Surgery for Malignancy
Radical Nephrectomy
Indications and Evaluation
Radical nephrectomy involves the en-bloc removal of the kidney outside the Gerota fascia together with the ipsilateral adrenal gland as well as complete regional lymphadenectomy from the crus of the diaphragm to the aortic bifurcation (Robson et al, 1969; Skinner et al, 1971). Whereas partial nephrectomy is the treatment of choice for T1 tumors when feasible, radical nephrectomy remains the definitive therapy for T2 or larger tumors.
Clinical staging of renal cell carcinoma involves radiologic evaluation of tumor size, extension beyond the Gerota fascia and into adjacent structures, venous involvement, and evaluation for nodal and metastatic disease. Renal cell carcinoma is staged with the 2010 American Joint Committee on Cancer (AJCC) TNM staging classification (Tables 54-10 and 54-11). Preoperative radiographic studies such as ultrasonography, CT, and MRI are important. Through preoperative imaging, one is able to determine the clinical stage of the tumor, define the anatomy of the affected kidney, and assess the structure or function of the contralateral kidney. All patients should undergo a metastatic evaluation including a chest radiograph and an abdominal CT scan. A bone scintiscan is indicated in patients with bone pain or elevated serum levels of alkaline phosphatase. Preoperative renal, cardiac, and pulmonary evaluations are essential, as described earlier.
Table 54–10 TNM Staging of Renal Cancer
| Primary Tumor (T) | |
| TX | Primary tumor cannot be assessed. |
| T0 | No evidence of primary tumor |
| T1 | Tumor ≤7 cm in greatest dimension, limited to the kidney |
| T1a | Tumor ≤4 cm in greatest dimension, limited to the kidney |
| T1b | Tumor >4 cm but not >7 cm in greatest dimension limited to the kidney |
| T2 | Tumor >7 cm in greatest dimension, limited to the kidney |
| T2a | Tumor >7 cm but ≤10 cm in greatest dimension, limited to the kidney |
| T2b | Tumor >10 cm, limited to the kidney |
| T3 | Tumor extends into the major veins or perinephric tissues but not into the ipsilateral adrenal gland and not beyond the Gerota fascia |
| T3a | Tumor grossly extends into the renal vein or its segmental (muscle-containing) branches or invades perirenal and/or renal sinus fat but not beyond the Gerota fascia |
| T3b | Tumor grossly extends into the vena cava below the diaphragm |
| T3c | Tumor grossly extends into the vena cava above the diaphragm or invades the wall of the vena cava |
| T4 | Tumor invades beyond the Gerota fascia (including contiguous extension into the ipsilateral adrenal gland) |
| Regional Lymph Nodes (N) | |
| NX | Regional lymph nodes cannot be assessed. |
| N0 | No regional lymph node metastasis |
| N1 | Metastasis in regional lymph node(s) |
| Distant Metastasis (M) | |
| M0 | No distant metastasis |
| M1 | Distant metastasis |
From the AJCC cancer staging manual. 7th ed. New York: Springer-Verlag; 2010. p. 482. Used with permission of the American Joint Committee on Cancer, Chicago, Illinois.
Tumor thrombus involving the IVC should be suspected in patients with lower extremity edema, varicocele, dilated superficial abdominal veins, proteinuria, pulmonary embolism, right atrial mass, or nonfunction of the involved kidney. MR venography is the authors’ preferred study for evaluating the presence and the distal extent of involvement of the IVC (Pritchett et al, 1987; Goldfarb et al, 1990) (Fig. 54–86). The use of invasive inferior vena cavography is limited to those patients with a nondiagnostic MR venogram or ultrasound evaluation, in those in whom MRI is contraindicated, and in patients who underwent preoperative angioinfarction with coils that would produce scatter leading to poor quality MR images (Fig. 54–87). Transesophageal echocardiography (Treiger et al, 1991; Glazer and Novick, 1997) and transabdominal Doppler ultrasonography (McGahan et al, 1993) are also useful. In patients with known IVC tumor thrombus the authors perform intraoperative transesophageal echocardiography to evaluate the distal extent of the tumor thrombus before making the incision.
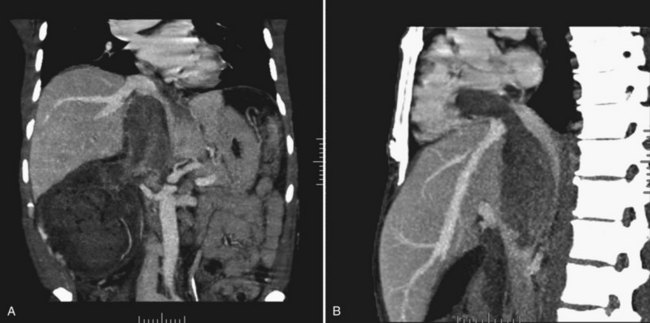
Figure 54–86 A, Coronal MR image demonstrates tumor extending to the level of the diaphragm. B, Sagittal MR reconstruction demonstrates thrombus that extends farther into the right atrium. The patient would eventually require cardiopulmonary bypass and deep hypothermic circulatory arrest.
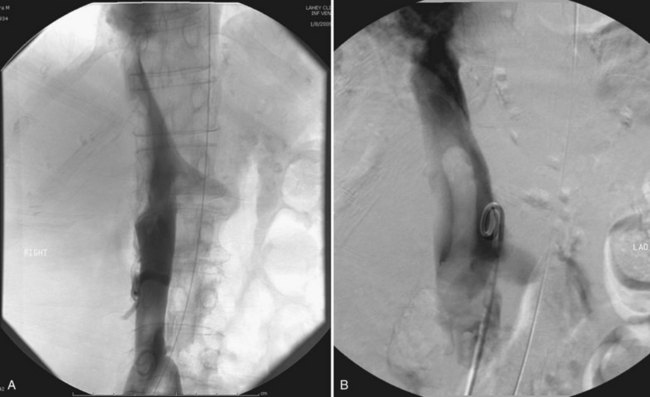
Figure 54–87 A and B, Preoperative cavogram demonstrating filling defect at the T10 level consistent with tumor thrombus in a patient with a large right renal mass. The patient required cardiopulmonary bypass and deep hypothermic circulatory arrest.
As discussed earlier, the senior author favors preoperative angioinfarction for large renal tumors with hypervascular characteristics or IVC tumor thrombus (Wotkowicz and Libertino, 2007). Proposed benefits of preoperative RAE include shrinkage of an arterialized tumor thrombus to ease surgical removal and reduce blood loss. When cardiopulmonary bypass (CPB) with deep hypothermic circulatory arrest is anticipated, surgeons at some institutions will also perform preoperative coronary angiography (Belis et al, 1989; Novick et al, 1990b). Coronary lesions can be repaired concurrently during CPB.
Technique
The key elements of radical nephrectomy include the surgical approach, early ligation of the renal artery and vein, removal of the kidney outside the Gerota fascia, removal of the ipsilateral adrenal gland, and a complete regional lymphadenectomy extending from the crus of the diaphragm to the aortic bifurcation.
Surgical approaches are determined by tumor location and size, body habitus, prior surgical history, and surgeon preference. Although simple nephrectomy is often accomplished through an extraperitoneal approach, radical nephrectomy is best performed through a transperitoneal incision. This permits abdominal exploration for metastatic disease and access to the pedicle before significant manipulation of the tumor. The senior author prefers either an anterior or a thoracoabdominal approach.
Anterior Approach
A transperitoneal anterior midline, anterior subcostal, or chevron incision is made. The abdomen is systematically examined for metastatic disease. The approach for left and right tumors is described.
Right
The right kidney is approached by taking down the hepatic flexure and incising the peritoneal reflection along the right paracolic gutter. This permits the colon to be reflected medially in a plane anterior to the Gerota fascia. A Kocher maneuver, in which the lateral peritoneal reflection of the duodenum is incised and the duodenum reflected medially, is used subsequently to expose the underlying IVC and pararenal aorta (see Fig. 54–90). These maneuvers typically provide adequate access to the right renal pedicle and great vessels.
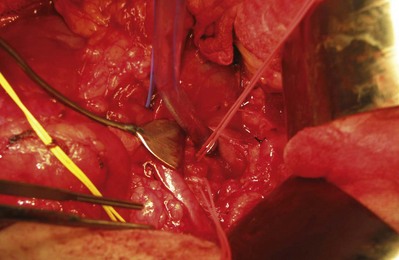
The right renal vein, which is relatively short and does not typically receive large tributaries, is mobilized. The gonadal vein enters the IVC and is ligated before the dissection of the right renal vein to avoid troublesome bleeding. The right renal artery lies posterior to the renal vein along its superior margin and normally runs a retrocaval course (see Fig. 54–37). The renal artery can be isolated to the right of the vena cava. Alternatively, in cases of a large medial tumor or significant nodal disease in the hilum, the renal artery can be controlled between the vena cava and aorta (Fig. 54–88).
Left
On the left side the white line of Toldt is incised and the colon is reflected medially to expose the great vessels. Division of the splenocolic ligaments reduces traction on and injury to the spleen. This approach permits access to the renal pedicle both anteriorly and posteriorly, which is the authors’ preference. The left renal artery can be isolated posteriorly and the venous system identified and controlled anteriorly.
The left renal vein passes anterior to the aorta and is longer than the right renal vein. Unlike the right side it receives gonadal, adrenal, and lumbar tributaries, which are ligated and divided. Identifying the left renal artery from the posterior approach is recommended to avoid inadvertent ligation of the superior mesenteric artery, which is on the anterior surface of the aorta 1 to 2 cm cephalad to the left renal vein. Alternatively, the artery can be isolated from an anterior approach by retracting the mobilized vein. The artery is mobilized toward the aorta, ligated, and divided. Two large Hem-o-Lok clips and a 0 silk suture ligature are used to secure the main renal artery with a silk tie on the specimen side, followed by the vein (see Fig. 54–89 [on the Expert Consult website![]() ] and Figs. 54-90 and 54-91). Preoperative arterial embolization permits venous ligation to be performed first, which is occasionally necessary in complex dissections.
] and Figs. 54-90 and 54-91). Preoperative arterial embolization permits venous ligation to be performed first, which is occasionally necessary in complex dissections.
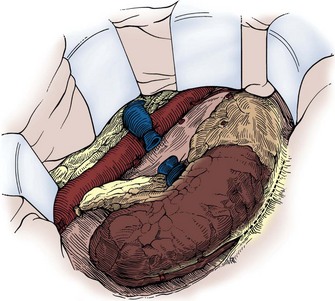
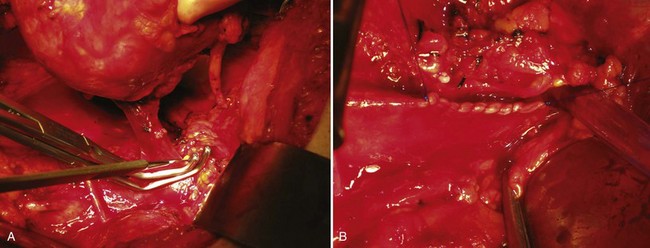
Figure 54–90 After the pedicle is secured and the ureter divided, the kidney is mobilized outside the Gerota fascia.
(Reproduced with permission from Novick AC, Streem SB, Pontes E, editors. Stewart’s operative urology. 2nd ed. Baltimore: Williams & Wilkins; 1989.)
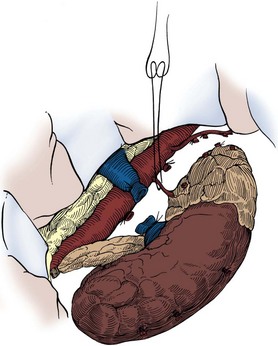
Figure 54–91 Remaining medial vascular attachments are secured and divided to complete the nephrectomy.
(Reproduced with permission from Novick AC, Streem SB, Pontes E, editors. Stewart’s operative urology. 2nd ed. Baltimore: Williams & Wilkins; 1989.)
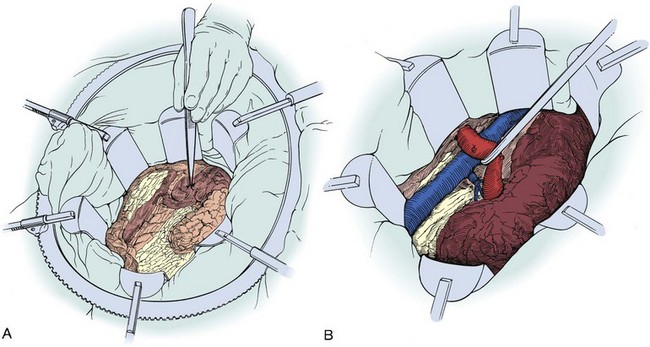
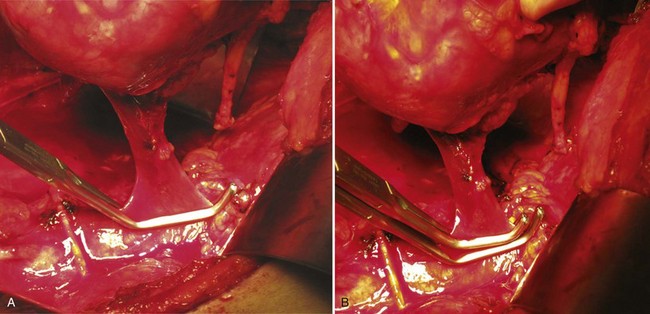
Figure 54–89 A, Self-retraining ring retractor is inserted to maintain exposure. B, Left renal vein is mobilized by ligation of its major branches to expose the artery posteriorly.
(Reproduced with permission from Novick AC, Streem SB, Pontes E, editors. Stewart’s operative urology. 2nd ed. Baltimore: Williams & Wilkins; 1989.)
Mobilization of Kidney and Completion of the Case
After dividing the renal pedicle, the kidney is mobilized outside the Gerota fascia with blunt and sharp dissection. Any remaining vascular attachments are ligated with nonabsorbable sutures and divided. The ureter is ligated and divided. The kidney and adrenal gland are removed. A Penrose drain is placed in the empty renal fossa and externalized through a separate stab incision. The abdomen is closed in layers as previously described.
Thoracoabdominal Approach
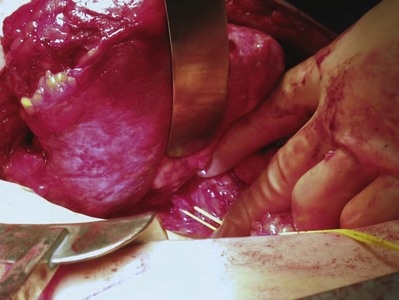
For large renal tumors, especially those of the upper pole, the thoracoabdominal approach is preferable to an anterior approach (Fig. 54–92; see Fig. 54–93 on the Expert Consult website![]() ). On the right side the liver is retracted cephalad. The hepatic flexure of the colon and the duodenum are reflected medially, exposing the IVC, the renal pedicle, and the anterior surface of the kidney (Figs. 54-94 to 54-96). The renal artery and vein are handled as described for the anterior approach. The ureter and right gonadal vein are ligated and divided. The kidney is mobilized outside the Gerota fascia down to the psoas fascia. The additional vascular attachments to the upper pole tumor and adrenal gland are exposed by medial retraction of the IVC along with caudal and lateral retraction of the kidney. They are ligated and divided. A curved-shaped clamp is passed along the superior portion of the adrenal gland to gain control of the phrenic vessels using a 0 silk suture ligature. The specimen is mobilized fully away from the liver and removed en bloc.
). On the right side the liver is retracted cephalad. The hepatic flexure of the colon and the duodenum are reflected medially, exposing the IVC, the renal pedicle, and the anterior surface of the kidney (Figs. 54-94 to 54-96). The renal artery and vein are handled as described for the anterior approach. The ureter and right gonadal vein are ligated and divided. The kidney is mobilized outside the Gerota fascia down to the psoas fascia. The additional vascular attachments to the upper pole tumor and adrenal gland are exposed by medial retraction of the IVC along with caudal and lateral retraction of the kidney. They are ligated and divided. A curved-shaped clamp is passed along the superior portion of the adrenal gland to gain control of the phrenic vessels using a 0 silk suture ligature. The specimen is mobilized fully away from the liver and removed en bloc.
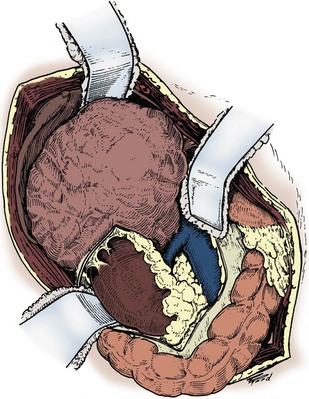
Figure 54–94 Exposure of large right upper pole tumor through a thoracoabdominal incision.
(Reproduced with permission from Novick AC, Streem SB, Pontes E, editors. Stewart’s operative urology. 2nd ed. Baltimore: Williams & Wilkins; 1989.)
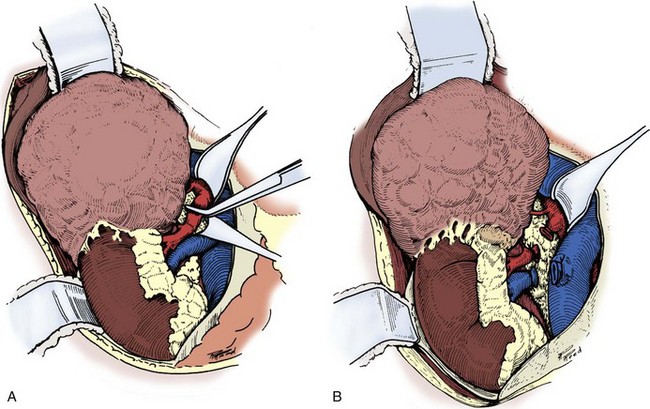
Figure 54–95 A and B, The renal artery and vein are secured and divided.
(Reproduced with permission from Novick AC, Streem SB, Pontes E, editors. Stewart’s operative urology. 2nd ed. Baltimore: Williams & Wilkins; 1989.)
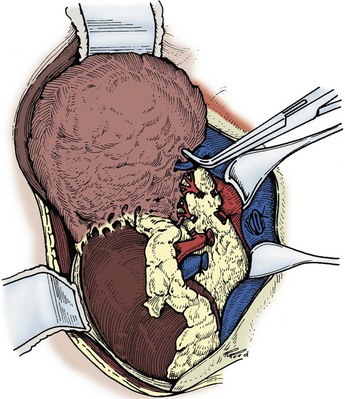
Figure 54–96 The vena cava is retracted medially to expose the remaining superior vascular attachments, which are secured and divided.
(Reproduced with permission from Novick AC, Streem SB, Pontes E, editors. Stewart’s operative urology. 2nd ed. Baltimore: Williams & Wilkins; 1989.)
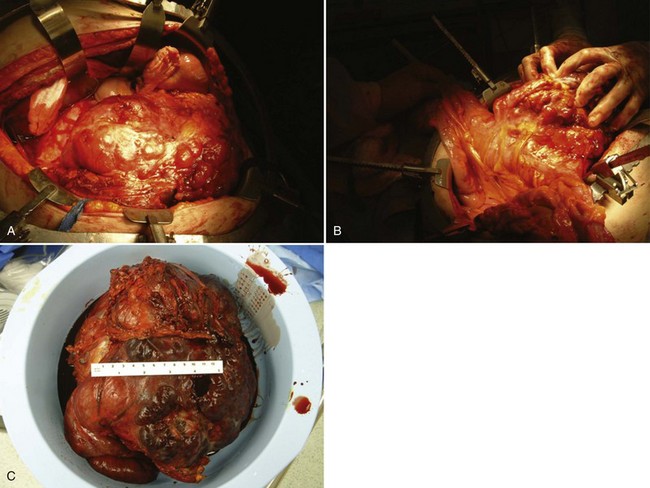
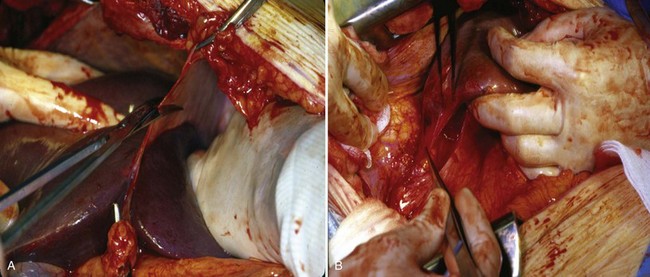
Figure 54–93 A, Operative photos demonstrate local invasion into the colon and gallbladder. B, Exposure of the entire retroperitoneum required for massive en-bloc resection. C, Surgical specimen.
On the left side the splenic flexure and the descending colon are mobilized. After the left renal pedicle is secured the kidney is mobilized outside the Gerota fascia. The upper border is dissected from the spleen, and the medial border is dissected from the tail of the pancreas. The ureter is ligated, and the specimen is removed.
Ipsilateral Adrenalectomy
Ipsilateral adrenalectomy may be associated with increased morbidity, adrenal insufficiency, and longer operative times. In 511 patients, Tsui and colleagues failed to demonstrate a correlation between tumor size and adrenal involvement, although presence of the tumor in the upper pole was a prognostic factor for adrenal involvement. Mordhorst and associates suggested that an adrenal-sparing approach should be taken in patients with tumors of less than 8 cm and no evidence of lymph node or organ metastasis. Contrary to the classic description of radical nephrectomy, removal of the ipsilateral adrenal gland is not routinely necessary unless the tumor either extensively involves the kidney or is located in the upper portion of the kidney (Sagalowsky et al, 1994).
Regional Lymphadenectomy
Complete regional lymphadenectomy, a traditional component of radical nephrectomy, encompasses removal of lymphatic tissue from the crus of the diaphragm to the aortic bifurcation. The lymph nodes can be removed en bloc with the kidney and adrenal gland or separately after completing the nephrectomy.
One enters and proceeds along a plane close to the adventitia of the aorta, which permits removal of all periaortic lymph tissue. One must be vigilant to avoid injury to the celiac trunk and superior mesenteric artery, which emanate from the anterior aorta. Proceeding downward to the origin of the inferior mesenteric artery, the sympathetic ganglia and nerves are removed together with the lymphatic tissue. The cisterna chyli, which is located medial to the right crus, is secured along with entering lymphatic vessels. After lymphadenectomy, the surgical field is copiously irrigated with sterile water. The argon laser beam can be helpful for hemostasis in extensive lymphadenectomies. Penrose drains are left for at least 48 hours.
Although lymphadenectomy is a recognized component of pathologic staging, the impact of lymphadenectomy on progression-free and overall survival is controversial. The European Organization for Research and Treatment of Cancer (EORTC) trial 30881, a randomized trial comparing radical nephrectomy with lymphadenectomy to radical nephrectomy alone in patients with clinical N0M0 renal cell carcinoma, did not demonstrate a survival benefit (Blom et al, 2009). The role of lymph node dissection in patients with locally advanced disease, clinical suspicion of nodal disease, or metastatic disease is unclear. There may be a subset of patients with micrometastatic lymph node involvement who can benefit from lymphadenectomy (Giuliani et al, 1990). Prospective trials of patients at higher risk of lymph node metastasis are needed.
Key Points: Radical Nephrectomy
Venous Extension and Tumor Thrombectomy
Renal cell carcinoma is associated with venous tumor thrombus in 4% to 10% of cases (Schefft et al, 1978). In the absence of metastases, an aggressive surgical approach with curative intent is justified. Five-year survival rates of 40% to 68% have been reported after radical nephrectomy and tumor thrombectomy (Libertino et al, 1987; Neves and Zincke, 1987; Skinner et al, 1989; Novick et al, 1990b). Although the level of IVC involvement is not predictive of survival, tumor thrombus limited to the renal vein is associated with improved survival compared with thrombus in the IVC (Moinzadeh and Libertino, 2004). The most favorable outcomes are achieved when the tumor does not involve the perinephric fat or regional lymph nodes (Cherrie et al, 1982). Although there are several systems for categorizing venous tumor thrombi based on the distal extent of the thrombus, a rational and commonly employed scheme that is preferred by the senior author is shown in Figure 54–97 and Table 54–12.
