37 Anaerobic, Non–Spore-Forming, Gram-Positive Bacteria
The non–spore-forming, gram-positive anaerobes are a miscellaneous collection of organisms that colonize the skin and mucous membranes of all humans.
1. With what disease is Mobiluncus associated?
2. What is characteristic about most infections with Actinomyces?
3. Lactobacillus causes what disease and how are Lactobacillus infections treated?
Answers
1. Mobiluncus is associated with bacterial vaginosis.
2. Infections with Actinomyces are characteristically chronic, requiring weeks to months to develop. The organisms also grow slowly in culture and respond slowly to antibiotic treatment.
3. Lactobacilli can cause transient bacteremia (typically from a genitourinary tract source), endocarditis, or opportunistic septicemia in an immunocompromised patient. Treatment of serious infections requires using the combination of penicillin with an aminoglycoside.
The anaerobic gram-positive cocci and non–spore-forming rods are a heterogeneous group of bacteria that characteristically colonize the skin and mucosal surfaces. These organisms are opportunistic pathogens, typically responsible for endogenous infections and usually recovered in mixtures of aerobic and anaerobic bacteria. In addition, most of these anaerobes have fastidious nutritional requirements and grow slowly on laboratory media. Thus the isolation and identification of individual strains are difficult and often time-consuming. Fortunately, the appropriate management and treatment of most infections with these organisms can be based on the knowledge that a mixture of aerobic and anaerobic organisms is present in the clinical specimen and does not require the isolation and identification of the individual organisms.
Anaerobic Gram-Positive Cocci (Table 37-1)
At one time, all clinically significant anaerobic cocci were included in the genus Peptostreptococcus. Unfortunately, it was recognized that these organisms were organized in a single genus based primarily on their Gram-stain morphology and inability to grow aerobically. More sophisticated methods have since been used to reclassify many of these species into six genera. The most common isolates are listed in Table 37-2. Although some anaerobic cocci are more virulent than others, and some are associated with specific diseases, specific identification of the different genera is generally unnecessary, and knowledge that anaerobic cocci are associated with an infection is typically sufficient.
Table 37-1 Important Anaerobic Gram-Positive Bacteria
| Organism | Historical Derivation |
|---|---|
| Anaerobic Cocci | |
| Anaerococcus | an, without; aer, air; coccus, berry or coccus (anaerobic coccus) |
| Finegoldia | Named after the American microbiologist Sid Finegold |
| Micromonas | micro, tiny; monas, cell (tiny cell) |
| Peptostreptococcus | pepto, cook or digest (the digesting streptococcus) |
| Schleiferella | Named after the German microbiologist K.H. Schleifer |
| Anaerobic Rods | |
| Actinomyces | aktinos, ray; mykes, fungus (ray fungus referring to the radial arrangement of filaments in granules) |
| Bifidobacterium | bifidus, cleft; bakterion, small rod (a small clefted or bifurcated rod) |
| Eubacterium | eu, good or beneficial (a beneficial rod; that is, a rod normally present) |
| Lactobacillus | lacto, milk (milk bacillus; organism originally recovered in milk; also, lactic acid is the primary metabolic product of fermentation) |
| Mobiluncus | mobilis, capable of movement or being active; uncus, hook (motile, curved rod) |
| Propionibacterium | propionicum, propionic acid (propionic acid is the primary metabolic product of fermentation) |
Table 37-2 New Classification of Selected Anaerobic Cocci Formerly in the Genus Peptostreptococcus
| Former Classification | New Classification |
|---|---|
| P. anaerobius | Unchanged |
| P. asaccharolyticus | Peptoniphilus asaccharolyticus |
| P. magnus | Finegoldia magna |
| P. micros | Unchanged |
| P. parvulus | Atopobium parvulum |
| P. prevotii | Anaerococcus prevotii |
The anaerobic gram-positive cocci normally colonize the oral cavity, gastrointestinal tract, genitourinary tract, and skin. They produce infections when they spread from these sites to normally sterile sites. For example, bacteria colonizing the upper airways can cause sinusitis and pleuropulmonary infections; bacteria in the intestines can cause intraabdominal infections; bacteria in the genitourinary tract can cause endometritis, pelvic abscesses, and salpingitis; bacteria on the skin can cause cellulitis and soft-tissue infections; and bacteria that invade the blood can produce infections in bones and solid organs (Figure 37-1).
