chapter 82 Management of Metastatic and Invasive Bladder Cancer
Clinical Presentation, Diagnosis, And Evaluation
Significant progress has been made in the management of invasive cancer, and, along with our increased knowledge of multimodal therapy, a multidisciplinary approach to the evaluation and management has become increasingly imperative. The urologist, medical oncologist, and radiation oncologist play a central role in integrating the disciplines of surgery, chemotherapy, and radiation therapy in order to achieve the goal of long-term cancer control. This chapter focuses on the evaluation and treatment of patients with muscle-invasive and metastatic bladder cancer.
Natural History
The majority (80%) of patients with bladder cancer present de novo with muscle-invasive disease as its first manifestation. The remaining 15% to 20% progress from non–muscle-invasive cancer after treatment with intravesical therapy. Deaths due to bladder cancer invariably occur as a result of distant metastases present at the time of loco-regional therapy. Progression of cancer after definitive loco-regional therapy commonly occurs within the first 2 years after treatment. Late recurrences are more common after perioperative systemic chemotherapy and often occur in unusual sites including the central nervous system, bowel serosa, and peritoneum. Muscle-invasive bladder cancer must be considered as a systemic disease and therapeutic strategies must be designed around the integration of treatment targeting the loco-regional disease (bladder and pelvic lymph nodes) and occult visceral metastatic disease according to pathologic risk factors.
Histology
Primary cancers of the bladder arise from the urothelium, and by far the most common cell type is transitional cell. Squamous cell predominates in Middle Eastern countries where bilharziasis is endemic. It occurs more frequently in Western countries in women and in the setting of chronic indwelling catheters and recurrent infection. As the prevalence of bilharziasis has diminished significantly, transitional cell carcinoma (TCC) now accounts for more than one half of bladder cancer diagnosed in Egypt (el-Mekresh et al, 2009). Adenocarcinomas of the bladder are uncommon but occur in the bladder base/trigone area. Patients with exstrophy are at increased risk. Surgical treatment of urachal adenocarcinoma is partial cystectomy, and the biology is similar to that of colorectal adenocarcinoma. Small cell neuroendocrine cancer is also a rare variant and may be the predominant histology or may be mixed with TCC (Mazzucchelli et al, 2009). This phenotype is easily distinguished by immunohistochemistry expressing synaptophysin and chromogranin. Treatment requires neoadjuvant chemotherapy (cisplatin and etoposide) followed by radical cystectomy or radiation therapy (Siefker-Radtke et al, 2004). Small cell tumors may also be associated with paraneoplastic syndromes including ectopic adrenocorticotropic hormone (ACTH) production, hypercalcemia, and hypophosphatemia. Other neuroendocrine tumors occur in the bladder and include carcinoid tumors, which may originate in the bladder or invade the bladder from the gastrointestinal tract, most commonly from the appendix. Large cell neuroendocrine tumors are rare with few cases reported in the literature and are believed to behave in a manner similar to that of small cell tumors (Akamatsu et al, 2008).
Other unusual variants reported have included lymphoepithelioma-like cancers, which have a better prognosis than high-grade invasive TCCs, although treatment is similar stage for stage. The micropapillary variant resembles ovarian papillary serous cancer and is aggressive. It tends toward high grade and high stage and is frequently associated with lymphatic and vascular invasion (Kamat et al, 2006). Radical cystectomy is the treatment of choice because this phenotype typically does not respond to chemotherapy directed at TCC. Rhabdomyosarcoma is seen in both children and adults, and leiomyosarcoma is seen in adults. Primary lymphoma of the bladder is rare, but the bladder may be involved in up to 10% of cases of systemic lymphoma.
Clinical and Pathologic Staging
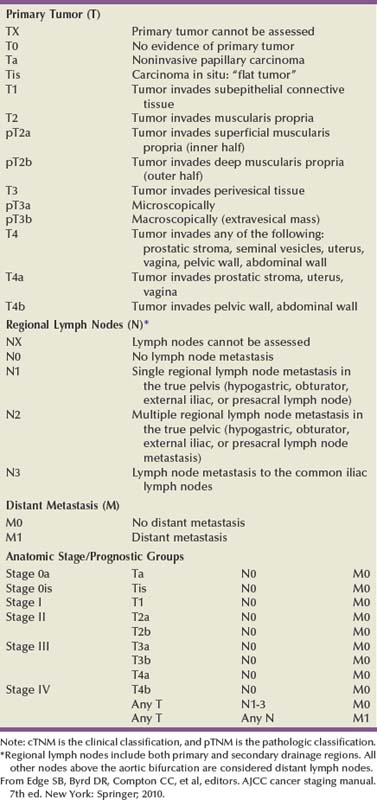
The T stage denotes the clinical stage, which is determined before definitive therapy. The depth of invasion determined on the transurethral resection of the bladder tumor (TURBT) specimen and the bimanual examination under anesthesia determine the clinical stage. Pathologic stage is derived from the cystectomy specimen. Clinical understaging is more common than overstaging (which does occur, however).
The 1997 TNM staging schema introduced a modification of prior staging schemes. Before 1997, T2 described disease in the inner half of muscle and T3a described disease in the outer half. T3b was used to identify all levels of perivesical fat invasion. In the current system T2a and T2b are used to designate the inner and outer half of the muscle, respectively, and the T3 category is split into T3a (microscopic) and T3b (macroscopic, or visible on gross inspection of the cystectomy specimen). The T4a prostate designation requires established stromal invasion, which can occur via the urethra or as direct extension via the bladder neck or posteriorly into the seminal vesicles or periprostatic tissue. Finding carcinoma in situ (CIS) in the prostatic urethra or ducts does not lead to upstaging because outcome is determined by the primary bladder cancer stage (Esrig et al, 1996; Pagano et al; 1996). Staging of nodes and metastases is similar to that for other genitourinary malignancies. Positive nodes above the common iliac bifurcation are classified as N+M1 disease. The UICC and AJCC staging systems were uniform in the sixth edition of the American Joint Committee on Cancer (AJCC) Staging Manual (Greene et al, 2002). An updated seventh edition includes the number of nodes removed as a quality measure of radical cystectomy. In addition, node metastases above the common iliac bifurcation are classified as N3 rather than M1 (Table 82–1) (Edge et al, 2010).
Staging Pitfalls
T1 versus T2
European Association of Urology (EAU) Guidelines require muscularis propria in the TURBT specimen for accurate staging of invasive cancers. In the absence of muscularis propria, understaging of T1 disease is common, and, unfortunately, this leads to worse outcomes for patients with T1 cancer (Dutta et al, 2001). True, T1 is often mistaken for T2 because there are fine muscle bundles called muscularis mucosa present within the lamina propria (Fig. 82–1A and B). There are blood vessels in this area as well. These muscle bundles may be confused with the muscularis propria and lead to overstaging of a T1 tumor as T2. In addition, recent data suggest that up to 40% of patients initially diagnosed with T1 who undergo cystectomy have pathologic T2 disease (Dutta et al, 2001). Because the management of patients with high-grade T1 tumors is challenging, it is imperative to stage these patients accurately before embarking on intravesical therapy. We therefore routinely re-resect T1G3 tumors before determining appropriate therapy even when we performed the original resection. Presence of persistent T1 cancer on re-resection is a highly significant predictor of poor outcome in patients treated with bacillus Calmette-Guérin (BCG) and is an indication for early cystectomy (Herr et al, 2007). Additional indications for initial cystectomy in patients with T1 disease include size greater than 3 cm, micropapillary histology, and lymphovascular invasion.
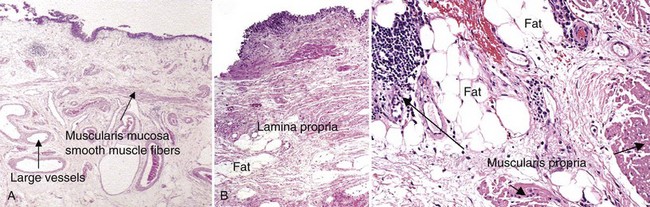
Figure 82–1 A, Smooth muscle fibers can be observed in the superficial and deep portion of the lamina propria and are not to be confused with muscularis propria invasion. Large vessels may be found throughout the lamina propria as well. B, Fat can be observed in lamina propria and deep portions of the muscularis propria and should not be confused with perivesical fat invasion.
T2 versus T3
Fat can be observed in the lamina propria and deep portions of the muscularis propria and should not be confused with perivesical fat invasion (see Fig. 82–1B). In stage T3, cancer appears in fat in the absence of muscle bundles.
Staging Tumors of the Bladder and Urethra
The goals in staging the primary tumor are to establish histology; look for lymphatic/vascular invasion, which is a risk factor for metastasis; and determine the depth of penetration through a combination of biopsy and bimanual examination. When cystectomy is anticipated, complete resection is not necessary and the risk of bladder perforation is avoided. Downstaging, however, may provide some benefit because retrospective studies and secondary analysis of randomized neoadjuvant chemotherapy trials suggest that survival may be improved when the pathologic tumor stage is less than the precystectomy clinical stage for clinical stage T2 patients (Grossman et al, 2003a; Sonpavde et al, 2009). Patients who are P0 at cystectomy may progress, but at a lower rate than patients with residual invasive cancer (Grossman et al, 2003a).
One must also determine whether the local tumor is causing upper tract obstruction and whether there is a concomitant upper tract tumor that also needs treatment. This is accomplished by imaging with computed tomography, magnetic resonance imaging (MRI), and/or retrograde ureteropyelography. With the patient anesthetized, the T stage of the tumor is identified with a combination of transurethral resection and bimanual examination. Findings described originally by Marshall in 1952 are T2a—nonpalpable; T2b—induration but no three-dimensional mass; T3—a three-dimensional mass that is mobile; T4a—invading adjacent structures such as the prostate, vagina, or rectum; T4b—fixed to pelvic sidewall and not mobile (Marshall, 1952). Because tumors of the trigone or those encroaching on the bladder neck in men may directly invade the prostatic stroma, additional tissue should be obtained at resection for accurate staging because this may affect decisions regarding neoadjuvant chemotherapy. The utility of site-directed biopsies or biopsy of normal-appearing, remote mucosa is controversial in non–muscle-invasive cancer (Kiemeney et al, 1994; van der Meijden et al, 1999). However, with high-grade muscle-invasive cancers, at least one half of patients have associated CIS that may affect treatment decisions and outcome if remote from the index tumor (Shariat et al, 2007). The last piece of information needed is the status of the bladder neck (female) or prostatic urethra (male) to serve as a basis for planning appropriate management of the urethra and urinary diversion.
Biopsies of the prostatic urethra may provide useful information before radical cystectomy (Lerner et al, 2008a). Using a resectoscope, the surgeon takes a full loop from the midprostate (or bladder neck in shorter prostates) to the mid to distal verumontanum and 5 and 7 o’clock adjacent to the verumontanum. This is the site of the highest concentration of prostatic ducts and the place where CIS is most likely to be found (Sakamoto et al, 1993). The full-thickness chip allows the pathologist to see the interface between the urethral mucosa, prostatic ducts, and stroma. This method is the most accurate means of staging the prostatic urethra (Wood et al, 1989). Negative prostatic urethra biopsies are associated with a negative apical urethral margin and obviate the need for intraoperative frozen section (Lerner and Shen, 2008b). In women, bladder neck biopsies are an accurate surrogate for urethral biopsy when removal of the distal two thirds of the urethra and orthotopic diversion is being considered.
Staging Evaluation: Regional and Metastatic Disease
Laboratory tests and radiographic imaging are directed at determining the effect of the local tumor on the upper urinary tract and the presence or absence of synchronous urothelial cancer of the ureter, renal pelvis, or urethra, and for evaluating the status of potential sites of metastatic disease. Laboratory testing at a minimum should include blood urea nitrogen (BUN), creatinine, electrolytes, complete blood count, and liver function tests. Imaging studies are directed to the most common sites of visceral metastasis, which include lung, liver, and bone. Following chemotherapy, metastasis may occur in unusual sites including the central nervous system and the peritoneum (with late presentation of bowel obstruction), but these sites are generally not a focus of staging at initial presentation.
Computed tomography (CT) has largely replaced intravenous urography for upper tract imaging. CT provides the added benefit of cross-sectional imaging of the pelvic, iliac, and retroperitoneal lymph nodes, as well as the liver. The incidence of pelvic node metastases is directly related to the depth of invasion of the primary tumor (Table 82–2) (Stein et al, 2001; Leissner et al, 2004; Vazina et al, 2004; Steven and Poulsen, 2007; Ghoneim et al, 2008) and the presence of lymphovascular invasion (Quek et al, 2005).
Table 82–2 Incidence of Pathologic Pelvic Node Metastasis (%) at Radical Cystectomy in Selected Contemporary Series (2000-2009)

Histologic confirmation of metastatic disease is desirable because its presence has a significant effect on treatment decisions and evaluating response to subsequent therapy. A chest radiograph is important for evaluating the lungs and mediastinum. A chest CT is obtained to further define abnormalities and should be obtained routinely in patients with muscle-invasive disease or N+ disease in whom the risk of visceral metastasis is greatest. Similarly, bone scintigraphy is obtained in these highest-risk patients and in patients with an elevated alkaline phosphatase or new-onset bone pain. An MRI may be useful for patients with an allergy to iodinated contrast or to resolve questions about an equivocal bone scan. The introduction of novel contrast agents with MRI may provide a more sensitive means for detecting nodal metastases. Ferromagnetic nanoparticles, for example, are taken up by macrophages, and dark areas represented by metastatic deposits can be detected with foci as small as 3 mm (Deserno et al, 2004). These studies, which require a 3 Tesla magnet, need further study and validation.
Positron emission tomography (PET) is used increasingly in oncology. Combination with CT is now standard and thus provides more accurate localization of abnormalities. Although there is as yet no defined role for routine staging of nodal or visceral metastases in invasive bladder cancer, PET/CT can be particularly helpful in discerning nodal or visceral metastatic disease when these findings will determine the use of chemotherapy or surgery (Kibel et al, 2009).
Several commercially available serum biomarkers (CEA, CA-125, CA-19.9) have been reported to be elevated in patients with advanced disease and may provide a marker that correlates with disease activity and response to therapy, but they are not standard (Margel et al, 2006; Kouba et al, 2009).
Treatment: Surgical
Radical Cystectomy and Bilateral Pelvic and Iliac Lymphadenectomy
The goal of therapy is complete eradication of the loco-regional disease including the primary tumor and regional lymph nodes. Neoadjuvant or adjuvant chemotherapy targeting occult regional node and/or visceral metastases should be integrated with loco-regional therapy when the risk of this occurrence is sufficiently high. Patients should have volitional control of urination via the retained urethra or by a continent catheterizable stoma in properly selected patients, and treatment-related morbidity should be minimized wherever possible.
Radical cystectomy provides excellent local control of the primary tumor and should include the bladder and surrounding perivesical soft tissue, prostate, and seminal vesicles in men and the ovaries, uterus/cervix, and anterior vagina in women. In sexually active women, vaginal preservation and/or reconstruction must be discussed and planned preoperatively. Recent reports challenge the dogma of removal of the internal female organs because involvement of the uterus, cervix, and ovaries is uncommon (Chang et al, 2002). Preservation of the vagina and uterus provides better support for a neobladder and the pelvic floor when the extent of the cancer or the age and general health status of the patient does not warrant anterior pelvic exenteration (Ali-El-Dein et al, 2002). Involvement of the urethra or bladder neck is an absolute contraindication to sparing the urethra in women, and a posterior-based invasive cancer is a relative contraindication.
Extent of Pelvic and Iliac Node Dissection
Lymph node (LN) metastasis is found in 20% to 25% of patients who undergo radical cystectomy (RC) and pelvic lymphadenectomy for bladder cancer, and it is the most important prognostic factor in these patients, predicting significantly decreased recurrence-free survival and overall survival compared with that for patients without nodal metastases (Lerner et al, 1993; Poulsen et al, 1998; Stein et al, 2001). In node-negative patients, the total number of LNs removed and the anatomic extent of the node dissection are both useful measures in evaluating the proper extent of surgery and predicting outcome. In patients with nodal metastasis, the number of nodes removed and the number and percent of positive nodes may both be independent predictors of recurrence and survival (Leissner et al, 2000; Herr et al, 2002; Herr, 2003; Stein et al, 2003b).
What Constitutes an Adequate Lymph Node Dissection?
The extent of the lymph node dissection as it relates to cancer control and detection of lymph node metastases is an important area for research. The minimum number of lymph nodes to be removed for accurate staging and therapeutic value in bladder cancer has yet to be defined (Mills et al, 2001; Vazina et al, 2004). The unfortunate reality revealed in the Surveillance, Epidemiology and End Results (SEER) database in 2003 was that the majority of patients in a population-based analysis had four or fewer nodes removed with cystectomy (Konety et al, 2003; Hollenbeck et al, 2008). Some improvement had occurred by 2004 with an increase in the number of nodes removed and fewer patients undergoing no node dissection at all (Hellenthal et al, 2009).
The wide range of total lymph node dissections reported may reflect a lack of standardization of the extent of node dissection, different sampling techniques, and differences in processing of tissue by the pathologist (en bloc removal vs. presentation in packets). Bochner and colleagues (2001, 2004) demonstrated that simply presenting the nodes to the pathologist in packets rather than en bloc with the cystectomy specimen increased the node count. This has been corroborated by others and should be considered the standard of care (Stein et al, 2007; Ather et al, 2008). Node dissection that includes the common iliac, presacral, and precaval/preaortic nodes may result in a higher number of lymph nodes removed (Vazina et al, 2004).
Patient’s age and comorbidities are factors that may affect a surgeon’s decision about the extent of LN dissection, thus influencing the number of LNs retrieved (Bochner et al, 2004; Vazina et al, 2004). A recent study, although small, suggested that in a single institution an individual surgeon had an independent impact on number of nodes removed (Kulkarni et al, 2008). This is not surprising because significant variability in the extent of the node dissection and node counts has been observed in prospective studies requiring a predefined anatomic extent of lymphadenectomy (Leissner et al, 2004; Lerner et al, 2009). Hospital and patient mix affecting comorbidities may affect node counts and mortality associated with cystectomy (Hollenbeck et al, 2008).
Staging
The current TNM staging system distinguishes between number of nodes with metastases, the size of nodal metastases, and the anatomic location, with nodal metastasis above the common iliac bifurcation denoting M1 disease and thus equivalence to visceral metastasis. The seventh edition of the American Joint Committee on Cancer (AJCC) Staging Manual (released October 2009) notes that at least 12 nodes should be removed. Most studies comparing extended and “standard” nodal dissection or packet presentation of the lymph nodes suggest that the overall incidence of node positivity is not altered, but, in fact, the number of nodes examined may be increased by extending the anatomic extent of the dissection (Poulsen et al, 1998; Vazina et al, 2004; Lerner et al, 2009; Edge et al, 2010).