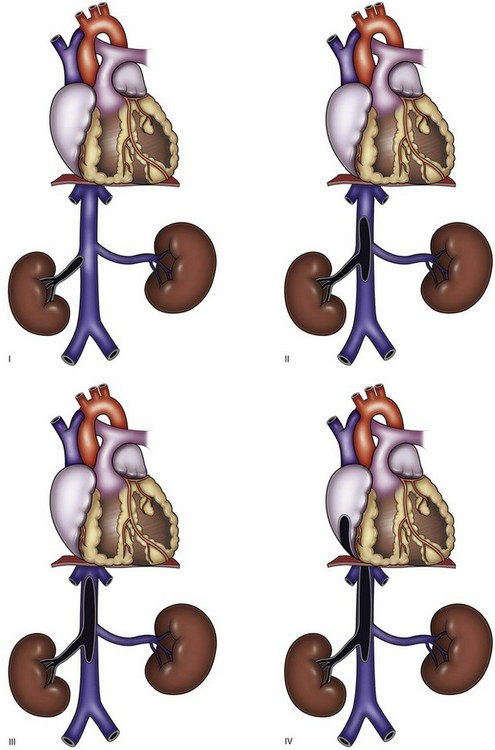
Figure 54–97 Classification of venous tumor thrombus extension.
(From Wang GJ, Carpenter JP, Fairman RM, et al. Single-center experience of caval thrombectomy in patients with renal cell carcinoma with tumor thrombus extension into the inferior vena cava. Vasc Endovasc Surg 2008;42[4]:335–40.)
Table 54–12 Classification of Venous Tumor Thrombus Extension
| LEVEL | DISTAL EXTENT OF THROMBUS |
|---|---|
| I | Limited to renal vein |
| II | Infrahepatic inferior vena cava |
| III | Retrohepatic inferior vena cava in proximity to renal veins |
| IV | Supradiaphragmatic inferior vena cava or right atrium |
Renal Vein Tumor Thrombus (Level I)
Tumor thrombus in the renal vein often can be approached using the techniques of a standard radical nephrectomy. The kidney, great vessels, and renal pedicle are exposed, mobilized, and controlled through a thoracoabdominal incision. After ligation of the renal artery the tumor thrombus is gently palpated to ensure that no further extension into the vena cava is present. Preoperative angioinfarction permits ligation of the venous system before taking the artery, which can occasionally be helpful.
The kidney is dissected free, leaving the renal vein as the only attachment point (see Figs. 54-98 and 54-99 on the Expert Consult website![]() ). Two Satinsky or carotid clamps are placed in close apposition to each other at the junction of the renal vein and IVC, sparing any lumbar tributaries (see Fig. 54–100 on the Expert Consult website
). Two Satinsky or carotid clamps are placed in close apposition to each other at the junction of the renal vein and IVC, sparing any lumbar tributaries (see Fig. 54–100 on the Expert Consult website![]() ). With a scalpel, the IVC is transected proximal to both clamps, leaving them on the IVC. The proximal clamp is removed, which leaves a margin of caval wall for reconstruction with the distal clamp still in place. The caval defect is closed with running 4-0 polypropylene suture, with caution taken to minimize reduction of the IVC diameter (see Fig. 54–101 on the Expert Consult website
). With a scalpel, the IVC is transected proximal to both clamps, leaving them on the IVC. The proximal clamp is removed, which leaves a margin of caval wall for reconstruction with the distal clamp still in place. The caval defect is closed with running 4-0 polypropylene suture, with caution taken to minimize reduction of the IVC diameter (see Fig. 54–101 on the Expert Consult website![]() ). Surgicel may be placed over the repair for additional hemostasis.
). Surgicel may be placed over the repair for additional hemostasis.
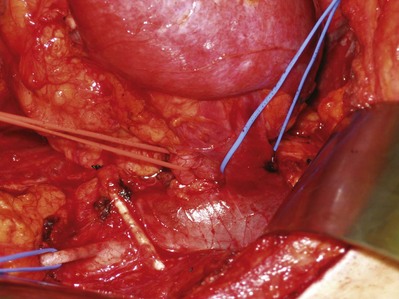
Figure 54–98 Vessel loops around left renal artery (red), left renal vein (blue) and gonadal (blue) vessel. Also pictured is a DeWeese clamp placed many years ago for pulmonary embolism prophylaxis in patient with deep venous thrombosis of the lower extremity.
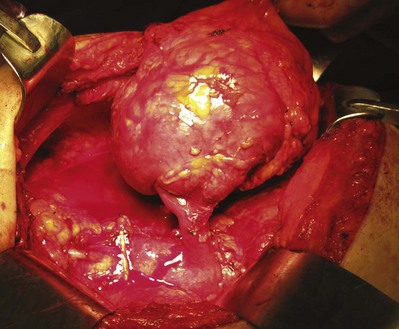
Figure 54–99 Left kidney with renal vein tumor thrombus mobilized before placement of a Satinsky clamp at renal vein/vena cava confluence. Renal artery and ureter have already been divided.
Perirenal or Infrahepatic Tumor Thrombus (Level II)
Transesophageal echocardiography is performed before making the incision to evaluate the cephalad extent of tumor thrombus (Fig. 54–102). Radical nephrectomy with removal of a perirenal or infrahepatic IVC thrombus is best approached through a chevron incision. For very large tumors involving the upper pole of the kidney a thoracoabdominal incision may be used instead. Most often these lesions are resected without CPB.
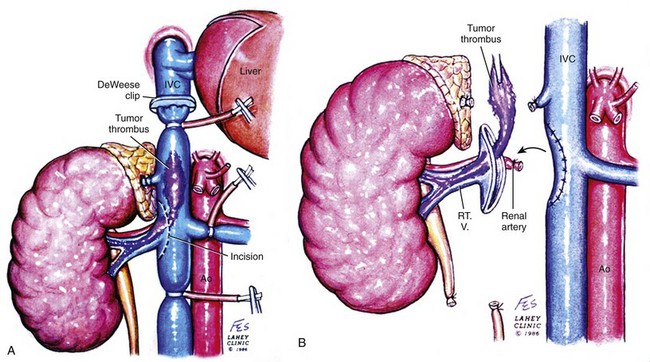
Figure 54–102 A and B, Technique for removing infrahepatic tumor thrombus with assistance of Rummel tourniquets avoiding cardiopulmonary bypass. Ao, aorta; IVC, inferior vena cava; RT. V, right vein.
(© The Lahey Clinic.)
The colon is reflected medially, and a Kocher maneuver is employed to mobilize the duodenum. A Bookwalter retractor is used for exposure. The caudate lobe of the liver can be exposed and a sweetheart retractor placed under it, taking caution to identify and safeguard the porta hepatis. Control of the IVC is obtained with limited manipulation to prevent embolization of tumor thrombus. Rummel tourniquets are placed above and below the thrombus and around the contralateral renal vein (see Figs. 54-103 and 54-104 on the Expert Consult website![]() ). The renal artery, associated lumbar and minor hepatic veins, as well as the contralateral renal vein are isolated and circumferentially dissected. One can sacrifice perforating veins to the caudate lobe of the liver, which permits the IVC to be separated from the caudate lobe and exposes several more centimeters of IVC (Novick, 2007).
). The renal artery, associated lumbar and minor hepatic veins, as well as the contralateral renal vein are isolated and circumferentially dissected. One can sacrifice perforating veins to the caudate lobe of the liver, which permits the IVC to be separated from the caudate lobe and exposes several more centimeters of IVC (Novick, 2007).
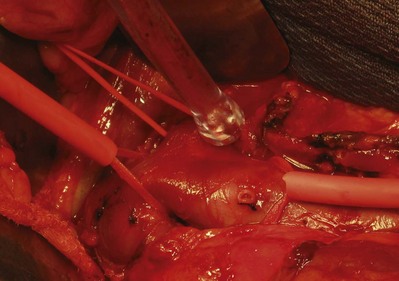
Figure 54–103 The inferior vena cava is exposed for a left renal mass with renal vein thrombus at the IVC confluence. The left renal vein and inferior vena cava are isolated with Rummel tourniquets. An oblique Rummel tourniquet is placed below the right renal vein allowing venous return. The gonadal vein has been divided (silk ties).
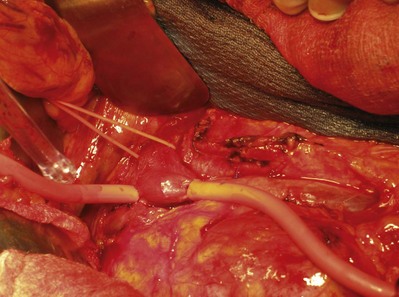
Figure 54–104 The inferior and oblique Rummel tourniquets are tightened to minimize blood loss during the thrombectomy.
The renal artery and the ureter are ligated and divided. The specimen is mobilized outside the Gerota fascia, leaving the renal vein as the sole attachment. Preoperative renal angioinfarction may produce an inflammatory response that precludes safe early mobilization of the renal artery. In this instance the authors defer ligation of the renal artery until the tumor thrombectomy has been completed.
Venous occlusion is obtained with the previously placed Rummel tourniquets. An alternative is to use a Satinsky clamp on the IVC and a bulldog vascular clamp on the contralateral renal vein (Novick, 2007). A longitudinal anterior cavotomy is made, and the thrombus is freed from the caval wall to the level of the renal vein ostium using a spatula and gentle manipulation. In most cases the thrombus is not attached to the wall of the cava. The IVC is gently flushed with heparinized saline and evaluated for residual fragments. The infrarenal clamp is released transiently to purge the system of debris and limit the risk of embolus. Alternatively the suprarenal clamp can be released while applying positive pulmonary pressure to flush the cava free of fragments. The cavotomy is closed with a continuous 4-0 polypropylene suture (Figs. 54-105 and 54-106 on the Expert Consult website![]() ). Radical nephrectomy is carried out after closure of the vena cava has been completed.
). Radical nephrectomy is carried out after closure of the vena cava has been completed.
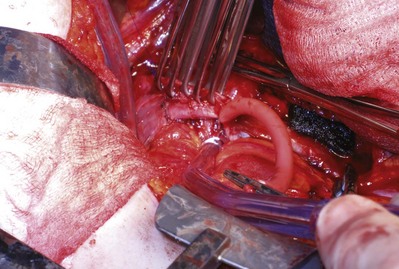
Figure 54–105 After removal of the left renal vein and tumor thrombus the caval defect is addressed with Allis clamps and oversewn with 4-0 polypropylene.
Whereas the senior author prefers to incise the anterior wall of the cava, an alternative is to open the anterior surface of the renal vein over the thrombus and extend the venotomy posteriorly with scissors (Fig. 54–107). With gentle downward traction the tumor thrombus is extracted from the cava through the venotomy, which is repaired as described previously.
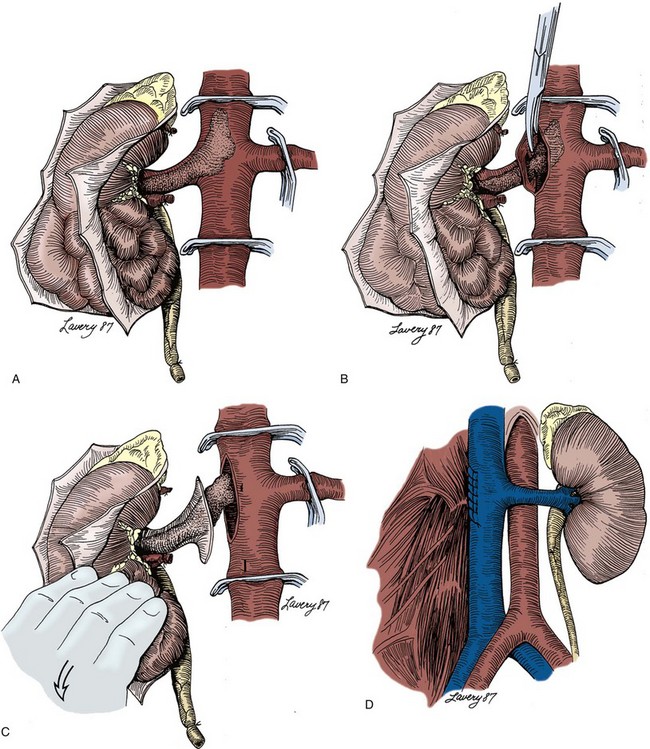
Figure 54–107 A to D, Technique of radical nephrectomy and venal caval tumor thrombectomy with infrahepatic tumor thrombus. The anterior surface of the renal vein is incised over the tumor thrombus, and the incision is continued posteriorly with scissors, passing just beneath the thrombus.
(Reproduced with permission from Novick AC, Streem SB, Pontes E, editors. Stewart’s operative urology. 2nd ed. Baltimore: Williams & Wilkins; 1989.)
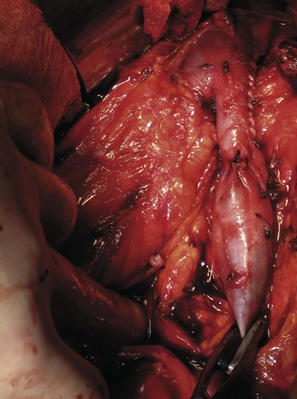
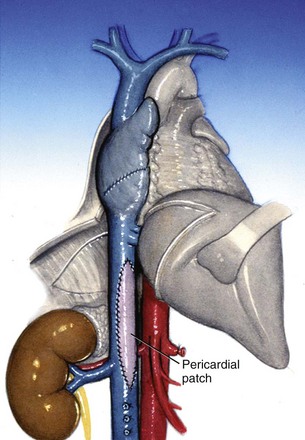
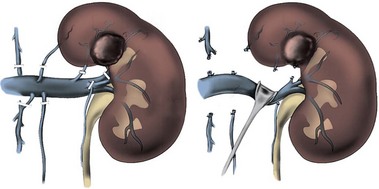
Occasionally the tumor thrombus invades the wall of the IVC necessitating caval wall resection. The lumen can be safely narrowed by half and closed primarily. To maintain the lumen in larger resections the cava can be reconstructed with PTFE or a pericardial graft in cases in which the chest is opened (Sarkar et al, 1998) (Fig. 54–108). When necessary, a segment of IVC can be excised without reconstruction. Resection of the infrarenal vena cava is usually safe and relies on the extensive venous collateral network that develops in the setting of tumor thrombus, particularly through the lumbar drainage system. In right radical nephrectomy with tumor thrombectomy the suprarenal vena cava can be resected, but only if the left renal vein has been ligated distal to its venous tributaries (i.e., gonadal, lumbar, and adrenal veins). These veins then provide venous drainage from the left kidney. Given the lack of venous tributaries on the right side the suprarenal vena cava should not be resected for a left-sided tumor unless one provides alternative venous drainage for the right kidney with autotransplantation or a saphenous vein graft to the splenic, portal, or inferior mesenteric vein.
Bland tumor thrombus completely occluding the cava can be managed with staple interruption to limit the risk of pulmonary embolism. Resection or ligation of the IVC should be prefaced by efforts to preserve the integrity of the lumbar drainage system.
Intrahepatic and Suprahepatic Tumor Thrombus (Levels III and IV)
The literature supports extirpative surgery for these tumors provided patients have a decent Eastern Cooperative Oncology Group (ECOG) performance status score and that there is not extensive locoregional invasion or metastatic disease (Staehler and Brkovic, 2000; Ficarra et al, 2001; Parekh et al, 2005; Blute et al, 2007; Klatte et al, 2007; Terakawa et al, 2007). Radical nephrectomy with excision of an intrahepatic or suprahepatic IVC thrombus is a challenging procedure requiring special techniques including CPB owing to the difficulty of obtaining vascular control above the tumor thrombus. Using traditional or minimally invasive bypass, which was pioneered at the Lahey Clinic, safe resection with excellent oncologic outcome is possible (Cummings et al, 1979; Foster et al, 1988; Skinner et al, 1989; Novick et al, 1990b; Burt, 1991; Fitzgerald et al, 1998; Ciancio et al, 2002; Wotkowicz et al, 2006).
Conventional Circulatory Bypass
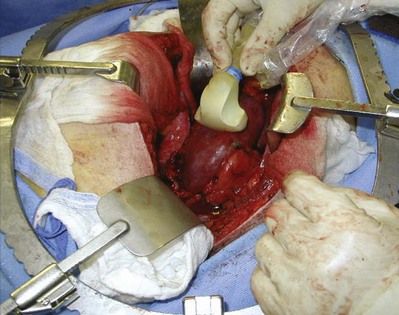
Transesophageal echocardiography is performed after induction of anesthesia to assess the distal extent of the thrombus. In general, the authors recommend CPB with deep hypothermic circulatory arrest for a thrombus within 2 cm of the hepatic veins and cephalad (Shahian et al, 1990). After a chevron incision is made the abdomen is evaluated for evidence of metastatic disease. Once respectability has been confirmed a median sternotomy is made (Fig. 54–109). The duodenum is kocherized. A Langenbeck maneuver, in which the right triangular and coronary ligaments of the liver are divided, is performed. This permits the right lobe of the liver to be rotated medially and cephalad, which exposes the retrohepatic IVC up to the diaphragm (Figs. 54-110 to 54-112).
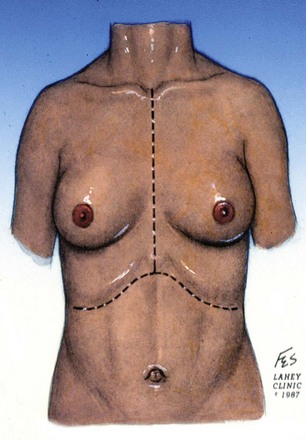
Figure 54–109 Traditional median sternotomy and chevron incisions for cardiopulmonary bypass and circulatory arrest.
(© The Lahey Clinic.)
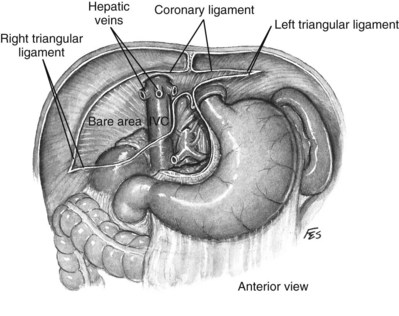
Figure 54–110 Relationship of liver and triangular and coronary ligaments to the vena cava. IVC, inferior vena cava.
(© The Lahey Clinic.)
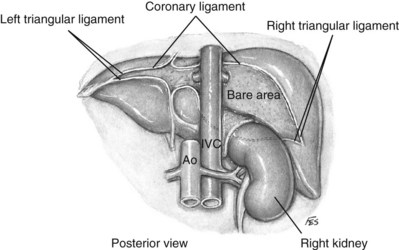
Figure 54–111 Posterior view of relationship of liver and coronary ligaments to the vena cava. Ao, aorta; IVC, inferior vena cava.
(© The Lahey Clinic.)
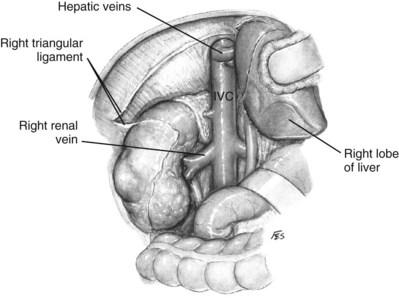
Figure 54–112 Access to vena cava by the Langenbeck maneuver. IVC, inferior vena cava.
(© The Lahey Clinic.)
The kidney is completely mobilized outside the Gerota fascia, with minimal traction to prevent embolism of the tumor thrombus. The gonadal vein and ureter are taken with Hem-o-Lok clips or 0 silk ties. The superior pole and adrenal are mobilized in a routine fashion along with the associated phrenic vessels. The last portion of dissection is the hilum. As stated earlier, isolating the renal artery from the posterior approach is recommended especially for left-sided tumors, given the risk of injury to the superior mesenteric artery. The main renal artery should be palpated in patients who had preoperative angioinfarction. If a pulse is present, the artery should be treated as unoccluded. The renal artery is taken with two large Hem-o-Lok clips and 0 silk suture ligatures, leaving the renal vein as the sole attachment (see Fig. 54–113 on the Expert Consult website![]() ). Few cases proceed in as straightforward a manner as described, because inflammation may make anatomic planes challenging to discern and the venous collateral vessels may be daunting.
). Few cases proceed in as straightforward a manner as described, because inflammation may make anatomic planes challenging to discern and the venous collateral vessels may be daunting.
The infrarenal vena cava and contralateral renal vein are exposed. The dissection is extended inferiorly to the bifurcation of the common iliac veins when bland or tumor thrombus extends there. Meticulous hemostasis is obtained before heparinization and initiation of CPB.
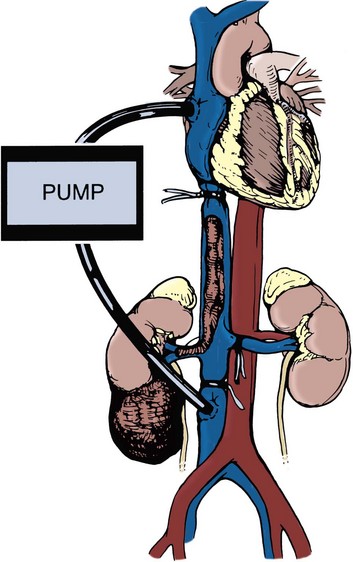
The patient is heparinized. The aortic arch and superior vena cava are cannulated. CPB is initiated (Fig. 54–114). A right atriotomy provides distal control of the tumor thrombus, which minimizes the risk of embolization. When the heart fibrillates, the aorta is clamped and crystalloid cardioplegic solution is infused. Inflow blood temperature is reduced as low as 10° C, resulting in deep hypothermia. The head and abdomen are packed in ice. After 15 to 30 minutes, a core temperature of 18° C to 20° C is achieved. Flow through the perfusion machine is stopped. Approximately 95% of the blood volume is drained into the pump, leaving no flow to any organ. Hypothermic circulatory arrest can be maintained for at least 40 minutes without incurring a cerebral ischemic event (Svensson et al, 1993). This interval can be extended by maintenance of “trickle” blood flow at a rate of 5 to 10 mL/kg/min (Mault et al, 1993) or by adjunctive retrograde cerebral perfusion (Pagano et al, 1995).
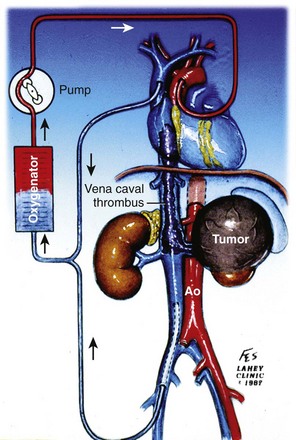
Figure 54–114 Traditional median sternotomy approach with cannulation of the aortic arch, superior vena cava, and right femoral vein for cardiopulmonary bypass. Ao, aorta.
(© The Lahey Clinic.)
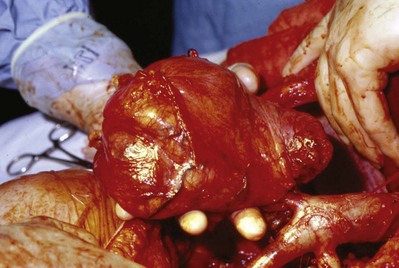
An anterior cavotomy is made using Potts forward and reverse scissors. The cavotomy should circumscribe the ostium of the renal vein and extend to the minor hepatic veins above the caudate lobe of the liver. The field will be nearly bloodless. For tumor extending into the right atrium the atrium is also opened (Fig. 54–115). The thrombus is extracted with the patient in Trendelenburg position and using positive-pressure respiration. If possible, the tumor thrombus is removed intact along with the kidney, although the thrombus may need to be removed in pieces if it is friable or adherent to the caval wall (Fig. 54–116). When necessary, a Fogarty catheter is passed from the atrium into the hepatic veins to retrieve portions of tumor thrombus. The authors ensure that tumor thrombus removal is complete by performing venacavoscopy with a flexible cystoscope. The cavotomy is closed with a double-armed 4-0 polypropylene running suture. The atriotomy is closed. Radical nephrectomy is carried out while rewarming to 37° C and coming off bypass. The patient is decannulated. Protamine is administered to reverse the heparin. Any leaking areas on the IVC are repaired with interrupted 4-0 polypropylene sutures. The abdomen is drained, and chest tubes are placed. Fresh frozen plasma, platelets, and desmopressin are used if needed to treat coagulopathy.
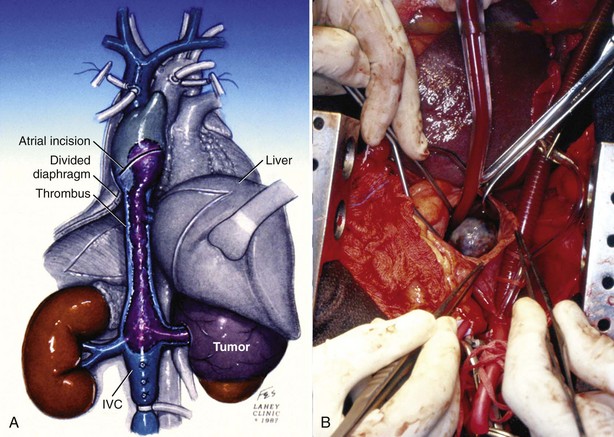
Figure 54–115 A and B, Tumor thrombus extension into right atrium removed using traditional median sternotomy approach and an atriotomy for retrieval of thrombus. IVC, inferior vena cava.
