Selected Extrinsic Causes of Ureteral Obstruction
A plethora of intraperitoneal and extraperitoneal disease processes may ultimately result in ureteral obstruction. Hydronephrosis, either symptomatic or incidental, may thus be the initial presentation of these conditions. Herein, selected extrinsic causes of ureteral obstruction are reviewed.
Retroperitoneal Fibrosis
Retroperitoneal fibrosis is an uncommon condition in which a fibrotic and inflammatory mass envelops and potentially obstructs retroperitoneal structures, including either or both ureters. The initial report is now attributed to Albarran (1905), although idiopathic retroperitoneal fibrosis has had the eponym Ormond disease, after that author’s description and definition of this clinical entity in 1948 (Albarran, 1905; Ormond, 1948). Of the two patients Ormond initially described, one had progressive renal deterioration and ultimately died of the disease, whereas the other underwent exploration and ureterolysis without subsequent signs or symptoms of recurrent ureteral obstruction during the ensuing 12 years, underscoring the need to recognize and address this process (Ormond, 1960).
Grossly, retroperitoneal fibrosis appears as a fibrous, whitish plaque that encases the aorta, inferior vena cava, and their major branches, and also the ureters, other retroperitoneal structures, and, at times, intraperitoneal structures including the gastrointestinal tract. Its longitudinal axis usually extends from the renal hilum to the pelvic brim, but it may extend into the pelvis, mediastinum, and even the optic orbit. Histologic features include a fibrous component and a chronic inflammatory infiltrate composed of lymphocytes, macrophages, plasma cells, and eosinophils. The infiltrate is both perivascular and diffuse. The fibrotic component consists of myofibroblasts and type-1 collagen (Corradi et al, 2007).
The etiology of retroperitoneal fibrosis is not well characterized. A number of mechanisms have been proposed. One is based on the development of vasculitis in the adventitial vessels of the aorta and perioaortic small vessels. This may lead to the release of antigens from atheromatous plaque such as ceroid, a complex lipoprotein, which induces an autoimmune antigenic response. The latter could induce local inflammation resulting in varying degrees of fibrosis (Baker, 2003). Antibodies for fibroblasts have been isolated in retroperitoneal fibrosis (Vaglio et al, 2006). In-vitro studies have demonstrated that such antibodies stimulate fibroblasts that could lead to the propagation of localized fibrosis (Chizzolini et al, 2002). Other immunologic events may play a role. Both CD3+ and CD20+ lymphocytes and IgG4-positive plasma cells have been identified (Corradi et al, 2007). The presence of clonal and oligoclonal B cells suggest that an errant B-cell disorder may play a role. Environmental toxins may play a role as suggested by the positive association between asbestos exposure and the development of retroperitoneal fibrosis (Uibu et al, 2004).
Limited information is available on its epidemiology. The incidence in Finland was reported to be 0.1 per 100,000 person-years and the prevalence to be 1.38 per 100,000 inhabitants of this country (Uibu et al, 2004). The incidence has been estimated to be 1 in 200,000 in the Netherlands (Debruyne et al, 1982). It is more common in males, with a 2 to 3 : 1 male-to-female ratio. The mean age at onset is typically 50 to 60 years, but it may manifest in children as well as the elderly (Wu et al, 2002; Miller et al, 2003; Vaglio et al, 2006). Genetic influence is not thought to play a major role, because there have been no reports of familial clustering. Retroperitoneal fibrosis is associated with HLA-DRB1*03, an allele linked to a number of autoimmune diseases (Martorana et al, 2006).
Numerous medications have been associated with the development of retroperitoneal fibrosis and are listed in Table 40–2, along with other etiologies of this process. A definitive etiology of retroperitoneal fibrosis is found in only 30% of cases (Koep et al, 1977). The term “idiopathic retroperitoneal fibrosis” should be used only when an inciting etiology is not defined. An underlying malignancy should always be considered, because one is reported to be present in 8% to 10% of such cases (Amis, 1991). Unique infections can cause retroperitoneal fibrosis such as tuberculosis, histoplasmosis, and actinomycosis. It can occur after abdominal or pelvic surgery as well as abdominal trauma. Radiation and systemic chemotherapy have also been reported to induce it (Fassina et al, 2007; Vaglio et al, 2006).
Table 40–2 Suspected Causes of Retroperitoneal Fibrosis
A number of associations between retroperitoneal fibrosis and systemic diseases have been reported including autoimmune and inflammatory processes. These include autoimmune pancreatitis, autoimmune thyroid disease, small- and medium-sized vessel vasculitis, ankylosing spondylitis, rheumatoid arthritis, glomerulonephritis, sclerosing cholangitis, primary biliary cirrhosis, uveitis, psoriasis, and amyloidosis and Erdheim-Chester disease (Vaglio et al 2006; Loddenkemper et al, 2008).
Patients usually have nonspecific symptoms, which may include back, abdominal, or flank pain, weight loss, anorexia, and malaise. Signs can be similarly nonspecific and include hypertension (in 50%), fever, and lower extremity edema. Because of its insidious nature, many patients do not present until late in the disease course, manifesting with signs or symptoms attributable to ureteral or vascular obstruction (Amis, 1991). Reported laboratory abnormalities are nonspecific and include elevations in acute phase reactants, such as erythrocyte sedimentation rate and C-reactive protein (in 80% to 100%), elevated serum creatinine, hypergammaglobulinemia, and normochromic, normocytic anemia (Monev, 2002; Vaglio et al, 2006). Tests for autoimmune diseases may also be positive, such as those for antinuclear antibodies, rheumatoid factor, and antibodies against smooth muscle, double-stranded DNA, extractable nuclear antigen and neutrophil cytoplasm (Vaglio et al, 2006).
Before the advent of cross-sectional imaging, radiologic diagnosis relied upon imaging not the disease process itself but its secondary effects on the ureters. The classic radiologic findings included medial deviation of extrinsically compressed ureters with hydronephrosis. Medial deviation is a nonspecific finding, because it has been demonstrated in up to 18% of normal subjects (Saldino and Palubinskas, 1972). Hydronephrosis is typically demonstrated on ultrasonography, and there may also be a smooth, well-demarcated, hypoechoic or isoechoic mass anterior to the lumbar or sacral spine (Amis, 1991). Retrograde pyelography typically demonstrates hydronephrosis, with medially deviated and segmentally narrowed ureters without filling defects (Fig. 40–10). Currently, cross-sectional imaging with CT or MRI is the modality of choice for evaluating patients with suspected retroperitoneal fibrosis. Both modalities have been demonstrated to delineate accurately the extent of the disease when correlated with surgical exploration (Mulligan et al, 1989). CT typically reveals a well-demarcated retroperitoneal mass, isodense with muscle on unenhanced studies (Fig. 40–11) (Vivas et al, 2000). It typically surrounds the lower abdominal aorta, inferior vena cava, and iliac vessels—frequently enveloping the ureters. In contrast, masses secondary to malignancy usually displace the aorta anteriorly and the ureters laterally. Variable degrees of contrast enhancement may be seen, typically more pronounced earlier in the course of the disease when the presumably more-immature plaque has increased vascularity (Amis, 1991). Some have advocated noncontrast multidetector CT imaging combined with antegrade CT pyelography for evaluation, because it can define the fibrotic process and the level and degree of ureteral obstruction without administration of intravenous contrast material (Ghersin et al, 2004). However, the invasive nature of this diagnostic approach limits its applicability.
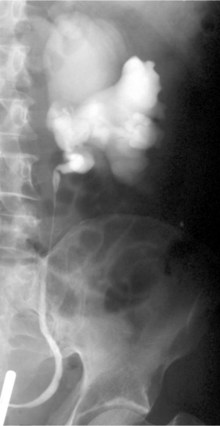
Figure 40–10 Retrograde pyelogram performed in a patient with retroperitoneal fibrosis. Note evenly narrowed midureteral segment with proximal hydronephrosis.
(Courtesy of Dr. Ronald Zagoria.)
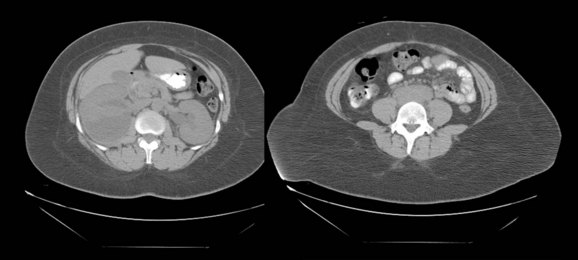
Figure 40–11 Unenhanced computed tomography performed in a patient with retroperitoneal fibrosis. Note hydronephrosis and soft tissue mass indistinguishable from the great vessels.
(Courtesy of Dr. Ronald Zagoria.)
MRI offers cross-sectional and multiplanar evaluation of the retroperitoneal fibrosis plaque (Fig. 40–12). Theoretically, MRI allows superior soft tissue discrimination and can more accurately distinguish the plaque from the great vessels than unenhanced CT. In addition, the disease can be followed without the radiation exposure of repeated CT (Burn et al, 2002). Fat saturation images are recommended to permit optimal discrimination from surrounding tissues (Elsayes et al, 2007; Cronin et al, 2008). Typically, T1-weighted images of the lesion reveal low to medium signal intensity (hypointense compared with muscle); T2-weighted imaging is variable. Low T2 signal intensity has been correlated with mature plaque (Mulligan et al, 1989). High signal intensity on T2-weighted images suggests increased water content and hypercellularity and may be due to inflammation associated with retroperitoneal fibrosis. However, the latter may be due to an underlying malignant process (Amis, 1991; Burn et al, 2002). A low intensity signal is present if the majority of the plaque is composed of fibrosis. With gadolinium administration, variable enhancement of the plaque may be seen, with a greater degree of enhancement in the more acute phases of the disease process. Enhancement ratios can be calculated with dynamic gadolinium enhancement, and investigators have used these ratios to assess disease activity and monitor treatment responses (Burn et al, 2002). 18F-fluorodeoxyglucose positron emission tomography (F-18 FDG PET) is an imaging modality that detects hypermetabolic activity. It may depict the degree of inflammation in the plaque, which may predict response to immunosuppressive therapy as well as its therapeutic response (Vaglio et al, 2006; Young et al, 2008). The utility of F-18 FDG PET for management of retroperitoneal fibrosis is not yet defined; thus, further studies are warranted. Gallium-67 single photon emission–computed tomography (SPECT) scintigraphy has been used for evaluating responses to medical therapy of retroperitoneal fibrosis and for detection of recurrent disease. It demonstrates both the mass lesion and its inflammatory component (van Bommel et al, 2007).
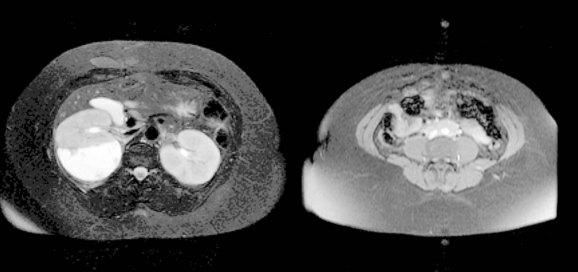
Figure 40–12 Unenhanced magnetic resonance imaging from the same patient as in Figure 40–9.
(Courtesy of Dr. Ronald Zagoria.)
If there is evidence of obstructive uropathy at presentation, therapy should be first directed at its correction. This may be accomplished with internalized ureteral stents, but percutaneous nephrostomy may be necessary if stenting is not possible or ineffective. Biopsy to exclude malignancy should be performed next. This can be attempted percutaneously with CT, MRI, or ultrasound guidance. Core tissue biopsy (histologic confirmation) is preferred over fine needle aspiration cytologic assessment. Although some have reported the latter to be an effective technique, others have noted pitfalls (Stein et al, 1997; Dash et al, 1999; Jogai, 2005). If this is not technically possible, or the tissue obtained is inadequate, an open surgical or laparoscopic biopsy is recommended with concomitant ureterolysis if malignancy is not suspected on frozen-section analysis. If the percutaneous biopsy demonstrates findings compatible with retroperitoneal fibrosis, possible inciting medications should be discontinued.
The potential morbidity of ureterolysis, accurate image-guided biopsy techniques, and more sensitive cross-sectional imaging studies for diagnosis and follow-up have made medical therapy the preferred approach in this setting. However, notably, there have been no randomized prospective studies addressing the merits of initial medical therapy versus a surgical approach. Patients should be informed about the side effects of the proposed medical therapy and the risk of disease progression or undiagnosed malignancy. Medical therapy has been directed to address the presumed autoimmune and inflammatory components of retroperitoneal fibrosis.
The administration of corticosteroids has been used for primary therapy. Although many steroid regimens have been described, appropriate dosing schedules and duration of therapy have not been clearly established. The sequence suggested by Kardar and colleagues (2002) has been demonstrated to be effective after long-term follow-up. Their algorithm for adults consists of prednisolone administered at an oral dose of 60 mg on alternate days for 2 months, tapered to 5 mg daily over the next 2 months. The latter dose is continued for a total of 2 years. Of the 11 patients who completed this regimen, at a median follow-up of 63 months, 9 maintained a good response, 1 required ureterolysis, and 1 recurred but responded to reinitiation of therapy. van Bommel and associates (2007) treated 24 patients with prednisone for 1 year. They were initially treated with 60 mg per day for 6 weeks and tapered to 10 mg over the next 2 to 3 months, a dosage which was maintained for the remainder of the year. Six subjects (25%) had no response to the therapy, while 12 of 16 responders (67%) had relapses, with the majority responding to reinitiation of prednisone or alternative medical therapy at a median follow-up of 55 months. Fry and colleagues (2008) treated 27 patients with 30 to 40 mg of prednisolone per day for a mean of 34 months. Ureteral stents were typically removed at 12 months. Twenty-four (89%) initially responded, and 6 recurred at a mean of 15 months after steroid withdrawal.
Tamoxifen, a nonsteroidal antiestrogen, has also been used for primary treatment. It is thought to alter TGF-β, potentially limiting fibrosis. van Bommel and associates (2007) treated 19 patients with 20 mg of tamoxifen per day. The median duration of treatment was 8.5 months (range 5.5 to 15 months). Fourteen responded to therapy (74%) and 5 failed (26%). Successful combination therapy using a tapered steroid regimen and daily tamoxifen has also been described (Tziomalos et al, 2004).
Other treatments have been used as primary or salvage therapy for those afflicted with retroperitoneal fibrosis. Marcolongo and colleagues (2004) reported 26 patients who were treated with a 6-month course of prednisone, combined with either 6 months of cyclophosphamide or 1 year of azathioprine. Severe adverse effects in the cyclophosphamide group included death in one, sepsis in one, and severe leukopenia in two patients. In the azathioprine group, activation of hepatitis, pancreatitis, and oral candidiasis developed. All surviving patients had disease resolution, although seven required a second course of treatment. Swartz and associates (2008) treated 16 patients with idiopathic retroperitoneal fibrosis with a 3- to 6-month course of prednisone in conjunction with mycophenolate mofetil for approximately 2 years. One half of these patients were subjected to ureterolysis or ureteral reconstruction, while the others just underwent ureteral stenting. All had resolution or improvement of symptoms and were rendered stent free. Other groups have reported success with this regimen. Adler and colleagues (2008) treated 9 patients having histologically confirmed retroperitoneal fibrosis with these same two drugs for 6 months. All had radiographic regression of the retroperitoneal fibrosis, and 7 were rendered stent free. Scheel and associates reported on 7 patients receiving a similar regimen (Scheel et al, 2007). Six of 7 had radiographic regression, and 10 of 11 with obstructed renal units became stent free. Vega and colleagues (2008) administered colchicine and prednisone or deflazacort (steroid preparation) to 7 patients and reported that the majority experienced a regression in their disease. The coadministration of steroids in all of these series does not permit the determination of the efficacy of these various respective agents.
Ureterolysis is undertaken if medical therapy fails or if the patient is not a candidate for medical therapy. Traditionally, this has been done by an open surgical approach, but successful laparoscopic and robotic ureterolysis have been reported and may be feasible options (Duchene et al 2007; Stifelman et al, 2008). Bilateral ureterolysis is recommended, even in the setting of unilateral disease, as the disease typically progresses to involve the contralateral side. The ureteral dissection should begin in a region free of disease, and care must be taken to avoid devascularization of the ureter. A biopsy of the fibrotic area should be repeated to limit the risk of sampling error associated with percutaneous biopsy. Several strategies have been employed to prevent recurrent ureteral involvement in the fibrotic process. The ureter may be displaced to a lateral position, brought to an intraperitoneal location by closing the peritoneum behind it, or wrapped within a sleeve of omentum. No single method has been definitively demonstrated to generate superior results (Barbalias and Liatsikos, 1999). If it is determined intraoperatively that ureterolysis is not technically possible, alternatives include ileal ureter replacement, autotransplantation, chronic ureteral stenting, or nephrostomy tube drainage.
Ureteral stents can generally be removed 6 to 8 weeks after ureterolysis (Varkarakis and Jarrett, 2005). Some have advocated postoperative medical therapy to expedite disease resolution or prevent recurrence (Varkarakis and Jarrett, 2005). Others have not found differences in outcomes for those who receive versus those who are not administered adjuvant therapy (Duchene et al, 2007).
Reported success rates for small series involving ureterolysis are 66% to 100%. The best success rate was reported by Elashry and colleagues (1996): 100% for seven laparoscopic and 100% in six open surgical procedures. Duchene and associates (2007) subjected 31 patients to laparoscopic or robotic ureterolysis and reported that this was successful in 38 of 46 obstructed renal units (83%). Alexopoulos and associates (1987), however, noted only 66% long-term success with ureterolysis combined with postoperative steroid administration in six patients. Of note, one of their patients failed 16 years after surgery and became dialysis dependent. Long-term follow-up in those subjected to medical or surgical therapy is warranted. Serial cross-sectional imaging should be obtained to assess for disease recurrence, progression, or findings suggestive of malignancy. Renal function should also be monitored.
Pelvic Lipomatosis
Pelvic lipomatosis is a rare, benign condition marked by exuberant pelvic overgrowth of nonmalignant but infiltrative adipose tissue, usually in the abdominal and pelvic cavities. The fat deposition generally occurs in the perivesical and perirectal spaces. Engels first described the condition in 1959, and it has been the subject of several reviews and many case reports in the interim. The largest review to date comprised 130 patients, whose mean age at presentation was 48 years. Racial and gender differences were noted, with 67% of the patients black and 33% white; there was an 18 : 1 male-to-female ratio (Heyns, 1991).
