59 Reoviruses
In January, a 6-month-old boy was seen in the emergency department after 2 days of persistent watery diarrhea and vomiting accompanied by a low-grade fever and mild cough. The infant appeared dehydrated and required hospitalization. The patient attended a day-care center.
1. In addition to rotavirus, what other viral agents must be considered in the differential diagnosis of this infant’s disease? What agents would need consideration if the patient were a teenager or an adult?
2. How would the diagnosis of rotavirus have been confirmed?
3. How was the virus transmitted? How long was the patient contagious?
Answers
1. Norovirus and adenovirus. These agents would also cause diarrhea in adults.
2. Rotavirus can be detected in stool by enzyme-linked immunosorbent assay. Reverse transcriptase polymerase chain reaction can also be used.
3. The virus is transmitted by the fecal-oral route. The patient is contagious for 2 to 5 days after the onset of diarrhea.
4. The baby is at high risk to dehydration due to its small size.
The Reoviridae consist of the orthoreoviruses, rotaviruses, orbiviruses, and coltiviruses (Table 59-1). The name reovirus was proposed in 1959 by Albert Sabin for a group of respiratory and enteric viruses that were not associated with any known disease (respiratory, enteric, orphan). The Reoviridae are nonenveloped viruses with double-layered protein capsids containing 10 to 12 segments of the double-stranded ribonucleic acid (dsRNA) genomes. These viruses are stable in detergents, over wide pH and temperature ranges, and in airborne aerosols. The orbiviruses and coltiviruses are spread by arthropods and are arboviruses.
Table 59-1 Reoviridae Responsible for Human Disease
| Virus | Disease |
|---|---|
| Orthoreovirus* | Mild upper respiratory tract illness, gastrointestinal tract illness, biliary atresia |
| Orbivirus/Coltivirus | Febrile illness with headache and myalgia (zoonosis) |
| Rotavirus | Gastrointestinal tract illness, respiratory tract illness (?) |
* Reovirus is the common name for the family Reoviridae and for the specific genus Orthoreovirus.
The orthoreoviruses, also referred to as mammalian reoviruses or simply reoviruses, were first isolated in the 1950s from the stools of children. They are the prototype of this virus family, and the molecular basis of their pathogenesis has been studied extensively. In general, these viruses cause asymptomatic infections in humans.
Rotaviruses cause human infantile gastroenteritis, a very common disease. In fact, rotaviruses account for approximately 50% of all cases of diarrhea in children requiring hospitalization because of dehydration (70,000 cases per year in the United States; 500,000 to 600,000 deaths per year worldwide). Rotaviruses are even more of a problem in underdeveloped countries, where before the development of vaccines they were responsible for at least 1 million deaths each year from uncontrolled viral diarrhea in undernourished children.
Structure
Rotaviruses and reoviruses share many structural, replicative, and pathogenic features. Reoviruses and rotaviruses have an icosahedral morphology with a double-layered capsid (60 to 80 nm in diameter) (Figure 59-1; Box 59-1) and a double-stranded segmented genome (“double:double”). The name rotavirus is derived from the Latin word rota, meaning “wheel,” which refers to the virion’s appearance in negative-stained electron micrographs (Figure 59-2). Proteolytic cleavage of the outer capsid (as occurs in the gastrointestinal tract) activates the virus for infection and produces an intermediate/infectious subviral particle (ISVP).

Figure 59-1 Computer reconstruction of cryoelectron micrographs of human reovirus type 1 (Lang). Top, left to right: Cross section of virion, intermediate/infectious subviral particle (ISVP), and core particle. The ISVP and core particles are generated by proteolysis of the virion and play important roles in the replication cycle. Center and bottom: Computer-generated images of the virions at different radii after the outer layers of features have been shaved off. The colors help one visualize the symmetry and molecular interactions within the capsid.
(Courtesy Tim Baker, Purdue University, West Lafayette, Ind.)
Box 59-1
Unique Features of Reoviridae
Double-layered capsid virion (60 to 80 nm) has icosahedral symmetry containing 10 to 12 (depending on the virus) unique double-stranded genomic segments (double:double virus).
Virion is resistant to environmental and gastrointestinal conditions (e.g., detergents, acidic pH, drying).
