POSTERIOR ABDOMINAL REGION
The posterior abdominal region is posterior to the abdominal part of the gastrointestinal tract, the spleen, and the pancreas (Fig. 4.127). This area, bounded by bones and muscles making up the posterior abdominal wall, contains numerous structures that not only are directly involved in the activities of the abdominal contents but also use this area as a conduit between body regions. Examples include the abdominal aorta and its associated nerve plexuses, the inferior vena cava, the sympathetic trunks, and lymphatics. There are also structures originating in this area that are critical to the normal function of other regions of the body (i.e., the lumbar plexus of nerves), and there are organs that associate with this area during development and remain in it in the adult (i.e., the kidneys and suprarenal glands).
Posterior abdominal wall
Bones
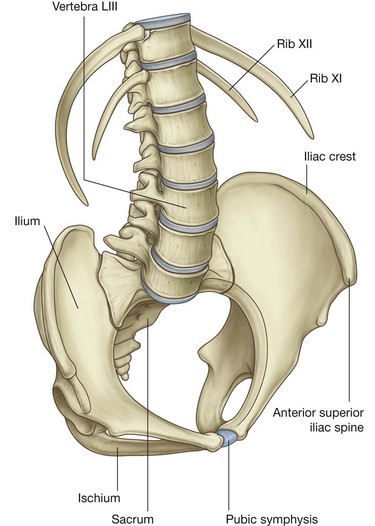
Lumbar vertebrae and the sacrum
Projecting into the midline of the posterior abdominal area are the bodies of the five lumbar vertebrae (Fig. 4.128). The prominence of these structures in this region is due to the secondary curvature (a forward convexity) of the lumbar part of the vertebral column.
The lumbar vertebrae can be distinguished from cervical and thoracic vertebrae because of their size. They are much larger than any other vertebrae in any other region. The vertebral bodies are massive and progressively increase in size from vertebra LI to LV. The pedicles are short and stocky, the transverse processes are long and slender, and the spinous processes are large and stubby. The articular processes are large and oriented medially and laterally, which promotes flexion and extension in this part of the vertebral column.
Between each lumbar vertebra is an intervertebral disc, which completes this part of the midline boundary of the posterior abdominal wall.
The midline boundary of the posterior abdominal wall, inferior to the lumbar vertebrae, consists of the upper margin of the sacrum (Fig. 4.128). The sacrum is formed by the fusion of the five sacral vertebrae into a single, wedge-shaped bony structure that is broad superiorly and narrows inferiorly. Its concave anterior surface and its convex posterior surface contain anterior and posterior sacral foramina for the anterior and posterior rami of spinal nerves to pass through.
Pelvic bones
The ilia, which are components of each pelvic bone, attach laterally to the sacrum at the sacro-iliac joints (Fig. 4.128). The upper part of each ilium expands outward into a thin wing-like area (the iliac fossa). The medial side of this region of each iliac bone, and the related muscles, are components of the posterior abdominal wall.
Ribs
Superiorly, ribs XI and XII complete the bony framework of the posterior abdominal wall (Fig. 4.128). These ribs are unique in that they do not articulate with the sternum, they have a single articular facet on their heads, and they do not have necks or tubercles.
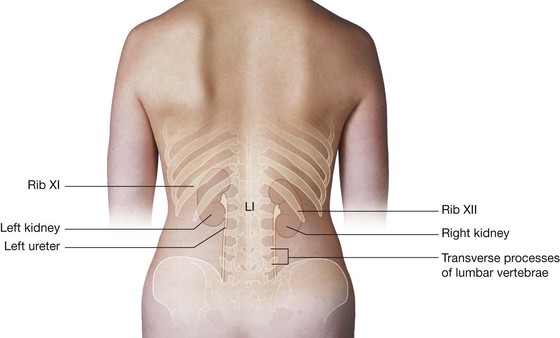
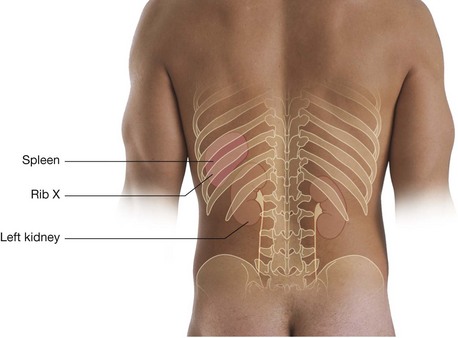
Rib XI is posterior to the superior part of the left kidney, and rib XII is posterior to the superior part of both kidneys. Also, rib XII serves as a point of attachment for numerous muscles and ligaments.
Muscles
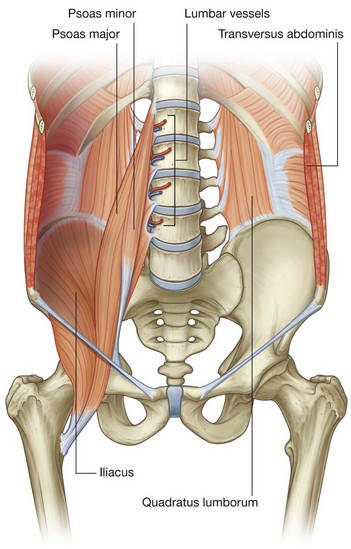
Muscles forming the medial, lateral, inferior, and superior boundaries of the posterior abdominal region fill in the bony framework of the posterior abdominal wall (Table 4.2). Medially are the psoas major and minor muscles, laterally is the quadratus lumborum muscle, inferiorly is the iliacus muscle, and superiorly is the diaphragm (Fig. 4.129).
Psoas major and minor
Medially, the psoas major muscles cover the anterolateral surface of the bodies of the lumbar vertebrae, filling in the space between the vertebral bodies and the transverse processes (Fig. 4.129). Each of these muscles arises from the bodies of vertebra TXII and all five lumbar vertebrae, from the intervertebral discs between each vertebra, and from the transverse processes of the lumbar vertebrae. Passing inferiorly along the pelvic brim, each muscle continues into the anterior thigh, under the inguinal ligament, to attach to the lesser trochanter of the femur.
The psoas major muscle flexes the thigh at the hip joint when the trunk is stabilized and flexes the trunk against gravity when the body is supine. It is innervated by anterior rami of nerves L1 to L3.
Associated with the psoas major muscle is the psoas minor muscle, which is sometimes absent. Lying on the surface of the psoas major when present, this slender muscle arises from vertebrae TXII and LI and the intervening intervertebral disc; its long tendon inserts into the pectineal line of the pelvic brim and the iliopubic eminence.
The psoas minor is a weak flexor of the lumbar vertebral column and is innervated by the anterior ramus of nerve L1.
Quadratus lumborum
Laterally, the quadratus lumborum muscles fill the space between ribs XII and the iliac crest on both sides of the vertebral column (Fig. 4.129). They are overlapped medially by the psoas major muscles; along their lateral borders are the transversus abdominis muscles.
Each quadratus lumborum muscle arises from the transverse process of vertebra LV, the iliolumbar ligament, and the adjoining part of the iliac crest. The muscle attaches superiorly to the transverse process of the first four lumbar vertebrae and the inferior border of rib XII.
The quadratus lumborum muscles depress and stabilize the twelfth ribs and contribute to lateral bending of the trunk. Acting together, the muscles may extend the lumbar part of the vertebral column. They are innervated by anterior rami of T12 and L1 to L4 spinal nerves.
Iliacus
Inferiorly, an iliacus muscle fills the iliac fossa on each side (Fig. 4.129). From this expansive origin covering the iliac fossa, the muscle passes inferiorly, joins with the psoas major muscle, and attaches to the lesser trochanter of the femur. As they pass into the thigh, these combined muscles are referred to as the iliopsoas muscle.
Like the psoas major muscle, the iliacus flexes the thigh at the hip joint when the trunk is stabilized and flexes the trunk against gravity when the body is supine. It is innervated by branches of the femoral nerve.
Diaphragm
Superiorly, the diaphragm forms the boundary of the posterior abdominal region. This musculotendinous sheet also separates the abdominal cavity from the thoracic cavity.
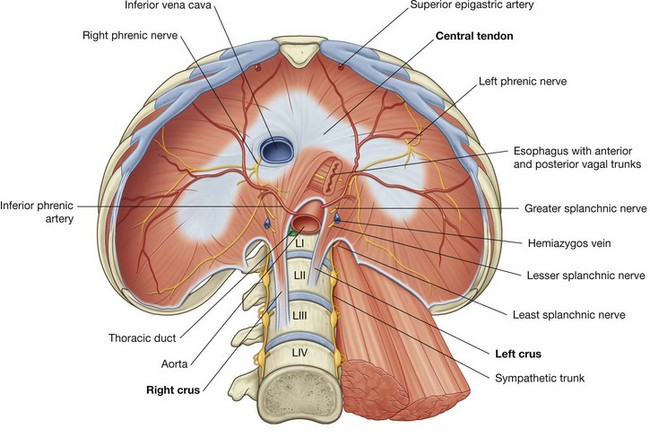
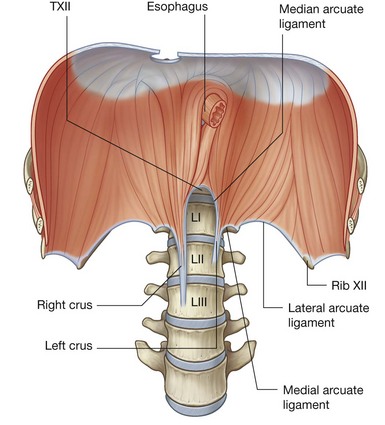
Structurally, the diaphragm consists of a central tendinous part into which the circumferentially arranged muscle fibers attach (Fig. 4.130). The diaphragm is anchored to the lumbar vertebrae by musculotendinous crura, which blend with the anterior longitudinal ligament of the vertebral column:
 the right crus is the longest and broadest of the crura and is attached to the bodies of vertebrae LI to LIII, and the intervening intervertebral discs (Fig. 4.131);
the right crus is the longest and broadest of the crura and is attached to the bodies of vertebrae LI to LIII, and the intervening intervertebral discs (Fig. 4.131); similarly, the left crus is attached to vertebrae LI and LII and the associated intervertebral disc.
similarly, the left crus is attached to vertebrae LI and LII and the associated intervertebral disc.The crura are connected across the midline by a tendinous arch (the median arcuate ligament), which passes anterior to the aorta (Fig. 4.131).
Lateral to the crura, a second tendinous arch is formed by the fascia covering the upper part of the psoas major muscle. This is the medial arcuate ligament, which is attached medially to the sides of vertebrae LI and LII and laterally to the transverse process of vertebra LI (Fig. 4.131).
A third tendinous arch, the lateral arcuate ligament, is formed by a thickening in the fascia that covers the quadratus lumborum. It is attached medially to the transverse process of vertebra LI and laterally to rib XII (Fig. 4.131).
The medial and lateral arcuate ligaments serve as points of origin for some of the muscular components of the diaphragm.