With the focus on number of nodes removed and number of positive nodes as prognostic factors, investigators have developed a combined variable of lymph node density (LND), which takes both of these variables into account. Separate reports from the University of Southern California (USC) and Memorial Sloan-Kettering Cancer Center (MSKCC) published in 2003 described this and determined that LND (positive nodes/total nodes removed) had prognostic significance independent of the prognostic value of the number of positive nodes, pathologic tumor stage, and history of adjuvant chemotherapy (Herr, 2003; Stein et al, 2003b). The prognostic importance of LND has been corroborated in several retrospectively evaluated series (Kassouf et al, 2008; Osawa et al, 2009). An important caveat is that LND is affected by all of the factors described earlier that potentially affect the quality of the node dissection and interpretation by the pathologist (Svatek and Shariat, 2008). Quek and Flanigan wrote an excellent review of this subject and concluded that the body of evidence, based largely on retrospective review, suggests that LN density is an important pathologic prognostic variable (Quek and Flanigan, 2009). As indicated later, prospective validation is important and results lead to improved prognostic models.
Is There a Threshold of Number of Nodes Removed in Order to Identify the Majority of N+ Patients?
Koppie and colleagues (2006) reported a study designed to determine if there is a minimum number of lymph nodes analyzed above which there is no improvement in survival. The cohort included 1121 patients from MSKCC accrued over a 14-year period, and the investigators determined that there was no plateau in the dose-response curve with increasing number of nodes up to 23 nodes because few had 24 or more nodes removed. The authors did not indicate the percent of patients who underwent an extended node dissection, and, in fact, 13% had no nodes identified in the pathology report. The median number of nodes removed was 9. Capitanio and colleagues (2009) reported a similar analysis from the Bladder Cancer Research Consortium (BCRC), a three-center database including Baylor College of Medicine, University of Texas Southwestern, and Johns Hopkins University. The median number of nodes removed was 19, and all patients underwent at least a bilateral pelvic node dissection. Using receiver operator curve analysis, the authors determined that analysis of 45 nodes resulted in detection of 90% of patients with nodal metastasis. Removal of 25 nodes resulted in detection of 75% (Fig. 82–2). The largest gain in sensitivity was between 15 and 30 nodes, and there was only modest improvement in sensitivity with examination of more than 30 nodes. They suggested that 25 nodes be the minimum number analyzed, and these findings provide some insight as to why a threshold was not observed in the MSKCC series.
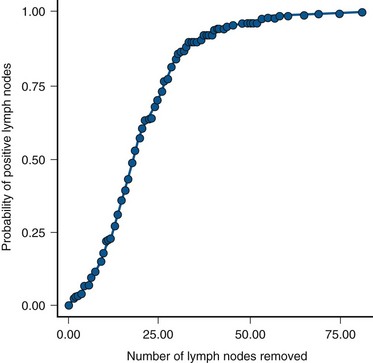
Figure 82–2 A plot describing the relationship between the number of lymph nodes removed and the probability of detecting lymph node metastasis.
(Reprinted with permission from Capitanio U, Suardi N, Shariat SF, et al. Assessing the minimum number of lymph nodes needed at radical cystectomy in patients with bladder cancer. BJU Int 2009;103:1359–62, Fig. 1, p. 1360.)
Outcome Related to Nodal Status
With so many factors contributing to the variability in node counts, one could argue that the anatomic extent and completeness of the node dissection deserve the most emphasis. It is clear that the more tissue removed, the more nodes a diligent pathologist will identify, and this should increase sensitivity in detecting nodal metastases. It is also clear that small-volume nodal disease is compatible with long-term survival, especially when the primary bladder cancer is organ confined. The integration of systemic chemotherapy either before or after radical cystectomy provides an incremental survival benefit, and a thorough node dissection is an integral component of this strategy (Herr et al, 2004). If there is a survival advantage associated with a more extensive node dissection, it is most likely due to the complete removal of all potential node-bearing tissue in the primary and secondary landing zones. This would lead to improved local and regional cancer control and to a more precise identification of patients with nodal metastasis who might benefit from adjuvant chemotherapy. Despite level 1 evidence supporting the use of neoadjuvant chemotherapy for muscle-invasive bladder cancer and a meta-analysis of adjuvant chemotherapy also supporting use of perioperative chemotherapy, recent estimates suggest that only 12% of patients are treated with an integrated treatment strategy of cystectomy and systemic chemotherapy (David et al, 2007). This reality requires that urologic surgeons performing radical cystectomy optimize the anatomic extent and completeness of the lymph node dissection.
Skinner and colleagues suggested in 1982 that “a meticulous pelvic node dissection can make a difference,” reporting 5-year survival of 36% in patients with node-positive disease (Skinner, 1982). In addition, they noted excellent local control and no increase in morbidity. These data were further supported in a larger series that also introduced the concept of lymph node density (Stein et al, 2003b). Poulsen and colleagues (1998) suggested that the extended node dissection as described by Skinner and colleagues was associated with better survival for patients with organ-confined cancers and patients with negative nodes than for patients with a pelvic dissection that included only the external and internal iliac and obturator nodes. Recently, Steven and Poulsen confirmed their earlier observation in a consecutive series of 336 patients who had an extended node dissection (Steven and Poulsen, 2007). Patients with lymph node metastases above the common iliac bifurcation had a survival probability similar to that of patients with nodal metastases confined to the pelvis (37% vs. 42%, respectively). An editorial commented that stage migration might account for some of the improvement in survival associated with an extended node dissection, particularly for patients with negative nodes with a more complete node dissection and meticulous pathologic evaluation (Poulsen et al, 1998). The question of whether an extended node dissection improves overall survival or whether that benefit applies only to particular subgroups should be addressed in the context of a randomized clinical trial.
Intraoperative Decision Making
Grossly Positive Nodes and T4b Disease
The management of patients with grossly involved lymph nodes includes treatment of systemic micrometastatic and loco-regional disease. If disease is recognized on preoperative imaging and outside the true pelvis, a CT-guided biopsy is performed to establish histology. If it is positive, then chemotherapy is initiated and followed by cystectomy. When adenopathy is encountered at the time of a cystectomy, a frozen section biopsy is undertaken to confirm the presence of metastasis, and extended lymph node dissection and radical cystectomy are performed. Cystectomy is not performed when (1) lymph node metastases are unresectable because of bulk or proximal extent above the common iliac vessels; (2) there is evidence of extensive periureteral disease; (3) the bladder is fixed to the pelvic sidewall; or (4) tumor is invading the rectosigmoid colon.
Carcinoma in Situ of the Ureter
Much has been written regarding the utility of intraoperative frozen section (FS) biopsy to identify CIS at the ureteral margin (Schoenberg et al, 1996; Silver et al, 1997; Balaji et al, 1999; Sved et al, 2004; Lee et al, 2006; Raj et al, 2006; Osman et al, 2007). Although previous retrospective studies suggested that FS lacked sufficient correlation with final margin status, contemporary studies both retrospective and prospective found high sensitivity and specificity and positive and negative predictive values for final margin status (Raj et al, 2006; Osman et al, 2007). There is little disagreement about whether every effort should be made to obtain a negative proximal margin before reimplantation when frank tumor is encountered at the margin. It is clear, moreover, that atypia and dysplasia do not require any action. In the case of CIS, an attempt is made to achieve a negative margin without compromising ureteral length because nephrectomy is not indicated for CIS of the ureter. Raj and colleagues, however, question the value of achieving a negative margin because this did not alter the risk of development of subsequent upper tract tumor (Raj et al, 2006). CIS of the ureter is not independently associated with a worse outcome following cystectomy (Lee et al, 2006). Cancer recurrence at the anastomosis is rare even with a positive margin showing CIS, but a positive margin is a risk factor for developing a second primary tumor of the ureter or renal pelvis (Lee et al, 2006; Raj et al, 2006). Upper tract tumors found after cystectomy are often at a more advanced stage and, particularly when symptomatic, may be associated with a worse prognosis (Balaji et al, 1999; Sved et al, 2004; Sanderson et al, 2007). Surveillance ureteroscopy is the most sensitive means for following patients with a positive ureteral margin, and long-term follow-up is required. The median time to occurrence in one recent series was 53 months (Wagner et al, 2008).
Prostatic Transitional Cell Carcinoma and Managing the Apical Urethral Margin
The risk of developing a second primary tumor of the retained urethra following cystectomy is influenced by the presence and extent of involvement of the prostatic urethra and prostatic stroma, with stromal invasion carrying the highest risk of approximately 30% (none < focal CIS < diffuse CIS < ductal/acinar involvement < stromal invasion) (Hardeman and Soloway, 1990). Urethrectomy should be considered in men if CIS is diffuse within the prostatic urethra or ducts or if there is invasion of prostatic stroma. One method for assessing the risk to the retained urethra and determining the appropriate choice of urinary diversion is to perform transurethral resection biopsies of the prostatic urethra (see “Staging the Bladder and Urethra” earlier) (Wood et al, 1989; Lerner and Shen, 2008b). Some experts, however, believe that the only absolute indication for consideration of urethrectomy is cancer at the apical urethral margin (Donat et al, 2001; Stein et al, 2005). Small low-grade papillary tumors of the urethra can be resected before cystectomy and the urethra retained if there is no other indication for urethrectomy. The probability of developing a second primary TCC of the retained urethra is lower with orthotopic diversion than with cutaneous diversion (Stein et al, 2005).
Managing the Female Urethra
Cystectomy in women has historically included the urethra. New data indicate that the distal two thirds of the urethra serves as an adequate sphincter mechanism innervated by the pudendal nerve and is infrequently involved with transitional cell cancer (Stenzl et al, 1995; Stein et al, 1998). Limiting dissection only to the soft tissue above the endopelvic fascia will preserve the innervation crucial to maintaining the sphincter function of the urethra. Cancer at the bladder neck or urethra or a T4 tumor involving the anterior vagina is a contraindication to urethral preservation and creation of an orthotopic neobladder. Posterior T3 tumors are a relative contraindication, particularly if the tumor involves the trigone, because achieving an adequate margin is difficult. Urethral cancer is always associated with bladder neck cancer, so findings of a biopsy of the bladder neck are an excellent surrogate for preoperative staging of the female urethra (Fig. 82–3). The proximal one third of the urethra is resected en bloc with the cystectomy specimen in order to reduce the risk of urinary retention, leaving the distal two thirds to provide adequate sphincter function.
Figure 82–3 Location of cancer in 65 female radical cystectomy specimens. Bladder neck involvement is a surrogate for urethra involvement (SPL, personal series). Biopsies of the bladder neck should be performed in order to determine the suitability of urethra preservation in women and orthotopic diversion. CIS, carcinoma in situ.
Survival and Treated Natural History
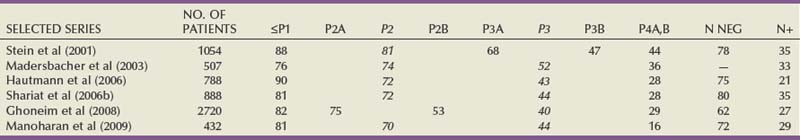
Pathologic tumor stage and nodal status are the primary variables affecting the risk of progression and survival probability following cystectomy (Table 82–3) (Stein et al, 2001; Madersbacher et al, 2003; Hautmann et al, 2006; Shariat et al, 2006b; Ghoneim et al, 2008; Manoharan et al, 2009). The incidence of nodal metastasis increases with depth of invasion, and lymphovascular invasion increases the risk of nodal metastasis and is an independent predictor of outcome (Quek et al, 2005; Algaba, 2006; Herrmann et al, 2008). Positive surgical margins have an adverse impact on outcome as well (Dotan et al, 2007; Sonpavde et al, 2009). Recent studies suggest that improved outcomes are associated with high-volume surgeons and hospitals and that patient’s age and body mass index (BMI) may also affect morbidity and survival (Konety et al, 2005; Nielsen et al, 2007; Bagrodia et al, 2009).
Table 82–3 Percentage 5-Year Disease-Specific Survival (DSS) by Pathologic Stage after Radical Cystectomy with and without Pelvic Lymph Node Metastasis: Selected Series Reporting DSS (2000-2009)
Up to 50% of patients with infiltrating disease develop metastases and ultimately succumb to their disease. Failure is usually due to occult metastatic disease present at the time of initial diagnosis. Long-term survival remains low and chemotherapy currently provides the potential for cure only in selected patients.
After metastases develop, there are few long-term survivors, although some 70% of patients have disease that is sensitive to chemotherapy. The suggestion is that systemic treatment in addition to radical cystectomy is required. Extensive lymph node dissection has both prognostic and therapeutic significance, and effective systemic therapies that eliminate micrometastases may improve outcome.
Perioperative chemotherapy is administered before (neoadjuvant) or after (adjuvant) cystectomy (bimodality therapy) to eradicate subclinical disease and primarily to improve survival. Concomitant chemotherapy and radiation therapy with thorough transurethral resection of the bladder (TURB) (trimodality therapy) is another method of treating patients with invasive bladder cancer and is particularly aimed at bladder preservation.
The majority of randomized clinical trials evaluating perioperative chemotherapy has been underpowered, used ineffective chemotherapy, or have had methodological flaws (Sylvester and Sternberg, 2000). However, more sufficiently powered neoadjuvant studies and meta-analyses provide further recommendations (Hall, 2002; Grossman et al, 2003b; Advanced Bladder Cancer Meta-analysis Collaboration, 2005b).
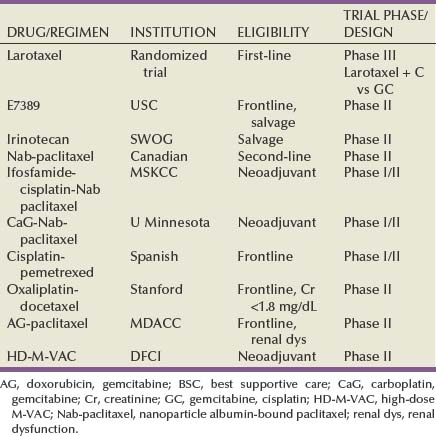
Neoadjuvant Chemotherapy
Neoadjuvant chemotherapy has been used for patients with operable clinical stage T2 to T4a muscle-invasive disease. The rationale for chemotherapy before cystectomy or, in some cases, full-dose radiation therapy (RT), is based on the intent to treat micrometastatic disease present at diagnosis.
Chemotherapy before surgery has several advantages. Therapy is better tolerated before surgery or radiation. Chemotherapy-related toxicities are considerably less in patients with localized disease than in those with metastatic disease on the basis of performance status. Patients are often able to tolerate a greater dose intensity and more cycles of chemotherapy preoperatively than postoperatively. Neoadjuvant chemotherapy allows in vivo drug sensitivity testing that may provide useful information for later therapy. The primary tumor can be evaluated for response, which also has major prognostic significance. In addition, preoperative chemotherapy may down-stage tumors, potentially allowing for technically easier surgery (Calabrò and Sternberg, 2009b).
Evaluating response to neoadjuvant chemotherapy can be complicated by discrepancies between clinical and pathologic staging, which have been reported in some 30% of cases (Herr and Scher, 1990; Sternberg et al, 2003). The major disadvantage of neoadjuvant chemotherapy is a delay in definitive local therapy in patients who do not respond or whose disease progresses. An interval longer than 12 weeks between the diagnosis of muscle invasion and cystectomy has even been associated with a poorer outcome (Sanchez-Ortiz et al, 2003; Stein, 2003a).
Another theoretic disadvantage of neoadjuvant chemotherapy is the possible increase in the incidence of perioperative morbidity. Few reports have studied this (Hall et al, 1996; Millikan et al, 2001). In a comparative study of neoadjuvant and adjuvant chemotherapy, neoadjuvant chemotherapy did not increase perioperative morbidity (Millikan et al, 2001). In the United States and in most of Europe, radical cystectomy is the preferred treatment for fit patients who have a good performance status.
Randomized Trials of Neoadjuvant Chemotherapy
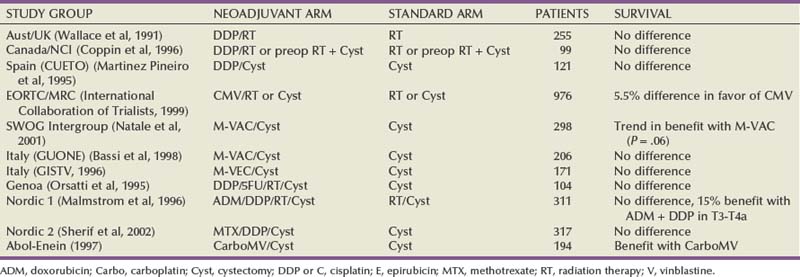
Many randomized trials have evaluated the impact of neoadjuvant chemotherapy on survival. These trials have shown either a trend toward a benefit or no survival advantage. The majority of these trials have, however, been underpowered. Randomized neoadjuvant chemotherapy trials are summarized in Table 82–4 (Sternberg et al, 2007).
The largest trial of neoadjuvant chemotherapy was performed by the European Organization for Research and Treatment of Cancer and the Medical Research Council (EORTC and MRC). A total of 976 patients who were accrued over 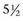 years from 106 institutions were randomized to receive cisplatin, methotrexate, and vinblastine (CMV) neoadjuvant chemotherapy or no chemotherapy. Cystectomy and/or radiation therapy was permitted as definitive management of the primary tumor. The study was designed to detect an absolute improvement in survival of 10% (from 50% to 60%) with a power of 90% and a type 1 error of 5%.
years from 106 institutions were randomized to receive cisplatin, methotrexate, and vinblastine (CMV) neoadjuvant chemotherapy or no chemotherapy. Cystectomy and/or radiation therapy was permitted as definitive management of the primary tumor. The study was designed to detect an absolute improvement in survival of 10% (from 50% to 60%) with a power of 90% and a type 1 error of 5%.
When results were first published, there was a nonsignificant trend toward improvement in survival in patients treated with neoadjuvant CMV chemotherapy (International Collaboration of Trialists, 1999). In an update presented at the American Society of Clinical Oncology (ASCO) meeting in 2002, with longer follow-up of 7.4 years, the data have barely reached statistical significance (P = .048). There was a 5.5% benefit in favor of patients treated with CMV (Hall, 2002). Survival at 5 years was 50% compared with 44%, and at 8 years was 43% versus 37%. Following neoadjuvant chemotherapy, no residual tumor or pathologic complete response (pCR) was found in 32.5% of cystectomy specimens. Chemotherapy-related mortality was 1%. Chemotherapy did not increase the rate of postoperative complications. Although patients treated with CMV had a survival benefit that was maintained over time, it was concluded that there was no change in absolute benefit.