Rotavirus and orthoreovirus virions are activated by mild proteolysis to intermediate/infectious subviral particles, increasing their infectivity.
Inner capsid contains a complete transcription system, including RNA-dependent RNA polymerase and enzymes for 5′ capping and polyadenylate addition.
Viral replication occurs in the cytoplasm. Double-stranded RNA remains in the inner core.
Inner capsid aggregates around (+) RNA and transcribes (–) RNA in the cytoplasm.
Rotavirus-filled inner capsid buds into the endoplasmic reticulum, acquiring its outer capsid and a membrane, which is then lost.
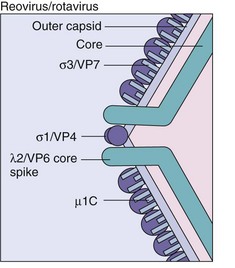
Figure 59-2 Structure of reovirus/rotavirus core and outer proteins. σ1/VP4, Viral attachment protein; σ3/VP7, major capsid component; λ2/VP6, major inner capsid protein; µ1C, minor outer capsid protein.
(Modified from Sharpe AH, Fields BN: Pathogenesis of viral infections. Basic concepts derived from the reovirus model, N Engl J Med 312:486–497, 1985.)
The outer capsid is composed of structural proteins (Figure 59-3), which surround a nucleocapsid core that includes enzymes for RNA synthesis and 10 (reo) or 11 (rota) different double-stranded RNA genomic segments. For rotavirus, the outer capsid has two layers, an intermediate layer consisting of the major capsid protein (VP6) and an outer layer that contains the viral attachment protein (VP4) and glycoprotein (VP7). As for the influenza virus, reassortment of gene segments can occur and thus create hybrid viruses.
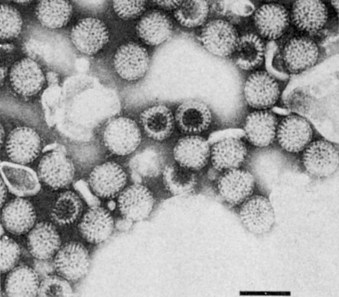
Figure 59-3 Electron micrograph of rotavirus. Bar = 100 nm.
(From Fields BN, et al: Virology, New York, 1985, Raven.)
Of interest, rotaviruses resemble enveloped viruses in that they (1) have glycoproteins (VP7, NSP4) that are on the outside of the virion, (2) acquire but then lose an envelope during assembly, and (3) appear to have a fusion protein activity that promotes direct penetration of the target cell membrane.
The genomic segments of rotaviruses and reoviruses encode structural and nonstructural proteins. The genomic segments of reovirus, the proteins they encode, and their functions are summarized in Table 59-2; those of rotavirus are summarized in Table 59-3. Core proteins include enzymatic activities required for the transcription of messenger RNA (mRNA). They include a 5′-methyl guanosine mRNA capping enzyme and an RNA polymerase. The σ1 protein (reo) and VP4 (rota) are located at the vertices of the capsid and extend from the surface like spike proteins. They have several functions, including viral attachment and hemagglutination, and they elicit neutralizing antibodies. VP4 is activated by protease cleavage into VP5 and VP8 proteins, exposing a structure similar to that of the fusion proteins of paramyxoviruses. Its cleavage facilitates productive entry of the virus into cells.
Table 59-2 Functions of Reovirus Gene Products
| Genomic Segments (Molecular Weight, Da) | Protein | Function (If Known) |
|---|---|---|
| Large Segments (2.8 × 106) | ||
| 1 | λ3 (inner capsid) | Polymerase |
| 2 | λ2 (outer capsid) | Capping enzyme |
| 3 | λ1 (inner capsid) | Transcriptase component |
| Medium Segments (1.4 × 106) | ||
| 1 | µ2 (inner capsid) | − |
| 2 | µ1C (outer capsid) | Cleaved from µ1, complexes with σ3, promotes entry |
| 3 | µNS | Promotes viral assembly* |
| Small Segments (0.7 × 106) | ||
| 1 | σ1 (outer capsid) | Viral attachment protein, hemagglutinin, determines tissue tropism† |
| 2 | σ2 (inner capsid) | Facilitates viral RNA synthesis |
| 3 | σNS | Facilitates viral RNA synthesis |
| 4 | σ3 (outer capsid) | Major component of outer capsid with µ1C |
* Proteins are not found in the virion.