Structures passing through or around the diaphragm
Numerous structures pass through or around the diaphragm (Fig. 4.130):
 the aorta passes posterior to the diaphragm and anterior to the vertebral bodies at the lower level of vertebra TXII; it is between the two crura of the diaphragm and posterior to the median arcuate ligament, just to the left of midline;
the aorta passes posterior to the diaphragm and anterior to the vertebral bodies at the lower level of vertebra TXII; it is between the two crura of the diaphragm and posterior to the median arcuate ligament, just to the left of midline; accompanying the aorta through the aortic hiatus is the thoracic duct and, sometimes, the azygos vein;
accompanying the aorta through the aortic hiatus is the thoracic duct and, sometimes, the azygos vein; the esophagus passes through the musculature of the right crus of the diaphragm at the level of vertebra TX, just to the left of the aortic hiatus;
the esophagus passes through the musculature of the right crus of the diaphragm at the level of vertebra TX, just to the left of the aortic hiatus; passing through the esophageal hiatus with the esophagus are the anterior and posterior vagal trunks, the esophageal branches of the left gastric artery and vein, and a few lymphatic vessels;
passing through the esophageal hiatus with the esophagus are the anterior and posterior vagal trunks, the esophageal branches of the left gastric artery and vein, and a few lymphatic vessels; the third large opening in the diaphragm is the caval opening through which the inferior vena cava passes from the abdominal cavity to the thoracic cavity (Fig. 4.130) at approximately vertebra TVIII in the central tendinous part of the diaphragm;
the third large opening in the diaphragm is the caval opening through which the inferior vena cava passes from the abdominal cavity to the thoracic cavity (Fig. 4.130) at approximately vertebra TVIII in the central tendinous part of the diaphragm; the left phrenic nerve passes through the muscular part of the diaphragm just anterior to the central tendon on the left side.
the left phrenic nerve passes through the muscular part of the diaphragm just anterior to the central tendon on the left side.Additional structures pass through small openings either in or just outside the diaphragm as they pass from the thoracic cavity to the abdominal cavity (Fig. 4.130):
Domes
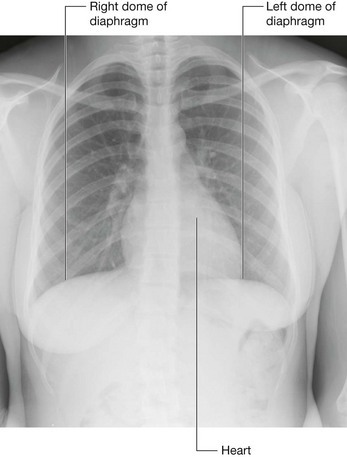
The classic appearance of the right and left domes of the diaphragm is caused by the underlying abdominal contents pushing these lateral areas upward, and by the fibrous pericardium, which is attached centrally, causing a flattening of the diaphragm in this area (Fig. 4.132).
 the liver on the right, with some contribution from the right kidney and the right suprarenal gland; and
the liver on the right, with some contribution from the right kidney and the right suprarenal gland; and the fundus of the stomach and spleen on the left, with contributions from the left kidney and the left suprarenal gland.
the fundus of the stomach and spleen on the left, with contributions from the left kidney and the left suprarenal gland.Although the height of these domes varies during breathing, a reasonable estimate in normal expiration places the left dome at the fifth intercostal space and the right dome at rib V. This is important to remember when percussing the thorax.
During inspiration, the muscular part of the diaphragm contracts, causing the central tendon of the diaphragm to be drawn inferiorly. This results in some flattening of the domes, enlargement of the thoracic cavity, and a reduction in intra-thoracic pressure. The physiological effect of these changes is that air enters the lungs and venous return to the heart is enhanced.
Blood supply
There is blood supply to the diaphragm on its superior and inferior surfaces:
 superiorly, the musculophrenic and pericardiacophrenic arteries, both branches of the internal thoracic artery, and the superior phrenic artery, a branch of the thoracic aorta, supply the diaphragm;
superiorly, the musculophrenic and pericardiacophrenic arteries, both branches of the internal thoracic artery, and the superior phrenic artery, a branch of the thoracic aorta, supply the diaphragm; inferiorly, the inferior phrenic arteries, branches of the abdominal aorta, supply the diaphragm (see Fig. 4.130).
inferiorly, the inferior phrenic arteries, branches of the abdominal aorta, supply the diaphragm (see Fig. 4.130).Venous drainage is through companion veins to these arteries.
Innervation
Innervation of the diaphragm is primarily by the phrenic nerves. These nerves, from the C3 to C5 spinal cord levels, provide all motor innervation to the diaphragm and sensory fibers to the central part. They pass through the thoracic cavity, between the mediastinal pleura and the pericardium, to the superior surface of the diaphragm. At this point, the right phrenic nerve accompanies the inferior vena cava through the diaphragm and the left phrenic nerve passes through the diaphragm by itself (see Fig. 4.130). Additional sensory fibers are supplied to the peripheral areas of the diaphragm by intercostal nerves.
In the clinic
At first glance, it is difficult to appreciate why the psoas muscle sheath is of greater importance than any other muscle sheath. The psoas muscle and its sheath arise not only from the lumbar vertebrae but also from the intervertebral discs between each vertebra. This disc origin is of critical importance. In certain types of infection, the intervertebral disc is preferentially affected (e.g., tuberculosis and salmonella discitis). As the infection of the disc develops, the infection spreads anteriorly and anterolaterally. In the anterolateral position, the infection passes into the psoas muscle sheath, and spreads within the muscle and sheath, and may appear below the inguinal ligament as a mass.
In the clinic
To understand why a hernia occurs through the diaphragm, it is necessary to consider the embryology of the diaphragm.
The diaphragm is formed from four structures—the septum transversum, the posterior esophageal mesentery, the pleuroperitoneal membrane, and the peripheral rim—which eventually fuse together, so separating the abdominal cavity from the thoracic cavity. The septum transversum forms the central tendon, which develops from a mesodermal origin in front of the embryo’s head and then moves to its more adult position during formation of the head fold.
Fusion of the various components of the diaphragm may fail, and hernias may occur through the failed points of fusion (Fig. 4.133). The commonest sites are:
 through an opening on the left when the pleuroperitoneal membrane fails to close the pericardioperitoneal canal (Bochdalek’s hernia).
through an opening on the left when the pleuroperitoneal membrane fails to close the pericardioperitoneal canal (Bochdalek’s hernia).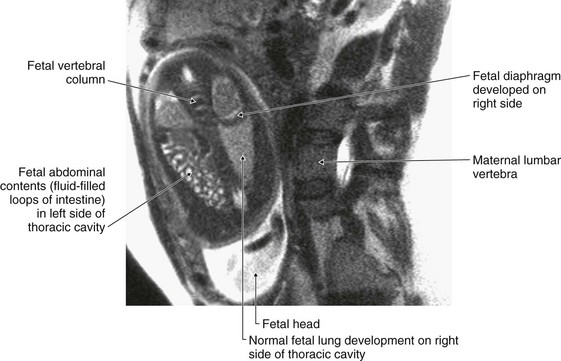
Fig. 4.133 Fetal diaphragmatic hernia in utero. T2-weighted MR image. Fetus in coronal plane, mother in sagittal plane.
Hernias may also occur through the central tendon and through a congenitally large esophageal hiatus.
Morgagni’s and Bochdalek’s hernias tend to appear at or around the time of birth or in early infancy. They allow abdominal bowel to enter the thoracic cavity, which may compress the lungs and reduce respiratory function. Most of these hernias require surgical closure of the diaphragmatic defect.
Occasionally, small defects within the diaphragm fail to permit bowel through, but do allow free movement of fluid. Patients with ascites may develop pleural effusions, while patients with pleural effusions may develop ascites when these defects are present.
In the clinic
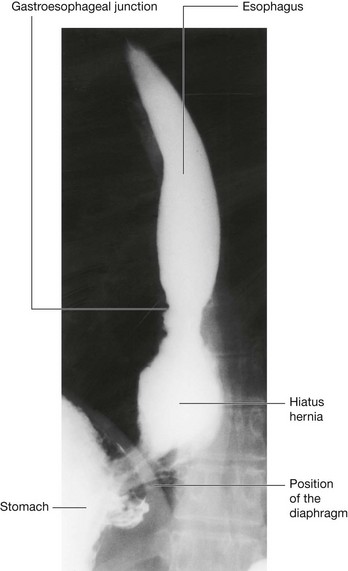
At the level of the esophageal hiatus, the diaphragm may be lax, allowing the fundus of the stomach to herniate into the posterior mediastinum (Fig. 4.134). This typically causes symptoms of acid reflux. Ulceration may occur and may produce bleeding and anemia. The diagnosis is usually made by barium studies or endoscopy. Treatment in the first instance is by medical management, although surgery may be necessary.
Viscera
Kidneys
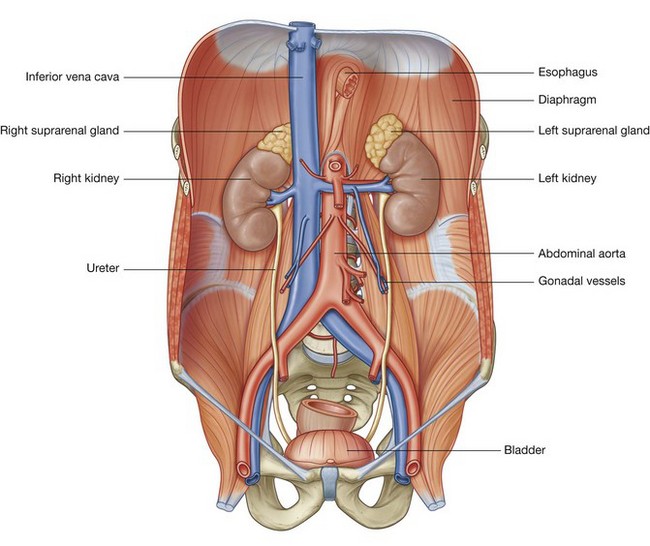
The bean-shaped kidneys are retroperitoneal in the posterior abdominal region (Fig. 4.135). They lie in the extraperitoneal connective tissue immediately lateral to the vertebral column. In the supine position, the kidneys extend from approximately vertebra TXII superiorly to vertebra LIII inferiorly, with the right kidney somewhat lower than the left because of its relationship with the liver. Although they are similar in size and shape, the left kidney is a longer and more slender organ than the right kidney, and nearer to the midline.
Relationships to other structures
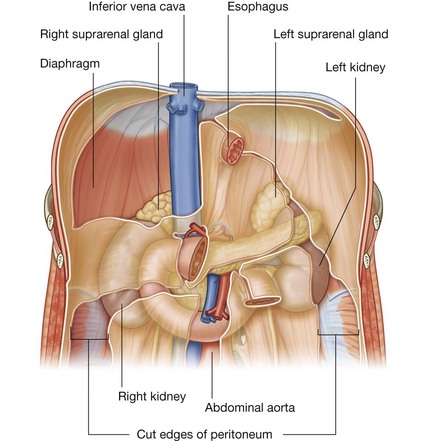
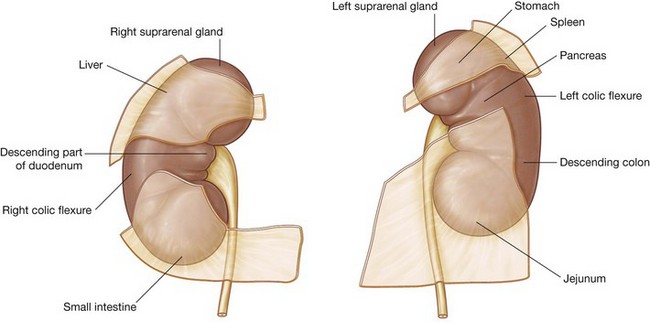
The anterior surface of the right kidney is related to numerous structures, some of which are separated from the kidney by a layer of peritoneum and some of which are directly against the kidney (Fig. 4.136):
 moving inferiorly, a large part of the rest of the upper part of the anterior surface is against the liver and is separated from it by a layer of peritoneum;
moving inferiorly, a large part of the rest of the upper part of the anterior surface is against the liver and is separated from it by a layer of peritoneum; the inferior pole of the kidney, on its lateral side, is directly associated with the right colic flexure and, on its medial side, is covered by a segment of the intraperitoneal small intestine.
the inferior pole of the kidney, on its lateral side, is directly associated with the right colic flexure and, on its medial side, is covered by a segment of the intraperitoneal small intestine.The anterior surface of the left kidney is also related to numerous structures, some with an intervening layer of peritoneum and some directly against the kidney (Fig. 4.136):
 on its lateral side, the lower half of the kidney is covered by the left colic flexure and the beginning of the descending colon, and, on its medial side, by the parts of the intraperitoneal jejunum.
