50 Adenoviruses
A 19-year-old army recruit complained that he had a high fever, chills, cough, runny nose, and sore throat. Several other members of his unit complained of similar symptoms.
1. How is adenovirus transmitted?
2. Which adenovirus types are most likely to cause acute respiratory distress syndrome?
3. What other diseases can adenoviruses cause?
4. What type of immune response would protect against infection?
5. Why did the military develop an attenuated vaccine for adenovirus strains 4 and 7?
Adenoviruses were first isolated in 1953 in a human adenoid cell culture. Since then, approximately 100 serotypes have been recognized, at least 52 of which infect humans. All human serotypes are included in a single genus within the family Adenoviridae. There are 7 subgroups for human adenoviruses (A through G) (Table 50-1). The viruses in each subgroup share many properties.
Table 50-1 Illnesses Associated with Adenoviruses
| Disease | Types | Patient Population |
|---|---|---|
| Respiratory Diseases | ||
| Febrile, undifferentiated upper respiratory tract infection | 1, 3, 5, 7, 14, 21, etc. | Infants, young children |
| Pharyngoconjunctival fever | 1, 2, 3, 4, 5, 7 | Children, adults |
| Acute respiratory disease | 4, 7, 14, 21 | Infants, young children; military recruits |
| Pertussis-like syndrome | 5 | Infants, young children |
| Pneumonia | 3, 4, 7, 21 | Infants, young children; military recruits; immunocompromised patients |
| Other Diseases | ||
| Acute hemorrhagic cystitis | 11, 21 | Children; immunocompromised patients |
| Epidemic keratoconjunctivitis | 8, 9,11, 19, 35, 37 | Any age |
| Gastroenteritis | 40, 41 | Infants, young children, immunocompromised patients |
| Hepatitis | 1-5, 7, 31 | Immunocompromised patients |
| Meningoencephalitis | 2, 7 | Children; immunocompromised patients |
The first human adenoviruses to be identified, numbered 1 to 7, are the most common. Common disorders caused by the adenoviruses include respiratory tract infection, pharyngoconjunctivitis (pinkeye), hemorrhagic cystitis, and gastroenteritis. Several adenoviruses have oncogenic potential in animals, but not humans, and for this reason have been extensively studied by molecular biologists. These studies have elucidated many viral and eukaryotic cellular processes. For example, analysis of the gene for the adenovirus hexon protein led to the discovery of introns and the splicing of eukaryotic messenger ribonucleic acid (mRNA). Adenovirus is also being used in genetic therapies to deliver deoxyribonucleic acid (DNA) for gene replacement therapy (e.g., cystic fibrosis), to express genes for other viruses (e.g., human immunodeficiency virus [HIV]) as a vaccine, and as oncolytic therapy.
Answers
1. The predominant route of transmission of adenovirus is aerosols, but adenovirus is also transmitted by contact and the fecal-oral route.
2. The most likely types are serotypes 4 and 7.
3. Conjunctivitis (pinkeye) and keratoconjunctivitis, pharyngoconjunctival fever, sore throat, common coldlike syndrome gastroenteritis, and systemic infection.
4. Replication of the virus will kill the infected cell, and so, antibody is sufficient to control the spread of adenoviruses. However, adenovirus can also establish chronic infection, and natural killer and T cells are important in killing and controlling the chronic and latent infection.
5. Adenovirus 4 and 7 are common causes of acute respiratory disease that spreads quickly to individuals in close proximity and under stress (military barracks). Infection of military personnel would rapidly spread and debilitate entire units, which would compromise their ability to serve their country.
Structure and Replication
Adenoviruses are double-stranded DNA viruses with a genome of approximately 36,000 base pairs, large enough to encode 30 to 40 genes. The adenovirus genome is a linear, double-stranded DNA with a terminal protein (molecular mass, 55 kDa) covalently attached at each 5′ end. The virions are nonenveloped icosadeltahedrons with a diameter of 70 to 90 nm (Figure 50-1 and Box 50-1). The capsid comprises 240 capsomeres, which consist of hexons and pentons. The 12 pentons, which are located at each of the vertices, have a penton base and a fiber. The fiber contains the viral attachment proteins and can act as a hemagglutinin. The penton base and fiber are toxic to cells. The pentons and fibers also carry type-specific antigens.