The etiology and incidence of this disorder are unknown. Obesity has been proposed to play a role, because radiographic improvement and worsening have been noted in response to weight loss and gain, respectively (Sacks et al, 1975). In addition, obesity has been noted in more than one half of the patients afflicted by pelvic lipomatosis (Heyns, 1991). However, if pelvic lipomatosis were simply a manifestation of obesity, its observed frequency should be far greater (Morretin and Wilson, 1971). The reported occurrence of pelvic lipomatosis in two brothers raises the possibility of an underlying genetic etiology (Tong et al, 2002). An abnormality in the chromatin-regulating high-mobility group A (HMGA) proteins has been implicated as a possible causative factor in pelvic lipomatosis, because transgenic mice with truncated HMGA developed pelvic lipomatosis (Fedele et al, 2001). Possible etiological genetic mutations have been traced to the HMGA2 gene located on the hypermobile cluster on chromosome 12 (Fedele et al, 2001). It is believed that pelvic lipomatosis may be a chronic inflammatory response triggered by urinary tract infection.
Approximately one half of patients present with lower urinary tract symptoms and one quarter with bowel symptoms, typically constipation. Suprapubic, back, flank, or perineal discomfort can also be an initial clinical manifestation. The nonspecific nature of these symptoms may result in a significant delay in diagnosis (Heyns, 1991). Physical findings may include a suprapubic mass, a high-riding prostate, and an indistinct pelvic mass. Hypertension has been reported in as many as one third of patients (Klein et al 1988; Heyns, 1991).
There are various radiologic signs of pelvic lipomatosis. On plain film, increased pelvic lucency may be noted (Fig. 40–13). On excretory urography, the bladder characteristically assumes a pear or gourd shape, extrinsically compressed and elongated, and the bladder base is frequently elevated (Fig. 40–14). At times, proliferation of the mature unencapsulated fat in the pelvis and retroperitoneum leads to severe bilateral hydronephrosis. In contrast to retroperitoneal fibrosis, which usually affects the proximal and midureters, pelvic lipomatosis causes significant displacement of the distal ureters medially (Lin et al, 2007). Hydroureteronephrosis may be seen (Heyns, 1991). CT is quite helpful in establishing the diagnosis, because it readily demonstrates pelvic fat (Fig. 40–15). Extrinsic compression of the rectum may also be demonstrated (Susmano and Dolin, 1979). CT may help in differentiating pelvic lipomatosis from other radiologically similar pelvic mass lesions such as lipomas, lipoplastic lymphadenopathy, and liposarcoma. Liposarcoma should be suspected if there is tissue heterogeneity, areas of positive attenuation coefficients, enhancement with contrast, and poor margination (Andac et al, 2003). MRI can also be used to make the diagnosis, because it permits characterization of fat deposits (Demas et al, 1988).
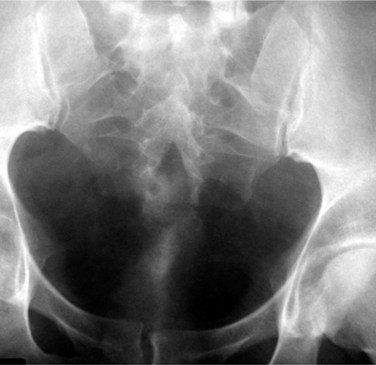
Figure 40–13 Plain radiograph of pelvis in a patient with pelvic lipomatosis. Note increased pelvic lucency.
(Courtesy of Dr. Ronald Zagoria.)
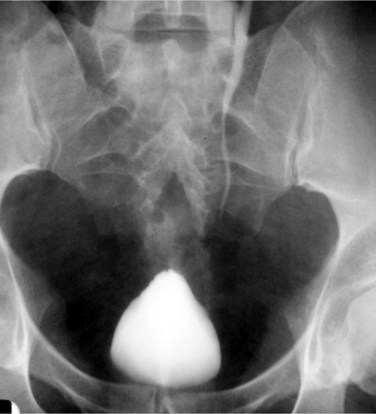
Figure 40–14 Excretory urography of a patient with pelvic lipomatosis. Note classic pear-shaped bladder with increased surrounding pelvic lucency.
(Courtesy of Dr. Ronald Zagoria.)
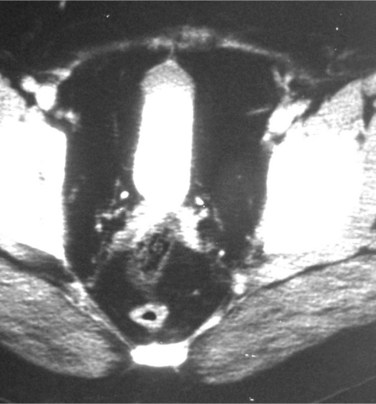
Figure 40–15 Computed tomography cystogram of a patient with pelvic lipomatosis. Note narrowed bladder and increased pelvic fat in perivesical and perirectal spaces.
(Courtesy of Dr. Ronald Zagoria.)
Evaluation should include cystoscopy, because some form of proliferative cystitis has been found in 75% of patients, including cystitis glandularis in up to 40% (Heyns, 1991). However, the majority of patients with cystitis glandularis do not have pelvic lipomatosis (Smith et al, 2008). The development of cystitis glandularis in this setting has been hypothesized to be due to accumulation of perivesical fat, causing compression of the bladder that leads to venous stasis, and the development of an edematous urothelium that subsequently sloughs off. As the urothelium regenerates, it may undergo a metaplastic change leading to development of cystitis glandularis. Continued cystoscopic surveillance is recommended in those with cystitis glandularis, because the development of bladder adenocarcinoma has been reported in this cohort (Sozen et al, 2004). Elongation of the prostatic urethra, elevation of the bladder neck, and pelvic fixation may impair cystoscopic access to the bladder. In Heyns’ review (1991) of 72 patients in whom cystoscopy was attempted, evaluation of the bladder was difficult in 24% and impossible in 18%. Hence, flexible cystoscopy may be required if anatomic distortion precludes rigid cystoscopy.
The clinical signs and symptoms include lower urinary tract symptoms, hematuria, passage of debris in the urine, and problems with defecation. Klein and associates (1988) suggested that there are two clinically separate groups of patients. The first is composed of young, stocky men with irritative lower urinary tract symptoms, vague pelvic complaints, hypertension, and proliferative cystitis. This group may be more susceptible to developing progressive ureteral obstruction. Pelvic lipomatosis has been found to be associated with progressive uremia in up to 40% of patients observed for five years from the time of initial diagnosis (Sozen et al, 2004). The second group includes older men with incidentally discovered pelvic lipomatosis who are reported to have a more indolent course. Others have not found age to affect disease progression (Heyns, 1991). The accumulation of fatty pelvic tissue causes a mass effect that is known to compress both pelvic as well as intra-abdominal organs, causing obstruction of the ureters, rectum and sigmoid colon, the pelvic veins, and even the inferior vena cava. Nevertheless, there is agreement that in light of the potential for progressive ureteral obstruction, long-term follow-up is mandated. Overall, 39% of patients in Klein’s series ultimately required operative intervention for upper tract obstruction.
Treatments with steroids, chemotherapy, radiotherapy, and chronic antibiotic therapy have not been reported to be successful. Surgical options in patients with obstructive uropathy secondary to pelvic lipomatosis include ureteral stenting, percutaneous nephrostomy, ureteral reimplantation, and urinary diversion with or without cystoprostatectomy (Sozen et al, 2004). Surgical intervention for removal of the pelvic lipomatous tissue is often limited, due to extensive deposits of fat, and their removal does not always lead to an improvement. Pelvic exploration should be approached cautiously, because there is obliteration of normal anatomic planes and increased vascularity within the fatty mass.
Obstetric and Gynecologic
Pregnancy
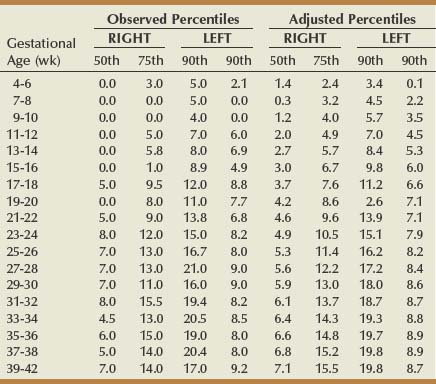
Hydronephrosis develops commonly during pregnancy, the reported occurrence varying between 43% and 100% (Faundes et al, 1998). This wide variability is due to the use of different definitions of hydronephrosis. The right kidney becomes hydronephrotic two to three times more commonly than the left (Cietak and Newton 1985; Faundes et al, 1998). Hydronephrosis may initially manifest in the first trimester, and it increasingly develops throughout pregnancy (Au et al, 1985; Loughlin, 1997). This progression was also reported in a large prospective study by Faundes and colleagues (1998), who found that hydronephrosis was present in 15%, 20%, and 50% of women during the first, second, and third trimesters, respectively. The degree of hydronephrosis also increased over time. Hydronephrosis has been demonstrated to be more common during a first-time pregnancy (Erickson et al, 1979). The maximal calyceal diameters during pregnancy were measured over time by Faundes and associates (1998). These parameters are provided (Table 40–3), because they may be useful when evaluating patients with hydronephrosis during pregnancy. After delivery, hydronephrosis gradually resolves. Approximately one third of patients may have persistent hydronephrosis during the first postpartum week, but it resolves in the majority within 6 weeks (Cietak and Newton, 1985).
Table 40–3 Observed and Adjusted Percentiles of Maximal Maternal Calyceal Diameter (mm) of Right and Left Kidneys According to Gestational Age
Two etiologies for hydronephrosis of pregnancy have been postulated, one hormonal and one mechanical. The 10% to 15% incidence of observed first-trimester ureteral dilatation, occurring before the uterus reaches the pelvic brim, supports a nonmechanical mechanism. Progesterone, a hormone in more abundance during pregnancy, has been hypothesized to promote ureteral dilatation and subsequent development of hydronephrosis (Faundes et al, 1988). The increased incidence and degree of hydronephrosis after the 20th week of gestation, a time when the uterus is large enough to compress the ureters extrinsically, supports a mechanical etiology. The significantly lower incidence of left-sided hydronephrosis also supports this hypothesis (Cietak and Newton, 1985). However, it is quite plausible that both mechanisms play a role.
Hydronephrosis of pregnancy is usually clinically silent. However, some with hydronephrosis of pregnancy may experience flank pain or develop pyelonephritis or renal failure. Renal ultrasonography should be undertaken in this setting. The typical ultrasonographic findings of this entity are hydroureteronephrosis extending to the pelvic brim. When ureteral dilation extends below this, a different etiology of obstruction, such as a ureteral stone, should be considered. If the latter is suspected, limited intravenous urography and low-energy CT are diagnostic options (White et al, 2007). However, MRI is now another consideration. Magnetic resonance urography has been advocated by some as a noninvasive, nonradiating method of assessing those with hydronephrosis of pregnancy. The presence of hydroureter below the pelvic brim is readily apparent on high-resolution T2 sequences, and filling defects representing stones or soft tissue may be seen (Spencer et al, 2004). In addition, MRI has proved useful in identifying other causes of acute abdominal pain in pregnancy, including appendicitis, ovarian torsion, and adrenal hemorrhage (Birchard et al, 2005). There is no current scientific evidence of definite adverse effects on the fetus or pregnancy due to the electromagnetic fields. MRI is currently viewed as a useful imaging technique for evaluating select pregnant patients with abdominal, flank, or pelvic pain (Fielding et al, 2006; Kanal et al, 2006). However, despite the lack of any proven adverse effects, the use of MRI is not advocated in the first trimester of pregnancy (Leyendecker et al, 2004). In addition, the administration of gadolinium contrast should be avoided in pregnant patients because it is known to cross the placental barrier (Shellock et al, 1999).
The majority of patients with symptomatic hydronephrosis of pregnancy can be managed with conservative measures, such as intravenous hydration, analgesics, and antibiotic therapy when clinically indicated. However, placement of ureteral stents may be necessary when these measures are unsuccessful, especially with signs of sepsis or compromised renal function. Rapid stent encrustation may be problematic, as urinary calcium excretion increases during pregnancy (Goldfarb et al, 1989). This may necessitate more frequent stent changes. Percutaneous nephrostomy is an alternative if this occurs, and it is also a consideration if pyonephrosis is suspected.
Benign Pelvic Abnormalities
Tubo-ovarian Abscess
Pelvic inflammatory disease (PID) is a common problem, affecting 10% of women during their lifetime. Tubo-ovarian abscess, occurring in approximately 15% of those afflicted with PID, may cause extrinsic ureteral obstruction, which may even lead to anuria (Aral et al, 1991; Conde Santos et al, 2003). This process may be demonstrated with ultrasonography, CT, or MRI. Ureteral obstruction may resolve with antibiotic therapy or transvaginal drainage of the abscess. However, an open surgical or laparoscopic intervention may be required to drain or excise the involved ovary and fallopian tube if the aforementioned measures are unsuccessful. Placement of an internalized ureteral stent or percutaneous nephrostomy should be considered if high-grade renal obstruction or urosepsis is suspected. Stent or nephrostomy tube removal is undertaken after the inflammatory process has resolved. Follow-up renal imaging is recommended, because some patients have persistent obstruction.
Endometriosis
Endometriosis is defined as the presence of functional endometrial tissue in an ectopic site. Although endometriosis is usually confined to the ovaries, uterosacral ligaments, and cul-de-sac, it has been documented in almost every organ system in the body (Jubanyik and Comite, 1997). Endometriosis has the potential for malignant transformation (Takeuchi et al, 2006). It may affect 10% to 20% of women of reproductive age, with a peak incidence in the mid-20s (Eskenazi and Warner, 1997; Vigano et al, 2004; Berkley et al, 2005). Rarely, endometriosis of the urinary tract has been reported to occur in postmenopausal women (Maeda et al, 2009). There is no relationship of endometriosis with race or socioeconomic status.
Genitourinary endometriosis is a rare condition (Antonelli et al, 2006a, 2006b), with involvement of the urinary tract in 1% to 5% of those afflicted with this disorder. Endometriosis of the urinary tract is predominantly found in the bladder, accounting for 70% to 80% of the cases. The ureter may be involved in 15% to 20% of the urinary tract cases (Williams, 1975; Giudice et al, 2004). Rarely, renal and urethral involvement may occur (Comiter, 2002; Giudice et al, 2004). Involvement may be either intrinsic or extrinsic. Intrinsic endometriosis is characterized by endometrial glands and stroma within the lamina propria, tunica muscularis, or ureteral lumen; extrinsic endometriosis is localized within periureteral tissue. Eighty percent of ureteral endometriosis is extrinsic and primarily involves the distal ureter (Klein and Cattolica, 1979). The left side is more often affected, and bilateral disease has been reported in up to 23% of cases (Antonelli et al, 2004). One theory for ureteral involvement is the occurrence of retrograde menstruation and implantation of endometrial cells. This is thought to occur more commonly on the left side due to relationships with the sigmoid colon and ipsilateral ovary and anatomic differences between the right and left side of the pelvis (Vercellini et al, 2000). Vesical endometriosis is usually solitary and typically involves the dome of the bladder (Schneider et al, 2006).
Classic symptoms and signs of urinary tract endometriosis include cyclical flank pain, dysuria, urgency, urinary tract infection, and hematuria (Jubanyik and Comite, 1997). Cyclical lower urinary tract symptoms, abdominal and pelvic pain are present in the majority of patients with bladder involvement. However, as many as one third are asymptomatic (Westney et al 2000; Schneider et al, 2006). Patients with intrinsic endometriosis experience these symptoms more than those with extrinsic disease. Notably, a significant portion of patients with ureteral endometriosis do not have genitourinary symptoms. For example, in one series, ureteral obstruction was silent in approximately 40% of those with severe ureteral endometriosis (Antonelli et al, 2004). Furthermore, silent loss of renal function has been reported in 25% to 43% of patients with ureteral endometriosis, which may result in total loss of function of the affected kidney (Watanabe et al, 2004). Historically, up to one third of kidneys affected by ureteral endometriosis were lost (Klein and Cattolica, 1979).
Because a large percentage of patients with ureteral endometriosis can have asymptomatic obstruction of the kidney with a loss of renal function, it has been recommended to image the upper urinary tract in all patients with pelvic endometriosis. An endoscopic endometriosis evaluation scale has been generated to quantitate the extent and depth of involvement. There is a positive correlation with this scale and genitourinary involvement (Giudice et al, 2004). Initial imaging may be done with ultrasonography as a noninvasive test to look for hydroureteronephrosis. An EXU may be a better test when the index of suspicion for ureteral involvement is high. Pyelographic findings of intrinsic disease include ureteral filling defects, whereas smooth strictured ureteral segments are seen with extrinsic disease (Stebbing and Notley, 1995). Retrograde ureteropyelography, CT, and MRI may help to ascertain the exact location and volume of the disease and may provide valuable information for planning treatment. On MRI the most frequent sign of ureteral endometriosis is the detection of a hypointense nodule along with hyperintense foci adjacent to the ureter on T2- and T1-weighted images (Balleyguier et al, 2004). MRI may be particularly useful for the detection of ureteric and/or bladder deep pelvic endometriosis (Kinkel et al, 2006; Marcelli et al, 2006). Cystoscopy and possible biopsy should be done in women with pelvic endometriosis, presenting with lower urinary tract symptoms, and in those with hematuria. Ureteroscopy and possible biopsy should be undertaken when upper urinary tract involvement is suspected (Comiter, 2002). Immunostaining of the biopsied tissue for ER, PR, CK7, CA125, and CD10 may help establish the diagnosis (Al-Khwaja et al, 2008).
Treatment has several goals: (1) preservation of renal function, with long-term relief of urinary obstruction, (2) management of the ongoing disease process by completely excising or ablating the endometriotic tissue with its reactive fibrotic component, (3) maintenance of the patient’s fertility, and (4) relief of the patient’s symptoms with the least invasive methods available. Treatment should be individualized according to age, reproductive desire, symptom severity, extent of lesion, and organ(s) involved (Pastor-Navarro et al, 2007).
If renal function is normal and there is minimal to mild hydronephrosis with no functional obstruction as determined by radionuclide renal scanning, hormone therapy may be prescribed. Ovarian hormonal ablation with gonadotropin-releasing hormone agonists has been used with success in some series. Rivlin and associates (1990) described three patients treated with leuprolide acetate for 6 to 9 months. One patient had BUO, and two had UUO. The obstruction resolved in the patient with BUO and in one of the patients with UUO. The failure occurred in the patient with intrinsic ureteral disease.
