chapter 80 Urothelial Tumors of the Bladder
The urothelium of the bladder is traditionally considered to be lined by transitional cells, which, as suggested by the name, can transform into a variety of benign and malignant tumors. Therefore the list of bladder tumors is long and includes those derived from the urothelium and mesenchyme. In this chapter we will discuss the epidemiology, etiology, pathology, staging, origin, recurrence, dissemination, molecular biology, detection, and prevention of the more common benign and malignant tumors of the bladder, with an emphasis on urothelial cancer.
Benign Tumors of the Bladder
There are numerous benign tumors of the bladder, but the more common ones include epithelial metaplasia, leukoplakia, inverted papilloma, nephrogenic adenoma, leiomyoma, cystitis cystica, and cystitis glandularis.
Epithelial Metaplasia
Epithelial metaplasia is focal areas of transformed urothelium with normal nuclear and cellular architecture surrounded by normal urothelium usually located on the trigone and composed of squamous (squamous metaplasia) or glandular (glandular metaplasia) cells. Squamous metaplasia often has a knobby appearance and is covered by white, flaky, easily disrupted material lying on the trigone. Glandular metaplasia appears as clumps of raised red areas that appear inflammatory and are often confused for cancer. Approximately 40% of women and 5% of men have squamous metaplasia of the bladder, which is usually related to infection, trauma, and surgery (Ozbey et al, 1999). There are no racial differences, and squamous metaplasia is more common in women of childbearing age. Spinal cord injury is associated with squamous metaplasia most likely from catheter trauma and urinary tract infections (Vaidyanathan et al, 2003). Glandular metaplasia can extensively involve the bladder, particularly the trigone, but biopsy is not required. Treatment is unnecessary, and a preventive agent has not been identified.
Leukoplakia
Leukoplakia of the bladder is similar to squamous metaplasia with the addition of keratin deposition that appears as a white flaky substance floating in the bladder (Staack et al, 2006). Leukoplakia occurs in other organs that are covered by squamous epithelium and is often premalignant (Zhang et al, 2009). However, cytogenetic studies on bladder leukoplakia are consistent with a benign lesion, and no treatment is necessary (Staack et al, 2006).
Inverted Papilloma
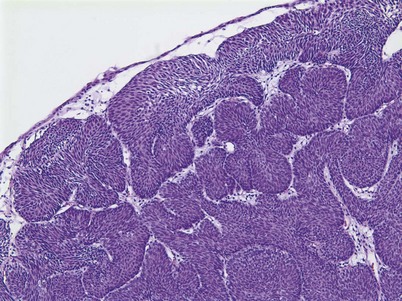
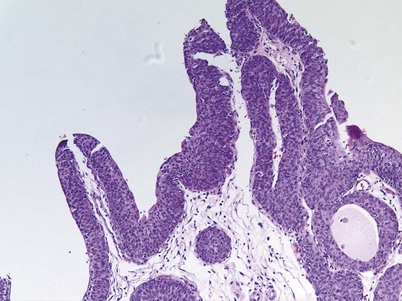
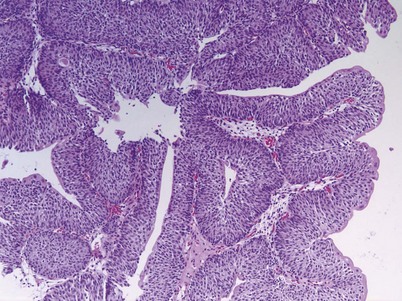
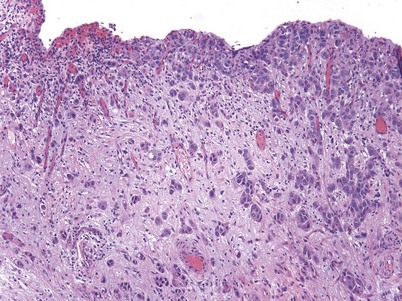
An inverted papilloma is a benign proliferative lesion that is associated with chronic inflammation or bladder outlet obstruction and can be located throughout the bladder but most commonly on the trigone, comprising less than 1% of all bladder tumors (Sung et al, 2006; Jones et al, 2007; Kilciler et al, 2008). Inverted papillomas demonstrate an inverted growth pattern composed of anastomosing islands of histologically and cytologically normal urothelial cells invaginating from the surface urothelium into the lamina propria but not into the muscularis propria (Fig. 80–1) (Sung et al, 2006). When diagnosed according to strictly defined criteria (e.g., lack of cytologic atypia), inverted papillomas behave in a benign fashion with only a 1% incidence of tumor recurrence (Sung et al, 2006; Kilciler et al, 2008). Occasionally, inverted papillomas are present with coexistent urothelial cancer elsewhere in the urinary system, occurring more commonly in the upper tract than the bladder (Asano et al, 2003). The use of fluorescent in-situ hybridization (FISH) to evaluate chromosomal changes can distinguish between an inverted papilloma and a urothelial cancer with an inverted growth pattern (Jones et al, 2007). Transurethral resection is the treatment of choice.
Papilloma
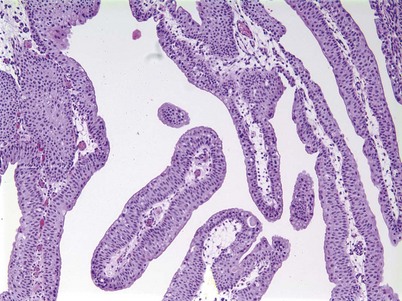
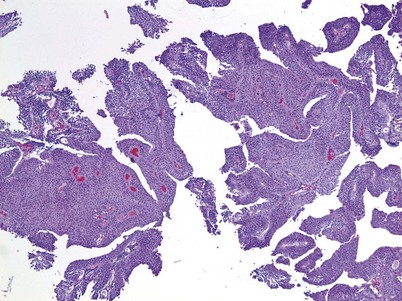
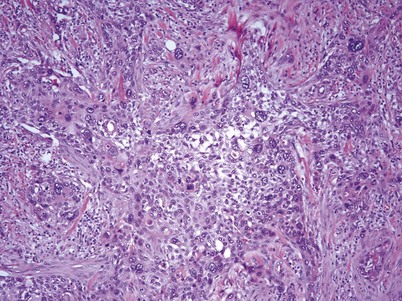
Urothelial papilloma is a benign proliferative growth in the bladder that is composed of delicate stalks lined by normal-appearing urothelium (Fig. 80–2) (Montironi and Lopez-Beltran, 2005). Papillomas had previously been categorized as grade 1 Ta tumors of the bladder until the World Health Organization (WHO) changed the classification of noninvasive bladder cancer in 1998 (Epstein et al, 1998). Papillomas rarely have mitotic figures and lack markers of aggressive growth such as TP53 or RB mutations, but 75% of these tumors will have mutations in the fibroblast growth factor receptor-3 (FGFR-3) (van Rhijn et al, 2004). Papillomas may recur, but they do not progress or invade.
Nephrogenic Adenoma
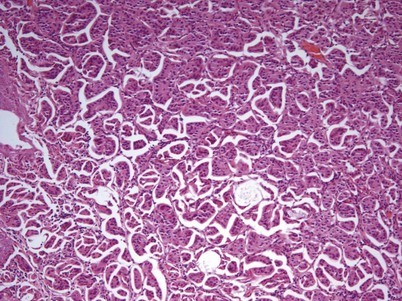
Nephrogenic adenoma is a rare tumor caused by chronic irritation of the urothelium; it arises from a variety of sources, including trauma, previous surgery, renal transplantation, intravesical chemotherapy, stones, catheters, and infection (Wood et al, 1988; Tse et al, 1997). Nephrogenic adenoma is composed of glandular-appearing tubules similar to renal tubules that involve the mucosa and submucosa of the bladder. These structures are covered by cuboidal cells with clear or eosinophilic cytoplasm with cytologically normal nuclei. The lesion may be vascular, which explains the presence of gross hematuria in most cases (Porcaro et al, 2001). There is no racial or gender association with the entity. The constant theme of chronic inflammation suggests metaplastic changes to the urothelium lead to nephrogenic adenoma, though some authors have proposed a theory that nephrogenic adenoma arises from nests of displaced mesonephric tissue in the urothelium that is activated with mucosal injury (Porcaro et al, 2001). The most frequent presenting symptom is gross hematuria, often in conjunction with a urinary tract infection. Treatment consists of transurethral resection and elimination of the chronic irritation.
Cystitis Cystica and Glandularis
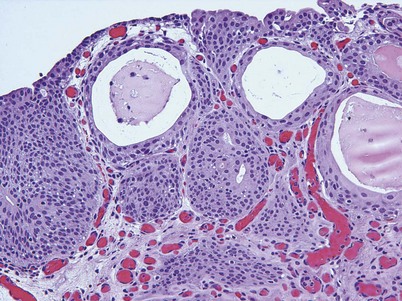
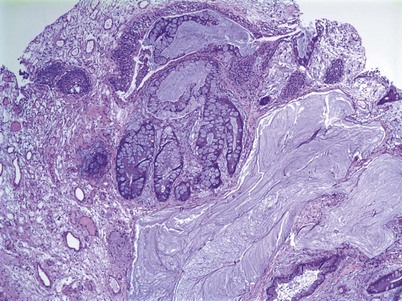
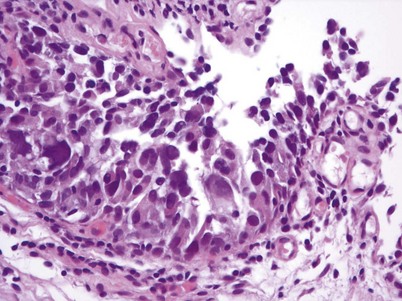
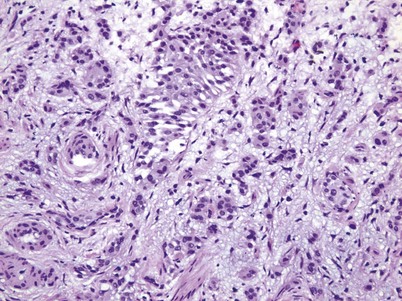
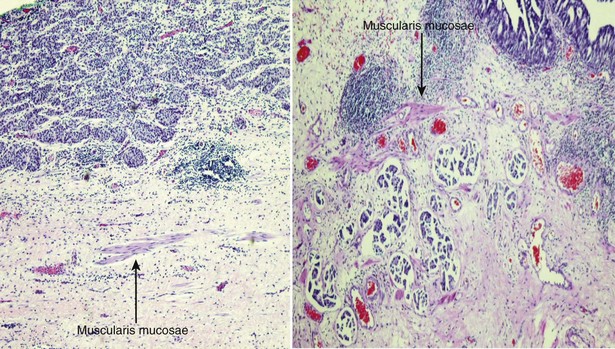
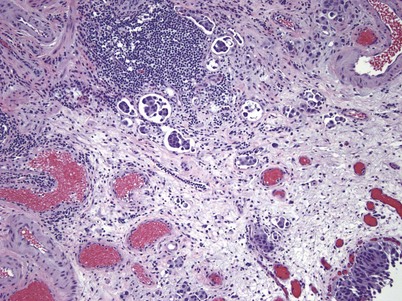
Cystitis cystica and/or glandularis is a common finding in normal bladders, usually associated with inflammation or chronic obstruction (Semins and Schoenberg, 2007). These benign tumors represent cystic nests that are lined by columnar or cuboidal cells and are generally associated with proliferation of Von Brunn nests (Figs. 80-3 and 80-4). Cystitis glandularis can be associated with pelvic lipomatosis and may occupy the majority of the bladder (Buckley et al, 2007). Cystitis glandularis may develop into or coexist with intestinal metaplasia, which are benign tumors characterized by goblet cells that are histologically similar to colonic epithelium. There have been a few case reports of cystitis cystica or glandularis transforming into adenocarcinoma, and therefore regular endoscopic evaluation of patients with these entities is recommended (Smith et al, 2008). The most common presenting feature of cystitis cystica or glandularis is irritative voiding symptoms and hematuria. Treatment is transurethral resection and relief of the obstruction or inflammatory condition.
Leiomyoma
Leiomyomas are the most common nonepithelial benign tumor of the bladder composed of benign smooth muscle. Several hundred cases have been reported, but this may under-represent the true prevalence. These tumors occur most commonly in women of childbearing age and are histologically similar to leiomyomas of the uterus (Castillo et al, 2008). Leiomyomas appear as smooth indentations of the bladder and can be confused with a bladder tumor except for the normal urothelium overlying the tumor (Fasih et al, 2008). Imaging, especially with magnetic resonance imaging (MRI), can confirm the diagnosis and spare invasive procedures (Fasih et al, 2008). Surgical resection is required if the leiomyoma is large or painful.
Urothelial Cancer
Epidemiology
The incidence rate of a cancer is defined as the number of new cancers diagnosed per 100,000 persons per year. The prevalence rate is the total number of cancers per 100,000 persons per year, not just new cases. Because urothelial cancer is a cancer of the environment and age, the incidence and prevalence rates increase with age, peaking in the 8th decade of life, and there is a strong association between environmental toxins and urothelial cancer formation (Jemal et al, 2008; Parkin, 2008). The incidence rate of urothelial cancer has been rising over the last 60 to 70 years, but the rate of rise has recently decreased significantly and in some geographic areas has leveled off (Parkin, 2008). Unfortunately, the incidence rate is rising the fastest in underdeveloped countries where industrialization has led to carcinogenic exposure.
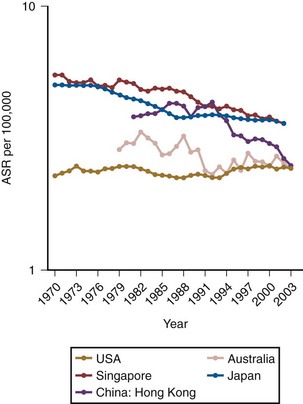
According to the latest American Cancer Society statistics, there were 68,810 total cases diagnosed in the United States in 2007, comprising 51,230 men and 17,580 women and accounting for 7% of all cancers (Jemal et al, 2008). Bladder cancer is a lethal disease with 14,100 deaths recorded in 2007, comprising 9950 men and 4150 women and accounting for 3% of all cancer deaths. Importantly, there has been a 5% decrease in bladder cancer mortality from 1990 to 2004 despite a continued rise in the incidence of the disease. Bladder cancer contributes to 0.7% of the absolute decrease in the cancer death rate seen during this time, when all cancers are considered (Jemal et al, 2008). Globally, the incidence rate of bladder cancer has been increasing, but due to smoking cessation programs, at a slower rate over the last decade (Parkin, 2008). The mortality rate from bladder cancer has decreased significantly from 1990 to 2004 with both men and women recording a 18.4% decrease in mortality rate (Fig. 80–5). This decrease in mortality rate is more striking in men than women because of the earlier peak when men begin to smoke, which occurred approximately 20 years before women. Because of the latency period with urothelial cancer–causing agents in tobacco, we should see a commensurate decrease in the mortality rate in women in 15 to 20 years as their smoking cessation programs become more widespread.
Gender/Racial/Age Differences
Males are 3 to 4 times more likely to develop bladder cancer than females, presumably because of an increased prevalence of smoking and exposure to environmental toxins (Jemal et al, 2008; Parkin, 2008). African-American males have a 19% higher incidence rate than white males for all cancers and a 37% higher death rate. However, for urothelial cancer, white males have a higher incidence and death rate than African-Americans (Jemal et al, 2008; Parkin, 2008). African-American women have a 6% lower incidence but a 17% higher death rate than white women in all cancers (Jemal et al, 2008). However, bladder cancer is roughly 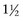 times more common in white women than in African-American women. A white male has a 3.7% chance of developing urothelial cancer in his lifetime, which is roughly 3 times the probability for white females or African-American males and more than
times more common in white women than in African-American women. A white male has a 3.7% chance of developing urothelial cancer in his lifetime, which is roughly 3 times the probability for white females or African-American males and more than 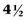 times the probability for African-American females (Hayat et al, 2007; Jemal et al, 2008). The risk of developing invasive bladder cancer is age dependant (Jemal et al, 2008). For men from birth to age 39 years, the incidence rate of invasive bladder cancer is 0.02%; ages 40 to 59 years, 0.41%; ages 60 to 69 years, 0.96%; ages 70 years and older, 3.5%; and from birth to death, 3.7%. The bladder cancer incidence for women from birth to age 39 years is 0.1%; ages 40 to 59 years, 0.13%; ages 60 to 69 years, 0.26%; ages 70 years and older, 0.99%; and from birth to death, 1.17%. In general, adolescents and young adults (less than age 40 years) tend to develop well-differentiated noninvasive, rather than invasive, bladder cancer (Linn et al, 1998). Unlike many other cancers where younger patients tend to develop more aggressive disease, the opposite appears to be true in bladder cancer, because they present more frequently with noninvasive low-grade tumors. However, the death rate, stage for stage, from bladder cancer is the same across all age groups (Wan and Grossman, 1989).
times the probability for African-American females (Hayat et al, 2007; Jemal et al, 2008). The risk of developing invasive bladder cancer is age dependant (Jemal et al, 2008). For men from birth to age 39 years, the incidence rate of invasive bladder cancer is 0.02%; ages 40 to 59 years, 0.41%; ages 60 to 69 years, 0.96%; ages 70 years and older, 3.5%; and from birth to death, 3.7%. The bladder cancer incidence for women from birth to age 39 years is 0.1%; ages 40 to 59 years, 0.13%; ages 60 to 69 years, 0.26%; ages 70 years and older, 0.99%; and from birth to death, 1.17%. In general, adolescents and young adults (less than age 40 years) tend to develop well-differentiated noninvasive, rather than invasive, bladder cancer (Linn et al, 1998). Unlike many other cancers where younger patients tend to develop more aggressive disease, the opposite appears to be true in bladder cancer, because they present more frequently with noninvasive low-grade tumors. However, the death rate, stage for stage, from bladder cancer is the same across all age groups (Wan and Grossman, 1989).
Global Burden of Bladder Cancer
There is a geographic difference in bladder cancer incidence rates across the world with the highest occurring in Southern and Eastern Europe parts of Africa, the Middle East, and North America and the lowest occurring in Asia and underdeveloped areas in Africa (Ferlay et al, 2007). Bladder cancer is the 9th most common cancer worldwide, with 357,000 cases recorded in 2002 (Parkin, 2008). Bladder cancer is the 13th most common cause of death, accounting for 145,000 deaths worldwide (Ferlay et al, 2007; Parkin, 2008). The incidence rate of bladder cancer has been rising in Asia and Russia because of an increased prevalence of smoking. Sixty-three percent of all bladder cancer cases occur in developed countries, with 55% from North America and Europe. In the United States, the highest bladder cancer incidence rate is in Rhode Island and the lowest is in the District of Columbia (Jemal et al, 2008). The histologic cell type of bladder cancer is very geographically dependent, but urothelial cancer is the most common. In North America and Europe, 95% to 97% of cases are urothelial carcinoma; in Africa 60% to 90% are urothelial and 10% to 40% are squamous cell; and Egypt has the highest rate of squamous cell carcinoma because of the endemic infections with Schistosoma species (Parkin, 2008).
Mortality
The mortality rate from bladder cancer in Egypt is 3 times higher than in Europe and 8 times greater than in North America because of the aggressive nature of squamous cell carcinoma that is highly prevalent in Egypt (Parekh et al, 2002). In the United States, death rates for all cancer sites combined decreased by 2.6% per year in males and by 1.8% in females from 2002 to 2004 compared with 1.5% and 0.8% per year in males and females, respectively, from 1992 to 2002 (Jemal et al, 2008). The mortality rate for bladder cancer has decreased by 5% during this period primarily because of smoking cessation, changes in environmental carcinogens, and healthier lifestyles. Mortality from bladder cancer is highest in elderly persons, particularly those over the age of 80 years, accounting for the third most common cause of cancer deaths in men older than age 80 years (Jemal et al, 2008). Whether this increase in mortality rate is related to tumor biology or changes in physician practices with the elderly is unclear. Recent evidence suggests that physician practices may be related to bladder cancer deaths in the elderly (Morris et al, 2009). These authors estimated that 31% of all bladder cancer deaths were avoidable, more commonly in noninvasive rather than invasive disease. Better chemotherapy has improved the survival rate in patients with metastatic bladder cancer, but changes in physician practices related to more timely care and more aggressive treatment in healthy patients could well lead to improvement in overall survival. Lee and colleagues (2006) reported that a delay of more than 12 weeks from the diagnosis of bladder cancer to cystectomy treatment was associated with a decrease in overall and cancer-specific survival. However, more intensive treatment (intravesical chemotherapy and cystoscopy) for patients with noninvasive bladder cancer did not correlate with the better survival or less need for invasive treatment (Hollenbeck et al, 2009; Morris et al, 2009).
Key Points: Epidemiology
Etiology
Bladder cancer is caused by genetic abnormalities and external risk factors, including carcinogen exposure, nutritional factors, fluid intake, alcohol, inflammation, infection, chemotherapy, radiation, and possibly artificial sweeteners.
Genetic
The higher incidence of bladder cancer in white compared with African-American males is probably not genetic but environmental, or it may be related to differential susceptibility to carcinogens (Bouchardy et al, 1995, 1996; Wanner et al, 1995). There are several polymorphisms that seem to be related to the formation of bladder cancer, in particular the susceptibility to environmental carcinogens. N-acetyl transferase (NAT) detoxifies nitrosamines, a known bladder carcinogen. Specifically, NAT-2 regulates the rate of acetylation of compounds such as caffeine, which are related to bladder cancer formation. The slow NAT-2 polymorphism is related to bladder cancer with an odds ratio of 1.4 compared with the fast polymorphism (Garcia-Closas et al, 2005). Glutathione-S-transferase (GSTM1) conjugates several reactive chemicals, including arylamines and nitrosamines. The null GSTM1 polymorphism is associated with an increased bladder risk with a relative risk of 1.5 (Garcia-Closas et al, 2005). The null GSTM1 and slow NAT-2 lead to high levels of 3-aminobiphenyl and higher risk of bladder cancer. These polymorphisms are present in 27% of white, 15% of African-American, and 3% of Asian males, thus partially explaining the different bladder cancer incidence rates across ethnic groups.