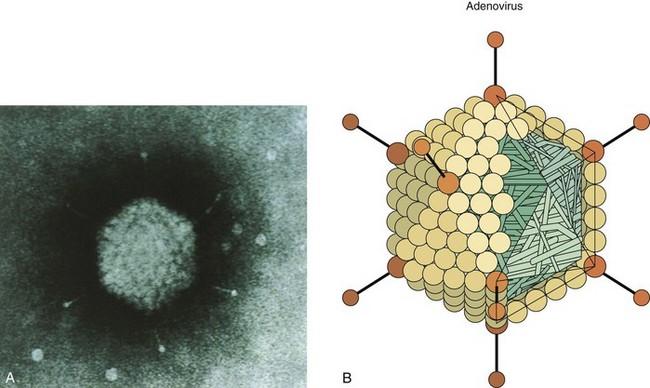
Figure 50-1 A, Electron micrograph of adenovirus virion with fibers. B, Model of adenovirus virion with fibers.
(A, From Valentine RC, Pereira HG: Antigens and structure of the adenovirus, J Mol Biol 13:13–20, 1965. B, From Armstrong D, Cohen J: Infectious diseases, St Louis, 1999, Mosby.)
Box 50-1
Unique Features of Adenovirus
Naked icosadeltahedral capsid has fibers (viral attachment proteins) at vertices.
Linear double-stranded genome has 5′ terminal proteins.
Synthesis of viral DNA polymerase activates a switch from early to late genes.
Virus encodes proteins to promote messenger RNA and DNA synthesis, including its own DNA polymerase.
Human adenoviruses are grouped A through G by DNA homologies and by serotype (more than 55 human types).
Serotype is mainly a result of differences in the penton base and fiber protein, which determine the nature of tissue tropism and disease.
Virus causes lytic, persistent, and latent infections in humans, and some strains can immortalize certain animal cells.
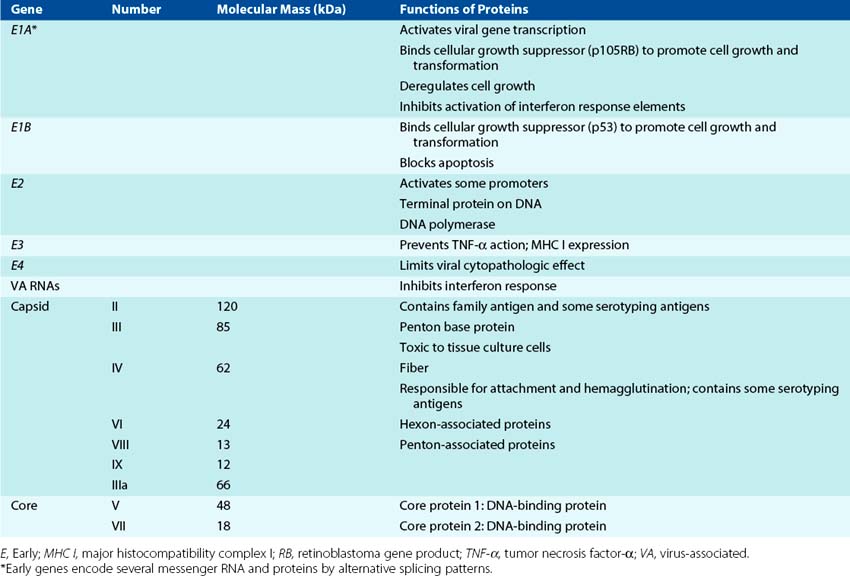
The core complex within the capsid includes viral DNA and at least two major proteins. There are at least 11 proteins in the adenovirus virion, 9 of which have an identified structural function (Table 50-2).
A map of the adenovirus genome shows the locations of the viral genes (Figure 50-2). The genes are transcribed from both DNA strands and in both directions at different times during the replication cycle. Genes for related functions are clustered together. Most of the RNA transcribed from the adenovirus genome is processed into several individual mRNAs in the nucleus. Early proteins promote cell growth and include a DNA polymerase that is involved in the replication of the genome. Adenovirus also encodes proteins that suppress apoptosis and host immune and inflammatory responses. Late proteins, which are synthesized after the onset of viral DNA replication, are primarily components of the capsid.
