chapter 50 Treatment of Advanced Renal Cell Carcinoma
Renal cell carcinoma (RCC) is a term that includes a variety of cancers arising in the kidney and comprises several histologically, biologically, and clinically distinct entities (Linehan et al, 2007, 2009). An estimated 58,240 new cases of cancer arising in the kidney or renal pelvis were diagnosed in 2010 in the United States (Jemal et al, 2010). Approximately one third of all newly diagnosed RCC patients present with synchronous metastatic disease and an additional 20% to 40% of patients with clinically localized disease at diagnosis will eventually develop metastases (Skinner et al, 1971; Rabinovitch et al, 1994; Bukowski, 1997). Metastatic RCC is almost always fatal, with 10-year survival rates of less than 5% (Bukowski, 1997; Motzer et al, 1999, 2000; Motzer and Russo, 2000; Négrier et al, 2002); patients with metastatic disease account for the majority of deaths (approximately 13,000 a year in the United States) related to RCC (Jemal et al, 2010).
Advances in our understanding of the genetic and molecular defects underlying the individual subtypes of RCC have led to the development of novel agents designed to reverse or modulate aberrant pathways contributing to renal oncogenesis. These “targeted” therapeutic strategies have largely supplanted other treatment modalities in the initial management of metastatic clear cell kidney cancer; however, surgery, irradiation, and cytokine therapy remain appropriate choices in the management of selected patients with advanced clear cell RCC. Although agents effective in clear cell RCC are often used in patients with other subtypes of RCC there is scant evidence from prospective studies demonstrating benefit in non–clear cell RCC variants. More recently, elucidation of aberrant oncogenic pathways in papillary, chromophobe, and other variants of RCC has paved the way for evaluation of targeted therapeutic approaches in these histologic subtypes (Linehan et al, 2009).
Prognostic Factors
Patients with metastatic RCC generally have a poor prognosis, with the majority succumbing to their disease. Ten-year survival in patients diagnosed with metastatic disease was estimated to be less than 5% in the era of cytokine therapy and is unlikely to change significantly with the advent of targeted therapy. However, several clinical features such as a long time interval between initial diagnosis and appearance of metastatic disease and presence of fewer sites of metastatic disease have been observed to be associated with better outcome. Conversely, poor performance status and the presence of lymph node and/or liver metastases are some factors associated with shorter survival. Investigators at the Memorial Sloan-Kettering Cancer Center evaluated a variety of clinical and laboratory parameters in 670 patients enrolled on various clinical trials of chemotherapy or immunotherapy in an effort to identify those pretreatment factors that were able to best predict outcome (Motzer et al, 1999). In a multivariate analysis, a poor performance status (Karnofsky score <80), an elevated serum lactate dehydrogenase (LDH) level (>1.5 times upper limit of normal), a low hemoglobin (less than the lower limit of normal), an elevated corrected calcium concentration (>10 g/dL), and lack of prior nephrectomy were independent predictors of a poor outcome (Table 50–1). Patients could be stratified into three distinct prognostic groups based on these five poor prognostic factors (Table 50–2). The overall survival (OS) times in patients with no adverse factors (favorable-risk group), one to two risk factors (intermediate-risk group), and more than three risk factors (poor-risk group) were 20 months, 10 months, and 4 months, respectively (Fig. 50–1) (Motzer et al, 1999). Subsequently, the same group identified poor performance status, high calcium level, low hemoglobin value, elevated LDH level, and short time interval from initial diagnosis to treatment as factors that could best predict a poor outcome in 463 patients receiving interferon-based therapy in the first-line setting (Motzer et al, 2002). This prognostic model was found to be predictive of survival in an independent data set derived from patients treated at the Cleveland Clinic and provides independent, external validation of the proposed model (Mekhail et al, 2005). Similar prognostic schemes have also been proposed by the Groupe Français d’Immunotherapie and by investigators from the University of California, Los Angeles (Tsui et al, 2000). Validated prognostic models are used in clinical practice to help make appropriate management decisions as well as in the design and interpretation of clinical trials. Modifications of these prognostic schemes as well as identification of reliable molecular markers are under investigation as suitable predictors of response to and survival after therapy with newer targeted agents against vascular endothelial growth factor (VEGF) and mammalian target of rapamycin (mTOR) pathway components (Choueiri et al, 2007, 2008b; Motzer et al, 2008a).
Table 50–1 Adverse Prognostic Factors in 670 Patients Treated with Chemotherapy or Immunotherapy at the Memorial Sloan-Kettering Cancer Center
Data from Motzer RJ, Mazumdar M, Bacik J, et al. Survival and prognostic stratification of 670 patients with advanced renal cell carcinoma. J Clin Oncol 1999;17:2530–40.
Table 50–2 Risk Stratification Based on Adverse Prognostic Factors in 670 Patients Treated with Chemotherapy or Immunotherapy at the Memorial Sloan-Kettering Cancer Center
| RISK GROUP | NO. OF ADVERSE PROGNOSTIC FACTORS | MEDIAN OVERALL SURVIVAL |
|---|---|---|
| Good | 0 | 20 months |
| Intermediate | 1-2 | 10 months |
| Poor | 3-5 | 4 months |
Data from Motzer RJ, Mazumdar M, Bacik J, et al. Survival and prognostic stratification of 670 patients with advanced renal cell carcinoma. J Clin Oncol 1999;17:2530–40.
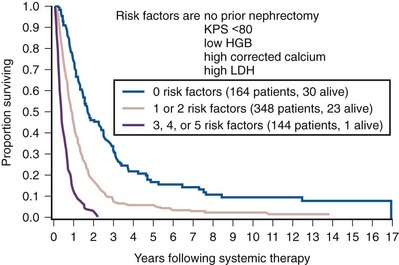
Figure 50–1 Survival analysis stratified according to risk group in 670 patients treated with chemotherapy or immunotherapy at the Memorial Sloan-Kettering Cancer Center (n = 656; 14 patients missing one or more of the five risk factors were excluded). KPS, Karnofsky performance score; HGB, hemoglobin; LDH, lactate dehydrogenase.
(From Motzer RJ, Mazumdar M, Bacik J, et al. Survival and prognostic stratification of 670 patients with advanced renal cell carcinoma. J Clin Oncol 1999;17:2530–40.)
Surgical Management of Metastatic Renal Cell Carcinoma
Debulking or Cytoreductive Nephrectomy in Patients with Metastatic RCC
The role of cytoreductive nephrectomy preceding systemic therapy has been extensively studied in the era of cytokine therapy. The impetus for exploring this approach in metastatic RCC was provided both by the perception that bulky tumors might inhibit key components of the immune system critical for combating cancer and by observations suggesting that removal of large primary tumors may provide clinical benefit. To support this practice early proponents of cytoreductive nephrectomy had cited (1) the rare but well-described occurrence of spontaneous regression of metastatic lesions after nephrectomy (Bloom, 1973; Middleton, 1980; Snow and Schellhammer, 1982; Marcus et al, 1993); (2) preclinical data suggesting that large primary tumors may inhibit T-cell function (Kudoh et al, 1997; Bukowski et al, 1998; Ling et al, 1998; Uzzo et al, 1999a, 1999b); and (3) the inability of systemic agents, particularly cytokines, to induce meaningful responses in primary renal tumors in most patients (Sella et al, 1993; Rackley et al, 1994; Wagner et al, 1999). However, the risk of perioperative morbidity and mortality and the inability of a significant proportion of patients undergoing nephrectomy to subsequently receive systemic therapy clearly underlined the need for unequivocal evidence of clinical benefit as well the ability to identify patients likely to benefit from this approach.
Nephrectomy as the sole therapeutic intervention in the context of metastatic disease is unlikely to alter outcome, as suggested by small retrospective analyses (DeKernion et al, 1978). However, several retrospective studies have demonstrated the feasibility of a combined modality approach in which nephrectomy is followed by cytokine therapy, with some suggesting that this approach may favorably impact response and survival. Investigators from the National Cancer Institute (NCI) reported their experience in 195 patients undergoing nephrectomy followed by high-dose interleukin-2 (IL-2) therapy between the years 1985 and 1996 (Walther et al, 1997b). An overall response rate of 18% (including a complete response rate of 4%) after IL-2 therapy was observed in this study. A notable finding that emerged from this study was that although the majority of patients underwent successful resection of the primary tumor only 107/195 (55%) went on to receive IL-2 therapy. Rapid postoperative disease progression and perioperative surgical and medical morbidity were the most common factors preventing delivery of systemic therapy, suggesting that careful patient selection may play an important role in the successful application of this combined modality approach. The impact of patient and/or disease characteristics on outcome is further highlighted by a retrospective study. In Bennett and associates’ (1995) series of 30 patients, which included several patients with unfavorable performance status (Eastern Cooperative Oncology Group [ECOG] 2) and multiple metastatic sites including patients with brain or liver metastases, only 7 (23%) were able to proceed with IL-2 after nephrectomy. Conversely, in a series that included only patients with favorable clinical/prognostic factors (e.g., good performance status, minimal comorbidity, no liver or brain metastases), the majority of patients were able to proceed to systemic therapy after nephrectomy, with high response rates (35% to 40%) and OS (median 20 to 22 months) after cytokine-based treatment (Fallick et al, 1997; Figlin et al, 1997). Although these retrospective analyses and small single-arm prospective studies confirmed the feasibility of a tandem surgical/cytokine therapeutic approach, their major contribution was in laying the foundation for controlled, prospective studies to determine if outcomes with cytokine therapy could be improved by prior nephrectomy.
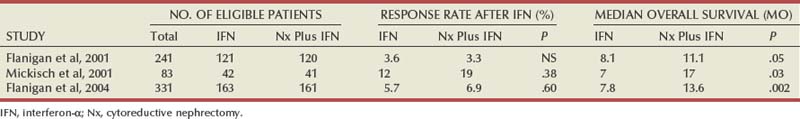
The most compelling evidence in support of cytoreductive nephrectomy is provided by two randomized phase III studies conducted by the Southwest Oncology Group (SWOG) and the European Organization for Research and Treatment of Cancer (EORTC). The larger of the two studies, SWOG trial 8949, randomized 241 patients with metastatic RCC to receive interferon-α-2b either as initial therapy or after cytoreductive nephrectomy (Flanigan et al, 2001). Salient eligibility criteria included a histologic diagnosis of kidney cancer (all histologic subtypes were allowed); good performance status (ECOG 0 or 1); presence of a resectable primary renal tumor; no prior chemotherapy, irradiation, or immunotherapy; and adequate organ function. Although there were no significant differences in the response rates to interferon observed in the two study arms, OS was improved in the surgery plus interferon arm (median 11.1 vs. 8.1 months for interferon alone, P = .05) (Fig. 50–2). These data were similar to those from a smaller EORTC trial (a total of 85 patients randomized to interferon alone or interferon after nephrectomy) that used a similar design and reported a survival advantage favoring the surgery plus interferon arm (median OS 17 vs. 7 months, P = .03) (Mickisch et al, 2001). A combined analysis of both trials revealed data that were consistent with those reported in the individual trials (Flanigan et al, 2004). These data support the use of cytoreductive nephrectomy in carefully selected patients with metastatic RCC who are likely candidates for subsequent cytokine therapy (data summarized in Table 50–3).
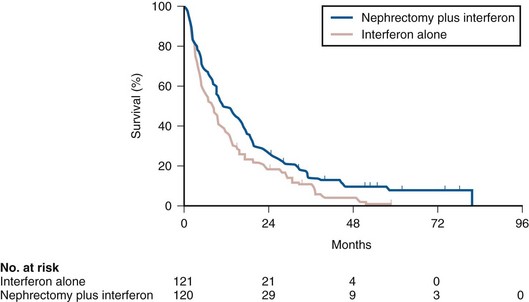
Figure 50–2 Actuarial survival among 241 patients with metastatic renal cell carcinoma randomized to either interferon-α alone or interferon-α after cytoreductive nephrectomy.
(From Flanigan RC, Salmon SE, Blumenstein BA, et al. Nephrectomy followed by interferon alfa-2b compared with interferon alfa-2b alone for metastatic renal-cell cancer. N Engl J Med 2001;345:1655–59.)
Table 50–3 Summary of Outcome in Randomized Studies of Interferon-α Alone or Interferon-α after Cytoreductive Nephrectomy in Patients with Metastatic Kidney Cancer
The impact of cytoreductive nephrectomy on outcome in patients receiving VEGF or mTOR pathway inhibitors remains to be determined and is the subject of ongoing trials. Similarly, although some patients with non–clear cell histologic subtypes of RCC were included in the aforementioned trials, the role of nephrectomy before systemic therapy in these patients is unclear.
Resection of Metastases
Most patients with metastatic RCC will not achieve a cure or long-term disease remission with currently available systemic agents. However, resection of limited metastatic disease has been reported by several groups to be associated with long disease-free intervals and OS. It should be emphasized that metastasectomy has not been evaluated systematically in a prospective, randomized fashion and that the favorable outcome ascribed to resection of solitary metastatic disease may reflect patient selection bias, inherent differences in tumor biology, and natural history or other confounding factors.
Most studies detailing outcome after metastasectomy are retrospective series. In most series, isolated pulmonary metastases were the lesions most commonly amenable to resection with curative intent. The OS of patients undergoing complete resection of limited metastatic disease is quite impressive, with reported median 5-year survival rates of 35% to 50% in many series (Middleton, 1967; Skinner et al, 1971; Tolia and Whitmore, 1975; O’Dea et al, 1978; Pogrebniak et al, 1992; Kierney et al, 1994; Friedel et al, 1999; Murthy et al, 2005; Russo et al, 2007). The larger of these studies have also attempted to identify patients most likely to benefit from this approach. In a series of 278 patients with recurrent RCC treated at the Memorial Sloan-Kettering Cancer Center, 211 were reported to have undergone either complete (141 patients) or incomplete (70 patients) resection of recurrent tumor from a variety of metastatic sites (Kavolius et al, 1998). In this series, complete or “curative” resection was associated with a longer OS (44% 5-year survival vs. 14% in patients undergoing incomplete resection); multivariate analysis also identified the presence of a solitary metastatic lesion, age younger than 60 years, and a disease-free interval of more than 1 year as favorable prognostic indicators. In addition, some studies have suggested that pulmonary metastases, smaller tumor size (<4 cm in one series), and metachronous lesions are predictors of better outcome after metastasectomy (Friedel et al, 1999; Piltz et al, 2002; Murthy et al, 2005). Although not supported by convincing evidence of survival benefit from prospective studies, resection of isolated metastatic lesions is a reasonable and widely employed practice in selected RCC patients.