Hormonal therapy is generally not as effective for patients with extensive endometriosis, as reflected by its high treatment failure and recurrence rates (Lam et al, 1992). Postmenopausal patients may be reasonably controlled with luteinizing hormone–releasing hormone (LHRH) analogues.
Surgical intervention is the treatment of choice for most patients with significant hydroureteronephrosis and periureteral disease. If the patient does not desire future pregnancy, treatment is total abdominal hysterectomy and bilateral salpingo-oophorectomy. If pregnancy is desired, lesser extirpative procedures can be performed, such as unilateral oophorectomy. Ureterolysis may correct ureteral obstruction in those with extrinsic disease. If laparoscopic ureterolysis is undertaken, a transperitoneal approach is preferable in that it allows a superior assessment of endometrial implants on the peritoneum overlying the ureter (Watanabe et al, 2004; Frenna et al, 2007). Success rates of 85% have been reported for this approach (Ghezzi et al, 2006). If patients are suspected to have intraluminal ureteral involvement, ureteroscopic resection or ablation is a consideration (Ghezzi et al, 2007). However, if these procedures are unsuccessful or extensive disease is present, distal ureterectomy with reimplantation should be performed. This can be done by an open surgical, laparoscopic- or robotic-assisted approach. This typically results in excellent long-term results with regard to renal preservation (Antonelli et al, 2004; Antonelli et al, 2006a, 2006b). The surgical treatment of those afflicted with vesical endometriosis is dependent on the extent and depth of the disease. Those with superficial involvement can be treated with transurethral resection (Fedele et al, 2005; Sanchez et al, 2005). Transurethral resection is not an optimal treatment for deep vesical endometriosis, because it frequently recurs (Le Tohic et al, 2009). Patients with invasive disease are best treated with open surgical, robotic, or laparoscopic partial cystectomy (Sanchez et al, 2005; Fedele et al, 2005; Granese et al, 2008). A recent long-term follow-up study focusing on the laparoscopic management of vesical and ureteric endometriosis has demonstrated a significant reduction in preoperative symptoms without relapse (Seracchioli et al, 2010).
Ureteral stenting is indicated prior to pelvic dissection for posterior vesical endometriotic nodules and prior to partial cystectomy (Marcelli et al, 2006).
Surveillance with sonography is advisable to detect recurrent ureteral obstruction in patients with ureteral endometriosis treated by hormones, endourological ablation, or surgical extirpation (Generao et al, 2005).
Ovarian Remnants
The ovarian remnant syndrome results from residual, viable ovarian tissue after bilateral salpingo-oophorectomy. Although uncommon, it is a recognized complication of a technically difficult oophorectomy, most typically in those with endometriosis, PID, or prior abdominal and pelvic surgery (Lafferty et al, 1996; Nezhat et al, 2005). Ovarian remnants may be quite large, up to 10 cm, and may be present in the normal ovarian bed but may be located in other areas as a consequence of dissemination and implantation occurring during surgery (Lafferty et al, 1996). Mass effect or a localized fibrosis may cause extrinsic ureteral obstruction. This process may be demonstrated with ultrasonography, CT, or MRI (Klutke et al, 1993).
Treatment is usually surgical excision of the mass and ureterolysis. Preoperative ureteral stenting may be considered if there is high-grade obstruction or as an aid to intraoperative identification of the ureter, although the latter has not been reported to be necessary by some investigators (Lafferty et al, 1996). In a series of 64 patients with ovarian remnant syndrome managed surgically, the majority of the ovarian remnants were found to be attached to the ureter, bowel, pelvic side wall, bladder, and uterosacral ligaments (Nezhat et al, 2005).
Medical management of ureteral obstruction is an option for poor surgical candidates. Successful resolution of the mass and hydronephrosis have been reported with leuprolide acetate therapy. However, chronic medical therapy may be required to prevent recurrence (Koch et al, 1994).
Mass Lesions of the Uterus and Ovaries
Other benign lesions of the uterus and ovary may become large enough to cause extrinsic ureteral obstruction. These include uterine fibroids, ovarian cysts, and ovarian fibromas. Uterine fibroids, the most common tumor of the upper female genital tract, are also the most common, benign gynecologic neoplasm causing ureteral obstruction (Greenberg and Kazamel, 1995). The most common site of extrinsic ureteral obstruction is at the level of the pelvic brim. Surgical resection or ablation of the leiomyomas should be considered in this setting.
Vascular Causes of Ureteral Obstruction
The ureters course adjacent to vascular structures and, as a consequence, may be affected by various vascular disease processes and anomalies that may result in their obstruction. An overview of several of these entities is subsequently provided.
Arterial Causes of Obstruction
Abdominal Aortic Aneurysm
Clinical signs and symptoms of ureteral obstruction may be the initial manifestation of an abdominal aortic aneurysm (AAA) (Labardini and Ratliff, 1967). The obstruction may be due to a mass effect or localized inflammation (Pennell et al, 1985).
Currently, CT scanning is most frequently used to evaluate patients with an AAA that may demonstrate associated hydronephrosis (Fig. 40–16A and B). Ureteral findings associated with AAA vary. The ureters may be pushed laterally on both sides, or one side may deviate laterally while the opposite side is drawn medially within the perianeurysmal inflammation, or both may be medially deviated. Some have noted that lateral deviation of the ureter with AAA is usually not associated with obstruction (Labardini and Ratliff, 1967). The medial deviation of the ureter associated with the desmoplastic reaction of inflammatory aneurysms (IAAA), however, carries a more significant risk of ureteral obstruction. Although IAAA represents only between 4% and 15% of AAAs, the associated retroperitoneal desmoplastic reaction significantly increases the risk of ureteral obstruction, which has been reported in 21% of this cohort (Pennell et al, 1985; Arroyo et al, 2003).
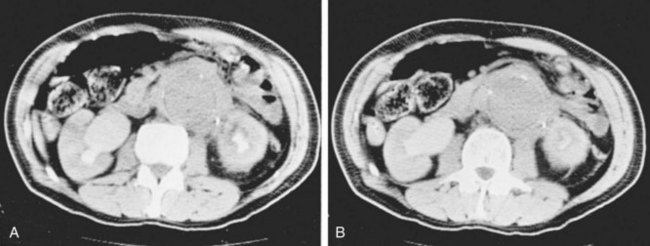
Figure 40–16 A and B, Computed tomography scans demonstrate extensive retroperitoneal fibrosis associated with aneurysm of the abdominal aorta.
At one time, ureterolysis was routinely performed during repair of IAAA associated with ureteral obstruction. However, this practice has generally been abandoned, because obstruction commonly resolves without ureterolysis. Placement of internalized ureteral stents is commonly undertaken prior to IAAA repair. Ureteral obstruction has been reported to resolve in 80% of patients managed with this approach (Arroyo et al, 2003). Some have advocated endovascular repair of the IAAA in this setting (Nevelsteen et al, 1999). Internalized ureteral stents are inserted prior to endovascular graft placement and are removed when it is thought that the associated fibrosis has resolved. A 50% resolution of ureteral obstruction has been reported with this approach, while less favorable results have been demonstrated by others (Rehring et al, 2001; Deleersnijder et al, 2002; van Bommel et al, 2008). In a systematic review of endovascular repair of IAAA it was associated with persistent periaortic fibrosis leading to significant ureteral obstruction. Patients with IAAA treated with open surgery or an endovascular approach warrant close monitoring for development of recurrent or de novo ureteral obstruction, because delayed ureteral obstruction has been reported to occur (Simons et al, 2002; Jetty and Barber, 2004; Lambie et al, 2004).
Iliac Artery Aneurysms
Iliac artery aneurysms can cause ureteral obstruction, which may even lead to renal failure (Ochoa Urdangarain et al, 2006). Ureteral obstruction has been reported in 35% of those with common iliac artery aneurysms and 19% of those involving the internal iliac artery. An iliac artery aneurysm should be suspected if a pulsatile mass is palpated on rectal examination (Marino et al, 1987). The diagnosis can be made with various imaging studies including ultrasonography, MRI, and CT (Fig. 40–17A and B). Ureteral obstruction may be due to extrinsic compression or localized inflammation (Mineta et al, 2004).
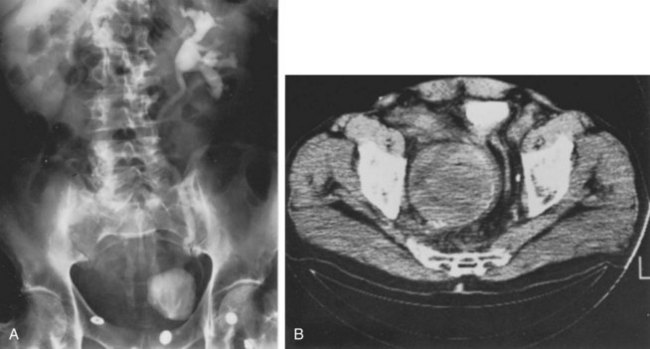
Figure 40–17 Right ureteral obstruction secondary to an aneurysm of the right hypogastric artery. A, Intravenous urogram demonstrates delayed visualization of the right kidney and marked deviation of the urinary bladder. B, Computed tomography scan demonstrates a large aneurysm. Note calcification of the wall.
Treatment options for common iliac artery aneurysms in the setting of ureteral obstruction include ureterolysis, aneurysm resection and graft placement, or endovascular graft placement. Options for obstructing internal iliac artery aneurysms include open surgical ligation and ureterolysis or initial endovascular occlusion and subsequent retrograde placement of an iliac artery endovascular graft. Internalized ureteral stents are typically placed before these procedures and are removed 6 to 8 weeks later. These patients need to be observed closely to determine whether ureteral obstruction resolves or recurs. If it recurs, management options include chronic internalized ureteral stenting, ureterolysis, or ureteral reconstruction (Marino et al, 1987).
An additional cause of ureteral obstruction is the retroiliac ureter (Corbus et al, 1960). Treatment of the ureteral obstruction involves transection of the ureter and transposition to the normal, anterior position. Other genitourinary abnormalities are associated with this condition, including renal hypoplasia, vesicoureteral reflux, ureteral ectopia, ectopic vas deferens, hypospadias, bifid scrotum, hypoplastic or duplex uterus, and hydrometrocolpos (Nguyen et al, 1989; Katz and Meirow, 1993). Therefore a search for other genitourinary abnormalities should be undertaken in patients with retroiliac ureter.
Ureteral Obstruction after Vascular Graft Placement
Hydronephrosis may occur in up to 10% to 20% of patients after aortic bypass graft surgery (Goldenberg et al, 1988; Schein and Saadia, 1991). There are several possible causes for this problem. Mechanical obstruction of the ureter may be due to graft placement anterior to the ureter, which is readily identified on axial imaging (Bouterie and Harbach, 1979). However, this is the cause in the minority of cases. Other possible etiologies include ureteral entrapment in perigraft fibrosis, ureteral devascularization, unidentified ureteral ligation, and ureteral compression from a postoperative pseudoaneurysm (Sant et al, 1983). Symptoms and signs of ureteral obstruction in this setting typically arise in the first postoperative year and include flank pain, anorexia, hypertension, anuria, and renal failure or insufficiency. A minority may remain asymptomatic or manifest delayed symptoms.
Management of this problem varies. If hydronephrosis is demonstrated early in the postoperative period and renal function is not compromised, close observation is initially indicated because hydronephrosis resolves spontaneously in the majority of cases (Goldenberg et al, 1988). Temporary internalized ureteral stent placement or percutaneous nephrostomy drainage may be required for those with renal impairment or severe symptoms while awaiting spontaneous resolution. If ureteral obstruction does not resolve spontaneously and the graft is placed anteriorly, transposition of the ureter and vascular graft should be a consideration. To reduce the risk of graft infection, however, it has been suggested that the vascular graft, rather than the ureter, should be transected and repositioned (Bouterie and Harbach, 1979). A trial of steroid or tamoxifen therapy, similar to that employed for retroperitoneal fibrosis, may be considered if persistent ureteral obstruction is ascribed to perigraft fibrosis. Although success has been reported with such regimens, careful follow-up is mandatory because delayed treatment failure has been reported (Huben and Schellhammer, 1981; Baskerville and Browse, 1987; Jetty and Barber, 2004). Ureterolysis, and ileal ureter substitution, if the former is not feasible, are considerations if medical therapy is not successful. Chronic ureteral stenting may be required in those who are not candidates for the aforementioned procedures (Sant et al, 1983; Matlaga et al, 2003).
Uterine Artery Aneurysms/Pseudoaneurysms (UAA)
UAA is a very uncommon problem that is typically associated with pregnancy. Symptomatic ureteral obstruction leading to hydronephrosis due to UAA has been reported (Leocadio et al, 2006). These conditions have been successfully managed by radiographic embolization or surgical ligation (Zimon et al, 1999).
Venous Causes of Obstruction
Puerperal Ovarian Vein Thrombophlebitis
Ureteral obstruction has been ascribed to a mechanical effect from a dilated ovarian vein, a condition called “ovarian vein syndrome.” The clinical relevance of this entity has since been questioned (Dure-Smith, 1979). However, ovarian vein pathology has been documented to be the cause of ureteral obstruction in the postpartum period. Puerperal ovarian vein thrombophlebitis, a rare but well-documented postpartum condition, has been reported to cause ureteral obstruction due to perivenous phlegmon formation and resultant periureteritis (Dure-Smith, 1979; Johnson et al, 1998; Brown et al, 1999). Although more common on the right, it may occur in either or both ovarian veins. The associated signs and symptoms are often nonspecific but include abdominal or flank pain and fever, usually beginning 2 to 3 days after birth. On physical examination, abdominal tenderness and guarding may be elicited. A tender indurated adnexal mass may be discerned in 50% of patients (Toland et al, 1993). CT or MRI can detect this condition and the associated ureteral obstruction (Twicker et al, 1997).
The majority of patients respond to conservative measures, including antibiotic therapy (Brown et al, 1999). Internalized ureteral stent placement may also be required for management of ureteral obstruction and symptoms (Toland et al, 1993). Laparoscopic or open surgical ureterolysis and ovarian vein resection are undertaken if ureteral obstruction persists (Almeida et al, 2003; del Valle González et al, 2006).
Testicular Vein Thrombophlebitis
Thrombophlebitis of the left testicular vein causing extrinsic obstruction of the left ureter has been reported (Kretkowski and Shah, 1977). Although the patient presented with the clinical picture of UPJO, surgical exploration revealed a dilated and thrombosed left testicular vein impinging upon the proximal ureter. Ureterolysis and excision of the thrombosed vein eradicated the obstruction. A similar event could also occur on the right side (Meyer et al, 1992).
Circumcaval Ureter
Obstruction of the ureter has been described in association with an anomalous course posterior, medial, anterior, and finally lateral to the inferior vena cava (IVC) (Bateson and Atkinson, 1969). This course may lead to extrinsic obstruction of the ureter. Although commonly referred to as circumcaval or retrocaval ureter, a more appropriate term may be preureteral vena cava, because it is due to a congenital abnormality in development of the vena cava. The term retrocaval is now primarily used to describe ureters that simply course behind the IVC and reemerge laterally.
Formation of the infrahepatic vena cava is based upon the development and regression of three pairs of embryonic veins: the posterior cardinal, the supracardinal, and the subcardinal. It is postulated that the normally developed IVC results from persistence of the right subcardinal vein suprarenally and the right supracardinal infrarenally. The posterior cardinal veins persist as the common iliac veins (Bass et al, 2000). The anastomosis between the right subcardinal and supracardinal vein crosses anterior to the fetal ureter. In normal development, this connection regresses, and the supracardinal vein persists as the infrarenal IVC. The prevailing theory of the development of this anomaly is that the subcardinal vein persists as the infrarenal IVC, thus crossing anterior to the midportion of the ureter and resulting in its circumcaval course (Schulman, 1997). Others have suggested that the persistence of the posterior cardinal vein as the infrarenal cava is responsible for this anomaly. However, failure of the supracardinal vein to develop into the infrarenal IVC is common to both theories (Bass et al, 2000) (Fig. 40–18A to C).
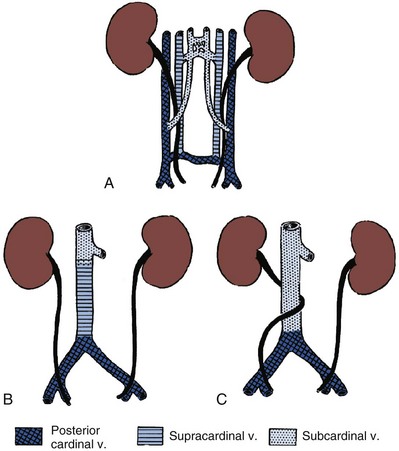
Figure 40–18 Relationship between the development of the infrarenal portion of the inferior vena cava and the retrocaval ureter. A, Primitive condition with the ureter winding among three cardinal veins. B, Usual method of formation of the vena cava from the right subcardinal vein (dorsal to ureter). C, Main portion of the vena cava formed from the subcardinal vein.
(From Hollinshead WH. Anatomy for surgeons, vol. 2. 2nd ed. New York: Harper & Row; 1971.)
The reported incidence of circumcaval ureter is approximately 1 in 1100. There is a 2.8-fold male predominance (Lutin et al, 1988). Symptomatic patients typically present in the third or fourth decade of life (Zhang et al, 1990). This vascular anomaly is not always associated with ureteral obstruction (Crosse et al, 1975). The majority involve the right ureter, although left-sided circumcaval ureter has been reported in association with a duplicated IVC and in association with situs inversus (Watanabe et al, 1991; Rubinstein et al, 1999). Other genitourinary anomalies may be present. Circumcaval right ureter with a retrocaval horseshoe isthmus has been reported (Knutson and Hawas, 2004).
Circumcaval ureter has been classified using different parameters based on intravenous urography or retrograde pyelographic findings. Bateson and Atkinson (1969) classified a ureter with an S-shaped, fish hook, or shepherd’s crook appearance as type I (Fig. 40–19A). The ureter typically overlies or is medial to the lower lumbar vertebral processes and eventually crosses anterior to the iliac vessels, where it then assumes a normal distal course in this setting. A less-angulated “sickle-shaped” ureteral deformity is classified as type II (Fig. 40–19B). The point of maximal obstruction in type I is lateral to the lateral margin of the IVC and is associated with a greater degree of hydronephrosis than type II, in which the point of obstruction is at the lateral border of the IVC (Bateson and Atkinson, 1969). Another classification scheme is based on the level of obstruction; type I crosses at the level of the third lumbar vertebra, and type II crosses at the level of the ureteropelvic junction. The latter may be confused with ureteropelvic junction obstruction (Kenawi and Williams, 1976). The obstruction has been attributed to extrinsic compression of the ureter by the IVC, its lumbar or gonadal branches, or the psoas muscle. However, an intrinsic ureteral abnormality may be contributory as histologic studies of involved ureteral segments have demonstrated fibrotic changes (Kumar and Bhandar, 1985).