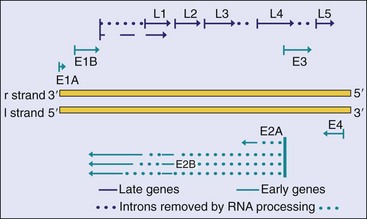
Figure 50-2 Simplified genome map of adenovirus type 2. Genes are transcribed from both strands (l and r) in opposite directions. The early genes are transcribed from four promoter sequences, and each generates several messenger RNAs by processing the primary RNA transcripts. This produces the full repertoire of viral proteins. The splicing pattern for only the E2 transcript is shown as an example. All of the late genes are transcribed from one promoter sequence. E, Early protein; L, late protein.
(Modified from Jawetz E, et al: Review of medical microbiology, ed 17, Norwalk, Conn, 1987, Appleton & Lange.)
The virus replication cycle takes approximately 32 to 36 hours and produces ≈10,000 virions. Attachment of the viral fiber proteins to a glycoprotein member of the immunoglobulin superfamily of proteins (approximately 100,000 fiber receptors are present on each cell) initiates infection for most adenoviruses. This same receptor is used by many Coxsackie B viruses; thus it is given the name Coxsackie adenovirus receptor. Some adenoviruses use the class I major histocompatibility complex (MHC I) molecule as a receptor. Then the penton base interacts with an αv integrin to promote internalization by receptor-mediated endocytosis in a clathrin-coated vesicle. The virus lyses the endosomal vesicle, and the capsid delivers the DNA genome to the nucleus. The penton and fiber proteins of the capsid are toxic to the cell and can inhibit cellular macromolecular synthesis.
Transcription of mRNA occurs in two phases. Early transcriptional events lead to the formation of proteins that can stimulate cell growth and promote viral DNA replication. As for the papovaviruses, several adenovirus mRNAs are transcribed from the same promoter and share initial sequences but are produced through the splicing out of different introns. Transcription of the early E1 gene, processing of the primary transcript (splicing out of introns to yield three mRNAs), and translation of the immediate early E1A transactivator protein are required for transcription of the early proteins. The early proteins include more DNA-binding proteins, the DNA polymerase, and proteins to help the virus escape the immune response. The E1A protein is also an oncogene, and together with the E1B protein, it can stimulate cell growth by binding to the cellular growth-suppressor proteins p105RB (p105RB retinoblastoma gene product) (E1A) and p53 (E1B). In permissive cells, stimulation of cell division facilitates transcription and replication of the genome, with cell death resulting from virus replication. In nonpermissive cells, the virus establishes latency, and the genome remains in the nucleus. For rodent cells, the E1A and E1B proteins may promote cell growth but without cell death, and therefore the virus oncogenically transforms the cell.
Viral DNA replication occurs in the nucleus and is mediated by the viral-encoded DNA polymerase. The polymerase uses the 55-kDa viral protein (terminal protein) with an attached cytosine monophosphate as a primer to replicate both strands of the DNA. The terminal protein remains attached to the DNA.
Late gene transcription starts after DNA replication. Most of the individual late mRNAs are generated from a large (83% of the genome) primary RNA transcript that is processed into individual mRNAs.
Capsid proteins are produced in the cytoplasm and then transported to the nucleus for viral assembly. Empty procapsids first assemble, and then the viral DNA and core proteins enter the capsid through an opening at one of the vertices. The replication and assembly process are inefficient and prone to error, producing as few as one infectious unit per 2300 particles. DNA, protein, and numerous defective particles accumulate in nuclear inclusion bodies. The virus remains in the cell and is released when the cell degenerates and lyses.
Pathogenesis and Immunity
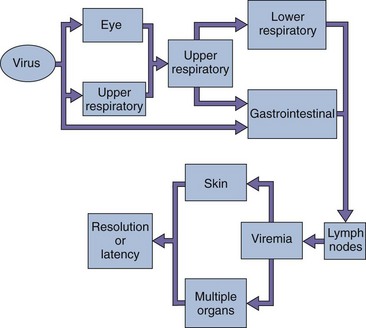
Adenoviruses are capable of causing lytic (e.g., mucoepithelial cells), latent (e.g., lymphoid and adenoid cells), and transforming (hamster, not human) infections. These viruses infect epithelial cells lining the oropharynx, as well as the respiratory and enteric organs (Box 50-2). The viral fiber proteins determine the target cell specificity. The toxic activity of the penton base protein can result in inhibition of cellular mRNA transport and protein synthesis, cell rounding, and tissue damage.
Box 50-2
Disease Mechanisms of Adenoviruses
Virus is spread by aerosol, close contact, or fecal-oral means to establish pharyngeal infection. Fingers spread virus to eyes.