(A, © The Lahey Clinic. B, From Wotkowicz C, Libertino JA, Sorcini A, Mourtzinos A. Management of renal cell carcinoma with vena cava and atrial thrombus: minimal access vs. median sternotomy with circulatory arrest. BJU Int 2006;98:289–97.)
Minimally Invasive Cardiopulmonary Bypass
Since 1998, the authors have used a minimally invasive approach to CPB originally developed for aortic valve replacement (Fig. 54–117; see Figs. 54-118 to 54-126 on the Expert Consult website![]() ) (Cosgrove and Sabik, 1996; Fitzgerald et al, 1998). Unlike the traditional approach the kidney is not mobilized as extensively before heparinization and bypass. A chevron incision is used. The colon is mobilized, and the IVC is exposed using the Kocher and Langenbeck maneuvers.
) (Cosgrove and Sabik, 1996; Fitzgerald et al, 1998). Unlike the traditional approach the kidney is not mobilized as extensively before heparinization and bypass. A chevron incision is used. The colon is mobilized, and the IVC is exposed using the Kocher and Langenbeck maneuvers.
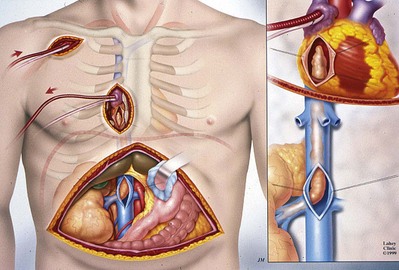
Figure 54–117 Minimal access technique utilizing a synthetic graft to the subclavian artery for arterial return and a two-stage venous cannula into the right atrium for venous return.
(© The Lahey Clinic.)
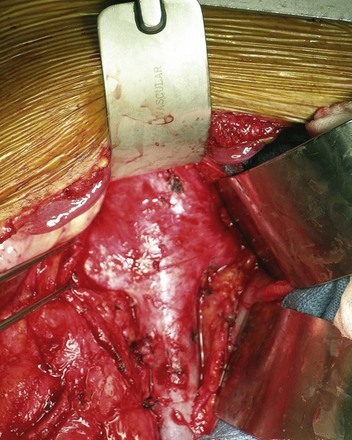
Figure 54–118 Following a chevron the anterior inferior vena cava and both renal veins are exposed without mobilizing the kidney and limiting trauma to the vena cava.
(From Wotkowicz C, Libertino JA, Sorcini A, Mourtzinos A. Management of renal cell carcinoma with vena cava and atrial thrombus: minimal access vs. median sternotomy with circulatory arrest. BJU Int 2006;98:289–97.)
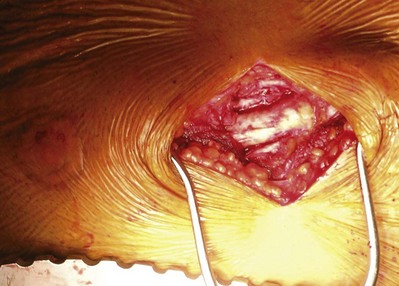
Figure 54–119 Right parasternal incision along the border of the third and fourth rib.
(From Wotkowicz C, Libertino JA, Sorcini A, Mourtzinos A. Management of renal cell carcinoma with vena cava and atrial thrombus: minimal access vs. median sternotomy with circulatory arrest. BJU Int 2006;98:289–97.)
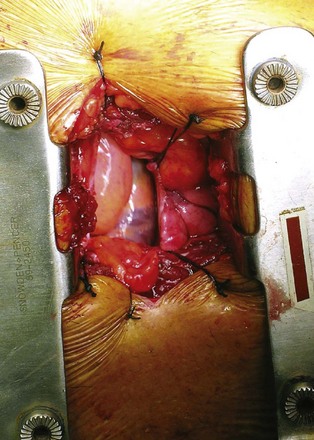
Figure 54–120 Small pericardial window is created, and a Finochietto retractor is placed, exposing the atrium prior to cannula placement.
(From Wotkowicz C, Libertino JA, Sorcini A, Mourtzinos A. Management of renal cell carcinoma with vena cava and atrial thrombus: minimal access vs. median sternotomy with circulatory arrest. BJU Int 2006;98:289–97.)
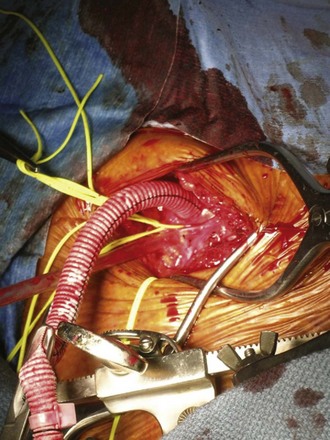
Figure 54–121 Right subclavian 8-mm synthetic graft is sewn into place for arterial return.
(From Wotkowicz C, Libertino JA, Sorcini A, Mourtzinos A. Management of renal cell carcinoma with vena cava and atrial thrombus: minimal access vs. median sternotomy with circulatory arrest. BJU Int 2006;98:289–97.)
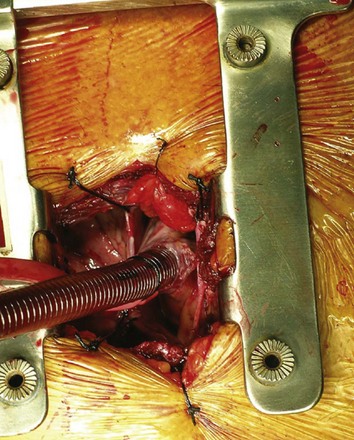
Figure 54–122 Right subclavian 8-mm synthetic graft is sewn into place for arterial return.
(From Wotkowicz C, Libertino JA, Sorcini A, Mourtzinos A. Management of renal cell carcinoma with vena cava and atrial thrombus: minimal access vs. median sternotomy with circulatory arrest. BJU Int 2006;98:289–97.)
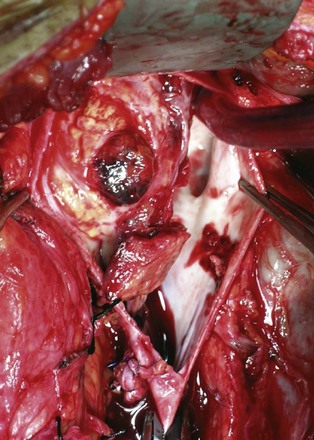
Figure 54–123 Anterior cavotomy with resection of tumor thrombus. Contralateral left renal vein ostium is free of disease.
(From Wotkowicz C, Libertino JA, Sorcini A, Mourtzinos A. Management of renal cell carcinoma with vena cava and atrial thrombus: minimal access vs. median sternotomy with circulatory arrest. BJU Int 2006;98:289–97.)
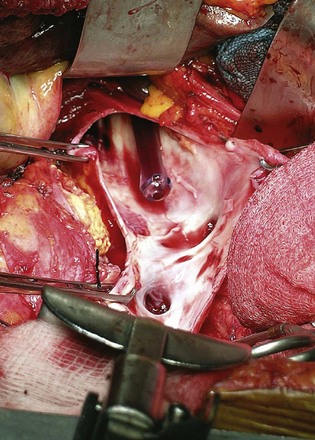
Figure 54–124 After removal of tumor thrombus Yankauer suction is passed from the atrium prior to the “shoe-shine” maneuver.
(From Wotkowicz C, Libertino JA, Sorcini A, Mourtzinos A. Management of renal cell carcinoma with vena cava and atrial thrombus: minimal access vs. median sternotomy with circulatory arrest. BJU Int 2006;98:289–97.)
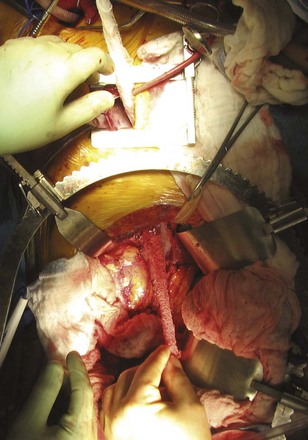
Figure 54–125 “Shoe-shine” maneuver performed with a gauze pad to remove small residual fragments in the vena cava.
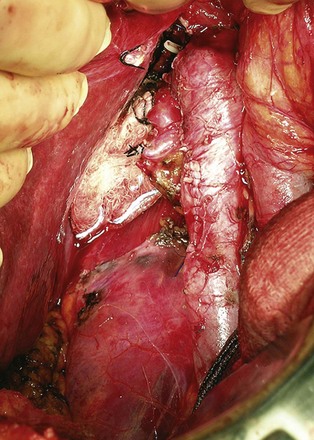
Figure 54–126 Inferior vena cava after tumor thrombectomy, right radical nephrectomy, and para-aortic lymph node dissection.
(From Wotkowicz C, Libertino JA, Sorcini A, Mourtzinos A. Management of renal cell carcinoma with vena cava and atrial thrombus: minimal access vs. median sternotomy with circulatory arrest. BJU Int 2006;98:289–97.)
The right subclavian artery is mobilized via an infraclavicular incision followed by a 2-inch right parasternal incision at the heads of the third and fourth ribs with resection of the rib cartilage and ligation of the right internal thoracic artery. Periosteum, muscles, and pleura are preserved for closure. The right pericardium is opened, exposing the superior vena cava and right atrium, and a Finochietto retractor is placed. Systemic heparinization is initiated as an 8-mm synthetic graft is sewn to the right subclavian artery for arterial return, while a two-stage venous cannula is positioned into the right atrium for venous outflow. CPB and circulatory arrest are initiated, and the patient is cooled. A right atriotomy is made to gain distal control of the thrombus and prevent passage into the pulmonary circulation. Tumor thrombus is removed from the cava, and a gauze pad is passed through the cava to the cardiothoracic team for removal of any loose debris (see Fig. 54–125 on the Expert Consult website![]() ). The remainder of the operation proceeds as described previously for a traditional median sternotomy.
). The remainder of the operation proceeds as described previously for a traditional median sternotomy.
Alternatives to Cardiopulmonary Bypass
Venovenous Bypass
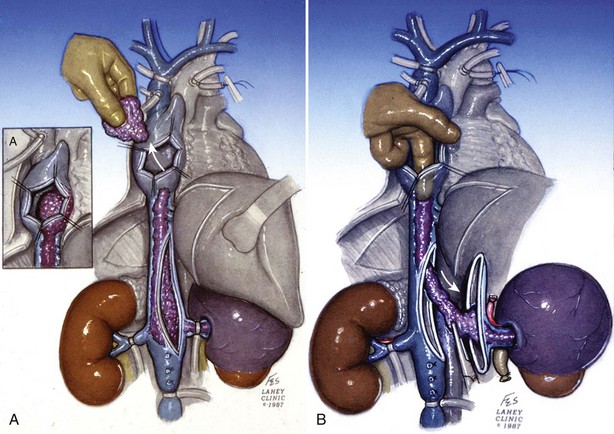
Patients with nonadherent supradiaphragmatic tumor thrombi that do not extend into the right atrium can be managed with venovenous bypass using a caval-atrial shunt (Foster et al, 1988; Burt, 1991). The intrapericardial vena cava, infrarenal vena cava, and contralateral renal vein are temporarily occluded. Cannulae are inserted into the right atrium or axillary vein and the femoral veins or infrarenal vena cava. Bypass is initiated using a primed pump to maintain flow to the right side of the heart (Fig. 54–127). This avoids the decline in cardiac output due to the reduced preload associated with occlusion of the intrapericardial and infrarenal vena cava. The cavotomy is made and the thrombectomy performed. Bleeding from the hepatic venous system, which can be brisk, is managed with a Pringle maneuver, cross clamping the hepatic veins, or both. Bleeding from the lumbar veins, which originates from the uncontrolled azygous and hemiazygous systems, is difficult to control and is a major downside of this technique. The cava is repaired and the nephrectomy performed as previously described. This technique helps to avoid some of the complexities of CPB with deep hypothermic circulatory arrest, albeit at the risk of hepatic venous bleeding.
Extensive Liver Mobilization and Other Techniques
Extensive liver mobilization, employed for decades at the Lahey Clinic, is another approach to avoid CPB (see Fig. 54–128 on the Expert Consult website![]() ) (Libertino, 1987; Ciancio et al, 2007). The liver is mobilized to the left after the division of the ligamentum teres, falciform ligament, triangular ligament, and superior coronary ligament of the liver. This provides excellent access to the retrohepatic portion of the vena cava, leaving on the major hepatic veins in continuity after a Pringle maneuver is performed. The surgeon palpates and milks the tumor thrombus caudally to the level of the hepatic veins, and a standard cavotomy is performed.
) (Libertino, 1987; Ciancio et al, 2007). The liver is mobilized to the left after the division of the ligamentum teres, falciform ligament, triangular ligament, and superior coronary ligament of the liver. This provides excellent access to the retrohepatic portion of the vena cava, leaving on the major hepatic veins in continuity after a Pringle maneuver is performed. The surgeon palpates and milks the tumor thrombus caudally to the level of the hepatic veins, and a standard cavotomy is performed.
D’Ancona and associates (2005) describe the removal of a suprarenal tumor thrombus using extracorporeal circulation and deep hypothermic arrest without violation of the thoracic cavity. After exposure of the vena cava the liver is retracted inferiorly to expose the pericardium at the level of the diaphragm. A pericardial window permits cardiac defibrillation. Extracorporeal circulation is established via the right femoral artery, right femoral vein, and subclavian vein. Core hypothermia to 20° C is achieved before circulatory arrest is begun. Infrahepatic and suprahepatic longitudinal incisions provide access for thrombus removal.
Chowdhury and associates (2007) discuss another alternative for intra-atrial tumor thrombus utilizing CPB, mild hypothermia, and an intermittent cross clamping of the supraceliac aorta to avoid the risks associated with circulatory arrest.
Outcomes
The incidence of complications in treating tumors of this magnitude has decreased with refinements in surgical techniques, intraoperative monitoring, and postoperative critical care (Boorjian et al, 2007; Wotkowicz and Libertino, 2007). Prolonged pulmonary support and renal replacement therapy may be necessary. The authors’ minimal access technique, used in more than 30 patients, avoids median sternotomy with equivalent oncology outcomes. Eliminating the median sternotomy reduces the dose of postoperative analgesics, avoids reoperative sternotomy in patients with a prior coronary artery bypass, and results in a shorter operative time, shorter hospital stays, and decreased transfusion and mechanical ventilatory support (Wotkowicz et al, 2006).
Key Points: Venous Extension and Tumor Thrombectomy
Cytoreductive Nephrectomy and Metastasectomy
Palliative nephrectomy is often performed for tumors that are very large with significant extrarenal disease in an effort to decrease symptoms. In contrast, cytoreductive nephrectomy is performed with the goal of improving cancer outcome. A combined analysis of two trials (Southwest Oncology Group S8949 and EORTC 30947) elucidated the benefits of cytoreductive nephrectomy and revealed a survival benefit for interferon-alfa combined with nephrectomy versus interferon alone (Lara et al, 2009; Mickisch et al, 2001). Additional studies show potential for cytoreductive nephrectomy in combination with tyrosine kinase inhibitors (sunitinib [Sutent] and sorafenib) (Thomas et al, 2009). Analyses indicate that excellent preoperative performance status (ECOG 0 to 1 or Karnofsky performance status <80) and lung-only metastatic disease are good prognostic variables (Tables 54-13 and 54-14). Proponents of cytoreductive therapy cite anecdotal cases of spontaneous metastasis resolution, referred to as the Lazarus effect. A reduction in angiogenesis and growth factors after nephrectomy may enhance the efficacy of systemic immunotherapy compared with immunotherapy without prior cytoreductive nephrectomy. Unresectable lung lesions can serve as markers for chemotherapy efficacy and may be resected if their volume decreases significantly. In the authors’ experience the presence of disease outside the chest and poor performance status are contraindications for surgical intervention, unless for pure palliative purposes such as with intractable edema, ascites, cardiac dysfunction, associated pain, or hematuria.
Table 54–13 Eastern Cooperative Oncology Group (ECOG) Status
| GRADE | ECOG |
|---|---|
| 0 | Fully active, able to carry on all pre-disease performance without restriction |
| 1 | Restricted in physically strenuous activity but ambulatory and able to carry out work of a light or sedentary nature (e.g., light housework and office work) |
| 2 | Ambulatory and capable of all self care but unable to carry out any work activities Up and about more than 50% of waking hours |
| 3 | Capable of only limited self care Confined to bed or chair more than 50% of waking hours |
| 4 | Completely disabled Cannot perform any self care Totally confined to bed or chair |
| 5 | Dead |
From Oken MM, Creech RH, Tormey DC, et al. Toxicity and response criteria of the Eastern Cooperative Oncology Group. Am J Clin Oncol 1982;5(6):649–55.
Table 54–14 Karnofsky Performance Score
| SCORE | DESCRIPTION |
|---|---|
| 100 | Normal no complaints; no evidence of disease |
| 90 | Able to carry on normal activity; minor signs or symptoms of disease |
| 80 | Normal activity with effort; some signs or symptoms of disease |
| 70 | Cares for self; unable to carry on normal activity or to do active work |
| 60 | Requires occasional assistance but is able to care for most personal needs |
| 50 | Requires considerable assistance and frequent medical care |
| 40 | Disabled; requires special care and assistance |
| 30 | Severely disabled; hospital admission is indicated although death not imminent |
| 20 | Very sick; hospital admission necessary; active supportive treatment necessary |
| 10 | Moribund, fatal processes progressing rapidly |
| 0 | Dead |
| Ranges | |
| 80-100 | Able to carry on normal activity and to work. No special care needed |
| 50-70 | Unable to work. Able to live at home and care for most personal needs |
| 0-40 | Unable to care for self. Requires institutional or hospital care |
From Schag CC, Heinrich RL, Ganz PA. Karnofsky performance status revisited: reliability, validity, and guidelines. J Clin Oncol 1984;2(3):187–93.
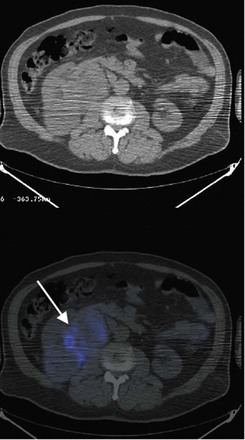
First described by Barney and Churchill in 1939, the resection of pulmonary metastasis remains an effective treatment for select patients. Utilizing a thoracoabdominal approach for the radical nephrectomy and metastasectomy, the authors resect anterior lower lobe lesions using Doyle clamps and endovascular staplers. Favorable prognostic factors for isolated pulmonary metastasis include preoperative performance status, completeness of resection, number of lesions (less than six), limited lymph node involvement, and disease-free interval. Properly selected patients may have 5-year survival rates exceeding 50% (Hofmann et al, 2005). Metastasectomy in nonpulmonary sites such as the liver and brain are controversial and should be considered investigational. Preoperative imaging using biologic markers, such as monoclonal antibodies to carbonic anhydrase IX, may better help to identify occult metastatic disease and better stratify these patients with complex disease (Divgi et al, 2007) (Fig. 54–129).
Partial Nephrectomy for Malignant Disease
From its 19th century roots, nephron-sparing surgery has emerged as a sophisticated and continually ground-breaking component of contemporary open renal surgery. Partial nephrectomy is a demanding endeavor that involves complete resection of a tumor with a negative margin, with maximal preservation of functioning renal parenchyma. Although radical nephrectomy is oncologically sound, technically straight forward, and boasts a favorable perioperative complication rate, the more challenging nephron-sparing approach is indicated in a growing number of patients.
Indications
Traditionally, the treatment of choice for renal cortical tumors was radical nephrectomy. Partial nephrectomy was reserved for patients in whom radical nephrectomy would result in dependence on dialysis, such as those patients with a solitary kidney, bilateral or familial renal tumors, or chronic renal insufficiency. In recent years, indications for nephron-sparing surgery have broadened. Partial nephrectomy has supplanted radical nephrectomy as the treatment of choice for T1 renal tumors when feasible, even in the absence of identifiable renal insufficiency or threat (Russo et al, 2002; Russo and Huang, 2008). Partial nephrectomy provides the same long-term oncologic results as radical nephrectomy for tumors of 7 cm or less (Fergany et al, 2000; Russo et al, 2002; Dash et al, 2006; Pahernik et al, 2008).
For tumors other than T1, partial nephrectomy may be indicated in patients with a contralateral kidney that is threatened by stone disease, chronic infection, renovascular disease, vesicoureteral reflux, or systemic diseases that threaten renal function such as hypertension or diabetes mellitus (Licht et al, 1994). In recent years, the adverse consequences of chronic renal insufficiency, even in the absence of dialysis, have become clear. Urologists are increasingly aware of the importance of preservation of renal function. The literature does not support partial nephrectomy for patients with a tumor that is T2 or higher with a normal contralateral kidney.
Contraindications to open partial nephrectomy include lymph node metastasis because the prognosis for these patients is poor. Biopsy of enlarged or suspicious-looking lymph nodes should be done before the renal resection is initiated. For renal cortical tumors in a solitary kidney with venous extension, partial nephrectomy with tumor thrombectomy may be feasible, but this remains investigational (Tollefson et al, 2005).
Nephron-Sparing Surgery and the Adverse Effects of Renal Insufficiency
In the past, partial nephrectomy was not thought to be necessary in patients with a normal contralateral kidney, owing to a mistaken belief that patients with a normal contralateral kidney would suffer no ill effect from radical nephrectomy. This idea dates back to the 19th century. In 1869, Simon performed the first nephrectomy in a human for a benign condition (Simon, 1870). He did so only after first removing a kidney in 30 dogs, thereby demonstrating that survival with a solitary kidney is possible. Nevertheless, 140 years later, it is apparent that the deficit in glomerular filtration due to radical nephrectomy can have far-reaching negative consequences.
The majority of patients with renal cortical tumors have unrecognized comorbid medicorenal disease. In a pathologic review of radical nephrectomy specimens, only 10% of specimens had normal renal parenchyma adjacent to the tumor (Bijol et al, 2006). More than 60% of patients had pathologic evidence of medicorenal disease and nearly 30% had demonstrable renal arteriosclerosis. In a study from Memorial Sloan-Kettering Cancer Center, 26% of patients with renal cortical tumors who underwent surgery had a preoperative estimated GFR less than 60 mL/min/1.73 m2 despite having two apparently normal kidneys and a serum creatinine concentration in the normal range (Huang et al, 2006).
In comparison to partial nephrectomy, radical nephrectomy is associated with a higher risk of postoperative renal insufficiency (Lau et al, 2000; McKiernan et al, 2002). In a retrospective study from Memorial Sloan-Kettering Cancer Center regarding patients with a renal cortical tumor 4 cm or smaller, normal preoperative serum creatinine concentration, and two intact kidneys, radical nephrectomy was an independent risk factor for new onset of chronic kidney disease. The 3-year probability of freedom from new onset of GFR less than 60 mL/min/1.73 m2 was 80% after partial nephrectomy but only 35% after radical nephrectomy (P < .01) (Huang et al, 2006).
There is a dose-response association between chronic kidney disease and hospitalizations, cardiovascular events, and death (Go et al, 2004). Recent efforts have demonstrated that radical nephrectomy may be associated with increased cardiovascular events and decreased survival. A study combining Surveillance, Epidemiology and End Results (SEER) cancer registry data with Medicare claims demonstrated that radical nephrectomy, in comparison to partial nephrectomy, is associated with a higher risk of overall mortality (HR 1.38, P < .01) and a 1.4-fold higher number of cardiovascular events (P < .05) (Huang et al, 2009). For patients younger than 65 years old with single unilateral renal cortical tumors measuring 4 cm or less with a normal contralateral kidney, data from the Mayo Clinic suggests that radical nephrectomy is associated with decreased overall survival in comparison to partial nephrectomy (response rate [RR] = 2.16; P = .02) (Thompson et al, 2008).
Partial Nephrectomy is Underutilized
Despite the growing number of small renal masses and ample evidence in favor of the oncologic efficacy of partial nephrectomy for T1 tumors and the potential adverse impact of radical nephrectomy, a large percentage of tumors amenable to partial nephrectomy are still removed with radical surgery (Nuttall et al, 2005; Hollenbeck et al, 2006). In a study from a tertiary care center, more than 70% of renal cortical tumors smaller than 4 cm were treated by laparoscopic radical nephrectomy in 2002 (Scherr et al, 2003). In a nationwide sample from 1988 to 2002, partial nephrectomy comprised fewer than 8% of operations for renal tumors (Hollenbeck et al, 2006). In another sample, only 20% of renal cortical tumors measuring 2 to 4 cm were treated with partial nephrectomy (Miller et al, 2006). The desire to use a minimally invasive modality may be contributing to excessive use of radical surgery (Scherr et al, 2003). This imbalance needs to be rectified.