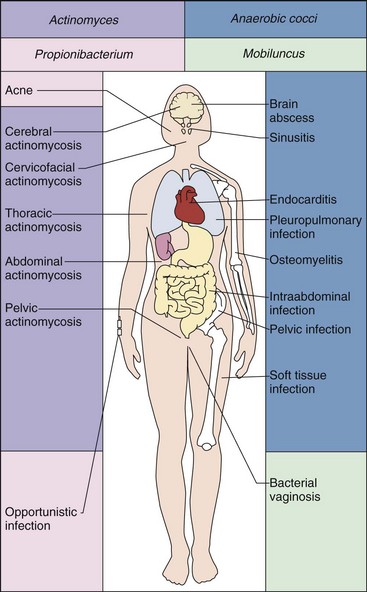
Figure 37-1 Diseases associated with anaerobic cocci and Actinomyces, Propionibacterium, and Mobiluncus, the latter three being anaerobic, non–spore-forming, gram-positive rods.
Laboratory confirmation of infections with anaerobic cocci is complicated by the following three factors: (1) care must be taken to prevent contamination of the clinical specimen with the anaerobic cocci that normally colonize the skin and mucosal surfaces; (2) the collected specimen must be transported in an oxygen-free container to prevent loss of the organisms; and (3) specimens should be cultured on nutritionally enriched media for a prolonged period (i.e., 5 to 7 days). In addition, some species of staphylococci and streptococci grow initially in an anaerobic atmosphere only and may be mistaken for anaerobic cocci. However, these organisms eventually grow well in air supplemented with 10% carbon dioxide (CO2), so they cannot be classified as anaerobes.
Anaerobic cocci are usually susceptible to penicillins and carbapenems (e.g., imipenem, meropenem); have intermediate susceptibility to broad-spectrum cephalosporins, clindamycin, erythromycin, and the tetracyclines; and are resistant to the aminoglycosides (as are all anaerobes). Specific therapy is generally indicated in monomicrobic infections; however, because most infections with these organisms are polymicrobic, broad-spectrum therapy against aerobic and anaerobic bacteria is usually selected.
Anaerobic, Non–Spore-Forming, Gram-Positive Rods (See Table 37-1)
The non–spore-forming, gram-positive rods are a diverse collection of facultatively anaerobic or strictly anaerobic bacteria that colonize the skin and mucosal surfaces (Table 37-3). Actinomyces, Mobiluncus, Lactobacillus, and Propionibacterium are well-recognized opportunistic pathogens, whereas other genera, such as Bifidobacterium and Eubacterium, can be isolated in clinical specimens but rarely cause human disease.
Table 37-3 Anaerobic, Non–Spore-Forming, Gram-Positive Rods
| Organism | Human Disease |
|---|---|
| Actinomyces spp. | Localized oral infections, actinomycosis (cervicofacial, thoracic, abdominal, pelvic, central nervous system) |
| Propionibacterium spp. | Acne, lacrimal canaliculitis, opportunistic infections |
| Mobiluncus spp. | Bacterial vaginosis, opportunistic infections |
| Lactobacillus spp. | Endocarditis, opportunistic infections |
| Eubacterium spp. | Opportunistic infections |
| Bifidobacterium spp. | Opportunistic infections |
Actinomyces
Physiology and Structure
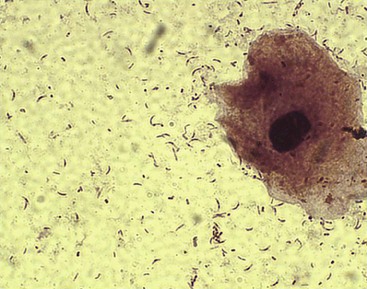
Actinomyces organisms are facultatively anaerobic or strictly anaerobic gram-positive rods. They are not acid-fast (in contrast to the morphologically similar Nocardia species), they grow slowly in culture, and they tend to produce chronic, slowly developing infections. They typically develop delicate filamentous forms or hyphae (resembling fungi) in clinical specimens or when isolated in culture (Figure 37-2). However, these organisms are true bacteria in that they lack mitochondria and a nuclear membrane, reproduce by fission, and are inhibited by penicillin but not antifungal antibiotics. Numerous species have been described; Actinomyces israelii, Actinomyces naeslundii, Actinomyces radingae, and Actinomyces turicensis are responsible for most human infections.