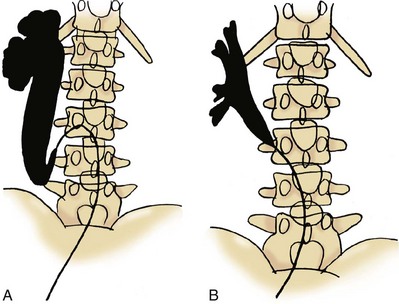
Figure 40–19 A, Diagram illustrating the radiologic features of the type 1 circumcaval ureter. B, Diagram illustrating the radiologic features of the type 2 circumcaval ureter.
(A and B, From Bateson EM, Atkinson D. Circumcaval ureter: a new classification. Clin Radiol 1969;20:173–7.)
Abdominal and flank pain, recurrent urinary tract infection, and hypertension are some of the initial symptoms and signs. A number of cases are now discovered on imaging studies performed to evaluate patients with nonurologic problems (Belis and Milam, 1980; Sener, 1993). The diagnosis should be suspected when the aforementioned pyelographic findings are present (Fig. 40–20A to C). The diagnosis can be confirmed with CT and MRI (Fig. 40–21A and B). Diuretic renography is used to confirm the presence and determine the functional impact of obstruction.
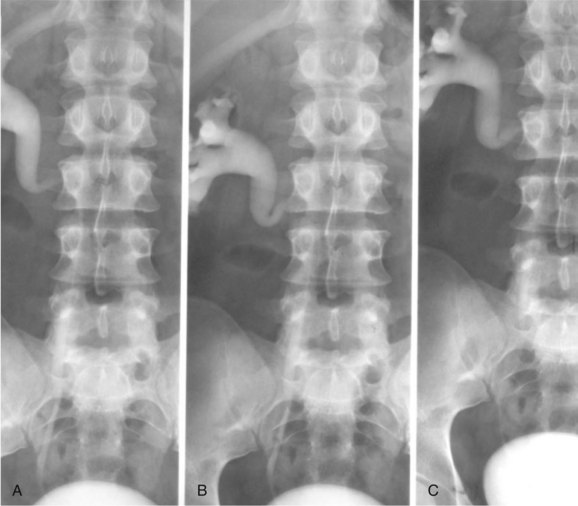
Figure 40–20 A to C. Excretory urography of a patient with type 1 circumcaval ureter.
(Courtesy of Dr. Ronald Zagoria.)
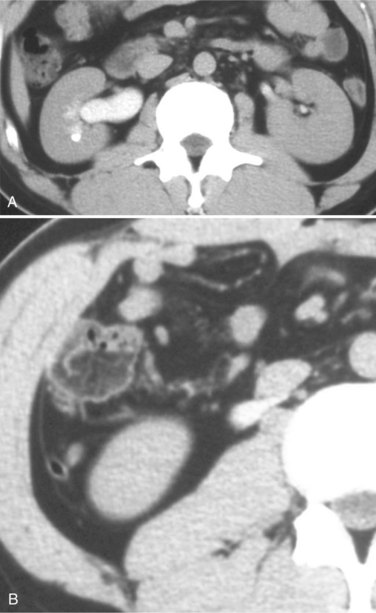
Figure 40–21 A, Contrast-enhanced computed tomography of a patient with circumcaval ureter. Note mild right-sided hydronephrosis. B, Lower section from same study and patient. Note ureter traversing posterior to inferior vena cava.
(A and B, Courtesy of Dr. Ronald Zagoria.)
Treatment is undertaken only in the presence of obstruction. The ureter is divided proximally and at the distal point, from which it emerges lateral to the IVC. A spatulated ureteroureterostomy is performed. This has been performed by open, laparoscopic, and retroperitoneoscopic approaches and is best dictated by the surgeon’s experience (Polascik and Chen, 1998; Gupta et al, 2001; Bagheri et al, 2009). Recently, pure robotic repair of retrocaval ureter has also been described successfully with its attendant ergonomic and technical benefits (Hemal et al, 2008). The most common complication associated with this procedure is ureteral stricture, reported to be 13% in one series (Hadzi-Djokic et al, 2009).
Key Points
Chevalier RL. Molecular and cellular pathophysiology of obstructive nephropathy. Pediatr Nephrol. 1999;13:612-619.
Corradi D, Maestri R, Palmisano A, et al. Idiopathic retroperitoneal fibrosis: clinicopathologic features and differential diagnosis. Kidney Int. 2007;72(6):742-753.
Dhillon HK. Prenatally diagnosed hydronephrosis: the Great Ormond Street experience. Br J Urol. 1998;81(Suppl. 2):39-44.
Faundes A, Bricola-Filho M, Pinto e Silva JL. Dilatation of the urinary tract during pregnancy: proposal of a curve of maximal caliceal diameter by gestational age. Am J Obstet Gynecol. 1998;178:1082-1086.
Frøkiaer J, Zeidel ML. Urinary tract obstruction. In Brenner BM, editor: Brenner and Rector’s the kidney, 8th ed, Philadelphia: WB Saunders, 2007.
Harris RH, Yarger WE. Renal function after release of unilateral ureteral obstruction in rats. Am J Physiol. 1974;227:806-815.
Hewitson TD. Renal tubulointersitial fibrosis: common but never simple. Am J Physiol Renal Physiol. 2009;296:F1239-F1244.
Heyns CF. Pelvic lipomatosis: a review of its diagnosis and management. J Urol. 1991;146:267-273.
Klahr S, Morrissey J. Obstructive nephropathy and renal fibrosis. Am J Physiol. 2002;283:F861-F875.
Li C, Wang W, Kwon TH, et al. Altered expression of major renal Na transporters with bilateral ureteral obstruction and release of obstruction. Am J Physiol. 2003;285:F889-F901.
Misseri R, Rink RC, Meldrum DR, et al. Research review: inflammatory mediators and growth factors in obstructive renal injury. J Surg Res. 2004;119:149-159.
Vaglio A, Salvarani C, Buzio C. Retroperitoneal fibrosis. Lancet. 2006;367(9506):241-251. 21
Varkarakis IM, Jarrett TW. Retroperitoneal fibrosis. AUA Update Series. 24, 2005. lesson 3
Vaughan ED, Gillenwater JY. Recovery following complete chronic unilateral ureteral occlusion: functional, radiographic, and pathologic alterations. J Urol. 1971;106:27-35.
Wamsley-Davis A, Padda R, Truong LD, et al. AT1A-mediated activation of kidney JNK1 and SMAD2 in obstructive uropathy: preservation of kidney tissue mass using candesartan. Am J Physiol. 2004;287:F474-F480.
Zagoria R, Tung G. Genitourinary radiology: the requisites. St Louis: Mosby; 1997.
Abodeely DA, Lee JB. Fuel of respiration of outer renal medulla. Am J Physiol. 1971;220:1693-1700.
Abou El-Ghar ME, Shokeir AA, El-Diasty TA, et al. Contrast enhanced spiral computerized tomography in patients with chronic obstructive uropathy and normal serum creatinine: a single session for anatomical and functional assessment. J Urol. 2004;172:985-988.
Abou El-Ghar ME, Shokeir A, Rafael H, El-Diasty T. Static T2w MRU in noncalcular urinary obstruction: comparison of its two techniques. Sci World J. 2008;8:454-462.
Abou El-Ghar ME, Shokeir AA, Refaie HF, El-Diasty TA. MRI in patients with chronic obstructive uropathy and compromised renal function: a sole method for morphological and functional assessment. Br J Radiol. 2008;81:624-629.
Abu-Zidan FM, Elgazzar AH, Bahar RH, et al. A new experimental model for partial ureteric obstruction in sheep. Int Urol Nephrol. 1999;31:149-155.
Adler S, Lodermeyer S, Gaa J, Heemann U. Successful mycophenolate mofetil therapy in nine patients with idiopathic retroperitoneal fibrosis. Rheumatology (Oxford). 2008;47(10):1535-1538.
Agostini S, Dedola GL, Gabbrielli S, Masi A. A new percutaneous nephrostomy technique in the treatment of obstructive uropathy. Radiol Med. 2003;105:454-461.
Albarran J. Rétention renale par périurétérite: libération externe de l’uretère. Assoc Fr Urol. 1905;9:511-517.
Alexopoulos E, Memmos D, Bakatselos S, et al. Idiopathic retroperitoneal fibrosis: a long-term follow up study. Eur Urol. 1987;13:313-317.
Al-Hunayan A, Al-Ateeqi A, Kehinde EO, et al. Diagnostic accuracy of urinary creatinine concentration in the estimation of differential renal function in patients with obstructive uropathy. Urol Int. 2008;80:300-305.
Al-Khwaja M, Tan PH, MacLennan GT, et al. Ureteral endometriosis: clinicopathological and immunohistochemical study of seven cases. Hum Pathol. 2008;39:954-959.
Allen JT, Vaughan EDJr, Gillenwater JY. The effect of indomethacin on renal blood flow and ureteral pressure in unilateral ureteral obstruction in awake dogs. Invest Urol. 1978;15:324-327.
Almeida A, Cavalcanti F, Barbosa S, et al. Laparoscopic approach in the ovarian vein syndrome. Int Braz J Urol. 2003;29:45-47.
Amis ES. Retroperitoneal fibrosis. AJR Am J Roentgenol. 1991;157:321-329.
Ammons WS. Renal afferent inputs to ascending spinal pathways. Am J Physiol. 1992;262:R165-R176.
Andac N, Baltacioglu F, Cimsit NC, et al. Fat necrosis mimicking liposarcoma in a patient with pelvic lipomatosis CT findings. J Clin Imaging. 2003;27:109-111.
Antonelli A, Simeone C, Canossi E, et al. Surgical approach to urinary endometriosis: experience on 28 cases. Arch Ital Urol Androl. 2006;78:35-38.
Antonelli A, Simeone C, Frego E, et al. Surgical treatment of ureteral obstruction from endometriosis: our experience with thirteen cases. Int Urogynecol J. 2004;15:407-412.
Antonelli A, Simeone C, Zani D, et al. Clinical aspects and surgical treatment of urinary tract endometriosis: our experience with 31 cases. Eur Urol. 2006;49:1093-1098.
Aral SO, Mosher WD, Cates W. Self reported pelvic inflammatory disease in the United States, 1988. JAMA. 1991;266:2570-2573.
Arendshorst WJ, Finn WF, Gottschalk CW. Nephron stop-flow pressure response to obstruction for 24 hours in the rat kidney. J Clin Invest. 1974;53:1497-1500.
Arroyo A, Rodriguez J, Porto J, Gesto R. Management and course of hydronephrosis secondary to inflammatory aneurysms of the abdominal aorta. Ann Vasc Surg. 2003;7:481-485.
Au KK, Woo JS, Tang LC, Liang ST. Aetiological factors in the genesis of pregnancy hydronephrosis. Aust N Z J Obstet Gynaecol. 1985;25:248-251.
Bae EH, Kim IJ, Park JW, et al. Altered regulation of renin-angiotensin, endothelin and natriuretic peptide systems in rat kidney with chronic unilateral ureteral obstruction. Urol Int. 2007;79(2):170-176.
Bagheri F, Pusztai C, Szántó A, et al. Laparoscopic repair of circumcaval ureter: one-year follow-up of three patients and literature review. Urology. 2009;74(1):148-153.
Bagnasco S, Good D, Balaban R, et al. Lactate production in isolated segments of the rat nephron. Am J Physiol. 1985;248:F522-F526.
Baker LR. Auto-allergic periaortitis (idiopathic retroperitoneal fibrosis). BJU Int. 2003;92:663-665.
Balleyguier C, Roupret M, Nguyen T, et al. Ureteral endometriosis: the role of magnetic reasonance imaging. J Am Assoc Gynecol Laprosc. 2004;4:530-536.
Bander SJ, Buerkert JE, Martin D, Klahr S. Long term effects of 24-hr unilateral obstruction on renal function in the rat. Kidney Int. 1985;28:614-620.
Barbalias GA, Liatsikos EN. Idiopathic retroperitoneal fibrosis revisited. Int Urol Nephrol. 1999;31:423-429.
Baskerville PA, Browse NL. Peri-aortic fibrosis: progression and regression. J Cardiovasc Surg. 1987;28:30-31.
Bass JE, Redwine MD, Kramer LA, et al. Spectrum of congenital anomalies of the inferior vena cava: cross sectional imaging findings. Radiographics. 2000;20:639-652.
Bateson EM, Atkinson D. Circumcaval ureter: a new classification. Clin Radiol. 1969;20:173-177.
Beaumont K, Vaughn DA, Maciejewski AR, Fanestil DD. Reversible downregulation of thiazide diuretic receptors by acute renal ischemia. Am J Physiol. 1989;256:F329-F334.
Beck N. Phosphaturia after release of bilateral ureteral obstruction in rats. Am J Physiol. 1979;237:F14-F19.
Beharrie A, Franc-Guimond J, Rodriguez MM, et al. A functional immature model of chronic partial ureteral obstruction. Kidney Int. 2004;65:1155-1161.
Belis JA, Milam F. Chronic abdominal pain caused by circumcaval ureter. South Med J. 1980;73:538-539.
Bell ET. Renal diseases. Philadelphia: Lea & Febiger; 1950.
Berkley KJ, Rapkin AJ, Papka RE. The pains of endometriosis. Science. 2005;308:1587-1589.
Berlyne G. Distal tubular function in chronic hydronephrosis. Q J Med. 1961;30:339-355.
Bhangdia DK, Gulmi FA, Mooppan UM, et al. Alterations of renal hemodynamics in unilateral ureteral obstruction mediated by activation of endothelin receptor subtypes. J Urol. 1998;159:S69.
Birchard KR, Brown MA, Hyslop WB, et al. MRI of acute abdominal and pelvic pain in pregnant patients. AJR Am J Roentgenol. 2005;184:452-458.
Blackshear JL, Wathen RL. Effects of indomethacin on renal blood flow and renin secretory responses to ureteral occlusion in the dog. Miner Electrolyte Metab. 1978;1:271-278.
Bouley R, Breton S, Sun T, et al. Nitric oxide and atrial natriuretic factor stimulate cGMP-dependent membrane insertion of aquaphorin 2 in renal epithelial cells. J Clin Invest. 2000;106:1115-1126.
Bouley R, Pastor-Soley N, Cohen O, et al. Stimulation of AQP-2 membrane insertion in renal epithelial cell in vitro and in vivo by the cGMP phosphodiesterase inhibitor sildenafil citrate (Viagra). Am J Physiol. 2005;288:F1103-F1112.
Bouterie RL, Harbach LB. Ureteral obstruction after aorto-femoral bypass surgery. Urology. 1979;14:273-275.
Brandel RA, Brock JW, Hamilton BD, et al. Unilateral hydronephrosis in infants: are measurements of contralateral renal length useful? J Urol. 1996;156:188-189.
Brenner BM, Ballermann BJ, Gunning ME, et al. Diverse biological actions of atrial natriuretic peptide. Physiol Rev. 1990;70:665-699.
Broome DR, Girguis MS, Baron PW, et al. Gadolinium-associated nephrogenic systemic fibrosis: why radiologists should be concerned. AJR Am J Radiol. 2007;188:586-592.
Brown CE, Stettler RW, Twickler D, Cunningham GF. Puerperal septic pelvic thrombophlebitis: incidence and response to heparin therapy. Am J Obstet Gynecol. 1999;181:143-148.
Burn PR, Singh S, Barbar S, et al. Role of gadolinium-enhanced magnetic resonance imaging in retroperitoneal fibrosis. Can Assoc Radiol J. 2002;53:168-170.
Campbell MF. Urinary obstruction. Campbell MF, Harrison JH. WB Saunders, Urology Philadelphia, 1970, 1772-1793.
Canbay A, Friedman S, Gores GJ. Apoptosis: the nexus of liver injury and fibrosis. Hepatology. 2004;39(2):273-278.
Carr MC, Peters CA, Retik AB, et al. Urinary levels of the renal tubular enzyme N-acetyl-beta-D-glucosaminidase in unilateral obstructive uropathy. J Urol. 1994;151:442-445.
Castle WN, McDougal WS. Contralateral renal hyperplasia and increased renal function after relief of chronic unilateral ureteral obstruction. J Urol. 1984;132:1016-1020.
Chen JH, Pu YS, Liu SP, Chiu TY. Renal hemodynamics in patients with obstructive uropathy evaluated by duplex Doppler sonography. J Urol. 1993;150:18-21.
Cheng X, Zhang H, Lee H, Park JM. Cyclooxygenase-2 inhibitor preserves medullary aquaphorin-2 expression and prevents polyuria after ureteral obstruction. J Urol. 2004;172:2387-2390.
Chevalier RL. Obstructive nephropathy: towards biomarker discovery and gene therapy. Nat Clin Pract Nephrol. 2006;2(3):157-168.
Chevalier RL, Goyal S, Kim A, et al. Renal tubulointerstitial injury from ureteral obstruction in the neonatal rat is attenuated by IGF-1. Kidney Int. 2000;57:882-890.
Chevalier RL, Goyal S, Wolstenholme JT, et al. Obstructive nephropathy in the neonatal rat is attenuated by epidermal growth factor. Kidney Int. 1998;54:38-47.
Chevalier RL, Kaiser DL. Chronic partial ureteral obstruction in the neonatal guinea pig. 1. Influence of uninephrectomy on growth and hemodynamics. Pediatr Res. 1984;18:1266-1271.
Chevalier RL, Thornhill BA, Wolstenholme JT, Kim A. Unilateral ureteral obstruction in early development alters renal growth: dependence on the duration of obstruction. J Urol. 1999;161:309-313.
Chizzolini C, Raschi E, Rezzonico R, et al. Autoantibodies to fibroblasts induce a proadhesive and proinflammatory fibroblast phenotype in patients with systemic sclerosis. Arthritis Rheum. 2002;46(6):1602-1613.
Choi YJ, Baranowska-Daca E, Nguyen V, et al. Mechanism of chronic obstructive uropathy: increased expression of apoptosis-promoting molecules. Kidney Int. 2000;58:1481-1491.
Chu WCW, Lam WWM, Chan KW, et al. Dynamic gadolinium-enhanced magnetic resonance urography for assessing drainage in dilated pelvicalyceal systems with moderate renal function: preliminary results and comparison with diuresis renography. BJU Int. 2004;93:830-834.
