chapter 135 Surgical Management of Pediatric Stone Disease
Prior to 1990, nephrolithiasis was responsible for 1 in 1000 to 1 in 7600 hospital admissions annually throughout the United States (Nimkin et al, 1992). In recent years, however, a dramatic increase in pediatric urolithiasis has been observed (Srivastava and Alon, 2005). The authors would concur with this observation, especially among adolescents without known metabolic disturbances. It has been speculated that diets rich in sodium and carbohydrates may be contributing to the etiology of urolithiasis in this cohort of children.
The management of upper urinary tract calculi has evolved dramatically in both adults and children in the last two decades. Progress in the management of pediatric stone disease has been motivated by surgeons adopting and applying adult endourologic methods. The most important catalyst in the evolution of surgical advancement has been the expansion of minimally invasive surgery, in general, with development of smaller and more durable endoscopic equipment.
Since its introduction in the 1980s, extracorporeal shock wave lithotripsy (ESWL) has transformed the management of pediatric upper tract calculus disease and remains very useful today. Other modalities of treatment, including ureteroscopy, percutaneous nephrolithotomy (PCNL), and combination treatment protocols allow flexibility in management. However, a consensus has not been reached as to the most effective and safe technique for upper tract urolithiasis, especially considering ESWL versus ureteroscopy. Despite the lack of compelling scientific data comparing these treatment methods in children, ureteroscopy has emerged in many centers as the first-line of treatment.
Pediatric stone disease has always been more prevalent in underdeveloped countries. Of concern is the observation that pediatric stone disease is becoming more prevalent in the western hemisphere. Many pediatric patients with urolithiasis have metabolic abnormalities (Jayanthi et al, 1999). Therefore there has been an increased demand on pediatric urologists to manage both simple and complex urolithiasis in all pediatric age groups.
Evaluation
Metabolic Workup
Often, a complete metabolic blood profile and a 24-hour urine collection are obtained once a stone is confirmed by imaging. Unfortunately, there are no accepted standard reference ranges for 24-hour urine analyses in the stone-forming pediatric population. Indeed, many pediatric urologists use adult standards to direct medical treatment, because specific reference ranges are not readily available or require additional complicated calculations. Regrettably, there are significant differences between the normal ranges of urine chemistries in children and adults (Battino et al, 2002; DeFoor et al, 2005a). In addition, conflicting reports exist as to the commonality of metabolic disturbances in the presence of calcium-based stones (Sternberg et al, 2005).
In the preliminary evaluation of pediatric stone disease, it is common to obtain 24-hour urine values for creatinine, sodium, calcium, oxalate, uric acid, and citrate. The very cumbersome nature of a 24-hour urine collection often limits its accuracy in the pediatric population. In light of this random urine spot sampling, ratios have been used. Urine calcium to urine creatine ratios (Uca : Ucr), for example, have been used with sensitivities and specificities up to 90% and 84%, respectively, in the evaluation of hypercalciuria, a known risk factor for urolithiasis (Mir and Serdaroglu, 2005). However, such a test is limited to one urinary metabolite and cannot be relied upon to monitor responses to various medical treatments. Furthermore, it is unreasonable to rely solely on urinary calcium excretion values as an indication of overall metabolic disturbances.
It has been suggested that measurement of urinary supersaturation products (calcium oxalate, urate) may help to improve identifying those children at risk for stone formation. DeFoor and colleagues (2006), for example, most recently determined that supersaturation levels of calcium oxalate, as well as calcium to creatinine levels, were significantly higher in children with stones compared with controls. This difference, however, may have been reflective of differences found in urinary volumes. Lande and colleagues (2005) have demonstrated that low urine volumes in children often negate the benefit of pursuing urine supersaturation products in a stone workup. Such conflicting data often confuses clinicians as to what urinary evaluation to pursue in the workup and management of pediatric stone disease.
Stone disease in the pediatric population has genetic, anatomic, metabolic, and dietary causes. There are numerous genetic causes of hypercalciuric nephrolithiasis alone that contribute to pediatric stone disease (Stechman et al, 2009). In this regard, the medical treatment always accompanies the endourologic management. To fully treat the pediatric stone patient, there must be a focus on prevention through diet and medication monitoring. For this reason, the pediatric nephrologist is a critical player in the management and surveillance of these children. Details regarding the metabolic workup and medical treatment of pediatric stone disease are beyond the scope of this chapter. Numerous pediatric nephrology resources are available. Alon (2009), for example, provides an excellent pediatric nephrology review, including medication dosing by weight. The website Litholink (www.litholink.com) can provide valuable information to parents and pediatric urologists alike.
Imaging
Radiographic assessment of the child with calculus disease must be accurate, economic, and safe. The goals include determination of stone location, size, density, and urinary tract anatomy. Indications for assessment include acutely symptomatic children with a suspected calculus or children with known calculus disease requiring follow-up evaluation to either determine stone burden or recurrence. Radiographic evaluation is also important in children with abnormal urinary tract anatomy that may predispose to calculus formation, requiring eventual surgical management.
Radiographic imaging in children is similar to imaging in the adult patient. The workup begins when the diagnosis of a calculus is suspected, and the radiographic study is determined by factors such as acuity of presentation and the practices of the treating hospital. Many, if not most, children with renal colic present to an emergency room, and unenhanced helical computed tomography (CT) has become the most accurate and efficient first choice in initial imaging. Typically, thin-slice helical protocol is defined by narrow collimation (≤5 mm). CT is not only able to visualize the entire urinary tract for the tiniest calculus, but it has the potential of ruling out alternative diagnosis. Unenhanced helical CT of the abdomen and pelvis to evaluate urinary tract calculi was first described in 1995 and is the mainstay for calculus imaging in adolescents and adults, replacing more traditional studies, such as intravenous pyelography (IVP), a kidney-ureter-bladder (KUB) film, and ultrasonography (Smith et al, 1995). With increased usage and familiarity with the technique, its application has expanded into the pediatric population with similar advantages. These advantages include high sensitivity and specificity, ready availability and speed in assessment, and intravenous (IV) contrast is not necessary. Because children are at significant lifetime risk for recurrence of urolithiasis, judicious use is necessary to limit radiation exposure, particularly gonadal exposure, and concerns of potential increase risk of future malignancy.
CT is highly accurate in stone assessment, exceeding 96% sensitivity and specificity independent of the location of the calculus (Smith et al, 1996b; Hamm et al, 2001; Heneghan et al, 2003; Palmer et al, 2005). CT is also helpful in demonstrating secondary signs of acute obstruction, such as hydroureteronephrosis, renal enlargement, and perinephric or periureteric stranding; however, the latter findings may be less obvious or absent in pediatric patients due to comparably less retroperitoneal fat (Smith et al, 1996a; Smergel et al, 2001; Strouse et al, 2002).
Progress has been made in developing unenhanced CT protocols that minimize the radiation dosage while not compromising diagnostic information (Heneghan et al, 2003; Cody et al, 2004; Singh et al, 2009). Experimental protocols using anthropomorphic pediatric phantoms have been used in a number of studies to determine specific organ doses, calculate effective dose, determine the lifetime attributable risk (LAR) for cancer incidence, and relative risk of cancer induction from a single scan under both standard- and low-dose modes (Brisse et al, 2009; Kim et al, 2010). Computer-simulated dose reduction has been useful in determining diagnostic thresholds in children. Compared with standard protocols, halving the dose to 40 mA for children weighing 50 kg or less does not significantly affect the diagnosis of pediatric renal stones (Karmazyn et al, 2009). Spielmann and colleagues (2002) found excellent detectability of calculi measuring 2 to 8 mm using much lower amperage, with an almost threefold decreased estimated radiation dose compared with standard protocols. Perhaps most importantly, radiation exposure can also be reduced effectively by reducing the number of CT examinations performed for poor clinical indications, scanning only the anatomic region of interest, and not performing either unenhanced and contrast-enhanced scanning unless absolutely necessary (Cohen, 2009).
Ultrasonography has a more limited role in the assessment of urolithiasis compared with CT but has the distinct advantage of no associated ionizing radiation. Therefore ultrasonography should be considered as a screening tool in the workup for nonemergent abdominal or flank pain. Ultrasonography, although useful in the evaluation of renal calculi or hydronephrosis, is technically limited for use in diagnosing a ureteral stone, with the possible exception of the very distal ureter or bladder. Especially in the acute setting, that is, symptomatic presentation with renal colic or hematuria, ultrasonography is less useful in detecting or directing management of urolithiasis. Palmer and colleagues (2005) demonstrated that ultrasonography was nondiagnostic in 41% of children compared with 5% using CT, and it failed to detect ureteral calculi in 62% of pediatric patients. Similar results have been reported in adults, confirming that ultrasonography is of limited value in the workup of urolithiasis (Fowler et al, 2002). Symptomatic children with a known history of renal urolithiasis, or children on observation protocols, may suffice with ultrasonography (US) and KUB alone, thereby limiting exposure to ionizing radiation. Additional support favoring US versus CT in the diagnosis of pediatric nephrolithiasis is also rooted in the ultimate clinical decision about how the child is to be treated. In a prospective study comparing CT and US, Passerotti and colleagues (2009) found that although CT was a more sensitive study in detecting stones in children, the difference in usefulness between the two radiologic techniques was not clinically significant.
Fluoroscopy has an important role in the real-time detection and management of urolithiasis in children, and the same important safety concerns exist regarding radiation exposure as with CT. C-arm fluoroscopy is used during the surgical setting to assist in antegrade percutaneous access of the upper urinary tract and retrograde access of the lower urinary tract. Manipulation of endoscopic instruments in vivo often requires fluoroscopic monitoring. Fluoroscopy is also used during ESWL to assist in stone localization and monitor the effectiveness of treatment. Urologists must have a working understanding of the principles of fluoroscopy and be aware of the intraprocedure fluoroscopy time and energy settings to limit radiation exposure to both the pediatric patient and operative staff.
Conservative Management
Conservative management of pediatric nephrolithiasis is considered first-line treatment, provided there is no evidence an obstructing stone is harboring an infection or a child is failing to thrive as the result of his or her stone disease. Clinical scenarios, including fever, anorexia greater than 24 hours, persistent nausea and vomiting, and/or pain refractory to conservative measures prompts endourologic intervention. In the instance of a stone in a solitary kidney, early intervention versus conservative treatment is favored. In managing stone disease in the pediatric population, it is important to note that renal calculi less than 3 mm are likely to spontaneously pass, and stones greater than or equal to 4 mm in the distal ureter are likely to require endourologic treatment (Van Savage et al, 2000). This information should be relayed to caregivers and parents. If a ureteral stent is placed acutely in children for the clinical circumstances described above, definitive endourologic therapy is delayed 2 to 4 weeks to allow for decompression, ureteric orifice dilation, resolution of edema, and proper treatment and clearance of any infection if need be.
There have been many studies in adults evaluating the efficacy of medical expulsive therapy to facilitate distal stone passage. Use of α antagonists, calcium channel blockers, and steroids has been shown to be effective. Based on efficacy demonstrated in the adult population (Porpiglia et al, 2004), α-receptor antagonists, such as tamsulosin, may be offered on an individualized basis as adjunctive therapy to facilitate ureteral expulsion in children. To date, however, there is a lack of published data capturing the pediatric population that prove the superiority of these agents versus standard pain medication. A Turkish study, for example, demonstrated that daily administration of 0.03 mg/kg of the α antagonist doxazosin versus an analgesic alone did not show superior expulsive results regarding distal ureteral stones up to 10 mm in children 2 to 14 years of age (Aydogdu et al, 2009).
