chapter 111 Normal Development of the Genitourinary Tract
The study of embryology provides a useful foundation for the understanding of definitive human anatomy and various congenital disease processes. During the past few decades, a torrent of molecular information and novel experimental techniques has revolutionized the field of embryology, and the knowledge base continues to expand at an exponential rate. From the urologic surgeon’s perspective, however, the classic, descriptive aspects of anatomic embryology continue to serve as an important reference point from which various congenital problems are solved clinically. The aim of this chapter is to provide a concise presentation of the essential facts of normal genitourinary system development, clarifying the important anatomic features and supplementing them with updated molecular information. Deliberate efforts have been made to separate the ever-expanding molecular information from that of the descriptive, anatomic embryology to keep the main “story” of genitourinary system development clear and understandable from a surgical point of view (Fig. 111–1). To help with visualization of the key events, various schematic drawings are provided. The goal of this chapter is not to provide potential explanations for every congenital defect that might occur in the genitourinary system but to select pertinent examples highlighting the fundamental concepts and principles.
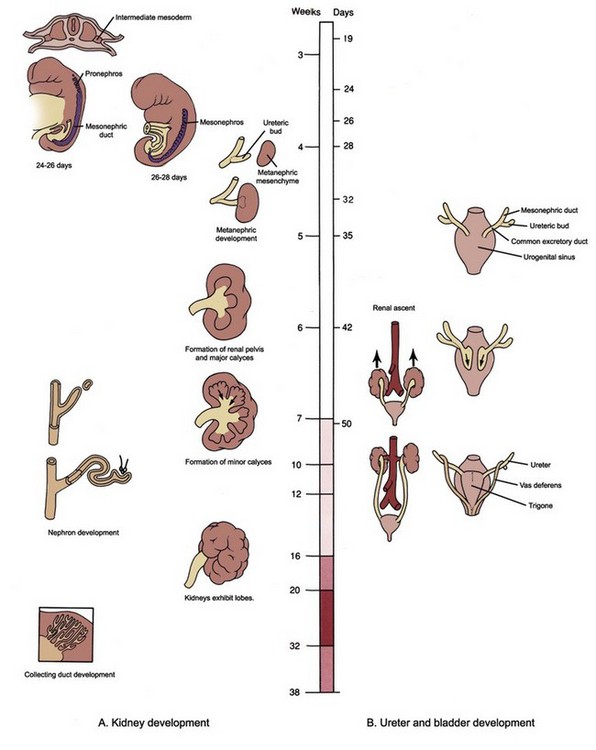
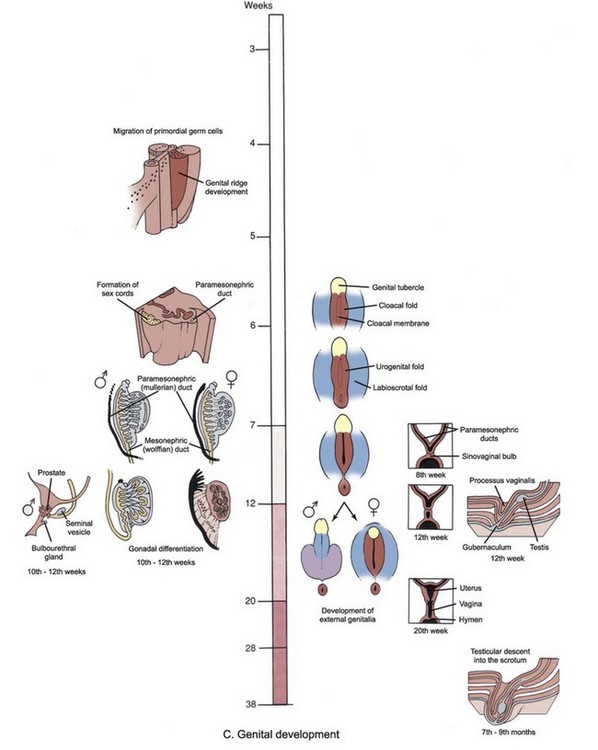
Figure 111–1 A to C, Time line and overview of genitourinary system development.
(Modified from Larsen WJ. Human embryology. New York: Churchill Livingstone; 1997.)
Kidney Development
Early Events
Mammals develop three kidneys in the course of intrauterine life. The embryonic kidneys are, in order of their appearance, the pronephros, the mesonephros, and the metanephros. The first two kidneys regress in utero, and the third becomes the permanent kidney. Embryologically, all three kidneys develop from the intermediate mesoderm. As the notochord and neural tube develop, the mesoderm located on either side of the midline differentiates into three subdivisions: paraxial (somite), intermediate, and lateral mesoderm (Fig. 111–2). As the embryo undergoes transverse folding, the intermediate mesoderm separates away from the paraxial mesoderm and migrates toward the intraembryonic coelom (the future peritoneum). At this time there is a progressive craniocaudal development of the bilateral longitudinal mesodermal masses, called nephrogenic cords. Each cord is seen bulging from the posterior wall of the coelomic cavity, producing the urogenital ridge.
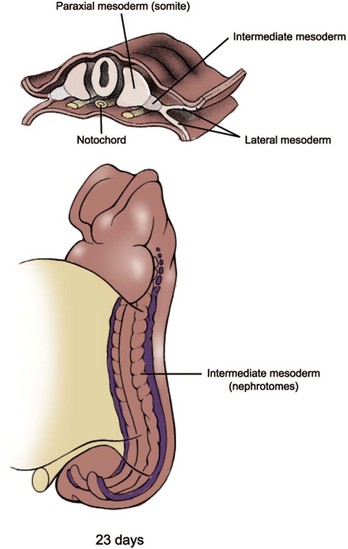
Figure 111–2 The intermediate mesoderm gives rise to paired, segmentally organized nephrotomes from cervical to sacral region. Cervical nephrotomes are formed early during the fourth week and are collectively referred to as the pronephros.
(Modified from Larsen WJ. Human embryology. New York: Churchill Livingstone; 1997.)
Pronephros and Mesonephros
The mammalian pronephros is a transitory, nonfunctional kidney, analogous to that of primitive fish. In humans, the first evidence of pronephros is seen late in the third week, and it completely degenerates by the start of the fifth week. The pronephros develops as five to seven paired segments in the region of the future neck and thorax (Fig. 111–3A). Development of the pronephric tubules starts at the cranial end of the nephrogenic cord and progresses caudally. As each tubule matures it immediately begins to degenerate along with the segment of the nephric duct to which the tubules are attached.
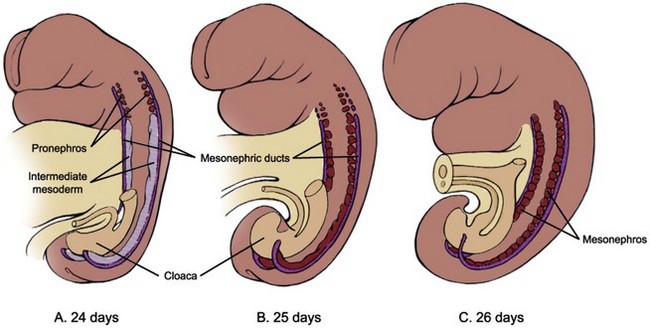
Figure 111–3 Development of pronephros and mesonephros. A, Pronephros develops in each of five to seven cervical segments, but this primitive renal structure degenerates quickly during the fourth week. The (meso)nephric ducts first appear on day 24. B and C, Mesonephric vesicles and tubules form in a craniocaudal direction throughout the thoracic and lumbar regions. The cranial pairs degenerate as caudal pairs develop, and the definitive mesonephros contains about 20 pairs confined to the first three lumbar segments.
(Modified from Larsen WJ. Human embryology. New York: Churchill Livingstone; 1997.)
The second kidney, the mesonephros, is also transient, but in mammals it serves as an excretory organ for the embryo while the definitive kidney, the metanephros, begins its development (see Fig. 111–3B and C). There is a gradual transition from the pronephros to the mesonephros at about the 9th and 10th somite levels. Development of the nephric ducts (also called the wolffian ducts) precedes the development of the mesonephric tubules. The nephric ducts can be seen as a pair of solid longitudinal tissue condensations at about the 24th day, developing parallel to the nephrogenic cords in the dorsolateral aspect of the embryo. Its blind distal ends grow toward the primitive cloaca and soon fuse with it at about the 28th day. As the ducts fuse with the cloaca they begin to form a lumen at the caudal end. This process of canalization then progresses cranially in a reverse direction, transforming the solid tissue condensations into the definitive nephric ducts with excretory capability. Soon after the appearance of the nephric ducts during the 4th week, mesonephric vesicles begin to form. Initially, several spherical masses of cells are found along the medial side of the nephrogenic cords at the cranial end. This differentiation progresses caudally and results in the formation of 40 to 42 pairs of mesonephric tubules, but only about 30 pairs are seen at any one time because the cranially located tubules start to degenerate starting at about the 5th week. By the 4th month, the human mesonephros has almost completely disappeared, except for a few elements that persist into maturity as part of the reproductive tract. In males, some of the cranially located mesonephric tubules become the efferent ductules of the testes. The epididymis and vas deferens are also formed from the nephric (wolffian) ducts. In females, remnants of cranial and caudal mesonephric tubules form small, nonfunctional mesosalpingeal structures termed the epoöphoron and paroöphoron.
The mesonephric tubules differentiate into excretory units that resemble an abbreviated version of an adult nephron. Shortly after the cell clusters are formed they develop lumens and take the shape of vesicles. As the vesicle elongates, each end curves in an opposite direction to form an S-shaped tubule. The lateral end forms a bud that connects with the nephric duct. The medial end lengthens and enlarges to form a cup-shaped sac, which eventually wraps around a knot of glomerular capillaries to form a renal corpuscle. The tuft of glomerular capillaries originating from a branch of the dorsal aorta invades the developing glomerulus while an efferent arteriole empties into a subcardinal sinus.
Metanephros
The definitive kidney, or the metanephros, forms in the sacral region as a pair of new structures, called the ureteric buds, sprout from the distal portion of the nephric duct and come in contact with the condensing blastema of metanephric mesenchyme at about the 28th day (Fig. 111–4). The ureteric bud penetrates the metanephric mesenchyme and begins to divide dichotomously. The tip of the dividing ureteric bud, called the ampulla, interacts with the metanephric mesenchyme to induce formation of future nephrons via mesenchymal-epithelial interaction. As the ureteric bud divides and branches, each new ampulla acquires a caplike condensation of metanephric mesenchyme, thereby giving the metanephros a lobulated appearance (Fig. 111–5).
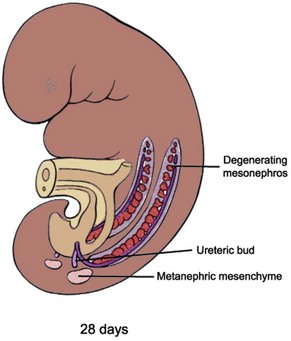
Figure 111–4 Metanephric mesenchyme condenses from the intermediate mesoderm during the early part of the fifth week and comes into contact with the ureteric bud, an outgrowth of the nephric duct, while the cranial mesonephros continues to degenerate.
(Modified from Larsen WJ. Human embryology. New York: Churchill Livingstone; 1997.)
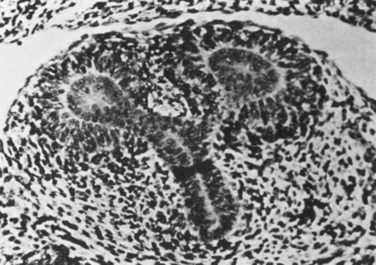
Figure 111–5 The ureteric bud divides to form enlarged tips, called ampullae, around which the metanephric mesenchyme condenses and begins nephron differentiation. The remaining mesenchymal cells remain stromal and continue to interact with tubular mesenchymal cells and dividing ureteric bud epithelial cells.
(From Potter EL. Normal and abnormal development of the kidney. Chicago: Year Book Medical Publishers; 1972.)
The ureteric bud and metanephric mesenchyme exert reciprocal inductive effects toward each other, and the proper differentiation of these primordial structures depends on these inductive signals (see Molecular Mechanism of Kidney Development, later). The metanephric mesenchyme induces the ureteric bud to branch and divide, and, in turn, the ureteric bud induces the metanephric mesenchyme to condense and undergo mesenchymal-epithelial conversion. The nephron, which consists of the glomerulus, proximal tubule, loop of Henle, and distal tubule, is thought to derive from the metanephric mesenchyme, while the collecting system, consisting of collecting ducts, calyces, pelvis and ureter, is formed from the ureteric bud (Fig. 111–6).
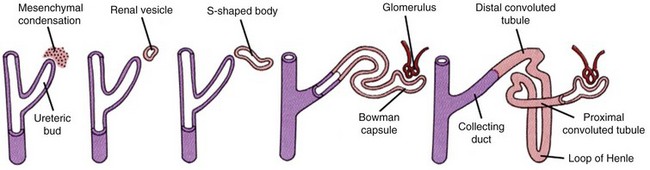
Figure 111–6 Development of the renal collecting ducts and nephrons. The tip of the dividing ureteric bud induces the metanephric mesenchyme (in pink) to condense, which then differentiates into a renal vesicle. This vesicle coils into an S-shaped tubule and ultimately forms a Bowman capsule as well as the proximal convoluted tubules, distal convoluted tubules, and loops of Henle. The ureteric bud (in purple) contributes to the formation of collecting ducts.
(Modified from Larsen WJ. Human embryology. New York: Churchill Livingstone; 1997.)
In principle, all nephrons are formed in the same way and can be classified into fairly well-defined developmental stages (Larsson et al, 1983) (Fig. 111–7). The metanephric mesenchyme first condenses to form a four- to five-cell layer condensate around the ampulla of the advancing ureteric bud. Near the interface of the ampulla and its adjacent ureteric branch, a cluster of cells separates from the condensate and forms an oval mass, called a pretubular aggregate. An internal cavity forms within the pretubular aggregate, at which point the structure is called a renal vesicle (stage I). Cells of the stage I renal vesicle are tall and columnar and are stabilized by their attachments to the newly formed basement membrane. It has not yet established a contact with the ampulla of the ureteric bud, but it subsequently forms a luminal connection. Multipotential precursors residing in renal vesicles ultimately give rise to all the epithelial cell types of the nephron (Herzlinger et al, 1992). Nephron segmentation into glomerular and tubular domains is initiated by the sequential formation of two clefts in the renal vesicle (stage II). Creation of a lower cleft, termed the vascular cleft, precedes formation of a comma-shaped body. Generation of an upper cleft in the comma-shaped body precedes formation of an S-shaped body. At this stage, the cup-shaped glomerular capsule is recognized in the lowest limb of the S-shaped tubule. Epithelial cells lining the inner wall of this cup will comprise the visceral glomerular epithelium, or podocyte layer. Cells lining the outer wall of the cup will form parietal glomerular epithelium, which lines the Bowman capsule. The glomerular capillary tuft is formed via recruitment and proliferation of endothelial and mesangial cell precursors. The rest of the S-shaped tubule develops into the proximal tubule, the loop of Henle, and the distal tubule. When the cup-shaped glomerular capsule matures into an oval structure the nephron has now passed into stage III of development. Now the nephron can be divided into identifiable proximal and distal tubules. The stage IV nephron is characterized by a round glomerulus that closely resembles the mature renal corpuscle. The morphology of the proximal tubule resembles that of a mature nephron, whereas the distal segments are still primitive. In some species, such as rodents, all stages of nephron development are present at birth, whereas in others, such as humans, all nephrons at birth are in varying steps of stage IV. Mesenchymal cells that do not become tubular epithelium give rise to interstitial stromal cells, which differentiate into a diverse population including fibroblasts, lymphocyte-like cells, and pericytes. Overall, these events are reiterated throughout the growing kidney so that older, more differentiated nephrons are located in the inner part of the kidney near the juxtamedullary region and newer, less differentiated nephrons are found at the periphery (Fig. 111–8). In humans, although renal maturation continues to take place postnatally, nephrogenesis is completed before birth at around 32 to 34 weeks of gestation.
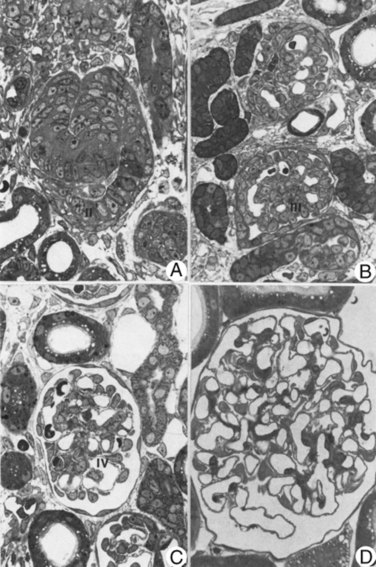
Figure 111–7 Nephron development as seen from a renal cortex of a 3-day-old rat. A, A developing nephron with S-shaped body (II). B, An oval glomeruli (III). C, Nephron now resembles that of mature tubules and glomeruli (IV). D, Mature superficial glomerulus from adult rat kidney.
(Reproduced from Larsson L, Maunsbach AB. The ultrastructural development of the glomerular filtration barrier in the rat kidney: a morphometric analysis. J Ultrastruct Res 1980;72:392.)
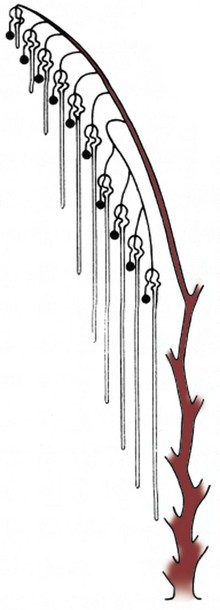
Figure 111–8 Schematic representation of progressive nephron differentiation. Older, more differentiated nephrons are located in the inner part of the kidney near the juxtamedullary region and newer, less differentiated nephrons are found at the periphery.
(Reproduced from Potter EL. Normal and abnormal development of the kidney. Chicago: Year Book Medical Publishers; 1972.)