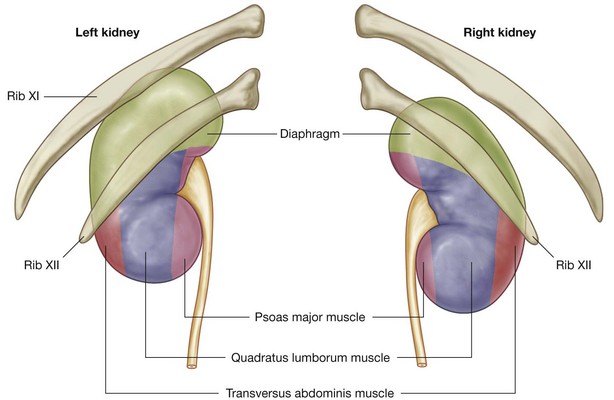
on its lateral side, the lower half of the kidney is covered by the left colic flexure and the beginning of the descending colon, and, on its medial side, by the parts of the intraperitoneal jejunum.Posteriorly, the right and left kidneys are related to similar structures (Fig. 4.137). Superiorly is the diaphragm and inferior to this, moving in a medial to lateral direction, are psoas major, quadratus lumborum, and transversus abdominis muscles.
The superior pole of the right kidney is anterior to rib XII, while the same region of the left kidney is anterior to ribs XI and XII. The pleural sacs, and specifically, the costodiaphragmatic recesses, therefore extend posterior to the kidneys.
Also passing posterior to the kidneys are the subcostal vessels and nerves and the iliohypogastric and ilio-inguinal nerves.
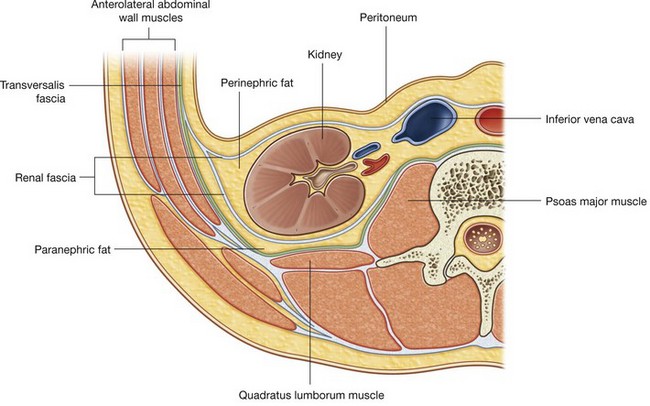
Renal fat and fascia
The kidneys are enclosed in and associated with a unique arrangement of fascia and fat. Immediately outside the renal capsule, there is an accumulation of extraperitoneal fat—the perinephric fat (perirenal fat), which completely surrounds the kidney (Fig. 4.138). Enclosing the perinephric fat is a membranous condensation of the extraperitoneal fascia (the renal fascia). The suprarenal glands are also enclosed in this fascial compartment, usually separated from the kidneys by a thin septum.
At the lateral margins of each kidney, the anterior and posterior layers of the renal fascia fuse (Fig. 4.138). This fused layer may connect with the transversalis fascia on the lateral abdominal wall.
Above each suprarenal gland, the anterior and posterior layers of the renal fascia fuse and blend with the fascia that covers the diaphragm.
Medially, the anterior layer of the renal fascia continues over the vessels in the hilum and fuses with the connective tissue associated with the abdominal aorta and the inferior vena cava (Fig. 4.138). In some cases, the anterior layer may cross the midline to the opposite side and blend with its companion layer.
The posterior layer of the renal fascia passes medially between the kidney and the fascia covering the quadratus lumborum muscle to fuse with the fascia covering the psoas major muscle.
Inferiorly, the anterior and posterior layers of the renal fascia enclose the ureters.
In addition to perinephric fat and the renal fascia, a final layer of paranephric fat (pararenal fat) completes the fat and fascias associated with the kidney (Fig. 4.138). This fat accumulates posterior and posterolateral to each kidney.
Kidney structure
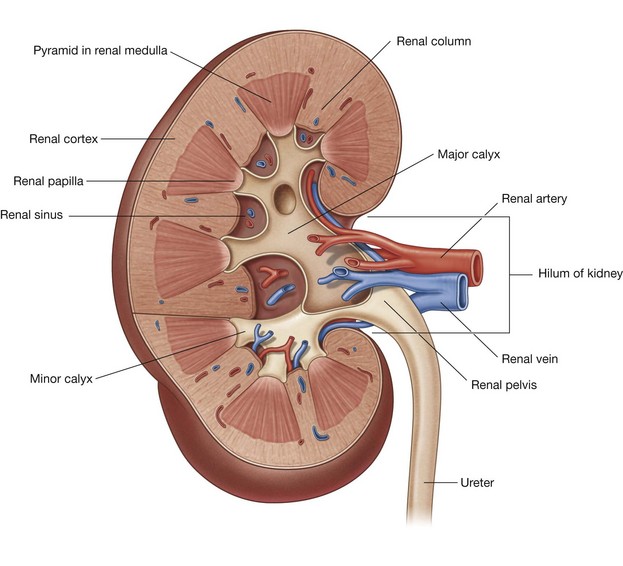
Each kidney has a smooth anterior and posterior surface covered by a fibrous capsule, which is easily removable except during disease.
On the medial margin of each kidney is the hilum of kidney, which is a deep vertical slit through which renal vessels, lymphatics, and nerves enter and leave the substance of the kidney (Fig. 4.139). Internally, the hilum is continuous with the renal sinus. Perinephric fat continues into the hilum and sinus and surrounds all structures.
Each kidney consists of an outer renal cortex and an inner renal medulla. The renal cortex is a continuous band of pale tissue that completely surrounds the renal medulla. Extensions of the renal cortex (the renal columns) project into the inner aspect of the kidney, dividing the renal medulla into discontinuous aggregations of triangular-shaped tissue (the renal pyramids).
The bases of the renal pyramids are directed outward, toward the renal cortex, while the apex of each renal pyramid projects inward, toward the renal sinus. The apical projection (renal papilla) is surrounded by a minor calyx.
The minor calices receive urine and represent the proximal parts of the tube that will eventually form the ureter (Fig. 4.139). In the renal sinus, several minor calices unite to form a major calyx, and two or three major calices unite to form the renal pelvis, which is the funnel-shaped superior end of the ureters.
Renal vasculature and lymphatics
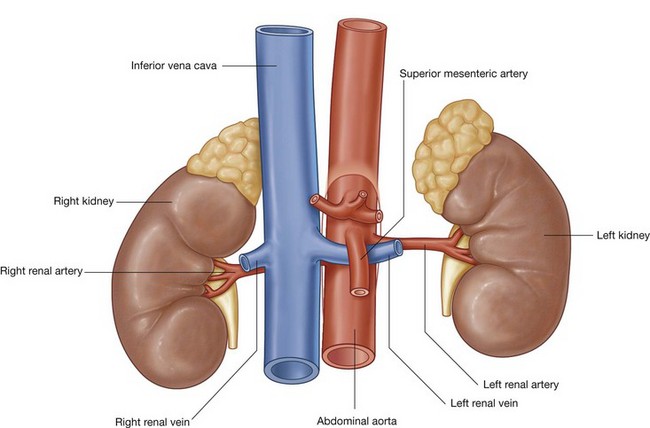
A single large renal artery, a lateral branch of the abdominal aorta, supplies each kidney. These vessels usually arise just inferior to the origin of the superior mesenteric artery between vertebrae LI and LII (Fig. 4.140). The left renal artery usually arises a little higher than the right, and the right renal artery is longer and passes posterior to the inferior vena cava.
As each renal artery approaches the renal hilum, it divides into anterior and posterior branches, which supply the renal parenchyma. Accessory renal arteries are common. They originate from the lateral aspect of the abdominal aorta, either above or below the primary renal arteries, enter the hilum with the primary arteries or pass directly into the kidney at some other level, and are commonly called extrahilar arteries.
Multiple renal veins contribute to the formation of the left and right renal veins, both of which are anterior to the renal arteries (Fig. 4.140).
Importantly, the longer left renal vein crosses the midline anterior to the abdominal aorta and posterior to the superior mesenteric artery and can be compressed by an aneurysm in either of these two vessels.
The lymphatic drainage of each kidney is to the lateral aortic (lumbar) nodes around the origin of the renal artery.
Ureters
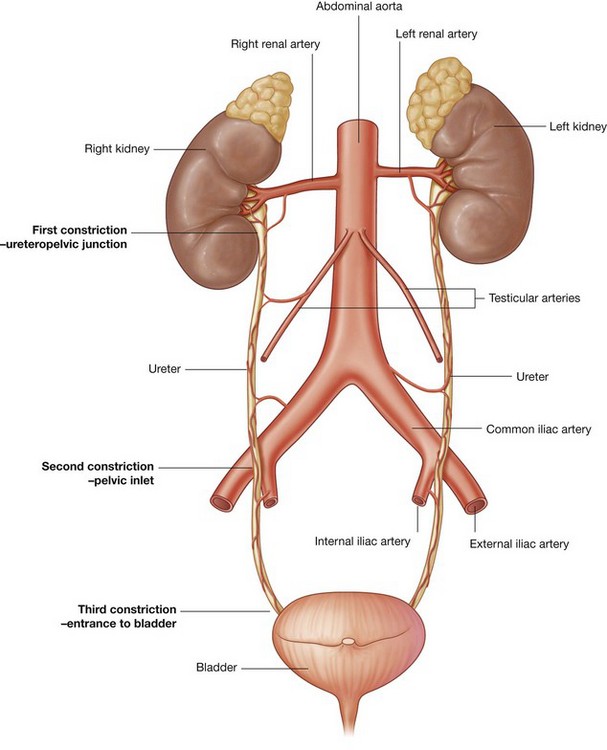
The ureters are muscular tubes that transport urine from the kidneys to the bladder. They are continuous superiorly with the renal pelvis, which is a funnel-shaped structure in the renal sinus. The renal pelvis is formed from a condensation of two or three major calices, which in turn are formed by the condensation of several minor calices (see Fig. 4.139). The minor calices surround a renal papilla.
The renal pelvis narrows as it passes inferiorly through the hilum of the kidney and becomes continuous with the ureter at the ureteropelvic junction (Fig. 4.141). Inferior to this junction, the ureters descend retroperitoneally on the medial aspect of the psoas major muscle. At the pelvic brim, the ureters cross either the end of the common iliac or the beginning of the external iliac arteries, enter the pelvic cavity, and continue their journey to the bladder.
At three points along their course the ureters are constricted (Fig. 4.141):
Ureteric vasculature and lymphatics
The ureters receive arterial branches from adjacent vessels as they pass towards the bladder (Fig. 4.141):
 the middle part may receive branches from the abdominal aorta, the testicular or ovarian arteries, and the common iliac arteries;
the middle part may receive branches from the abdominal aorta, the testicular or ovarian arteries, and the common iliac arteries; in the pelvic cavity, the ureters are supplied by one or more arteries from branches of the internal iliac arteries.
in the pelvic cavity, the ureters are supplied by one or more arteries from branches of the internal iliac arteries.In all cases, arteries reaching the ureters divide into ascending and descending branches, which form longitudinal anastomoses.
