagulation current pose the greatest threat for electrosurgical injury, especially through the mechanism of capacitive coupling and insulation failure. Lastly, an active electrode monitoring system (Encision, Boulder, CO) is extremely helpful; with this system, any sudden break in the insulation of the electrosurgical instrument (e.g., with scissors or hook electrode) results in immediate shutdown of the electrosurgical current, thereby precluding an electrosurgical injury.
Bowel Injury: Mechanical
Inadvertent mechanical damage can be caused by a wide variety of sharp and blunt instruments (e.g., laparoscopic graspers, scissors, retractors). This type of injury is more visible to the surgeon and is usually discovered intraoperatively or at the end of the procedure when the surgical site is irrigated. Direct visual identification during the procedure allows the surgeon to repair the injury laparoscopically, even though the patient has not had a formal bowel preparation. Given its localized nature, bowel resection is rarely necessary. The abdomen should be irrigated copiously at the end of the procedure with 4 to 5 L of an antibiotic-containing solution.
If the situation is missed during the procedure, then postoperatively symptoms develop much earlier than with an electrosurgical injury. Fever, nausea, ileus, and peritonitis develop in the very early postoperative period. Diagnosis is confirmed by an abdominal CT scan with oral contrast material. This type of injury should be managed with immediate return to the operating room to correct the problem by local excision or resection of bowel with subsequent end-to-end anastomosis and copious irrigation of the abdomen (see later).
Delicate handling of tissue with laparoscopic instruments by the main surgeon and the assistants is essential to avoiding this complication. Likewise, it is important that introduction of laparoscopic instruments into the peritoneal cavity be done under strict visual control. Instruments should never be left untended; if they are not in use, they should be withdrawn from the abdominal cavity. Attentiveness, economy of motion, and deftness of touch are essential characteristics of both the successful open and laparoscopic surgeon.
Vascular Injury
Fortunately, direct vascular injury during laparoscopic dissection is a rare event. The use of only blunt trocars, the small nature of the instrumentation, the limitations on surgical speed, and the magnification of the surgical field by the laparoscope all combine to decrease this potential problem.
During right renal dissection, in particular, the chance of a vena cava or renal vein injury is heightened. When this occurs the surgeon can undertake several steps to resolve the bleeding. First, the pneumoperitoneum pressure can be raised to 25 mm Hg, thereby slowing or stopping any venous bleeding. With the use of the irrigator/aspirator, the blood can be cleared and the bleeding site identified; if necessary a second insufflator can be brought into the room and connected to maintain pneumoperitoneum pressure even during use of suction at the site of injury. Next, through one of the 12-mm ports, a gauze sponge or a 4 × 18-inch laparotomy pad can be introduced into the abdomen and handled with a grasping forceps, thereby allowing the surgeon to identify and tamponade the area of bleeding. If the injury is small, then it may respond to simple tamponade; alternatively, a hemostatic patch and/or fibrin glue or thrombin-impregnated granules (e.g., Floseal) may be applied. If the injury is larger, then the surgeon must decide whether to obtain a vascular consult and proceed to convert to an open procedure or to attempt securing the injury with a laparoscopic Satinsky clamp and proceed with intracorporeal suturing, either freehand or with an Endo Stitch and 4 inches of a 4-0 Vicryl suture with a Lapra-Ty clip secured at its end. Care should be taken to use a laparoscopic grasper to introduce the Lapra-Ty clip into the abdomen first, followed by the Endo Stitch device; the shaft of the Endo Stitch cannot be passed alongside the Lapra-Ty clip because the two together are too broad to pass side by side through a 12-mm cannula. With the Endo Stitch device a running suture is done, with the initial Lapra-Ty clip serving as the distal “knot”; after the defect is closed, the suture is again secured with a Lapra-Ty clip. One caveat is important: specifically, owing to the larger holes in the tissue caused by the Endo Stitch needle and suture passing side by side the sewn tissue may ooze slightly after the repair; this can be nicely controlled by the application of thrombin-impregnated granules (e.g., Floseal). Throughout this period, it is essential for the anesthesiologist to administer sufficient fluids or blood replacement to preclude a hypovolemic state because the hypovolemic patient has a higher risk of possible air embolism at these higher intra-abdominal pressures (O’Sullivan et al, 1997).
If the surgeon is able to gain temporary control of the vessel with a grasper then it is often helpful to place an extra 5-mm port that can be used by the assistant for suction/irrigation or to optimize the plane of approach to the injury, thereby facilitating laparoscopic suturing. Either way the additional port allows the surgeon to repair the bleeding site using two hands and with excellent visualization of the surgical field.
Alternatively, the surgeon can convert from standard laparoscopy to a hand-assisted approach. The hand in this case is valuable because it can rapidly tamponade the bleeding site. In this regard it is recommended, if at all possible, to pinch the sidewalls of the vein (e.g., inferior vena cava) closed rather than just putting direct pressure on the top of the injury; the latter approach has a tendency to result in a gradual enlargement of the hole in the vein. Also, by pinching the hole closed, a Satinsky clamp can be more easily passed beneath the surgeon’s fingers to provide reliable control of the injury in preparation for a sutured repair.
Minor arterial injuries usually respond to tamponade. Larger aortic or renal artery injuries are much more difficult to resolve laparoscopically. Although the latter, if it occurs during a planned nephrectomy, can be handled by expeditiously taking the renal artery with a vascular stapler, the former almost invariably leads to immediate conversion and open repair. In this case, the area of injury should be tamponaded with a laparoscopic forceps. A vascular surgeon can be called into the room and the surgeon can proceed to rapidly make a midline incision by swinging one of the midline ports up to the underside of the abdominal wall and cutting down on the shaft of the port. The tamponading laparoscopic forceps directs the surgeon immediately to the site of injury, which can then be properly repaired.
As mentioned previously in this chapter, because most bleeding episodes are unexpected, it is wise to have in the room a hemorrhage tray equipped with all instruments necessary to control bleeding. In most cases, a successful outcome is heavily dependent on a rapid and effective response on the part of the surgeon, which is only possible if the equipment is immediately available and the operating room team is knowledgeable and efficient. A hemorrhage tray that can quickly be opened in case of a vascular injury containing all needed instruments is highly recommended (see Table 9–6). The authors recommend that this tray be wrapped in a red cloth or other highly visible unique wrapping so it is always readily identifiable in the operating room.
Injury to the Urinary Tract
Bladder Injury
Electrocautery dissection, blunt and sharp dissection (with laparoscopic scissors), and laser dissection have been identified as intraoperative causes of bladder injury (Ostrzenski and Ostrzenska, 1998). Concomitant bladder or pelvic anomalies or pathologic conditions (acute or chronic inflammation, prior pelvic or bladder surgery, endometriosis, malignant infiltration, bladder diverticula, amyloidosis, or previous radiation) are predisposing factors that increase the chances of this complication (Ostrzenski and Ostrzenska, 1998).
When a bladder injury has occurred, the intraoperative signs may be subtle. One of the first signs is the presence of blood or gas in the Foley catheter bag. Also, the surgeon may notice clear fluid welling up in the pelvis, although this sign is often obscured if irrigation has been used during the procedure.
Postoperatively, if the bladder injury was missed, the patient may develop oliguria and urinary ascites; this may be accompanied by hyponatremia and, rarely, hyperkalemia with mild elevation of the serum creatinine concentration owing to the peritoneal absorption of urine. Patients who have been discharged from the hospital because of the minor nature of their laparoscopic procedure may contact their physician complaining of lower abdominal discomfort, abdominal swelling, fever, and, in the case of a gynecologic procedure, vaginal discharge.
The intraoperative suspicion of a bladder injury may first arise due to the recognition of an air-expanded urine collection bag; this presumptive diagnosis can be confirmed by the injection of saline mixed with indigo carmine through the Foley catheter. Postoperatively, the diagnosis can be made by radiologic examinations (pelvic ultrasonography, pelvic CT, and/or voiding cystourethrography). Similarly, an endoscopic examination with the injection of 5 mL of indigo carmine intravenously is helpful if a vesical fistula is suspected; the surgeon can then look for blue-tinged vaginal or rectal discharge (Chapron et al, 1995; Ostrzenski and Ostrzenska 1998).
The intraoperative diagnosis of a bladder injury can be followed by laparoscopic repair: suturing with absorbable suture, closing the defect with a laparoscopic stapler, or, if the injury is quite small, using preformed suture loops to encircle and secure the cystotomy (Poffenberger, 1996; Ostrzenski and Ostrzenska, 1998). More extensive defects may require open incisional repair.
When bladder injury is diagnosed postoperatively, the key factor is whether the drainage is extraperitoneal or intraperitoneal, which is largely dependent on the preceding laparoscopic access. Extraperitoneal extravasation without any complicating additional problems may be treated by simple placement of a transurethral indwelling Foley catheter. Intraperitoneal drainage is an indication for subsequent laparoscopic or open repair.
Prevention of bladder injury requires preoperative placement of a Foley catheter. Strict adherence to basic laparoscopic principles remains the hallmark of an uncomplicated laparoscopic procedure; in this regard, avoidance of excessive coagulation near the bladder and dissection with exact knowledge of bladder anatomy (urachus, medial umbilical, and vesicocervical ligaments) are key.
Ureteral Injury
Ureteral injury is usually a result of thermal damage caused by dissection using monopolar electrocautery in the immediate vicinity of the ureter. Its incidence in laparoscopic hysterectomy is 1%; it may also occur during laparoscopic endometriosis ablation and tubal ligation and has been reported during pelvic lymphadenectomy and laparoscopic radical prostatectomy (Baumann et al, 1988; Grainger et al, 1990; Poffenberger, 1996; Liu et al, 1997; Ostrzenski and Ostrzenska, 1998; Guillonneau et al, 2002).
The intraoperative diagnosis is made by the astute laparoscopist when urine is seen to be welling up in the wound. However, if irrigation has been used during the procedure this sign is invariably obscured. As opposed to a bladder injury, macroscopic hematuria or pneumaturia is distinctly unusual with this injury.
Typically, ureteral injuries remain unnoticed throughout the laparoscopic procedure. Within 2 to 3 days after surgery, patients may present with abdominal and/or flank pain, fever, signs of peritonitis, and leukocytosis (Grainger et al, 1990; Liu and McFadden, 2000).
Most of these diagnoses are made during the postoperative period when an intravenous pyelogram or abdominal/pelvic CT is ordered owing to the patient’s complaint of flank pain, abdominal swelling, and/or the physical signs of urinary ascites. Depending on the function of the contralateral kidney and the amount of urine leakage, serum chemistries may reveal hyponatremia and, rarely, hyperkalemia with a mild elevation in the serum creatinine concentration.
If identified intraoperatively, the injury can be repaired laparoscopically. If the injury is due to mechanical trauma, simple closure of the defect can be accomplished with laparoscopic suturing techniques followed by stent placement. If the injury is due to monopolar electrosurgical current, then a formal resection of the affected area and an end-to-end spatulated ureteroureterostomy and stent placement are indicated; ureteral reimplantation is indicated if the level of injury is at the ureterovesical junction. This can be done laparoscopically but may require conversion to an open procedure. An indwelling ureteral stent is placed.
If the problem is detected in the postoperative period, the first step is to place an indwelling ureteral stent and a bladder drainage catheter. If a stent cannot be placed, then a laparoscopic or open surgical intervention is indicated to repair the area of injury; alternatively one can consider a percutaneous nephrostomy and an antegrade attempt at stent placement before seeking a laparoscopic/open solution. Once a cystogram reveals reflux without extravasation, the bladder drainage catheter can be removed. The stent is left in place for 6 to 8 weeks. Careful follow-up is necessary to rule out the development of a ureteral stricture that may require endourologic or formal surgical repair.
Prevention of this injury again harkens back to the importance of the surgeon’s knowledge of laparoscopic anatomy and the course of the ureter with regard to its topographic relation to other anatomic structures (medial umbilical ligament, round ligament or vas deferens, and common iliac artery). During dissection, the use of monopolar electrosurgical coagulation current should be used with great discretion around the ureter. In particular, a “cutting” rather than “coagulation” mode should be used whenever possible; when employing “coagulation” current the wattage should not exceed 30 watts. In addition, fine-tipped electrosurgical instruments should be employed and the duration of discharge should be brief (i.e., multiple short bursts of current rather than a continuous multisecond discharge).
Pancreatic Injury
Injury to the pancreas during left-sided laparoscopic adrenalectomy or radical nephrectomy is most commonly associated with mechanical retraction. The tail of the pancreas overlies the adrenal and may be injured during dissection of the medial aspect of the adrenal and the securing of the splenorenal ligament. The incidence of this complication is 2.1% for radical left nephrectomy and 8.6% for left adrenalectomy (Varkarakis et al, 2004). Of note, the diagnosis is rarely made intraoperatively; indeed, 75% of pancreatic injuries are diagnosed during the postoperative period.
Among the few in whom this diagnosis is made intraoperatively, general surgery consultation can be obtained and the injury possibly repaired by freeing up the tail of the pancreas and excising the injured portion with an Endo GIA stapler. However, more commonly the patient presents postoperatively with abdominal discomfort. Evaluation reveals elevated serum lipase and amylase levels, as well as leukocytosis. CT reveals a fluid collection that can often be drained percutaneously. A nasogastric tube is placed and oral intake is stopped. When drainage drops below 50 mL/24 hr, the drain can be removed, followed by removal of the nasogastric tube and initiation of a low fat diet. Of note, the average hospital stay among patients with this complication was 18 days.