Management of Upper Urinary Tract Calculi
Pediatric Considerations
Special considerations in the endourologic management of stone disease in children include preservation of renal development and function, prevention of radiation exposure, and minimizing the need for re-treatment. Despite advances in endourologic equipment and technique, controversy remains regarding the contribution of SWL to future development of diabetes or hypertension, and whether ureteric orifice dilation during ureteroscopy (URS) leads to ureteral stricture formation or development of vesicoureteral reflux. International consensus is lacking as to the most effective surgical management of pediatric stone disease due to lack of prospective randomized trials comparing treatment modalities and disparity in the access to emerging technologies. Regardless of treatment modality, the presence of residual stone fragments is associated with adverse clinical outcomes (Afshar et al, 2004), and every attempt should be made to achieve a stone-free status. The surgeon’s experience is paramount to facilitate complete stone clearance and minimize re-treatment rates. The decision regarding the most efficacious primary treatment modality must be individualized per child based on age, anatomy, location, and composition of the stone burden.
Antibiotic Use
In line with the 2008 American Urologic Association’s best practice statement on antibiotic prophylaxis, less than or equal to 24 hours of perioperative antibiotics are indicated in all patients undergoing upper tract instrumentation (Wolf et al, 2008). In children, appropriate agents include trimethoprim-sulfamethazole, first- and second-generation cephalosporins, and ampicillin in combination with an aminoglycoside. A urine culture is mandatory before all upper tract procedures to determine if the urine is sterile, and culture results are used to guide preoperative antibiotic therapy, particularly for percutaneous procedures, patients with high-grade obstruction, or patients with an indwelling stent (Wu and Docimo, 2004). In the authors’ practice, children with a negative urine culture undergoing uncomplicated URS procedures receive perioperative cefazolin, and all children undergoing a percutaneous procedure or who have a preexisting ureteral stent/nephrostomy tube receive a fluoroquinolone or ampicillin/gentamicin. Use of postoperative antibiotics is controversial and is determined on an individual basis, especially with recent data demonstrating an increased risk of developing resistant bacterial strains with prolonged use of antibiotic prophylactic therapy (Conway et al, 2007).
Shock Wave Lithotripsy
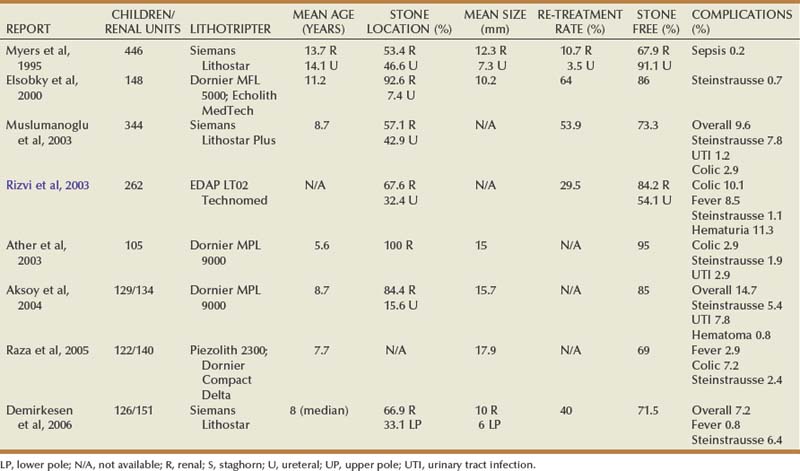
The emergence of shock wave lithotripsy (SWL) revolutionized the minimally invasive treatment of adult urolithiasis during the early 1980s. Since the initial report on successful SWL use in children in 1986 (Newman et al, 1986), large series have reported complication, safety, and stone-free rates comparable to adult cohorts (Table 135–1) (Myers et al, 1995; Elsobky et al, 2000; Ather and Noor, 2003; Muslumanoglu et al, 2003; Rizvi et al, 2003; Aksoy et al, 2004; Raza et al, 2005; Demirkesen et al, 2006). When used as a primary treatment option for upper tract calculi, SWL efficacy ranges from 68% to 84% (Rizvi et al, 2003; Myers et al, 1995; DeFoor et al, 2005b). SWL has been a preferred treatment modality for uncomplicated renal and proximal calculi less than or equal to 15 mm in the pediatric population. In a contemporary series of 216 children (mean age 6.6 years) with a mean stone size of 14.9 mm and who were undergoing SWL with the Dornier HM3 lithotripter, Landau and colleagues (2009) reported a 3-month stone-free rate of 80%, demonstrating that efficacious stone-free rates can be achieved in appropriate candidates. Complications rates are minimal and range in severity from hematuria and ecchymosis to obstruction with sepsis (Farhat and Kropp, 2007).
Although well tolerated in children, current stone-free rates with SWL are difficult to interpret from the existing body of data due to discrepancies between studies regarding type of lithotriptor, number of shocks administered, and re-treatment rates. Recent data suggest that stone-free rates in children with a history of urologic condition or urinary tract reconstruction are quite low (12.5%), and, with alternative surgical techniques available, children may be better served with URS or PCNL (Nelson et al, 2008). Despite encouraging results, SWL has not been approved by the Food and Drug Administration for use in children, although it is a widely accepted treatment modality.
Shock Wave Lithotripsy Technique in Children
General anesthesia is administered in a majority of smaller children to avoid both patient and stone motion and the need for repeated repositioning. With modern lithotriptors, intravenous sedation has been successfully employed in select older children (Aldridge et al, 2006). Bowel preparation is seldom used in order to avoid dehydration and electrolyte imbalance postoperatively. The number of shocks delivered and the kilovoltage used vary per lithotripter, but the current consensus is that low-power settings (17 to 22 kV) should be used to prevent stone migration during the procedure, with 3000 shock waves per session (<2000 in very young children) (Farhat and Kropp, 2007). A recent report assessed and compared the number and intensity of shock waves required for stone fragmentation in 44 children (mean age 5.9 years) and 562 adults (mean age 40.9 years). With an equivalent number of sessions (1.1 vs. 1.1), the mean number of shock waves (950 vs. 1262, P < .001) and the kV required (11.8 vs. 12.4, P < .001) were significantly reduced in the pediatric cohort (Kurien et al, 2009).
Whether or not to place a ureteral stent prior to ESWL in children remains controversial and is a matter of personal preference. It is currently unclear if placement of a ureteral stent prior to SWL facilitates fragment passage and improves stone-free outcomes. Although prestenting rates are not consistent across series, current relative indications include cases of solitary kidneys, staghorn calculi, large ureteral calculi, obstruction, or abnormal anatomy and are not based on total stone burden. Ureteral catheters with retrograde opacification are occasionally employed by some to aid in the localization of radiolucent calculi.
Stone Size, Location, Composition, and Patient Age
Although early series focused primarily on the feasibility, safety, and efficacy of SWL in children, recent efforts have centered on identifying demographic, anatomic, and stone-related prognostic factors for treatment success. SWL is currently considered the primary treatment for upper tract calculi less than or equal to 15 mm in children (Farhat and Kropp, 2007), but evidence supporting this stone size cutoff is lacking. Ather and Noor (2003), for example, analyzed the correlation between stone size and clearance in 105 children younger than 14 years. They reported an overall stone-free rate of 95% after a mean of 1.7 SWL treatments, with 5% of patients requiring additional procedures as adjuncts to SWL. In this cohort, mean stone size in the treatment success group was 14 mm compared with 16 mm in the treatment failure group. By contrast, Elsobky and colleagues (2000) reported a 91% stone-free rate versus 75% stone-free rate for a mean stone diameter less than 10 mm and greater than 10 mm, respectively. Recently, Shouman and colleagues (2009) reported on a series of 24 children with a mean stone size of 31 mm undergoing SWL with the Dornier DoLi S device. In 53 sessions requiring a mean number of 3489 shock waves per session, stone-free and complication rates were 83.3% and 25%, respectively. Although it is possible to treat very large stone burdens with SWL, concerns include the necessity of more shock treatments, more frequent re-treatment sessions, and increased risk of postoperative obstruction. Further study delineating a clear size cutoff for uncomplicated upper tract stone burden is required to effectively counsel parents regarding the most effective first-line therapy for renal calculi between 1 and 1.5 cm.
Renal anatomy and stone location has been the subject of recent interest. The subject of frequent debate in the adult population, the most effective management of lower pole calculi in children has yet to be determined. Stone-free rates from initial small retrospective SWL series range from 56% to 61% (Ozgur Tan et al, 2003; Onal et al, 2004) with re-treatment rates as high as 40% (Onal et al, 2004). SWL failure and re-treatment rates were associated with increased mean stone burden (Onal et al, 2004), increased infundibular length (Ozgur Tan et al, 2003), and an infundibulopelvic angle greater than 45 degrees (Ozgur Tan et al, 2003).
Staghorn calculi are uncommon in children and represent a management challenge. Although monotherapy success rates are low in adults, acceptable stone-free rates in children have been achieved with SWL. In 23 children with a mean stone burden of 1.6 cm who were stratified by age, Lottmann and colleagues (2001) reported an overall stone-free rate of 82.6%, with only one case of symptomatic obstruction. A ureteral stent was placed in 22% of children, and these authors reported an 88% stone-free rate in children less than 2 years old compared with 71% in children 6 to 11 years old. In 42 children with a mean stone burden of 3.2 cm and stratified by ureteral stent placement, Al-Busaidy and colleagues (2003) reported an overall stone-free rate of 79%. Although stent placement did not affect stone-free rates, they found that stent placement significantly reduced the major complication rate. The superior success rates with SWL monotherapy in children compared with adults have been attributed to softer stone composition, smaller relative stone volume, increased ureteral compliance to accommodate stone fragments, and smaller body volume to facilitate shock transmission.
SWL safety and efficacy have been demonstrated even in very young children. McLorie and colleagues (2003) treated 34 children younger than 3.5 years (mean age 23 months) and reported an 86% overall stone-free rate (66% after one treatment) without major complications. Treatment of proximal ureteral stones has achieved similar success rates compared with treatment of renal stones in most pediatric series, although ureteral stenting is more commonly employed to aid in stone localization and clearance (Myers et al, 1995). Treatment of mid- to distal ureteral calculi has historically been avoided in children due to difficulties with localization over the sacroiliac joint and concern regarding possible injury to developing reproductive systems. The greater and lesser sciatic foramen has been explored as a potential blast path to treat distal stones in children.
SWL success by stone composition is similar between the adult and pediatric populations. Cystine stones are uniquely challenging due to their durability and high recurrence rates. Although SWL monotherapy has demonstrated variable results in adults, there are few reports in the pediatric population. In a small recent series, Slavkovic and colleagues (2002) reported a 50% stone-free rate in 6 children with a cystine stone burden ranging from 0.2 to 2.5 cm. Although stone-free rates were low, fragmentation was achieved in 100% of patients, and the stone dissolution was achieved with medical therapy in the remaining children following SWL. Some authors have proposed that cystine stones formed within 2 years of therapy may be more easily fragmented with SWL and that stone number, and not diameter, may be more predictive of success (Farhat and Kropp, 2007).