Lymphatic drainage of the ureters follows a pattern similar to that of the arterial supply. Lymph from:
Ureteric innervation
Ureteric innervation is from the renal, aortic, superior hypogastric, and inferior hypogastric plexuses through nerves that follow the blood vessels.
Visceral efferent fibers come from both sympathetic and parasympathetic sources, whereas visceral afferent fibers return to T11 to L2 spinal cord levels. Ureteric pain, which is usually related to distention of the ureter, is therefore referred to cutaneous areas supplied by T11 to L2 spinal cord levels. These areas would most likely include the posterior and lateral abdominal wall below the ribs and above the iliac crest, the pubic region, the scrotum in males, the labia majora in females, and the proximal anterior aspect of the thigh.
In the clinic
Urinary tract stones
Urinary tract stones (calculi) occur more frequently in men than in women, are most common in people aged between 20 and 60 years, and are usually associated with sedentary lifestyles. The stones are polycrystalline aggregates of calcium, phosphate, oxalate, urate, and other soluble salts within an organic matrix. The urine becomes saturated with these salts, and small variations in the pH cause the salts to precipitate.
Typically the patient has pain that radiates from the infrascapular region (loin) into the groin, and even into the scrotum or labia majora. Blood in the urine (hematuria) may also be noticed.
Infection must be excluded because certain species of bacteria are commonly associated with urinary tract stones.
The complications of urinary tract stones include infection, urinary obstruction, and renal failure. Stones may also develop within the bladder and produce marked irritation, causing pain and discomfort.
The diagnosis of urinary tract stones is based upon history and examination. Stones are often visible on abdominal radiographs. Special investigations include:
In the clinic
Urinary tract cancer
Most tumors that arise in the kidney are renal cell carcinomas. These tumors develop from the proximal tubular epithelium. Approximately 5% of tumors within the kidney are transitional cell tumors, which arise from the urothelium of the renal pelvis. Most patients typically have blood in the urine (hematuria), pain in the infrascapular region (loin), and a mass.
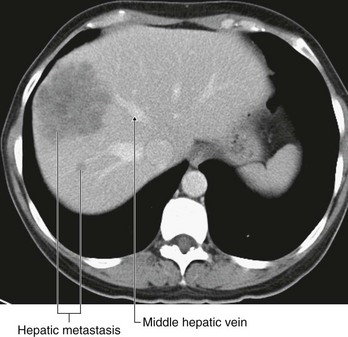
Renal cell tumors are unusual because not only do they grow outward from the kidney, invading the fat and fascia, but they also spread into the renal vein. This venous extension is rare for any other type of tumor, so, when seen, renal cell carcinoma should be suspected. In addition, the tumor may spread along the renal vein and into the inferior vena cava, and in rare cases can grow into the right atrium across the tricuspid valve and into the pulmonary artery (Figs. 4.142 and 4.143).
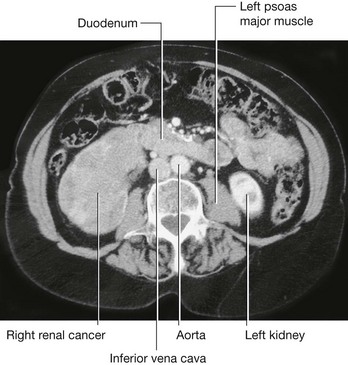
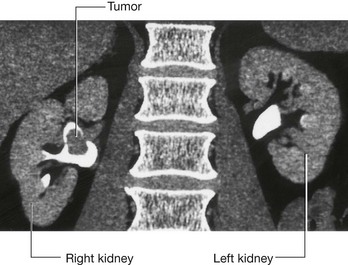
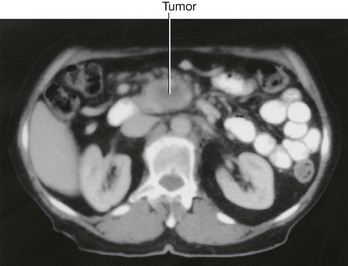
Fig. 4.142 Tumor in the right kidney growing toward, and possibly invading, the duodenum. Computed tomogram in the axial plane.
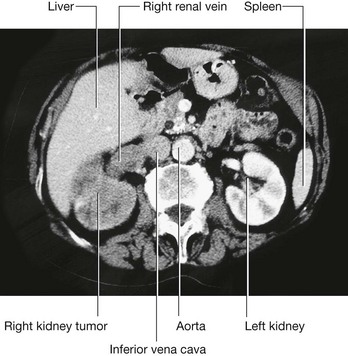
Fig. 4.143 Tumor in the right kidney spreading into the right renal vein. Computed tomogram in the axial plane.
Treatment for most renal cancers is surgical removal, even when metastatic spread is present, because some patients show regression of metastases.
Transitional cell carcinoma arises from the urothelium. The urothelium is present from the calices to the urethra and behaves as a “single unit.” Therefore, when patients develop transitional carcinomas within the bladder, similar tumors may also be present within upper parts of the urinary tract. In patients with bladder cancer, the whole of the urinary tract must always be investigated to exclude the possibility of other tumors (Fig. 4.144).
In the Clinic
Nephrostomy
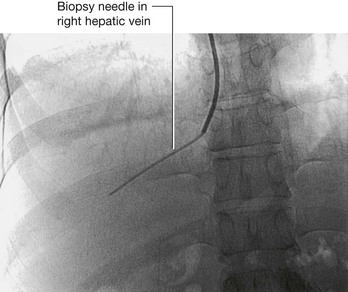
A nephrostomy is a procedure where a tube is placed through the lateral or posterior abdominal wall into the renal cortex to lie within the renal pelvis. The function of this tube is to allow drainage of urine from the renal pelvis through the tube externally (Fig. 4.145).
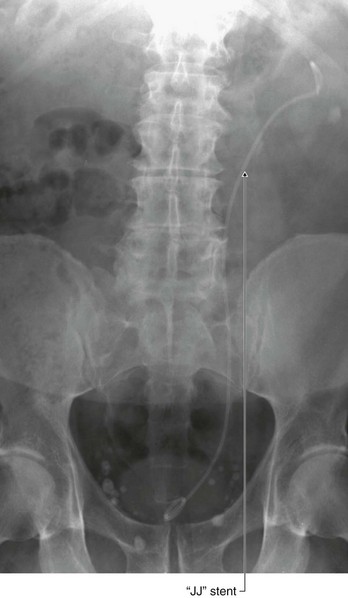
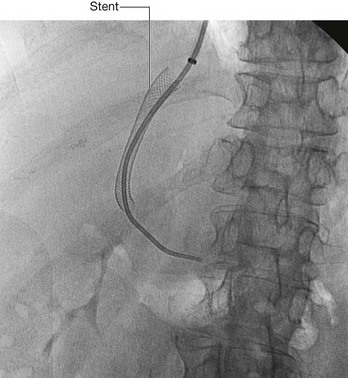
Fig. 4.145 This radiograph, anterior-posterior view, demonstrates a double J stent. The superior aspect of the double J stent is situated within the renal pelvis. The stent passes through the ureter describing the path of the ureter, and the tip of the double J stent is projected over the bladder, which appears as a slightly dense area on the radiograph.
The kidneys are situated on the posterior abdominal wall, and in thin healthy subjects may be only up to 2–3 cm from the skin. Access to the kidney is relatively straightforward, because the kidney can be easily visualized under ultrasound guidance. Using local anesthetic, a needle can be placed, under ultrasound direction, through the skin into the renal cortex and into the renal pelvis. A series of wires and tubes can be passed through the needle to position the drainage catheter.
The indications of such a procedure are many. In patients with distal ureteric obstruction the back pressure of urine within the ureters and the kidney significantly impairs the function of the kidney, which will fail to function. This will produce renal failure and ultimately death. Furthermore, a dilated obstructed system is also susceptible to infection. In many cases, there is not only obstruction producing renal failure but also infected urine within the system.
In the clinic
Kidney transplant
Kidney transplantation began in the United States in the 1950s. Since the first transplant, the major problem for kidney transplantation has been tissue rejection. A number of years have passed since this initial procedure and there have been significant breakthroughs in transplant rejection medicine. Renal transplantation is now a common procedure undertaken in patients with end stage renal failure.
Transplant kidneys are obtained from either living or deceased donors. The living donors are carefully assessed, because harvesting a kidney from a normal healthy individual, even with modern day medicine, carries a small risk.
Deceased kidney donors are brain dead or have suffered cardiac death. The donor kidney is harvested with a small cuff of aortic and venous tissue. The ureter is also harvested.
An ideal place to situate the transplant kidney is in the left or the right iliac fossa (Fig. 4.146). A curvilinear incision is made paralleling the iliac crest and pubic symphysis. The external oblique muscle, internal oblique muscle, transverse abdominis muscle, and transversalis fascia are divided. The surgeon identifies the parietal peritoneum but does not enter the peritoneal cavity. The parietal peritoneum is medially retracted to reveal the external iliac artery, external iliac vein, and the bladder. In some instances the internal iliac artery of the recipient is mobilized and anastomosed directly as an end-to-end procedure onto the renal artery of the donor kidney. Similarly the internal iliac vein is anastomosed to the donor vein. In the presence of a small aortic cuff of tissue the donor artery is anastomosed to the recipient external iliac artery and similarly for the venous anastomosis. The ureter is easily tunneled obliquely through the bladder wall with a straightforward anastomosis.
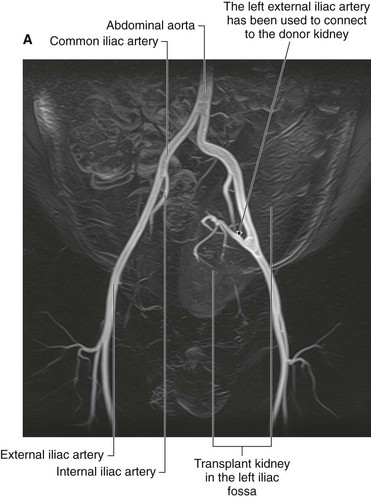
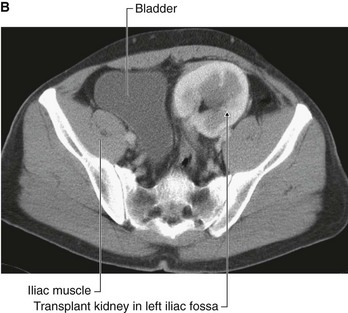
Fig. 4.146 Kidney transplant. A. This image demonstrates an MR angiogram of the bifurcation of the aorta. Attaching to the left external iliac artery is the donor artery for a kidney that has been transplanted into the left iliac fossa. B. Abdominal computed tomogram, in the axial plane, showing the transplanted kidney in the left iliac fossa.
The left and right iliac fossae are ideal locations for the transplant kidney, because a new space can be created without compromise to other structures. The great advantage of this procedure is the proximity to the anterior abdominal wall, which permits easy ultrasound visualization of the kidney and permits Doppler vascular assessment. Furthermore, in this position biopsies are easily obtained. The extraperitoneal approach enables patients to make a swift recovery.
Suprarenal glands
The suprarenal glands are associated with the superior pole of each kidney (Fig. 4.147). They consist of an outer cortex and an inner medulla. The right gland is shaped like a pyramid, whereas the left gland is semilunar in shape and the larger of the two.
Anterior to the right suprarenal gland is part of the right lobe of the liver and the inferior vena cava, whereas anterior to the left suprarenal gland is part of the stomach, pancreas, and, on occasion, the spleen. Parts of the diaphragm are posterior to both glands.