Prevention of this complication requires the surgeon to widely dissect the line of Toldt and the splenophrenic attachments. The latter are freed to the level of the diaphragm. The colon is broadly reflected from the Gerota fascia, and the splenocolic and splenorenal ligaments are divided. With these maneuvers the spleen and colon rotate medially carrying with them the tail of the pancreas, well away from the medial surface of the adrenal gland.
Splenic Injury
The advent of laparoscopic surgery has been associated with a significant decrease in incidental splenectomy, with a fall from 4.3% to only 1.5%. These injuries are invariably discovered intraoperatively and can effectively be treated with argon beam coagulation or with one of the newer hemostatic agents such as fibrin glue or thrombin-impregnated granules. Avoidance of this complication is best achieved by wide mobilization of the spleen during left radical/total nephrectomy. To this end the splenophrenic attachments as well as the splenocolic and splenorenal ligaments need to be divided. Retraction in the area of the spleen should never be on the spleen itself but beneath the area of the splenocolic ligament such that the retractor is placed on these thicker tissues (Cooper et al, 1996; Simon et al, 2004).
Also, the laparoscopic surgeon must ever be vigilant of the splenic vein. This often tortuous vein may run parallel and within millimeters of the superomedial surface of the adrenal gland and the upper pole of the left kidney.
Nerve Injury
There is no problem more vexing to physician and patient alike than a postoperative nerve injury; a technically and surgically successful procedure is marred by an acute complication in a totally unrelated area. This problem is invariably due to patient positioning in combination with the duration of the procedure. The exact incidence of this problem is not known, but in a recent survey of neuromuscular injuries associated with laparoscopic urologic surgery completed by 18 urologists from 15 institutions in the United States it was found that of a total of 1651 procedures there were 46 neuromuscular injuries in 45 patients (2.7%). This included abdominal wall neuralgia (14), extremity sensory deficit (12), extremity motor deficit (8), clinical rhabdomyolysis (6), shoulder contusion (4), and back spasm (2) (Wolf et al, 2000). If the patient is inadequately positioned and/or padded, nerve damage may result owing to abnormal nerve stretching or compression. Among position-related nerve injuries, the brachial plexus appears to be most at risk. Injury may be inflicted in several ways: (1) abduction of the arm beyond 90 degrees, (2) extreme outward rotation of the head of the humerus, and (3) compression damage when shoulder braces are used in the Trendelenburg position, which pushes the clavicle into the retroclavicular space. Other nerves that can be affected by positioning include the femoral nerve, owing to extreme lateral rotation and abduction of the hip joint, and the sciatic nerve, owing to stretching along the superior leg when the patient is in the lateral decubitus position (Hershlag et al, 1990; Abdel-Meguid and Gomella, 1996). In addition, nerves may be injured during the surgery itself because of either direct mechanical injury or monopolar electrosurgical current. In this regard, the nerves most susceptible to damage in urologic laparoscopy are the obturator nerve during pelvic lymphadenectomy and the genitofemoral nerve during radical nephrectomy/nephroureterectomy. The diagnosis is invariably made postoperatively. On awakening from anesthesia, the patient complains of weakness or inability to contract the affected musculature, paresthesias, and/or anesthesia of the innervated skin areas.
Mechanical or electrosurgical injury to a nerve during laparoscopy may be recognized intraoperatively. A common example is transection of the obturator nerve during pelvic lymphadenectomy. A neurosurgical consultation can be obtained, and the nerve can be repaired with 6-0 suture using an open or laparoscopic approach.
In contrast, nerve palsy owing to positioning is recognized only postoperatively, often in the postanesthesia recovery room or on the first postoperative day when the patient tries to ambulate. From both a medical and a legal standpoint, a neurology consultation should be obtained as soon as the patient calls the surgeon’s attention to a possible nerve injury. Neurologic examination with possible nerve conduction studies to document acute damage is important. Physical therapy may facilitate recovery. However, recovery in these cases, if it does not occur within the first few postoperative days, is often slow, requiring months.
Prevention is paramount. Table-mounted accessories for all major commercial operating room tables now exist that aid in safely and effectively positioning patients in the lateral decubitus position and in the prone position. The kidney rest, if used, should be lowered after the pneumoperitoneum or retroperitoneum is obtained and trocars placed. Special head supports are also available. If the arms are to be at the patient’s side, they should be pronated to protect the brachial plexus. If the patient is to be in a lateral decubitus position, all bony prominences should be padded with additional gel pads (i.e., hip, knee, and ankle on the downside leg); a pillow is always placed between the legs. Padding should also be placed beneath Velcro straps and tape which may be used on the upside hip and shoulder. Shoulder braces should not be used owing to risk of brachial plexus injury. Instead, a well-padded wide strap directly across the upper chest is an excellent way to secure the patient when in the extreme Trendelenburg position, which is often required during laparoscopic radical prostatectomy. Extreme abduction of the hip is also to be avoided. Padding must be checked each time the table position is changed. Lastly, as with all areas of laparoscopy, judicious use of monopolar coagulation current and knowledge of anatomy are key to avoiding direct nerve injury during the procedure.
Complications Related to Exiting the Abdomen
Bowel Entrapment
During removal of laparoscopic ports and release of the pneumoperitoneum, omentum, or bowel may be entrapped at one of the port sites. If missed during the process of cannula removal, then in the early postoperative period, usually on the second or third postoperative day, the patient develops an ileus and point tenderness at the port site incision.
The treatment is laparoscopic. The pneumoperitoneum is reestablished through one of the unaffected port sites, and three ports are replaced: one for the camera and two for grasping forceps. The entrapped bowel is visualized, and an atraumatic bowel clamp is placed on the bowel on either side of the area of herniation. Once this is done, the skin sutures of the affected port site are carefully cut and the wound is opened; the surgeon uses a fingertip to manually reduce the bowel into the abdominal cavity. The bowel can then be carefully inspected; if it appears viable, which is usually the case, it can be left in place and the port site closed. Rarely is formal bowel resection and anastomosis required.
This particular problem is the result of a technical error. Indeed, most laparoscopic ports have a hole drilled into the side of the port within a few millimeters of the end of the port’s shaft. This hole equalizes the pressure in the port and the abdomen as the port is pulled out of the abdomen, thereby precluding any bowel being withdrawn with the port. Furthermore, if each port site is endoscopically inspected at the time of cannula removal, bowel or omentum that may have entered the port site can be readily identified and positioned back into the abdominal cavity. When the last endoscope-bearing port is removed, the assistant should pull up on the closure sutures or on a fascial clamp, and the surgeon should back the cannula out of the wound and up onto the shaft of the endoscope so that the endoscope is the last thing to leave the abdomen. This maneuver, again, visually assures the surgeon that no bowel or omentum has entered the track of the final trocar.
Bleeding at the Sheath Site
This problem has been previously discussed under the heading of trocar placement. However, there are times when this problem does not become apparent until the end of the procedure owing to tamponade from the trocar itself. Again, it is essential to inspect each trocar site at 5 mm Hg to rule out this problem. Its resolution and avoidance are as previously described.
Early Postoperative Complications
Acute Hydrocele
When significant amounts of irrigation fluid are used during a laparoscopic case in a male, it is not unusual for this fluid to accumulate in the scrotum if the patient has a patent processus vaginalis or a small hernia. The problem is usually not recognized until late in the first postoperative day when the patient is ambulating. The scrotum enlarges noticeably, and there may be dull, aching scrotal discomfort.
Direct visual inspection and transillumination suffice to make the diagnosis. If a possible underlying testicular pathologic process is a concern, scrotal ultrasonography can be done.
Treatment of acute hydrocele is simply observation and scrotal support. Reabsorption of fluid occurs within a week. Preventative effort with regard to this problem involves aspiration of irrigation fluid at the end of the procedure.
Scrotal and Abdominal Ecchymosis
This is another problem that may not become apparent until the second postoperative day. It is usually a result of delayed subcutaneous bleeding from one of the port sites. There are no specific symptoms, but the purplish discoloration of a large area of skin surrounding a port site is very disconcerting to the patient and family. Indeed, with pelvic ports, the discoloration in the male patient may involve the scrotum, too. There is no specific treatment other than observation and reassurance. The purplish discoloration of the skin eventually turns yellow and then disappears. The patient should be informed of these expected changes in color to preclude further alarm. The best method of prevention is to observe each port entry site after port removal to check for evidence of bleeding.
Pain
Pain may be localized or diffuse. If postoperative pain is limited to a port site, it may be secondary to herniation (immediate or late) or to infection (late). Localized pain combined with a subcutaneous bulge may indicate a rectus sheath hematoma, bleeding and hematoma formation at a port site, or a port site hernia. Pain at a port site without swelling may be due to a particularly broad fascial suture or palpation of the knot of a port site fascial suture in a thin patient. Early in the postoperative course, port site discomfort is to be expected; however, if it appears to be increasing on subsequent postoperative days, then herniation should be suspected.
Immediate, severe, diffuse abdominal pain may be related to the release of noxious material during the procedure (e.g., cyst fluid in patients with autosomal-dominant polycystic kidney disease) or to a bowel injury. Immediate postoperative scapular discomfort may be a result of the CO2 pneumoperitoneum itself causing some irritation of the diaphragm; rarely, this discomfort may be sufficiently severe to mimic the symptoms of a pulmonary embolus. Of note is that this pain is invariably along the area of the right posterior shoulder region. Delayed diffuse abdominal discomfort and development of peritoneal signs or simply ongoing abdominal discomfort accompanied by low-grade fever may be due to an unsuspected bowel injury; usually, this is the result of a monopolar electrosurgical injury and may present as late as 18 days after the procedure. Of note, these patients may not have a leukocytosis; however, the differential usually shows a left shift.
The cause of localized pain can usually be discerned by the astute surgeon. A fascial knot or hematoma causing localized port site pain can be readily palpated; similarly, a port site hematoma is discernible by the firm mass it produces. When none of these signs is present in a patient complaining of localized pain over a port site, a hernia should be suspected; CT readily reveals the diagnosis.
For diffuse pain, an associated low-grade fever and mild leukocytosis/leukopenia with a left shift are hallmarks of bowel injury. This is best diagnosed by CT with oral contrast material and repeat CT 6 to 8 hours later. If chest pain is present, an electrocardiogram plus cardiac enzymes as well as spiral CT of the chest with intravenous administration of a contrast agent or ventilation-perfusion scan of the lungs plus arterial blood gas tests should be performed to rule out myocardial infarction and pulmonary embolism, respectively.
The treatment of localized pain is directed by the diagnosis. Thus, for localized pain owing to a hematoma or a fascial suture, time, reassurance, and a heating pad are all that is necessary. For a port site hernia, laparoscopic reduction, as previously described, is the next step. The treatment of diffuse pain is guided by the CT results. For suspected bowel injury, if it is early in the postoperative course (i.e., if the patient is still in the hospital), reexploration and surgical correction are indicated. If the bowel injury is detected late and CT shows that it is already confined, then it can be managed as a closed fistula with observation and an elemental or parenteral diet; it often takes months for the problem to completely resolve when managed nonsurgically.
Prevention of most of these causes of postoperative pain requires a meticulous, careful inspection of the entire abdomen before ending the procedure. Various methods have been tried, with variable success, to decrease discomfort owing to the pneumoperitoneum; these have included placement of a drain to help expel all the CO2, flushing of the abdomen with nitrous oxide at the end of the procedure to expel the CO2 and replace it with a less irritating gas, maintaining the pneumoperitoneum pressure as low as possible (i.e., 10 to 12 mm Hg), and selective bathing of the surgical site with a solution of local anesthetic (e.g., 30 mL of  % bupivacaine).
% bupivacaine).
Incisional Hernia
In adults, the occurrence of an incisional hernia is usually confined to port sites larger than 10 mm. However, in the pediatric population, this complication can occur even with 5-mm ports. The patient usually complains of localized discomfort accompanied by nausea and signs of ileus. Rarely, diffuse abdominal pain and/or signs of a complete bowel obstruction may be present. On examination there is tenderness and, at times, swelling overlying the affected port site. A plain film of the abdomen may show an ileus pattern; however, the definitive study is abdominal CT, which can actually reveal the bowel protruding above the fascial level.
Laparoscopic repair with dissection of the hernia and subsequent intra-abdominal closure can be accomplished. The method for performing this procedure has already been described. In complicated cases in which a strangulated hernia is suspected or confirmed laparoscopically, open surgical repair is indicated.
This problem is most easily avoided by performing a meticulous fascial suture closure of all “bladed” trocar entry sites equal to or larger than 10 mm in all adults. This should always be done under direct endoscopic monitoring. In children, it is advisable to perform fascial closure of any “bladed” port site 5 mm or larger. The fascial layer is usually closed with an absorbable 0-0 suture as previously described. For patients in whom only nonbladed trocars have been used, fascial closure is indicated only of the midline ports or any port site that has been unduly stretched (e.g., passage of a LapSac entrapment sack). Indeed, some authors recommend no closure even of midline nonbladed trocar sites. Although there has been one report of a hernia developing after use of a nonbladed trocar, this is distinctly rare (Lowry et al, 2003). Indeed, the incidence of postoperative hernia formation has been noted to fall from 1.8% with bladed trocars to only 0.19% for nonbladed larger trocars (Boike et al, 1995; Hashizume and Sugimachi, 1997; Thomas et al, 2003). Of note, with midline hand-assist approaches, a higher incidence of hernia formation has been identified than would otherwise be expected: 4.0% to 7.3%. As such, some authors have recommended closure of this midline incision with interrupted nonabsorbable suture rather than a running, absorbable closure (Troxel and Das, 2005).