Chung SY, Stein RJ, Landsittel D, et al. 15-year experience with the management of extrinsic ureteral obstruction with indwelling ureteral stents. J Urol. 2004;172:592-595.
Cietak KA, Newton JR. Serial qualitative maternal nephrosonography in pregnancy. Br J Radiol. 1985;58:399-404.
Cogan MG. Renal effects of atrial natriuretic factor. Annu Rev Physiol. 1990;52:699-708.
Cohen JJ. Is the function of the renal papilla coupled exclusively to an anaerobic pattern of metabolism? Am J Physiol. 1979;236:F423-F433.
Colon I, Gulmi F, Chou S-Y, et al. Alterations of renal hemodynamics after release of unilateral ureteral obstruction by blocking of endothelin receptor subtypes ETa and ETb. J Urol. 2000;163:S83.
Comiter CV. Endometriosis of the urinary tract. Urol Clin North Am. 2002;29:625-635.
Conde Santos G, Bielsa Gali O, Arango Toro O, et al. Extrinsic ureteral obstruction secondary to inflammatory gynecologic pathology. Arch Esp Urol. 2003;56:181-185.
Corbus BC, Estrem RD, Hunt W. Retro-iliac ureter. J Urol. 1960;84:67-68.
Corradi D, Maestri R, Palmisano A, et al. Idiopathic retroperitoneal fibrosis: clinicopathologic features and differential diagnosis. Kidney Int. 2007;72(6):742-753.
Cozzi F, Zani A, Schiavetti A, et al. Compensatory renal growth in children with unilateral renal tumor treated by nephron-sparing surgery or nephrectomy. Eur J Pediatr Surg. 2007;17(6):382-386.
Cronin CG, Lohan DG, Blake MA, et al. Retroperitoneal fibrosis: a review of clinical features and imaging findings. AJR Am J Roentgenol. 2008;191(2):423-431.
Crosse JE, Soderdahl DW, Teplick SK, Clark RE. Nonobstructive circumcaval (retrocaval) ureter: a report of two cases. Radiology. 1975;116:69-71.
Dal Canton A, Corradi A, Stanziale R, et al. Glomerular hemodynamics before and after release of 24-hour bilateral ureteral obstruction. Kidney Int. 1980;17:491-496.
Danilovic A, Antonopoulos IM, Mesquita JL, Lucon AM. Likelihood of retrograde double-J stenting according to ureteral obstructing pathology. Int Braz J Urol. 2005;31:431-436.
Das D, Teece S. Best evidence topic report. Intravenous NSAIDs in the management of renal colic. Emerg Med J. 2006;23:225.
Dash RC, Liu K, Sheafor DH, Dodd LG. Fine-needle aspiration findings in idiopathic retroperitoneal fibrosis. Diagn Cytopathol. 1999;21(1):22-26.
Davenport K, Timoney AG, Keeley FX. Conventional and alternative methods for providing analgesia in renal colic. BJU Int. 2005;95:297.
Debruyne FM, Bogman MJ, Ypma AF. Retroperitoneal fibrosis in the scrotum. Eur Urol. 1982;8:45-48.
de Cavanagh EM, Ferder M, Inserra F, Ferder L. Angiotensin II, mitochondria, cytoskeletal, and extracellular matrix connections: an integrating viewpoint. Am J Physiol Heart Circ Physiol. 2009;296(3):H550-H558.
Deleersnijder R, Daenens K, Fourneau I, et al. Endovascular repair of inflammatory abdominal aortic aneurysms with special reference to concomitant ureteric obstruction. Eur J Vasc Endovasc Surg. 2002;24:146-149.
del Valle González N, Estébanez Zarranz J, Escudero Caro T, et al. Laparoscopic treatment of ovarian vein syndrome. Actas Urol Esp. 2006;30:85-89.
Demas BE, Avallone A, Hricak H. Pelvic lipomatosis: diagnosis and characterization by magnetic resonance imaging. Urol Radiol. 1988;10:198-202.
Denstedt JD, Razvi H. Management of urinary calculi during pregnancy. J Urol. 1992;148:1072-1075.
Dhillon HK. Prenatally diagnosed hydronephrosis: the Great Ormond Street experience. Br J Urol. 1998;81(Suppl. 2):39-44.
Diamond JR, Kees-Folts D, Ding G, et al. Macrophages, monocyte chemoattractant peptide-1, and TGF-beta 1 in experimental hydronephrosis. Am J Physiol. 1994;266:F926-F933.
Dicker SE, Shirley DG. Compensatory renal growth after unilateral nephrectomy in the newborn rat. J Physiol. 1973;228:193-202.
Docimo SG, Dewolf WC. High failure rate of indwelling ureteral stents in patients with existing obstruction: experience at 2 institutions. J Urol. 1989;142:277-279.
Duchene DA, Winfield HN, Cadeddu JA, et al. Multi-institutional survey of laparoscopic ureterolysis for retroperitoneal fibrosis. Urology. 2007;69(6):1017-1021.
Dure-Smith P. Ovarian syndrome: is it a myth? Urology. 1979;13:355-363.
Edgren J, Laasonen L, Kock B, et al. Kidney function and compensatory growth of the kidney in living donors. Scand J Urol. 1976;10:134-136.
el-Dahr SS, Gee J, Dipp S, et al. Upregulation of renin-angiotensin system and downregulation of kallikrein in obstructive nephropathy. Am J Physiol. 1993;264:F874-F881.
Elashry OM, Nakada SY, Wolf JS, et al. Ureterolysis for extrinsic ureteral obstruction: a comparison of laparoscopic and open surgical techniques. J Urol. 1996;156:1403-1410.
El-Nahas AR, Abou El-Ghar ME, Refae HF, et al. Magnetic resonance imaging in the evaluation of pelvi-ureteric junction obstruction: an all-in-one approach. BJU Int. 2007;99(3):641-645.
El-Nahas AR, Abou-El-Ghar M, Shoma AM, et al. Role of multiphasic helical CT in planning surgical treatment for pelviureteric junction obstruction. BJU Int. 2004;94:582-587.
Elsayes KM, Staveteig PT, Narra VR, et al. Retroperitoneal masses: magnetic resonance imaging findings with pathologic correlation. Curr Probl Diagn Radiol. 2007;36(3):97-106.
Engeler DS, Schmid S, Schmid HP. The ideal analgesic treatment for acute renal colic—theory and practice. Scand J Urol Nephrol. 2008;42(2):137-142.
Engels EP. Sigmoid colon and urinary bladder in high fixation: roentgen changes simulating pelvic tumor. Radiology. 1959;72:419.
English PJ, Testa HJ, Lawson RS, et al. Modified method of diuresis renography for the assessment of equivocal pelviureteric junction obstruction. Br J Urol. 1987;59:10-14.
Ergen FB, Hussain HK, Carlos RC, et al. 3D excretory MR urography: improved image quality with intravenous saline and diuretic administration. J Magn Reson Imaging. 2007;25(4):783-789.
Erickson LM, Nicholson SF, Lewall DB, et al. Ultrasound evaluation of hydronephrosis of pregnancy. J Clin Ultrasound. 1979;7(2):128-132.
Eskenazi B, Warner ML. Epidemiology of endometriosis. Obstet Gynecol Clin North Am. 1997;24:235-258.
Eskild-Jensen A, Frøkiær J, Djurhuus JC, et al. Reduced number of glomeruli in kidneys with neonatally induced partial ureteropelvic obstruction in pigs. J Urol. 2002;167:1435-1439.
Eskild-Jensen A, Jacobsen L, Christensen H, et al. Renal functional outcome in unilateral hydronephrosis in newborn pigs. II. Function and volume of contralateral kidneys. J Urol. 2001;165:205-209.
Eskild-Jensen A, Paulsen LF, Wogensen L, et al. AT1 receptor blockade prevents interstitial and glomerular apoptosis but not fibrosis in pigs with neonatal induced partial unilateral ureteral obstruction. Am J Physiol Renal Physiol. 2007;292(6):F1771-F1781.
Everaert K, Kerckhaert W, Delanghe J, et al. Elevated tubular proteinuria, albuminuria and decreased urinary N-acetyl-beta-D-glucosaminidase activity following unilateral total ureteral obstruction in rats. Urol Res. 1998;26:285-289.
Fassina A, Boscolo Berto R, El Mazloum R, et al. Retroperitoneal fibrosis after chemotherapy. Eur Urol. 2007;51(1):270-271.
Faundes A, Bricola-Filho M, Pinto e Silva JL. Dilatation of the urinary tract during pregnancy: proposal of a curve of maximal caliceal diameter by gestational age. Am J Obstet Gynecol. 1998;178:1082-1086.
Fedele L, Bianchi S, Zanconato G, et al. Long-term follow up after conservative surgery for bladder endometriosis. Fertil Steril. 2005;83:1729-1733.
Fedele M, Battista S, Manfioletti G, et al. Role of the high mobility group A proteins in human lipomas. Carcinogenesis. 2001;22:1583-1591.
Feder MT, Blitstein J, Mason B, Hoenig DM. Predicting differential renal function using computerized tomography measurements of renal parenchymal area. J Urol. 2008;180:2110-2115.
Fielding JR, Chin BM. Magnetic resonance imaging of abdominal pain during pregnancy. Top Magn Reson Imaging. 2006;6:409-416.
Francisco LL, Hoversten LG, Dibona GF. Renal nerves in the compensatory adaptation to ureteral occlusion. Am J Physiol. 1980;238:F229-F234.
Frenna V, Santos L, Ohana E, et al. Laparoscopic management of ureteral endometriosis: our experience. J Min Inv Gynecol. 2007;14:169-171.
Fried TA, Ayon MA, McDonald G, et al. Atrial natriuretic peptide, right atrial pressure, and sodium excretion rate in the rat. Am J Physiol. 1987;253:F969-F975.
Frøkiaer J, Eskild-Jensen A, Dissing T. Antenatally detected hydronephrosis: the nuclear medicine techniques. In: Prigent A, Piepez A, editors. Functional imaging in nephrourology. London: Taylor and Francis, 2005.
Frøkiaer J, Marples D, Knepper MA, Nielsen S. Bilateral ureteral obstruction downregulates expression of vasopressin-sensitive AQP-2 water channel in rat kidney. Am J Physiol. 1996;270:F657-F668.
Frøkiaer J, Nielsen AS, Knudsen L, et al. The effect of indomethacin infusion on renal hemodynamics and on the renin-angiotensin system during unilateral ureteral obstruction of the pig. J Urol. 1993;150:1557-1563.
Frøkiær J, Sørensen SS. Eicosanoid excretion from the contralateral kidney in pigs with complete unilateral ureteral obstruction. J Urol. 1995;154:1205-1209.
Frøkiaer J, Zeidel ML. Urinary tract obstruction. In Brenner BM, editor: Brenner and Rector’s the kidney, 8th ed, Philadelphia: WB Saunders, 2007.
Fry AC, Singh S, Gunda SS, et al. Successful use of steroids and ureteric stents in 24 patients with idiopathic retroperitoneal fibrosis: a retrospective study. Nephron Clin Pract. 2008;108(3):C213-C220.
Fukasawa H, Yamamoto T, Togawa A, et al. Down-regulation of Smad-7 expression by ubiquitin-dependent degradation contributes to renal fibrosis in obstructive nephropathy in mice. Proc Natl Acad Sci U S A. 2004;101:8687-8692.
Fung LCT, Khoury AE, McLorie GA, et al. Evaluation of pediatric hydronephrosis using individualized pressure flow criteria. J Urol. 1995;154:671-676.
Fung LCT, Steckler RE, Khoury AE, et al. Intrarenal resistive index correlates with renal pelvis pressure. J Urol. 1994;152:607-611.
Gallagher PE, Chappell MC, Ferrario CM, Tallant EA. Distinct roles for ANG II and ANG-(1-7) in the regulation of angiotensin-converting enzyme 2 in rat astrocytes. Am J Physiol Cell Physiol. 2006;290:C420-C426.
Gasparich JP, Mayo ME. Comparative effects of four prostaglandin synthesis inhibitors on the obstructed kidney in the dog. J Urol. 1986;135:1088-1090.
Gaudio KM, Siegel NJ, Hayslett JP, et al. Renal perfusion and intratubular pressure during ureteral occlusion in the rat. Am J Physiol. 1980;238:F205-F209.
Generao SE, Keene KD, Das S. Endoscopic diagnosis and management of ureteral endometriosis. J Endourol. 2005;19:1177-1179.
Gentle DL, Stoller ML, Jarrett TW, et al. Protease inhibitor-induced urolithiasis. Urology. 1997;50:508-511.
Gesek FA, Friedman PA. Mechanism of calcium transport stimulated by chlorothiazide in mouse distal convoluted tubule cells. J Clin Invest. 1992;90:429-438.
Ghezzi F, Cromi A, Bergamini V, et al. Outcome of laparoscopic ureterolysis for ureteral endometriosis. Fertil Steril. 2006;86:418-422.
Ghezzi F, Cromi A, Bergamini V, Bolis P. Management of ureteral endometriosis: areas of controversy. Curr Opin Obstet Gynecol. 2007;19:319-324.
Ghersin E, Brook OR, Meretik S, et al. Antegrade MDCT pyelography for the evaluation of patients with obstructed urinary tract. AJR Am J Roentgenol. 2004;183:1691-1696.
Gillenwater JY, Westervelt FBJr, Vaughan EDJr, Howards SS. Renal function after release of chronic unilateral hydronephrosis in man. Kidney Int. 1975;7:179-186.
Giudice LC, Kao LC. Endometriosis. Lancet. 2004;364:1789-1799.
Gobet R, Park JM, Nguyen HT, et al. Renal renin-angiotensin system dysregulation caused by partial bladder outlet obstruction in fetal sheep. Kidney Int. 1999;56:1654-1661.
Goldenberg SL, Gordon PB, Cooperberg PL, McLoughlin MG. Early hydronephrosis following aortic bifurcation graft surgery: a prospective study. J Urol. 1988;140:1367-1369.
Goldfarb RA, Neerhut GJ, Lederer E. Management of acute hydronephrosis of pregnancy by ureteral stenting: risk of stone formation. J Urol. 1989;141:921-922.
Gosling JA, Dixon JS. Functional obstruction of the ureter and renal pelvis. A histological and electron microscopic study. Br J Urol. 1978;50:145-152.
Granese R, Candiani M, Perino A, et al. Bladder endometriosis: laparoscopic treatment and follow up. Eur J Obstet Gynecol Reprod Biol. 2008;140:114-117.
Greenberg MD, Kazamel TI. Medical and socioeconomic impact of uterine fibroids. Obstet Gynecol Clin North Am. 1995;22:625-636.
Grossman EB, Hebert SC. Modulation of Na-K-ATPase activity in the mouse medullary thick ascending limb of Henle. Effects of mineralocorticoids and sodium. J Clin Invest. 1988;81:885-892.
Guan Y, Chang M, Cho W, et al. Cloning, expression, and regulation of rabbit cyclooxygenase 2 in renal medullary interstitial cells. Am J Physiol. 1997;273:F18-F26.
Guder WG, Schmidt U. Substrate and oxygen dependence of renal metabolism. Kidney Int. 1976;6:S32-S38.
Gulmi FA, Matthews GJ, Marion D, et al. Volume expansion enhances the recovery of renal function and prolongs the diuresis and natriuresis after release of bilateral ureteral obstruction: a possible role for atrial natriuretic peptide. J Urol. 1995;153:1276-1283.
Gulmi FA, Mooppan UM, Chou S, Kim H. Atrial natriuretic peptide in patients with obstructive uropathy. J Urol. 1989;142:268-272.
Gupta NP, Hemal AK, Singh I, Khaitan MS. Retroperitoneoscopic ureterolysis and reconstruction of retrocaval ureter. J Endourol. 2001;15:291-293.
Hackstein N, Wiegand C, Rau WS, et al. Glomerular filtration rate measured by using triphasic helical CT with a two-point Patlak plot technique. Radiology. 2004;230(1):221-226.
Hadzi-Djokic J, Basic D, Dzamic Z, et al. Retrocaval ureter based on a series of 16 cases. Prog Urol. 2009;19:33-38.
Hanley MJ, Davidson K. Isolated nephron segments from rabbit models of obstructive nephropathy. J Clin Invest. 1982;69:165-174.
Harris KP, Schreiner GF, Klahr S. Effects of leukocyte depletion on the function of the postobstructed kidney in the rat. Kidney Int. 1989;36:210-215.
Harris RH, Yarger WE. Renal function after release of unilateral ureteral obstruction in rats. Am J Physiol. 1974;227:806-815.
Harris RH, Yarger WE. The pathogenesis of post-obstructive diuresis. The role of circulating natriuretic and diuretic factors, including urea. J Clin Invest. 1975;56:880-887.
Harris RH, Yarger WE. Urine-reinfusion natriuresis: evidence for potent natriuretic factors in rat urine. Kidney Int. 1977;11:93-105.
He W, Fischman AJ. Nuclear imaging in the genitourinary tract: recent advances and future directions. Radiol Clin North Am. 2008;46(1):25-43.
Hemal AK, Rao R, Sharma S, Clement RGE. Pure robotic retrocacval ureter. Int Braz J Urol. 2008;34:734-738.
Hendrikx A, Nadorp S, De Beer NA, et al. The use of endoluminal ultrasonography for preventing significant bleeding during endopyelotomy: evaluation of helical computed tomography vs endoluminal ultrasonography for detecting crossing vessels. BJU Int. 2006;97:786-789.
Hengartner MO. The biochemistry of apoptosis. Nature. 2000;407:770-776.
Hewitson TD. Renal tubulointersitial fibrosis: common but never simple. Am J Physiol Renal Physiol. 2009;296:F1239-F1244. Epub Jan 14, 2009
Heyns CF. Pelvic lipomatosis: a review of its diagnosis and management. J Urol. 1991;146:267-273.
Hinman F. The condition of renal counterbalance and the theory of renal atrophy of disuse. J Urol. 1943;49:392-400.
Hodson CJ. The effects of disturbance of flow on the kidney. J Infect Dis. 1969;120:54-60.
Holdgate A, Oh CM. Is there a role for antimuscarinics in renal colic? A randomised controlled trial. J Urol. 2005;174:572-575.
Holdgate A, Pollock T. Systematic review of the relative efficacy of non-steroidal anti-inflammatory drugs and opioids in the treatment of acute renal colic. BMJ. 2004;328:1401-1408.
