Opioid (Narcotic) Analgesics and Antagonists
1 Explain the classification, mechanism of action, and pharmacokinetics of opioids.
2 List and describe the pharmacologic effects and potential adverse reactions of opioids.
3 Discuss the addiction potential of opioids, including treatment.
4 Name and explain the analgesic actions of the most common opioid agonists.
5 Discuss the actions of and provide examples of the mixed opioids.
The opioid analgesics are often used to manage dental pain in patients in whom nonsteroidal antiinflammatory drugs (NSAIDs) are contraindicated. The dental hygienist and the dentist should be aware of the opioid groups, side effects, relative potency, and proper place in the management of dental pain.
HISTORY
Opium is the dried juice from the unripe seed capsules of the opium poppy. As early as 4000 bc, many cultures had recognized the euphoric effect of the poppy plant. In the early 1800s, morphine and codeine were isolated from opium. Until about 1920, patent medicines (medicines whose efficacy and safety were questionable) containing opium were promoted for numerous uses. When these agents, used orally, became unlawful, narcotic (opioid) abuse by injection began and has continued until the present.
TERMINOLOGY
The terms used to refer to this drug group have changed over the years. Narcotics, the original name for this group of drugs, is derived from the Greek word that means “stupor.” At first, the term narcotics was used to refer to drugs that are derivatives of opium poppy. Drugs in different pharmacologic classes with central nervous system (CNS) depressant effects also began to be lumped into the narcotic group because they caused stupor. This designation then became confusing because the drugs in it had different properties. Opiates was the next term that was used. It refers to drugs that are derived from the substances in the opioid poppy. Other chemical agents that produced opiate effects but did not have a structure like the opiates were synthesized but were not opiate-like. To be more inclusive, the term opioids was then used to include not only the former opiates but also other structurally different agents, their antagonists, and the receptors stimulated by the opioids. The old term narcotic is still used in older publications or by older practitioners.
CLASSIFICATION
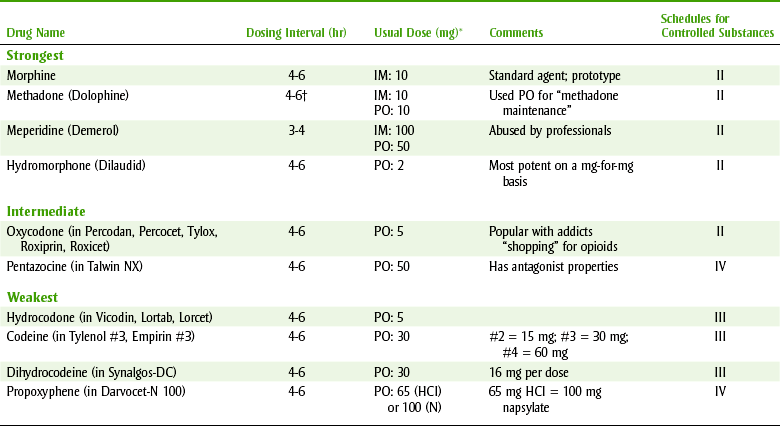
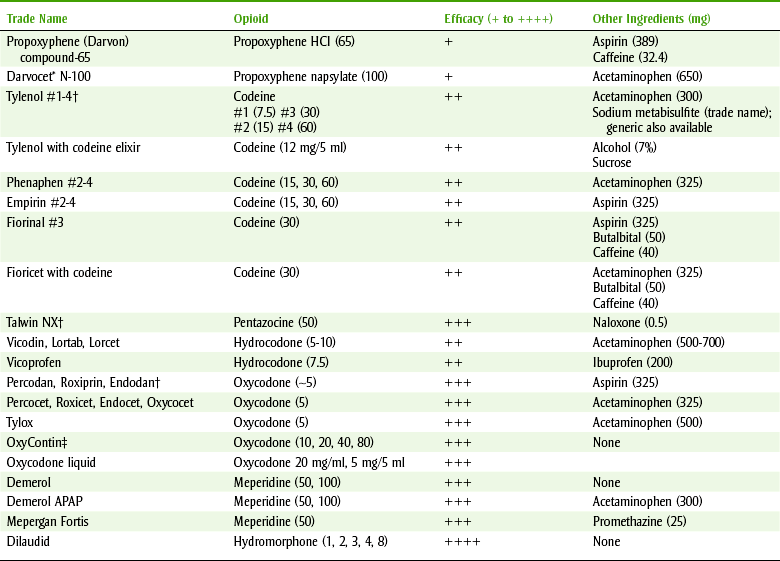
The clinically useful opioids may be divided in several different ways. One way to divide these agents is by their mechanism of action at the receptor sites: agonists, mixed opioids, and antagonists. Table 6-1 shows the classifications.
TABLE 6-1
CLASSIFICATION OF THE OPIOIDS BY RECEPTOR ACTION
| Group | Subgroup | Example |
| Opioid agonists | Morphine, codeine | |
| Mixed opioids | Agonist antagonists | Pentazocine |
| Partial agonists | Buprenorphine | |
| Antagonists | Naloxone |
The opioids may also be classified by their chemical structure (Box 6-1). Structural classification is useful when the patient has a history of an allergy. Agents with the most similar chemical structure are more likely to be cross-allergenic; conversely, those with very different structures are much less likely to exhibit cross-allergenicity. The chemical structure groups include morphine/codeine, methadone, morphinan, meperidine, and others. The largest group is the morphine/codeine group, which includes codeine. A patient with a true allergy to codeine should not be given an analgesic in that group.