This study demonstrated an improvement with CMV chemotherapy, but the improvement was slightly less than the magnitude originally sought in the trial. Several factors may explain why the experimental arm in this study achieved somewhat lower survival than anticipated. The chemotherapeutic regimen used in this study is not considered standard of care, although CMV has never been compared with methotrexate, vinblastine, doxorubicin, and cisplatin (M-VAC) in a randomized trial. Local therapy included cystectomy or radiation or both, with 42% and 43% of patients receiving radiation alone as local therapy in the neoadjuvant chemotherapy group and the no-chemotherapy group, respectively. Although in Europe radiation therapy is believed to be equivalent to cystectomy, no prospective randomized trial has ever proven this.
In the Southwest Oncology Group (SWOG) Intergroup trial, 317 patients with clinical T2 to T4a bladder cancer were randomized to receive cystectomy or three cycles of neoadjuvant M-VAC chemotherapy followed by cystectomy (Grossman et al, 2003a). Enrollment took place at 126 institutions over 11 years. Patients were stratified according to age (younger than 65 years old or 65 years and older) and stage (cT2 vs. cT3 or cT4a). The aim of the study was to detect an increase in survival with M-VAC chemotherapy. The original study was planned for one-sided testing, which means that the investigators were planning to test not for a difference but only for improvement because the hypothesis was that medical practice would change only if there was an improvement with chemotherapy.
Median survival among patients randomized to surgery alone was 46 months, as compared with 77 months for patients who received neoadjuvant M-VAC chemotherapy (P = .06; two-sided stratified log rank test). In part because of the very long accrual period and follow-up, these results have achieved borderline statistical significance. The estimated risk of death was decreased by 25% (HR = 1.33) in patients treated with neoadjuvant M-VAC (Grossman et al, 2003b). The neoadjuvant chemotherapy group had a significantly higher proportion of patients with pCR than the cystectomy-alone group (38% vs. 15%, P < .001). Patients with locally advanced (cT3 or T4a) disease had the greatest survival benefit from neoadjuvant chemotherapy (65 vs. 24 months). Other randomized trials of neoadjuvant chemotherapy are found in Table 82–4 (Malmstrom et al, 1996; Bassi et al, 1998; Sherif et al, 2002).
Meta-Analyses of Randomized Neoadjuvant Chemotherapy Trials
The majority of neoadjuvant randomized trials have failed to show an unequivocal survival benefit in favor of chemotherapy. However, the majority of the studies have been hampered by inadequate sample sizes, suboptimal chemotherapy, premature closure, or inadequate follow-up (Sylvester and Sternberg, 2000). For this reason, meta-analyses have attempted to explain and interpret these data (International Collaboration of Trialists, 1999; Winquist et al, 2004).
The Advanced Bladder Cancer (ABC) Meta-analysis included data on 3005 patients from 11 randomized neoadjuvant chemotherapy trials (Advanced Bladder Cancer Meta-analysis Collaboration, 2005b).The overall analysis was in favor of chemotherapy (HR = .89, 95% CI .81 to .98, P = .022). In patients who received single-agent cisplatin chemotherapy, neoadjuvant chemotherapy appeared detrimental, but the numbers were too small to support definitive conclusions (P = .264). However, in the subset of patients who received cisplatin-based combination neoadjuvant chemotherapy, the meta-analysis showed a reduction in the risk of death of 14% (HR = .86, 95% CI .77 to .95, P = .003), translating into a 5% absolute survival benefit at 5 years (95% CI: +2% to +9%). The results are similar to the results obtained in the EORTC and MRC trial, which was the largest trial in the analysis (Hall, 2002). The authors noted no difference in the relative risk reduction either in relation to the type of local treatment (cystectomy, radiotherapy or the combination) or in relation to patient characteristics. However, apart from this study, neoadjuvant chemotherapy before radiation therapy has not been shown to improve survival (Zietman et al, 1997).
Despite the 5% survival benefit, the neoadjuvant approach before cystectomy has not been widely accepted. In an analysis of stage III bladder cancer from the National Bladder Cancer Database (n = 11,328), only 0.7% of patients received neoadjuvant chemotherapy (David et al, 2007). Part of the reason may be that physicians feel that it is not worthwhile to give chemotherapy to all patients to achieve a 5% benefit (Sternberg and Collette, 2006a). The studies on which recommendations have been made do not contain information on toxicity and quality of life. In most of the trials elderly patients (median age 63 to 65 years in EORTC, SWOG, Nordic) or those with poor renal function or performance status have not been included, making it difficult to generalize the results to all of the elderly patients who are affected with bladder cancer (Droz, 2005). Information is lacking on subgroups of patients to indicate who would derive the most benefit from treatment. In some studies (Nordic) both neoadjuvant and adjuvant chemotherapy were used. And of perhaps most importance, the impact of cystectomy and the quality of surgery have been shown to be as important as whether or not chemotherapy was administered (Herr, 2004). The delay in definitive surgery in patients who do not respond to treatment raises concerns regarding compromise of curability, and many urologists simply believe that a potential 5% advantage in overall survival is not great enough to justify giving toxic chemotherapy to all patients before surgery.
Many oncologists prefer to use newer agents such as gemcitabine or the taxanes, which have not been evaluated in randomized studies. A single-institution retrospective report of neoadjuvant gemcitabine and cisplatin before cystectomy reported that 26% of patients achieved a pathologic complete response to chemotherapy, which may be comparable with other combination cisplatin-based regimens (Dash et al, 2008).
In conclusion, cystectomy is the “gold standard” of treatment for patients with localized muscle-invasive bladder cancer. Neoadjuvant chemotherapy is intended for patients with operable clinical stage T2 to T4a muscle-invasive disease. The rationale for chemotherapy before cystectomy is based on the intent to treat micrometastatic disease present at diagnosis. Although the data available support the use of M-VAC or CMV as neoadjuvant chemotherapy, these regimens are less frequently used today because the regimen of gemcitabine and cisplatin has been widely used for metastatic disease. However, the efficacy of the gemcitabine and cisplatin combination in the neoadjuvant setting has not been proven, suggesting that M-VAC or CMV should still be used on the basis of level 1 evidence. Toxicity and mortality associated with neoadjuvant chemotherapy are acceptable because these patients are generally less debilitated than patients with metastatic disease.
Discrepancies between clinical and pathologic staging can be expected. Available data suggest that for “average-risk” patients with cT2 cancer, the benefit of adding chemotherapy to local therapy is at best modest. Studies suggest a more substantial benefit for patients with cT3b disease. The quality of the surgery is an important factor that cannot be underestimated. Meta-analysis of cisplatin-containing combination neoadjuvant chemotherapy trials reveals a 5% difference in favor of neoadjuvant chemotherapy. Unfortunately, however, quality of life data are not available.
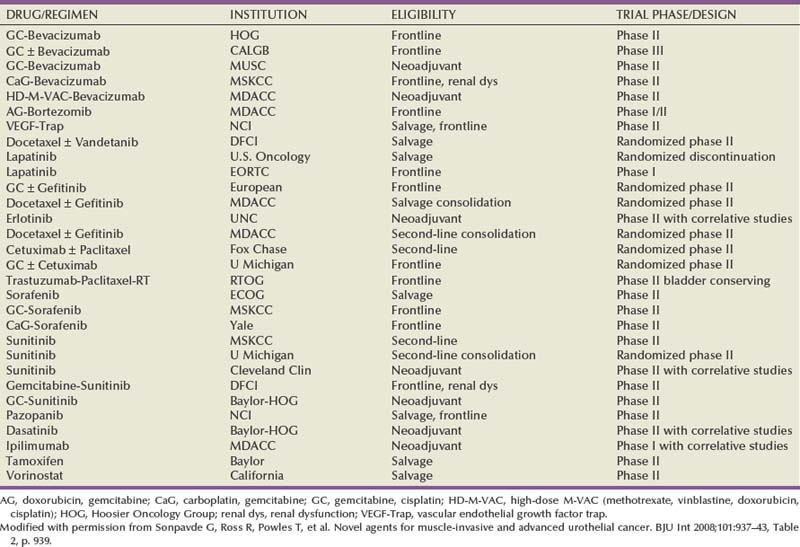
Adjuvant Chemotherapy
In patients with pT3-4 and/or N+M0 disease, 5-year survival after radical cystectomy is only 25% to 35% at best. As a result, adjuvant chemotherapy has been advocated for high-risk patients in an effort to delay recurrence and prolong survival. Delivery of chemotherapy postoperatively has potential advantages. An adjuvant approach allows selection of patients at highest risk of metastatic or recurrent disease on the basis of an accurate pathologic evaluation (Calabrò and Sternberg, 2009b). Surgery is performed without delay, and the advent of orthotopic neobladders and continent urinary diversions has improved quality of life in patients after cystectomy, favoring immediate cystectomy. There is evidence that delaying cystectomy can be detrimental (Chang et al, 2003; Sanchez-Ortiz et al, 2003), and no time is wasted in those patients who do not respond to chemotherapy. The availability of sufficient tissue for increasingly sophisticated analysis of molecular prognostic and predictive markers is also a potential advantage. If micrometastases are present, they can be treated with chemotherapy when at a low volume, rather than after there is overt metastatic disease.
The major disadvantages are that the bladder is not preserved and that there is a delay in starting systemic therapy for occult metastases while the focus is first on the primary tumor. Response cannot be easily evaluated, and the only clinical end point that can be assessed is the time to tumor recurrence.
Several factors contribute to the difficulty in accrual to bladder cancer trials. Patients are often elderly with significant comorbidities that are related to smoking and/or compromised renal function (Jin et al, 2006). An additional disadvantage is the difficulty in administering chemotherapy to those with surgical morbidities following cystectomy. A prospective standardized reporting methodology was used to quantify and characterize the risk of perioperative morbidity following cystectomy at MSKCC, a center with high-volume, fellowship-trained urologists. Staggering rates of postoperative complications were observed within 90 days of surgery (Donat et al, 2009; Shabsigh et al, 2009), further confirming prior observations at another high-volume center in California (Stein and Skinner, 2006). These often underreported postoperative morbidities can lead to long delays and difficulties in administering adjuvant chemotherapy.
Randomized Trials of Adjuvant Chemotherapy
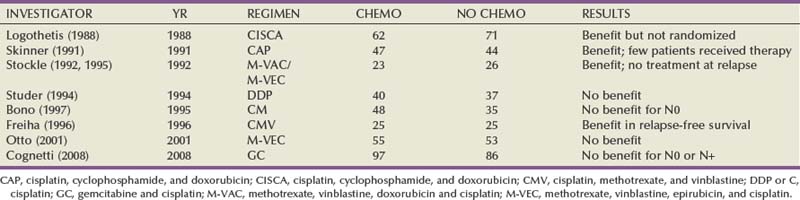
Despite its appeal, there have been few randomized trials evaluating adjuvant chemotherapy. Table 82–5 shows the results of randomized trials of adjuvant chemotherapy after cystectomy (Logothetis et al, 1988; Skinner et al, 1991; Stockle et al, 1992; Studer et al, 1994; Stockle et al, 1995; Freiha F et al, 1996; Bono et al, 1997; Otto et al, 2001; Cognetti et al, 2008). All of these trials were relatively small, enrolling only 49 to 108 patients. Nonetheless, two trials suggest a survival benefit with adjuvant chemotherapy.
In the USC trial, 91 patients with pT3-4a or node-positive bladder cancer were randomized to adjuvant chemotherapy for 4 cycles or no further treatment after cystectomy (Skinner et al, 1991). The chemotherapy given was heterogeneous and included a cisplatin, cyclophosphamide, and doxorubicin hydrochloride (Adriamycin) (CISCA)-like regimen, single-agent cisplatin, and chemotherapy regimens selected by clonal assays. Three-year time to progression was significantly delayed (P = .01) with adjuvant chemotherapy (70%) compared with time to progression with observation only (46%). Median survival was also significantly improved (P = .0062) in the chemotherapy arm (4.3 years) compared with results in the observation arm (2.4 years). However, 3-year survival with adjuvant chemotherapy (66%) was not statistically superior (P = .09) to survival after surgery alone (50%).
This trial has been criticized both for its statistical design and for its treatment plan. Its small size resulted in a lack of statistical power, limiting its ability to detect a clinically meaningful survival difference.
Another adjuvant chemotherapy trial was conducted in Germany (Stockle et al, 1992; Stockle et al, 1995). High-risk patients were randomized to M-VAC/M-VEC (methotrexate, vinblastine, epirubicin, and cisplatinum) or to no further therapy after cystectomy. The study was terminated early with only 49 patients enrolled when an interim analysis showed significant improvement in 3-year disease-free survival (63% vs. 13%, P = .002). Ten-year survival data from this trial still favored adjuvant chemotherapy over surgery alone for progression-free survival (44% vs. 13%, P = .002), tumor-specific survival (42% vs. 17%, P = .007), and a trend in overall survival (27% vs. 17%, P = .07). Significant benefit was noted in patients with positive lymph nodes who received adjuvant chemotherapy. Only 27% of patients who received chemotherapy had evidence of tumor progression compared with 92% of those treated with cystectomy alone.
This trial has also been heavily criticized. The study took 3.5 years to accrue 49 patients and was stopped early when an interim analysis revealed a significant advantage in favor of chemotherapy. Many patients in the cystectomy alone group were not offered chemotherapy when disease progressed, and some patients randomized to the chemotherapy group never received chemotherapy.
Other randomized trials have not shown a survival benefit with adjuvant chemotherapy. A systematic review of randomized trials of adjuvant cisplatin-containing combination chemotherapy in locally advanced bladder cancer revealed that adjuvant trials have been hampered by serious methodological flaws. Major deficiencies have been observed with small sample sizes, early closure of trials, and limitations of statistical analyses, reporting of results, and poorly substantiated conclusions (Sylvester and Sternberg, 2000). These trials provide insufficient evidence to support the routine use of adjuvant chemotherapy.
The ABC Meta-analysis Collaboration (2005a) identified 11 randomized trials of adjuvant chemotherapy, but individual patient survival data from only 6 adjuvant trials comprising 283 events in 491 patients were eventually included, and 4 of these 6 trials were stopped early. Although the results suggest a 9% improvement in absolute survival at 3 years (HR = .75, 95% CI .60 to .96, P = .019), the analysis was severely compromised by underpowered survival curves. In publishing their results, the authors clearly acknowledged the problems with interpretation of these data (Sternberg and Collette, 2006a).
A randomized adjuvant trial from Italy failed to reach its planned accrual (Cognetti et al, 2008). In this trial patients with G3T2 were also eligible; however, the trial was closed with only 190 patients accrued. The study was underpowered, and no difference in survival for either pN0 or pN+ patients was observed in patients treated with two different regimens of gemcitabine and cisplatin (GC). The Spanish Oncology Genitourinary Group (SOGUG) accrued 146 of the 340 patients planned and in July 2007 closed a phase III study comparing surgery followed by four cycles of GCP (gemcitabine, cisplatin, and paclitaxel) or observation.
The role of adjuvant chemotherapy continues to be unresolved; yet it is widely administered in the United States and Europe. The EORTC evaluated early versus delayed chemotherapy using M-VAC, high-density M-VAC, or the gemcitabine-cisplatin combination. The trial was closed with only 278 patients of a planned 660 patients accrued as of June 2008, making it unlikely that there will ever be a definitive answer to this important question.
Finally, the USC/SWOG International trial has completed accrual to a study evaluating adjuvant M-VAC in patients with organ-confined (pT1-pT2) high-risk bladder cancer as defined by altered p53 status (Stadler et al, 2009). With 511 patients registered and 110 randomized, the data safety monitoring committee recommended study closure on the basis of a futility analysis, suggesting that the probability of detecting a significant difference in time to relapse would be highly unlikely. The study was unable to confirm the prognostic or predictive value of p53 as measured by immunohistochemistry (IHC).
Unfortunately, the question of the true benefit of adjuvant chemotherapy in high-risk patients with pT3, pT4, and N+ disease is likely to remain unanswered. Perhaps a meta-analysis of the EORTC, Spanish, and Italian studies may someday be of interest. Defining optimal therapy in all patients with muscle-invasive bladder cancer is a difficult task. Neoadjuvant and adjuvant chemotherapy probably have similar efficacy, although only one randomized trial has looked at this comparison (Logothetis et al, 1996).
Some patients have been cured by TURB or radiation alone, and selection of optimal therapy for a given patient should be individualized. Current data appear to favor neoadjuvant chemotherapy followed by radical cystectomy with adequate complete pelvic lymph node dissection (PLND). More patients are likely to receive effective chemotherapy before rather than after surgery. All clinical stages of invasive bladder cancer may benefit from this combined strategy. However, a 5% absolute survival benefit for chemotherapy-treated patients is only a small improvement, and development of more effective regimens is necessary.
Bladder Preservation And Combined Modality Approaches
Radical Transurethral Resection of Bladder Tumor (TURBT)
For selection of candidates, strict criteria are required and include (1) initial occurrence of bladder cancer; (2) no CIS; (3) size less than or equal to 3 cm; (4) stage T2 (no palpable mass); and (5) not in the dome or high posterior wall because of the risk of bowel injury. Patients with T3b tumors are not candidates for complete TUR, and those with T4 tumors are not eligible because, by definition, the tumors are invading surrounding structures. When these criteria are used, long-term survival is comparable with that of radical cystectomy for the small fraction of potentially eligible patients. Herr (2001) and Solsona and colleagues (1998) reported 10-year survival probabilities for 99 and 130 patients, respectively, with disease-progression rates ranging from 30% to 34%, disease-specific survival 76% to 80%, and cystectomy rates of 9% to 34%.
All patients must undergo re-resection 3 months after radical TURBT, and if tumor persists at this time, radical cystectomy or radiation is considered. Leibovici and colleagues (2007) reported the experience from the M.D. Anderson Cancer Center, where they adopted a policy of re-resecting tumors in all patients with muscle-invasive cancer and offered bladder preservation to those patients whose cancer was stage T0. Of the 35 eligible patients, 27 opted for preservation. Of these, 15 had recurrence and 8 of these patients underwent radical cystectomy. Overall and cancer-specific survival rates exceeded 80% in this highly selected group of patients.
The advantage of this approach is that at least a small portion of patients with invasive bladder cancer preserve their native bladder and optimum quality of life. The disadvantages are the absence of pathologic staging of the pelvic and iliac lymph nodes and the requirement for lifelong surveillance because up to 25% ultimately require cystectomy.