Deep Venous Thrombosis and Pulmonary Embolism
Although it seems reasonable to expect decreased venous return and, hence, increased stasis with concomitant higher risk of deep venous thrombosis (DVT) and pulmonary embolism (PE) in patients undergoing laparoscopy, this is remarkably not the case. Indeed, there is no evidence that this complication occurs more often during laparoscopic procedures versus open incisional surgery (Abdel-Meguid and Gomella, 1996; Secin et al, 2008).
Signs of DVT include localized calf tenderness with associated swelling. However, most patients with postoperative DVT have a subclinical course. Indeed, unfortunately, the most common clinical scenario is detection of a DVT only after a patient has developed a PE. Signs of PE include acute shortness of breath, decreased oxygen saturation, and pleuritic chest pain. Additionally, an electrocardiogram might show a right-sided heart strain pattern. The diagnosis of a DVT is made clinically and confirmed using Doppler ultrasonography of the lower extremities. PE is diagnosed most expeditiously by contrast-enhanced chest CT.
Treatment is immediate anticoagulation, initially with heparin and then with warfarin. In patients with a pulmonary embolus who are not candidates for anticoagulation, a vena caval filter can be placed under radiographic control.
Thus far there have been no randomized controlled trials addressing the issue of DVT prophylaxis in patients undergoing laparoscopic and robotic surgery. The American Urological Association has released a “Best Practice Policy Statement” for the prevention of DVT in patients undergoing urologic surgery. Noting the lack of data available directly pertaining to laparoscopic and robotic surgery, the panel recommended the use of pneumatic compression stockings placed before the procedure for all patients. Additionally, they acknowledged that certain high-risk groups may require the use of low-dose unfractionated heparin or low molecular weight heparin before, during, or after surgery.
Pneumatic compression stockings should be placed preoperatively and continued for 48 to 72 hours postoperatively. Additionally, in morbidly obese patients or in individuals at high risk for thrombosis, the addition of unfractionated perioperative heparin (5000 units 2 hours preoperatively and then every 12 hours postoperatively) has also been recommended (Clagett et al, 1995; Capan and Miller, 1999). However, with specific respect to upper retroperitoneal (renal/adrenal/ureter) laparoscopic procedures, at least one recent study by Montgomery and Wolf (2005) indicated that sequential pneumatic compressive stockings provide equivalent DVT prophylaxis compared with subcutaneous fractionated heparin and that the use of subcutaneous heparin may increase the incidence of hemorrhagic complications. Three-hundred and forty-four patients (172 in each group) nonrandomly received heparin or sequential pneumatic compressive stockings as DVT prophylaxis beginning on the day of surgery. In both groups the rate of thrombotic complication was 1.2%. The rate of hemorrhagic complication was 9.3% in the heparinized group, of which 7.0% were considered major. The hemorrhagic complication rate was only 3.5% in the sequential pneumatic compressive stockings group, with 2.9% considered major (P = .045). Thus, after urologic laparoscopy of the upper retroperitoneum, subcutaneous heparin may be associated with increased hemorrhagic complications, without a reduction in thrombotic complications, compared with sequential pneumatic compressive stockings; however, it must be stressed that this was a nonrandomized study obtained from a prospective database augmented by retrospective chart review. Another retrospective multi-institutional review of DVT and PE comprised 5951 patients undergoing laparoscopic or robotic radical prostatectomy; 31 (0.5%) developed symptomatic thrombotic complications (22 DVT, 4 PE, and 5 with both). Two patients died of PE (Secin et al, 2008). These results are similar to the rates of thromboembolic complication with open radical retropubic prostatectomy (Lepor and Kaci, 2003).
Wound Infections
Superficial wound infections may occur at any of the port entry sites. This problem usually presents as local tenderness, redness, and swelling; rarely, it is accompanied by a low-grade fever. Incision and drainage of the port site are both diagnostic and therapeutic; antibiotic therapy is routinely administered.
Prevention of this complication is similar to that done in open surgery and includes attention to antiseptic preparation and sterile draping of the abdominal wall, irrigation of each port site at the end of the procedure, and meticulous closure of the wound. Overall, this is a rare complication with standard laparoscopy. However, with the hand-assist approach a higher incidence of wound infections has been noted. In one report the postoperative wound infection rate at the hand-assist site was 9% (Nelson and Wolf, 2002).
Rhabdomyolysis
Rhabdomyolysis is a devastating complication after laparoscopic surgery. The exact incidence of this problem is not known, but in one survey of neuromuscular injuries associated with laparoscopic urologic surgery completed by 18 urologists from 15 institutions in the United States it was found that of a total of 1651 procedures there were 6 cases (0.4%) of clinical rhabdomyolysis (Wolf et al, 2000). In a more recent study, it was estimated that the occurrence of this problem among patients undergoing retroperitoneal laparoscopic procedures (e.g., renal, ureteral, adrenal) was higher, 1% (Reisiger et al, 2005). Rhabdomyolysis is invariably associated with male patients undergoing longer laparoscopic renal procedures, especially if the kidney rest has been employed for the entire case. Although extra padding applied to pressure areas can be useful in avoiding nerve compression and rhabdomyolysis, gel pads and foam “egg crate” padding have not been conclusively demonstrated to significantly reduce pressure on the patient’s flank in the lateral position (Deane et al, 2008). Women have significantly lower interface pressures than men. A BMI of 25 or greater, use of a kidney rest, and full-table flexion as opposed to half-table flexion were all associated with increases in interface pressures; of these, use of the kidney rest was believed to be the most detrimental and its use beyond 20 to 30 minutes was disparaged. Therefore, male patients with a BMI of 25 or greater undergoing laparoscopic surgery in the lateral position with the kidney rest elevated and the table completely flexed are at highest risk of developing rhabdomyolysis due to flank pressure. In this study the unaugmented operating table mattress was superior to egg crate or gel padding as an augmenting surface material; of note, egg crate padding was equal or superior to the more expensive gel padding.
Rhabdomyolysis presents immediately in the postanesthesia recovery room with the patient complaining of severe pain in the downside hip area. Brown urine may also be noted. The serum creatine kinase value invariably exceeds 5000 units/dL. The treatment is hydration and alkalinization; these maneuvers are undertaken to try to prevent subsequent renal failure. Acute renal failure and the need for transient dialysis occurs in a minority of patients. The more difficult and ominous problem is that of long-term disability caused by muscle necrosis. Extended physical therapy is often required.
Prevention of this problem is essential. This can, to some extent, be done by avoiding use of the kidney rest or employing it for only the earliest part of the case (i.e., less than an hour) and avoiding prolonged periods of hypotension during the procedure (Cadeddu et al, 2001; Kuang et al, 2002; Parsons et al, 2004; Reisiger et al, 2005). Lastly, with increasing skill and experience, few procedures should proceed beyond 5 hours.
Late Postoperative Complications
Complications beyond the 3-week postoperative period are rare. These include primarily lymphatic complications and incisional hernia. The latter has been addressed in the prior discussion because it can also appear as an early postoperative complication.
Lymphocele Formation
Lymphocele formation is most commonly associated with pelvic procedures, such as pelvic lymph node dissection (1.3% incidence) (Kavoussi et al, 1993) or with renal transplantation. The lymphocele may take weeks to develop and may occur despite a transperitoneal approach. Presentation may be by a mass effect, but the astute clinician should realize that it may also present due to local compression causing lower extremity edema, and in rare cases, venous thrombosis and pulmonary embolism. The lymphocele is diagnosed readily by CT. Treatment is immediate percutaneous drainage. Sclerosant therapy can be used to treat the lymphocele; however, if unsuccessful or if the lymphocele is not amenable to percutaneous drainage, then a transperitoneal laparoscopic marsupialization procedure is usually successful. At the time of the procedure, a tag of omentum can be placed into the opening made in the lymphocele to try to prevent closure of the opening and recurrence.
Prevention of lymphocele formation requires marked attention to clipping suspected lymphatic structures. Monopolar electrocoagulation does not work well to seal lymphatics. Bipolar or harmonic devices have been shown to effectively seal lymphatic channels; the bipolar devices created seals that were 5- to 10-fold stronger than the harmonic device (Box et al, 2009).
Chylous Ascites
After left-sided retroperitoneal surgery (i.e., left radical nephrectomy, donor nephrectomy, left-sided retroperitoneal lymph node dissection), the patient may return complaining of a distended abdomen. Although this complaint is common in the initial few days after a laparoscopic procedure, owing to the pneumoperitoneum, irrigation fluid, and/or ileus, it is distinctly rare late in the patient’s course. There is no associated fever, pain, or bowel dysfunction, and routine laboratory studies are within normal limits. If the suspicion is high for chylous ascites, one may simply prescribe a low fat, medium-chain triglyceride diet and choose to observe the patient. Usually the condition is self-limited and resolves without any intervention.
If the presentation is not straightforward, then abdominal CT will reveal the underlying problem and diagnose the presence of ascites. If tapped, the fluid, which is commonly turbid, should be sent for culture, complete blood cell count, and determination of triglyceride, cholesterol, and electrolyte levels. The chylous ascitic fluid may well have elevations in the level of lymphocytes, cholesterol, and triglycerides, the latter two being particularly more likely if following a fatty meal.
Treatment is usually dietary, as previously noted. One may also consider giving somatostatin (Leibovitch et al, 2002). In the rare situation in which the fluid does not resolve with these conservative measures or if the patient is symptomatic, the fluid can be tapped once or twice. If it continues to accumulate, then an open or laparoscopic exploration is undertaken; before the procedure, the patient should be given a “fatty” meal or an intravenous load of hyperalimental lipids, which should create a whitish discoloration of the lymphatic fluid and make the point of lymphatic leakage more visible. Once identified, the leaking lymphatic channel is dissected and secured with either suture or clips (Molina et al, 2003).
Prevention of this type of problem is extremely difficult because the lymphatics are not readily visible during the procedure. In addition, with the recent advent of avoiding clips around the renal hilum, the problem may become more prevalent. To date, instances of post-laparoscopic chylous ascites have been cited only sparingly in the literature (Leibovitch et al, 2002; Molina et al, 2003). However, the authors believe that because the condition is usually self-limited and responds well to dietary measures, it is likely underreported.
Limitations and Advantages of Transperitoneal Versus Extraperitoneal Approach to the Flank and Pelvis
Laparoscopy versus Retroperitoneoscopy
Limitations
Retroperitoneoscopy is associated with unique anatomic orientation and a relatively restricted initial working area compared with transperitoneal laparoscopy. This results in a steeper learning curve with the former technique. Moreover, the fact that a comparatively limited space is available necessitates precise accuracy regarding the strategic placement of ports. The degree of technical difficulty increases in the presence of large-sized specimens. Additionally, retroperitoneoscopic entrapment of these larger specimens may be difficult. The latter problem can be overcome by laparoscopically creating an intentional peritoneotomy at the end of the procedure to allow entrapment of the specimen within the larger peritoneal cavity. Laparoscopic reconstruction and intracorporeal suturing are technically demanding procedures; in the retroperitoneal space, reconstruction can be more challenging compared with transperitoneal laparoscopy due to suboptimal instrument angulation and a smaller working space. One potential way to overcome some of these limitations is to use a robotic approach to the retroperitoneum. Kaouk and coworkers (2008c) reported the Cleveland Clinic experience using a robotic retroperitoneal approach to perform dismembered pyeloplasty in 10 patients. They noted that the enhanced dexterity of the robotic platform did facilitate reconstructive suturing in a smaller working space. Although a retroperitoneal approach might not be advantageous for every surgery there are certain instances in which this approach might be more appropriate (as in the case of multiple prior transabdominal surgeries).
If a patient has a history of prior open retroperitoneal surgery, subsequent attempts at retroperitoneoscopy should be approached with extreme caution. However, as mentioned earlier in this chapter, prior percutaneous renal procedures do not necessarily constitute a contraindication to subsequent retroperitoneoscopy. Lastly, of interest is an increase in pulmonary complications after retroperitoneoscopy versus transperitoneal laparoscopy (see later).
Advantages
With retroperitoneoscopy, morbidity that is usually associated with the transperitoneal approach is commonly avoided (Kavoussi et al, 1993). The risks of inadvertent bowel injury and postoperative ileus are minimized, although not eliminated. This results in a slightly more rapid postoperative recovery. During retroperitoneoscopy and extraperitoneoscopy the bowel can be effectively and safely retracted within its peritoneal cover. Although out of sight, the bowel should never be out of mind, because bowel injuries can still occur. Transperitoneal laparoscopy may be associated with an increased incidence of postoperative shoulder-tip pain, a feature rarely associated with retroperitoneoscopy. In addition, extraperitoneoscopy and retroperitoneoscopy are associated with a significantly lower incidence of postoperative trocar site hernias.
A significant benefit of this technique is rapid and direct access to the renal hilum, with the renal artery being the first hilar structure encountered and controlled. Furthermore, if a partial nephrectomy is planned, it may be more effective to approach a posterior tumor from the retroperitoneal approach whereas anterior tumors are often more easily approached from the transperitoneal approach.