† Target of neutralizing antibodies.
Modified from Field BN, et al: Virology, ed 3, New York, 1996, Lippincott-Raven.
Table 59-3 Functions of Rotavirus Gene Products
| Gene Segment | Protein (Location) | Function |
|---|---|---|
| 1 | VP1 (inner capsid) | Polymerase |
| 2 | VP2 (inner capsid) | Transcriptase component |
| 3 | VP3 (inner capsid) | mRNA capping |
| 4 | VP4 (outer capsid spike protein at vertices of virion) | Activation by protease to VP5 and VP8 in ISVP, hemagglutinin, viral attachment protein* |
| 5 | NSP1 (NS53) | RNA binding |
| 6 | VP6 (inner capsid) | Major structural protein of inner capsid, binding to NSP4 at ER to promote assembly of outer capsid |
| 7 | NSP3 (NS34) | RNA binding |
| 8 | NSP2 (NS35) | RNA binding, important for genome replication and packaging |
| 9 | VP7 (outer capsid) | Type-specific antigen, major outer capsid component that is glycosylated in ER and facilitates attachment and entry* |
| 10 | NSP4 (NS28) | Glycosylated protein in ER that promotes inner capsid binding to ER, transient envelopment, and addition of outer capsid; acts as enterotoxin to mobilize calcium and cause diarrhea |
| 11 | NSP5 (NS26) | RNA binding |
| 11 | NSP6 | Binds to NSP5 |
ER, Endoplasmic reticulum; ISVP, intermediate/infectious subviral particle; mRNA, messenger ribonucleic acid.
Replication
The replication of reoviruses and rotaviruses starts with ingestion of the virus (Figure 59-4). The virion outer capsid protects the inner nucleocapsid and core from the environment, especially the acidic environment of the gastrointestinal tract. The complete virion is then partially digested in the gastrointestinal tract and activated by protease cleavage and loss of the external capsid proteins (σ3/VP7) and cleavage of the σ1/VP4 protein to produce the ISVP. The σ1/VP4 protein at the vertices of the ISVP binds to sialic acid–containing glycoproteins on epithelial and other cells. Additional receptors include the β-adrenergic receptor for reovirus and integrin molecules for rotavirus. The σ1/VP4 of rotavirus also promotes the penetration of the virion into the cell. Whole virions of reovirus and rotavirus can also be taken up by receptor-mediated endocytosis.
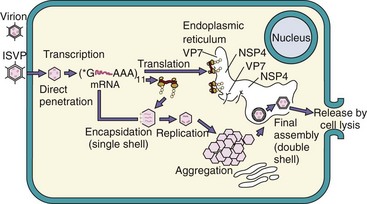
Figure 59-4 Replication of rotavirus. Rotavirus virions can be activated by protease (e.g., in the gastrointestinal tract) to produce an intermediate/infectious subviral particle (ISVP). The virion or ISVP binds, penetrates the cell, and loses its outer capsid. The inner capsid contains the enzymes for messenger ribonucleic acid (mRNA) transcription using the (±) strand as a template. Some mRNA segments are transcribed early; others are transcribed later. Enzymes in the virion cores attach 5′-methyl capped guanosine (*G) and 3′ polyadenylate sequence (poly A [AAA]) to mRNA. (+) RNA is mRNA and is also enclosed into inner capsids as a template to replicate the ± segmented genome. VP7 and NSP4 are synthesized as glycoproteins and expressed in the endoplasmic reticulum. The capsids aggregate and “dock” onto the NSP4 protein in the endoplasmic reticulum, acquiring VP7 and its outer capsid and an envelope. The virus loses the envelope and leaves the cell on cell lysis.
The ISVP releases the core into the cytoplasm, and the enzymes in the core initiate mRNA production. The dsRNA always remains in the core. Transcription of the genome occurs in two phases, early and late. In a manner similar to a negative-sense RNA virus, each of the negative-sense (−) RNA strands is used as a template by virion core enzymes, which synthesize individual mRNAs. Virus-encoded enzymes within the core add a 5′-methyl guanosine cap and a 3′-polyadenylate tail. The 5′-methyl guanosine cap was first discovered for reovirus mRNA and then shown to occur for celluar mRNA. The mRNA then leaves the core and is translated. Later, virion proteins and positive-sense (+) RNA segments associate together into corelike structures that aggregate into large cytoplasmic inclusions. The (+) RNA segments are copied to produce (−) RNAs in the new cores, replicating the double-stranded genome. The new cores either generate more (+) RNA or are assembled into virions.