Open Partial Nephrectomy: An Increasingly Challenging Endeavor
Because the majority of renal cortical lesions are now discovered incidentally on cross-sectional imaging, tumors of 4 cm or less now make up the principal share of newly diagnosed masses (Nguyen et al, 2006). Despite this general trend, in the senior author’s experience the lesions referred for open partial nephrectomy have become more complex. Lesions managed with open partial nephrectomy at the Lahey Clinic are now larger, more often hilar, more central, deeper, and involve the collecting system to a greater degree than in the past. This observation is consistent with experience at other tertiary care centers. When compared with the era before laparoscopy, tumors undergoing open partial nephrectomy in the laparoscopic era at the Cleveland Clinic are more often in a solitary kidney (40.0% vs. 15.6%), centrally located (55.6% vs. 37.3%), and higher grade (Fuhrman 3 or 4) (43.1% vs. 27.8%, each P < .01) (Weight et al, 2008).
The increasing complexity of open partial nephrectomy is attributable, at least in part, to widespread adoption of minimally invasive techniques (e.g., laparoscopic radical and partial nephrectomy, cryoablation, and radiofrequency ablation) (Weight et al, 2008). In a multi-institutional comparison of 1800 open and laparoscopic partial nephrectomies, patients undergoing open partial nephrectomy had larger tumors, which were more centrally located and more often malignant. Patients undergoing open surgery were more often symptomatic at presentation and more frequently had impaired renal function, worse performance status, and solitary kidney (Gill et al, 2007).
New surgical techniques, which will be integrated into the technical descriptions in this chapter, have been adopted so the nephron-sparing approach can be applied to these increasingly complex masses with limited impact on renal function. Contemporary open partial nephrectomy is a cutting-edge operation that is rooted in powerful evidence favoring both oncologic efficacy and renal preservation.
Preoperative Evaluation and Preparation
Patients undergoing partial nephrectomy should have an extensive preoperative workup as described earlier in the chapter, including cardiac, pulmonary, and renal evaluation. Autologous blood donation should be offered to patients because perioperative blood transfusion may be necessary. A preoperative urine culture is mandatory. It is the practice of the senior author to treat bacteriuria, even if asymptomatic, before surgery. Urine should be sterile before partial nephrectomy given the risk of entry into the collecting system. Patients should be intravascularly fluid replete before surgery. Although it is no longer the authors’ practice to admit patients for volume resuscitation on the day before partial nephrectomy, it is the belief of the senior author that patients benefit from vigorous intravenous hydration on the morning of surgery.
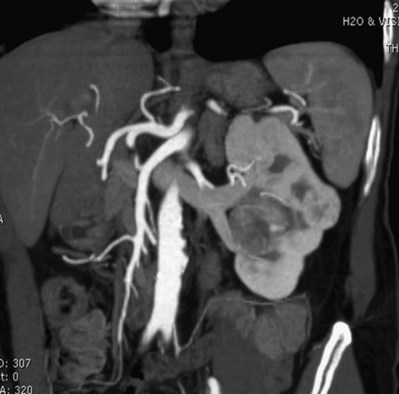
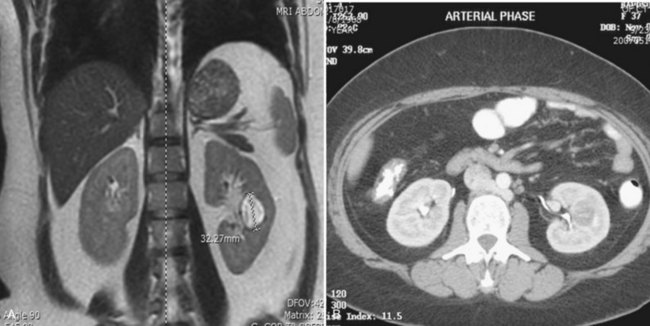
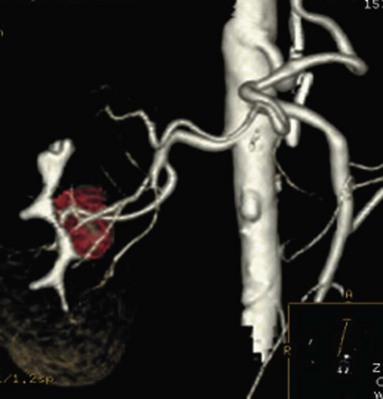
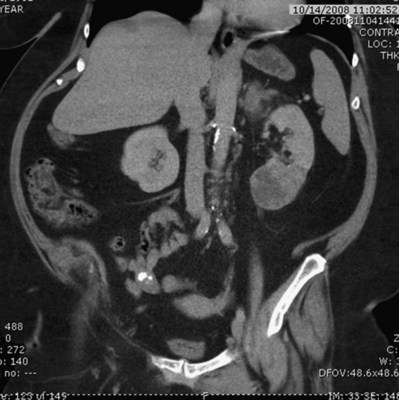
Before partial nephrectomy, evaluation of patients with renal cortical tumors should include imaging to identify locally extensive or metastatic disease. Preoperative imaging should also delineate relevant regional anatomy, intrarenal anatomy, and vasculature to minimize blood loss and damage to adjacent renal parenchyma. Invasive renal arteriography has largely been replaced by noninvasive 3D CT angiography, which offers excellent anatomic detail. 3D CT integrates information from arteriography, venography, excretory urography, and conventional two-dimensional CT into a single imaging modality (Fig. 54–130). In addition to tumor staging, 3D reconstructions of contrast-enhanced CT angiograms, MR images, or MR venograms are useful for planning site and extent of planned resection and to determine proximity to the renal hilum, intrarenal vasculature, and collecting system (Fig. 54–131). 3D CT accurately demonstrates involvement of the collecting system by tumor (Derweesh et al, 2003).
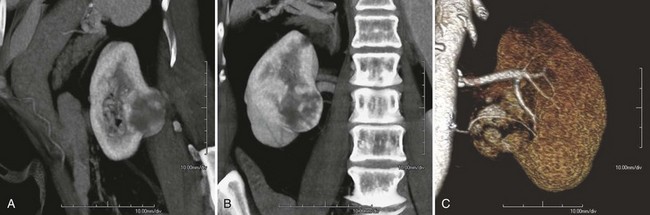
Figure 54–131 A, Contrast-enhanced CT scan demonstrating right renal mass. B, Three-dimensional reconstruction demonstrates a large intrarenal component to the mass. C, Arterial reconstruction shows lower pole renal artery in close proximity to the renal mass.
In general, the authors prefer not to administer an intravenous contrast load within 1 week of surgery. Appropriate precautions including oral N-acetylcysteine, vigorous hydration with normal saline, and use of low osmolality contrast agents should be taken in patients at risk of contrast nephropathy (Barrett and Parfrey, 2006). After administration of a contrast medium, renal function is reevaluated before partial nephrectomy to exclude contrast nephropathy.
Bilateral Tumors
Bilateral tumors represent a special circumstance that is considered a firm indication for nephron-sparing surgery. The simpler partial nephrectomy is performed first. A MAG3 renogram is used to document preservation of renal function in the postoperative kidney. Once the patient has fully recovered from the initial surgery, the more complicated partial or radical nephrectomy is performed on the contralateral kidney.
Fundamentals of Partial Nephrectomy
Open partial nephrectomy is a technically demanding procedure. Although individual techniques may vary, the fundamental steps begin with the surgical approach, vascular control, and mobilization of the kidney. After complete excision of the tumor with maximal preservation of normal parenchyma and minimization of ischemic injury, renorrhaphy is completed. Renorrhaphy includes hemostasis, watertight closure of the collecting system, and repair of the renal defect. The basic techniques are outlined here, followed by a more detailed discussion of the various types of partial nephrectomy.
Approach
Good functional exposure through a properly selected incision is essential to the success of the procedure. For partial nephrectomy the incision should be chosen to optimize exposure of the tumor, kidney, and renal pedicle. The authors most often employ an extrapleural, extraperitoneal supra-11th rib incision. The level of incision is determined by the position of the kidney in relation to the ribs as seen on preoperative radiographic studies and by the location and size of the tumor. The thoracoabdominal approach is useful in large or upper pole tumors. Partial nephrectomy can also be accomplished through an anterior subcostal incision, although this typically affords inferior exposure. The rib-resecting approach is not employed. In rare circumstances, extracorporeal partial nephrectomy with autotransplantation is necessary, in which case a supracostal 11th rib incision is used to harvest the kidney and a semilunar Gibson incision is used to autotransplant the kidney.
Prompt Vascular Control and Mobilization of the Kidney
With few exceptions, the first priority in partial nephrectomy is identification, dissection, and nonocclusive control of the renal artery and vein with vessel loops (see Fig. 54–132 on the Expert Consult website![]() ). This protects the renal pedicle in the ensuing dissection and, in emergencies, permits the surgeon to rapidly occlude the renal artery to stop life-threatening hemorrhage. One may be able to safely resect small, peripheral renal tumors without first obtaining control of the main renal artery.
). This protects the renal pedicle in the ensuing dissection and, in emergencies, permits the surgeon to rapidly occlude the renal artery to stop life-threatening hemorrhage. One may be able to safely resect small, peripheral renal tumors without first obtaining control of the main renal artery.
The ureter is identified and encircled with a vessel loop. Dissection of the ureter is carried superiorly to the renal pelvis, with special care to avoid devascularization. The uninvolved portion of the kidney is mobilized within the Gerota fascia, while the perirenal fat overlying the tumor is left undisturbed. The kidney must be adequately mobilized to permit resection of a renal tumor with negative margins and minimal injury to adjacent vasculature, parenchyma, and collecting system.
Fully Perfused Partial Nephrectomy: A New Model of Nephron Sparing
Although clamping of the renal artery and vein may reduce the technical difficulty of the procedure by providing a nearly bloodless field, this maneuver leads to renal ischemia that may adversely affect renal function (Thompson et al, 2007). At the Lahey Clinic, the senior author prefers to perform fully perfused partial nephrectomy.
Duration of renal ischemia is the strongest modifiable risk factor for renal insufficiency after partial nephrectomy (Lane et al, 2008). In patients with a solitary kidney, clamping is associated with greater risk of urine leak, renal failure, and temporary dialysis (Thompson et al, 2007). The senior author considers clamping of the pedicle to be antithetical to the philosophy of nephron-sparing surgery. The authors perform the majority of open partial nephrectomies with the kidney fully perfused. In a retrospective review of 112 patients with a solitary kidney or preoperative renal dysfunction (serum creatinine concentration >1.5 mg/dL) who underwent partial nephrectomy by the senior author, only 18% required arterial clamping (Smith et al, 2008). The fully perfused partial nephrectomy technique may result in excellent sparing of renal function. The majority of patients had either no change in serum creatinine concentration (37%) or a less than a 50% change (46%). Others have advocated a similar approach when feasible (Russo and Huang, 2008).
In the nonclamping method, a vessel loop is placed around the renal artery after it has been mobilized but the vessels are never clamped or compressed. Although this nonclamping technique has frequently been coupled with enucleation, a method of excision characterized by minimal blood loss and trauma to surrounding parenchyma, the senior author has expanded nonclamping to more difficult tumors requiring complex resection. In a retrospective review of 273 patients at the Lahey Clinic who underwent fully perfused partial nephrectomy, 37% of tumors were 4 cm or smaller and 33% were hilar or central. There were multiple tumors in 17% of patients, and 26% of patients had a solitary kidney (Kurteva et al, 2008). The median estimated blood loss was 685 mL (mean 1100 mL). With fully perfused partial nephrectomy, the authors believe the risk of durable deficit in renal function is lowered, as demonstrated in patients with solitary kidneys or preoperative renal dysfunction, albeit at the cost of an increased risk of blood loss and a higher level of technical difficulty (Kurteva et al, 2008).
On rare occasions, some form of transient compression may be required to aid in tumor excision, pelvicalyceal suture repair, and hemostasis. Even in cases of brisk bleeding, the authors prefer not to clamp the main renal artery with instruments or tourniquets, given the threat of ischemia and the small but real risk of intimal damage, thrombosis, and loss of the kidney. It is often sufficient to have the assistant gently compress the renal parenchyma adjacent to the cut surface or use a Kitner to compress the bleeding vessel while the surgeon places shallow 4-0 monocryl figure-of-eight sutures into the transected vessels.
Traditional Partial Nephrectomy with Vascular Clamping
Although the authors have largely abandoned vascular clamping, partial nephrectomy has traditionally been performed with temporary occlusion of the main renal artery or a segmental artery supplying the tumor. Proposed benefits of clamping include limitation of intraoperative bleeding, improved visualization, and reduction in renal tissue turgor, which may improve access to intrarenal structures (Novick, 2007).
When clamping of the renal artery is required, in-situ renal hypothermia is used to protect against ischemic renal injury (Novick, 2007). Surface cooling of the kidney with ice slush allows up to 3 hours of safe ischemia without permanent renal injury. The entire kidney should be covered with ice slush for 10 to 15 minutes immediately after the renal artery is occluded and before the partial nephrectomy is begun (Fig. 54–133). This amount of time is needed to obtain core renal cooling to a temperature (approximately 20° C) that optimizes in-situ renal preservation. During excision of the tumor, invariably, large portions of the kidney are no longer covered with ice slush; and in the absence of adequate prior renal cooling, rapid rewarming and ischemic renal injury can occur. Cooling by perfusion of the kidney with a cold solution instilled through the renal artery is not recommended because of the theoretical risk of tumor dissemination. Mannitol is given intravenously 5 to 10 minutes before temporary renal arterial occlusion. Systemic or regional anticoagulation to prevent intrarenal vascular thrombosis is not necessary (Novick, 2007).
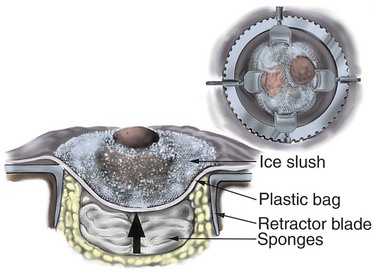
Figure 54–133 Elevation of the mobilized kidney to skin level is demonstrated by placement of sponges under the plastic bag containing the kidney and ice slush.
Even with the artery clamped it is preferable to leave the renal vein patent to reduce ischemia and permit identification of venous bleeding. When large veins are transected, such as in large centrally located tumors, some find it necessary to clamp the renal vein (Novick, 2007).
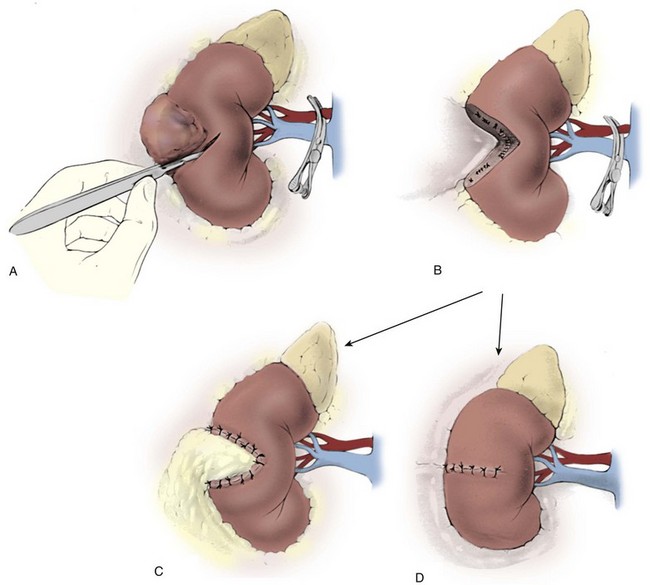
Tumor Excision
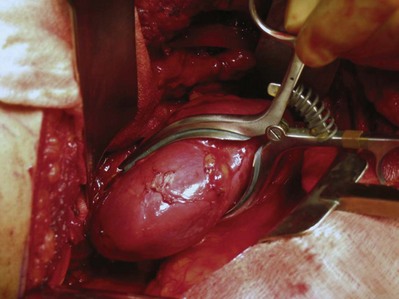
The goal of partial nephrectomy is complete excision of the tumor with negative margins and maximal preservation of benign adjacent parenchyma. Various techniques can be employed to remove the tumor, including enucleation, polar segmental nephrectomy, transverse resection, wedge resection, and extracorporeal partial nephrectomy with renal autotransplantation (see later for a detailed discussion). When performing partial nephrectomy for a suspected malignancy, the overlying renal capsule is not preserved for renorrhaphy but is excised along with the tumor.
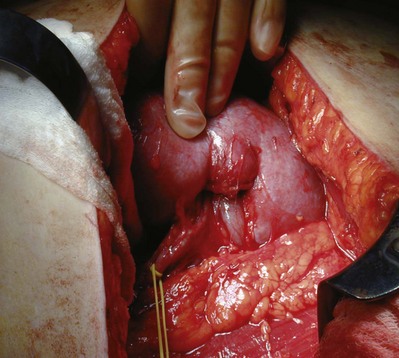
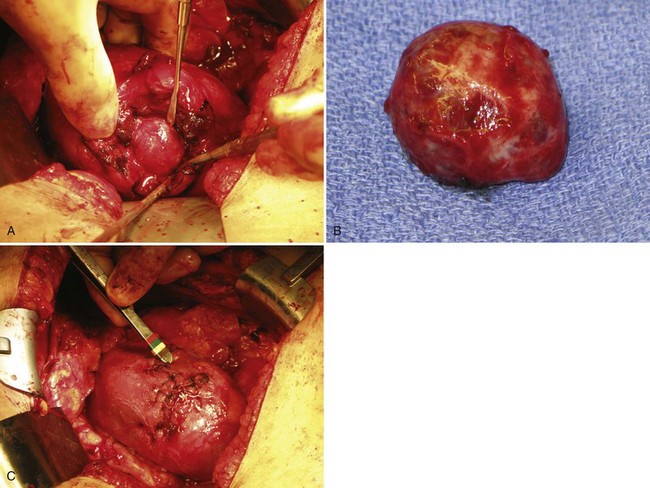
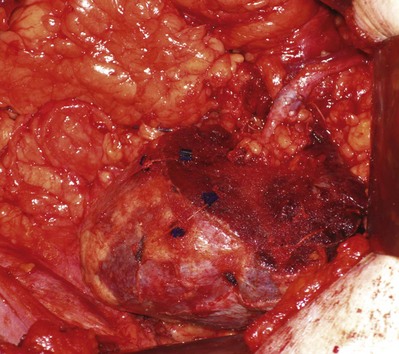
Complete resection must be ensured in the operating room. Local recurrence may be due to incomplete resection of the primary lesion or synchronous lesions that were unrecognized at the time of surgery. Intraoperative ultrasonography is used to delineate tumor from normal parenchyma and may help localize intrarenal lesions that are not visible or palpable from the surface of the kidney (Assimos et al, 1991; Campbell et al, 1996) (see Fig. 54–134 on the Expert Consult website![]() ; see also Fig. 54–135). Intraoperative ultrasonography is especially useful in the patient with von Hippel-Lindau disease with multiple lesions. The tumor should be inked and cut by the pathologist to be certain that the capsule has not been violated. Intraoperative biopsies should be obtained from the tumor bed if the capsule has been violated. Frozen sections should be examined, and additional tissue should be excised if the frozen section reveals persistent tumor. The width of a tumor-free resection margin has no biologic or prognostic significance (Castilla et al, 2002).
; see also Fig. 54–135). Intraoperative ultrasonography is especially useful in the patient with von Hippel-Lindau disease with multiple lesions. The tumor should be inked and cut by the pathologist to be certain that the capsule has not been violated. Intraoperative biopsies should be obtained from the tumor bed if the capsule has been violated. Frozen sections should be examined, and additional tissue should be excised if the frozen section reveals persistent tumor. The width of a tumor-free resection margin has no biologic or prognostic significance (Castilla et al, 2002).
Renorrhaphy: Hemostasis, Closure of the Collecting System, and Repair of the Renal Defect
After excision of the tumor the transected blood vessels on the renal surface are secured with figure-of-eight 4-0 monocryl sutures. The argon beam coagulator can be used to achieve hemostasis on the cut renal surface. At the Lahey Clinic, use of Floseal hemostatic matrix (Baxter International, Deerfield, IL) has obviated use of the argon beam coagulator, which can disrupt the figure-of-eight sutures or devitalize the collecting system (Fig. 54–136).
Figure 54–136 Floseal is used to provide hemostasis after removal of the tumor and to identify areas of collecting system leaks after intravenous administration of indigo carmine.
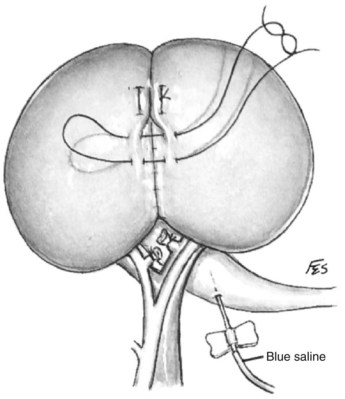
Collecting system defects can be identified by administration of methylene blue or indigo carmine, either intravascularly or directly into the renal pelvis, to dye the urine (see Fig. 54–137 on the Expert Consult website![]() ). The collecting system is closed with interrupted or running 4-0 monocryl suture. A ureteral stent can be placed in a retrograde fashion at the start of the procedure if significant repair of the intrarenal collecting system is anticipated. Alternatively, before repair of the collecting system, a wire can be placed antegrade into the bladder through the opening in the collecting system followed by a double-J stent (see Fig. 54–138 on the Expert Consult website
). The collecting system is closed with interrupted or running 4-0 monocryl suture. A ureteral stent can be placed in a retrograde fashion at the start of the procedure if significant repair of the intrarenal collecting system is anticipated. Alternatively, before repair of the collecting system, a wire can be placed antegrade into the bladder through the opening in the collecting system followed by a double-J stent (see Fig. 54–138 on the Expert Consult website![]() ).
).
Figure 54–137 A and B, Floseal application before and after administration of indigo carmine. Small collecting system openings are addressed with 2-0 polyglactin figure-of-eight sutures.
Figure 54–138 A and B, Antegrade placement of JJ ureteral stent before closure of a larger opening in the collecting system during resection of a hilar mass. Before placing the stent, a 5-Fr ureteral catheter is placed over the guide wire and aspirated to confirm wire position. The bladder is filled with methylene blue via a three-way Foley catheter.
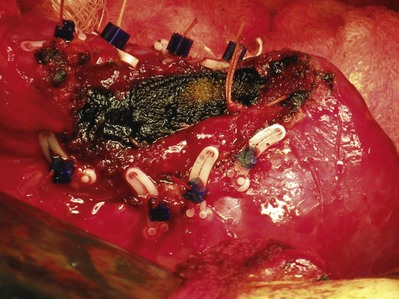
After closure of the renal vasculature and collecting system, a hemostatic bolster composed of rolled Surgicel is placed at the base of the defect. Floseal hemostatic matrix is applied to the resection site around the bolster. The cortical edges are approximated over the bolster with simple, pledgeted interrupted 2-0 polyglactin sutures (Fig. 54–139; see Fig. 54–140 on the Expert Consult website![]() ). The suture line should be tension free, and the kidney should be reconstructed in such a way that the renal pedicle is not kinked. If the renal artery was clamped, circulation is restored after the renorrhaphy is complete. Nephropexy to the posterior musculature with interrupted 2-0 polydioxanone may prevent postoperative migration or rotation of the remnant kidney (Fig. 54–141). A retroperitoneal drain is externalized through a counter incision and is left in place approximately 1 week, or longer if it is draining urine.
). The suture line should be tension free, and the kidney should be reconstructed in such a way that the renal pedicle is not kinked. If the renal artery was clamped, circulation is restored after the renorrhaphy is complete. Nephropexy to the posterior musculature with interrupted 2-0 polydioxanone may prevent postoperative migration or rotation of the remnant kidney (Fig. 54–141). A retroperitoneal drain is externalized through a counter incision and is left in place approximately 1 week, or longer if it is draining urine.
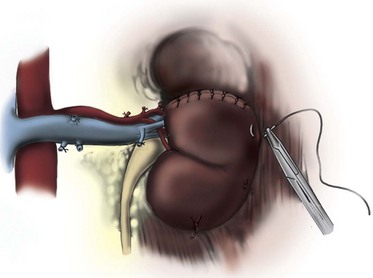
Figure 54–141 Nephropexy of the remnant kidney to the retroperitoneum is achieved with several interrupted sutures.