Opioids may be classified by their efficacy (Table 6-2). Efficacy classification assists in selection of the proper opioid based on the amount of pain relief needed. The amount of pain experienced is usually related to the individual patient and his or her reaction to the dental procedure and the specific dental procedure being performed. Although “bigger” procedures may elicit more pain, the characteristics of the patient play a more important role.
MECHANISM OF ACTION
The opioids bind to receptors located in both the CNS and the spinal cord, producing an altered perception of reaction to pain. Receptors that mediate specific pharmacologic effects and adverse reactions are stimulated to varying degrees by individual opioids.
The discovery of three groups of endogenous substances with opioid-like action, the enkephalins, endorphins, and dynorphins, has helped explain the presence of these receptors. These naturally occurring peptides possess analgesic action and have addiction potential. They probably function as neurotransmitters, but their exact function has not been elucidated. They may be involved in the analgesic action of a placebo and the enhancement of well-being that occurs with running (an increase in β-endorphins).
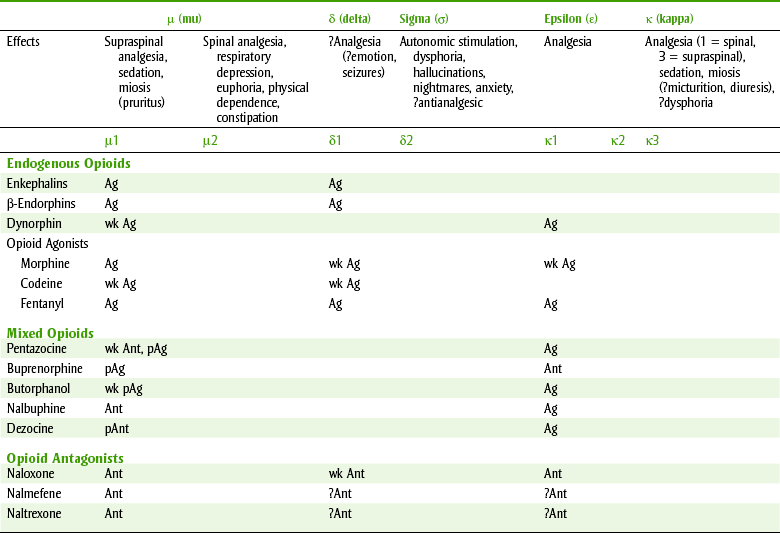
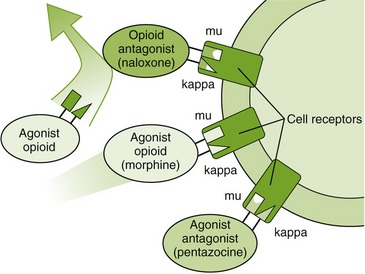
Table 6-3 describes the pharmacologic effects of selected opioid receptors and the effect of some opioids on these receptors. Opioids may be complete agonists, partial agonists, agonist-antagonists, or antagonists. The three opioid receptors that have been characterized in more detail and that are stimulated by the opioids are the mu (µ), kappa (κ), and delta (δ) receptors. Differences in affinity for and action of different opioids in tolerance to pain might even be the result of variations in the endogenous levels of the neurotransmitters. Differences in affinity for and action of different opioids at these and other specific receptors explain some of the distinctions among the different opioids’ adverse reactions. For example, stimulation of µ-receptors produce analgesia. The k-receptor is responsible for dysphoria. Pentazocine, a κ-receptor agonist, produces dysphoria; morphine has no effect on the κ-receptor and produces less dysphoria than pentazocine. Naloxone is an antagonist at the three receptor sites (Figure 6-1). More opioid receptors are sure to be identified and characterized. As more subreceptor types are elucidated, it will be possible to further separate beneficial (analgesic) effects of the opioids from their side effects (e.g., respiratory depression, constipation, or drug dependence).
PHARMACOKINETICS
ADME, which is an acronym of the first letters of each component of drug handling, refers to absorption, distribution, metabolism, and excretion:
• Absorption: Most opioid analgesic agents are absorbed well when taken orally; absorption occurs from the lungs and from the nasal and oral mucosa. Absorption occurs through the mucous membranes of the nose and the intact skin. A nasal spray for one opioid, butorphanol (Stadol NS), is available. Absorption through the skin is used to advantage with transdermal patches of fentanyl (Duragesic).
• Distribution: After absorption, the opioids undergo variable first-pass metabolism in the liver or intestinal cell wall, which reduces their bioavailability. The oral-to-parenteral ratio determines the difference in bioavailability between an opioid administered orally and one given parenterally. For example, the ratio is 0.2 to 0.3 for morphine, 0.25 to 0.7 for meperidine, and 0.4 to 0.7 for codeine. Therefore about two-thirds of codeine administered orally reaches the systemic circulation, whereas only about one-fourth of morphine does. The opioids are bound to plasma proteins to varying degrees (morphine 35%, meperidine 60%). The opioids are also distributed to the fetus in pregnant women, accounting for the respiratory depression produced in the fetus when the mother is given opioids near term.