Virus infects mucoepithelial cells in the respiratory tract, gastrointestinal tract, and conjunctiva or cornea, causing cell damage directly.
Disease is determined by the tissue tropism of the specific group or serotype of the virus strain.
Virus persists in lymphoid tissue (e.g., tonsils, adenoids, Peyer patches).
Antibody is important for prophylaxis and resolution but cell-mediated immunity is also important.
The histologic hallmark of adenovirus infection is a dense, central intranuclear inclusion (that consists of viral DNA and protein) within an infected epithelial cell (Figure 50-3). These inclusions may resemble those seen in cells infected with cytomegalovirus, but adenovirus does not cause cellular enlargement (cytomegaly). Mononuclear cell infiltrates and epithelial cell necrosis are seen at the site of infection.
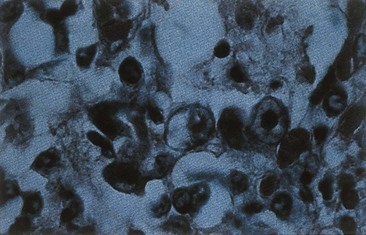
Figure 50-3 Histologic appearance of adenovirus-infected cells. Inefficient assembly of virions yields dark basophilic nuclear inclusion bodies containing DNA, proteins, and capsids.
Viremia may occur after local replication of the virus, with subsequent spread to visceral organs (Figure 50-4). This dissemination is more likely to occur in immunocompromised patients than in immunocompetent ones. The virus has a propensity to become latent and persist in lymphoid and other tissue, such as adenoids, tonsils, and Peyer patches, and can be reactivated in immunosuppressed patients. Although certain adenoviruses (groups A and B) are oncogenic in certain rodents, adenovirus transformation of human cells has not been observed.
Antibody is important for resolving lytic adenovirus infections and protects the person from reinfection with the same serotype but not other serotypes. Cell-mediated immunity is important in limiting virus outgrowth, and immunosuppressed people suffer more serious and recurrent disease. Adenoviruses have several mechanisms to evade host defenses and help them persist in the host. They encode small virus-associated RNAs (VA RNA) that prevent the activation of the interferon-induced protein kinase R–mediated inhibition of viral protein synthesis. The viral E3 and E1A proteins block apoptosis induced by cellular responses to the virus or by T-cell or cytokine (e.g., tumor necrosis factor-α [TNF-α]) actions. Some strains of adenoviruses can inhibit CD8+ cytotoxic T-cell action by preventing proper expression of MHC I molecules and therefore antigen presentation.
Epidemiology
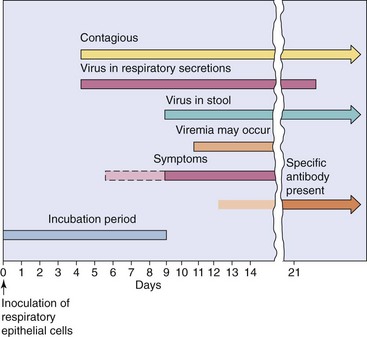
Adenovirus virions resist drying, detergents, gastrointestinal tract secretions (acid, protease, and bile), and even mild chlorine treatment (Box 50-3). These virions are spread in aerosols and by the fecal-oral route, by fingers, by fomites (including towels and medical instruments), and in poorly chlorinated swimming pools. Crowds and close proximity, as occurs in classrooms and military barracks, promotes spread of the virus. Adenoviruses may be shed intermittently and over long periods from the pharynx and especially in feces. Most infections are asymptomatic, a feature that greatly facilitates their spread in the community.
Adenoviruses 1 through 7 are the most prevalent serotypes. From 5% to 10% of cases of pediatric respiratory tract disease is caused by adenovirus types 1, 2, 5, and 6, and the infected children shed virus for months after infection. Adenovirus causes 15% of the cases of gastroenteritis requiring hospitalization. Serotypes 4 and 7 seem especially able to spread among military recruits because of their close proximity and rigorous lifestyle.
Clinical Syndromes (Box 50-4)
Adenoviruses primarily infect children and, less commonly, infect adults. Disease from reactivated virus occurs in immunocompromised children and adults. Specific clinical syndromes are associated with specific adenovirus types (see Table 50-1). The time course of adenovirus respiratory infection is shown in Figure 50-5.