Palliative Surgery
Cytoreductive nephrectomy can be performed with palliative intent in patients with intractable pain, hematuria, constitutional symptoms, or a variety of paraneoplastic manifestations such as hypercalcemia, erythrocytosis, secondary thrombocytosis, or hypertension. Symptoms such as pain and laboratory abnormalities including hypercalcemia can often be effectively managed medically, whereas symptoms such as hematuria may be amenable to alternative treatment approaches (e.g., angioembolization). Furthermore, resection of the primary renal tumor does not always result in clinical benefit; for instance, in one series, only a little over half the patients (7/12) with hypercalcemia experienced clinically meaningful reductions in serum calcium levels (Walther et al, 1997a). Cytoreductive nephrectomy with palliative intent is therefore performed relatively infrequently but is appropriate in some patients.
Resection of metastases to alleviate pain or to forestall potentially life-threatening or debilitating complications is often indicated in a variety of situations. Patients who may benefit from noncurative resection of metastatic lesions include those with solitary brain metastases, metastatic lesions in weight-bearing bones or joints, or vertebral metastatic lesions with impending spinal cord or radicular compromise (Sundaresan et al, 1986; Kollender et al, 2000; Sheehan et al, 2003). Surgical resection is often combined with radiation and/or systemic therapy in many of the aforementioned situations.
Key Points: Cytoreductive Nephrectomy in Metastatic Renal Cell Carcinoma
Key Points: Metastasectomy
Key Points: Palliative Surgery in Advanced Renal Cell Carcinoma
Immunologic Approaches in the Management of Advanced Clear Cell Renal Cell Carcinoma
The host immune system has long been believed to play an important role in the causation and control of renal cell cancer. A report detailing spontaneous regression of metastatic lesions after radical nephrectomy provides perhaps the earliest evidence implicating the immune system in the regulation of kidney cancer. The phenomenon of spontaneous regression, thought to represent T- or B-cell mediated antitumor immunity, has sparked great enthusiasm over the years and generated several reports describing this phenomenon (Braren et al, 1974; Silber et al, 1975; Middleton, 1980; Robson, 1982; Snow and Schellhammer, 1982; Kavoussi et al, 1986; Marcus et al, 1993; Edwards et al, 1996). Although rare (it is estimated that the true incidence of spontaneous regression is less than 1%) and often transient, the presumed immunologic mechanisms underlying this event have nonetheless played an important part in the development of immunotherapeutic approaches in kidney cancer. The presence of immune cells, notably cytotoxic T lymphocytes, in resected tumors and the identification of tumor-associated antigens that can serve as human leukocyte antigen (HLA)-restricted targets on tumor cells for T cell—mediated cytotoxicity have also kindled interest in immune-based strategies in renal cell cancer (Finke et al, 1994; Boon et al, 1997; Ada, 1999; Rosenberg, 1999). Early clinical studies explored the efficacy of agents believed to act as nonspecific stimulators of the host immune system components, such as cytokines, with or without adoptive cellular therapy. More recently, investigators have evaluated a variety of novel approaches including allogeneic immunotherapy, vaccines, and modulators of T-cell function. Most immunotherapy strategies have been directed at clear cell RCC, and the utility of these approaches in non–clear cell variants remains to be explored.
Interferons
The interferons are a group of proteins with diverse biologic functions, including immunomodulatory properties. Interferon-α was one of the earliest cytokines to be evaluated for activity in RCC. Initial trials with interferon utilized leukocyte-derived interferon. The subsequent availability of recombinant interferon-α in the early to mid 1980s allowed investigators to evaluate higher doses of this cytokine in a series of phase 2 trials. Initial trials demonstrated overall response rates of 16% to 26% in patients treated with interferon-α, and several subsequent trials have confirmed the activity of this agent, with response rates generally in the 10% to 15% range (deKernion et al, 1983; Quesada et al 1983, 1985, 1989; Umeda and Niijima, 1986; Muss et al, 1987; Rosenberg et al, 1987; Figlin et al, 1988; Minasian et al, 1993; Motzer et al, 2002). The limited long-term survival data available suggest that durable complete responses with this agent are relatively rare (<2%). A variety of dosing schedules and routes have been evaluated to determine the optimal interferon regimen; no single mode of administration or dosing schedule has so far demonstrated superiority over others (Kirkwood et al, 1985; Umeda and Niijima, 1986; Muss et al, 1987; Minasian et al, 1993). Similarly, the addition of chemotherapy or other cytokines to interferon-α has failed to improve the outcomes seen with single-agent therapy (Rosenberg et al, 1989b; Sella et al, 1992; Ravaud et al, 1994, 1998; Ellerhorst et al, 1997; Tourani et al, 1998, 2003; Dorval et al, 1999; Négrier et al, 2000a, 2000b).
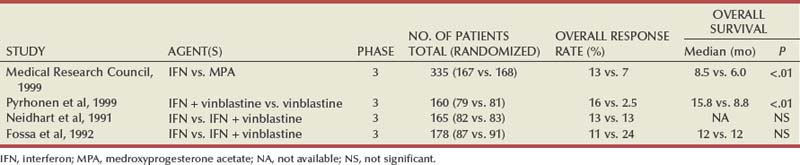
Several prospective, randomized trials evaluating the efficacy of interferon-α have demonstrated a modest but statistically significant improvement in outcome after treatment with this agent. A randomized phase III study that assigned 335 patients to receive either interferon-α or medroxyprogesterone demonstrated a higher response rate (14% vs. 2%) and OS (median 8.5 vs. 6 months, hazard ratio 0.72, P = .017) favoring the interferon arm of the study (Medical Research Council Renal Cancer Collaborators, 1999). A second study randomized 160 patients with metastatic RCC to receive vinblastine alone or in combination with interferon-α; a higher response rate (16% vs. 2.5%) and improved OS (median 16 vs. 9 months, P = .0049) with the addition of interferon was observed in this study (Table 50–4) (Pyrhonen et al, 1999). Two additional randomized studies suggested that vinblastine is unlikely to have contributed significantly to the activity of this combination by showing that survival was not improved with the addition of vinblastine to interferon-α (Neidhart et al, 1991; Fossa et al, 1992). Lastly, a meta-analysis of randomized trials of interferon against a variety of agents suggested that interferon-based therapy conferred a survival advantage (Coppin et al, 2005). Despite its relatively modest activity, based on the just described data and relative ease of administration compared with IL-2, interferon was commonly the agent of choice in the initial treatment of metastatic RCC until the advent of VEGF pathway antagonists.
Interleukin-2
Clinical trials in the early 1980s initially identified IL-2 as an active agent in RCC, with some IL-2–based regimens leading to objective response rates in excess of 30% (Rosenberg et al, 1989a, 1993). In a subsequent report detailing 255 patients treated on a series of phase 2 trials at the NCI, a more modest overall response rate of 15% (37/255 patients) was noted (Fyfe et al, 1996). Several trials conducted by the NCI and the Cytokine Working Group as well as meta-analyses of published data have consistently demonstrated response rates in the range of 15% to 20% (Lotze et al, 1986; Rosenberg et al, 1987, 1989a, 1993; Fisher et al, 1988; Dutcher et al, 1997a). More importantly, 7% to 9% of patients receiving high-dose IL-2 are reported to have achieved complete regression of all metastatic tumor, with the majority of complete responders (>60%) demonstrating no evidence of disease recurrence on long-term follow-up (Fisher et al, 1997, 2000; Rosenberg et al, 1998). There have also been reports of long-term disease-free remission in partial responders whose limited disease burden after IL-2 therapy rendered them amenable to resection of localized metastases. High-dose IL-2 was approved by the U.S. Food and Drug Administration (FDA) for the treatment of metastatic kidney cancer in 1992, based largely on its ability to induce durable complete responses in some patients.
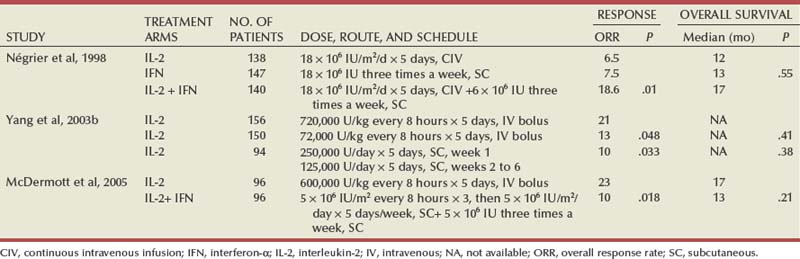
The initial studies with IL-2 were conducted using an intravenous bolus regimen with doses of 600,000 or 720,000 IU/kg administered every 8 hours as tolerated to a maximum of 15 doses. A major limitation of this dosing regimen is the considerable associated toxicity that has limited its widespread use. Vascular leak syndrome, and the resulting hypotension, third-space fluid retention, respiratory compromise, and multiorgan damage are some of the more problematic concomitants of IL-2 therapy and led to an unacceptably high treatment-related mortality rate (2% to 5%) in early studies with this agent (Rosenberg et al, 1987; Kammula et al, 1998). Subsequently, careful patient selection, intensive monitoring schemes, and early interventions with IV fluids, vasopressors, and antibiotics have served to significantly reduce mortality associated with IL-2 (Kammula et al, 1998). However, the significant morbidity and expense associated with delivering bolus high-dose IL-2 have led several investigators to explore alternative regimens aimed at reducing toxicity without compromising efficacy. Numerous single-arm phase 2 studies have evaluated a variety of alternative regimens, including daily subcutaneous administration and continuous intravenous infusion (Escudier et al, 1994a, 1994b, 1995; Atkins et al, 2001; Négrier et al, 2000b, 2005, 2008). Many of these studies have reported overall response rates of 10% to 30%, suggesting comparable efficacy to high-dose bolus administration based on data from historical controls. However, two randomized studies have demonstrated that, although well tolerated, lower-dose regimens are associated with lower overall response rates, as well with fewer durable, complete responses (Table 50–5) (Yang et al, 2003b; McDermott et al, 2005).
Table 50–5 Summary of Results from Selected Randomized Trials of Interleukin-2 in Metastatic Renal Cell Carcinoma
Attempts to enhance the efficacy of IL-2 therapy have led investigators to explore combination therapy with other cytokines, cytotoxic chemotherapy, and adoptive cellular immunotherapy. Early experience with combination cytokine therapy was promising, with one study reporting a 31% overall response rate in patients treated with high-dose bolus IL-2 and interferon (Rosenberg et al, 1989b). However, subsequent studies have indicated that this combination is more toxic and no more effective than IL-2 alone (Ravaud et al, 1994; Bukowski et al, 1997; Dutcher et al, 1997b; Tourani et al, 1998, 2003). A multicenter, randomized phase III study compared the efficacy of intermediate-dose IL-2 administered by continuous intravenous infusion with interferon-α or the combination in 425 patients with metastatic clear cell RCC (Négrier et al, 1998). Although the combination of IL-2 and interferon resulted in a higher response rate (18.6%) and 1-year event-free survival (EFS 20%) compared with IL-2 (overall response rate 6.5%, 1-year EFS 15%) or interferon (overall response rate 7.5%, 1-year EFS 12%) alone, there was no significant difference in survival between the groups (Fig. 50–3). Combination therapy also resulted in higher toxicity than either agent given alone. Regimens combining IL-2 and cytotoxic chemotherapy (particularly 5-fluorouracil [5-FU]) have been the subject of numerous studies. Unfortunately, reports of high response rates in initial studies (49% in a study using IL-2, interferon, and 5-FU) could not be reproduced in later studies (Atzpodien et al, 1993; Ellerhorst et al, 1997; Dutcher et al, 2000; Négrier et al, 2000a). Similarly, despite the promise of preclinical and early clinical studies, the addition of ex-vivo expanded tumor-infiltrating lymphocytes or lymphokine-activated killer cells to high-dose IL-2 has not reliably demonstrated improved clinical benefit and these approaches have been largely abandoned (Rosenberg et al, 1993; Law et al, 1995; Figlin et al, 1999).
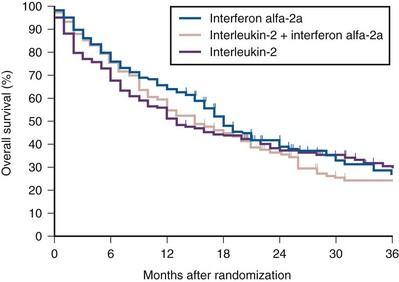
Figure 50–3 Kaplan-Meier curves for overall survival among 425 patients randomized to receive interleukin-2 alone, interferon-α alone, or both.
(From Négrier S, Escudier B, Lasset C, et al. Recombinant human interleukin-2, recombinant human interferon alfa-2a, or both in metastatic renal-cell carcinoma. Groupe Français d’Immunotherapie. N Engl J Med 1998;338:1272–78.)
Given the considerable toxicity associated with high dose IL-2 and the relatively small proportion of patients who derive benefit from this therapy, identification of predictors of response and long-term outcome has received considerable attention. The predictive value of a variety of histologic, clinical, laboratory, and molecular parameters has been studied. Patients with clear cell RCC appear most likely to benefit from IL-2 therapy, although the exceedingly small number of patients with non–clear cell histologies typically enrolled in studies of IL-2 makes it difficult to draw definitive conclusions about efficacy in this subgroup of patients. Patient performance status, number of metastatic sites, site of metastases, prior nephrectomy, and time from nephrectomy to systemic therapy are some of the factors that may impact outcome. In one study, patients with more than one site of metastasis, those with metastatic disease within 1 year of diagnosis, and those with liver metastases had the worst outcome, with a median survival of 6 months (Négrier et al, 2002). Overexpression of carbonic anhydrase IX (CAIX or G250) has been observed in a retrospective analysis to be associated with a higher probability of response to IL-2 (Atkins et al, 2005). When combined with histologic features, CAIX expression was found to be predictive of IL-2 responsiveness and OS. The expression of CAIX is regulated by hypoxia-inducible factor (HIF), and both HIF and CAIX are upregulated in tumors with VHL loss; it is therefore unclear if CAIX expression influences the behavior of these tumors or is merely a surrogate for VHL dysfunction. Although intriguing, these data should be validated in prospective trials before CAIX and/or histologic parameters can be reliably used to select patients most likely to benefit from cytokine therapy.
The role of cytokine therapy in the current management of kidney cancer has changed with the availability of novel inhibitors of VEGF and mTOR pathways with activity in clear cell RCC. Single-agent interferon, the previous standard in many institutions, is no longer used in the first-line treatment of clear cell RCC. However, given the inability of newer targeted agents to induce durable responses, high-dose intravenous IL-2 remains a reasonable option in the initial therapy of carefully selected patients with metastatic clear cell RCC.