Collecting System
The dichotomous branching of the ureteric bud determines the eventual pelvicalyceal patterns and their corresponding renal lobules (Cebrian et al, 2004) (Fig. 111–9). In humans, the first nine branch generations are formed by approximately 15 weeks’ gestation. By 20 to 22 weeks, ureteric bud branching is completed. Thereafter, collecting duct development occurs by extension of peripheral branch segments. Between 22 and 24 weeks of human fetal gestation the peripheral (cortical) and central (medullary) domains of the developing kidney are established. The renal cortex, which represents 70% of total kidney volume at birth, becomes organized as a relatively compact, circumferential rim of tissue surrounding the periphery of the kidney. The renal medulla, which represents 30% of total kidney volume at birth, has a modified cone shape with a broad base contiguous with cortical tissue. The apex of the cone is formed by convergence of collecting ducts in the inner medulla and is termed the papilla. Distinct morphologic differences emerge between collecting ducts located in the medulla compared with those located in the renal cortex. Medullary collecting ducts are organized into elongated linear arrays that converge centrally in a region devoid of glomeruli. In contrast, collecting ducts located in the renal cortex continue to induce metanephric mesenchyme. The most central segments of the collecting system, formed from the first five generations of ureteric bud branching, undergo remodeling by increased growth and dilatation of these tubules to form the pelvis and calyces.
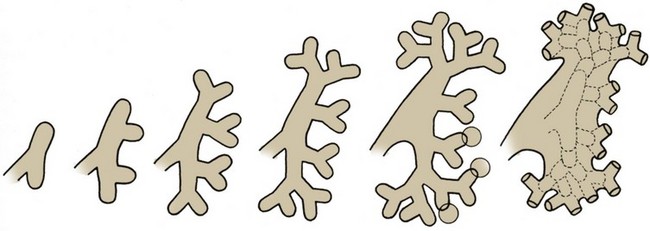
Figure 111–9 Dichotomous branching of the ureteric bud and subsequent fusion of the ampullae to form the renal pelvis and calyces. Circles indicate possible sites of infundibular development between the third, fourth, or fifth generations of branches and their subsequent expansions to give rise to the calyces.
(Reproduced from Potter EL. Normal and abnormal development of the kidney. Chicago: Year Book Medical Publishers; 1972.)
Renal Ascent
Between the sixth and ninth weeks the kidneys ascend to a lumbar site just below the adrenal glands (Fig. 111–10). The precise mechanism responsible for renal ascent is not known, but it is speculated that the differential growth of the lumbar and sacral regions of the embryo plays a major role. As the kidneys migrate they are vascularized by a succession of transient aortic sprouts that arise at progressively higher levels. These arteries do not elongate to follow the ascending kidneys but instead degenerate and are replaced by successive new arteries. The final pair of arteries forms in the upper lumbar region and becomes the definitive renal arteries. Occasionally, a more inferior pair of arteries persists as accessory lower pole arteries. When the kidney fails to ascend properly its location becomes ectopic. If its ascent fails completely it remains as a pelvic kidney. The inferior poles of the kidneys may also fuse, forming a horseshoe kidney that crosses over the ventral side of the aorta. During ascent the fused lower pole becomes trapped under the inferior mesenteric artery and thus does not reach its normal site. Rarely, the kidney fuses to the contralateral one and ascends to the opposite side, resulting in a cross-fused ectopy.
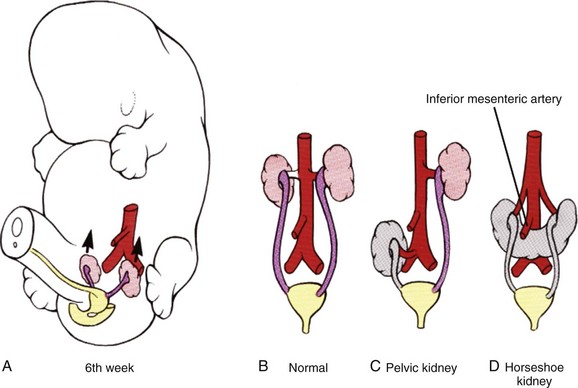
Figure 111–10 Normal and abnormal ascent of the kidneys. A and B, The metanephros normally ascends from the sacral region to its definitive lumbar location between the sixth and ninth weeks. C, Rarely, a kidney may fail to ascend, resulting in a pelvic kidney. D, If the inferior poles of the kidneys fuse before ascent, the resulting horseshoe kidney does not ascend to a normal position owing to entrapment by the inferior mesenteric artery.
(Modified from Larsen WJ. Human embryology. New York: Churchill Livingstone; 1997.)
Molecular Mechanism of Kidney Development
The details of inductive interactions among metanephric mesenchyme, ureteric bud epithelia, and, more recently, the stroma, are becoming clearer and provide insights into the complex regulatory mechanisms underlying renal development. Formation of renal tubules and collecting system occurs sequentially and requires dynamic interactions among epithelial, mesenchymal, and stromal cells. Many of the early events in embryonic kidney development were first elucidated by manipulating lower vertebrate embryos and by utilizing a mammalian in-vitro organ culture system. Clifford Grobstein’s pioneering work in the 1950s led to an organ culture technique (Grobstein, 1956) whereby the metanephric mesenchyme is separated from the ureteric bud during the early part of kidney development and grown in vitro on a filter. An inducer tissue, such as ureter or spinal cord, cultured on the opposite side of the filter then provides the inductive signal (Fig. 111–11). This ingenious experimental approach has established the kidney as a model system for studying the role of epithelial-mesenchymal interaction in organ development. The development of many other organs, including lung, salivary glands, gonads, prostate, and bladder, also require epithelial-mesenchymal interaction for the controlled differentiation and proliferation of tissues.
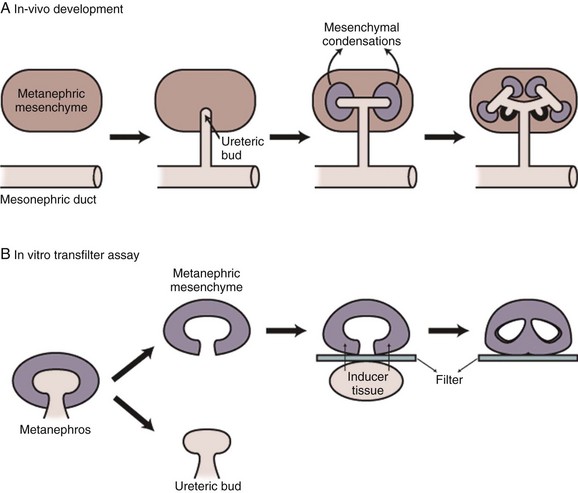
Figure 111–11 Schematic representation of in-vivo kidney development (A) and an in-vitro transfilter organ culture system of Grobstein (B). At an early stage of renal development, the metanephric mesenchyme is separated from the ureteric bud and cultured on a filter. If there is an inducer tissue grown on the opposite side of the filter, such as ureter and spinal cord, the metanephric mesenchyme will continue to differentiate into nephron structures. In the absence of inducer tissue, the metanephric mesenchyme will degenerate via apoptosis.
(Modified from Vainio S, Muller U. Inductive tissue interactions, cell signaling, and the control of kidney organogenesis. Cell 1997;90[6]:975.)
Formation of Nephric Ducts
The first recognizable event in renal development may be the formation of nephric ducts within the region of the intermediate mesoderm. The molecular signals responsible for this early event, in which seemingly unorganized mesenchymal cells aggregate to become an epithelial duct, remain essentially unknown, but details are beginning to emerge. The early intermediate mesoderm destined to become nephric ducts is distinguished by expression of the transcription factors LIM1, PAX2, and SIM1, but only LIM1 appears to be absolutely essential for nephric duct formation (Shawlot and Behringer, 1995). PAX2 may be important for maintaining other marker gene expression in the nephric ducts (Torres et al, 1995). Available data suggest a model in which few opposing secreted factors from the surrounding tissues cumulatively restrict LIM1 expression to the intermediate mesoderm. LIM1 then activates PAX2 expression to further orchestrate the formation of nephric ducts.
Ureteric Bud Outgrowth toward Metanephric Mesenchyme
The outgrowth of the ureteric bud from the nephric duct and its invasion into the condensing blastema of metanephric mesenchyme is a crucial initiating event in the development of the adult kidney (metanephros). Many candidate genes have been identified to play a critical role in this process (see http://golgi.ana.ed.ac.uk/kidhome.html). In particular, several lines of experimental evidence have revealed a crucial role of the RET-GDNF-GFRα1 pathway in the ureteric bud outgrowth (Fig. 111–12). Glial cell line–derived neurotrophic factor (GDNF) is a secreted peptide expressed in the metanephric mesenchyme that activates the RET receptor, which is expressed along the nephric duct. GDNF activation of RET requires the glycosylphosphatidylinositol (GPI)-linked protein GFRα1, which is expressed in both metanephric mesenchyme and nephric duct. Gene knockout mutations in Ret, GDNF (Moore et al, 1996; Pichel et al, 1996; Sanchez et al, 1996), and GFRα1 (Cacalano et al, 1998) inhibit ureteric bud outgrowth. In organ culture systems, recombinant GDNF is sufficient to induce ectopic ureteric bud outgrowth (Sainio et al, 1997) (Fig. 111–13). However, the competence of the nephric duct to respond to GDNF is restricted along the anteroposterior axis. This anteroposterior restriction might be mediated by suppressors of RET signaling within the surrounding tissue, such as bone morphogenetic protein-4 (BMP4). Mice that are deficient for BMP4 show more broadened ureteric buds and/or secondary anterior buds, suggesting that full BMP4 activity is required to limit RET signaling to the caudal aspect adjacent to the developing metanephric mesenchyme (Miyazaki et al, 2000). Similarly, in organ culture, BMP4 can suppress the activity of GDNF to induce ectopic ureteric bud formation (Brophy et al, 2001). Proper positioning of the ureteric bud is also controlled by the localized expression of GDNF within the metanephric mesenchyme. Both positive and negative regulators have been described for GDNF localization. Homozygous mutation of a transcription factor Eya1 causes failure of ureteric bud outgrowth, and its metanephric mesenchyme lacks GDNF expression, suggesting that Eya1 regulates GDNF expression (Xu et al, 1999). In humans, haploinsufficiency of Eya1 results in a dominantly inherited disorder called branchio-oto-renal syndrome, which involves kidney and urinary tract anomalies (Abdelhak et al, 1997). Expression of PAX2 in the metanephric mesenchyme is also required for GDNF activation (Brophy et al, 2001). GDNF expression is also suppressed at the anterior boundary of the metanephric mesenchyme through the concerted action of the FoxC1 and FoxC2 transcription factors (Kume et al, 2000). Mutations in either Fox gene result in an expansion of GDNF expression and the formation of ectopic ureteric buds. Most FoxC1 homozygous mutants have duplex kidneys, in which the upper ureter is dilated and connects aberrantly to ectopic nephric duct derivatives in males such as seminal vesicles and vas deferens. In the developing kidneys, Slit2 is primarily expressed in the nephric duct whereas Robo2 is expressed in the metanephric mesenchyme (Piper et al, 2000). Mice deficient in Slit2 or Robo2 exhibit ectopic ureteric bud formation, multiple ureters and hydroureter, and anterior expansion of GDNF expression (Grieshammer et al, 2004). SPRY1 negatively regulates GDNF-RET signaling. Loss of SPRY1 function in mice results in renal malformations, including multiple ureters, duplex kidneys and hydroureter, and increased expression of GDNF in the metanephric mesenchyme (Basson et al, 2006). The data therefore suggest that multiple factors regulate, both positively and negatively, the precise timing and localization of GDNF, which then functions as a guidance cue to activate RET.
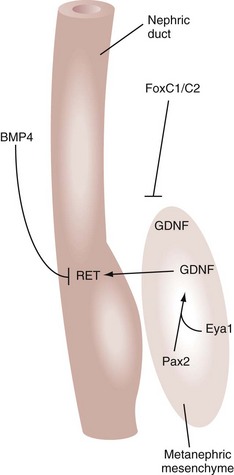
Figure 111–12 Inductive interactions during early kidney development. Glial cell line–derived neurotrophic factor (GDNF) is secreted from the metanephric mesenchyme and activates the RET receptor tyrosine kinase in the ureteric bud epithelium. The expression and localization of GDNF are positively regulated by Eya1 and Pax2 and negatively by FoxC transcription factors. The inducibility of nephric ducts to GDNF signaling is restricted by the action of BMP4.
(Modified from Dressler GR. Tubulogenesis in the developing mammalian kidney. Trends Cell Biol 2002;12:390–5.)
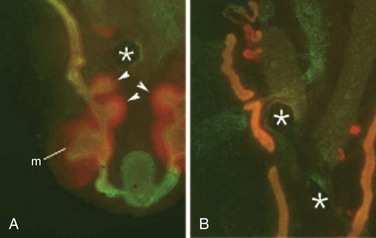
Figure 111–13 A, Stimulation of ectopic ureteric buds by GDNF. To determine whether GDNF is sufficient to promote outgrowth of nephric duct epithelial cells, heparin acrylamide beads preadsorbed with recombinant GDNF (asterisks) were placed between two nephric duct organ cultures. The native metanephros (m) is seen anteriorly. GDNF alone induces multiple ectopic ureteric buds in the posterior nephric ducts (arrowheads). B, However, the effect of GDNF is suppressed when Bmp4 is also added to the beads (asterisks). Cultures were stained with anti-cytokeratin (green) and anti-Pax2 (red) antibodies.
(From Dressler GR. Tubulogenesis in the developing mammalian kidney. Trends Cell Biol 2002;12:390–5.)
Ureteric Bud Branching
Once the ureteric bud has contacted the condensing metanephric mesenchyme it undergoes a dichotomous branching morphogenesis (Cebrian et al, 2004). Many of the same factors that regulate the initial outgrowth of the ureteric bud also appear to be essential for the subsequent branching of the ureteric bud. Ureteric bud branching is positively regulated by genetic and nutritional factors. PAX2, a paired-box type transcription factor that is mutated in humans with renal coloboma syndrome, is a positive regulator of ureteric bud branching. During renal development, Pax2 is expressed in the nephric duct, ureteric bud, and metanephric blastema induced by ureteric bud branch tips. Mice with Pax2 mutation exhibit decreased ureteric bud branching and renal hypoplasia (Porteous et al, 2000). Ureteric branching is also positively regulated by vitamin A and its retinoic acid receptor signaling, which promote Ret expression. Rarα and Rarβ2 are expressed in stromal cells surrounding Ret-expressing ureteric bud branch tips. Mice deficient in these receptors exhibit a decreased number of ureteric bud branches and diminished expression of Ret (Batourina et al, 2001). Certain markers such as Wnt11 might already be compartmentalized to opposing poles of the dilated bud tips, even before a morphologic branch point is evident (Pepicelli et al, 1997). In mice deficient for the homeobox gene Emx2 (Miyamoto et al, 1997), ureteric bud outgrowth into the metanephric mesenchyme appears normal but the leading edge never dilates and branching is suppressed. Thus ureteric development is arrested before the first branching event, and the resulting metanephric mesenchyme does not express any markers for induction. Similarly, mice with mutation of Sall1 exhibit developmental arrest just after ureteric bud outgrowth and before dilation of the leading edge (Nishinakamura et al, 2001). In normal murine embryos, Sall1 is expressed in the metanephric mesenchyme. Thus Sall1 might control mesenchyme-derived signals that are necessary for ureteric bud dilation and the early branch point determination. Clearly, the pattern of ureteric bud branching and the expression of ureteric bud–specific genes are influenced by the metanephric mesenchyme. Indeed, the heterologous mesenchyme derived from lung primordia can not only change the pattern of ureteric bud branching to that of lung epithelia but also induce the ureteric bud tissues to express lung-specific genes (Lin et al, 2001). Recent studies demonstrate that BMP/activin-like kinase-2 (ALK3) signaling negatively regulates early ureteric bud branching in vivo (Hartwig et al, 2008). The cell surface receptor ALK3 binds BMP2 and BMP4 with high affinity and is expressed in the nephric duct. Inactivation of ALK3 changes the pattern of primary ureteric bud branching from bifid to trifid and increases the number of first- and second-generation branches. These defects are associated with decreased formation of subsequent branch generations, resulting in a decreased complement of collecting ducts. These observations suggest that the pattern of early ureteric bud branching is a critical determinant of subsequent branching morphogenesis. Thus ureteric bud epithelial branching morphogenesis is controlled by both intrinsic and extrinsic factors working in concert to generate a kidney-specific branching pattern.