External Risk Factors
In addition to the skin and lungs, the bladder is the main internal organ affected by occupational carcinogens. The primary culprits are the aromatic amines that bind to DNA (Delclos and Lerner, 2008; Reulen et al, 2008). Twenty percent to 27% of all bladder cancers are associated with industrial exposure of some type, primarily in areas with a heavy concentration of chemical industries (Case and Hosker, 1954; Blot and Fraumeni, 1978; Reulen et al, 2008). Among the first chemical agents implicated in the formation of bladder cancer in dye and rubber workers were benzidine and β-naphthylamine (Case and Hosker, 1954). Activation of these amines to allow DNA binding occurs with enzymes that are selectively expressed in the population, making some subjects more susceptible to cancer formation as described above for the NAT-2 and GSTM1 polymorphisms. There are 11 specific aromatic amines implicated in bladder cancer formation; they are broken into three groups—definite, probable, and possible—as seen in Table 80–1. Other industrial agents implicated in bladder cancer formation include polycyclic aromatic hydrocarbons (PAH), diesel exhaust, and paint substances (Zeegers et al, 2001a).
Table 80–1 Aromatic Amines Associated with Urothelial Cancer Formation
| DEFINITE | PROBABLE | POSSIBLE |
|---|---|---|
| 4-Chloro-ortho-toluidine |
Data from Delclos GL, Lerner SP. Occupational risk factors. Scand J Urol Nephrol Suppl 2008;218:58–63; and IARC. Tobacco smoke and involuntary smoking. Lyon (France): IARC Press; 2004.
Environmental carcinogens can enter the system and cause bladder cancer from inhalation or through skin absorption. In general, there is a long latency period of 10 to 20 years between the industrial exposure and the formation of the bladder cancer, thus proving definitive causative relationships is difficult (Dryson et al, 2008). However, there are a variety of occupations statistically associated with bladder cancer formation, and all are industrial in nature (Table 80–2) (Reulen et al, 2008). The overall increased risk of bladder cancer formation in industrial workers is 30%, with agriculture workers having the lowest and rubber workers the highest risk of bladder cancer formation.
Table 80–2 Relative Risk of Occupations Associated with Urothelial Cancer Formation
| OCCUPATION | RR (95% CI) |
|---|---|
| Armed forces | 1.09 (0.94-1.26) |
| Managers | 1.17 (0.96-1.44) |
| Architects and engineers | 1.07 (0.91-1.25) |
| Health professionals | 1.02 (0.92-1.13) |
| Nurses | 1.07 (0.91-1.26) |
| Teaching professionals | 0.90 (0.74-0.09) |
| Writers and artists | 1.20 (1.05-1.36) |
| Clerks | 1.01 (0.92-1.10) |
| Cooks | 1.14 (0.86-1.50) |
| Hairdressers | 1.23 (1.11-1.37) |
| Protective service occupations | 1.07 (0.96-1.19) |
| Firefighters | 1.17 (0.92-1.49) |
| Police officers and guards | 1.10 (0.95-1.29) |
| Agricultural workers | 0.86 (0.79-0.93) |
| Gardeners | 1.04 (0.78-1.40) |
| Forestry workers | 0.93 (0.73-1.17) |
| Fishery workers | 0.95 (0.83-1.07) |
| Miners | 1.31 (1.09-1.57) |
| Building-frame workers | 1.06 (0.97-1.16) |
| Bricklayers | 1.03 (0.97-1.10) |
| Carpenters | 1.04 (0.90-1.21) |
| Building finishers | 1.10 (1.05-1.15) |
| Plumbers | 1.14 (1.03-1.25) |
| Electricians | 1.07 (1.02-1.13) |
| Painters | 1.17 (1.10-1.23) |
| Metalworkers | 1.10 (1.02-1.20) |
| Welders | 1.09 (0.98-1.20) |
| Sheet-metal workers | 1.14 (0.98-1.33) |
| Blacksmiths and toolmakers | 1.16 (1.06-1.26) |
| Blacksmiths | 1.27 (1.02-1.58) |
| Toolmakers | 1.10 (0.90-1.34) |
| Machine-tool setters | 1.24 (1.09-1.42) |
| Mechanics | 1.21 (1.12-1.31) |
| Motor mechanics | 1.27 (1.10-1.46) |
| Glassmakers | 1.31 (0.94-1.81) |
| Printers | 1.13 (0.96-1.33) |
| Food processors | 1.02 (0.93-1.11) |
| Butchers | 0.99 (0.78-1.26) |
| Bakers | 1.05 (0.87-1.26) |
| Cabinetmakers | 1.10 (0.94-1.27) |
| Textile workers | 1.12 (0.97-1.29) |
| Tailors | 1.28 (0.99-1.65) |
| Leather workers | 1.27 (1.07-1.49) |
| Machinist | 1.18 (1.06-1.30) |
| Metal processors | 1.18 (1.06-1.32) |
| Furnace operators | 1.17 (0.96-1.42) |
| Paper-pulp workers | 1.08 (0.90-1.29) |
| Petroleum workers | 1.15 (0.97-1.36) |
| Rubber workers | 1.29 (1.06-1.58) |
| Motor vehicle drivers | 1.11 (1.06-1.17) |
| Car, taxi, and van drivers | 1.20 (1.03-1.39) |
| Bus drivers | 1.29 (1.08-1.53) |
| Truck drivers | 1.18 (1.06-1.33) |
| Domestic helpers | 1.19 (1.01-1.42) |
| Cleaners | 1.06 (0.80-1.41) |
| Launderers | 1.27 (0.95-1.71) |
| Building caretakers | 1.17 (0.92-1.49) |
| Hand packers | 1.07 (0.93-1.23) |
| Freight handlers | 1.03 (0.96-1.11) |
| Dye makers | 1.10 (1.06-1.13) |
| Sales workers | 1.05 (0.98-1.13) |
| Woodworkers | 1.05 (0.89-1.24) |
| Chemical workers | 1.19 (0.98-1.43) |
CI, confidence interval; RR, relative risk.
From Reulen RC, Kellen E, Buntinx F, et al. A meta-analysis on the association between bladder cancer and occupation. Scand J Urol Nephrol Suppl 2008;218:64–78.
Smoking
Tobacco is the main known cause for urothelial cancer formation, particularly cigarette smoking, accounts for 60% and 30% of all urothelial cancers in males and females, respectively (Brennan et al, 2000, 2001; Boffetta, 2008; Gandini et al, 2008). The relative risk of developing urothelial cancer from smoking is 2.8 and 2.73 in men and women, respectively (Gandini et al, 2008). Overall there is a 2- to 6-times greater chance of developing urothelial cancer with smoking, and the intensity and duration of smoking is linearly related to the increased risk, with no clear plateau level (Brennan et al, 2000; Smoke, 2004; Boffetta, 2008). If a person smokes 1 to 9 cigarettes versus more than 21 cigarettes per day, the relative risk of bladder cancer is 1.5 versus 5.4, respectively (Weir and Dunn, 1970). If a person smokes 1 to 10 years versus more than 40 years, the relative risk of bladder cancer is 1.2 versus 3.0, respectively (Burns and Swanson, 1991). If a person smokes more than 60 years, they have a sixfold increased risk of developing urothelial cancer compared with a nonsmoker (Burch et al, 1989). The type of tobacco smoked appears to be associated with bladder cancer formation because of different carcinogens present within the tobacco. Black tobacco appears to be worse than blonde tobacco because of a greater amount of aromatic amines in the former (Boffetta, 2008). Cigars and pipes are probably associated with bladder cancer formation, but there are too few studies evaluating only cigar and pipe smokers because of the high probability that these subjects also smoke cigarettes. The risk of bladder cancer formation in those who chew tobacco is unclear. Again, most studies did not restrict their subjects to just chewing tobacco, but in general, there is an increased risk of bladder cancer formation associated with chewing tobacco. The risk of secondhand smoke in bladder cancer formation is low and not statistically different than that for nonsmokers (Zeegers et al, 2002). Importantly, smoking cessation does make a difference in urothelial cancer formation. Smokers who have stopped for 1 to 3 years have a 2.6 relative risk, and those stopping for more than 15 years have a 1.1 relative risk of bladder cancer formation (Wynder and Goldsmith, 1977; Smoke, 2004). Smoking is responsible for 30% of all deaths from bladder cancer in males and accounts for 46% of all bladder cancer deaths in high-income countries and 28% in low- to middle-income countries (Brennan et al, 2000; Parkin, 2008). The impact of smoking on mortality is more startling in women, in whom smoking is responsible for 13% of bladder cancer deaths, with 29% in high-income countries compared with only 3% in low- to middle-income countries.
Nutritional Factors
Most nutrients or other metabolites are excreted in the urine and have prolonged contact with the urothelium, particularly in the bladder; therefore nutrition plays a role in urothelial cancer formation (Steinmaus et al, 2000; Brinkman and Zeegers, 2008). However, there are inconsistent reports regarding the exact fruits and vegetables that are beneficial in preventing urothelial cancer, suggesting that epidemiologic factors are at play. In general, a Mediterranean diet leads to the lowest urothelial cancer risk. In a case-controlled study, there were fewer cases of urothelial cancer in the group given a Mediterranean diet versus a standard Western diet, probably because of the increased ingestion of fruits and vegetables (de Lorgeril et al, 1998). Both fruits and vegetables—specifically citrus, apples, berries, tomatoes, carrots, and cruciferous vegetables—contain several active compounds that are important in detoxification. These compounds include polyphenols, antioxidants, and enzymes that modulate the detoxification of nitrous amines and can prevent DNA adduct formation and oxidative damage (Boffetta, 2008; De Stefani et al, 2008; Lumbreras et al, 2008). Micronutrients associated with a preventive effect on urothelial cancer formation are mainly antioxidants, including vitamins A, C, and E; selenium; and zinc (Michaud et al, 2000; Zeegers et al, 2002; Schabath et al, 2005; Brinkman and Zeegers, 2008). Fish, rice, and cereal do not seem to have a protective or detrimental effect on urothelial cancer formation (Radosavljevic et al, 2005). Foods that have an indeterminate effect on bladder cancer formation include beef, eggs, and processed meats (Balbi et al, 2001; Wakai et al, 2004; Brinkman and Zeegers, 2008). Nutritional factors that have been associated with causing or promoting the formation of urothelial cancer include salted and barbequed meat, pork, total fat, pickled vegetables, soy, and spices (Balbi et al, 2001).
Occurrence of urothelial cancer is moderately higher in coffee and tea drinkers, but this may be compounded by smoking or other dietary factors associated with people who drink coffee or tea (Pelucchi et al, 2008). There is no apparent intensity or duration association of coffee and tea ingestion, suggesting an indirect causative effect, unlike for smoking. Coffee and tea do not appear to have any carcinogenic effect in animals; however, one study did show a twofold increase in urothelial cancer formation in nonsmokers who drank large amounts of coffee in western New York (Vena et al, 1993a). Conversely, a pooled analysis of 10 studies of nonsmokers related to coffee or tea ingestion showed no increased risk of cancer formation in coffee or tea drinkers (Sala et al, 2000). In conclusion, there are inconsistencies regarding nutritional factors related to urothelial cancer formation, in part because of confounding effects and associations, including coffee ingestion and smoking, ingestion of fruits and vegetables without involvement of smoking, and epidemiologic factors. However, even if not directly causative, there is a very clear association between a healthy diet and a decreased risk of urothelial cancer formation.
Fluid Intake
Urogenesis theory prescribes that decreased fluid intake leads to less micturition and higher concentrations of potential carcinogens in the urine and thus an increased risk of bladder cancer (Braver et al, 1987). A meta-analysis concluded that approximately 50% of the studies on fluid intake and bladder cancer risk showed an association and 50% did not find an association (Brinkman and Zeegers, 2008). Adding to the controversy, Vena and colleagues (1993b) reported a decreased risk of bladder cancer with increased fluid intake in women, but the opposite was true in men. In conclusion, although it makes sense that increased fluid intake would decrease the concentration of potential carcinogens and thus decrease the risk of bladder cancer, the scientific data supporting this theory is inconclusive.
Alcohol
Alcohol intake is related to several cancers, including those of the oral cavity, esophagus, liver, larynx, and breast, accounting for 3.6% of all cancers (Boffetta and Hashibe, 2006; Pelucchi et al, 2008). However, a meta-analysis of available literature from the last 20 years did not show an association between alcohol intake and bladder cancer, with a relative risk of 1.2 overall—1.3 in men and 1.0 in women (Zeegers et al, 2001b).
Artificial Sweeteners
Some animal studies have shown that large doses of saccharine or cyclamates may influence the development of bladder cancer (Allen et al, 1957; Sontag, 1980). These studies are controversial because of the high doses of saccharine and cyclamates provided to the animals and the altered composition of these compounds, which may have influenced the carcinogenic activity found in animal studies (Cohen et al, 1995). Epidemiologic studies in humans have shown no evidence of an increased risk of bladder cancer in consumers of artificial sweeteners (Armstrong and Doll, 1975; Morrison et al, 1982).
Analgesic Abuse
Acetaminophen is the active metabolite of phenacetin, a commonly used antipyretic and analgesic. Consumptions of large quantities of acetaminophen or phenacetin (5 to 15 kg during a 10-year period) have been associated with an increased risk of renal cancer and, perhaps, bladder cancer (Piper et al, 1985). However, these studies relied on interviews and questionnaires to ascertain drug exposure rather than actual determination of analgesic use. Kaye and colleagues (2001) performed a nested match case–controlled study and found no association between acetaminophen or other nonsteroidal anti-inflammatory drug ingestion and bladder cancer.
Inflammation/Infection
Infection is clearly a contributor to the formation of squamous cell carcinoma in patients chronically infected with Schistosoma hematobium and will be covered in the section on squamous cell carcinoma of the bladder (Abol-Enein, 2008). There is a possible link between human papillomavirus (HPV) and urothelial cancer formation. HPV encodes two oncoproteins, E6 and E7. E6 interacts with TP53, which has a role in bladder cancer progression and formation (Westenend et al, 2001). A meta-analysis supports a possible association between HPV infection and bladder cancer, reporting a 2.3 relative risk with confidence intervals of 1.3 to 4.1 (Gutierrez et al, 2006). However, the association between HPV and bladder cancer depends significantly on the method of analysis, the statistical evaluation of the data, proving infection status, and recall bias by the individual. The BK virus is oncogenic in newborn hamsters and can immortalize mammalian cells in vitro (Newton et al, 2005). BK virus can cause severe hemorrhagic cystitis in bone marrow transplant recipients; however, there is no consistent link between BK virus infection and urothelial carcinoma.
Bacterial
Several investigators have suggested that chronic bacterial infections may play a role in bladder cancer formation (Davis et al, 1991). Clinically, chronic catheter use, stones, and infections are associated with bladder carcinoma, but the mechanism of neoplastic formation is not well understood (Abol-Enein, 2008). The mechanism of action may be related to the production of carcinogens like nitrosamines that can be produced with chronic urinary tract infections (Radomski et al, 1978). In an animal model, rats with a urinary tract infection produce increased urinary levels of N1 N-dimethylnitrosamine over a 24-week period, and this was associated with urothelial hyperplasia and early neoplastic changes in the urothelium. The potential carcinogens were produced primarily with infections caused by Escherichia coli and Pseudomonas. A retrospective review of published literature suggests that chronic urinary tract infections are associated with bladder cancer, reporting a 14 to 16 relative risk of developing bladder cancer for any history of urinary tract infection versus none (Abol-Enein, 2008). However, there has been no prospective study examining the association between urinary tract infections and bladder cancer risk. It remains possible that the positive association between infection and urothelial cancer is driven by detection bias or preferential recall between cases and controls. Finally, a large case control study from the United States based on data from the National Bladder Cancer Study Group reported a 4.8 relative risk (CI, 1.9 to 11.5) of bladder cancer formation for subjects with greater than or equal to three urinary tract infections versus none (Kantor et al, 1984).
Previous gonorrhea infections have been associated with urothelial cancer formation. In a case-controlled study adjusting for age, gender, smoking, and other confounders, the relative risk of urothelial cancer formation was 2.1 to 2.4 (La Vecchia et al, 1991). A prospective study found a 1.92 relative risk (CI, 1.0 to 3.3) of bladder cancer formation in men with a history of gonorrhea (Michaud et al, 2007). Previous gonorrhea infection was most associated with invasive rather than noninvasive cancers. These studies suggest that gonorrhea infection may be associated with bladder cancer formation, and further prospective studies would be warranted.
Radiation
The potential association between radiation exposure and bladder cancer formation is primarily based on atomic bomb survivors during World War II (Ron et al, 1994; Thompson et al, 1994; Pierce et al, 1996; Hall, 2008). Since 1950, 86,572 people who were exposed to atomic bomb radiation have been followed. Seventy-three percent had a low dose of exposure (less than 50 mSv), and 6% had exposure to very high doses of radiation (more than 500 mSv). There is a significant increased risk of dying from any cancer if a person is exposed to greater than 50 mSv. For urothelial cancer, the relative risk of urothelial cancer formation is 1.63 in men and 1.74 in women. Interestingly, urothelial cancer formation after radiation is not age related, but the latency period is 15 to 30 years. Further support that radiation can cause bladder cancer is an increased risk of urothelial cancer in those patients with prostate or cervical cancer who were treated with radiation therapy (Boice et al, 1988; Neugut et al, 1997; Brenner et al, 2000). Even the radiation from more than six CT scans in a given year is associated with second malignancies (Hansen and Jurik, 2009). However, there is no association with low-dose or industrial exposure of radiation therapy and bladder cancer formation. Importantly, urologic technicians or nuclear radiation workers do not have an increased risk of urothelial cancer formation (Sont et al, 2001; Mohan et al, 2003; Sigurdson et al, 2003).
Chemotherapy
Chemotherapy destroys malignant cells by causing significant DNA and cellular damage, but can also have a profound effect on rapidly dividing normal epithelium such as in the bladder. The only chemotherapeutic agent that has been proven to cause bladder cancer is cyclophosphamide (Travis et al, 1995; Nilsson and Ullen, 2008). The risk of bladder cancer formation is linearly related to the duration and intensity of cyclophosphamide treatment, supporting a causative role. Phosphoramide mustard is the primary mutagenic metabolite that causes bladder cancer in patients exposed to cyclophosphamide.
Heredity
First-degree relatives of patients with bladder cancer have a twofold increased risk of developing urothelial cancer themselves, but high-risk of urothelial cancer families are relatively rare (Aben et al, 2002; Murta-Nascimento et al, 2007; Kiemeney, 2008). The hereditary risk seems to be higher for women and nonsmokers, but it is not related to secondhand exposure to smoking in families. The inherited risk of bladder cancer formation appears to affect all stages of urothelial carcinoma and is not associated with bladder cancer formation at an earlier age. Unfortunately, there are no clear mendelian inheritance patterns, making classic linkage studies impossible. Most likely, there are a variety of low-penetrance genes that can be inherited to make a person more susceptible to carcinogenic exposure, thus increasing the risk of bladder cancer formation. There are two high-penetrance genes that are associated with urothelial carcinoma: RB1 and CDC91L1 (Fletcher et al, 2004; Guo et al, 2004). In a recent study, there were five bladder cancer cases from 144 patients with known retinoblastoma, resulting in a relative risk of 26.3 compared with those without retinoblastoma (Fletcher et al, 2004). CDC91L1 was discovered in 1996 and is a germline translocation of t(5;20) (p15;q11) that is located on 20q11 (Guo et al, 2004). This gene encodes phosphatidylinositol glycan class U and is amplified in one third of urothelial carcinomas. The role of the gene is unclear, and the protein expressed is unknown at this time. Thelen and Schaeuble were the first to report familial clustering of urothelial carcinomas in 1957 by identifying identical twins with urothelial papillomas. This finding has led to further evaluations of looking for an inherited form of urothelial cancer, which to date has proven elusive.
Key Points: External Risk Factors
Pathology
Histologically, 90% of bladder cancers are of urothelial origin, 5% are squamous cell carcinomas, and less than 2% are adenocarcinoma or other variants (Lopez-Beltran, 2008). Urothelial carcinoma is the most common malignancy of the urinary tract and is the second most common cause of death among genitourinary tumors. At initial presentation, 80% of urothelial tumors are non–muscle invasive. There are multiple growth patterns of urothelial cancer, including flat carcinoma in situ (CIS), papillary tumors that can be low or high grade, and sessile tumors with a solid growth pattern. Non–muscle-invasive cancers can be very large because of lack of genetic alterations required for invasion. Likewise, invasive tumors can be quite small if early genetic changes occur within the tumor cell, allowing for an invasive phenotype.
The World Health Organization (WHO) first determined nomenclature for the histologic classification of urothelial cancer in 1973. In 1998, the International Society of Urological Pathology (ISUP) developed a new nomenclature to better reflect the recurrence and progression rates of urothelial cancer (Epstein et al, 1998). The two main changes were recognition that papillary Ta grade 1 urothelial cancers should not be considered cancers because of their indolent growth and lack of invasion, and the second was elimination of “grade 2” cancers, which became a grey zone encompassing grade 1 and grade 3 cancers causing interobserver variation. In 2004, the WHO adopted the ISUP recommended staging system and is the standard histologic nomenclature for urothelial carcinoma (Sauter et al, 2004). The clinical pattern of presentation is related to the cytologic and architectural alterations that occur within the tumor. Table 80–3 lists the histologic changes that occur from normal epithelium to high-grade muscle-invasive disease. Table 80–4 lists the neoplasms that can occur in the bladder, and Table 80–5 lists the WHO 2004 classification of urothelial neoplasms (Sauter et al, 2004; Montironi and Lopez-Beltran, 2005).
Table 80–3 Histologic Characteristics of Noninvasive Papillary Urothelial Tumors of the Bladder According to the WHO 2004 Classification
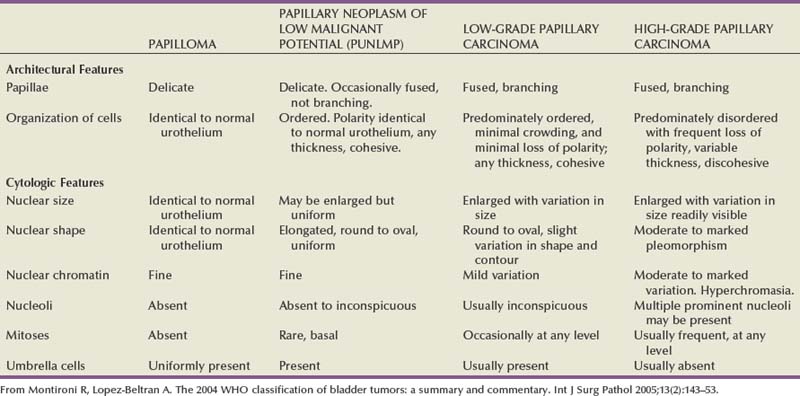
Table 80–4 Histologic Type of Tumors of the Urinary Bladder (World Health Organization, 2004)
|
Undifferentiated carcinomas*
|
NOS, not otherwise specified.
* Refers to tumors that are undifferentiated by light microscopy.
