Pregnancy and Breast Feeding
1 List the two main concerns in the administration of drugs during pregnancy.
2 Define teratogenicity and outline the Food and Drug Administration’s categories of drugs for pregnancy.
3 Name several types of local anesthetic, antiinfective, and antianxiety agents and state their indications or contraindications for pregnant women.
Dental treatment of the pregnant or nursing woman is always of special concern to dental health care workers. Pregnant women often need additional dental treatment during their pregnancies, and in addition, that treatment must be carefully planned. Many questions about drug therapy for the pregnant or breast-feeding woman arise. The literature, unfortunately, does not provide all the answers. This chapter attempts to offer guidelines for determining the relative risk when prescribing drugs for the pregnant woman or nursing mother. No unnecessary drug should be administered to the pregnant woman. If a drug is to be administered, the risk to the fetus must be weighed against the benefit to the woman. An adequate health history, including whether a woman might be pregnant (puberty to menopause) should be taken at each dental appointment. Close coordination with the patient’s obstetric health care professional is recommended when questions about her potential use of drugs arise. Consultations should be documented in the patient’s chart. Box 24-1 lists the dental implications involved in managing a pregnant dental patient.
GENERAL PRINCIPLES
Two main concerns must be addressed when considering whether to give a drug to a pregnant woman. The first is that the drug may be teratogenic. The term teratogen is derived from the Greek prefix terato-, meaning “monster,” and the suffix -gen, meaning “producing.” These two combine to give rise to the meaning of teratogen: “producing a monster.” The second is that the drug can affect the near-term fetus, causing the newborn infant to have an adverse reaction, such as respiratory depression or jaundice. A relatively new concern is the long-term (physiologic and psychological) consequences of in utero exposure to agents not evident at birth.
History
In 1941, a relationship between getting German measles during pregnancy and blindness, deafness, and death of the offspring was noted. Scientists recognized that exogenous agents could affect the unborn fetus, producing congenital abnormalities. In 1961, a “harmless” sedative, thalidomide, available over the counter (OTC) in Europe, was taken by pregnant women. An increase in the rare birth defect phocomelia (short or absent limbs) occurred shortly thereafter. Thalidomide was later implicated in these birth defects. Environmental factors are also thought to contribute to birth defects.
PREGNANCY
Pregnancy involves three trimesters, each 3 months long. During the first trimester, the organs in the fetus are forming. This is considered the most critical time for teratogenicity. If abnormalities occur very early in development, spontaneous abortion is the usual outcome. With later exposure, abnormalities occur in the fetus. Often, a woman is unaware that she is pregnant for at least one-half of this trimester. Dental prophylaxis with detailed instructions and a visual examination of the oral cavity without x-rays should be performed if the patient is pregnant. Because this is the time when the woman may feel nauseated at any time during the day or night (often referred to as morning sickness), other elective dental treatment should be avoided during this time.
The second trimester is an excellent time for the patient to receive both oral health instructions and another dental prophylaxis, if needed. The patient’s periodontal status should be carefully evaluated during this time. The patient is most comfortable during this trimester.
The third trimester is closest to delivery. The woman is beginning to feel uncomfortable, and it is difficult for her to lie prone for any length of time.
If dental treatment is needed, she may feel more comfortable sitting or with the right hip elevated. In addition, this is the time when premature labor is most likely to begin. Drugs that may affect the newborn child should not be given during this trimester.
Teratogenicity
It is difficult to prove that a drug is teratogenic in humans (Box 24-2).