Recently, the senior author has utilized an alternative method of approximating the cortical edges that is adapted from the laparoscopic technique. Interrupted 2-0 polyglactin sutures are used to approximate the cortical margins, which are secured in a novel, knotless fashion (see Fig. 54–142 on the Expert Consult website![]() ). The suture is secured at the first cortical surface with a Weck ligating clip (Pilling Weck Canada, L.P., Markham, ON, Canada), followed by a Lapra-Ty clip (Ethicon Endosurgery, Cincinnati, OH). A Weck clip is then placed on the suture where it exits the other cortical surface. It is cinched snugly into position, causing the cortical margins to come together, and secured with a second Lapra-Ty clip. This may permit tighter closure of the cortical margins than the traditional repair, provide good closure of parenchyma over the repaired collecting system, and eliminate the need for a bolster.
). The suture is secured at the first cortical surface with a Weck ligating clip (Pilling Weck Canada, L.P., Markham, ON, Canada), followed by a Lapra-Ty clip (Ethicon Endosurgery, Cincinnati, OH). A Weck clip is then placed on the suture where it exits the other cortical surface. It is cinched snugly into position, causing the cortical margins to come together, and secured with a second Lapra-Ty clip. This may permit tighter closure of the cortical margins than the traditional repair, provide good closure of parenchyma over the repaired collecting system, and eliminate the need for a bolster.
Techniques of Tumor Excision
Enucleation
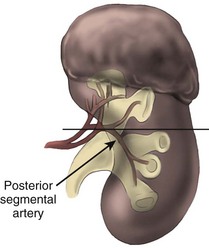
Pure enucleation involves removing a renal tumor with blunt dissection along the natural plane provided by the tumor pseudocapsule. Although this technique is convenient and is associated with minimal blood loss, several studies have demonstrated that tumor may extend beyond the pseudocapsule and might therefore be left behind after a pure enucleation. For instance, an analysis of 82 radical nephrectomy specimens for tumors measuring less than 4 cm demonstrated that nearly 20% of specimens had tumor beyond the pseudocapsule, at a mean of 0.5 mm from the capsule. No cancer was found to extend beyond 5 mm, and fewer than 3% extended beyond 4 mm (Li et al, 2003). Other studies have also suggested a higher risk of leaving cancer in the kidney when pure enucleation is performed (Rosenthal et al, 1984; Marshall et al, 1986; Blackley et al, 1988). In response, some authors have advocated using pure enucleation followed by ablation of the tumor base with argon beam or laser energy, such as with the neodymium:yttrium-aluminum-garnet (Nd-YAG) laser, to destroy remaining tumor cells (Kutikov et al, 2008).
Given the risk of incomplete resection with pure enucleation, it is the practice of the senior author to enucleate tumors along with a thin rim of grossly normal renal parenchyma. It is a rapid, precise technique with the benefit of limited blood loss and excellent preservation of normal parenchyma. The kidney is approached and mobilized as described previously, leaving the perirenal fat overlying the tumor intact. The limits of the tumor are defined by palpation and ultrasonography. An incision is made in the renal capsule around the base of the tumor, using the coagulation setting of the Bovie device (see Fig. 54–143 on the Expert Consult website![]() ). The Bovie device is inserted into the cut renal capsule and the tumor is circumferentially inscribed. A small tenotomy scissor is used to divide the renal parenchyma. A Penfield spatula is used to retract the normal parenchyma from the specimen. Small vessels in the cortex are palpated with the Frazer pediatric suction device by gently moving it along the cut surface of the parenchyma. These vessels are then grasped with a delicate Gerald forceps, coagulated, and divided with the tenotomy scissors (see Fig. 54–144 on the Expert Consult website
). The Bovie device is inserted into the cut renal capsule and the tumor is circumferentially inscribed. A small tenotomy scissor is used to divide the renal parenchyma. A Penfield spatula is used to retract the normal parenchyma from the specimen. Small vessels in the cortex are palpated with the Frazer pediatric suction device by gently moving it along the cut surface of the parenchyma. These vessels are then grasped with a delicate Gerald forceps, coagulated, and divided with the tenotomy scissors (see Fig. 54–144 on the Expert Consult website![]() ). Cautery should not be used deeper than the corticomedullary junction or near the collecting system because it will cause ischemic damage. Nonhealing of the collecting system due to cautery injury may result in a urinary fistula.
). Cautery should not be used deeper than the corticomedullary junction or near the collecting system because it will cause ischemic damage. Nonhealing of the collecting system due to cautery injury may result in a urinary fistula.
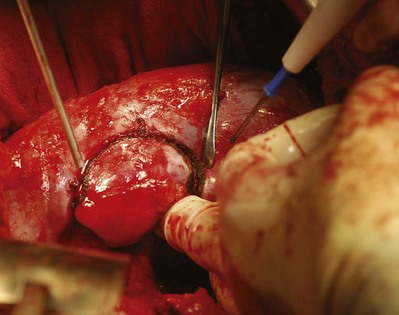
Figure 54–143 After ultrasonography the peripheral tumor margins are palpated and a 2- to 3-mm margin is marked using electrocautery.
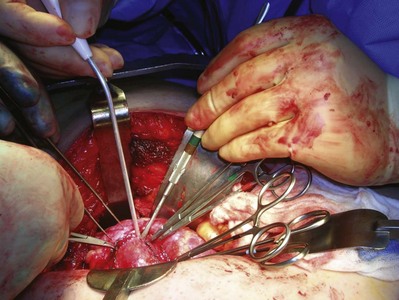
Figure 54–144 Using a spatula and fine forceps the tumor is enucleated, placing curved snaps and small right angles on vessels and collecting system components.
Closer to the base of the lesion the tumor is meticulously dissected with the surgeon’s thumb protecting the specimen. Larger intrarenal vessels are clamped with pediatric right-angle clamps, divided, and ligated (see Fig. 54–145 on the Expert Consult website![]() ). Frozen sections are obtained to be certain that the margins are negative. If there is any question about the margin, additional tissue at the margins is resected and reevaluated. The specimen is inked, and the integrity of the capsule is determined. Floseal is applied to the cut surface of the kidney. In the absence of renal artery clamping, indigo carmine can be given intravenously (see Fig. 54–146 on the Expert Consult website
). Frozen sections are obtained to be certain that the margins are negative. If there is any question about the margin, additional tissue at the margins is resected and reevaluated. The specimen is inked, and the integrity of the capsule is determined. Floseal is applied to the cut surface of the kidney. In the absence of renal artery clamping, indigo carmine can be given intravenously (see Fig. 54–146 on the Expert Consult website![]() ). An opening in the collecting system is identified by the appearance of a blue dot on the surface of the Floseal, which is then closed with 4-0 monocryl suture (see Fig. 54–138). If possible, the renal parenchyma should be closed over the opening in the collecting system. Otherwise, perinephric fat or omentum can be used to cover the renal parenchymal defect. The perinephric fat is approximated over the kidney. This is accomplished without clamping the renal artery or manual compression of the parenchyma. A Penrose drain is externalized through a separate stab incision.
). An opening in the collecting system is identified by the appearance of a blue dot on the surface of the Floseal, which is then closed with 4-0 monocryl suture (see Fig. 54–138). If possible, the renal parenchyma should be closed over the opening in the collecting system. Otherwise, perinephric fat or omentum can be used to cover the renal parenchymal defect. The perinephric fat is approximated over the kidney. This is accomplished without clamping the renal artery or manual compression of the parenchyma. A Penrose drain is externalized through a separate stab incision.
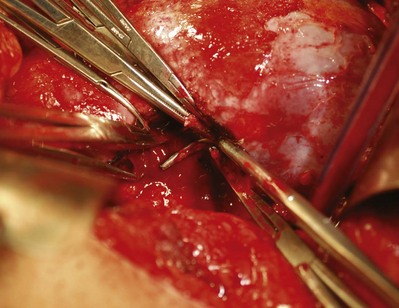
Figure 54–145 Vessels are tied off using 4-0 polyglactin suture. Larger vessels are ligated with 2-0 polyglactin suture.
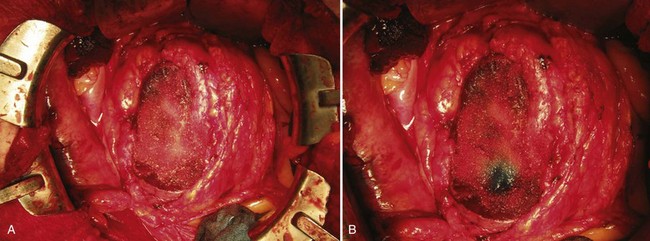
Figure 54–146 Floseal application before (A) and after (B) administration of indigo carmine. Small collecting system openings are addressed with 2-0 polyglactin figure-of-eight sutures.
Deep-seated tumors can be resected using a similar approach. After the tumor is located with ultrasound, a Y-shaped incision is made in the parenchyma over the tumor. The incision is opened, and the tumor is dissected meticulously from the kidney using the techniques described earlier. The incision is then closed with absorbable suture (see Fig. 54–147 on the Expert Consult website![]() ).
).
Polar Nephrectomy
Polar nephrectomy is useful for tumors confined to the upper or lower pole of the kidney. The kidney is approached and mobilized as previously explained. The authors typically use a vessel loop to obtain nonocclusive control of the segmental artery supplying the tumor and perform the polar nephrectomy with the kidney fully perfused, as described earlier. The limits of the resection are determined by the tumor rather than by the territory supplied by the segmental apical or basilar artery. This technique permits resection of a polar tumor with a thin margin of normal parenchyma and preservation of uninvolved polar parenchyma.
A more traditional approach is to isolate, ligate, and divide the segmental apical or basilar artery, leaving the remainder of the kidney perfused (Fig. 54–148). The corresponding venous branch is also divided. A line marking the ischemic segment will appear. If this ischemic line is not apparent, injection of methylene blue into the distal aspect of the ligated artery will delineate the involved renal segment (Fig. 54–149). The renal cortex is incised at the line of ischemia, which should be at least several millimeters away from the visible edge of the tumor. Renal parenchyma is divided with a small tenotomy scissor. A neurosurgical spatula is used to provide exposure. Whereas renal parenchyma is easily divided, vessels and collecting system structures are sturdier and elastic. With careful technique, this difference can aid in identification of vessels and the collecting system structures before they are divided. By placing gentle tension on the polar segment to be excised, the gap between specimen and remnant kidney is enlarged, which helps identify larger blood vessels before they are transected. The polar segment is removed. Renorrhaphy with hemostasis, closure of the collecting system, and repair of the renal defect proceeds as described earlier. Surgicel is placed at the base of the defect. If the cortical margins cannot be approximated to each other, a hemostatic bolster is employed.
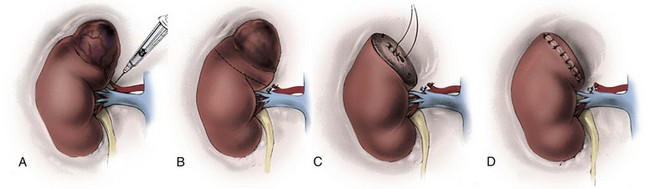
Figure 54–148 A to D, Technique of segmental (apical) polar nephrectomy with preliminary ligation of apical arterial and venous branches.
(From Novick AC. Partial nephrectomy for renal cell carcinoma. Urol Clin North Am 1987;14:419.)
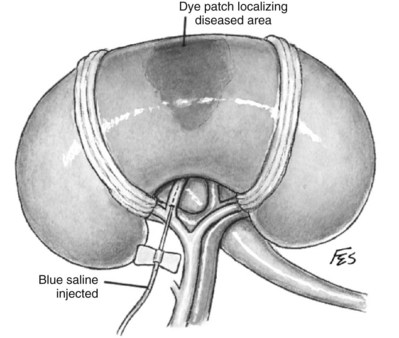
Figure 54–149 Identification of the small blood vessel, such as one that supplies part of the diseased parenchyma, is achieved by injecting a diluted solution of indigo carmine dye into the lumen with a scalpel vein needle and correlating the region stained with that seen angiographically.
(© The Lahey Clinic.)
Knowledge of the renovascular anatomy is essential for polar nephrectomy. If the segmental artery supplying the tumor has been identified with a preoperative CT angiogram, it can be isolated and controlled with a minimal amount of dissection. Alternatively, one is obligated to dissect the main renal artery and identify the various branches. The main renal artery often enters the upper third of the kidney and branches downward to supply the rest of the kidney. In upper pole partial nephrectomy it is therefore possible to damage the main renal artery during tumor excision or occlude a segmental artery supplying the lower portion of the kidney during renorrhaphy.
Heminephrectomy
Heminephrectomy, also referred to as a transverse resection, is used to remove large tumors that extensively involve the upper or lower portion of the kidney (see Figs. 54-150 and 54-152 on the Expert Consult website![]() ; see also Fig. 54–151). It is the authors’ preference to perform transverse resections without clamping to maintain perfusion to what will be the remnant parenchyma. The kidney is approached and mobilized as previously described. The main renal artery is isolated and surrounded with a vessel loop. With nonocclusive control of the renal artery the parenchyma is sharply divided, leaving several millimeters of grossly normal parenchyma around the tumor (Fig. 54–153). Transected vessels are controlled with shallow 4-0 monocryl figure-of-eight sutures. If needed, bleeding can be slowed transiently by gentle manual compression on the cut parenchyma of the kidney. For more severe bleeding the bleeding vessel can be compressed digitally or with a Kitner. Alternatively, Libertino clamps can be briefly applied until hemostasis has been obtained (Fig. 54–154). Renorrhaphy proceeds as previously described.
; see also Fig. 54–151). It is the authors’ preference to perform transverse resections without clamping to maintain perfusion to what will be the remnant parenchyma. The kidney is approached and mobilized as previously described. The main renal artery is isolated and surrounded with a vessel loop. With nonocclusive control of the renal artery the parenchyma is sharply divided, leaving several millimeters of grossly normal parenchyma around the tumor (Fig. 54–153). Transected vessels are controlled with shallow 4-0 monocryl figure-of-eight sutures. If needed, bleeding can be slowed transiently by gentle manual compression on the cut parenchyma of the kidney. For more severe bleeding the bleeding vessel can be compressed digitally or with a Kitner. Alternatively, Libertino clamps can be briefly applied until hemostasis has been obtained (Fig. 54–154). Renorrhaphy proceeds as previously described.
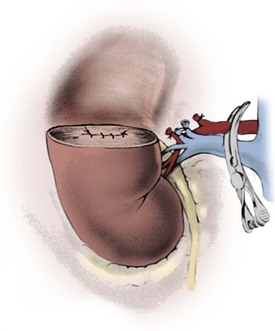
Figure 54–153 Technique of transverse resection for a tumor involving the upper half of the kidney.
(From Novick AC. Partial nephrectomy for renal cell carcinoma. Urol Clin North Am 1987;14:419.)
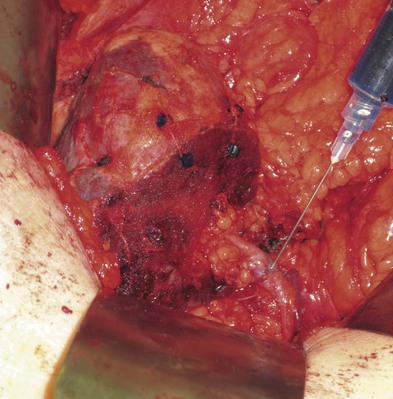
Figure 54–150 After left heminephrectomy, the renal pelvis is gently dilated with a 25-gauge needle using methylene blue or indigo carmine to evaluate for any collecting system leaks.
A more traditional approach is to perform a major transverse resection with arterial clamping and renal hypothermia. Before clamping the main renal artery, major branches of the renal artery and vein supplying the tumor-bearing portion of the kidney are identified in the renal hilum, ligated, and divided (Fig. 54–155). Renal artery clamping and tissue hypothermia are then obtained. Once adequate hypothermia has been obtained, the transverse resection proceeds as described previously.
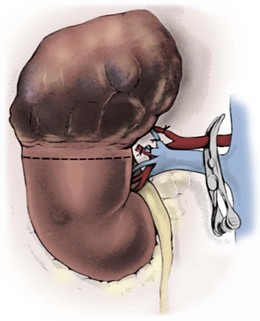
Figure 54–155 Technique of transverse resection for a tumor involving the upper half of the kidney.
(From Novick AC: Partial nephrectomy for renal cell carcinoma. Urol Clin North Am 1987;14:419.)
The relevant renovascular anatomy should be identified on a preoperative CT angiogram. The posterior segmental renal artery can be injured during transverse resection. The basilar renal artery, which typically arises from the anterior segmental renal artery, may occasionally arise from the posterior segmental artery. In that case, injury to the posterior segmental artery would devascularize a major portion of the remnant kidney during an upper pole transverse resection (Fig. 54–156).
Midsegmental Partial Nephrectomy
Midsegmental partial nephrectomy is a complicated and rarely employed nephron-sparing technique that is utilized for predominantly endophytic interpolar tumors. The kidney is approached and mobilized as described. The hilar structures are dissected free from each other and cleaned of adherent fat. The arterial branches are carefully identified. Vessel loops or loose loops of silk are placed around the primary and secondary arterial branches supplying the segment of parenchyma to be removed. The secondary arterial branches are injected with a dilute solution of indigo carmine or methylene blue, which demarcates the territory supplied by the vessels. If the demarcated area includes the tumor with an acceptable margin, the appropriate segmental arteries are ligated. The corresponding veins are doubly tied and divided. The caliceal infundibulum draining the targeted tissue is transected and ligated.
The main renal artery is clamped and surface hypothermia is obtained as previously described. A fingertip is placed under the midparenchyma to protect the hilar vessels. With a scalpel, a central section of the kidney that includes the tumor and a clean margin is excised from the convex through to the concave surfaces of the kidney (Fig. 54–157). Hemostasis is obtained with figure-of-eight sutures of 4-0 monocryl. The collecting system is closed. No calyces of the remaining lower or upper pole should be left open. The clamp is removed from the main renal artery. The cut surfaces and hilar branches are checked for significant bleeding. The two poles are approximated with horizontal mattress sutures (Fig. 54–158). During renorrhaphy, one must ensure that the blood vessels supplying the remnant kidney are free of significant angulation or kinking.
Wedge Resection
Wedge resection can be employed to excise large tumors that are predominantly located on the surface of the kidney and that are not limited to the upper or lower pole. A wedge shape that contains the tumor along with several millimeters of grossly normal renal parenchyma is removed. Wedge resection is traditionally performed with clamping of the renal artery and surface hypothermia, although it is the authors’ practice to avoid clamping whenever possible. The parenchyma is divided by a combination of sharp and blunt dissection, as previously described. The tumor is excised. The collecting system and vasculature are repaired as previously described.
Surgicel is placed at the base of the defect, which is then obliterated by closing the kidney on itself using simple interrupted 2-0 polyglactin sutures to approximate the cut cortical edges (Fig. 54–159). Alternatively, if closing the kidney would produce tension on the suture line or kinking of the blood supply, the base of the renal defect is covered with perirenal fat, which is sutured to the parenchymal margins with interrupted 4-0 monocryl sutures. The renal artery is then unclamped.
Central Tumors
Centrally located tumors present unique challenges for partial nephrectomy given their proximity to the hilum and the collecting system (see Fig. 54–160 on the Expert Consult website![]() ). The kidney is approached and mobilized as described. With central tumors extending into the renal hilum there may be no overlying perirenal fat to preserve. As mentioned previously, very deep-seated tumors can be resected without clamping by making a Y-shaped incision in the parenchyma overlying the tumor (see Figs. 54-147 and 54-161 on the Expert Consult website
). The kidney is approached and mobilized as described. With central tumors extending into the renal hilum there may be no overlying perirenal fat to preserve. As mentioned previously, very deep-seated tumors can be resected without clamping by making a Y-shaped incision in the parenchyma overlying the tumor (see Figs. 54-147 and 54-161 on the Expert Consult website![]() ). The tumor and a thin rim of normal parenchyma is meticulously dissected and extracted from the kidney using the techniques described in the enucleation section. The incision is closed with absorbable suture.
). The tumor and a thin rim of normal parenchyma is meticulously dissected and extracted from the kidney using the techniques described in the enucleation section. The incision is closed with absorbable suture.
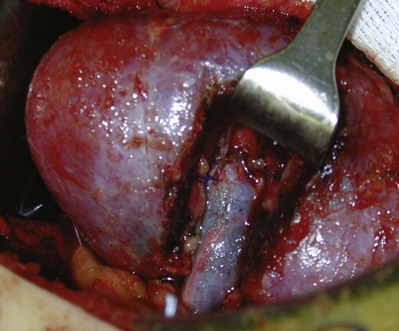
Figure 54–160 After meticulous resection, intrarenal arterial and collecting components can be identified.
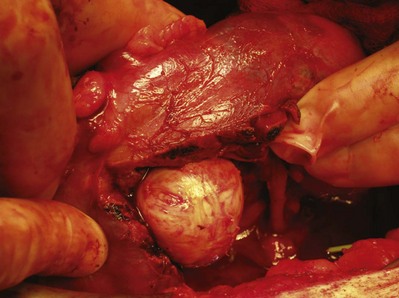
Figure 54–161 Nephrotomy to remove a larger intrarenal tumor with nonclamp, nonischemic partial nephrectomy.
A more conventional technique for tumors adjacent to the hilum is to perform partial nephrectomy with temporary occlusion of both the renal artery and vein using individual vascular clamps. Before clamping the main renal artery, segmental renal vessels adjacent to the tumor that supply the uninvolved portion of kidney are dissected away from the tumor and preserved. Because intrarenal venous branches freely anastomose, providing collateral venous drainage, one may divide small venous branches to access central tumors in the renal hilum without causing ischemia. In this way the main renal vein can be mobilized and retracted in a fairly extensive fashion to expose a central tumor (Fig. 54–162). Segmental arterial branches that directly supply the tumor are divided. The renal artery and vein are clamped, and the kidney is cooled. The tumor is excised using a combination of blunt and sharp dissection as described previously. These tumors should be removed with neighboring renal sinus fat and with a 3- to 4-mm margin of surrounding normal parenchyma where feasible. Hilar tumors may border the central collecting system; this may limit the margin of normal tissue. Renorrhaphy proceeds with the previously described methods. The vessels are unclamped. As mentioned previously, one must be vigilant to avoid injury to the posterior segmental renal artery
Extracorporeal Partial Nephrectomy and Autotransplantation
Extracorporeal partial nephrectomy with autotransplantation of the renal remnant can facilitate excision of large complex tumors involving the renal hilum (see Fig. 54–76). Proposed benefits of an extracorporeal approach include excellent exposure, a bloodless surgical field, maximal conservation of renal parenchyma, and greater protection of the kidney from prolonged ischemia. The technique, which is associated with longer operative time, need for renovascular and ureteral anastomoses, and risk of renal insufficiency, is infrequently employed and is discussed previously in this chapter.
Key Points: Partial Nephrectomy for Malignant Disease
Complications of Open Renal Surgery
Although much can be done to avoid surgical complications, some complications are due to the nature of the disease being treated and may be unavoidable. Meticulous attention to preoperative evaluation, surgical technique, and postoperative care will minimize avoidable morbidity and mortality. Complications are to be avoided at all costs, but they also should be regarded as an invaluable teaching tool for surgeons in training.
Patients with significant comorbidity may be at higher risk of complications, and some procedures are inherently perilous. Ideal surgical outcomes and safety should be relentlessly pursued, but the risk of complications should not deter surgeons from performing an indicated operation for a patient in need. Unlike vigilance and confidence, timidity and apprehension on the part of a surgeon do not benefit the patient.
Pulmonary and Thoracic Complications
Pulmonary complications of open renal surgery include atelectasis, pneumothorax, pneumonia, and injury to the lung parenchyma. As discussed previously, use of the flank positions can have deleterious effects on cardiopulmonary function. Postoperative atelectasis, which is cited as a common cause of postoperative fever, is best managed with early ambulation, vigilant fluid management, deep breathing, and incentive spirometry. Atelectasis and pneumonia may be prevented by postoperative pulmonary toilet, including incentive spirometry, bronchodilators when indicated, and postural drainage.
In addition, the pleura or parenchyma of the lung can be injured during open renal surgery, especially through a thoracoabdominal or flank incision. When the pleura is injured, the authors prefer to leave a 8-Fr “dart” chest drain one or two rib levels above the incision. On the contrary, after a nephrectomy with wedge resection of the lung for metastasectomy, a standard-caliber chest tube is used.