Lopez-Beltran A. Bladder cancer: clinical and pathological profile. Scand J Urol Nephrol Suppl 2008;218:95–109.
Table 80–5 2004 World Health Organization Classification of Noninvasive and Invasive Urothelial Neoplasia
| Noninvasive Urothelial Neoplasia |
| Invasive Urothelial Neoplasia |
From Montironi R, Lopez-Beltran A. The 2004 WHO classification of bladder tumors: a summary and commentary. Int J Surg Pathol 2005;13(2):143–53.
Precursor Lesions to Urothelial Cancer
Normal bladder urothelium is multilayered and less than seven cells thick (Epstein et al, 1998; Montironi and Lopez-Beltran, 2005) (Fig. 80–6). The cells mature from the basement membrane to the surface cells in an orderly fashion. The surface has large umbrella cells that may have nuclear atypia and form asymmetrical units. Their membrane is composed of uroplakin proteins and is rigid. These umbrella cells are part of the urine bladder barrier preventing toxins within the urine from transforming urothelial cells. Previously, normal urothelium was called “transitional cells” because of their ability to transition into other cell types, including squamous cells and adenocolumnar cells.
Precursor lesions are a continuum from hyperplasia to atypia to dysplasia and finally cancer. Different pathologists have their own interpretation of important cellular or structural changes that separate these histologic changes into different categories. Hyperplasia is characterized by markedly thicker mucosa with or without atypia (Fig. 80–7). The urothelium is more than seven cells thick, and there is some disorganization of the cellular architecture. Hyperplasia is often adjacent to low-grade tumors and thought to be a precursor lesion (Epstein et al, 1998). Loss of parts of chromosome 9 can occur in hyperplasia, particularly if adjacent to low-grade tumors (Hartmann et al, 1999). Urothelial hyperplasia appears as undulating or papillary growths that are best seen on flexible cystoscopy (Sauter et al, 2004). The presence of hyperplasia does not increase the risk of developing cancer; however, if present in someone with a history of urothelial carcinoma, it may herald a recurrence in the future (Montironi and Lopez-Beltran, 2005; Lopez-Beltran, 2008).
Reactive atypia can have nuclear changes associated with inflammation, regeneration, or reaction from noxious stimuli and is characterized by large cells with prominent nucleoli (Lopez-Beltran et al, 2002). Mitotic activity without abnormal forms can be present. Endoscopically, reactive atypia is edematous, especially in the lamina propria and associated with previous urinary instrumentation, stones, infection, or intravesical therapy (Lopez-Beltran, 2008).
Urothelial dysplasia has abnormal cytologic and nuclear changes that are preneoplastic but are not sufficient to be characterized as CIS (Sauter et al, 2004). Dysplasia is characterized by cohesive cells with mildly abnormal nuclear changes. There is nuclear crowding, prominent nucleoli, and abnormal mitotic figures may be present. Umbrella cells can be present with dysplasia, but not with CIS. Most cellular abnormalities are in the basal or early cell layers, with appropriate maturation toward the luminal surface. The urothelium is more than seven cells thick at times, but there can be sloughing of the more mature urothelium, exposing the less mature early cell layers. There is allelic loss of chromosome 9 and occasional TP53 abnormalities (Harnden et al, 1996; Hartmann et al, 1999). It is difficult to quantitate the clinical significance of dysplasia secondary to the interobserver reproducibility of this entity (Montironi and Lopez-Beltran, 2005). However, dysplasia is a good indication of urothelial instability and a marker of recurrence and progression in those with known urothelial cancer. Isolated dysplasia progressing to CIS occurs in approximately 19% of cases, but dysplasia in the face of previous history of urothelial cancer will form CIS in approximately 60% of cases (Cheng et al, 1999a).
Urothelial Cancer Histology
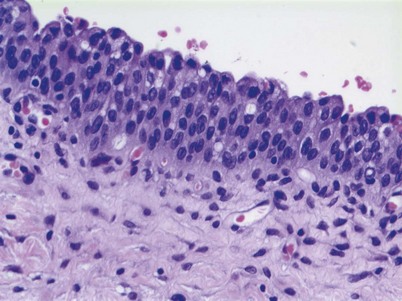
Non–muscle-invasive bladder cancer (NMIBC) includes CIS, papillary urothelial neoplasia of low-malignant potential (PUNLMP), and low- and high-grade urothelial cancer that previously had been called “superficial bladder cancer,” which is a misnomer. The clinical significance of the WHO grading classification is shown in Table 80–6. The grade distribution of NMIBC is 25% PUNLMP, 50% low grade, and 25% high grade (including CIS) (Holmang et al, 2001; Samaratunga et al, 2002). CIS is characterized as nonpapillary, flat, high-grade tumors in which the surface epithelium contains cancer cells (Fig. 80–8) (Sauter et al, 2004). There is severe nuclear atypia, loss of cellular polarity, and a noncohesive cellular structure. The cells are large, pleomorphic, chromatin clumping, and abnormal mitotic figures are common. Loss of umbrella cells is a characteristic, separating CIS from dysplasia. All CIS is high grade by definition. The genetic abnormalities associated with CIS include alterations to the RB, TP53, and PTEN genes (Cordon-Cardo et al, 2000; Lopez-Beltran et al, 2002; Cordon-Cardo, 2008). CIS is immunoreactive for cytokeratin 20, and NMP22 is present in the cells. CIS is a precursor lesion for invasive cancer and can spread to the distal ureters and prostatic urethra on the surface or in a pagetoid manner, undermining normal adjacent urothelium (Montironi et al, 2002). Endoscopically, CIS is reddish with heaped-up mucosa and can be mistaken for inflammatory changes or radiation cystitis. CIS in association with invasive tumors have a worse prognosis, with a 45% to 65% 5-year death rate (Lopez-Beltran et al, 2002).
Table 80–6 Clinical Significance of Different Non–Muscle-Invasive Urothelial Cancer Categories in WHO 2004 Grading System
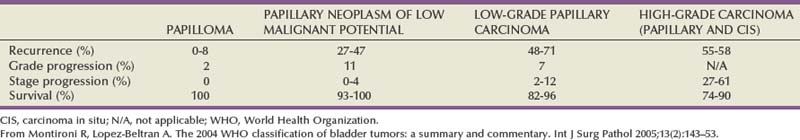
PUNLMP is a papillary growth with minimal cytological atypia that is more than seven cells thick and is generally solitary and located on the trigone (Fig. 80–9) (Holmang et al, 2001; Sauter et al, 2004). PUNLMP is composed of thin papillary stalks, where the polarity of the cells is maintained and the nuclei are minimally enlarged. PUNLMP has a low proliferation rate and is not associated with invasion or metastases, but almost 80% will have loss of chromosome 9 (Cheng et al, 2004). PUNLMP is different from a benign papilloma in that a PUNLMP has a thicker cell layer and large nuclei with occasionally mitotic figures. The male to female ratio for PUNLMP is 5 : 1, and the mean age is 65 (Holmang et al, 2001). PUNLMP can recur within the bladder in 35% of cases, but progression is rare, occurring in less than 4% (Oosterhuis et al, 2002).
Low-grade urothelial carcinoma is typically papillary in nature with a fibrovascular stalk and frequent papillary branching with increased cellular size, some nuclear atypia (more than in PUNLMP), and occasional mitotic figures (Fig. 80–10) (Epstein et al, 1998). Genetic abnormalities associated with low-grade cancer include deletion of 9q and alterations of FGFR-3, HRAS, and PI3K (Holmang et al, 2001; Cordon-Cardo, 2008). Low-grade carcinomas are immunoreactive for cytokeratin-20 and CD-44. The architectural and histologic changes that separate low-grade urothelial carcinoma from a PUNLMP include multiple stalks, more cytologic atypia, and the multifocal nature of low-grade carcinomas compared with the solitary PUNLMP.
High-grade papillary urothelial cancer is composed of fused papillary stalks with high-grade cancer in the urothelial layer. There is a disordered growth pattern, numerous mitotic figures present, and there are pleomorphic cells with exaggerated nuclei. Over 80% of high-grade cancers will invade the underlying stroma if left untreated. Genetic abnormalities associated with high-grade papillary urothelial cancer include similar abnormalities seen in the low-grade type plus alterations in the INK4A gene (Cordon-Cardo, 2008). In addition, there can be deletions of 2q, 5q, 10q, 18q, and gains of 5q and 20q (Knowles, 2008b). Alterations of TP21 and TP27 along with TP53 have been reported (Simon et al, 2004). High-grade urothelial carcinoma stains are immunoreactive for cytokeratin-20 and are likely aneuploid.
There are key genetic and phenotypic changes that occur in cancer cells, thus providing the ability to invade the underlying stroma. Invasive urothelial carcinoma is divided into two groups: lamina propria and deep muscle invasion. Lamina propria invasive tumors are high-grade cancers that can be in clusters or in single cells, with single-cell invasion having a worse prognosis (Fig. 80–11). Rarely, low-grade cancers can invade the lamina propria. Vascular invasion can occur within the lamina propria because of the large vascular network within this tissue layer; however, it is frequently overcalled because of retraction artifact around tumor nests. There is a subdivision of lamina propria invasion into T1a (invasion above muscularis mucosa) and T1b (invasion below the muscularis mucosa). This subject will be dealt with more extensively in the Staging section.
Muscle-invasive urothelial carcinoma is by definition high grade and is composed of cancerous cells extending through the lamina propria into the deep muscle bundles. There are rare cases of metastases to the deep muscle, but the overwhelming majority of cases are a direct extension from a surface urothelial carcinoma through the lamina propria into the deep muscle.
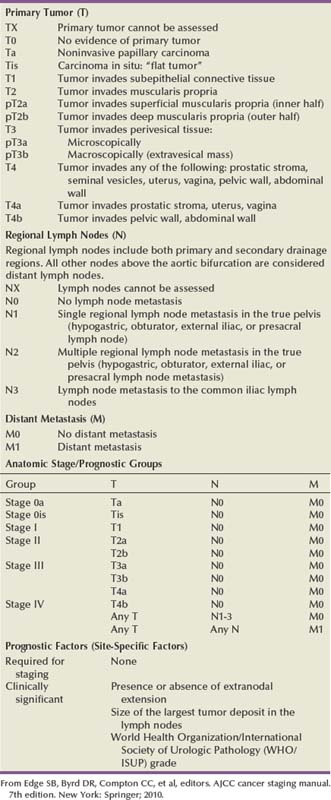
Staging
The American Joint Commission on Cancer in combination with the International Union Cancer Consortium meets on a regular basis to determine the tumor, nodes, and metastases (TNM) staging classifications. The 2009 staging system is shown in Table 80–7 (Edge et al, 2010). Ta and CIS disease have no invasion of the basement membrane, but endophytic growth of low-grade tumors into the lamina propria is possible, and cancer can occur in Von Brunn nests (Sung et al, 2006, Jones et al, 2007). T1 disease, as mentioned earlier, can be divided into T1a and T1b disease (Smits et al, 1998). The subdivision is based on the muscularis mucosa, which comprises thin wavy vesicles of muscle within the lamina propria that are associated with large vessels and lymphatics (Fig. 80–12). Some investigators substitute the presence of this large-vessel lymphatic layer for the muscularis mucosa when dividing T1a and T1b disease (Lopez-Beltran and Cheng, 2003). Unfortunately, muscularis mucosa is only identified in 15% to 80% of bladder biopsy specimens and in 90% of radical cystectomy specimens (Cheng et al, 1999; Sozen et al, 2002; Lopez-Beltran and Cheng, 2003; Lopez-Beltran, 2008). The prognostic significance of T1a and T1b disease is inconsistent because of the lack of muscularis mucosa in many bladder biopsy specimens. Essentially, the T1a and T1b stratifications suggest that the deeper the tumor invades in the lamina propria, the worse the survival.
Muscle-invasive disease is subdivided into T2a and T2b. T2a includes invasion into the inner half of the muscularis propria, while T2b is deeper into the outer half. Analysis of the American Joint Committee on Cancer (AJCC) data would suggest that there is a disease-free survival difference between T2a and T2b disease (Edge et al, 2010). On the contrary, Cheng and colleagues (1999b) showed that depth of muscle invasion was not predictive of disease-free survival, but the size of the tumor was the key element. T3 disease constitutes invasion outside the bladder proper into the periadipose tissue. T3a disease involves microscopic extension, while T3b involves macroscopic extension. Clinically, T3a disease is identified by a palpable mass at the time of examination under anesthesia during the initial transurethral resection and subsequently is nonpalpable after the tumor is resected. T3b disease has a persistent palpable mass after transurethral resection of the tumor. Pathologically, T3a disease is microscopic extension into the periadipose tissue, while T3b disease is macroscopic extension. T4a disease is invasion of the prostatic stroma, uterus, or vagina, and T4b disease is invasion of the pelvic wall or abdominal wall. Extension of the tumor into the prostatic urethra without stromal invasion is currently classified under the prostatic urethral section and does not carry an adverse prognosis for patients with known bladder cancer (Pagano et al, 1996; Edge et al, 2010). Prostatic stromal invasion, however, particularly if it is a direct extension from the bladder through the muscle into the prostate, does have a poor prognostic factor, with a 5-year overall survival of less than 25% (Esrig et al, 1996; Pagano et al, 1996).
A major problem in bladder cancer is understaging, occurring in 34% to 64% of cases. Chang and colleagues (2001) reported that 27% of T1 tumors were upstaged after radical cystectomy, and 49% of T2 tumors were upstaged to T3. Because of this understaging, the American Urological Association (AUA) guidelines call for a repeat transurethral resection in patients with T1 tumors to assess for muscle invasive disease even if muscle was present in the specimen (Hall et al, 2007). The significance of this restaging is shown in an article by Herr and colleagues (2007). Of 701 patients with T1 disease on the first transurethral resection, a second transurethral resection found 22% had no residual tumor, 23% had Ta or TIs disease, 25% had T1 disease, and 30% had muscle-invasive T2 cancer. If muscle was present in the original specimen, only 15% were upstaged to muscle-invasive disease, but if there was no muscle in the specimen, 40% were upstaged to T2 disease. This suggests that even if muscle is present in the initial specimen, an additional transurethral resection will detect muscle-invasive disease in up to 15% of cases.
Key Points: History and Staging
Origin, Recurrence, and Dissemination of Urothelial Cancer
Primary Tumors
The formation of primary urothelial carcinoma is a combination of environmental, genetic, and epigenetic causes. The main environmental factor associated with primary urothelial cancer is cigarette smoking, occurring in one third to one half of all bladder cancers in men and in 30% in women (Brennan et al, 2001; Boffetta, 2008). Smokers have a 2.77 relative risk of developing bladder cancer compared to nonsmokers (Gandini et al, 2008). Other external risk factors are listed above in the Etiology section. These external environmental factors then cause genetic and epigenetic instability that ultimately result in the formation of urothelial carcinoma. The deletion of parts or all of chromosome 9 is most likely the earliest mutation seen in low-malignant potential, non–muscle-invasive bladder cancer (Obermann et al, 2003). There is a known tumor suppressor gene at 9p21 that is a negative regulator of RB (Berggren et al, 2003). Another genetic abnormality mutated in 75% of low–malignant potential, non–muscle-invasive bladder cancer is FGFR-3 (Billerey et al, 2001; Gomez-Roman et al, 2005). High–malignant potential, non–muscle-invasive bladder cancer is more likely associated with deletions of tumor suppressor genes such as TP53 and RB (Chatterjee et al, 2004a; George et al, 2007; Sanchez-Carbayo et al, 2007). There are a large number of genes associated with stage progression because of the general genetic instability seen in high-grade tumors. Deletions of 3p, 5q, 6q, 11p, 16q, 18q, and TP53 with RB are all important for stage progression (Cordon-Cardo et al, 2000). It is the accumulation of these multiple genetic alterations, most often caused by external risk factors, that leads to the formation of urothelial cancer.
Recurrent Tumors
A hallmark of urothelial cancer is the high recurrence rate that approaches 80% for high–malignant potential, non–muscle-invasive bladder cancer. The two primary theories for recurrent tumor formation are field change effects within the bladder and tumor implantation. Using a cDNA expression library, genetic abnormalities seen in CIS can be found in endoscopically appearing normal urothelium away from the primary tumor (Dyrskjot et al, 2007). Similarly, alterations in certain urine markers that are associated with bladder cancer can be found in biopsy-negative bladders from patients with a history of urothelial carcinoma, suggesting that normal-appearing urothelium has the capacity to produce these various tumor markers (Keesee et al, 1996; Black et al, 2006). Because of these field-change genetic and epigenetic alterations, random bladder biopsies taken from patients after intravesical therapies for non–muscle-invasive bladder cancer or in those with positive urine cytology and yet endoscopically negative bladders are useful to detect incipient disease. Mufti and Singh (1992) evaluated the utility of random mucosal biopsies in the management of noninvasive bladder cancer and found 23% of normal-appearing bladder mucosa biopsies had dysplasia, CIS, or low–malignant potential papillary tumors (Mufti and Singh, 1992). Multifocality and rapidly recurring tumors are strong prognostic factors associated with finding histologically abnormal urothelium in endoscopically normal-appearing bladder tissue. In addition, the effectiveness of maintenance bacillus Calmette-Guérin (BCG) therapy in preventing tumor recurrence compared with an induction course only is supportive of field-change effects in the normal-appearing urothelium (Lamm et al, 2000). This suggests that treating the “nonvisible” tumors can prevent or delay the formation of visible cancer.
Tumor implantation during a transurethral resection of the bladder tumor has been suggested as a possible cause for recurrent tumor formation (Soloway and Masters, 1980; Pode et al, 1986). In experimental mouse models, 54% of transurethrally placed inoculates of tumor cells implanted successfully and grew in mice that underwent simultaneous bladder cauterization, in contrast with 12% of mice with an intact urothelial surface (Soloway and Masters, 1980). The immediate introduction of intravesical therapy, after the tumor inoculation and cauterization, significantly reduced the implantation rate. This finding has been confirmed by phase III clinical trials, showing that the immediate instillation of intravesical agents after transurethral resection of the bladder tumor can significantly decrease the risk of subsequent tumor recurrence (Kurth et al, 2000; Sylvester et al, 2004). However, for patients with a normal random bladder biopsy, the risk of subsequent tumor recurrence was significantly lower than those presenting with multiple tumors or single tumors with an abnormal random bladder biopsy (Mufti and Singh, 1992). The authors suggest that performing random bladder biopsies do not lead to an increased risk of bladder cancer formation; however, a strict control group was not used. Data from studies combining a transurethral resection of the prostate with a simultaneous transurethral resection of the bladder tumor did not find an increase risk of tumor implantation in the bladder neck or prostatic fossa (Ugurlu et al, 2007; Ham et al, 2009). Interestingly, Ham and colleagues (2009) showed a decreased risk of tumor recurrence in patients who had a simultaneous transurethral resection of the prostate and bladder tumor presumably because of a lower postvoid residual volume and, consequently, less urothelial exposure to carcinogens. It is unclear whether this was a cause and effect, and further studies are required to confirm this result.
Angiolymphatic Invasion
The key phenotypic change that occurs in urothelial cancer that is destined to metastasize is the ability to invade the angiolymphatic system, which is seen in approximately 25% of invasive urothelial carcinoma (Fig. 80–13) (Kunju et al, 2008). Angiolymphatic invasion is a poor prognostic sign with a 40% risk of nodal disease and is an independent predictor of overall and cancer specific survival (Abdel-Latif et al, 2004; Lotan et al, 2005). A transurethral resection of the bladder tumor can detect angiolymphatic invasion that is subsequently found in the radical cystectomy specimen 65% of the time. It is critical to use CD-31 and CD-34 monoclonal antibodies with immunohistochemistry to accurately identify the blood vessels in contrast to tumor retraction artifacts (Lotan et al, 2005; Kunju et al, 2008).
Pagetoid Spread
Pagetoid spread occurs when cancer cells grow underneath a layer of normal-appearing surface urothelium (Montironi et al, 2002). Pagetoid spread is primarily seen in urothelial CIS and was first described by Melicow and Hollowell in 1952. Detection of pagetoid spread is difficult because it occurs in approximately 15% of bladders that contain CIS and decreases to 11% in patients with papillary high–malignant potential, non–muscle-invasive bladder cancer (Orozco et al, 1993; McKenney et al, 2001). Pagetoid spread of urothelial cancer can occur into the prostatic urethra and distal ureters. This is more common after repeated doses of intravesical therapies, and therefore biopsies of normal-appearing prostatic urothelium are needed in the evaluation of patients with positive urine cytology and yet endoscopically normal bladder (Wood et al, 1989a, 1989b).
Direct Extension
Direct extension of tumors into the basal lamina, connective tissue, and, ultimately, the angiolymphatic system is caused by genetic and epigenetic changes that produce substances capable of invading these tissues. These substances include collagenases, motility and growth factors, and cell adhesion molecules. There are several collagenases associated with tumor invasion, primarily collagenase type IV (Bianco et al, 1998). Several members of the metalloproteinase (MMP) family are also associated with the invasive phenotype. MMPs are a family of proteases that chelate zinc and calcium. They are inhibited by tissue inhibitors of metalloproteinase called TIMPS. The ratio of MMP to TIMP is increased in invasive bladder cancer and associated with a poor clinical outcome (Bianco et al, 1998; Ozdemir et al, 1999). Urokinase plasminogen activator (UPA) is a serum protease that cleaves plasminogen to plasmin (Hanke et al, 2007). Plasmin degrades laminin, which is a major component of the basal lamina layer. High expression of UPA is seen invasive urothelial cancer and is an independent predictor of survival (Hasui and Osada, 1997).
There are numerous motility and growth factors present within the extracellular matrix that increase tumor growth. Proepithelin may play a critical role as an autocrine growth factor in the establishment and progression of bladder cancer, and studies suggest that proepithelin may be a novel biomarker for the diagnosis and prognosis of bladder neoplasms (Lovat et al, 2009). The ability of cancer cells to migrate and invade through the extracellular matrix is a critical step for tumor metastasis. Other growth factors associated with bladder cancer invasion include epidermal growth factors (EGF), transforming growth factor-α, heparin-binding growth factor, and insulin-like growth factor (Brown et al, 1993; Theodorescu et al, 1998). Cell adhesion molecules are critical for integrity of cell–cell junctions and the inhibition of cell growth. Cell adhesion molecules associated with invasive urothelial carcinoma include E-cadherin, integrins, CD-44 and NCD-44 (Kashibuchi et al, 2007). Decreased levels of these cell adhesion molecules are seen in invasive tumors and are related to cancer specific survival. A combination of genetic and epigenetic changes causing increased production of substances to invade underlying stromal tissue in combination with a lack of cell–cell binding and increased cellular growth all lead to the invasive phenotype and subsequent metastasis.