Limitations and Concerns
In children, there is currently no consensus regarding the maximum size of residual stone fragments (RF) that are considered clinically significant, and as a result, there is no clear definition as to what constitutes “stone-free” status (Wu and Docimo, 2004; Farhat and Kropp, 2007). Although children have been shown to have a greater capacity to clear fragments than adults (Gofrit et al, 2001), the presence of RFs have been correlated with adverse clinical outcomes (Afshar et al, 2004). Afshar and colleagues (2004) followed 26 renal units with RFs less than or equal to 5 mm and reported that although 31% were asymptomatic with no fragment growth, 69% had adverse clinical outcomes, including RF growth or clinical symptoms. Patients with RF had a significant increase in adverse clinical outcome compared with stone-free subjects, and the presence of metabolic disorders was associated with RF growth (Afshar et al, 2004). For these reasons, metabolic evaluations are now routinely being performed in children with a history of calculi, and every attempt should be made to achieve stone-free status.
Although SWL is well tolerated in children with few complications, stone-free rates following single-session monotherapy can remain as low as 44% (Muslumanoglu et al, 2003). As a result, children are subjected to multiple treatments requiring general anesthesia (Aldridge et al, 2006). The need for multiple treatment sessions is concerning, because the effects of shock waves on renal tissue are unclear. A growing body of evidence in adults indicates that shock waves result in renal vessel vasoconstriction and that renal tubular injury and subcapsular hematoma from cavitation and shear forces are dependent on the kilovoltage applied (Lingeman et al, 2003). In a large series of 340 adult patients with a mean follow-up of 19 years post-SWL, (Krambeck and colleagues (2006) reported an increased risk of hypertension and diabetes mellitus related to bilateral treatment, number of administered shocks, and treatment intensity. Although these results are concerning, differences between pediatric and adult populations and limitations inherent to a questionnaire-based retrospective study make application of these data in children difficult.
Retrospective studies with limited follow-up in children have reported that SWL and PCNL do not cause renal morphologic or functional alteration as measured by glomerular flow rate (GFR) and serial dimercaptosuccinic acid (DMSA) functional studies (Wadhwa et al, 2007), but long-term data are unavailable to date. To eliminate confounding variables and fully address the risks of chronic renal damage from SWL, long-term prospective data in children are clearly required.
Ureteroscopic Management of Upper Urinary Tract Calculi
The indications for primary ureteroscopic management of upper tract calculi in children have significantly expanded. In combination with new endourologic instrumentation and holmium : YAG lasers, access and treatment of calculus disease throughout the entire pediatric urinary tract is effective, safe, and readily available in the acute setting. Adoption of ureteroscopy in the treatment of pediatric upper tract calculus disease lagged behind the adult experience, because of concerns regarding large ureteroscope caliber size in children. However, evolving experience with ureteroscopy and recent large single-center retrospective series have demonstrated comparable stone-free and complication rates to SWL in children, validating its growing application in children.
Indications
Since the mid-1980s, with the acceptance of SWL as primary therapy for upper tract calculi less than 1.5 cm, ureteroscopy has been historically used for calculi found below the iliac crests and for upper tract calculi after SWL failure (Wu and Docimo, 2004). Ureteroscopy was not considered primary therapy for upper tract stones in children due to complications from ureteral ischemia, perforation, stricture formation, and development of vesicoureteral reflux as a result of dilation of small-caliber ureteric orifices.
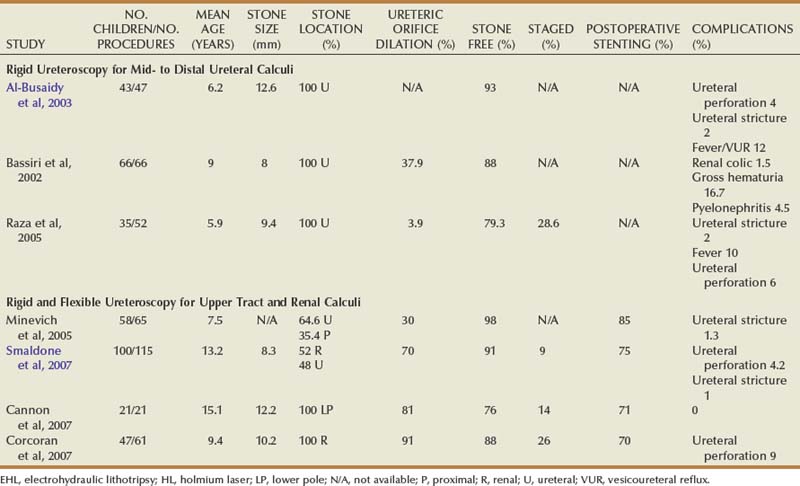
With significant improvements in both the miniaturization and durability of endoscopic equipment and the acceptance of the holmium laser, ureteroscopy has become a more attractive option in young children. Early series using rigid ureteroscopy for distal stones reported stone-free rates ranging from 86% to 100% with minimal complications (Table 135–2) (Ritchey et al, 1988; Van Savage et al, 2000; Schuster et al, 2002; Rizvi et al, 2003; De Dominicis et al, 2005; Minevich et al, 2005; Tan et al, 2005). Children randomized to either ureteroscopy or SWL as primary therapy for distal ureteral stones were reported to have a significantly higher stone-free rate after one treatment (94% vs. 43%) for those treated with ureteroscopy (De Dominicis et al, 2005). Another group reported experience using 4.5-, 6-, and 8-Fr rigid ureteroscopy in treating proximal ureteral stones, with a mean age of 10.7 years. Ureteral dilation was not performed in any of the cases, and 100% of children were rendered stone free (Lesani and Palmer, 2006). The results of these retrospective studies have begun to refute the notion that dilation of the pediatric ureter will result in vesicoureteral reflux or the development of ureteral strictures. In a systematic review of the literature encompassing 221 pediatric ureteroscopies, Schuster and colleagues (2002) noted only two ureteral strictures and a low incidence of vesicoureteral reflux. Newer 4.5-Fr semirigid ureteroscopes, with working ports that accommodate 2.4-Fr endosurgical instrumentation, extend the versatility of managing distal ureteral calculi in most pediatric patients without predilation.
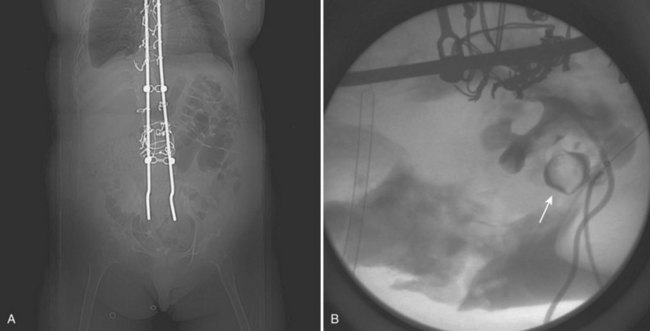
Standardization in the treatment of distal calculi in children has led a number of centers to expand ureteroscopy to the treatment of upper tract calculi (Table 135–2). Comparable stone-free rates approaching 100%, with complication rates similar to that of the adult population, have recently been reported (Minevich et al, 2005; Lesani and Palmer, 2006; Cannon et al, 2007; Corcoran et al, 2007; Smaldone et al, 2007). Smaldone and colleagues (2007) reported a 91% stone-free rate with a mean follow-up of 10 months in a series of 100 children with a mean stone diameter of 8.3 mm, 52% of whom had upper tract calculi. Nine percent of these children underwent staged procedures. They reported a 4.2% perforation rate that was managed with ureteral stenting and one distal ureteral stricture requiring open neocystostomy. Corcoran and colleagues (2007) reviewed a cohort of 47 children (mean age 9.4 years) with upper tract calculi managed with flexible ureteroscopy and holmium laser lithotripsy. An 88% stone-free rate with a mean stone burden of 10.2 mm was reported, with 26% requiring staged procedures. These authors concluded that because of improved ureteroscopic access to the pediatric upper tract, calculi up to 15 mm were as safely and effectively treated in children as in adults. Even with outstanding pediatric ureteroscopic technology, pediatric ureters may require a period of stenting prior to embarking on endoscopic treatment. For example, a smaller-caliber ureter that is resistant to instrumentation may be passively dilated with an indwelling stent during 6 to 8 weeks (Fig. 135–1).
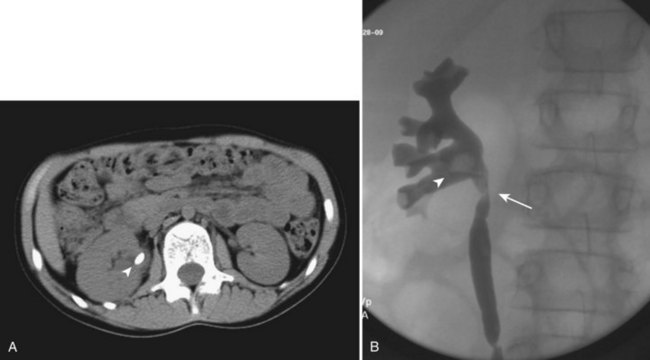
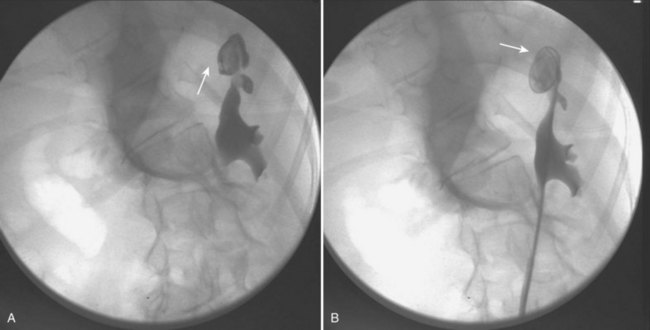
Figure 135–1 CT scan (A) and retrograde pyelogram (B) in a 10-year-old female with glucose transport deficiency demonstrating a 9-mm renal pelvic stone (arrowheads). Nephrolithiasis was secondary to profound hypocitruria and hypercalciuria. Primary ureteroscopy could not be accomplished because of a narrowed segment of proximal ureter (arrow). Following 8 weeks of ureteral accommodation with an indwelling stent, flexible ureteroscopy with laser lithotripsy and stone basketing was performed.
Adoption of techniques used in the adult population, most notably sequential coaxial and balloon dilation of the ureteric orifice and use of ureteral access sheaths, has further facilitated access to the pediatric urinary tract. Initially described in eight children by Singh and colleagues (2006), ureteral access sheaths have been shown to facilitate repetitive upper tract access, reduce intrarenal pressures, decrease operative time, and improve stone-free rates in adults. Access for the treatment of lower pole calculi is possible with use of ureteral access sheaths and the 6.9-Fr flexible ureteroscope, a stone location that would have previously required SWL or PCNL. Cannon and colleagues (2007) reported a 76% stone-free rate in 21 children with lower pole calculi and a mean stone diameter of 12.2 mm. After a mean of 11.4 months, no major complications were observed. Therefore with the transition from SWL to ureteroscopy as a primary treatment modality at the authors’ institution, current relative contraindications to ureteroscopic management include staghorn stones in recurrent stone formers more amenable to PCNL, anatomic anomalies making retrograde access difficult, and previous endoscopic failure.