The suprarenal glands are surrounded by the perinephric fat and enclosed in the renal fascia, though a thin septum separates each gland from its associated kidney.
In the clinic
Investigation of the urinary tract
After an appropriate history and examination of the patient, including a digital rectal examination to assess the prostate in men, special investigations are required.
IVU (intravenous urogram)
An IVU is one of the most important and commonly carried out radiological investigations. The patient is injected with iodinated contrast medium. Most contrast media contain three iodine atoms spaced around a benzene ring. The relatively high atomic number of iodine compared to the atomic number of carbon, hydrogen, and oxygen, attenuates the radiation beam. After intravenous injection, contrast media are excreted predominantly by glomerular filtration, although some are secreted by the renal tubules. This allows visualization of the collecting system as well as the ureters and bladder.
Ultrasound
Ultrasound can be used to assess kidney size and the size of the calices, which may be dilated when obstructed. Although the ureters are poorly visualized using ultrasound, the bladder can be easily seen when full. Ultrasound measurements of bladder volume can be obtained before and after micturition.
Computed tomography
Computed tomography can be used to assess the kidneys, ureters, bladder, and adjacent structures and is a powerful tool for staging primary urinary tract tumors.
Nuclear medicine
Nuclear medicine is an extremely useful tool for investigating the urinary tract because radioisotope compounds can be used to estimate renal cell mass and function and assess the parenchyma for renal scarring. These tests are often very useful in children when renal scarring and reflux disease is suspected.
Suprarenal vasculature
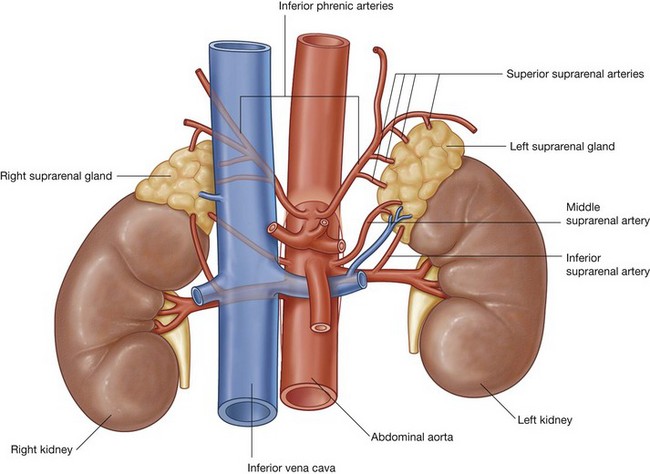
The arterial supply to the suprarenal glands is extensive and arises from three primary sources (Fig. 4.147):
 as the bilateral inferior phrenic arteries pass upward from the abdominal aorta to the diaphragm, they give off multiple branches (superior suprarenal arteries) to the suprarenal glands;
as the bilateral inferior phrenic arteries pass upward from the abdominal aorta to the diaphragm, they give off multiple branches (superior suprarenal arteries) to the suprarenal glands; a middle branch (middle suprarenal artery) to the suprarenal glands usually arises directly from the abdominal aorta;
a middle branch (middle suprarenal artery) to the suprarenal glands usually arises directly from the abdominal aorta; inferior branches (inferior suprarenal arteries) from the renal arteries pass upward to the suprarenal glands.
inferior branches (inferior suprarenal arteries) from the renal arteries pass upward to the suprarenal glands.In contrast to this multiple arterial supply is the venous drainage, which usually consists of a single vein leaving the hilum of each gland. On the right side, the right suprarenal vein is short and almost immediately enters the inferior vena cava; while on the left side, the left suprarenal vein passes inferiorly to enter the left renal vein.
Vasculature
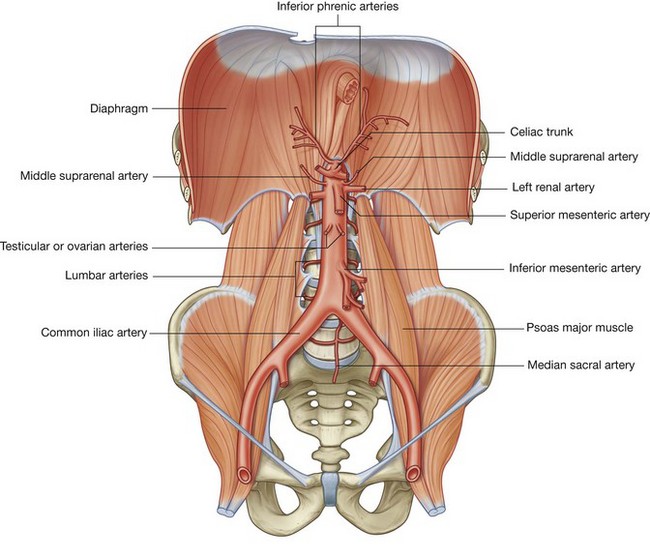
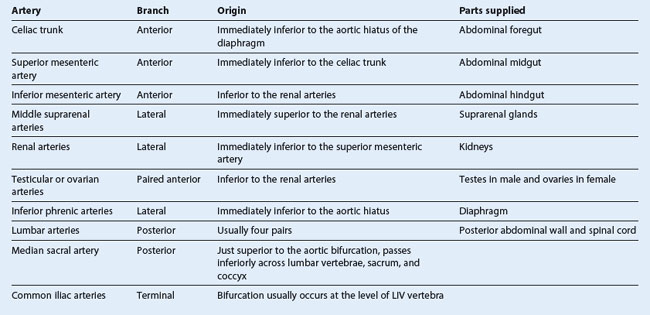
Abdominal aorta
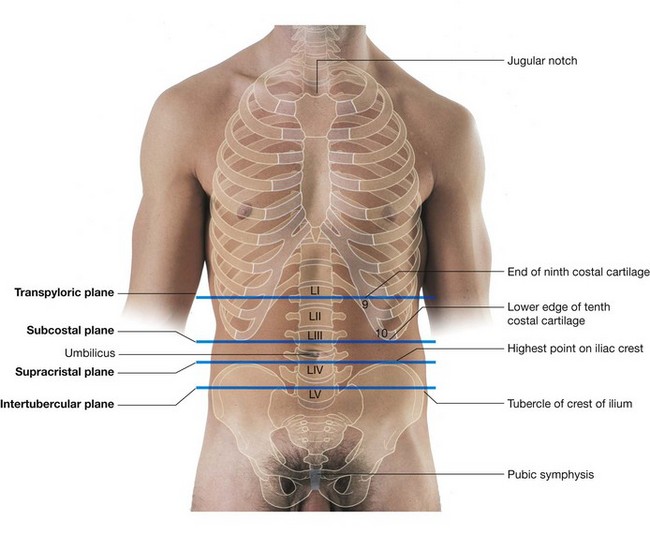
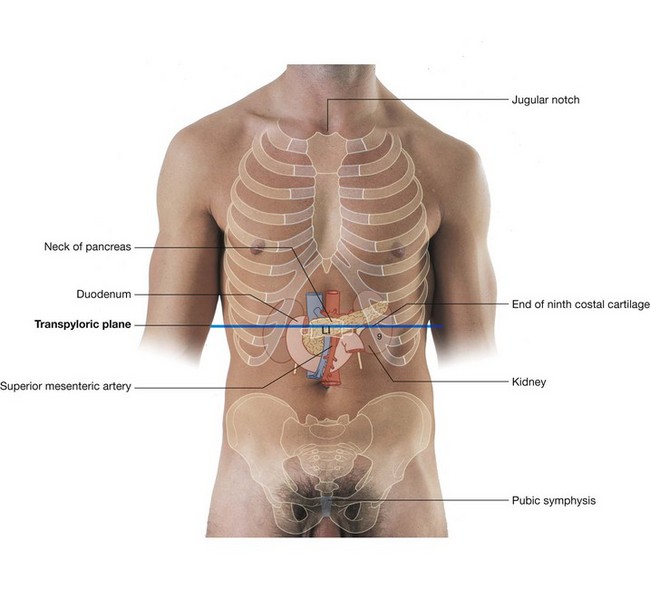
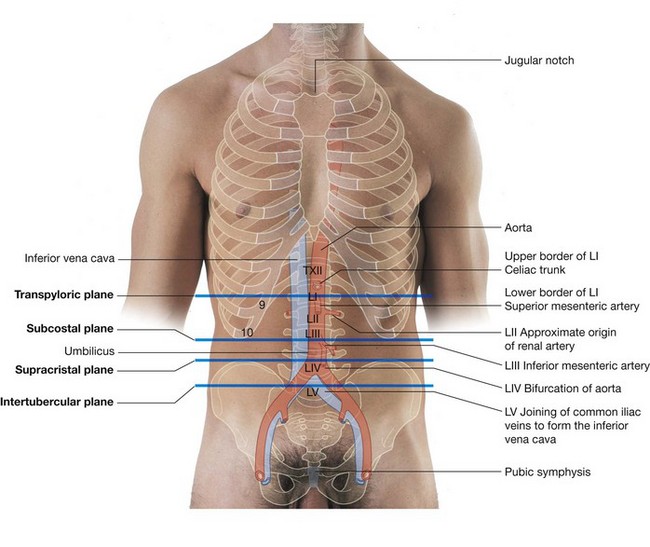
The abdominal aorta begins at the aortic hiatus of the diaphragm as a midline structure at approximately the lower level of vertebra TXII (Fig. 4.148). It passes downward on the anterior surface of the bodies of vertebrae LI to LIV, ending just to the left of midline at the lower level of vertebra LIV. At this point, it divides into the right and left common iliac arteries. This bifurcation can be visualized on the anterior abdominal wall as a point approximately 2.5 cm below the umbilicus or even with a line extending between the highest points of the iliac crest.
As the abdominal aorta passes through the posterior abdominal region, the prevertebral plexus of nerves and ganglia covers its anterior surface. It is also related to numerous other structures:
 anterior to the abdominal aorta, as it descends, are the pancreas and splenic vein, the left renal vein, and the inferior part of the duodenum;
anterior to the abdominal aorta, as it descends, are the pancreas and splenic vein, the left renal vein, and the inferior part of the duodenum; on its right side are the cisterna chyli, thoracic duct, azygos vein, right crus of the diaphragm, and the inferior vena cava;
on its right side are the cisterna chyli, thoracic duct, azygos vein, right crus of the diaphragm, and the inferior vena cava;Branches of the abdominal aorta (Table 4.3) can be classified as:
Visceral branches
The visceral branches are either unpaired or paired vessels.
The three unpaired visceral branches that arise from the anterior surface of the abdominal aorta (Fig. 4.148) are:
The paired visceral branches of the abdominal aorta (Fig. 4.148) include:
 the middle suprarenal arteries—small, lateral branches of the abdominal aorta arising just above the renal arteries that are part of the multiple vascular supply to the suprarenal gland;
the middle suprarenal arteries—small, lateral branches of the abdominal aorta arising just above the renal arteries that are part of the multiple vascular supply to the suprarenal gland;Posterior branches
The posterior branches of the abdominal aorta are vessels supplying the diaphragm or body wall. They consist of the inferior phrenic arteries, the lumbar arteries, and the median sacral artery (Fig. 4.148).
Inferior phrenic arteries
The inferior phrenic arteries arise immediately inferior to the aortic hiatus of the diaphragm either directly from the abdominal aorta, as a common trunk from the abdominal aorta, or from the base of the celiac trunk (Fig. 4.148). Whatever their origin, they pass upward, provide some arterial supply to the suprarenal gland, and continue onto the inferior surface of the diaphragm.