Tubulogenesis
Classic tissue recombination experiments focused almost exclusively on the relationship between metanephric mesenchymal cells and ureteric bud epithelial cells. It is now clear that at least three cell types are involved in the control of renal development: the ureteric bud tip cells, the condensed mesenchymal cells, and the stromal or interstitial mesenchymal cells (Fig. 111–14). It is not known whether the mesenchyme is a homogeneous cell population before its interaction with the ureteric bud. It is clear, however, that once induced by the ureteric bud the metanephric mesenchyme patterns itself into at least two different cell populations, a tubular one and a stromal one. The tubular cell population is thought to derive from mesenchymal cells in direct contact with the ureteric bud ampulla (Vainio et al, 1989; Stark et al, 1994; Torres et al, 1995), whereas the stromal cell population surrounds the tubular cells (Hatini et al, 1996). Once the mesenchyme has been patterned, the cells in the tubular zone undergo morphogenesis to become renal tubular epithelial cells. There is evidence that this process is dependent not only on signals from the ureteric bud but also on signals from the mesenchyme itself. One of these autocrine signals may be Wnt4, whose expression is induced in cells of the tubular zone on interaction with the ureteric bud. In Wnt4 gene knockout mice the ureteric bud forms and invades the metanephric mesenchyme but subsequent development of epithelial tubules is abolished (Stark et al, 1994). This suggests that two signals are essential for renal tubule formation—initial ureteric bud derived signals activating Wnt4 expression in the metanephric mesenchyme and Wnt4 itself as a mesenchymal autocrine signal. Signals from the stromal cell population also contribute to tubule formation as well, because tubulogenesis is perturbed in Bf2 gene knockout mice (Hatini et al, 1996). The discovery that Wnt4 acts as a downstream signal during the induction cascade leading to renal tubulogenesis leads to the question regarding the nature of the initial ureteric bud–derived signals. In-vitro data suggest a role for fibroblast growth factor-2 (FGF2) and other uncharacterized factors secreted by the ureteric bud (Karavanova et al, 1996). Candidate molecules that may cooperate with FGF2 are Wnt11 and BMP7 (Kispert et al, 1996; Vukicevic et al, 1996). Localization of RET protein to the ureteric bud tips is reinforced by both GDNF (Pepicelli et al, 1997) and signals emanating from surrounding stromal cells. For example, retinoic acid receptors are expressed in the stromal cells and are required for stromal cell–mediated signaling to maintain high levels of RET expression in the bud tips (Mendelsohn et al, 1999; Batourina et al, 2001). Consistent with the role of retinoic acid receptors in maintaining RET expression in the dividing ureteric bud, rats suffering from vitamin A deficiency have smaller kidneys and fewer nephrons (Lelievre-Pegorier et al, 1999). The cellular crosstalk among stromal, mesenchymal, and ureteric bud cells is further highlighted by gain- and loss-of-function experiments involving FGFs and BMPs. Fgf7 null mutant mice have fewer branch points and correspondingly fewer nephrons, whereas ectopic FGF7 in organ culture can stimulate branching (Qiao et al, 1999). FGF1 and FGF10 affect elongation of the ureteric bud stalk before the branch-point decision is made (Qiao et al, 2001). Null mutations in Bmp7 are associated with even more severe phenotypic anomalies, exhibiting limited branching morphogenesis and complete renal developmental arrest. Yet it is difficult to assess how FGFs and BMPs exert their collective effects on branching given the interplay among all the cell types present in the early kidney (Dudley et al, 1999). In addition to the proteins just mentioned, a growing list of growth factors, secreted peptides, and their receptors have been implicated in the control of branching morphogenesis, most by using a variety of in-vitro model systems (Pohl et al, 2000; Davies, 2001). For many of these factors, however, genetic studies have not proved conclusive in assigning specific functional roles during ureteric bud branching in vivo, either because of potential redundancies or embryonic lethalities before the onset of kidney development. Nevertheless, the role of these factors in the renal development must be considered.
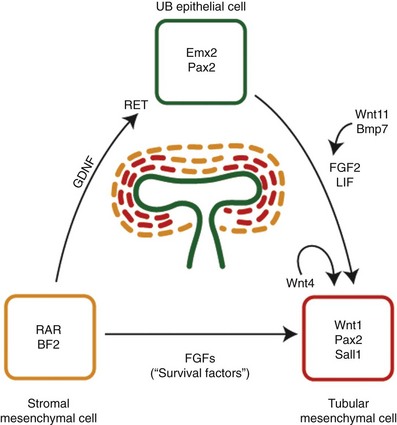
Figure 111–14 Cell-cell interactions promote nephrogenesis. Three major cell types—ureteric bud (UB) epithelial cells, condensing tubular mesenchymal cells, and stromal mesenchymal cells—are thought to play a critical role. At the UB tips, cells express unique markers such as Emx2 and Pax2. The stromal cell lineage is marked by expression of retinoic acid receptors (RAR) and BF2. Presence of Pax2, WT1, and Sall1 appear to be important for continued branching morphogenesis of the UB. Wnt4 is activated in the tubular mesenchymal cells by the invading UB and stimulates the development of polarized epithelium in an autocrine fashion. Finally, fibroblast growth factors (FGFs), such as FGF2, along with LIF, may be critical as survival factors for the developing renal tubular epithelial cells.
(Modified from Dressler GR. Tubulogenesis in the developing mammalian kidney. Trends Cell Biol 2002;12:390–5.)
Mesenchymal-Epithelial Conversion
The inductive signals emanating from the ureteric bud promote condensation of the metanephric mesenchymal cells around the ureteric bud tips and subsequent tubulogenesis. Mice with null mutations of Pax2 or Wt1 fail to exhibit ureteric bud outgrowth and, in both cases, the metanephric mesenchyme does not respond to induction even when recombined with strong inducers in vitro (Kreidberg et al, 1993; Brophy et al, 2001). The establishment of glomerular versus tubular cell fates is dependent on negative feedback between Wt1 and Pax2 (Ryan et al, 1995). During early kidney development, the expression domain of Pax2 is complementary to that of Wt1 in S-shaped bodies. Wt1 expression is restricted to glomerular epithelial precursors (Pelletier et al, 1991), whereas Pax2 expression is restricted to the portion that gives rise to tubular epithelial precursors of the proximal and distal nephron segments and later repressed in differentiated tubular epithelium (Dressler and Douglass, 1992). Evidence in support of Wnt proteins as mesenchyme inducers has been gained from in-vitro induction assays using Wnt-expressing cell lines (Herzlinger et al, 1994; Kispert et al, 1998). Of the Wnt mutants examined to date, only Wnt4, which is expressed in the mesenchyme and not the ureteric bud, is crucial for propagation of the inductive signals. Although Wnt4 mutant mesenchyme is able to aggregate in response to ureteric bud contact, these mutant aggregates do not form polarized epithelia. Rat ureteric bud cells secrete tubulogenic factors, such as leukocyte-inhibitory factor (LIF), which, together with FGF2, appears to stimulate growth and tubulogenesis in vitro (Plisov et al, 2001). Once induced to form aggregates, metanephric mesenchyme becomes polarized into an early renal vesicle. This vesicle is closely associated with the branching ureteric bud and will eventually connect to the ureteric bud epithelium to form a continuous tubule. Profound changes take place in the expression of cell adhesion molecules such as cadherins. Shortly after induction, metanephric mesenchyme expresses R-cadherin, cadherin-6, and E-cadherin, along with suppression of the mesenchyme-specific cadherin-11. Both R-cadherin and cadherin-6 mutants show defects in the rate of mesenchymal condensation and polarization (Mah et al, 2000; Dahl et al, 2002). Some renal vesicles in cadherin-6 mutants also fail to fuse to the ureteric bud epithelia, resulting in “dead end” tubules and a subsequent loss of nephrons.
Renal Vascular Development
The origin of intrarenal vasculature is not completely understood. Until recently it was thought that renal vasculature derived exclusively from branches off the aorta and other pre-existing extrarenal vessels (“angiogenic” hypothesis). There is evidence, however, that the renal vessels may originate in situ, within the embryonic kidney from vascular progenitor cells (“vasculogenic” hypothesis) (Loughna et al, 1996; Tufro et al, 1999). Using antibodies to Flk-1, a vascular endothelial growth factor (VEGF) receptor present in angioblasts and mature endothelial cells, it was demonstrated that endothelial cell precursors were already present in the prevascular rodent kidneys before any vessels were discernible from a morphologic standpoint. When embryonic kidneys are cultured at the usual atmospheric oxygen concentration, vessels do not develop. However, if the explants are cultured in a hypoxic atmosphere containing 5% oxygen, capillary sprouts develop within and outside the glomeruli, an effect that is inhibited by anti-VEGF antibodies (Tufro-McReddie et al, 1997). Depending on the developmental potential of the cells involved, both angiogenesis and vasculogenesis may play a role in the development of renal vasculature (Abrahamson et al, 1998).
Bladder and Ureter Development
Formation of Urogenital Sinus
At the third week of gestation the cloacal membrane remains a bilaminar structure composed of endoderm and ectoderm. During the fourth week the neural tube and the tail of the embryo grow dorsally and caudally, projecting itself over the cloacal membrane; and this differential growth of the body results in embryo folding. The cloacal membrane is now turned to the ventral aspect of the embryo, and the terminal portion of the endoderm-lined yolk sac dilates and becomes the cloaca (Fig. 111–15). According to the embryonic theories of Rathke and Tourneux, the partition of the cloaca into an anterior urogenital sinus and a posterior anorectal canal occurs by the midline fusion of two lateral ridges of the cloacal wall and by a descending urorectal septum. This process is thought to occur during the fifth and sixth weeks, and it is culminated by the fusion of this urorectal septum with the cloacal membrane. Recently, however, some investigators have challenged this classic view with evidence that there is neither a descending septum nor fusing lateral ridges of the cloacal wall (van der Putte, 1986; Kluth et al, 1995). There is further evidence that the urorectal septum never fuses with the cloacal membrane (Nievelstein et al, 1998). According to these observations the congenital cloacal and anorectal malformations, which were previously thought to occur due to a failure of septum formation and its fusion with the cloacal membrane, may in fact occur from an abnormal development of the cloacal membrane itself (Nievelstein et al, 1998) (Fig. 111–16).
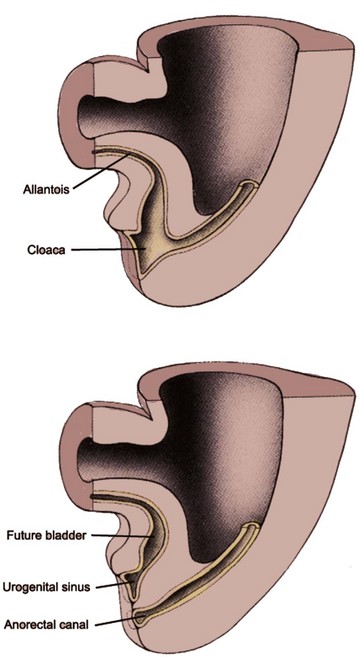
Figure 111–15 Development of the urogenital sinus. Between the fourth and sixth weeks the cloaca is divided into an anterior urogenital sinus and a posterior anorectal canal. The superior part of the urogenital sinus, continuous with the allantois, forms the bladder. The constricted narrowing at the base of the urogenital sinus forms the pelvic urethra. The distal expansion of the urogenital sinus forms the vestibule of the vagina in females and the penile urethra in males.
(Modified from Larsen WJ. Human embryology. New York: Churchill Livingstone; 1997.)
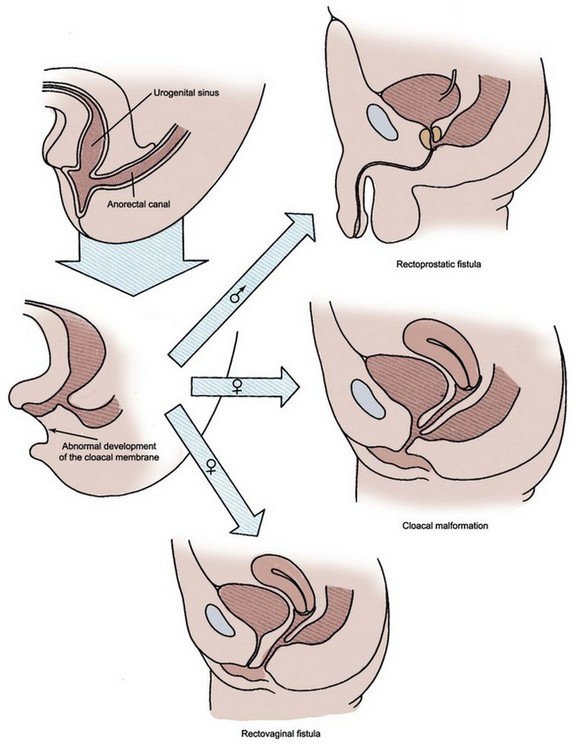
Figure 111–16 Abnormal development of cloacal membrane results in characteristic anomalies of the urogenital and lower gastrointestinal tract.
(Modified from Larsen WJ. Human embryology. New York: Churchill Livingstone; 1997.)
The nephric (wolffian) duct fuses with the cloaca by the 24th day and remains with the urogenital sinus during the cloacal separation. The entrance of the nephric duct into the primitive urogenital sinus serves as a landmark distinguishing the cephalad vesicourethral canal from the caudal urogenital sinus. The vesicourethral canal gives rise to the bladder and pelvic urethra, whereas the caudal urogenital sinus forms the phallic urethra for males and distal vaginal vestibule for females.
Formation of Trigone
By day 33 of gestation, the common excretory ducts (the portion of nephric ducts distal to the origin of ureteric buds) dilate and connect to the urogenital sinus. The formation of these final connections involves apoptosis, which enables the ureters to disconnect from the nephric ducts, and fusion, in which the ureteral orifice inserts into the urogenital sinus epithelium at the level of the trigone (Batourina et al, 2005). According to the classic view (Weiss 1988), the right and left common excretory ducts fuse in the midline as a triangular area, forming the primitive trigone, structurally different from bladder and urethra. The ureteral orifice extrophies and evaginates into the bladder by day 37 and begins to migrate in a cranial and lateral direction within the floor of the bladder. During this process the nephric duct orifice diverges away from the ureteral orifice and migrates caudally, flanking the paramesonephric (müllerian) duct at the level of the urogenital sinus. This is the site of the future verumontanum in males and vaginal canal in females. Recent studies, however, have challenged this classic mechanism of trigone development. Using the cell lineage studies in mice, the trigone was found to form mostly from bladder smooth muscle cells with only a minor contribution from the ureters (Viana et al, 2007).
The embryonic pattern of ureteral orifice incorporation into the developing bladder is inferred primarily from clinical observations of duplex kidneys. The upper pole ureteral orifice rotates posteriorly relative to the lower pole orifice and assumes a more caudal and medial position. Weigert and Meyer recognized the regularity of this relationship between upper and lower pole ureteral orifices, which has come to be known as the Weigert-Meyer rule. According to this concept, an abnormally lateral lower pole ureteral orifice may result from a ureteric bud arising too low on the nephric duct, therefore resulting in premature incorporation and migration within the developing bladder. In such a ureteral orifice, vesicoureteral reflux is more likely to occur due to an inadequate intramural tunnel. In contrast, an abnormally caudal upper pole ureteral orifice may result from a ureteric bud arising too high on the nephric duct. It may drain at the bladder neck and verumontanum or remain connected to the nephric (wolffian) duct derivatives such as the vas deferens in males (Mackie and Stephens, 1977; Schwarz and Stephens, 1978). In females, the ectopic upper pole ureter may insert into the remnants of the nephric ducts (e.g., Gartner duct cyst) or vaginal vestibule (Fig. 111–17).
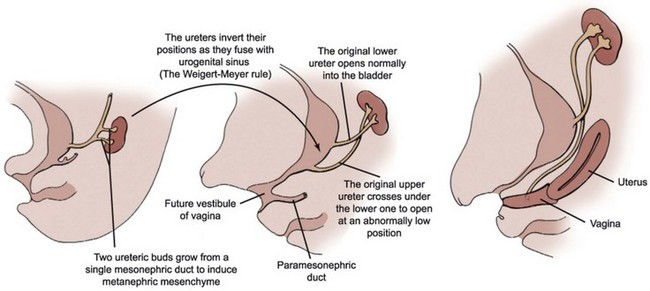
Figure 111–17 Development of an ectopic upper pole ureter draining into the vagina.
(Modified from Larsen WJ. Human embryology. New York: Churchill Livingstone; 1997.)
Anomalous development of the common excretory duct may lead to an ectopic vas deferens. In certain clinical situations the vas deferens is connected to the ureter rather than the verumontanum, so that both the ureter and vas drain into a common duct. This situation may occur when the ureteric bud arises too high on the nephric duct and the subsequent common excretory duct becomes too long, resulting in incomplete absorption into the developing bladder. This anomaly, although rare, should be kept in mind when evaluating males with recurrent epididymitis and ipsilateral hydronephrosis.
Development of the Ureter
In contrast to the previous discussion regarding the molecular aspects of renal development, little is understood at the molecular level concerning the events of ureteral development. There is only a small amount of descriptive information and speculative theories regarding the molecular mechanism of smooth muscle cell and urothelial differentiation. Morphologically, the ureter begins as a simple cuboidal epithelial tube surrounded by loose mesenchymal cells that acquires a complete lumen at 28 days of gestation in human. It was suggested that the developing ureter undergoes a transient luminal obstruction between 37 and 40 days’ gestation and subsequently recanalizes (Alcaraz et al, 1991). It appears that this recanalization process begins in the midureter and extends in a bidirectional manner both cranially and caudally. In addition, another source of physiologic ureteral obstruction may exist as the Chwalla membrane, a two-cell-thick layer over the ureteral orifice that is seen between 37 and 39 days’ gestation. In humans, urine production is followed by proliferative changes in the ureteral epithelium (bilaminar by 10 weeks of gestation). The epithelium attains a transitional configuration by 14 weeks. The first signs of ureteral muscularization and development of elastic fibers are seen at 12 weeks of gestation. In both rat and human the ureteral smooth muscle phenotype appears later than that of the bladder. Smooth muscle differentiation is first detected in the subserosal region of the bladder dome and extends toward the bladder base and urethra, whereas smooth muscle differentiation of the ureter occurs later within the subepithelial region in the ureterovesical junction, ascending toward the intrarenal collecting system (Baker and Gomez, 1998). In the embryonic ureter and bladder it is likely that epithelial-mesenchymal interactions are important in the development of urothelium, lamina propria, and muscular compartments, but the exact nature of this induction process is unknown. Before 10 weeks of gestation elastic fibers are few in number, poorly developed, and randomly arranged. After 12 weeks these fibers become more numerous throughout the ureter and are seen with specific orientation (Escala et al, 1989).