Pathogenesis and Immunity
Actinomyces organisms colonize the upper respiratory, gastrointestinal, and female genital tracts. These bacteria are not normally present on the skin surface. The organisms have a low virulence potential and cause disease only when the normal mucosal barriers are disrupted by trauma, surgery, or infection.
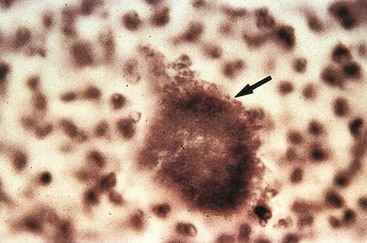
Classic disease caused by actinomyces is termed actinomycosis (in keeping with the original idea that these organisms were fungi or “mycoses”). Actinomycosis is characterized by the development of chronic granulomatous lesions that become suppurative and form abscesses connected by sinus tracts. Macroscopic colonies of organisms resembling grains of sand can frequently be seen in the abscesses and sinus tracts. These colonies, called sulfur granules because they appear yellow or orange, are masses of filamentous organisms bound together by calcium phosphate (Figure 37-3). The areas of suppuration are surrounded by fibrosing granulation tissue, which gives the surface overlying the involved tissues a hard or woody consistency. Actinomycosis is now relatively uncommon. Currently, most infections involving actinomyces are polymicrobic, oral infections such as endodontic infections, odontogenic abscesses, and dental implant–associated infections.
Epidemiology
Infections caused by actinomyces are endogenous, with no evidence of person-to-person spread or disease originating from an external source, such as soil or water. Cervicofacial infections are seen in patients who have poor oral hygiene or have undergone an invasive dental procedure or oral trauma. In these patients, the actinomyces that are present in the mouth invade into the diseased tissue and initiate the infectious process.
Patients with thoracic infections generally have a history of aspiration, with the disease becoming established in the lungs and then spreading to adjoining tissues. Abdominal infections most commonly occur in patients who have undergone gastrointestinal surgery or have suffered trauma to the bowel. Pelvic infection can be a secondary manifestation of abdominal actinomycosis or may be a primary infection in a woman with an intrauterine device (Figure 37-4). Central nervous system infections usually represent hematogenous spread from another infected tissue, such as the lungs.
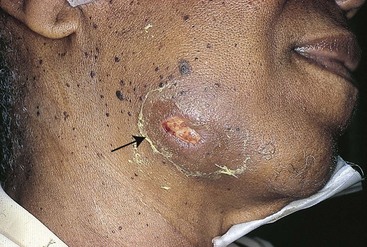
Clinical Diseases (Clinical Case 37-1)
Most actinomyces infections are the cervicofacial type (Figure 37-5). The disease may occur as an acute, pyogenic infection or as a slowly evolving, relatively painless process. The finding of tissue swelling with fibrosis and scarring, as well as draining sinus tracts along the angle of the jaw and neck, should alert the physician to the possibility of actinomycosis. Symptoms of thoracic actinomycosis are nonspecific. Abscesses may form in the lung tissue early in the disease and then spread into adjoining tissues as the disease progresses. Abdominal actinomycosis can spread throughout the abdomen, potentially involving virtually every organ system. Pelvic actinomycosis can occur as a relatively benign form of vaginitis or, more commonly, there can be extensive tissue destruction, including the development of tuboovarian abscesses or ureteral obstruction. The most common manifestation of central nervous system actinomycosis is a solitary brain abscess, but meningitis, subdural empyema, and epidural abscess are also seen. Actinomycosis in patients with chronic granulomatous disease, presenting as a nonspecific febrile illness, has recently been described.