Lumbar arteries
There are usually four pairs of lumbar arteries arising from the posterior surface of the abdominal aorta (Fig. 4.148). They run laterally and posteriorly over the bodies of the lumbar vertebrae, continue laterally, passing posterior to the sympathetic trunks and between the transverse processes of adjacent lumbar vertebrae, and reach the abdominal wall. From this point onward, they demonstrate a branching pattern similar to a posterior intercostal artery, which includes providing segmental branches that supply the spinal cord.
Median sacral artery
The final posterior branch is the median sacral artery (Fig. 4.148). This vessel arises from the posterior surface of the abdominal aorta just superior to the bifurcation and passes in an inferior direction, first over the anterior surface of the lower lumbar vertebrae and then over the anterior surface of the sacrum and coccyx.
In the clinic
Abdominal aortic stent graft
An abdominal aortic aneurysm is a dilatation of the aorta and generally tends to occur in the infrarenal region (the region at or below the renal arteries). As the aorta expands, the risk of rupture increases, and it is now generally accepted that when an aneurysm reaches 5.5 cm or greater an operation will significantly benefit the patient.
With the aging population, the number of abdominal aortic aneurysms is increasing. Moreover, with the increasing use of imaging techniques a number of abdominal aortic aneurysms are identified in asymptomatic patients.
For many years the standard treatment for repair was an open operative technique, which involved a large incision from xiphoid process of the sternum to symphysis pubis and dissection of the aneurysm. The aneurysm was excised and a tubular woven graft was sewn into place. Recovery may take a number of days, even weeks, and most patients would be placed in the intensive care unit after the operation
Further developments and techniques have led to a new type of procedure being performed to treat abdominal aortic aneurysms—the endovascular graft. The idea of placing the graft into the aortic aneurysm and lining the dilated vessel is not new and was first described over 10 years ago. Since the original description the devices have been modified on a number of occasions (Fig. 4.149).
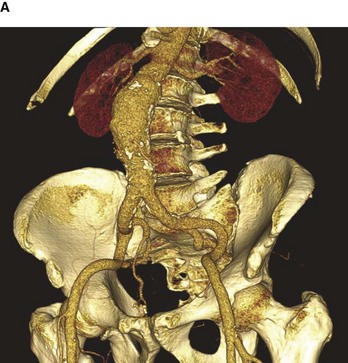
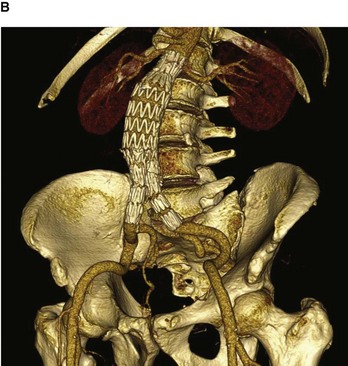
Fig. 4.149 Volume-rendered reconstruction using multidetector computed tomography of patient with an infrarenal abdominal aortic aneurysm before (A) and after (B) endovascular aneurysm repair. Note the image only demonstrates the intraluminal contrast and not the entire vessel. White patches in the aorta (A) represent intramural calcium.
The technique involves surgically dissecting the femoral artery below the inguinal ligament. A small incision is made in the femoral artery and the pre-loaded compressed graft with metal support struts is passed on a large catheter into the abdominal aorta through the femoral artery. Using X-ray for guidance the graft is opened, lining the inside of the aorta. Limb attachments are made to the graft that extend into the common iliac vessels. This bifurcated tube device effectively excludes the abdominal aortic aneurysm.
This type of device is not suitable for all patients. Patients who receive this device do not need to go to the intensive care unit. Many patients leave hospital within 24–48 hours. Importantly, this device can be used for patients who were deemed unfit for open surgical repair.
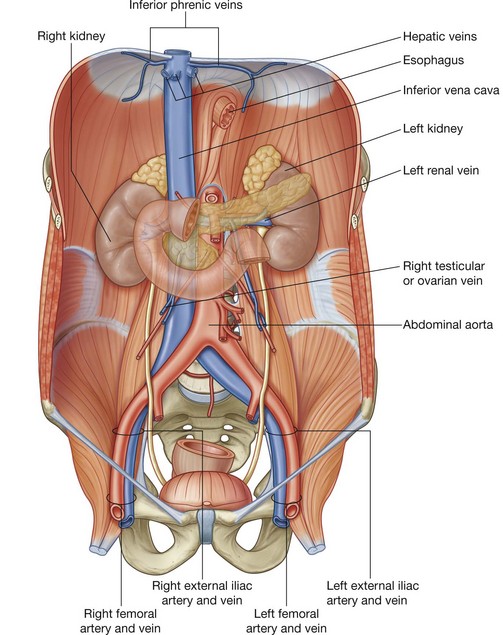
Inferior vena cava
The inferior vena cava returns blood from all structures below the diaphragm to the right atrium of the heart. It is formed when the two common iliac veins come together at the level of vertebra LV, just to the right of midline. It ascends through the posterior abdominal region anterior to the vertebral column immediately to the right of the abdominal aorta (Fig. 4.150), continues in a superior direction, and leaves the abdomen by piercing the central tendon of the diaphragm at the level of vertebra TVIII.
During its course, the anterior surface of the inferior vena cava is crossed by the right common iliac artery, the root of the mesentery, the right testicular or ovarian artery, the inferior part of the duodenum, the head of the pancreas, the superior part of the duodenum, the bile duct, the portal vein, and the liver, which overlaps and on occasion completely surrounds the vena cava (Fig. 4.150).
Tributaries to the inferior vena cava include the:
There are no tributaries from the abdominal part of the gastrointestinal tract, the spleen, the pancreas, or the gallbladder, because veins from these structures are components of the portal venous system, which first passes through the liver.
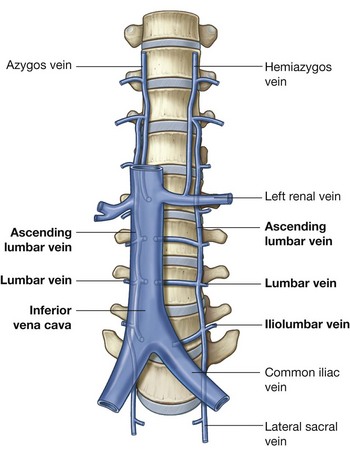
Of the venous tributaries mentioned above, the lumbar veins are unique in their connections and deserve special attention. Not all of the lumbar veins drain directly into the inferior vena cava (Fig. 4.151):
 the fifth lumbar vein generally drains into the iliolumbar vein, a tributary of the common iliac vein;
the fifth lumbar vein generally drains into the iliolumbar vein, a tributary of the common iliac vein;The ascending lumbar veins are long, anastomosing venous channels that connect the common iliac, iliolumbar, and lumbar veins with the azygos and hemiazygos veins of the thorax.
If the inferior vena cava becomes blocked the ascending lumbar veins become important collateral channels between the lower and upper parts of the body.
In the clinic
Inferior vena cava filter
Deep vein thrombosis is a potentially fatal condition where a clot (thrombus) is formed in the deep venous system of the legs and the veins of the pelvis. Virchow described the reasons for thrombus formation as decreased blood flow, abnormality of the constituents of blood, and abnormalities of the vessel wall. Common predisposing factors include hospitalization and surgery, the oral contraceptive pill, smoking, and air travel. Other abnormalities also include clotting abnormalities (e.g., protein S and protein C deficiency).
The diagnosis of deep vein thrombosis may be difficult to establish, with symptoms including leg swelling and pain and discomfort in the calf. It may also be an incidental finding.
In practice, patients with suspected deep vein thrombosis undergo a D-dimer blood test, which measures levels of a fibrin degradation product. If this is positive there is a high association with deep vein thrombosis.
The consequences of deep vein thrombosis are two-fold. Occasionally the clot may dislodge and pass into the venous system through the right side of the heart and into the main pulmonary arteries. If the clots are of significant size they obstruct blood flow to the lung and may produce instantaneous death. Secondary complications include destruction of the normal valvular system in the legs, which may lead to venous incompetency and chronic leg swelling with ulceration.
The treatment for deep vein thrombosis is prevention. In order to prevent deep vein thrombosis, patients are optimized by removing all potential risk factors. Subcutaneous heparin may be injected and the patient wears compression stockings to prevent venous stasis while in the hospital.
In certain situations it is not possible to optimize the patient with prophylactic treatment, and it may be necessary to insert a filter into the inferior vena cava that traps any large clots. It may be removed after the risk period has ended.
Lymphatic system
Lymphatic drainage from most deep structures and regions of the body below the diaphragm converges mainly on collections of lymph nodes and vessels associated with the major blood vessels of the posterior abdominal region (Fig. 4.152; Table 4.4). The lymph then predominantly drains into the thoracic duct.
| Lymphatic vessel | Area drained |
|---|---|
| Right jugular trunk | Right side of head and neck |
| Left jugular trunk | Left side of head and neck |
| Right subclavian trunk | Right upper limb, superficial regions of thoracic and upper abdominal wall |
| Left subclavian trunk | Left upper limb, superficial regions of thoracic and upper abdominal wall |
| Right bronchomediastinal trunk | Right lung and bronchi, mediastinal structures, thoracic wall |
| Left bronchomediastinal trunk | Left lung and bronchi, mediastinal structures, thoracic wall |
| Thoracic duct | Lower limbs, abdominal walls and viscera, pelvic walls and viscera, thoracic wall |
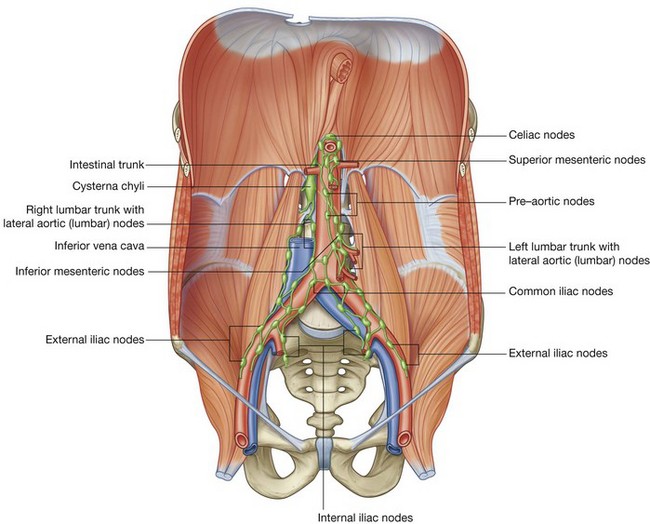
Pre-aortic and lateral aortic or lumbar nodes (para-aortic nodes)
Approaching the aortic bifurcation, the collections of lymphatics associated with the two common iliac arteries and veins merge, and multiple groups of lymphatic vessels and nodes associated with the abdominal aorta and inferior vena cava pass superiorly. These collections may be subdivided into pre-aortic nodes, which are anterior to the abdominal aorta, and right and left lateral aortic or lumbar nodes (para-aortic nodes), which are positioned on either side of the abdominal aorta (Fig. 4.152).
As these collections of lymphatics pass through the posterior abdominal region, they continue to collect lymph from a variety of structures. The lateral aortic or lumbar lymph nodes (para-aortic nodes) receive lymphatics from the body wall, the kidneys, the suprarenal glands, and the testes or ovaries.
The pre-aortic nodes are organized around the three anterior branches of the abdominal aorta that supply the abdominal part of the gastrointestinal tract, as well as the spleen, pancreas, gallbladder, and liver. They are divided into celiac, superior mesenteric, and inferior mesenteric nodes, and receive lymph from the organs supplied by the similarly named arteries.
