11 Antimicrobial Vaccines
Immunity, whether generated in reaction to immunization or administered as therapy, can prevent or lessen the serious symptoms of disease by blocking the spread of a bacterium, bacterial toxin, virus, or other microbe to its target organ or by acting rapidly at the site of infection. The memory immune responses activated upon challenge of an immunized individual are faster and stronger than for an unimmunized individual. The immunization of a population, like personal immunity, stops the spread of the infectious agent by reducing the number of susceptible hosts (herd immunity). Immunization programs on national and international levels have achieved the following goals:
1. Protection of population groups from the symptoms of pertussis, diphtheria, tetanus, and rabies
2. Protection and control of the spread of measles, mumps, rubella, varicella-zoster virus, Haemophilus influenzae type B (Hib), and Streptococcus pneumoniae
3. Elimination of wild-type poliomyelitis in most of the world and smallpox worldwide
In conjunction with immunization programs, measures can be taken to prevent disease by limiting the exposure of healthy people to infected people (quarantine) and by eliminating the source (e.g., water purification) or means of spread (e.g., mosquito eradication) of the infectious agent. Smallpox is an example of an infection that was controlled by such means. As of 1977, natural smallpox was eliminated through a successful World Health Organization (WHO) program that combined vaccination and quarantine. Polio and measles have also been targeted for elimination.
Vaccine-preventable diseases still occur, however, where immunization programs (1) are unavailable or too expensive (developing countries) or (2) are neglected (e.g., the United States). An example is measles, which causes 2 million deaths annually worldwide for the first reason and outbreaks of which continue to occur in the United States for the second reason.
Types of Immunization
The injection of purified antibody or antibody-containing serum to provide rapid, temporary protection or treatment of a person is termed passive immunization. Newborns receive natural passive immunity from maternal immunoglobulin that crosses the placenta or is present in the mother’s milk.
Active immunization occurs when an immune response is stimulated because of challenge with an immunogen, such as exposure to an infectious agent (natural immunization) or through exposure to microbes or their antigens in vaccines. On subsequent challenge with the virulent agent, a secondary immune response is activated that is faster and more effective at protecting the individual, or antibody is present to block the spread or function of the agent.
Passive Immunization
Passive immunization may be used as follows:
1. To prevent disease after a known exposure (e.g., needlestick injury with blood that is contaminated with hepatitis B virus [HBV])
2. To ameliorate the symptoms of an ongoing disease
3. To protect immunodeficient individuals
4. To block the action of bacterial toxins and prevent the diseases they cause (i.e., as therapy)
Immune serum globulin preparations derived from seropositive humans or animals (e.g., horses) are available as prophylaxis for several bacterial and viral diseases (Table 11-1). Human serum globulin is prepared from pooled plasma and contains the normal repertoire of antibodies for an adult. Special high-titer immune globulin preparations are available for hepatitis B virus (HBIg), varicella-zoster virus (VZIg), rabies (RIg), and tetanus (TIg). Human immunoglobulin is preferable to animal immunoglobulin because there is little risk of a hypersensitivity reaction (serum sickness).
Table 11-1 Immune Globulins Available for Postexposure Prophylaxis*
| Disease | Source |
|---|---|
| Hepatitis A | Human |
| Hepatitis B | Human |
| Measles | Human |
| Rabies | Human† |
| Chickenpox, varicella-zoster | Human† |
| Cytomegalovirus | Human |
| Tetanus | Human,† equine |
| Botulism | Equine |
| Diphtheria | Equine |
* Immune globulins to other agents may also be available.
† Specific high-titer antibody is available and is the preferred therapy.
Monoclonal antibody preparations are being developed for protection against various agents and diseases. In addition to infectious diseases, monoclonal antibodies are being used as therapy to block overzealous cytokine responses in inflammation and sepsis and for other therapies.