Partial Cystectomy
The indications for partial cystectomy are similar to those for radical TURBT with one exception—the location. Tumors must be in a location suitable for bladder preservation—usually on the dome and away from the ureteral orifices. Bilateral pelvic lymphadenectomy is performed at the time of surgery for pathologic staging of the nodes. Although early series reported a high rate of progression and mortality, contemporary series report long-term relapse rates that are comparable with those for radical cystectomy when strict selection criteria are applied (Holzbeierlein et al, 2004; Kassouf et al, 2006). Smaldone and colleagues (2008) recently reported on a series of 25 patients accrued over 11 years in whom partial cystectomy was performed for patients with primary solitary T2 or high-grade T1 tumors and no CIS. All patients received preoperative radiation for 5 days and a single dose of intraoperative intravesical chemotherapy followed by partial cystectomy. Follow-up included intravesical BCG, and the 5-year cancer-specific survival probability was 84%. These authors emphasize the need for lifelong cystoscopic surveillance and abdominal imaging.
This surgical option also has a definite role in patients who have had an objective response to neoadjuvant chemotherapy, with or without radiation therapy (Herr et al, 1998; Koga et al, 2009; Sternberg et al, 2003). The tumor site can be resected in patients with a clinical complete response (CR) in order to ensure pathologically that the tumor has been completely removed. This may also be an option for patients with a partial response (PR) if the tumor location is amenable and there is no CIS. However, radical cystectomy remains the gold standard in patients who experience a PR. In the unique situation of urachal adenocarcinoma, partial cystectomy with en bloc resection of the urachus is the preferred surgical method for local control (Siefker-Radtke, 2006; Gopalan et al, 2009). This is undertaken using a closed technique in order to avoid tumor spill of this highly implantable tumor, with en bloc resection of the posterior rectus sheath, urachus, and dome of the bladder.
Neoadjuvant or Concomitant Chemotherapy and Bladder Preservation
Advantages of bladder preservation are that there is less surgery, there is no need for a urinary diversion, sexual function is preserved, and a better quality of life results. Neoadjuvant chemotherapy or concomitant chemotherapy with radiation therapy has been given to patients with muscle-invasive bladder cancer as a bladder-preserving strategy. The common theme has been to select patients with a complete pathologic response to treatment for bladder preservation. Appropriate candidates for bladder preservation include those with solitary T2 or early T2 tumors less than 6 cm in size, no tumor-associated hydronephrosis, tumors allowing a visibly complete TURB, invasive tumors not associated with CIS, and renal function adequate to allow use of cisplatin concurrent with radiation.
Although this strategy appears promising, bladder preservation protocols have never been compared with radical cystectomy in randomized studies. Therefore these techniques are often applied in highly selected patients with low-volume disease or in those who refuse or who are poor candidates for radical cystectomy.
Can we save the bladder and obtain the same survival as with radical cystectomy? Up to 15% of patients with muscle-invasive tumors have no residual disease at the time of cystectomy, indicating that TURB alone can potentially cure some patients (Herr, 2009; Solsona et al, 2009). However, the absence of residual disease at TURB does not mean that patients are necessarily cured. Understaging with TURB occurs in approximately 30% of cases (Herr and Scher, 1990; Frazier et al, 1993; Amling et al, 1994; Stein, 2000; Sternberg et al, 2003). The limited efficacy of surgery alone and the introduction of effective combination chemotherapy have led increasingly to a multidisciplinary approach to bladder preservation. However, bladder preservation is usually achieved with trimodality (chemoradiation and thorough TURB) and occasionally with bimodality protocols.
Bimodality Therapy
In Italy, three cycles of neoadjuvant M-VAC chemotherapy, with extensive TURB before chemotherapy, were used in 104 cT2-T4 N0M0 bladder tumor patients with the intent of bladder preservation (Sternberg et al, 2003). At the TURB following M-VAC, 49 (49%) patients were T0. Responding patients underwent TURB or partial cystectomy. Median survival was 7.5 years (95% CI, 4.9 to 10 years); 60% of the patients treated with M-VAC and TURB alone were alive at a median follow-up of 56 months (10 to 160+), and 44% in the TURB alone group maintained an intact bladder. Patients who were down-staged to pathologic CR or superficial disease after chemotherapy had a 5-year survival of 69%, whereas those with persistent muscle-invasive disease after chemotherapy had a 5-year survival of only 26%. Bladder sparing was possible in selected patients on the basis of response to neoadjuvant chemotherapy.
Likewise, investigators at MSKCC have used neoadjuvant M-VAC in 111 T2-T3N0M0 bladder cancer patients followed by transurethral bladder-sparing surgery (Herr et al, 1998). Of these patients, 60 (54%) achieved pCR (pT0). The majority of patients who achieved pCR after TURB and neoadjuvant chemotherapy benefitted from long-term survival with the bladder preserved.
From these single-institution studies, one could conclude that patients with cT0 status after chemotherapy who forego cystectomy have a survival rate similar to that of patients undergoing immediate cystectomy. However, because these data are from highly specialized single institutions, this practice must be considered investigational and this approach cannot be considered as standard methodology.
In an intergroup SWOG study, neoadjuvant paclitaxel, carboplatin, and gemcitabine produced a promising 46% cT0 rate in 77 patients with histologically confirmed T2 to T4a muscle-invasive disease. There was, however, an unacceptably high rate (60%) of persistent cancer at cystectomy in patients presumed to have pT0 status. The authors recommended that patients completing neoadjuvant chemotherapy should strongly consider definitive local therapy rather than cystoscopic surveillance regardless of postchemotherapy cT0 status (DeVere White et al, 2009).
Neoadjuvant chemotherapy can be given safely, and patients who respond to chemotherapy have a favorable outcome. However, in a multi-institutional setting after neoadjuvant chemotherapy, TURB cannot be safely relied on to determine which patients require cystectomy. Definitive local therapy should be a mandatory component of the treatment paradigm. Perioperative chemotherapy has a role in treating this disease, but the exact timing of delivery and the appropriate patient population require further definition.
Trimodality Therapy
Combining systemic chemotherapy with RT can permit bladder preservation while sensitizing the tumor to RT and perhaps also treating occult metastases. The optimal combined-modality bladder-sparing regimen is not well defined. The majority of modern series have used single-agent cisplatin as the radiosensitizing agent. Trimodality therapy for clinical stages T2 to T4a includes maximal TURB of the bladder tumor, modern external beam XRT with radiosensitizing chemotherapy given concurrently, and a careful urology-based surveillance program with prompt cystectomy for persistent or recurrent invasive tumors. This approach is best illustrated by the Radiation Therapy Oncology Group (RTOG) series of trials, in which complete responses in the bladder were observed in 59% to 87% of patients (Fernando and Sandler, 2007).
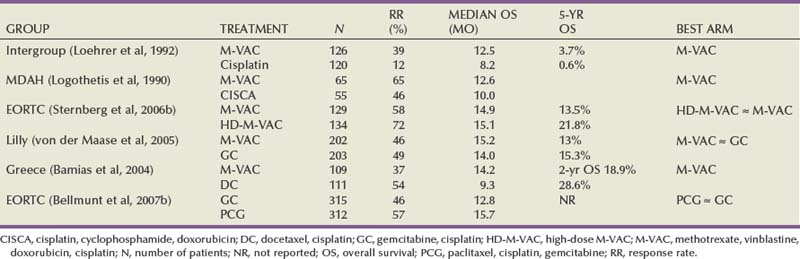
Trials of concomitant neoadjuvant chemotherapy and RT are shown in Table 82–6 (Kaufman et al, 1993; Housset et al, 1997; Kachnic et al, 1997; Shipley et al, 1998; Rodel et al, 2002). In a study at the Massachusetts General Hospital, 106 patients with muscle-invasive stage T2 to T4a bladder cancer underwent chemoradiation following TURB (Kaufman et al, 1993; Kachnic et al, 1997). Patients received two cycles of CMV followed by radiotherapy (4000 cGy) with two cycles of concurrent cisplatin every 3 weeks. Those with persistent disease underwent radical cystectomy, and those who attained CR were selected for additional radiation (2480 cGy) and one additional cycle of cisplatin. After a median follow-up of 48 months, the 5-year overall survival (OS) and disease-specific survivals were 52% and 60%, respectively. The 5-year OS rate with an intact functioning bladder was 43%. These investigators contend that TURB plus chemoradiation can be considered as an alternative to radical cystectomy.
However, RTOG investigators reported a randomized study of 123 patients treated with two cycles of neoadjuvant CMV before concomitant cisplatin and RT (39.6 Gy) (Shipley et al, 1998). Those who achieved a CR received an additional 25.2 Gy plus one concurrent cycle of cisplatin. Patients with persistent disease underwent cystectomy. Neoadjuvant CMV did not improve 5-year survival (48% vs. 49%) or survival rate with preserved bladders (36% vs. 40%). An unfavorable toxicity profile was observed, and the study did not complete accrual.
Paclitaxel as a radiosensitizer has also been evaluated, as well as adjuvant chemotherapy with GC. The results of RTOG protocol 99-06 were reported with more than 4 years of median follow-up in 80 patients, showing an 81% complete response rate and actuarial 5-year overall and disease-specific survival rates of 56% and 71%, respectively (Kaufman et al, 2009b). The RTOG has also performed a randomized phase II trial to identify the optimum concurrent chemotherapy regimen in conjunction with radiation. Patients were randomized to radiation twice daily with either concurrent paclitaxel and cisplatin or concurrent infusional fluorouracil and cisplatin during the induction and consolidation phases. Additionally, the adjuvant chemotherapy triplet regimen of gemcitabine, paclitaxel, and cisplatin was investigated (Kaufman et al, 2009a).
What appears to emerge from these studies is that neoadjuvant chemotherapy before radiation therapy is not beneficial in terms of survival (Zietman et al, 1997). Concomitant chemoradiation therapy with chemotherapy as a sensitizing agent is an alternative to cystectomy. It is difficult to interpret the contribution to survival of each component of these multimodality bladder-sparing approaches. Because restaging TURB has not been performed as standard practice in all series, it is difficult to know the true impact of TURB alone. One expects patients who have been rendered clinical p0, either by TURB alone or by chemoradiation before TURB or cystectomy, to have improved long-term survival (Herr, 1996; Herr et al, 1998; Sternberg, 2005).
Cystectomy series do not report by “intention to treat” and exclude those patients for whom cystectomy is found to be inappropriate. Additionally, many patients in cystectomy series do not have preoperative proof of muscle invasion by tumor. Most cystectomy reports include 25% to 40% of the patients having less than P2-stage tumors. Although survival may be similar to that in contemporary cystectomy series, it is extremely difficult to compare methodologies (Shipley et al, 2001).
The combined morbidities of chemotherapy and radiation must also be considered. Evaluation of quality of life is important because patients are often candidates for immediate cystectomy with continent diversions. Late adverse effects of trimodality chemoradiation therapy may include poor bladder function (6%), occasional rectal bleeding (2% to 5%), rectal urgency (19%), impotence (20% to 50%), vaginal dryness (rare), and temporary neuropathy (common) (Heney et al, 2009). Available data regarding quality of life indicate that urinary, bowel, and sexual function after bladder preservation seem acceptable. Patients who undergo chemoradiation and bladder preservation should be carefully selected and well informed, willing to undergo frequent follow-up and multiple cystoscopies, and understand the possibility that cystectomy may become necessary.
Current protocols incorporate newer chemotherapeutic agents such as paclitaxel and gemcitabine in an attempt to improve efficacy. Various radiation schedules such as hypofractionation and twice-daily treatments are under evaluation. Carefully selected patients with small T2 tumors who undergo complete TURB, have no evidence of urethral obstruction, and have complete response to induction chemoradiation are the most suitable candidates.
No prospective randomized trials have compared survival with chemotherapy and TURB alone or with concomitant chemoradiation and radical cystectomy in muscle-invasive disease. Factors associated with maintaining the bladder include (1) clinical stage (organ-confined), (2) tumor size less than 3 to 5 cm, (3) absence of hydronephrosis, (4) absence of a palpable mass, and (5) unifocal disease. Patients with residual disease at first cystoscopy (within 3 months) after TURB alone or neoadjuvant chemotherapy plus TURB are at risk and should undergo cystectomy.
Molecular Markers In Invasive And Advanced Bladder Cancer
Peripheral Blood
Despite accurate pathologic staging and tissue biomarkers, pretreatment markers that reflect the overall biology of the patient’s cancer are lacking. Because 40% to 50% of patients die within 5 years despite aggressive surgery and perioperative chemotherapy, clinically useful markers that are associated with risk of progression and response to treatment are necessary.
Early studies, perhaps the earliest, evaluated carbohydrate-based cell surface markers including CEA, CA19-9, and CA-125. Several reports indicate that these may be elevated in locally advanced and/or metastatic disease and may correlate with response to therapy (Cook et al, 2000; Izes et al, 2001; Margel et al, 2006). Margel and colleagues (2006, 2007) prospectively evaluated these three markers and found that CEA elevations were modest and not associated with pathologic stage but that CA19-9 and CA-125 were associated with non-organ-confined disease and with overall and/or disease-specific survival. Cook and colleagues (2000) evaluated 74 patients with metastatic disease and found that 58% had at least one elevated marker and that marker response correlated with clinical response to treatment. Margel and colleagues (2009) incorporated these three peripheral biomarkers into a predictive model that included four clinical parameters—hydronephrosis, clinical stage, presence of CIS, and tumor size greater than 3 cm—for predicting non-organ-confined cancer and demonstrated 85% accuracy.
Recent studies suggest that several growth factors and cytokines that are measurable in peripheral blood are elevated in invasive bladder cancer and are associated with metastatic disease and increased risk of progression. These include growth factors such as transforming growth factor beta-1 (TGF-β1), insulin growth factor-1 (IGF-1) and IGF binding protein-3, and the cytokine receptor interleukin 6 (IL-6) and its soluble receptor (IL6-sR) (Shariat et al, 2001; Andrews et al, 2002; Shariat et al, 2003a). Other classes include the soluble proteolytic cleavage product of the cell adhesion molecule E-cadherin. Loss of expression of E-cadherin in bladder cancer is associated with a higher risk of progression, and high blood levels of the soluble fraction are associated with a high risk of progression after cystectomy (Byrne et al, 2001; Matsumoto et al, 2003). Still other markers include urokinase type plasminogen activator (UPA) (Shariat et al, 2003b). Several of these biomarkers are associated with lymphovascular invasion and nodal metastases. When analyzed in a COX proportional hazards analysis, IL6-sR and UPA were independently associated with progression after cystectomy (Sanderson et al, 2004). Clearly, before incorporation into clinical practice, more research with prospective validation is necessary.
Proteomic profiling using mass spectrometry is beginning to be explored as an alternative to tissue-based genomic profiling. Schiffer and colleagues (2009) identified a panel of urinary polypeptide bladder cancer biomarkers in 127 patients with bladder cancer and then evaluated the panel prospectively in 130 patients in order to discriminate between noninvasive and muscle-invasive cancers. Multivariate analysis revealed that the proteomic panel and tumor grade, but not cytology, were associated with muscle invasion. This work may also serve as a basis for discovery of novel proteins associated with bladder cancer progression.
With the first FDA approval of techniques to detect rare circulating tumor cells (CTCs) in breast, colorectal, and prostate cancer, several investigators are conducting studies to determine if CTCs can be detected in various stages of invasive bladder cancer (Cristofanilli et al, 2004; Goodman et al, 2009). Preliminary data suggest that the presence and number of CTCs are associated with metastatic disease and are measurable using a variety of platforms (Naoe et al, 2007; Gallagher et al, 2009; Nezos et al, 2009).
Tissue Markers
A substantial volume of research has examined molecular alterations in bladder cancer. The initial focus was on the cell cycle regulatory gene p53 and the retinoblastoma tumor suppressor gene (Rb), both of which are altered in about 50% of high-grade transitional cell cancers (Chatterjee et al, 2004a; Shariat et al, 2004). In addition, genes downstream from p53 and Rb including p21, p27, and p16 are known to be altered frequently and to contribute prognostic information in combination with p53 and Rb (Benedict et al, 1999; Chatterjee et al, 2004b; Shariat et al, 2008). A clinical trial, initiated by investigators at USC who were later joined by SWOG, was the first multicenter multinational trial designed to validate p53 alterations as a prognostic marker and to use it to define a high-risk group of patients with organ-confined disease after radical cystectomy (see “Adjuvant Chemotherapy” earlier) (Lerner et al, 2009). Patients with altered p53 IHC were offered randomization to M-VAC adjuvant chemotherapy or observation. Recently reported results of the trial were negative because adjuvant chemotherapy appeared to offer no benefit, at least in part because of a lower than predicted progression rate in p53 positive (altered) patients (Stadler et al, 2009). The study does, however, contribute a rich repository of biologic tissues and long-term clinical follow-up to aid in determining the influence of a variety of immunohistochemical markers on progression after radical cystectomy. A more contemporary study analyzed both p53 IHC and gene mutation status and found that each was predictive of outcome after cystectomy but that the two techniques combined provided the most accurate determination of outcome (George et al, 2007).
Other tissue markers that are associated with outcome after cystectomy include the calcium-dependent cell adhesion marker E-cadherin (Byrne et al, 2001). Loss of expression is associated with a higher probability of advanced pathologic stage and worse outcome following radical cystectomy. Proliferation markers such as Ki-67 (Margulis et al, 2009), markers of angiogenesis (vascular endothelial growth factor [VEGF], thrombospondin, microvessel density) (Bochner et al, 1997; Grossfeld et al, 1997; Slaton et al, 2004), and markers involved in apoptosis (Caspase 3, survivin, bcl-2) (Karam et al, 2007) have been analyzed primarily in cystectomy specimens and demonstrate that increased proliferation rates, increased angiogenesis, and loss of expression of apoptosis markers or inhibitors are associated with worse outcomes. A multicenter group has recently reported a nomogram incorporating several of these tissue markers with better predictive accuracy than TNM staging (see “Nomograms” later) (Shariat et al, 2008). To date, however, no marker or combination of markers has been validated as a predictive biomarker for progression or survival.
Advances in high-throughput genomic profiling have delivered results in identifying gene expression profiles associated with invasive bladder cancer. Mitra and colleagues (2009) evaluated 69 genes involved in known cancer pathways and identified a 4-gene panel including JUN, MAP2K6, STAT3, and ICAM 1 that predicted recurrence and survival in patients with Ta-T4 bladder cancer. A group of investigators in Denmark pioneered applications of single nucleotide polymorphism (SNP) array analysis to identify patterns of allelic imbalance associated with invasive cancer (Zieger et al, 2009). Several studies using microarray analysis analyzed gene expression profiles, and Wang and colleagues (2009) recently reported a meta-analysis of 12 studies designed to identify a 59-gene signature that distinguished invasive from noninvasive cancers. These techniques, which can be applied to predict progression and response to chemotherapy or radiotherapy, herald the age of personalized genomic medicine.
