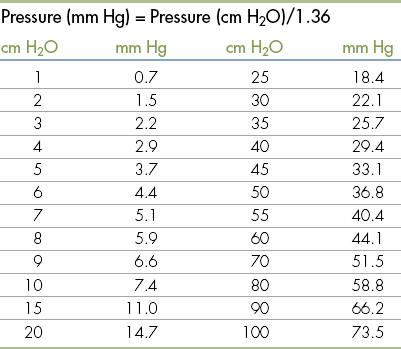
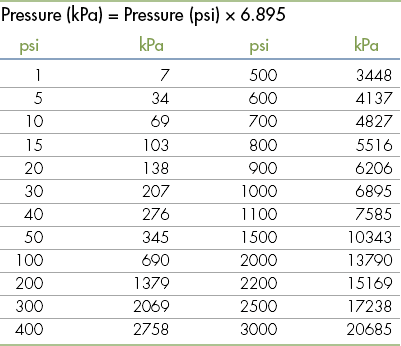
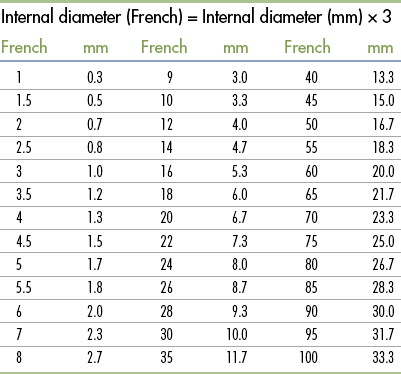
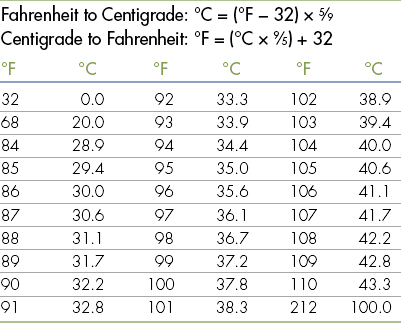
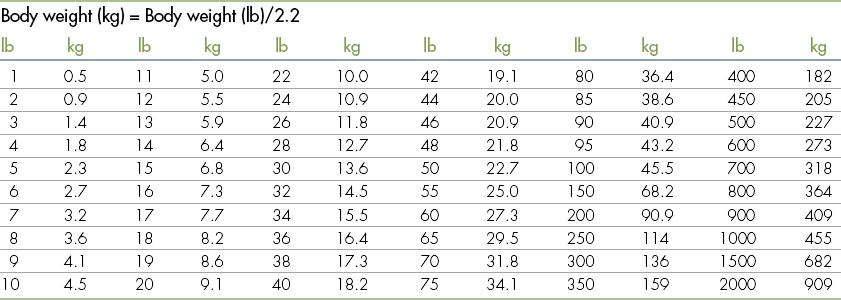
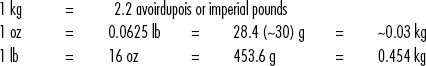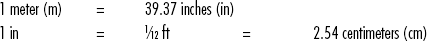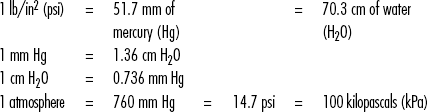Standard Volumes, Weights, Measures, and Equivalents
METRIC SYSTEM
| “kilo” | means | One thousand |
| “milli” | means | One thousandth |
| “micro” | means | One thousandth of one thousandth, or one millionth |
Weight to Volume Equivalents
1 gram (g) = the weight of 1 cc (cubic centimeter) of water at 4 degrees centigrade
Volume Equivalents
| 1 cc | = | 1 milliliter (mL) |
| 1000 mL or cc | = | 1 liter (L) |
| 1 mL or cc | = | 0.001 L |
| 1 deciliter (dL) | = | 100 mL |
Weight Equivalents
| 1000 microgram (mcg) | = | 1 milligram (mg) |
| 1 mcg | = | 0.001 mg |
| 1000 mg | = | 1 gram (g) |
| 1 mg | = | 0.001 g |
| 1 million mcg | = | 1 g |
| 1000 g | = | 1 kilogram (kg) |
| 1 g | = | 0.001 kg |
Solution Equivalents
| 1 part in 10 (1:10) | = | 10% (1 mL contains 100 mg) |
| 1 part in 100 (1:100) | = | 1% (1 mL contains 10 mg) |
| 1 part in 500 (1:500) | = | 0.20% (1 mL contains 2 mg) |
| 1 part in 1000 (1:1000) | = | 0.10% (1 mL contains 1 mg) |
| 1 part in 5000 (1:5000) | = | 0.02% (1 mL contains 0.2 mg) |
| 1 part in 10,000 (1:10,000) | = | 0.01% (1 mL contains 0.1 mg) |
The number of milligrams in 1 mL of any solution of known percentage strength is obtained by moving the decimal one place to the right. For example, a 1% solution contains 10 mg/mL. By definition, a percent solution contains the specified weight (in grams) of the solute in 100 mL of total solution. For example, a 5% dextrose and water solution contains 5 g of dextrose dissolved in each 100 mL of water.