Active Immunization
The term vaccine is derived from vaccinia virus, a less virulent member of the poxvirus family that is used to immunize people against smallpox. Classical vaccines can be subdivided into two groups on the basis of whether they elicit an immune response on infection (live vaccines such as vaccinia) or not (inactivated–subunit-killed vaccines) (Figure 11-1). Deoxyribonucleic acid (DNA) vaccines represent a new means of immunization. In this approach, plasmid DNA is injected into muscle or skin, then taken up by dendritic, muscle, or macrophage cells, which express the gene for the immunogen as if for a natural infection. DNA vaccination stimulates T-cell immune responses, which can be boosted with antigen to elicit mature antibody responses.
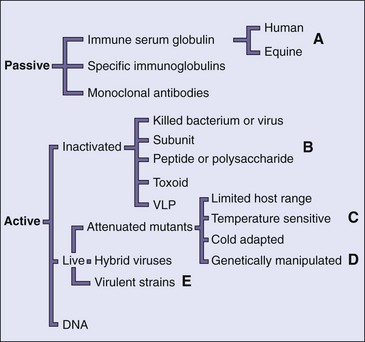
Figure 11-1 Types of immunizations. Antibodies (passive immunization) can be provided to block the action of an infectious agent, or an immune response can be elicited (active immunization) by natural infection or vaccination. The different forms of passive and active immunization are indicated. A, Equine antibodies can be used if human antibody is not available. B, Vaccine can consist of components purified from the infectious agent or can be developed through genetic engineering (virus-like particle [VLP]). C, Vaccine selected by passage at low or high temperature, in animals, embryonated eggs, or tissue culture cells. D, Deletion, insertion, reassortment, and other laboratory-derived mutants. E, Vaccine composed of a virus from a different species, which has a common antigen with the human virus.
Inactivated Vaccines
Inactivated vaccines utilize a large amount of antigen to produce a protective antibody response but without the risk of infection by the agent. Inactivated vaccines can be produced by chemical (e.g., formalin) or heat inactivation of bacteria, bacterial toxins, or viruses, or by purification or synthesis of the components or subunits of the infectious agents. Inactivated vaccines usually generate antibody (TH2 responses) and limited cell-mediated immune reponses.
These vaccines are usually administered with an adjuvant, which boosts their immunogenicity by enhancing uptake by or stimulating dendritic cells (DCs) and macrophages. Many adjuvants stimulate Toll-like receptors to activate these antigen-presenting cells. Most vaccines are precipitated onto alum to promote uptake by DCs and macrophages. MF59 (squalene microfluidized in an oil and water emulsion) and monophosphoryl lipid A (MPL) are adjuvants used in some newer vaccines. Experimental adjuvants include emulsions, virus-like particles, liposomes (defined lipid complexes), bacterial cell wall components, molecular cages for antigen, polymeric surfactants, and attenuated forms of cholera toxin and Escherichia coli lymphotoxin. These latter molecules are potent adjuvants for secretory antibody (immunoglobulin [Ig] A) after intranasal or oral immunization.
Inactivated, rather than live, vaccines are used to confer protection against most bacteria and viruses that cannot be attenuated, may cause recurrent infection, or have oncogenic potential. Inactivated vaccines are generally safe, except in people who have allergic reactions to vaccine components. For example, many antiviral vaccines are produced in eggs and therefore cannot be administered to people who are allergic to eggs. The disadvantages of inactivated vaccines are listed below and compared to live vaccines in Table 11-2.
1. Immunity is not usually lifelong.
2. Immunity may be only humoral (TH2) and not cell mediated.
3. The vaccine does not elicit a local IgA response.
Table 11-2 Advantages and Disadvantages of Live versus Inactivated Vaccines
| Property | Live | Inactivated |
|---|---|---|
| Route of administration | Natural* or injection | Injection |
| Dose of virus, cost | Low | High |
| Number of doses, amount | Single,† low | Multiple, high |
| Need for adjuvant | No | Yes‡ |
| Duration of immunity | Long-term | Short-term |
| Antibody response | IgG, IgA§ | IgG |
| Cell-mediated immune response | Good | Poor |
| Heat lability in tropics | Yes‖ | No |
| Interference¶ | Occasional | None |
| Side effects | Occasional mild symptoms** | Occasional sore arm |
| Reversion to virulence | Rarely | None |
Ig, Immunoglobulin.