When an air leak is not anticipated, a well-described alternative to a chest tube is to evacuate the air from the pleural cavity with a red rubber catheter while the anesthesiologist hyperinflates the lungs. With hyperinflation and suction on the catheter, air and fluid in the hemithorax are forced out through the catheter, which is then removed as a preplaced purse-string suture is tied. Chest films are obtained in the postanesthesia care unit. A significant pneumothorax (15%-20%) or tension pneumothorax requires standard (28 to 32 Fr) chest tube placement. The less common complications of urinothorax or hemothorax are best managed with drainage of the chest and treatment of the root causes of urine leak and bleeding, respectively.
Injured lung parenchyma can be identified by gently pouring water over the parenchyma while the anesthesiologist hyperinflates the lungs. One can oversew the injured areas with 3-0 chromic sutures while the lungs are hyperinflated. Alternatively, a wedge resection of the injured lung using a gastrointestinal stapler can be performed. In the absence of definitive injury to the lung parenchyma, a clinically significant pneumothorax will rarely occur.
Diaphragmatic palsy is a hazard of any approach that puts the phrenic nerve at risk of injury. In addition, renal tumors may be adherent to and require partial resection of the diaphragm, especially when performing a thoracoabdominal incision. Small- to medium-sized defects can be repaired with figure-of-eight 2-0 silk sutures. A PTFE patch can be used to reconstruct larger defects along with chest tube drainage. Omental flaps interposed between the diaphragm and abdominal organs provide additional vasculature to promote healing.
Renal Complications
Renal insufficiency is a risk of any renal surgery, from ischemic injury from vascular clamping, excision of functional renal tissue, or renal unit loss. Hemodialysis, either temporary or permanent, may be necessary. As discussed earlier, duration of renal ischemia is the strongest modifiable risk factor for renal insufficiency after partial nephrectomy (Lane et al, 2008). In addition, clamping of the renal artery is associated with greater risk of urine leak, renal failure, and temporary dialysis (Thompson et al, 2007).
Ligation of the right renal vein during radical nephrectomy and IVC thrombectomy will result in failure of the right renal unit due to lack of venous collateral vessels. On the left, the renal insufficiency should be transient and resolve once adequate venous collateral vessels have developed (Clark, 1961; Pathak, 1971).
Urine leak is a risk of any renal surgery involving entrance into the colleting system, such as partial nephrectomy. This complication should resolve with placement of a ureteral stent, and urinomas should be managed with percutaneous drainage.
Neurologic Complications
Patients undergoing CPB and circulatory arrest are at risk for cerebral ischemic insults when the total arrest time exceeds 40 minutes. Retrograde cerebral perfusion and “trickle” maintenance blood flow (5 to 10 mL/kg/min) can be employed when the ischemic period is expected to be long. When the foramen ovale remains patent, tumor thrombus that dislodges can cause massive stroke. After a single instance of this devastating complication, the authors abandoned the median sternotomy technique to gain distal control via the minimally access approach, which was hoped to reduce the risk of embolism of the thrombus.
Compression neurapraxia is a recognized complication of the flank position for nephrectomy (Garland and Moorhouse, 1952). In addition, intraoperative hypothermia, which is used in IVC thrombectomy, is a risk factor for development of nerve injury (Graham et al, 1981). Nerve injury due to intraoperative positioning is caused by stretch, ischemia, or compression. Risk factors include thin body habitus, diabetes mellitus, existing neuropathy, peripheral vascular disease, malnutrition, and intraoperative hypothermia or hypotension (Winfree and Kline, 2005). Particular care should be taken, especially in high-risk patients, to properly pad and position all areas at risk (Winfree and Kline, 2005). The authors advocate a team approach with the anesthesiologists for positioning and make an effort to reposition the arms every 2 to 3 hours during prolonged operations.
The bulk of neurapraxia injuries resolve with conservative measures. Postoperative complaints should be addressed with physical therapy and neurology evaluations if symptoms persist or worsen. Pain is treated with analgesics. Physical therapy will help preserve range of motion and joint flexibility in the presence of motor deficits. The physical examination and electromyography findings should normalize over time, although surgical intervention may be indicated if the deficit does not improve.
The flank approaches risk injury to the intercostal nerves and subcostal nerve, which can result in denervation and paresis of the flank musculature. Consequences can include chronic pain or flank bulge, as discussed earlier.
Intra-Abdominal Complications
Abdominal complications include injury to adjacent structures such as the colon, liver, spleen, pancreas, and duodenum. Injury to the bowel can be a devastating complication of renal surgery. Intraoperative vigilance can help prevent and recognize bowel injury, which should be repaired and drained. Adhesions from prior surgery increase the risk of injury to the small bowel. The small bowel can suffer injury when packed away under a retractor. The authors close simple serosal tears or enterotomies with interrupted polyglactin 3-0 sutures in a two-layered Lembert fashion. When necessary, small bowel resection is performed using gastrointestinal staplers and a side-to-side anastomosis. The large bowel can be injured when it is mobilized, especially during adhesiolysis or resection of locally advanced tumors invading the colon or mesocolon.
Right radical nephrectomy requiring Kocher mobilization can lead to injuries of the second or third portions of the duodenum. A duodenal injury should be treated with sharp debridement of nonviable tissue. Two-layered closure in Lembert fashion with an omental patch is the optimal closure to limit leaks and provide additional blood support for healing. Complete mobilization of the small bowel mesentery can maximize exposure of the duodenum and the degree of medial reflection. Use of moist sponges and gentle retraction will limit undue pressure. The same approach should be taken on left-sided tumors to minimize damage to the fourth portion of the duodenum and the tail of the pancreas.
Left-sided tumors requiring mobilization and division of the splenorenal ligament can lead to inadvertent splenic injury. Simple lacerations can be addressed with electrocautery or the argon laser. Larger defects can be managed with bolster repairs utilizing Surgicel or Gelfoam. Omental patches can provide additional support. Persistent bleeding necessitates splenectomy. The spleen is first mobilized anterior and medially, and the short gastric vessels are ligated with 2-0 silk sutures or controlled with Harmonic shears (Ethicon Endo Surgery, Inc., Cincinnati, Ohio). The splenic artery and vein should be ligated individually with suture ligatures of 2-0 silk. Postoperative immunizations to encapsulated bacteria are given before discharge.
Liver lacerations can be very troublesome depending on the location. Superficial lacerations can be managed with electrocautery or an argon beam laser. Alternatively, hemostatic bolsters of Surgicel can be sewn over defects. In the case of severe bleeding from liver injury, partial hepatectomy with assistance of hepatobiliary surgeons may be necessary.
Pancreatic injuries for the most part tend to result from inadequate mobilization or excess traction during exposure. Pancreatic injuries recognized at the time of surgery require that the capsule of the pancreas be closed with nonabsorbable suture and drained with a Jackson-Pratt suction device. In the rare instance that a pancreatectomy is necessary, a consult with hepatobiliary or general surgeons should be obtained. The tail of the pancreas, which can be injured in left nephrectomy, can be resected in a straightforward manner. The pancreas is mobilized and the tail resected with a gastrointestinal stapler. The resected edge may be oversewn with nonabsorbable nylon or polypropylene suture and use of a Surgicel bolster.
Postoperative ileus with significant pain should alert one to possible pancreatitis. Conservative management with parental nutrition and bowel rest remains the primary therapy. A pancreatic fistula usually presents as acute pancreatitis and is confirmed by measuring the amylase level and pH of the draining fluid. Serum chemistries should be obtained and CT performed to delineate the extent of disease. Pancreatic leak is managed by percutaneous or surgical drainage of the fluid collection, hyperalimentation, and administration of somatostatin until the drainage ends or requires surgical management. Most fistulas close spontaneously with adequate drainage. Early drainage of peripancreatic collections can limit pseudocyst or abscess formation. Surgical closure by excision of the fistulous tract and construction of an anastomosis between the pancreas and a Roux-en-Y limb of the jejunum is only occasionally necessary in patients with prolonged drainage.
Prolonged ileus and small bowel obstruction are potential complications of any surgery, especially the transperitoneal approach.
Incisional Complications
Incisional hernias can be a vexing complication. Anterior midline hernias may result from a technical error in failing to identify and reapproximate the rectus fascia. Patients with multiple prior incisions may be best served by closure with mesh or retention sutures.
Flank bulges after nephrectomy are often not hernias but rather result from paresis of the flank musculature due to intercostal or subcostal nerve injury. An effort should be made to spare the intercostal nerve, which lies between the internal oblique and the transverse abdominal muscles, by padding and retracting the nerve out of the surgical field.
Flank incisional hernias are rare and may result from inappropriate closure of the fascia of the oblique muscles. A fascial defect is often palpable. Closing flank incisions in multiple layers will reduce the incidence of hernia.
Partial or complete dehiscence of the operative wound can occur in 1% to 4% of midline incisions and less than 1% of all flank incisions. Most occur between postoperative days 6 to 8 when tensile strength is relatively weak. Risk factors include poor surgical technique, immunosuppression, malnutrition, poorly controlled diabetes, and sepsis (Riou et al, 1992). The key to management of this complication is prevention with a well-planned incision and solid tissue approximation. Retention sutures should be considered in those patients at risk. A complete revision with retention sutures and synthetic mesh reinforcement should be considered in treating wound dehiscences.
Infectious Complications
Preoperative antibiotics are administered per guidelines discussed in earlier sections. Superficial wound infections are treated by opening the wound to allow purulent material to drain. Deep wound infections, and those accompanied by cellulitis or systemic symptoms, are treated with drainage and antibiotics. Small wounds can be managed with wet-to-dry packing two to three times per day to allow for secondary intention healing. Larger wounds with intact fascia heal faster with vacuum devices.
Asymptomatic lymph or serous collections in the surgical site are best managed conservatively. Infection or respiratory impingement from mass effect are indications to percutaneously drain the collection. Infected hematomas or urinomas should be drained. Prolonged drainage should raise the possibility of a retained foreign body or enteric fistula.
Vascular Injury and Hemorrhage
Blood loss in renal surgery can be significant due to the high percentage of cardiac output delivered to the kidneys. Preparation is essential. Cross-matched blood should be available. Two larger-bore peripheral intravenous catheters and an arterial catheter are standard. Any patient with an extensive cardiac or pulmonary history is managed with a pulmonary artery catheter to facilitate fluid management and determine the need for inotropic support. Vascular instruments should be open before the initial incision is made. Preoperative images should be reviewed for anomalous vasculature.
Injury to the main renal artery often will require proximal exposure at the aortic takeoff to get adequate control. In patients without significant atherosclerotic disease a side clamp can be placed for temporary occlusion until a 4-0 polypropylene repair suture can be placed. Inadvertent injury to the aorta is best repaired with gentle pressure over the aortotomy and polypropylene figure-of-eight repair sutures. Very small holes are best treated with Surgicel, absorbable gelatin sponge such as Gelfoam, or microfibrillar collagen such as Avitene, along with gentle pressure and patience. Segmental branch injuries can be difficult to manage and may require excellent hilar exposure with proximal control for clamping.
The vascular nature of renal tumors and the presence of venous tumor thrombus can lead to extensive venous collateral patterns. These veins are fragile and easily torn. Lumbar tributaries lie in difficult positions and may be injured before they are detected. If the distal end can be identified, the authors like to use a small Allis clamp for hemostasis with figure-of-eight polypropylene sutures. If complete circumferential control cannot be gained, a small snap can be used along with a gentle twisting motion followed by suture ligation. In the rare instance that a lumbar vein retracts into the deep musculature of the retroperitoneum, a large figure-of-eight suture is placed through the muscle in wide bites. Likewise, these vessels can retract into the foramina of vertebral bodies requiring figure-of-eight sutures into the vertebral periosteum. Large lesions with parasitizing vessels can bleed extensively and may be best treated with tumor extirpation and a damage control approach to bleeding after exposure is increased.
Injuries to the IVC are manageable if recognized early with adequate exposure. As a general rule the key principle is to avoid making more than one hole at a time. When a venotomy is encountered the initial move is get proximal and distal control with gentle pressure using one’s fingers or sponge sticks. An Allis clamp(s) can be used for hemostasis and a running polypropylene suture can be placed. For larger rents one can temporary occlude the vena cava using Fogarty clamps.
Postoperative bleeding after open renal surgery may be characterized by pain, signs of shock, abdominal or flank swelling, and drainage of blood through the incision or a drain site. Potential sources of postoperative bleeding include the kidney itself, the renal pedicle, and unrecognized injury to the spleen, the liver, or a mesenteric vessel. In some cases it may be best to explore the surgical site, evacuate the hematoma, and obtain hemostasis. In the event of diffuse bleeding from a clotting disorder it may be necessary to temporarily pack the wound with gauze, which can then be removed after 24 to 48 hours. Arteriovenous malformations are an additional significant complication of renal surgery that may present in a delayed fashion. They can often be treated percutaneously (Fig. 54–163). Patients should be given blood and fluid replacement as needed.
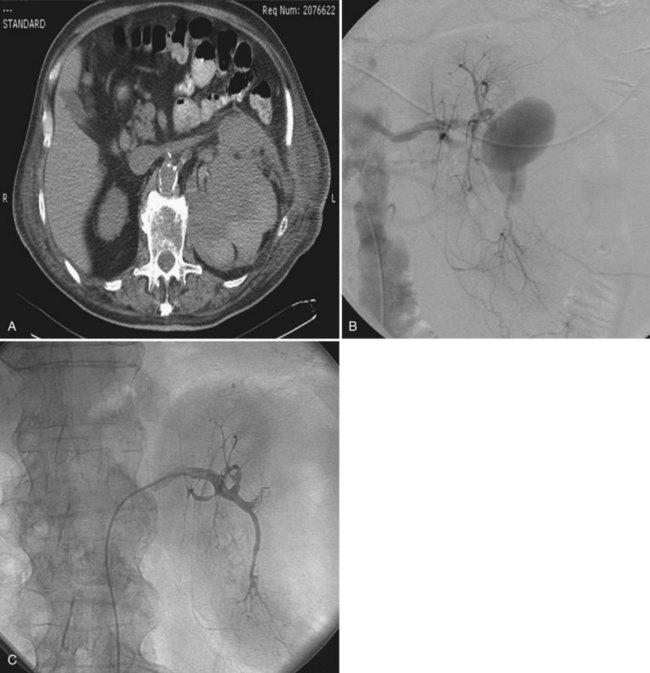
Figure 54–163 A, Perirenal hematoma 2 months after left partial nephrectomy for a large midpolar renal cell carcinoma. Patient presented with clot retention and hypotension. B, Larger 4-cm pseudoaneurysm is noted on the angiogram. C, Resolution of pseudoaneurysm after placement of coil.
Assimos DG, Boyce H, Woodruff RD, et al. Intraoperative renal ultrasonography: a useful adjunct to partial nephrectomy. J Urol. 1991;146(5):1218-1220.
Bakal CW, Cynamon J, Lakritz PS, Sprayregen S. Value of preoperative renal artery embolization in reducing blood transfusion requirements during nephrectomy for renal cell carcinoma. J Vasc Interv Radiol. 1993;4(6):727-731.
Barney JD, Churchill CE. Adenocarcinoma of the kidney with metastasis to the lung. J Urol. 1939;42:269-276.
Belzer FO, D’Alessandro AM, Hoffmann RM, et al. The use of UW solution in clinical transplantation: a 4-year experience. Ann Surg. 1992;215(6):579-583. discussion 584–5
Benincasa AB, Clements LW, Herrell SD, Galloway RL. Feasibility study for image-guided kidney surgery: assessment of required intraoperative surface for accurate physical to image space registrations. Med Phys. 2008;35(9):4251-4261.
Blackley SK, Ladaga L, Woolfitt RA, Schellhammer PF. Ex situ study of the effectiveness of enucleation in patients with renal cell carcinoma. J Urol. 1988;140(1):6-10.
Blom JH, van Poppel H, Marechal JM, et al. Radical nephrectomy with and without lymph-node dissection: final results of European Organization for Research and Treatment of Cancer (EORTC) randomized phase 3 trial 30881. Eur Urol. 2009;55(1):28-34.
Blute ML, Boorjian SA, Leibovich BC, et al. Results of inferior vena caval interruption by Greenfield filter, ligation or resection during radical nephrectomy and tumor thrombectomy. J Urol. 2007;178(2):440-445. discussion 444
Burt M. Inferior vena caval involvement by renal cell carcinoma: use of venovenous bypass as adjunct during resection. Urol Clin North Am. 1991;18(3):437-444.
Castilla EA, Liou LS, Abrahams NA, et al. Prognostic importance of resection margin width after nephron-sparing surgery for renal cell carcinoma. Urology. 2002;60(6):993-997.
Centenera LV, Hirsch JA, Choi IS, et al. Wide-necked saccular renal artery aneurysm: endovascular embolization with the Guglielmi detachable coil and temporary balloon occlusion of the aneurysm neck. J Vasc Interv Radiol. 1998;9(3):513-516.
Cherrie RJ, Goldman DG, Lindner A, deKernion JB. Prognostic implications of vena caval extension of renal cell carcinoma. J Urol. 1982;128(5):910-912.
Chowdhury UK, Mishra AK, Seth A, et al. Novel techniques for tumor thrombectomy for renal cell carcinoma with intraatrial tumor thrombus. Ann Thorac Surg. 2007;83(5):1731-1736.
Chung BI, Hamawy KJ, Zinman LN, Libertino JA. The use of bowel for ureteral replacement for complex ureteral reconstruction: long-term results. J Urol. 2006;175(1):179-183. discussion 183–4
Ciancio G, Livingstone AS, Soloway M. Surgical management of renal cell carcinoma with tumor thrombus in the renal and inferior vena cava: the University of Miami experience in using liver transplantation techniques. Eur Urol. 2007;51(4):988-994. discussion 994–5
Coll DM, Uzzo RG, Herts BR, et al. 3-Dimensional volume rendered computerized tomography for preoperative evaluation and intraoperative treatment of patients undergoing nephron sparing surgery. J Urol. 1999;161(4):1097-1102.
Dash A, Vickers AJ, Schachter LR, et al. Comparison of outcomes in elective partial vs radical nephrectomy for clear cell renal cell carcinoma of 4–7 cm. BJU Int. 2006;97(5):939-945.
Diblasio CJ, Snyder ME, Russo P. Mini-flank supra-11th rib incision for open partial or radical nephrectomy. BJU Int. 2006;97(1):149-156.
Divgi CR, Pandit-Taskar N, Jungbluth AA, et al. Preoperative characterisation of clear-cell renal carcinoma using iodine-124-labelled antibody chimeric G250 (124I-cG250) and PET in patients with renal masses: a phase I trial. Lancet Oncol. 2007;8(4):304-310.
Eklof H, Bergqvist D, Hagg A, Nyman R. Outcome after endovascular revascularization of atherosclerotic renal artery stenosis. Acta Radiol. 2009;50(3):256-264.
Fergany AF, Hafez KS, Novick AC. Long-term results of nephron sparing surgery for localized renal cell carcinoma: 10-year followup. J Urol. 2000;163(2):442-445.
Ficarra V, Righetti R, D’Amico A, et al. Renal vein and vena cava involvement does not affect prognosis in patients with renal cell carcinoma. Oncology. 2001;61(1):10-15.
Fitzgerald JM, Tripathy U, Svensson LG, Libertino JA. Radical nephrectomy with vena caval thrombectomy using a minimal access approach for cardiopulmonary bypass. J Urol. 1998;159(4):1292-1293.
Fleisher LA, Beckman JA, Brown KA, et al. ACC/AHA 2007 guidelines on perioperative cardiovascular evaluation and care for noncardiac surgery: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Writing Committee to Revise the 2002 Guidelines on Perioperative Cardiovascular Evaluation for Noncardiac Surgery): developed in collaboration with the American Society of Echocardiography, American Society of Nuclear Cardiology, Heart Rhythm Society, Society of Cardiovascular Anesthesiologists, Society for Cardiovascular Angiography and Interventions, Society for Vascular Medicine and Biology, and Society for Vascular Surgery. Circulation. 2007;116(17):e418-e499.
Foster RS, Mahomed Y, Bihrle R, Strup S. Use of a caval-atrial shunt for resection of a caval tumor thrombus in renal cell carcinoma. J Urol. 1988;140(6):1370-1371.
Geerts WH, Bergqvist D, Pineo GF, et al. Prevention of venous thromboembolism: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. 8th ed. Chest. 2008;133(6 Suppl.):381S-453S.
Gill IS, Kavoussi LR, Lane BR, et al. Comparison of 1,800 laparoscopic and open partial nephrectomies for single renal tumors. J Urol. 2007;178(1):41-46.
Giuliani L, Giberti C, Martorana G, Rovida S. Radical extensive surgery for renal cell carcinoma: long-term results and prognostic factors. J Urol. 1990;143(3):468-473. discussion 473–4
Go AS, Chertow GM, Fan D, et al. Chronic kidney disease and the risks of death, cardiovascular events, and hospitalization. N Engl J Med. 2004;351(13):1296-1305.
Gruntzig A, Hopff H. Percutaneous recanalization after chronic arterial occlusion with a new dilator-catheter (modification of the Dotter technique) (author’s transl). Dtsch Med Wochenschr. 1974;99(49):2502-2510. [in German]
Herr HW. Surgical management of renal tumors: a historical perspective. Urol Clin North Am. 2008;35(4):543-549. v
Herts BR. Role of three-dimensional imaging in surgical planning for kidney surgery. BJU Int. 2005;95(Suppl. 2):16-20.
Hollenbeck BK, Taub DA, Miller DC, et al. National utilization trends of partial nephrectomy for renal cell carcinoma: a case of underutilization? Urology. 2006;67(2):254-259.
Huang WC, Elkin EB, Levey AS, et al. Partial nephrectomy versus radical nephrectomy in patients with small renal tumors—is there a difference in mortality and cardiovascular outcomes? J Urol. 2009;181(1):55-61. discussion 62
Huang WC, Levey AS, Serio AM, et al. Chronic kidney disease after nephrectomy in patients with renal cortical tumours: a retrospective cohort study. Lancet Oncol. 2006;7(9):735-740.
Huang WC, Rhee HK, Mourtzinos AP, et al. Hyponatremia is a potentially serious complication following renal angioinfarction. J Urol. 169(4 Suppl.), 2003. abstract 115
Israel GM, Bosniak MA. An update of the Bosniak renal cyst classification system. Urology. 2005;66(3):484-488.
Kalman D, Varenhorst E. The role of arterial embolization in renal cell carcinoma. Scand J Urol Nephrol. 1999;33(3):162-170.
Kurteva T, Cohen MC, Smith GL, Libertino JA. Non-clamped, non-ischemic partial nephrectomy: the new gold standard [abstract]. J Urol. 2008;179(4):476-477.
Kutikov A, Vanarsdalen KN, Gershman B, et al. Enucleation of renal cell carcinoma with ablation of the tumour base. BJU Int. 2008;102(6):688-691.
Lacombe M, Ricco JB. Surgical revascularization of renal artery after complicated or failed percutaneous transluminal renal angioplasty. J Vasc Surg. 2006;44(3):537-544.
Lane BR, Babineau DC, Poggio ED, et al. Factors predicting renal functional outcome after partial nephrectomy. J Urol. 2008;180(6):2363-2368. discussion 2368–9
Lara PNJr, Tangen CM, Conlon SJ, et al. Predictors of survival of advanced renal cell carcinoma: long-term results from Southwest Oncology Group Trial S8949. J Urol. 2009;181(2):512-516. discussion 516–7
Lau WK, Blute ML, Weaver AL, et al. Matched comparison of radical nephrectomy vs nephron-sparing surgery in patients with unilateral renal cell carcinoma and a normal contralateral kidney. Mayo Clin Proc. 2000;75(12):1236-1242.
Lawrie GM, Morris GCJr, Glaeser DH, DeBakey ME. Renovascular reconstruction: factors affecting long-term prognosis in 919 patients followed up to 31 years. Am J Cardiol. 1989;63(15):1085-1092.
Levey AS, Bosch JP, Lewis JB, et al. A more accurate method to estimate glomerular filtration rate from serum creatinine: a new prediction equation. Modification of Diet in Renal Disease Study Group. Ann Intern Med. 1999;130(6):461-470.
Li QL, Guan HW, Zhang QP, et al. Optimal margin in nephron-sparing surgery for renal cell carcinoma 4 cm or less. Eur Urol. 2003;44(4):448-451.
Libertino J. Renovascular surgery. In: Campbell MF, Walsh PC, editors. Campbell’s urology. 6th ed. Philadelphia: Saunders; 1992:2521-2551.