The assembly processes for reovirus and rotavirus differ. In the assembly of reovirus, the outer capsid proteins associate with the core, and the virion leaves the cell upon cell lysis. Assembly of rotavirus resembles that of an enveloped virus, in that the rotavirus cores associate with the NSP4 viral protein on the outside of the endoplasmic reticulum (ER); on budding into the ER, they acquire its VP7 outer capsid glycoprotein. The membrane is lost in the ER, and the virus leaves the cell during cell lysis. Reovirus inhibits cellular macromolecular synthesis within 8 hours of infection.
Orthoreoviruses (Mammalian Reoviruses)
The orthoreoviruses are ubiquitous. The virions are very stable and have been detected in sewage and river water. The mammalian reoviruses occur in three serotypes, referred to as reovirus types 1, 2, and 3; these serotypes are based on neutralization and hemagglutination-inhibition tests. All three serotypes share a common complement-fixing antigen.
Pathogenesis and Immunity
Orthoreoviruses do not cause significant disease in humans. However, studies of reovirus disease in mice have advanced our understanding of the pathogenesis of viral infections in humans. Depending on the reovirus strain, the virus can be neurotropic or viscerotropic in mice. The functions and virulence properties of the reovirus proteins were identified through comparison of the activities of interstrain hybrid viruses that differ in only one genomic segment (encoding one protein). With this approach, the new activity is attributable to the genomic segment from the other virus strain.
After ingestion and proteolytic production of the ISVP, the orthoreoviruses bind to M cells in the small intestine, which then transfer the virus to the lymphoid tissue of Peyer patches lining the intestines. The viruses then replicate and initiate a viremia. Although the virus is cytolytic in vitro, it causes few if any symptoms before entering the circulation and producing infection at a distant site. In the mouse model, the viral attachment protein (δ1) facilitates viral spread to the mesenteric lymph nodes and determines whether the virus is neurotropic.
Mice, and presumably humans, mount protective humoral and cellular immune responses to outer capsid proteins. Although orthoreoviruses are normally lytic, they can also establish persistent infection in cell culture.
Epidemiology
As already mentioned, the orthoreoviruses have been found worldwide. Most people are probably infected during childhood because approximately 75% of adults have antiviral antibody. Most animals, including chimpanzees and monkeys, are infected with reoviruses that are serologically related to human reovirus. It is not known whether animals are a reservoir for human infections.
Clinical Syndromes
Orthoreoviruses infect people of all ages, but linking specific diseases to these agents has been difficult. Most infections are thought to be asymptomatic or are so mild they go undetected. Thus far, these viruses have been linked to common coldlike, mild, upper respiratory tract illness (low-grade fever, rhinorrhea, and pharyngitis), gastrointestinal tract disease, and biliary atresia.
Laboratory Diagnosis
Human orthoreovirus infection can be detected through assay of the viral antigen or RNA in clinical material, virus isolation, or serologic assays for virus-specific antibody. Throat, nasopharyngeal, and stool specimens from patients with suspected upper respiratory tract or diarrheal disease are used as samples. Human orthoreoviruses can be isolated using mouse L-cell fibroblasts, primary monkey kidney cells, and HeLa cells. Serologic assays can be performed for epidemiologic purposes.
Rotaviruses
Rotaviruses are common agents of infantile diarrhea worldwide. The rotaviruses are a large group of gastroenteritis-causing viruses infecting many different mammals and birds.
Rotavirus virions are relatively stable to environmental abuse, including treatment with detergents, pH extremes of 3.5 to 10, and even repeated freezing and thawing. Within the intestine, proteolytic enzymes, such as trypsin, enhance infectivity.
Human and animal rotaviruses are divided into serotypes, groups, and subgroups. Serotypes are distinguished primarily by the VP7 (glycoprotein, G) and VP4 (protease-sensitive protein, P) outer capsid proteins. Groups are determined primarily on the basis of the antigenicity of VP6 and the electrophoretic mobility of the genomic segments. Seven groups (A to G) of human and animal rotaviruses have been identified on the basis of the VP6 inner capsid protein. Human disease is caused by group A rotavirus and occasionally group B and C rotaviruses.