* Oral or respiratory, in certain cases.
† A single booster may be required (yellow fever, measles, rubella) after 6 to 10 years.
‡ However, the commonly used alum is inefficient.
§ IgA if delivered via the oral or respiratory route.
‖ Magnesium chloride and other stabilizers and cold storage assist preservation.
¶ Interference from other viruses or diseases may prevent sufficient infection and immunity.
** Especially rubella and measles.
From White DO, Fenner FJ: Medical virology, ed 3, New York, 1986, Academic.
There are three major types of inactivated bacterial vaccines: toxoid (inactivated toxins), inactivated (killed) bacteria, and capsule or protein subunits of the bacteria. The bacterial vaccines currently available are listed in Table 11-3. Most antibacterial vaccines protect against the pathogenic action of toxins.
Table 11-3 Bacterial Vaccines*
| Bacteria (Disease) | Vaccine Components | Who Should Receive Vaccinations |
|---|---|---|
| Corynebacterium diphtheriae (diphtheria) | Toxoid | Children and adults |
| Clostridium tetani (tetanus) | Toxoid | Children and adults |
| Bordetella pertussis (pertussis) | Acellular | Children and teens |
| Haemophilus influenzae B (Hib) | Capsule polysaccharide-protein conjugate | Children |
| Neisseria meningitidis A and C (meningococcal disease) | Capsule polysaccharide-protein conjugate, capsule polysaccharide | People at high risk (e.g., those with asplenia), travelers to epidemic areas (e.g., military personnel), children |
| Streptococcus pneumoniae (pneumococcal disease; meningitis) | Capsule polysaccharides; capsule polysaccharide-protein conjugate | Children, people at high risk (e.g., those with asplenia), the elderly |
| Vibrio cholerae (cholera) | Killed cell | Travelers at risk to exposure |
| Salmonella typhi (typhoid) | Killed cell; polysaccharide | Travelers at risk to exposure, household contacts, sewage workers |
| Bacillus anthracis (anthrax) | Killed cell | Handlers of imported fur, military personnel |
| Yersinia pestis (plague) | Killed cell | Veterinarians, animal handlers |
| Francisella tularensis (tularemia) | Live attenuated | Animal handlers in endemic areas |
| Coxiella burnetii (Q fever) | Inactivated | Sheep handlers, laboratory personnel working with C. burnetii |
| Mycobacterium tuberculosis (tuberculosis) | Live attenuated bacillus Calmette-Guérin Mycobacterium bovis | Not recommended in United States |
* Listed in order of frequency of use.
Inactivated viral vaccines are available for polio, hepatitis A, influenza, and rabies, among other viruses. The Salk polio vaccine (inactivated poliomyelitis vaccine [IPV]) is prepared through the formaldehyde inactivation of virions. In the past, a rabies vaccine was prepared by means of formalin inactivation of infected rabbit neurons or duck embryos. Now, however, it is prepared through the chemical inactivation of virions grown in human diploid tissue culture cells. Because of the slow course of rabies, the vaccine can be administered immediately after a person is exposed to the virus and still elicit a protective antibody response.
A subunit vaccine consists of the bacterial or viral components that elicit a protective immune response. Surface structures of bacteria and the viral attachment proteins (capsid or glycoproteins) elicit protective antibodies. T-cell antigens may also be included in a subunit vaccine. The immunogenic component can be isolated from the bacterium, virus, or virally infected cells by biochemical means, or the vaccine can be prepared through genetic engineering by the expression of cloned viral genes in bacteria or eukaryotic cells. For example, the HBV subunit vaccine was initially prepared from surface antigen obtained from human sera of chronic carriers of the virus. Today HBV vaccine is obtained from yeast bearing the HBsAg gene. The antigen is purified, chemically treated, and absorbed onto alum to be used as a vaccine. The subunit proteins used in the HBV and the human papillomavirus (HPV) vaccines form virus-like particles (VLPs), which are more immunogenic than individual proteins.