Transperitoneal versus Extraperitoneal Pelvic Surgery
The transperitoneal approach to the pelvis offers easy access to all areas of the pelvic cavity after which the space of Retzius, iliac/obturator fossa, or the rectovesical space is entered directly through the peritoneum. The working space is maximized because the peritoneum does not limit camera or instrument motion once it is incised. The potential limitations associated with this approach, however, are that it is possible to cause mechanical or thermal injury to the bowel and it may be necessary to utilize more extreme Trendelenburg positioning or additional retraction of the bowel to prevent it from impinging on the operative field. The possibility of prolonged ileus has also been suggested; however, in a large prospective study comparing 165 transperitoneal versus 165 extraperitoneal laparoscopic radical prostatectomy patients, Ruiz and colleagues (2004) did not find significant differences between the two groups in terms of hospital stay or medical and surgical complications. Operative time was half an hour shorter in the extraperitoneal group (220.0 vs. 248.5 minutes, P < .0001); however, there was also a trend toward higher positive margins in patients with pT2 (organ confined) cancers in the extraperitoneal group (17.0% vs. 13.0%; P = .42). In two other studies comparing the two approaches, patients undergoing radical prostatectomy were matched with regard to important preoperative attributes and the authors found no significant differences between the extraperitoneal and transperitoneal approaches regarding all important parameters except that the operative time for the extraperitoneal approach is generally a little shorter (30 minutes) (Erdogru et al, 2004; Madi et al, 2007). Interestingly, in one study comparing the two approaches with 50 patients in each arm, in addition to there being no difference between the groups with regard to intraoperative and postoperative parameters, free intraperitoneal air was found on all extraperitoneal patients on the kidney-ureter-bladder radiograph taken on postoperative day 1 (Bivalacqua et al, 2008). Thus this finding should not necessarily be interpreted as an ominous sign of bowel injury unless the patient has other signs and symptoms present (i.e., fever, pain, nausea, elevated white blood cell count). One undeniable advantage in favor of the extraperitoneal approach is that it avoids the necessity of lysis of adhesions in patients who have had prior abdominal surgery. In contrast, for patients who have had prior laparoscopic inguinal hernia repair, a transperitoneal approach is more advantageous.
In summary, for most cases there does not seem to be a clear advantage to using a transperitoneal versus extraperitoneal approach for laparoscopic pelvic surgery. In selected cases described earlier there may be advantages of one approach over the other. The usual determinant for the laparoscopic approach is that of surgeon preference.
Training and Practicing Laparoscopic and Robotic Surgery
Surgeons learning how to perform laparoscopic procedures face several hurdles, including an altered 2D view of a 3D space, counterintuitive control of instruments, different ergonomic constraints, and an altered perspective of surgical anatomy (Piechaud and Pansadoro, 2006). Robotic surgical systems such as the da Vinci Robotic System overcome several of these issues by providing 3D vision and intuitive instrument motion but they, too, have hurdles, such as complete lack of tactile feedback and a learning curve regarding the syncopation of the surgeon’s console. Additionally, much like life in general, the only thing constant about laparoscopic and robotic surgery is change. It is therefore necessary for minimally invasive surgeons, no matter how experienced, to master new skills and familiarize themselves with new technologies as they develop. With an ever-increasing emphasis on patient safety and operating room efficiency it is widely accepted that new surgical skills and techniques should be developed and honed outside the operating room and then transferred into the operating room once proficiency has been developed.
Equipment for Practicing Laparoscopic and Robotic Surgery
A wide variety of practice models exist for both laparoscopic and robotic surgery ranging from inanimate laparoscopic training boxes to live animal and human cadaveric models. There has also been heightened interest in the development of computer-driven virtual reality trainers that can be used to practice standard techniques and whole procedures. As a general premise, nonliving models should be used unless it is absolutely essential to have a live model.
Standard Laparoscopic Training Boxes
The items necessary for a basic laparoscopic training box are a box with an opaque top that is suitable for placement of laparoscopic ports and a vision system consisting of a video monitor, a source of light, and a camera or camera/lens combination. Several commercially available training boxes are available that contain all of the necessary components (Fig. 9–31 on the Expert Consult website![]() ). Alternatively, one can construct a training box from relatively inexpensive materials such as a cardboard box and a web camera with a computer screen or video camera. Although it adds a hint of reality, trocars are not mandatory. In general the instruments and camera should be configured in a triangular fashion such that the camera lies between the right- and left-hand instruments. For optimal ergonomic positioning the angle formed between the right- and left-hand instruments should be between 25 and 45 degrees. Additionally, the angle between the instruments and the horizontal plane should be less than 55 degrees (Frede et al, 1999). Some more formal training boxes will have built-in camera holders such that one person can practice alone without the assistance of a “camera driver.” When possible, however, a camera driver can adjust the lens/camera in, out, up, and down to match the motions of the surgeon. This is most helpful during more delicate maneuvers such as suturing in which panning the view from wide to closeup is essential. This gives the assistant the opportunity to practice “driving camera,” which is also an essential skill for laparoscopy.
). Alternatively, one can construct a training box from relatively inexpensive materials such as a cardboard box and a web camera with a computer screen or video camera. Although it adds a hint of reality, trocars are not mandatory. In general the instruments and camera should be configured in a triangular fashion such that the camera lies between the right- and left-hand instruments. For optimal ergonomic positioning the angle formed between the right- and left-hand instruments should be between 25 and 45 degrees. Additionally, the angle between the instruments and the horizontal plane should be less than 55 degrees (Frede et al, 1999). Some more formal training boxes will have built-in camera holders such that one person can practice alone without the assistance of a “camera driver.” When possible, however, a camera driver can adjust the lens/camera in, out, up, and down to match the motions of the surgeon. This is most helpful during more delicate maneuvers such as suturing in which panning the view from wide to closeup is essential. This gives the assistant the opportunity to practice “driving camera,” which is also an essential skill for laparoscopy.

Figure 9–31 A, Example of a standard laparoscopic training box with a monitor, camera/lens combo, and light source. B, LapEd EZ Trainer is a portable briefcase-like box that opens to accommodate a laptop computer that is then connected through the USB port to the contained cam-camera thereby allowing the laparoscopic skills practice platform to be visualized on the laptop screen.
Instruments
Necessary instruments might include laparoscopic graspers, scissors, and dissectors, essentially any instrument necessary to meet the requirements of the training drill. Practice for basic suturing requires a pair of laparoscopic needle drivers and some suture. A 3-0 suture on an SH needle is best for general practice. Other suture and needle types can be used for practicing more specialized procedures.
Inanimate Training Models
Training models for basic tasks such as touching areas of the box at different depths and heights within space, passing objects between instruments and through and around obstacles, and other helpful maneuvers such as cutting and tissue manipulation can be constructed from common materials such as paper, sponges, rubber gloves, rubber tubing, and other commonly found, abundant materials (Fig. 9–32 on the Expert Consult website![]() ). Alternatively, a wide variety of commercially available models exist that are more realistic looking and may mimic actual operative scenarios more closely (Fig. 9–33 on the Expert Consult website
). Alternatively, a wide variety of commercially available models exist that are more realistic looking and may mimic actual operative scenarios more closely (Fig. 9–33 on the Expert Consult website![]() ). Whether simple or complex, training models can be placed within standard training boxes and act as the basis for general skills acquisition and progression.
). Whether simple or complex, training models can be placed within standard training boxes and act as the basis for general skills acquisition and progression.

Figure 9–32 Example of an inexpensive suture model made from a household sponge and a marker. The dots can be used to practice placing sutures at different angles.
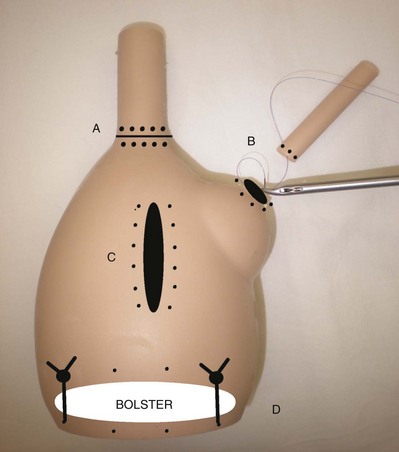
Figure 9–33 Example of a task-specific training model LapEd (4-in-1) model: This model can be used to practice four different urology specific tasks: (A) vesicourethral anastomosis, (B) dismembered pyeloplasty anastomosis, (C) closure of cystotomy, and (D) renorrhaphy.
(From Abraham JB, Abdelshehid CS, Lee HJ, et al: LapED 4-in-1 silicone training aid for practicing laparoscopic skills and tasks: a preliminary evaluation. J Endourol 2008;22:1351–7.)
Nonliving Animal Tissue Models
The advantage of using real but nonliving animal tissue to create training models is that it creates a more realistic environment providing true to life tissue handling. Generally the tissues used are inexpensive and can be purchased at a butcher shop or market. The disadvantage is that it requires more intense cleanup after practice. Generally the field inside the training box is set up to mimic a specific surgical scenario and develop specific skills. For instance, the stomach and esophagus of a chicken can be divided and then sewn back together laparoscopically to practice the fashioning of a vesicourethral anastomosis or pyeloplasty (Laguna et al, 2006; Ramachandran et al, 2008).
Live Animal Models
The obvious advantage of live animal models is the opportunity to train under more realistic conditions, including more accurate anatomic relationships of tissue and the necessity for hemostasis that does not exist with inanimate models. The disadvantages of using live animal models is that it requires extensive resources such as a veterinary facility, personnel for care of and providing anesthesia for the animals, and, eventually, euthanasia of the animals. For these reasons, practicing surgical skills on live animals is not appropriate for daily practice. Instead, these opportunities are usually reserved for formal training courses or when the need for a realistic environment warrants the necessary resources. In general the species of animal used for practice must have a sufficient peritoneal compartment to provide an adequate working space. For fine procedures, animals as small as rabbits can be used. Swine are the most common animals used for laparoscopic and robotic training because they are readily available, they have the appropriate size peritoneal compartment, and their anatomy mimics relatively well that of humans. It must be noted, however, that the amount of bleeding that comes from a pig kidney (in partial nephrectomy, for example) is not realistic compared with that of a human. Additionally, the porcine bladder is almost completely intraperitoneal.
Cadaveric Models
One of the first experiences most medical students have in training is anatomy, and this entails cadaveric dissection. The reason is simple. Short of operating on real patients there is no better or more realistic way to learn human anatomy. This holds true for surgical practice as well: however, owing to the major resources necessary to have an anatomic gift program and the significantly high cost for a cadaver compared with alternative models, it is often not practical to use cadavers. If, however, facilities and funding are available and there is a need for practicing a procedure in the most realistic environment, there is no substitute for cadaveric models.
Virtual Reality Trainers
Virtual reality (VR) trainers are computer-based simulators that offer the opportunity to practice laparoscopic and robotic skills through specific tasks as well as whole procedures. Typically, trainees view the computer-generated operative field or training drill on a computer monitor and perform tasks using laparoscopic manipulators. The instruments are depicted on the viewer and correspond with the trainee’s movements. A major advantage of VR trainers is that they can collect information regarding trainee performance and track progress over time in terms of precision, economy of motion, and other parameters of interest. Because the trainers are computer based it is possible to develop training programs and curricula including didactics, videos, and live practice sessions. The trainee can progress through the curriculum based on performance. VR trainers have been shown to improve the skills of trainees helping to prepare them for better performance during live surgery (Seymour et al, 2002; Lucas et al, 2008). Several commercially produced VR trainers for laparoscopy are available but are quite costly owing to the advanced technology and software development necessary. An experimental VR trainer for robotic surgery is also under development (Albani and Lee, 2007).
Formal Training Programs
Surgical competence is defined by technical and nontechnical skills (Hoznek et al, 2006). Traditionally these skills have been passed down from teacher to apprentice in the process of caring for and operating on real patients during residency training. The technical skills were taught during real operative procedures and the nontechnical skills such as surgical judgment, problem anticipation and avoidance, economy of motion, and so on were gained by experience of the trainee with supervision from a more senior instructor. In this way, traditional residency programs have been more “learning” programs than “teaching” programs. With ever-increasing emphasis being placed on patient safety, operating room efficiency, and cost containment there has been a great interest in evaluating the efficacy of formal training programs in laparoscopic and robotic surgery. Such programs offer the opportunity to receive formal instruction in a safe, stress-free environment with the intention of honing both technical and nontechnical skills.
With this concept in mind one method of concentrated instruction and skills acquisition is the “mini-residency” or “mini-fellowship.” In this training model surgeons spend a period of several days (typically 5 days) receiving concentrated instruction on a given set of surgical skills or procedures including didactic sessions, clinical case observation, and laboratory training using both inanimate and animate models. Typically, the attendees also gain access to expert proctors during their initial learning curve for the new procedure. Mini-residency training has been shown to be effective in helping postgraduate urologists incorporate new laparoscopic and robotic procedures into their clinical practices. In one study of laparoscopic renal surgery mini-fellowship trainees at the University of California, Irvine, 73% of attendees were performing laparoscopic renal surgery 3 years later (Kolla et al, 2010). This was also found with robotic surgical training: of 47 urologists who took a 5-day training course for robotic radical prostatectomy, 90% were performing the procedure in their own practice at 3-year follow-up (Gamboa et al, 2009).
Conclusion
Yesterday’s humor is today’s reality. Cartoons in the 1990s often depicted several surgeons standing around an operating table with a patient in whom numerous laparoscopic ports had been placed. The caption was “Can anyone open?” While this concern has been a rallying cry for the maximally invasive surgeon, the less invasive surgeon sees this as a manifestation of the progress that has occurred in the past 15 years. Laparoscopic procedures unthinkable in early 1990 are now routine. The indications for performing these procedures through a single site are expanding. The use of a robotic surgical platform to perform complex reconstructive procedures is improving the precision and often the outcomes of these operations and makes it possible for a greater number of urologists to perform high-quality laparoscopic procedures. As robotics and flexible endoscopically guided technologies advance in the future we may see adoption of NOTES and our patients may undergo operations that leave no visible scar. The benefit will be more proficient laparoscopic and robotic surgeons who can handle any procedure and any complication without the massive incisions of yore. It is hoped that in the near future a similar cartoon may show a group of urologists in an image-guidance suite or in an office gathered around a patient with the caption: “Can anyone do laparoscopy?” Such should be the next step of our surgical and pharmacologic paths of discovery. At that point Osler’s 20th century admonition, “Diseases that harm require treatments that harm less,” will be truly fulfilled. We are as yet still on the bridge to the future.