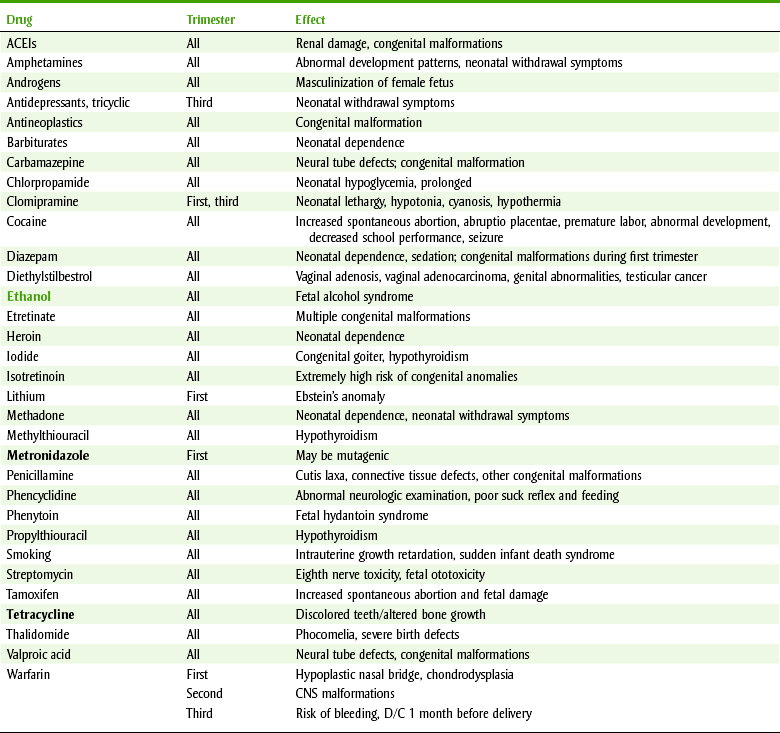
Drugs that are known teratogens include drugs such as thalidomide, certain vitamin A analogs (isotretinoin), antineoplastic agents (busulfan, cyclophosphamide), oral anticoagulants (warfarin), lithium, methimazole, penicillamine, some antiepileptic agents (phenytoin, trimethadione, and valproic acid), the tetracyclines, certain steroids (diethylstilbestrol, androgens), and ethyl alcohol. Table 24-1 lists selected drugs with adverse effects on the fetus.
Food and Drug Administration Pregnancy Categories
The U.S. Food and Drug Administration (FDA) has developed pregnancy categories A, B, C, D, and X. Each drug that is the subject of FDA regulation for pregnancy labeling is given a category based on its known potential for risk. Table 24-2 gives a summary of the criteria for the different categories. One should note that the availability of animal or human studies is a criterion. Category A is the safest, and category X should not be used in pregnant women. Categories B, C, and D fall in between these two criteria.
TABLE 24-2
FDA PREGNANCY CATEGORIES FOR DRUGS*
| Category | Description | Examples |
| A | Adequate studies have failed to demonstrate a risk to fetus (in first trimester) and no evidence of risk in later trimesters; possibility of fetal harm appears remote. | Thyroid supplements (levothyroxine), vitamins (folic acid, riboflavin; vitamins A, D, and C†), potassium chloride |
| B | Animal studies have failed to demonstrate a risk to the fetus, and there are no adequate studies in pregnant women; or animal studies show an adverse effect on the fetus but well-controlled studies in pregnant women have failed to demonstrate a risk to the fetus. | Acetaminophen, acyclovir, opioids,‡ penicillins, cephalosporins, erythromycin,§ caffeine, cimetidine, insulin, NSAIDs¶ |
| C | Animal studies have shown an adverse effect on the fetus and there are no adequate studies in humans, or no studies are available in either animals or women. Potential benefits may warrant its use. | Epinephrine, phenylpropanolamine, trimethobenzamide, aspirin,¶ atropine, promethazine, theophylline, lisinopril, disulfiram, propranolol, fluoxetine, amitriptyline, sulfonamides,| prednisone |
| D | Positive evidence of human fetal risk based on adverse reaction data, but potential benefits in serious situations may warrant its use. | Warfarin, tetracycline, phenytoin, diazepam, trimethadione, lorazepam |
| X | Studies in animals or humans have demonstrated fetal abnormalities and/or there is positive evidence of human fetal risk, and the risks clearly outweigh any potential benefits. | Isotretinoin, diethylstilbestrol, phencyclidine (PCP), triazolam |
FDA, Food and Drug Administration; NSAIDs, nonsteroidal antiinflammatory drugs.