Clinical Case 37-1
Pelvic Actinomycosis
Quercia and associates (Med Mal Infect 36:393–395, 2006) described a classic presentation of pelvic actinomycosis associated with an intrauterine contraceptive device (IUD). The patient is a 41-year-old woman who presented with a 5-month history of abdominal and pelvic pain, weight loss, malaise, and a yellow vaginal discharge. Since 1994 she had used an IUD, which was removed in June 2004. Her symptoms began soon after removal of the IUD. A computed tomography scan revealed a large pelvic mass involving the fallopian tubes as well as numerous hepatic abscesses. A surgical biopsy was performed, and Actinomyces was recovered in culture. She underwent surgical debridement and received oral therapy with a penicillin antibiotic for 1 year. The medical team thought the woman’s pelvis was infected with Actinomyces at the time the IUD was removed. This episode illustrates the chronic nature of actinomycosis and the need for surgical drainage and long-term antibiotic therapy.
Laboratory Diagnosis
Laboratory confirmation of actinomycosis is often difficult. Care must be used during collection of clinical specimens that they not become contaminated with Actinomyces that are part of the normal bacterial population on mucosal surfaces. The significance of Actinomyces isolated from contaminated specimens cannot be determined. Because the organisms are concentrated in sulfur granules and are sparse in involved tissues, a large amount of tissue or pus should be collected. If sulfur granules are detected in a sinus tract or in tissue, the granule should be crushed between two glass slides, stained, and examined microscopically. Thin, gram-positive, branching rods can be seen along the periphery of the granules.
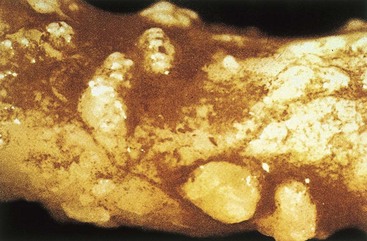
Actinomyces are fastidious and grow slowly under anaerobic conditions; it can take 2 weeks or more for the organisms to be isolated. Colonies appear white and have a domed surface that can become irregular after incubation for a week or more, resembling the top of a molar (Figure 37-6). The individual species of Actinomyces can be differentiated by biochemical tests; however, this process can be time consuming. In general, it is necessary to determine only that the isolate is a member of the genus Actinomyces.
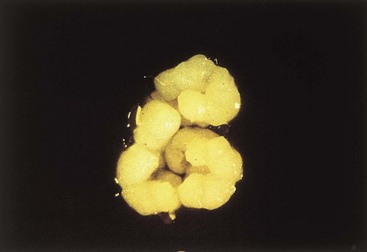
Figure 37-6 Molar tooth appearance of Actinomyces israelii after incubation for 1 week. This colonial morphology serves as a reminder that the bacteria are normally found in the mouth.
Recovery of Actinomyces in blood cultures should be evaluated carefully. Most isolates represent transient, insignificant bacteremia from the oropharynx or gastrointestinal tract. If the isolate is clinically significant, evidence of tissue pathology should be obtained.
Treatment, Prevention, and Control
Treatment for actinomycosis involves the combination of drainage of a localized abscess or surgical debridement of the involved tissues and the prolonged administration of antibiotics. Actinomyces are uniformly susceptible to penicillin (considered the antibiotic of choice), carbapenems, macrolides, and clindamycin. Most species are resistant to metronidazole, and the tetracyclines have variable activity. An undrained focus should be suspected in patients with infections that do not appear to respond to prolonged therapy (e.g., 4 to 12 months). The clinical response is generally good even in patients who have suffered extensive tissue destruction. Maintenance of good oral hygiene and the use of appropriate antibiotic prophylaxis when the mouth or gastrointestinal tract is penetrated can lower the risk of these infections.
Propionibacterium
Propionibacteria are small gram-positive rods often arranged in short chains or clumps (Figure 37-7). They are commonly found on the skin (in contrast with the Actinomyces), conjunctiva, external ear, and in the oropharynx and female genital tract. The organisms are anaerobic or aerotolerant, nonmotile, catalase positive, and capable of fermenting carbohydrates, producing propionic acid as their major byproduct (hence the name). The two most commonly isolated species are Propionibacterium acnes and Propionibacterium propionicum.