Key Points
Abdel-Meguid T, Gomella L. Prevention and management of complications. St. Louis: Quality Medical Publishing; 1996.
Berryhill RJr, Jhaveri J, Yadav R, et al. Robotic prostatectomy: a review of outcomes compared with laparoscopic and open approaches. Urology. 2008;72(1):15-23.
Bongard F, Planim M, Liu SY, et al. Using helium for insufflation during laparoscopy. JAMA. 266(3131), 1991.
Breda A, Bui MH, Liao JC, Schulam PG. Association of bowel rest and ketorolac analgesia with short hospital stay after laparoscopic donor nephrectomy. Urology. 2007;69(5):828-831.
Brown SL, Woo EK. Surgical stapler-associated fatalities and adverse events reported to the Food and Drug Administration. J Am Coll Surg. 2004;199(3):374-381.
Cadeddu JA, Wolfe JSJr, Nakada SY, et al. Complications of laparoscopic procedures after concentrated training in urological laparoscopy. J Urol. 2001;166(6):2109-2111.
Chapron C, Querleu D, Bruhat MA, et al. Surgical complications of diagnostic and operative gynecological laparoscopy: a series of 29,966 cases. Hum Reprod. 1998;13:867-872.
Chiu A, Azadzoi K, Hatzichristou DG, et al. Effects of intra-abdominal pressure on renal tissue perfusion during laparoscopy. J Endourol. 1994;8:99-103.
Erdogru T, Teber D, Frede T, et al. Comparison of transperitoneal and extraperitoneal laparoscopic radical prostatectomy using match-pair analysis. Eur Urol. 2004;46(3):312-319. discussion 320
Fahlenkamp D, Rassweiler J, Fornara P, et al. Complications of laparoscopic procedures in urology: experience with 2,407 procedures at 4 German centers. J Urol. 1999;162:765-770.
Frede T, Stock C, Renner C, et al. Geometry of laparoscopic suturing and knotting techniques. J Endourol. 1999;13(3):191-198.
Gill IS, Schweizer D, Hobart MG, et al. Retroperitoneal laparoscopic radical nephrectomy: the Cleveland Clinic experience. J Urol. 2000;163:1665-1670.
Guillonneau B, Rozet F, Cathelineau X, et al. Perioperative complications of laparoscopic radical prostatectomy: the Montsouris 3-year experience. J Urol. 2002;167(1):51-56.
Landman J, Kerbl K, Rehman J, et al. Evaluation of a vessel sealing system, bipolar electrosurgery, harmonic scalpel, titanium clips, endoscopic gastrointestinal anastomosis vascular staples and sutures for arterial and venous ligation in a porcine model. J Urol. 2003;169(2):697-700.
Landman J, Olweny E, Sundaram CP, et al. Prospective comparison of the immunological and stress response following laparoscopic and open surgery for localized renal cell carcinoma. J Urol. 2004;171(4):1456-1460.
Liu CD, McFadden DW. Laparoscopic port sites do not require fascial closure when nonbladed trocars are used. Am Surg. 2000;66(9):853-854.
Montgomery JS, Wolf JSJr. Venous thrombosis prophylaxis for urological laparoscopy: fractionated heparin versus sequential compression devices. J Urol. 2005;173(5):1623-1626.
Orvieto MA, Chien GW, Laven B, et al. Eliminating knot tying during warm ischemia time for laparoscopic partial nephrectomy. J Urol. 2004;172(6 Pt. 1):2292-2295.
Parsons JK, Varkarakis I, Rha KH, et al. Complications of abdominal urologic laparoscopy: longitudinal five-year analysis. Urology. 2004;63(1):27-32.
Patel R, Stifelman MD. Hand-assisted laparoscopic devices: the second generation. J Endourol. 2004;18(7):649-653.
Patel VR, Palmer KJ, Coughlin G, Samavedi S. Robot-assisted laparoscopic radical prostatectomy: perioperative outcomes of 1500 cases. J Endourol. 2008;22(10):2299-2305.
Pearle M. Physiologic effects of pneumoperitoneum. St. Louis: Quality Medical Publishing; 1996.
Ponsky L, Cherullo E, Moinzadeh A, et al. The Hem-o-lok clip is safe for laparoscopic nephrectomy: a multi-institutional review. Urology. 2008;71(4):593-596.
Rambourg P, Saint-Remy JM, Sturm C, Watson N. Evaluating the differences between fibrin sealants: recommendations from an international advisory panel of hospital pharmacists. Eur J Hosp Pharm Sci. 2006;12(1):3-9.
Recart A, Duchene D, White PF, et al. Efficacy and safety of fast-track recovery strategy for patients undergoing laparoscopic nephrectomy. J Endourol. 2005;19(10):1165-1169.
Secin FP, Jiborn T, Bjartell AS, et al. Multi-institutional study of symptomatic deep venous thrombosis and pulmonary embolism in prostate cancer patients undergoing laparoscopic or robot-assisted laparoscopic radical prostatectomy. Eur Urol. 2008;53(1):134-145.
Vallancien G, Cathelineau X, Baumert H, et al. Complications of transperitoneal laparoscopic surgery in urology: review of 1,311 procedures at a single center. J Urol. 2002;168(1):23-26.
Wolf JS. Tips and tricks for hand-assisted laparoscopy. AUA Update Ser. 2005;24(2):10-15.
Wolf JS, Stoller M. The physiology of laparoscopy: basic principles, complications and other considerations. J Urol. 1994;152:294-302.
Abassi Z, Bishara B, et al. Adverse effects of pneumoperitoneum on renal function: involvement of the endothelin and nitric oxide systems. Am J Physiol Regul Integr Comp Physiol. 2008;294(3):R842-R850.
Abbou CC, Cicco D, et al. Retroperitoneal laparoscopic versus open radical nephrectomy. J Urol. 1999;161:1776-1780.
Abdel-Meguid T, Gomella L. Prevention and management of complications. St. Louis: Quality Medical Publishing; 1996.
Abreu SC, Sharp DS, et al. Thoracic complications during urological laparoscopy. J Urol. 2004;171(4):1451-1455.
Adams J, Moore R, et al. Laparoscopic genitourinary surgery utilizing 20 mm Hg intra-abdominal pressure. J Laparoendosc Adv Surg Tech. 1999;9:131-134.
Adams JB, Micali S, et al. Complications of extraperitoneal balloon dilation. J Endourol. 1996;10(4):375-378.
Adams JB, Schulam PG, et al. New laparoscopic suturing device: initial clinical experience. Urology. 1995;46:242-245.
Ahlering TE, Eichel L, et al. Robotic radical prostatectomy: a technique to reduce pT2 positive margins. Urology. 2004;64(6):1224-1228.
Ahlering TE, Eichel L, et al. Impact of obesity on clinical outcomes in robotic prostatectomy. Urology. 2005;65(4):740-744.
Albani JM, Lee DI. Virtual reality-assisted robotic surgery simulation. J Endourol. 2007;21(3):285-287.
Alexander G, Noe F, et al. Anesthesia for pelvic laparoscopy. Anesth Analg. 1969;48:14-18.
Amaral J, Chrotstek C. Experimental comparison of the ultrasonically-activated scalpel to electrosurgery and laser surgery for laparoscopic use. Minim Invasive Ther Allied Technol. 1997;6:324-331.
Ames C, Perrone JM, Frisella AM, et al. Holding strength of suture anchors for hepatic and renal parenchyma. J Endourol. 2005;19(10):1221-1225.
Aneman A, Svensson M, et al. Intestinal perfusion during pneumoperitoneum with carbon dioxide, nitrogen, and nitric oxide during laparoscopic surgery. Eur J Surg. 2000;166(1):70-76.
Arthur H. Laparoscopy hazard. BMJ. 1970;4:492-493.
Badger WJ, Gallagher BL, et al. Hurdles to helium gas laparoscopy and a readily available alternative. J Endourol. 2008;22(11):2455-2459.
Basting RF, Djakovic N, et al. Use of water jet resection in organ-sparing kidney surgery. J Endourol. 2000;14(6):501-505.
Baumann H, Jaeger P, et al. Ureteral injury after laparoscopic tubal sterilization by bipolar electrocoagulation. Obstet Gynecol. 1988;71:483-485.
Berryhill RJr, Jhaveri J, Yadav R, et al. Robotic prostatectomy: a review of outcomes compared with laparoscopic and open approaches. Urology. 2008;72(1):15-23.
Bivalacqua TJ, Schaeffer EM, et al. Intraperitoneal effects of extraperitoneal laparoscopic radical prostatectomy. Urology. 2008;72(2):273-277.
Boike GM, Miller CE, et al. Incisional bowel herniations after operative laparoscopy: a series of nineteen cases and review of the literature. Am J Obstet Gynecol. 1995;172(6):1726-1731. discussion 1731–3
Bolla G, Tuzzato G. Immunologic postoperative competence after laparoscopy versus laparotomy. Surg Endosc. 2003;17(8):1247-1250.
Bongard F, Planim M, Liu SY, et al. Using helium for insufflation during laparoscopy. JAMA. 266(3131), 1991.
Bonjer HJ, Hazebroek EJ, et al. Open versus closed establishment of pneumoperitoneum in laparoscopic surgery. Br J Surg. 1997;84(5):599-602.
Bordelon B, Hunter J. Laparoscopy in the pregnant patient. Laparosc Surg. 69, 1994.
Bouvy N, Marquet R, et al. Laparoscopic surgery is associated with less tumor growth stimulation than conventional surgery: an experimental study. Br J Surg. 1997;84:358-361.
Box GN, Lee HJ, et al. Rapid communication: robot-assisted NOTES nephrectomy: initial report. J Endourol. 2008;22(3):503-506.
Box GN, Lee HJ, et al. Comparative study of in vivo lymphatic sealing capability of the porcine thoracic duct using laparoscopic dissection devices. J Urol. 2009;181(1):387-391.
Brackman MR, Finelli FC, et al. Helium pneumoperitoneum ameliorates hypercarbia and acidosis associated with carbon dioxide insufflation during laparoscopic gastric bypass in pigs. Obes Surg. 2003;13(5):768-771.
Breda A, Bui MH, Liao JC, Schulam PG. Association of bowel rest and ketorolac analgesia with short hospital stay after laparoscopic donor nephrectomy. Urology. 2007;69(5):828-831.
Brooks P. New laparoscopic retractors. J Am Assoc Gynecol Laparosc. 1993;1:60-61.
Brown JA, Chenven E, et al. Hand-assisted laparoscopic nephroureterectomy (HALNU): an assessment of the impact of obesity in 50 procedures. J Laparoendosc Adv Surg Tech A. 2008;18(1):61-68.
Brown SL, Woo EK. Surgical stapler-associated fatalities and adverse events reported to the Food and Drug Administration. J Am Coll Surg. 2004;199(3):374-381.
Cadeddu J, Chan D, et al. Retroperitoneal access for transperitoneal laparoscopy in patients at high risk for intra-abdominal scarring. J Endourol. 1999;13:567-571.
Cadeddu JA, Wolfe JSJr, Nakada SY, et al. Complications of laparoscopic procedures after concentrated training in urological laparoscopy. J Urol. 2001;166(6):2109-2111.
Caldwell C, Ricotta J. Changes in visceral blood flow with elevated intra-abdominal pressure. J Surg Res. 1987;43:14-20.
Capan L, Miller S. Perioperative embolic complications. In: Benumof JL, Saidman LJ, editors. Anesthesia and perioperative complications. 2nd ed. St Louis: Mosby; 1999:685-738.
Carbonell AM, Joels CS, et al. A comparison of laparoscopic bipolar vessel sealing devices in the hemostasis of small-, medium-, and large-sized arteries. J Laparoendosc Adv Surg Tech A. 2003;13(6):377-380.
Carter J. A new technique of fascial closure for laparoscopic incisions. J Laparoendosc Surg. 1994;4:143-148.
Castellucci SA, Curcillo PG, et al. Single port access adrenalectomy. J Endourol. 2008;22(8):1573-1576.
Chang D, Kirsch A, et al. Oliguria during laparoscopic surgery. J Endourol. 1994;8:349-352.
Chapron C, Dubuisson J, Bruhat MA, et al. Bladder injuries during total laparoscopic hysterectomy: diagnosis, management and prevention. J Gynecol Surg. 1995;11:95-98.
Chapron C, Querleu D, et al. Surgical complications of diagnostic and operative gynecological laparoscopy: a series of 29,966 cases. Hum Reprod. 1998;13:867-872.
Chen RN, Moore RG, et al. Laparoscopic pyeloplasty. Indications, technique, and long-term outcome. Urol Clin North Am. 1998;25(2):323-330.
Chiu A, Azadzoi K, Hatzichristou DG, et al. Effects of intra-abdominal pressure on renal tissue perfusion during laparoscopy. J Endourol. 1994;8:99-103.
Chiu A, Chen K, et al. Direct needle insufflation for pneumoretroperitoneum: anatomic confirmation and clinical experience. Urology. 1995;46:432-437.
Chung HJ, Meng MV, et al. Upper quadrant access for urologic laparoscopy. Urology. 2003;62(6):1117-1119.
Clagett P, Anderson F, et al. Prevention of venous thromboembolism. Chest. 1995;108:312.
Clayman R, Kavoussi L, et al. Laparoscopic nephrectomy: a review of 16 cases. Surg Laparosc Endosc. 1992;2:29-34.