*Any unnecessary medication should be avoided in pregnant women.
†When used at recommended daily allowance (RDA) levels.
§Except erythromycin estolate.
¶Except near term, when dystocia and delayed parturition can be produced and then categorized as D.
BREAST FEEDING
Questions about the safety of a certain drug given to a nursing mother are appearing more often because nursing is becoming more popular. As during pregnancy, the risk-to-benefit ratio should be carefully considered before drugs are given to the nursing mother. Drugs without strong indications for use should not be taken. Almost all drugs given to the mother can pass into the breast milk in varying concentrations. While nursing, the baby ingests the drug, which may produce an effect in the infant. The amount of drug that appears in the milk depends on the plasma concentration of the drug, lipid solubility, degree of ionization, and binding to plasma proteins.
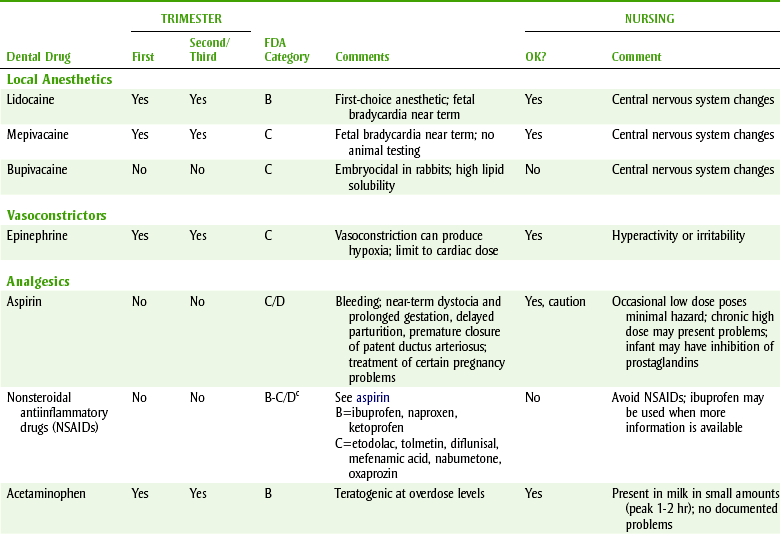
For a few drugs, nursing is clearly contraindicated. If these drugs must be given, breast feeding should be discontinued or the milk expressed and discarded until the mother stops taking the contraindicated drug. For drugs that are not contraindicated, the timing of nursing can further reduce the dose to which an infant is exposed. Table 24-3 summarizes the available data on the use of dental drugs for nursing mothers.
TABLE 24-3
DENTAL DRUG USE DURING PREGNANCY,a FOOD AND DRUG ADMINISTRATION (FDA) CATEGORIES,b AND NURSING SAFETY
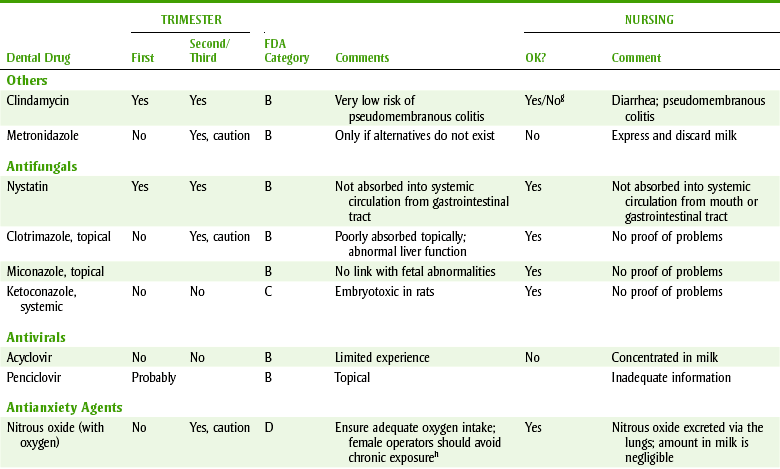
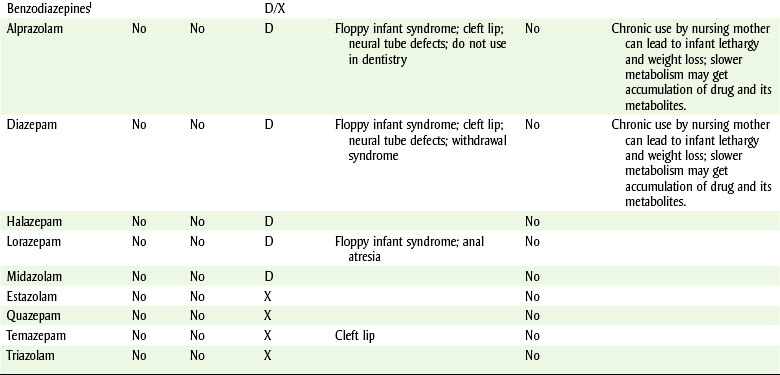
aDo not administer any drug that is not absolutely necessary; potential risk to the fetus must be weighed against the benefit to the woman; consult the patient’s health care provider before using drugs.