Nomograms In The Management Of Patients With Invasive Bladder Cancer
Clinical decision making has evolved over time from physician judgment, to use of overall average outcome prediction, to risk group stratification, and to use of prediction models using Cox multivariate or logistic regression, to use of nomograms, which graphically represent a prediction model for individualized prediction of an end point (Kattan, 2005). The most accurate prediction of outcomes after radical cystectomy (RC) has been based on pathologic staging of the primary tumor and regional lymph node status. Several large, contemporary radical cystectomy studies have documented risks of recurrence and death based on either TNM staging or pathologic groupings that separate organ-confined, extravesical, and node-positive tumors (Stein et al, 2001; Shariat et al, 2006b). Although these traditional prognostic groupings have provided useful estimates for recurrence risk and survival outcomes, significant variation has been observed within each prognostic group. The observed variation in outcomes is related to the heterogeneity of tumor biology and patient characteristics within each pathologic subgroup. Other factors that may affect outcome after stratification by stage and nodal status include lymphovascular invasion, patient age and gender, and histology. Patient comorbidities may also influence outcome. Alternative prognostic models capable of simultaneous evaluation of multiple relevant variables have been shown to provide improved estimates of outcome that can be applied more accurately to an individual patient (Kattan, 2003; Lerner et al, 2007).
Two consortia first reported nomograms for predicting recurrence after radical cystectomy (Fig. 82–4) (Bochner et al, 2006; Karakiewicz et al, 2006a). The Bladder Cancer Research Consortium (BCRC), a collaboration among Baylor College of Medicine, University of Texas Southwestern Medical Center, Johns Hopkins University, and the University of Montreal, studied outcomes following radical cystectomy. The resulting data set was composed of 958 patients from three institutions with 728 evaluable for recurrence. The BCRC group has also developed a suite of nomograms for predicting overall and cancer-specific survival for patients with advanced pathologic T and N stage disease (available online at www.nomogram.org) (Karakiewicz et al, 2006a; Karakiewicz et al, 2006b; Shariat et al, 2006a). The International Bladder Cancer Nomogram was developed by a team from MSKCC and based on data from 4840 evaluable patients (available online at www.mskcc.org/mskcc/html/73854.cfm) (Bochner et al, 2006). The accuracy for predicting recurrence was remarkably similar between groups with concordance indices of 0.78 and 0.75, respectively. Both were significantly more accurate than AJCC TNM and standard pathologic stage groupings.

Figure 82–4 To obtain nomogram-predicted probability of recurrence, locate patient values at each axis. Draw a vertical line to the “Point” axis to determine how many points are attributed for each variable value. Sum the points for all variables. Locate the sum on the “Total Points” line. Draw a vertical line toward the 2YrRecFP, 5YrRecFP, and 8YrRecFP axes to respectively determine the 2-year recurrence-free probability, 5-year recurrence-free probability, and 8-year recurrence-free probability. A, Postoperative nomogram for predicting recurrence after radical cystectomy. B, Postoperative nomogram for predicting recurrence after radical cystectomy incorporating tissue biomarkers. CIS, carcinoma in situ; LVI, lymphovascular invasion.
(A, Reprinted with permission from Karakiewicz PI, Shariat SF, Palapattu GS, et al. Nomogram for predicting disease recurrence after radical cystectomy for transitional cell carcinoma of the bladder. J Urol 2006;176:1354–62, Fig. 3, p. 1359. B, Reprinted with permission from Shariat SF, Karakiewicz PI, Ashfaq R, et al. Multiple biomarkers improve prediction of bladder cancer recurrence and mortality in patients undergoing cystectomy. Cancer 2008;112:315–25, Fig. 2A, p. 321.)
In order to improve on the accuracy of these “first-generation” nomograms, Shariat and colleagues (2008) incorporated several tissue biomarkers. This work, built on research described earlier in the “Molecular Markers in Invasive and Advanced Bladder Cancer” section, demonstrated that altered expression of multiple cell cycle regulatory gene products was independently associated with outcome following radical cystectomy. This “second-generation” nomogram incorporated p53, pRB, p21, p27, and cyclin E1 measured by immunohistochemistry into a nomogram that predicted recurrence and survival for patents with pTa-3N0M0 disease at cystectomy. With the addition of the biomarkers, the accuracy for predicting recurrence and cancer-specific survival compared with accuracy of a nomogram based on TNM stage was increased by 10.9% (83.4% vs. 72.5%, P < .001) and 8.6% (86.9% vs. 78.3, P < .001), respectively. These instruments are based on data acquired retrospectively and must be validated prospectively in order for their utility in clinical practice to be determined.
Management Of Metastatic Bladder Cancer
Systemic combination chemotherapy is the primary treatment modality for patients with metastatic urothelial cancer. Cisplatin-based first-line chemotherapy regimens for urothelial or transitional cell carcinoma include M-VAC or high-density (HD) M-VAC and GC (Sternberg et al, 1989; Loehrer et al, 1992; Saxman et al, 1997; von der Maase et al, 2005; Sternberg et al, 2006b). Despite initial high response rates (RRs) of 40% to 70% in advanced disease, chemotherapy is usually not curative and overall 5-year survival is only approximately 5% to 20% (Table 82–7) (Saxman et al, 1997; von der Maase et al, 2005; Sternberg et al, 2006b; Bellmunt et al, 2007b). A contemporary EORTC/Intergroup randomized trial failed to demonstrate an OS benefit with the addition of paclitaxel to GC (Bellmunt et al, 2007b).
Renal dysfunction (usually defined as a creatinine clearance of <60 mL/min), poor performance status, and advanced age are relatively common and often preclude cisplatin chemotherapy. Carboplatin-based combination regimens are feasible for such patients but appear to be suboptimal compared with cisplatin-based regimens (Petrioli et al, 1996; Bellmunt et al, 1997; Dogliotti et al, 2007).
Ongoing randomized trials specifically evaluate regimens for elderly patients or poor performance status or patients with poor renal function. These studies include comparisons of carboplatin and gemcitabine with methotrexate, vinblastine, and carboplatin (De Santis et al, 2008) and studies of gemcitabine compared with gemcitabine and vinflunine (Culine et al, 2006).
Salvage chemotherapy for advanced TCC with conventional chemotherapeutic agents (taxanes, gemcitabine) usually yields suboptimal response rates and variable median survivals (Table 82–8) (Dreicer et al, 1996; McCaffrey et al, 1997; Lorusso et al, 1998; Sternberg et al, 2001; Albers et al, 2002; Vaughn et al, 2002). However, a nonplatinum taxane-gemcitabine regimen may in some cases be a reasonable alternative for such patients (Dreicer et al, 2003; Calabrò et al, 2009a).
The development of novel and tolerable agents for TCC is clearly warranted, coupled with utilization of factors that may be predictive of response such as ERCC1 (Bellmunt et al, 2007a).
Prognostic Factors
Comparisons among phase II and III studies of metastatic TCC require that prognostic factors be well balanced. Two key factors have been shown to be predictors of a poor prognosis for patients treated with first-line cisplatin-based chemotherapy. A poor performance status (PS) (KPS <80) and the presence of visceral metastases adversely affect survival in patients with locally advanced or metastatic TCC (Bajorin et al, 1999).
In the analysis of patients treated at MSKCC with M-VAC, median survival of patients with zero, one, or two risk factors ranged from 33 to 13.4 to 9.3 months, respectively (Bajorin et al, 1999). In a recent presentation from MSKCC, a nomogram was constructed incorporating four parameters: hemoglobin, serum albumin, KPS, and visceral metastasis (Bajorin et al, 2007). This nomogram needs further validation.
Novel Agents for Advanced Transitional Cell Carcinoma
Several novel agents that are discussed later have been evaluated in patients with advanced and metastatic TCC (Sonpavde and Sternberg, 2008b).
Vinflunine
Vinflunine ditartrate is a novel antitubulin agent obtained from a vinca alkaloid. Salvage therapy with vinflunine plus BSC (best supportive care) was compared with BSC in a multinational randomized trial. Unfortunately, no improvement in survival was demonstrated (Bellmunt Molins et al, 2008). After subset analyses, this agent was approved in the second-line setting by the EMEA. Another ongoing randomized trial compares the combination of frontline vinflunine and gemcitabine with gemcitabine alone in patients ineligible for cisplatin.
Pemetrexed
Pemetrexed is a multitargeted antifolate agent approved for pleural mesothelioma and non–small cell lung cancer. Supplementation of vitamin B12 and folate attenuate its toxicities. Although initial phase II reports with this agent were promising (Paz-Ares et al, 1998; Sweeney et al, 2006) (Table 82–9), enthusiasm has waned after a trial from MSKCC revealed an overall RR of 8% (Galsky et al, 2007). Combined frontline treatment of pemetrexed and gemcitabine also failed to reveal results that were better than those achieved with gemcitabine alone (von der Maase et al, 2006). A phase II trial is evaluating the combination of cisplatin and pemetrexed as frontline therapy for advanced TCC (Table 82–10).
Ixabepilone
The epothilones are novel nontaxane tubulin polymerization agents, and aza-epothilone B is a semisynthetic analog of the natural product, epothilone B. Ixabepilone has been evaluated as second-line therapy of metastatic TCC in a phase II trial in patients who previously had received taxanes (Dreicer et al, 2007) (see Table 82–9).
Larotaxel
Larotaxel is a novel, semisynthetic taxoid with several advantages over docetaxel and paclitaxel. It is active in tumor cells sensitive to docetaxel but also in tumor cells expressing P-glycoprotein, which plays a key role in taxane resistance. Several specific toxicities related to taxanes (nail changes, fluid retention, and allergic reaction) appear to be less frequent with larotaxel. In addition, larotaxel has the potential to cross the blood-brain barrier (Investigator’s Brochure Edition 12, dated April 3, 2006). A randomized study of larotaxel and cisplatin (LC) versus GC in the firstline treatment of locally advanced/metastatic urothelial tract or bladder cancer is under way. The primary end point is overall survival.
Nanoparticle Albumin-Bound (Nab) Paclitaxel
Nab-paclitaxel is a novel solvent-free, albumin-bound formulation of paclitaxel designed to avoid solvent-related toxicities and to deliver paclitaxel to tumors via molecular pathways involving an endothelial cell-surface albumin receptor and an albumin-binding protein expressed by tumor cells and secreted into the tumor interstitium (secreted protein acid rich in cysteine [SPARC]) (Nyman et al, 2005). Nab-paclitaxel is being evaluated both for the salvage therapy of progressive TCC and as a component of combination regimens in the neoadjuvant setting (see Table 82–10).
Oxaliplatin
Oxaliplatin is a non-nephrotoxic third-generation platinum analogue that has been studied in previously-treated metastatic TCC in a phase II trial (Winquist et al, 2005) (see Table 82–9). Combinations of oxaliplatin and gemcitabine and of docetaxel and oxaliplatin may be of some interest (see Table 82–10).
Monoclonal Antibodies
Trastuzumab
HER2/neu expression in urothelial cancers is variable and may be associated with a more aggressive clinical course (Lonn et al, 1995). In a phase II trial, patients with metastatic TCC or squamous cell carcinoma that expressed HER2/neu (by immunohistochemistry f, serology, or fluorescence in situ hybridization [FISH]) were treated with trastuzumab in combination with paclitaxel, carboplatin, and gemcitabine (Hussain et al, 2007). Thirty-one (70%) of 44 HER2/neu-positive patients responded (5 CRs and 26 PRs), with 25 (57%) confirmed responses. These outcomes are promising, and a randomized trial of gemcitabine and cisplatin with or without trastuzumab has been conducted in France. One thousand patients have been screened, and 60 have actually been included in this trial with an HER2 positivity of only around 5% to 10%. Trastuzumab is also being evaluated in combination with paclitaxel and radiotherapy in a bladder preservation strategy (Table 82–11).
Bevacizumab
VEGF (vascular endothelial growth factor) receptors are expressed in TCC, and preclinical evidence supports the antitumor efficacy of targeting this pathway in combination with chemotherapy (Inoue et al, 2000). A phase II trial by the Hoosier Oncology Group (HOG) has evaluated frontline GC plus bevacizumab for metastatic TCC (Hahn et al, 2009). Gemcitabine was reduced to 1000 mg/m2 intravenous days 1 and 8 after the first 17 patients were entered because of 7 thrombotic DVT or PE events. The Cancer and Leukemia Group B (CALGB) is nonetheless planning a frontline randomized phase III trial of GC versus GC and bevacizumab (see Table 82–11).
Cetuximab
Human TCCs overexpress epidermal growth factor receptor (EGFR), which confers a poor prognosis (Sriplakich et al, 1999). Cetuximab is an intravenously administered EGFR monoclonal antibody commonly used in colorectal cancer and in head and neck cancers. A trial is planned to evaluate the combination of cetuximab with frontline GC, as well as with salvage paclitaxel (see Table 82–11).
Small-Molecule Biologic Agents
EGFR and HER2 Receptor Tyrosine Kinase Inhibitors (TKIs)
The CALGB reported a phase II trial of frontline GC and gefitinib (an orally bioavailable EGFR TKI) in metastatic urothelial cancer, with an RR of 51% and median survival of 14.4 months (Philips et al, 2006). This is now under evaluation in a randomized study of GC with or without gefitinib (see Table 82–9). Erlotinib, another oral EGFR TKI, used commonly in the treatment of non–small cell lung cancer, is being studied in the neoadjuvant setting before cystectomy with primarily correlative and pharmacodynamic end points (see Table 82–11).
Lapatinib is an oral TKI that targets EGFR and HER2 and has been successful in combination with capecitabine chemotherapy in breast cancer. In a preliminary report of a phase II trial of 59 patients with EGFR and/or HER2 expression (1+ to 3+ by IHC), lapatinib was active as salvage therapy for advanced TCC with PRs in 3% and clinical benefit (response + stability ≥ 16 weeks) in 12% of patients (Wuelfing et al, 2005). The median time to progression was 8.6 weeks, and there was a trend toward clinical benefit in those with EGFR or HER2 2+/3+ by IHC. A randomized discontinuation trial being conducted by the U.S. Oncology Research network is evaluating salvage therapy with lapatinib in HER2 expressing (by FISH) metastatic TCC. The EORTC is evaluating the combination of GC and lapatinib in a phase I dose escalation study (see Table 82–11).
VEGF Receptor TKIs
Sorafenib was the first orally bioavailable multitargeted receptor TKI to be approved for use as second-line therapy in advanced renal cancer. It was designed as a c- and b-raf kinase inhibitor since the ras/raf signaling pathway mediates tumor cell proliferation and angiogenesis. Sorafenib also inhibits several receptor tyrosine kinases, among them VEGF receptor (R)-2, PDGFR-β, Flt-3, and c-KIT. Sorafenib as a single agent was ineffective as second-line therapy for advanced TCC (Dreicer, 2008). However, the combination of sorafenib with carboplatin/cisplatin and gemcitabine is being evaluated for frontline therapy (see Table 82–11).
Sunitinib, another small-molecule, multitargeted receptor TKI taken orally, is also approved for the therapy of renal cell cancer. It targets VEGFR-2, PDGFR-β, KIT, and Flt-3 receptors. A preclinical study recently demonstrated significant activity of sunitinib both as a single agent and in combination with cisplatin (Sonpavde et al, 2007). In addition, activity has been demonstrated in a phase II trial of sunitinib as salvage therapy for TCC (Gallagher et al, 2007). Another trial compares sunitinib with placebo in patients who are stable or responding to frontline chemotherapy (see Table 82–9). The Cleveland Clinic is evaluating neoadjuvant sunitinib alone with primarily correlative studies, and another neoadjuvant phase II trial of sunitinib in combination with GC is planned by the HOG.
Axitinib, a perhaps more potent VEGFR TKI, caused regression of subcutaneous human TCC xenografts and inhibited angiogenesis and VEGFR-2 and PDGFR-β phosphorylation (Zhu et al, 2006). This molecule is clearly of interest for future studies. Another randomized phase II trial led by the Dana Farber Cancer Institute is evaluating salvage docetaxel alone or with vandetanib, a dual EGFR and VEGFR-TKI in the second-line setting (see Table 82–9).
Other Novel Agents
On the basis of ER-β expression in urothelial cancers, which increases with increasing stage and grade, and the inhibitory effect of selective estrogen receptor modulators in preclinical models, salvage therapy with oral tamoxifen is being evaluated in a multi-institutional phase II trial of advanced UC at Baylor College of Medicine in collaboration with colleagues in Los Angeles and Rome (see Table 82–9).
Neoadjuvant Therapy To Develop Novel Agents
Among novel therapies, neoadjuvant therapy before cystectomy is an interesting model. Pathologic complete remission (pCR) is used as a surrogate for the efficacy of neoadjuvant chemotherapy and long-term outcomes (Grossman et al, 2003b). Neoadjuvant therapy with cisplatin-based regimens (M-VAC, GC) has been shown to be safe and does not increase the risk of postoperative complications (Grossman et al, 2003b; Dash et al, 2008). Therefore the neoadjuvant paradigm may accelerate the development of novel agents. Owing to the availability of tissue before and after chemotherapy, it may be possible to determine molecular and biologic characteristics that predict chemosensitivity.
The choice of novel agents should be based on the knowledge of potential molecular targets emerging from studies examining urothelial cancer biology. If biologic activity can be demonstrated in initial small pilot trials (e.g., 15 to 25 patients with an appropriate statistical design to detect a biologic effect), additional larger phase II and randomized phase II studies of novel agents alone or in combination may be planned with more rigorous efficacy end points (pCR). Additional goals may be discovering markers predictive for efficacy including global gene expression assays. Before initiation of trials that require large sample sizes, careful consideration of potential activity may be achieved using a neoadjuvant strategy.
Predictive Factors
In order to guide optimal patient selection, the discovery of predictive factors should proceed with the development of novel agents. Levels of DNA-repair genes including ERCC1, RRM1, BRCA1, and caveolin-1 have been evaluated in 57 patients with advanced bladder cancer treated with either GC or PCG (Bellmunt et al, 2007a). Median survival was significantly higher in patients with low ERCC1 levels (25.4 vs. 15.4 months; P = .03). A trend toward longer time to progression was observed in patients with tumors, expressing low levels of all markers. On multivariate analysis of pretreatment prognostic factors, ERCC1 emerged as an independent predictive factor for survival. The results indicate that ERCC1 may predict survival in urothelial cancer treated with platinum-based therapy.
