53 Parvoviruses
A 6-year-old girl had a viral respiratory infection and then became very pale, weak, and tired and became severely anemic because of a transient aplastic crisis.
1. What predisposing condition exacerbated the relatively benign disease in this child?
2. What cell type is the host for this virus, and what determines this tropism?
3. What disease signs occur following infection of an adult? Of a fetus?
Answers
1. Chronic hemolytic anemia, such as sickle cell anemia, puts an individual at risk because it compounds the loss of erythrocyte production resulting from the virus infection of erythroid precursors.
2. Erythroid precursors are the host cell for the virus. The virus requires a growing cell to replicate and binds to the blood group P antigen (globoside).
3. Infection of an adult may result in acute polyarthritis because of immune complex–mediated inflammatory reactions. Infection of the fetus can result in hydrops fetalis. The virus infects the the erythroid precursors of the fetus, killing them and causing anemia and congestive heart failure.
The Parvoviridae are the smallest of the deoxyribonucleic acid (DNA) viruses. Their small size and limited genetic repertoire make them more dependent than any other DNA virus on the host cell, or they require the presence of a helper virus to replicate. B19 and bocavirus are the only parvoviruses known to cause human disease.
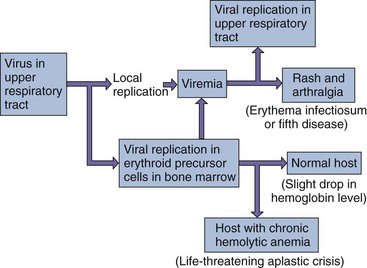
B19 normally causes erythema infectiosum, or fifth disease, a mild febrile exanthematous disease that occurs in children. It goes by the latter name because it was counted as one of five classic childhood exanthems (the first four being varicella, rubella, roseola, and measles). B19 is also responsible for episodes of aplastic crisis in patients with chronic hemolytic anemia and is associated with acute polyarthritis in adults. Intrauterine infection of a fetus may cause abortion.
Bocavirus is a recently discovered virus that can cause acute respiratory disease, which may become severe in young children.
Other parvoviruses, such as RA-1 (isolated from a person with rheumatoid arthritis) and fecal parvoviruses, have not been shown to cause human disease. Feline and canine parvoviruses do not cause human disease and are preventable with vaccination of the pet.
Adeno-associated viruses (AAVs) are members of the genus Dependovirus. They commonly infect humans but replicate only in association with a second “helper” virus, usually an adenovirus. Dependoviruses neither cause illness nor modify infection by their helper viruses. These properties and the propensity of AAVs to integrate into the host chromosome have made genetically modified AAVs candidates for use in gene-replacement therapy. A third genus of the family, Densovirus, infects only insects.
Structure and Replication
The parvoviruses are extremely small (18 to 26 nm in diameter) and have a nonenveloped, icosahedral capsid (Box 53-1 and Figure 53-1). The B19 virus genome contains one linear, single-stranded DNA molecule with a molecular mass of 1.5 to 1.8 × 106 Da (5500 bases in length) (Box 53-2). Plus or minus DNA strands are packaged separately into virions. The genome encodes three structural and two major nonstructural proteins. Unlike larger DNA viruses, the parvoviruses must infect mitotically active cells because they do not encode the means to stimulate cell growth or a polymerase. Only one serotype of B19 is known to exist.
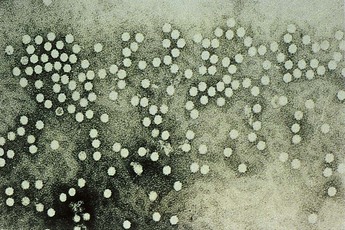
Figure 53-1 Electron micrograph of parvovirus. Parvoviruses are small (18 to 26 nm), nonenveloped viruses with single-stranded DNA.
(Courtesy Centers for Disease Control and Prevention, Atlanta.)