Allogeneic Hematopoietic Stem Cell Transplantation
Allogeneic hematopoietic stem cell transplantation allows the replacement of host or recipient immune and hematopoietic systems with those of a healthy, HLA-compatible donor. The therapeutic potential of hematopoietic stem cell transplantation lies largely in the ability of the transplanted donor graft to generate an allogeneic antitumor immune response known as the graft-versus-tumor effect. This approach has been used successfully with curative intent in a variety of hematologic malignancies (Thomas et al, 1977; Weiden et al, 1979, 1981). The ability of a variety of immune-based approaches to induce remissions in patients with RCC and evidence suggesting that the host immune system in these patients may be compromised and/or tolerant to tumor cells have led several investigators to evaluate allogeneic hematopoietic stem cell transplantation in kidney cancer.
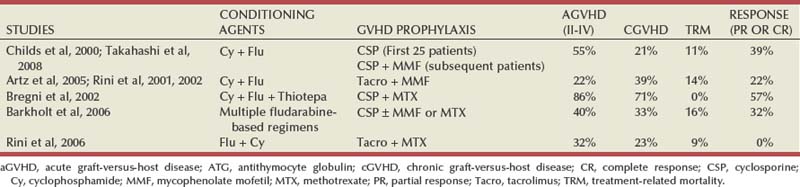
The approach was initially studied by investigators at the National Heart, Lung and Blood Institute exploring the efficacy of reduced intensity hematopoietic stem cell transplantation in patients with treatment-refractory metastatic RCC. Eligible patients underwent reduced intensity conditioning with cyclophosphamide (120 mg/kg) and fludarabine (125 mg/m2) followed by infusion of a granulocyte colony-stimulating factor–mobilized peripheral blood stem cell graft from a 5/6 or 6/6 HLA-matched sibling donor. The initial experience with hematopoietic stem cell transplantation in metastatic RCC was published by Childs and colleagues (2000). Ten of the first 19 patients treated with this transplant approach had tumor shrinkage, including 3 who had a complete response and 7 who had a partial response. As more recently reported, 74 patients have undergone hematopoietic stem cell transplantation for RCC at the National Institutes of Health. Of these, 73 patients have demonstrated durable engraftment, achieving 100% donor T-cell chimerism by day 100 posttransplant. Twenty-nine of 74 (39%) patients have had a disease response, including 7 complete (9%) and 22 partial responders (30%) (Takahashi et al, 2008). Preliminary data suggest that disease response after hematopoietic stem cell transplantation is a clinically meaningful phenomenon because regression of metastatic RCC appears to be associated with a trend toward improved survival. Survival in nonresponders has been less than 6 months, in contrast to those achieving a partial response who survived a median 2.5 years posttransplant. Several durable responses have been noted, and the first patient who underwent a transplant remains in complete remission more than 10 years after the procedure. Hematopoietic stem cell transplantation is associated with a variety of adverse events typically associated with conditioning chemotherapy (e.g., pancytopenia), a variety of opportunistic infections, and graft-versus-host disease (GVHD). Eight patients in the just-mentioned series died of transplant-related causes (transplant-related mortality of 11%), most due to GVHD and its attendant infectious complications. Several other trials have since confirmed the efficacy of this approach in RCC (Table 50–6) (Rini et al, 2001, 2002; Bregni et al, 2002; Artz et al, 2005; Barkholt et al, 2006). However, a Cancer and Leukemia Group B (CALGB) intergroup trial evaluating the feasibility of performing hematopoietic stem cell transplantation for metastatic RCC in a multi-institutional setting in the United States reported no responses in 22 patients undergoing hematopoietic stem cell transplantation from an HLA-matched sibling donor after cyclophosphamide/fludarabine–based conditioning (Rini et al, 2006). Median OS was only 5.5 months, with most patients dying of disease progression (median time to progression of 3 months). Inclusion of a number of patients with multiple adverse prognostic factors, sparing use of donor lymphocyte infusions (only 2/22 patients received donor lymphocyte infusions despite disease progression in the majority), and inclusion of patients with non–clear cell histology are some of the factors that may account for the poor outcome observed in this trial. This trial clearly highlights the importance of appropriate patient selection and the need for identifying prognostic factors likely to predict for a favorable outcome. In a European multicenter study of 106 patients undergoing transplantation, chronic GVHD, good performance status (Karnofsky score = 80), administration of donor lymphocyte infusions, and fewer than three sites of metastatic disease were identified as factors favorably impacting survival (Barkholt et al, 2006). Given the high morbidity and mortality with this approach, careful patient selection is of great importance. Hematopoietic stem cell transplantation remains an experimental approach in the management of RCC; strategies for minimizing the toxicity associated with this procedure and maximizing antitumor effects are being explored and are required to render this approach more widely applicable.
Molecular Basis for Targeted Approaches in Clear Cell Renal Cell Carcinoma
The development of antagonists of the VEGF pathway in the treatment of clear cell RCC is an often-cited paradigm in the evolution of rational targeted therapeutic strategies and represents a logical progression from identification of a defective cellular pathway to rational drug design and structured clinical evaluation. The earliest clues to the central role played by VEGF in renal oncogenesis came from studies attempting to identify the genetic basis of the von Hippel-Lindau familial kidney cancer syndrome. In the early 1990s, investigators at the National Cancer Institute studying families with von-Hippel-Lindau disease used genetic linkage analysis to identify germline mutations or deletions in the VHL gene as the basis for this disease (Latif et al, 1993). Individuals carrying germline VHL mutations are at increased risk for developing tumors in multiple organs, including bilateral, multifocal clear cell kidney cancer. The VHL gene is a classic tumor suppressor gene, with inactivation of the normal VHL allele in affected tissues by a somatic “second hit” required for tumor formation.
The VHL gene resides on the short arm of chromosome 3 and encodes the VHL protein, which can be synthesized as two alternatively spliced variants (Latif et al, 1993; Gnarra et al, 1996; Iliopoulos et al, 1998; Linehan, 2003). One of the better-understood functions of the VHL protein is its association with elongins B and C and CUL2 to form a protein complex that serves to tag certain cellular proteins for delivery to and degradation by the ubiquitin system (Duan et al, 1995; Pause et al, 1997; Stebbins et al, 1999; Linehan, 2003; Kaelin, 2004). Proteins targeted for ubiquitin-mediated degradation include the α subunits of a group of transcriptionally active proteins known as hypoxia-inducible factors (HIFs) (Iliopoulos et al, 1996; Pause et al, 1997; Maxwell et al, 1999; Cockman et al, 2000; Ohh et al, 2000; Ivan et al, 2001; Jaakkola et al, 2001). In cells with intact VHL function, HIF levels are primarily controlled by ambient oxygen tension. In normoxic conditions, hydroxylation of key proline residues on HIF promotes its association with the VHL/elongin/CUL complex and subsequent degradation; conversely, hypoxia impedes prolyl hydroxylation of HIF and its subsequent degradation, leading to intracellular accumulation of HIF α subunits. Mutations in VHL interfere with its binding to either HIF or elongin/CUL 2 and promote HIF accumulation even under normoxia. HIF accumulation, in turn, leads to the upregulation of a variety of proangiogenic and growth factors, including VEGF, platelet-derived growth factor (PDGF), transforming growth factor-α, Glut-1, and erythropoietin, which are believed to play critical roles in the development and progression of clear cell RCC (Fig. 50–4) (Duan et al, 1995; Iliopoulos et al, 1996; Kaelin, 2004; Linehan et al, 2007). Although originally identified as a germline defect in von Hippel-Lindau families, evidence of somatic VHL inactivation by mutation or promoter hypermethylation has been observed in a high proportion of sporadic clear cell tumors (91% in a recent series) (Herman et al, 1994; Gnarra et al, 1996; Zhuang et al, 1996a, 1996b; Nickerson et al, 2008). The recognition of VHL loss as a central event in renal oncogenesis has paved the way for the development of novel agents targeting components of this pathway in the management of metastatic clear cell RCC.
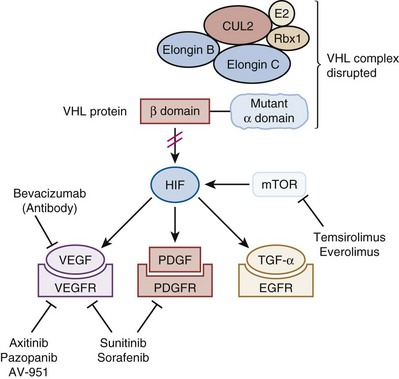
Figure 50–4 Targeting the VHL pathway in clear cell renal cell carcinoma. Loss of VHL activity leads to accumulation of HIF and several proangiogenic and growth factors that can serve as targets for anticancer drugs. HIF can also be upregulated by mTOR, which promotes HIF translation.
(Adapted from Linehan WM, Bratslavsky G, Pinto PA, et al. Molecular diagnosis and therapy of kidney cancer. Annu Rev Med 2010;61:329–43.)
Targeted Molecular Agents in Clear Cell Renal Cell Carcinoma
Antagonists of the Vascular Endothelial Growth Factor Pathway
Bevacizumab
Bevacizumab, a humanized monoclonal antibody against VEGF-A, was the first VEGF pathway antagonist used in clinical trials to test the hypothesis that modulation of aberrantly expressed components of the VHL pathway would be associated with clinical activity in clear cell RCC. In a three-arm phase 2 study, patients with metastatic clear cell RCC whose disease had progressed after prior cytokine therapy were randomized to receive either one of two dose levels of bevacizumab (10 mg/kg or 3 mg/kg administered every 2 weeks intravenously) or placebo (Table 50–7) (Yang et al, 2003a). An interim efficacy analysis performed after 116 patients (of a planned 240 patients) were enrolled in the study demonstrated a significantly longer progression-free survival (PFS) in patients assigned to 10 mg/kg bevacizumab compared with those receiving placebo (4.8 vs 2.5 months, P < .001) (Fig. 50–5). There was no corresponding improvement in OS, although the crossover design used in this trial (patients progressing on placebo were allowed to cross over to the bevacizumab arm) may have influenced this outcome. The overall response rate in patients receiving bevacizumab was modest (objective response rate of 10%, all in patients assigned to the 10 mg/kg dose). The agent was well tolerated, with bleeding, hypertension, fatigue, and proteinuria being some of the more common adverse events reported. Single-agent bevacizumab has not been compared with either cytokines (often used in first-line treatment before the advent of VEGF pathway antagonists) or other VEGF pathway antagonists such as sunitinib in prospective trials.
Table 50–7 Summary of Selected Studies of Bevacizumab-Based Regimens in Metastatic Renal Cell Carcinoma
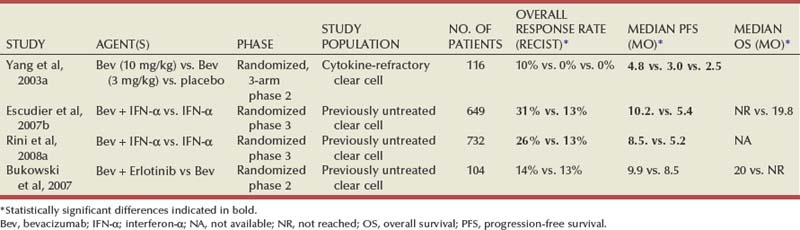
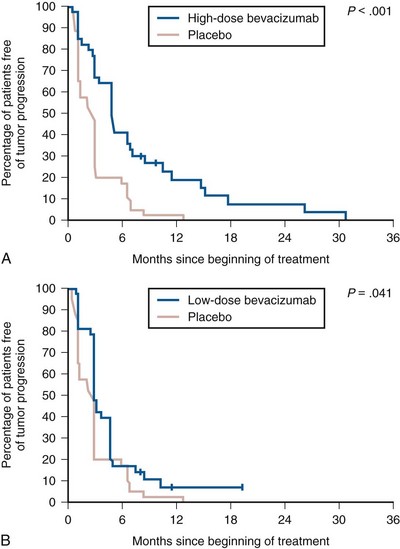
Figure 50–5 Kaplan-Meier analysis of 39 patients assigned to high-dose bevacizumab (10 mg/kg, A) and of 37 patients assigned to low-dose bevacizumab (3 mg/kg, B) compared with 40 patients assigned to receive placebo.
(From Yang JC, Haworth L, Sherry RM, et al. A randomized trial of bevacizumab, an anti-vascular endothelial growth factor antibody, for metastatic renal cancer. N Engl J Med 2003a;349:427–34.)
Several strategies for improving the efficacy of bevacizumab have been explored, including combination with cytokines (interferon-α) and other targeted agents (e.g., erlotinib, a small molecule inhibitor of epidermal growth factor receptor tyrosine kinase activity). A single-arm phase 2 study of bevacizumab plus erlotinib showed promising activity with an overall response rate of 25% (higher than would be expected with bevacizumab alone based on historical data) and median PFS of 11 months (Hainsworth et al, 2005). However, a subsequent randomized phase 2 study failed to demonstrate the superiority of this combination (median PFS 9.9 months) over bevacizumab alone (median PFS 8.5 months, P = .58) (Bukowski et al, 2007). Two large randomized phase 3 studies have compared the combination of bevacizumab and interferon-α to interferon-α alone (Table 50–8). In a Cancer and Leukemia Group B study (CALGB 90206) of 752 patients with previously untreated metastatic clear cell RCC randomized to one of two treatment arms, a higher response rate (25.5% vs. 13.1%, P < .0001) and PFS (8.5 months vs. 5.2 months, P < .0001) were observed in patients assigned to bevacizumab plus interferon-α compared with those receiving interferon-α alone (Rini et al, 2008a). A multicenter European trial (AVOREN) with a similar design also reported similar results with PFS (10.2 vs. 5.4 months, P = .0001) and objective response rate (31% vs. 13%) favoring the combination arm (Fig. 50–6) (Escudier et al, 2007b). Updated survival data from both trials were recently reported, demonstrating no improvement in overall survival associated with the addition of bevacizumab to interferon (Escudier et al, 2010; Rini et al, 2010). While neither study permitted crossover of patients assigned to interferon-α at progression, a significant number of patients (>60%) subsequently received anticancer therapy off protocol, potentially confounding overall survival analysis. Both trials reported a higher incidence of some grade 3 adverse events such as hypertension, fatigue, anorexia, and asthenia in patients receiving combination therapy. Both the CALGB and Avoren trials were large, well-designed, and well-conducted multicenter studies that suggest that the addition of bevacizumab to interferon may be associated with clinical benefit (i.e., improvement in PFS). However, these trials did not include an arm with bevacizumab alone (owing to insufficient evidence of single-agent activity at the time these trials were designed to justify a bevacizumab-only arm), making it difficult to determine if inclusion of interferon in this regimen, with its attendant toxicities, adds meaningful clinical benefit. Interestingly, the median PFS observed in the combination arm of the CALGB trial (8.5 months) is similar to that observed with bevacizumab alone in a phase 2 trial of previously untreated patients with clear cell RCC (Bukowski et al, 2007). Lastly, bevacizumab (either alone or in combination with interferon) has not been compared prospectively with VEGF receptor (VEGFR) antagonists such as sunitinib, arguably the current standard of care for most newly diagnosed patients with metastatic clear cell RCC. Bevacizumab is not widely used as a single agent in the initial therapy for metastatic clear cell RCC but may have a role in patients who have failed standard therapy with first-line VEGFR antagonists, either alone or in combination with other agents, particularly interferon. Ongoing and future trials may help determine the best use of this agent in the overall management of patients with clear cell RCC.