Prognostic Factors
There are genetic, pathologic, and phenotypic changes in bladder cancer that are characteristic of poor cancer-specific survival. Overall genetic instability is the hallmark of invasive urothelial cancer, but specifically alterations of TP53, RB, and PTEN carry a very poor prognosis (Chatterjee et al, 2004a). Wang and colleagues (2009) developed a gene expression signature that could accurately segregate poor- and good-risk noninvasive and invasive bladder cancers. They were able to make the segregation even within similar pathologically staged tumors. However, despite the major advances in understanding the genetics of urothelial carcinoma, the stage and grade of the primary tumor is still the strongest predictor of survival. Grade is indicative of the growth potential of the cell and stage describes the extent of the cancer and the ability to invade. The ability of high-grade tumors to invade and thus metastasize is due to micrometastatic disease from angiolymphatic invasion. As mentioned above, there are numerous genetic and epigenetic causes for the invasive phenotype, but because of genetic instability, it is difficult to determine a true cause and effect for any given genetic change. New markers of proliferation and invasion are being developed that can better define the invasive phenotype. Proliferation markers, such as MIB-1 and PCNA, are found in high-grade tumors and are associated with worse prognosis (Lopez-Beltran et al, 2004). Alteration in cell cycle regulators, such as cyclins, TP53, and TP27, leads to increased proliferation as seen by MIB-1 staining. Ultimately, it will be the integration of stage, grade, and molecular markers that will greatly improve the prognostic determination of urothelial carcinomas and hopefully provide new therapeutic targets.
Key Points: Origin, Recurrence, and Invasion
Molecular Biology
Cancer is abnormal, unregulated, and excessive cell growth occurring after an initial transforming event that eventually results in cellular changes that, in turn, allow widespread dissemination. The abnormal inciting events are a combination of somatic and hereditary genetic changes that result in genetic instability characteristic of cancer formation (Vogelstein and Kinzler, 2004). Somatic mutations are more common than germline mutations, and if germline mutations occur, they are associated with a specific type of cancer such as von Hippel-Lindau disease. These somatic and germline genetic changes result in an altered phenotype that can be enhanced or, at times, be caused by epigenetic alterations, such as promoter methylation or protein degradation, that suppress gene function involved in bladder cancer formation (Wolff et al, 2005).
Genes associated with cancer formation are divided into two groups: proto-oncogenes and tumor suppressor genes. Proto-oncogenes are generally activated by point mutations in the genetic code, gene amplification, and gene translocation. The activated proto-oncogenes become oncogenes that can cause cancer and are considered a positive or dominant growth effect (Lengauer et al, 1998; Wolff et al, 2005; Cordon-Cardo, 2008). Tumor suppressor genes are mainly activated by allelic deletions followed by point mutations of the remaining allele. Tumor suppressor genes are recessive or have a negative effect resulting in unregulated cellular growth. Both oncogene and tumor suppressor gene activation occur with the same frequency. The key to cancer formation is the accumulation of the genetic events that ultimately cause enough phenotypic changes to result in unregulated cell growth and invasion (Lengauer et al, 1998; Vogelstein and Kinzler, 2004).
There are specific genetic changes that occur between each stage of urothelial tumor development (Cordon-Cardo et al, 2000; Simon et al, 2004; Cordon-Cardo, 2008). Traditionally, there are two pathways in urothelial cancer formation: normal urothelium to low-grade noninvasive disease and normal urothelium to CIS and subsequent muscle-invasive disease. A proposed third pathway involves normal urothelium to hyperplasia/dysplasia to high-grade papillary carcinoma and subsequent muscle-invasive disease (Fig. 80–14). Low-grade papillary carcinoma with additional genetic alterations can develop into high-grade papillary disease and subsequent muscle-invasive disease, but it is rare for high-grade tumors to mutate into low-grade cancers. In general, low-grade papillary tumors have genomic stability that allows tumor recurrence but rarely progression. High-grade papillary cancer and CIS have unstable genomes that more readily allow additional genetic alterations needed for muscle-invasive or metastatic disease (Spruck et al, 1994; Knowles, 2006; Lindgren et al, 2006).
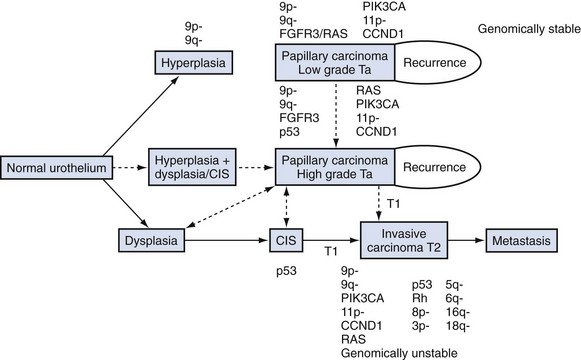
Figure 80–14 Pathogenesis of urothelial cancer formation.
(From Knowles MA. Bladder cancer subtypes defined by genomic alterations. Scand J Urol Nephrol Suppl 2008;218:116–30.)
The genetic alterations that are the hallmark of low-grade non–muscle-invasive disease are alterations in the fibroblast growth factor receptor–3 (FGFR-3) and deletions of chromosome regions 9p and 9q. High-grade invasive disease has infrequent FGFR-3 mutations but a high rate of TP53 mutations that approaches 60%. Noninvasive tumors that have both FGFR-3 and TP53 mutations are rare and constitute the third pathway to the formation of invasive disease by forming papillary rather than sessile T1 cancer that is derived from CIS (Knowles, 2006; van der Kwast, 2008). Genetic changes that occur in noninvasive and invasive bladder cancers are listed in Tables 80-8 and 80-9, respectively.
Table 80–8 Genetic Changes Identified in Ta Bladder Tumors
| GENE (CYTOGENETIC LOCATION) | ALTERATION | FREQUENCY |
|---|---|---|
| Oncogenes | ||
| HRAS (11p15)/NRAS | Activating mutations | 15% |
| (1p13)/KRAS2 (12p12) | ||
| FGFR-3 (4p16) | Activating mutations | 60%-80% |
| CCND1 (11q13) | Amplification/overexpression | 10%-20% |
| PIK3CA (3q26) | Activating mutations | 25% PUNLMP; 16% Ta |
| MDM2 (12q13) | Overexpression | ≈30% overexpression |
| Tumor Suppressor Genes | ||
| CDKN2A (9P21) | Homozygous deletion/methylation/mutation | HD 20%-30%; LOH ≈60% |
| PTCH (9q22) | Deletion/mutation | LOH ≈60%; mutation frequency low |
| DBCI (9q32-33) | Deletion/methylation | LOH ≈60% |
| TSCI (9q34) | Deletion/mutation | LOH ≈60%; mutation ≈12% |
| DNA Copy Number Changes; Target Gene(S) Unkown* | ||
| 2q | Deletion | 10% |
| 8p | Deletion | 16% |
| 9p | Deletion | 36%-47% |
| 9q | Deletion | 44%-66% |
| 10p | Deletion | 20% |
| 10q | Deletion | 20% |
| 11p | Deletion/LOH | 10%-24% |
| 13q | Deletion | 17% |
| 17q | Deletion | 15% |
| 18q | Deletion | 13% |
| Y | Deletion | 24%-28% |
| 1q | Gain | 11%-14% |
| 17q | Gain | 14% |
| 20q | Gain | 13%-17% |
| 8p12 | Amplification | Occasional |
| 11q13 (including CCND1) | Amplification | Occasional |
HD, homozygously deleted; LOH, loss of heterozygosity; PUNLMP, papillary urothelial neoplasm of low malignant potential.
* Comparative genomic hybridization analyses.
From Knowles MA. Bladder cancer subtypes defined by genomic alterations. Scand J Urol Nephrol Suppl 2008;218:116–30.
Table 80–9 Genetic Changes Found in Invasive (≥T2) Bladder Tumors
| GENE (CYTOGENETIC LOCATION) | ALTERATION | FREQUENCY |
|---|---|---|
| Oncogenes | ||
| HRAS (11p15)/NRAS | Activating mutations | 10%-15% |
| (1p13)/KRAS2 (12p12) | ||
| FGFR-3 (4p16) | Activating mutations | 0%-34% |
| ERBB2 (17q) | Amplification/overexpression | 10%-14% amplification |
| CCND1 (11q13) | Amplification/overexpression | 10%-20% |
| MDM2 (12q13) | Amplification/overexpression | 4% amplification |
| E2F3 (6p22) | Amplification/overexpression | 9%-11% amplification in ≥T1 |
| Tumor Suppressor Genes | ||
| CDKN2A (9P21) | Homozygous deletion/methylation/mutation | HD 20%-30%; LOH ≈60% |
| PTCH (9q22) | Deletion/mutation | LOH ≈60%; mutation frequency low |
| DBCI (9q32-33) | Deletion/methylation | LOH ≈60% |
| TSCI (9q34) | Deletion/mutation | LOH ≈60%; mutation ≈12% |
| PTEN (10q23) | Homozygous deletion/mutation | LOH 30%-35%; mutation 17% |
| RB1 (13q14) | Deletion | 37% |
| TP53 (17p13) | Deletion/mutation | 70% |
| DNA Copy Number Changes; Target Gene(S) Unkown* | ||
| 2q | Deletion | 12% |
| 5q | Deletion | 15%-24% |
| 6q | Deletion | 15%-28% |
| 8p | Deletion | 29%-34% |
| 9p | Deletion | 21%-30% |
| 9q | Deletion | 17% |
| 10q | Deletion | 16%-21% |
| 11p | Deletion | 18%-24% |
| 11q | Deletion | 22% |
| 13q | Deletion | 19% |
| 15q | Deletion | 13% |
| 16q | Deletion | 15% |
| 17q | Deletion | 17%-24% |
| 18q | Deletion | 16%-17% |
| Y | Deletion | 21% |
| 1q | Gain | 17%-33% |
| 3q | Gain | 18% |
| 5p | Gain | 24%-37% |
| 7p | Gain | 20% |
| 8q | Gain | 23%-34% |
| 10p | Gain | 12% |
| 17q | Gain | 30% |
| 20p | Gain | 21% |
| 20q | Gain | 26%-28% |
| 1q22 | Amplification | <5% |
| 3p24 | Amplification | <5% |
| 6p22 | Amplification | 5%-10% |
| 8p12 | Amplification | <5% |
| 8q21-22 and q24 | Amplification | <5% |
| 10p13-14 | Amplification | <5% |
| 12q15 | Amplification | <5% |
| 17q21 | Amplification | <5% |
| 20q13 | Amplification | <5% |
HD, homozygously deleted; LOH, loss of heterozygosity.
* Comparative genomic hybridization analyses.
From Knowles MA. Bladder cancer subtypes defined by genomic alterations. Scand J Urol Nephrol Suppl 2008;218:116–30.
Normal urothelium transforms into low-grade papillary cancer through activation of proto-oncogenes, resulting in phenotypic changes that are histologically named papilloma, papillary urothelial neoplasia of low-malignant potential (PUNLMP), hyperplasia, and low-grade urothelial cancer. Papillomas lack genetic alterations and have no FGFR-3 mutations, thus distinguishing this growth pattern from low-grade urothelial carcinoma and therefore are probably not a precursor lesion to cancer (Knowles, 2006). Conversely, PUNLMP does contain the same genetic abnormalities seen in low-grade urothelial carcinoma, has a proliferation rate similar to low-grade carcinoma, and is likely to be a precursor to low-grade bladder cancer. Thus there are no clear DNA markers that can distinguish PUNLMP from low-grade cancer (Dyrskjot et al, 2007). Urothelial hyperplasia is considered a precursor of low-grade carcinoma, and the most frequent genetic deletion is of chromosome 9 and is most likely the earliest mutation seen in low-grade urothelial cancer formation (Obermann et al, 2003). There are a variety of tumor suppressor genes present on both regions 9p and 9q. At 9p21, CDKN2A codes for TP16 (INK4A), which is a negative regulator of RB, and TP14ARF, which is a negative regulator of TP53 (Berggren et al, 2003). In addition, CDKN2B is located at 9p21 and codes for TP15. It is less clear which genes located on 9q are deleted. PTCH is located at 9q22, and DBC1 and TSC1 are located on 9q33-34 (Aboulkassim et al, 2003; Adachi et al, 2003). The multiple gene losses on chromosome 9 cumulatively lead to the formation of low-grade urothelial neoplasia. It is rare in low-grade papillary cancers to have markers of aggressiveness, such as loss of chromosome 17p, 2q, 4, or 11p (Cordon-Cardo, 2008). Urothelial hyperplastic lesions associated with papillary low-grade urothelial carcinoma in the same bladder have the same alterations in the majority of cases (Obermann et al, 2003; van Oers et al, 2005). This suggests that some papillary hyperplastic lesions have the genetic abnormalities to form low-grade carcinomas, while others that are histologically similar do not and instead stay as hyperplastic lesions even if they are near a papillary tumor.
FGFR-3 is related to EGFR, which is not mutated in low-grade cancers but can be overexpressed in low- or high-grade urothelial cancer. FGFR-3 is a tyrosine kinase that causes enhanced growth of bladder cells and is altered in up to 75% of noninvasive low-grade tumors (Gomez-Roman et al, 2005). Altered FGFR-3 can be found in CIS and muscle-invasive disease, although to a much lesser extent, supporting the overlapping nature of tumor formation with multiple genetic changes destined to make either noninvasive or invasive disease. Interestingly, FGFR-3 mutations are also found in seborrheic keratosis, which is a benign papillary skin wart that can be multiple, recur, but does not invade (Logie et al, 2005). FGFR-3 mRNA is overexpressed eightfold in noninvasive disease and up to fourfold in muscle-invasive disease. Normal urothelium does not produce the FGFR-3 protein, but over 71% of noninvasive bladder cancer will be immunopositive for the FGFR-3 receptor (Billerey et al, 2001). Because of the differential FGFR-3 expression between normal and cancerous tissue, it could be a therapeutic target (Knowles, 2008c). FGRF-3 and HRAS mutations are mutually exclusive, because they are in the same RAS-MEK-ERK pathway. Mutations of FGFR-3 cause constitutive activation of the FGFR-3 receptor and thus signaling through the MAPK pathway (Cordon-Cardo, 2008; Knowles, 2008c). FGFR-3 and TP53 mutations are virtually mutually exclusive as well, but not because of a common signaling pathway. It is unclear why these two mutations are mutually exclusive, but it does highlight the different pathways in the formation of urothelial carcinoma (Bakkar et al, 2003).
RAS genes are from a family of transforming oncogenes that were originally identified in T24 bladder cancer cell lines (McBride et al, 1982). The most frequent site of HRAS activations are mutations in codons 12, 13, 59, and 61 that result in increased enzymatic activity. HRAS mutations occur in 40% of urothelial carcinoma, and mutations in codon 12 are the most common (Czerniak et al, 1992). Point mutations in PIK3CA are found in 10% of noninvasive bladder cancer and are critical in the PTEN pathway (Cairns et al, 1998). FGFR-3 and PIK3CA work together in a similar pathway, and thus both mutations are often seen in the same tumor. Deletions on 9q activate the tumor suppressor genes TSCI, PTCH, and DBC1 (Hornigold et al, 1999; Louhelainen et al, 2006). TSCI is a major negative regulator of the PTEN signaling pathway, encodes hamartian, is located on 9q34, and is mutated in 10% of noninvasive bladder cancer. TSCI controls the activity of RHEB, which in turn regulates MTOR and the downstream events of the pathway. Deletion of TSC1 activates the oncogenic potential of the signaling network. Deletions of PTCH located on 9q22 and loss of heterozygosity is seen in 40% of noninvasive tumors and cause increased cellular proliferation (McGarvey et al, 1998). EBC1 is located on 9q33 and is mutated in 50% of noninvasive bladder cancer and also causes increased cellular proliferation (Louhelainen et al, 2006).
The transformation of normal urothelium to high-grade invasive bladder cancer is a continuum from dysplasia to CIS and then invasive disease. The conversion of normal urothelium to dysplasia is associated with chromosome 9 deletions in 75% of cases, abnormal TP53 accumulation in 50%, and increased cellular growth in all cases (Mallofre et al, 2003). The subsequent genetic changes to CIS are most likely deletions of tumor suppressor genes rather than activation of oncogenes, and alterations of TP53 are the main hallmark of high-grade disease (George et al, 2007; Sanchez-Carbayo et al, 2007). TP53 is located on chromosome 17p and controls the expression of multiple apoptosis-related genes. The presence of TP53 nuclear protein overexpression and TP53 gene mutations occur in approximately 80% of high-grade tumors, including CIS. Primary CIS, which is defined as CIS not associated with a papillary or invasive lesion, has TP53 overexpression but lacks deletion of chromosome 9. This is in contrast to secondary CIS, which is associated with papillary lesions, can display alterations in chromosome 9, and has molecular expression profiles similar to the adjacent papillary tumor, thus highlighting the overlapping pathways to invasive disease from flat CIS or the hyperplasia to papillary high-grade disease, and then subsequent muscle-invasive disease (Hopman et al, 2002). Also, the same expression signature that exists for secondary CIS can be found in biopsies of morphologically normal urothelium in the same bladder (Dyrskjot et al, 2004). The retinoblastoma gene (RB) encodes for a 110 kD nuclear phosphoprotein that functions as a negative cell cycle regulator (Chatterjee et al, 2004a, 2004b). Undetectable levels of the RB protein are associated with poor prognoses and increased tumor growth, presumably by affecting the E2F gene pathway. Decreased PTEN expression is seen in advanced urothelial cancers and CIS. PTEN genetic alterations are an independent predictor of survival in advanced cancer, and altered PTEN and TP53 mutations are associated with aggressive tumor growth (Puzio-Kuter et al, 2009).
The genetic changes that occur between noninvasive and invasive high-grade lesions are essentially genes needed to invade rather than to grow. The earliest changes seen in T1 disease are deletions of 3p, 5q, 6q, 11p, 16q, and 18q (Cordon-Cardo, 2008). Alterations in the TP53, RB, and PTEN pathways are hallmarks of invasive disease, and the combined alterations of all three pathways carries a much worse prognosis (Chatterjee et al, 2004a, 2004b). Finally, the general genetic instability seen in muscle-invasive disease to metastatic disease makes identification of specific genes associated with this progression difficult to clearly delineate (Knowles, 2008a).
Many investigators are using the genetic changes identified in urothelial cancer to determine the malignant potential of small noninvasive cancers by using molecular staging (van der Kwast, 2008). Through the formation of cDNA expression libraries, 80% of Ta urothelial cancers are correctly staged by their genetic profile. Of the 20% of Ta tumors that were wrongly classified as T1 or T2 by genetic profiling, most had a significantly worse prognosis than the correctly staged Ta tumors (Dyrskjot et al, 2003, 2007; Blaveri et al, 2005). A 16-gene expression chip of CIS has been developed to discriminate CIS from normal bladder urothelium, with an 80% sensitivity and a 68% specificity. Unfortunately, gene abnormalities for CIS can be found in normal urothelium, confirming the field-chain effect seen in bladder cancer (Dyrskjot et al, 2007). The main genetic changes that separate noninvasive from invasive disease, as mentioned above, include high FGFR-3 and low TP53 mutation rates and general genetic stability. T1 disease has low FGFR3 and high TP53 mutation rates and loss of 17p, 13q, and 8p (Knowles, 2008a). Many of these genetic changes are not necessarily related to invasive potential but, instead, reflect the different grade of tumor seen in Ta and T1 disease. The separation of T1 and T2 disease by genetic analysis is more difficult because of the overall worsening genetic instability and because both are high-grade tumors. However, T2 tumors have more frequent allelic imbalance of chromosomes 6, 10p, and 22 (Koed et al, 2005). Finally, response to chemotherapy in T2 tumors may be predicted by cDNA profiling of the emmprin and survivin genes, although further prospective studies are required (Als et al, 2007). The accumulation of genetic changes in cell cycle regulation (TP53), angiogenesis (NRP2), and metastases suppressors (RhoGDI2) eventually leads to the highly malignant phenotype of high-grade urothelial cancer (Aaboe et al, 2006). On the horizon, these genetic signatures of aggressive disease will be used for prognostic and therapeutic intent. Wang and colleagues (2009) developed a multiplex quantitative polymerase chain reaction (PCR) assay that accurately segregated Ta, T1, and T2 bladder cancer into high- and low-risk groups. Using a 57-gene mRNA expression profile, at 2 years, the high-risk groups had greater stage progression in each pathologic group. A prospective multi-institutional study is needed to confirm these results but may herald a new staging method.
Detection of Urothelial Carcinoma
Gross, painless hematuria is the primary symptom in 85% of patients with a newly diagnosed bladder tumor, and microscopic hematuria occurs in virtually all patients (Khadra et al, 2000; Alishahi et al, 2002; Edwards et al, 2006). The hematuria is usually intermittent and can be related to Valsalva maneuvers; therefore any episode of gross hematuria should be evaluated even if subsequent urinalysis is negative. Fifty percent of patients with gross hematuria will have a demonstrable cause, 20% will have a urologic malignancy, and 12% will have a bladder tumor (Khadra et al, 2000). The risk of malignancy in patients with recurrent gross or microscopic hematuria who had a full, negative evaluation is nearly zero within the first 6 years (Khadra et al, 2000). This should be considered when recommending repeat evaluations for patients with recurrent hematuria.
A full hematuria evaluation for bladder cancer includes cystoscopy, urine cytology, upper-tract imaging (primarily a CT scan of the abdomen and pelvis), and a prostate-specific antigen (PSA) blood test. A PSA blood test is recommended, because 10% of patients with recurrent gross hematuria will have prostate cancer (Mishriki et al, 2009). Microscopic hematuria is typically asymptomatic and carries a 5.4% risk of urologic malignancy and a 4.1% risk of bladder cancer (Mishriki et al, 2008). For patients with a negative microscopic hematuria evaluation, 84.5% never had a recurrence, and of those with repeat microscopic hematuria, none had a urologic malignancy with a 13-year follow-up (Mishriki et al, 2008). The AUA guidelines for microscopic hematuria evaluation include a cystoscopy, upper tract imaging, and urine cytology (Grossfeld et al, 2001). The guidelines recommend consideration for re-evaluation of low-risk patients with microscopic hematuria, but repeat evaluation every 6 months with urinalysis, cytology, and blood pressure (to detect renal disease) is recommended for high-risk patients (Table 80–10).