Finally, the lateral aortic or lumbar nodes form the right and left lumbar trunks, whereas the pre-aortic nodes form the intestinal trunk (Fig. 4.152). These trunks come together and form a confluence that, at times, appears as a saccular dilation (the cisterna chyli). This confluence of lymph trunks is posterior to the right side of the abdominal aorta and anterior to the bodies of vertebrae LI and LII. It marks the beginning of the thoracic duct.
In the clinic
Retroperitoneal lymph node surgery
From a clinical perspective, retroperitoneal lymph nodes are arranged in two groups. The pre-aortic lymph node group drains lymph from the embryological midline structures, such as the liver, bowel, and pancreas. The para-aortic lymph node group (the lateral aortic or lumbar nodes), on either side of the aorta, drain lymph from bilateral structures, such as the kidneys and adrenal glands. Organs embryologically derived from the posterior abdominal wall also drain lymph to these nodes. These organs include the ovaries and the testes (importantly, the testes do not drain lymph to the inguinal regions).
In general, lymphatic drainage follows standard predictable routes; however, in the presence of disease, alternate routes of lymphatic drainage will occur.
There are a number of causes for enlarged retroperitoneal lymph nodes. In the adult, massively enlarged lymph nodes are a feature of lymphoma, and smaller lymph node enlargement is observed in the presence of infection and metastatic malignant spread of disease (e.g., colon cancer).
The treatment for malignant lymph node disease is based upon a number of factors, including the site of the primary tumor (e.g., bowel) and its histological cell type. Normally, the primary tumor is surgically removed and the lymph node spread and metastatic organ spread (e.g., to the liver and the lungs) are often treated with chemotherapy and radiotherapy.
In certain instances it may be considered appropriate to resect the lymph nodes in the retroperitoneum (e.g., for testicular cancer).
The surgical approach to retroperitoneal lymph node resection involves a lateral paramedian incision in the midclavicular line. The three layers of the anterolateral abdominal wall (external oblique, internal oblique, and transversus abdominis) are opened and the transversalis fascia is divided. The next structure the surgeon sees is the parietal peritoneum. Instead of entering the parietal peritoneum, which is standard procedure for most intra-abdominal surgical operations, the surgeon gently pushes the parietal peritoneum toward the midline, which moves the intra-abdominal contents and allows a clear view of the retroperitoneal structures. On the left, the para-aortic lymph node group (lateral aortic or lumbar nodes) are easily demonstrated with a clear view of the abdominal aorta and kidney. On the right the inferior vena cava is demonstrated, which has to be retracted to access to the right para-aortic lymph node chain (lateral aortic or lumbar nodes).
The procedure of the retroperitoneal lymph node dissection is extremely well tolerated and lacks the problems of entering the peritoneal cavity (e.g., paralytic ileus). Unfortunately, the complication of a vertical incision in the midclavicular line is to divide the segmental nerve supply to the rectus abdominis muscle. This produces muscle atrophy and asymmetric proportions to the anterior abdominal wall.
Nervous system in the posterior abdominal region
Several important components of the nervous system are in the posterior abdominal region. These include the sympathetic trunks and associated splanchnic nerves, the plexus of nerves and ganglia associated with the abdominal aorta, and the lumbar plexus of nerves.
Sympathetic trunks and splanchnic nerves
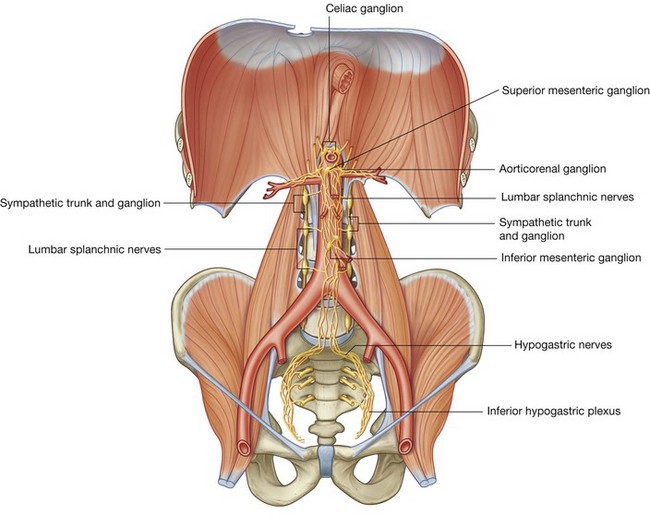
The sympathetic trunks pass through the posterior abdominal region anterolateral to the lumbar vertebral bodies, before continuing across the sacral promontory and into the pelvic cavity (Fig. 4.153). Along their course, small raised areas are visible. These represent collections of neuronal cell bodies—primarily postganglionic neuronal cell bodies—which are located outside the central nervous system. They are sympathetic paravertebral ganglia. There are usually four ganglia along the sympathetic trunks in the posterior abdominal region.
Also associated with the sympathetic trunks in the posterior abdominal region are the lumbar splanchnic nerves (Fig. 4.153). These components of the nervous system pass from the sympathetic trunks to the plexus of nerves and ganglia associated with the abdominal aorta. Usually two to four lumbar splanchnic nerves carry preganglionic sympathetic fibers and visceral afferent fibers.
Abdominal prevertebral plexus and ganglia
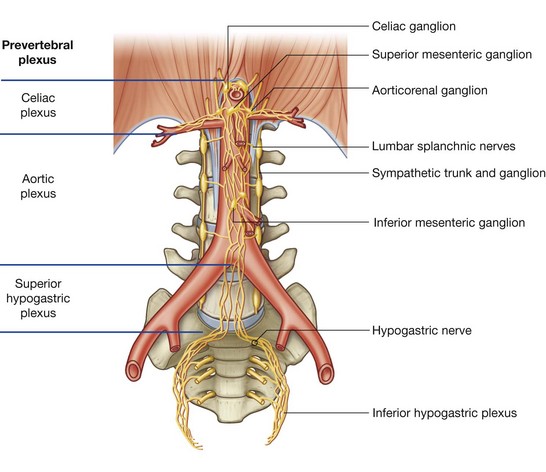
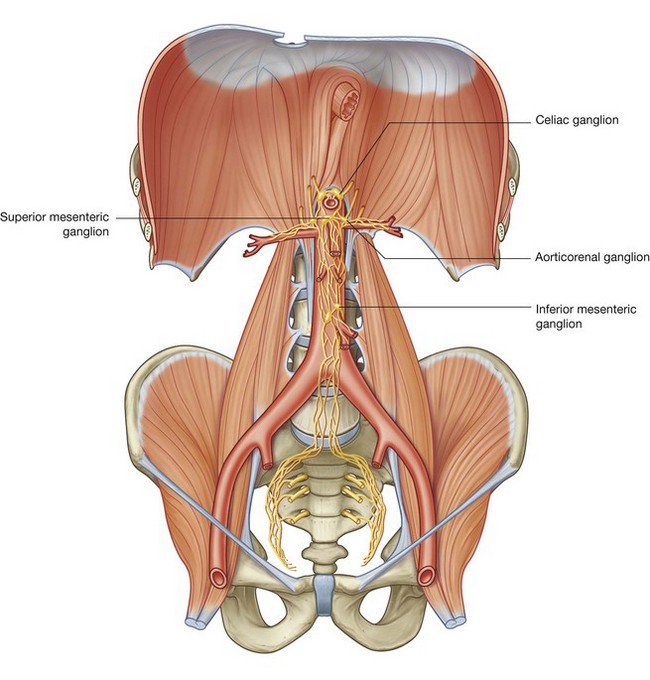
The abdominal prevertebral plexus is a network of nerve fibers surrounding the abdominal aorta. It extends from the aortic hiatus of the diaphragm to the bifurcation of the aorta into the right and left common iliac arteries. Along its route, it is subdivided into smaller, named plexuses (Fig. 4.154):
 beginning at the diaphragm and moving inferiorly, the initial accumulation of nerve fibers is referred to as the celiac plexus—this subdivision includes nerve fibers associated with the roots of the celiac trunk and superior mesenteric artery;
beginning at the diaphragm and moving inferiorly, the initial accumulation of nerve fibers is referred to as the celiac plexus—this subdivision includes nerve fibers associated with the roots of the celiac trunk and superior mesenteric artery; continuing inferiorly, the plexus of nerve fibers extending from just below the superior mesenteric artery to the aortic bifurcation is the abdominal aortic plexus (Fig. 4.155);
continuing inferiorly, the plexus of nerve fibers extending from just below the superior mesenteric artery to the aortic bifurcation is the abdominal aortic plexus (Fig. 4.155); at the bifurcation of the abdominal aorta, the abdominal prevertebral plexus continues inferiorly as the superior hypogastric plexus.
at the bifurcation of the abdominal aorta, the abdominal prevertebral plexus continues inferiorly as the superior hypogastric plexus.Throughout its length, the abdominal prevertebral plexus is a conduit for:
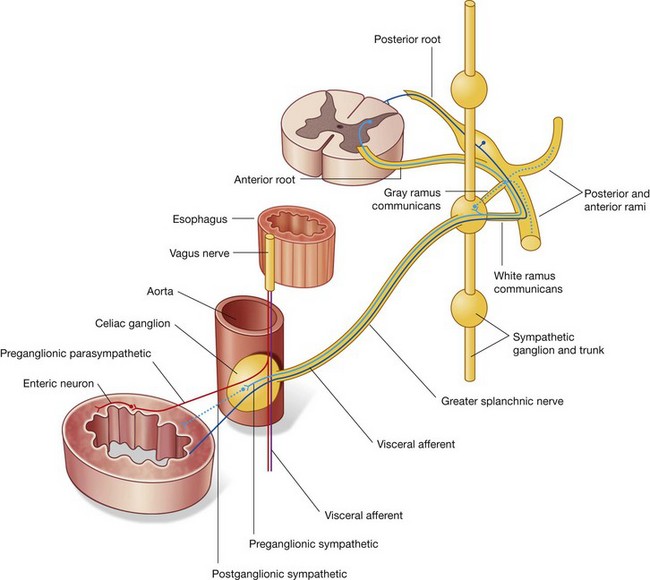
 preganglionic sympathetic and visceral afferent fibers from the thoracic and lumbar splanchnic nerves;
preganglionic sympathetic and visceral afferent fibers from the thoracic and lumbar splanchnic nerves;Associated with the abdominal prevertebral plexus are clumps of nervous tissue (the prevertebral ganglia), which are collections of postganglionic sympathetic neuronal cell bodies in recognizable aggregations along the abdominal prevertebral plexus; they are usually named after the nearest branch of the abdominal aorta. They are therefore referred to as celiac, superior mesenteric, aorticorenal, and inferior mesenteric ganglia (Fig. 4.156). These structures, along with the abdominal prevertebral plexus, play a critical role in the innervation of the abdominal viscera.
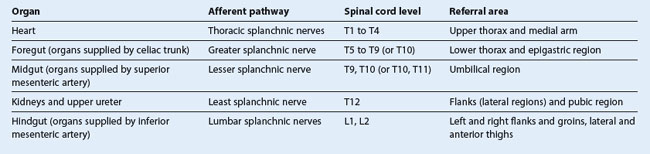
Common sites for pain referred from the abdominal viscera are given in Table 4.5.