The inactivated influenza vaccine consists of either a mixture of strains of viruses grown in embryonated eggs and then inactivated, or their protein subunits (hemagglutinin and neuraminidase). Tissue culture, cell-derived, and genetically engineered vaccines are in development. The vaccine is formulated annually to elicit protection from the virus strains predicted to threaten the population in the coming year.
Vaccines against H. influenzae B, Neisseria meningitidis, Salmonella typhi, and S. pneumoniae (23 strains) are prepared from capsular polysaccharides. Unfortunately, polysaccharides are generally poor immunogens (T-independent antigens). The meningococcal vaccine contains the polysaccharides of four major serotypes (A, C, Y, and W-135). The pneumococcal vaccine contains polysaccharides from 23 serotypes. The immunogenicity of polysaccharides can be enhanced by chemical linkage to a protein carrier (conjugate vaccine) (e.g., diphtheria toxoid, N. meningitidis outer membrane protein, or Corynebacterium diphtheriae protein) (Figure 11-2). The H. influenzae B (Hib) polysaccharide-diphtheria toxoid carrier complex is approved for administration to infants and children. An S. pneumoniae “pneumococcal” conjugate vaccine has been developed in which polysaccharide from the thirteen most prevalent strains in the United States is attached to a nontoxic form of the diphtheria toxin. This vaccine is available for use in infants and young children. The other polysaccharide vaccines are less immunogenic and should be administered to individuals older than 2 years.
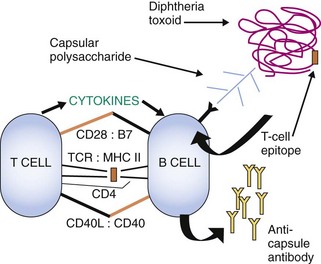
Figure 11-2 Capsular polysaccharide conjugate vaccines. Capsular polysaccharides are poor immunogens, do not elicit T-cell help, and only elicit IgM without memory. Capsule polysaccharide conjugated to a protein (e.g., diphtheria toxoid) binds to surface antipolysaccharide IgM on the B cell, the complex is internalized, processed and then a peptide is presented on major histocompatibility complex II (MHC II) to CD4 T cells. The T cells become activated, produce cytokines, and promote immunoglobulin class switching for the polysaccharide specific B cell. The B cell can become activated, make IgG, and memory cells will develop. TCR, T-cell receptor.
Live Vaccines
Live vaccines are prepared with organisms limited in their ability to cause disease (e.g., avirulent or attenuated organisms). Live vaccines are especially useful for protection against infections caused by enveloped viruses, which require T-cell immune responses for resolution of the infection. Immunization with a live vaccine resembles the natural infection in that the immune response progresses through the natural innate, TH1, and then TH2 immune responses, and humoral, cellular, and memory immune responses are developed. Immunity is generally long lived and, depending on the route of administration, can mimic the normal immune response to the infecting agent. However, the following list includes three problems with live vaccines:
1. The vaccine virus may still be dangerous for immunosuppressed people or pregnant women, who do not have the immunologic resources to resolve even a weakened virus infection.
Live bacterial vaccines include the orally administered live, attenuated S. typhi strain (Ty2la) vaccine for typhoid; the bacillus Calmette-Guérin (BCG) vaccine for tuberculosis, which consists of an attenuated strain of Mycobacterium bovis; and an attenuated tularemia vaccine. A combination of antibody and cell-mediated immune responses elicited by a live vaccine may be required against intracellularly growing bacteria. The BCG vaccine is not used in the United States because immunization is not always protective and people vaccinated with it show a false-positive reaction to the purified protein derivative (PPD) test, which is the screening test used to control tuberculosis in the United States.
Live virus vaccines consist of less virulent mutants (attenuated) of the wild-type virus, viruses from other species that share antigenic determinants (vaccinia for smallpox, bovine rotavirus), or genetically engineered viruses lacking virulence properties (see Figure 11-1). Wild-type viruses are attenuated by growth in embryonated eggs or tissue culture cells at nonphysiologic temperatures (25° C to 34° C) and away from the selective pressures of the host immune response. These conditions select for or allow the growth of viral strains (mutants) that (1) are less virulent because they grow poorly at 37° C (temperature-sensitive strains [e.g., measles vaccine] and cold-adapted strains [influenza vaccine]), (2) do not replicate well in any human cell (host-range mutants), (3) cannot escape immune control, or (4) can replicate at a benign site but do not disseminate, bind, or replicate in the target tissue characteristically affected by the disease (e.g., polio vaccine replicates in the gastrointestinal tract but does not reach or infect neurons). Table 11-4 lists examples of attenuated live virus vaccines currently in use.