Although more than 30 genes are found to be involved in the development of mammalian kidneys, only a few genes have been thus far demonstrated to concurrently cause both kidney and ureteral anomalies—Agtr2, Bmp4, FoxC1, Pax2, and Eya1 (see Molecular Mechanism of Kidney Development, earlier). A mutation of PAX2 gene has been identified in a human family carrying renal coloboma syndrome, a rare autosomal dominant syndrome characterized by optic nerve coloboma, renal anomalies, and vesicoureteral reflux (Sanyanusin et al, 1996). EYA1 is mutated in patients with dominantly inherited disorder, branchio-oto-renal syndrome, which includes a duplex collecting system, renal hypoplasia/dysplasia, and renal agenesis (Abdelhak et al, 1997). Pax2 is required for the growth and elongation of nephric ducts prior to ureteric bud formation, whereas Eya1 appears to regulate the GDNF expression, which is a prerequisite for ureteric bud outgrowth. Bmp4 and FoxC1 appear to play a suppressive role in the ureteric bud outgrowth.
The renin-angiotensin system is present and active during fetal life. It is generally thought that the major role of this system in the fetus is to maintain fetal glomerular filtration and to ensure an adequate urine production (Lumbers, 1995). There is growing evidence, however, that the renin-angiotensin system is also important for normal growth and development of the kidney and ureter. The kidney is able to produce all components of this system, and thus the local (intrarenal) production of angiotensin II may play a critical role in this regard. Renin mRNA is detectable in the human mesonephros at about 30 days of gestation and in the metanephros at about 56 days of gestation (Schutz et al, 1996). A similar profile of expression is seen for angiotensinogen and angiotensin-converting enzyme (ACE). Mutant mice lacking ACE are found to have abnormal renal vasculature and tubules as well as increased renin synthesis in interstitial and perivascular cells (Hilgers et al, 1997). Pharmacologic inhibition of ACE in the neonatal rat produces irreversible abnormalities in renal function and morphology (Guron et al, 1997), supporting that an intact renin-angiotensin system is crucial for normal kidney development and maturation. In addition to the high rate of fetal loss, infants born to human mothers treated with ACE inhibitors during pregnancy have increased rates of oligohydramnios, hypotension, and anuria (Shotan et al, 1994; Sedman et al, 1995).
Both subtypes of angiotensin II receptors, AT1 and AT2, are expressed in the developing mesonephros and metanephros. AT2 expression predominates in the undifferentiated mesenchymal cells that surround the nephric duct at the time of ureteric bud outgrowth and declines with maturation, and this pattern of expression suggests AT2’s role in embryonic renal development. AT1 is expressed in more differentiated structures and may be involved in modulating later stages of renal vascular development and acquisition of classic angiotensin II–mediated effects of vasoconstriction and sodium reabsorption. The function of AT2 receptor is not defined completely, but when its gene, Agtr2, was inactivated genetically in mice these mutants demonstrated a significant incidence of anomalies in the kidney and urinary tract. Abnormal phenotype in these mice mimicked all the key features of human congenital anomalies of the kidney and urinary tract, such as ureteropelvic junction obstruction, hypoplastic kidney, vesicoureteral reflux, megaureter, and duplicated collecting system (Nishimura et al, 1999).
Because of its embryonic expression pattern, it was initially speculated that AT2 might play a role in regulating the initial outgrowth of the ureteric bud. Analysis of whole tissue sections showed that ectopic ureteric budding occurred in Agtr2-deficient mutant mice (Oshima et al, 2001). It was thus postulated that similar to Bmp4, AT2 might have a role in directing the site of ureteric bud outgrowth through its inhibitory effect. In other words, a defect in this process may lead to an abnormal timing and location of the ureteric bud outgrowth, resulting in congenital ureteral anomalies.
Recent evidence suggest that BMPs control formation of smooth muscle in the proximal ureter and pelvis. BMP4, expressed in the caudal mesenchyme cells, induces ureteral morphogenesis including smooth muscle differentiation and urothelial development (Brenner-Anantharam et al, 2007). Consistent with such a role, Bmp4- and Bmp5-mutant mice display hydronephrosis and hydroureter (Miyazaki et al, 2003).
Development of the Bladder and Continence Mechanism
By the 10th week of gestation the bladder is a cylindrical tube lined by a single layer of cuboidal cells surrounded by loose connective tissue. The apex tapers as the urachus, which is contiguous with the allantois. By the 12th week the urachus involutes to become a fibrous cord, which becomes the median umbilical ligament. The bladder epithelium consists of bilayered cuboidal cells between the 7th and 12th weeks, and it begins to acquire mature urothelial characteristics between the 13th and 17th weeks. By the 21st week it becomes four- to five-cell layers thick and demonstrates ultrastructural features similar to the fully differentiated urothelium. Between the 7th and 12th weeks the surrounding connective tissues condense and smooth muscle fibers begin to appear, first at the region of the bladder dome and later proceeding toward the bladder base. Collagen fibers first appear in the lamina propria and then later extend into the deeper wall between the muscle fibers (Newman and Antonakopoulos, 1989).
Bladder compliance is thought to change during development. When studied in whole organ preparation using fetal sheep bladders, bladder compliance is very low during early gestation and increases gradually thereafter (Coplen et al, 1994). The mechanism of these changes in bladder compliance is not known but may involve alterations in both smooth muscle tone and connective tissue composition. This phenomenon is also observed in developing human bladders (Kim et al, 1991). During gestation the bladder wall muscle thickness increases and the relative collagen content decreases. The ratio of thick-to-thin collagen fibers also decreases, whereas the amount of elastic fibers increases. These changes in compliance seem to coincide with the time of fetal urine production, suggesting a possible role for mechanical distention (Baskin et al, 1994). Using fetal mouse bladders as organ culture explants, bladder distention promoted a more orderly development of collagen fiber bundles within the lamina propria in comparison to decompressed bladder explants, suggesting that mechanical factors from accumulating urine may play a role during bladder development (Beauboeuf et al, 1998).
Similar to other organ development, the epithelial-mesenchymal inductive interactions appear to be necessary for orderly differentiation and proper development of the bladder. A modified Grobstein technique was applied to study the mechanism of bladder smooth muscle cell differentiation (Baskin et al, 1996). Undifferentiated rat bladder epithelial and mesenchymal rudiments were separated before bladder smooth muscle cell differentiation and then recombined to grow within the immunologically compromised host (athymic nude mouse). In the presence of epithelial cells, the mesenchymal cells differentiated into smooth muscle cells with sequential expression of appropriate smooth muscle markers, whereas in the absence of epithelial cells they involuted with evidence of apoptosis.
No functional study has been done to assess fetal continence mechanism. Only a handful of ontogenic descriptions are available using human fetal specimens, providing a basis for speculative theories. A mesenchymal condensation forms around the caudal end of the urogenital sinus after the division of the cloaca and the rupture of the cloacal membrane. Striated muscle fibers can be seen clearly by the 15th week. At this time the smooth muscle layer becomes thicker at the level of bladder neck and forms the inner part of the urethral musculature. The urethral sphincter, composed of central smooth muscle fibers and peripheral striated muscle fibers, develops in the anterior wall of the urethra (Bourdelat et al, 1992). Beyond this point, sexual dimorphism develops in conjunction with the formation of the prostate in males and the vagina in females (Tichy, 1989). The urethral sphincter muscle fibers extend to the posterior wall of the urethra. In males these fibers project to the lateral wall of the prostate, whereas in females the muscle fibers attach to the lateral wall of the vagina.
Genital and Reproductive Tract Development
Formation of Genital Ridges and Paramesonephric Ducts
During the fifth week, primordial germ cells migrate from the yolk sac along the dorsal mesentery to populate the mesenchyme of the posterior body wall near the 10th thoracic level (Fig. 111–18). In both sexes the arrival of primordial germ cells in the area of future gonads serves as the signal for the existing cells of the mesonephros and the adjacent coelomic epithelium to proliferate and form a pair of genital ridges just medial to the developing mesonephros (Fig. 111–19). During the sixth week the cells of the genital ridge invade the mesenchyme in the region of future gonads to form aggregates of supporting cells called the primitive sex cords. The primitive sex cords will subsequently invest the germ cells and support their development. The genital ridge mesenchyme containing the primitive sex cords is divided into the cortical and medullary regions. Both regions develop in all embryos, but after the sixth week they pursue different fates in the male and female.
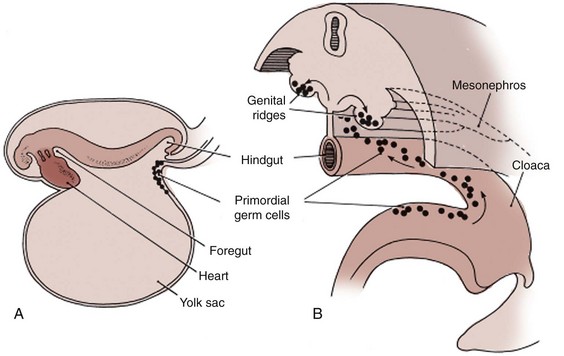
Figure 111–18 A, The site of the primordial germ cell origin in the wall of the yolk sac in a 3-week-old embryo. B, Migratory path of the primordial germ cells along the wall of the yolk sac and dorsal mesentery into the developing genital ridges.
(Modified from Sadler TW. Langman’s medical embryology. Baltimore: Williams & Wilkins; 1985.)
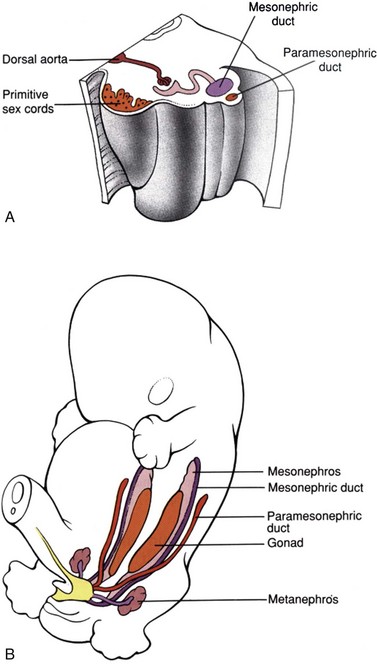
Figure 111–19 Formation of genital ridges and paramesonephric ducts. A, During the fifth and sixth weeks the genital ridges form in the posterior abdominal wall just medial to the developing mesonephros. The primordial germ cells induce the coelomic epithelial cells lining the peritoneal cavity and the cells of the mesonephros to proliferate and form the primitive sex cords. B, During the sixth week, the paramesonephric ducts develop lateral to the mesonephros. The caudal tips of the paramesonephric ducts fuse with each other as they connect with the urogenital sinus.
(Modified from Larsen WJ. Human embryology. New York: Churchill Livingstone; 1997.)
During this time a new pair of ducts, called the paramesonephric (müllerian) ducts, begins to form just lateral to the nephric ducts in both male and female embryos (Fig. 111–20). These ducts arise by the craniocaudal invagination of thickened coelomic epithelium, extending all the way from the third thoracic segment to the posterior wall of the developing urogenital sinus. The caudal tips of the paramesonephric ducts adhere to each other as they connect with the urogenital sinus between the openings of the right and left nephric ducts. The cranial ends of the paramesonephric ducts form funnel-shaped openings into the coelomic cavity, which is the future peritoneum.
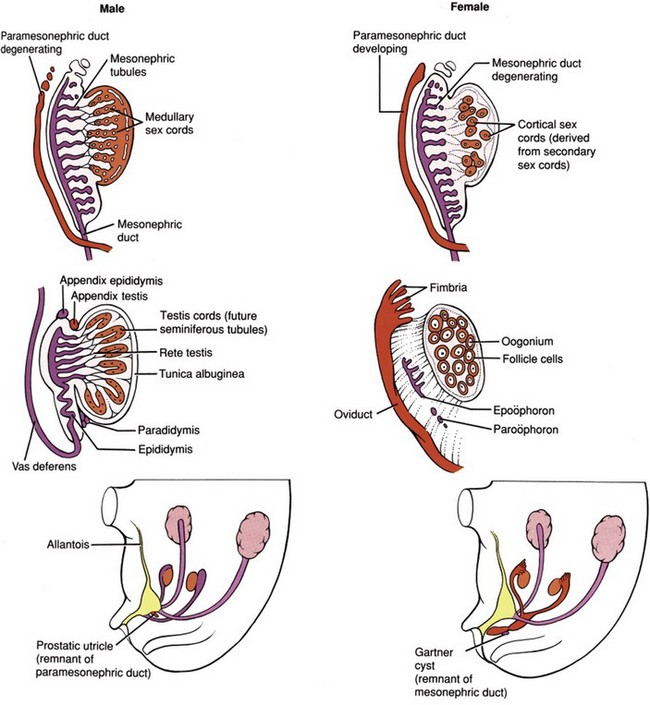
Figure 111–20 Male and female gonad and genital development. The male and female genital structures are virtually identical through the seventh week. In males, SRY protein produced by the Sertoli cells causes the medullary sex cords to become presumptive seminiferous tubules and causes the cortical sex cords to regress. Müllerian-inhibiting substance (MIS), a glycoprotein hormone produced by the Sertoli cells, then causes the paramesonephric ducts to regress, leaving behind appendix testis and prostatic utricle as remnants. Appendix epididymis and paradidymis arise from the mesonephric ducts. In females, cortical sex cords invest the primordial germ cells and become the ovarian follicles. In the absence of MIS, the mesonephric ducts degenerate and the paramesonephric ducts give rise to the fallopian tubes, uterus, and the upper vagina. The remnants of the mesonephric ducts are found in the ovarian mesentery as the epoöphoron and paroöphoron, and in the anterolateral vaginal wall as the Gartner’s duct cysts.
(Modified from Larsen WJ. Human embryology. New York: Churchill Livingstone; 1997.)
Development of Male Genital Structures
Under the influence of SRY (the Sex-determining Region of the Y chromosome), cells in the medullary region of the primitive sex cords begin to differentiate into Sertoli cells, while the cells of the cortical sex cords degenerate. Sex cord cells differentiate into Sertoli cells only if they contain the SRY protein; otherwise the sex cords differentiate into ovarian follicles. During the seventh week, the differentiating Sertoli cells organize to form the testis cords. At puberty these testis cords associated with germ cells undergo canalization and differentiate into seminiferous tubules. Direct cell-to-cell contact between developing Sertoli cells and primordial germ cells is thought to play a key role in the proper development of male gametes. This interaction occurs shortly after the arrival of the primordial germ cells in the presumptive genital ridge. The testis cords distal to the presumptive seminiferous tubules also develop lumen and differentiate into a set of thin-walled ducts called the rete testis. Just medial to the developing gonad the tubules of rete testis connect with 5 to 12 residual tubules of nephric ducts, called efferent ductules. The vas deferens also develops from the nephric duct. At this time the testis begins to become round, reducing its area of contact with the surrounding mesonephros. As the testis continues to develop the degenerating cortical sex cords become separated from the coelomic (peritoneal) epithelium by an intervening layer of connective tissue called the tunica albuginea (see Fig. 111–20).
As the developing Sertoli cells begin their differentiation in response to the SRY protein they also begin to secrete a glycoprotein hormone called müllerian-inhibiting substance (MIS). MIS causes the paramesonephric (müllerian) ducts to regress rapidly between the 8th and 10th weeks. Small müllerian duct remnants can be detected in the developed male as a small tissue protrusion at the superior pole of the testis, called the appendix testis, and as a posterior expansion of the prostatic urethra, called the prostatic utricle. In female embryos, MIS is absent; therefore the müllerian ducts do not regress. Occasionally, genetic males have persistent müllerian duct structures (uterus and fallopian tubes), a condition known as hernia uteri inguinale. In these individuals either MIS production by Sertoli cells is deficient or the müllerian ducts do not respond to normal MIS levels.
During the 9th and 10th weeks, Leydig cells differentiate from mesenchymal cells of the genital ridge in response to the SRY protein. These endocrine cells produce testosterone. At an early stage of development testosterone secretion is regulated by placental chorionic gonadotropin, but eventually the pituitary gonadotropins assume control of androgen production. Between the 8th and 12th weeks, testosterone secretion by Leydig cells stimulates the nephric (wolffian) ducts to transform into the vas deferens. The cranial portions of the nephric ducts degenerate, leaving a small remnant of tissue protrusion called the appendix epididymis, and the region of nephric ducts adjacent to the presumptive testis differentiate into the epididymis. During the 9th week, 5 to 12 nephric ducts in the region of the epididymis make contact with the sex cords of the future rete testis. It is not until the third month, however, that these tubules actually establish communication with the rete testis as the efferent ductules. Meanwhile, the nephric duct–derived tubules near the inferior pole of the developing testis degenerate, sometimes leaving a remnant of tissue protrusion called the paradidymis.
Prostate and Seminal Vesicle Development
The seminal vesicles sprout from the distal nephric ducts, whereas the prostate and bulbourethral glands develop from the urogenital sinus (Fig. 111–21). They therefore have different embryologic origins. The initial event in prostatic development is an outgrowth of solid epithelial cords from the urogenital sinus epithelium into the surrounding mesenchyme during weeks 10 to 12 of gestation. This prostatic bud growth and subsequent branching morphogenesis occur in a specific spatial pattern that eventually establishes the lobar subdivisions of the mature prostate gland (Sugimura et al, 1986; Timms et al, 1994). The solid prostatic ducts are subsequently canalized from their urethral connections, proceeding distally toward the ductal tips. As the solid epithelial cords canalize, the epithelium organizes itself into two distinct cell types—luminal and basal cells (Hayward et al, 1996). At this time the prostatic mesenchyme differentiates into a layer of smooth muscle cells that surround the prostatic ducts (Hayward et al, 1996). At puberty, corresponding to a rise in circulating testosterone, the prostate size increases rapidly, along with functional cytodifferentiation of luminal cells, as evidenced by the expression of prostate-specific secretory proteins (Hayward et al, 1996).
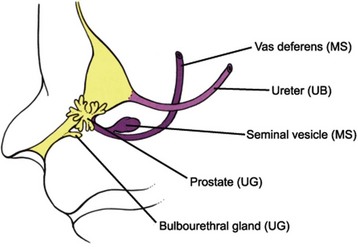
Figure 111–21 Development of male accessory sex glands. During the 10th week, the seminal vesicles sprout from the distal mesonephric ducts in response to testosterone whereas the prostate and bulbourethral glands develop from the urethra in response to dihydrotestosterone. Thus the vas deferens and seminal vesicle derive from the mesonephric ducts (MS) while prostate and bulbourethral glands develop from the urogenital sinus (UG). UB, ureteric bud.