Table 80–10 High-Risk Characteristics of Patients with Microscopic Hematuria
From Grossfeld GD, Litwin MS, Wolf JS Jr, et al. Evaluation of asymptomatic microscopic hematuria in adults: the American Urological Association best practice policy—part II: patient evaluation, cytology, voided markers, imaging, cystoscopy, nephrology evaluation, and follow-up. Urology 2001;57(4):604–10.
The main diagnostic test for bladder cancer is cystoscopy and biopsy. White light cystoscopy (WLC) is the gold standard, and flexible office cystoscopy is as reliable as a rigid endoscopy (Grossfeld et al, 2001). White light cystoscopy has an excellent sensitivity and specificity for papillary tumors but is relatively poor for CIS. Cystoscopy with porphyrin dye (commonly referred to as blue light cystoscopy) may be more sensitive in the detection of CIS (Fradet et al, 2007; Grossman et al, 2007). Porphyrin-induced fluorescence cystoscopy uses photoactive porphyrins, such as hexaminolevulinate, that accumulate preferentially in neoplastic tissue and emit red fluorescence under blue-wavelength light. This may improve the detection of small papillary lesions and CIS. A phase III trial evaluating white and blue light cystoscopy in patients with known or suspected tumors was recently completed (Grossman et al, 2007). Blue light cystoscopy detected 58% of CIS compared with 15% using white light. However, at the patient level, the sensitivity of blue light was 87% and 83% for white light. Blue light cystoscopy has a false-positive rate of 39% (Fradet et al, 2007). The true impact of blue light cystoscopy on the detection of bladder cancer is unclear and further studies are required to determine its exact clinical role.
Narrow-band imaging (NBI) is an endoscopic optical image enhancement technique that enhances the contrast between mucosal surfaces and microvascular structures without the use of dyes. The depth of light penetration into the bladder wall increases with increasing wavelength. NBI illuminates the mucosal surface with light of a narrow bandwidth in the blue (415 nm) and green (540 nm) light spectrum, which are strongly absorbed by hemoglobin. Consequently, the vascular structures appear dark brown or green against a pink or white mucosal background. Commercially available systems have integrated NBI and WLC, allowing activation of the NBI wavelengths with the push of a button. Herr et al. performed white light cystoscopy with subsequent NBI cystoscopy in 427 consecutive patients with a history of NMIBC (Herr and Donat, 2008). Of the 103 patients with a tumor recurrence, 56% had additional tumors identified with NBI compared with use of white light cystoscopy, and in 12% of patients, the recurrent tumor was only found with NBI. For white light and NBI cystoscopy, the overall sensitivity was 87% and 100% and the overall specificity 85% and 82%, respectively. A recent study suggests that NBI more accurately detects tumor recurrence after BCG therapy than do urine cytology or white light cystoscopy, and NBI can obviate the need for random bladder biopsies in post-BCG bladders (Herr, 2008). NBI detected tumor recurrence in 21 of 22 patients with tumor recurrence after BCG, but another 10 patients had a false-positive NBI resulting in unnecessary biopsies. Because the NBI and white light cystoscopy is performed by the same urologist, observation bias may skew these results.
Random bladder biopsies are recommended to detect unsuspected CIS or small papillary tumors in endoscopically normal urothelium. Overall, there is a 2.5% detection rate of CIS or small papillary tumors in random biopsies of patients with known or suspected bladder tumors (Fradet et al, 2007). For patients with concurrent bladder tumors, a random biopsy will detect dysplasia or CIS in up to 23% of cases (Mufti and Singh, 1992). It is reasonable to perform random biopsies in high-risk individuals, such as for those given postintravesical therapy or for those with a positive cytology and an endoscopically negative bladder. Urine cytology, first introduced by Papanicolaou in 1945, evaluates the morphologic changes associated with bladder cancer and is the gold standard urinary marker against which other markers are held (Papanicolaou and Marshall, 1945). Overall, the sensitivity and specificity for cytology in detecting bladder cancer is 40% to 62% and 94% to 100%, respectively (van Rhijn et al, 2005; Volpe et al, 2008). Positive urine cytology is virtually diagnostic of a bladder tumor, though there are cases in which the tumor is not endoscopically visible. The sensitivity and specificity of urine cytology is dependent on the cytopathologist, number of samples evaluated, and the stage and grade of the tumor (Volpe et al, 2008). Instrumented urine during cystoscopy has improved sensitivity and specificity, but an invasive procedure is required (Badalament et al, 1987). Fifteen percent of patients with atypical cytology that is not diagnostic of cancer will have an underlying malignancy (Novicki et al, 1998). Thus patients with an atypical cytology need more frequent evaluation or repeat random bladder biopsies.
Urine Markers for Urothelial Cancer
Van Rhijn and colleagues (2005) did a systematic literature review evaluating urine marker studies for surveillance only and included those markers that had at least two studies published from two separate institutions (Table 80–11). We will discuss the markers that had at least 70% sensitivity and specificity plus novel markers that may be important in the future. The sensitivity and specificity of these tests are lower in patients undergoing surveillance evaluations with no active tumor or those with low-grade cancers (van Rhijn et al, 2005; Zwarthoff, 2008).
NMP-22 is a nuclear matrix protein that is used to form the cell nuclei. NMP-22 is shed into the urine and has a 20-times higher concentration in the urine of bladder cancer patients than in noncancer controls (Keesee et al, 1996). There are a variety of NMP-22 cutoff levels for bladder cancer detection, but typically 10 units/mL is used to identify patients with or without cancer (Soloway et al, 1996; Grossman et al, 2006). A lower cutoff level of 5 units/mL improves the sensitivity but significantly worsens the specificity. The cutoff level does not appear to be related to stage or grade of the disease. False positives with NMP-22 can occur from patients with an active urinary tract infection or significant hematuria (Atsu et al, 2002). Grossman and colleagues (2006) reported a large multi-institutional study evaluating the efficacy of using NMP-22 (Grossman et al, 2006) as a marker. Using a cutoff level of 10 units/mL, the overall sensitivity and specificity for detecting urothelial cancer was 49% and 87%, respectively. The sensitivity for Ta, T1, and T2 tumors was 36%, 65%, and 88%, respectively. A combination of cystoscopy and NMP-22 detected all but one of the 103 tumors seen in this study. NMP-22 picked up eight of the nine tumors missed by white light cystoscopy. NMP-22 appears to be an adjunct to white light cystoscopy in approximately 10% of patients.
The Lewis blood group antigen X is usually absent from urothelial cells in adults except for occasional umbrella cells (Sheinfeld et al, 1990). There is increased Lewis X expression in bladder cancers, and it is independent of secretor status, grade, and stage. The sensitivity and specificity for the detection of bladder cancer is 75% and 85%, respectively. There is no commercially available test to date. CK 20 and CYFRA 21.1 are fragments of cytoskeletal proteins that can be detected in the urine of bladder cancer patients by either protein or mRNA detection (Ramos et al, 2003). CK 20 has a sensitivity and specificity of 85% and 76%, respectively. A recent multicenter study of 446 patients evaluating the role of CYFRA 21.1, with a cutoff value of 4 ng/mL, found a sensitivity and specificity of 43% and 68%, respectively (Fernandez-Gomez et al, 2007). Unfortunately, none of the Ta tumors were identified at the 4 ng/mL cutoff. Decreasing the CYFRA 21.1 cutoff to 1.5 ng/mL increased Ta detection to 33%, but the specificity dropped to an unacceptable 43%. Therefore it is not felt to be a useful marker in the current form, or at least for low-grade disease.
Fluorescence in-situ hybridization (FISH) identifies fluorescently labeled DNA probes that bind to intranuclear chromosomes. The current commercially available probes evaluate aneuploidy for chromosomes 3, 7, and 17 and homozygous loss of 9p 21 (Zwarthoff, 2008). The median sensitivity and specificity of FISH analysis is 79% and 70%, respectively (van Rhijn et al, 2005). A recent prospective study of 250 patients evaluated the role of FISH analysis for identifying recurrent urothelial cancer (Yoder et al, 2007). FISH detected 25 of 39 concurrent tumors, and 35 tumors occurred later in 56 patients who initially had a positive FISH test. The authors suggested that this was an anticipatory finding. Another study by Moonen and colleagues (2007) evaluated 105 patients with urothelial cancer. The sensitivity for Ta, T1, and T2 tumors was 26.7%, 60%, and 50%, respectively. These lower sensitivity findings were confirmed (Gudjonsson et al, 2008). It appears FISH analysis is moderately useful for high-grade disease and may be anticipatory of new tumor formation; however, because of the recurrent nature of noninvasive bladder cancer, it is difficult to tell if the FISH test was identifying chromosomal abnormalities present in normal-appearing urothelium or if it was false-positive test. There are multiple markers available to identify short DNA repeats present throughout the chromosomes that are lost in some tumor cells. Microsatellite analysis amplifies these repeats in the genome that are highly polymorphic, and PCR amplification can detect tumor-associated loss of heterozygosity by comparing the peak ratio of the two alleles in tumor DNA in the urine sample with the presence of the alleles in a blood sample from the same individual (Steiner et al, 1997, Wang et al, 1997). The sensitivity and specificity of microsatellite analysis for the detection of urothelial carcinoma range from 72% to 97% and 80% to 100%, respectively (Steiner et al, 1997; Wang et al, 1997). A recent European study evaluated microsatellite analysis in voided urine samples for the detection of low–malignant potential non–muscle-invasive urothelial carcinoma (van der Aa et al, 2009). They report sensitivity and specificity of 58% and 72%, respectively. Microsatellite analysis missed only one T1 high-grade urothelial cancer. Interestingly, if the microsatellite analysis is persistently positive, there was an 83% 2-year recurrence rate, but if the analysis was persistently negative, only 22% of patients had recurrent tumors. Hopefully, standardization of the test will allow analysis without a blood sample, and this will significantly improve the patient’s acceptance.
CpG dinucleotide islands cluster around promoters in an unmethylated state to allow gene expression (Knowles, 2007). Methylation of the CpG islands shuts down the promoter, and if the promoter in question is part of a tumor suppressor gene then cancer can form. Examples of promoter methylation of CpG islands causing epigenetic changes in urothelial cancer include the P16/CDKN2A gene (Gonzalez-Zulueta et al, 1995). The sensitivity of gene methylation for the detection of bladder cancer is 75%; however, methylated CpG islands can be found in the normal urothelial cells of older patients (Yates et al, 2006). FGFR-3 point mutations are found in 75% of non–muscle-invasive bladder cancer, particularly in Ta tumors (van Rhijn et al, 2004; Wolff et al, 2005). Unfortunately, there are over 11 different point mutation sites within this gene, and therefore identification of all possible mutations is difficult in a single urine sample. Single-strand conformational polymorphism can detect these point mutations, and a snapshot assay has been produced that holds the possibility for rapid identification of FGFR-3 mutations (van Oers et al, 2005). Surface-enhanced laser desorption ionization (SELDI) mass spectroscopy of urine samples has a sensitivity and specificity for detecting bladder cancer of 50% to 90% and 60% to 90%, respectively (Vlahou et al, 2001). Tumor grade, patient age, and type of analysis are confounders of SELDI analysis, and a multi-institutional trial is needed to determine its effectiveness in identifying urothelial carcinoma. Survivin is an antiapoptotic protein that has a high expression in urothelial cancer (Smith et al, 2001). Survivin is found in 10% to 30% of bladder cancers and is readily shed into the urine. The sensitivity and specificity of survivin in the detection of urothelial tumors is 64% to 100% and 87% to 93%, respectively (Smith et al, 2001; Shariat et al, 2004). This test may be useful in predicting which patients will respond to intravesical therapy (Hausladen et al, 2003). Survivin was relatively poor at detecting advanced-stage or high-grade tumors, with a sensitivity of 71% for stage T2 tumors and 80% for high-grade cancers (Shariat et al, 2004). Hylauronic acid controls intercellular communications and cell replication. Urothelial cancer induces hylauronic acid production from fibroblasts, and the amount correlates with the stage of the disease. The sensitivity and specificity of hylauronic acid for detection of bladder cancer is 91% to 100% and 84% to 90%, respectively (Pham et al, 1997; Lokeshwar et al, 2002). The sensitivity and specificity for discriminating between low-grade and high-grade lesions is unclear. Telomerase resides at the terminal ends of the chromosomes and duplicates random DNA repeats to prevent cell death (Rhyu, 1995). Telomerase activity is measured in telomeric repeat application protocol (TRAP) and is detected in 80% of urine from patients with bladder cancer with no grade differential. The sensitivity and specificity is 90% and 88%, respectively (Sanchini et al, 2005). The ImmunoCyt test (Diagnocure, Québec, Canada) detects mucin-based antigens that are present on most bladder cancer cells. The sensitivity and specificity is 61% to 92% and 71% to 90%, respectively (Halling et al, 2000; Pfister et al, 2003).
Virtually all patients complain of pain and discomfort with an office cystoscopy. Urine markers studies could forgo this pain in select situations as described above. However, patients reported that a urine marker study would need 90% sensitivity in order to replace office cystoscopy (Vriesema et al, 2000). The primary concern is missing tumor cells by relying on the urinary marker. None of the currently available urinary markers meet this 90% sensitivity on a reliable basis, and therefore a combination of cystoscopy with urine markers, in select situations, is appropriate for surveillance of patients with non–muscle-invasive bladder cancer.
Key Points: Detection of Urothelial Cancer
Prevention/Complementary Medicine Treatments for Urothelial Cancer
Bladder cancer is primarily caused by urothelial exposure to carcinogens in the environment. The long lag time between carcinogenic exposure and subsequent bladder cancer formation makes testing of preventive measures difficult. Prevention of urothelial cancer is a high priority because it is the most expensive cancer to treat for several reasons, including that most urothelial cancer patients do not die from the disease, there is a high recurrence rate, and the primary mode of cancer control is repeated surgical procedures (Botteman et al, 2003; Jemal et al, 2008). There are three avenues to cancer prevention: primary/avoidance, prevention of malignant transformation of premalignant lesions, and prevention of tumor recurrence. Because there are no well-defined premalignant lesions in bladder cancer formation, most research has centered on primary tumor prevention and prevention of recurrent tumor.
Smoking cessation is the primary mode to prevent urothelial carcinoma. Smoking is responsible for 30% to 50% of all bladder cancers in males, and smokers have a two- to sixfold greater chance of getting bladder cancer than nonsmokers (Brennan et al, 2000; Boffetta, 2008). Smoking cessation will decrease the risk of eventual urothelial cancer formation in a linear fashion. After 15 years of not smoking, the risk of cancer formation is the same as for someone who never smoked (Smoke IAfRoCT, 2004). The strong influence of smoking in bladder cancer formation prevents accurate determination of other less significant dietary, micronutrient, or lifestyle changes that may alter bladder cancer formation. There have been several animal studies that show calorie restriction prolongs life and prevents cancer (Kuska, 2000). The mechanism is unclear but may be mediated through insulin-like growth factor-1 (IGF-1), because calorie-restricted mice given IGF-1 had cancer formation similar to normal-calorie mice but more than calorie-restricted mice without IGF-1 supplementation (Dunn et al, 1997). However, it is difficult to translate these calorie-restriction animal studies to the human setting, because exercise level cannot be controlled. The key seems to be less caloric intake rather than total body weight, although the two are indirectly related. A randomized breast cancer study evaluated low- versus normal-fat diets in women with recently resected breast cancer. Women on the low-fat diet with aggressive breast cancer had a significantly lower risk of recurrence. This could be due to a variety of factors, including weight loss, lower caloric intake, and lower insulin levels (Chlebowski et al, 2006). Fruit and vegetable ingestion has been postulated to prevent a variety of cancers, including urothelial cancer. There have been many prospective and case-control studies evaluating fruit and vegetable intake. Most show equivocal results, although one showed a statistically significant difference in bladder cancer rates only in male subjects (Michaud et al, 2000). Citrus, apples, berries, and tomatoes have been shown in some, but not all studies, to result in a lower risk of urothelial cancer (Brinkman and Zeegers, 2008). Carrots and cruciferous vegetables, particularly cabbage, cauliflower, and kale, have also been implicated to have an inverse relationship with bladder cancer formation. None of these studies have been a randomized trial, therefore whether ingestion of fruits and vegetables leads to a healthier lifestyle that is associated with lower urothelial cancer formation, or if these particular agents have a direct effect on urothelial cancer formation, is unclear.
There have been several studies evaluating the role of vitamin ingestion in bladder cancer formation. None of the large vitamin trials have shown a decreased risk in primary urothelial cancer prevention (Grossman et al, 2008). A randomized trial of patients with urothelial cancer did show that BCG plus high-dose vitamins significantly prolonged the time of recurrence (Lamm et al, 1994). In this randomized trial of active urothelial cancer patients, all received intravesical BCG plus the recommended dietary-allowed vitamin doses, and half were randomly assigned to high-dose vitamins that included 4000 units of vitamin A, 100 mg vitamin B6, 2000 mg of vitamin C, 400 international units of vitamin E, and 90 mg of zinc daily. The high-dose vitamin group had a significantly prolonged time to urothelial cancer recurrence, with a 5-year disease survival rate of 91% compared with 41% in the standard-dose vitamin group (P = .0014). To date, follow-up trials have not been conducted to corroborate these results. Micronutrient ingestion has been suggested to decrease the bladder cancer risk. There is one ongoing randomized placebo-controlled trial of 200 µg of selenium plus 154 mg of α-tocopherol daily that should be completed by 2011.
The urogenesis theory suggests that high fluid intake leads to more micturition and lower concentrations of potential carcinogens in the urine and thus a lower risk of urothelial cancer (Braver et al, 1987). There are conflicting results showing an inverse relationship between fluid intake and urothelial cancer formation, but it is strongest in women (Vena et al, 1993b). The health professional follow-up study of 48,000 participants found that total fluid intake was inversely related to urothelial cancer formation but only when comparing the highest quintile (>2500 mL) and lowest quintile (<1300 mL) groups (Michaud et al, 2007). Green tea contains polyphenolic compounds that are potent antioxidants—specifically, epigallocatechin-3 gallate, which inhibits urothelial cell growth in vitro (Qin et al, 2007). A large Japanese study of 49,566 men and 54,874 women evaluated the effect of smoking, caffeine, and green tea on urothelial cancer formation during a 15-year period (Kurahashi et al, 2008). As expected, smoking was strongly associated with urothelial cancer formation. Caffeine, either in the form of coffee or green tea, may be associated with urothelial cancer formation, and this effect was strongest in women. Caffeine is a known risk factor for urothelial cancer formation, and the carcinogenic affect of caffeine on urothelial cancer cells may be stronger than the antioxidant affect found in green tea (Pelucchi et al, 2008).
Histologic Variants of Urothelial Cancer
Urothelial carcinoma of the bladder had previously been labeled transitional cell carcinoma of the bladder because of its known propensity for cellular differentiation into other tumor types, such as squamous cell carcinoma, adenocarcinoma, and clear cell carcinoma. Recently the wider spectrum of histologic variance of urothelial cancer has been identified to include distinct growth patterns of urothelial carcinoma, altered cellular differentiation of urothelial carcinoma, mixed cellular growth patterns, and unusual stromal reactions combined with urothelial cancer growth (Table 80–12) (Lopez-Beltran and Cheng, 2006). The more common histologic variants will be discussed.
Table 80–12 Variants of Invasive Urothelial Carcinoma
Lopez-Beltran A, Cheng L. Histologic variants of urothelial carcinoma: differential diagnosis and clinical implications. Hum Pathol 2006;37(11):1371–88.
Micropapillary Urothelial Carcinoma
Micropapillary urothelial carcinoma was first described in the early 1990s as a tumor growth pattern that occurs in many organs, including bladder, breast, lung, and ovary, usually presenting at an advanced stage (Amin et al, 1994; Samaratunga and Khoo, 2004). The incidence of micropapillary urothelial carcinoma is 0.7% to 2.2% of all urothelial tumors, with a male to female ratio of 10 : 1 and an average age at diagnosis of 65 years (Amin et al, 1994). Because of the advanced stage at diagnosis, the 5- and 10-year overall survival rate of patients with micropapillary urothelial carcinoma is 51% and 24%, respectively (Kamat et al, 2007). The cancer-specific and overall survival rates are similar, suggesting that most patients died from their cancer (Johansson et al, 1999). The histologic characteristics of micropapillary urothelial carcinoma are similar to papillary serous carcinoma of the ovary that develops delicate filiform processes with infiltrating clusters of micropapillary tumors lacking vascular stalks (Fig. 80–15). Micropapillary urothelial carcinoma is associated with conventional urothelial carcinoma in 80% of cases (Lopez-Beltran and Cheng, 2006). Angiolymphatic invasion is common even in non–muscle-invasive disease, highlighting the aggressive nature of this cancer. There is a high progression rate from non–muscle-invasive to muscle-invasive bladder cancer approaching 70%, with a high subsequent metastatic rate despite treatment (Johansson et al, 1999; Kamat et al, 2007). The most effective treatment for all stages of micropapillary urothelial carcinoma is surgical resection. Treatment with transurethral resection and BCG therapy is ineffective unless the tumor is completely resected (Kamat et al, 2007). Similar to ovarian cancer, neoadjuvant chemotherapy does not appear effective in micropapillary urothelial carcinoma (Bristow et al, 2002; Kamat et al, 2007). Neoadjuvant chemotherapy may actually worsen survival by delaying therapy when compared with immediate cystectomy. Even in the best of situations, immediate cystectomy for non–muscle-invasive micropapillary bladder cancer has a cancer-specific survival rate of 72% compared with 60% for those treated with transurethral resection of the bladder tumor and BCG followed by cystectomy at the time of progression (Kamat et al, 2007). The best outcomes were in those patients who had no residual micropapillary tumor at the time of cystectomy, suggesting that complete resection of the tumor is the key procedure. Patients with locally advanced disease do poorly despite aggressive chemotherapy and surgical resection, with a cancer-specific survival rate of less than 22% at 4 years. There are no specific molecular markers associated with micropapillary urothelial carcinoma; however, these tumors are immunoreactive for epithelial membrane antigen and cytokeratins 7, 20, and 34 (Samaratunga and Khoo, 2004). Micropapillary urothelial carcinoma presents at an advanced stage, with less than 9% having noninvasive disease and more than 50% presenting as muscle-invasive, nodal, or metastatic disease (Johansson et al, 1999).
Nested Variant of Urothelial Carcinoma
This is a rare but aggressive cancer that has a male to female ratio of 6 : 1 and can be confused with benign lesions such as Von Brunn nests that are in the lamina propria, cystitis cystica, and inverted papillomas (Holmang and Johansson, 2001). There is little nuclear atypia in nested variant urothelial carcinoma, but the tumor cells will often contain areas with large nuclei and mitotic figures (Fig. 80–16). Despite aggressive therapy, the mortality rate from nested variant urothelial carcinoma is significant, with 70% dying of their disease within three years (Paik and Park, 1996).