Box 53-2
Parvovirus Genome
Single-stranded linear DNA genome
Approximately 5.5 kilobases in length
Plus and minus strands packaged into separate B19 virions
Ends of the genome have inverted repeats that hybridize to form hairpin loops and a primer for DNA synthesis
Separate coding regions for nonstructural (NS) and structural proteins (VP)
B19 virus replicates in mitotically active cells and prefers cells of the erythroid lineage, such as fresh human bone marrow cells, erythroid cells from fetal liver, and erythroid leukemia cells (Figure 53-2). After binding to the erythrocyte blood group P antigen (globoside) and its internalization, the virion is uncoated, and the single-stranded DNA genome is delivered to the nucleus. Factors available only during the S phase of the cell’s growth cycle and cellular DNA polymerases are required to generate a complementary DNA strand.
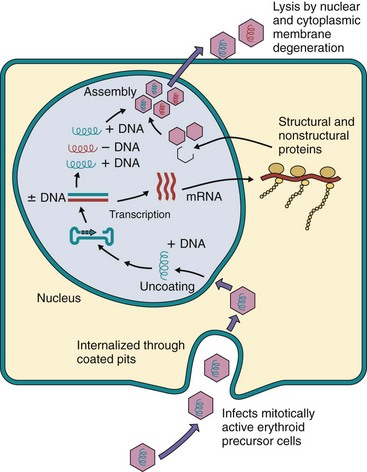
Figure 53-2 Postulated replication of parvovirus (B19) based on information from related viruses (minute virus of mice). The internalized parvovirus delivers its genome to the nucleus, where the single-stranded (plus or minus) DNA is converted to double-stranded DNA by host factors and DNA polymerases present only in growing cells. Transcription, replication, and assembly occur in the nucleus. Virus is released by cell lysis.
The single-stranded DNA virion genome is converted to a double-stranded DNA version, which is required for transcription and replication. Inverted repeat sequences of DNA at both ends of the genome fold back and hybridize with the genome to create a primer for the cell’s DNA polymerase. This creates the complementary strand and replicates the genome. The two major nonstructural proteins and the VP1 and VP2 structural capsid proteins are synthesized in the cytoplasm, and the structural proteins go to the nucleus, where the virion is assembled. The VP2 protein is cleaved later to produce VP3. The nuclear and cytoplasmic membrane degenerates, and the virus is released on cell lysis.
Pathogenesis and Immunity
B19 targets and is cytolytic for erythroid precursor cells (Box 53-3). B19 disease is determined by the direct killing of these cells and the subsequent immune response to the infection (rash and arthralgia).
Box 53-3
Disease Mechanisms of B19 Parvovirus
Virus spreads by respiratory and oral secretions.
Virus infects mitotically active erythroid precursor cells in bone marrow and establishes lytic infection.
Virus establishes large viremia and can cross the placenta.
Antibody is important for resolution and prophylaxis.
Virus causes biphasic disease.
Depletion of erythroid precursor cells and destabilization of erythrocytes initiate aplastic crisis in persons with chronic anemia.
Studies performed in volunteers suggest that B19 virus first replicates in the nasopharynx or upper respiratory tract then spreads by viremia to the bone marrow and elsewhere, where it replicates and kills erythroid precursor cells (Figure 53-3). Bocavirus also intiates infection in the respiratory tract, replicates in the respiratory epithelium, and causes disease.
B19 viral disease has a biphasic course. The initial febrile stage is the infectious stage. During this time, erythrocyte production is stopped for approximately 1 week because of the viral killing of erythroid precursor cells. A large viremia occurs within 8 days of infection and is accompanied by nonspecific flulike symptoms. Large numbers of virus are also released into oral and respiratory secretions. Antibody stops the viremia and is important for resolution of the disease but contributes to the symptoms.
The second, symptomatic, stage is immune mediated. The rash and arthralgia seen in this stage coincide with the appearance of virus-specific antibody, the disappearance of detectable B19 virus, and the formation of immune complexes.
Hosts with chronic hemolytic anemia (e.g., sickle cell anemia) who are infected with B19 are at risk for a life-threatening reticulocytopenia, which is referred to as an aplastic crisis. The reticulocytopenia results from the combination of (1) B19 depletion of the red blood cell precursors and (2) shortened life span of the erythrocytes caused by the underlying anemia.