(Modified from Larsen WJ. Human embryology. New York: Churchill Livingstone; 1997.)
Circulating androgens produced by fetal testes play a critical role in the development of the prostate. Cellular responses to circulating androgens are mediated by nuclear androgen receptors that are activated by either testosterone or dihydrotestosterone (DHT). The evidence for the requirement of androgens in establishing the prostate specificity of the urogenital sinus comes primarily from the absence of prostate development in mice and humans that lack functional androgen receptors (Lubahn et al, 1989; He et al, 1991), as well as from the development of the prostate in the female urogenital sinus exposed to androgens (Takeda et al, 1986). In the urogenital sinus, testosterone could activate androgen receptors by directly binding to the receptor and also through a local conversion of circulating testosterone into the more potent DHT by the enzyme 5α-reductase (Russell and Wilson, 1994). DHT has a 10-fold greater affinity for the androgen receptor than testosterone (Deslypere et al, 1992). When 5α-reductase is deficient, the urogenital sinus is specified to become the prostate but the overall prostatic growth and development are severely compromised (Andersson et al, 1991). Tissue recombination and grafting experiments using testicular feminization mice that lack functional androgen receptor have shown that the presence of androgen receptors in the urogenital sinus mesenchyme is required for prostate specification and differentiation (Cunha and Lung, 1978). The fact that mesenchymal but not epithelial androgen receptors are required for prostate-specific ductal growth and branching suggests that paracrine signals from the urogenital sinus mesenchyme mediate the action of androgens on the epithelium. Prostate development appears to be affected by the levels of estrogenic compounds as well (vom Saal et al, 1997; Timms et al, 1999), but their specific role has not been fully elucidated.
Prostate development requires inductive and reciprocal interactions between the urogenital sinus epithelium and mesenchyme. In addition to mediating the effect of androgens to the developing prostatic epithelium, paracrine signals from the urogenital sinus mesenchyme also appear to direct lobe-specific patterning of juxtaposed epithelium (Timms et al, 1995). The urogenital sinus mesenchyme when combined with either embryonic or adult bladder epithelium (also a derivative of endodermal cloaca) will stimulate formation of prostatic ducts. In contrast, the urogenital sinus mesenchyme combined with epithelia of other anatomic origins, such as seminal vesicle (a mesodermal derivative), salivary gland, or esophagus, forms tissues with epithelial characteristics that resemble the anatomic origin of the partnering epithelium (Cunha et al, 1987). These observations suggest that prostate development is spatially restricted by prostate-inducing paracrine signals from the urogenital sinus mesenchyme and that epithelial potential to respond to signals from the urogenital sinus mesenchyme is restricted to the endodermal epithelia of similar embryonic origin as the prostate. The interactions between epithelium and mesenchyme are reciprocal. The presence of prostate epithelium plays a critical role in the differentiation of mesenchymal cells into the periductal smooth muscle cells (Hayward et al, 1998).
Several candidate genes have been implicated in the prostate development, but the nature of paracrine mesenchymal factors that drive urogenital sinus epithelial transformation into prostatic ducts remains unknown. Furthermore, the precise relationship and the embryologic sequence of these candidate molecules have not been clearly defined. The Hox family of homeobox genes may be involved in the proper differentiation of male accessory sex glands, including the prostate (Podlasek et al, 1997, 1999b). In particular, Hoxa-13 and Hoxd-13 transcription factors are expressed in both urogenital sinus and nephric ducts, and the loss of function mutation of these genes in mice results in agenesis of bulbourethral glands and defective morphogenesis of the prostate and seminal vesicles. Two members of the FGF family of secreted proteins, FGF7 and FGF10, are expressed in the urogenital sinus mesenchyme. In-vitro organ culture experiments have shown that exogenous FGF7 and FGF10 can stimulate proliferation and branching of developing prostate tissue, but these factors do not appear to be androgen responsive (Thomson and Cunha, 1999). There is also evidence that secreted factor activin-A and its antagonistic binding protein follistatin may be important in the regulation of prostate epithelial development (Cancilla et al, 2001). Activin-A is expressed in both urogenital sinus epithelium and mesenchyme, whereas its receptors are found in the epithelium. Follistatin, an activin-A antagonist, is expressed in the urogenital sinus epithelium. Prostatic ductal growth and branching might therefore be a result of balanced interplay between activin-A and follistatin. Other implicated molecules in prostate development include Bmp4 (Lamm et al, 2001), growth hormone receptor (Ruan et al, 1999), insulin-like growth factor-1 (Ruan et al, 1999), Nkx3.1 (Bhatia-Gaur et al, 1999), sonic hedgehog (Podlasek et al, 1999a), P63 (Signoretti et al, 2000), prolactin (Steger et al, 1998), hyaluronan (Gakunga et al, 1997), fucosyltransferase-1 (Marker et al, 2001), and urokinase plasminogen activator (Elfman et al, 2001).
Development of Female Genital Structures
In female embryos the primitive sex cords do not contain the Y chromosome, do not elaborate SRY protein, and therefore do not differentiate into Sertoli cells. In the absence of Sertoli cells and SRY protein, therefore, MIS synthesis, Leydig cell differentiation, and androgen production do not occur. Consequently, male development of the genital ducts and accessory glands is not stimulated and female development ensues. In females the primitive sex cords degenerate and the mesothelium of the genital ridge forms the secondary cortical sex cords. These secondary sex cords invest the primordial germ cells to form the ovarian follicles. The germ cells differentiate into oogonia and enter the first meiotic division as primary oocytes. The follicle cells then arrest further germ cell development until puberty, at which point individual oocytes resume gametogenesis in response to a monthly surge of gonadotropins.
In the absence of MIS and androgens, the nephric (wolffian) ducts degenerate, and the paramesonephric (müllerian) ducts give rise to the fallopian tubes, uterus, and upper two thirds of the vagina. The remnants of nephric ducts are found in the mesentery of the ovary as the epoöphoron and paroöphoron and near the vaginal introitus and anterolateral vaginal wall as Gartner duct cysts. The distal tips of the paramesonephric ducts adhere to each other just before they contact the posterior wall of the urogenital sinus. The wall of the urogenital sinus at this point forms a small thickening called the sinusal tubercle. As soon as the fused tips of the paramesonephric ducts connect with the sinusal tubercle the paramesonephric ducts begin to fuse in a caudal to cranial direction, forming a tube with a single lumen. This tube, called the uterovaginal canal, becomes the superior portion of the vagina and the uterus. The unfused, superior portions of the paramesonephric ducts become the fallopian tubes (oviducts), and the funnel-shaped superior openings of the paramesonephric ducts become the infundibula.
While the uterovaginal canal is forming during the third month, the endodermal tissue of the sinusal tubercle in the posterior urogenital sinus continues to thicken, forming a pair of swellings called the sinovaginal bulbs. These structures give rise to the lower third of the vagina. The most inferior portion of the uterovaginal canal becomes occluded transiently by a block of tissue called the vaginal plate. The origin of the vaginal plate is not clear; it may arise from the sinovaginal bulbs, from the walls of the paramesonephric ducts, from the nearby mesonephric ducts, or from a combination of these tissues. The vaginal plate elongates between the third to fifth month and subsequently becomes canalized to form the inferior vaginal lumen (Fig. 111–22).
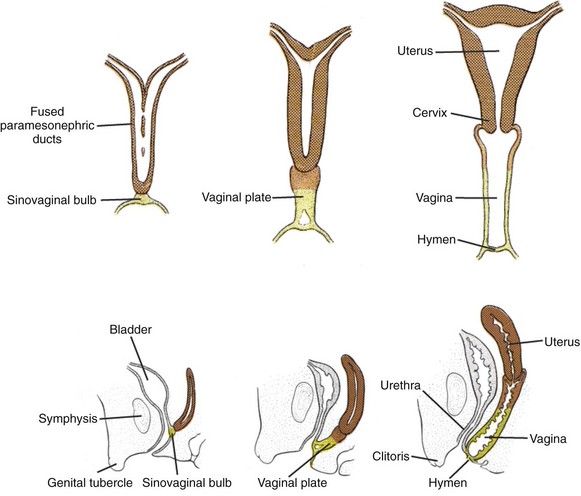
Figure 111–22 Development of uterus and vagina. During the 10th week the paramesonephric ducts fuse at their caudal ends to establish a common channel and come into contact with a thickened portion of the posterior urogenital sinus called the sinovaginal bulb. This is followed by development of the vaginal plate, which elongates between the third to fifth months and becomes canalized to form the inferior vaginal lumen.
(Modified from Sadler TW. Langman’s medical embryology. Baltimore: Williams & Wilkins, 1985.)
As the vaginal plate forms, the lower end of the vagina lengthens, and its junction with the urogenital sinus migrates caudally until it comes to rest on the posterior wall of definitive urogenital sinus (future vestibule of the vagina) during the fourth month. An endodermal membrane temporarily separates the vaginal lumen from the cavity of the definitive urogenital sinus. This barrier degenerates partially after the fifth month, but its remnant persists as the vaginal hymen. The mucous membrane that lines the vagina and cervix may also derive from the endodermal epithelium of the definitive urogenital sinus.
Development of External Genitalia
Unlike the rest of the developing embryo the cloacal membrane, along with the oropharyngeal membrane (future oral cavity), is a bilayered structure, in which the outer ectoderm remains in close contact with the underlying endoderm without the intervening mesoderm. Initially, the cloacal membrane represents an elongated midline structure, which extends from the root of the umbilical cord to the future site of perineum distally. During the subsequent development this bilayered cloacal membrane “retracts” into the perineum as a result of cranial and medial migration of mesodermal cells into the anterior body wall between the ectoderm and the endoderm layers of the cloacal membrane. This mesenchymal migration brings about the closure of the inferior part of the anterior abdominal wall and causes the caudal portion of the cloacal membrane to position itself in the perineal region. These migrating mesodermal cells give rise to the musculature of the medial portion of the anterior abdominal wall, the mesenchymal portion of the anterior bladder wall, the pubic symphysis, and the rudiments of the external genitalia (Vermeij-Keers et al, 1996). Failure of migration of these mesodermal cells into the midline results in bladder exstrophy and other associated genital defects (Langer, 1993; Vermeij-Keers et al, 1996).
The early development of the external genitalia is similar in both sexes. Migrating mesenchymal cells spread themselves around the cloacal membrane and pile up to form swellings. Early in the fifth week, a pair of swellings called cloacal folds develops on either side of the cloacal membrane. These folds meet just anterior to the cloacal membrane to form a midline swelling called the genital tubercle (Fig. 111–23). During the cloacal division into the anterior urogenital sinus and the posterior anorectal canal the portion of the cloacal folds flanking the opening of the urogenital sinus becomes the urogenital folds and the portion flanking the opening of the anorectal canal becomes the anal folds. A new pair of swellings, called the labioscrotal folds, appears on either side of the urogenital folds.
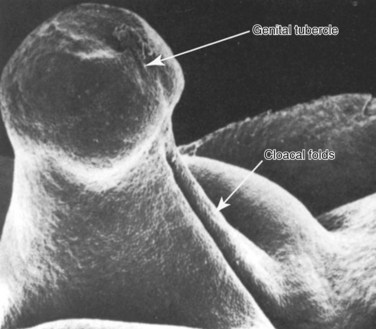
Figure 111–23 The early stages of cloacal fold development.
(Reproduced from Hamilton WJ, Mossman HW. Human embryology prenatal development of form and function. New York: Macmillan; 1976; and from Waterman RE. Human embryo and fetus. In: Hafez ESE, Kenemans P, editors. Atlas of human reproduction. Hinghman [MA]: Kluwer Boston; 1982.)
The most popular hypothesis of external genital and urethral development is based on work performed in the early part of the 20th century. Most embryology texts today quote the mechanism of urethral development proposed by Glenister (1954). As the genital tubercle elongates in males, a groove appears on its ventral aspect (called the urethral groove) during the sixth week. In both sexes an ectodermal epithelial tag is present at the tip of the genital tubercle. The urethral groove is defined laterally by urethral folds, which are continuations of the previous urogenital folds surrounding the urogenital membrane. Initially, the urethral groove extends only part of the way distally along the shaft of the elongating genital tubercle. The distal portion of the urethral groove terminates in a solid epithelial plate called the urethral plate that extends into the glans penis. The solid urethral plate canalizes and thus extends the urethral groove distally toward the glans. The urethral groove is thought to be lined by endoderm. Likewise, the solid urethral plate, the distal precursor of the urethral groove, is also believed to derive from the endodermal source. Clearly, fusion of the urethral folds is the key step in the formation of penile urethra. A prerequisite of urethral fold fusion is the canalization of solid urethral plate and formation of the urethral groove bounded on each side by the urethral folds. If the urethral groove and urethral fold formations are abnormal, then the urethral fold fusion is likely to be impaired as well (Figs. 111-24 and 111-25).
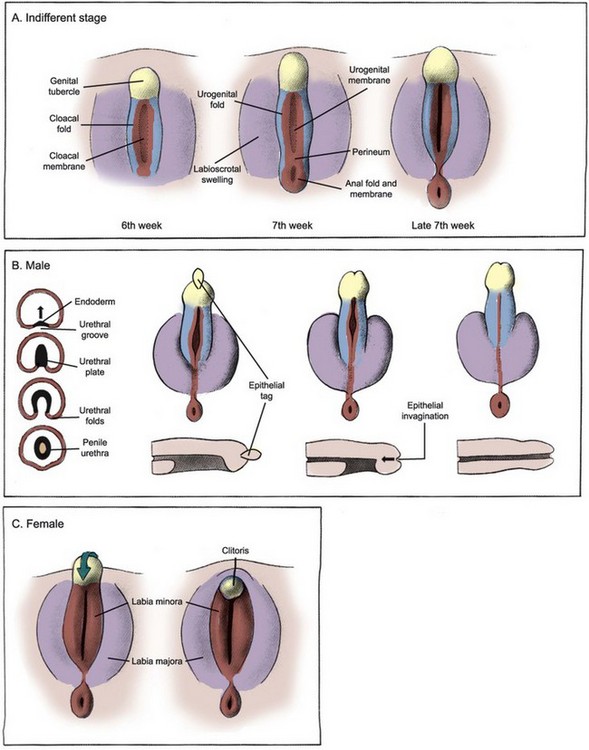
Figure 111–24 Development of external genitalia in male and female. A, The external genitalia derive from a pair of labioscrotal swellings, a pair of urogenital folds, and an anterior genital tubercle. Male and female genitalia are morphologically indistinguishable until the seventh week. B, In males, the urogenital folds fuse and the genital tubercle elongates to form the penile shaft and glans. A small region of the distal urethra in the glans is formed by the invagination of surface epithelial tag. The fused labioscrotal folds give rise to the scrotum. C, In females the genital tubercle bends inferiorly to form the clitoris and the urogenital folds remain separate to become the labia minora. The unfused labioscrotal folds form the labia majora.
(Modified from Larsen WJ. Human embryology. New York: Churchill Livingstone; 1997.)
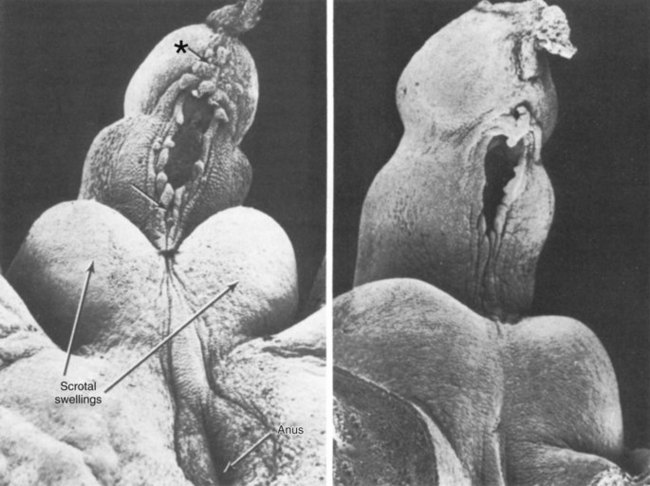
Figure 111–25 The epithelial tag (asterisk) and fusing urethral folds (arrows) in the developing male external genitalia.
(Reproduced from Waterman RE. Human embryo and fetus. In: Hafez ESE, Kenemans P, editors. Atlas of human reproduction. Hinghman [MA]: Kluwer Boston; 1982.)
The formation of the distal glanular urethra may occur by a combination of two separate processes—the fusion of urethral folds proximally and the ingrowth of ectodermal cells distally. It is generally thought that the stratified squamous epithelium of the fossa navicularis results from an ingrowth of surface ectoderm as far proximally as the valve of Guérin. The lacuna magna (also known as the sinus of Guérin), which can give symptoms of hematuria and dysuria in some boys, may form as a result of dorsal extension of this ectodermal ingrowth. It was suggested recently that the entire penile urethra might differentiate from the fusion of the endodermal urethral groove via the mechanism of epithelial-mesenchymal interactions (Kurzrock et al, 1999).
The molecular basis of the sexual dimorphism in genital development is based on the presence or absence of the signaling via the androgen receptor. As the fetal testis produces testosterone, cells within the fetal external genitalia express the enzyme 5α-reductase, which converts testosterone to DHT. Androgen receptors are present in cells of the developing external genitalia and prominently expressed in the genital tubercle mesenchymal cells. The action of DHT acting via androgen receptors masculinizes the developing external genitalia. The key role of androgen in sexually dimorphic development of the external genitalia has been corroborated through many experimental studies. In-utero exposure of rodents to antiandrogenic compounds reduces the size of the genital tubercle and prevents the development of the scrotum. Likewise, in-utero exposure of rats to 5α-reductase inhibitors leads to the development of hypospadias. Mice and humans with functional loss of androgen receptors via mutations demonstrate a complete feminization of the external genitalia.