Holdgate A, Pollock T. Nonsteroidal anti-inflammatory drugs (NSAIDs) versus opioids for acute renal colic. Cochrane Database Syst Rev 2005;(2):CD004137.
Holdgate A, Pollock T. Nonsteroidal anti-inflammatory drugs (NSAIDs) versus opioids for acute renal colic. Cochrane Library, Issue 1. Chichester (UK): John Wiley & Sons; 2006.
Holmlund D. The pathophysiology of ureteral colic. Scand J Urol Nephrol Suppl. 1983;75:25-27.
Hu Z, Wang C, Xiao Y, et al. NDST1-dependent heparan sulfate regulates BMP signaling and internalization in lung development. J Cell Sci. 2009;122(Pt. 8):1145-1154.
Huben RP, Schellhammer PF. Steroid therapy for ureteral obstruction after aortoiliac graft surgery. J Urol. 1981;125:881-883.
Hwang S, Hu G, Charness ME, et al. Regulation of Na/K-ATPase expression in obstructive uropathy [abstract]. Clin Res. 1993;41:141A.
Hwang SJ, Haas M, Harris HWJr, et al. Transport defects of rabbit medullary thick ascending limb cells in obstructive nephropathy. J Clin Invest. 1993;91:21-28.
Ichikawa I, Brenner BM. Local intrarenal vasoconstrictor-vasodilator interactions in mild partial ureteral obstruction. Am J Physiol. 1979;236:F131-F140.
Ichikawa I, Purkerson ML, Yates J, et al. Dietary protein intake conditions the degree of renal vasoconstriction in acute renal failure caused by ureteral obstruction. Am J Physiol. 1985;249:F54-F61.
Ishidoya S, Morrissey J, McCracken R, et al. Angiotensin II receptor antagonist ameliorates renal tubulointerstitial fibrosis caused by unilateral ureteral obstruction. Kidney Int. 1995;47:1285-1294.
Ito K, Chen J, Chaar M, et al. Renal damage progresses despite improvement of renal function after relief of unilateral ureteral obstruction in adult rats. Am J Physiol. 2004;287:F1283-F1293.
Ito K, Chen J, Khodadadian JJ, et al. Liposome-mediated transfer of nitric oxide synthase gene improves renal function in ureteral obstruction in rats. Kidney Int. 2004;66(4):1365-1375.
Ito K, Chen J, Vaughan EDJr, et al. Dietary L-arginine supplementation improves glomerular filtration rate and renal blood flow after 24 hours of unilateral ureteral obstruction in rats. J Urol. 2004;171:926-930.
Iwai T, Kitamoto K, Teramoto K, et al. Cobalt protoporphyrin attenuates rat obstructive nephropathy: role of cellular infiltration. Urology. 2008;72(2):432-438.
Jaenike J, Bray G. Effects of acute transitory urinary obstruction in the dog. Am J Physiol. 1960;199:1219-1222.
Jaenike JR. The renal functional defect of postobstructive nephropathy. The effects of bilateral ureteral obstruction in the rat. J Clin Invest. 1972;51:2999-3006.
Jarrard DJ, Gerber GS, Lyon ES. Management of acute ureteral obstruction in pregnancy utilizing ultrasound-guided placement of ureteral stents. Urology. 1993;42:263-268.
Jensen AM, Li C, Praetorius HA, et al. Angiotensin II mediates downregulation of aquaporin water channels and key renal sodium transporters in response to urinary tract obstruction. Am J Physiol Renal Physiol. 2006;291(5):F1021-F1032.
Jetty P, Barber GG. Aortitis and bilateral ureteral obstruction after endovascular repair of abdominal aortic aneurysm. J Vasc Surg. 2004;39:1344-1347.
Jogai S, Al-Jassar A, Temmin L. Idiopathic retroperitoneal fibrosis—a potential pitfall for fine needle aspiration cytology. Cytopathology. 2005;16(1):49-50.
Johnson SC, Esclapes M. Sonography of postpartum ovarian vein thrombophlebitis. J Clin Ultrasound. 1998;26:143-149.
Jones DA, George NJ, O’Reilly PH, Barnard RJ. The biphasic nature of renal functional recovery following relief of chronic obstructive uropathy. Br J Urol. 1988;61:192-197.
Jones RA, Schmotzer B, Little SB, Grattan-Smith JD. MRU post-processing. Pediatr Radiol. 2008;38(Suppl. 1):S18-S27.
Jorgensen B, Keller AK, Radvanska E, et al. Reproducibility of contrast enhanced MR renography in children. J Urol. 2006;176:1171-1176.
Joshi HB, Adams S, Obadeyi OO, Rao PN. Nephrostomy tube or ‘JJ’ ureteric stent in ureteric obstruction: assessment of patient perspectives using quality-of-life survey and utility analysis. Eur Urol. 2001;39:695-701.
Jubanyik KJ, Comite F. Extrapelvic endometriosis. Obstet Gynecol Clin. 1997;24:411-440.
Kalika V, Bard RH, Iloreta A, et al. Prediction of renal functional recovery after relief of upper urinary tract obstruction. J Urol. 1981;126:301-305.
Kanal E, Borgstede JP, Barkovich AJ, et al. ACR white paper on magnetic resonance (MR) safety: combined papers of 2002 and 2004. http://www.acr.org. [accessed 08.12.10]
Kaneto H, Morrissey J, Klahr S. Increased expression of TGF-beta 1 mRNA in the obstructed kidney of rats with unilateral ureteral ligation. Kidney Int. 1993;44:313-321.
Kaneto H, Morrissey J, McCracken R, et al. The expression of mRNA for tumor necrosis factor-alpha increases in the obstructed kidney of rats soon after unilateral ureteral ligation. Nephrology. 1996;2:161-166.
Kaplon DM, Lasser MS, Sigman M, et al. Renal parenchyma thickness: a rapid estimation of renal function on computed tomography. Int Braz J Urol. 2009;35(1):3-8.
Karabacakoglu A, Karakose S, Ince O, et al. Diagnostic value of diuretic enhanced excretory MR urography in patients with obstructive uropathy. Eur J Radiol. 2004;52:320-327.
Karam M, Feustel PJ, Goldfarb CR, Kogan BA. Diuretic renogram clearance half times in the diagnosis of obstructive uropathy: effect of age and previous surgery. Nucl Med Commun. 2003;24:797-807.
Kardar AH, Kattan S, Lindstedt E, Hanash K. Steroid therapy for idiopathic retroperitoneal fibrosis: dose and duration. J Urol. 2002;168:550-555.
Kashi SH, Irving HC, Sadek SA. Does the Whitaker test add to antegrade pyelography in the investigation of collecting system dilatation in renal allografts? Br J Radiol. 1993;66:877-881.
Kasinath BS, Mariappan MM, Sataranatarajan K, et al. mRNA translation: unexplored territory in renal science. J Am Soc Nephrol. 2006;17(12):3281-3292.
Katz G, Meirow D. An incompletely identified combined urogynecological malformation presenting as anuria. J Urol. 1993;149:610-612.
Kelleher JP, Shah V, Godley ML, et al. Urinary endothelin (ET1) in complete ureteric obstruction in the miniature pig. Urol Res. 1992;20:63-65.
Kemper J, Regier M, Stork A, et al. Improved visualization of the urinary tract in multidetector CT urography (MDCTU): analysis of individual acquisition delay and opacification using furosemide and low-dose test images. J Comput Assist Tomogr. 2006;30(5):751-757.
Kenawi MM, Williams DI. Circumcaval ureter: a report of four cases in children with a review of the literature and a new classification. Br J Urol. 1976;48:183-192.
Kerr WSJr. Effect of complete ureteral obstruction for one week on kidney function. J Appl Physiol. 1954;6:762-772.
Khalaf IM, Shokeir AA, El-Gyoushi FI, et al. Recoverability of renal function after treatment of adult patients with unilateral obstructive uropathy and normal contralateral kidney: a prospective study. Urology. 2004;64:664-668.
Khanna PC, Karnik ND, Jankharia BG, et al. Magnetic resonance urography (MRU) versus intravenous urography (IVU) in obstructive uropathy: a prospective study of 30 cases. J Assoc Physicians India. 2005;53:527-534.
Kim DS, Noh JY, Jeong HJ, et al. Elastin content of the renal pelvis and ureter determines post-pyeloplasty recovery. J Urol. 2005;173(3):962-966.
Kim SW, Cho SH, Oh BS, et al. Diminished renal expression of aquaporin water channels in rats with experimental bilateral ureteral obstruction. J Am Soc Nephrol. 2001;12:2019-2028.
Kim SW, Lee JU, Kim SZ, et al. Enhanced Dendroaspis natriuretic peptide immunoreactivity in experimental ureteral obstruction. Nephron. 2002;92:369-372.
Kim SW, Lee JU, Park JW, et al. Increased expression of atrial natriuretic peptide in the kidney of rats with bilateral ureteral obstruction. Kidney Int. 2001;59:1274-1282.
King LS, Choi M, Fernandez PC, et al. Defective urinary concentrating ability due to a complete deficiency of aquaporin-1. N Engl J Med. 2001;345:175-179.
Kinkel K, Frei KA, Balleyguier C, Chapron C. Diagnosis of endometriosis with imaging: a review. Eur Radiol. 2006;16:285-298.
Kiratli PO, Orhan D, Gedik GK, Tekgul S. Relation between radionuclide imaging and pathologic findings of ureteropelvic junction obstruction in neonatal hydronephrosis. Scand J Urol Nephrol. 2008;42(3):249-256.
Klahr S, Morrissey J. Obstructive nephropathy and renal fibrosis: the role of bone morphogenic protein-7 and hepatocyte growth factor. Kidney Int. 2003;87:S105-S112.
Klahr S, Morrissey J. L-Arginine as a therapeutic tool in kidney disease. Semin Nephrol. 2004;24:389-394.
Klahr S, Schwab S, Stokes TJ. Metabolic adaptations of the nephron in renal disease. Kidney Int. 1986;29:80-89.
Klein FA, Smith MJV, Kasenetz I. Pelvic lipomatosis: 35 year experience. J Urol. 1988;139:998-1001.
Klein RS, Cattolica EV. Ureteral endometriosis. Urology. 1979;13:477-482.
Klotman PE, Smith SR, Volpp BD, et al. Thromboxane synthetase inhibition improves function of hydronephrotic rat kidneys. Am J Physiol. 1986;250:F282-F287.
Klutke J, Kavoussi LR, Albala DM, Clayman RV. Laparoscopic treatment of ureteral obstruction secondary to ovarian remnant syndrome. J Urol. 1993;149:827-829.
Knoers NAVM. Hyperactive vasopressin receptors and disturbed water homeostasis. N Engl J Med. 2005;352:1847-1850.
Knutson T, Hawas B. Horseshoe kidney with a circumcaval ureter. Scand J Urol Nephrol. 2004;38:348-350.
Koch MO, Coussens D, Burnett L. The ovarian remnant syndrome and ureteral obstruction: medical management. J Urol. 1994;152:158-160.
Koep L, Zuidema GD. The clinical significance of retroperitoneal fibrosis. Surgery. 1977;81:250-257.
Koff SA, Peller PA. Diagnostic criteria for assessing obstruction in the newborn with unilateral hydronephrosis using the renal growth-renal function index. J Urol. 1995;154:662-666.
Kretkowski R, Shah N. Testicular vein syndrome unusual cause of hydronephrosis. Urology. 1977;10:253-254.
Krishnamurthi G, Stantz KM, Steinmetz R, et al. Functional imaging in small animals using x-ray computed tomography: study of physiologic measurement reproducibility. IEEE Trans Med Imaging. 2005;24(7):832-843.
Ku JH, Lee SW, Jeon HG, et al. Percutaneous nephrostomy versus indwelling ureteral stents in the management of extrinsic ureteral obstruction in advanced malignancies: are there differences? Urology. 2004;64:895-899.
Kuhnle U, Gusariso G, Sonega M, et al. Transient pseudohypoaldosteronism in obstructive renal disease with transient reduction of lymphocytic aldosterone receptors. Results in two affected infants. Horm Res. 1993;39:152-155.
Kumar S, Bhandar M. Selection of operative procedure for circumcaval ureter (type I). Br J Urol. 1985;57:399-401.
Kuwabara N, Tamada S, Iwai T, et al. Attenuation of renal fibrosis by curcumin in rat obstructive nephropathy. Urology. 2006;67(2):440-446.
Kwon TH, Frøkiær J, Han JS, et al. Decreased abundance of major Na+ transporters in kidneys of rats with ischemia-induced acute renal failure. Am J Physiol. 2000;278:F925-F939.
Labardini MM, Ratliff RK. The abdominal aortic aneurysm and the ureter. J Urol. 1967;98:590-596.
Ladefoged O, Djurhuus JC. Morphology of the upper urinary tract in experimental hydronephrosis in pigs. Acta Chir Scand. 1976;472:29-35.
Lafferty HW, Angioli R, Rudolph J, Penalver M. Ovarian remnant syndrome: experience at Jackson Memorial Hospital, University of Miami, 1985 through 1993. Am J Obstet Gynecol. 1996;174:641-645.
Laing FC, Jeffrey RB, Wing VW. Ultrasound versus excretory urography in evaluating acute flank pain. Radiology. 1985;154:613-616.
Lam A, French M, Charnock FM. Bilateral ureteric obstruction due to recurrent endometriosis associated with hormone replacement therapy. Aust N Z J Obstet Gynaecol. 1992;32:83-84.
Lambie SH, DeNinzio M, Williams JH, McIntyre CW. Obstructive uropathy associated with endovascular repair of an inflammatory abdominal aortic aneurysm. Nephrol Dial Transplant. 2004;19:745-746.
Lanzone JA, Gulmi FA, Chou SY, et al. Renal hemodynamics in acute unilateral ureteral obstruction: contribution of endothelium-derived relaxing factor. J Urol. 1995;153:2055-2059.
Lasky J. Pirfenidone. IDrugs. 2004;7:166-172.
Lasoye TA, Sedgwick PM, Patel N, et al. Management of acute renal colic in the UK: a questionnaire survey. BMC Emerg Med. 2004;4:5.
Le Tohic A, Chis C, Yazbeck C, et al. Bladder endometriosis: diagnosis and treatment. A series of 24 patients. Gynecol Obstet Fertil. 2009;37(3):216-221.
Leahy AL, Ryan PC, McEntee GM, et al. Renal injury and recovery in partial ureteric obstruction. J Urol. 1989;142:199-203.
Lee C, Gnanasegaram D, Maloba M. Best evidence topic report. Rectal or intravenous non-steroidal anti-inflammatory drugs in acute renal colic. Emerg Med J. 2005;22(9):653-654.
Leocadio DE, Ruckle HC, Curhan DJ. Ureteral obstruction by uterine artery aneurysm in third trimester of pregnancy. Urol. 2006;67:1290.e7-1290.e9.
Leyendecker JR, Gorengaut V, Brown JJ. MR imaging of maternal diseases of the abdomen and pelvis during pregnancy and the immediate postpartum period. Radiographics. 2004;24(5):1301-1316.
Li C, Wang W, Knepper MA, et al. Downregulation of renal aquaporins in response to unilateral ureteral obstruction. Am J Physiol. 2003;284:F1066-F1079.
Li C, Wang W, Kwon TH, et al. Downregulation of AQP1, -2, and -3 after ureteral obstruction is associated with a long-term urine-concentrating defect. Am J Physiol. 2001;281:F163-F171.
Li C, Wang W, Norregaard R, et al. Altered expression of epithelial sodium channel in rats with bilateral or unilateral ureteral obstruction. Am J Physiol Renal Physiol. 2007;293(1):F333-F341.
Lin EP, Bhatt S, Dogra VS, Rubens DJ. Sonography of urolithiasis and hydronephrosis. Ultrasound Clin. 2007;2:1-16.
Lin L, Bagley DH, Liu JB. Role of endoluminal sonography in evaluation of obstruction of the ureteropelvic junction. AJR Am J Roentgenol. 2008;4:1250-1254.
Liu JS, Hrebinko RL. The use of 2 ipsilateral ureteral stents for relief of ureteral obstruction from extrinsic compression. J Urol. 1998;159:179-181.
Liu Y, Ghesani NV, Shurick JH, et al. The F + 0 protocol for diuretic renography results in fewer interrupted studies due to voiding than the F − 15 protocol. J Nucl Med. 2005;46(8):1317-1320.
Loddenkemper K, Hoyer B, Lodenkemper C, et al. A case of Erdheim-Chester disease initially mistaken for Ormond’s disease. Nat Clin Pract Rheumatol. 2008;4:50-55.
Loo MH, Egan D, Vaughan EDJr, et al. The effect of the thromboxane A2 synthesis inhibitor OKY-046 on renal function in rabbits following release of unilateral ureteral obstruction. J Urol. 1987;137:571-576.
Loo MH, Vaughan ED. Obstructive nephropathy and postobstructive diuresis. AUA Update Series. 4, 1985. lesson 9
Loughlin KR. Management of urologic problems in the pregnant patient. AUA Update Series. 1997:10-15. lesson 16
Lutaif NA, Yu L, Abdulkader RC. Factors influencing the non-recovery of renal function after the relief of urinary tract obstruction in women with cancer of cervix. Ren Fail. 2003;25:215-223.
Lutin EM, Haramati N, Frager D, et al. CT diagnosis of circumcaval ureter. AJR Am J Roentgenol. 1988;150:591-594.
Ma MC, Huang HS, Chen CF. Impaired renal sensory responses after unilateral ureteral obstruction in the rat. J Am Soc Nephrol. 2002;13:1008-1016.
Maeda T, Uchida Y, Nakajima F. Vesical endometriosis following the menopause. Int Urogynecol J Pelvic Floor Dysfunct. 2009;20:1515-1517.
Mandell J, Peters CA, Estroff JA, et al. Human fetal compensatory renal growth. J Urol. 1993;150:790-792.
Manucha W, Carrizo L, Alvarez S, et al. Effect of losartan pretreatment on kidney lipid content after unilateral obstruction in rats. Cell Mol Biol (Noisy-le-grand). 2005;51(6):539-545.
Manucha W, Oliveros L, Carrizo L, et al. Losartan modulation on NOS isoforms and COX-2 expression in early renal fibrogenesis in unilateral obstruction. Kidney Int. 2004;65:2091-2107.
Marcelli F, Collinet P, Vinatier D, et al. Ureteric and bladder involvement of deep pelvic endometriosis. Value of multidisciplinary surgical management. Prog Urol. 2006;16:588-593.
Marcolongo R, Tavolini IM, Laveder F, et al. Immunosuppressive therapy for idiopathic retroperitoneal fibrosis: a retrospective analysis of 26 cases. Am J Med. 2004;116:194-197.