P. acnes is responsible for two types of infections: (1) acne vulgaris (as the name implies) in teenagers and young adults and (2) opportunistic infections (Clinical Case 37-2) in patients with prosthetic devices (e.g., artificial heart valves or joints) or intravascular lines (e.g., catheters, cerebrospinal fluid shunts). Propionibacteria are also commonly isolated in blood cultures, but this finding usually represents contamination with bacteria on the skin at the phlebotomy site.
Clinical Case 37-2
Shunt Infected with Propionibacterium
Chu and associates (Neurosurgery 49:717–720, 2001) reported three patients with central nervous system infections with Propionibacterium acnes. The following patient illustrates the problems with this organism. A 38-year-old woman with congenital hydrocephalus presented with a 1-week history of decreased level of consciousness, headaches, and emesis. She had undergone numerous ventriculoperitoneal shunt placements in the past, with the last one placed 5 years before this presentation. The patient was afebrile and had no meningeal signs, but she was somnolent and arousable only by deep stimuli. Cerebrospinal fluid (CSF) collected from the shunt contained no erythrocytes but had 55 WBCs; protein levels were high and glucose slightly low. Pleomorphic, gram-positive rods were observed on Gram stain and P. acnes grew in the anaerobic culture of the CSF. After 1 week of therapy with high dose penicillin, the CSF remained positive by Gram stain and culture. The patient was taken to surgery, where all foreign material was removed, and the patient was treated with penicillin for an additional 10 weeks. This patient illustrates the chronic, relatively asymptomatic nature of this disease, the need to remove the shunt and other foreign bodies, and the need to treat for a prolonged period of time.
The central role of P. acnes in acne is to stimulate an inflammatory response. Production of a low-molecular-weight peptide by the bacteria residing in sebaceous follicles attracts leukocytes. The bacteria are phagocytized and, after release of bacterial hydrolytic enzymes (lipases, proteases, neuraminidase, and hyaluronidase), stimulate a localized inflammatory response. P. propionicum is associated with endodontic abscesses and lacrimal canaliculitis (inflammation of the tear duct).
Propionibacteria can grow on most common media, although it may take 2 to 5 days for growth to appear. Care must be taken to avoid contamination of the specimen with the organisms normally found on the skin. The significance of the recovery of an isolate must also be interpreted in light of the clinical presentation (e.g., a catheter or other foreign body can serve as a focus for these opportunistic pathogens).
Acne is unrelated to the effectiveness of skin cleansing because the lesion develops within the sebaceous follicles. For this reason, acne is managed primarily through the topical application of benzoyl peroxide and antibiotics. Antibiotics such as erythromycin and clindamycin have proved effective for treatment.
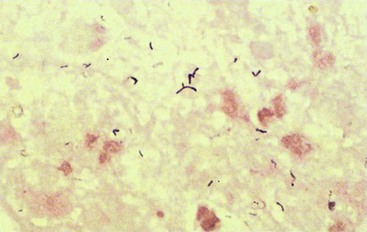
Mobiluncus
Members of the genus Mobiluncus are obligate anaerobic, gram-variable or gram-negative, curved rods with tapered ends. Despite their appearance in Gram-stained specimens (Figure 37-8), they are classified as gram-positive rods because they (1) have a gram-positive cell wall, (2) lack endotoxin, and (3) are susceptible to vancomycin, clindamycin, erythromycin, and ampicillin but resistant to colistin. The organisms are fastidious, growing slowly even on enriched media supplemented with rabbit or horse serum.
Of the two species of Mobiluncus, M. curtisii is rarely found in the vaginas of healthy women but is abundant in women with bacterial vaginosis (vaginitis). Their microscopic appearance is a useful marker for this disease, but the precise role of these organisms in the pathogenesis of bacterial vaginosis is unclear.