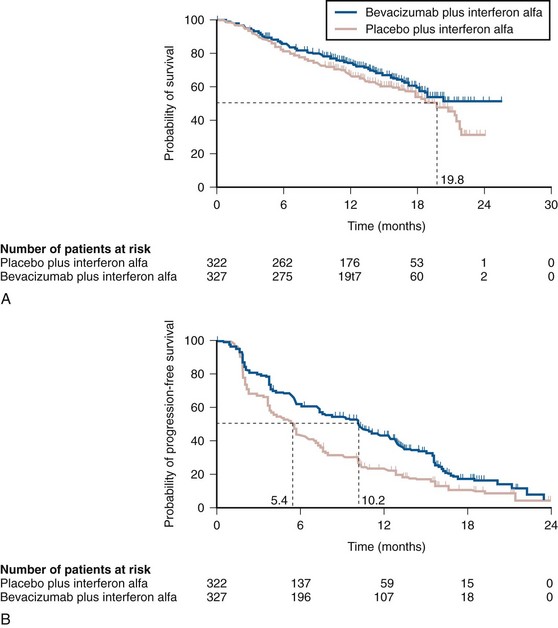
Figure 50–6 Kaplan-Meier analysis of (A) overall survival and (B) progression-free survival in 649 patients assigned to receive interferon-α alone or in combination with bevacizumab.
(From Escudier B, Pluzanska A, Koralewski P, et al. Bevacizumab plus interferon alfa-2a for treatment of metastatic renal cell carcinoma: a randomised, double-blind phase III trial. Lancet 2007b;370:2103–11.)
Sorafenib
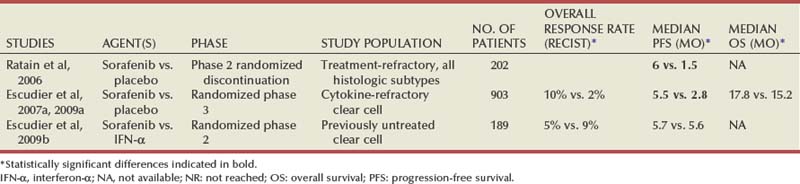
Sorafenib is an oral receptor kinase inhibitor with activity against VEGFR2, PDGF receptor-β, and raf-1. A phase 2 trial using a randomized discontinuation design provided initial evidence that sorafenib is active in RCC (see Table 50–8) (Ratain et al, 2006). Two hundred and two patients were enrolled on this trial, and all of them received 400 mg of sorafenib twice daily for the first 12 weeks. Sixty-five patients who demonstrated stable disease at the end of this period (defined in this trial as within ± 25% of baseline) were then randomized to either continued treatment with sorafenib or to placebo. Patients randomized to sorafenib had a superior PFS (24 weeks) versus those receiving placebo (6 weeks, P = .0087). A global, multicenter, placebo-controlled randomized phase 3 trial with 903 patients (the TARGET study) was subsequently undertaken to evaluate the efficacy of sorafenib in patients with metastatic RCC who had previously received cytokine therapy (Fig. 50–7) (Escudier et al, 2007a). This trial echoed the results seen in the phase 2 trial, with patients randomized to receive sorafenib experiencing a longer PFS (median 5.5 months) versus those receiving placebo (2.8 months, P < .01) at a planned interim analysis. After this interim analysis patients progressing on placebo were allowed to cross over to receive sorafenib. Mature survival data from this trial were recently presented and demonstrated no significant difference in OS between the two groups (17.8 months for sorafenib vs. 15.3 months with placebo, P = .146) in an intent-to-treat analysis (see Fig. 50–7) (Escudier et al, 2009a). However, if patients who crossed over from placebo to sorafenib were censored, the survival data demonstrated a statistically significant advantage favoring sorafenib (median OS 17.8 vs. 14.3 months, P = .029). Results from this exploratory “pre-crossover” survival analysis suggest that the survival end point may have been influenced by the crossover design. Overall response rate to sorafenib in this trial was relatively low, with 10% of patients experiencing a partial response; however, more than 70% of patients receiving sorafenib had some degree of tumor regression. The side effect profile of sorafenib is comparable to that of other agents in this class and includes hypertension, fatigue, rash, hand-foot syndrome, and diarrhea. The efficacy of sorafenib in the first-line setting was investigated in a randomized phase 2 study that assigned 189 previously untreated patients with metastatic clear cell RCC to receive either sorafenib or interferon-α (Escudier et al, 2009b). Although patients receiving sorafenib had a higher likelihood of achieving tumor regression (68% vs. 39%), PFS in the two groups was nearly identical (median 5.7 months for sorafenib vs. 5.6 months for interferon, P = NS) (Table 50–9). Although sorafenib is FDA-approved for the treatment of advanced kidney cancer, its precise role in the management of these patients remains to be determined in view of the plethora of VEGFR antagonists currently available or under investigation. Dose-intensive regimens of sorafenib (up to 800 mg twice daily) are being explored in an attempt to enhance the efficacy of this agent, but to date there is no convincing evidence that higher doses offer additional clinical benefit. Currently, sorafenib is infrequently used in the first-line setting. Anecdotal evidence and small case series suggest that patients whose disease has progressed on other VEGFR inhibitors may respond favorably to sorafenib, and the agent is commonly used in patients whose disease has progressed on sunitinib or similar agents.
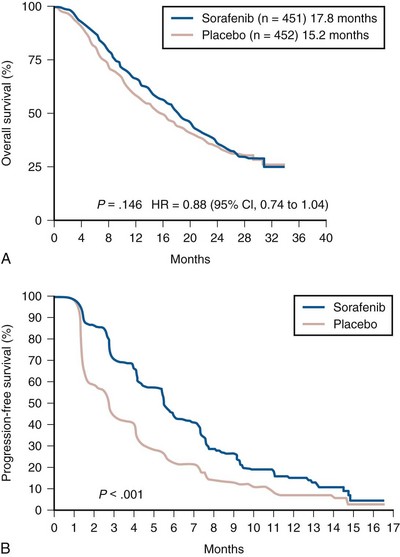
Figure 50–7 Kaplan-Meier analysis of overall survival (A) and progression-free survival (B) in 903 metastatic renal cell carcinoma patients randomized to sorafenib or placebo.
(From Escudier B, Eisen T, Stadler WM, et al. Sorafenib for treatment of renal cell carcinoma: final efficacy and safety results of the phase III treatment approaches in renal cancer global evaluation trial. J Clin Oncol 2009a;27:3312–18; and Escudier B, Eisen T, Stadler WM, et al. Sorafenib in advanced clear-cell renal-cell carcinoma. N Engl J Med 2007a;356:125–34.)
Sunitinib
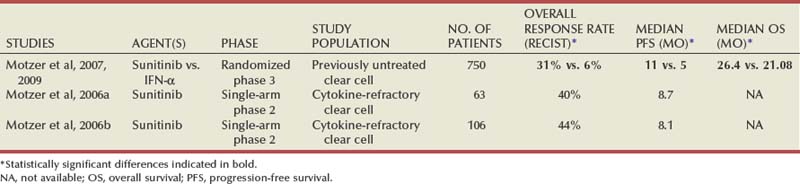
Sunitinib is currently the most widely used oral VEGFR kinase inhibitor in the initial treatment of metastatic clear cell RCC. It is a potent inhibitor of VEGFR2, PDGFR-β, c-KIT, and fms-like tyrosine kinase-3. As with sorafenib, simultaneous targeting of VEGF and PDGF pathways by this agent is likely to act synergistically in inhibiting tumor angiogenesis, by directly disrupting VEGF-mediated vascular endothelial development and proliferation and by interfering with vascular pericyte function, which is dependent on the integrity of PDGF signaling. Initial evaluation of sunitinib was undertaken with two single-arm open-label phase 2 trials in patients with metastatic RCC, most of whom had previously received cytokine therapy (Motzer et al, 2006a, 2006b). Sunitinib was administered orally at a dose of 50 mg/day during the first 4 weeks of a 6-week cycle on both trials (see Table 50–9). Remarkably high overall response rates (30% to 40%) with a median PFS of more than 8.5 months were observed in these trials, leading to the approval of this agent for treatment of advanced RCC by the FDA. A landmark phase 3 randomized trial comparing sunitinib with interferon-α as first-line therapy in patients with metastatic clear cell RCC further demonstrated the activity of sunitinib in this patient population (Motzer et al, 2007). In this study, 750 patients were randomized to receive either sunitinib or interferon-α. An interim analysis based on independent, third-party radiologic assessment demonstrated a significantly superior PFS (median 11 months vs, 5 months, P = .001) and overall response rate (31% vs. 6%, P = .001) favoring the sunitinib arm (Fig. 50–8). Gastrointestinal events, particularly diarrhea, dermatologic manifestations such as rash and hand-foot syndrome, constitutional symptoms such as fatigue and asthenia, and hypertension were the most commonly adverse events associated with sunitinib, whereas bone marrow suppression, decline in left ventricular ejection fraction, and hypothyroidism were other notable side effects. Sunitinib also performed better than interferon in a quality of life assessment conducted as part of the study. Mature survival data from this study were recently reported and demonstrated superior OS in the sunitinib study arm based on an intent-to-treat analysis (median OS 26.4 vs. 21.8 months, P = .051) (Motzer et al, 2009). Based on these data, sunitinib has become the most widely used agent in the initial management of metastatic clear cell RCC patients in the United States.
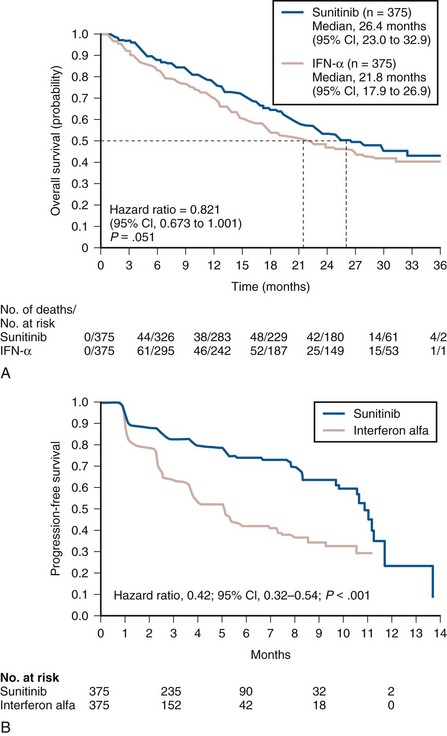
Figure 50–8 Kaplan-Meier analysis of overall survival (A) and progression-free survival (B) in 750 previously untreated patients with metastatic renal cell carcinoma receiving either sunitinib or interferon-α.
(From Motzer RJ, Hutson TE, Tomczak P, et al. Overall survival and updated results for sunitinib compared with interferon alfa in patients with metastatic renal cell carcinoma. J Clin Oncol 2009;27:3584–90; and Motzer RJ, Hutson TE, Tomczak P, et al. Sunitinib versus interferon alfa in metastatic renal-cell carcinoma. N Engl J Med 2007;356:115–24.)
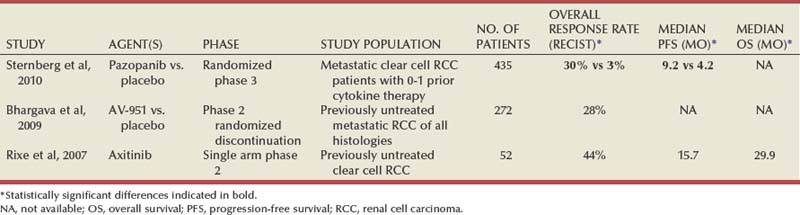
Other VEGF Pathway Antagonists
Agents such as sunitinib and sorafenib have activity against a wide array of target molecules, some of which may not be relevant in clear cell RCC. Although these agents are relatively well tolerated when compared with conventional cytotoxic chemotherapy, dose reductions and termination of treatment due to toxicity are not infrequently warranted in patients receiving these drugs. A variety of newer agents with selective activity against the VEGFR family have recently gained attention as a possible means of diminishing the side effects associated with therapy without compromising efficacy (Table 50–10). Axitinib is an oral small molecule tyrosine kinase inhibitor of VEGFR1, VEGFR2, and VEGFR3. In a phase 2 trial of axitinib in 52 patients with advanced kidney cancer, an overall response of 44%, including two complete responses (4%), and a median time to progression of 15.7 months were reported (Rixe et al, 2007). Most patients treated on this study displayed some degree of tumor shrinkage, and many had been pretreated with either IL-2 or interferon. Diarrhea, fatigue, and hypertension were the most commonly encountered grade 3 and 4 events and were amenable to medical management in most patients. Several other agents with selective activity against the VEGFR family are also under investigation. One agent in this class, pazopanib, was the subject of a recent randomized, double-blind, placebo-controlled phase 3 trial in patients with metastatic clear cell RCC who had received no or one prior cytokine-based therapy (Sternberg et al, 2010). In the 405 patients randomized (2 : 1) to receive either pazopanib or placebo, PFS (median 9.4 months vs. 4.2 months, P = .0000001) and response rates (30% vs. 3%) clearly favored the pazopanib study arm. Furthermore, reported toxicities were mild, with very few grade 3 and 4 adverse events encountered. Tivozanib (AV-951), yet another inhibitor of VEGFR1 to VEGRF3, has been evaluated in a phase 2 randomized discontinuation trial with demonstration of activity in RCC as well as an appealing adverse event profile (Bhargava et al, 2009). The approach directed at a single type of growth factor receptor has the potential to provide a strategy for precisely targeting a component of the VHL pathway in clear cell kidney cancer while avoiding the toxicity of multiple kinase inhibitors. Prospective randomized trials of selective VEGFR inhibitors are required to determine if these agents offer tangible advantages over currently FDA-approved agents.