Conclusions
Invasive bladder cancer should be viewed as diseases with both loco-regional and systemic implications requiring an integrated multidisciplinary treatment approach. Attention to accurate staging and surgical quality including the extent and completeness of the node dissection are significant determinants of long-term survival. Quality of life can be maintained with appropriate sexual function–preserving surgical techniques and continent urinary diversion in motivated patients. Both neoadjuvant and adjuvant chemotherapy continue to play a role in treatment of locally advanced bladder cancer, although meta-analyses reported in currently available studies lean more toward the use of neoadjuvant chemotherapy. Concomitant chemoradiation is an excellent strategy for selected patients in whom bladder preservation can be considered. Future investigation will focus on optimizing radiation techniques including the possibility of radiosensitization. Progress in the treatment of muscle-invasive and advanced TCC hopefully will be made with the addition and rational use of novel biologic agents to current cisplatin-based combination chemotherapeutic strategies.
Thorough understanding of biology is necessary in order to improve outcomes. Employment of the neoadjuvant paradigm may be a powerful tool to accelerate drug development and discovery of factors predictive of efficacy. Factors predictive of response to specific agents need to be better defined in order to facilitate more personalized strategies. Close coordination among all disciplines is required to achieve optimal results, incorporating more effective systemic chemotherapy and the proper selection of patients based on predictive molecular markers.
Key Points
Advanced Bladder Cancer (ABC) Meta-analysis Collaboration. Adjuvant chemotherapy in invasive bladder cancer: a systematic review and meta-analysis of individual patient data. Eur Urol. 2005;48(2):189-199. discussion 199–201
Advanced Bladder Cancer (ABC) Meta-analysis Collaboration. Neoadjuvant chemotherapy in invasive bladder cancer: update of a systematic review and meta-analysis of individual patient data advanced bladder cancer (ABC) meta-analysis collaboration. Eur Urol. 2005;48(2):202-205.
Bochner BH, Herr HW, Reuter VE. Impact of separate versus en bloc pelvic lymph node dissection on the number of lymph nodes retrieved in cystectomy specimens. J Urol. 2001;166(6):2295-2296.
Dutta SC, Smith JAJr, Shappell SB, et al. Clinical under staging of high risk nonmuscle invasive urothelial carcinoma treated with radical cystectomy. J Urol. 2001;166(2):490-493.
George B, Datar RH, Wu L, et al. p53 gene and protein status: the role of p53 alterations in predicting outcome in patients with bladder cancer. J Clin Oncol. 2007;25(34):5352-5358.
Grossman HB, Natale RB, Tangen CM, et al. Neoadjuvant chemotherapy plus cystectomy compared with cystectomy alone for locally advanced bladder cancer. N Engl J Med. 2003;349(9):859-866.
Hautmann RE, Gschwend JE, de Petriconi RC, et al. Cystectomy for transitional cell carcinoma of the bladder: results of a surgery only series in the neobladder era. J Urol. 2006;176:486-492. discussion 491–2
Herr HW. Superiority of ratio based lymph node staging for bladder cancer. J Urol. 2003;169:943-945.
Saxman SB, Propert KJ, Einhorn LH, et al. Long-term follow-up of a phase III intergroup study of cisplatin alone or in combination with methotrexate, vinblastine, and doxorubicin in patients with metastatic urothelial carcinoma: a cooperative group study. J Clin Oncol. 1997;15(7):2564-2569.
Shabsigh A, Korets R, Vora KC, et al. Defining early morbidity of radical cystectomy for patients with bladder cancer using a standardized reporting methodology. Eur Urol. 2009;55(1):164-176.
Skinner DG. Management of invasive bladder cancer: a meticulous pelvic node dissection can make a difference. J Urol. 1982;128(1):34-36.
Stein JP, Lieskovsky G, Cote R, et al. Radical cystectomy in the treatment of invasive bladder cancer: long-term results in 1,054 patients. J Clin Oncol. 2001;19(3):666-675.
von der Maase H, Sengelov L, Roberts JT, et al. Long-term survival results of a randomized trial comparing gemcitabine plus cisplatin, with methotrexate, vinblastine, doxorubicin, plus cisplatin in patients with bladder cancer. J Clin Oncol. 2005;23(21):4602-4608.
Abol-Enein H, El Makresh M, El Baz M, Ghoneim M. Neo-adjuvant chemotherapy in treatment of invasive transitional bladder cancer: a controlled, prospective randomised study. Br J Urol. 1997;80(Suppl. 2):49.
Advanced Bladder Cancer (ABC) Meta-analysis Collaboration. Adjuvant chemotherapy in invasive bladder cancer: a systematic review and meta-analysis of individual patient data. Eur Urol. 2005;48(2):189-199. discussion 199–201
Advanced Bladder Cancer (ABC) Meta-analysis Collaboration. Neoadjuvant chemotherapy in invasive bladder cancer: update of a systematic review and meta-analysis of individual patient data advanced bladder cancer (ABC) meta-analysis collaboration. Eur Urol. 2005;48(2):202-205.
Akamatsu S, Kanamaru S, Ishihara M, et al. Primary large cell neuroendocrine carcinoma of the urinary bladder. Int J Urol. 2008;15(12):1080-1083.
Albers P, Siener R, Perabo FG, et al. Gemcitabine monotherapy as 2nd line treatment in cisplatin refractory transitional cell carcinoma—prognostic factors for response and improvement of quality of life. Onkologie. 2002;25:47-52.
Algaba F. Lymphovascular invasion as a prognostic tool for advanced bladder cancer. Curr Opin Urol. 2006;16(5):367-371.
Ali-El-Dein B, Gomha M, Ghoneim MA. Critical evaluation of the problem of chronic urinary retention after orthotopic bladder substitution in women. J Urol. 2002;168(2):587-592.
Amling CL, Thrasher JB, Frazier HA, et al. Radical cystectomy for stages Ta, Tis and T1 transitional cell carcinoma of the bladder. J Urol. 1994;151(1):31-35.
Andrews B, Shariat SF, Kim JH, et al. Preoperative plasma levels of interleukin-6 and its soluble receptor predict disease recurrence and survival of patients with bladder cancer. J Urol. 2002;167(3):1475-1481.
Ather MH, Alam Z, Jamshaid A, et al. Separate submission of standard lymphadenectomy in 6 packets versus en bloc lymphadenectomy in bladder cancer. Urol J. 2008;5(2):94-98.
Bagrodia A, Grover S, Srivastava A, et al. Impact of body mass index on clinical and cost outcomes after radical cystectomy. BJU Int. 2009;104(3):326-330.
Bajorin DF, Dodd PM, Mazumdar M, et al. Long-term survival in metastatic transitional-cell carcinoma and prognostic factors predicting outcome of therapy. J Clin Oncol. 1999;17(10):3173-3181.
Bajorin DF, Ostrovnaya I, Iasonos A, et al. A nomogram predicting survival of patients (pts) with metastatic or unresectable urothelial cancer (UC) treated with cisplatin-based chemotherapy. Proc Annu Meet Am Soc Clin Oncol. 25(18S), 2007.
Balaji KC, McGuire M, Grotas J, et al. Upper tract recurrences following radical cystectomy: an analysis of prognostic factors, recurrence pattern and stage at presentation. J Urol. 1999;162(5):1603-1606.
Bamias A, Aravantinos G, Deliveliotis C. Docetaxel and cisplatin with granulocyte colony-stimulating factor (G-CSF) versus M-VAC with G-CSF in advanced urothelial carcinoma: a multicenter, randomized, phase III study from the Hellenic Cooperative Oncology Group. J Clin Oncol. 2004;22:220-228.
Bassi P, Pagano F, Pappagallo G, et al. Neo-adjuvant M-VAC of invasive bladder cancer: the G.U.O.N.E. multicenter phase III trial. Eur Urol. 1998;33(Suppl. 1):142.
Bellmunt J, Paz-Ares L, Cuello M, et al. Gene expression of ERCC1 as a novel prognostic marker in advanced bladder cancer patients receiving cisplatin-based chemotherapy. Ann Oncol. 2007;18(3):522-528.
Bellmunt J, Ribas A, Eres N, et al. Carboplatin-based versus cisplatin-based chemotherapy in the treatment of surgically incurable advanced bladder carcinoma. Cancer. 1997;80:1966-1972.
Bellmunt J, von der Maase H, Mead GM, et al. Randomized phase III study comparing paclitaxel/cisplatin/gemcitabine and gemcitabine/cisplatin in patients with locally advanced or metastatic urothelial cancer without prior systemic therapy: EORTC 30987/Intergroup Study. Proc Annu Meet Am Soc Clin Oncol. 2007;25(18S):242s.
Bellmunt Molins J, von der Maase H, Theodore C, et al. Randomised phase III trial of vinflunine (V) plus best supportive care (B) vs B alone as 2nd line therapy after a platinum-containing regimen in advanced transitional cell carcinoma of the urothelium (TCCU). Proc Annu Meet Am Soc Clin Oncol. 2008:26.
Benedict WF, Lerner SP, Zhou J, et al. Level of retinoblastoma protein expression correlates with p16 (MTS-1/INK4A/CDKN2) status in bladder cancer. Oncogene. 1999;18(5):1197-1203.
Bochner BH, Cho D, Herr HW, et al. Prospectively packaged lymph node dissections with radical cystectomy: evaluation of node count variability and node mapping. J Urol. 2004;172(4 Pt. 1):1286-1290.
Bochner BH, Esrig D, Groshen S, et al. Relationship of tumor angiogenesis and nuclear p53 accumulation in invasive bladder cancer. Clin Cancer Res. 1997;3(9):1615-1622.
Bochner BH, Herr HW, Reuter VE. Impact of separate versus en bloc pelvic lymph node dissection on the number of lymph nodes retrieved in cystectomy specimens. J Urol. 2001;166(6):2295-2296.
Bochner BH, Kattan MW, Vora KC. Postoperative nomogram predicting risk of recurrence after radical cystectomy for bladder cancer. J Clin Oncol. 2006;24(24):3967-3972.
Bono AV, Benvenuti C, Gibba A, et al. Adjuvant chemotherapy in locally advanced bladder cancer. Final analysis of a controlled multicentre study. Acta Urol Ital. 1997;11:5-8.
Byrne RR, Shariat SF, Brown R, et al. E-cadherin immunostaining of bladder transitional cell carcinoma, carcinoma in situ and lymph node metastases with long-term followup. J Urol. 2001;165(5):1473-1479.
Calabrò F, Lorusso V, Rosati G, et al. Gemcitabine and paclitaxel every 2 weeks in patients with previously untreated urothelial carcinoma. Cancer. 2009;115(12):2652-2659.
Calabrò F, Sternberg CN. Neoadjuvant and adjuvant chemotherapy in muscle-invasive bladder cancer. Eur Urol. 2009;55(2):348-358.
Capitanio U, Suardi N, Shariat SF, et al. Assessing the minimum number of lymph nodes needed at radical cystectomy in patients with bladder cancer. BJU Int. 2009;103(10):1359-1362.
Chang SS, Cole E, Smith JAJr, Cookson MS. Pathological findings of gynecologic organs obtained at female radical cystectomy. J Urol. 2002;168(1):147-149.
Chang SS, Hassan JM, Cookson MS, et al. Delaying radical cystectomy for muscle invasive bladder cancer results in worse pathological stage. J Urol. 2003;170(4 Pt. 1):1085-1087.
Chatterjee SJ, Datar R, Youssefzadeh D, et al. Combined effects of p53, p21, and pRb expression in the progression of bladder transitional cell carcinoma. J Clin Oncol. 2004;22(6):1007-1013.
Chatterjee SJ, George B, Goebell PJ, et al. Hyperphosphorylation of pRb: a mechanism for RB tumour suppressor pathway inactivation in bladder cancer. J Pathol. 2004;203(3):762-770.
Cognetti F, Ruggeri EM, Felici A, et al. Adjuvant chemotherapy (AC) with cisplatin + gemcitabine (CG) versus chemotherapy (CT) at relapse (CR) in patients (pts) with muscle-invasive bladder cancer (MIBC) submitted to radical cystectomy (RC). An Italian multicenter randomised phase III trial. Proc Annu Meet Am Soc Clin Oncol. 2008:26.
Cook AM, Huddart RA, Jay G, et al. The utility of tumour markers in assessing the response to chemotherapy in advanced bladder cancer. Br J Cancer. 2000;82(12):1952-1957.
Coppin CM, Gospodarowicz MK, James K, et al. Improved local control of invasive bladder cancer by concurrent cisplatin and preoperative or definitive radiation. The National Cancer Institute of Canada Clinical Trials Group. J Clin Oncol. 1996;14(11):2901-2907.
Cristofanilli M, Budd GT, Ellis MJ, et al. Circulating tumor cells, disease progression, and survival in metastatic breast cancer. N Engl J Med. 2004;351(8):781-791.
Culine S, Theodore C, De Santis M, et al. A phase II study of vinflunine in bladder cancer patients progressing after first-line platinum-containing regimen. Br J Cancer. 2006;94(10):1395-1401.
Dash A, Pettus JAIV, Herr HW, et al. A role for neoadjuvant gemcitabine plus cisplatin in muscle-invasive urothelial carcinoma of the bladder: a retrospective experience. Cancer. 2008;113(9):2471-2477.
David KA, Milowsky MI, Ritchey J, et al. Low incidence of perioperative chemotherapy for stage III bladder cancer 1998 to 2003: a report from the National Cancer Data Base. J Urol. 2007;178(2):451-454.
De Santis M, Bellmunt Molins J, de Wit R, et al. Randomized phase II/III trial assessing gemcitabine/carboplatin (GC) and methotrexate/carboplatin/vinblastine (M-CAVI) in patients (pts) with advanced urothelial cancer (UC) “unfit” for cisplatin based chemotherapy (CHT): updated phase II results and risk group analysis of EORTC study 30986. Proc Annu Meet Am Soc Clin Oncol. 2008:26.
Deserno WM, Harisinghani MG, Taupitz M, et al. Urinary bladder cancer: preoperative nodal staging with ferumoxtran-10-enhanced MR imaging. Radiology. 2004;233(2):449-456.
DeVere White RW, Lara PNJr, Goldman B, et al. A sequential treatment approach to myoinvasive urothelial cancer: a phase II Southwest Oncology Group trial (S0219). J Urol. 2009;181(6):2476-2480.
Dogliotti L, Cartenì G, Siena S, et al. Gemcitabine plus cisplatin versus gemcitabine plus carboplatin as first-line chemotherapy in advanced transitional cell carcinoma of the urothelium: results of a randomized phase 2 trial. Eur Urol. 2007;52(1):134-141.
Donat SM, Wei DC, McGuire MS, Herr HW. The efficacy of transurethral biopsy for predicting the long-term clinical impact of prostatic invasive bladder cancer. J Urol. 2001;165(5):1580-1584.
Donat SM, Shabsigh A, Savage C, et al. Potential impact of postoperative early complications on the timing of adjuvant chemotherapy in patients undergoing radical cystectomy: a high-volume tertiary cancer center experience. Eur Urol. 2009;55(1):177-186.
Dotan ZA, Kavanagh K, Yossepowitch O, et al. Positive surgical margins in soft tissue following radical cystectomy for bladder cancer and cancer specific survival. J Urol. 2007;178(6):2308-2312. discussion 2313
Dreicer R, DiPaola P, Eleff M, et al. Phase II trial of sorafenib in advanced carcinoma of the urothelium (E 1804): a trial of the Eastern Cooperative Oncology Group. Proc Annu Meet Am Soc Clin Oncol. 2008;26(15S):270s.
Dreicer R, Gustin DM, See WA, Williams RD. Paclitaxel in advanced urothelial carcinoma: its role in patients with renal insufficiency and as salvage therapy. J Urol. 1996;156(5):1606-1608.
Dreicer R, Li S, Manola J, et al. Phase II trial of epothilone B analog BMS-247550 (ixabepilone) in advanced carcinoma of the urothelium (E3800): a trial of the Eastern Cooperative Oncology Group. Cancer. 2007;110:759-763.
Dreicer R, Manola J, Schneider DJ, et al. Phase II trial of gemcitabine and docetaxel in patients with advanced carcinoma of the urothelium: a trial of the Eastern Cooperative Oncology Group. Cancer. 2003;97(11):2743-2747.
Droz JP. Editorial comment: neoadjuvant chemotherapy in invasive bladder cancer: update of a systematic review and meta-analysis of individual patient data: Advanced Bladder Cancer (ABC) Meta-analysis Collaboration. Eur Urol. 2005;48(2):205-206.
Dutta SC, Smith JAJr, Shappell SB, et al. Clinical under staging of high risk nonmuscle invasive urothelial carcinoma treated with radical cystectomy. J Urol. 2001;166(2):490-493.
Edge SB, Byrd DR, Compton CC, et al, editors. AJCC cancer staging manual, 7th ed, New York: Springer, 2010.
el-Mekresh M, Akl A, Mosbah A, et al. Prediction of survival after radical cystectomy for invasive bladder carcinoma: risk group stratification, nomograms or artificial neural networks? J Urol. 2009;182(2):466-472. discussion 472
Esrig D, Freeman JA, Elmajian DA, et al. Transitional cell carcinoma involving the prostate with a proposed staging classification for stromal invasion. J Urol. 1996;156(3):1071-1076.
Fernando SA, Sandler HM. Multimodality bladder preservation therapy for muscle-invasive bladder tumors. Sem Oncol. 2007;34:129-134.
Frazier HA, Robertson JE, Dodge RK, Paulson DF. The value of pathologic factors in predicting cancer-specific survival among patients treated with radical cystectomy for transitional cell carcinoma of the bladder and prostate. Cancer. 1993;71(12):3993-4001.
Freiha F, Reese J, Torti FM. A randomized trial of radical cystectomy versus radical cystectomy plus cisplatin, vinblastine and methotrexate chemotherapy for muscle invasive bladder cancer. J Urol. 1996;155(2):495-499.
Gallagher DJ, Milowsky MI, Gerst SR, et al. Phase II study of sunitinib in patients (pts) with relapsed or refractory urothelial carcinoma (UC). Proc Annu Meet Am Soc Clin Oncol. 2007;25(18S):318s.
Gallagher DJ, Milowsky MI, Ishill N, et al. Detection of circulating tumor cells in patients with urothelial cancer. Ann Oncol. 2009;20(2):305-308.
Galsky MD, Mironov S, Iasonos A, et al. Phase II trial of pemetrexed as second-line therapy in patients with metastatic urothelial carcinoma. Invest New Drugs. 2007;25(3):265-270.
George B, Datar RH, Wu L, et al. p53 gene and protein status: the role of p53 alterations in predicting outcome in patients with bladder cancer. J Clin Oncol. 2007;25(34):5352-5358.
Ghoneim MA, Abdel-Latif M, el-Mekresh M, et al. Radical cystectomy for carcinoma of the bladder: 2,720 consecutive cases 5 years later. J Urol. 2008;180(1):121-127.