• Metabolism: The major route of metabolism for the opioids is conjugation with glucuronic acid in the liver. Given orally, most opioids have a similar duration of action for analgesia—4 to 6 hours.
• Excretion: Metabolized opioids are excreted by glomerular filtration as their metabolites. The metabolites and the unchanged drug are excreted in the urine.
The dosing interval and usual dose of some opioids are listed in Table 6-2. In general, their onset is within 1 hour and their duration necessitates dosing every 4 to 6 hours.
PHARMACOLOGIC EFFECTS
Although the pharmacologic effects and adverse reactions of the opioids are closely related, they are discussed separately. A pharmacologic effect may also be an adverse reaction, depending on the clinical use of the agent. In general, the severity of the side effects is proportional to the agent’s efficacy (strength).
Analgesia
The opioid analgesics provide varying degrees of analgesia, depending on the strength of the agent. Figure 6-2 shows the relative analgesic efficacy of selected opioids. Morphine is the opioid agonist by which other opioids are measured. The strongest opioids can reduce even the most severe pain; the weaker agents mixed with nonopioids are equivalent to the NSAIDs in their ability to relieve pain; and the analgesic potency of the weakest agent (codeine) is low (see Table 6-2).
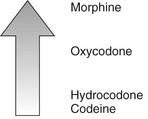
FIGURE 6-2 Comparing strengths of opioids. Opioids vary in efficacy (maximal effect attained) from codeine (low) to morphine (high).
Codeine raises the pain threshold and affects the cerebral cortex to depress the reaction to pain. Both m-receptors and k-receptors are involved in producing analgesia. The opioids alter the patient’s reaction to painful stimuli, possibly by altering the release of certain central neurotransmitters.
Sedation and Euphoria
In the usual therapeutic doses, the opioid analgesics generally produce sedation by k-receptor stimulation. This may potentiate their analgesic effect and relieve anxiety. This effect is additive with other CNS depressants such as alcohol. With larger doses, or if the pain is suddenly removed, euphoria can result. CNS excitation rarely occurs.
Cough Suppression
The opioids exert their antitussive action by depressing the cough center, located in the medulla. The dose that produces the antitussive effect is much lower than that required for analgesia, so the least potent agents are effective (e.g., codeine). Related compounds, such as dextromethorphan, are often used as antitussives.
Gastrointestinal Effects
The opioids increase the smooth muscle tone of the intestinal tract and markedly decrease its propulsive contractions and motility. This effect has made opioids useful in the symptomatic treatment of diarrhea. Opioid-like agents without analgesic properties, such as diphenoxylate (in Lomotil), are used to treat diarrhea.
ADVERSE REACTIONS
Unlike many other drugs, the adverse reactions of the opioids are not related to a direct damaging effect on hepatic, renal, or hematologic tissues but instead are an extension of their pharmacologic effects. Like the pharmacologic effects, the adverse reactions of the opioid analgesics are proportional to their analgesic strength. Table 6-4 lists contraindications and cautions for the use of opioids.
TABLE 6-4
CONTRAINDICATIONS AND CAUTIONS TO THE USE OF OPIOIDS
| Condition | Comment |
| Alcoholic or addict | Greater potential for abuse |
| Head injury | Can increase intracranial pressure |
| Chronic pain | Addiction potential limits (e.g., TMD) duration |
| Respiratory disease | Respiratory depression can occur |
| Pregnancy | Respiratory depression near term (fetus) |
| Nursing | No problem: watch infant |
| Nausea | Additive nausea |
| Constipation | Exacerbates or produces constipation |
Respiratory Depression
The opioid analgesic agonists depress the respiratory center in a dose-related manner. This is usually the cause of death with an overdose. The depression is related to a decrease in the sensitivity of the brainstem to carbon dioxide. Both the rate and depth of breathing are reduced. In elderly or debilitated patients, the usual therapeutic dose of morphine can produce a significant decrease in pulmonary ventilation. Reduced ventilation produces vasodilation, which results in an increase in intracranial pressure. Opioids should not be used in patients with head injuries. Opioids may also mask CNS diagnostic symptoms. Patients with hyperthyroidism are more tolerant of the depression, whereas patients with hypothyroidism are more sensitive.
Nausea and Emesis
Analgesic doses of opioid analgesics often produce nausea and vomiting. This is the result of their direct stimulation of the chemoreceptor trigger zone (CTZ), located in the medulla. This side effect is reduced if the patient does not ambulate. Administration of repeated, regular doses of an opioid can prevent vomiting by depressing the vomiting center (VC), another area in the CNS distinct from the CTZ.
Constipation
The opioids produce constipation by causing a tonic contraction of the gastrointestinal tract. Small doses of even weak opioids often have this effect, and their duration outlasts their analgesic effect. Even with continued administration, tolerance does not develop to this effect.
Miosis
The opioid analgesics cause miosis, an important sign (pinpoint pupils) in diagnosing an opioid overdose or identifying an addict. Tolerance does not develop to this effect.
Urinary Retention
The opioids increase the smooth muscle tone in the urinary tract, thereby causing urinary retention. They also produce an antidiuretic effect by stimulating the release of antidiuretic hormone (ADH) from the pituitary gland. This reaction may pose a problem in patients with prostatic hypertrophy.