Equipment
Having an updated and well-stocked system that is familiar to the operative staff is vital to efficient management. It usually falls on the nursing staff to care for the delicate instruments and keep up on repairs so that the instruments are operational when needed; maintenance is often needed after 20 to 30 uses. A “basic” ureteroscopic kit is listed below; however, variation exists based on the manufacturer. An array of semirigid and flexible pediatric ureteroscopes, in additional to ureteral access sheaths, is available from numerous companies (Buscarini and Conlin, 2008). In general, semirigid ureteroscopes usually have a 2.4- to 3.5-Fr working port and flexible ureteroscopes a 1.8- to 3.5-Fr working port. With flexible ureteroscopes, distal tip deflection up to 270 degrees can facilitate access to most lower pole stones (Figs. 135-2 and 135-3). However, smaller working areas and difficulties in exchanging instruments through the working element of a deflected ureteroscope may limit this treatment modality in some cases (Fig. 135–4).

Figure 135–2 Pediatric semirigid self-dilating ureteroscopes may be used for rigid ureteroscopy in toddlers. A 4.5-Fr distal beak allows easier passage to facilitate dilation with the 6.5-Fr proximal component.
(Photo provided by Richard Wolf; © Richard Wolf, all rights reserved.)

Figure 135–3 Flexible ureteroscopes offer thumb control (A) for biplanar 270-degree deflection (B). Working channels from 1.8 to 3.6 Fr can accommodate working elements such as a holmium : YAG laser fiber (C).
(Photos provided by Richard Wolf; © Richard Wolf, all rights reserved.)
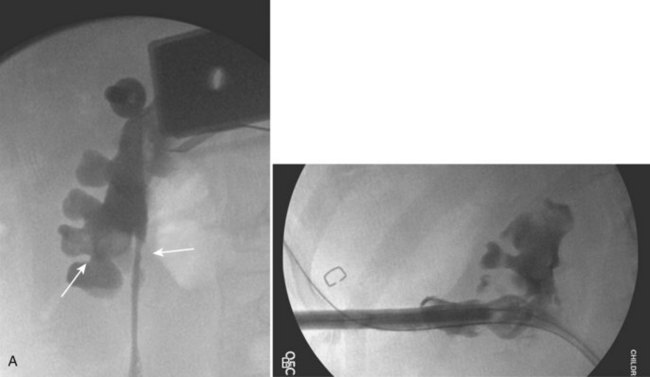
Figure 135–4 A, A 7-year-old boy with two separate 7- to 9-mm stones within lower pole calices (left arrow). A ureteroscopic approach was attempted, but a lower pole infundibular–ureteropelvic junction angle greater than 270 degrees (right arrow) limited ureteroscopic access and visibility. B, A percutaneous nephrolithotomy was subsequently performed, clearing all stones in one sitting.
Ureteroscopic Technique in Children
All ureteroscopic procedures are performed under general anesthesia with paralysis to prevent patient movement and minimize the risk of ureteral perforation. When clinically appropriate, a urine culture should be confirmed prior to surgery. Following broad-spectrum antibiotic prophylaxis, patients are placed in the lithotomy position, and a rigid cystoscope (7.5, 11, or 18 Fr) is used to perform a retrograde pyelogram and to place a safety or working wire under fluoroscopic guidance. Initial ureteric orifice dilation is performed under C-arm fluoroscopic guidance with 8/10-Fr coaxial dilators in ureters that have not been prestented or when the rigid/flexible ureteroscope cannot easily be advanced. The authors generally do not use balloon dilation of the ureteric orifice due to less control and cognitive “feel” during active dilation, as well as concern for development of ureteral stricture from ischemia. If the authors encounter difficulty easily passing the 8/10 dilator, their preference is to place a stent and return for a second procedure rather than dilate more aggressively.
The decision to use a flexible (6.9-Fr) or semirigid (7.5-Fr) ureteroscope is determined on size and location of stone, anatomic factors, and individual surgeon preference. Rigid or semirigid ureteroscopy for ureteral calculi is routinely performed with a safety wire in place, while flexible ureteroscopy is performed with both a safety and working wire in place. Ureteral access sheaths (internal diameter of 9.5 Fr) are routinely used to facilitate flexible ureteroscopy, especially in cases of large proximal ureteral stones and heavy renal pelvis stone burdens. Access sheaths may also serve to facilitate flexible ureteroscopy when altered anatomy or torturous ureters are encountered (Fig. 135–5).
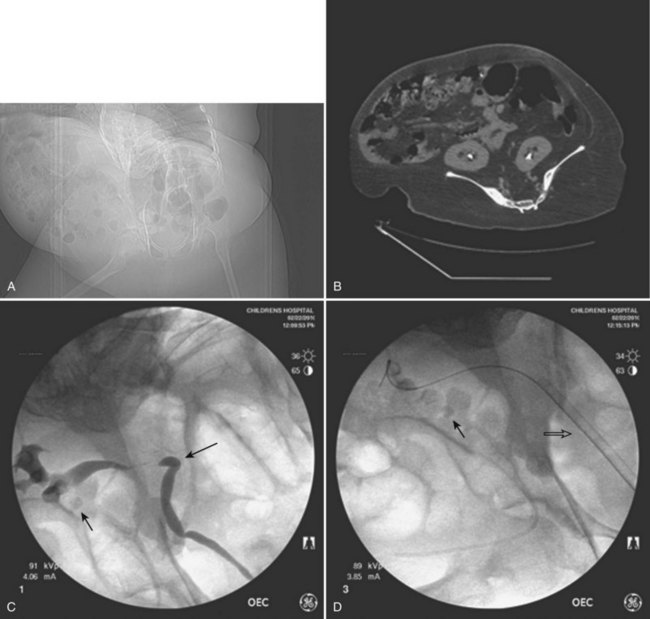
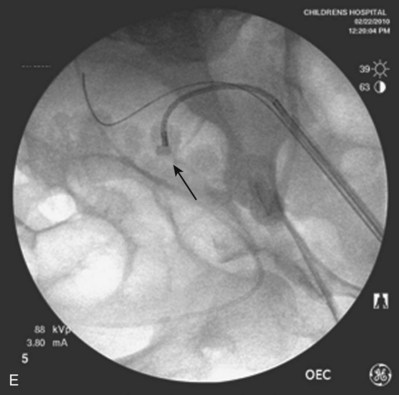
Figure 135–5 Altered anatomy in a 17-year-old male spina bifida patient who underwent a high thoracic myelomeningocele closure at birth (A). A computed tomography (CT) scan performed for a gross hematuria workup revealed bilateral nephrolithiasis in pelvic kidneys (B). The right retrograde pyelogram (C) demonstrated the lower pole stone (short arrow) and a torturous ureter almost taking a perpendicular course (long arrow). Placement of a ureteral access sheath (D) straightened the ureter (white arrow). This allowed easier access to address the stone (short arrow in D) with a flexible ureteroscope (E).
Irrigating fluid, which may be used under pressure, should be isotonic and at body temperature to avoid hypothermia and hyponatremia. Calculi are basket-extracted when feasible or fragmented using the holmium : YAG laser to facilitate removal. The decision to place a ureteral stent postoperatively is made based on the duration of the procedure, number of passes with the ureteroscope, and degree of visible ureteral trauma or edema at the conclusion of the procedure. If the child can tolerate leaving a urethral string in place for 3 days to 1 week, the patient’s parents are asked to remove the stent at home; otherwise, the stent is removed under brief anesthetic after 7 days.
Limitations and Complications
As ureteroscopy is becoming more prominent in the pediatric endourologist’s armamentarium, complications need to be anticipated and unanswered questions addressed. The most common complications involve unrecognized ureteral injury, including mucosal flaps and tears, perforation, false passage, and partial to complete avulsion. Mitigating the damage by early recognition and temporizing with immediate discontinuation of the procedure and passage of a ureteral stent may avoid complications related to shear force injury on the ureter, ischemic damage, and extravasation of irrigant or urine. Injury can occur during introduction of the ureteroscope or passage, either antegrade or retrograde, of instrumentation (guidewires, baskets, dilators), especially in the area of an impacted ureteral calculus. Attempting to withdraw a basket-entrapped calculus too large for the ureter to accommodate can result in any of the above-mentioned injuries. In postpubertal children with an adult body mass, ureteroscopic access is technically similar to the adult population. However, in the prepubertal child, whether or not to attempt primary treatment without ureteric orifice dilation, to perform dilation at the time of definitive therapy, or to place a stent and allow the ureter to passively dilate prior to definitive therapy remains in question. Herndon and colleagues (2006) performed semirigid ureteroscopy (4.5 and 6.5 Fr for distal ureteral calculi) in 29 children with a mean age of 11 years. With no ureter actively dilated and only 14% of children prestented, the ureter was accessed in 100% of cases for a stone-free rate of 96%. The authors can further validate this report with their own institutional experience using 6.9-Fr flexible and 4.5- and 6.5-Fr semirigid ureteroscopy; however, it is their preference to sequentially dilate with the 8/10 coaxial dilator even in very young children. If the authors encounter difficulties, a stent is placed rather than dilated more aggressively. This approach appears to minimize the immediate risks and potential long-term complications, particularly in the management of upper tract calculi; however, it increases the number of children who will require a second anesthetic and procedure to achieve stone-free status. Recent reports suggest that up to 40% of pediatric patients will require at least two procedures to treat upper tract calculi, suggesting that the likelihood of achieving a stone-free status after one ureteroscopic procedure may not be significantly better than with shock wave lithotripsy (Corcoran et al, 2007). Additional procedures were reported with more than half of the stones that were 6 mm or larger (Tanaka et al, 2008).
The necessity of placing a stent after ureteroscopy in all children has also been debated. Although, in large series, the tendency has been to leave a stent in place after ureteroscopic manipulation in a majority of children (Smaldone et al, 2007), several authors have reported no acute or long-term sequelae despite leaving a postoperative stent in less than 20% of cases (Herndon et al, 2006). In the authors’ experience, the decision to place a postprocedural stent is made on an individual patient basis and is dependent on the surgeon’s experience and degree of visible ureteral trauma at the conclusion of the procedure.