Pathogenesis and Immunity
The rotavirus can survive the acidic environment in a buffered stomach or in a stomach after a meal and is converted to the ISVP by proteases (Box 59-2). Viral replication occurs after adsorption of the ISVP to columnar epithelial cells covering the villi of the small intestine. Approximately 8 hours after infection, cytoplasmic inclusions that contain newly synthesized proteins and RNA are seen. As many as 1010 viral particles per gram of stool may be released during disease. Studies of the small intestine, either of experimentally infected animals or in biopsy specimens from infants, show shortening and blunting of the microvilli and mononuclear cell infiltration into the lamina propria.
Box 59-2
Disease Mechanisms of Rotavirus
Virus is spread by the fecal-oral route and possibly the respiratory route.
Cytolytic and toxin-like action on the intestinal epithelium causes loss of electrolytes and prevents reabsorption of water.
Disease can be significant in infants younger than 24 months, but it is asymptomatic in adults.
Large amounts of virus are released during the diarrheal phase.
Similar to cholera, rotavirus infection prevents the absorption of water, causing a net secretion of water and loss of ions, which together result in a watery diarrhea. The NSP4 protein of rotavirus acts in a toxin-like manner to promote calcium ion influx into enterocytes, release of neuronal activators, and a neuronal alteration in water absorption. The loss of fluids and electrolytes can lead to severe dehydration and even death if therapy does not include electrolyte replacement. Of interest, the diarrhea also promotes spread and transmission of the virus.
Immunity to infection requires the presence of antibody, primarily immunoglobulin A (IgA), in the lumen of the gut. Antibodies to the VP7 and VP4 neutralize the virus. Actively or passively acquired antibody (including antibody in colostrum and mothers’ milk) can lessen the severity of disease but does not consistently prevent reinfection. In the absence of antibody, the inoculation of even small amounts of virus causes infection and diarrhea. Infection in infants and small children is generally symptomatic, whereas in adults, it is usually asymptomatic.
Epidemiology
Rotaviruses are ubiquitous worldwide, with 95% of children infected by 3 to 5 years of age (Box 59-3). Rotaviruses are passed from person to person by the fecal-oral route. Maximal shedding of the virus occurs 2 to 5 days after the start of diarrhea but can occur without symptoms. The virus survives well on fomites (e.g., furniture and toys) and on hands because it can withstand drying. Although domestic animals (e.g., cows) are known to harbor serologically related rotaviruses, they are not a common source of human infection. Outbreaks occur in preschools and day-care centers and among hospitalized infants.
Box 59-3
Epidemiology of Rotavirus
Rotaviruses are one of the most common causes of serious diarrhea in young children worldwide, affecting more than 18 million infants and children and accounting for close to 1600 deaths per day resulting from dehydration. In North America, outbreaks occur during the autumn, winter, and spring. More severe disease occurs in severely malnourished children. Rotavirus diarrhea is a very contagious, severe, life-threatening disease for infants in developing countries, and it occurs year-round. Several outbreaks of group B rotavirus have occurred in China because of contaminated water supplies that affected millions of people.
Clinical Syndromes (Clinical Case 59-1; Box 59-4)
Rotavirus is a major cause of gastroenteritis. The incubation period for rotavirus diarrheal illness is estimated to be 48 hours. The major clinical findings in hospitalized patients are vomiting, diarrhea, fever, and dehydration. Neither fecal leukocytes nor blood occurs in stool for this form of diarrhea. Rotavirus gastroenteritis is a self-limited disease, and recovery is generally complete and without sequelae. However, the infection may prove fatal in infants who live in developing countries and who are malnourished and dehydrated before the infection.