Box 50-4
Clinical Summaries
Pharyngoconjunctival fever: A 7-year-old student develops sudden onset of red eyes, sore throat, and a fever of 38.9° C (102° F). Several children in the local elementary school have similar symptoms.
Gastroenteritis: An infant has diarrhea and is vomiting. Adenovirus serotype 41 is identified by polymerase chain reaction analysis of stool for epidemiologic reasons.
Acute Febrile Pharyngitis and Pharyngoconjunctival Fever
Adenovirus causes pharyngitis, which is often accompanied by conjunctivitis and pharyngoconjunctival fever. Pharyngitis alone occurs in young children, particularly those younger than 3 years, and may mimic streptococcal infection. Affected patients have mild, flulike symptoms (including nasal congestion, cough, coryza, malaise, fever, chills, myalgia, and headache) that may last 3 to 5 days. Pharyngoconjunctival fever occurs more often in outbreaks involving older children.
Acute Respiratory Disease
Acute respiratory disease is a syndrome consisting of fever, runny nose, cough, pharyngitis, and possible conjunctivitis (Clinical Case 50-1). The high incidence of infection of military recruits stimulated the development and use of a vaccine for these serotypes.
Clinical Case 50-1
Pathogenic Adenovirus 14
The Centers for Disease Control and Prevention (Morb Mortal Wkly Rep 56:1181–1184, 2007) reported that analysis of isolates from trainees during an outbreak of febrile respiratory infection at Lackland Air Force Base showed 63% resulting from adenovirus, and 90% of these were adenovirus 14. Of the 423 cases, 27 were hospitalized with pneumonia, 5 required admission to the intensive care unit, and 1 patient died. In an analogous case reported by CNN (www.cnn.com/2007/HEALTH/conditions/12/19/killer.cold/index.html), an 18-year-old high-school athlete complained of flulike symptoms, with vomiting, chills, and fever of 104° F that progressed to life-threatening pneumonia within days. The adenovirus causing these infections is a mutant of the adenovirus 14 that was first identified in 1955. The adenovirus 14 mutant has spread around the United States, putting adults at risk to severe disease. Adenovirus 14 infection usually causes a benign respiratory infection in adults, with newborns and the elderly at higher risk for severe outcomes. Although most virus mutations produce a weaker virus, occasionally, a more virulent, antibody-escape, or antiviral-drug–resistant virus may occur.
Other Respiratory Tract Diseases
Adenoviruses cause coldlike symptoms, laryngitis, croup, and bronchiolitis. They can also cause a pertussis-like illness in children and adults that consists of a prolonged clinical course and true viral pneumonia.
Conjunctivitis and Epidemic Keratoconjunctivitis
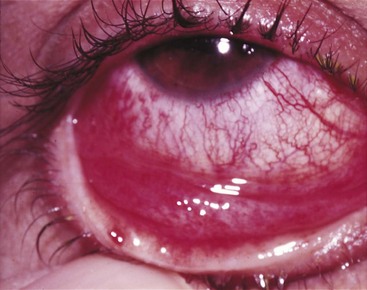
Adenoviruses cause a follicular conjunctivitis in which the mucosa of the palpebral conjunctiva becomes pebbled or nodular, and both conjunctivae (palpebral and bulbar) become inflamed (Figure 50-6). Such conjunctivitis may occur sporadically or in outbreaks that can be traced to a common source. Swimming pool conjunctivitis is a familiar example of a common-source adenovirus infection. Epidemic keratoconjunctivitis may be an occupational hazard for industrial workers. The most striking such epidemic occurred in people working in the naval shipyards of Pearl Harbor in Hawaii, where it caused more than 10,000 cases during 1941 and 1942. Irritation of the eye by a foreign body, dust, debris, and so forth is a risk factor for the acquisition of this infection.
Gastroenteritis and Diarrhea
Adenovirus is a major cause of acute viral gastroenteritis especially in infants. The enteric adenoviruses (types 40 to 42) do not replicate in the same tissue culture cells as do other adenoviruses and rarely cause fever or respiratory tract symptoms.
Other Manifestations
Adenovirus has also been associated with intussusception in young children, acute hemorrhagic cystitis with dysuria and hematuria in young boys, musculoskeletal disorders, and genital and skin infections. Adenovirus (type 36) is also associated with obesity.
Systemic Infection in Immunocompromised Patients
Immunocompromised patients are at risk for serious adenovirus infections, although not as much as they are for infections caused by herpesviruses. Adenoviral disease in immunocompromised patients includes pneumonia and hepatitis. Infection can originate from exogenous or endogenous (reactivation) sources.