bSee Table 24-2 for definition of FDA categories.
cCategory D in third trimester of pregnancy.
dOxycodone, meperidine, hydrocodone.
eCodeine.
fAugmentin = amoxicillin + clavulanic acid.
gReferences differ.
hCheck levels in the dental operatory, minimize risk, increase ventilation exchanges.
iClorazepate, flurazepam, oxazepam—no category.
DENTAL DRUGS
Questions relating to drug administration in conjunction with dental treatment refer to whether a specific drug may be safely given to the pregnant woman. In general, a drug should be used in a pregnant woman only if the benefits to the pregnant woman outweigh the risks to the fetus and a definite indication exists. Table 24-3 summarizes the information about which dental drugs can be used in pregnant women.
Local Anesthetic Agents
No drug is used more often in the dental office than a local anesthetic agent. Local anesthetic amides have been reported to produce fetal bradycardia and neonatal depression when given in very large doses near to term. High doses may produce uterine vascular constriction, leading to fetal heart rate changes. Lidocaine, prilocaine, and etidocaine have been tested in animals without teratogenic effects (category B). Bupivacaine has been shown to be teratogenic in rats and rabbits (category C), whereas mepivacaine has not been tested (category C). Small doses used by careful, slow injection have not been associated with any problems in the fetus. Lidocaine is the local anesthetic of choice for the pregnant woman because it is a category B drug and is not associated with methemoglobinemia (as is prilocaine) and not highly lipid soluble (as is etidocaine), prolonging its effect.
Epinephrine
Small doses of epinephrine, administered with appropriate care, are similar to those produced endogenously. Large doses could produce adverse effects in the fetus, including anoxia from vasoconstriction. If procedures are to be short, then local anesthetics without epinephrine are preferred. These comments also apply to other vasoconstrictor substances contained in local anesthetic solutions.
Analgesics
Analgesics should be given in the lowest possible dose and for the shortest duration possible to control pain. In dentistry, adjunctive therapy (incision, drainage, and curettage) should be used first.
 ASPIRIN
ASPIRIN
Studies in animals have shown that aspirin can cause a variety of birth defects involving the eyes, central nervous system (CNS), gastrointestinal tract, and skeleton. In humans, controlled studies have not been able to demonstrate that aspirin use during pregnancy increases the incidence of birth defects. During the third trimester, aspirin can prolong gestation, complicate delivery, decrease placental function, or increase the risk of maternal or fetal hemorrhage. Premature closure of the patent ductus arteriosus may occur (see Chapter 5). These effects have been reported with chronic high-dose aspirin use. Abuse of aspirin may increase stillbirths or neonatal death.