Clinical Case 59-1
Rotavirus Infection of Adults
Mikami and associates (J Med Virol 73:460–464, 2004) described an outbreak of acute gastroenteritis that occurred over a 5-day period in 45 of 107 children (aged 11 to 12 years) after a 3-day school trip. The source person for the outbreak was ill at the start of the trip. A case of rotavirus acute gastroenteritis is defined as three or more episodes of diarrhea and/or two or more episodes of vomiting per day. Other symptoms included fever, nausea, fatigue, abdominal pain, and headache. The rotavirus responsible for the outbreak was identified from stool of several individuals as serotype G2 group A rotavirus by comparison of the genomic ribonucleic acid migration pattern by electrophoresis, by reverse transcriptase polymerase chain reaction, and by enzyme-linked immunosorbent assay of virus obtained from stool samples. Although rotavirus is the most common cause of infantile diarrhea, this virus, especially the G2 strain, also causes gastroenteritis in adults. This article illustrated the different laboratory methods available for detection of a virus that is difficult to grow in tissue culture.
Laboratory Diagnosis
The clinical findings in patients with rotavirus infection resemble those of other viral diarrheas (e.g., Norwalk virus). Most patients have large quantities of virus in stool, making the direct detection of viral antigen the method of choice for diagnosis. Enzyme immunoassay and latex agglutination are quick, easy, and relatively inexpensive ways to detect rotavirus in stool. Viral particles in specimens can also be readily detected on electron microscopy or by immunoelectron microscopy. Reverse transcriptase polymerase chain reaction is useful to detect and distinguish the genotypes of rotavirus.
Cell culture of rotavirus requires pretreatment of the virus with trypsin to generate the ISVP for infection to occur but is not used for diagnostic purposes. Serologic studies are primarily used for research and epidemiologic purposes. Because so many people have rotavirus-specific antibody, a fourfold rise in antibody titer is necessary for the diagnosis of recent infection or active disease.
Treatment, Prevention, and Control
Rotaviruses are acquired very early in life. Their ubiquitous nature makes it difficult to limit the spread of the virus and infection. Hospitalized patients with disease must be isolated to limit spread of the infection to other susceptible patients.
No specific antiviral therapy is available for a rotavirus infection. The morbidity and mortality associated with rotavirus diarrhea result from dehydration and electrolyte imbalance. The purpose of supportive therapy is to replace fluids so that blood volume and electrolyte and acid-base imbalances are corrected.
Development of a safe rotavirus vaccine was a high priority for protecting children, especially those in underdeveloped countries, from potentially fatal disease. Animal rotaviruses, such as the rhesus monkey rotavirus and the Nebraska calf diarrhea virus, share antigenic determinants with human rotaviruses and do not cause disease in humans. A human–rhesus monkey reassortant vaccine (Rotashield) was recalled in 1999 because of the incidence of intussusception (misfolding of the bowel possibly resulting from inflammatory reactions to the vaccine) in a small number of infants. Two new safer rotavirus vaccines have since been developed and are U.S. Food and Drug Administration–approved in the United States and elsewhere. RotaTeq consists of five reassortant bovine rotaviruses containing the VP4 or VP7 of five different human rotaviruses. The RotaRix vaccine is a single-strain attenuated human rotavirus. The vaccines are administered as young as possible, at 2, 4, and 6 months of age.
Coltiviruses and Orbiviruses
The coltiviruses and orbiviruses infect vertebrates and invertebrates. The coltiviruses cause Colorado tick fever and related human disease. The orbiviruses mainly cause disease in animals, including blue tongue disease of sheep, African horse sickness, and epizootic hemorrhagic disease of deer.
Colorado tick fever, an acute disease characterized by fever, headache, and severe myalgia, was originally described in the 19th century and is now believed to be one of the most common tick-borne viral diseases in the United States. Although hundreds of infections occur annually, the exact number is not known, because Colorado tick fever is not a reportable disease.
The structure and physiology of the coltiviruses and orbiviruses are similar to those of the other Reoviridae, with the following major exceptions:
1. The outer capsid of the orbiviruses has no discernible capsomeric structure, even though the inner capsid is icosahedral.
2. The virus causes viremia, infects erythrocyte precursors, and remains in the mature red blood cells, protected from the immune response.
3. The orbivirus life cycle includes vertebrates and invertebrates (insects).
Colorado tick fever viruses have 12 double-stranded RNA genomic segments, and orbiviruses have 10.
Pathogenesis
Colorado tick fever virus infects erythroid precursor cells without severely damaging them. The virus remains within the cells, even after they mature into red blood cells; this factor protects the virus from clearance. The resulting viremia can persist for weeks or months, even after cessation of symptoms. Both of these factors promote transmission of the virus to the tick vector.