Marino R, Mooppan UM, Zein TA, et al. Urological manifestations of isolated iliac artery aneurysms. J Urol. 1987;137:232-234.
Martorana D, Vaglio A, Greco P, et al. Chronic periaortitis and HLA-DRB1*03: another clue to an autoimmune origin. Arthritis Rheum. 2006;55(1):126-130.
Marver D, Bernabe J. Inhibition of Na+/K+-ATPase by PGE2 [abstract]. J Am Soc Nephrol. 1992;3:500A.
Matlaga BR, Shah OD, Hart LJ, Assimos DG. Ileal ureter substitution: a contemporary series. Urology. 2003;62:998-1001.
McAleer SJ, Loughlin KR. Nephrolithiasis and pregnancy. Curr Opin Urol. 2004;14:123-127.
McCullough TC, May NR, Metro MJ, et al. Serum creatinine predicts success in retrograde ureteral stent placement in patients with pelvic malignancies. Urology. 2008;72:370-373.
Mearini L, Rosi P, Zucchi A, et al. Color Doppler ultrasonography in the diagnosis of vascular abnormalities associated with ureteropelvic junction obstruction. J Endourol. 2003;17:745-750.
Meister B, Lippoldt A, Bunnemann B, et al. Cellular expression of angiotensin type-1 receptor mRNA in the kidney. Kidney Int. 1993;44:331-336.
Melzi ML, Guez S, Sersale G, et al. Acute pyelonephritis as a cause of hyponatremia/hyperkalemia in young infants with urinary tract malformations. Pediatr Infect Dis J. 1995;14:56-59.
Mense S. Basic neurobiologic mechanisms of pain and analgesia. Am J Med. 1983;75:4-14.
Meyer JI, Wilbur AC, Lichtenberg R. Ureteric obstruction by the right testicular vein: CT diagnosis. Urol Radiol. 1992;13:233-236.
Micali S, Grande M, Sighinolfi MC, et al. Medical therapy of urolithiasis. J Endourol. 2006;20:841-847.
Micali S, Grande M, Sighinolfi MC, et al. Efficacy of expulsive therapy using nifedipine or tamsulosin, both associated with ketoprofene, after shock wave lithotripsy of ureteral stones. Urol Res. 2007;35:133-137.
Middleton GW, Beamon CR, Panko WB, et al. Effects of ureteral obstruction on the renal metabolism of alpha-ketoglutarate and other substrates in vivo. Invest Urol. 1977;14:255-262.
Miller OF, Smith LJ, Ferrara EX, et al. Presentation of idiopathic retroperitoneal fibrosis in the pediatric population. J Pediatr Surg. 2003;38(11):1685-1688.
Mineta K, Nomura M, Fujimoto N, et al. Ureteral obstruction due to retroperitoneal fibrosis secondary to a solitary internal iliac aneurysm. Int J Urol. 2004;11:1024-1027.
Misseri R, Rink RC, Meldrum DR, et al. Research review: inflammatory mediators and growth factors in obstructive renal injury. J Surg Res. 2004;119:149-159.
Mitterberger M, Pinggera GM, Neururer R, et al. Comparison of contrast-enhanced color Doppler imaging (CDI), computed tomography (CT), and magnetic resonance imaging (MRI) for the detection of crossing vessels in patients with ureteropelvic junction obstruction (UPJO). Eur Urol. 2008;6:1254-1260.
Miyajima A, Chen J, Poppas DP, et al. Role of nitric oxide in renal tubular apoptosis of unilateral ureteral obstruction. Kidney Int. 2001;59:1290-1303.
Mokhmalji H, Braun PM, Martinez Portillo FJ, et al. Percutaneous nephrostomy versus ureteral stents for diversion of hydronephrosis caused by stones: a prospective, randomized clinical trial. J Urol. 2001;165:1088-1092.
Moller JC. Proximal tubules in long term compensatory renal growth. Quantitative light and electron microscopic analyses. APMIS Suppl. 1988;4:82-86.
Monev S. Idiopathic retroperitoneal fibrosis: prompt diagnosis preserves organ function. Cleve Clin J Med. 2002;69:160-166.
Moody TE, Vaughan EDJr, Wyker AT, et al. The role of intrarenal angiotensin II in the hemodynamic response to unilateral obstructive uropathy. Invest Urol. 1977;14:390-397.
Moody TE, Vaughn EDJr, Gillenwater JY. Relationship between renal blood flow and ureteral pressure during 18 hours of total unilateral uretheral occlusion. Implications for changing sites of increased renal resistance. Invest Urol. 1975;13:246-251.
Moon JA, Kim HT, Cho IS, et al. IN-1130, a novel transforming growth factor-beta type I receptor kinase (ALK5) inhibitor, suppresses renal fibrosis in obstructive nephropathy. Kidney Int. 2006;70(7):1234-1243.
Morretin LB, Wilson M. Pelvic lipomatosis. AJR Am J Roentgenol. 1971;113:181.
Morrissey J, Klahr S. Rapid communication. Enalapril decreases nuclear factor kappa B activation in the kidney with ureteral obstruction. Kidney Int. 1997;52:926-933.
Morrissey JJ, Klahr S. Differential effects of ACE and AT1 receptor inhibition on chemoattractant and adhesion molecule synthesis. Am J Physiol. 1998;274:F580-F586.
Morrissey J, Klahr S. Transcription factor NF-kappaB regulation of renal fibrosis during ureteral obstruction. Semin Nephrol. 1998;18:603-611.
Mortensen J, Djurhuus JC, Laursen H, Bisballe S. The relationship between pressure and flow in the normal pig renal pelvis: An experimental study of the range of normal pressures. Scand J Urol Nephrol. 1983;17:369-372.
Mulligan SA, Holley HC, Koehler RE, et al. CT and MR imaging in the evaluation of retroperitoneal fibrosis. J Comput Assist Tomogr. 1989;13:277-281.
Mulroney SE, Haramati A, Roberts CTJr, LeRoith D. Renal IGF1 mRNA levels are enhanced following unilateral nephrectomy in immature rats but not adult rats. Endocrinology. 1991;128:2660-2662.
Murakumo M, Nonomuran K, Yamashita T, et al. Structural changes of collagen components and diminution of nerves in congenital ureteropelvic junction obstruction. J Urol. 1997;157:1963-1968.
Nakada SY, Jerde TJ, Jacobson LM, et al. Cyclooxygenase-2 expression is up-regulated with obstruction. J Urol. 2002;168:1226-1229.
Nam UC, Chang SG. Clinical study of insulin-like growth factor 1 and compensatory renal growth in patients with donor nephrectomy. Transplant Proc. 1999;31:2114-2117.
Nevelsteen A, Lacroix H, Stockx L, et al. Inflammatory abdominal aortic aneurysm and bilateral complete ureteral obstruction: treatment by endovascular graft and bilateral ureteral stenting. Ann Vasc Surg. 1999;13:222-224.
Nezhat C, Kearney S, Malik S, et al. Laparoscopic management of ovarian remnant. Fertil Steril. 2005;83:973-978.
Nguyen DH, Koleilat N, Gonzalez R. Retroiliac ureter in a male newborn with multiple genitourinary anomalies: case report and review of the literature. J Urol. 1989;141:1400-1403.
Nielsen S, Agre P. The aquaporin family of water channels in kidney. Kidney Int. 1995;48:1057-1068.
Nishimoto G, Zelenina M, Li D, et al. Arginine vasopressin stimulates phosphorylation of aquaphorin-2 in rat renal tissue. Am J Physiol. 1999;276:F254-F259.
Nito H, Descoeudres C, Kurokawa K, Massry SG. Effect of unilateral obstruction on renal cell metabolism and function. J Lab Clin Med. 1978;91:60-71.
Nolte-Ernsting C, Staatz G, Wildberger J, et al. MR-urography and CT-urography: principles, examination techniques, applications. Rofo. 2003;175(2):211-222.
Nørregaard R, Jensen BL, Topcu SO, et al. Cyclooxygenase type 2 is increased in obstructed rat and human ureter and contributes to pelvic pressure increase after obstruction. Kidney Int. 2006;70(5):872-881.
Nørregaard R, Jensen BL, Topcu SO, et al. COX-2 activity transiently contributes to increased water and NaCl excretion in the polyuric phase after release of ureteral obstruction. Am J Physiol Renal Physiol. 2007;292(5):F1322-F1333.
Nyman MA, Schwenk NM, Silverstein MD. Management of urinary retention: rapid versus gradual decompression and risk of complications. Mayo Clin Proc. 1997;72:951-956.
O’Dell-Anderson KJ, Twardock R, Grimm JB, et al. Determination of glomerular filtration rate in dogs using contrast-enhanced computed tomography. Vet Radiol Ultrasound. 2006;47(2):127-135.
Ochoa Urdangarain O, Hermida Pérez JA, et al. Obstructive anuria secondary to left external iliac artery aneurysm. Case report. Arch Esp Urol. 2006;59:281-284.
Ormond JK. Bilateral ureteral obstruction due to envelopment and compression by inflammatory retroperitoneal process. J Urol. 1948;59:1072-1079.
Ormond JK. Idiopathic retroperitoneal fibrosis: an established clinical entity. JAMA. 1960;174:1561-1568.
Ortapamuk H, Naldoken S, Tekdogan UY, et al. Differential renal function in the prediction of recovery in adult obstructed kidneys after pyeloplasty. Ann Nucl Med. 2003;17(8):663-668.
Pastor-Navarro H, Gimnez-Bachs JM, Onate Moreno MJ, et al. Update on the diagnosis and treatment of bladder endometriosis. Int Urogynecol J. 2007;18:949-954.
Pearle MS, Pierce HL, Miller GL, et al. Optimal method of urgent decompression of the collecting system for obstruction and infection due to ureteral calculi. J Urol. 1998;160:1260-1264.
Pennell RC, Hollier LH, Lie JT, et al. Inflammatory abdominal aortic aneurysms: a thirty-year review. J Vasc Surg. 1985;2:859-869.
Perlmutter A, Miller L, Trimble LA, et al. Toradol, an NSAID used for renal colic, decreases renal perfusion and ureteral pressure in a canine model of unilateral ureteral obstruction. J Urol. 1993;149:926-930.
Peters CA, Gaertner RC, Carr MC, Mandell J. Fetal compensatory renal growth due to unilateral ureteral obstruction. J Urol. 1993;150:597-600.
Petty KJ, Kokko JP, Marver D. Secondary effect of aldosterone on Na+-K+-ATPase activity in the rabbit cortical collecting tubule. J Clin Invest. 1981;68:1514-1521.
Pimental JLJr, Sundell CL, Wang S, et al. Role of angiotensin II in the expression and regulation of transforming growth factor-beta in obstructive nephropathy. Kidney Int. 1995;48:1233-1246.
Platt JF, Ellis JH, Rubin JM. Examination of native kidneys with duplex Doppler ultrasound. Semin Ultrasound CT MR. 1991;12:308-318.
Platt JF, Rubin JM, Ellis JH. Acute renal obstruction: evaluation with intrarenal duplex Doppler and conventional US. Radiology. 1993;186:685-688.
Pohl HG, Joyce GF, Wise M, Cilento BGJr. Pediatric urological disorders. In: Litwin MS, Saigal CS, editors. Urological diseases in America. Washington (DC): National Institutes of Health; 2007:408-411.
Polascik T, Chen RN. Laparoscopic ureteroureterostomy for retrocaval ureter. J Urol. 1998;160:121-122.
Prcić A, Aganović D, Junuzović D, Kulovac B. Comparative efficiency study, safety and usage of tested drugs in patients with renal colic. Med Arh. 2006;60:37-40.
Purcell H, Bastani B, Harris KP, et al. Cellular distribution of H+-ATPase following acute unilateral ureteral obstruction in rats. Am J Physiol. 1991;261:F365-F376.
Purkerson ML, Blaine EH, Stokes TJ, et al. Role of atrial peptide in the natriuresis and diuresis that follows relief of obstruction in rat. Am J Physiol. 1989;256:F583-F589.
Purkerson ML, Klahr S. Prior inhibition of vasoconstrictors normalizes GFR in postobstructed kidneys. Kidney Int. 1989;35:1305-1314.
Rehring TF, Brewster DC, Kaufman JA, et al. Regression of perianeurysmal fibrosis and ureteral dilatation following endovascular repair of inflammatory abdominal aortic aneurysm. Ann Vasc Surg. 2001;15:591-593.
Reyes A, Klahr S. Role of platelet-activating factor in renal function in normal rats and rats with bilateral ureteral obstruction. Proc Soc Exp Biol Med. 1991;198:572-578.
Reyes A, Klahr S. Renal function after release of ureteral obstruction: role of endothelin and the renal artery endothelium. Kidney Int. 1992;42:632-638.
Rivlin M, Miller JD, Krueger RP, et al. Leuprolide acetate in the management of ureteral obstruction caused by endometriosis. Obstet Gynecol. 1990;75:532-536.
Rohrschneider WK, Haufe S, Wiesel M, et al. Functional and morphologic evaluation of congenital urinary tract dilatation by using combined static-dynamic MR urography: findings in kidneys with a single collecting system. Radiology. 2002;224:683-694.
Rokaw MD, Sarac E, Lechman E, et al. Chronic regulation of transepithelial Na+ transport by the rate of apical Na+ entry. Am J Physiol. 1996;270:C600-C607.
Rouprêt M, Daudon M, Hupertan V, et al. Can ureteral stent encrustation analysis predict urinary stone composition? Urology. 2005;66(2):246-251.
Rosevear HM, Kim SP, Wenzler DL, et al. Retrograde ureteral stents for extrinsic ureteral obstruction: nine years’ experience at University of Michigan. Urology. 2007;70(5):846-850.
Rubinstein I, Cavalcanti AG, Canalini AF, et al. Left retrocaval ureter associated with inferior vena cava duplication. J Urol. 1999;162:1373-1374.
Ruegg CE, Mandel LJ. Bulk isolation of renal PCT and PST. I. Glucose-dependent metabolic differences. Am J Physiol. 1990;259:F164-F175.
Ryan PC, Fitzpatrick JM. Partial ureteric obstruction: a new variable canine experimental model. J Urol. 1987;137:1034-1038.
Ryan PC, Maher K, Hurley GD, Fitzpatrick JM. The Whitaker test: experimental analysis in a canine model of partial ureteric obstruction. J Urol. 1989;141:387-390.
Ryndin I, Gulmi FA, Chou SY, et al. Renal responses to atrial natriuretic peptide are preserved in bilateral ureteral obstruction and augmented by neural endopeptidase inhibition. J Urol. 2005;173:651-656.
Sacks SA, Drenick EJ. Pelvic lipomatosis: effect of diet. Urology. 1975;6:609-615.
Sadowski EA, Bennett LK, Chan MR, et al. Nephrogenic systemic fibrosis: risk factors and incidence estimation. Radiology. 2007;243:148-157.
Saldino RM, Palubinskas AJ. Medial placement of the ureter: a normal variant, which may simulate retroperitoneal fibrosis. J Urol. 1972;107:582-585.
Salvemini D, Seibert K, Masferrer JL, et al. Endogenous nitric oxide enhances prostaglandin production in a model of renal inflammation. J Clin Invest. 1994;93:1940-1947.
Sanchez Merino JM, Guillan MC, Garcia AJ. The treatment of bladder endometriosis. Arch Esp Urol. 2005;58:189-194.
Sant GR, Heaney JA, Parkhurst EC, Blaivas JG. Obstructive uropathy—a potentially serious complication of reconstruction vascular surgery. J Urol. 1983;129:16-22.
Satin AJ, Seiken GL, Cunningham FG. Reversible hypertension in pregnancy caused by obstructive uropathy. Obstet Gynecol. 1993;81:823-825.
Schein M, Saadia R. Ureteral obstruction after abdominal aortic surgery. Am J Surg. 1991;162:86-89.
Scheel PJJr, Piccini J, Rahman MH, et al. Combined prednisone and mycophenolate mofetil treatment for retroperitoneal fibrosis. J Urol. 2007;178(1):140-143.
Schlossberg SM, Vaughan EDJr. The mechanism of unilateral post-obstructive diuresis. J Urol. 1984;131:534-536.
Schneider A, Toulopidis S, Papatsoris AG, et al. Endometriosis of the urinary tract in women of reproductive age. Int J Urol. 2006;13:902-904.
Schreiner GF, Harris KP, Purkerson ML, et al. Immunological aspects of acute ureteral obstruction: Immune cell infiltrate in the kidney. Kidney Int. 1988;34:487-493.
Schulam PG, Litwin MS, Saigal CS, editors. Urological diseases in America. National Institutes of Health, Washington (DC), 2007, 323-332.
Schulman CC. The ureter. In: O’Donnell B, Koff SA, editors. Pediatric urology. 3rd ed. Oxford (UK): Butterworth-Heinemann; 1997:409-410.
Schwiebert EM, Kishore BK. Extracellular nucleotide signaling along the renal epithelium. Am J Physiol. 2001;280:F945-F963.
Sechi LA, Grady EF, Griffin CA, et al. Distribution of angiotensin II receptor subtypes in rat and human kidney. Am J Physiol. 1992;262:F236-F240.
Sener RN. Nonobstructive right circumcaval ureter associated with double inferior vena cava. Urology. 1993;41:356-360.
Seracchioli R, Mabrouk M, Montanari G, et al. Conservative laparoscopic management of urinary tract endometriosis (UTE): surgical outcome and long-term follow-up. Fertil Steril. 2010;94(3):856-861.
Serel TA, Savas C, Delibas N, et al. Free insulin like growth factor-1 and unilateral complete ureteral obstruction in the rat. Urology. 2000;56:863-866.
Shapiro SR, Bennett AH. Recovery of renal function after prolonged unilateral ureteral obstruction. J Urol. 1976;115:136-140.
Sharma AK, Mauer SM, Kim Y, Michael AF. Interstitial fibrosis in obstructive nephropathy. Kidney Int. 1993;44:774-788.
Shellock FG, Kanal E. Safety of magnetic resonance imaging contrast agents. J Magn Reson Imaging. 1999;10:477-484.
Shine S. Urinary calculus: IVU vs. CT renal stone? A critically appraised topic. Abdom Imaging. 2008;1:41-43.
Shokeir AA, Shoma AM, Abubieh EA, et al. Recoverability of renal function after relief of acute complete ureteral obstruction: clinical prospective study of the role of renal resistive index. Urology. 2002;59:506-510.