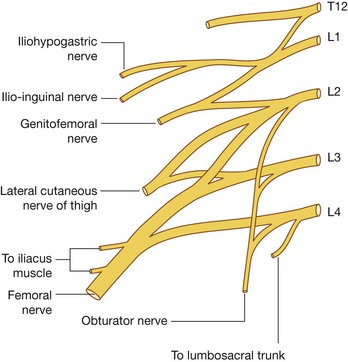
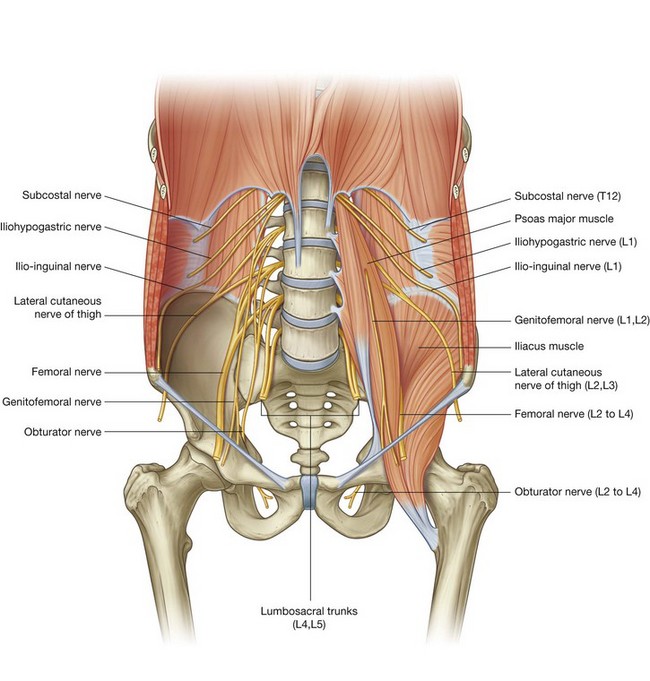
Lumbar plexus
The lumbar plexus is formed by the anterior rami of nerves L1 to L3, and most of the anterior ramus of L4 (Fig. 4.157 and Table 4.6). It also receives a contribution from the T12 (subcostal) nerve.
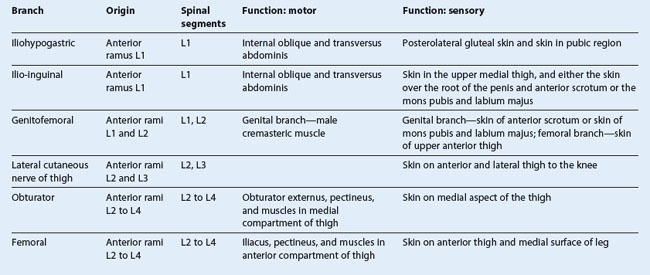
Branches of the lumbar plexus include the iliohypogastric, ilio-inguinal, genitofemoral, lateral cutaneous nerve of thigh (lateral femoral cutaneous), femoral, and obturator nerves. The lumbar plexus forms in the substance of the psoas major muscle anterior to its attachment to the transverse processes of the lumbar vertebrae (Fig. 4.158). Therefore, relative to the psoas major muscle, the various branches emerge either:
 lateral—iliohypogastric, ilio-inguinal, and femoral nerves, and the lateral cutaneous nerve of the thigh.
lateral—iliohypogastric, ilio-inguinal, and femoral nerves, and the lateral cutaneous nerve of the thigh.Iliohypogastric and ilio-inguinal nerves (L1)
The iliohypogastric and ilio-inguinal nerves arise as a single trunk from the anterior ramus of nerve L1 (Fig. 4.157). Either before or soon after emerging from the lateral border of the psoas major muscle, this single trunk divides into the iliohypogastric and the ilio-inguinal nerves.
Iliohypogastric nerve
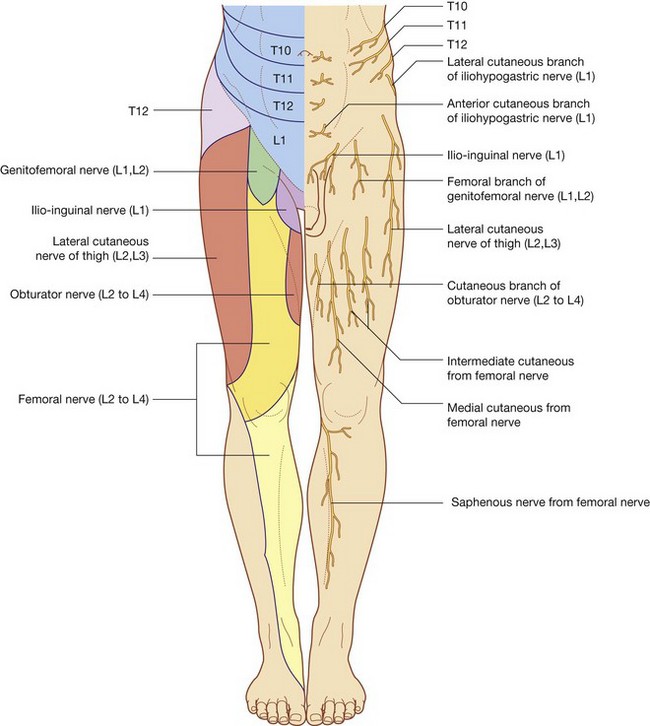
The iliohypogastric nerve passes across the anterior surface of the quadratus lumborum muscle, posterior to the kidney. It pierces the transversus abdominis muscle and continues anteriorly around the body between the transversus abdominis and internal oblique muscles. Above the iliac crest, a lateral cutaneous branch pierces the internal and external oblique muscles to supply the posterolateral gluteal skin (Fig. 4.159).
The remaining part of the iliohypogastric nerve (the anterior cutaneous branch) continues in an anterior direction, piercing the internal oblique just medial to the anterior superior iliac spine as it continues in an obliquely downward and medial direction. Becoming cutaneous, just above the superficial inguinal ring, after piercing the aponeurosis of the external oblique, it distributes to the skin in the pubic region (Fig. 4.159). Throughout its course, it also supplies branches to the abdominal musculature.
Ilio-inguinal nerve
The ilio-inguinal nerve is smaller than, and inferior to, the iliohypogastric nerve as it crosses the quadratus lumborum muscle. Its course is more oblique than that of the iliohypogastric nerve, and it usually crosses part of the iliacus muscle on its way to the iliac crest. Near the anterior end of the iliac crest, it pierces the transversus abdominis muscle, and then pierces the internal oblique muscle and enters the inguinal canal.
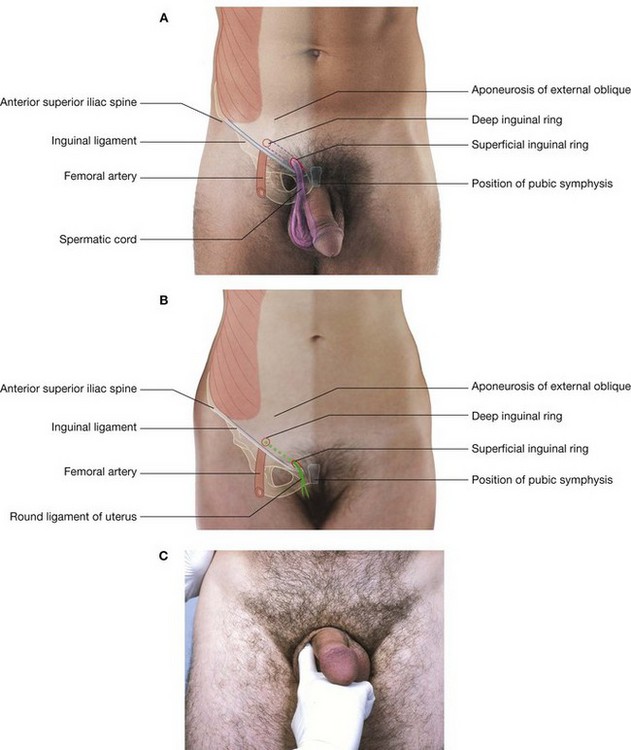
The ilio-inguinal nerve emerges through the superficial inguinal ring, along with the spermatic cord, and provides cutaneous innervation to the upper medial thigh, the root of the penis, and the anterior surface of the scrotum in men, or the mons pubis and labium majus in women (Fig. 4.159). Throughout its course, it also supplies branches to the abdominal musculature.
Genitofemoral nerve (L1 and L2)
The genitofemoral nerve arises from the anterior rami of the nerves L1 and L2 (Fig. 4.157). It passes downward in the substance of the psoas major muscle until it emerges on the anterior surface of psoas major. It then descends on the surface of the muscle, in a retroperitoneal position, passing posterior to the ureter. It eventually divides into genital and femoral branches.
The genital branch continues downward and enters the inguinal canal through the deep inguinal ring. It continues through the canal and:
 in men, innervates the cremasteric muscle and terminates on the skin in the upper anterior part of the scrotum; and
in men, innervates the cremasteric muscle and terminates on the skin in the upper anterior part of the scrotum; and in women, accompanies the round ligament of the uterus and terminates on the skin of the mons pubis and labium majus.
in women, accompanies the round ligament of the uterus and terminates on the skin of the mons pubis and labium majus.The femoral branch descends on the lateral side of the external iliac artery and passes posterior to the inguinal ligament, entering the femoral sheath lateral to the femoral artery. It pierces the anterior layer of the femoral sheath and the fascia lata to supply the skin of the upper anterior thigh (Fig. 4.159).
Lateral cutaneous nerve of thigh (L2 and L3)
The lateral cutaneous nerve of thigh arises from the anterior rami of nerves L2 and L3 (Fig. 4.157). It emerges from the lateral border of the psoas major muscle, passing obliquely downward across the iliacus muscle toward the anterior superior iliac spine (Fig. 4.159). It passes posterior to the inguinal ligament and enters the thigh.
The lateral cutaneous nerve of thigh supplies the skin on the anterior and lateral thigh to the level of the knee (Fig. 4.159).
Obturator nerve (L2 to L4)
The obturator nerve arises from the anterior rami of the nerves L2 to L4 (Fig. 4.157). It descends in the psoas major muscle, emerging from its medial side near the pelvic brim (Fig. 4.158).
The obturator nerve continues posterior to the common iliac vessels, passes across the lateral wall of the pelvic cavity, and enters the obturator canal, through which the obturator nerve gains access to the medial compartment of the thigh.
In the area of the obturator canal, the obturator nerve divides into anterior and posterior branches. On entering the medial compartment of the thigh, the two branches are separated by the obturator externus and adductor brevis muscles. Throughout their course through the medial compartment, these two branches supply:
 muscular branches to obturator externus, pectineus, adductor longus, gracilis, adductor brevis, and adductor magnus muscles;
muscular branches to obturator externus, pectineus, adductor longus, gracilis, adductor brevis, and adductor magnus muscles; in association with the saphenous nerve, cutaneous branches to the medial aspect of the upper part of the leg, and articular branches to the knee joint (Fig. 4.159).
in association with the saphenous nerve, cutaneous branches to the medial aspect of the upper part of the leg, and articular branches to the knee joint (Fig. 4.159).Femoral nerve (L2 to L4)
The femoral nerve arises from the anterior rami of nerves L2 to L4 (Fig. 4.157). It descends through the substance of the psoas major muscle, emerging from the lower lateral border of the psoas major (Fig. 4.158). Continuing its descent, the femoral nerve lies between the lateral border of the psoas major and the anterior surface of the iliacus muscle. It is deep to the iliacus fascia and lateral to the femoral artery as it passes posterior to the inguinal ligament and enters the anterior compartment of the thigh. Upon entering the thigh, it immediately divides into multiple branches.
Cutaneous branches of the femoral nerve include:
 medial and intermediate cutaneous nerves supplying the skin on the anterior surface of the thigh; and
medial and intermediate cutaneous nerves supplying the skin on the anterior surface of the thigh; andMuscular branches innervate the iliacus, pectineus, sartorius, rectus femoris, vastus medialis, vastus intermedius, and vastus lateralis muscles. Articular branches supply the hip and knee joints.