Table 11-4 Viral Vaccines*
| Virus | Vaccine Components | Who Should Receive Vaccinations |
|---|---|---|
| Polio, inactivated | Trivalent (Salk vaccine) | Children |
| Attenuated polio | Live (oral polio vaccine, Sabin vaccine) | Children |
| Measles | Attenuated | Children |
| Mumps | Attenuated | Children |
| Rubella | Attenuated | Children |
| Varicella-zoster | Attenuated | Children |
| Rotavirus | Human-bovine hybrids | Infants |
| Attenuated | ||
| Human papilloma-virus | VLP | Girls aged 9-26 yr |
| Influenza | Inactivated | Children, adults, especially medical personnel, and the elderly |
| Attenuated (nasal spray) | 2-50 yr | |
| Hepatitis B | Subunit (VLP) | Newborns, health care workers, high-risk groups (e.g., sexually promiscuous, intravenous drug users) |
| Hepatitis A | Inactivated | Children, child care workers, travelers to endemic areas, Native Americans, and Alaskans |
| Adenovirus | Attenuated | Military personnel |
| Yellow fever | Attenuated | Travelers at risk to exposure, military personnel |
| Rabies | Inactivated | Anyone exposed to virus Preexposure: veterinarians, animal handlers |
| Smallpox | Live vaccinia virus | Protection from bioterrorism, military |
| Japanese encephalitis | Inactivated | Travelers at risk to exposure |
VLP, Virus-like particle.
* Listed in order of frequency of use.
The first vaccine—that for smallpox—was developed by Edward Jenner. The idea for the vaccine came to him when he noted that cowpox (vaccinia), a virulent virus from another species that shares antigenic determinants with smallpox, caused benign infections in humans but conferred protective immunity against smallpox. Similarly, a mixture of genetic reassortant human and bovine rotaviruses are the basis for one of the current vaccines administered to protect infants against human rotavirus.
Albert Sabin developed the first live oral polio vaccine (OPV) in the 1950s. The attenuated virus vaccine was obtained by multiple passages of the three types of poliovirus through monkey kidney tissue culture cells. At least 57 mutations accumulated in the polio type 1 vaccine strain. When this vaccine is administered orally, IgA is secreted in the gut and IgG in the serum, providing protection along the normal route of infection by the wild-type virus. This vaccine is inexpensive, easy to administer, and relatively stable and can spread to contacts of the immunized individual. Effective immunization programs have led to the elimination of wild-type polio in most of the world. The IPV is used in most of the world for routine well-baby immunizations because of the risk of vaccine-virus–induced polio disease by the OPV (see Figure 11-2).
The HBV and HPV vaccines are genetically engineered and grown in yeast cells. The viral attachment proteins, the surface antigen of HBV, and the L protein of HPV form viral-like particles. By limiting the spread of these viruses, these vaccines are also preventing their associated cancers (cervical carcinoma: HPV; primary hepatocellular carcinoma: HBV).
Live vaccines for measles, mumps, and rubella (administered together as the MMR vaccine), varicella-zoster, and now influenza have been developed. Protection against these infections requires a potent cellular immune response. To elicit a mature T-cell response, the vaccine must be administered after 1 year of age, when there will be no interference by maternal antibodies and cell-mediated immunity is sufficiently mature. A killed measles vaccine proved to be a failure because it conferred an incomplete immunity that induced more serious symptoms (atypical measles) on challenge with wild-type measles virus than the symptoms associated with the natural infection.