Clear Cell Variant of Urothelial Carcinoma
Seventy percent of urothelial cancer will have foci of clear cells within the tumor (Lopez-Beltran and Cheng, 2006). These clear cells contain glycogen-rich vacuoles and may be confused with metastatic clear cell carcinoma of the kidney; however, the clear cell variant does not portent a significantly worse prognosis for urothelial cancers.
Glandular/Adeno Differentiation
Mixed tumor differentiation is most common with squamous cell cancer, but glandular differentiation only occurs in 6% of urothelial cancer cases (Lopez-Beltran and Cheng, 2006). Glandular differentiation is defined as the presence of two glandular spaces within the tumor. It is important that these glandular spaces are not confused with lymphatic invasion or with processing artifacts. Mucin production can occur, and tumor cells seem to be floating in the mucin. Adjuvant chemotherapy plus radical cystectomy is the best method of treatment for glandular-differentiated urothelial cancer. Previous data has suggested that neoadjuvant chemotherapy was relatively ineffective against glandular and squamous differentiation for muscle-invasive disease. A recent secondary analysis of the Southwest Oncology Group Trial 8710 of neoadjuvant muscle methotrexate, vinblastine, doxorubicin, and cisplatin (MVAC) followed by cystectomy versus cystectomy alone for muscle-invasive bladder cancer evaluated the effect of neoadjuvant therapy on mixed cellular differentiation of urothelial cancer. Among the patients with mixed tumors, the survival benefit from chemotherapy appeared to be greater in magnitude (hazard ratio [HR] = 0.46, P = .02) than among patients with pure urothelial carcinoma (HR = 0.9, P = .48) compared with immediate cystectomy (Scosyrev et al, 2010). This suggests that neoadjuvant chemotherapy is an appropriate treatment prior to radical cystectomy for patients with invasive mixed differentiation urothelial cancer.
Nonurothelial Malignancies
Sarcomas
Sarcomas are the most common mesenchymal tumor of the bladder but comprise less than 1% of all bladder cancers (Berkmen and Celebioglu, 1997; Parekh et al, 2002; Dotan et al, 2006). Subclassification of sarcoma is based on histologic variations, depending on the specific malignant cell type (Parekh et al, 2002; Spiess et al, 2007). Leiomyosarcoma is the most common histologic subtype, followed by rhabdomyosarcoma and then, rarely, angiosarcoma, osteosarcoma, and carcinosarcoma. The male to female ratio is 2 : 1, and the average age at presentation is in the 6th decade of life. There are no clear agents that cause bladder sarcomas, although there is an association with pelvic radiation and systemic chemotherapy for other malignancies (Spiess et al, 2007). Importantly, bladder sarcomas are not smoking related. Genetic abnormalities of leiomyosarcoma are inconsistent and not used in staging or identification. The majority of sarcomas are high-grade, and greater than 75% will be confined to bladder muscle (Rosser et al, 2003; Dotan et al, 2006). The most common presenting symptom is gross painless hematuria in 79%, followed by local irritative symptoms in 16%. Transurethral resection of the tumor, which may appear to have normal overlying urothelium, is necessary for diagnosis along with abdominal and chest imaging. The grade of the sarcoma is the primary prognostic factor and incorporated into the overall sarcoma staging system (Dotan et al, 2006). Treatment for localized disease includes radical cystectomy, with a premium on obtaining negative surgical margins because the local recurrence rate is 2.4 times higher in patients with a positive surgical margin (Dotan et al, 2006). The overall 5-year disease-free survival rate for leiomyosarcoma of the bladder is 52% to 62% (Rosser et al, 2003). Other poor prognostic factors associated with sarcomas include angiolymphatic invasion and metastatic disease at the time of presentation. Active chemotherapeutic regimens are lacking for bladder sarcomas, but doxorubicin, ifosphamide, and cisplatin are the most effective agents (Dotan et al, 2006; Spiess et al, 2007). The most common site for metastatic disease is lung, followed by bone, liver, and, rarely, soft tissue organs. Rhabdomyosarcomas may occur at any age, but in young children, they produce polyploid lesions at the base of the bladder, described as botryoides tumors. Multi-modality therapy combining chemotherapy with surgical resection and radiation are used in the treatment of pediatric rhabdomyosarcomas (Zanetta et al, 1999).
Signet Ring Cell Carcinoma
Primary signet ring cell carcinoma of the bladder is extremely rare, occupying less than 1% of all epithelial bladder neoplasms (Morelli et al, 2006). Signet ring cell carcinoma can be of urachal origin and directly extend into the bladder. These tumors generally present as high-grade, high-stage tumors and have a uniformly poor prognosis. The primary treatment is radical cystectomy; however, in the majority of cases there are regional or distant metastases at the time of presentation, and the mean survival time is less than 20 months (Torenbeek et al, 1996). There are reports of elevated carcinoembryonic antigen (CEA) in patients with signet ring cell carcinoma. The prognostic significance of this elevated serum marker is unclear (Morelli et al, 2006). Understaging is very common in signet ring cell carcinoma, with peritoneal studding common at the time of surgical exploration.
Small Cell Carcinoma
Small cell carcinoma primarily arises in the lung, but can occur in extrapulmonary sites, including the bladder, prostate, and colon (Blomjous et al, 1989). Small cell carcinoma of the bladder should be considered and treated as metastatic disease, even if there is no radiologic evidence of disease outside the bladder. Small cell carcinoma of the bladder accounts for much less than 1% of all primary bladder tumors. In general, small cell carcinoma of the bladder is very chemosensitive, and the primary mode of therapy is chemoradiation therapy. The tumor affects men older than 70 years, and there is a slightly higher prevalence in smokers (Choong et al, 2005). The origin of extrapulmonary small cell carcinoma is unclear, but may be related to multipotential stem cells that can develop into small cell carcinoma within extrapulmonary organs (Blomjous et al, 1989). The most common presenting symptom is painless gross hematuria; however, local irritation and pain are relatively frequent. At transurethral resection the mass is indistinguishable from urothelial carcinoma, and resection is required to make a histologic diagnosis. Histologically, the common cellular pattern is diffuse sheets of dark blue cells with necrosis and mitosis. Chromogranin A staining is the primary method to distinguish high-grade urothelial carcinoma from small cell cancer of the bladder (Iczkowski et al, 1999). A variety of chemotherapeutic regimens have been used, but carboplatin or cisplatin and etoposide is the current treatment of choice (Choong et al, 2005). It is common to have a complete response from initial chemotherapy; however, clinical relapse occurs in greater than 80% of cases. It is not uncommon to see small cell carcinoma admixed with other histologic types of bladder cancer, including urothelial, adenocarcinoma, and squamous cell cancer (Choong et al, 2005). This supports the stem cell theory of histogenesis with multipotential undifferentiated stem cells producing small cell carcinoma and other histologic types of bladder cancer. Identical patterns of allelic loss in small cell carcinoma and coexisting urothelial cancers suggest a common clonal origin. Small cell carcinoma of the bladder exhibits both epithelial and neuroendocrine differentiation. Neuron-specific enolase, chromagranin A, and synaptophysin markers help differentiate from urothelial carcinoma (Mukesh et al, 2009). Although chemo-radiation therapy is the primary treatment for small cell carcinoma of the bladder, experience combining chemotherapy with radical cystectomy for primary small cell cancers of the bladder has shown equal, or perhaps better, local control and disease-free survival than found with chemoradiation (Quek et al, 2005). However, with 5-year cancer-specific survival rates of 16% to 18% with chemoradiation or chemotherapy and radical cystectomy, respectively, the primary method to improve survival will be more effective systemic therapy.
Squamous Cell Cancer
Chronic infection with Schistosoma hematobium or other bacteria, to a lesser degree, leads to squamous cell formation of the bladder (Abol-Enein, 2008). The Schistosoma ova are deposited in the wall of the bladder and produce chronic inflammation that converts the urothelium to a squamous cell epithelium. Squamous cell epithelium has a much greater proliferation rate, and, with the presence of chronic inflammation, over time this greater proliferation rate leads to cancer formation. The exact mechanism by which Schistosoma ova can cause squamous cell carcinoma is unclear, but two factors are suspected. One is the increased proliferation rate, and the second is the chronic inflammation and exposure to environmental agents. The increased proliferation of the squamous epithelium leads to a higher risk of spontaneous genetic alterations that can cause cancer (Cohen and Ellwein, 1990). A chronic inflammatory process and exposure to environmental agents can combine to generate genotoxic substances in the urine, such as N-butyl-N-(4-hydroxybutyl)nitrosamine. This substance is generated in very high levels in the urine of patients chronically infected with Schistosoma organisms and is a known bladder cancer carcinogen in bladder cancer models (Abol-Enein, 2008). In addition, chronic infection with Schistosoma hematobium converts nitrates to nitrites and subsequently to nitrosamines, which are known bladder carcinogens. Chronic schistosomiasis leads predominantly to squamous cell carcinoma rather than urothelial carcinoma, with 70% of infected patients who develop bladder cancer having squamous cell carcinoma, although many will have both urothelial and squamous cell cancer (Cohen and Johansson, 1992) (Fig. 80–17). Spinal cord–injured patients are also at risk for developing squamous cell carcinoma, most likely because of chronic catheter irritation and infection. Older studies have suggested a 2.5% to 10% incidence of squamous cell carcinoma in the spinal cord–injured population, with a mean delay of 17 years after the spinal cord injury (Kaufman et al, 1977). More recent analysis of the association of spinal cord injury and bladder cancer formation has shown a remarkably lower risk of bladder cancer formation of 0.38%, most likely because of better catheter care (Bickel et al, 1991). This supports the concept that chronic infection and foreign bodies can lead to bladder cancer formation.
Key Points: Histologic Variants
Prostatic Urethral Cancer
Ortega was the first to describe urothelial carcinoma involving the prostatic urethra (Ortega et al, 1953). Prostatic urethral cancer is associated with urothelial cancer in 90% of cases, primarily CIS, and most will have multifocal bladder tumors. However, the incidence of prostatic urethral disease in patients with primary urothelial cancer is only 3% (Rikken et al, 1987; Millan-Rodriguez et al, 2000). Secondary prostatic urethral involvement in patients with a history of urothelial cancer is approximately 15% at 5 years and 30% at 15 years, almost uniformly associated with extensive intravesical therapy (Herr and Donat, 1999). For patients undergoing radical cystectomy for urothelial cancer, the risk of identifying prostatic urethral disease is 40%. Risk factors for prostatic urethral involvement are CIS of the bladder neck and a history of intravesical chemotherapy (Wood et al, 1989a). Most patients with prostatic urethral disease will have direct extension of their bladder cancer into the prostatic urethra; however, some patients will have pagetoid spread underneath normal-appearing urothelium at the bladder neck.
Transurethral resection of the prostatic urethra is the primary method for detecting prostatic urethral carcinoma, with a sensitivity and specificity of greater than 90% (Wood et al, 1989; Sakamoto et al, 1993; Donat et al, 2001a, 2001b). A transurethral resection of the prostatic urethra should be performed on all patients with positive urine cytology but a negative bladder biopsy, or on those with recurrent bladder cancer after multiple courses of intravesical chemotherapy. For patients with noninvasive prostatic urethral cancer, transurethral resection of the prostate with BCG therapy is appropriate (Palou et al, 2007). For patients with prostatic ductal disease, a complete transurethral prostatectomy (TURP) is warranted, plus BCG therapy. The current 2009 bladder cancer staging system now excludes noninvasive prostatic urethral disease from the T4 category. These tumors have a relatively good prognosis not consistent with T4 staging status. Prostatic urethral cancer is now staged in a prostatic urethral category in the AJCC staging manual (Edge et al, 2010). Only patients with prostatic stromal invasion, either direct or indirect, are considered to have T4a-staged disease. Prostatic stromal invasion occurs in 7.6% to 25% of patients with prostatic urethral cancer (Wood et al, 1989b; Herr and Donat, 1999). Ninety percent of these cases are associated with previous bladder tumors, and almost all have had previous BCG therapy. Transurethral biopsy of the prostate is recommended, however, if patients have direct extension of the tumor through the bladder wall into the prostate, a prostate needle biopsy may be warranted (Donat et al, 2001). Patients with prostatic stromal disease are staged at T4a, and a better prognosis is associated with those who have stromal invasion through the prostatic urethra compared with invasion directly through the bladder wall into the prostate (Esrig et al, 1996). Multimodal therapy combining chemotherapy with radical cystectomy is the appropriate treatment (Palou et al, 2007).
Dyrskjot L, Zieger K, Real FX, et al. Gene expression signatures predict outcome in non-muscle-invasive bladder carcinoma: a multicenter validation study. Clin Cancer Res. 2007;13(12):3545-3551.
Grossfeld GD, Litwin MS, Wolf JSJr, et al. Evaluation of asymptomatic microscopic hematuria in adults: the American Urological Association best practice policy—part II: patient evaluation, cytology, voided markers, imaging, cystoscopy, nephrology evaluation, and follow-up. Urology. 2001;57(4):604-610.
Grossman HB, Gomella L, Fradet Y, et al. A phase III, multicenter comparison of hexaminolevulinate fluorescence cystoscopy and white light cystoscopy for the detection of superficial papillary lesions in patients with bladder cancer. J Urol. 2007;178(1):62-67.
Herr HW, Donat SM, Dalbagni G. Can restaging transurethral resection of T1 bladder cancer select patients for immediate cystectomy? J Urol. 2007;177(1):75-79. discussion 79
Jemal A, Siegel R, Ward E, et al. Cancer statistics, 2008. CA Cancer J Clin. 2008;58(2):71-96.
Kamat AM, Dinney CP, Gee JR, et al. Micropapillary bladder cancer: a review of the University of Texas M.D. Anderson Cancer Center experience with 100 consecutive patients. Cancer. 2007;110(1):62-67.
Knowles MA. Bladder cancer subtypes defined by genomic alterations. Scand J Urol Nephrol Suppl. 2008;218:116-130.
Lopez-Beltran A. Bladder cancer: clinical and pathological profile. Scand J Urol Nephrol Suppl. 2008;218:95-109.
Reulen RC, Kellen E, Buntinx F, et al. A meta-analysis on the association between bladder cancer and occupation. Scand J Urol Nephrol Suppl. 2008;218:64-78.
Spiess PE, Kassouf W, Steinberg JR, et al. Review of the M.D. Anderson experience in the treatment of bladder sarcoma. Urol Oncol. 2007;25(1):38-45.
van Rhijn BW, van der Poel HG, van der Kwast TH. Urine markers for bladder cancer surveillance: a systematic review. Eur Urol. 2005;47(6):736-748.
Wang R, Morris DS, Tomlins SA, et al. Development of a multiplex quantitative PCR signature to predict progression in non-muscle-invasive bladder cancer. Cancer Res. 2009;69(9):3810-3818.
Wood DPJr, Montie JE, Pontes JE, et al. Transitional cell carcinoma of the prostate in cystoprostatectomy specimens removed for bladder cancer. J Urol. 1989;141(2):346-349.
Aaboe M, Birkenkamp-Demtroder K, Wiuf C. SOX4 expression in bladder carcinoma: clinical aspects and in vitro functional characterization. Cancer Res. 2006;66:3434-3442.
Abdel-Latif M, Abol-Enein H, El-Baz M, Ghoneim MA. Nodal involvement in bladder cancer cases treated with radical cystectomy: incidence and prognosis. J Urol. 2004;172(1):85-89.
Aben KK, Witjes JA, Schoenberg MP, et al. Familial aggregation of urothelial cell carcinoma. Int J Cancer. 2002;98(2):274-278.
Abol-Enein H. Infection: is it a cause of bladder cancer? Scand J Urol Nephrol Suppl. 2008;218:79-84.
Aboulkassim TO, LaRue H, Lemieux P, et al. Alteration of the PATCHED locus in superficial bladder cancer. Oncogene. 2003;22(19):2967-2971.
Adachi H, Igawa M, Shiina H, et al. Human bladder tumors with 2-hit mutations of tumor suppressor gene TSC1 and decreased expression of p27. J Urol. 2003;170(2 Pt. 1):601-604.
Alishahi S, Byrne D, Goodman CM, Baxby K. Haematuria investigation based on a standard protocol: emphasis on the diagnosis of urological malignancy. J R Coll Surg Edinb. 2002;47:422-427.
Allen MJ, Boyland E, Dukes CE, et al. Cancer of the urinary bladder induced in mice with metabolites of aromatic amines and tryptophan. Br J Cancer. 1957;11(2):212-228.
Als AB, Dyrskjot L, von der Maase H. Emmprin and survivin predict response and survival following cisplatin-containing chemotherapy in patients with advanced bladder cancer. Clin Cancer Res. 2007;13:4407-4414.
Amin MB, Ro JY, el-Sharkawy T, et al. Micropapillary variant of transitional cell carcinoma of the urinary bladder. Histologic pattern resembling ovarian papillary serous carcinoma. Am J Surg Pathol. 1994;18(12):1224-1232.
Armstrong B, Doll R. Bladder cancer mortality in diabetics in relation to saccharin consumption and smoking habits. Br J Prev Soc Med. 1975;29(2):73-81.
Asano K, Miki J, Maeda S, et al. Clinical studies on inverted papilloma of the urinary tract: report of 48 cases and review of the literature. J Urol. 2003;170(4 Pt. 1):1209-1212.
Atsu N, Ekici S, Oge OO, et al. False-positive results of the NMP22 test due to hematuria. J Urol. 2002;167(2 Pt. 1):555-558.
Badalament RA, Kimmel M, Gay H, et al. The sensitivity of flow cytometry compared with conventional cytology in the detection of superficial bladder carcinoma. Cancer. 1987;59(12):2078-2085.
Bakkar AA, Wallerand H, Radvanyi F, et al. FGFR3 and TP53 gene mutations define two distinct pathways in urothelial cell carcinoma of the bladder. Cancer Res. 2003;63(23):8108-8112.
Balbi JC, Larrinaga MT, De Stefani E, et al. Foods and risk of bladder cancer: a case-control study in Uruguay. Eur J Cancer Prev. 2001;10(5):453-458.
Berggren P, Kumar R, Sakano S, et al. Detecting homozygous deletions in the CDKN2A(p16(INK4a))/ARF(p14(ARF)) gene in urinary bladder cancer using real-time quantitative PCR. Clin Cancer Res. 2003;9(1):235-242.
Berkmen F, Celebioglu AS. Adult genitourinary sarcomas: a report of seventeen cases and review of the literature. J Exp Clin Cancer Res. 1997;16(1):45-48.
Bianco FJJr, Gervasi DC, Tiguert R, et al. Matrix metalloproteinase-9 expression in bladder washes from bladder cancer patients predicts pathological stage and grade. Clin Cancer Res. 1998;4(12):3011-3016.
Bickel A, Culkin DJ, Wheeler JSJr. Bladder cancer in spinal cord injury patients. J Urol. 1991;146(5):1240-1242.
Billerey C, Chopin D, Aubriot-Lorton MH, et al. Frequent FGFR3 mutations in papillary non-invasive bladder (pTa) tumors. Am J Pathol. 2001;158(6):1955-1959.
Black PC, Brown GA, Dinney CP. Molecular markers of urothelial cancer and their use in the monitoring of superficial urothelial cancer. J Clin Oncol. 2006;24:5528-5535.
Blaveri E, Simko JP, Korkola JE, et al. Bladder cancer outcome and subtype classification by gene expression. Clin Cancer Res. 2005;11:4044-4055.
Blomjous CE, Vos W, De Voogt HJ, et al. Small cell carcinoma of the urinary bladder. A clinicopathologic, morphometric, immunohistochemical, and ultrastructural study of 18 cases. Cancer. 1989;64(6):1347-1357.
Blot WJ, Fraumeni JFJr. Geographic patterns of bladder cancer in the United States. J Natl Cancer Inst. 1978;61(4):1017-1023.
Boffetta P. Tobacco smoking and risk of bladder cancer. Scand J Urol Nephrol Suppl. 2008;218:45-54.
Boffetta P, Hashibe M. Alcohol and cancer. Lancet Oncol. 2006;7(2):149-156.
Boice JDJr, Engholm G, Kleinerman RA, et al. Radiation dose and second cancer risk in patients treated for cancer of the cervix. Radiat Res. 1988;116(1):3-55.
Botteman MF, Pashos CL, Redaelli A, et al. The health economics of bladder cancer: a comprehensive review of the published literature. Pharmacoeconomics. 2003;21:1315-1330.
Bouchardy C, Parkin DM, Wanner P, Khlat M. Cancer mortality among north African migrants in France. Int J Epidemiol. 1996;25(1):5-13.
Bouchardy C, Wanner P, Parkin DM. Cancer mortality among sub-Saharan African migrants in France. Cancer Causes Control. 1995;6(6):539-544.
Braver DJ, Modan M, Chetrit A, et al. Drinking, micturition habits, and urine concentration as potential risk factors in urinary bladder cancer. J Natl Cancer Inst. 1987;78(3):437-440.
Brennan P, Bogillot O, Cordier S, et al. Cigarette smoking and bladder cancer in men: a pooled analysis of 11 case-control studies. Int J Cancer. 2000;86(2):289-294.
Brennan P, Bogillot O, Greiser E, et al. The contribution of cigarette smoking to bladder cancer in women (pooled European data). Cancer Causes Control. 2001;12(5):411-417.
Brenner DJ, Curtis RE, Hall EJ, Ron E. Second malignancies in prostate carcinoma patients after radiotherapy compared with surgery. Cancer. 2000;88(2):398-406.
Bristow RE, Gossett DR, Shook DR, et al. Micropapillary serous ovarian carcinoma: surgical management and clinical outcome. Gynecol Oncol. 2002;86:163-170.
Brinkman M, Zeegers MP. Nutrition, total fluid and bladder cancer. Scand J Urol Nephrol Suppl. 2008;218:25-36.
Brown LF, Berse B, Jackman RW, et al. Increased expression of vascular permeability factor (vascular endothelial growth factor) and its receptors in kidney and bladder carcinomas. Am J Pathol. 1993;143(5):1255-1262.
Buckley O, Geoghegan T, Feeney J, et al. Answer to case of the month #126. Pelvic lipomatosis in association with cystitis glandularis. Can Assoc Radiol J. 2007;58(5):300-302.
Burch JD, Rohan TE, Howe GR, et al. Risk of bladder cancer by source and type of tobacco exposure: a case-control study. Int J Cancer. 1989;44(4):622-628.