Percutaneous Nephrolithotomy
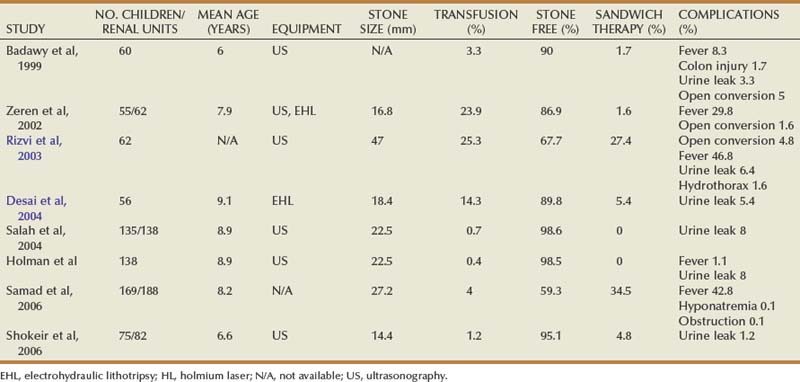
The safety and efficacy of percutaneous nephrolithotomy for large stone burdens have been well established in adults. Initially, urologists were reluctant to perform PCNL in children due to concerns regarding the use of large instruments in pediatric kidneys, parenchymal damage and the associated effects on renal function, radiation exposure with fluoroscopy, and the risks of major complications, including sepsis and bleeding. Another deterrent to performing PCNL in children was the potential resultant sequelae of hypothermia (i.e., coagulopathy) from prolonged exposure to irrigation. With significantly accumulated experience, however, PCNL is currently being used as monotherapy and in combination with SWL (sandwich therapy) in children, achieving stone-free rates ranging from 68% to 100% (Table 135–3) (Rizvi et al, 2003; Mahmud and Zaidi, 2004). Although there is no current international consensus, relative indications for PCNL as the primary treatment modality in children include large upper tract stone burden (>1.5 cm), lower pole calculi greater than 1 cm, concurrent anatomic abnormality impairing urinary drainage and stone clearance, or known cystine or struvite composition (Wu and Docimo, 2004; Farhat and Kropp, 2007).
The earliest pediatric PCNL series were performed using standard adult-sized instruments. In 1985, Woodside and colleagues (1985) first reported on a series of seven patients (ages 5 to 18 years old) who were rendered stone free without complications when using adult percutaneous techniques and equipment. Years later in 1997, Mor and colleagues (1997) also reported on using adult-sized instruments in their pediatric PCNL series. The use of PCNL in these early series was avoided in very small children (<5 years old) due to concerns regarding parenchymal damage. In contrast to these concerns, multiple series subsequently used adult-sized instruments and reported high efficacy rates with acceptable complication rates even when the dilating tract size as high as 30 Fr (Salah et al, 2004; Samad et al, 2006; Bilen et al, 2007). In fact, recent data have suggested that PCNL is possible in very young children using adult-sized equipment (Nouralizadeh et al, 2009). Investigations regarding the risk of renal damage in pediatric patients treated with PCNL have revealed that there is no significant risk of renal functional loss. Mor and colleagues (1997) performed radioisotope scans on 10 children before and after PCNL and found no change in differential function and no evidence of significant scarring.
Despite early successes employing adult equipment, early efforts focused on developing technology to minimize percutaneous tract size without affecting PCNL efficacy. Jackman and colleagues (1998), developed a novel percutaneous access technique (“mini-perc”) using an 13-Fr peel-away vascular access sheath and reported an 85% stone-free rate for 11 procedures in seven children with a mean age of 3.4 years and an average stone burden of 1.2 cm2 (Fig. 135–6). With this technique, PCNL could now be performed in preschool-aged children. The benefits of minimal tract dilation included increased maneuverability, decreased blood loss, and shorter hospital stay. However, theoretical limitations, including prolonged operative times and impaired visualization from bleeding, suggests that this technique may not be adequate for very large stone burdens.
Figure 135–6 The “mini-perc” set uses an 11- to 13-Fr peel-away vascular access sheath to accommodate a ureteroscope or cystoscope, serving as a nephroscope, to perform percutaneous nephrolithotomy (PCNL) in children of preschool age.
Recent advances in instrumentation, such as smaller nephroscopes (15- to 18-Fr) and more efficient energy sources for intracorporeal lithotripsy, including the holmium : YAG laser and smaller pneumatic lithoclast and ultrasound probes, have greatly facilitated percutaneous treatment techniques in pediatric patients (Fig. 135–7). As a result, PCNL has now replaced open surgery as the treatment of choice for large stone burdens in children of all ages.

Figure 135–7 Smaller-caliber nephroscopes (15 to 18 Fr) with offset lenses have greatly facilitated standard percutaneous treatment techniques in pediatric patients.
(From Wolf JS Jr, et al. Best practice policy statement on urologic surgery antimicrobial prophylaxis. J Urol 2008;179[4]:1379–90.)
Planning for PCNL in Children
Much planning is required prior to embarking on PCNL in children. Films must first be reviewed scrupulously to determine if the stone(s) is/are amenable to a percutaneous procedure. Nephrocalcinosis in children, for example, may be confused with staghorn calculi; the etiology and treatment are very different (Fig. 135–8). Infection is the most common causative agent in the formation of a staghorn calculus, and PCNL is a first-line therapy. The most frequent causes of nephrocalcinosis are hereditary tubulopathies and vitamin D intoxication (Ammenti et al, 2009). Medullary sponge kidney is a renal malformation characterized by cystic anomalies of precalyceal ducts, which is frequently associated with nephrocalcinosis and stone formation (Gambaro et al, 2006). Nephrocalcinosis is most often not amenable to successful endourologic treatment(s) because calculi are intraparenchymal and thus outside the collecting system. Management is often medical and aimed at prevention of further nephrocalculi, a cause of worsening renal function.
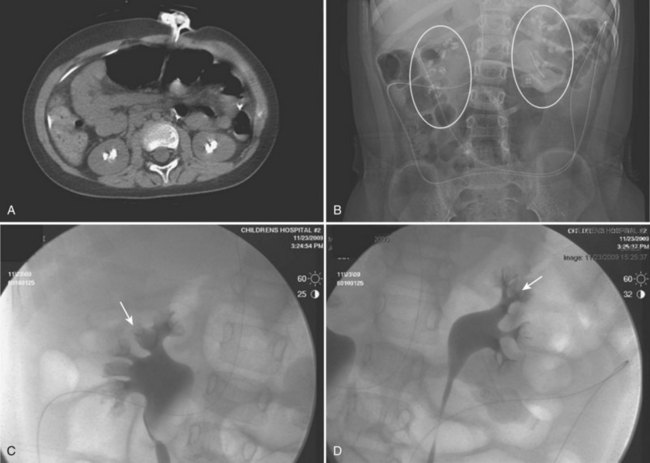
Figure 135–8 A 9-year-old boy with cerebral palsy and gross hematuria. CT scan demonstrated bilateral nephrolithiasis (A) thought to be staghorn calculi. Plain radiograph (B) and bilateral retrograde pyelograms (C and D) correlate to show nephrocalcinosis (white circles and white arrows, respectively) reflective of medullary sponge kidney disease. Subsequent metabolic workup revealed renal tubular acidosis (RTA) with hypercalciuria.
The risks of the PCNL must be reviewed with the consenting parent or guardian. It must be understood that a percutaneous procedure carries risks that are not limited to bleeding (requiring transfusion), delayed renal hemorrhage (requiring angioablation), sepsis, pneumothorax, hemothorax, urothorax, incomplete stone treatment, and injuries to organs adjacent to each respective kidney.
Every attempt should be made to treat a urinary tract infection and/or minimize bacteriuria prior to the procedure. A urine culture, with antibiotic sensitivities, should be checked 3 weeks prior to the procedure. Any positive culture requires a full course of antibiotics and repeat culture to confirm sterility. Three to 5 days of prophylactic antibiotics is recommended prior to the procedure, even with a negative preoperative culture. Broad-spectrum IV antibiotics (i.e., ampicillin and gentamicin) should be given at the time of the procedure.
All percutaneous procedures are performed using general anesthesia. A warm operating room, warmed isotonic irrigation solution, short operative times (not to exceed 1.5 hours), proper draping, and monitoring of body temperature should decrease the incidence of hypothermia and hyponatremia. After induction of anesthesia with the patient in the lithotomy position, a retrograde pyelogram is performed to outline the collecting system, and an occlusive balloon or a 5-Fr open-ended ureteral catheter is left in situ to opacify the collecting system during percutaneous access. The patient is then repositioned in the prone position with the torso elevated at 30 degrees from the table surface with a towel roll (Farhat and Kropp, 2007).
Circumstances that require special consideration involve children with spinal cord injuries and congenital anomalies such as spina bifida. In these patients, positioning can be a challenge due to existing spinal hardware and limb contracture (Fig. 135–9) (Ost and Lee, 2006a). Patients who have had prior spinal surgery consisting of vertebral fusion or Harrington rod placement will have restricted spinal mobility, spinal curvature, and/or atrophic or contracted extremities (Fig. 135–10). As the result of scoliosis, lordosis, and/or kyphosis, renal anatomy is altered. Consequently, the risk of injuries to adjacent organs (i.e., pneumothorax) during percutaneous procedures increases. Assessing the degree of mobility in the trunk and extremities is crucial in planning for PCNL in these patients (Fig. 135–11). These patients must be placed in the most comfortable position possible without excessive contortion or flexion of the joints. Special attention must be paid to latex precautions in the myelomeningocele population, and, as in all cases, proper padding of pressure points is mandatory.
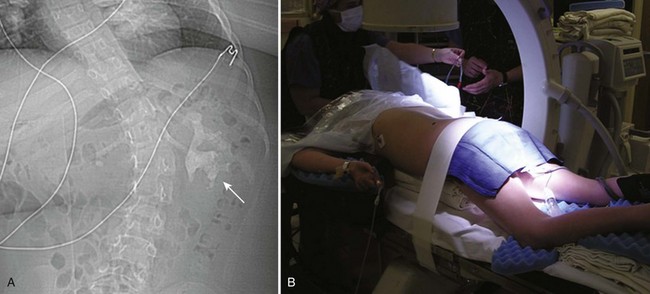
Figure 135–9 A, Severe scoliosis and complete staghorn calculus (arrow) in a 10-year-old partial quadriplegic. B, Spinal curvature and limb contractures (left arm) prevented optimal prone positioning. Care is taken to pad all joints in the prone position.
Percutaneous Nephrolithotomy Technique in Children
After selection of the desired calyx, a 16- or 18-gauge spinal needle is placed with the assistance of fluoroscopy in the 30-degree position. When fluoroscopy is used, the collecting system is opacified through an open-ended ureteral catheter that has been inserted cystoscopically prior to positioning in the prone position. Alternatively, ultrasound guidance can be used to puncture the calyx of choice. The ideal tract is one that provides the shortest and most direct access to the stone. For complex calculi occupying multiple calices, including the lower pole, a supracostal posterior access is preferred to provide visualization of the superior calyx and pelvis, access to the pelvis and ureter, and straight access to the inferior calices, allowing easier manipulation of the working instruments and minimizing torque on the collecting system (El-Nahas et al, 2008). Following initial puncture, to avoid trauma, no attempt should be made to redirect the needle while it is located within the cortex of the kidney. With the C-arm in the 90-degree plane, the depth and medial extension of the needle is checked. After access is confirmed with urine or irrigation return, a flexible guidewire is placed into the collecting system through the needle and directed down the ureter into the bladder. A small skin incision is made with a No. 11 scalpel and 8- and 10-Fr coaxial dilators are passed over the guidewire into the collecting system. Once in place, an Amplatz Super Stiff guidewire (AGA Medical, Plymouth, MN) is placed as a working wire.