Inhibitors of the Mammalian Target of Rapamycin
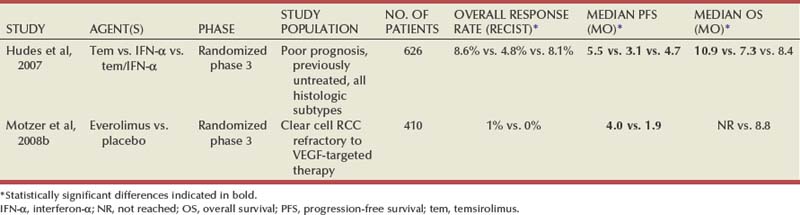
mTOR is a key intracellular protein that is a component of several signaling cascades including those mediating the effects of some growth factors. It appears to play a role in regulating translation and stability of HIF-1α, and preclinical models have suggested that growth inhibition occurring in response to mTOR inhibitors correlates with a block in HIF-1α translation (Hudson et al, 2002; Thomas et al, 2006). Two analogues of sirolimus, temsirolimus and everolimus, have been clinically evaluated with demonstrable activity in RCC.
A phase 2 trial of temsirolimus evaluated three different doses (25 mg, 75 mg, and 250 mg per week administered intravenously) in 111 patients assigned randomly to one of the dose levels (Table 50–11). The overall response rate observed on this trial was modest (7%, including one patient with a complete response), and the median PFS was 15 months (Atkins et al, 2004). Responses and PFS appeared to be independent of temsirolimus dose. An exploratory subgroup analysis based on stratification of patients according to MSKCC risk groups revealed that patients in the poor-risk group had a longer median OS compared with historical controls receiving interferon-α (8.2 vs. 4.9 months). Based on this observation, a three-arm randomized phase 3 trial of 626 patients with three or more predefined poor risk features was undertaken (Hudes et al, 2007). Patients with previously untreated metastatic kidney cancer of all histologic subtypes were eligible and randomized to receive temsirolimus alone (25 mg intravenously every week), interferon-α alone (up to 18 million units subcutaneously three times a week) or temsirolimus (15 mg intravenously every week) plus interferon-α (6 million units subcutaneously three times a week). PFS was superior in both temsirolimus-containing arms compared with interferon alone (median 3.8 months vs. 1.9 months). Importantly, the trial also demonstrated a significantly higher OS in the temsirolimus arm compared with the interferon-only arm (median OS 10.9 vs. 7.3 months, P = .008), while the addition of temsirolimus did not appear to significantly alter OS compared with interferon alone (median OS 8.4 vs. 7.3 months, P = .70) (Fig. 50–9). Temsirolimus was fairly well tolerated, and most common adverse events such as mucositis, fatigue, rash, hyperglycemia, hypophosphatemia, hypercholesterolemia, and pulmonary complications were amenable to medical and/or supportive measures. Based on these data, temsirolimus was approved by the U.S. FDA for the treatment of patients with metastatic RCC, and the agent is a reasonable front-line choice in patients presenting with poor-risk features.
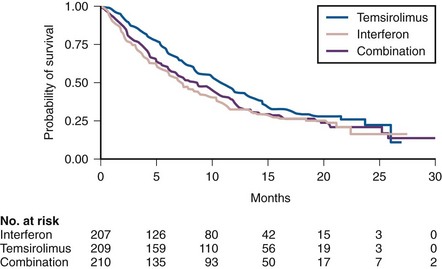
Figure 50–9 Kaplan-Meier estimates of overall survival in 626 metastatic renal cell carcinoma patients with adverse prognostic features randomized to receive temsirolimus alone, interferon-α alone, or combination therapy.
(From Hudes G, Carducci M, Tomczak P, et al. Temsirolimus, interferon alfa, or both for advanced renal-cell carcinoma. N Engl J Med 2007;356:2271–81.)
Everolimus is an orally bioavailable inhibitor of mTOR. It was the subject of a phase 3 trial in patients with metastatic clear cell RCC whose disease had progressed after therapy with sunitinib, sorafenib, or both (Motzer et al, 2008b). Patients were randomized to receive either everolimus, 10 mg once daily (n = 272), or placebo (n = 138) (see Table 50–11). The trial was stopped after an interim analysis demonstrated superior PFS in the everolimus arm (median 4 months) compared with the placebo arm (median 1.9 months) (Fig. 50–10). At the time of this analysis, median survival had not been reached in the everolimus group and was 8.8 months for the placebo group; OS was not significantly different in the two groups. Although the agent offers only a modest improvement in PFS in patients progressing on first-line VEGFR antagonists, it is nonetheless a reasonable therapeutic option and has been approved by the U.S. FDA for this indication.
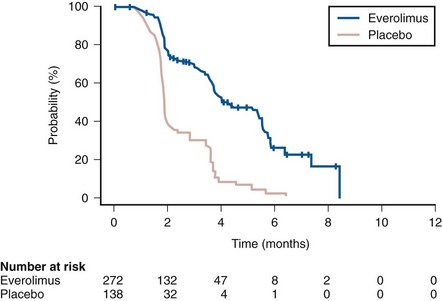
Figure 50–10 Kaplan-Meier estimates of progression-free survival in 410 patients with metastatic renal cell carcinoma randomized to everolimus or placebo.
(From Motzer RJ, Escudier B, Oudard S, et al. Efficacy of everolimus in advanced renal cell carcinoma: a double-blind, randomised, placebo-controlled phase III trial. Lancet 2008b;372:449–56.)
Combination and Sequential Therapy with Agents Targeting the VHL Pathway
Combinations of two or more classes of agents with activity centered on different components of the VHL pathway provides an attractive strategy that might serve to increase efficacy as well as eliminating potential mechanisms of resistance. A major limitation of this approach is the overlapping toxicity profile of several drugs, necessitating significant dose reductions of individual drugs (Feldman et al, 2009; Patel et al, 2009). Ongoing trials will determine if the synergy engendered by combination therapy can adequately offset the effects of dose reduction.
Given the profusion of agents with single-agent activity in RCC, identifying the optimal sequencing of these agents is a question of considerable importance. A randomized study has demonstrated that everolimus can prolong PFS compared with placebo in patients who have progressed on first-line VEGFR antagonists (Motzer et al, 2008b). Smaller retrospective analyses have suggested that a proportion of patients who have progressed on one VEGF pathway inhibitor may benefit from a second agent targeting the same pathway (Rini et al, 2008b; Tamaskar et al, 2008). Carefully designed clinical trials are required to identify the most effective strategies for sequencing multiple agents, as well as to categorize patients most likely to benefit from a given sequence based on molecular characterization of their tumors.
Other Treatment Options in Patients with Clear Cell Renal Cell Carcinoma
Chemotherapy
Conventional cytotoxic chemotherapy has been largely ineffective in the management of clear cell RCC. Numerous chemotherapeutic agents including 5-FU, platinum compounds, gemcitabine, vinblastine, and bleomycin have been evaluated as single agents in this disease but have failed to demonstrate clinically meaningful activity; a wide array of combination chemotherapy regimens have fared little better (Haas et al, 1976; Hahn et al, 1977; Zaniboni et al, 1989; Mertens et al, 1993, 1994; Amato, 2000; Rini et al, 2000, 2005; Stadler et al, 2006). Comprehensive meta-analyses of chemotherapy trials in RCC indicate the overall response rate is 5.5% to 6.0% (Yagoda et al, 1995). Although the mechanisms underlying this profound chemoresistance have not been fully elucidated, overexpression of the multidrug resistance gene (MDR) has been proposed as one possible culprit (Fojo et al, 1987). However, the addition of MDR inhibitors such as toremefine, cyclosporine, and verapamil to conventional chemotherapeutic agents has failed to improve their efficacy, suggesting that other, as yet unrecognized, factors may be at play (Braybrooke et al, 2000). Cytotoxic chemotherapy has no role in the current management of most patients with clear cell RCC. One situation in which chemotherapy may bear further investigation is in patients whose tumors demonstrate a sarcomatoid component; a small case series has suggested promising activity for gemcitabine-based chemotherapy in this setting, prompting further study of this approach (Nanus et al, 2004).
Hormonal Therapy
Hormonal therapy had been the subject of trials in RCC in the 1970s and 1980s, preceding the advent of cytokines. These studies were prompted by both the lack of effective therapies for kidney cancer and by the belief that a male preponderance (kidney cancer occurs approximately twice as frequently in males) implied a hormonal basis for this malignancy. Hormonal agents such as medroxyprogesterone have been noted to induce tumor regressions in a small minority of patients, but overall response rates are too low (approximately 2%) to have meaningful clinical impact in most patients (Schomburg et al, 1993; Harris, 1983; Medical Research Council Renal Cancer Collaborators, 1999; Braybrooke et al, 2000). Progestational and other hormonal agents have no role in the current management of renal cell cancer.
Systemic Therapy for Non–Clear Cell Variants of Renal Cell Carcinoma
Non–clear cell subtypes of kidney cancer are relatively rare (constituting approximately 15% of all kidney cancer) and have been the subject of few prospective studies. Given the unavailability of agents of proven efficacy in papillary, chromophobe, and other rare histologic subtypes, patients with non–clear cell renal tumors often receive agents with activity in clear cell RCC.
Recent advances in our understanding of some subtypes of RCC are bound to lead to a more rational approach to their management. As with clear cell RCC, hereditary forms of papillary and chromophobe RCC may hold the key to the identification of critical molecular events leading to these tumors both in familial and sporadic settings (Linehan, 2003; Linehan et al, 2009). Hereditary papillary renal cancer (HPRC) is a familial condition characterized by the predisposition of affected individuals to develop bilateral, multifocal papillary type I RCC. HPRC is characterized by the presence of activating germline mutations in the tyrosine kinase domain of the proto-oncogene c-MET, which is located on chromosome 7; this is usually accompanied by nonrandom duplication of the chromosome containing the mutated allele (Schmidt et al, 1997, 1998, 1999, 2004; Zhuang et al, 1998; Linehan, 2003; Linehan et al, 2009). c-MET is a cell surface receptor that is normally activated on binding its ligand, hepatocyte growth factor (HGF) but is rendered constitutionally active in the presence of mutations in the kinase domain of this protein (Bottaro et al, 1991; Dharmawardana et al, 2004; Peruzzi and Bottaro, 2006; Giubellino et al, 2009). The HGF/c-MET pathway is involved in regulating a variety of biologic functions, including cell growth, proliferation, and motility (Fig. 50–11) (Jeffers et al, 1998; Giubellino et al, 2009). Somatic c-MET alterations have been noted in a proportion of patients with sporadic papillary RCC, with activating mutations identified in approximately 13% of papillary tumors in one series. Trisomy 7 (both HGF and c-MET are located on chromosome 7) has been described in more than two thirds of papillary tumors and may represent an alternative mechanism contributing to activation of the c-MET pathway (Kovacs et al, 1991; Henke and Erbersdobler, 2002).
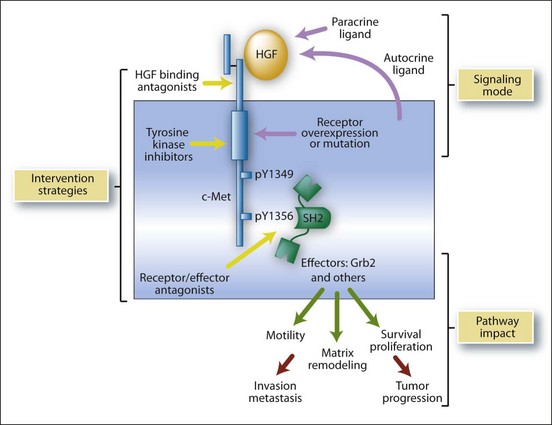
Figure 50–11 c-MET oncogenic signaling pathway. Important downstream biochemical and biologic consequences of c-MET activation (by HGF binding or by constitutive activation of the receptor tyrosine kinase activity by mutation) are represented. The c-MET ligand (HGF), c-MET tyrosine kinase activity, and downstream effectors (such as Grb-2) are potential targets for drugs in tumors with aberrant pathway activation.
(From Peruzzi B, Bottaro DP. Targeting the c-Met signaling pathway in cancer. Clin Cancer Res 2006;12:3657–60.)
VEGFR inhibitors active in clear cell RCC do not seem to be effective in papillary RCC. Sorafenib and sunitinib were shown to have minimal activity in retrospective studies of papillary RCC patients (Choueiri et al, 2008a). Prospective phase 2 trials with sunitinib have demonstrated that this agent is associated with low response rates (5% to 10%) in papillary RCC. The Southwest Oncology Group reported an 11% overall response rate with erlotinib, an oral EGFR inhibitor, in 52 patients with metastatic papillary RCC with a 6-month PFS of only 29%. More recently, the realization that c-MET activation may play an important role in some forms of papillary RCC has led to the evaluation of foretinib (formerly known as XL880 and GSK1363089), a novel tyrosine kinase inhibitor with activity against c-MET and VEGFR2 in this RCC subtype. A multicenter, open-label phase 2 trial studied two dosing regimens (an intermittent regimen in which the drug was administered orally at a dose of 240 mg/day for the first 5 days of every 14-day cycle and a continuous or daily dosing regimen at a dose of 80 mg/day) in patients with metastatic papillary RCC and those with bilateral multifocal disease (Srinivasan et al, 2009). A recent interim analysis in the first 53 patients enrolled on trial (including seven HPRC patients with germline c-MET mutations and bilateral multifocal tumors) has been reported in abstract form. Of 44 evaluable patients in both dosing regimens, 6 had a confirmed partial response (objective response rate 14%) and an additional 34 (77%) had stable disease as their best response. Striking responses were seen in some patients, including regression of renal tumors in patients with bilateral multifocal disease. The adverse event profile associated with this drug is similar to that observed with other VEGFR antagonists. Patients on this trial will be retrospectively stratified based on the presence or absence of c-MET pathway activation (germline or somatic c-MET mutation or trisomy 7 or 7q31 amplification in tumor tissue) to help explore the correlation between c-MET activation and response to foretinib. Foretinib and other agents targeting the c-MET pathway warrant further evaluation in patients with papillary RCC.
An exploratory subgroup analysis of patients enrolled in large randomized phase 3 study evaluating the efficacy of temsirolimus versus interferon in “poor prognosis” patients suggests that mTOR inhibitors may have activity in some non–clear cell variants. Approximately 20% of the patients enrolled in this trial had non–clear cell histologies (predominantly papillary RCC). The outcome of 37 patients with non–clear cell RCC treated with temsirolimus (both OS and PFS) was found to be better than that of 36 patients receiving interferon in this subgroup analysis. Although these data suggest that temsirolimus may have activity in some non–clear cell variants, these conclusions are limited by the fact that this is a subgroup analysis (Dutcher et al, 2009). Prospective studies evaluating the efficacy of mTOR inhibitors are required to clarify the role of these agents in different subsets of RCC.