Burns PB, Swanson GM. Risk of urinary bladder cancer among blacks and whites: the role of cigarette use and occupation. Cancer Causes Control. 1991;2(6):371-379.
Cairns P, Evron E, Okami K, et al. Point mutation and homozygous deletion of PTEN/MMAC1 in primary bladder cancers. Oncogene. 1998;16(24):3215-3218.
Case RA, Hosker ME. Tumour of the urinary bladder as an occupational disease in the rubber industry in England and Wales. Br J Prev Soc Med. 1954;8(2):39-50.
Castillo O, Foneron A, Vitagliano G, et al. Bladder leiomyoma: case report. Arch Esp Urol. 2008;61(1):87-91.
Chang BS, Kim HL, Yang XJ, Steinberg GD. Correlation between biopsy and radical cystectomy in assessing grade and depth of invasion in bladder urothelial carcinoma. Urology. 2001;57(6):1063-1066. discussion 6–7
Chatterjee SJ, Datar R, Youssefzadeh D, et al. Combined effects of p53, p21, and pRb expression in the progression of bladder transitional cell carcinoma. J Clin Oncol. 2004;22(6):1007-1013.
Chatterjee SJ, George B, Goebell PJ, et al. Hyperphosphorylation of pRb: a mechanism for RB tumour suppressor pathway inactivation in bladder cancer. J Pathol. 2004;203(3):762-770.
Cheng L, Cheville JC, Neumann RM, Bostwick DG. Natural history of urothelial dysplasia of the bladder. Am J Surg Pathol. 1999;23(4):443-447.
Cheng L, MacLennan GT, Zhang S, et al. Laser capture microdissection analysis reveals frequent allelic losses in papillary urothelial neoplasm of low malignant potential of the urinary bladder. Cancer. 2004;101(1):183-188.
Cheng L, Neumann RM, Weaver AL, et al. Predicting cancer progression in patients with stage T1 bladder carcinoma. J Clin Oncol. 1999;17(10):3182-3187.
Cheng L, Weaver AL, Neumann RM, et al. Substaging of T1 bladder carcinoma based on the depth of invasion as measured by micrometer: a new proposal. Cancer. 1999;86(6):1035-1043.
Chlebowski RT, Blackburn GL, Thomson CA, et al. Dietary fat reduction and breast cancer outcome: interim efficacy results from the Women’s Intervention Nutrition Study. J Natl Cancer Inst. 2006;98:1767-1776.
Choong NW, Quevedo JF, Kaur JS. Small cell carcinoma of the urinary bladder. The Mayo Clinic experience. Cancer. 2005;103:1172-1178.
Cohen SM, Cano M, St John MK, et al. Effect of sodium saccharin on the neonatal rat bladder. Scanning Microsc. 1995;9(1):137-147.
Cohen SM, Ellwein LB. Cell proliferation in carcinogenesis. Science. 1990;249(4972):1007-1011.
Cohen SM, Johansson SL. Epidemiology and etiology of bladder cancer. Urol Clin North Am. 1992;19(3):421-428.
Cordon-Cardo C. Molecular alterations associated with bladder cancer initiation and progression. Scand J Urol Nephrol Suppl. 2008;218:154-165.
Cordon-Cardo C, Cote RJ, Sauter G. Genetic and molecular markers of urothelial premalignancy and malignancy. Scand J Urol Nephrol Suppl. 2000;205:82-93.
Czerniak B, Cohen GL, Etkind P, et al. Concurrent mutations of coding and regulatory sequences of the Ha-ras gene in urinary bladder carcinomas. Hum Pathol. 1992;23(11):1199-1204.
Davis CP, Cohen MS, Hackett RL, et al. Urothelial hyperplasia and neoplasia. III. Detection of nitrosamine production with different bacterial genera in chronic urinary tract infections of rats. J Urol. 1991;145(4):875-880.
de Lorgeril M, Salen P, Martin JL, et al. Mediterranean dietary pattern in a randomized trial: prolonged survival and possible reduced cancer rate. Arch Intern Med. 1998;158(11):1181-1187.
De Stefani E, Boffetta P, Ronco AL, et al. Dietary patterns and risk of bladder cancer: a factor analysis in Uruguay. Cancer Causes Control. 2008;19(10):1243-1249.
Delclos GL, Lerner SP. Occupational risk factors. Scand J Urol Nephrol Suppl. 2008;218:58-63.
Donat SM, Genega EM, Herr HW, Reuter VE. Mechanisms of prostatic stromal invasion in patients with bladder cancer: clinical significance. J Urol. 2001;165:1117-1120.
Donat SM, Wei DC, McGuire MS, Herr HW. The efficacy of transurethral biopsy for predicting the long-term clinical impact of prostatic invasive bladder cancer. J Urol. 2001;165:1580-1584.
Dotan ZA, Tal R, Golijanin D, et al. Adult genitourinary sarcoma: the 25-year Memorial Sloan-Kettering experience. J Urol. 2006;176(5):2033-2038. discussion 2038–9
Dryson E, Mannetje A, Walls C, et al. Case-control study of high risk occupations for bladder cancer in New Zealand. Int J Cancer. 2008;122(6):1340-1346.
Dunn SE, Kari FW, French J, et al. Dietary restriction reduces insulin-like growth factor I levels, which modulates apoptosis, cell proliferation, and tumor progression in p53-deficient mice. Cancer Res. 1997;57:4667-4672.
Dyrskjot L, Kruhoffer M, Thykjaer T, et al. Gene expression in the urinary bladder: a common carcinoma in situ gene expression signature exists disregarding histopathological classification. Cancer Res. 2004;64(11):4040-4048.
Dyrskjot L, Thykjaer T, Kruhoffer M, et al. Identifying distinct classes of bladder carcinoma using microarrays. Nat Genet. 2003;33:90-96.
Dyrskjot L, Zieger K, Real FX, et al. Gene expression signatures predict outcome in non-muscle-invasive bladder carcinoma: a multicenter validation study. Clin Cancer Res. 2007;13(12):3545-3551.
Edge SB, Byrd DR, Compton CC, et al, editors. AJCC cancer staging manual, 7th ed, New York: Springer, 2010.
Edwards TJ, Dickinson AJ, Natale S. A prospective analysis of the diagnostic yield resulting from the attendance of 4020 patients at a protocol-driven haematuria clinic. BJU Int. 2006;97:301-305.
Epstein JI, Amin MB, Reuter VR, Mostofi FK. The World Health Organization/International Society of Urological Pathology consensus classification of urothelial (transitional cell) neoplasms of the urinary bladder. Bladder Consensus Conference Committee. Am J Surg Pathol. 1998;22(12):1435-1448.
Esrig D, Freeman JA, Elmajian DA, et al. Transitional cell carcinoma involving the prostate with a proposed staging classification for stromal invasion. J Urol. 1996;156(3):1071-1076.
Fasih N, Prasad Shanbhogue AK, et al. Leiomyomas beyond the uterus: unusual locations, rare manifestations. Radiographics. 2008;28(7):1931-1948.
Ferlay J, Autier P, Boniol M, et al. Estimates of the cancer incidence and mortality in Europe in 2006. Ann Oncol. 2007;18(3):581-592.
Fernandez-Gomez J, Rodriguez-Martinez JJ, Barmadah SE, et al. Urinary CYFRA 21.1 is not a useful marker for the detection of recurrences in the follow-up of superficial bladder cancer. Eur Urol. 2007;51:1267-1274.
Fletcher O, Easton D, Anderson K, et al. Lifetime risks of common cancers among retinoblastoma survivors. J Natl Cancer Inst. 2004;96(5):357-363.
Fradet Y, Grossman HB, Gomella L, et al. A comparison of hexaminolevulinate fluorescence cystoscopy and white light cystoscopy for the detection of carcinoma in situ in patients with bladder cancer: a phase III, multicenter study. J Urol. 2007;178(1):68-73. discussion 73
Gandini S, Botteri E, Iodice S, et al. Tobacco smoking and cancer: a meta-analysis. Int J Cancer. 2008;122(1):155-164.
Garcia-Closas M, Malats N, Silverman D, et al. NAT2 slow acetylation, GSTM1 null genotype, and risk of bladder cancer: results from the Spanish Bladder Cancer Study and meta-analyses. Lancet. 2005;366:649-659.
George B, Datar RH, Wu L, et al. p53 gene and protein status: the role of p53 alterations in predicting outcome in patients with bladder cancer. J Clin Oncol. 2007;25(34):5352-5358.
Gomez-Roman JJ, Saenz P, Molina M, et al. Fibroblast growth factor receptor 3 is overexpressed in urinary tract carcinomas and modulates the neoplastic cell growth. Clin Cancer Res. 2005;11(2 Pt. 1):459-465.
Gonzalez-Zulueta M, Bender CM, Yang AS, et al. Methylation of the 5′ CpG island of the p16/CDKN2 tumor suppressor gene in normal and transformed human tissues correlates with gene silencing. Cancer Res. 1995;55:4531-4535.
Grossman HB, Soloway M, Messing E, et al. Surveillance for recurrent bladder cancer using a point-of-care proteomic assay. JAMA. 2006;295(3):299-305.
Grossman HB, Stenzl A, Moyad MA, Droller MJ. Bladder cancer: chemoprevention, complementary approaches and budgetary considerations. Scand J Urol Nephrol Suppl. 2008;218:213-233.
Gudjonsson S, Isfoss BL, Hansson K, et al. The value of the UroVysion assay for surveillance of non-muscle-invasive bladder cancer. Eur Urol. 2008;54(2):402-408.
Guo Z, Linn JF, Wu G, et al. CDC91L1 (PIG-U) is a newly discovered oncogene in human bladder cancer. Nat Med. 2004;10(4):374-381.
Gutierrez J, Jimenez A, de Dios Luna J, et al. Meta-analysis of studies analyzing the relationship between bladder cancer and infection by human papillomavirus. J Urol. 2006;176(6 Pt. 1):2474-2481. discussion 2481
Hall MC, Chang SS, Dalbagni G, et al. Guideline for the management of nonmuscle invasive bladder cancer (stages Ta, T1, and Tis): 2007 update. J Urol. 2007;178(6):2314-2330.
Hall P. Radiation-associated urinary bladder cancer. Scand J Urol Nephrol Suppl. 2008;218:85-88.
Halling KC, King W, Sokolova IA, et al. A comparison of cytology and fluorescence in situ hybridization for the detection of urothelial carcinoma. J Urol. 2000;164:1768-1775.
Ham WS, Kim WT, Jeon HJ, et al. Long-term outcome of simultaneous transurethral resection of bladder tumor and prostate in patients with nonmuscle invasive bladder tumor and bladder outlet obstruction. J Urol. 2009;181(4):1594-1599.
Hanke M, Kausch I, Dahmen G, et al. Detailed technical analysis of urine RNA-based tumor diagnostics reveals ETS2/urokinase plasminogen activator to be a novel marker for bladder cancer. Clin Chem. 2007;53(12):2070-2077.
Hansen J, Jurik AG. Survival and radiation risk in patients obtaining more than six CT examinations during one year. Acta Oncol. 2009;48(2):302-307.
Harnden P, Eardley I, Joyce AD, Southgate J. Cytokeratin 20 as an objective marker of urothelial dysplasia. Br J Urol. 1996;78(6):870-875.
Hartmann A, Moser K, Kriegmair M, et al. Frequent genetic alterations in simple urothelial hyperplasias of the bladder in patients with papillary urothelial carcinoma. Am J Pathol. 1999;154(3):721-727.
Hasui Y, Osada Y. Urokinase-type plasminogen activator and its receptor in bladder cancer. J Natl Cancer Inst. 1997;89(10):678-679.
Hausladen DA, Wheeler MA, Altieri DC, et al. Effect of intravesical treatment of transitional cell carcinoma with bacillus Calmette-Guerin and mitomycin C on urinary survivin levels and outcome. J Urol. 2003;170:230-234.
Hayat MJ, Howlader N, Reichman ME, Edwards BK. Cancer statistics, trends, and multiple primary cancer analyses from the Surveillance, Epidemiology, and End Results (SEER) Program. Oncologist. 2007;12(1):20-37.
Herr HW, Donat SM. A comparison of white-light cystoscopy and narrow-band imaging cystoscopy to detect bladder tumour recurrences. BJU Int. 2008;102(9):1111-1114.
Herr HW, Donat SM. Prostatic tumor relapse in patients with superficial bladder tumors: 15-year outcome. J Urol. 1999;161(6):1854-1857.
Herr HW, Donat SM, Dalbagni G. Can restaging transurethral resection of T1 bladder cancer select patients for immediate cystectomy? J Urol. 2007;177(1):75-79. discussion 79
Hollenbeck BK, Ye Z, Dunn RL, et al. Provider treatment intensity and outcomes for patients with early-stage bladder cancer. J Natl Cancer Inst. 2009;101(8):571-580.
Holmang S, Andius P, Hedelin H, et al. Stage progression in Ta papillary urothelial tumors: relationship to grade, immunohistochemical expression of tumor markers, mitotic frequency and DNA ploidy. J Urol. 2001;165(4):1124-1128.
Holmang S, Johansson SL. The nested variant of transitional cell carcinoma—a rare neoplasm with poor prognosis. Scand J Urol Nephrol. 2001;35(2):102-105.
Hopman AH, Kamps MA, Speel EJ, et al. Identification of chromosome 9 alterations and p53 accumulation in isolated carcinoma in situ of the urinary bladder versus carcinoma in situ associated with carcinoma. Am J Pathol. 2002;161(4):1119-1125.
Hornigold N, Devlin J, Davies AM, et al. Mutation of the 9q34 gene TSC1 in sporadic bladder cancer. Oncogene. 1999;18(16):2657-2661.
Iczkowski KA, Shanks JH, Allsbrook WC, et al. Small cell carcinoma of urinary bladder is differentiated from urothelial carcinoma by chromogranin expression, absence of CD44 variant 6 expression, a unique pattern of cytokeratin expression, and more intense gamma-enolase expression. Histopathology. 1999;35:150-156.
Jemal A, Siegel R, Ward E, et al. Cancer statistics, 2008. CA Cancer J Clin. 2008;58(2):71-96.
Johansson SL, Borghede G, Holmang S. Micropapillary bladder carcinoma: a clinicopathological study of 20 cases. J Urol. 1999;161(6):1798-1802.
Jones TD, Zhang S, Lopez-Beltran A, et al. Urothelial carcinoma with an inverted growth pattern can be distinguished from inverted papilloma by fluorescence in situ hybridization, immunohistochemistry, and morphologic analysis. Am J Surg Pathol. 2007;31(12):1861-1867.
Kantor AF, Hartge P, Hoover RN, et al. Urinary tract infection and risk of bladder cancer. Am J Epidemiol. 1984;119(4):510-515.
Kashibuchi K, Tomita K, Schalken JA, et al. The prognostic value of E-cadherin, alpha-, beta- and gamma-catenin in bladder cancer patients who underwent radical cystectomy. Int J Urol. 2007;14(9):789-794.
Kaufman JM, Fam B, Jacobs SC, et al. Bladder cancer and squamous metaplasia in spinal cord injury patients. J Urol. 1977;118(6):967-971.
Kaye JA, Myers MW, Jick H. Acetaminophen and the risk of renal and bladder cancer in the general practice research database. Epidemiology. 2001;12(6):690-694.
Keesee SK, Briggman JV, Thill G, Wu YJ. Utilization of nuclear matrix proteins for cancer diagnosis. Crit Rev Eukaryot Gene Expr. 1996;6(2-3):189-214.
Khadra MH, Pickard RS, Charlton M, et al. A prospective analysis of 1,930 patients with hematuria to evaluate current diagnostic practice. J Urol. 2000;163(2):524-527.
Kiemeney LA. Hereditary bladder cancer. Scand J Urol Nephrol Suppl. 2008;218:110-115.
Kilciler M, Bedir S, Erdemir F, et al. Evaluation of urinary inverted papillomas: a report of 13 cases and literature review. Kaohsiung J Med Sci. 2008;24(1):25-30.
Knowles MA. Molecular subtypes of bladder cancer: Jekyll and Hyde or chalk and cheese? Carcinogenesis. 2006;27(3):361-373.
Knowles MA. Tumor suppressor loci in bladder cancer. Front Biosci. 2007;12:2233-2251.
Knowles MA. Bladder cancer subtypes defined by genomic alterations. Scand J Urol Nephrol Suppl. 2008;218:116-130.
Knowles MA. Molecular pathogenesis of bladder cancer. Int J Clin Oncol. 2008;13(4):287-297.
Knowles MA. Novel therapeutic targets in bladder cancer: mutation and expression of FGF receptors. Future Oncol. 2008;4(1):71-83.
Koed K, Wiuf C, Christensen LL, et al. High-density single nucleotide polymorphism array defines novel stage and location-dependent allelic imbalances in human bladder tumors. Cancer Res. 2005;65:34-45.
Kunju LP, You L, Zhang Y, et al. Lymphovascular invasion of urothelial cancer in matched transurethral bladder tumor resection and radical cystectomy specimens. J Urol. 2008;180(5):1928-1932. discussion 32
Kurahashi N, Inoue M, Iwasaki M, et al. Coffee, green tea, and caffeine consumption and subsequent risk of bladder cancer in relation to smoking status: a prospective study in Japan. Cancer Sci. 2008. Dec 4 [Epub ahead of print]
Kurth KH, Bouffioux C, Sylvester R, et al. Treatment of superficial bladder tumors: achievements and needs. The EORTC Genitourinary Group. Eur Urol. 2000;37(Suppl. 3):1-9.
Kuska B. Calories and cancer: can we starve our way to health? J Natl Cancer Inst. 2000;92(18):1466-1469.
La Vecchia C, Negri E, D’Avanzo B, et al. Genital and urinary tract diseases and bladder cancer. Cancer Res. 1991;51(2):629-631.
Lamm DL, Blumenstein BA, Crissman JD, et al. Maintenance bacillus Calmette-Guerin immunotherapy for recurrent TA, T1 and carcinoma in situ transitional cell carcinoma of the bladder: a randomized Southwest Oncology Group Study. J Urol. 2000;163(4):1124-1129.
Lamm DL, Riggs DR, Shriver JS, et al. Megadose vitamins in bladder cancer: a double-blind clinical trial. J Urol. 1994;151(1):21-26.
Lee CT, Madii R, Daignault S, et al. Cystectomy delay more than 3 months from initial bladder cancer diagnosis results in decreased disease specific and overall survival. J Urol. 2006;175(4):1262-1267. discussion 1267
Lengauer C, Kinzler KW, Vogelstein B. Genetic instabilities in human cancers. Nature. 1998;396(6712):643-649.
Lindgren D, Liedberg F, Andersson A, et al. Molecular characterization of early-stage bladder carcinomas by expression profiles, FGFR3 mutation status, and loss of 9q. Oncogene. 2006;25(18):2685-2696.
Linn JF, Sesterhenn I, Mostofi FK, Schoenberg M. The molecular characteristics of bladder cancer in young patients. J Urol. 1998;159(5):1493-1496.
Logie A, Dunois-Larde C, Rosty C, et al. Activating mutations of the tyrosine kinase receptor FGFR3 are associated with benign skin tumors in mice and humans. Hum Mol Genet. 2005;14(9):1153-1160.
Lokeshwar VB, Schroeder GL, Selzer MG, et al. Bladder tumor markers for monitoring recurrence and screening comparison of hyaluronic acid-hyaluronidase and BTA-Stat tests. Cancer. 2002;95:61-72.
Lopez-Beltran A. Bladder cancer: clinical and pathological profile. Scand J Urol Nephrol Suppl. 2008;218:95-109.
Lopez-Beltran A, Cheng L. Stage pT1 bladder carcinoma: diagnostic criteria, pitfalls and prognostic significance. Pathology. 2003;35(6):484-491.
Lopez-Beltran A, Cheng L. Histologic variants of urothelial carcinoma: differential diagnosis and clinical implications. Hum Pathol. 2006;37(11):1371-1388.
Lopez-Beltran A, Cheng L, Andersson L, et al. Preneoplastic non-papillary lesions and conditions of the urinary bladder: an update based on the Ancona International Consultation. Virchows Arch. 2002;440(1):3-11.
Lopez-Beltran A, Luque RJ, Alvarez-Kindelan J, et al. Prognostic factors in stage T1 grade 3 bladder cancer survival: the role of G1-S modulators (p53, p21Waf1, p27kip1, Cyclin D1, and Cyclin D3) and proliferation index (ki67-MIB1). Eur Urol. 2004;45:606-612.
Lotan Y, Gupta A, Shariat SF, et al. Lymphovascular invasion is independently associated with overall survival, cause-specific survival, and local and distant recurrence in patients with negative lymph nodes at radical cystectomy. J Clin Oncol. 2005;23(27):6533-6539.
Louhelainen JP, Hurst CD, Pitt E, et al. DBC1 re-expression alters the expression of multiple components of the plasminogen pathway. Oncogene. 2006;25(16):2409-2419.
Lovat F, Bitto A, Xu SQ, et al. Proepithelin is an autocrine growth factor for bladder cancer. Carcinogenesis. 2009;30(5):861-868.
Lumbreras B, Garte S, Overvad K, et al. Meat intake and bladder cancer in a prospective study: a role for heterocyclic aromatic amines? Cancer Causes Control. 2008;19(6):649-656.
Mallofre C, Castillo M, Morente V, Sole M. Immunohistochemical expression of CK20, p53, and Ki-67 as objective markers of urothelial dysplasia. Mod Pathol. 2003;16(3):187-191.
McBride OW, Swan DC, Santos E, et al. Localization of the normal allele of T24 human bladder carcinoma oncogene to chromosome 11. Nature. 1982;300(5894):773-774.
McGarvey TW, Maruta Y, Tomaszewski JE, et al. PTCH gene mutations in invasive transitional cell carcinoma of the bladder. Oncogene. 1998;17(9):1167-1172.
McKenney JK, Gomez JA, Desai S, et al. Morphologic expressions of urothelial carcinoma in situ: a detailed evaluation of its histologic patterns with emphasis on carcinoma in situ with microinvasion. Am J Surg Pathol. 2001;25:356-362.
Melicow MM, Hollowell JW. Intra-urothelial cancer: carcinoma in situ, Bowen’s disease of the urinary system: discussion of thirty cases. J Urol. 1952;68(4):763-772.
Michaud DS, Platz EA, Giovannucci E. Gonorrhoea and male bladder cancer in a prospective study. Br J Cancer. 2007;96(1):169-171.
Michaud DS, Spiegelman D, Clinton SK, et al. Prospective study of dietary supplements, macronutrients, micronutrients, and risk of bladder cancer in US men. Am J Epidemiol. 2000;152(12):1145-1153.