Libertino JA. Pediatric and adult reconstructive urologic surgery, 2nd ed. Baltimore: Williams & Wilkins; 1987.
Libertino JA. Reconstructive urologic surgery, 3rd ed. St. Louis: Mosby; 1998.
Libertino JA, Bosco PJ, Ying CY, et al. Renal revascularization to preserve and restore renal function. J Urol. 1992;147(6):1485-1487.
Libertino JA, Selman FJJr. Alternatives to aortorenal revascularization. J Cardiovasc Surg (Torino). 1982;23(4):318-322.
Libertino JA, Zinman L, Breslin DJ, Swinton NWJr. Hepatorenal artery bypass in the management of renovascular hypertension. J Urol. 1976;115(4):369-372.
Libertino JA, Zinman L, Breslin DJ, et al. Renal artery revascularization: restoration of renal function. JAMA. 1980;244(12):1340-1342.
Libertino JA, Zinman L, Watkins EJr. Long-term results of resection of renal cell cancer with extension into inferior vena cava. J Urol. 1987;137(1):21-24.
Marshall FF, Taxy JB, Fishman EK, Chang R. The feasibility of surgical enucleation for renal cell carcinoma. J Urol. 1986;135(2):231-234.
Matin SF, Novick AC. Renal dysfunction associated with staged bilateral partial nephrectomy: the importance of operative positioning. J Urol. 2001;165(3):880-881.
McKiernan J, Simmons R, Katz J, Russo P. Natural history of chronic renal insufficiency after partial and radical nephrectomy. Urology. 2002;59(6):816-820.
Meng MV, Freise CE, Stoller ML. Expanded experience with laparoscopic nephrectomy and autotransplantation for severe ureteral injury. J Urol. 2003;169(4):1363-1367.
Mohr SJ, Whitesel JA. Spontaneous regression of renal cell carcinoma metastases after preoperative embolization of primary tumor and subsequent nephrectomy. Urology. 1979;14(1):5-8.
Moinzadeh A, Libertino JA. Prognostic significance of tumor thrombus level in patients with renal cell carcinoma and venous tumor thrombus extension: is all T3b the same? J Urol. 2004;171(2 Pt. 1):598-601.
Nguyen MM, Gill IS, Ellison LM. The evolving presentation of renal carcinoma in the United States: trends from the Surveillance, Epidemiology, and End Results program. J Urol. 2006;176(6 Pt. 1):2397-2400. discussion 2400
Novick AC. Percutaneous transluminal angioplasty and surgery of the renal artery. Eur J Vasc Surg. 1994;8(1):1-9.
Novick AC. Open surgery of the kidney. In: Campbell MF, Wein AJ, Kavoussi LR, editors. Campbell-Walsh urology. 9th ed. Philadelphia: WB Saunders; 2007:1686-1758.
Novick AC, Jackson CL, Straffon RA. The role of renal autotransplantation in complex urological reconstruction. J Urol. 1990;143(3):452-457.
Pagano D, Carey JA, Patel RL, et al. Retrograde cerebral perfusion: clinical experience in emergency and elective aortic operations. Ann Thorac Surg. 1995;59(2):393-397.
Pahernik S, Roos F, Rohrig B, et al. Elective nephron sparing surgery for renal cell carcinoma larger than 4 cm. J Urol. 2008;179(1):71-74. discussion 74
Parekh DJ, Cookson MS, Chapman W, et al. Renal cell carcinoma with renal vein and inferior vena caval involvement: clinicopathological features, surgical techniques and outcomes. J Urol. 2005;173(6):1897-1902.
Paty PS, Darling RC3rd, Lee D, et al. Is prosthetic renal artery reconstruction a durable procedure? An analysis of 489 bypass grafts. J Vasc Surg. 2001;34(1):127-132.
Qaseem A, Snow V, Fitterman N, et al. Risk assessment for and strategies to reduce perioperative pulmonary complications for patients undergoing noncardiothoracic surgery: a guideline from the American College of Physicians. Ann Intern Med. 2006;144(8):575-580.
Russo P, Huang W. The medical and oncological rationale for partial nephrectomy for the treatment of T1 renal cortical tumors. Urol Clin North Am. 2008;35(4):635-643. vii
Sarkar R, Eilber FR, Gelabert HA, Quinones-Baldrich WJ. Prosthetic replacement of the inferior vena cava for malignancy. J Vasc Surg. 1998;28(1):75-81. discussion 82–3
Shahian DM, Libertino JA, Zinman LN, et al. Resection of cavoatrial renal cell carcinoma employing total circulatory arrest. Arch Surg. 1990;125(6):727-731. discussion 731–2
Skinner DG, Pritchett TR, Lieskovsky G, et al. Vena caval involvement by renal cell carcinoma: surgical resection provides meaningful long-term survival. Ann Surg. 1989;210(3):387-392. discussion 392–4
Smith GL, Cohen MC, Kurteva T, Libertino JA. Non-clamped, non-ischemic partial nephrectomy in patients with compromised preoperative renal function or with a solitary kidney [asbtract]. J Urol. 2008;179(4):377.
Terakawa T, Miyake H, Takenaka A, et al. Clinical outcome of surgical management for patients with renal cell carcinoma involving the inferior vena cava. Int J Urol. 2007;14(9):781-784.
Thomas AA, Rini BI, Lane BR, et al. Response of the primary tumor to neoadjuvant sunitinib in patients with advanced renal cell carcinoma. J Urol. 2009;181(2):518-523. discussion 523
Thompson RH, Boorjian SA, Lohse CM, et al. Radical nephrectomy for pT1a renal masses may be associated with decreased overall survival compared with partial nephrectomy. J Urol. 2008;179(2):468-471. discussion 472–3
Thompson RH, Frank I, Lohse CM, et al. The impact of ischemia time during open nephron sparing surgery on solitary kidneys: a multi-institutional study. J Urol. 2007;177(2):471-476.
Turner Warwick RT. The supracostal approach to the renal area. Br J Urol. 1965;37(6):671-672.
van der Velden JJ, van Bockel JH, Zwartendijk J, et al. Long-term results of surgical treatment of renal carcinoma in solitary kidneys by extracorporeal resection and autotransplantation. Br J Urol. 1992;69(5):486-490.
Weight CJ, Fergany AF, Gunn PW, et al. The impact of minimally invasive techniques on open partial nephrectomy: a 10-year single institutional experience. J Urol. 2008;180(1):84-88.
Wolf JSJr, Bennett CJ, Dmochowski RR, et al. Best practice policy statement on urologic surgery antimicrobial prophylaxis. J Urol. 2008;179(4):1379-1390.
Wotkowicz C, Libertino JA. Renal autotransplantation. BJU Int 2004;93(3):253–7.
Wotkowicz C, Libertino JA. Renal cell cancer: radical nephrectomy. BJU Int. 2007;99(5 Pt. B):1231-1238.
Wotkowicz C, Libertino JA, Sorcini A, Mourtzinos A. Management of renal cell carcinoma with vena cava and atrial thrombus: minimal access vs median sternotomy with circulatory arrest. BJU Int. 2006;98(2):289-297.
Wszolek MF, Wotkowicz C, Libertino JA. Surgical management of large renal tumors. Nat Clin Pract Urol. 2008;5(1):35-46.
Wunderlich H, Reichelt O, Schubert R, et al. Preoperative simulation of partial nephrectomy with three-dimensional computed tomography. BJU Int. 2000;86(7):777-781.
Zinman L, Libertino J. Revascularization of the totally occluded renal artery [abstract]. Circulation. 48(4), 1973.
Zinman L, Libertino JA. Revascularization of the chronic totally occluded renal artery with restoration of renal function. J Urol. 1977;118(4):517-521.
Almgard LE, Fernstrom I, Haverling M, Ljungqvist A. Treatment of renal adenocarcinoma by embolic occlusion of the renal circulation. Br J Urol. 1973;45(5):474-479.
Andaloro VA, Lilien OM. Posterior approach to the kidney. Urology. 1975;5(5):600-605.
Anderson GHJr, Blakeman N, Streeten DH. The effect of age on prevalence of secondary forms of hypertension in 4429 consecutively referred patients. J Hypertens. 1994;12(5):609-615.
Aoi W, Akahoshi M, Seto S, et al. Correction of hypertension by partial nephrectomy in segmental renal artery stenosis and electron microscopic studies of renin. Jpn Heart J. 1981;22(4):679-687.
April EW. Clinical anatomy, 3rd ed. Baltimore: Williams & Wilkins; 1997.
Armstrong EP, Patrick KL, Erstad BL. Comparison of preoperative skin preparation products. Pharmacotherapy. 2001;21(3):345-350.
Assimos DG, Boyce H, Woodruff RD, et al. Intraoperative renal ultrasonography: a useful adjunct to partial nephrectomy. J Urol. 1991;146(5):1218-1220.
Assouad J, Riquet M, Foucault C, et al. Renal lymphatic drainage and thoracic duct connections: implications for cancer spread. Lymphology. 2006;39(1):26-32.
Bakal CW, Cynamon J, Lakritz PS, Sprayregen S. Value of preoperative renal artery embolization in reducing blood transfusion requirements during nephrectomy for renal cell carcinoma. J Vasc Interv Radiol. 1993;4(6):727-731.
Bakke A, Gothlin JH, Haukaas SA, Kalland T. Augmentation of natural killer cell activity after arterial embolization of renal carcinomas. Cancer Res. 1982;42(9):3880-3883.
Bapoje SR, Whitaker JF, Schulz T, et al. Preoperative evaluation of the patient with pulmonary disease. Chest. 2007;132(5):1637-1645.
Barney JD, Churchill CE. Adenocarcinoma of the kidney with metastasis to the lung. J Urol. 1939;42:269-276.
Barrett BJ, Parfrey PS. Clinical practice: preventing nephropathy induced by contrast medium. N Engl J Med. 2006;354(4):379-386.
Bates RJ, Heaney JA, Kerr WSJr. Segmental calculus disease: potential of partial nephrectomy. Urology. 1981;17(5):409-414.
Belis JA, Pae WEJr, Rohner TJJr, et al. Cardiovascular evaluation before circulatory arrest for removal of vena caval extension of renal carcinoma. J Urol. 1989;141(6):1302-1307.
Belzer FO, D’Alessandro AM, Hoffmann RM, et al. The use of UW solution in clinical transplantation: a 4-year experience. Ann Surg. 1992;215(6):579-583. discussion 584–5
Belzer FO, Keaveny TV, Reed TW, Pryor JP. A new method of renal artery reconstruction. Surgery. 1970;68(4):619-624.
Benincasa AB, Clements LW, Herrell SD, Galloway RL. Feasibility study for image-guided kidney surgery: assessment of required intraoperative surface for accurate physical to image space registrations. Med Phys. 2008;35(9):4251-4261.
Berg AA. Malignant hypernephroma of the kidney, its clinical course and diagnosis, with a description of the author’s method of radical operative cure. Surg Gynecol Obstet. 1913:17.
Bijol V, Mendez GP, Hurwitz S, et al. Evaluation of the nonneoplastic pathology in tumor nephrectomy specimens: predicting the risk of progressive renal failure. Am J Surg Pathol. 2006;30(5):575-584.
Blackley SK, Ladaga L, Woolfitt RA, Schellhammer PF. Ex situ study of the effectiveness of enucleation in patients with renal cell carcinoma. J Urol. 1988;140(1):6-10.
Blom JH, van Poppel H, Marechal JM, et al. Radical nephrectomy with and without lymph-node dissection: final results of European Organization for Research and Treatment of Cancer (EORTC) randomized phase 3 Trial 30881. Eur Urol. 2009;55(1):28-34.
Blute ML, Boorjian SA, Leibovich BC, et al. Results of inferior vena caval interruption by Greenfield filter, ligation or resection during radical nephrectomy and tumor thrombectomy. J Urol. 2007;178(2):440-445. discussion 444
Bodner H, Briskin HL. Subdiaphragmatic renal exposure by resection of the eleventh rib; a brief review of all previous incisions for renal exposure. Urol Cutaneous Rev. 1950;54(5):272-276.
Boijsen E. Angiographic studies of the anatomy of single and multiple renal arteries. Acta Radiol Suppl. 1959;183:1-135.
Boorjian SA, Sengupta S, Blute ML. Renal cell carcinoma: vena caval involvement. BJU Int. 2007;99(5 Pt. B):1239-1244.
Bratzler DW, Houck PM. Antimicrobial prophylaxis for surgery: an advisory statement from the National Surgical Infection Prevention Project. Am J Surg. 2005;189(4):395-404.
Brodel M. The intrinsic blood-vessels of the kidney and their significance in nephrotomy. Fourteenth annual session of the Association of American Anatomists, Johns Hopkins University, 1900. Baltimore: Beresford; 1900. p. 251–60
Burt M. Inferior vena caval involvement by renal cell carcinoma: use of venovenous bypass as adjunct during resection. Urol Clin North Am. 1991;18(3):437-444.
Buse S, Gilfrich C, Wagener N, et al. Thoraco-abdominal approach to large retroperitoneal tumours. BJU Int. 2006;98(5):969-972.
Campbell SC, Fichtner J, Novick AC, et al. Intraoperative evaluation of renal cell carcinoma: a prospective study of the role of ultrasonography and histopathological frozen sections. J Urol. 1996;155(4):1191-1195.
Castilla EA, Liou LS, Abrahams NA, et al. Prognostic importance of resection margin width after nephron-sparing surgery for renal cell carcinoma. Urology. 2002;60(6):993-997.
Centenera LV, Hirsch JA, Choi IS, et al. Wide-necked saccular renal artery aneurysm: endovascular embolization with the Guglielmi detachable coil and temporary balloon occlusion of the aneurysm neck. J Vasc Interv Radiol. 1998;9(3):513-516.
Chatterjee S, Nam R, Fleshner N, Klotz L. Permanent flank bulge is a consequence of flank incision for radical nephrectomy in one half of patients. Urol Oncol. 2004;22(1):36-39.
Cherrie RJ, Goldman DG, Lindner A, deKernion JB. Prognostic implications of vena caval extension of renal cell carcinoma. J Urol. 1982;128(5):910-912.
Chowdhury UK, Mishra AK, Seth A, et al. Novel techniques for tumor thrombectomy for renal cell carcinoma with intraatrial tumor thrombus. Ann Thorac Surg. 2007;83(5):1731-1736.
Chung BI, Hamawy KJ, Zinman LN, Libertino JA. The use of bowel for ureteral replacement for complex ureteral reconstruction: long-term results. J Urol. 2006;175(1):179-183. discussion 183–4
Chute R, Baron JAJr, Olsson CA. The transverse upper abdominal “chevron” incision in urological surgery. Trans Am Assoc Genitourin Surg. 1967;59:14-17.
Chute R, Soutter L, Kerr WSJr. The value of the thoracoabdominal incision in the removal of kidney tumors. N Engl J Med. 1949;241(24):951-960. illust
Ciancio G, Livingstone AS, Soloway M. Surgical management of renal cell carcinoma with tumor thrombus in the renal and inferior vena cava: the University of Miami experience in using liver transplantation techniques. Eur Urol. 2007;51(4):988-994. discussion 994–5
Ciancio G, Vaidya A, Savoie M, Soloway M. Management of renal cell carcinoma with level III thrombus in the inferior vena cava. J Urol. 2002;168(4 Pt. 1):1374-1377.
Clark CD. Survival after excision of a kidney, segmental resection of the vena cava, and division of the opposite renal vein. Lancet. 1961;2(7210):1015-1016.
Clarke BG, Rudy HA, Leadbetter WF. The thoracoabdominal incision for surgery of renal, adrenal, and testicular neoplasms. Surg Gynecol Obstet. 1958;106(3):363-367.
Coll DM, Uzzo RG, Herts BR, et al. 3-Dimensional volume rendered computerized tomography for preoperative evaluation and intraoperative treatment of patients undergoing nephron sparing surgery. J Urol. 1999;161(4):1097-1102.
Cooper CS, Cohen MB, Donovan JFJr. Splenectomy complicating left nephrectomy. J Urol. 1996;155(1):30-36.
Cooper JF, Leadbetter WF, Chute R. The thoracoabdominal approach for retroperitoneal gland dissection: its application to testis tumors. 1950. J Urol. 2002;167(2 Pt. 2):920-926. discussion 927
Cosgrove DM3rd, Sabik JF. Minimally invasive approach for aortic valve operations. Ann Thorac Surg. 1996;62(2):596-597.
Craven WM, Redmond PL, Kumpe DA, et al. Planned delayed nephrectomy after ethanol embolization of renal carcinoma. J Urol. 1991;146(3):704-708.
Crawford ED, Skinner DG, Mallamo JT. Renal arteriovenous fistulas secondary to bilateral renal cell carcinoma. Urology. 1979;13(5):525-528.
Cummings KB, Li WI, Ryan JA, et al. Intraoperative management of renal cell carcinoma with supradiaphragmatic caval extension. J Urol. 1979;122(6):829-832.
D’Ancona CA, Petrucci OJr, Otsuka R. Renal cell carcinoma with thrombus in the inferior vena cava: extracorporeal circulation and deep hypothermia without open-chest surgery. Int Braz J Urol. 2005;31(1):49-50.
Dash A, Vickers AJ, Schachter LR, et al. Comparison of outcomes in elective partial vs radical nephrectomy for clear cell renal cell carcinoma of 4–7 cm. BJU Int. 2006;97(5):939-945.
Derweesh IH, Herts B, Novick AC. Three-dimensional image reconstruction for preplanning of renal surgery. Urol Clin North Am. 2003;30(3):515-528.
Diblasio CJ, Snyder ME, Russo P. Mini-flank supra-11th rib incision for open partial or radical nephrectomy. BJU Int. 2006;97(1):149-156.
Divgi CR, Pandit-Taskar N, Jungbluth AA, et al. Preoperative characterisation of clear-cell renal carcinoma using iodine-124-labelled antibody chimeric G250 (124I-cG250) and PET in patients with renal masses: a phase I trial. Lancet Oncol. 2007;8(4):304-310.
Dos Santos JC. Note sur la désobstruction des anciennes thromboses artérielles. Presse Med. 1949:57.
Dotter CT, Judkins MP. Transluminal treatment of arteriosclerotic obstruction: description of a new technic and a preliminary report of its application. Circulation. 1964;30:654-670.
Eklof H, Bergqvist D, Hagg A, Nyman R. Outcome after endovascular revascularization of atherosclerotic renal artery stenosis. Acta Radiol. 2009;50(3):256-264.
Ellenhorn JD, Smith DD, Schwarz RE, et al. Paint-only is equivalent to scrub-and-paint in preoperative preparation of abdominal surgery sites. J Am Coll Surg. 2005;201(5):737-741.
Fabrizio MD, Kavoussi LR, Jackman S, et al. Laparoscopic nephrectomy for autotransplantation. Urology. 2000;55(1):145.
Fergany AF, Hafez KS, Novick AC. Long-term results of nephron sparing surgery for localized renal cell carcinoma: 10-year followup. J Urol. 2000;163(2):442-445.
Ficarra V, Righetti R, D’Amico A, et al. Renal vein and vena cava involvement does not affect prognosis in patients with renal cell carcinoma. Oncology. 2001;61(1):10-15.
Fitzgerald JM, Tripathy U, Svensson LG, Libertino JA. Radical nephrectomy with vena caval thrombectomy using a minimal access approach for cardiopulmonary bypass. J Urol. 1998;159(4):1292-1293.
Fleisher LA, Beckman JA, Brown KA, et al. ACC/AHA 2007 guidelines on perioperative cardiovascular evaluation and care for noncardiac surgery: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Writing Committee to Revise the 2002 Guidelines on Perioperative Cardiovascular Evaluation for Noncardiac Surgery): developed in collaboration with the American Society of Echocardiography, American Society of Nuclear Cardiology, Heart Rhythm Society, Society of Cardiovascular Anesthesiologists, Society for Cardiovascular Angiography and Interventions, Society for Vascular Medicine and Biology, and Society for Vascular Surgery. Circulation. 2007;116(17):e418-e499.
Forrest JB, Clemens JQ, Finamore P, et al. AUA Best Practice Statement for the prevention of deep vein thrombosis in patients undergoing urologic surgery. J Urol. 2009;181(3):1170-1177.
Foster RS, Mahomed Y, Bihrle R, Strup S. Use of a caval-atrial shunt for resection of a caval tumor thrombus in renal cell carcinoma. J Urol. 1988;140(6):1370-1371.
Gardiner RA, Naunton-Morgan TC, Whitfield HN, et al. The modified lumbotomy versus the oblique loin incision for renal surgery. Br J Urol. 1979;51(4):256-259.
Garland H, Moorhouse D. Compressive lesions of the external popliteal (common peroneal) nerve. BMJ. 1952;2(4799):1373-1378.
Geerts WH, Bergqvist D, Pineo GF, et al. Prevention of venous thromboembolism. In: American College of Chest Physicians evidence-based clinical practice guidelines. 8th ed. Chest. 2008;133(6 Suppl.):381S-453S.
Gibson S, Kuzmarov IW, McClure DR, Morehouse DD. Blunt renal trauma: the value of a conservative approach to major injuries in clinically stable patients. Can J Surg. 1982;25(1):25-26.
Gil-Vernet JM. La lombotomie verticale posterieure: considerations a propos de 366 cas. Acta Urol Belg. 1964;32:391-400.
Gill IS, Kavoussi LR, Lane BR, et al. Comparison of 1,800 laparoscopic and open partial nephrectomies for single renal tumors. J Urol. 2007;178(1):41-46.
Giuliani L, Giberti C, Martorana G, Rovida S. Radical extensive surgery for renal cell carcinoma: long-term results and prognostic factors. J Urol. 1990;143(3):468-473. discussion 473–4
Glazer A, Novick AC. Preoperative transesophageal echocardiography for assessment of vena caval tumor thrombi: a comparative study with venacavography and magnetic resonance imaging. Urology, 49, 1997, 32-34.
Go AS, Chertow GM, Fan D, et al. Chronic kidney disease and the risks of death, cardiovascular events, and hospitalization. N Engl J Med. 2004;351(13):1296-1305.
Goldfarb DA, Novick AC, Lorig R, et al. Magnetic resonance imaging for assessment of vena caval tumor thrombi: a comparative study with venacavography and computerized tomography scanning. J Urol. 1990;144(5):1100-1103. discussion 1103–4
Graham JG, Pye IF, McQueen IN. Brachial plexus injury after median sternotomy. J Neurol Neurosurg Psychiatry. 1981;44(7):621-625.
Graves FT. The anatomy of the intrarenal arteries and its application to segmental resection of the kidney. Br J Surg. 1954;42(172):132-139.
Grégoire W. Les propagations du cancer du rein. Bull Med Soc Anat (Paris). 1903;78:764-769.
Gruntzig A, Hopff H. Percutaneous recanalization after chronic arterial occlusion with a new dilator-catheter (modification of the Dotter technique) (author’s transl). Dtsch Med Wochenschr. 1974;99(49):2502-2510. [in German]
Hardy JD. High ureteral injuries. Management by autotransplantation of the kidney. JAMA. 1963;184:97-101.
Herr HW. Surgical management of renal tumors: a historical perspective. Urol Clin North Am. 2008;35(4):543-549.
Herts BR. Role of three-dimensional imaging in surgical planning for kidney surgery. BJU Int. 2005;95(Suppl. 2):16-20.
Hess E. Resection of the rib in renal operations. J Urol. 1939:42.
Hiatt JR, Gabbay J, Busuttil RW. Surgical anatomy of the hepatic arteries in 1000 cases. Ann Surg. 1994;220(1):50-52.
Hofmann HS, Neef H, Krohe K, et al. Prognostic factors and survival after pulmonary resection of metastatic renal cell carcinoma. Eur Urol. 2005;48(1):77-81. discussion 82
Hollenbeck BK, Taub DA, Miller DC, et al. National utilization trends of partial nephrectomy for renal cell carcinoma: a case of underutilization? Urology. 2006;67(2):254-259.
Huang WC, Elkin EB, Levey AS, et al. Partial nephrectomy versus radical nephrectomy in patients with small renal tumors—is there a difference in mortality and cardiovascular outcomes? J Urol. 2009;181(1):55-61. discussion 62
Huang WC, Levey AS, Serio AM, et al. Chronic kidney disease after nephrectomy in patients with renal cortical tumours: a retrospective cohort study. Lancet Oncol. 2006;7(9):735-740.
Huang WC, Rhee HK, Mourtzinos AP, et al. Hyponatremia is a potentially serious complication following renal angioinfarction. J Urol. 169(4 Suppl.), 2003. abstract 115
Hughes FA. Resection of the 12th rib in the surgical approach to the renal fossa. J Urol. 1949;61(2):159-162.