Central Nervous System Effects
Occasionally, opioids may produce CNS stimulation, exhibited by anxiety, restlessness, or nervousness. Dysphoria can also occur from the opioids.
Cardiovascular Effects
The opioids may depress the vasomotor center and stimulate the vagus nerve. With high doses, postural hypotension, bradycardia, and even syncope may result.
Biliary Tract Constriction
In high doses, the opioids may constrict the biliary duct, resulting in biliary colic (pain associated with gallstones). This effect is important in patients passing gallstones who are being treated with opioids.
Histamine Release
Because the opioids can stimulate the release of histamine, itching and urticaria can result from their administration. This effect can occur at the site of intramuscular injection or at remote sites (e.g., itchy nose).
Pregnancy and Nursing Considerations
Opioids have not been shown to be teratogenic, although they may prolong labor or depress fetal respiration if given near term. Infants born to mothers using high-dose opioids, such as an addict, can have marked depressed respiration and experience withdrawal symptoms. The amount of opioid excreted in the mother’s milk when therapeutic doses are given to the mother would pose no problem to the normal infant. Morphine and codeine are classified as Food and Drug Administration (FDA) pregnancy category C. Acetaminophen is a pregnancy category B drug. Caution is urged because acetaminophen is often combined with opioid analgesics.
Addiction
The degree of addiction potential of opioids is proportional to their analgesic strength. This fact limits the usefulness of the strongest of these agents. Because the duration of use in dentistry is usually short, addiction does not often pose a problem for the dentist. NSAIDs should be used to control dental pain in the addict. An addict will develop tolerance to the effects of the opioids, except for miosis and constipation. The rate of development of tolerance is related to the strength of the opioid and its frequency of use.
 OVERDOSE
OVERDOSE
The major symptom of opioid overdose is respiratory depression. In addition to pinpoint pupils and coma, this symptom is characteristic of opioid overdose. Opioid overdose is treated with an antagonist, naloxone, discussed later in this chapter.
 WITHDRAWAL
WITHDRAWAL
After abruptly discontinuing the opioids, a withdrawal syndrome occurs. The symptoms include yawning, lacrimation, perspiration, rhinorrhea, gooseflesh (“cold turkey”), irritability, nausea, vomiting, tachycardia, tremors, and chills. The name cold turkey comes from the symptom of piloerection (like when a person is cold); this reaction reminded addicts of the way a turkey looks (little bumps).
 IDENTIFICATION OF AN ADDICT
IDENTIFICATION OF AN ADDICT
“Shoppers” are addicts who try to find a physician or dentist who will prescribe their drug of choice. There have even been organized groups of shoppers headed by an individual. The members of the groups are directed to physicians and dentists with complaints whose symptoms are taught to them. Prescriptions for controlled substances that are given to these “patients” are returned to the leader, and the “patients” are paid for their time. New dentist offices are often targets for “shopping.” If a prescription for a controlled substance is obtained, more addicts will be contacting the office. This is not the type of “practice builder” that any dental office needs. Dental practitioners should become suspicious if any of the following shopper symptoms are present in a patient:
• Requests a certain drug and says it is better; he or she may stumble over the name
• Claims many allergies and says lots of pain medications do not work
• Cancels dental appointments because he or she claims to be going out of town on business
• Experiences pain for days after scaling and root planing
• Moves from dental office to dental office because “others do not understand”
• Claims a “low pain threshold”
• Needs refills several days after a dental procedure without complications
 TREATMENT
TREATMENT
The following four general methods are used for treating opioid addiction:
1. One method involves substituting the equivalent amount of an oral opioid (usually methadone) for the injectable form that the addict had been using (e.g., heroin) and then gradually withdrawing that oral form.
2. Another method involves going cold turkey by abruptly withdrawing the opioid and using adjunctive medication to alleviate the symptoms of withdrawal, such as phenothiazines, clonidine, or benzodiazepines.
3. A third method involves maintaining a patient on high doses of methadone, termed methadone maintenance. With this method, the patient takes supervised large oral doses of methadone on a daily basis. Because the patient develops a tolerance for the effects of the opioids, a block is produced that prevents heroin-like agents from producing the “rush” feeling after injecting.
4. The last method involves administering an orally effective, long-acting antagonist, naltrexone (Trexan). Naltrexone blocks the action of usual doses of opioid administered illicitly. No treatment for opioid addiction is successful in all patients.