Millan-Rodriguez F, Chechile-Toniolo G, Salvador-Bayarri J. Multivariate analysis of the prognostic factors of primary superficial bladder cancer. J Urol. 2000;163:73-78.
Mishriki SF, Grimsley SJ, Nabi G. Incidence of recurrent frank hematuria and urological cancers: prospective 6.9 years of followup. J Urol. 2009;182:1294-1298.
Mishriki SF, Nabi G, Cohen NP. Diagnosis of urologic malignancies in patients with asymptomatic dipstick hematuria: prospective study with 13 years’ follow-up. Urology. 2008;71(1):13-16.
Mohan AK, Hauptmann M, Freedman DM, et al. Cancer and other causes of mortality among radiologic technologists in the United States. Int J Cancer. 2003;103(2):259-267.
Montironi R, Carmona E, Bollito E, et al. The pagetoid variant of bladder urothelial carcinoma in situ A clinicopathological study of 11 cases. Virchows Archiv. 2002;441(2):148-153.
Montironi R, Lopez-Beltran A. The 2004 WHO classification of bladder tumors: a summary and commentary. Int J Surg Pathol. 2005;13(2):143-153.
Moonen PM, Merkx GF, Peelen P, et al. UroVysion compared with cytology and quantitative cytology in the surveillance of non-muscle-invasive bladder cancer. Eur Urol. 2007;51:1275-1280. discussion 1280
Morelli O, Castellani D, Asciutti S, et al. Colon and gastric metastases from a primary signet-ring cell carcinoma of the urinary bladder. Dig Liver Dis. 2006;38(8):609-611.
Morris DS, Weizer AZ, Ye Z, et al. Understanding bladder cancer death: tumor biology versus physician practice. Cancer. 2009;115(5):1011-1020.
Morrison AS, Verhoek WG, Leck I, et al. Artificial sweeteners and bladder cancer in Manchester, U.K., and Nagoya, Japan. Br J Cancer. 1982;45(3):332-336.
Mufti GR, Singh M. Value of random mucosal biopsies in the management of superficial bladder cancer. Eur Urol. 1992;22(4):288-293.
Mukesh M, Cook N, Hollingdale AE, et al. Small cell carcinoma of the urinary bladder: a 15-year retrospective review of treatment and survival in the Anglian Cancer Network. BJU Int. 2009;103(6):747-752.
Murta-Nascimento C, Silverman DT, Kogevinas M, et al. Risk of bladder cancer associated with family history of cancer: do low-penetrance polymorphisms account for the increase in risk? Cancer Epidemiol Biomarkers Prev. 2007;16(8):1595-1600.
Neugut AI, Ahsan H, Robinson E, Ennis RD. Bladder carcinoma and other second malignancies after radiotherapy for prostate carcinoma. Cancer. 1997;79(8):1600-1604.
Newton R, Ribeiro T, Casabonne D, et al. Antibody levels against BK virus and prostate, kidney and bladder cancers in the EPIC-Oxford cohort. Br J Cancer. 2005;93(11):1305-1306.
Nilsson S, Ullen A. Chemotherapy-induced bladder cancer. Scand J Urol Nephrol Suppl. 2008;218:89-92.
Novicki DE, Stern JA, Nemec R, Lidner TK. Cost-effective evaluation of indeterminate urinary cytology. J Urol. 1998;160(3 Pt. 1):734-736.
Obermann EC, Junker K, Stoehr R, et al. Frequent genetic alterations in flat urothelial hyperplasias and concomitant papillary bladder cancer as detected by CGH, LOH, and FISH analyses. J Pathol. 2003;199(1):50-57.
Oosterhuis JW, Schapers RF, Janssen-Heijnen ML, et al. Histological grading of papillary urothelial carcinoma of the bladder: prognostic value of the 1998 WHO/ISUP classification system and comparison with conventional grading systems. J Clin Pathol. 2002;55(12):900-905.
Orozco RE, Vander Zwaag R, Murphy WM. The pagetoid variant of urothelial carcinoma in situ. Hum Pathol. 1993;24:1199-1202.
Ortega LG, Whitmore WFJr, Murphy AI. In situ carcinoma of the prostate with intraepithelial extension into the urethra and bladder. Cancer. 1953;6(5):898-923.
Ozbey I, Aksoy Y, Polat O, et al. Squamous metaplasia of the bladder: findings in 14 patients and review of the literature. Int Urol Nephrol. 1999;31(4):457-461.
Ozdemir E, Kakehi Y, Okuno H, Yoshida O. Role of matrix metalloproteinase-9 in the basement membrane destruction of superficial urothelial carcinomas. J Urol. 1999;161(4):1359-1363.
Pagano F, Bassi P, Ferrante GL, et al. Is stage pT4a (D1) reliable in assessing transitional cell carcinoma involvement of the prostate in patients with a concurrent bladder cancer? A necessary distinction for contiguous or noncontiguous involvement. J Urol. 1996;155(1):244-247.
Paik SS, Park MH. The nested variant of transitional cell carcinoma of the urinary bladder. Br J Urol. 1996;78(5):793-794.
Palou J, Baniel J, Klotz L, et al. Urothelial carcinoma of the prostate. Urology. 2007;69(Suppl. 1):50-61.
Papanicolaou GN, Marshall VF. Urine sediment smears as a diagnostic procedure in cancers of the urinary tract. Science. 1945;101(2629):519-520.
Parekh DJ, Jung C, O’Conner J, et al. Leiomyosarcoma in urinary bladder after cyclophosphamide therapy for retinoblastoma and review of bladder sarcomas. Urology. 2002;60(1):164.
Parkin DM. The global burden of urinary bladder cancer. Scand J Urol Nephrol Suppl. 2008;218:12-20.
Pelucchi C, Tavani A, La Vecchia C. Coffee and alcohol consumption and bladder cancer. Scand J Urol Nephrol Suppl. 2008;218:37-44.
Pfister C, Chautard D, Devonec M, et al. Immunocyt test improves the diagnostic accuracy of urinary cytology: results of a French multicenter study. J Urol. 2003;169:921-924.
Pham HT, Block NL, Lokeshwar VB. Tumor-derived hyaluronidase: a diagnostic urine marker for high-grade bladder cancer. Cancer Res. 1997;57:778-783.
Pierce DA, Shimizu Y, Preston DL, et al. Studies of the mortality of atomic bomb survivors. Report 12, Part I. Cancer: 1950-1990. Radiat Res. 1996;146(1):1-27.
Piper JM, Tonascia J, Matanoski GM. Heavy phenacetin use and bladder cancer in women aged 20 to 49 years. N Engl J Med. 1985;313(5):292-295.
Pode D, Alon Y, Horowitz AT, et al. The mechanism of human bladder tumor implantation in an in vitro model. J Urol. 1986;136(2):482-486.
Porcaro AB, D’Amico A, Ficarra V, et al. Nephrogenic adenoma of the urinary bladder: our experience and review of the literature. Urol Int. 2001;66(3):152-155.
Puzio-Kuter AM, Castillo-Martin M, Kinkade CW, et al. Inactivation of p53 and Pten promotes invasive bladder cancer. Genes Dev. 2009;23(6):675-680.
Qin J, Xie LP, Zheng XY, et al. A component of green tea, (-)-epigallocatechin-3-gallate, promotes apoptosis in T24 human bladder cancer cells via modulation of the PI3K/Akt pathway and Bcl-2 family proteins. Biochem Biophys Res Commun. 2007;354:852-857.
Quek ML, Nichols PW, Yamzon J, et al. Radical cystectomy for primary neuroendocrine tumors of the bladder: the University of Southern California experience. J Urol. 2005;174:93-96.
Radomski JL, Greenwald D, Hearn WL, et al. Nitrosamine formation in bladder infections and its role in the etiology of bladder cancer. J Urol. 1978;120(1):48-50.
Radosavljevic V, Jankovic S, Marinkovic J, Dokic M. Diet and bladder cancer: a case-control study. Int Urol Nephrol. 2005;37(2):283-289.
Ramos D, Navarro S, Villamon R. Cytokeratin expression patterns in low-grade papillary urothelial neoplasms of the urinary bladder. Cancer. 2003;97:1876-1883.
Reulen RC, Kellen E, Buntinx F, et al. A meta-analysis on the association between bladder cancer and occupation. Scand J Urol Nephrol Suppl. 2008;218:64-78.
Rhyu MS. Telomeres, telomerase, and immortality. J Natl Cancer Inst. 1995;87(12):884-894.
Rikken CH, van Helsdingen PJ, Kazzaz BA. Are biopsies from the prostatic urethra useful in patients with superficial bladder carcinoma? Br J Urol. 1987;59:145-147.
Ron E, Preston DL, Mabuchi K, et al. Cancer incidence in atomic bomb survivors. Part IV: comparison of cancer incidence and mortality. Radiat Res. 1994;137(Suppl. 2):S98-112.
Rosser CJ, Slaton JW, Izawa JI, et al. Clinical presentation and outcome of high-grade urinary bladder leiomyosarcoma in adults. Urology. 2003;61(6):1151-1155.
Sakamoto N, Tsuneyoshi M, Naito S, Kumazawa J. An adequate sampling of the prostate to identify prostatic involvement by urothelial carcinoma in bladder cancer patients. J Urol. 1993;149:318-321.
Sala M, Cordier S, Chang-Claude J, et al. Coffee consumption and bladder cancer in nonsmokers: a pooled analysis of case-control studies in European countries. Cancer Causes Control. 2000;11(10):925-931.
Samaratunga H, Khoo K. Micropapillary variant of urothelial carcinoma of the urinary bladder; a clinicopathological and immunohistochemical study. Histopathology. 2004;45(1):55-64.
Samaratunga H, Makarov DV, Epstein JI. Comparison of WHO/ISUP and WHO classification of noninvasive papillary urothelial neoplasms for risk of progression. Urology. 2002;60(2):315-319.
Sanchez-Carbayo M, Socci ND, Richstone L, et al. Genomic and proteomic profiles reveal the association of gelsolin to TP53 status and bladder cancer progression. Am J Pathol. 2007;171(5):1650-1658.
Sanchini MA, Gunelli R, Nanni O, et al. Relevance of urine telomerase in the diagnosis of bladder cancer. JAMA. 2005;294(16):2052-2056.
Sauter G, Algaba F, Amin A, et al. Nonivasive urothelial neoplasias: WHO classification of noninvasive papillary urothelial tumors. In: Eble J, Sauter G, Epstein JI, Sesterhenn I, editors. World Health Organization classification of tumors. Lyon (France): IARC Press, 2004.
Schabath MB, Spitz MR, Lerner SP, et al. Case-control analysis of dietary folate and risk of bladder cancer. Nutr Cancer. 2005;53(2):144-151.
Scosyrev E, Ely BW, Messing EM, et al. Do mixed histological features affect survival benefit from neoadjuvant platinum-based combination chemotherapy in patients with locally advanced bladder cancer? A secondary analysis of Southwest Oncology Group-Directed Intergroup Study (S8710). BJU Int. 2010. [Epud ahead of print]
Semins MJ, Schoenberg MP. A case of florid cystitis glandularis. Nat Clin Pract Urol. 2007;4(6):341-345.
Shariat SF, Casella R, Khoddami SM, et al. Urine detection of survivin is a sensitive marker for the noninvasive diagnosis of bladder cancer. J Urol. 2004;171(2 Pt. 1):626-630.
Sheinfeld J, Reuter VE, Melamed MR, et al. Enhanced bladder cancer detection with the Lewis X antigen as a marker of neoplastic transformation. J Urol. 1990;143(2):285-288.
Sigurdson AJ, Doody MM, Rao RS, et al. Cancer incidence in the US radiologic technologists health study, 1983-1998. Cancer. 2003;97(12):3080-3089.
Simon R, Jones PA, Sidransky D, et al. Genetics and predictive factors of noninvasive urothelial neoplasias. WHO classification of noninvasive papillary urothelial tumors. In: Eble J, Sauter G, Epstein JI, Sesterhenn I, editors. World Health Organization classification of tumors. Lyon (France): IARC Press, 2004.
Smith AK, Hansel DE, Jones JS. Role of cystitis cystica et glandularis and intestinal metaplasia in development of bladder carcinoma. Urology. 2008;71(5):915-918.
Smith SD, Wheeler MA, Plescia J, et al. Urine detection of survivin and diagnosis of bladder cancer. JAMA. 2001;285:324-328.
Smits G, Schaafsma E, Kiemeney L, et al. Microstaging of pT1 transitional cell carcinoma of the bladder: identification of subgroups with distinct risks of progression. Urology. 1998;52(6):1009-1013. discussion 1013–14
Smoke IAfRoCT. Tobacco Smoke and Involuntary Smoking. Lyon (France): IARC Press; 2004.
Soloway MS, Briggman V, Carpinito GA, et al. Use of a new tumor marker, urinary NMP22, in the detection of occult or rapidly recurring transitional cell carcinoma of the urinary tract following surgical treatment. J Urol. 1996;156:363-367.
Soloway MS, Masters S. Urothelial susceptibility to tumor cell implantation: influence of cauterization. Cancer. 1980;46(5):1158-1163.
Sont WN, Zielinski JM, Ashmore JP, et al. First analysis of cancer incidence and occupational radiation exposure based on the National Dose Registry of Canada. Am J Epidemiol. 2001;153(4):309-318.
Sontag JM. Experimental identification of genitourinary carcinogens. Urol Clin North Am. 1980;7(3):803-814.
Sozen S, Akbal C, Sokmensuer C, et al. Microstaging of pT1 transitional cell carcinoma of the bladder. Does it really differentiate two populations with different prognoses? (pT1 subcategory). Urol Int. 2002;69(3):200-206.
Spiess PE, Kassouf W, Steinberg JR, et al. Review of the M.D. Anderson experience in the treatment of bladder sarcoma. Urol Oncol. 2007;25(1):38-45.
Spruck CH3rd, Ohneseit PF, Gonzalez-Zulueta M, et al. Two molecular pathways to transitional cell carcinoma of the bladder. Cancer Res. 1994;54(3):784-788.
Staack A, Schlechte H, Sachs M, et al. Clinical value of vesical leukoplakia and evaluation of the neoplastic risk by mutation analyses of the tumor suppressor gene TP53. Int J Urol. 2006;13(8):1092-1097.
Steiner G, Schoenberg MP, Linn JF, et al. Detection of bladder cancer recurrence by microsatellite analysis of urine. Nat Med. 1997;3:621-624.
Steinmaus CM, Nunez S, Smith AH. Diet and bladder cancer: a meta-analysis of six dietary variables. Am J Epidemiol. 2000;151(7):693-702.
Sung M-T, Maclennan GT, Lopez-Beltran A, et al. Natural history of urothelial inverted papilloma. Cancer. 2006;107(11):2622-2627.
Sylvester RJ, Oosterlinck W, van der Meijden AP. A single immediate postoperative instillation of chemotherapy decreases the risk of recurrence in patients with stage Ta T1 bladder cancer: a meta-analysis of published results of randomized clinical trials. J Urol. 2004;171:2186-2190.
Thelen A, Schaeuble J. [Simultaneous occurrence of bladder papilloma in uniovular twins.]. Z Urol. 1957;50(4):188-195.
Theodorescu D, Laderoute KR, Calaoagan JM, Guilding KM. Inhibition of human bladder cancer cell motility by genistein is dependent on epidermal growth factor receptor but not p21ras gene expression. Int J Cancer. 1998;78(6):775-782.
Thompson DE, Mabuchi K, Ron E, et al. Cancer incidence in atomic bomb survivors. Part II: solid tumors, 1958–1987. Radiat Res. 1994;137(Suppl. 2):S17-S67.
Torenbeek R, Koot RA, Blomjous CE, et al. Primary signet-ring cell carcinoma of the urinary bladder. Histopathology. 1996;28(1):33-40.
Travis LB, Curtis RE, Glimelius B, et al. Bladder and kidney cancer following cyclophosphamide therapy for non-Hodgkin’s lymphoma. J Natl Cancer Inst. 1995;87(7):524-530.
Tse V, Khadra M, Eisinger D, et al. Nephrogenic adenoma of the bladder in renal transplant and non-renal transplant patients: a review of 22 cases. Urology. 1997;50(5):690-696.
Ugurlu O, Gonulalan U, Adsan O, et al. Effects of simultaneous transurethral resection of prostate and solitary bladder tumors smaller than 3 cm on oncologic results. Urology. 2007;70(1):55-59.
Vaidyanathan S, McDicken IW, Mansour P, et al. Detection of early squamous metaplasia in bladder biopsies of spinal cord injury patients by immunostaining for cytokeratin 14. Spinal Cord. 2003;41(8):432-434.
van der Aa MN, Zwarthoff EC, Steyerberg EW, et al. Microsatellite analysis of voided-urine samples for surveillance of low-grade non-muscle-invasive urothelial carcinoma: feasibility and clinical utility in a prospective multicenter study (Cost-Effectiveness of Follow-up of Urinary Bladder Cancer trial [CEFUB]). Eur Urol. 2009;55(3):659-667.
van der Kwast TH. How to combine the molecular profile with the clinicopathological profile of urothelial neoplastic lesions. Scand J Urol Nephrol Suppl. 2008;218:175-184.
van Oers JM, Lurkin I, van Exsel AJ, et al. A simple and fast method for the simultaneous detection of nine fibroblast growth factor receptor 3 mutations in bladder cancer and voided urine. Clin Cancer Res. 2005;11(21):7743-7748.
van Rhijn BW, van der Kwast TH, Vis AN, et al. FGFR3 and P53 characterize alternative genetic pathways in the pathogenesis of urothelial cell carcinoma. Cancer Res. 2004;64(6):1911-1914.
van Rhijn BW, van der Poel HG, van der Kwast TH. Urine markers for bladder cancer surveillance: a systematic review. Eur Urol. 2005;47(6):736-748.
Vena JE, Freudenheim J, Graham S, et al. Coffee, cigarette smoking, and bladder cancer in western New York. Ann Epidemiol. 1993;3(6):586-591.
Vena JE, Graham S, Freudenheim J, et al. Drinking water, fluid intake, and bladder cancer in western New York. Arch Environ Health. 1993;48(3):191-198.
Vlahou A, Schellhammer PF, Mendrinos S, et al. Development of a novel proteomic approach for the detection of transitional cell carcinoma of the bladder in urine. Am J Pathol. 2001;158:1491-1502.
Vogelstein B, Kinzler KW. Cancer genes and the pathways they control. Nat Med. 2004;10(8):789-799.
Volpe A, Racioppi M, D’Agostino D, et al. Bladder tumor markers: a review of the literature. Int J Biol Markers. 2008;23(4):249-261.
Vriesema JL, Poucki MH, Kiemeney LA, Witjes JA. Patient opinion of urinary tests versus flexible urethrocystoscopy in follow-up examination for superficial bladder cancer: a utility analysis. Urology. 2000;56(5):793-797.
Wakai K, Hirose K, Takezaki T, et al. Foods and beverages in relation to urothelial cancer: case-control study in Japan. Int J Urol. 2004;11(1):11-19.
Wan J, Grossman HB. Bladder carcinoma in patients age 40 years or younger. Cancer. 1989;64(1):178-181.
Wang R, Morris DS, Tomlins SA, et al. Development of a multiplex quantitative PCR signature to predict progression in non-muscle-invasive bladder cancer. Cancer Res. 2009;69(9):3810-3818.
Wang Y, Hung SC, Linn JF, et al. Microsatellite-based cancer detection using capillary array electrophoresis and energy-transfer fluorescent primers. Electrophoresis. 1997;18:1742-1749.
Wanner P, Bouchardy C, Parkin DM. [Cancer mortality among Swiss migrants in France]. Rev Epidemiol Sante Publique. 1995;43(1):26-36.
Weir JM, Dunn JEJr. Smoking and mortality: a prospective study. Cancer. 1970;25(1):105-112.
Westenend PJ, Stoop JA, Hendriks JG. Human papillomaviruses 6/11, 16/18 and 31/33/51 are not associated with squamous cell carcinoma of the urinary bladder. BJU Int. 2001;88(3):198-201.
Wolff EM, Liang G, Jones PA. Mechanisms of disease: genetic and epigenetic alterations that drive bladder cancer. Nat Clin Pract Urol. 2005;2(10):502-510.
Wood DPJr, Montie JE, Pontes JE, Levin HS. Identification of transitional cell carcinoma of the prostate in bladder cancer patients: a prospective study. J Urol. 1989;142(1):83-85.
Wood DPJr, Montie JE, Pontes JE, et al. Transitional cell carcinoma of the prostate in cystoprostatectomy specimens removed for bladder cancer. J Urol. 1989;141(2):346-349.
Wood DPJr, Streem SB, Levin HS. Nephrogenic adenoma in patients with transitional cell carcinoma of the bladder receiving intravesical thiotepa. J Urol. 1988;139(1):130-131.
Wynder EL, Goldsmith R. The epidemiology of bladder cancer: a second look. Cancer. 1977;40(3):1246-1268.
Yates DR, Rehman I, Meuth M, et al. Methylational urinalysis: a prospective study of bladder cancer patients and age stratified benign controls. Oncogene. 2006;25(13):1984-1988.
Yoder BJ, Skacel M, Hedgepeth R, et al. Reflex UroVysion testing of bladder cancer surveillance patients with equivocal or negative urine cytology: a prospective study with focus on the natural history of anticipatory positive findings. Am J Clin Pathol. 2007;127:295-301.
Zanetta G, Rota SM, Lissoni A, et al. Conservative treatment followed by chemotherapy with doxorubicin and ifosfamide for cervical sarcoma botryoides in young females. Br J Cancer. 1999;80(3-4):403-406.
Zeegers MP, Goldbohm RA, van den Brandt PA. A prospective study on active and environmental tobacco smoking and bladder cancer risk (The Netherlands). Cancer Causes Control. 2002;13(1):83-90.
Zeegers MP, Swaen GM, Kant I, et al. Occupational risk factors for male bladder cancer: results from a population based case cohort study in the Netherlands. Occup Environ Med. 2001;58(9):590-596.
Zeegers MP, Volovics A, Dorant E, et al. Alcohol consumption and bladder cancer risk: results from The Netherlands Cohort Study. Am J Epidemiol. 2001;153(1):38-41.
Zhang S, Li Y, Li L, et al. Phase I study of repeated intraepithelial delivery of adenoviral p53 in patients with dysplastic oral leukoplakia. J Oral Maxillofac Surg. 2009;67(5):1074-1082.
Zwarthoff EC. Detection of tumours of the urinary tract in voided urine. Scand J Urol Nephrol Suppl. 2008;218:147-153.