GISTV (Italian Bladder Cancer Study Group). Neoadjuvant treatment for locally advanced bladder cancer: a randomized prospective clinical trial. J Chemother. 1996;8:345-346.
Goodman OBJr, Fink LM, Symanowski JT, et al. Circulating tumor cells in patients with castration-resistant prostate cancer baseline values and correlation with prognostic factors. Cancer Epidemiol Biomarkers Prev. 2009;18(6):1904-1913.
Gopalan A, Sharp DS, Fine SW, et al. Urachal carcinoma: a clinicopathologic analysis of 24 cases with outcome correlation. Am J Surg Pathol. 2009;33(5):659-668.
Greene FL, Page DL, Fleming ID, et al. AJCC cancer staging manual, 6th ed. Berlin: Springer Verlag; 2002.
Grossfeld GD, Ginsberg DA, Stein JP, et al. Thrombospondin-1 expression in bladder cancer: association with p53 alterations, tumor angiogenesis, and tumor progression. J Natl Cancer Inst. 1997;89(3):219-227.
Grossman HB, Natale RB, Tangen CM, et al. Neoadjuvant chemotherapy plus cystectomy compared with cystectomy alone for locally advanced bladder cancer. N Engl J Med. 2003;349(9):859-866.
Grossman HB, Natale RB, Tangen CM, et al. Errata Corrige for: Neoadjuvant chemotherapy plus cystectomy compared with cystectomy alone for locally advanced bladder cancer. N Engl J Med. 2003;349(9):1880.
Hahn NM, Stadler WM, Zon RT, et al. A multicenter phase II study of cisplatin (C), gemcitabine (G), and bevacizumab (B) as first-line therapy for metastatic urothelial carcinoma (UC): Hoosier Oncology Group GU04-75. Proc Annu Meet Am Soc Clin Oncol. 2009;27(15S, I):239s.
Hall MC, Swanson DA, Dinney CP. Complications of radical cystectomy: impact of the timing of perioperative chemotherapy. Urology. 1996;47(6):826-830.
Hall RR. Updated results of a randomised controlled trial of neoadjuvant cisplatin (C), methotrexate (M) and vinblastine (V) chemotherapy for muscle-invasive bladder cancer. International Collaboration of Trialists of the MRC Advanced Bladder Cancer Group. Proc Annu Meet Am Soc Clin Oncol. 2002;21(1):178a.
Hardeman SW, Soloway MS. Urethral recurrence following radical cystectomy. J Urol. 1990;144:666-669.
Hautmann RE, Gschwend JE, de Petriconi RC, et al. Cystectomy for transitional cell carcinoma of the bladder: results of a surgery only series in the neobladder era. J Urol. 2006;176:486-492. discussion 491–2
Hellenthal NJ, Ramirez ML, Evans CP, et al. Trends in pelvic lymphadenectomy at the time of radical cystectomy: 1988 to 2004. J Urol. 2009;181(6):2490-2495.
Heney NM, Kaufman DS, Shipley WU. Surgery: selective bladder-preserving therapy for muscle-invasive cancer. Nat Rev Clin Oncol. 2009;6(4):193-194.
Herr HW. Uncertainty and outcome of invasive bladder tumors. Urol Oncol. 1996;2:92-95.
Herr HW. Transurethral resection of muscle-invasive bladder cancer: 10-year outcome. J Clin Oncol. 2001;19:89-93.
Herr HW. Superiority of ratio based lymph node staging for bladder cancer. J Urol. 2003;169:943-945.
Herr HW. Editorial comment: bladder preservation in selected patients with muscle-invasive bladder cancer by complete transurethral resection of the bladder plus systemic chemotherapy: long-term follow-up of a phase 2 nonrandomized comparative trial with radical cystectomy. Eur Urol. 2009;55(4):920-921.
Herr HW, Bajorin DF, Scher HI. Neoadjuvant chemotherapy and bladder sparing surgery for invasive bladder cancer: ten-year outcome. J Clin Oncol. 1998;16(4):1298-1301.
Herr HW, Bochner BH, Dalbagni G, et al. Impact of the number of lymph nodes retrieved on outcome in patients with muscle invasive bladder cancer. J Urol. 2002;167:1295-1298.
Herr HW, Donat SM, Dalbagni G. Can restaging transurethral resection of T1 bladder cancer select patients for immediate cystectomy? J Urol. 2007;177:75-79. discussion 79
Herr HW, Faulkner JR, Grossman HB, et al. Surgical factors influence bladder cancer outcomes: a cooperative group report. J Clin Oncol. 2004;22(14):2781-2789.
Herr HW, Scher HI. Surgery of invasive bladder cancer: is pathologic staging necessary? Sem Oncol. 1990;17:590-597.
Herrmann E, Stoter E, van Ophoven A, et al. The prognostic impact of pelvic lymph node metastasis and lymphovascular invasion on bladder cancer. Int J Urol. 2008;15(7):607-611.
Hollenbeck BK, Ye Z, Wong SL, et al. Hospital lymph node counts and survival after radical cystectomy. Cancer. 2008;112(4):806-812.
Holzbeierlein JM, Lopez-Corona E, Bochner BH, et al. Partial cystectomy: a contemporary review of the Memorial Sloan-Kettering Cancer Center experience and recommendations for patient selection. J Urol. 2004;172:878-881.
Housset M, Dufour B, Maulard-Durdux C, et al. Concomitant fluorouracil (5-FU)-cisplatin (CDDP) and bifractionated split course radiation therapy (BSCRT) for invasive bladder cancer. Proc Am Soc Clin Oncol. 1997;16:319A.
Hussain MH, MacVicar GR, Petrylak DP, et al. Trastuzumab, paclitaxel, carboplatin, and gemcitabine in advanced human epidermal growth factor receptor-2/neu-positive urothelial carcinoma: results of a multicenter phase II National Cancer Institute trial. J Clin Oncol. 2007;25(16):2118-2124.
Inoue K, Slaton JW, Davis DW, et al. Treatment of human metastatic transitional cell carcinoma of the bladder in a murine model with the anti-vascular endothelial growth factor receptor monoclonal antibody DC101 and paclitaxel. Clin Cancer Res. 2000;6(7):2635-2643.
International Collaboration of Trialists. Neoadjuvant cisplatin, methotrexate, and vinblastine chemotherapy for muscle-invasive bladder cancer: a randomised controlled trial. Lancet. 1999;354(9178):533-540.
Izes JK, Dyer MW, Callum MG, et al. Ca 125 as a marker of tumor activity in advanced urothelial malignancy. J Urol. 2001;165:1908-1913.
Jin JO, Boyar M, Petrylak DP, Stadler W. Adjuvant chemotherapy for invasive bladder cancer. In: Lerner SP, Schoenberg MP, Sternberg CN, editors. Textbook of bladder cancer. London: Abingdon, Taylor & Francis; 2006:665-670.
Kachnic LA, Kaufman DS, Heney NM. Bladder preservation by combined modality therapy for invasive bladder cancer. J Clin Oncol. 1997;15(3):1022-1029.
Kamat AM, Gee JR, Dinney CP, et al. The case for early cystectomy in the treatment of nonmuscle invasive micropapillary bladder carcinoma. J Urol. 2006;175:881-885.
Karakiewicz PI, Shariat SF, Palapattu GS, et al. Nomogram for predicting disease recurrence after radical cystectomy for transitional cell carcinoma of the bladder. J Urol. 2006;176(4):1354-1362.
Karakiewicz PI, Shariat SF, Palapattu GS, et al. Precystectomy nomogram for prediction of advanced bladder cancer stage. Eur Urol. 2006;50(6):1254-1260. discussion 1261–2
Karam JA, Lotan Y, Karakiewicz PI, et al. Use of combined apoptosis biomarkers for prediction of bladder cancer recurrence and mortality after radical cystectomy. Lancet Oncol. 2007;8:128-136.
Kassouf W, Agarwal PK, Herr HW, et al. Lymph node density is superior to TNM nodal status in predicting disease-specific survival after radical cystectomy for bladder cancer: analysis of pooled data from MDACC and MSKCC. J Clin Oncol. 2008;26:121-126.
Kassouf W, Swanson D, Kamat AM, et al. Partial cystectomy for muscle invasive urothelial carcinoma of the bladder: a contemporary review of the M.D. Anderson Cancer Center experience. J Urol. 2006;175:2058-2062.
Kattan MW. Nomograms are superior to staging and risk grouping systems for identifying high-risk patients: preoperative application in prostate cancer. Curr Opin Urol. 2003;13(2):111-116.
Kattan MW. When and how to use informatics tools in caring for urologic patients. Nat Clin Pract Urol. 2005;2:183-190.
Kaufman DS, Shipley WU, Griffin PP, et al. Selective bladder preservation by combination treatment of invasive bladder cancer. N Engl J Med. 1993;4(329):1377-1382.
Kaufman DS, Shipley WU, Feldman AS. Bladder cancer. Lancet. 2009;374(9685):239-249.
Kaufman DS, Winter KA, Shipley WU, et al. A phase I/II RTOG study (99-06) in patients with muscle-invading bladder cancer of transurethral surgery plus paclitaxel, cisplatin and twice daily irradiation followed by either selective bladder preservation or radical cystectomy and adjuvant chemotherapy. Urology. 2009;73:833-837.
Kibel AS, Dehdashti F, Katz MD, et al. Prospective study of [18f]fluorodeoxyglucose positron emission tomography/computed tomography for staging of muscle-invasive bladder carcinoma. J Clin Oncol. 2009;27(26):4314-4320.
Kiemeney LA, Witjes JA, Heijbroek RP, et al. Should random urothelial biopsies be taken from patients with primary superficial bladder cancer? A decision analysis. Members of the Dutch South-East Co-Operative Urological Group. Br J Urol. 1994;73:164-171.
Koga F, Kihara K, Fujii Y, et al. Favourable outcomes of patients with clinical stage T3N0M0 bladder cancer treated with induction low-dose chemo-radiotherapy plus partial or radical cystectomy vs immediate radical cystectomy: a single-institutional retrospective comparative study. BJU Int. 2009;104(2):189-194.
Konety BR, Dhawan V, Allareddy V, Joslyn SA. Impact of hospital and surgeon volume on in-hospital mortality from radical cystectomy: data from the health care utilization project. J Urol. 2005;173(5):1695-1700.
Konety BR, Joslyn SA, O’Donnell MA. Extent of pelvic lymphadenectomy and its impact on outcome in patients diagnosed with bladder cancer: analysis of data from the surveillance, epidemiology and end results program data base. J Urol. 2003;169:946-950.
Koppie TM, Vickers AJ, Vora K, et al. Standardization of pelvic lymphadenectomy performed at radical cystectomy: can we establish a minimum number of lymph nodes that should be removed? Cancer. 2006;107:2368-2374.
Kouba EJ, Lentz A, Wallen EM, Pruthi RS. Clinical use of serum CA-125 levels in patients undergoing radical cystectomy for transitional cell carcinoma of the bladder. Urol Oncol. 2009;27:486-490.
Kulkarni GS, Finelli A, Lockwood G, et al. Effect of healthcare provider characteristics on nodal yield at radical cystectomy. Urology. 2008;72:128-132.
Lee SE, Byun SS, Hong SK, et al. Significance of cancer involvement at the ureteral margin detected on routine frozen section analysis during radical cystectomy. Urol Int. 2006;77(1):13-17.
Leibovici D, Kassouf W, Pisters LL, et al. Organ preservation for muscle-invasive bladder cancer by transurethral resection. Urology. 2007;70(3):473-476.
Leissner J, Ghoneim MA, Abol-Enein H, et al. Extended radical lymphadenectomy in patients with urothelial bladder cancer: results of a prospective multicenter study. J Urol. 2004;171(1):139-144.
Leissner J, Hohenfellner R, Thuroff JW, Wolf HK. Lymphadenectomy in patients with transitional cell carcinoma of the urinary bladder; significance for staging and prognosis. BJU Int. 2000;85(7):817-823.
Lerner SP, Bochner B, Kibel AS. The use and abuse of data: nomograms and talking to patients about clinical medicine. Urol Oncol. 2007;25(4):333-337.
Lerner SP, Colen J, Shen S. Prostatic biology, histologic patterns and clinical consequences of transitional cell carcinoma. Curr Opin Urol. 2008;18:508-512.
Lerner SP, Shen S. Pathologic assessment and clinical significance of prostatic involvement by transitional cell carcinoma and prostate cancer. Urol Oncol. 2008;26(5):481-485.
Lerner SP, Skinner DG, Lieskovsky G, et al. The rationale for en bloc pelvic lymph node dissection for bladder cancer patients with nodal metastases: long-term results. J Urol. 1993;149:758-764. discussion 764–5
Lerner SP, Stein JP, Stadler WM, et al. Variability in surgical quality in a randomized clinical trial of adjuvant chemotherapy following radical cystectomy and pelvic lymphadenectomy. J Urol. 2009;181(4):630. (Abstract 1747)
Loehrer P, Einhorn LH, Elson PJ, et al. A randomized comparison of cisplatin alone or in combination with methotrexate, vinblastine, and doxorubicin in patients with metastatic urothelial carcinoma: a cooperative group study. J Clin Oncol. 1992;10:1066-1073.
Logothetis CJ, Dexeus F, Finn L, et al. A prospective randomized trial comparing CISCA to M-VAC chemotherapy in advanced metastastic urothelial tumors. J Clin Oncol. 1990;8:1050-1055.
Logothetis CJ, Johnson DE, Chong C, et al. Adjuvant cyclophosphamide, doxorubicin, and cisplatin chemotherapy for bladder cancer: an update. J Clin Oncol. 1988;6:1590-1596.
Logothetis CJ, Swanson D, Amato R, et al. Optimal delivery of perioperative chemotherapy: preliminary results of a randomized, prospective, comparative trial of preoperative and postoperative chemotherapy for invasive bladder carcinoma. J Urol. 1996;155(4):1241-1245.
Lonn U, Lonn S, Friberg S, et al. Prognostic value of amplification of c-erb-B2 in bladder carcinoma. Clin Cancer Res. 1995;1:1189-1194.
Lorusso V, Pollera CF, Antimi M, et al. A phase II study of gemcitabine in patients with transitional cell carcinoma of the urinary tract previously treated with platinum. Eur J Cancer. 1998;34(8):1208-1212.
Madersbacher S, Hochreiter W, Burkhard F, et al. Radical cystectomy for bladder cancer today—a homogeneous series without neoadjuvant therapy. J Clin Oncol. 2003;21(4):690-696.
Malmstrom PU, Rintala E, Wahlqvist R, et al. Five year follow-up of a prospective trial of radical cystectomy and neoadjuvant chemotherapy. J Urol. 1996;155:1903-1906.
Manoharan M, Ayyathurai R, Soloway MS. Radical cystectomy for urothelial carcinoma of the bladder: an analysis of perioperative and survival outcome. BJU Int. 2009;104(9):1227-1232.
Margel D, Harel A, Yossepowitch O, Baniel J. A novel algorithm to improve pathologic stage prediction of clinically organ-confined muscle-invasive bladder cancer. Cancer. 2009;115(7):1459-1464.
Margel D, Tal R, Baniel J. Serum tumor markers may predict overall and disease specific survival in patients with clinically organ confined invasive bladder cancer. J Urol. 2007;178:2297-2300. discussion 2300–1
Margel D, Tal R, Neuman A, et al. Prediction of extravesical disease by preoperative serum markers in patients with clinically organ confined invasive bladder cancer. J Urol. 2006;175(4):1253-1257. discussion 1257
Margulis V, Lotan Y, Karakiewicz PI, et al. Multi-institutional validation of the predictive value of Ki-67 labeling index in patients with urinary bladder cancer. J Natl Cancer Inst. 2009;101:114-119.
Marshall VF. The relation of the preoperative estimate to the pathologic demonstration of the extent of vesical neoplasms. J Urol. 1952;68(4):714-723.
Martinez Pineiro JA, Gonzalez Martin M, Arocena F. Neoadjuvant cisplatin chemotherapy before radical cystectomy in invasive transitional cell carcinoma of the bladder: prospective randomized phase III study. J Urol. 1995;153:964-973.
Matsumoto K, Shariat SF, Casella R, et al. Preoperative plasma soluble E-cadherin predicts metastases to lymph nodes and prognosis in patients undergoing radical cystectomy. J Urol. 2003;170(6 Pt. 1):2248-2252.
Mazzucchelli R, Morichetti D, Lopez-Beltran A, et al. Neuroendocrine tumours of the urinary system and male genital organs: clinical significance. BJU Int. 2009;103:1464-1470.
McCaffrey JA, Hilton S, Mazumdar M. Phase II trial of docetaxel in patients with advanced or metastatic transitional cell carcinoma. J Clin Oncol. 1997;15(5):1853-1857.
Millikan R, Dinney C, Swanson D, et al. Integrated therapy for locally advanced bladder cancer: final report of a randomized trial of cystectomy plus adjuvant M-VAC versus cystectomy with both preoperative and postoperative M-VAC. J Clin Oncol. 2001;19:4005-4013.
Mills RD, Turner WH, Fleischmann A, et al. Pelvic lymph node metastases from bladder cancer: outcome in 83 patients after radical cystectomy and pelvic lymphadenectomy. J Urol. 2001;166(1):19-23.
Mitra AP, Pagliarulo V, Yang D, et al. Generation of a concise gene panel for outcome prediction in urinary bladder cancer. J Clin Oncol. 2009;27:3929-3937.
Naoe M, Ogawa Y, Morita J, et al. Detection of circulating urothelial cancer cells in the blood using the CellSearch System. Cancer. 2007;109:1439-1445.
Natale RB, Grossman HB, Blumenstein B, et al. SWOG 8710 (INT-0080): randomized phase III trial of neoadjuvant M-VAC and cystectomy versus cystectomy alone in patients with locally advanced bladder cancer. Proc Am Soc Clin Oncol. 2001;20:2a.
Nezos A, Pissimisis N, Lembessis P, et al. Detection of circulating tumor cells in bladder cancer patients. Cancer Treat Rev. 2009;35:272-279.
Nielsen ME, Shariat SF, Karakiewicz PI, et al. Advanced age is associated with poorer bladder cancer-specific survival in patients treated with radical cystectomy. Eur Urol. 2007;51(3):699-706. discussion 706–8
Nyman DW, Campbell KJ, Hersh E, et al. Phase I and pharmacokinetics trial of ABI-007, a novel nanoparticle formulation of paclitaxel in patients with advanced nonhematologic malignancies. J Clin Oncol. 2005;23(31):7785-7793.
Orsatti M, Curotto A, Canobbio L. Alternating chemo-radiotherapy in bladder cancer: a conservative approach. Int J Radiat Oncol Biol Phys. 1995;33:173-178.