Allergic Reactions
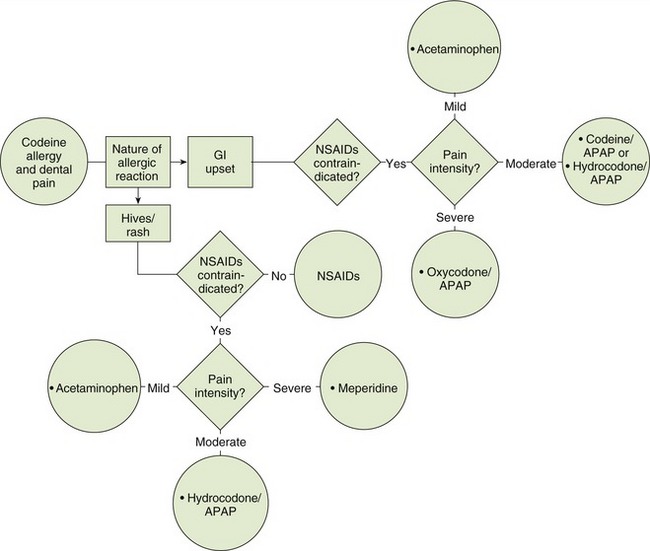
The most common type of true allergic reactions to the opioids is dermatologic in nature, including skin rashes and urticaria. Reports of gastrointestinal side effects are often reported as allergies but are side effects of the opioids. Contact dermatitis can occur with topical exposure. These allergic reactions have to be differentiated from the symptoms related to the histamine-releasing properties of the opioids. If a patient gives a history of a true allergic reaction to an opioid, an opioid from a different chemical class should be chosen (see Box 6-1). Figure 6-3 shows choices of analgesics for the patient allergic to codeine. Some brands of opioid analgesic combinations are formulated with sodium bisulfite. In patients with sulfite hypersensitivity, reference sources should be consulted to determine which brand contains sulfites.
Drug Interactions
Some of the drug interactions of the opioids are listed in Table 6-5. The most common outcome is sedation.
TABLE 6-5
DRUG INTERACTIONS OF THE OPIOIDS
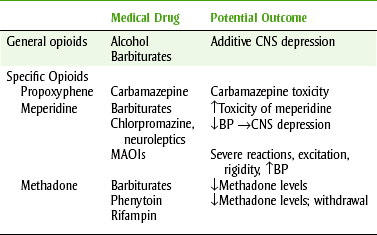
BP, Blood pressure; CNS, central nervous system; MAOIs, monoamine oxidase inhibitors.
The respiratory depression produced by the opioids is additive with that produced by other CNS depressants. Alcohol or sedative-hypnotic agents can potentiate the opioids’ respiratory depressant effect. When promethazine or hydroxyzine (antihistamines) is added to an opioid regimen, the opioid dose should be reduced.
All opioids can interact with the monoamine oxidase inhibitors (MAOI), a group of drugs used to treat depression. CNS excitation, hypertension, and hypotension have been reported. The accumulation of a metabolite of meperidine, normeperidine, may be responsible for the increased effect of meperidine in the presence of the antipsychotic agents such as chlorpromazine.
SPECIFIC OPIOIDS
The analgesic action of the most commonly used opioids (agonists) is related to their action on the µ-receptors and κ-receptors (see morphine in Table 6-3). These agonist opioids are discussed first.
 MORPHINE
MORPHINE
Morphine (more-FEEN) is considered to be the prototype opioid agonist against which other opioids are measured. An equivalent number of milligrams of each opioid is compared with 10 mg of morphine. Morphine is used parenterally to control postoperative pain in hospitalized patients. It is also used orally, primarily in the treatment of terminal illnesses. Sustained-release morphine tablets are the most commonly used form of morphine for outpatient use in the terminally ill. Few, if any, sustained-release analgesics are useful in dentistry because the patient needs immediate relief not future relief. The usual oral dosing interval and route of administration are listed in Table 6-2.
 OXYCODONE
OXYCODONE
Oxycodone (ox-i-KOE-done) is used alone or combined with either aspirin (in Percodan) or acetaminophen (in Percocet and Tylox) (Table 6-6) to provide relief of moderate-to-severe pain. Combining an opioid with a nonopioid analgesic produces an additive analgesic effect with fewer adverse reactions. Oxycodone retains about two-thirds of its action when given orally. It bridges the gap between codeine and morphine in terms of strength of analgesic action.
 HYDROCODONE
HYDROCODONE
There are many combinations of hydrocodone (hye-droe-KOE-done) and acetaminophen, including the original, which contains 5 mg of hydrocodone and 500 mg of acetaminophen (5/500). Various combinations of the two ingredients, hydrocodone and acetaminophen, include ranges of 2.5 to 10 mg of hydrocodone and 500 to 750 mg of acetaminophen. The companies offering brand name combinations seem to manufacture new combinations to thwart the development of generic equivalents. The original product combination (hydrocodone 5 mg and acetaminophen 500 mg) is recommended for the majority of dental patients with pain. To change the dose of this drug combination, the number of tablets per dose can be altered (from one-half to two tablets). This strength is available generically and is inexpensive. As with any opioid product containing acetaminophen, the total dose of acetaminophen should not exceed 4 gm (see Chapter 5). Upper limits for the total daily dose per day must not be exceeded. With combination products, the maximum daily dose (for a person who does not drink) is six tablets for products containing 650 mg and five tablets for those with 750 mg per dose form.
 CODEINE
CODEINE
Codeine (KOE-deen) is the most commonly used opioid in dentistry, and it is combined with acetaminophen (Tylenol #3) for oral administration. Other constituents, including caffeine and aluminum/magnesium hydroxides (antacid), are often included in these analgesic combinations. Codeine has a relatively weak analgesic action compared with morphine, hydromorphone, hydrocodone, or even oxycodone. Some commonly used analgesic combinations are listed in Table 6-6. Evidence for hydrocodone’s efficacy being more than that of codeine is lacking because hydrocodone was previously used (and therefore tested) as an antitussive.