Epidemiology
Approximately 65% of the adult population has been infected with B19 by 40 years of age (Box 53-4). Erythema infectiosum is most common in children and adolescents aged 4 to 15 years, who are a source of contagion. Arthralgia and arthritis are likely to occur in adults. Respiratory droplets and oral secretions most probably transmit the virus. Disease usually occurs in late winter and spring. Parenteral transmission of the virus by a blood-clotting–factor concentrate has also been described.
Bocavirus is found worldwide and causes disease in children younger than 2 years old. The virus is transmitted in respiratory secretions but can also be isolated from stool.
Clinical Syndromes (Clinical Case 53-1)
B19 virus, as stated earlier, is the cause of erythema infectiosum (fifth disease) (Box 53-5). Infection starts with an unremarkable prodromal period of 7 to 10 days, during which the person is contagious. Infection of a normal host may cause either no noticeable symptoms or fever and nonspecific symptoms, such as sore throat, chills, malaise, and myalgia, as well as a slight decrease in hemoglobin levels (Figure 53-4). This period is followed by a distinctive rash on the cheeks, which appear to have been slapped. The rash then usually spreads, especially to exposed skin such as the arms and legs (Figure 53-5), and then subsides over 1 to 2 weeks. Relapse of the rash is common.
Clinical Case 53-1
B19 Infection of a Transplant Recipient
Persistent, rather than transient, anemia occurs upon human parvovirus B19 infection of immunosuppressed individuals. One such case was reported by Pamidi and associates (Transplantation 69:2666–2669, 2000). After 1 year of immunosuppressive therapy (mycophenolate mofetil, prednisone, and tacrolimus) after a kidney transplant, a 46-year-old man complained of dyspnea, lightheadedness, and fatigue upon exercise. Laboratory tests confirmed a diagnosis of anemia. Bone marrow analysis indicated erythroid hyperplasia with a predominance of immature erythroblasts. Proerythroblasts could be found, with deep basophilic cytoplasm and intranuclear inclusions that immunohistologically stained for B19 antigen. The patient received 16 units of packed red blood cells over 6 weeks, with continued anemia. Serology indicated the presence of IgM (1 : 10) but insignificant IgG anti-B19 antibody. Treatment with intravenous IgG for 5 days resulted in a dramatic improvement. Immunosuppressive therapy of this patient prevented expansion and class switch to an IgG antibody response because of the lack of helper T cells. Resolution of the encapsidated parvovirus is dependent upon a robust antibody response, and in its absence, the normal transient anemia resulting from virus replication in erythroid precursors cannot be resolved.
Box 53-5
Clinical Consequences of Parvovirus (B19) Infection
Mild, flulike illness (fever, headache, chills, myalgia, malaise)
Erythema infectiosum (fifth disease)
Aplastic crisis in persons with chronic anemia
Arthropathy (polyarthritis: symptoms in many joints)
Risk of fetal loss as a result of B19 virus crossing the placenta, causing anemia-related disease but not congenital anomalies
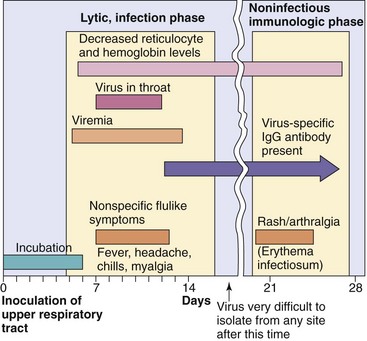
Figure 53-4 Time course of parvovirus (B19) infection. B19 causes biphasic disease: first, an initial lytic infection phase characterized by febrile, flulike symptoms and then a noninfectious immunologic phase characterized by a rash and arthralgia.
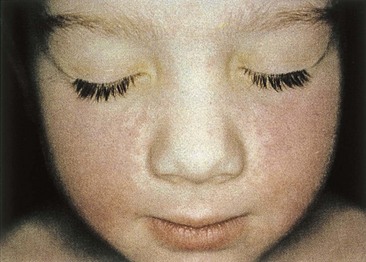
Figure 53-5 A “slapped-cheek” appearance is typical of the rash for erythema infectiosum.