Cytotoxic chemotherapy has been used with modest success in collecting duct carcinoma, a rare kidney cancer variant with similarities to urothelial malignancies. In a series of 23 patients with metastatic collecting duct carcinoma, a response rate of 26% (including one complete response) was reported with a regimen comprising gemcitabine and carboplatin. Median PFS (7.1 months) and OS (10.5 months) were modest (Oudard et al, 2007). In summary there is no standard approach of proven efficacy for most patients with non–clear cell RCC, although some promising approaches are being evaluated. Enrollment in suitable trials should be considered for all patients with non–clear cell RCC.
Atkins MB, Dutcher J, Weiss G, et al. Kidney cancer: the Cytokine Working Group experience (1986-2001): part I. IL-2-based clinical trials. Med Oncol. 2001;18:197-207.
Escudier B, Eisen T, Stadler WM, et al. Sorafenib in advanced clear-cell renal-cell carcinoma. N Engl J Med. 2007;356:125-134.
Escudier B, Pluzanska A, Koralewski P, et al. Bevacizumab plus interferon alfa-2a for treatment of metastatic renal cell carcinoma: a randomised, double-blind phase III trial. Lancet. 2007;370:2103-2111.
Flanigan RC, Salmon SE, Blumenstein BA, et al. Nephrectomy followed by interferon alfa-2b compared with interferon alfa-2b alone for metastatic renal-cell cancer. N Engl J Med. 2001;345:1655-1659.
Fyfe GA, Fisher RI, Rosenberg SA, et al. Long-term response data for 255 patients with metastatic renal cell carcinoma treated with high-dose recombinant interleukin-2 therapy. J Clin Oncol. 1996;14:2410-2411.
Hudes G, Carducci M, Tomczak P, et al. Temsirolimus, interferon alfa, or both for advanced renal-cell carcinoma. N Engl J Med. 2007;356:2271-2281.
Motzer RJ, Hutson TE, Tomczak P, et al. Sunitinib versus interferon alfa in metastatic renal-cell carcinoma. N Engl J Med. 2007;356:115-124.
Motzer RJ, Mazumdar M, Bacik J, et al. Survival and prognostic stratification of 670 patients with advanced renal cell carcinoma. J Clin Oncol. 1999;17:2530-2540.
Négrier S, Escudier B, Gomez F, et al. Prognostic factors of survival and rapid progression in 782 patients with metastatic renal carcinomas treated by cytokines: a report from the Groupe Francais d’Immunotherapie. Ann Oncol. 2002;13:1460-1468.
Rini BI, Halabi S, Rosenberg JE, et al. Bevacizumab plus interferon alfa compared with interferon alfa monotherapy in patients with metastatic renal cell carcinoma: CALGB 90206. J Clin Oncol. 2008;26:5422-5428.
Ada G. The coming of age of tumour immunotherapy. Immunol Cell Biol. 1999;77:180-185.
Amato RJ. Chemotherapy for renal cell carcinoma. Semin Oncol. 2000;27:177-186.
Artz AS, Van Besien K, Zimmerman T, et al. Long-term follow-up of nonmyeloablative allogeneic stem cell transplantation for renal cell carcinoma: the University of Chicago Experience. Bone Marrow Transplant. 2005;35:253-260.
Atkins M, Regan M, McDermott D, et al. Carbonic anhydrase IX expression predicts outcome of interleukin 2 therapy for renal cancer. Clin Cancer Res. 2005;11:3714-3721.
Atkins MB, Dutcher J, Weiss G, et al. Kidney cancer: the Cytokine Working Group experience (1986-2001): part I. IL-2-based clinical trials. Med Oncol. 2001;18:197-207.
Atkins MB, Hidalgo M, Stadler WM, et al. Randomized phase II study of multiple dose levels of CCI-779, a novel mammalian target of rapamycin kinase inhibitor, in patients with advanced refractory renal cell carcinoma. J Clin Oncol. 2004;22:909-918.
Atzpodien J, Kirchner H, Hanninen EL, et al. Interleukin-2 in combination with interferon-alpha and 5-fluorouracil for metastatic renal cell cancer. Eur J Cancer. 1993;29A(Suppl. 5):S6-S8.
Barkholt L, Bregni M, Remberger M, Blaise D, et al. Allogeneic haematopoietic stem cell transplantation for metastatic renal carcinoma in Europe. Ann Oncol. 2006;17:1134-1140.
Bennett RT, Lerner SE, Taub HC, et al. Cytoreductive surgery for stage IV renal cell carcinoma. J Urol. 1995;154:32-34.
Bhargava P, Esteves DA, Nosov O, Lipatov AA. Updated activity and safety results of a phase II randomized discontinuation trial (RDT) of AV-951, a potent and selective VEGFR1, 2, and 3 kinase inhibitor, in patients with renal cell carcinoma (RCC). J Clin Oncol. 27(Suppl.), 2009. 15s [abstract 5032]
Bloom HJ. Proceedings: Hormone-induced and spontaneous regression of metastatic renal cancer. Cancer. 1973;32:1066-1071.
Boon T, Coulie PG, Van den Eynde B. Tumor antigens recognized by T cells. Immunol Today. 1997;18:267-268.
Bottaro DP, Rubin JS, Faletto DL, et al. Identification of the hepatocyte growth factor receptor as the c-met proto-oncogene product. Science. 1991;251:802-804.
Braren V, Taylor JN, Pace W. Regression of metastatic renal carcinoma following nephrectomy. Urology. 1974;3:777-778.
Braybrooke JP, Vallis KA, Houlbrook S, et al. Evaluation of toremifene for reversal of multidrug resistance in renal cell cancer patients treated with vinblastine. Cancer Chemother Pharmacol. 2000;46:27-34.
Bregni M, Dodero A, Peccatori J, et al. Nonmyeloablative conditioning followed by hematopoietic cell allografting and donor lymphocyte infusions for patients with metastatic renal and breast cancer. Blood. 2002;99:4234-4236.
Bukowski RM. Natural history and therapy of metastatic renal cell carcinoma: the role of interleukin-2. Cancer. 1997;80:1198-1220.
Bukowski RM, Kabbinavar FF, Figlin RA, et al. Randomized phase II study of erlotinib combined with bevacizumab compared with bevacizumab alone in metastatic renal cell cancer. J Clin Oncol. 2007;25:4536-4541.
Bukowski RM, Olencki T, Wang Q, et al. Phase II trial of interleukin-2 and interferon-alpha in patients with renal cell carcinoma: clinical results and immunologic correlates of response. J Immunother. 1997;20:301-311.
Bukowski RM, Rayman P, Uzzo R, et al. Signal transduction abnormalities in T lymphocytes from patients with advanced renal carcinoma: clinical relevance and effects of cytokine therapy. Clin Cancer Res. 1998;4:2337-2347.
Childs R, Chernoff A, Contentin N, et al. Regression of metastatic renal-cell carcinoma after nonmyeloablative allogeneic peripheral-blood stem-cell transplantation. N Engl J Med. 2000;343:750-758.
Choueiri TK, Garcia JA, Elson P, et al. Clinical factors associated with outcome in patients with metastatic clear-cell renal cell carcinoma treated with vascular endothelial growth factor-targeted therapy. Cancer. 2007;110:543-550.
Choueiri TK, Plantade A, Elson P, et al. Efficacy of sunitinib and sorafenib in metastatic papillary and chromophobe renal cell carcinoma. J Clin Oncol. 2008;26:127-131.
Choueiri TK, Vaziri SA, Jaeger E, et al. von Hippel-Lindau gene status and response to vascular endothelial growth factor targeted therapy for metastatic clear cell renal cell carcinoma. J Urol. 2008;180:860-865. discussion 865–6
Cockman ME, Masson N, Mole DR, et al. Hypoxia inducible factor-alpha binding and ubiquitylation by the von Hippel-Lindau tumor suppressor protein. J Biol Chem. 2000;275:25733-25741.
Coppin C, Porzsolt F, Awa A, et al. Immunotherapy for advanced renal cell cancer. Cochrane Database Syst Rev 2005: CD001425.
Dekernion JB, Ramming KP, Smith RB. The natural history of metastatic renal cell carcinoma: a computer analysis. J Urol. 1978;120:148-152.
deKernion JB, Sarna G, Figlin R, et al. The treatment of renal cell carcinoma with human leukocyte alpha-interferon. J Urol. 1983;130:1063-1066.
Dharmawardana PG, Giubellino A, Bottaro DP. Hereditary papillary renal carcinoma type I. Curr Mol Med. 2004;4:855-868.
Dorval T, Négrier S, Chevreau C, et al. Randomized trial of treatment with cisplatin and interleukin-2 either alone or in combination with interferon-alpha-2a in patients with metastatic melanoma: a Federation Nationale des Centres de Lutte Contre le Cancer Multicenter, parallel study. Cancer. 1999;85:1060-1066.
Duan DR, Humphrey JS, Chen DY, et al. Characterization of the VHL tumor suppressor gene product: localization, complex formation, and the effect of natural inactivating mutations. Proc Natl Acad Sci U S A. 1995;92:6459-6463.
Dutcher JP, Atkins M, Fisher R, et al. Interleukin-2-based therapy for metastatic renal cell cancer: the Cytokine Working Group experience, 1989-1997. Cancer J Sci Am. 1997;3(Suppl. 1):S73-S78.
Dutcher JP, de Souza P, McDermott D, et al. Effect of temsirolimus versus interferon-alpha on outcome of patients with advanced renal cell carcinoma of different tumor histologies. Med Oncol. 2009;26:202-209.
Dutcher JP, Fisher RI, Weiss G, et al. Outpatient subcutaneous interleukin-2 and interferon-alpha for metastatic renal cell cancer: five-year follow-up of the Cytokine Working Group Study. Cancer J Sci Am. 1997;3:157-162.
Dutcher JP, Logan T, Gordon M, et al. Phase II trial of interleukin 2, interferon alpha, and 5-fluorouracil in metastatic renal cell cancer: a cytokine working group study. Clin Cancer Res. 2000;6:3442-3450.
Edwards MJ, Anderson JA, Angel JR, Harty JI. Spontaneous regression of primary and metastatic renal cell carcinoma. J Urol. 1996;155:1385.
Ellerhorst JA, Sella A, Amato RJ, et al. Phase II trial of 5-fluorouracil, interferon-alpha and continuous infusion interleukin-2 for patients with metastatic renal cell carcinoma. Cancer. 1997;80:2128-2132.
Escudier B, Bellmunt J, Negrier S, et al. Phase III trial of bevacizumab plus interferon alfa-2a in patients with metastatic renal cell carcinoma (AVOREN): final analysis of overall survival. J Clin Oncol. 2010;28(13):2144-2150.
Escudier B, Eisen T, Stadler WM, et al. Sorafenib in advanced clear-cell renal-cell carcinoma. N Engl J Med. 2007;356:125-134.
Escudier B, Eisen T, Stadler WM, et al. Sorafenib for treatment of renal cell carcinoma: final efficacy and safety results of the phase III treatment approaches in renal cancer global evaluation trial. J Clin Oncol. 2009;27:3312-3318.
Escudier B, Farace F, Angevin E, et al. Immunotherapy with interleukin-2 (IL2) and lymphokine-activated natural killer cells: improvement of clinical responses in metastatic renal cell carcinoma patients previously treated with IL2. Eur J Cancer. 1994;30A:1078-1083.
Escudier B, Farace F, Theodore C, et al. [Treatment of metastatic kidney neoplasms with a new interleukin 2 protocol: the experience of the Gustave-Roussy Institute. Bull Cancer. 1995;82:296-302.
Escudier B, Pluzanska A, Koralewski P, et al. Bevacizumab plus interferon alfa-2a for treatment of metastatic renal cell carcinoma: a randomised, double-blind phase III trial. Lancet. 2007;370:2103-2111.
Escudier B, Ravaud A, Fabbro M, et al. High-dose interleukin-2 two days a week for metastatic renal cell carcinoma: a FNCLCC multicenter study. J Immunother Emphasis Tumor Immunol. 1994;16:306-312.
Escudier B, Szczylik C, Hutson TE, et al. Randomized phase II trial of first-line treatment with sorafenib versus interferon alfa-2a in patients with metastatic renal cell carcinoma. J Clin Oncol. 2009;27:1280-1289.
Fallick ML, McDermott DF, LaRock D, et al. Nephrectomy before interleukin-2 therapy for patients with metastatic renal cell carcinoma. J Urol. 1997;158:1691-1695.
Feldman DR, Baum MS, Ginsberg MS, et al. Phase I trial of bevacizumab plus escalated doses of sunitinib in patients with metastatic renal cell carcinoma. J Clin Oncol. 2009;27:1432-1439.
Figlin RA, deKernion JB, Mukamel E, et al. Recombinant interferon alfa-2a in metastatic renal cell carcinoma: assessment of antitumor activity and anti-interferon antibody formation. J Clin Oncol. 1988;6:1604-1610.
Figlin RA, Pierce WC, Kaboo R, et al. Treatment of metastatic renal cell carcinoma with nephrectomy, interleukin-2 and cytokine-primed or CD8 (+) selected tumor infiltrating lymphocytes from primary tumor. J Urol. 1997;158:740-745.
Figlin RA, Thompson JA, Bukowski RM, et al. Multicenter, randomized, phase III trial of CD8 (+) tumor-infiltrating lymphocytes in combination with recombinant interleukin-2 in metastatic renal cell carcinoma. J Clin Oncol. 1999;17:2521-2529.
Finke JH, Rayman P, Hart L, et al. Characterization of tumor-infiltrating lymphocyte subsets from human renal cell carcinoma: specific reactivity defined by cytotoxicity, interferon-gamma secretion, and proliferation. J Immunother Emphasis Tumor Immunol. 1994;15:91-104.
Fisher RI, Coltman CAJr, Doroshow JH, et al. Metastatic renal cancer treated with interleukin-2 and lymphokine-activated killer cells: a phase II clinical trial. Ann Intern Med. 1988;108:518-523.
Fisher RI, Rosenberg SA, Fyfe G. Long-term survival update for high-dose recombinant interleukin-2 in patients with renal cell carcinoma. Cancer J Sci Am. 2000;6(Suppl. 1):S55-S57.
Fisher RI, Rosenberg SA, Sznol M, et al. High-dose aldesleukin in renal cell carcinoma: long-term survival update. Cancer J Sci Am. 1997;3(Suppl. 1):S70-S72.
Flanigan RC, Mickisch G, Sylvester R, et al. Cytoreductive nephrectomy in patients with metastatic renal cancer: a combined analysis. J Urol. 2004;171:1071-1076.