Tract dilation can be performed by several techniques. Serial dilation with Amplatz dilators over working wires and subsequent sheath placement under fluoroscopic guidance is the most common technique employed. For smaller children and lower stone burdens, an 11- to 13-Fr peel-away sheath (Docimo Mini-Perc, Cook Urological, Spencer, IN) and trocar are passed over the wire and through the calyx under fluoroscopic guidance. For balloon dilation, either the Bard X-Force or NephroMax High-Pressure Nephrostomy Balloon Catheter (Bard Medical, Covington, GA; Boston Scientific, Natick, MA) may be used. Either will facilitate dilation of a 30-Fr tract at a pressure of 17 atmospheres. This technique permits dilation and sheath placement in a single step, thereby minimizing potential parenchymal trauma and bleeding from sequential dilation with rigid dilators. Although the decision to proceed with mini-perc or dilation is individualized based on the child’s age, anatomy, and stone burden, familiarity with all of the above techniques facilitates complete access with minimal morbidity (Fig. 135–12A and B).
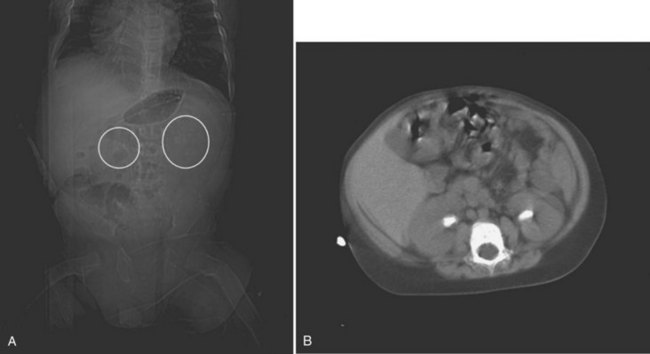
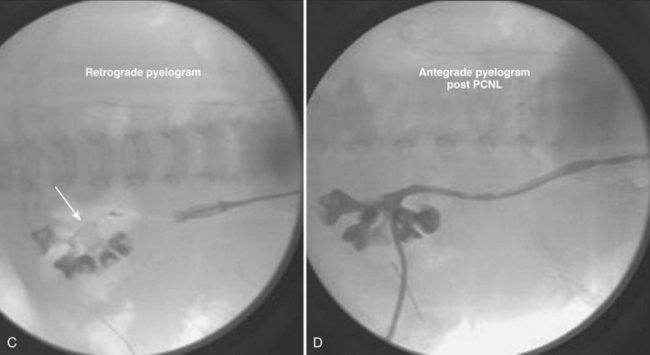
Figure 135–12 A 22-month-old boy with intractable diarrhea of infancy from tufting enteropathy who awaited a small bowel transplant. His risks for nephrolithiasis included total parenteral nutrition dependency, osteomalacia, chronic dehydration, hypocitruria, and hyperoxaluria. A, A kidney-ureter-bladder film demonstrated mildly radiopaque stones, and a subsequent CT scan (B) confirmed bilateral staghorn calculi. C, Prone view of left retrograde pyelogram of child in A, demonstrating staghorn calculi as a filling defect (arrow). A “mini-perc” technique was used to address his stones. D, Prone view of the antegrade nephrostogram through a 6-Fr nephrostomy tube demonstrating “stone-free” status.
Once access is obtained, nephroscopy and nephrolithotomy can be performed with a variety of energy sources for stone fragmentation. The outer diameter of nephroscopes range from 15 to 26 Fr, and a 15-Fr flexible nephroscope with a 6-Fr working channel has also been developed. In addition, 7- and 8-Fr offset cystoscopes with 5-Fr working ports and 7- to 9-Fr flexible ureteroscopes can be used through an 11-Fr access sheath with enough clearance to allow low pressure irrigation (Wu and Docimo, 2004). Energy sources currently used include ultrasonic lithotripsy, electrohydraulic lithotripsy (EHL), and the holmium laser, although individual preference is determined by availability and the surgeon’s experience. Postoperative stenting and/or placement of a nephrostomy tube are both patient and surgeon dependent and vary between series. Similar to adult procedures, tubeless PCNL has theoretical advantages, including decreased postoperative pain and a short hospital stay, but currently, data is limited in the pediatric population (Khairy Salem et al, 2007).
Percutaneous Nephrolithotomy Outcomes
Recent large retrospective series of PCNL monotherapy have demonstrated high efficacy rates approaching 90% (see Table 135–3) (Zeren et al, 2002; Desai et al, 2004; Bilen et al, 2007). In 56 children (mean age 9.1 years) with a mean stone burden of 337.5 mm2, Desai and colleagues (2004) reported a stone-free rate of 89.8% using EHL through a 14-Fr nephroscope and a 20- to 24-Fr sheath. Of these, 61% required multiple tracts, and 45% were staged procedures. Findings demonstrated that the number and size of tracts were significantly associated with postoperative hemoglobin decrease (mean 1.9 g/dL) and an overall transfusion rate of 14%. In 52 children with a mean age of 7.9 years and a mean stone burden of 282 mm2, Zeren and colleagues (2002) reported an 87% stone-free rate using US and EHL for fragmentation and tract dilation from 18 to 30 Fr. Complications included postoperative fever (30%) and the need for transfusion (24%). Transfusion was associated with operative time, sheath size, and stone burden. In 135 children of mean age 8.9 years with a mean stone burden of 507 mm2, Salah and colleagues (2004) reported a 98.5% stone-free rate using US through a 26-Fr nephroscope. Complications were low (8% urine leak rate and 0.7% transfusion rate), with only one patient requiring a second procedure. In a recent series of 46 children with a mean stone burden of 332 mm2, Bilen and colleagues (2007) reported an 88% stone-free rate using EHL, US, and the holmium laser. When stratified by tract size (14, 20, and 24 Fr), efficacy rates were similar in all groups, but there were no complications or transfusions in the 14-Fr tract group.
In an effort to reduce the number of tracts and associated morbidity, some centers have chosen to follow primary PCNL with adjunctive SWL therapy to clear residual stone fragments. In a small series of 29 children with a mean age of 3.8 years and a mean stone burden of 2.4 cm, Mahmud and Zaidi (2004) reported a 60% stone-free rate after PCNL monotherapy using EHL through a 17-Fr angled nephroscope. Only one tract was used in all patients, and after SWL sandwich therapy, the stone-free rate increased to 100%. In a larger series of 169 children with a mean stone burden of 3.1 cm, Samad and colleagues (2006) reported a 59% monotherapy stone-free rate with 96% of cases performed through a single tract. Approximately one third (34.5%) of primary failures were treated with SWL; the cumulative stone-free rate in all patients was 93.8% with a 3.6% transfusion rate. When stratified by age, anatomy, bilaterality, and renal function, stone-free outcomes were equivalent in all groups. The decision to follow PCNL with SWL is related to operator experience with percutaneous technique and available technology. It is the authors’ preference to perform a second-look nephroscopy through the original tract to ensure stone-free status during the initial hospital admission, rather than progress to SWL sandwich therapy. Endoscopic surveillance during the initial procedure can determine the need for second-look nephroscopy without relying on additional imaging and the associated risks of radiation exposure (Roth et al, 2009). With continued improvement in technology and technique, the indications for PCNL in children will continue to increase. Given the technically challenging nature of PCNL, the surgeon’s experience is paramount in developing individualized treatment plans to optimize efficacy with minimal morbidity.
Laparoscopic and Robotic-Assisted Pyelolithotomy
Treatment of large stone burdens in children is technically challenging, often requiring multiple procedures. Laparoscopy and robotic-assisted laparoscopy have been used successfully in adults for treatment of calculi during the concomitant treatment of ureteropelvic junction obstruction and in the primary treatment of staghorn calculi. Small series using these techniques in children have only recently been described. In eight children (mean age 4 years) with a mean stone burden of 2.9 cm and who underwent transperitoneal laparoscopic pyelolithotomy, Casale and colleagues (2004) reported a 100% success rate, a mean hospital stay of 2.15 days, and a mean operative time of 1.6 hours with no major complications. In the first report of robotic-assisted laparoscopic pyelolithotomy, Lee and colleagues (2007) described their experience in five patients, four with cystine staghorn calculi refractory to PCNL and SWL and one with calcium oxalate calculi and concurrent ureteropelvic junction obstruction. Of these cases, four were completed robotically, with one patient having a residual 6-mm lower pole stone and one patient requiring conversion to an open procedure. Mean operative time in this series was 315 minutes, mean estimated blood loss was less than 20 cc, and the mean hospital length of stay was 3.8 days. These early experiences demonstrate that laparoscopic pyelolithotomy is feasible, safe, and efficacious as an alternative to open pyelolithotomy in children and warrants further study. However, due to their demanding technical nature, these procedures will likely be limited to endourologic management failures in academic centers with abundant expertise in laparoscopic and robotic pediatric surgery. The indications for use of these technologies are also limited to particularly complex and large stone burden patients.
Percutaneous Cystolithotripsy for Bladder Stones
Bladder stones are more often found in children from developing countries and are thought to be related endemically to malnutrition. It is thought that diets low in animal protein and phosphorous (breast milk as opposed to cow’s milk), in addition to vitamin A deficiency, are contributory (Kancha and Anasuya, 1992). Bladder stones from children in these developing countries are most often composed of ammonium acid urate. In contrast, among children from industrialized nations, bladder stones are most often found in those with spinal cord injuries or congenital abnormalities such as spina bifida. Very often, these children have undergone augmentation cystoplasty and/or manage their bladders by clean intermittent catheterization. It has been reported that up to 50% of children with reconstructed bladders will develop a bladder stone(s) in their lifetime (Palmer et al, 1993). Urinary stasis, bacterial colonization or infection with urea-splitting organisms, retained mucus, and foreign bodies can all contribute to the formation of bladder stones, the majority of which are struvite.
Open cystolithotomy has been the traditional modality to treat bladder stones. Transurethral cystolithotripsy is an alternative, although not ideal in the pediatric population. In children, a smaller-caliber urethra limits effective treatment of large bladder stone burdens. Percutaneous cystolithotripsy, however, is now used worldwide with the advantage of shorter hospital stays, smaller scars, and less indwelling catheter time postoperatively (Al-Marhoon et al, 2009). Presently, percutaneous cystolithotripsy is the preferred method to treat bladder stones that have formed in reconstructed bladders (Paez et al, 2007). In developing countries as well, percutaneous lithotripsy has become the first-line treatment modality to address bladder stones in bladders that have not been augmented. Salah and colleagues (2005) reported on their experience with cystolithotripsy in 155 children from Pakistan and Yemen with a mean age of 4.5 years and average bladder stone burden of 2.3 cm (range 0.7 to 4 cm). All children were treated safely and successfully using a 26-Fr nephroscope through a 30-Fr sheath placed through a 1-cm suprapubic incision.
Percutaneous cystolithotripsy has also been used effectively in infants less than 1 year old to clear bladder stones. Gan and colleagues (2010), reported on their experience using a 16-Fr peel- away sheath with a ureteroscope to treat bladder stones with an average size of 1.4 cm in 15 boys with a mean age of 8.2 months.
Percutaneous cystolithotripsy in children may be performed under ultrasound or fluoroscopic guidance and is most often an outpatient procedure. With either modality, the bladder is first filled to capacity with water or contrast, respectively. The child is placed in the Trendelenburg position to minimize the risk of bowel injury during access and tract dilation/formation. An 18-gauge needle is placed into the distended bladder midline, 1 to 2 fingerbreadths above the pubic bone. Once proper placement is confirmed with return of fluid, a wire is passed through the needle into the bladder. A tract may then be established reflective of stone diameter and the child’s size. Most often, a tract is established with a balloon dilator to accompany a 30-Fr sheath. Amplatz dilators may also be used. A 26-Fr nephroscope is used to extract stones less than 1 cm using a rigid stone forceps, or an ultrasonic lithotripter may be used to fragment stones greater than 1 cm. At the conclusion of the percutaneous cystolithotripsy procedure, a Foley catheter is left per urethra or per continent catheterizable stoma for 1 week. The rectus fascia defect is closed with a 2-0 Vicryl suture (Fig. 135–13).