The amount of codeine in combination products is designated by #2 (15 mg; 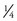 gr), #3 (30 mg;
gr), #3 (30 mg; 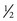 gr), and #4 (60 mg; 1 gr). Generally, doses greater than 30 to 60 mg of codeine are poorly tolerated by the patient (too much nausea). In pain studies, it is difficult to show that 30 mg of codeine is any better than a placebo, and 60 mg of codeine has an analgesic strength about the same as 650 mg of aspirin, 650 mg of acetaminophen, or 200 mg of ibuprofen. Because of codeine’s weak analgesic efficacy, prescription doses of NSAIDs often produce better results in the management of dental pain. When codeine is combined with nonopioid analgesics, there is additive analgesic activity. In the combination products, lower doses of each analgesic may be used and there is a potential for a reduction in adverse reactions.
gr), and #4 (60 mg; 1 gr). Generally, doses greater than 30 to 60 mg of codeine are poorly tolerated by the patient (too much nausea). In pain studies, it is difficult to show that 30 mg of codeine is any better than a placebo, and 60 mg of codeine has an analgesic strength about the same as 650 mg of aspirin, 650 mg of acetaminophen, or 200 mg of ibuprofen. Because of codeine’s weak analgesic efficacy, prescription doses of NSAIDs often produce better results in the management of dental pain. When codeine is combined with nonopioid analgesics, there is additive analgesic activity. In the combination products, lower doses of each analgesic may be used and there is a potential for a reduction in adverse reactions.
 PROPOXYPHENE
PROPOXYPHENE
An opioid that is synthetic and is chemically similar to methadone is propoxyphene (proe-POX-i-feen), which is in Darvocet N-100. Its analgesic efficacy has been questioned (see Table 6-6), and it is certainly no more efficacious than two tablets of aspirin or acetaminophen. It is available combined with aspirin and caffeine or acetaminophen, which adds to its puny efficacy. Its adverse effects include nausea, vomiting, dizziness, and physical dependence. These adverse effects are significantly higher and more dangerous in the elderly. Also, its active metabolite is cardiotoxic. Hundreds of deaths have been associated with its overdose, often in combination with alcohol. With the availability of other agents, it is difficult to justify the use of propoxyphene. However, propoxyphene is very commonly prescribed in medicine. The reason for this is that it acts as the bridge (in the provider’s perception) when “waffling” on whether to give an opioid. (There must be lots of “wafflers.”)
 MEPERIDINE
MEPERIDINE
Meperidine (me-PAIR-i-deen) (Demerol) is intended for the acute management of moderate-to-severe pain. It requires 100 mg to equal about 10 mg of morphine. However, its use has resulted in inadequate pain control and adverse effects for many patients over the years. Morphine, hydromorphone, or oxycodone should be used in its place. The drug interactions between meperidine and both the MAOIs and phenothiazines must be considered before using meperidine. Meperidine is a poor choice for oral use because it has a high first-pass effect, a short duration of action, and more drug interactions. It may be useful as an ingredient in an anxiolytic “medley” given as an oral preoperative. Although occasionally used in outpatient dentistry, it has little if any use.
 HYDROMORPHONE
HYDROMORPHONE
An orally effective opioid, hydromorphone (hye-droe-MORE-fone) (Dilaudid) is reserved for the management of severe pain. It is more potent than morphine and better absorbed orally, but it tends to produce similar adverse reactions. Its use in dentistry should be limited to rare situations, limited numbers, and careful monitoring. It is a favorite of the addict because of its strength (see Table 6-6).
 METHADONE
METHADONE
Methadone (METH-e-done) (Dolophine) is used primarily in the treatment of opioid addicts. It is used either to withdraw the patient gradually or for methadone maintenance. Because it has a longer duration of action, withdrawal from methadone is easier than from heroin. However, because it is an opioid analgesic the risk for dependence still exists. Lately, methadone has been used as an analgesic in the treatment of chronic pain because it can be dosed less frequently than short-acting opioids such as morphine or hydrocodone. It also has a long half-life, good bioavailability, and it is cost-effective. The downside to methadone use is the risk of death and life-threatening changes in breathing and heart rates. These incidences have been reported in persons newly starting methadone or in persons switching to methadone after using stronger opioid analgesics. As a result, only low doses should be prescribed for pain.
 FENTANYL FAMILY
FENTANYL FAMILY
Fentanyl (FEN-ta-nil) (Duragesic, Sublimaze), sufentanil (sue-FEN-ta-nil) (Sufenta), and alfentanil (al-FEN-ta-nil) (Alfenta) are short-acting parenterally administered agonist opioid analgesics that are used perioperatively or during general anesthesia. They provide analgesia during and immediately after general anesthesia. Fentanyl is used in combination with droperidol (droe-PER-i-dol) (Inapsine) to induce or supplement general or regional anesthesia and to produce general anesthesia. Postoperative ventilation and observation are needed when these agents are used. Fentanyl is also available as a patch (Fentanyl [Duragesic] Transdermal System) for application to the skin every 3 days. The patches provide constant pain relief for the terminally ill. Sometimes, oral morphine is used concomitantly as needed to control “breakthrough” pain.