Flanigan RC, Salmon SE, Blumenstein BA, et al. Nephrectomy followed by interferon alfa-2b compared with interferon alfa-2b alone for metastatic renal-cell cancer. N Engl J Med. 2001;345:1655-1659.
Fojo AT, Shen DW, Mickley LA, et al. Intrinsic drug resistance in human kidney cancer is associated with expression of a human multidrug-resistance gene. J Clin Oncol. 1987;5:1922-1927.
Fossa SD, Martinelli G, Otto U, et al. Recombinant interferon alfa-2a with or without vinblastine in metastatic renal cell carcinoma: results of a European multi-center phase III study. Ann Oncol. 1992;3:301-305.
Friedel G, Hurtgen M, Penzenstadler M, et al. Resection of pulmonary metastases from renal cell carcinoma. Anticancer Res. 1999;19:1593-1596.
Fyfe GA, Fisher RI, Rosenberg SA, et al. Long-term response data for 255 patients with metastatic renal cell carcinoma treated with high-dose recombinant interleukin-2 therapy. J Clin Oncol. 1996;14:2410-2411.
Giubellino A, Linehan WM, Bottaro DP. Targeting the Met signaling pathway in renal cancer. Expert Rev Anticancer Ther. 2009;9:785-793.
Gnarra JR, Duan DR, Weng Y, et al. Molecular cloning of the von Hippel-Lindau tumor suppressor gene and its role in renal carcinoma. Biochim Biophys Acta. 1996;1242:201-210.
Haas CD, Coltman CAJr, Gottlieb JA, et al. Phase II evaluation of bleomycin: a Southwest Oncology Group study. Cancer. 1976;38:8-12.
Hahn DM, Schimpff SC, Ruckdeschel JC, Wiernik PH. Single-agent therapy for renal cell carcinoma: CCNU, vinblastine, thioTEPA, or bleomycin. Cancer Treat Rep. 1977;61:1585-1587.
Hainsworth JD, Sosman JA, Spigel DR, et al. Treatment of metastatic renal cell carcinoma with a combination of bevacizumab and erlotinib. J Clin Oncol. 2005;23:7889-7896.
Harris DT. Hormonal therapy and chemotherapy of renal-cell carcinoma. Semin Oncol. 1983;10:422-430.
Henke RP, Erbersdobler A. Numerical chromosomal aberrations in papillary renal cortical tumors: relationship with histopathologic features. Virchows Arch. 2002;440:604-609.
Herman JG, Latif F, Weng Y, et al. Silencing of the VHL tumor-suppressor gene by DNA methylation in renal carcinoma. Proc Natl Acad Sci U S A. 1994;91:9700-9704.
Hudes G, Carducci M, Tomczak P, et al. Temsirolimus, interferon alfa, or both for advanced renal-cell carcinoma. N Engl J Med. 2007;356:2271-2281.
Hudson CC, Liu M, Chiang GG, et al. Regulation of hypoxia-inducible factor 1 alpha expression and function by the mammalian target of rapamycin. Mol Cell Biol. 2002;22:7004-7014.
Iliopoulos O, Levy AP, Jiang C, et al. Negative regulation of hypoxia-inducible genes by the von Hippel-Lindau protein. Proc Natl Acad Sci U S A. 1996;93:10595-10599.
Iliopoulos O, Ohh M, Kaelin WGJr. pVHL19 is a biologically active product of the von Hippel-Lindau gene arising from internal translation initiation. Proc Natl Acad Sci U S A. 1998;95:11661-11666.
Ivan M, Kondo K, Yang H, et al. HIFalpha targeted for VHL-mediated destruction by proline hydroxylation: implications for O2 sensing. Science. 2001;292:464-468.
Jaakkola P, Mole DR, Tian YM, et al. Targeting of HIF-alpha to the von Hippel-Lindau ubiquitylation complex by O2-regulated prolyl hydroxylation. Science. 2001;292:468-472.
Jeffers M, Fiscella M, Webb CP, et al. The mutationally activated Met receptor mediates motility and metastasis. Proc Natl Acad Sci U S A. 1998;95:14417-14422.
Jemal A, Siegel R, Xu J, et al. Cancer statistics, 2010. CA Cancer J Clin. 2010;60(5):277-300. Erratum in CA Cancer J Clin 2011;61(2):133–4
Kaelin WGJr. The von Hippel-Lindau tumor suppressor gene and kidney cancer. Clin Cancer Res. 2004;10:6290S-6295S.
Kammula US, White DE, Rosenberg SA. Trends in the safety of high dose bolus interleukin-2 administration in patients with metastatic cancer. Cancer. 1998;83:797-805.
Kavolius JP, Mastorakos DP, Pavlovich C, et al. Resection of metastatic renal cell carcinoma. J Clin Oncol. 1998;16:2261-2266.
Kavoussi LR, Levine SR, Kadmon D, Fair WR. Regression of metastatic renal cell carcinoma: a case report and literature review. J Urol. 1986;135:1005-1007.
Kierney PC, van Heerden JA, Segura JW, Weaver AL. Surgeon’s role in the management of solitary renal cell carcinoma metastases occurring subsequent to initial curative nephrectomy: an institutional review. Ann Surg Oncol. 1994;1:345-352.
Kirkwood JM, Ernstoff MS, Davis CA, et al. Comparison of intramuscular and intravenous recombinant alpha-2 interferon in melanoma and other cancers. Ann Intern Med. 1985;103:32-36.
Kollender Y, Bickels J, Price WM, et al. Metastatic renal cell carcinoma of bone: indications and technique of surgical intervention. J Urol. 2000;164:1505-1508.
Kovacs G, Fuzesi L, Emanual A, Kung HF. Cytogenetics of papillary renal cell tumors. Genes Chromosomes Cancer. 1991;3:249-255.
Kudoh S, Redovan C, Rayman P, et al. Defective granzyme B gene expression and lytic response in T lymphocytes infiltrating human renal cell carcinoma. J Immunother. 1997;20:479-487.
Latif F, Tory K, Gnarra J, et al. Identification of the von Hippel-Lindau disease tumor suppressor gene. Science. 1993;260:1317-1320.
Law TM, Motzer RJ, Mazumdar M, et al. Phase III randomized trial of interleukin-2 with or without lymphokine-activated killer cells in the treatment of patients with advanced renal cell carcinoma. Cancer. 1995;76:824-832.
Linehan WM. Molecular targeting of VHL gene pathway in clear cell kidney cancer. J Urol. 2003;170:593-594.
Linehan WM, Bratslavsky G, Pinto PA, et al. Molecular diagnosis and therapy of kidney cancer. Annu Rev Med. 2010;61:329-343.
Linehan WM, Pinto PA, Bratslavsky G, et al. Hereditary kidney cancer: unique opportunity for disease-based therapy. Cancer. 2009;115:2252-2261.
Linehan WM, Pinto PA, Srinivasan R, et al. Identification of the genes for kidney cancer: opportunity for disease-specific targeted therapeutics. Clin Cancer Res. 2007;13:671s-679s.
Ling W, Rayman P, Uzzo R, et al. Impaired activation of NFkappaB in T cells from a subset of renal cell carcinoma patients is mediated by inhibition of phosphorylation and degradation of the inhibitor, IkappaBalpha. Blood. 1998;92:1334-1341.
Lotze MT, Matory YL, Rayner AA, et al. Clinical effects and toxicity of interleukin-2 in patients with cancer. Cancer. 1986;58:2764-2772.
Marcus SG, Choyke PL, Reiter R, et al. Regression of metastatic renal cell carcinoma after cytoreductive nephrectomy. J Urol. 1993;150:463-466.
Maxwell PH, Wiesener MS, Chang GW, et al. The tumour suppressor protein VHL targets hypoxia-inducible factors for oxygen-dependent proteolysis. Nature. 1999;399:271-275.
McDermott DF, Regan MM, Clark JI, et al. Randomized phase III trial of high-dose interleukin-2 versus subcutaneous interleukin-2 and interferon in patients with metastatic renal cell carcinoma. J Clin Oncol. 2005;23:133-141.
Medical Research Council. Interferon-alpha and survival in metastatic renal carcinoma: early results of a randomised controlled trial. Medical Research Council Renal Cancer Collaborators. Lancet. 1999;353:14-17.
Mekhail TM, Abou-Jawde RM, Boumerhi G, et al. Validation and extension of the Memorial Sloan-Kettering prognostic factors model for survival in patients with previously untreated metastatic renal cell carcinoma. J Clin Oncol. 2005;23:832-841.
Mertens WC, Eisenhauer EA, Jolivet J, et al. Docetaxel in advanced renal carcinoma: a phase II trial of the National Cancer Institute of Canada Clinical Trials Group. Ann Oncol. 1994;5:185-187.
Mertens WC, Eisenhauer EA, Moore M, et al. Gemcitabine in advanced renal cell carcinoma: a phase II study of the National Cancer Institute of Canada Clinical Trials Group. Ann Oncol. 1993;4:331-332.
Mickisch GH, Garin A, van Poppel H, et al. Radical nephrectomy plus interferon-alfa–based immunotherapy compared with interferon alfa alone in metastatic renal-cell carcinoma: a randomised trial. Lancet. 2001;358:966-970.
Middleton AWJr. Indications for and results of nephrectomy for metastatic renal cell carcinoma. Urol Clin North Am. 1980;7:711-717.
Middleton RG. Surgery for metastatic renal cell carcinoma. J Urol. 1967;97:973-977.
Minasian LM, Motzer RJ, Gluck L, et al. Interferon alfa-2a in advanced renal cell carcinoma: treatment results and survival in 159 patients with long-term follow-up. J Clin Oncol. 1993;11:1368-1375.
Motzer RJ, Bacik J, Murphy BA, et al. Interferon-alfa as a comparative treatment for clinical trials of new therapies against advanced renal cell carcinoma. J Clin Oncol. 2002;20:289-296.
Motzer RJ, Bukowski RM, Figlin RA, et al. Prognostic nomogram for sunitinib in patients with metastatic renal cell carcinoma. Cancer. 2008;113:1552-1558.
Motzer RJ, Escudier B, Oudard S, et al. Efficacy of everolimus in advanced renal cell carcinoma: a double-blind, randomised, placebo-controlled phase III trial. Lancet. 2008;372:449-456.
Motzer RJ, Hutson TE, Tomczak P, et al. Overall survival and updated results for sunitinib compared with interferon alfa in patients with metastatic renal cell carcinoma. J Clin Oncol. 2009;27:3584-3590.
Motzer RJ, Hutson TE, Tomczak P, et al. Sunitinib versus interferon alfa in metastatic renal-cell carcinoma. N Engl J Med. 2007;356:115-124.
Motzer RJ, Mazumdar M, Bacik J, et al. Survival and prognostic stratification of 670 patients with advanced renal cell carcinoma. J Clin Oncol. 1999;17:2530-2540.
Motzer RJ, Mazumdar M, Bacik J, et al. Effect of cytokine therapy on survival for patients with advanced renal cell carcinoma. J Clin Oncol. 2000;18:1928-1935.
Motzer RJ, Michaelson MD, Redman BG, et al. Activity of SU11248, a multitargeted inhibitor of vascular endothelial growth factor receptor and platelet-derived growth factor receptor, in patients with metastatic renal cell carcinoma. J Clin Oncol. 2006;24:16-24.
Motzer RJ, Rini BI, Bukowski RM, et al. Sunitinib in patients with metastatic renal cell carcinoma. JAMA. 2006;295:2516-2524.
Motzer RJ, Russo P. Systemic therapy for renal cell carcinoma. J Urol. 2000;163:408-417.
Murthy SC, Kim K, Rice TW, et al. Can we predict long-term survival after pulmonary metastasectomy for renal cell carcinoma? Ann Thorac Surg. 2005;79:996-1003.
Muss HB, Costanzi JJ, Leavitt R, et al. Recombinant alfa interferon in renal cell carcinoma: a randomized trial of two routes of administration. J Clin Oncol. 1987;5:286-291.
Nanus DM, Garino A, Milowsky MI, et al. Active chemotherapy for sarcomatoid and rapidly progressing renal cell carcinoma. Cancer. 2004;101:1545-1551.
Négrier S, Caty A, Lesimple T, et al. Treatment of patients with metastatic renal carcinoma with a combination of subcutaneous interleukin-2 and interferon alfa with or without fluorouracil. Groupe Français d’Immunotherapie, Fédération Nationale des Centres de Lutte Contre le Cancer. J Clin Oncol. 2000;18:4009-4015.
Négrier S, Escudier B, Gomez F, et al. Prognostic factors of survival and rapid progression in 782 patients with metastatic renal carcinomas treated by cytokines: a report from the Groupe Français d’Immunotherapie. Ann Oncol. 2002;13:1460-1468.
Négrier S, Escudier B, Lasset C, et al. Recombinant human interleukin-2, recombinant human interferon alfa-2a, or both in metastatic renal-cell carcinoma. Groupe Français d’Immunotherapie. N Engl J Med. 1998;338:1272-1278.
Négrier S, Gomez F, Douillard JY, et al. Prognostic factors of response or failure of treatment in patients with metastatic renal carcinomas treated by cytokines: a report from the Groupe Français d’Immunotherapie. World J Urol. 2005;23:161-165.
Négrier S, Maral J, Drevon M, et al. Long-term follow-up of patients with metastatic renal cell carcinoma treated with intravenous recombinant interleukin-2 in Europe. Cancer J Sci Am. 2000;6(Suppl. 1):S93-S98.
Négrier S, Perol D, Ravaud A, et al. Randomized study of intravenous versus subcutaneous interleukin-2, and IFNalpha in patients with good prognosis metastatic renal cancer. Clin Cancer Res. 2008;14:5907-5912.
Neidhart JA, Anderson SA, Harris JE, et al. Vinblastine fails to improve response of renal cancer to interferon alfa-n1: High response rate in patients with pulmonary metastases. J Clin Oncol. 1991;9:832-836.
Nickerson ML, Jaeger E, Shi Y, et al. Improved identification of von Hippel-Lindau gene alterations in clear cell renal tumors. Clin Cancer Res. 2008;14:4726-4734.
O’Dea MJ, Zincke H, Utz DC, Bernatz PE. The treatment of renal cell carcinoma with solitary metastasis. J Urol. 1978;120:540-542.
Ohh M, Park CW, Ivan M, et al. Ubiquitination of hypoxia-inducible factor requires direct binding to the beta-domain of the von Hippel-Lindau protein. Nat Cell Biol. 2000;2:423-427.
Oudard S, Banu E, Vieillefond A, et al. Prospective multicenter phase II study of gemcitabine plus platinum salt for metastatic collecting duct carcinoma: results of a GETUG (Groupe d’Études des Tumeurs Uro-Genitales) study. J Urol. 2007;177:1698-1702.