The initial live measles vaccine consisted of the Edmonston B strain, which was developed by Enders and colleagues. This virus underwent extensive passage at 35° C through primary human kidney cells, human amnion cells, and chicken embryo cells. The currently used Moraten (United States) and Schwarz (other countries) vaccine strains of measles were obtained by further passage of the Edmonston B strain in chick embryos at 32° C.
The mumps vaccine (Jeryl Lynn strain) and rubella vaccine (Wistar RA 27/3) viruses were also attenuated by extensive passage of the virus in cell culture. The varicella-zoster vaccine uses the Oka strain, an attenuated virus. The varicella-zoster vaccine is administered along with the MMR vaccine, or a stronger version is administered to adults to prevent zoster (shingles).
The live trivalent influenza vaccine is administered nasally within a mist and is cold adapted to 25° C. Unlike the previous inactivated vaccine, T- and B-cell responses and mucosal immunity are elicited by this vaccine. This vaccine can only be administered to individuals between ages 2 and 49 years.
Future Directions for Vaccination
Molecular biology techniques are being used to develop new vaccines. New live vaccines can be created by genetic engineering mutations to inactivate or delete a virulence gene instead of through random attenuation of the virus by passage through tissue culture. Genes from infectious agents that cannot be properly attenuated can be inserted into safe viruses (e.g., vaccinia, canarypox, attenuated adenovirus) to form hybrid virus vaccines. This approach holds the promise of allowing the development of a polyvalent vaccine to many agents in a single, safe, inexpensive, and relatively stable vector. On infection, the hybrid virus vaccine need not complete a replication cycle but simply promote the expression of the inserted gene to initiate an immune response to the antigens. The vaccinia, canarypox, and adenovirus virus vector systems have been used in several experimental hybrid vaccines. A canarypox human immunodeficiency virus (HIV) vaccine followed by two booster immunizations with recombinant HIV glycoprotein 120 showed modest but promising results. A vaccinia-based vaccine is used to immunize forest animals against rabies. Other viruses have also been considered as vectors.
Genetically engineered subunit vaccines are being developed through cloning of genes that encode immunogenic proteins into bacterial and eukaryotic vectors. The greatest difficulties in the development of such vaccines are (1) identifying the appropriate subunit or peptide immunogen that can elicit protective antibody and, ideally, T-cell responses and (2) presenting the antigen in the correct conformation. Once identified, the gene can be isolated, cloned, and expressed in bacteria or yeast cells, and then large quantities of these proteins can be produced. The envelope protein gp120 of HIV, the hemagglutinin of influenza, the G antigen of rabies, and the glycoprotein D of herpes simplex virus have been cloned, and their proteins have been generated in bacteria or eukaryotic cells for use (or potential use) as subunit vaccines.
Peptide subunit vaccines contain specific epitopes of microbial proteins that elicit neutralizing antibody or desired T-cell responses. To generate such a response, the peptide must contain sequences that bind to MHC I or MHC II (class I or class II major histocompatibility complex) proteins on DCs for presentation and recognition by T cells to initiate an immune response. The immunogenicity of the peptide can be enhanced by its covalent attachment to a carrier protein (e.g., tetanus toxoid or keyhole limpet hemocyanin [KLH]), a ligand for a Toll-like receptor (e.g., flagellin) or an immunologic peptide that can specifically present the epitope to the appropriate immune response. Better vaccines are being developed as the mechanisms of antigen presentation and T-cell receptor-specific antigens are better understood.
Adjuvants in addition to alum are being developed to enhance the immunogenicity and direct the response of vaccines to a TH1- or TH2-type of response. These include activators of Toll-like receptors, such as oligodeoxynucleotides of CpG, derivatives of lipid A from lipopolysaccharide, cytokines, liposomes, nanoparticles, etc. Use of MF59 in a new influenza vaccine (not available in the United States) allows reduction in the amount of antigen required to elicit protective immunity.
DNA vaccines offer great potential for immunization against infectious agents that require T-cell responses but are not appropriate for use in live vaccines. For these vaccines, the gene for a protein that elicits protective responses is cloned into a plasmid that allows the protein to be expressed in eukaryotic cells. The naked DNA is injected into the muscle or skin of the vaccine recipient, where the DNA is taken up by cells, the gene is expressed, and the protein is produced, presented to, and activates T-cell responses. DNA vaccines usually require a boost with antigenic protein to produce antibody.