 NONSTEROIDAL ANTIINFLAMMATORY DRUGS
NONSTEROIDAL ANTIINFLAMMATORY DRUGS
The nonsteroidal antiinflammatory agents (NSAIDs) produce effects similar to aspirin; therefore if they are given near term, the outcome on the fetus would be expected to be the same as aspirin’s effects. They can delay delivery and make it more difficult and can constrict the ductus arteriosus. NSAIDs also potentiate vasoconstriction if hypoxia exists. All NSAIDs carry a warning to avoid use during pregnancy. For ibuprofen and naproxen, studies in animals have not shown adverse effects on the fetus. Diflunisal (category C), but not naproxen (category B), has been shown to be teratogenic in rabbits in large doses. Ibuprofen is the NSAID of choice for the nursing mother.
 ACETAMINOPHEN
ACETAMINOPHEN
Although no controlled studies in humans have been done, acetaminophen (APAP) is generally considered to be safe in pregnancy. In large doses, it may be associated with fetal renal changes similar to those that occur in adults.
 OPIOIDS
OPIOIDS
Doses used by addicts have been demonstrated to produce problems. The opioids, with the exception of codeine, have not been associated with teratogenicity. Retrospective studies have associated the use of codeine during the first trimester with fetal abnormalities involving the respiratory, gastrointestinal, cardiac, and circulatory systems and with inguinal hernia and cleft lip and palate (Figure 24-1). These studies suggest that codeine or other opioids should not be used indiscriminately during the first trimester. Whether the birth defects associated with codeine are related to its ubiquitous use or to some difference it possesses is not known. Near-term administration can produce respiratory depression in the infant. If the mother is addicted, the infant will experience withdrawal symptoms after birth. The use of codeine in limited quantities for a limited duration of time is common in clinical practice. Although opioids appear in breast milk when analgesic doses are administered, the small amounts appear to be insignificant. By properly timing the doses of analgesic, the dose the infant receives is reduced further. The infant should be observed for signs of sedation and constipation.
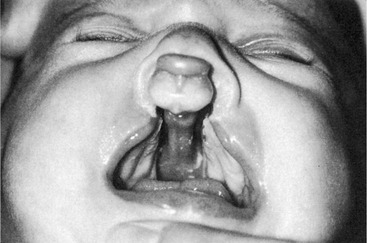
FIGURE 24-1 A 3-week-old female infant with a typical bilateral cleft lip and palate deformity. Certain drugs used during pregnancy, including opioids and benzodiazepines, have been known to cause this deformity. (From Sanders RC, Blackman LR, Hogge WA: Structural fetal abnormalities, ed 2, Philadelphia, 2002, Mosby.)
Antiinfective Agents
Antiinfective agents should only be used when a definite indication for their use exists. Prophylactic use, use when no indication exists, and use when an infection can be locally treated are inappropriate.
 AMOXICILLIN
AMOXICILLIN
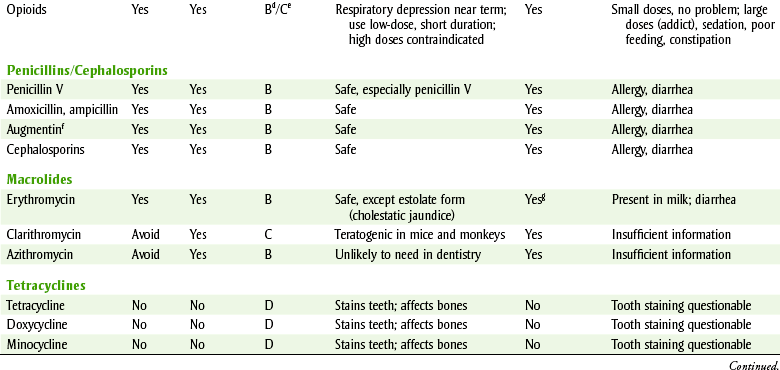
The most common antiinfective agent used in dentistry is amoxicillin. It is generally agreed that the amoxicillin is safe to use during pregnancy. Using amoxicillin for a dental infection that is not controlled by local measures would be acceptable. Amoxicillin appears in breast milk, and infants should be observed for signs of diarrhea, candidiasis, and allergic reactions.