Cochrane J, Forsling M, et al. Arginine vasopressin release following surgical operations. Br J Surg. 1981;68:209-213.
Collins J. Inert gas exchange of subcutaneous and intraperitoneal gas pockets in piglets. Respir Physiol. 1981;46:391-404.
Colombo JRJr, Haber GP, et al. Laparoscopic partial nephrectomy in obese patients. Urology. 2007;69(1):44-48.
Cooper CS, Cohen MB, et al. Splenectomy complicating left nephrectomy. J Urol. 1996;155(1):30-36.
Cooper CS, Donovan JF, et al. Hip and knee replacement as a relative contraindication to laparoscopic pelvic lymph node dissection. J Urol. 1997;158(1):128-130.
Cooper G, Scoggins A, et al. Laparoscopy—a stressful procedure. Anesthesia. 1982;37:266-269.
Corica FA, Boker JR, et al. Short-term impact of a laparoscopic mini-residency experience on postgraduate urologists’ practice patterns. J Am Coll Surg. 2006;203(5):692-698.
Crabtree J, Fishman A. Videoscopic surgery under local and regional anesthesia with helium abdominal insufflation. Surg Endosc. 1999;13:1035-1039.
Cristaldi M, Rovati M, et al. Lymphocytic subpopulation changes after open and laparoscopic cholecystectomy: a prospective and comparative study on 38 patients. Surg Laparosc Endosc. 1997;3:255-261.
Dahn S, Schwalbach P, et al. Influence of different gases used for laparoscopy (helium, carbon dioxide, room air, and xenon) on tumor volume, histomorphology, and leukocyte-tumor-endothelium interaction in intravital microscopy. Surg Endosc. 2005;19(1):65-70.
Deane LA, Lee HJ, et al. Third place: flank position is associated with higher skin-to-surface interface pressures in men versus women: implications for laparoscopic renal surgery and the risk of rhabdomyolysis. J Endourol. 2008;22(6):1147-1151.
Derossis A, Bothwell J, et al. The effect of practice on performance in a laparoscopic simulator. Surg Endosc. 1998;12:1117-1120.
Diakun T. Carbon dioxide embolism: successful resuscitation with cardiopulmonary bypass. Anesthesiology. 1991;74:1151-1153.
Diamant M, Benumof J, et al. Hemodynamics of increased intra-abdominal pressure: interaction with hypovolemia and halothane anesthesia. Anesthesiology. 1978;48:23-27.
Doctor N, Hussain Z. Bilateral pneumothorax associated with laparoscopy. Anesthesia. 1973;28:75-81.
Doyle D, Mark P. Laparoscopy and vagal arrest. Anesthesia. 1989;44:448.
Duffey B. Regurgitation during pelvic laparoscopy. Br J Anaesth. 1979;51:1089-1090.
Eichel L, Abdelshehid C, et al. In vitro comparison of burst tension and puncture pressure in commonly used organ retrieval bags. J Am Coll Surg. 2004;199(1):166-169.
Ekman L, Abrahamsson J, et al. Hemodynamic changes during laparoscopy with positive end-expiratory pressure ventilation. Acta Anaesth Scand. 1988;32:447-453.
El-Minawi M, Wahbi O, et al. Physiologic changes during CO2 and N2O pneumoperitoneum in diagnostic laparoscopy. J Reprod Med. 1981;26:338-346.
Elashry O, Wolf JJ, et al. Comparative clinical study of port closure techniques following laparoscopic surgery. J Am Coll Surg. 1996;183:335-344.
Erdogru T, Teber D, Frede T, et al. Comparison of transperitoneal and extraperitoneal laparoscopic radical prostatectomy using match-pair analysis. Eur Urol. 2004;46(3):312-319. discussion 320
Esposito MP, Vincent JL, Jovallo G. Extraperitoneal robotic radical prostatectomy. In: Stock JA, Esposito MP, Lanteri V, editors. Urologic robotic surgery. Totowa, NJ: Humana Press; 2008:75.
Fahlenkamp D, Rassweiler J, Fornara P, et al. Complications of laparoscopic procedures in urology: experience with 2,407 procedures at 4 German centers. J Urol. 1999;162:765-770.
Falabella A, Moore-Jeffries E, et al. Cardiac function during steep Trendelenburg position and CO2 pneumoperitoneum for robotic-assisted prostatectomy: a trans-oesophageal Doppler probe study. Int J Med Robot. 2007;3(4):312-315.
Fazeli-Matin S, Gill I, et al. Laparoscopic renal and adrenal surgery in obese patients: comparison to open surgery. J Urol. 1999;162:665-669.
Fentie DD, Barrett PH, et al. Metastatic renal cell cancer after laparoscopic radical nephrectomy: long-term follow-up. J Endourol. 2000;14(5):407-411.
Fischer J. The metabolic response to laparoscopic cholecystectomy. Ann Surg. 1995;221:211-213.
Fitzgerald S, Andrus C, et al. Hypercarbia during carbon dioxide pneumoperitoneum. Am J Surg. 1992;163:186-190.
Frede T, Stock C, Renner C, et al. Geometry of laparoscopic suturing and knotting techniques. J Endourol. 1999;13(3):191-198.
Friedman AL, Peters TG, et al. Fatal and nonfatal hemorrhagic complications of living kidney donation. Ann Surg. 2006;243(1):126-130.
Fugita OE, Chan DY, et al. Laparoscopic radical nephrectomy in obese patients: outcomes and technical considerations. Urology. 2004;63(2):247-252. discussion 252
Gamboa AJ, Santos RT, et al. Long-term impact of a robot assisted laparoscopic prostatectomy mini fellowship training program on postgraduate urological practice patterns. J Urol. 2009;181(2):778-782.
Garzotto M, Newman R, et al. Closure of laparoscopic trocar sites using a spring-loaded needle. J Urol. 1995;45:310-312.
Gaur D. Laparoscopic operative retroperitoneoscopy: use of a new device. J Urol. 1992;148:1137-1139.
Geers J, Holden C. Major vascular injury as a complication of laparoscopic surgery: a report of three cases and review of the literature. Am Surg. 1996;62:377-379.
Gettman MT, Lotan Y, et al. Transvaginal laparoscopic nephrectomy: development and feasibility in the porcine model. Urology. 2002;59(3):446-450.
Giebler R, Kabatnik M, et al. Retroperitoneal and intraperitoneal CO2 insufflation have markedly different cardiovascular effects. J Surg Res. 1997;68:153-160.
Gill IS. Retroperitoneal laparoscopic nephrectomy. Urol Clin North Am. 1998;25:343-360.
Gill IS, Canes D, et al. Single port transumbilical (E-NOTES) donor nephrectomy. J Urol. 2008;180(2):637-641. discussion 641
Gill IS, Munch L, et al. Retroperitoneal laparoscopic nephroureterectomy: double-balloon technique. Urology. 1995;46:747-750.
Gill IS, Schweizer D, Hobart MG, et al. Retroperitoneal laparoscopic radical nephrectomy: the Cleveland Clinic experience. J Urol. 2000;163:1665-1670.
Glascock J, Winfield H, et al. Carbon dioxide homeostasis during transperitoneal or extraperitoneal laparoscopic pelvic lymphadenopathy: a real-intraoperative comparison. J Endourol. 1996;10:319-323.
Glerup H, Heindorff H, et al. Elective laparoscopic cholecystectomy nearly abolishes the postoperative hepatic catabolic stress response. Ann Surg. 1995;221:214-219.
Goel RK, Kaouk JH. Single port access renal cryoablation (SPARC): a new approach. Eur Urol. 2008;53(6):1204-1209.
Gofrit ON, Mikahail AA, et al. Surgeons’ perceptions and injuries during and after urologic laparoscopic surgery. Urology. 2008;71(3):404-407.
Goitein D, Papasavas P, Yeaney W, et al. Microsphere intestinal blood flow analysis during pneumoperitoneum using carbon dioxide and helium. Surg Endosc. 2005;19(4):541-545.
Gossot D, Buess A, et al. Ultrasonic dissection for endoscopic surgery. Surg Endosc. 1999;13:412-417.
Grainger D, Soderstrom R, et al. Ureteral injuries at laparoscopy: insights into diagnosis, management, and prevention. Obstet Gynecol. 1990;75:839-843.
Guillonneau B, Rozet F, Cathelineau X, et al. Perioperative complications of laparoscopic radical prostatectomy: the Montsouris 3-year experience. J Urol. 2002;167(1):51-56.
Guillonneau B, Vallancien G. Laparoscopic radical prostatectomy: the Montsouris experience. J Urol. 2000;163(2):418-422.
Gunenc MZ, Yesildaglar N, et al. The safety and efficacy of direct trocar insertion with elevation of the rectus sheath instead of the skin for pneumoperitoneum. Surg Laparosc Endosc Percutan Tech. 2005;15(2):80-81.
Haber GP, Crouzet S, et al. Robotic NOTES (Natural Orifice Translumenal Endoscopic Surgery) in reconstructive urology: initial laboratory experience. Urology. 2008;71(6):996-1000.
Hagberg C, Boin M. Management of the airway. In: Benumof JL, Saidman LJ, editors. Anesthesia and perioperative complications. 2nd ed. St Louis: Mosby; 1999:3-24.
Halevy A, Kais H, et al. Continuous esophageal pH monitoring during laparoscopic cholecystectomy. Surg Endosc. 1994;8:1294-1296.
Hanley E. Anesthesia for laparoscopic surgery. Surg Clin North Am. 1992;72:1013-1019.
Hanney R, Alle K, et al. Major vascular injury and laparoscopy. Aust N Z J Surg. 1995;65:533-535.
Hanney R, Carmalt H, et al. Use of the Hasson cannula producing major vascular injury at laparoscopy. Surg Endosc. 1999;13:1238-1240.
Harkki-Siren P, Sjoberg J, et al. Major complications of laparoscopy: a follow-up Finnish study. Obstet Gynecol. 1999;94:94-98.
Harmann P, Kron I, et al. Elevated intra-abdominal pressure and renal function. Ann Surg. 1982;196:594-597.
Harris M, Plantevin O, et al. Cardiac arrhythmias during anesthesia for laparoscopy. Br J Anaesth. 1984;56:1213-1217.
Hashikura Y, Kawasaki S, et al. Effects of peritoneal insufflation on hepatic and renal blood flow. Surg Endosc. 1994;8:759-761.
Hashizume M, Sugimachi K. Needle and trocar injury during laparoscopic surgery in Japan. Surg Endosc. 1997;11(12):1198-1201.
Hazebroek EJ, Haitsma JJ, et al. Mechanical ventilation with positive end-expiratory pressure preserves arterial oxygenation during prolonged pneumoperitoneum. Surg Endosc. 2002;16(4):685-689.
Hershlag A, Loy R, et al. Femoral neuropathy after laparoscopy. J Reprod Med. 1990;35:575-576.
Hirano D, Minei S, et al. Retroperitoneoscopic adrenalectomy for adrenal tumors via a single large port. J Endourol. 2005;19(7):788-792.
Hoznek A, Salomon L, et al. Simulation training in video-assisted urologic surgery. Curr Urol Rep. 2006;7(2):107-113.
Hsu TH, Sung GT, et al. Retroperitoneoscopic approach to nephrectomy. J Endourol. 1999;13:713-718.
Isariyawongse JP, McGee MF, et al. Pure natural orifice transluminal endoscopic surgery (NOTES) nephrectomy using standard laparoscopic instruments in the porcine model. J Endourol. 2008;22(5):1087-1091.
Ishizaki Y, Bandai Y, et al. Safe intra-abdominal pressure of carbon dioxide pneumoperitoneum during laparoscopic surgery. Surgery. 1993;114:549-554.
Ivankovich A, Miletich D, et al. Cardiovascular effects of intraperitoneal insufflation with carbon dioxide and nitrous oxide in the dog. Anesthesiology. 1975;42:281-287.
Iwamura M, Tsumura H, et al. Port site recurrence of renal cell carcinoma following retroperitoneoscopic radical nephrectomy with manual extraction without using entrapment sac or wound protector. J Urol. 2004;171(3):1234-1235.
Iwase K, Takenaka H, et al. Serial changes in renal function during laparoscopic cholecystectomy. Gastroenterol Jpn. 1992;27:686.
Jacobi C, Ordemann J, et al. Impact of laparoscopy with carbon dioxide versus helium on local and systemic inflammation in an animal model of peritonitis. J Laparoendosc Adv Surg Tech. 1999;3:305-312.
Jacobi C, Sabat R, et al. Pneumoperitoneum with CO2 stimulates malignant colonic cells. Surgery. 1997;121:72-78.
Jakeways M, Mitchell V, et al. Metabolic and inflammatory responses after open or laparoscopic cholecystectomy. Br J Surg. 1994;81:127-131.
Jorgensen J, Hanel K, et al. Thromboembolic complications of laparoscopic cholecystectomy. BMJ. 1993;306:518-519.
Joris J, Noirot D, et al. Hemodynamic changes during laparoscopic cholecystectomy. Anesth Analg. 1993;76:1067-1071.
Kalhan S, Reaney J, et al. Pneumomediastinum and subcutaneous emphysema during laparoscopy. Cleve Clin J Med. 1990;47:639-642.
Kaouk J, Desai MM, Stein R, et al. Single-port laparoscopic and robotic surgery: a scarless approach. Urol Kidney Dis News. 2008;17(2008–2009):24-25.
Kaouk JH, Haber GP, et al. Single-port laparoscopic surgery in urology: initial experience. Urology. 2008;71(1):3-6.
Kaouk JH, Hafron J, et al. Is retroperitoneal approach feasible for robotic dismembered pyeloplasty: initial experience and long-term results. J Endourol. 2008;22(9):2153-2159.