The elongating phallus is covered externally by ectoderm that gives rise to the penile skin, whereas most of the substance of the penis is derived from mesodermal cells forming the corporeal bodies, connective tissue, and dermis. Corporeal tissue is first recognized as distinct dense mesenchymal condensations within the shaft of the developing penis. Little is known regarding the molecular regulatory mechanisms of the differentiation of penile mesenchyme into its various derivatives, but it is likely that this process is dependent on epithelial-mesenchymal interactions.
In the female, owing to absence of androgen receptor signaling via DHT, the primitive perineum does not lengthen and the labioscrotal and urethral folds do not fuse across the midline. The phallus bends inferiorly, becoming the clitoris, and the ostium of the urogenital membrane becomes the vestibule of the vagina. The urethral folds become the labia minora, and the labioscrotal folds become the labia majora. The external genitalia develop in a similar manner in genetic males who are deficient in 5α-reductase and therefore lack DHT.
Gonadal Descent
Morphologically, the human urogenital ridge is identical in both sexes at 7 to 8 weeks of gestation. Before gonadal differentiation, the testis lies near the developing kidney, loosely held in place by two ligamentous structures. The dorsal ligament is referred to as the cranial suspensory ligament (CSL), whereas the ventral ligament later develops into the gubernaculum (Fig. 111–26). Between 10 and 15 weeks, the testis remains close to the future inguinal region during the enlargement of the abdominal cavity while the ovary moves more cranially. The testis is anchored near the inguinal region by enlargement of the gubernaculum and regression of the CSL. As early as the 1700s, enlargement of the gubernaculum in males was observed to tether the testis near the groin while the kidney migrated cranially (Wyndham, 1943; van der Schoot, 1993). In females, the CSL continues to develop, keeping the ovary close to the kidney while the gubernaculum involutes. In males, androgen induces resorption of the CSL while the gubernaculum enlarges to become a plump ligamentous body, “holding” the testis close to the inguinal region. Starting in the seventh month, the gubernaculum begins to bulge beyond the external inguinal ring and descends to the scrotal location, while simultaneously it is hollowed out by the evaginating peritoneal diverticulum called the processus vaginalis (Heyns, 1987). The processus vaginalis allows the intra-abdominal testis to exit the abdominal cavity. The bulky distal end of the gubernaculum (known as the bulb) is resorbed in humans after completion of inguinoscrotal migration.
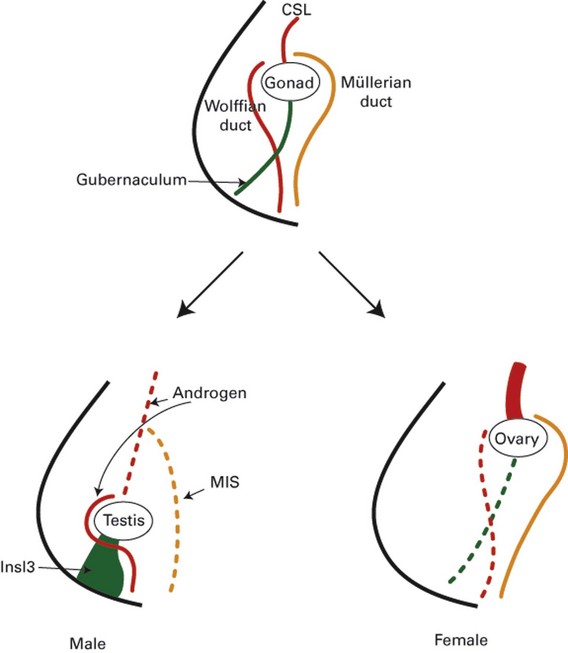
Figure 111–26 Mechanism of gonadal descent. The undifferentiated gonad is initially located high in the abdomen, anchored by the cranial suspensory ligament (CSL). In males, Insl3 will cause the swelling and enlargement of gubernaculum to pull the developing testis toward the inguinal region, whereas androgens will cause an involution of CSL. Owing to the action of müllerian-inhibiting substance (MIS), müllerian ducts will regress while androgens continue to stimulate the development of wolffian ducts into male genital ductal structures. In females, CSL persists due to the absence of androgens and the gubernaculum remains thin due to the absence of Insl3 activity, thereby keeping the ovary well within the pelvis.
Caudal enlargement of the gubernaculum during the early relative transabdominal movement of the testis is known as the “swelling reaction” or “gubernacular outgrowth.” The proximal gubernacular cord appears to shorten during this process, as it becomes incorporated into the enlarging bulb (Wensing, 1986). Shortening of the cord may be an important mechanism to position the testis over the inguinal ring to permit abdominal pressure to push the testis out of the abdomen (Quinlan et al, 1988; Attah and Hutson, 1993; Husmann and Levy, 1995). Transection of the gubernacular cord can lead to either accidental testicular descent into the contralateral inguinal canal or aberrant intra-abdominal location (Frey and Rajfer, 1984; Beasley and Hutson, 1988; Attah and Hutson, 1993).
Although intra-abdominal pressure may not be a factor during the initial transabdominal descent, it is thought to be important during transit through the inguinal canal and the subsequent scrotal migration. Inguinoscrotal descent requires migration of the gubernaculum over a considerable distance, along with an increase in the length of the processus vaginalis. The force for movement may come from the intra-abdominal pressure, transmitted directly and indirectly to the testis via the lumen of the processus vaginalis and the gubernacular cord, respectively.
Although patients with defective androgen production or metabolism show varied manifestations of cryptorchidism, the exact role of androgen in testicular descent still remains unclear. During intra-abdominal testicular descent, androgen appears to play a role in the regression of the CSL (van der Schoot, 1992). Gubernacular enlargement, in contrast, seems to occur independent of androgen activity, because it occurs in androgen-resistant mice and humans normally, being able to keep the testis close to the inguinal region (Hutson, 1985). The second migratory step—the inguinoscrotal phase—is thought to be more androgen dependent. Migration of the gubernaculum beyond the inguinal region is absent in gonadotropin-deficient mice (Grocock et al, 1988) and those with complete androgen resistance (Hutson, 1986). Regression of the gubernacular bulb after the completion of scrotal descent also appears to be androgen dependent because in humans with androgen resistance the gubernaculum remains enlarged (Hutson, 1986).
Müllerian-inhibiting substance is a glycoprotein produced and secreted by Sertoli cells and is responsible for regression of the müllerian ducts (Josso et al, 1993; Lee and Donahoe, 1993). Evidence for the role of MIS in testicular descent is conflicting. Some clinical observations support the role of MIS, including patients with persistent müllerian duct syndrome caused by genetic defects of MIS or its receptor gene (Josso et al, 1983). In this clinical scenario the testes are undescended and the gubernaculum is thin and elongated. Transgenic mice with MIS deficiency show a variable testicular position depending on their androgenic status: those with normal androgen receptors have normally descended testes whereas those with androgen resistance have completely undescended testes (Behringer et al, 1994). A recent study on MIS-receptor knockout mice, however, failed to show any defect in gubernacular development and testicular positions (Bartlett et al, 2002).
INSL3 was identified as a novel gene product of the Leydig cells in 1993 (Adham et al, 1993). INSL3 is similar in structure to the peptide hormones relaxin or insulin and is expressed in both fetal and adult Leydig cells in a differentiation-dependent manner (Balvers et al, 1998). Mice lacking a functional Insl3 gene demonstrate intra-abdominal cryptorchidism but otherwise no obvious defects in other male reproductive organs. More importantly, early surgical correction of the cryptorchidism in these mice can restore normal fertility potential (Nef and Parada, 1999; Zimmermann et al, 1999). These are important findings because they reflect the phenotype most commonly observed in classic cryptorchidism in humans. Recently, a G protein–coupled receptor—LGR8—has been cloned with binding and functional response to INSL3 in transfected cells (Hsu et al, 2002; Kumagai et al, 2002). Moreover, mutations in this receptor in mice can lead to the development of cryptorchidism and have been linked to a case of cryptorchidism in the human (Overbeek et al, 2001; Gorlov et al, 2002). Treatment of rat gubernacular explant with exogenous INSL3 leads to a rapid growth of the ligament, an effect that synergizes with androgen treatment (Kubota et al, 2002). It was also demonstrated that LGR8 was expressed in the rat gubernaculum (Kubota et al, 2002). Although INSL3 appears to be a good candidate for a responsible gene for cryptorchidism, to date no causative mutations have been identified in the human INSL3 gene. Furthermore, because INSL3 is expressed in a differentiated testis, any factor that influences Leydig cell differentiation may also affect INSL3 expression and thereby cause cryptorchidism.
Treatment of pregnant mothers with diethylstilbestrol as a hormonal support during pregnancy was abandoned owing to a high rate of cryptorchidism and other genital defects (Stillman, 1982). The effect of environmental xenoestrogenic compounds has also been linked to the recent rise in cryptorchidism in humans (Toppari and Skakkebaek, 1998). In one study, mice were treated with diethylstilbestrol to induce cryptorchidism in male neonates (Emmen et al, 2000; Nef et al, 2000). Interestingly, the treated animals demonstrated a complete suppression of testicular Insl3 expression on embryonic days 16 and 18.
The male knockout mice for the transcription factor Hoxa-10 gene are viable but infertile. While they are normally virilized, they are bilaterally cryptorchid with severely underdeveloped gubernaculum. Fetal localization studies have shown that Hoxa-10 is expressed in the gubernaculum, as well as in the kidneys, but not in other reproductive tissues. Although its function and role are not yet established, it appears to be another candidate regulatory gene for gubernacular development and testicular descent.
The spinal nucleus of the genitofemoral nerve (GFN) is located at L1-2 in the spinal cord and is sexually dimorphic (Goh et al, 1994). Transection of GFN produced cryptorchidism (Lewis, 1948), and the initial thought—now proven faulty—was that the cremasteric muscle paralysis caused by denervation led to an abnormal traction of the testis through the inguinal canal. When this observation was revisited many years later it was speculated that androgens may act via the GFN (Beasley and Hutson, 1987). The GFN innervates the gubernaculum from its posterior and caudal surface, so that distal transection would cause denervation of the gubernaculum (Tayakkanonta, 1963). Additional supporting evidence for the role of the GFN comes from the analysis of patients with spina bifida and animals with spinal cord transection (Hutson et al, 1988). In more than 300 boys with spina bifida, 23% had cryptorchidism, with a higher incidence found in those whose defect was higher than L4. In rats with neonatal spinal cord transection, approximately 40% had cryptorchidism when the lesions were midlumbar. Anatomic studies of the GFN identified calcitonin gene–related peptide (CGRP) as the primary neurotransmitter (Goh et al, 1994). The effect of CGRP on the rodent gubernaculum has been studied extensively. In male neonatal rats under anesthesia the gubernaculum, which has not yet reached the scrotum, contracts rhythmically, and this is enhanced by increased intra-abdominal pressure and direct application of exogenous CGRP (Park and Hutson, 1991). In organ culture, neonatal rat gubernaculum responds to CGRP in a dose-dependent manner but not to other neuropeptides (Park and Hutson, 1991). Although these findings are raising a strong speculation for the role of CGRP, its significance in human testicular descent remains uncertain.
The ovaries also descend and become suspended within the broad ligaments of the uterus. As in males, the female embryos develop a gubernaculum-like structure extending initially from the inferior pole of the gonad to the subcutaneous fascia of the presumptive labioscrotal folds. This “female gubernaculum” later penetrates the abdominal wall as part of a fully formed inguinal canal and becomes the round ligament. In females, although the gubernaculum does not shorten like that in males, it still causes the ovaries to descend during the third gestational month (by anchoring the ovaries in the pelvis) and places them into a peritoneal fold (the broad ligament of the uterus). This translocation of ovaries appears to occur during the seventh week when the gubernaculum becomes attached to the developing paramesonephric (müllerian) ducts. As the paramesonephric ducts fuse together in their caudal ends they sweep out the broad ligaments and simultaneously pull the ovaries into these peritoneal folds. In the absence of androgens, the female gubernaculum remains intact and grows in step with the rest of the body. The inferior gubernaculum becomes the round ligament of the uterus and attaches the fascia of the labia majora to the uterus, while the superior gubernaculum becomes the ligament of the ovary, connecting the uterus to the ovary. As in males, the processus vaginalis of the inguinal canal is normally obliterated, but occasionally it remains patent to become an indirect inguinal hernia.
Molecular Mechanism of Sex Development
Mammalian sex development involves a complex interplay of multiple cell types, which occurs in a narrow window of time. Thus it is important to understand the temporal and spatial pattern of gene expression as well as the anatomic sequence of tissue movement and differentiation. In both sexes, before the expression of the male-determining gene SRY, a number of factors appear to play a role in urogenital ridge specification (Fig. 111–27). Because the urogenital ridge is the primordium for gonad, kidney, and reproductive tract, multiple organs are affected simultaneously by mutations of these genes.
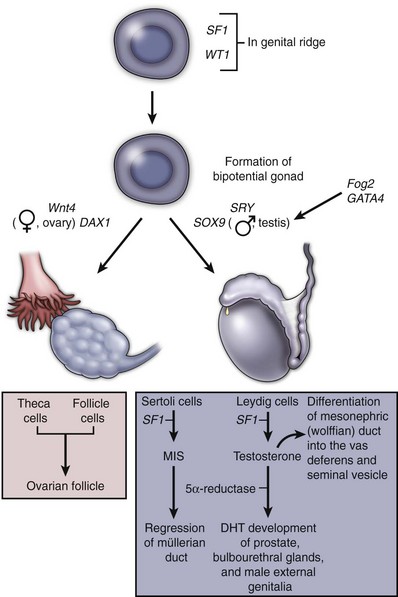
Figure 111–27 Molecular mechanism of male and female genital development. SF1 and WT1 expression is critical for genital ridge specification. SRY and SOX9, influenced by GATA4 and Fog2, are important factors for specifying the differentiation of Sertoli cells. SF1 is also critical in the regulation of müllerian-inhibiting substance (MIS) and other genes involved in androgen synthesis. No specific female factors have been identified, but Wnt4 and DAX1 are expressed with unique female patterns. DNT, dihydrotestosterone.
In the mouse gonad, Wt1 is expressed early, suggesting a role in specifying coelomic epithelial cells in the development of urogenital ridge. The Wt1 homozygous knockout mice do not form kidney, adrenal, or gonad cells (Kreidberg et al, 1993). Humans heterozygous for mutations in WT1 exhibit abnormalities of the genital system in addition to abnormalities in renal development, including WAGR syndrome, Denys-Drasch syndrome, and Frasier syndrome. WT1 appears to function upstream of SF1 (steroidogenic factor 1) and DAX1 (dosage-sensitive sex reversal, adrenal hypoplasia congenita, X chromosome) (Wilhelm and Englert, 2002). Wt1 and Sf1 enhance transcription of mouse MIS gene, whereas Dax1 appears to suppress this interaction (Nachtigal et al, 1998). SF1 also regulates the expression of other genes involved in male differentiation, steroidogenesis, and reproduction (Achermann et al, 2001). Although SF1 stimulates DAX1 transcription (Ikeda et al, 1996), DAX1 in turn acts as a transcriptional repressor of SF1-regulated genes (Ito et al, 1997). In Sf1 knockout mice, neither XX nor XY animals form gonads, and cells in gonadal remnant undergo apoptosis, suggesting that SF1 is a necessary survival factor for early progenitors of a developing gonad (Luo et al, 1994). Data suggest that SF1 and DAX1 are both independently important for normal male gonadal differentiation. Other candidate genes have also emerged based on mouse gene deletion models and embryonic expression studies, although their functional significance in gonadal development is not fully elucidated yet, and they include M33, Lhx9, Pod1, Dmrt1, Mro, Pn1, and Vn1 (Park and Jameson, 2005).
Mammalian embryos remain sexually undifferentiated until the time of sex determination. When the Y-linked master regulatory gene, called SRY (the sex-determining region of the Y chromosome), is expressed in the male, the epithelial cells of the primitive sex cords differentiate into Sertoli cells, and this critical morphogenetic event triggers subsequent testicular development. Once the testes are established they produce androgens to give rise to the male phenotype. In the female gonads no morphologic change is observable at the time of SRY expression. It follows from this general picture that in mammals, sex determination is synonymous with testicular development, with the differentiation of Sertoli cells being the key event (McLaren, 1991). After 3 decades of search for the elusive mammalian testis-determining gene, the SRY gene was discovered in 1990 by Sinclair and colleagues. Since then, research has focused on identifying the putative regulatory mechanism operating downstream of SRY and the genetic control of SRY expression.