 ERYTHROMYCIN
ERYTHROMYCIN
Erythromycins, other than the estolate form, also appear to be safe for use during pregnancy. The estolate form (Ilosone) should not be used in pregnant women because it has been associated with reversible hepatic toxicity in the mother. Erythromycin is concentrated in breast milk but has not been documented to produce problems.
 CEPHALOSPORINS
CEPHALOSPORINS
The first- and second-generation cephalosporins have not been associated with teratogenicity. These cephalosporins should be used in dentistry only if a specific indication exists.
 TETRACYCLINES
TETRACYCLINES
All tetracyclines, including tetracycline and doxycycline, are contraindicated during pregnancy because of the potential for adversely affecting the fetus. They cross the placenta and are deposited in the fetal teeth and bones. Deciduous teeth may become stained (see Color Plate 15), and fetal bone growth inhibited. Hepatotoxicity can occur in the pregnant woman treated with large doses of tetracycline. Whether the amount excreted in milk, after it is complexed with the calcium in milk, can produce problems in the nursing infant is not known.
 CLINDAMYCIN
CLINDAMYCIN
Clindamycin should be used for dental infections during pregnancy for susceptible anaerobic infections not sensitive to penicillin. It is also indicated for prophylaxis of endocarditis in penicillin-allergic patients. No adverse fetal problems have been reported. Because clindamycin is excreted in breast milk if it is given to nursing mothers, the infant should be monitored for diarrhea.
 METRONIDAZOLE
METRONIDAZOLE
In animals, metronidazole can produce birth defects. Metronidazole should be used carefully during the first trimester. It would be difficult to encounter a dental situation in which the risk to the fetus would not be greater than the benefit to the mother. Because animal studies have shown metronidazole to be carcinogenic, the nursing mother should only be given metronidazole if the breast milk is expressed and discarded during treatment and for 48 hours after the last dose.
 NYSTATIN
NYSTATIN
Nystatin is safe to use during pregnancy to treat oral Candida infections. When applied topically or taken orally, it is not absorbed into the systemic circulation. It may also be used by either the pregnant woman or the nursing infant to treat thrush.
 CLOTRIMAZOLE
CLOTRIMAZOLE
Small amounts of clotrimazole are absorbed from topical administration of this agent. No occurrences of abnormality have been reported, but nystatin is safer.
 KETOCONAZOLE
KETOCONAZOLE
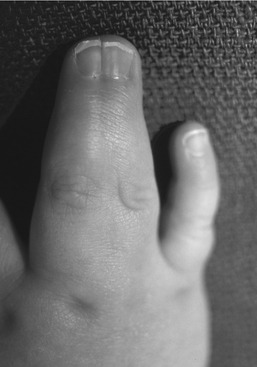
Ketoconazole is classified by the FDA as a category C drug. It has been shown to be teratogenic in rats, producing an abnormal number of digits (syndactyly [Figure 24-2] and oligodactyly). Dystocia during delivery has been demonstrated in animals. Ketoconazole appears in breast milk and may increase the chance that kernicterus (jaundice) may occur in the nursing infant. If ketoconazole must be used, breast milk must be expressed and discarded during therapy and for 72 hours after cessation of therapy. Fluconazole, like ketoconazole, is also scheduled as category C.
Antianxiety Agents
Operating room personnel exposed to trace amounts of nitrous oxide (N2O) have a significantly higher incidence of spontaneous abortion and birth defects in their children, regardless of whether the man or woman was exposed. These data suggest that methods for reducing the environmental exposure, especially chronically, should be explored and implemented. Pregnant dental health care workers should have knowledge of the levels of N2O that are present in the dental offices in which they practice.
 BENZODIAZEPINES
BENZODIAZEPINES
First-trimester use of the benzodiazepines (chlordiazepoxide and diazepam) has been reported to increase the risk of congenital malformations. Cleft palate and lip and neural tube defects (Figure 24-3) have been seen. Other benzodiazepines may be associated with this increase in risk also. Temazepam and triazolam are FDA pregnancy category X drugs, and alprazolam, halazepam, and lorazepam are category D drugs. Benzodiazepines are indicated during pregnancy only for the treatment of status epilepticus (no dental use).