(From Hart CA, Broadhead RL: A color atlas of pediatric infectious diseases, London, 1992, Wolfe.)
B19 infection in adults causes polyarthritis, (with or without a rash) that can last for weeks, months, or longer. Arthritis of the hands, wrists, knees, and ankles predominates. The rash may precede the arthritis but often does not occur. B19 infection of immunocompromised people may result in chronic disease.
The most serious complication of parvovirus infection is the aplastic crisis that occurs in patients with chronic hemolytic anemia (e.g., sickle cell anemia). Infection in these people causes a transient reduction in erythropoiesis in the bone marrow. The reduction results in a transient reticulocytopenia that lasts 7 to 10 days and a decrease in hemoglobin level. An aplastic crisis is accompanied by fever and nonspecific symptoms, such as malaise, myalgia, chills, and itching. A maculopapular rash with arthralgia and some joint swelling may also be present.
B19 infection of a seronegative mother increases the risk for fetal death. The virus can infect the fetus and kill erythrocyte precursors, causing anemia and congestive heart failure (hydrops fetalis). Infection of seropositive pregnant women often has no adverse effect on the fetus. There is no evidence that B19 causes congenital abnormalities (Box 53-6; see Box 53-5).
Box 53-6
Clinical Summary
A 10-year-old patient has a 5-day history of a flulike illness (headache, fever, muscle pain, feels tired) then develops an intensely red rash over the cheeks and a fainter “lacy” rash over the trunk and extremities.
Bocavirus may cause mild or severe acute respiratory disease. The more severe disease occurs in children younger than 2 years old who may have bronchiolitis with wheezing and with a viremia that extends long beyond the disease. A fatal case of bocavirus bronchiolitis has been reported.
Laboratory Diagnosis
The diagnosis of erythema infectiosum is usually based on the clinical presentation. For B19 disease to be definitively diagnosed, however, specific immunoglobulin M (IgM) or viral DNA must be detected (i.e., to distinguish the rash of B19 from that of rubella in a pregnant woman). Enzyme-linked immunosorbent assays for B19 IgM and IgG are available. The polymerase chain reaction test is a very sensitive method for detecting the B19 and bocavirus genomes in clinical samples. Virus isolation is not performed.
Treatment, Prevention, and Control
No specific antiviral treatment or means of control is available. Vaccines are available for dog and cat parvoviruses.
Mrs. Doe brought her daughter to the pediatrician with the complaint of a rash. The daughter’s face appeared as if it had been slapped, but she had no fever or other notable symptoms. On questioning, Mrs. Doe reported that her daughter had had a mild cold within the previous 2 weeks and that she herself was currently having more joint pain than usual and felt very tired.
1. What features of this history indicate a parvovirus B19 etiology?
2. Was the child infectious at presentation? If not, when was she contagious?
4. Were the symptoms of the mother and daughter related?
5. What underlying condition would put the daughter at increased risk for serious disease after B19 infection? The mother?
6. Why is quarantine a poor means of limiting the spread of B19 parvovirus?
1. The biphasic nature of the disease and the slapped-face rash are notable symptoms but are not unique to B19. B19 also causes arthralgia in adults because of immune complexes. A somewhat similar course of disease would occur with human herpesvirus 6 induction of exanthema subitum (roseola), although the time course may be different.
2. The child is infectious during the initial disease signs, which resemble a mild cold. The rash is immune mediated.
3. The initial nonspecific disease signs are caused by interferon and other innate responses to the infection. The rash is caused by immune responses, most likely associated with antibody and immune complexes.
4. The rash of the daughter and the arthralgia of the mother are due to the presence of antibody, formation of immune complexes, and type 2 and 3 hypersensitivity reactions.
5. Individuals with chronic hemolytic anemia (e.g., sickle cell anemia) are at risk for serious disease because B19 replicates in erythrocyte precursors and prevents the development of new erythrocytes or shortens their lifetime. Pregnant women are at risk for B19 infection, which causes hydrops fetalis and loss of the fetus.
6. Quarantine would not be effective because the virus is spread before the onset of the classic disease signs of erythema infectiosum (fifth disease).