Mixed Opioids
Mixed opioids include the agonist-antagonist opioid analgesics and the partial agonists. The only mixed opioid available for oral use is the agonist-antagonist pentazocine. Butorphanol (Stadol), available as a nasal spray, is also in this group. This group is ripe for research to develop opioids with adequate analgesic potency and fewer side effects, such as respiratory depression and addiction potential, than the agonist opioids. At present, their place in dental therapeutics is unclear.
 AGONIST-ANTAGONIST OPIOIDS
AGONIST-ANTAGONIST OPIOIDS
The only agonist-antagonist opioid available in oral form is pentazocine (pen-TAZ-oh-seen) (Talwin). It produces CNS effects not unlike the opioid agonists, including analgesia, sedation, and respiratory depression. The type of analgesia it produces is somewhat different from that produced by the agonist opioids. This may be the result of its agonist action at the κ-receptors and δ-receptors and its antagonist action at the µ-receptor. (References differ in attributing the dysphoric and psychomimetic adverse reactions—some say δ, and others say σ.)
The adverse reactions of pentazocine include sedation, dizziness, nausea, vomiting, and headache. Opioid-like effects on the gastrointestinal tract occur with pentazocine. Psychomimetic effects, including nightmares, hallucinations, and dysphoria, have been reported. With high doses, respiratory depression can occur. Unlike the agonist opioids, increasing the dose of pentazocine does not result in a commensurate increase in respiratory depression, that is, respiratory depression is nonlinear. Unlike the opioid agonists, pentazocine can increase both the blood pressure and heart rate. This may be related to its catecholamine-releasing properties.
The drug of choice to treat pentazocine overdose is naloxone. With abuse, repeated injections in the same location can result in severe sclerosis, fibrosis, and ulceration. Because pentazocine was initially thought not to have abuse potential, many pentazocine abusers have been produced. A popular mixture termed “Ts and Blues” is a combination of pentazocine (Talwin) and pyribenzamine (blue-colored tablet), an antihistamine. Because of its weak antagonist property, it may precipitate withdrawal in the addict.
Pentazocine is available as tablets containing 50 mg of pentazocine and 0.5 mg of naloxone, a pure opioid antagonist (Talwin-NX). Naloxone, a Schedule IV opioid, was added to pentazocine to reduce its addiction potential. How does it do this? First, naloxone, a pure antagonist, is effective parenterally but not orally because it is inactivated. Second, if the tablet is taken by the intended oral route, the naloxone will not affect its analgesic potency because it is rapidly inactivated when taken orally. Third, if the contents of the tablet are injected parenterally, the active naloxone will counteract the action of pentazocine, reducing its positive effects. This combination tablet has resulted in a tablet that is more difficult to abuse and whose street value was cut in half. (One cannot say that drug addicts do not know their pharmacology.)
Parenterally available agonist-antagonists include dezocine (DEZ-oh-seen) (Dalgan), nalbuphine (NAL-byoo-feen) (Nubain), and butorphanol (byoo-TOR-fa-nol) (Stadol). Dezocine has agonist action at the κ-receptor and antagonist action at the µ-receptor. Its analgesic strength is comparable to morphine at usual therapeutic doses. Like pentazocine, these other agonist-antagonists demonstrate nonlinear respiratory depression. Sedation, nausea, vomiting, xerostomia, and headache are side effects of these drugs. These agents produce fewer psychomimetic effects than pentazocine but more than the agonists.
When originally marketed, these agonist-antagonists were said to have much less addiction potential or even none at all. They were not placed on any narcotic schedule by the Drug Enforcement Administration (DEA). Butorphanol, available as a nasal spray, has been marketed for some time. Because of the nature of the patient use of this agent in clinical practice, most pharmacists consider this product to be “addicting.” The current literature and clinical practice has determined that these agents do in fact have addiction potential. If they are added to the list, it will not be surprising.
 PARTIAL AGONISTS
PARTIAL AGONISTS
The first and only available partial agonist is buprenorphine (byoo-pre-NOR-feen) (Buprenex, Subutex). It is a partial m-receptor agonist but has no d-receptor action. In abstinent morphine-dependent patients, buprenorphine suppresses withdrawal; in stabilized opioid-dependent patients, it precipitates withdrawal. Its abuse potential appears to be moderate, and it is classified as a Schedule III drug. It is available for oral and parenteral use.
 OPIOID ANTAGONISTS
OPIOID ANTAGONISTS
Naloxone (nal-OX-zone) (Narcan) is an essentially pure opioid antagonist that is active parenterally. It antagonizes the µ-receptors, κ-receptors, and δ-receptors. When given alone, it produces few pharmacologic effects in the usual therapeutic doses. Naloxone is the drug of choice for treating agonist or mixed opioid overdoses. It will reverse opioid-induced respiratory depression. If another agent, such as a barbiturate, is responsible for the depression, naloxone does not add to the respiratory depression. If administered to an addict who has taken an overdose of an opioid, small doses must be carefully titrated or opioid withdrawal may be produced. It also serves as a useful tool in research to determine the role of the opioid receptors in hypnosis, acupuncture, and the placebo effect.
Naloxone is given intravenously or intramuscularly with an average adult dose of 2 mg and a range of doses between 0.4 and 10 mg. Doses should be repeated if the duration of action of the opioid is longer than that of naloxone.