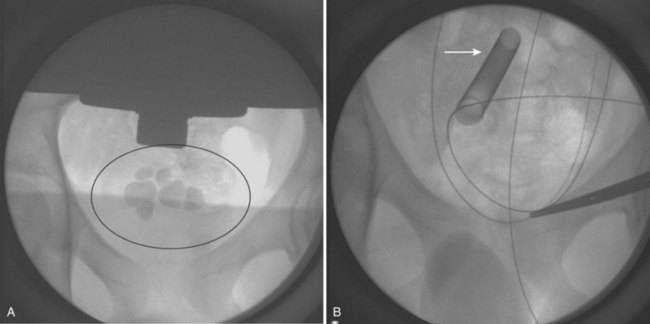
Figure 135–13 A 12-year-old male with spina bifida with an augmented bladder (ilealcystoplasty) found to have multiple sizeable bladder stones (A) on a workup for recurrent Proteus mirabilis infections. B, Percutaneous cystolithotripsy with an ultrasonic lithotripter was performed through a 30-Fr access sheath (arrow), clearing all stones from the augmented bladder.
Determination of Stone-Free Status
As the surgical management of pediatric stone disease evolves, the lack of a consistent definition of “stone free” following definitive therapy is an issue that remains unaddressed. Although controversial, in select adult patients, all stone fragments can be considered clinically significant and can lead to stone recurrence (Krambeck et al, 2008). Likewise, the presence of residual stone fragments in children has been associated with poor outcomes (Afshar et al, 2004), and any size stone fragment in a young stone former may result in the need for repeat surgical procedures. However, these fragments often are not detected on US or KUB, thus necessitating reliance on CT imaging in select children.
Balancing the risks of radiation exposure for post-treatment stone detection and the risks of anesthesia for secondary procedures is a challenging dilemma for contemporary pediatric endourologists. Newer, high-speed helical CT scanners reduce radiation exposure and rarely require intravenous sedation. In addition, maximizing intraoperative fragment detection by direct visualization in URS and PCNL and continued development of high-resolution real-time fluoroscopy may result in less reliance on postoperative imaging and decreased need for second-look nephroscopy/URS, SWL, or sandwich therapy (Ost and Smith, 2006b). Until the risks of radiation exposure in children are more clearly defined, surveillance in these children will be individualized based on age, anatomy, stone burden, and underlying metabolic abnormalities.
Conclusions
Evolution of techniques and miniaturization of instruments have changed the management of pediatric stone disease. However, despite encouraging results, concern remains regarding the safety of endourologic treatment in smaller patients and its subsequent effects on the developing kidney. Although SWL is still considered first-line therapy for upper tract calculi less than 1.5 cm, evidence is accumulating that ureteroscopy with laser lithotripsy and stone basketing may be more efficacious in treating upper tract stone disease in children. Although PCNL remains the most effective technique for large upper tract stone burdens, there are now reports of laparoscopic and robotic-assisted laparoscopic pyelolithotomy in major pediatric academic centers with extensive laparoscopic and robotic experience. Prospective studies designed to determine “the preferred” endourologic approach to upper tract calculi in children would be helpful, albeit difficult to conduct. In this regard, the individual surgeon’s experience and comfort level will weigh heavily in choosing a treatment modality. Pediatric urologists’ familiarity with percutaneous renal access and the full spectrum of endourologic equipment and techniques will continue to facilitate efficacious minimally invasive approaches for the entire pediatric urinary tract.
Acknowledgment
The authors would like to acknowledge Marc C. Smaldone, MD for his help in preparing this chapter.
Al-Busaidy SS, Prem AR, Medhat M. Pediatric staghorn calculi: the role of extracorporeal shock wave lithotripsy monotherapy with special reference to ureteral stenting. J Urol. 2003;169(2):629-633.
Alon US. Medical treatment of pediatric urolithiasis. Pediatr Nephrol. 2009;24(11):2129-2135.
Ammenti A, et al. Nephrocalcinosis in children: a retrospective multi-centre study. Acta Paediatr. 2009;98(10):1628-1631.
Desai MR, et al. Percutaneous nephrolithotomy for complex pediatric renal calculus disease. J Endourol. 2004;18(1):23-27.
Gofrit ON, et al. Is the pediatric ureter as efficient as the adult ureter in transporting fragments following extracorporeal shock wave lithotripsy for renal calculi larger than 10 mm? J Urol. 2001;166(5):1862-1864.
Lottmann HB, et al. Monotherapy extracorporeal shock wave lithotripsy for the treatment of staghorn calculi in children. J Urol. 2001;165(6 Pt. 2):2324-2327.
McLorie GA, et al. Safety and efficacy of extracorporeal shock wave lithotripsy in infants. Can J Urol. 2003;10(6):2051-2055.
Palmer JS, et al. Diagnosis of pediatric urolithiasis: role of ultrasound and computerized tomography. J Urol. 2005;174(4 Pt. 1):1413-1416.
Rizvi SA, et al. Management of pediatric urolithiasis in Pakistan: experience with 1,440 children. J Urol. 2003;169(2):634-637.
Smaldone MC, et al. Is ureteroscopy first line treatment for pediatric stone disease? J Urol. 2007;178(5):2128-2131. discussion 2131
Van Savage JG, et al. Treatment of distal ureteral stones in children: similarities to the American Urological Association guidelines in adults. J Urol. 2000;164(3 Pt. 2):1089-1093.
Afshar K, et al. Outcome of small residual stone fragments following shock wave lithotripsy in children. J Urol. 2004;172(4 Pt. 2):1600-1603.
Aksoy Y, et al. Extracorporeal shock wave lithotripsy in children: experience using a mpl-9000 lithotriptor. World J Urol. 2004;22(2):115-119.
Al-Busaidy SS, Prem AR, Medhat M. Pediatric staghorn calculi: the role of extracorporeal shock wave lithotripsy monotherapy with special reference to ureteral stenting. J Urol. 2003;169(2):629-633.
Al-Marhoon MS, et al. Comparison of endourological and open cystolithotomy in the management of bladder stones in children. J Urol. 2009;181(6):2684-2687. discussion 2687–8
Aldridge RD, Aldridge RC, Aldridge LM. Anesthesia for pediatric lithotripsy. Paediatr Anaesth. 2006;16(3):236-241.
Alon US. Medical treatment of pediatric urolithiasis. Pediatr Nephrol. 2009;24(11):2129-2135.
Ammenti A, et al. Nephrocalcinosis in children: a retrospective multi-centre study. Acta Paediatr. 2009;98(10):1628-1631.
Ather MH, Noor MA. Does size and site matter for renal stones up to 30-mm in size in children treated by extracorporeal lithotripsy? Urology. 2003;61(1):212-215. discussion 215
Aydogdu O, et al. Effectiveness of doxazosin in treatment of distal ureteral stones in children. J Urol. 2009;182(6):2880-2884.
Badawy H, et al. Percutaneous management of renal calculi: experience with percutaneous nephrolithotomy in 60 children. J Urol. 1999;162(5):1710-1713.
Bassiri A, et al. Transureteral lithotripsy in pediatric practice. J Endurol. 2002;16(4):257-260.
Battino BS, et al. Metabolic evaluation of children with urolithiasis: are adult references for supersaturation appropriate? J Urol. 2002;168(6):2568-2571.
Bilen CY, et al. Percutaneous nephrolithotomy in children: lessons learned in 5 years at a single institution. J Urol. 2007;177(5):1867-1871.
Brisse HJ, et al. The relevance of image quality indices for dose optimization in abdominal multi-detector row CT in children: experimental assessment with pediatric phantoms. Phys Med Biol. 2009;54(7):1871-1892.
Buscarini M, Conlin M. Update on flexible ureteroscopy. Urol Int. 2008;80(1):1-7.
Cannon GM, et al. Ureteroscopic management of lower-pole stones in a pediatric population. J Endourol. 2007;21(10):1179-1182.
Casale P, et al. Transperitoneal laparoscopic pyelolithotomy after failed percutaneous access in the pediatric patient. J Urol. 2004;172(2):680-683. discussion 683
Cody DD, et al. Strategies for formulating appropriate MDCT techniques when imaging the chest, abdomen, and pelvis in pediatric patients. AJR Am J Roentgenol. 2004;182(4):849-859.
Cohen MD. Pediatric CT radiation dose: how low can you go? AJR Am J Roentgenol. 2009;192(5):1292-1303.
Conway PH, et al. Recurrent urinary tract infections in children: risk factors and association with prophylactic antimicrobials. JAMA. 2007;298(2):179-186.
Corcoran AT, et al. Flexible ureteroscopy for proximal stones in pediatric patients: how complete access simplifies the surgical approach. J Endourol. 2007;21(Suppl 1):A84.
De Dominicis M, et al. Retrograde ureteroscopy for distal ureteric stone removal in children. BJU Int. 2005;95(7):1049-1052.
DeFoor W, et al. Results of a prospective trial to compare normal urine supersaturation in children and adults. J Urol. 2005;174(4 Pt. 2):1708-1710.
DeFoor W, et al. Use of mobile extracorporeal shock wave lithotripter: experience in a pediatric institution. Urology. 2005;65(4):778-781.
DeFoor W, et al. Urinary metabolic evaluations in normal and stone forming children. J Urol. 2006;176(4 Pt. 2):1793-1796.
Demirkesen O, et al. Efficacy of extracorporeal shock wave lithotripsy for isolated lower caliceal stones in children compared with stones in other renal locations. Urology. 2006;67(1):170-174. discussion 174–5
Desai MR, et al. Percutaneous nephrolithotomy for complex pediatric renal calculus disease. J Endourol. 2004;18(1):23-27.
El-Nahas AR, et al. Safety and efficacy of supracostal percutaneous nephrolithotomy in pediatric patients. J Urol. 2008;180(2):676-680.
Elsobky E, et al. Extracorporeal shock wave lithotripsy in children: experience using two second-generation lithotripters. BJU Int. 2000;86(7):851-856.
Farhat WA, Kropp BP. Surgical treatment of pediatric urinary stones. AUA Update Series. 2007;26(3):22-28.
Fowler KA, et al. US for detecting renal calculi with nonenhanced CT as a reference standard. Radiology. 2002;222(1):109-113.
Gambaro G, et al. Medullary sponge kidney (Lenarduzzi-Cacchi-Ricci disease): a Padua Medical School discovery in the 1930s. Kidney Int. 2006;69(4):663-670.
Gan W, et al. Minimally invasive percutaneous cystolithotomy: an effective treatment for bladder stones in infants aged <1 year. BJU Int. 2010;106(2):275-277.
Gofrit ON, et al. Is the pediatric ureter as efficient as the adult ureter in transporting fragments following extracorporeal shock wave lithotripsy for renal calculi larger than 10 mm? J Urol. 2001;166(5):1862-1864.