Osawa T, Abe T, Shinohara N, et al. Role of lymph node density in predicting survival of patients with lymph node metastases after radical cystectomy: a multi-institutional study. Int J Urol. 2009;16:274-278. discussion 278
Osman Y, El-Tabey N, Abdel-Latif M, et al. The value of frozen-section analysis of ureteric margins on surgical decision-making in patients undergoing radical cystectomy for bladder cancer. BJU Int. 2007;99:81-84.
Otto T, Börgemann C, Krege S, et al. Adjuvant chemotherapy in locally advanced bladder cancer (PT3/PN1-2,M0)—a phase III study. Eur Urol. 2001;39(Suppl. 5):147.
Pagano F, Bassi P, Ferrante GL, et al. Is stage pT4a (D1) reliable in assessing transitional cell carcinoma involvement of the prostate in patients with a concurrent bladder cancer? A necessary distinction for contiguous or noncontiguous involvement. J Urol. 1996;155(1):244-247.
Paz-Ares L, Tabernero J, Moyano A, et al. Significant activity of the multi-targeted antifolate MTA (LY231514) in advanced transitional cell carcinoma (TCC) of the bladder: results of a phase II trial. Ann Oncol. 1998;9:292.
Petrioli R, Frediani B, Manganelli A, et al. Comparison between a cisplatin-containing regimen and a carboplatin-containing regimen for recurrent or metastatic cancer patients: a randomized phase II study. Cancer. 1996;77:344-351.
Philips G, Sanford B, Halabi S, et al. Phase II study of cisplatin (C), gemcitabine (G) and gefitinib for advanced urothelial carcinoma (UC): analysis of the second cohort of CALGB 90102. Proc Annu Meet Am Soc Clin Oncol. 2006;24:18S.
Poulsen AL, Horn T, Steven K. Radical cystectomy: extending the limits of pelvic lymph node dissection improves survival for patients with bladder cancer confined to the bladder wall. J Urol. 1998;160(6 Pt. 1):2015-2019. discussion 2020
Quek ML, Flanigan RC. The role of lymph node density in bladder cancer prognostication. World J Urol. 2009;27(1):27-32.
Quek ML, Stein JP, Nichols PW, et al. Prognostic significance of lymphovascular invasion of bladder cancer treated with radical cystectomy. J Urol. 2005;174:103-106.
Raj GV, Tal R, Vickers A, et al. Significance of intraoperative ureteral evaluation at radical cystectomy for urothelial cancer. Cancer. 2006;107(9):2167-2172.
Rodel C, Grabenbauer GG, Kuhn R, et al. Combined-modality treatment and selective organ preservation in invasive bladder cancer: long-term results. J Clin Oncol. 2002;20:3061-3071.
Sakamoto N, Tsuneyoshi M, Naito S, Kumazawa J. An adequate sampling of the prostate to identify prostatic involvement by urothelial carcinoma in bladder cancer patients. J Urol. 1993;149:318-321.
Sanchez-Ortiz RF, Huang WC, Mick R, et al. An interval longer than 12 weeks between the diagnosis of muscle invasion and cystectomy is associated with worse outcome in bladder carcinoma. J Urol. 2003;169(1):110-115.
Sanderson KM, Cai J, Miranda G, Skinner DG, Stein JP. Upper tract urothelial recurrence following radical cystectomy for transitional cell carcinoma of the bladder: an analysis of 1,069 patients with 10-year followup. J Urol. 2007;177:2088-2094.
Sanderson KM, Stein JP, Skinner DG. The evolving role of pelvic lymphadenectomy in the treatment of bladder cancer. Urol Oncol. 2004;22:205-211. discussion 212–13
Saxman SB, Propert KJ, Einhorn LH, et al. Long-term follow-up of a phase III intergroup study of cisplatin alone or in combination with methotrexate, vinblastine, and doxorubicin in patients with metastatic urothelial carcinoma: a cooperative group study. J Clin Oncol. 1997;15(7):2564-2569.
Schiffer E, Vlahou A, Petrolekas A, et al. Prediction of muscle-invasive bladder cancer using urinary proteomics. Clin Cancer Res. 2009;15:4935-4943.
Schoenberg MP, Carter HB, Epstein JI. Ureteral frozen section analysis during cystectomy: a reassessment. J Urol. 1996;155:1218-1220.
Shabsigh A, Korets R, Vora KC, et al. Defining early morbidity of radical cystectomy for patients with bladder cancer using a standardized reporting methodology. Eur Urol. 2009;55(1):164-176.
Shariat SF, Karakiewicz PI, Ashfaq R, et al. Multiple biomarkers improve prediction of bladder cancer recurrence and mortality in patients undergoing cystectomy. Cancer. 2008;112(2):315-325.
Shariat SF, Karakiewicz PI, Palapattu GS, et al. Nomograms provide improved accuracy for predicting survival after radical cystectomy. Clin Cancer Res. 2006;12(22):6663-6676.
Shariat SF, Karakiewicz PI, Palapattu GS, et al. Outcomes of radical cystectomy for transitional cell carcinoma of the bladder: a contemporary series from the Bladder Cancer Research Consortium. J Urol. 2006;176:2414-2422. discussion 2422
Shariat SF, Kim J, Nguyen C, et al. Correlation of preoperative levels of IGF-I and IGFBP-3 with pathologic parameters and clinical outcome in patients with bladder cancer. Urology. 2003;61:359-364.
Shariat SF, Kim JH, Andrews B, et al. Preoperative plasma levels of transforming growth factor beta(1) strongly predict clinical outcome in patients with bladder carcinoma. Cancer. 2001;92(12):2985-2992.
Shariat SF, Monoski MA, Andrews B, et al. Association of plasma urokinase-type plasminogen activator and its receptor with clinical outcome in patients undergoing radical cystectomy for transitional cell carcinoma of the bladder. Urology. 2003;61:1053-1058.
Shariat SF, Palapattu GS, Karakiewicz PI, et al. Concomitant carcinoma in situ is a feature of aggressive disease in patients with organ-confined TCC at radical cystectomy. Eur Urol. 2007;51:152-160.
Shariat SF, Tokunaga H, Zhou J, et al. p53, p21, pRB, and p16 expression predict clinical outcome in cystectomy with bladder cancer. J Clin Oncol. 2004;22(6):1014-1024.
Sherif A, Rintala E, Mestad O, et al. Neoadjuvant cisplatin-methotrexate chemotherapy of invasive bladder cancer—Nordic cystectomy trial 2. Scand J Urol Nephrol. 2002;36(6):419-425.
Shipley WU, Winter KA, Kaufman DS, et al. Phase III trial of neoadjuvant chemotherapy in patients with invasive bladder cancer treated with selective bladder preservation by combined radiation therapy and chemotherapy: initial results of Radiation Therapy Oncology Group 89-03. J Clin Oncol. 1998;16(11):3576-3583.
Shipley WU, Zietman AL, Kaufman DS, et al. Selective bladder preservation by combined modality treatment of invasive bladder cancer: 10-year outcome. Int J Radiat Oncol Biol Phys. 2001;51(3 Suppl. 1):13-14.
Siefker-Radtke AO. Urachal carcinoma: surgical and chemotherapeutic options. Expert Rev Anticancer Ther. 2006;6:1715-1721.
Siefker-Radtke AO, Dinney CP, Abrahams NA, et al. Evidence supporting preoperative chemotherapy for small cell carcinoma of the bladder: a retrospective review of the M. D. Anderson cancer experience. J Urol. 2004;172(2):481-484.
Silver DA, Stroumbakis N, Russo P, et al. Ureteral carcinoma in situ at radical cystectomy: does the margin matter? J Urol. 1997;158(3 Pt. 1):768-771.
Skinner DG. Management of invasive bladder cancer: a meticulous pelvic node dissection can make a difference. J Urol. 1982;128(1):34-36.
Skinner DG, Daniels JR, Russell CA, et al. The role of adjuvant chemotherapy following cystectomy for invasive bladder cancer: a prospective comparative trial. J Urol. 1991;145:459-467.
Slaton JW, Millikan R, Inoue K, et al. Correlation of metastasis related gene expression and relapse-free survival in patients with locally advanced bladder cancer treated with cystectomy and chemotherapy. J Urol. 2004;171(2 Pt. 1):570-574.
Smaldone MC, Jacobs BL, Smaldone AM, Hrebinko RLJr. Long-term results of selective partial cystectomy for invasive urothelial bladder carcinoma. Urology. 2008;72(3):613-616.
Solsona E, Climent MA, Iborra I, et al. Bladder preservation in selected patients with muscle-invasive bladder cancer by complete transurethral resection of the bladder plus systemic chemotherapy: long-term follow-up of a phase 2 nonrandomized comparative trial with radical cystectomy. Eur Urol. 2009;55(4):911-919.
Solsona E, Iborra I, Ricos JV, et al. Feasibility of transurethral resection for muscle infiltrating carcinoma of the bladder: long-term followup of a prospective study. J Urol. 1998;159(1):95-98. discussion 98–9
Sonpavde G, Goldman BH, Speights VO, et al. Quality of pathologic response and surgery correlate with survival for patients with completely resected bladder cancer after neoadjuvant chemotherapy. Cancer. 2009;115(18):4104-4109.
Sonpavde G, Jian W, Lerner SP. Sunitinib malate is active and synergistic with cisplatin against human urothelial carcinoma in a preclinical model. Proc Annu Meet Am Soc Clin Oncol. 25(18S), 2007.
Sonpavde G, Ross R, Powles T, et al. Novel agents for muscle-invasive and advanced urothelial cancer. BJU Int. 2008;101(8):937-943.
Sonpavde G, Sternberg CN. Treatment of metastatic urothelial cancer: opportunities for drug discovery and development. BJU Int. 2008;102(9 Pt. B):1354-1360.
Sriplakich S, Jahnson S, Karlsson MG. Epidermal growth factor receptor expression: predictive value for the outcome after cystectomy for bladder cancer? BJU Int. 1999;83:498-503.
Stadler WM, Lerner SP, Groshen S, et al. Randomized trial of p53 targeted adjuvant therapy for patients (pts) with organ-confined node-negative urothelial bladder cancer (UBC). Proc Annu Meet Am Soc Clin Oncol. 2009;27(15S):239s.
Stein JP. Indications for early cystectomy. Sem Urol Oncol. 2000;18:289-295.
Stein JP. Contemporary concepts of radical cystectomy and the treatment of bladder cancer. J Urol. 2003;169(1):116-117.
Stein JP, Cai J, Groshen S, Skinner DG. Risk factors for patients with pelvic lymph node metastases following radical cystectomy with en bloc pelvic lymphadenectomy: concept of lymph node density. J Urol. 2003;170(1):35-41.
Stein JP, Clark P, Miranda G, et al. Urethral tumor recurrence following cystectomy and urinary diversion: clinical and pathological characteristics in 768 male patients. J Urol. 2005;173(4):1163-1168.
Stein JP, Esrig D, Freeman JA, et al. Prospective pathologic analysis of female cystectomy specimens: risk factors for orthotopic diversion in women. Urology. 1998;51(6):951-955.
Stein JP, Lieskovsky G, Cote R, et al. Radical cystectomy in the treatment of invasive bladder cancer: long-term results in 1,054 patients. J Clin Oncol. 2001;19(3):666-675.
Stein JP, Penson DF, Cai J, et al. Radical cystectomy with extended lymphadenectomy: evaluating separate package versus en bloc submission for node positive bladder cancer. J Urol. 2007;177(3):876-881. discussion 881–2
Stein JP, Skinner DG. Radical cystectomy—technique and outcome. In: Lerner SP, Schoenberg MP, Sternberg CN, editors. Textbook of bladder cancer. London: Abingdon, Taylor & Francis; 2006:445-469.
Stenzl A, Draxl H, Posch B, et al. The risk of urethral tumors in female bladder cancer: can the urethra be used for orthotopic reconstruction of the lower urinary tract? J Urol. 1995;153(3 Pt. 2):950-955.
Sternberg CN. Chemotherapy for local treatment of bladder cancer. Semin Radiat Oncol. 2005;15(1):60-65.
Sternberg CN, Calabrò F, Pizzocaro G, et al. Chemotherapy with an every-2-week regimen of gemcitabine and paclitaxel in patients with transitional cell carcinoma who have received prior cisplatin-based therapy. Cancer. 2001;92(12):2993-2998.
Sternberg CN, Collette L. What has been learned from meta-analyses of neoadjuvant and adjuvant chemotherapy in bladder cancer? BJU Int. 2006;98(3):487-489.
Sternberg CN, de Mulder P, Schornagel JH, et al. Seven year update of an EORTC phase III trial of high dose intensity M-VAC chemotherapy and G-CSF versus classic M-VAC in advanced urothelial tract tumors (EORTC protocol 30924). Eur J Cancer. 2006;42(1):50-54.
Sternberg CN, Donat SM, Bellmunt J, et al. Chemotherapy for bladder cancer: treatment guidelines for neoadjuvant chemotherapy, bladder preservation, adjuvant chemotherapy, and metastatic cancer. Urology. 2007;69(1 Suppl.):62-79.
Sternberg CN, Pansadoro V, Calabrò F, et al. Can patient selection for bladder preservation be based on response to chemotherapy? Cancer. 2003;97(7):1644-1652.
Sternberg CN, Yagoda A, Scher HI, et al. M-VAC for advanced transitional cell carcinoma of the urothelium: efficacy, and patterns of response and relapse. Cancer. 1989;64:2448-2458.
Steven K, Poulsen AL. Radical cystectomy and extended pelvic lymphadenectomy: survival of patients with lymph node metastasis above the bifurcation of the common iliac vessels treated with surgery only. J Urol. 2007;178(4 Pt. 1):1218-1223. discussion 1223–4
Stockle M, Meyenburg W, Wellek S, et al. Advanced bladder cancer (stages pT3b, pT4a, pN1 nad pN2): improved survival after radical cystectomy and 3 adjuvant cycles of chemotherapy results of a controlled prospective study. J Urol. 1992;148:302-307.
Stockle M, Meyenburg W, Wellek S. Adjuvant polychemotherapy of nonorgan-confined bladder cancer after radical cystectomy revisited: long term results of a controlled prospective study and further clinical experience. J Urol. 1995;153:47-52.
Studer UE, Bacchi M, Biedermann C. Adjuvant cisplatin chemotherapy following cystectomy for bladder cancer: results of a prospective randomized trial. J Urol. 1994;152:81-84.
Svatek R, Shariat SF. Re: lymph node density is superior to TNM nodal status in predicting disease-specific survival after radical cystectomy for bladder cancer: analysis of pooled data from MDACC and MSKCC. Eur Urol. 2008;54(3):690-691.
Sved PD, Gomez P, Nieder AM, et al. Upper tract tumour after radical cystectomy for transitional cell carcinoma of the bladder: incidence and risk factors. BJU Int. 2004;94(5):785-789.
Sweeney CJ, Roth BJ, Kabbinavar FF, et al. Phase II study of pemetrexed for second-line treatment of transitional cell cancer of the urothelium. J Clin Oncol. 2006;24(21):3451-3457.
Sylvester R, Sternberg C. The role of adjuvant combination chemotherapy after cystectomy in locally advanced bladder cancer: what we do not know and why. Ann Oncol. 2000;11(7):851-856.
van der Meijden A, Oosterlinck W, Brausi M, et al. Significance of bladder biopsies in Ta,T1 bladder tumors: a report from the EORTC Genito-Urinary Tract Cancer Cooperative Group. EORTC-GU Group Superficial Bladder Committee. Eur Urol. 1999;35(4):267-271.
Vaughn DJ, Broome CM, Hussain M, et al. Phase II trial of weekly paclitaxel in patients with previously treated advanced urothelial cancer. J Clin Oncol. 2002;20(4):937-940.
Vazina A, Dugi D, Shariat SF, et al. Stage specific lymph node metastasis mapping in radical cystectomy specimens. J Urol. 2004;171(5):1830-1834.
von der Maase H, Lehmann J, Gravis G, et al. A phase II trial of pemetrexed plus gemcitabine in locally advanced and/or metastatic transitional cell carcinoma of the urothelium. Ann Oncol. 2006;17(10):1533-1538.
von der Maase H, Sengelov L, Roberts JT, et al. Long-term survival results of a randomized trial comparing gemcitabine plus cisplatin, with methotrexate, vinblastine, doxorubicin, plus cisplatin in patients with bladder cancer. J Clin Oncol. 2005;23(21):4602-4608.
Wagner KR, Schoenberg MP, Bianco FJJr, Jarrett TW. Prospective intermediate follow-up of carcinoma in situ involving the distal ureter at cystectomy: is there a role for ureteroscopy? J Endourol. 2008;22(6):1241-1246.
Wallace DM, Raghavan D, Kelly KA, et al. Neo-adjuvant (pre-emptive) cisplatin therapy in invasive transitional cell carcinoma of the bladder. Br J Urol. 1991;67:608-615.
Wang R, Morris DS, Tomlins SA, et al. Development of a multiplex quantitative PCR signature to predict progression in non-muscle-invasive bladder cancer. Cancer Res. 2009;69(9):3810-3818.
Winquist E, Kirchner TS, Segal R, et al. Neoadjuvant chemotherapy for transitional cell carcinoma of the bladder: a systematic review and meta-analysis. Genitourinary Cancer Disease Site Group, Cancer Care Ontario Program in Evidence-based Care Practice Guidelines Initiative. J Urol. 2004;171(2 Pt. 1):561-569.
Winquist E, Vokes E, Moore MJ, et al. A phase II study of oxaliplatin in urothelial cancer. Urol Oncol. 2005;23:150-154.
Wood DPJr, Montie JE, Pontes JE, Levin HS. Identification of transitional cell carcinoma of the prostate in bladder cancer patients: a prospective study. J Urol. 1989;142(1):83-85.
Wuelfing C, Machiels J, Richel D, et al. A single arm, multicenter, open label, phase II study of lapatinib as 2L treatment of pts with locally advanced/metastatic transitional cell carcinoma (TCC) of the urothelial tract. Proc Annu Meet Am Soc Clin Oncol. 23(16S), 2005.
Zhu K, Pino MS, Siefker-Radtke AO, et al. AG-013736, a novel VEGF receptor and PDGF receptor inhibitor with potent activity against human bladder carcinoma in vitro and in vivo. Proc Annu Meet Am Soc Clin Oncol. 24(16S), 2006.
Zieger K, Wiuf C, Jensen KM, et al. Chromosomal imbalance in the progression of high-risk non-muscle invasive bladder cancer. BMC Cancer. 2009;9:149.
Zietman AL, Prince EA, Nakfoor BM, Shipley WU. Neoadjuvant androgen suppression with radiation in the management of locally advanced adenocarcinoma of the prostate: experimental and clinical results. Urol. 1997;49(3A Suppl.):74-83.