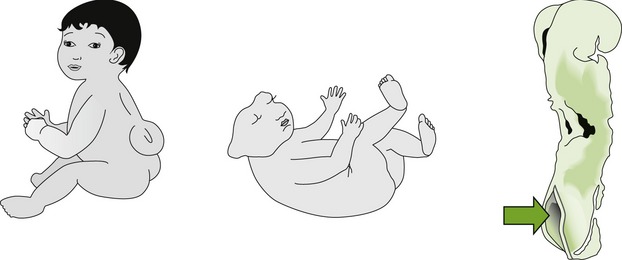
FIGURE 24-3 Neural tube defects include spina bifida and anencephaly. Administration of benzodiazepines during pregnancy can cause neural tube defects. (From Schlenker E, Long S: Williams’ essentials of nutrition & diet therapy, ed 9, St Louis, 2007, Mosby. Redrawn from Centers for Disease Control and Prevention, Atlanta.)
Chronic ingestion of the benzodiazepines can produce physical dependence in the infant. Floppy infant syndrome, or neonatal flaccidity, has been seen at birth, with inadequate sucking reflex or apnea. Use of benzodiazepines in the nursing mother, which may accumulate in the neonate because of slower metabolism, may cause sedation and feeding difficulties. Therefore if they are needed, the infant should be monitored for sedation.
 ALCOHOL
ALCOHOL
Although alcohol is not a dental drug, it is mentioned here because the evidence for the teratogenicity of alcohol is strong. Fetal alcohol syndrome (FAS) is the syndrome associated with the changes that occur in an infant exposed to excessive alcohol intake by the mother. FAS involves abnormalities in three areas: growth retardation (prenatal or postnatal), CNS abnormalities (neurologic or intellectual), and facial dysmorphology (e.g., microcephaly, microphthalmia or short palpebral fissures, and flat maxillary area or a thin lip [Figure 24-4]). Infants born to mothers who drank throughout pregnancy show more tremors, hypertonia, restlessness, crying, and abnormal reflexes compared with control groups after birth.
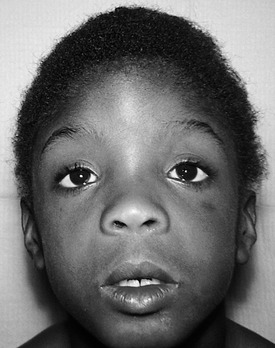
FIGURE 24-4 A child with fetal alcohol syndrome. (From Zitelli BJ, Davis HW: Atlas of pediatric physical diagnosis, ed 5, Philadelphia, 2007, Mosby.)
Pregnant dental patients should be encouraged to abstain from the ingestion of alcohol. No safe threshold level for the pregnant woman is known. Well-documented studies show that adverse effects on the fetus are dose related and can extend for years after the birth of the baby. The dental health care worker, as a health care professional, is in a position to remind the pregnant woman to care for her oral cavity and also her baby’s development.
CLINICAL SKILLS ASSESSMENT
1. Describe the proper method for the dental hygienist to obtain information about possible pregnancy or breastfeeding patients. State the information to be obtained.
2. Explain the three trimesters and the special risks for each one.
3. Define teratogenicity and describe why identifying drugs that produce it is so difficult.
4. Explain the FDA pregnancy categories and state their significance.
5. Determine the factors that are important when a woman who is breast feeding is to receive drugs.
6. For the commonly used dental drugs, such as local anesthetics, antibiotics, and analgesics, state the agents in each group that are the least safe.
7. Describe two activities that the dental health care worker should perform before giving a pregnant woman any medications to minimize future legal problems.
![]() Please visit http://evolve.elsevier.com/Haveles/pharmacology for review questions and additional practice and reference materials.
Please visit http://evolve.elsevier.com/Haveles/pharmacology for review questions and additional practice and reference materials.