Patel PH, Senico PL, Curiel RE, Motzer RJ. Phase I study combining treatment with temsirolimus and sunitinib malate in patients with advanced renal cell carcinoma. Clin Genitourin Cancer. 2009;7:24-27.
Pause A, Lee S, Worrell RA, et al. The von Hippel-Lindau tumor-suppressor gene product forms a stable complex with human CUL-2, a member of the Cdc53 family of proteins. Proc Natl Acad Sci U S A. 1997;94:2156-2161.
Peruzzi B, Bottaro DP. Targeting the c-Met signaling pathway in cancer. Clin Cancer Res. 2006;12:3657-3660.
Piltz S, Meimarakis G, Wichmann MW, et al. Long-term results after pulmonary resection of renal cell carcinoma metastases. Ann Thorac Surg. 2002;73:1082-1087.
Pogrebniak HW, Haas G, Linehan WM, et al. Renal cell carcinoma: resection of solitary and multiple metastases. Ann Thorac Surg. 1992;54:33-38.
Pyrhonen S, Salminen E, Ruutu M, et al. Prospective randomized trial of interferon alfa-2a plus vinblastine versus vinblastine alone in patients with advanced renal cell cancer. J Clin Oncol. 1999;17:2859-2867.
Quesada JR. Role of interferons in the therapy of metastatic renal cell carcinoma. Urology. 1989;34:80-83. discussion 87–96
Quesada JR, Swanson DA, Gutterman JU. Phase II study of interferon alpha in metastatic renal-cell carcinoma: a progress report. J Clin Oncol. 1985;3:1086-1092.
Quesada JR, Swanson DA, Trindade A, Gutterman JU. Renal cell carcinoma: antitumor effects of leukocyte interferon. Cancer Res. 1983;43:940-947.
Rabinovitch RA, Zelefsky MJ, Gaynor JJ, Fuks Z. Patterns of failure following surgical resection of renal cell carcinoma: implications for adjuvant local and systemic therapy. J Clin Oncol. 1994;12:206-212.
Rackley R, Novick A, Klein E, et al. The impact of adjuvant nephrectomy on multimodality treatment of metastatic renal cell carcinoma. J Urol. 1994;152:1399-1403.
Ratain MJ, Eisen T, Stadler WM, et al. Phase II placebo-controlled randomized discontinuation trial of sorafenib in patients with metastatic renal cell carcinoma. J Clin Oncol. 2006;24:2505-2512.
Ravaud A, Audhuy B, Gomez F, et al. Subcutaneous interleukin-2, interferon alfa-2a, and continuous infusion of fluorouracil in metastatic renal cell carcinoma: a multicenter phase II trial. Groupe Français d’Immunotherapie. J Clin Oncol. 1998;16:2728-2732.
Ravaud A, Négrier S, Cany L, et al. Subcutaneous low-dose recombinant interleukin 2 and alpha-interferon in patients with metastatic renal cell carcinoma. Br J Cancer. 1994;69:1111-1114.
Rini BI, Halabi S, Barrier R, et al. Adoptive immunotherapy by allogeneic stem cell transplantation for metastatic renal cell carcinoma: a CALGB intergroup phase II study. Biol Blood Marrow Transplant. 2006;12:778-785.
Rini BI, Halabi S, Rosenberg JE, et al. Bevacizumab plus interferon alfa compared with interferon alfa monotherapy in patients with metastatic renal cell carcinoma: CALGB 90206. J Clin Oncol. 2008;26:5422-5428.
Rini B, Halabi S, Rosenberg JE, et al. Phase III trial of bevacizumab plus interferon alfa monotherapy in patients with metastatic renal cell carcinoma: final results of CALGB 90206. J Clin Oncol. 2010;28:2137-2143.
Rini BI, Michaelson MD, Rosenberg JE, et al. Antitumor activity and biomarker analysis of sunitinib in patients with bevacizumab-refractory metastatic renal cell carcinoma. J Clin Oncol. 2008;26:3743-3748.
Rini BI, Vogelzang NJ, Dumas MC, et al. Phase II trial of weekly intravenous gemcitabine with continuous infusion fluorouracil in patients with metastatic renal cell cancer. J Clin Oncol. 2000;18:2419-2426.
Rini BI, Weinberg V, Small EJ. A phase I trial of fixed dose rate gemcitabine and capecitabine in metastatic renal cell carcinoma. Cancer. 2005;103:553-558.
Rini BI, Zimmerman T, Stadler WM, et al. Allogeneic stem-cell transplantation of renal cell cancer after nonmyeloablative chemotherapy: feasibility, engraftment, and clinical results. J Clin Oncol. 2002;20:2017-2024.
Rini BI, Zimmerman TM, Gajewski TF, et al. Allogeneic peripheral blood stem cell transplantation for metastatic renal cell carcinoma. J Urol. 2001;165:1208-1209.
Rixe O, Bukowski RM, Michaelson MD, et al. Axitinib treatment in patients with cytokine-refractory metastatic renal-cell cancer: a phase II study. Lancet Oncol. 2007;8:975-984.
Robson CJ. The natural history of renal cell carcinoma. Prog Clin Biol Res. 1982;100:447-452.
Rosenberg SA. A new era for cancer immunotherapy based on the genes that encode cancer antigens. Immunity. 1999;10:281-287.
Rosenberg SA, Lotze MT, Muul LM, et al. A progress report on the treatment of 157 patients with advanced cancer using lymphokine-activated killer cells and interleukin-2 or high-dose interleukin-2 alone. N Engl J Med. 1987;316:889-897.
Rosenberg SA, Lotze MT, Yang JC, et al. Experience with the use of high-dose interleukin-2 in the treatment of 652 cancer patients. Ann Surg. 1989;210:474-484. discussion 484–5
Rosenberg SA, Lotze MT, Yang JC, et al. Combination therapy with interleukin-2 and alpha-interferon for the treatment of patients with advanced cancer. J Clin Oncol. 1989;7:1863-1874.
Rosenberg SA, Lotze MT, Yang JC, et al. Prospective randomized trial of high-dose interleukin-2 alone or in conjunction with lymphokine-activated killer cells for the treatment of patients with advanced cancer. J Natl Cancer Inst. 1993;85:622-632.
Rosenberg SA, Yang JC, White DE, Steinberg SM. Durability of complete responses in patients with metastatic cancer treated with high-dose interleukin-2: identification of the antigens mediating response. Ann Surg. 1998;228:307-319.
Russo P, Synder M, Vickers A, et al. Cytoreductive nephrectomy and nephrectomy/complete metastasectomy for metastatic renal cancer. Sci World J. 2007;7:768-778.
Schmidt L, Duh FM, Chen F, et al. Germline and somatic mutations in the tyrosine kinase domain of the MET proto-oncogene in papillary renal carcinomas. Nat Genet. 1997;16:68-73.
Schmidt L, Junker K, Nakaigawa N, et al. Novel mutations of the MET proto-oncogene in papillary renal carcinomas. Oncogene. 1999;18:2343-2350.
Schmidt L, Junker K, Weirich G, et al. Two North American families with hereditary papillary renal carcinoma and identical novel mutations in the MET proto-oncogene. Cancer Res. 1998;58:1719-1722.
Schmidt LS, Nickerson ML, Angeloni D, et al. Early onset hereditary papillary renal carcinoma: germline missense mutations in the tyrosine kinase domain of the met proto-oncogene. J Urol. 2004;172:1256-1261.
Schomburg A, Kirchner H, Fenner M, et al. Lack of therapeutic efficacy of tamoxifen in advanced renal cell carcinoma. Eur J Cancer. 1993;29A:737-740.
Sella A, Logothetis CJ, Fitz K, et al. Phase II study of interferon-alpha and chemotherapy (5-fluorouracil and mitomycin C) in metastatic renal cell cancer. J Urol. 1992;147:573-577.
Sella A, Swanson DA, Ro JY, et al. Surgery following response to interferon-alpha-based therapy for residual renal cell carcinoma. J Urol. 1993;149:19-21. discussion 21–2
Sheehan JP, Sun MH, Kondziolka D, et al. Radiosurgery in patients with renal cell carcinoma metastasis to the brain: long-term outcomes and prognostic factors influencing survival and local tumor control. J Neurosurg. 2003;98:342-349.
Silber SJ, Chen CY, Gould F. Regression of metastases after nephrectomy for renal cell carcinoma. Br J Urol. 1975;47:259-261.
Skinner DG, Colvin RB, Vermillion CD, et al. Diagnosis and management of renal cell carcinoma: a clinical and pathologic study of 309 cases. Cancer. 1971;28:1165-1177.
Snow RM, Schellhammer PF. Spontaneous regression of metastatic renal cell carcinoma. Urology. 1982;20:177-181.
Srinivasan R, Linehan WM, Vaishampayan U, et al. A phase II study of two dosing regimens of GSK 1363089 (GSK089), a dual MET/VEGFR2 inhibitor, in patients (pts) with papillary renal carcinoma (PRC). J Clin Oncol. 27(Suppl.), 2009. 15s [abstract 5103]
Stadler WM, Halabi S, Rini B, et al. A phase II study of gemcitabine and capecitabine in metastatic renal cancer: a report of Cancer and Leukemia Group B protocol 90008. Cancer. 2006;107:1273-1279.
Stebbins CE, Kaelin WGJr, Pavletich NP. Structure of the VHL-ElonginC-ElonginB complex: implications for VHL tumor suppressor function. Science. 1999;284:455-461.
Sternberg CN, Davis ID, Mardiak J, et al. Pazopanib in locally advanced or metastatic renal cell carcinoma: results of a randomized phase III trial. J Clin Oncol. 2010;28(6):1061-1068.
Sundaresan N, Scher H, DiGiacinto GV, et al. Surgical treatment of spinal cord compression in kidney cancer. J Clin Oncol. 1986;4:1851-1856.
Takahashi Y, Harashima N, Kajigaya S, et al. Regression of human kidney cancer following allogeneic stem cell transplantation is associated with recognition of an HERV-E antigen by T cells. J Clin Invest. 2008;118:1099-1109.
Tamaskar I, Garcia JA, Elson P, et al. Antitumor effects of sunitinib or sorafenib in patients with metastatic renal cell carcinoma who received prior antiangiogenic therapy. J Urol. 2008;179:81-86. discussion 86
Thomas ED, Buckner CD, Banaji M, et al. One hundred patients with acute leukemia treated by chemotherapy, total body irradiation, and allogeneic marrow transplantation. Blood. 1977;49:511-533.
Thomas GV, Tran C, Mellinghoff IK, et al. Hypoxia-inducible factor determines sensitivity to inhibitors of mTOR in kidney cancer. Nat Med. 2006;12:122-127.
Tolia BM, Whitmore WFJr. Solitary metastasis from renal cell carcinoma. J Urol. 1975;114:836-838.
Tourani JM, Pfister C, Berdah JF, et al. Outpatient treatment with subcutaneous interleukin-2 and interferon alfa administration in combination with fluorouracil in patients with metastatic renal cell carcinoma: results of a sequential nonrandomized phase II study. Subcutaneous Administration Propeukin Program Cooperative Group. J Clin Oncol. 1998;16:2505-2513.
Tourani JM, Pfister C, Tubiana N, et al. Subcutaneous interleukin-2 and interferon alfa administration in patients with metastatic renal cell carcinoma: final results of SCAPP III, a large, multicenter, phase II, nonrandomized study with sequential analysis design—the Subcutaneous Administration Propeukin Program Cooperative Group. J Clin Oncol. 2003;21:3987-3994.
Tsui KH, Shvarts O, Smith RB, et al. Prognostic indicators for renal cell carcinoma: a multivariate analysis of 643 patients using the revised 1997 TNM staging criteria. J Urol. 2000;163:1090-1095. quiz 1295
Umeda T, Niijima T. Phase II study of alpha interferon on renal cell carcinoma: summary of three collaborative trials. Cancer. 1986;58:1231-1235.
Uzzo RG, Clark PE, Rayman P, et al. Alterations in NFkappaB activation in T lymphocytes of patients with renal cell carcinoma. J Natl Cancer Inst. 1999;91:718-721.
Uzzo RG, Rayman P, Kolenko V, et al. Mechanisms of apoptosis in T cells from patients with renal cell carcinoma. Clin Cancer Res. 1999;5:1219-1229.
Wagner JR, Walther MM, Linehan WM, et al. Interleukin-2 based immunotherapy for metastatic renal cell carcinoma with the kidney in place. J Urol. 1999;162:43-45.
Walther MM, Patel B, Choyke PL, et al. Hypercalcemia in patients with metastatic renal cell carcinoma: effect of nephrectomy and metabolic evaluation. J Urol. 1997;158:733-739.
Walther MM, Yang JC, Pass HI, et al. Cytoreductive surgery before high dose interleukin-2 based therapy in patients with metastatic renal cell carcinoma. J Urol. 1997;158:1675-1678.
Weiden PL, Flournoy N, Thomas ED, et al. Antileukemic effect of graft-versus-host disease in human recipients of allogeneic-marrow grafts. N Engl J Med. 1979;300:1068-1073.
Weiden PL, Sullivan KM, Flournoy N, et al. Antileukemic effect of chronic graft-versus-host disease: contribution to improved survival after allogeneic marrow transplantation. N Engl J Med. 1981;304:1529-1533.
Yagoda A, Abi-Rached B, Petrylak D. Chemotherapy for advanced renal-cell carcinoma: 1983-1993. Semin Oncol. 1995;22:42-60.
Yang JC, Haworth L, Sherry RM, et al. A randomized trial of bevacizumab, an anti-vascular endothelial growth factor antibody, for metastatic renal cancer. N Engl J Med. 2003;349:427-434.
Yang JC, Sherry RM, Steinberg SM, et al. Randomized study of high-dose and low-dose interleukin-2 in patients with metastatic renal cancer. J Clin Oncol. 2003;21:3127-3132.
Zaniboni A, Simoncini E, Marpicati P, et al. Phase II trial of 5-fluorouracil and high-dose folinic acid in advanced renal cell cancer. J Chemother. 1989;1:350-351.
Zhuang Z, Emmert-Buck MR, Roth MJ, et al. von Hippel-Lindau disease gene deletion detected in microdissected sporadic human colon carcinoma specimens. Hum Pathol. 1996;27:152-156.
Zhuang Z, Gnarra JR, Dudley CF, et al. Detection of von Hippel-Lindau disease gene mutations in paraffin-embedded sporadic renal cell carcinoma specimens. Mod Pathol. 1996;9:838-842.
Zhuang Z, Park WS, Pack S, et al. Trisomy 7-harbouring non-random duplication of the mutant MET allele in hereditary papillary renal carcinomas. Nat Genet. 1998;20:66-69.