Although it has been known since 1921 that human males have an X and Y chromosome, the role of these “sex” chromosomes in human sex development was not elucidated until 1959. This question was answered by the examination of two individuals with unique chromosome abnormalities: one female with Turner syndrome (45,XO karyotype) and one male with Klinefelter syndrome (47,XXY karyotype). By 1966, analysis of many structurally aberrant Y chromosomes in humans led to the conclusion that the information necessary to initiate the male phenotypic development was present on the short arm of the Y chromosome. The identity of the protein encoded by the testis-determining region of the Y chromosome proved elusive. In the mid 1980s, the DNA of sex-reversed males with 46,XX karyotype was examined. The genome in these individuals was found to contain small amounts of Y chromosome that had been translocated onto the X chromosome. Analysis of this DNA narrowed the location of the SRY to a relatively small region within the short arm of the chromosome. The role of SRY in human sex development has been further supported by studies using mice (Greenfield and Koopman, 1996). The comparable genetic locus in mice (Sry) is activated and expressed in the genital ridge 11.5 days after coitus, just before the initiation of testicular development. Moreover, when the DNA of female XY mouse chromosomes was analyzed with specific DNA probes for Sry, this locus was absent. Most importantly, it has been demonstrated that insertion of Sry into one of the X chromosomes of genetically female mouse embryos converted these mice to phenotypic males (Koopman et al, 1991). These transgenic “female” mice exhibited testes, vas deferens, and an absence of the female reproductive tract. It was thought that identification of the SRY protein would rapidly lead to the identity of downstream elements regulating male sexual development. However, the binding of SRY protein to other genes or factors has not been demonstrated, and the molecular mechanism by which genes interact to determine sex remains speculative. Deletions of the SRY gene in humans cause XY male-to-female sex reversal, whereas SRY translocations to the X chromosome lead to XX female-to-male sex reversal (Harley et al, 2003). In the mouse, Sry expression occurs in a narrow temporal window in the developing Sertoli cells. The central region of the gonad demonstrates Sry expression first, followed by cephalad to caudal progression along the entire length of the gonad (Bullejos and Koopman, 2001). A closely related gene, SOX9, is the other definitive male-determining gene identified thus far. SRY and SOX9 expression overlap in cells of the Sertoli lineage. As the SRY expression diminishes, SOX9 expression increases in the male. SOX9, which is weakly expressed in the undifferentiated gonad, is downregulated in the female (Sekido et al, 2004). Transgenic expression of Sox9 in XX mice is sufficient to induce female-to-male sex reversal and male differentiation (Vidal et al, 2001). Heterozygous human SOX9 mutation leads to campomelic dysplasia, a severe skeletal disorder with defective cartilage development, and many of these patients have dysgenetic gonads (Foster et al, 1994). These individuals possess a normal SRY but may exhibit completely feminized genital structures. Sox9 knockout mouse embryos have elevated levels of Sry expression, suggesting a possible negative feedback regulatory loop that downregulates Sry (Chaboissier et al, 2004). GATA4 and Fog2 are important in cardiac development but also appear to affect gonadal development as well. GATA4 mutation eliminates expression of male differentiation markers Sox9 and MIS. Fog2 knockout mice have decreased expression of Sry, loss of Sox9, MIS, and desert hedgehog, but persistence of female gonad marker Wnt4 (Tevosian et al, 2002). Similarly, triple mutants for insulin receptor, insulin-related receptor, and Igf-1 receptor have low Sry and Sox9 expression and exhibit male-to-female sex reversal, implicating a role for an insulin signaling pathway (Nef et al, 2003).
Proliferation of Sertoli progenitor cells is an important event in male gonad development, a process driven by SRY expression (Schmahl et al, 2000). One paracrine factor linked to this proliferation is FGF9. Fgf9 knockout mice demonstrate varying degrees of male-to-female sex reversal (Colvin et al, 2001). In addition, FGF9 is a candidate male gonad-specific chemoattractant paracrine signal that induces migration of cells from the mesonephros (endothelial and peritubular myoid cells) into the gonad (Martineau et al, 1997). Migration of these cells is critical for the development of testis cord formation (Buehr et al, 1993) as well as SOX9 expression (Tilmann and Capel, 1999). Such migration is absent in females, presumably owing to absence of chemoattractants.
Once sex determination has occurred the subsequent phenotypic differentiation depends mostly on the production of androgens. As bipotential gonad differentiates into testis, Sf1 expression becomes restricted to Leydig cells and mediates expression of several gene-encoding enzymes that are required for testosterone biosynthesis, including StAR, Cyp11a1, Cyp17, and 3βHSD. Factors for Leydig cell determination are not known, and, moreover, Leydig cell origin, whether from immigrant mesonephric cells or progenitors within the gonad, remain speculative. There is evidence that Leydig cell fate is dependent on paracrine signals (Yao et al, 2002; Brennan et al, 2003).
Relatively few genes have been shown to exhibit a female-specific pattern of gene expression early in gonadal development. Thus far, no female-determining gene has been identified. The DAX1 gene was initially suggested as a pro-ovarian (or antitestis) candidate gene because its duplication on an XY background was associated with impaired testicular development (Bardoni et al, 1994; Swain et al, 1998). However, loss of DAX1 in XX background does not prevent ovary development (Yu et al, 1998). A growing list of genes demonstrates an ovary-specific expression patterns, including Fst and Stra8 (Park and Jameson, 2005). There has been a postulate for a “Z-factor” that suppresses protesticular events in both XX and XY backgrounds (McElreavey et al, 1993). According to this hypothesis, the Z-factor, which normally suppresses testicular determination, is repressed by SRY in males, and in females, owing to absence of SRY, it will inhibit testicular development. Loss of Z-factor in XX background will result in female-to-male sex reversal, but gain-of-function in XY background may or may not result in male-to-female sex reversal, depending on whether the Z-factor can override the suppression effect of SRY-driven signaling. One candidate for such Z-factor is Wnt4. XX mice deficient for Wnt4 develop testis-like differentiation and nephric (wolffian) duct derivative (Vainio et al, 1999). Curiously, their external genitalia remain female. Furthermore, Wnt4 is downregulated in males whereas its expression remains strong in females (Yao et al, 2004).
Key Points
Baskin LS, Hayward SW, Young P, et al. Role of mesenchymal-epithelial interactions in normal bladder development. J Urol. 1996;156(5):1820-1827.
Dressler GR. Tubulogenesis in the developing mammalian kidney. Trends Cell Biol. 2002;12(8):390-395.
Hutson JM, Hasthorpe S, Heyns CF. Anatomical and functional aspects of testicular descent and cryptorchidism. Endocr Rev. 1997;18(2):259-280.
Ichikawa I, Kuwayama F, Pope JCIV, et al. Paradigm shift from classic anatomic theories to contemporary cell biological views of CAKUT. Kidney Int. 2002;61:889-898.
Marker PC, Donjacour AA, Dahiya R, et al. Hormonal, cellular, and molecular control of prostatic development. Dev Biol. 2003;253:165-174.
Park SY, Jameson JL. Transcriptional regulation of gonadal development and differentiation. Endocrinology. 2005;146(3):1035-1042.
Abdelhak S, Kalatzis V, et al. A human homologue of the Drosophila eyes absent gene underlies branchio-oto-renal (BOR) syndrome and identifies a novel gene family. Nat Genet. 1997;15(2):157-164.
Abrahamson DR, Robert B, et al. Origins and formation of microvasculature in the developing kidney. Kidney Int Suppl. 1998;67:S7-11.
Achermann JC, Meeks JJ, et al. Phenotypic spectrum of mutations in DAX-1 and SF-1. Mol Cell Endocrinol. 2001;185(1-2):17-25.
Adham IM, Burkhardt E, et al. Cloning of a cDNA for a novel insulin-like peptide of the testicular Leydig cells. J Biol Chem. 1993;268(35):26668-26672.
Alcaraz A, Vinaixa F, et al. Obstruction and recanalization of the ureter during embryonic development. J Urol. 1991;145(2):410-416.
Andersson S, Berman DM, et al. Deletion of steroid 5 alpha-reductase 2 gene in male pseudohermaphroditism. Nature. 1991;354(6349):159-161.
Attah AA, Hutson JM. The role of intra-abdominal pressure in cryptorchidism. J Urol. 1993;150:994-996.
Baker LA, Gomez RA. Embryonic development of the ureter and bladder: acquisition of smooth muscle. J Urol. 1998;160(2):545-550.
Balvers M, Spiess AN, et al. Relaxin-like factor expression as a marker of differentiation in the mouse testis and ovary. Endocrinology. 1998;139(6):2960-2970.
Bardoni B, Zanaria E, et al. A dosage sensitive locus at chromosome Xp21 is involved in male to female sex reversal. Nat Genet. 1994;7(4):497-501.
Bartlett JE, Lee SM, et al. Gubernacular development in mullerian inhibiting substance receptor-deficient mice. BJU Int. 2002;89(1):113-118.
Baskin L, Meaney D, et al. Bovine bladder compliance increases with normal fetal development. J Urol. 1994;152(2 Pt 2):692-695. discussion 696–7
Baskin LS, Hayward SW, et al. Role of mesenchymal-epithelial interactions in normal bladder development. J Urol. 1996;156(5):1820-1827.
Basson MA, Watson-Johnson J, et al. Branching morphogenesis of the ureteric epithelium during kidney development is coordinated by the opposing functions of GDNF and Sprouty1. Dev Biol. 2006;299(2):466-477.
Batourina E, Gim S, et al. Vitamin A controls epithelial/mesenchymal interactions through Ret expression. Nat Genet. 2001;27(1):74-78.
Batourina E, Tsai S, et al. Apoptosis induced by vitamin A signaling is crucial for connecting the ureters to the bladder. Nat Genet. 2005;37(10):1082-1089.
Beasley SW, Hutson JM. Effect of division of genitofemoral nerve on testicular descent in the rat. Aust NZ J Surg. 1987;57(1):49-51.
Beasley SW, Hutson JM. The role of the gubernaculum in testicular descent. J Urol. 1988;140:1191-1193.
Beauboeuf A, Ordille S, et al. In vitro ligation of ureters and urethra modulates fetal mouse bladder explants development. Tissue Cell. 1998;30(5):531-536.
Behringer RR, Finegold MJ, et al. Mullerian-inhibiting substance function during mammalian sexual development. Cell. 1994;79(3):415-425.
Bhatia-Gaur R, Donjacour AA, et al. Roles for Nkx3.1 in prostate development and cancer. Genes Dev. 1999;13(8):966-977.
Bourdelat D, Barbet JP, et al. Fetal development of the urethral sphincter. Eur J Pediatr Surg. 1992;2(1):35-38.
Brennan J, Tilmann C, et al. PDGFR-alpha mediates testis cord organization and fetal Leydig cell development in the XY gonad. Genes Dev. 2003;17(6):800-810.
Brenner-Anantharam A, Cebrian C, et al. Tailbud-derived mesenchyme promotes urinary tract segmentation via BMP4 signaling. Development. 2007;134(10):1967-1975.
Brophy PD, Ostrom L, et al. Regulation of ureteric bud outgrowth by Pax2-dependent activation of the glial derived neurotrophic factor gene. Development. 2001;128(23):4747-4756.
Buehr M, Gu S, et al. Mesonephric contribution to testis differentiation in the fetal mouse. Development. 1993;117(1):273-281.
Bullejos M, Koopman P. Spatially dynamic expression of Sry in mouse genital ridges. Dev Dynam. 2001;221(2):201-205.
Cacalano G, Farinas I, et al. GFRalpha1 is an essential receptor component for GDNF in the developing nervous system and kidney. Neuron. 1998;21(1):53-62.
Cancilla B, Jarred RA, et al. Regulation of prostate branching morphogenesis by activin A and follistatin. Dev Biol. 2001;237(1):145-158.
Cebrian C, Borodo K, et al. Morphometric index of the developing murine kidney. Dev Dynam. 2004;231(3):601-608.
Chaboissier MC, Kobayashi A, et al. Functional analysis of Sox8 and Sox9 during sex determination in the mouse. Development. 2004;131(9):1891-1901.
Colvin JS, Green RP, et al. Male-to-female sex reversal in mice lacking fibroblast growth factor 9. Cell. 2001;104(6):875-889.
Coplen DE, Macarak EJ, et al. Developmental changes in normal fetal bovine whole bladder physiology. J Urol. 1994;151(5):1391-1395.
Cunha GR, Donjacour AA, et al. The endocrinology and developmental biology of the prostate. Endocr Rev. 1987;8(3):338-362.
Cunha GR, Lung B. The possible influence of temporal factors in androgenic responsiveness of urogenital tissue recombinants from wild-type and androgen-insensitive (Tfm) mice. J Exp Zool. 1978;205(2):181-193.
Dahl U, Sjodin A, et al. Genetic dissection of cadherin function during nephrogenesis. Mol Cell Biol. 2002;22(5):1474-1487.
Davies J. Intracellular and extracellular regulation of ureteric bud morphogenesis. J Anat. 2001;198(Pt 3):257-264.
Deslypere JP, Young M, et al. Testosterone and 5 alpha-dihydrotestosterone interact differently with the androgen receptor to enhance transcription of the MMTV-CAT reporter gene. Mol Cell Endocrinol. 1992;88(1-3):15-22.
Dressler GR, Douglass EC. Pax-2 is a DNA-binding protein expressed in embryonic kidney and Wilms tumor. Proceedings of the National Academy of Sciences of the United States of America. 1992;89(4):1179-1183.
Dudley AT, Godin RE, et al. Interaction between FGF and BMP signaling pathways regulates development of metanephric mesenchyme. Genes Dev. 1999;13(12):1601-1613.
Elfman F, Bok R, et al. Urokinase plasminogen activator amino-terminal peptides inhibit development of the rat ventral prostate. Differentiation. 2001;69(2-3):108-120.
Emmen JM, McLuskey A, et al. Involvement of insulin-like factor 3 (Insl3) in diethylstilbestrol-induced cryptorchidism. Endocrinology. 2000;141(2):846-849.
Escala JM, Keating MA, et al. Development of elastic fibres in the upper urinary tract. J Urol. 1989;141(4):969-973.
Foster JW, Dominguez-Steglich MA, et al. Campomelic dysplasia and autosomal sex reversal caused by mutations in an SRY-related gene. Nature. 1994;372(6506):525-530.
Frey HL, Rajfer J. Role of gubernaculum and intraabdominal pressure in the process of testicular descent. J Urol. 1984;131:574-579.
Gakunga P, Frost G, et al. Hyaluronan is a prerequisite for ductal branching morphogenesis. Development. 1997;124(20):3987-3997.
Glenister TW. The origin and fate of the urethral plate in man. J Anat. 1954;88:413-424.
Goh DW, Middlesworth W, et al. Prenatal androgen blockade with flutamide inhibits masculinization of the genitofemoral nerve and testicular descent. J Pediatr Surg. 1994;29(6):836-838.
Gorlov IP, Kamat A, et al. Mutations of the GREAT gene cause cryptorchidism. Hum Mol Genet. 2002;11(19):2309-2318.
Greenfield A, Koopman P. SRY and mammalian sex determination. Curr Topics Dev Biol. 1996;34:1-23.
Grieshammer U, Le M, et al. SLIT2-mediated ROBO2 signaling restricts kidney induction to a single site. Dev Cell. 2004;6(5):709-717.
Grobstein C. Trans-filter induction of tubules in mouse metanephric mesenchyme. Exp Cell Res. 1956;10:424-440.
Grocock CA, Charlton HM, et al. Role of the fetal pituitary in cryptorchidism induced by exogenous maternal oestrogen during pregnancy in mice. J Reprod Fertil. 1988;83(1):295-300.
Guron G, Adams MA, et al. Neonatal angiotensin-converting enzyme inhibition in the rat induces persistent abnormalities in renal function and histology. Hypertension. 1997;29(1 Pt 1):91-97.
Harley VR, Clarkson MJ, et al. The molecular action and regulation of the testis-determining factors, SRY (sex-determining region on the Y chromosome) and SOX9 (SRY-related high-mobility group [HMG] box 9). Endocr Rev. 2003;24(4):466-487.
Hartwig S, Bridgewater D, et al. BMP receptor ALK3 controls collecting system development. J Am Soc Nephrol. 2008;19(1):117-124.
Hatini V, Huh SO, et al. Essential role of stromal mesenchyme in kidney morphogenesis revealed by targeted disruption of Winged Helix transcription factor BF-2. Genes Dev. 1996;10(12):1467-1478.
Hayward SW, Baskin LS, et al. Epithelial development in the rat ventral prostate, anterior prostate and seminal vesicle. Acta Anat. 1996;155(2):81-93.
Hayward SW, Haughney PC, et al. Interactions between adult human prostatic epithelium and rat urogenital sinus mesenchyme in a tissue recombination model. Differentiation. 1998;63(3):131-140.
He WW, Kumar MV, et al. A frame-shift mutation in the androgen receptor gene causes complete androgen insensitivity in the testicular-feminized mouse. Nucl Acids Res. 1991;19(9):2373-2378.
Herzlinger D, Koseki C, et al. Metanephric mesenchyme contains multipotent stem cells whose fate is restricted after induction. Development. 1992;114(3):565-572.
Herzlinger D, Qiao J, et al. Induction of kidney epithelial morphogenesis by cells expressing Wnt-1. Dev Biol. 1994;166(2):815-818.
Heyns CF. The gubernaculum during testicular descent in the human fetus. J Anat. 1987;153:93-112.
Hilgers KF, Reddi V, et al. Aberrant renal vascular morphology and renin expression in mutant mice lacking angiotensin-converting enzyme. Hypertension. 1997;29(1 Pt 2):216-221.
Hsu SY, Nakabayashi K, et al. Activation of orphan receptors by the hormone relaxin. Science. 2002;295(5555):671-674.
Husmann DA, Levy JB. Current concepts in the pathophysiology of testicular descent. Urology. 1995;46:267-276.
Hutson JM. A biphasic model for the hormonal control of testicular descent. Lancet. 1985;2:419-421.
Hutson JM. Testicular feminization: a model for testicular descent in mice and men. J Pediatr Surg. 1986;21:195-198.
Hutson JM, Beasley SW, et al. Cryptorchidism in spina bifida and spinal cord transection: a clue to the mechanism of transinguinal descent of the testis. J Pediatr Surg. 1988;23(3):275-277.
Ikeda Y, Swain A, et al. Steroidogenic factor 1 and Dax-1 colocalize in multiple cell lineages: potential links in endocrine development. Mol Endocrinol. 1996;10(10):1261-1272.
Ito M, Yu R, et al. DAX-1 inhibits SF-1-mediated transactivation via a carboxy-terminal domain that is deleted in adrenal hypoplasia congenita. Mol Cell Biol. 1997;17(3):1476-1483.
Josso N, Cate RL, et al. Anti-mullerian hormone: the Jost factor. Recent Prog Horm Res. 1993;48:1-59.
Josso N, Fekete C, et al. Persistence of mullerian ducts in male pseudohermaphroditism, and its relationship to cryptorchidism. Clin Endocrinol. 1983;19(2):247-258.
Karavanova ID, Dove LF, et al. Conditioned medium from a rat ureteric bud cell line in combination with bFGF induces complete differentiation of isolated metanephric mesenchyme. Development. 1996;122(12):4159-4167.