Effects should occur within 1 to 2 minutes. Doses may be repeated at 2- to 3-minute intervals. If no response occurs after 10 mg is administered, the diagnosis of opioid overdose must be questioned. If any opioid is used in the dental office, the dental office emergency kit should contain naloxone.
Nalmefene (NAL-me-feen) (Revex) is another parenteral opioid antagonist used to reverse opioid overdose.
Naltrexone: A long-acting, orally effective opioid antagonist, naltrexone (nal-TREKS-zone) (ReVia, Vivitrol) is indicated for the maintenance of the opioid-free state in detoxified, formerly opioid-dependent patients. It should not be administered until the patient has remained opioid free for at least 1 week and has had a negative naloxone challenge. It is also used in the management of alcohol abstinence. Its adverse reactions include insomnia, nervousness, headache, abdominal cramping, nausea, vomiting, and arthralgia. Acute hepatitis and liver failure have been associated with naltrexone. It is administered daily or in some instances three times weekly. Patients on naltrexone should not be given opioid analgesic agents for management of dental pain.
Tramadol
Tramadol (Ultram) is a unique analgesic with an interesting mechanism of action. It has µ-opioid agonist action and inhibits the reuptake of norepinephrine and serotonin (modifies the ascending pain pathways). It has some but not all properties of an opioid (like codeine and hydrocodone) because of its µ-agonist activity and it does not affect the other two opioid receptors the κ-receptors and δ-receptors. Tramadol’s other mechanism involves the inhibition of reuptake of norepinephrine and serotonin, similar to the mechanism of the antidepressants.
Adverse reactions of tramadol include CNS effects such as dizziness, somnolence, headache, and stimulation. Gastrointestinal tract side effects include nausea, diarrhea, constipation, and vomiting. Palpitations, diaphoresis, and seizures have been reported in patients taking tramadol. One should watch for signs of addiction (currently this drug is not scheduled).
Tramadol use is associated with physical dependency and withdrawal symptoms. Tramadol causes typical opiate withdrawal symptoms and atypical symptoms including anxiety, palpitations, and anguish.
Tramadol’s analgesic efficacy is difficult to assess because most studies are unpublished and therefore cannot be judged; studies comparing tramadol with nonsteroidal agents or stronger opioids have not been done. Tramadol efficacy is stated in Table 6-7.
TABLE 6-7
RELATIVE EFFICACY OF TRAMADOL (ULTRAM)
| Tramadol Dose (mg) | Compared with Codeine (mg) | Comparison with Combinations |
| 50 | = 60 | <#2 ASA/#3 codeine (total = 650/60) |
| 75 | ≥APAP/propoxyphene 650/100 | |
| 100 | >60 | <#2 ASA/#3 codeine(total = 650/60) |
| 150 | >60 | ≥APAP/propoxyphene 650/100 |
| 5 times a day | ||
| 50 | = #1 tablet acetaminophen 300/codeine 30 mg | |
| = #1 tablet of aspirin 300/codeine 30 mg |
The usual adult dose is 50 to 100 mg every 4 to 6 hours, not to exceed 400 mg/day. Because of its weak analgesic activity and its high cost compared with established analgesics, its use is difficult to justify.
DENTAL USE OF OPIOIDS
The advent of the NSAIDs has produced a change in the use of the opioids in dental practice. Most dental pain can be better managed by the use of NSAIDs. In the patient in whom NSAIDs are contraindicated, the dentist has a wide range of opioids from which to choose. By beginning with codeine or hydrocodone combinations and progressing to oxycodone combinations, almost all dental pain can be managed. Only in rare cases and for very short periods (1 to 2 days) should stronger opioids be prescribed for outpatient dental pain. Box 6-2 lists the patient instructions for opioid analgesics.
Patients with chronic pain should be managed with nonopioid therapies and referred to appropriate specialists, depending on the nature of their chronic pain. In the treatment of chronic pain, opioids are not indicated. New patients with a complaint of pain should be seen in the dental office, and definitive treatment rendered. Opioid prescriptions should be given only for small amounts without refills and only if the patient has dental treatment performed. If dental pain persists, the patient should be seen in the dental office for evaluation and local treatment. If the patient demands opioids repeatedly, the patient should be referred to a pain clinic for evaluation. Temporomandibular disease (TMD), formerly called temporomandibular joint (TMJ) disease, often produces chronic pain. When pain becomes chronic, the mechanisms producing the pain differ from that of acute pain. Treatment of TMD should include NSAIDs and possibly muscle relaxants and tricyclic antidepressants.
CLINICAL SKILLS ASSESSMENT
1. What are some red flags associated with opioid addiction?
2. Is any one opioid more addicting than another? If so, what is this based on?
3. Is there a need for concern with opioid addiction for patients taking these drugs to treat or manage dental pain? Why or why not?
4. What is hydrocodone and how effective is it in treating or managing dental pain?
5. Compare and contrast hydrocodone with ibuprofen.
6. What are the adverse reactions associated with hydrocodone?
7. Are there drug interactions with hydrocodone?
8. What are the dental concerns associated with hydrocodone?
![]() Please visit http://evolve.elsevier.com/Haveles/pharmacology for review questions and additional practice and reference materials.
Please visit http://evolve.elsevier.com/Haveles/pharmacology for review questions and additional practice and reference materials.