A new approach, termed reverse vaccinology, was used to develop a vaccine for N. meningitidis B. Based on protein properties predicted from the gene sequence, thousands of proteins were tested for their ability to confer protection against infection to identify protein candidates. With the advent of this and other new technology, it should be possible to develop vaccines against infectious agents, such as Streptococcus mutans (to prevent tooth decay), the herpesviruses, HIV, and parasites, such as Plasmodium falciparum (malaria) and Leishmania. In fact, it should be possible to produce a vaccine to almost any infectious agent once the appropriate protective immunogen is identified and its gene isolated.
Immunization Programs
An effective vaccine program can save millions of dollars in health care costs. Such a program not only protects each vaccinated person against infection and disease but also reduces the number of susceptible people in the population, thereby preventing the spread of the infectious agent within the population. Although immunization may be the best means of protecting people against infection, vaccines cannot be developed for all infectious agents. One reason is that it is very time consuming and costly to develop vaccines. Box 11-1 lists the considerations that are weighed in the choice of a candidate for a vaccine program.
Natural smallpox was eliminated by means of an effective vaccine program because it was a good candidate for such a program; the virus existed in only one serotype, symptoms were always present in infected people, and the vaccine was relatively benign and stable. However, its elimination came about only as the result of a concerted, cooperative effort on the part of the WHO and local health agencies worldwide. Rhinovirus is an example of a poor candidate for vaccine development, because the viral disease is not serious and there are too many serotypes for vaccination to be successful. Practical aspects of and problems with vaccine development are listed in Box 11-2.
Box 11-2
Problems with Vaccine Use
Live vaccine can occasionally revert to virulent forms
Interference by other organisms may prevent the infection produced by a live virus vaccine (e.g., rubella prevents replication of poliovirus)
Vaccinating an immunocompromised person with a live vaccine can be life threatening
Side effects to vaccination can occur; these include hypersensitivity and allergic reactions to the antigen, to nonmicrobial material in the vaccine, and to contaminants (e.g., eggs)
Vaccine development is high risk and very expensive
Misinformation about safety causes underutilization of important vaccines
Microbes with many serotypes are difficult to control with vaccination
From the standpoint of the individual, the ideal vaccine should elicit dependable, lifelong immunity to infection without serious side effects. Factors that influence the success of an immunization program include not only the composition of the vaccine but also the timing, site, and conditions of its administration. Misinformation regarding safety issues with vaccines has deterred some individuals from being vaccinated putting them at risk to disease.
The recommended schedules of vaccinations for children are given in Figure 11-3. Tables of recommended schedules for vaccination of children, teens, adults, and for special cases are provided annually by the Advisory Committee on Immunization Practices (ACIP) of the Centers for Disease Control and Prevention. Booster immunizations of inactivated vaccines and the live measles vaccine are required later in life. Women younger than age 26 should receive the HPV vaccine, and college students should receive the meningococcal vaccine or a booster. Adults should be immunized with vaccines for S. pneumoniae (pneumococcus), influenza, rabies, HBV, and other diseases, depending on their jobs, the type of traveling they do, and other risk factors that may make them particularly susceptible to specific infectious agents. Further discussion of each of the vaccines is presented in later chapters with the disease they prevent.
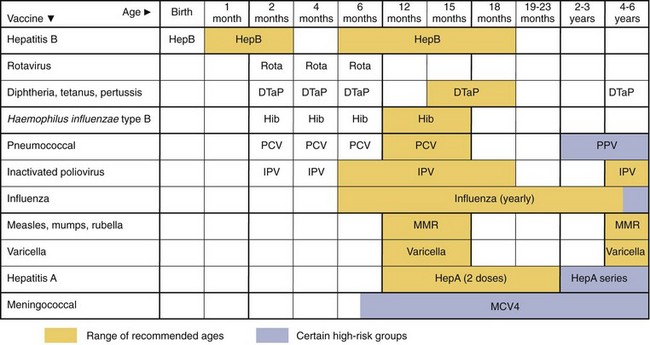
Figure 11-3 Recommended childhood immunization schedule from the Centers for Disease Control and Prevention. Vaccines are listed at the ages routinely recommended for their administration. Bars indicate the range of acceptable ages for vaccination. DTaP, Diphtheria, tetanus, and acellular pertussis; HepA, hepatitis A; HepB, hepatitis B; Hib, Haemophilus influenzae type B; IPV, inactivated poliovirus; MMR, measles, mumps, and rubella; MCV4, quadrivalent conjugated meningococcal; PCV, pneumococcal conjugate; PPV, pneumococcal polysaccharide; Rota, rotavirus.