Kim KM, Kogan BA, et al. Collagen and elastin in the normal fetal bladder. J Urol. 1991;146(2 Pt 2):524-527.
Kispert A, Vainio S, et al. Proteoglycans are required for maintenance of Wnt-11 expression in the ureter tips. Development. 1996;122(11):3627-3637.
Kispert A, Vainio S, et al. Wnt-4 is a mesenchymal signal for epithelial transformation of metanephric mesenchyme in the developing kidney. Development. 1998;125(21):4225-4234.
Kluth D, Hillen M, et al. The principles of normal and abnormal hindgut development. J Pediatr Surg. 1995;30(8):1143-1147.
Koopman P, Gubbay J, et al. Male development of chromosomally female mice transgenic for Sry. Nature. 1991;351(6322):117-121.
Kreidberg JA, Sariola H, et al. WT-1 is required for early kidney development. Cell. 1993;74(4):679-691.
Kubota Y, Temelcos C, et al. The role of insulin 3, testosterone, mullerian inhibiting substance and relaxin in rat gubernacular growth. Mol Hum Reprod. 2002;8(10):900-905.
Kumagai J, Hsu SY, et al. INSL3/Leydig insulin-like peptide activates the LGR8 receptor important in testis descent. J Biol Chem. 2002;277(35):31283-31286.
Kume T, Deng K, et al. Murine forkhead/winged helix genes Foxc1 (Mf1) and Foxc2 (Mfh1) are required for the early organogenesis of the kidney and urinary tract. Development. 2000;127(7):1387-1395.
Kurzrock EA, Baskin LS, et al. Ontogeny of the male urethra: theory of endodermal differentiation. Differentiation. 1999;64(2):115-122.
Lamm ML, Podlasek CA, et al. Mesenchymal factor bone morphogenetic protein 4 restricts ductal budding and branching morphogenesis in the developing prostate. Dev Biol. 2001;232(2):301-314.
Langer JC. Fetal abdominal wall defects. Semin Pediatr Surg. 1993;2(2):121-128.
Larsson L, Aperia A, et al. Structural and functional development of the nephron. Acta Paediatr Scand Suppl. 1983;305:56-60.
Lee MM, Donahoe PK. Mullerian inhibiting substance: a gonadal hormone with multiple functions. Endocr Rev. 1993;14(2):152-164.
Lelievre-Pegorier M, Vilar J, et al. Mild vitamin A deficiency leads to inborn nephron deficit in the rat. Kidney Int. 1999;54(5):1455-1462.
Lewis LG. Cryptorchidism. J Urol. 1948;60:345-356.
Lin Y, Zhang S, et al. Induced repatterning of type XVIII collagen expression in ureter bud from kidney to lung type: association with sonic hedgehog and ectopic surfactant protein C. Development. 2001;128(9):1573-1585.
Loughna S, Landels E, et al. Growth factor control of developing kidney endothelial cells. Exp Nephrol. 1996;4(2):112-118.
Lubahn DB, Brown TR, et al. Sequence of the intron/exon junctions of the coding region of the human androgen receptor gene and identification of a point mutation in a family with complete androgen insensitivity [erratum appears in Proc Natl Acad Sci U S A 1990 Jun;87(11):4411]. Proc Natl Acad Sci U S A. 1989;86(23):9534-9538.
Lumbers ER. Functions of the renin-angiotensin system during development. Clin Exp Pharmacol Physiol. 1995;22(8):499-505.
Luo X, Ikeda Y, et al. A cell-specific nuclear receptor is essential for adrenal and gonadal development and sexual differentiation. Cell. 1994;77(4):481-490.
Mackie GG, Stephens FD. Duplex kidneys: a correlation of renal dysplasia with position of the ureteric orifice. Birth Defects Orig Article Ser. 1977;13(5):313-321.
Mah SP, Saueressig H, et al. Kidney development in cadherin-6 mutants: delayed mesenchyme-to-epithelial conversion and loss of nephrons. Dev Biol. 2000;223(1):38-53.
Marker PC, Stephan JP, et al. Fucosyltransferase1 and H-type complex carbohydrates modulate epithelial cell proliferation during prostatic branching morphogenesis. Dev Biol. 2001;233(1):95-108.
Martineau J, Nordqvist K, et al. Male-specific cell migration into the developing gonad. Curr Biol. 1997;7(12):958-968.
McElreavey K, Vilain E, et al. A regulatory cascade hypothesis for mammalian sex determination: SRY represses a negative regulator of male development. Proc Natl Acad Sci U S A. 1993;90(8):3368-3372.
McLaren A. Development of the mammalian gonad: the fate of the supporting cell lineage. Bioessays. 1991;13(4):151-156.
Mendelsohn C, Batourina E, et al. Stromal cells mediate retinoid-dependent functions essential for renal development. Development. 1999;126(6):1139-1148.
Miyamoto N, Yoshida M, et al. Defects of urogenital development in mice lacking Emx2. Development. 1997;124(9):1653-1664.
Miyazaki Y, Oshima K, et al. Bone morphogenetic protein 4 regulates the budding site and elongation of the mouse ureter. J Clin Invest. 2000;105(7):863-873.
Miyazaki Y, Oshima K, et al. Evidence that bone morphogenetic protein 4 has multiple biological functions during kidney and urinary tract development. Kidney Int. 2003;63(3):835-844.
Moore MW, Klein RD, et al. Renal and neuronal abnormalities in mice lacking GDNF. Nature. 1996;382(6586):76-79.
Nachtigal MW, Hirokawa Y, et al. Wilms’ tumor 1 and Dax-1 modulate the orphan nuclear receptor SF-1 in sex-specific gene expression. Cell. 1998;93(3):445-454.
Nef S, Parada LF. Cryptorchidism in mice mutant for Insl3. Nat Genet. 1999;22(3):295-299.
Nef S, Shipman T, et al. A molecular basis for estrogen-induced cryptorchidism. Dev Biol. 2000;224(2):354-361.
Nef S, Verma-Kurvari S, et al. Testis determination requires insulin receptor family function in mice. Nature. 2003;426(6964):291-295.
Newman J, Antonakopoulos GN. The fine structure of the human fetal urinary bladder: development and maturation: a light, transmission and scanning electron microscopic study. J Anat. 1989;166:135-150.
Nievelstein RA, van der Werff JF, et al. Normal and abnormal embryonic development of the anorectum in human embryos. Teratology. 1998;57(2):70-78.
Nishimura H, Yerkes E, et al. Role of the angiotensin type 2 receptor gene in congenital anomalies of the kidney and urinary tract, CAKUT, of mice and men. Mol Cell. 1999;3(1):1-10.
Nishinakamura R, Matsumoto Y, et al. Murine homolog of SALL1 is essential for ureteric bud invasion in kidney development. Development. 2001;128(16):3105-3115.
Oshima K, Miyazaki Y, et al. Angiotensin type II receptor expression and ureteral budding. J Urol. 2001;166(5):1848-1852.
Overbeek PA, Gorlov IP, et al. A transgenic insertion causing cryptorchidism in mice. Genesis. 2001;30(1):26-35.
Park SY, Jameson JL. Minireview: transcriptional regulation of gonadal development and differentiation. Endocrinology. 2005;146(3):1035-1042.
Park WH, Hutson JM. The gubernaculum shows rhythmic contractility and active movement during testicular descent. J Pediatr Surg. 1991;26(5):615-617.
Pelletier J, Schalling M, et al. Expression of the Wilms’ tumor gene WT1 in the murine urogenital system. Genes Dev. 1991;5(8):1345-1356.
Pepicelli CV, Kispert A, et al. GDNF induces branching and increased cell proliferation in the ureter of the mouse. Dev Biol. 1997;192(1):193-198.
Pichel JG, Shen L, et al. Defects in enteric innervation and kidney development in mice lacking GDNF. Nature. 1996;382(6586):73-76.
Piper M, Georgas K, et al. Expression of the vertebrate Slit gene family and their putative receptors, the Robo genes, in the developing murine kidney. Mech Dev. 2000;94(1-2):213-217.
Plisov SY, Yoshino K, et al. TGF beta 2, LIF and FGF2 cooperate to induce nephrogenesis. Development. 2001;128(7):1045-1057.
Podlasek CA, Barnett DH, et al. Prostate development requires Sonic hedgehog expressed by the urogenital sinus epithelium. Dev Biol. 1999;209(1):28-39.
Podlasek CA, Clemens JW, et al. Hoxa-13 gene mutation results in abnormal seminal vesicle and prostate development. J Urol. 1999;161(5):1655-1661.
Podlasek CA, Duboule D, et al. Male accessory sex organ morphogenesis is altered by loss of function of Hoxd-13. Dev Dynam. 1997;208(4):454-465.
Pohl M, Stuart RO, et al. Branching morphogenesis during kidney development. Annu Rev Physiol. 2000;62:595-620.
Porteous S, Torban E, et al. Primary renal hypoplasia in humans and mice with PAX2 mutations: evidence of increased apoptosis in fetal kidneys of Pax2(1Neu) +/− mutant mice. Hum Mol Genet. 2000;9(1):1-11.
Qiao J, Bush KT, et al. Multiple fibroblast growth factors support growth of the ureteric bud but have different effects on branching morphogenesis. Mech Dev. 2001;109(2):123-135.
Qiao J, Uzzo R, et al. FGF-7 modulates ureteric bud growth and nephron number in the developing kidney. Development. 1999;126(3):547-554.
Quinlan DM, Gearhart JP, et al. Abdominal wall defects and cryptorchidism: an animal model. J Urol. 1988;140:1141-1144.
Ruan W, Powell-Braxton L, et al. Evidence that insulin-like growth factor I and growth hormone are required for prostate gland development. Endocrinology. 1999;140(5):1984-1989.
Russell DW, Wilson JD. Steroid 5 alpha-reductase: two genes/two enzymes. Annu Rev Biochem. 1994;63:25-61.
Ryan G, Steele-Perkins V, et al. Repression of Pax-2 by WT1 during normal kidney development. Development. 1995;121(3):867-875.
Sainio K, Suvanto P, et al. Glial-cell-line-derived neurotrophic factor is required for bud initiation from ureteric epithelium. Development. 1997;124(20):4077-4087.
Sanchez MP, Silos-Santiago I, et al. Renal agenesis and the absence of enteric neurons in mice lacking GDNF. Nature. 1996;382(6586):70-73.
Sanyanusin P, Schimmenti LA, et al. Mutation of the gene in a family with optic nerve colobomas, renal anomolies and vesicoureteral reflux. Nat Genet. 1996;13(1):129.
Schmahl J, Eicher EM, et al. Sry induces cell proliferation in the mouse gonad. Development. 2000;127(1):65-73.
Schutz S, Le Moullec JM, et al. Early expression of all the components of the renin-angiotensin-system in human development [see comments]. Am J Pathol. 1996;149(6):2067-2079.
Schwarz R, Stephens FD. The persisting mesonephric duct: high junction of vas deferens and ureter. J Urol. 1978;120(5):592-596.
Sedman AB, Kershaw DB, et al. Recognition and management of angiotensin converting enzyme inhibitor fetopathy. Pediatr Nephrol. 1995;9(3):382-385.
Sekido R, Bar I, et al. SOX9 is up-regulated by the transient expression of SRY specifically in Sertoli cell precursors. Dev Biol. 2004;274(2):271-279.
Shawlot W, Behringer RR. Requirement for Lim1 in head-organizer function. Nature. 1995;374(6521):425-430.
Shotan A, Widerhorn J, et al. Risks of angiotensin-converting enzyme inhibition during pregnancy: experimental and clinical evidence, potential mechanisms, and recommendations for use. Am J Med. 1994;96(5):451-456.
Signoretti S, Waltregny D, et al. p63 is a prostate basal cell marker and is required for prostate development. Am J Pathol. 2000;157(6):1769-1775.
Sinclair AH, Berta P, et al. A gene from the human sex-determining region encodes a protein with homology to a conserved DNA-binding motif [see comments]. Nature. 1990;346(6281):240-244.
Stark K, Vainio S, et al. Epithelial transformation of metanephric mesenchyme in the developing kidney regulated by Wnt-4. Nature. 1994;372(6507):679-683.
Steger RW, Chandrashekar V, et al. Neuroendocrine and reproductive functions in male mice with targeted disruption of the prolactin gene. Endocrinology. 1998;139(9):3691-3695.
Stillman RJ. In utero exposure to diethylstilbestrol: adverse effects on the reproductive tract and reproductive performance and male and female offspring. Am J Obstet Gynecol. 1982;142(7):905-921.
Sugimura Y, Cunha GR, et al. Morphogenesis of ductal networks in the mouse prostate. Biol Reprod. 1986;34(5):961-971.
Swain A, Narvaez V, et al. Dax1 antagonizes Sry action in mammalian sex determination. Nature. 1998;391(6669):761-767.
Takeda H, Lasnitzki I, et al. Analysis of prostatic bud induction by brief androgen treatment in the fetal rat urogenital sinus. J Endocrinol. 1986;110(3):467-470.
Tayakkanonta K. The gubernaculum testis and its nerve supply. Aust NZ J Surg. 1963;33:61-67.
Tevosian SG, Albrecht KH, et al. Gonadal differentiation, sex determination and normal Sry expression in mice require direct interaction between transcription partners GATA4 and FOG2. Development. 2002;129(19):4627-4634.
Thomson AA, Cunha GR. Prostatic growth and development are regulated by FGF10. Development. 1999;126(16):3693-3701.
Tichy M. The morphogenesis of human sphincter urethrae muscle. Anat Embryol. 1989;180(6):577-582.
Tilmann C, Capel B. Mesonephric cell migration induces testis cord formation and Sertoli cell differentiation in the mammalian gonad. Development. 1999;126(13):2883-2890.
Timms BG, Lee CW, et al. Instructive induction of prostate growth and differentiation by a defined urogenital sinus mesenchyme. Microsc Res Tech. 1995;30(4):319-332.
Timms BG, Mohs TJ, et al. Ductal budding and branching patterns in the developing prostate. J Urol. 1994;151(5):1427-1432.
Timms BG, Petersen SL, et al. Prostate gland growth during development is stimulated in both male and female rat fetuses by intrauterine proximity to female fetuses. J Urol. 1999;161(5):1694-1701.
Toppari J, Skakkebaek NE. Sexual differentiation and environmental endocrine disrupters. Baillieres Clin Endocrinol Metab. 1988;12(1):143-156.
Torres M, Gomez-Pardo E, et al. Pax-2 controls multiple steps of urogenital development. Development. 1995;121(12):4057-4065.
Tufro A, Norwood VF, et al. Vascular endothelial growth factor induces nephrogenesis and vasculogenesis. J Am Soc Nephrol. 1999;10(10):2125-2134.
Tufro-McReddie A, Norwood VF, et al. Oxygen regulates vascular endothelial growth factor-mediated vasculogenesis and tubulogenesis. Dev Biol (Orlando). 1997;183(2):139-149.
Vainio S, Heikkila M, et al. Female development in mammals is regulated by Wnt-4 signalling. Nature. 1999;397(6718):405-409.
Vainio S, Lehtonen E, et al. Epithelial-mesenchymal interactions regulate the stage-specific expression of a cell surface proteoglycan, syndecan, in the developing kidney. Dev Biol (Orlando). 1989;134(2):382-391.
van der Putte SC. Normal and abnormal development of the anorectum. J Pediatr Surg. 1986;21(5):434-440.
van der Schoot P. Androgen-induced prevention of the outgrowth of cranial suspensory ligaments in fetal rats. J Androl. 1992;13:534-542.
van der Schoot P. The name cranial ovarian suspensory ligaments in mammalian anatomy should only be used to indicate the structures derived from the foetal cranial mesonephric and gonadal ligaments. Anat Rec. 1993;237:434-438.
Vermeij-Keers C, Hartwig NG, et al. Embryonic development of the ventral body wall and its congenital malformations. Semin Pediatr Surg. 1996;5(2):82-89.
Viana R, Batourina E, et al. The development of the bladder trigone, the center of the anti-reflux mechanism. Development. 2007;134(20):3763-3769.
Vidal VP, Chaboissier MC, et al. Sox9 induces testis development in XX transgenic mice. Nat Genet. 2001;28(3):216-217.
vom Saal FS, Timms BG, et al. Prostate enlargement in mice due to fetal exposure to low doses of estradiol or diethylstilbestrol and opposite effects at high doses. Proc Natl Acad Sci U S A. 1997;94(5):2056-2061.
Vukicevic S, Kopp JB, et al. Induction of nephrogenic mesenchyme by osteogenic protein 1 (bone morphogenetic protein 7). Proc Natl Acad Sci U S A. 1996;93(17):9021-9026.
Weiss JP. Embryogenesis of ureteral anomalies: a unifying theory. Aust NZ J Surg. 1988;58(8):631-638.
Wensing CJG. Testicular descent in the rat and a comparison of this process in the rat with that in the pig. Anat Rec. 1986;214:154-160.
Wilhelm D, Englert C. The Wilms tumor suppressor WT1 regulates early gonad development by activation of Sf1. Genes Dev. 2002;16(14):1839-1851.
Wyndham NR. A morphological study of testicular descent. J Anat. 1943;77:179-188.
Xu PX, Adams J, et al. Eya1-deficient mice lack ears and kidneys and show abnormal apoptosis of organ primordia. Nat Genet. 1999;23(1):113-117.
Yao HH, Matzuk MM, et al. Follistatin operates downstream of Wnt4 in mammalian ovary organogenesis. Dev Dynam. 2004;230(2):210-215.
Yao HH, Whoriskey W, et al. Desert Hedgehog/Patched 1 signaling specifies fetal Leydig cell fate in testis organogenesis. Genes Dev. 2002;16(11):1433-1440.
Yu RN, Ito M, et al. Role of Ahch in gonadal development and gametogenesis. Nat Genet. 1998;20(4):353-357.
Zimmermann S, Steding G, et al. Targeted disruption of the Insl3 gene causes bilateral cryptorchidism. Mol Endocrinol. 1999;13(5):681-691.