Serious hemorrhagic disease can result from the infection of vascular endothelial and vascular smooth muscle cells and pericytes, thereby weakening capillary structure. The weakness leads to leakage, hemorrhage, and potentially hypotension and shock. Neuronal infection can lead to meningitis and encephalitis.
Epidemiology
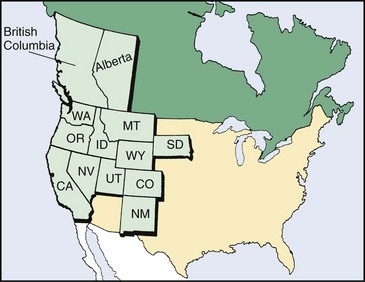
Colorado tick fever occurs in western and northwestern areas of the United States and western Canada, where the wood tick Dermacentor andersoni is distributed (elevations of 4000 to 10,000 feet) (Figure 59-5). Ticks acquire the virus by feeding on a viremic host and subsequently transmit the virus in saliva when feeding on a new host. Natural hosts of this virus constitute many mammals, including squirrels, chipmunks, rabbits, and deer. Human disease is observed during the spring, summer, and autumn, seasons when humans are more likely to invade the habitat of the tick.
Clinical Syndromes
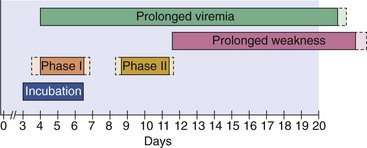
Colorado tick fever virus generally causes mild or subclinical infection. The symptoms of the acute disease resemble those of dengue fever. After a 3- to 6-day incubation period, symptomatic infections start with the sudden onset of fever, chills, headache, photophobia, myalgia, arthralgia, and lethargy (Figure 59-6). Characteristics of the infection include a biphasic fever, conjunctivitis, and possibly lymphadenopathy, hepatosplenomegaly, and a maculopapular or petechial rash. A leukopenia involving both neutrophils and lymphocytes is an important hallmark of the disease. Children occasionally have a more severe hemorrhagic disease. Colorado tick fever must be differentiated from Rocky Mountain spotted fever, a tick-borne rickettsial infection characterized by a rash, because the latter disease may require antibiotic treatment.
Laboratory Diagnosis
A diagnosis of Colorado tick fever can be established through the direct detection of viral antigens, virus isolation, or serologic tests. The best, most rapid method is detection of viral antigen on the surfaces of erythrocytes in a blood smear through the use of immunofluorescence. Laboratory tests may be available through state public health departments or the Centers for Disease Control and Prevention.
The titers of antibody in acute and convalescent specimens must be compared for a serologically based diagnosis to be rendered because subclinical infections can occur and antibody may persist for a lifetime. Specific IgM is present for approximately 45 days after the onset of illness, and its detection is also presumptive evidence of an acute or very recent infection. Immunofluorescence is the best technique, but complement fixation, neutralization, and enzyme immunoassay are also used to detect Colorado tick fever antibody.
Treatment, Prevention, and Control
No specific treatment is available for Colorado tick fever. The disease is generally self-limited, indicating that supportive care is sufficient. The viremia is long lasting, implying that infected patients should not donate blood soon after recovery. Prevention consists of (1) avoiding tick-infested areas, (2) using protective clothing and tick repellents, and (3) removing ticks before they bite. Unlike tick-borne rickettsial disease, in which prolonged feeding is required for the bacteria to be transmitted, the coltivirus from the tick’s saliva can enter the bloodstream rapidly. A formalinized Colorado tick fever vaccine has been developed and evaluated, but because of the mildness of the disease, its distribution to the general public is not warranted.
A 10-month-old Pakistani infant has watery diarrhea, vomiting, and fever for 4 days. The baby becomes very dehydrated and dies.
1. Commercially available enzyme-linked immunosorbent assays detect rotavirus in stool.
2. The NSP4 protein of rotavirus has a toxin-like (e.g., cholera) activity to promote secretory diarrhea.
3. Treatment is fluid replacement.
4. There are two commercially available vaccines administered during the first year of life.
5. Dehydration occurs very rapidly in babies because of their small size and the rapid fluid loss. Lack of access to a hospital and ability to rapidly rehydrate put this baby at greater risk.