Lactobacillus
Lactobacillus species are facultatively anaerobic or strictly anaerobic rods. They are found as part of the normal flora of the mouth, stomach, intestines, and genitourinary tract. The organisms are most commonly isolated in urine specimens and blood cultures. Because lactobacilli are the most common organism in the urethra, their recovery in urine cultures usually is a result of contamination of the specimen, even when large numbers of the organisms are present. The reason lactobacilli rarely cause infections of the urinary tract is their inability to grow in urine. Invasion into blood occurs in one of the following three settings: (1) transient bacteremia from a genitourinary source (e.g., after childbirth or a gynecologic procedure), (2) endocarditis (Clinical Case 37-3), and (3) opportunistic septicemia in an immunocompromised patient. Strains of lactobacilli are used as probiotics and have occasionally been associated with human infections, most commonly in immunocompromised patients.
Clinical Case 37-3
Lactobacillus Endocarditis
The following is a classical description of endocarditis caused by Lactobacillus (Salvana and Frank, J Infect 53;5–10, 2006). A 62-year-old woman was admitted for atrial fibrillation and a 2-week history of flulike symptoms. The patient had had dental work performed 4 weeks before this admission and did not take antibiotic prophylaxis despite a history of rheumatic fever in childhood, with resultant mitral valve prolapse and regurgitation. On examination, the patient was afebrile, tachycardic, and mildly tachypneic. Cardiac examination was significant for a systolic murmur. Three blood cultures were collected, all of which yielded Lactobacillus acidophilus upon culture. The patient was treated with the combination of penicillin and gentamicin for a total of 6 weeks, resulting in complete recovery. This case illustrates the need for antibiotic prophylaxis during dental procedures for patients with underlying damaged heart valves, and the requirement for combined antibiotic therapy for successful treatment of serious infections caused by lactobacilli.
Treatment of endocarditis and opportunistic infections is difficult because lactobacilli are resistant to vancomycin (an antibiotic commonly active against gram-positive bacteria) and are inhibited but not killed by other antibiotics. A combination of penicillin with an aminoglycoside is required for bactericidal activity.
Bifidobacterium and Eubacterium
Bifidobacterium and Eubacterium species are commonly found in the oropharynx, large intestine, and vagina. These bacteria can be isolated in clinical specimens but have a very low virulence potential and usually represent clinically insignificant contaminants. Confirmation of their etiologic role in an infection requires their repeated isolation in large numbers from multiple specimens and the absence of other pathogenic organisms.
A 41-year-old man entered the university hospital for the treatment of a chronically draining wound in his jaw. The patient had undergone extraction of many teeth 3 months before admission and had poor oral hygiene and fetid breath at the time of admission. Multiple pustular nodules were observed overlying the carious teeth, and some nodules had ruptured. The drainage material consisted of serosanguineous fluid containing small, hard granules.
1. The diagnosis of actinomycosis is considered. How would you collect and transport specimens for confirmation of this diagnosis? What diagnostic tests can be performed?
2. Describe the epidemiology of actinomycosis. What is the risk factor for this patient?
3. What diseases does Propionibacterium cause? What is the most common source of this organism?
1. The diagnosis of actinomycosis can be difficult to confirm. Specimens that avoid oral contamination must be collected because Actinomyces are part of the normal oropharyngeal flora. Furthermore, relatively few organisms may be present in the specimen because this is a chronic infection, and cultures may need to be incubated for a week or more. For these reasons, many diagnoses of actinomycosis are not confirmed. Granules present in the specimens (referred to as “sulfur granules”) should be crushed and examined microscopically. Gram-positive rods embedded in amorphous mineral deposits should be observed.
2. Actinomyces colonize the oropharynx, gastrointestinal tract, and vagina. Infections with these organisms are commonly chronic, developing slowly after trauma to the colonized mucosa introduces the organisms into deep tissues. Infection is characterized by the development of chronic granulomatous lesions that become suppurative and form abscesses connected by sinus tracts. Pelvic actinomycosis is frequently associated with the presence of an intrauterine device. This patient’s poor oral hygiene predisposed him to cervicofacial actinomycosis.
3. Propionibacterium acnes is responsible for acne and opportunistic infections in patients with prosthetic devices or intravascular lines. Propionibacterium propionicus causes lacrimal canaliculitis (inflammation of the tear duct) and abscesses. Both organisms colonize the surface of the skin and mucosal membranes.