Laboratory Diagnosis
For the results of virus isolation to be significant, the isolate should be obtained from a site or secretion relevant to the disease symptoms. The presence of adenovirus in the throat of a patient with pharyngitis is usually diagnostic if laboratory findings eliminate other common causes of pharyngitis, such as Streptococcus pyogenes.
Direct analysis of the clinical sample without virus isolation can be used for rapid detection and identification of adenoviruses. Immunoassays, including fluorescent antibody and enzyme-linked immunosorbent assay, and genome assays, including different variations of the polymerase chain reaction (PCR) and DNA probe analysis, can be used to detect, type, and group the virus in clinical samples and tissue cultures. These approaches must be used for enteric adenovirus serotypes 40 to 42, which do not grow readily in available cell cultures. Serologic testing is rarely used except for epidemiologic purposes.
The isolation of most adenovirus types is best accomplished in cell cultures derived from epithelial cells (e.g., primary human embryonic kidney cells, continuous [transformed] lines, such as HeLa and human epidermal carcinoma cells). Within 2 to 20 days, the virus causes a lytic infection with characteristic inclusion bodies and cell death. Recovery of virus from cell culture requires an average of 6 days. The characteristic intranuclear inclusions can be seen in infected tissue during histologic examination. However, such inclusions are rare and must be distinguished from those produced by cytomegalovirus.
Treatment, Prevention, and Control
Careful handwashing and chlorination of swimming pools can reduce transmission of adenovirus. There is no approved treatment for adenovirus infection. Live oral vaccines have been used to prevent infections with adenovirus types 4 and 7 in military recruits but are not used in civilian populations.
Therapeutic Adenoviruses
Adenoviruses have been used and are being considered for gene delivery for correction of human diseases, including immune deficiencies (e.g., adenosine deaminase deficiency), cystic fibrosis, and lysosomal storage diseases. The virus is inactivated by deletion or mutation of the E1 and other viral genes (e.g., E2, E4). The appropriate gene is inserted into the viral genome, replacing this DNA, and is controlled by an appropriate promoter. The resultant virus vector must be grown in a cell that expresses the missing viral functions (E1, E4) to complement the deficiency and allow production of virus. Types 4 and 7 and replication defective mutants of types 5, 26, and 35 are being developed to carry genes of HIV, Ebola, and other viruses as attenuated vaccines for these deadly viruses. Adenoviruses lacking a functional E1B gene creates a virus that selectively grows and kills tumor cells that lack p53 providing oncolytic therapy. Despite the genetically engineered attenuation, these viruses still may cause serious disease in immunocompromised individuals.
A 7-year-old boy attending summer camp complains of sore throat, headache, cough, red eyes, and tiredness and is sent to the infirmary. His temperature is 40° C. Within hours, other campers and counselors visit the infirmary with similar symptoms. Symptoms last for 5 to 7 days. All the patients have gone swimming in the camp pond. More than 50% of the people in the camp complain of symptoms similar to those in the initial case. The Public Health Department identifies the agent as adenovirus serotype 3.
1. Toward which adenovirus syndrome do the symptoms point?
2. An outbreak as large as this indicates a common source of infection. What was the most likely source or sources? What were the most likely routes by which the virus was spread?
3. What physical properties of the virus facilitate its transmission?
4. What precautions should the camp owners take to prevent other outbreaks?
5. What sample or samples would have been used by the Public Health Department to identify the infectious agent, and what tests would be required to diagnose the infection?
1. The patient has disease signs consistent with pharyngoconjunctival fever.
2. The most likely source of this outbreak is the unchlorinated water in the camp pond. The virus is very hardy and can endure relatively harsh conditions.
3. The capsid of the adenovirus protects the virus from harsh conditions of drying and even the acid and bile of the gastrointestinal tract to allow the virus to be transmitted by fecal-oral and respiratory routes, through contact, and on fomites.
4. Contamination of the pond would be difficult to eliminate. There is no vaccine to protect the campers. However, greater care with sewage may prevent further contamination of the pond. Also, campers should not share towels or other items that may come into contact with virus.
5. An eye swab, a fecal sample, and a nasal wipe could be tested for the virus in the infected child. Pond water could be concentrated to allow detection of virus as a common source of the infection. The presence of adenovirus and its type would be analyzed by PCR.