Husberg BS, Bakshandeh K, Lilly J, et al. Five cases and five unusual indications for autogenic renal transplantation. Acta Chir Scand. 1975;141(6):557-563.
Ishikawa K. Natural history and classification of occlusive thromboaortopathy (Takayasu’s disease). Circulation. 1978;57(1):27-35.
Israel GM, Bosniak MA. An update of the Bosniak renal cyst classification system. Urology. 2005;66(3):484-488.
Iversen P, Brun C. Aspiration biopsy of the kidney. Am J Med. 1951;11(3):324-330.
Johnson G, Kalland T. Characterization of effector cells mediating the augmentation of spontaneous cell-mediated cytotoxicity induced by therapeutic infarction of human renal carcinomas. Urol Int. 1984;39(4):236-240.
Jordan ML, Novick AC, Cunningham RL. The role of renal autotransplantation in pediatric and young adult patients with renal artery disease. J Vasc Surg. 1985;2(3):385-392.
Kalman D, Varenhorst E. The role of arterial embolization in renal cell carcinoma. Scand J Urol Nephrol. 1999;33(3):162-170.
Kerbl K, Rehman J, Landman J, et al. Current management of urolithiasis: progress or regress? J Endourol. 2002;16(5):281-288.
Kerr WK, Kyle VN, Keresteci AG, Smythe CA. Renal hypothermia. J Urol. 1960;84:236-242.
Khoury EN. Thoraco-abdominal approach in lesions of the kidney, adrenal and testis: morbidity studies. J Urol. 1966;96(5):631-634.
Kimbrough JC, Morse WH. Subcapsular nephrectomy. Surg Gynecol Obstet. 1953;96(2):235-239.
Kittredge WE, Fridge JC. Subcapsular nephrectomy: comparison of fifty-three cases with three hundred eighty-five classic nephrectomies. JAMA. 1958;168(6):758-760.
Klatte T, Pantuck AJ, Riggs SB, et al. Prognostic factors for renal cell carcinoma with tumor thrombus extension. J Urol. 2007;178(4 Pt. 1):1189-1195. discussion 1195
Klimberg I, Hunter P, Hawkins IF, et al. Preoperative angioinfarction of localized renal cell carcinoma using absolute ethanol. J Urol. 1985;133(1):21-24.
Knipp BS, Dimick JB, Eliason JL, et al. Diffusion of new technology for the treatment of renovascular hypertension in the United States: surgical revascularization versus catheter-based therapy, 1988–2001. J Vasc Surg. 2004;40(4):717-723.
Kocher T, Langham T. Ein Nephrotome wegen Nierensarkom. Zugleich ein Beitrag zur Histologie des Nierenkrebses. Dtsch Z Chir (Leipzig). 1878:9. [in German]
Kottra JJ, Castellino RA. The circumaortic left renal vein: angiographic appearance. Radiology. 1970;95(1):141-143.
Kurteva T, Cohen MC, Smith GL, Libertino JA. Non-clamped, non-ischemic partial nephrectomy: the new gold standard [abstract]. J Urol. 2008;179(4):476-477.
Kuster EGF. Die Chirugirschen Krankheiten der Nieren. Stuttgart: Deutsche Chirugie; 1896–1902.
Kutikov A, Vanarsdalen KN, Gershman B, et al. Enucleation of renal cell carcinoma with ablation of the tumour base. BJU Int. 2008;102(6):688-691.
Lacombe M. Renal arteriovenous fistula following nephrectomy. Urology. 1985;25(1):13-16.
Lacombe M, Ricco JB. Surgical revascularization of renal artery after complicated or failed percutaneous transluminal renal angioplasty. J Vasc Surg. 2006;44(3):537-544.
Lane BR, Babineau DC, Poggio ED, et al. Factors predicting renal functional outcome after partial nephrectomy. J Urol. 2008;180(6):2363-2368. discussion 2368–9
Lara PNJr, Tangen CM, Conlon SJ, et al. Predictors of survival of advanced renal cell carcinoma: long-term results from Southwest Oncology Group Trial S8949. J Urol. 2009;181(2):512-516. discussion 516–7
Lau WK, Blute ML, Weaver AL, et al. Matched comparison of radical nephrectomy vs nephron-sparing surgery in patients with unilateral renal cell carcinoma and a normal contralateral kidney. Mayo Clin Proc. 2000;75(12):1236-1242.
Lawrie GM, Morris GCJr, Glaeser DH, DeBakey ME. Renovascular reconstruction: factors affecting long-term prognosis in 919 patients followed up to 31 years. Am J Cardiol. 1989;63(15):1085-1092.
Leach GE, Lieber MM. Partial nephrectomy: Mayo Clinic experience 1957–1977. Urology. 1980;15(3):219-228.
Lee TH, Marcantonio ER, Mangione CM, et al. Derivation and prospective validation of a simple index for prediction of cardiac risk of major noncardiac surgery. Circulation. 1999;100(10):1043-1049.
Levey AS, Bosch JP, Lewis JB, et al. A more accurate method to estimate glomerular filtration rate from serum creatinine: a new prediction equation. Modification of Diet in Renal Disease Study Group. Ann Intern Med. 1999;130(6):461-470.
Li QL, Guan HW, Zhang QP, et al. Optimal margin in nephron-sparing surgery for renal cell carcinoma 4 cm or less. Eur Urol. 2003;44(4):448-451.
Libertino J. Renovascular surgery. In: Campbell MF, Walsh PC, editors. Campbell’s urology. 6th ed. Philadelphia: WB Saunders; 1992:2521-2551.
Libertino JA. Pediatric and adult reconstructive urologic surgery, 2nd ed. Baltimore: Williams & Wilkins; 1987.
Libertino JA. Reconstructive urologic surgery, 3rd ed. St. Louis: Mosby; 1998.
Libertino JA, Bosco PJ, Ying CY, et al. Renal revascularization to preserve and restore renal function. J Urol. 1992;147(6):1485-1487.
Libertino JA, Flam TA, Zinman LN, et al. Changing concepts in surgical management of renovascular hypertension. Arch Intern Med. 1988;148(2):357-359.
Libertino JA, Lagneau P. A new method of revascularization of the right renal artery by the gastroduodenal artery. Surg Gynecol Obstet. 1983;156(2):221-223.
Libertino JA, Selman FJJr. Alternatives to aortorenal revascularization. J Cardiovasc Surg (Torino). 1982;23(4):318-322.
Libertino JA, Zinman L. Renal revascularization using aortorenal saphenous vein bypass grafting. Surg Clin North Am. 1980;60(3):487-501.
Libertino JA, Zinman L, Breslin DJ, Swinton NWJr. Hepatorenal artery bypass in the management of renovascular hypertension. J Urol. 1976;115(4):369-372.
Libertino JA, Zinman L, Breslin DJ, et al. Renal artery revascularization: restoration of renal function. JAMA. 1980;244(12):1340-1342.
Libertino JA, Zinman L, Watkins EJr. Long-term results of resection of renal cell cancer with extension into inferior vena cava. J Urol. 1987;137(1):21-24.
Licht MR, Novick AC, Goormastic M. Nephron sparing surgery in incidental versus suspected renal cell carcinoma. J Urol. 1994;152(1):39-42.
Longnecker DE. Principles and practice of anesthesiology. New York: McGraw-Hill; 2007.
Maatman TJ, Novick AC, Tancinco BF, et al. Renal oncocytoma: a diagnostic and therapeutic dilemma. J Urol. 1984;132(5):878-881.
Mangram AJ, Horan TC, Pearson ML, et al. Guideline for prevention of surgical site infection, 1999. Centers for Disease Control and Prevention (CDC) Hospital Infection Control Practices Advisory Committee. Am J Infect Control. 1999;27(2):97-132. quiz 133–4; discussion 96
Marshall FF, Taxy JB, Fishman EK, Chang R. The feasibility of surgical enucleation for renal cell carcinoma. J Urol. 1986;135(2):231-234.
Matin SF, Novick AC. Renal dysfunction associated with staged bilateral partial nephrectomy: the importance of operative positioning. J Urol. 2001;165(3):880-881.
Matlaga BR, Assimos DG. Changing indications of open stone surgery. Urology. 2002;59(4):490-493. discussion 493–4
Mault JR, Ohtake S, Klingensmith ME, et al. Cerebral metabolism and circulatory arrest: effects of duration and strategies for protection. Ann Thorac Surg. 1993;55(1):57-63. discussion 64
McGahan JP, Blake LC, deVere White R, et al. Color flow sonographic mapping of intravascular extension of malignant renal tumors. J Ultrasound Med. 1993;12:403-409.
McKiernan J, Simmons R, Katz J, Russo P. Natural history of chronic renal insufficiency after partial and radical nephrectomy. Urology. 2002;59(6):816-820.
Mejean A, Vogt B, Quazza JE, et al. Mortality and morbidity after nephrectomy for renal cell carcinoma using a transperitoneal anterior subcostal incision. Eur Urol. 1999;36(4):298-302.
Meng MV, Freise CE, Stoller ML. Expanded experience with laparoscopic nephrectomy and autotransplantation for severe ureteral injury. J Urol. 2003;169(4):1363-1367.
Menon M. Dorsal lumbotomy. In: Hinman F, editor. Atlas of urologic surgery. 2nd ed. Philadelphia: WB Saunders; 1998:896-901.
Merklin RJ, Michels NA. The variant renal and suprarenal blood supply with data on the inferior phrenic, ureteral and gonadal arteries: a statistical analysis based on 185 dissections and review of the literature. J Int Coll Surg. 1958;29(1 Pt. 1):41-76.
Mickisch GH, Garin A, van Poppel H, et alEuropean Organisation for Research and Treatment of Cancer (EORTC) Genitourinary Group. Radical nephrectomy plus interferon-alfa-based immunotherapy compared with interferon-alfa alone in metastatic renal-cell carcinoma: a randomised trial. Lancet. 2001;358:966.
Middleton RG, Presto AJ3rd. Radical thoracoabdominal nephrectomy for renal cell carcinoma. J Urol. 1973;110(1):36-37.
Miller DC, Hollingsworth JM, Hafez KS, et al. Partial nephrectomy for small renal masses: an emerging quality of care concern? J Urol. 2006;175(3 Pt. 1):853-857. discussion 858
Mohr SJ, Whitesel JA. Spontaneous regression of renal cell carcinoma metastases after preoperative embolization of primary tumor and subsequent nephrectomy. Urology. 1979;14(1):5-8.
Moinzadeh A, Libertino JA. Prognostic significance of tumor thrombus level in patients with renal cell carcinoma and venous tumor thrombus extension: is all T3b the same? J Urol. 2004;171(2 Pt. 1):598-601.
Morris H. A case of nephrolithotomy; or on the extraction of a calculus from an undilated kidney. Trans Clin Soc Lond. 1881:14.
Mortensen H. Transthoracic nephrectomy. J Urol. 1948;60(6):855-858.
Nakano H, Nihira H, Toge T. Treatment of renal cancer patients by transcatheter embolization and its effects on lymphocyte proliferative responses. J Urol. 1983;130(1):24-27.
Neves RJ, Zincke H. Surgical treatment of renal cancer with vena cava extension. Br J Urol. 1987;59(5):390-395.
Nguyen MM, Gill IS, Ellison LM. The evolving presentation of renal carcinoma in the United States: trends from the Surveillance, Epidemiology, and End Results program. J Urol. 2006;176(6 Pt. 1):2397-2400. discussion 2400
Novick AC. Posterior surgical approach to the kidney and ureter. J Urol. 1980;124(2):192-195.
Novick AC. Percutaneous transluminal angioplasty and surgery of the renal artery. Eur J Vasc Surg. 1994;8(1):1-9.
Novick AC. Open surgery of the kidney. In: Campbell MF, Wein AJ, Kavoussi LR, editors. Campbell-Walsh urology. 9th ed. Philadelphia: WB Saunders; 2007:1686-1758.
Novick AC, Jackson CL, Straffon RA. The role of renal autotransplantation in complex urological reconstruction. J Urol. 1990;143(3):452-457.
Novick AC, Kaye MC, Cosgrove DM, et al. Experience with cardiopulmonary bypass and deep hypothermic circulatory arrest in the management of retroperitoneal tumors with large vena caval thrombi. Ann Surg. 1990;212(4):472-476. discussion 476–7
Novick AC, Magnusson M, Braun WE. Multiple-artery renal transplantation: emphasis on extracorporeal methods of donor arterial reconstruction. J Urol. 1979;122(6):731-735.
Nuttall M, Cathcart P, van der Meulen J, et al. A description of radical nephrectomy practice and outcomes in England: 1995–2002. BJU Int. 2005;96(1):58-61.
Okamoto M, Hashimoto M, Akita T, et al. Congestive heart failure caused by aortocaval fistula after nephrectomy. Intern Med. 2001;40(11):1113-1116.
Olin JW, Melia M, Young JR, et al. Prevalence of atherosclerotic renal artery stenosis in patients with atherosclerosis elsewhere. Am J Med. 1990;88(1N):46N-51N.
Olsson CA, Idelson B. Renal autotransplantation for recurrent renal colic. J Urol. 1980;123(4):467-474.
Ota K, Mori S, Awane Y, Ueno A. Ex situ repair of renal artery for renovascular hypertension. Arch Surg. 1967;94(3):370-373.
Pagano D, Carey JA, Patel RL, et al. Retrograde cerebral perfusion: clinical experience in emergency and elective aortic operations. Ann Thorac Surg. 1995;59(2):393-397.
Pahernik S, Roos F, Rohrig B, et al. Elective nephron sparing surgery for renal cell carcinoma larger than 4 cm. J Urol. 2008;179(1):71-74. discussion 74
Papathanassiadis S, Swinney J. Results of partial nephrectomy compared with pyelolithotomy and nephrolithotomy. Br J Urol. 1966;38(4):403-409.
Parekh DJ, Cookson MS, Chapman W, et al. Renal cell carcinoma with renal vein and inferior vena caval involvement: clinicopathological features, surgical techniques and outcomes. J Urol. 2005;173(6):1897-1902.
Parker A. Studies on the main posterior lymph channels of the abdomen and their connections with the lymphatics of the genito-urinary system. Am J Anat. 1935:56.
Parrott TS, Woodard JR, Trulock TS, Glenn JF. Segmental renal vein renins and partial nephrectomy for hypertension in children. J Urol. 1984;131(4):736-739.
Paster SB, Bergreen P, Schwarz H. Percutaneous catheter-aided infarction of renal tumors: a preliminary report. J Urol. 1975;114(3):351-354.
Pathak IC. Survival after right nephrectomy, excision of infrahepatic vena cava and ligation of left renal vein: a case report. J Urol. 1971;106(4):599-602.
Paty PS, Darling RC3rd, Lee D, et al. Is prosthetic renal artery reconstruction a durable procedure? An analysis of 489 bypass grafts. J Vasc Surg. 2001;34(1):127-132.
Paul R, Mordhorst J, Busch R, et al. Adrenal sparing surgery during radical nephrectomy in patients with renal cell cancer: a new algorithm. J Urol. 2001;166(1):59-62.
Pettus JA, Eggener SE, Shabsigh A, et al. Perioperative clinical thromboembolic events after radical or partial nephrectomy. Urology. 2006;68(5):988-992.
Pritchett TR, Raval JK, Benson RC, et al. Preoperative magnetic resonance imaging of vena caval tumor thrombi: experience with 5 cases. J Urol. 1987;138(5):1220-1222.
Putnam CW, Halgrimson CG, Stables DP, et al. Ex vivo renal perfusion and autotransplantation in treatment of calculous disease or abdominal aortic aneurysm. Urology. 1975;05(3):337-342.
Qaseem A, Snow V, Fitterman N, et al. Risk assessment for and strategies to reduce perioperative pulmonary complications for patients undergoing noncardiothoracic surgery: a guideline from the American College of Physicians. Ann Intern Med. 2006;144(8):575-580.
Rehn E. Gefäßkomplikationen und ihre Beherrschung bei dem Hypernephrom. Z Urol Chir. 1922:10. [in German]
Rihal CS, Textor SC, Breen JF, et al. Incidental renal artery stenosis among a prospective cohort of hypertensive patients undergoing coronary angiography. Mayo Clin Proc. 2002;77(4):309-316.
Riou JP, Cohen JR, Johnson HJr. Factors influencing wound dehiscence. Am J Surg. 1992;163(3):324-330.
Robson CJ, Churchill BM, Anderson W. The results of radical nephrectomy for renal cell carcinoma. J Urol. 1969;101(3):297-301.
Rosenstein P. Pneumoradiology of kidney position-a new technique for the radiological representation of the kidneys and neighboring organs (suprarenal gland, spleen, liver). J Urol. 1921;15:447.
Rosenthal CL, Kraft R, Zingg EJ. Organ-preserving surgery in renal cell carcinoma: tumor enucleation versus partial kidney resection. Eur Urol. 1984;10(4):222-228.
Roy C, Tuchmann C, Morel M, et al. Is there still a place for angiography in the management of renal mass lesions? Eur Radiol. 1999;9(2):329-335.
Russo P, Goetzl M, Simmons R, et al. Partial nephrectomy: the rationale for expanding the indications. Ann Surg Oncol. 2002;9(7):680-687.
Russo P, Huang W. The medical and oncological rationale for partial nephrectomy for the treatment of T1 renal cortical tumors. Urol Clin North Am. 2008;35(4):635-643. vii
Sagalowsky AI, Kadesky KT, Ewalt DM, Kennedy TJ. Factors influencing adrenal metastasis in renal cell carcinoma. J Urol. 1994;151(5):1181-1184.
Sarkar R, Eilber FR, Gelabert HA, Quinones-Baldrich WJ. Prosthetic replacement of the inferior vena cava for malignancy. J Vasc Surg. 1998;28(1):75-81. discussion 82–3
Schefft P, Novick AC, Straffon RA, Stewart BH. Surgery for renal cell carcinoma extending into the inferior vena cava. J Urol. 1978;120(1):28-31.
Scherr DS, Ng C, Munver R, et al. Practice patterns among urologic surgeons treating localized renal cell carcinoma in the laparoscopic age: technology versus oncology. Urology. 2003;62(6):1007-1011.
Schwartz MJ, Smith EB, Trost DW, Vaughan EDJr. Renal artery embolization: clinical indications and experience from over 100 cases. BJU Int. 2007;99(4):881-886.
Shahian DM, Libertino JA, Zinman LN, et al. Resection of cavoatrial renal cell carcinoma employing total circulatory arrest. Arch Surg. 1990;125(6):727-731. discussion 731–2
Simon G. Extirpation einer Niere am Menschen Dtsch Klin 1870;22 [in German].
Skinner DG, Colvin RB, Vermillion CD, et al. Diagnosis and management of renal cell carcinoma: a clinical and pathologic study of 309 cases. Cancer. 1971;28(5):1165-1177.
Skinner DG, Pritchett TR, Lieskovsky G, et al. Vena caval involvement by renal cell carcinoma: surgical resection provides meaningful long-term survival. Ann Surg. 1989;210(3):387-392. discussion 392–4
Smith GL, Cohen MC, Kurteva T, Libertino JA. Non-clamped, non-ischemic partial nephrectomy in patients with compromised preoperative renal function or with a solitary kidney [asbtract]. J Urol. 2008;179(4):377.
Smith MJ, Boyce WH. Anatrophic nephrotomy and plastic calyrhaphy. J Urol. 1968;99(5):521-527.
Staehler G, Brkovic D. The role of radical surgery for renal cell carcinoma with extension into the vena cava. J Urol. 2000;163(6):1671-1675.
Svensson LG, Crawford ES, Hess KR, et al. Deep hypothermia with circulatory arrest: determinants of stroke and early mortality in 656 patients. J Thorac Cardiovasc Surg. 1993;106(1):19-28. discussion 31
Swanson DA, Johnson DE, von Eschenbach AC, et al. Angioinfarction plus nephrectomy for metastatic renal cell carcinoma—an update. J Urol. 1983;130(3):449-452.
Tanner J, Woodings D, Moncaster K. Preoperative hair removal to reduce surgical site infection. Cochrane Database Syst Rev. 2006;3:CD004122.
Terakawa T, Miyake H, Takenaka A, et al. Clinical outcome of surgical management for patients with renal cell carcinoma involving the inferior vena cava. Int J Urol. 2007;14(9):781-784.
Tessler AN, Yuvienco F, Farcon E. Paramedian extraperitoneal incision for total nephroureterectomy. Urology. 1975;05(3):397-398.
Thomas AA, Rini BI, Lane BR, et al. Response of the primary tumor to neoadjuvant sunitinib in patients with advanced renal cell carcinoma. J Urol. 2009;181(2):518-523. discussion 523
Thompson RH, Boorjian SA, Lohse CM, et al. Radical nephrectomy for pT1a renal masses may be associated with decreased overall survival compared with partial nephrectomy. J Urol. 2008;179(2):468-471. discussion 472–3
Thompson RH, Frank I, Lohse CM, et al. The impact of ischemia time during open nephron sparing surgery on solitary kidneys: a multi-institutional study. J Urol. 2007;177(2):471-476.
Tollefson MK, Kawashima A, Blute ML. In situ partial nephrectomy and tumor thrombectomy for renal cell carcinoma with level II vena cava extension in a solitary kidney. Urology. 2005;66(4):882.
Treiger BF, Humphrey LS, Peterson CVJr, et al. Transesophageal echocardiography in renal cell carcinoma: an accurate diagnostic technique for intracaval neoplastic extension. J Urol. 1991;145(6):1138-1140.
Tsui K-H, Shvarts O, Barbaric Z, et al. Is adrenalectomy a necessary component of radical nephrectomy? UCLA experience with 511 radical nephrectomies. J Urol. 2000;163(2):437-441.
Turner-Warwick RT. The supracostal approach to the renal area. Br J Urol. 1965;37(6):671-672.
van der Velden JJ, van Bockel JH, Zwartendijk J, et al. Long-term results of surgical treatment of renal carcinoma in solitary kidneys by extracorporeal resection and autotransplantation. Br J Urol. 1992;69(5):486-490.
Ward JN, Lavengood RWJr, Subramaniam AP, Draper JW. Lumbar approaches to kidney: complications associated with procedure. Urology. 1974;3(2):163-167.
Weckermann D, Schlotmann R, Tietze W, Hackel T. Gas formation after renal artery embolisation: genesis and clinical relevance. Urol Int. 1992;49(4):211-214.
Weight CJ, Fergany AF, Gunn PW, et al. The impact of minimally invasive techniques on open partial nephrectomy: a 10-year single institutional experience. J Urol. 2008;180(1):84-88.
Wells S. Successful removal of two solid circumrenal tumors. BMJ. 1884:1.
Willscher MK, Conway JFJr. Retrograde nephrostomy tube placement during open surgical procedures requiring urinary diversion. J Urol. 1987;138(5):1196-1197.
Winfree CJ, Kline DG. Intraoperative positioning nerve injuries. Surg Neurol. 2005;63(1):5-18. discussion 518
Wolf JSJr, Bennett CJ, Dmochowski RR, et al. Best practice policy statement on urologic surgery antimicrobial prophylaxis. J Urol. 2008;179(4):1379-1390.
Wotkowicz C, Libertino JA. Renal autotransplantation. BJU Int. 2004;93(3):253-257.
Wotkowicz C, Libertino JA. Renal cell cancer: radical nephrectomy. BJU Int. 2007;99(5 Pt. B):1231-1238.
Wotkowicz C, Libertino JA, Sorcini A, Mourtzinos A. Management of renal cell carcinoma with vena cava and atrial thrombus: minimal access vs. median sternotomy with circulatory arrest. BJU Int. 2006;98(2):289-297.
Wszolek MF, Wotkowicz C, Libertino JA. Surgical management of large renal tumors. Nat Clin Pract Urol. 2008;5(1):35-46.
Wunderlich H, Reichelt O, Schubert R, et al. Preoperative simulation of partial nephrectomy with three-dimensional computed tomography. BJU Int. 2000;86(7):777-781.
Yang JC, Hye RJ. Ruptured renal artery aneurysm during pregnancy. Ann Vasc Surg. 1996;10(4):370-372.
Zielinski H, Szmigielski S, Petrovich Z. Comparison of preoperative embolization followed by radical nephrectomy with radical nephrectomy alone for renal cell carcinoma. Am J Clin Oncol. 2000;23(1):6-12.
Zinman L, Libertino J. Revascularization of the totally occluded renal artery [abstract]. Circulation. 48(4), 1973.
Zinman L, Libertino JA. Revascularization of the chronic totally occluded renal artery with restoration of renal function. J Urol. 1977;118(4):517-521.