Kaouk JH, Palmer JS. Single-port laparoscopic surgery: initial experience in children for varicocelectomy. BJU Int. 2008;102(1):97-99.
Kapoor A, Nassir A, et al. Comparison of laparoscopic radical renal surgery in morbidly obese and non-obese patients. J Endourol. 2004;18(7):657-660.
Karayiannakis A, Makri G, et al. Systemic stress response after laparoscopic or open cholecystectomy: a randomized trial. Br J Surg. 1997;84:467-471.
Kashtan J, Green J, et al. Hemodynamic effects of increased abdominal pressure. J Surg Res. 1981;30:249-255.
Kavoussi L, Sosa E, et al. Complications of laparoscopic pelvic lymph node dissection. J Urol. 1993;149:322-325.
Keith L, Silver A, et al. Anesthesia for laparoscopy. J Reprod Med. 1974;12:227-233.
Kelman G, Swapp GH, et al. Cardiac output and arterial blood-gas tension during laparoscopy. Br J Anaesth. 1972;44:1155-1162.
Kemen M, Bein N, et al. [Postoperative small intestinal motility after abdominal surgery]. Infusionstherapie. 1991;18:233-235.
Kerbl K, Figenshau RS, et al. Retroperitoneal laparoscopic nephrectomy: laboratory and clinical experience. J Endourol. 1993;7(1):23-26.
Khoury W, Jakowlev K, et al. Renal apoptosis following carbon dioxide pneumoperitoneum in a rat model. J Urol. 2008;180(4):1554-1558.
Kim FJ, Chammas MFJr, et al. Temperature safety profile of laparoscopic devices: Harmonic ACE (ACE), LigaSure V (LV), and plasma trisector (PT). Surg Endosc. 2008;22(6):1464-1469.
Kloosterman T, von Blomberg M, et al. Unimpaired immune function after laparoscopic cholecystectomy. Surgery. 1994;115:424-428.
Knos G, Sung Y, et al. Pneumopericardium associated with laparoscopy. J Clin Anesth. 1991;3:56-59.
Kolla SB, Gamboa AJ, Li R, et al. Impact of a laparoscopic renal surgery mini-fellowship program on postgraduate urologist practice patterns at 3-year followup. J Urol. 2010;184(5):2089-2093.
Kuang W, Ng CS, et al. Rhabdomyolysis after laparoscopic donor nephrectomy. Urology. 2002;60(5):911.
Laguna MP, Arce-Alcazar A, et al. Construct validity of the chicken model in the simulation of laparoscopic radical prostatectomy suture. J Endourol. 2006;20(1):69-73.
Lamberton GR, Hsi RS, et al. Prospective comparison of four laparoscopic vessel ligation devices. J Endourol. 2008;22(10):2307-2312.
Landman J, Kerbl K, Rehman J, et al. Evaluation of a vessel sealing system, bipolar electrosurgery, harmonic scalpel, titanium clips, endoscopic gastrointestinal anastomosis vascular staples and sutures for arterial and venous ligation in a porcine model. J Urol. 2003;169(2):697-700.
Landman J, Olweny E, Sundaram CP, et al. Prospective comparison of the immunological and stress response following laparoscopic and open surgery for localized renal cell carcinoma. J Urol. 2004;171(4):1456-1460.
Landman J, Venkatesh R, et al. Modified renal morcellation for renal cell carcinoma: laboratory experience and early clinical application. Urology. 2003;62(4):632-634. discussion 635
Lavery HJ, Thaly R, et al. Robotic equipment malfunction during robotic prostatectomy: a multi-institutional study. J Endourol. 2008;22(9):2165-2168.
Lee C. Acute hypotension during laparoscopy: a case report. Anesth Analg. 1975;54:142-143.
Lefebvre G, Thirion A, et al. [Laparoscopic surgery versus laparotomy. Comparative analysis of stress markers]. J Gynecol Obstet Biol Reprod. 1992;21:507-511. [in French]
Lehtinen A, Laatikainen T, et al. Modifying effects of epidural analgesia or general anesthesia on the stress hormone response to laparoscopy for in vitro fertilization. J Vitro Fert Embryo Trans. 1987;4:23-29.
Leibovitch I, Mor Y, et al. The diagnosis and management of postoperative chylous ascites. J Urol. 2002;167(2 Pt 1):449-457.
Leighton T, Liu S, et al. Comparative cardiopulmonary effects of carbon dioxide versus helium pneumoperitoneum. Surgery. 1993;113:527-531.
Lepor H, Kaci L. Contemporary evaluation of operative parameters and complications related to open radical retropubic prostatectomy. Urology. 2003;62(4):702-706.
Lewis D, Ryder W, et al. Laparoscopy—an investigation during spontaneous ventilation with halothane. Br J Anaesth. 1972;44:685-691.
Lima E, Rolanda C, et al. Transvesical endoscopic peritoneoscopy: a novel 5 mm port for intra-abdominal scarless surgery. J Urol. 2006;176(2):802-805.
Lima E, Rolanda C, et al. Third-generation nephrectomy by natural orifice transluminal endoscopic surgery. J Urol. 2007;178(6):2648-2654.
Lin P, Grow D. Complications of laparoscopy. Strategies for prevention and cure. Obstet Gynecol Clin North Am. 1999;26:23-38.
Liu C, Wang P, et al. Ureteral injury after laparoscopic surgery. J Am Assoc Gynecol Laparosc. 1997;4:503-506.
Liu CD, McFadden DW. Laparoscopic port sites do not require fascial closure when nonbladed trocars are used. Am Surg. 2000;66(9):853-854.
Loffer FD, Pent D. Laparoscopy in the obese patient. Am J Obstet Gynecol. 1976;125(1):104-107.
Loris J. Anesthetic management of laparoscopy. New York: Churchill Livingstone; 1994.
Lowry PS, Moon TD, et al. Symptomatic port-site hernia associated with a non-bladed trocar after laparoscopic live-donor nephrectomy. J Endourol. 2003;17(7):493-494.
Lucas SM, Zeltser IS, et al. Training on a virtual reality laparoscopic simulator improves performance of an unfamiliar live laparoscopic procedure. J Urol. 2008;180(6):2588-2591. discussion 2591
Madi R, Daignault S, Wood DP. Extraperitoneal v intraperitoneal robotic prostatectomy: analysis of operative outcomes. J Endourol. 2007;21(12):1553-1557.
Makarov DV, Kainth D, et al. Physiologic changes during helium insufflation in high-risk patients during laparoscopic renal procedures. Urology. 2007;70(1):35-37.
Marshall R, Jebson P, et al. Circulatory effects of carbon dioxide insufflation of the peritoneal cavity for laparoscopy. Br J Anaesth. 1972;44:680-684.
McDougall E, Figenshau R, et al. Laparoscopic pneumoperitoneum: impact of body habitus. J Laparoendosc Surg. 1994;4:385-391.
McDougall E, Monk T, et al. The effect of prolonged pneumoperitoneum on renal function in an animal model. J Am Coll Surg. 1996;182:317-328.
McGrath R, Zimmerman J, et al. Carbon dioxide embolism treated with hyperbaric oxygen. Can J Anaesth. 1989;36:586-589.
McKernan J. Extraperitoneal prosthetic inguinal hernia repair using an endoscopic approach. Int Surg. 1995;80:26-28.
Meininger D, Byhahn C, et al. Prolonged intraperitoneal versus extraperitoneal insufflation of carbon dioxide in patients undergoing totally endoscopic robot-assisted radical prostatectomy. Surg Endosc. 2004;18(5):829-833.
Melville R, Frizis H, et al. The stimulus for vasopressin release during laparoscopy. Surg Gynecol Obstet. 1985;161:253-256.
Mendoza D, Newman RC, et al. Laparoscopic complications in markedly obese urologic patients (a multi-institutional review). Urology. 1996;48(4):562-567.
Meng MV, Freise CE, et al. Techniques to optimize vascular control during laparoscopic donor nephrectomy. Urology. 2003;61(1):93-97. discussion 97–8
Meraney A, Gill I. Extraperitoneoscopic pelvic surgery. AUA Update Ser. 2001;20(38):298-303.
Mertens zur Borg IR, Lim A, et al. Effect of intraabdominal pressure elevation and positioning on hemodynamic responses during carbon dioxide pneumoperitoneum for laparoscopic donor nephrectomy: a prospective controlled clinical study. Surg Endosc. 2004;18(6):919-923.
Micali S, Celia A, et al. Tumor seeding in urological laparoscopy: an international survey. J Urol. 2004;171(6 Pt 1):2151-2154.
Minoli G, Terruzi V, et al. The influence of carbon dioxide and nitrous oxide on pain during laparoscopy: a double-blind, controlled trial. Gastrointest Endosc. 1982;28:173-175.
Mintz M. Risks and prophylaxis in laparoscopy: a survey of 100,000 cases. J Reprod Med. 1977;18:269-272.
Molina WR, Desai MM, et al. Laparoscopic management of chylous ascites after donor nephrectomy. J Urol. 2003;170(5):1938.
Monga M, Premoli J, et al. Forearm compression by laparoscopic hand-assist devices. J Endourol. 2004;18(7):654-656.
Monk B, Gordon N, et al. Closure of fascial incision made at the time of laparoscopy: development of a device. J Laparoendosc Surg. 1994;4:257-259.
Montgomery JS, Johnston WK3rd, et al. Wound complications after hand assisted laparoscopic surgery. J Urol. 2005;174(6):2226-2230.
Montgomery JS, Wolf JSJr. Venous thrombosis prophylaxis for urological laparoscopy: fractionated heparin versus sequential compression devices. J Urol. 2005;173(5):1623-1626.
Motew M, Ivankovich A, et al. Cardiovascular effects and acid-base and blood gas changes during laparoscopy. Am J Obstet Gynecol. 1973;115:1002-1012.
Munro M. Complications of laparoscopic electrosurgery. In: Ponsky JL, editor. Complications of endoscopic and laparoscopic surgery. Philadelphia: Lippincott Williams & Wilkins; 1997:265-284.
Myles P. Bradyarrhythmias and laparoscopy: a prospective study of heart rate changes with laparoscopy. Aust N Z J Obstet Gynaecol. 1991;31:171-173.
Nelson CP, Wolf JSJr. Comparison of hand assisted versus standard laparoscopic radical nephrectomy for suspected renal cell carcinoma. J Urol. 2002;167(5):1989-1994.
Neuberger T, Andrus C, et al. Prospective comparison of helium versus carbon dioxide pneumoperitoneum. Gastrointest Endosc. 1994;40:30.
Nezhat F, Tazuke S, et al. Laparoscopy during pregnancy: a literature review. J Soc Laparoendosc Surg. 1997;1:17-27.
Ng C, Gill I, et al. Retroperitoneoscopic surgery is not associated with increased carbon dioxide absorption. J Urol. 1999;162:1268-1272.
Nguyen N, Luketich J, et al. Effect of open and laparoscopic surgery on cellular immunity in a swine model. Surg Laparosc Endosc. 1999;3:176-180.
O’Connor JP, Biyani CS, et al. Laparoscopic nephrectomy for renal-cell carcinoma during pregnancy. J Endourol. 2004;18(9):871-874.
Ono Y, Kinukawa T, et al. Laparoscopic radical nephrectomy for renal cell carcinoma: a five-year experience. Urology. 1999;53:280-286.
O’Sullivan D, Micali S, et al. Risk of carbon dioxide embolism from laparoscopic venous injuries. J Urol. 1997;157:401.
Ortega I. The Carter-Thomason needle suture passer to correct cannula induced defects and vascular injuries in the abdominal wall during laparoscopy. J Am Assoc Gynecol Laparosc. 3(37), 1996.
Orvieto MA, Chien GW, Lavan B, et al. Eliminating knot tying during warm ischemia time for laparoscopic partial nephrectomy. J Urol. 2004;172(6 Pt 1):2292-2295.
Ostrzenski A, Ostrzenska K. Bladder injury during laparoscopic surgery. Obstet Gynecol Surv. 1998;53:175-180.
Ott D. Laparoscopic hypothermia. J Laparoendosc Surg. 1991:127-131.
Ott D. Correction of laparoscopic insufflation hypothermia. J Laparoendosc Surg. 1991;1:183-186.
Parsons JK, Varkarakis I, Rha KH, et al. Complications of abdominal urologic laparoscopy: longitudinal five-year analysis. Urology. 2004;63(1):27-32.
Pascual J, Baranda M, et al. Subcutaneous emphysema, pneumomediastinum, bilateral pneumothorax, and pneumopericardium after laparoscopy. Endoscopy. 22(59), 1990.
Patel R, Stifelman MD. Hand-assisted laparoscopic devices: the second generation. J Endourol. 2004;18(7):649-653.
Patel VR, Palmer KJ, Coughlin G, Samavedi S. Robot-assisted laparoscopic radical prostatectomy: perioperative outcomes of 1500 cases. J Endourol. 2008;22(10):2299-2305.
Pearle M. Physiologic effects of pneumoperitoneum. St. Louis: Quality Medical Publishing; 1996.
Perez J, Taura P, et al. Role of dopamine in renal dysfunction during laparoscopic surgery. Surg Endosc. 2002;16(9):1297-1301.
Phillips JM, Narula N, et al. Histological evaluation of cold versus hot cutting: clinical impact on margin status for laparoscopic partial nephrectomy. J Urol. 2008;180(6):2348-2352.
Piechaud PT, Pansadoro A. Transfer of skills from the experimental model to the patients. Curr Urol Rep. 2006;7(2):96-99.
Poffenberger R. Laparoscopic repair of intraperitoneal bladder injury: a simple new technique. Urology. 1996;47:248-249.