Hamm M, et al. Unenhanced helical computed tomography in the evaluation of acute flank pain. Eur Urol. 2001;39(4):460-465.
Heneghan JP, et al. Helical CT for nephrolithiasis and ureterolithiasis: comparison of conventional and Viamonte L, Joseph reduced radiation-dose techniques. Radiology. 2003;229(2):575-580.
Herndon CD, Viamonte L, Joseph DB. Ureteroscopy in children: is there a need for ureteral dilation and postoperative stenting? J Pediatr Urol. 2006;2(4):290-293.
Jackman SV, et al. Percutaneous nephrolithotomy in infants and preschool age children: experience with a new technique. Urology. 1998;52(4):697-701.
Jayanthi VR, Arnold PM, Koff SA. Strategies for managing upper tract calculi in young children. J Urol. 1999;162(3 Pt. 2):1234-1237.
Kancha RK, Anasuya A. Contribution of vitamin A deficiency to calculogenic risk factors of urine: studies in children. Biochem Med Metab Biol. 1992;47(1):1-9.
Karmazyn B, et al. CT with a computer-simulated dose reduction technique for detection of pediatric nephroureterolithiasis: comparison of standard and reduced radiation doses. AJR Am J Roentgenol. 2009;192(1):143-149.
Khairy Salem H, et al. Tubeless percutaneous nephrolithotomy in children. J Pediatr Urol. 2007;3(3):235-238.
Kim S, et al. Radiation dose from cone beam CT in a pediatric phantom: risk estimation of cancer incidence. AJR Am J Roentgenol. 2010;194(1):186-190.
Krambeck AE, et al. Diabetes mellitus and hypertension associated with shock wave lithotripsy of renal and proximal ureteral stones at 19 years of followup. J Urol. 2006;175(5):1742-1747.
Krambeck AE, et al. Long-term outcomes of percutaneous nephrolithotomy compared to shock wave lithotripsy and conservative management. J Urol. 2008;179(6):2233-2237.
Kurien A, et al. Extracorporeal shock wave lithotripsy in children: equivalent clearance rates to adults is achieved with fewer and lower energy shock waves. BJU Int. 2009;103(1):81-84.
Landau EH, et al. Extracorporeal shock wave lithotripsy in prepubertal children: 22-year experience at a single institution with a single lithotriptor. J Urol. 2009;182(Suppl 4):1835-1839.
Lande MB, et al. Role of urinary supersaturation in the evaluation of children with urolithiasis. Pediatr Nephrol. 2005;20(4):491-494.
Lee RS, et al. Early results of robot assisted laparoscopic lithotomy in adolescents. J Urol. 2007;177(6):2306-2309. discussion 2309–10
Lesani OA, Palmer JS. Retrograde proximal rigid ureteroscopy and pyeloscopy in prepubertal children: safe and effective. J Urol. 2006;176(4 Pt. 1):1570-1573.
Lingeman JE, et al. Shockwave lithotripsy: anecdotes and insights. J Endourol. 2003;17(9):687-693.
Lottmann HB, et al. Monotherapy extracorporeal shock wave lithotripsy for the treatment of staghorn calculi in children. J Urol. 2001;165(6 Pt. 2):2324-2327.
Mahmud M, Zaidi Z. Percutaneous nephrolithotomy in children before school age: experience of a Pakistani centre. BJU Int. 2004;94(9):1352-1354.
McLorie GA, et al. Safety and efficacy of extracorporeal shock wave lithotripsy in infants. Can J Urol. 2003;10(6):2051-2055.
Minevich E, et al. Ureteroscopy is safe and effective in prepubertal children. J Urol. 2005;174(1):276-279. discussion 279
Mir S, Serdaroglu E. Quantification of hypercalciuria with the urine calcium osmolality ratio in children. Pediatr Nephrol. 2005;20(11):1562-1565.
Mor Y, et al. The role of percutaneous nephrolithotomy in the management of pediatric renal calculi. J Urol. 1997;158(3 Pt. 2):1319-1321.
Muslumanoglu AY, et al. Extracorporeal shock wave lithotripsy as first line treatment alternative for urinary tract stones in children: a large scale retrospective analysis. J Urol. 2003;170(6 Pt. 1):2405-2408.
Myers DA, et al. Pediatric low energy lithotripsy with the Lithostar. J Urol. 1995;153(2):453-457.
Nelson CP, et al. Extracorporeal shock wave lithotripsy in pediatric patients using a late generation portable lithotriptor: experience at Children’s Hospital Boston. J Urol. 2008;180(Suppl 4):1865-1868.
Newman DM, et al. Extracorporeal shock wave lithotripsy experience in children. J Urol. 1986;136(1 Pt. 2):238-240.
Nimkin K, et al. Urolithiasis in a children’s hospital: 1985-1990. Urol Radiol. 1992;14(3):139-143.
Nouralizadeh A, et al. Experience of percutaneous nephrolithotomy using adult-size instruments in children less than 5 years old. J Pediatr Urol. 2009;5(5):351-354.
Onal B, et al. The impact of caliceal pelvic anatomy on stone clearance after shock wave lithotripsy for pediatric lower pole stones. J Urol. 2004;172(3):1082-1086.
Ost MC, Lee BR. Urolithiasis in patients with spinal cord injuries: risk factors, management, and outcomes. Curr Opin Urol. 2006;16(2):93-99.
Ost MC, Smith AD. Editorial comment: intraoperative fragment detection during percutaneous nephrolithotomy: evaluation of high magnification rotational fluoroscopy combined with aggressive nephroscopy. J Urol. 2006;175(1):165-166.
Ozgur Tan M, et al. The impact of radiological anatomy in clearance of lower calyceal stones after shock wave lithotripsy in paediatric patients. Eur Urol. 2003;43(2):188-193.
Paez E, et al. Percutaneous treatment of calculi in reconstructed bladder. J Endourol. 2007;21(3):334-336.
Palmer JS, et al. Diagnosis of pediatric urolithiasis: role of ultrasound and computerized tomography. J Urol. 2005;174(4 Pt. 1):1413-1416.
Palmer LS, et al. Urolithiasis in children following augmentation cystoplasty. J Urol. 1993;150(2 Pt. 2):726-729.
Passerotti C, Chow JS, Silva A, et al. Ultrasound versus computerized tomography for evaluating urolithiasis. J Urol. 2009;182:1829-1834.
Porpiglia F, et al. Nifedipine versus tamsulosin for the management of lower ureteral stones. J Urol. 2004;172(2):568-571.
Raza A, et al. Pediatric urolithiasis: 15 years of local experience with minimally invasive endourological management of pediatric calculi. J Urol. 2005;174(2):682-685.
Ritchey M, et al. A case of pediatric ureteroscopic lasertripsy. J Urol. 1988;139(6):1272-1274.
Rizvi SA, et al. Management of pediatric urolithiasis in Pakistan: experience with 1,440 children. J Urol. 2003;169(2):634-637.
Roth CC, et al. Use of second look nephroscopy in children undergoing percutaneous nephrolithotomy. J Urol. 2009;181(2):796-800.
Salah MA, et al. Percutaneous nephrolithotomy in children: experience with 138 cases in a developing country. World J Urol. 2004;22(4):277-280.
Salah MA, et al. Percutaneous cystolithotomy for pediatric endemic bladder stone: experience with 155 cases from 2 developing countries. J Pediatr Surg. 2005;40(10):1628-1631.
Samad L, Aquil S, Zaidi Z. Paediatric percutaneous nephrolithotomy: setting new frontiers. BJU Int. 2006;97(2):359-363.
Schuster TG, et al. Ureteroscopy for the treatment of urolithiasis in children. J Urol. 2002;167(4):1813. discussion 1815–16
Shokeir AA, et al. Treatment of renal stones In children: a comparison between percutaneous nephrolithotomy and shock wave lithotripsy. J Urol. 2006;176(2):706-710.
Shouman AM, et al. Extracorporeal shock wave lithotripsy monotherapy for renal stones >25 mm in children. Urology. 2009;74(1):109-111.
Singh A, et al. Ureteral access sheath for the management of pediatric renal and ureteral stones: a single center experience. J Urol. 2006;175(3 Pt. 1):1080-1082. discussion 1082
Singh S, et al. Dose reduction and compliance with pediatric CT protocols adapted to patient size, clinical indication, and number of prior studies. Radiology. 2009;252(1):200-208.
Slavkovic A, et al. Extracorporeal shock wave lithotripsy for cystine urolithiasis in children: outcome and complications. Int Urol Nephrol. 2002;34(4):457-461.
Smaldone MC, et al. Is ureteroscopy first line treatment for pediatric stone disease? J Urol. 2007;178(5):2128-2131. discussion 2131
Smergel E, et al. CT urograms in pediatric patients with ureteral calculi: do adult criteria work? Pediatr Radiol. 2001;31(10):720-723.
Smith RC, et al. Acute flank pain: comparison of non-contrast-enhanced CT and intravenous urography. Radiology. 1995;194(3):789-794.
Smith RC, et al. Acute ureteral obstruction: value of secondary signs of helical unenhanced CT. AJR Am J Roentgenol. 1996;167(5):1109-1113.
Smith RC, et al. Diagnosis of acute flank pain: value of unenhanced helical CT. AJR Am J Roentgenol. 1996;166(1):97-101.
Spielmann AL, et al. Decreasing the radiation dose for renal stone CT: a feasibility study of single- and multidetector CT. AJR Am J Roentgenol. 2002;178(5):1058-1062.
Srivastava T, Alon US. Urolithiasis in adolescent children. Adolesc Med Clin. 2005;16(1):87-109.
Stechman MJ, Loh NY, Thakker RV. Genetic causes of hypercalciuric nephrolithiasis. Pediatr Nephrol. 2009;24(12):2321-2332.
Sternberg K, et al. Pediatric stone disease: an evolving experience. J Urol. 2005;174(4 Pt. 2):1711-1714. discussion 1714
Strouse PJ, et al. Non-contrast thin-section helical CT of urinary tract calculi in children. Pediatr Radiol. 2002;32(5):326-332.
Tan AH, et al. Ureteroscopy for pediatric urolithiasis: an evolving first-line therapy. Urology. 2005;65(1):153-156.
Tanaka ST, et al. Pediatric ureteroscopic management of intrarenal calculi. J Urol. 2008;180(5):2150-2153. discussion 2153–4
Van Savage JG, et al. Treatment of distal ureteral stones in children: similarities to the American Urological Association guidelines in adults. J Urol. 2000;164(3 Pt. 2):1089-1093.
Wadhwa P, et al. Critical prospective appraisal of renal morphology and function in children undergoing shockwave lithotripsy and percutaneous nephrolithotomy. J Endourol. 2007;21(9):961-966.
Wolf JrJS, et al. Best practice policy statement on urologic surgery antimicrobial prophylaxis. J Urol. 2008;179(4):1379-1390.
Woodside JR, et al. Percutaneous stone removal in children. J Urol. 1985;134(6):1166-1167.
Wu HY, Docimo SG. Surgical management of children with urolithiasis. Urol Clin North Am. 2004;31(3):589-594. xi
Zeren S, et al. Percutaneous nephrolithotomy in the management of pediatric renal calculi. J Endourol. 2002;16(2):75-78.