Simons PCG, van Overhagen H, Bruijninckx CMA, et al. Periaortitis with ureteral obstruction after endovascular repair of an abdominal aortic aneurysm. AJR Am J Roentgenol. 2002;179:118-120.
Singh A, Alter HJ, Littlepage A. A systematic review of medical therapy to facilitate passage of ureteral calculi. Ann Emerg Med. 2007;50:552-563.
Singh I, Gupta NP, Hemal AK, et al. Severely encrusted polyurethane ureteral stents: management and analysis of potential risk factors. Urology. 2001;58:526-531.
Sinuani I, Averbukh Z, Gitelman I, et al. Mesangial cells initiate compensatory renal tubular hypertrophy via IL-10-induced TGF-beta secretion: effect of the immunomodulator AS101 on this process. Am J Physiol Renal Physiol. 2006;291(2):F384-F394.
Sjodin JG, Wahlberg J, Persson EG. The effect of indomethacin on glomerular capillary pressure and pelvic pressure during ureteral obstruction. J Urol. 1982;127:1017-1020.
Smith AK, Hansel DE, Jones JS. Role of cystitis cystica et glandularis and intestinal metaplasia in development of bladder carcinoma. Oncology. 2008;71(5):915-918.
Smith RC, Verga M, Dalrymple N, et al. Acute ureteral obstruction: value of secondary signs of helical unenhanced CT. AJR Am J Roentgenol. 1996;167:1109-1113.
Solez K, Ponchak S, Buono RA, et al. Inner medullary plasma flow in the kidney with ureteral obstruction. Am J Physiol. 1976;231:1315-1321.
Sonnenberg H, Wilson DR. The role of the medullary collecting ducts in postobstructive diuresis. J Clin Invest. 1976;57:1564-1574.
Sozen S, Gurocak S, Uzum N, et al. The importance of reevaluation in patients with cystitis glandularis associated with pelvic lipomatosis: a case report. Urol Oncol. 2004;22:428-430.
Spencer JA, Chahal R, Kelly A, et al. Evaluation of painful hydronephrosis in pregnancy: magnetic resonance urographic patterns in physiological dilatation versus calculous obstruction. J Urol. 2004;171:256-260.
Sperling H, Becker G, Heemann U, et al. The Whitaker test, a useful tool in renal grafts? Urology. 2000;56:49-52.
Springhart WP, Marguet CG, Sur RL, et al. Forced versus minimal intravenous hydration in the management of acute renal colic: a randomized trial. J Endourol. 2006;20:713-716.
Stacul F, Rossi A, Cova MA. CT urography: the end of IVU? Radiol Med. 2008;5:658-669.
Stebbing J, Notley RG. Noncalculous obstruction due to involvement of the ureter in endometriosis. Eur Urol. 1995;28:122-125.
Stecker JFJr, Vaughan EDJr, Gillenwater JY. Alteration in renal metabolism occurring in ureteral obstruction in vivo. Surg Gynecol Obstet. 1971;133:846-848.
Stenberg A, Bohman SO, Morsing P, et al. Back-leak of pelvic urine to the bloodstream. Acta Physiol Scand. 1988;134:223-234.
Stein AL, Bardawil RG, Silverman SG, Cibas ES. Fine needle aspiration biopsy of idiopathic retroperitoneal fibrosis. Acta Cytol. 1997;41(2):461-466.
Stephan M, Conrad S, Eggert T, et al. Urinary concentration and tissue messenger RNA expression of monocyte chemoattractant protein-1 as an indicator of the degree of hydronephrotic atrophy in partial ureteral obstruction. J Urol. 2002;167:1497-1502.
Sterrett SP, Nakada SY. Medical expulsive therapy. Semin Nephrol. 2008;28:192-199.
Stifelman MD, Shah O, Mufarrij P, Lipkin M. Minimally invasive management of retroperitoneal fibrosis. Urology. 2008;71(2):201-204.
Storch S, Saggi S, Megyesi J, et al. Ureteral obstruction decreases renal prepro-epidermal growth factor and Tamm-Horsfall expression. Kidney Int. 1992;42:89-94.
Strandhoy JW, Ott CE, Schneider EG, et al. Effects of prostaglandins E1 and E2 on renal sodium reabsorption and Starling forces. Am J Physiol. 1974;226:1015-1021.
Swartz RD, Lake AM, Roberts WW, et al. Idiopathic retroperitoneal fibrosis: a role for mycophenolate mofetil. Clin Nephrol. 2008;69(4):260-268.
Susmano DE, Dolin EH. Computed tomography in diagnosis of pelvic lipomatosis. Urology. 1979;13:215-220.
Sutaria PM, Ohebshalom M, McCaffrey TA, et al. Transforming growth factor-beta receptor types I and II are expressed in renal tubules and are increased after chronic unilateral ureteral obstruction. Life Sci. 1998;62:1965-1972.
Syed N, Gulmi F, Chou S, et al. Renal actions of endothelin-1 under endothelin receptor blockade by BE-18257B. J Urol. 1998;159:563-566.
Takeuchi M, Matsuzaki K, Uehara H, Nishitani H. Malignant transformation of pelvic endometriosis: MR imaging findings and pathologic correlation. Radiographics. 2006;26(2):407-417.
Taki M, Goldsmith DI, Spitzer A. Impact of age on effects of ureteral obstruction on renal function. Kidney Int. 1983;24:602-609.
Tamada S, Asai T, Kuwabara N, et al. Molecular mechanisms and therapeutic strategies of chronic renal injury: the role of nuclear factor kappaB activation in the development of renal fibrosis. J Pharmacol Sci. 2006;100(1):17-21.
Tan PH, Chang GS, Tay AH. Pathology of urinary tract malformations in a paediatric autopsy series. Ann Acad Med Singapore. 1994;23:838-843.
Tanner GA. Tubuloglomerular feedback after nephron or ureteral obstruction. Am J Physiol. 1985;248:F688-F697.
Tartaglia LA, Weber RF, Figari IS, et al. The two different receptors for tumor necrosis factor mediate distinct cellular responses. Proc Natl Acad Sci U S A. 1991;88:9292-9296.
Thirakomen K, Kozlov N, Arruda JA, et al. Renal hydrogen ion secretion after release of unilateral ureteral obstruction. Am J Physiol. 1976;231:1233-1239.
Thompson A, Gough DCS. The use of renal scintigraphy in assessing the potential for recovery in the obstructed renal tract in children. BJU Int. 2001;87:853-856.
Thornhill BA, Burt LE, Chen C, et al. Variable chronic partial ureteral obstruction in the neonatal rat: a new model of uretoropelvic junction obstruction. Kidney Int. 2005;67:42-52.
Toland KC, Pelander WM, Mohr SJ. Postpartum ovarian vein thrombosis presenting as ureteral obstruction: a case report and review of the literature. J Urol. 1993;149:1538-1540.
Tong RSK, Larner T, Finlay M, et al. Pelvic lipomatosis associated with proliferative cystitis occurring in two brothers. Urology. 2002;59:602xvii-xx.
Trachtman H, Weiser AC, Valderrama E, et al. Prevention of renal fibrosis by spironolactone in mice with complete unilateral ureteral obstruction. J Urol. 2004;172:1590-1594.
Tremel F, Caravel JP, Siche JP, et al. Diagnostic value of renal scintigraphy with MAG 3 and DTPA in the diagnosis of renal artery stenosis. Arch Mal Coeur Vaiss. 1996;89(8):1035-1039.
Tripathi M, Chandrashekar N, Phom H, et al. Evaluation of dilated upper renal tracts by technetium-99m ethylenedicysteine F+O diuresis renography in infants and children. Ann Nucl Med. 2004;18(8):681-687.
Truong LD, Choi YJ, Tsao CC. Renal cell apoptosis in chronic obstructive uropathy: the roles of caspases. Kidney Int. 2001;60:924-934.
Truong LD, Sheikh-Hamad D, Chakraborty S, et al. Cell apoptosis and proliferation in obstructive uropathy. Semin Nephrol. 1998;18:641-651.
Tublin ME, Dodd GD, Verdile VP. Acute renal colic: diagnosis with duplex Doppler US. Radiology. 1994;193:697-701.
Tucci SJr, Molina CA, Cologna AJ, et al. Compensatory renal growth and mitochondrial function: the influence of warm ischemia and reperfusion. Acta Cir Bras. 2008;23(Suppl. 1):31-35.
Turkolmez S, Atasever T, Turkolmez K, Gogus O. Comparison of three different diuretic renal scintigraphy protocols in patients with dilated upper urinary tracts. Clin Nucl Med. 2004;29:154-160.
Twicker DM, Setiawan AT, Evans RS, et al. Imaging of puerperal septic thrombophlebitis: Prospective comparison of MR imaging, CT, and sonography. AJR Am J Roentgenol. 1997;169:1039-1043.
Tziomalos K, Krikis N, Karagiannis A, et al. Treatment of idiopathic retroperitoneal fibrosis with combined administration of corticosteroids and tamoxifen. Clin Nephrol. 2004;62:74-76.
Uchida S, Endou H. Substrate specificity to maintain cellular ATP along the mouse nephron. Am J Physiol. 1988;255:F977-F983.
Uibu T, Oksa P, Auvinen A, et al. Asbestos exposure as a risk factor for retroperitoneal fibrosis. Lancet. 2004;363(9419):1422-1426.
Ulahannan D, Blakeley CJ, Jeyadevan N, Hashemi K. Benefits of CT urography in patients presenting to the emergency department with suspected ureteric colic. Emerg Med J. 2008;9:569-571.
Ulm A, Miller F. An operation to produce experimental, reversible hydronephrosis in dogs. J Urol. 1962;88:337-341.
Upsdell SM, Leeson SM, Brooman PJ, O’Reilly PH. Diuretic induced urinary flow rates at varying clearances and their relevance to the performance and interpretation of diuresis renography. Br J Urol. 1988;61:14-18.
Vaglio A, Salvarani C, Buzio C. Retroperitoneal fibrosis. Lancet. 2006;367(9506):241-251. 21
Valles P, Manucha W. H+-ATPase activity on unilateral ureteral obstruction: interaction of endogenous nitric oxide and angiotensin II. Kidney Int. 2000;58:1641-1651.
Van Balkom BWM, Savelkoul PJM, Markovich D, et al. The role of putative phosphorylation sites in the targeting and shuttling of the aquaphorin-2 water channel. J Biol Chem. 2002;277:41473-41479.
van Bommel EF, Siemes C, van der Veer SJ, et al. Clinical value of gallium-67 SPECT scintigraphy in the diagnostic and therapeutic evaluation of retroperitoneal fibrosis: a prospective study. J Intern Med. 2007;262(2):224-234.
van Bommel EF, van der Veer SJ, Hendriksz TR, Bleumink GS. Persistent chronic peri-aortitis (‘inflammatory aneurysm’) after abdominal aortic aneurysm repair: systematic review of the literature. Vasc Med. 2008;13:293-303.
Varkarakis IM, Jarrett TW. Retroperitoneal fibrosis. AUA Update Series. 24, 2005. lesson 3
Vaughan ED, Gillenwater JY. Recovery following complete chronic unilateral ureteral occlusion: functional, radiographic, and pathologic alterations. J Urol. 1971;106:27-35.
Vaughan ED, Gillenwater JY. Diagnosis, characterization, and management of post-obstructive diuresis. J Urol. 1973;109:286-292.
Vaughan EDJr, Shenasky JH2nd, Gillenwater JY. Mechanism of acute hemodynamic response to ureteral occlusion. Invest Urol. 1971;9:109-118.
Vaughan EDJr, Sweet RC, Gillenwater JY. Peripheral renin and blood pressure changes following complete unilateral ureteral occlusion. J Urol. 1970;104:89-92.
Vaughan EJ, Sosa R. Hypertension and hydronephrosis. In Laragh J, Brenner B, editors: Hypertension: pathophysiology, diagnosis, and management, 2nd ed, New York: Raven, 1995.
Vega J, Goecke H, Tapia H, et al. Treatment of idiopathic retroperitoneal fibrosis with colchicine and steroids: a case series. Am J Kidney Dis. 2008;53(4):628-637.
Vercellini P, Pisacreta A, Pesole A, et al. Is endometriosis an asymmetric disease? BJOG. 2000;107:559-561.
Vigano P, Parazzini F, Somigliana E, Vercellini P. Endometriosis: epidemiology and aetiological factors. Best Pract Res Clin Obstet Gynaecol. 2004;18:177-200.
Villar SR, Brandoni A, Quaglia NB, et al. Renal elimination of organic anions in rats with bilateral ureteral obstruction. Biochim Biophys Acta. 2004;1688:204-209.
Vivas I, Nicolas AI, Velazquez P, et al. Retroperitoneal fibrosis: typical and atypical manifestations. Br J Radiol. 2000;73:214-222.
Wagner M, Mayr J, Häcker FM. Improvement of renal split function in hydronephronephrosis with less than 10% function. Eur J Pediatr Surg. 2008;18(3):156-159.
Wahlin N, Magnusson A, Persson EG, et al. Pressure flow measurement of hydronephrosis in children: a new approach to definition and quantification of obstruction. J Urol. 2001;166:1842-1847.
Walton G, Buttyan R, Garcia-Montes E, et al. Renal growth factor expression during the early phase of experimental hydronephrosis. J Urol. 1992;148:510-514.
Wamsley-Davis A, Padda R, Truong LD, et al. AT1A-mediated activation of kidney JNK1 and SMAD2 in obstructive uropathy: preservation of kidney tissue mass using candesartan. Am J Physiol. 2004;287:F474-F480.
Wang CJ, Huang SW, Chang CH. Efficacy of an alpha1 blocker in expulsive therapy of lower ureteral stones. J Enourol. 2008;22:41-46.
Wang G, Li C, Kim SW, et al. Ureter obstruction alters expression of renal acid-base transport proteins in rat kidney. Am J Physiol Renal Physiol. 2008;295(2):F497-F506.
Wang L, Lee JY, Kwak JH, et al. Protective effects of low-dose carbon monoxide against renal fibrosis induced by unilateral ureteral obstruction. Am J Physiol Renal Physiol. 2008;294(3):F508-F517.
Watanabe M, Kawamura S, Nakada T, et al. Left preureteral vena cava (retrocaval or circumcaval ureter) associated with partial situs inversus. J Urol. 1991;145:1047-1048.
Watanabe Y, Ozawa H, Uematsu K, et al. Hydronephrosis due to ureteral endometriosis treated by transperitoneal laparoscopic ureterolysis. Int J Urol. 2004;11:560-562.
Watkin S, Bowra J, Sharma P, et al. Validation of emergency physician ultrasound in diagnosing hydronephrosis in ureteric colic. Emerg Med Australas. 2007;19:188-195.
Weinreb S, Hruska KA, Klahr S, et al. Uptake of Pi in brush border vesicles after release of unilateral ureteral obstruction. Am J Physiol. 1982;243:F29-F35.
Wen JG, Ringaard S, Frokier J, et al. Contralateral compensatory kidney growth in rats with partial unilateral ureteral obstruction monitored by magnetic resonance imaging. J Urol. 1999;162:1084-1089.
Westenfelder C, Loghman-Adham M, Baranowski RL, et al. Increased reabsorptive capacity after ureteral obstruction reduces the ability of glucose to inhibit phosphate reabsorption in rat kidney. Nephrol Dial Transplant. 1998;13:1675-1681.
Westney OL, Amundsen CL, McGuire EJ. Bladder endometriosis. Conservative management. J Urol. 2000;163:1814-1817.
Whitaker RH. Methods of assessing obstruction in dilated ureters. Br J Urol. 1973;45:15-22.
White WM, Zite NB, Gash J, et al. Low-dose computed tomography for the evaluation of flank pain in the pregnant population. J Endourol. 2007;21(11):1255-1260.
Williams TJ. The role of surgery in the management of endometriosis. Mayo Clin Proc. 1975;50:198-203.
Worster A, Richards C. Fluids and diuretics for acute ureteric colic. Cochrane Database Syst Rev 2005;(3):CD004926.
Wright FS, Briggs JP. Feedback control of glomerular blood flow, pressure, and filtration rate. Physiol Rev. 1979;59:958-1006.
Wu J, Catalano E, Coppola D. Retroperitoneal fibrosis (Ormond’s disease): clinical pathologic study of eight cases. Cancer Control. 2002;9:432-437.
Yanagisawa H, Morrissey J, Morrison AR, et al. Eicosanoid production by isolated glomeruli of rats with unilateral ureteral obstruction. Kidney Int. 1990;37:1528-1535.
Yarger WE, Aynedjian HS, Bank N. A micropuncture study of postobstructive diuresis in the rat. J Clin Invest. 1972;51:625-637.
Yarger WE, Griffith LD. Intrarenal hemodynamics following chronic unilateral ureteral obstruction in the dog. Am J Physiol. 1974;227:816-826.
Yarger WE, Schocken DD, Harris RH. Obstructive nephropathy in the rat: possible roles for the renin-angiotensin system, prostaglandins, and thromboxanes in postobstructive renal function. J Clin Invest. 1980;65:400-412.
Yildiz B, Kural N, Colak O, et al. IGF-1, IGFBP-3, VEGF and MMP-9 levels and their potential relationship with renal functions in patients with compensatory renal growth. Clin Physiol Funct Imaging. 2008;28(2):107-112.
Yossepowitch O, Lifshitz DA, Dekel Y, et al. Predicting the success of retrograde stenting for managing ureteral obstruction. J Urol. 2001;166:1746-1749.
Young PM, Peterson JJ, Calamia KT. Hypermetabolic activity in patients with active retroperitoneal fibrosis on F-18 FDG PET: report of three cases. Ann Nucl Med. 2008;22(1):87-92.
Zagoria R, Tung G. Genitourinary radiology: the requisites. St Louis: Mosby; 1997. p. 2–6
Zeidel ML. Hormonal regulation of inner medullary collecting duct sodium transport. Am J Physiol. 1993;265:F159-F173.
Zeidel ML, Pirtskhalaishvili G. Urinary tract obstruction. In: Brenner BM, editor. Brenner and Rector’s the kidney. 7th ed. Philadelphia: WB Saunders; 2004:1867-1893.
Zhang XD, Hou SK, Zhu JH, et al. Diagnosis and treatment of retrocaval ureter. Eur Urol. 1990;18:207-210.
Zimon AE, Hwang JK, Principle DL. Pseudoaneurysm of the uterine artery. Obstet Gynecol. 1999;94:827-830.
Zook TE, Strandhoy JW. Inhibition of ADH-enhanced transepithelial urea and water movement by prostaglandins. Prostaglandins. 1980;20:1-13.