Ponsky L, Cherullo E, Moinzadeh A, et al. The Hem-o-lok clip is safe for laparoscopic nephrectomy: a multi-institutional review. Urology. 2008;71(4):593-596.
Ponsky LE, Cherullo EE, et al. Single access site laparoscopic radical nephrectomy: initial clinical experience. J Endourol. 2008;22(4):663-666.
Prentice J, Martin J. The Trendelenburg position: anesthesiologic considerations. In: Martin J, editor. Positioning in anesthesia and surgery. 2nd ed. Philadelphia: Saunders; 1987:127-145.
Puri G, Singh H. Ventilatory effects of laparoscopy under general anaesthesia. Br J Anaesth. 1992;68:211-213.
Quinlan D, Naslund M, et al. Application of argon beam coagulation in urological surgery. J Urol. 1992;147:410-412.
Rademaker B, Bannenberg J, et al. Effects of pneumoperitoneum with helium on hemodynamics and oxygen transport: a comparison with carbon dioxide. J Laparoendosc Surg. 1995;1:15-20.
Ramachandran A, Kurien A, et al. A novel training model for laparoscopic pyeloplasty using chicken crop. J Endourol. 2008;22(4):725-728.
Raman JD, Bagrodia A, Cadeddu JA, et al. Single-incision, umbilical laparoscopic versus conventional laparoscopic nephrectomy: a comparison of perioperative outcomes and short-term measures of convalescence. Eur Urol. 2008;55(5):1198-1204.
Rane A, Rao P. Single-port-access nephrectomy and other laparoscopic urologic procedures using a novel laparoscopic port (R-port). Urology. 2008;72(2):260-263. discussion 263–4
Rassweiler J, Frede T. Robotics, telesurgery and telementoring—their position in modern urological laparoscopy. Arch Esp Urol. 2002;55(6):610-628.
Rassweiler J, Seemann O, et al. Retroperitoneoscopy: experience with 200 cases. J Urol. 1998;160:1265-1269.
Rassweiler J, Stock C, et al. Organ retrieval system for endoscopic nephrectomy: a comparative study. J Endourol. 1998;12:325-333.
Recart A, Duchene D, White PF, et al. Efficacy and safety of fast-track recovery strategy for patients undergoing laparoscopic nephrectomy. J Endourol. 2005;19(10):1165-1169.
Reisiger K, Landman J, Kibel A, Clayman RV. Laparoscopic renal surgery and the risk of rhabdomyolysis: diagnosis and treatment. Urol. 2005;66(5 Suppl.):29-35.
Richards W, Scovill W, et al. Acute renal failure associated with increased intra-abdominal pressure. Ann Surg. 1983;197:183-187.
Romero FR, Rais-Bahrami S, et al. Laparoscopic partial nephrectomy in obese and non-obese patients: comparison with open surgery. Urology. 2008;71(5):806-809.
Rosenberg H, Frank S. Causes and consequences of hypothermia and hyperthermia. In: Benumof JL, Saidman LJ, editors. Anesthesia and perioperative complications. 2nd ed. St Louis: Mosby; 1999:338-356.
Ruiz L, Salomon L, et al. Comparison of early oncologic results of laparoscopic radical prostatectomy by extraperitoneal versus transperitoneal approach. Eur Urol. 2004;46(1):50-54. discussion 54–6
Sainsbury DC, Dorkin TJ, et al. Laparoscopic radical nephrectomy in first-trimester pregnancy. Urology. 2004;64(6):1231.
Sarli L, Costi R, et al. Prospective randomized trial of low-pressure pneumoperitoneum for reduction of shoulder-tip pain following laparoscopy. Br J Surg. 2000;87(9):1161-1165.
Schippers E, Ottinger A, et al. Intestinal motility after laparoscopic vs conventional cholecystectomy. An animal experiment study and clinical observation. Langenbecks Arch Chir. 1992;377:14-18. [in German]
Schorr R. Regarding laparoscopy-associated mesenteric vascular events: description of an evolving clinical syndrome. J Laparoendosc Adv Surg Tech. 1998;8:249-250.
Scott D, Julian D. Observations on cardiac arrhythmias during laparoscopy. BMJ. 1972;1:411-413.
Sebesta MJ, Bishoff JT. Octylcyanoacrylate skin closure in laparoscopy. JSLS. 2004;8(1):9-14.
Secin FP, Jiborn T, Bjartell AS, et al. Multi-institutional study of symptomatic deep venous thrombosis and pulmonary embolism in prostate cancer patients undergoing laparoscopic or robot-assisted laparoscopic radical prostatectomy. Eur Urol. 2008;53(1):134-145.
See W, Monk T, et al. Complications of laparoscopy. Laparosc Urol. 1993:183-206.
Seed R, Shakespeare T, et al. Carbon dioxide homeostasis during anesthesia for laparoscopy. Anesthesia. 1970;25:223-231.
Semm K. Morcellement and suturing using pelviscopy—not a problem any more. Geburtshilfe Frauenheilk. 1991;51:843-846. [in German]
Seymour NE, Gallagher AG, et al. Virtual reality training improves operating room performance: results of a randomized, double-blinded study. Ann Surg. 2002;236(4):458-463. discussion 463–4
Sezeur A, Benoit J, et al. [Elective cholecystectomy by celioscopy versus subcostal approach cholecystectomy. Comparative study of postoperative pain and discomfort.]. Gastroenterol Clin Biol. 1993;17:833-838. [in French]
Sharp J, Pierson W, et al. Comparison of CO2- and N2O-induced discomfort during peritoneoscopy under local anesthesia. Gastroenterology. 1982;82:453-456.
Shekarriz B, Upadhyay J, et al. Nerve-sparing retroperitoneal lymphadenectomy using hydro-jet dissection: initial experience. J Endourol. 2004;18(3):273-276.
Shifren J, Adlestein L, et al. Asystolic cardiac arrest: a rare complication of laparoscopy. Obstet Gynecol. 1992;79:840-841.
Shulman B, Aronson H. Capnography in the early diagnosis of carbon dioxide embolus during laparoscopy. Can Anaesth Soc J. 1984;31:455-459.
Simon SD, Castle EP, et al. Routine postoperative chest x-ray following laparoscopic nephrectomy. JSLS. 2005;9(2):205-207.
Simon SD, Castle EP, et al. Complications of laparoscopic nephrectomy: the Mayo Clinic experience. J Urol. 2004;171(4):1447-1450.
Siqueira TMJr, Paterson RF, et al. The use of blunt-tipped 12-mm trocars without fascial closure in laparoscopic live donor nephrectomy. JSLS. 2004;8(1):47-50.
Slim K, Bousquet J, et al. Effect of CO(2) gas warming on pain after laparoscopic surgery: a randomized double-blind controlled trial. Surg Endosc. 1999;13(11):1110-1114.
Society of American Gastrointestinal and Endoscopic Surgeons. Framework for post-residency surgical education & training. http://www.sages.org/publication/id/17/, 2003. [accessed 04.02.11]
Solis Herruzo J, Castellano G, et al. Plasma arginine vasopressin concentration during laparoscopy. Hepatogastroenterology. 1989;36:499-503.
Soong YK, Yu HT, et al. Urinary tract injury in laparoscopic-assisted vaginal hysterectomy. J Minim Invasive Gynecol. 2007;14(5):600-605.
Stern JA, Simon SD, et al. TissueLink device for laparoscopic nephron-sparing surgery. J Endourol. 2004;18(5):455-456.
Stifelman M, Nieder AM. Prospective comparison of hand-assisted laparoscopic devices. Urology. 2002;59(5):668-672.
Strate T, Bloechle C, et al. Hemostasis with the ultrasonically activated scalpel. Surg Endosc. 13(727), 1999.
Strup S, Garrett J, et al. Laparoscopic partial nephrectomy: hand-assisted technique. J Endourol. 2005;19(4):456-459. discussion 459–60
Sukhotnik I, Bejar J, et al. Adverse effects of increased intra-abdominal pressure on small bowel structure and bacterial translocation in the rat. J Laparoendosc Adv Surg Tech A. 2006;16(4):404-410.
Sundaram CP, Ono Y, et al. Hydrophilic guide wire technique to facilitate organ entrapment using a laparoscopic sack during laparoscopy. J Urol. 2002;167(3):1376-1377.
Sung GT, Gill IS. Laparoscopic adrenalectomy. Semin Laparosc Surg. 2000;7(3):211-222.
Suzuki K, Fujita K, et al. Efficacy of an ultrasonic surgical system for laparoscopic adrenalectomy. J Urol. 1995;154:484-486.
Swanstrom LL, Khajanchee Y, Abbas M. A. Natural orifice transluminal endoscopic surgery: the future of gastrointestinal surgery. Permanente J. 2008;12(2):42-47.
Swanstrom LL, Whiteford M, et al. Developing essential tools to enable transgastric surgery. Surg Endosc. 2008;22(3):600-604.
Takeda M, Go H, et al. Retroperitoneal laparoscopic adrenalectomy for functioning adrenal tumors: comparison with conventional transperitoneal adrenalectomy. J Urol. 1997;157:19-23.
Tasch M. Pulmonary aspiration. In: Atlee JL, editor. Complications in anesthesia. Philadelphia: Saunders; 1999:167-170.
Thomas MA, Rha KH, et al. Optical access trocar injuries in urological laparoscopic surgery. J Urol. 2003;170(1):61-63.
Thorell A, Nygren J, et al. The metabolic response to cholecystectomy: open versus laparoscopic surgery. Clin Nutr. 1993;12:1-2.
Thorington J, Schmidt C. A study of urinary output and blood-pressure changes resulting in experimental ascites. Am J Med Sci. 1923;165:880-890.
Tolksdorf W, Strang C, et al. The effects of the carbon dioxide pneumoperitoneum in laparoscopic cholecystectomy on postoperative spontaneous respiration. Anesthetist. 1992;41:199-203.
Tredree R, Debrix BWI, Eisert A, et al. Evaluating the differences between fibrin sealants: recommendations from an international advisory panel of hospital pharmacists. Eur J Hosp Pharm Sci. 2006;12(1):3-9.
Trokel M, Bessler M, et al. Preservation of immune response after laparoscopy. Surg Endosc. 1994;8:1385-1388.
Troxel SA, Das S. Incisional hernia following hand-assisted laparoscopic surgery for renal cell cancer. JSLS. 2005;9(2):196-198.
Truong H, Gamboa LMA, Lin R, et al. In vivo comparative study of vessel and ureteral seal strengths and pathologic examination of tissue margins between the LigaSure V and the Harmonic Ace in the acute porcine model. J Endourol. 22(Suppl.), 2008. Abstract 1584
Urban D, Kerbl K, et al. Organ entrapment and renal morcellation: permeability studies. J Urol. 1993;150:1792-1794.
Urena R, Mendez F, et al. Laparoscopic partial nephrectomy of solid renal masses without hilar clamping using a monopolar radio frequency device. J Urol. 2004;171(3):1054-1056.
Usal H, Sayad P, et al. Major vascular injuries during laparoscopic cholecystectomy: an institutional review of experience with 2589 procedures and literature review. Surg Endosc. 1998;12:960-962.
Vallancien G, Cathelineau X, Baumert H, et al. Complications of transperitoneal laparoscopic surgery in urology: review of 1,311 procedures at a single center. J Urol. 2002;168(1):23-26.
Varkarakis J, Bartsch G, et al. Long-term morbidity and mortality of transurethral prostatectomy: a 10-year follow-up. Prostate. 2004;58(3):248-251.
Watson R, Redmond H, et al. Exposure of the peritoneal cavity to air regulates early inflammatory responses to surgery in a murine model. Br J Surg. 1995;82:1060-1065.
Webb D, Redgrave N, et al. Extraperitoneal laparoscopy: early experience and evaluation. Aust N Z J Surg. 1993;63:554-557.
Weld KJ, Arzola J, Montiglio C, et al. Lapra-Ty holding strength and slippage with various suture types and sizes. Urology. 2008;71(1):32-35.
Williams M, Murr P. Laparoscopic insufflation of the abdomen depresses cardiopulmonary function. Surg Endosc. 1993;7:12-16.
Winfield H, Donovan J, et al. Urological laparoscopic surgery. J Urol. 1991;146:941-948.
Wolf JS. Tips and tricks for hand-assisted laparoscopy. AUA Update Ser. 2005;24(2):10-15.
Wolf JS, Marcovich R, et al. Survey of neuromuscular injuries to the patient and surgeon during urologic laparoscopic surgery. Urology. 2000;55(6):831-836.
Wolf JS, Monk T. Anesthetic considerations. St. Louis: Quality Medical Publishing; 1996.
Wolf JS, Monk TG, et al. The extraperitoneal approach and subcutaneous emphysema are associated with greater absorption of carbon dioxide during laparoscopic renal surgery. J Urol. 1995;154(3):959-963.
Wolf JS, Stoller M. The physiology of laparoscopy: basic principles, complications and other considerations. J Urol. 1994;152:294-302.
Zhao LC, Han JS, et al. Thoracic complications of urologic laparoscopy: correlation between radiographic findings and clinical manifestations. J Endourol. 2008;22(4):607-614.
Zorn KC, Gofrit ON, et al. Da Vinci robot error and failure rates: single institution experience on a single three-arm robot unit of more than 700 consecutive robot-assisted laparoscopic radical prostatectomies. J Endourol. 2007;21(11):1341-1344.
Zucker K, Martin D, et al. Complications of instrumentation and equipment. In: Bailey RW, editor. Complications of laparoscopic surgery. St. Louis: Quality Medical Publishing; 1995:58-74.