(From the Centers for Disease Control and Prevention Advisory Committee on Immunization Practices: Recommended immunization schedule for persons aged 0 through 6 years--United States, 2012 (PDF). www.cdc.gov/vaccines/recs/schedules/downloads/child/0-6yrs-schedule-pr.pdf. Accessed May 25, 2012.)
1. Why is an inactivated rather than a live vaccine used for the following immunizations: rabies, influenza, tetanus, HBV, HiB, diphtheria, polio, and pertussis?
2. Tetanus is treated with passive immunization and prevented by active immunization. Compare the nature and function of each of these therapies.
3. The inactivated polio vaccine is administered intramuscularly, whereas the live polio vaccine is administered as an oral vaccine. How do the course of the immune response and the immunoglobulins produced in response to each vaccine differ? What step in poliovirus infection is blocked in a person vaccinated by each vaccine?
4. Why have large-scale vaccine programs not been developed for rhinovirus, herpes simplex virus, and respiratory syncytial virus?
5. Describe the public or personal health benefits that justify the development of the following major vaccine programs: measles, mumps, rubella, polio, smallpox, tetanus, and pertussis.
1. Inactivated vaccines are used when attenuated vaccines cannot be generated safely or when an antibody response is sufficient for protection. Although the inactivated vaccine is predominantly used, a live vaccine is now licensed for influenza.
2. Treatment by passive immunization with antibody is like treating the infection with a drug that blocks the action of the tetanus toxin; it is immediate but lasts only approximately 2 months, until the antibody is cleared from the system. Active immunization establishes cells that produce an immune response that lasts longer and is stronger but takes time to establish.
3. The inactivated polio vaccine elicits a predominantly antibody (TH2) response. This antibody does not prevent infection but is sufficient to block the progression of a polio virus in the bloodstream from reaching its target tissue (muscle and brain) and hence prevents disease.
The oral vaccine infects the individual with attenuated mutants of the three types of poliovirus to initiate a natural response to each virus, including a secretory IgA response. The development of memory cells is stronger and more permanent.
4. Vaccines to these microbes have not been developed for the following reasons:
Rhinovirus: too many serotypes; other viruses cause similar disease; and the disease is not life threatening.
Herpes simplex virus: protection requires antibody- and cell-mediated immunity but must block the spread from the initial site of infection to the neuron and virus may be hidden from antibody at this time (other vaccines need only block viremic spread).
Respiratory syncytial virus: antibody- and cell-mediated immunity must be elicited; the virus can spread from cell to cell and escape antibody control; although there are limited strains, multiple viruses can cause similar disease.
5. These agents cause significant morbidity and mortality in the infected individual. There are limited serotypes for these agents, and stabile, safe, and relatively inexpensive vaccines can be developed.
Measles and smallpox are major killers for which there is only one serotype of virus. In addition, smallpox always causes visible disease, which allows quarantine to facilitate the success of a vaccine program.
Mumps is problematic but usually not life threatening, but there is only one serotype and an effective live vaccine was developed and can be administered with the measles and rubella vaccines.
The rubella vaccine was developed to reduce the onset of congenital disease. Again, there is only one serotype.
Tetanus vaccine is a toxoid that elicits antibody that prevents the action of the toxin. Tetanus is a prevalent life-threatening disease.
The acellular pertussis vaccine prevents whooping cough, a deadly infection in young children. Increased onset of this disease in teens and adults has prompted the development of a booster shot.